Abstract
The preparation of low-generation dendrimers based on a simple calix[4]arene scaffold by insertion of the iminosugar-analogue C2-symmetric 3,4-dihydroxypyrrolidine is described. This methodology allows a rapid incorporation of a considerable number of iminosugar-like moieties in a reduced volume and in a well-defined geometry. The inclusion of alkali-metal ions (sodium and potassium) in the polar cavity defined by the acetamide moieties at the lower rim of the calixarene was demonstrated, which allows also the rigidification of the dendrimer structure and the iminosugar presentation in the clusters. The combination of the supramolecular properties of calixarenes with the advantage of a dendrimeric presentation of repetitive units opens up the possibility of generating well-defined multivalent and multifaceted systems with more complex and/or biologically relevant iminosugars.
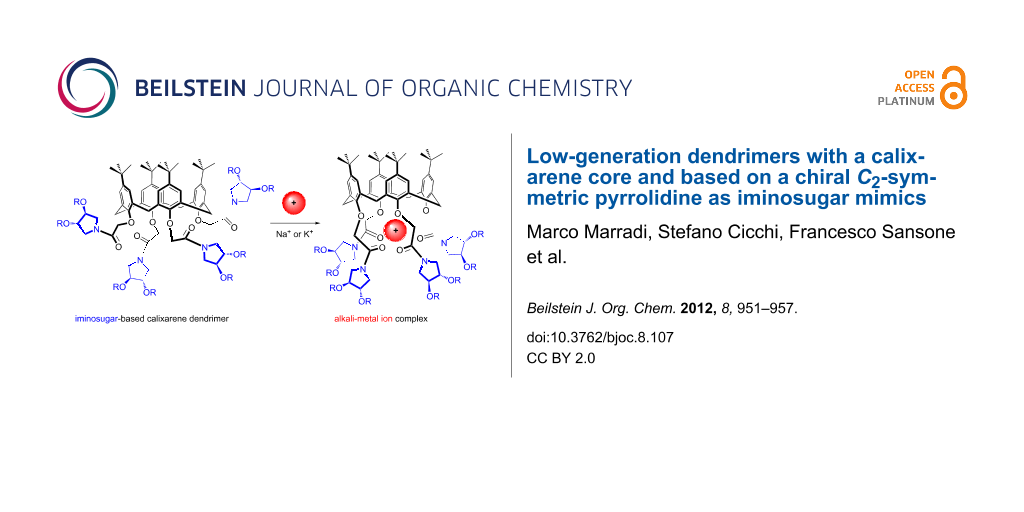
Graphical Abstract
Introduction
Polyhydroxylated pyrrolidines are one of the main classes of naturally occurring sugar mimics [1,2] and belong to the so-called iminosugars [3]. Several iminosugars have shown potential as therapeutics due to the ability to inhibit glycosidases and other enzymes associated with the metabolism of polysaccharides and the processing of glycoproteins [3]. Conjugation of iminosugars onto a polyvalent skeleton has been investigated only occasionally, and their properties as multivalent enzyme inhibitors gave contrasting results. Early findings indicated in fact that poor multivalent phenomena take place [4-6], but more recently moderate [7] to remarkable [8,9] effects have been reported on glycosidases. Unlike lectin-mediated interactions [10], the effect of iminosugar-based multivalent inhibitors on enzyme activity is difficult to rationalise. However, the introduction of several copies of an N-alkyl analogue of iminosugar 1-deoxynojirimycin onto a fullerene ball [8] or onto β-cyclodextrin [9] afforded the first pieces of evidence for a significant multivalent effect in glycosidase inhibition. These results highlight the interest in synthesizing multivalent iminosugar-conjugates with well-defined structures. In this context, we aimed at studying the feasibility of combining the supramolecular properties of multivalent scaffolds, such as calixarenes, with the advantages of a dendrimeric presentation of iminosugar analogues.
Calixarenes [11] have been widely employed in host–guest chemistry, first as ligands for small ions and neutral molecules [12,13] and, more recently, for biologically relevant molecules and macromolecules [14]. Multivalent calixarenes functionalised with carbohydrate units (glycocalixarenes) [15] have been extensively reported in the literature and represent examples of sugar clustering on macrocyclic structures [16,17]. Thanks to the “glycoside cluster effect” [18-20], glycocalixarenes can enhance the avidity of interactions between glycans and lectins [15]. Some glycocalixarenes have shown remarkable inhibition properties towards galectins [21,22] or Pseudomonas Aeruginosa lectin [23], the inhibition ability being dependent on the macrocyclic conformation and presentation of the glycoside units. With the purpose of expanding the valency and increasing the glycoside density, glycodendrimers have also been synthesised and their properties in protein–carbohydrate interactions have been studied [24-26]. However, the innovative frontier of combining glycodendrimeric arrangements onto a calixarene core has only occasionally been explored. To the best of our knowledge, only one example in which a glycodendrimer was built on a calixarene core has been published [27], while no examples of iminosugar-based calixarene dendrimers have been reported so far.
We report herein the synthesis of low-generation iminosugar-type calixarene-based dendrimers, demonstrating the feasibility of increasing the valency of the cluster by combining a dendrimeric arrangement of iminosugar ligands with a multivalent calixarene core.
Results and Discussion
We chose to address the conjugation of iminosugar-analogue dendrimers, based on the C2-symmetric (3S,4S)-3,4-dihydoxypyrrolidine (1) unit, to a simple calix[4]arene scaffold (Figure 1). This allows a rapid increase of the valency of the iminosugar dendrimer in a reduced volume. The C2 symmetry of pyrrolidine 1, its ready accessibility from the “chiral pool” (L-tartaric acid) [28], and its functional groups (an anchoring amine and two transformable hydroxy groups) make it a convenient chiral AB2 building block for the construction of dendrimers [29]. The linkable hydroxy groups of the pyrrolidine rings open up the possibility of constructing calixarene-based dendrimers [30] of higher generation. In addition to its properties as a chiral building block, pyrrolidine 1 may be considered an elemental iminosugar, and it is, by far, easier to handle and to characterise as a consequence of its symmetry. The synthesis of model iminosugar-based calixarene dendrimers may open the way for the construction of more complex systems, decorated with biologically active polyhydroxylated pyrrolidines or piperidines.
Figure 1: First (2) and second (3) generation of dendrimers based on chiral C2-symmetric pyrrolidine 1 and having p-tert-butyl calix[4]arene as the scaffold.
Figure 1: First (2) and second (3) generation of dendrimers based on chiral C2-symmetric pyrrolidine 1 and ha...
The synthesis of first- and second-generation iminosugar-based calixarene-dendrimers 2 and 3 (Figure 1) was addressed in order to prove the viability of such a strategy. A convergent synthetic approach was chosen for the conjugation of pyrrolidine 4 and pyrrolidine-based dendron 5 (see Scheme 1) to the calix[4]arene core, via intermediate 11, in order to obtain iminosugar-based calixarene-dendrimers 2 and 3 (see Scheme 2). The starting calixarene used in this work was the commercially available p-tert-butyl calix[4]arene, which has the phenolic hydroxy groups at the lower rim and the tert-butyl groups at the upper rim. Functionalization of p-tert-butyl calix[4]arene was performed by standard conversion of the hydroxy groups of the lower rim to activated acetic acid succinimide ester (COOSu) moieties (compound 11, see Scheme S2 in Supporting Information File 1). These moieties allowed the subsequent conjugation by amide bond formation between 11 and the suitably protected pyrrolidine derivatives 4 and 5 in order to obtain the calixarene-dendrimers 2 and 3, respectively. The synthesis of (3S,4S)-3,4-bis(tert-butyldimethylsilyloxy)pyrrolidine (4) was achieved by following a reported procedure [31], via the key intermediate 6 (Scheme 1 and Supporting Information File 1). Tripyrrolidine 5 required for the second generation calixarene-dendrimer 3 was prepared starting again from the key intermediate 6, but in this case a modified multistep synthetic procedure was necessary. First, N-Boc dihydroxypyrrolidine 7 was obtained from 6 through a “one pot” change of protecting group from benzyl to tert-butoxycarbonyl, which occurred in excellent yield by performing the catalytic hydrogenation in methanol under reflux and in the presence of Boc2O (Scheme 1). This simple process is unprecedented and may result in a new straightforward method to convert N-benzyl amines into N-Boc amines once a series of similar compounds are screened. On the other hand, alkylation of the hydroxy groups of 7 with the 2-ethoxycarbonylmethyl linker was problematic. In fact, when sodium hydride was used as a deprotonating agent, no reaction occurred after in situ addition of ethyl bromoacetate. However, a mixture of mono-(8') and di-(8) substituted products was obtained by using metallic potassium (Scheme 1). In spite of the poor yield (35%) of the desired product 8, the starting material 7 and the mono-derivative 8' could be recovered after column chromatography over silica gel, and used again for the same reaction. The N-protected pyrrolidine 8 was then activated for the coupling with pyrrolidine 4 in order to obtain the pyrrolidine-based dendron 5 to be used in assembling the second-generation calixarene-dendrimer 3. In particular, succinimidyl-activated pyrrolidine 9 was obtained by hydrolysis of ester groups in 8 followed by treatment with DCC/NHS (two steps, 80% overall yield). The amide coupling was then performed by reaction of 9 with pyrrolidine 4 using DIPEA as a base, and this afforded the branched tripyrrolidine 10 in 77% yield after column chromatography over neutral aluminium oxide. The tert-butoxycarbonyl group was then removed in the presence of TFA to obtain the pyrrolidine-based dendron 5 (Scheme 1), which was used in the next step without further purification.
Scheme 1: Use of the key intermediate (3S,4S)-1-benzyl-3,4-dihydroxypyrrolidine (6) [31] for the synthesis of pyrrolidine 4 and the pyrrolidine-based dendron 5. Reagents and conditions: i. Pd(OH)2/C, MeOH, HCOO−NH4+, Boc2O, reflux, 3 h, 95%; ii. K, THF, 0 °C to rt, 16 h; iii. BrCH2CO2Et, 4 h, 35% (8) and 17% (8'); iv. KOH 1 N, EtOH, reflux, 3.5 h, quantitative; v. N-hydroxysuccinimide (NHS), AcOEt, dicyclohexylcarbodiimide (DCC), 30 °C, 72 h, 80%, (Su: succinimidyl); vi. N,N-diisopropylethylamine (DIPEA), CH2Cl2, 30 °C, 5 d, 77%; vii. trifluoroacetic acid (TFA), CH2Cl2, rt, 16 h, quantitative.
Scheme 1: Use of the key intermediate (3S,4S)-1-benzyl-3,4-dihydroxypyrrolidine (6) [31] for the synthesis of pyr...
Using the SuO-activated calix[4]arene 11 as the scaffold, the planned convergent synthetic approach was completed to afford the first (2) and second (3) generation iminosugar-based calixarene dendrimers. The coupling between calixarene 11 and pyrrolidine derivatives 4 or 5 was performed as reported before for the synthesis of compound 10 (Scheme 1) and is shown in Scheme 2. The identity of both final compounds 2 and 3, obtained in good yields (77% and 68%, respectively), was assessed by their spectroscopic data and elemental analysis (see Supporting Information File 1). In particular, integration ratios of the calixarene aromatic (8H, δ ≈ 7 ppm) and tert-butyl signals (36H, δ ≈ 1 ppm) versus those of the CH3 of TBDMS at ca. 0 ppm were in good agreement with the number of the expected protons for dendrimers 2 and 3 (48H and 96H, respectively). Thus, an increase of the valency of a model iminosugar dihydroxypyrrolidine 4 in a controlled manner and geometry, by its conjugation to the calixarene scaffold 11 in a dendrimeric fashion, could be demonstrated. Quite interestingly, we could also prove the feasibility of the iminosugar deprotection on the calixarene dendrimer 2. By treatment with CsF in EtOH, the eight tert-butyldimethylsilyl groups could be removed and the deprotected derivative 12 was obtained in 56% yield after trituration with CH2Cl2 and washing several times with water.
Scheme 2: Synthesis of calixarene-based dendrimers 2 and 3. Reagents and conditions: DIPEA, CH2Cl2, 30 °C, 5 d, (Su: succinimidyl; TBDMS: tert-butyldimethylsilyl).
Scheme 2: Synthesis of calixarene-based dendrimers 2 and 3. Reagents and conditions: DIPEA, CH2Cl2, 30 °C, 5 ...
The presence of acetamide moieties at the lower rim of the dendrimers 2 and 3 prompted us to explore the possibility to use alkali metal salts as allosteric effectors in the modulation of the shape and rigidity of the iminosugar presentation by the calixarene scaffold. A common way, in fact, used to rigidify the “mobile cone” structure of tetraalkoxycalix[4]arenes is to introduce strong donating groups, such as amide or ester [32,33], on the phenolic oxygen atoms and an alkali-metal ion. The cation, strongly coordinated by eight oxygen atoms in the polar region created at the lower rim, blocks the calixarene in a “rigid cone” structure [34], also controlling the convergence of the iminosugars.
The ability of first-generation calixarene dendrimer 2 to bind alkali-metal cations was tested by means of NMR, by solid–liquid extraction of solid alkali picrate salts into a CDCl3 solution of ligand 2. A mixture of 0.5 equiv of sodium or potassium picrate and ligand 2 showed the simultaneous presence of the peaks of the complex and of the free ligand indicating a slow exchange regime on the NMR timescale. On the other hand, the NMR analysis of a CDCl3 solution of ligand 2 in the presence of an excess of metal picrate (see Figure S1 in Supporting Information File 1) allowed the stoichiometry of the complex to be established. As the picrate salts are scarcely soluble in CDCl3, the comparison of the integrals of the picrate signal (a singlet of 2H, around 8.8 ppm) and of the calixarene aromatic protons (a signal of 8H at 7.00–7.10 ppm) indicated that the complexes (both with sodium and potassium) have a 1:1 stoichiometry. As it can be seen in Figure 2, and as reported also for other alkali-metal ion complexes of similar tetramide ligands [35], the cation complexation induces a strong deshielding effect on the aromatic protons of about 0.3 ppm with respect to the free calixarene (as indicated by the asterisks) as a consequence of the electron-withdrawing effect of the metal ion coordinated to the phenolic oxygen.
![[1860-5397-8-107-2]](/bjoc/content/figures/1860-5397-8-107-2.png?scale=2.0&max-width=1024&background=FFFFFF)
Figure 2: Expansion (about 7 to 3 ppm) of the 1H NMR spectra of (A) the free ligand 2, (B) the sodium picrate complex, and (C) the potassium picrate complex. The full spectra are reported in Supporting Information File 1 (Figure S1). Asterisks: aromatic protons; empty circles: OCH2CON protons; filled circles: methylene bridge protons.
Figure 2: Expansion (about 7 to 3 ppm) of the 1H NMR spectra of (A) the free ligand 2, (B) the sodium picrate...
Quite remarkable is the up-field shift exerted by the OCH2CON protons (empty circles in Figure 2), which is due to their positioning close to the shielding cone of the aromatic nuclei as a consequence of the C=O coordination to the metal ion (Figure 3). Interesting, and not observed for achiral calixarene tetramide ligands, is the splitting of the OCH2CON methylene protons into an AB system (empty circles in Figure 2), which, especially in the case of the Na+ complex, indicates a quite different chemical environment for the two geminal protons and could be the consequence of a quite twisted square antiprism of coordination around the cation [36]. Consistent shifts are observed for the methylene bridge protons (filled circles in Figure 2), which are in line with previous observations but more difficult to be rationalised also as a consequence of a slight conformational rearrangement of the calixarene scaffold. Less important shifts are obviously observed for the pyrrolidine ring protons, which are quite far from the binding region.
Figure 3: Schematic of the inclusion of alkali-metal ions (sodium and potassium) in the polar cavity defined by the acetamide moieties at the lower rim of the calixarene, and its effect on the rigidification of the calixarene scaffold and organisation of iminosugars.
Figure 3: Schematic of the inclusion of alkali-metal ions (sodium and potassium) in the polar cavity defined ...
These results show, therefore, that, in spite of the bulky substituents present on the pyrrolidine nuclei, the amide groups can still bind alkali-metal ions quite efficiently. This encourages the use of such compounds not only to study the metal-ion effect on the organisation of iminosugars and on the rigidification of the calixarene scaffold, but also to exploit the ability of these chiral ligands to enantioselectively recognise chiral salts [37] or the ability of their transition-metal complexes to catalyse enantioselective syntheses [38,39], which are objects of current investigations.
Conclusion
The coupling of low-generation dendrons, based on the chiral C2-symmetric dihydroxypyrrolidine 1, with a calix[4]arene scaffold allowed the construction of novel supramolecular architectures, i.e., iminosugar-analogue-based calixarene dendrimers. The convergent synthetic approach used permits the rapid increase of the valency of the iminosugar presentation. Complexation studies with sodium or potassium picrate, carried out on the calixarene-dendrimer 2, indicated an effective binding of the metal cation at the calixarene lower rim that induces a rigidification of the macrocyclic scaffold and, as a consequence, the convergence of the iminosugars into a restricted region of space. These chiral multivalent dendrimers could thus be interestingly engaged in enantioselective recognition and catalysis. Up to now, pyrrolidine 1 has been used both as a dendrimeric building block and iminosugar model; its substitution at the hydroxy groups with more complex and biologically active iminosugars, such as polyhydroxylated pyrrolidines, would also allow the investigation of the corresponding calixarene dendrimers in interactions with enzymatic receptors.
Supporting Information
| Supporting Information File 1: Experimental procedures; spectroscopic and analytical data. | ||
| Format: PDF | Size: 518.3 KB | Download |
Acknowledgements
Cassa di Risparmio di Firenze is acknowledged for granting a 400 MHz spectrometer. Maurizio Passaponti and Brunella Innocenti (Università degli Studi di Firenze) are acknowledged for technical assistance. MM thanks the Networking Research Center on Bioengineering, Biomaterials and Nanomedicine (CIBER-BBN). AC and MM acknowledge COST action CM1102. AC and FS also acknowledge PRIN200858SA98 for partial support.
References
-
Asano, N.; Nash, R. J.; Molyneux, R. J.; Fleet, G. W. J. Tetrahedron: Asymmetry 2000, 11, 1645–1680. doi:10.1016/S0957-4166(00)00113-0
Return to citation in text: [1] -
Elbein, A. D. Annu. Rev. Biochem. 1987, 56, 497–523. doi:10.1146/annurev.bi.56.070187.002433
Return to citation in text: [1] -
Compain, P.; Martin, O. R. Iminosugars: From Synthesis to Therapeutic Applications; John Wiley & Sons, Ltd: Chichester, U. K., 2007.
Return to citation in text: [1] [2] -
Johns, B. A.; Johnson, C. R. Tetrahedron Lett. 1998, 39, 749–752. doi:10.1016/S0040-4039(97)10616-5
Return to citation in text: [1] -
Lohse, A.; Jensen, K. B.; Lundgren, K.; Bols, M. Bioorg. Med. Chem. 1999, 7, 1965–1971. doi:10.1016/S0968-0896(99)00116-9
Return to citation in text: [1] -
Wennekes, T.; van den Berg, R. J. B. H. N.; Bonger, K. M.; Donker-Koopman, W. E.; Ghisaidoobe, A.; van der Marel, G. A.; Strijland, A.; Aerts, J. M. F. G.; Overkleeft, H. S. Tetrahedron: Asymmetry 2009, 20, 836–846. doi:10.1016/j.tetasy.2009.02.043
Return to citation in text: [1] -
Diot, J.; García-Moreno, M. I.; Gouin, S. G.; Ortiz Mellet, C.; Haupt, K.; Kovensky, J. Org. Biomol. Chem. 2009, 7, 357–363. doi:10.1039/b815408b
Return to citation in text: [1] -
Compain, P.; Decroocq, C.; Iehl, J.; Holler, M.; Hazelard, D.; Mena Barragán, T.; Ortiz Mellet, C.; Nierengarten, J.-F. Angew. Chem., Int. Ed. 2010, 49, 5753–5756. doi:10.1002/anie.201002802
Return to citation in text: [1] [2] -
Decroocq, C.; Rodríguez-Lucena, D.; Russo, V.; Mena Barragán, T.; Ortiz Mellet, C.; Compain, P. Chem.–Eur. J. 2011, 17, 13825–13831. doi:10.1002/chem.201102266
Return to citation in text: [1] [2] -
Kiessling, L. L.; Gestwicki, J. E.; Strong, L. E. Angew. Chem., Int. Ed. 2006, 45, 2348–2368. doi:10.1002/anie.200502794
Return to citation in text: [1] -
Gutsche, C. D. Calixarenes: An Introduction; Royal Society of Chemistry: Cambridge, U. K., 2008.
Return to citation in text: [1] -
Ungaro, R.; Arduini, A.; Casnati, A.; Pochini, A.; Ugozzoli, F. Pure Appl. Chem. 1996, 68, 1213–1218. doi:10.1351/pac199668061213
Return to citation in text: [1] -
Casnati, A.; Sansone, F.; Ungaro, R. Calixarene Receptors in Ion Recognition and Sensing. In Advances in Supramolecular Chemistry; Gokel, G. W., Ed.; Cerberus Press Inc.: Miami, FL, U. S. A., 2003; Vol. 9, pp 165–218.
Return to citation in text: [1] -
Sansone, F.; Baldini, L.; Casnati, A.; Ungaro, R. New J. Chem. 2010, 34, 2715–2728. doi:10.1039/c0nj00285b
Return to citation in text: [1] -
Marra, A.; Scherrmann, M.-C.; Dondoni, A.; Ungaro, R.; Casnati, A.; Minari, P. Angew. Chem., Int. Ed. Engl. 1995, 33, 2479–2481. doi:10.1002/anie.199424791
Return to citation in text: [1] [2] -
Dondoni, A.; Marra, A. Chem. Rev. 2010, 110, 4949–4977. doi:10.1021/cr100027b
Return to citation in text: [1] -
Sansone, F.; Rispoli, G.; Casnati, A.; Ungaro, R. Multivalent Glycocalixarenes. In Synthesis and Biological Applications of Glycoconjugates; Renaudet, O.; Spinelli, N., Eds.; Bentham Science Publishers: eBook, 2011; pp 36–63. doi:10.2174/978160805277611101010036
Return to citation in text: [1] -
Lee, Y. C.; Lee, R. T. Acc. Chem. Res. 1995, 28, 321–327. doi:10.1021/ar00056a001
Return to citation in text: [1] -
Kiessling, L. L.; Pohl, N. L. Chem. Biol. 1996, 3, 71–77. doi:10.1016/S1074-5521(96)90280-X
Return to citation in text: [1] -
Lundquist, J. J.; Toone, E. J. Chem. Rev. 2002, 102, 555–578. doi:10.1021/cr000418f
Return to citation in text: [1] -
André, S.; Sansone, F.; Kaltner, H.; Casnati, A.; Kopitz, J.; Gabius, H.-J.; Ungaro, R. ChemBioChem 2008, 9, 1649–1661. doi:10.1002/cbic.200800035
Return to citation in text: [1] -
André, S.; Grandjean, C.; Gautier, F.-M.; Bernardi, S.; Sansone, F.; Gabius, H.-J.; Ungaro, R. Chem. Commun. 2011, 47, 6126–6128. doi:10.1039/c1cc11163a
Return to citation in text: [1] -
Cecioni, S.; Lalor, R.; Blanchard, B.; Praly, J.-P.; Imberty, A.; Matthews, S. E.; Vidal, S. Chem.–Eur. J. 2009, 15, 13232–13240. doi:10.1002/chem.200901799
Return to citation in text: [1] -
Chabre, Y. M.; Roy, R. Adv. Carbohydr. Chem. Biochem. 2010, 63, 165–393. doi:10.1016/S0065-2318(10)63006-5
Return to citation in text: [1] -
Turnbull, W. B.; Stoddart, J. F. Rev. Mol. Biotechnol. 2002, 90, 231–255. doi:10.1016/S1389-0352(01)00062-9
Return to citation in text: [1] -
Röckendorf, N.; Lindhorst, T. K. Glycodendrimers. In Dendrimers IV. Metal Coordination, Self Assembly, Catalysis; Vögtle, F.; Schalley, C. A., Eds.; Topics in Current Chemistry, Vol. 217; Springer: Berlin, 2001; pp 201–238.
Return to citation in text: [1] -
Roy, R.; Kim, J. M. Angew. Chem., Int. Ed. 1999, 38, 369–372. doi:10.1002/(SICI)1521-3773(19990201)38:3<369::AID-ANIE369>3.0.CO;2-1
Return to citation in text: [1] -
Nagel, U.; Kinzel, E.; Andrade, J.; Prescher, G. Chem. Ber. 1986, 119, 3326–3342. doi:10.1002/cber.19861191112
Return to citation in text: [1] -
Cicchi, S.; Goti, A.; Rosini, C.; Brandi, A. Eur. J. Org. Chem. 1998, 2591–2597. doi:10.1002/(SICI)1099-0690(199811)1998:11<2591::AID-EJOC2591>3.0.CO;2-Q
Return to citation in text: [1] -
Baklouti, L.; Cheriaa, N.; Mahouachi, M.; Abidi, R.; Kom, J. S.; Kim, Y.; Vicens, J. J. Inclusion Phenom. Macrocyclic Chem. 2006, 54, 1–7. doi:10.1007/s10847-005-6994-4
Return to citation in text: [1] -
Arakawa, Y.; Yoshifuji, S. Chem. Pharm. Bull. 1991, 39, 2219–2224. doi:10.1248/cpb.39.2219
Return to citation in text: [1] [2] -
Arduini, A.; Ghidini, E.; Pochini, A.; Ungaro, R.; Andreetti, G. D.; Calestani, G.; Ugozzoli, F. J. Inclusion Phenom. Mol. Recognit. Chem. 1988, 6, 119–134. doi:10.1007/BF00663045
Return to citation in text: [1] -
Arnaud-Neu, F.; Barboso, S.; Berny, F.; Casnati, A.; Muzet, N.; Pinalli, A.; Ungaro, R.; Schwing-Weill, M.-J.; Wipff, G. J. Chem. Soc., Perkin Trans. 2 1999, 1727–1738. doi:10.1039/a902001b
Return to citation in text: [1] -
Arduini, A.; Fabbi, M.; Mantovani, M.; Mirone, L.; Pochini, A.; Secchi, A.; Ungaro, R. J. Org. Chem. 1995, 60, 1454–1457. doi:10.1021/jo00110a055
Return to citation in text: [1] -
Arduini, A.; Pochini, A.; Reverberi, S.; Ungaro, R. Tetrahedron 1986, 42, 2089–2100. doi:10.1016/S0040-4020(01)87626-2
Return to citation in text: [1] -
Casnati, A.; Cavallo, G.; Metrangolo, P.; Resnati, G.; Ugozzoli, F.; Ungaro, R. Chem.–Eur. J. 2009, 15, 7903–7912. doi:10.1002/chem.200900771
Return to citation in text: [1] -
Yakovenko, A. V.; Boyko, V. I.; Kalchenko, V. I.; Baldini, L.; Casnati, A.; Sansone, F.; Ungaro, R. J. Org. Chem. 2007, 72, 3223–3231. doi:10.1021/jo062410x
Return to citation in text: [1] -
Casolari, S.; Cozzi, P. G.; Oriolo, P.; Tagliavini, E.; Umani-Ronchi, A. J. Chem. Soc., Chem. Commun. 1997, 2123–2124. doi:10.1039/a705045c
Return to citation in text: [1] -
Pinkhassik, E.; Stibor, I.; Casnati, A.; Ungaro, R. J. Org. Chem. 1997, 62, 8654–8659. doi:10.1021/jo9706990
Return to citation in text: [1]
| 35. | Arduini, A.; Pochini, A.; Reverberi, S.; Ungaro, R. Tetrahedron 1986, 42, 2089–2100. doi:10.1016/S0040-4020(01)87626-2 |
| 36. | Casnati, A.; Cavallo, G.; Metrangolo, P.; Resnati, G.; Ugozzoli, F.; Ungaro, R. Chem.–Eur. J. 2009, 15, 7903–7912. doi:10.1002/chem.200900771 |
| 37. | Yakovenko, A. V.; Boyko, V. I.; Kalchenko, V. I.; Baldini, L.; Casnati, A.; Sansone, F.; Ungaro, R. J. Org. Chem. 2007, 72, 3223–3231. doi:10.1021/jo062410x |
| 1. | Asano, N.; Nash, R. J.; Molyneux, R. J.; Fleet, G. W. J. Tetrahedron: Asymmetry 2000, 11, 1645–1680. doi:10.1016/S0957-4166(00)00113-0 |
| 2. | Elbein, A. D. Annu. Rev. Biochem. 1987, 56, 497–523. doi:10.1146/annurev.bi.56.070187.002433 |
| 7. | Diot, J.; García-Moreno, M. I.; Gouin, S. G.; Ortiz Mellet, C.; Haupt, K.; Kovensky, J. Org. Biomol. Chem. 2009, 7, 357–363. doi:10.1039/b815408b |
| 18. | Lee, Y. C.; Lee, R. T. Acc. Chem. Res. 1995, 28, 321–327. doi:10.1021/ar00056a001 |
| 19. | Kiessling, L. L.; Pohl, N. L. Chem. Biol. 1996, 3, 71–77. doi:10.1016/S1074-5521(96)90280-X |
| 20. | Lundquist, J. J.; Toone, E. J. Chem. Rev. 2002, 102, 555–578. doi:10.1021/cr000418f |
| 4. | Johns, B. A.; Johnson, C. R. Tetrahedron Lett. 1998, 39, 749–752. doi:10.1016/S0040-4039(97)10616-5 |
| 5. | Lohse, A.; Jensen, K. B.; Lundgren, K.; Bols, M. Bioorg. Med. Chem. 1999, 7, 1965–1971. doi:10.1016/S0968-0896(99)00116-9 |
| 6. | Wennekes, T.; van den Berg, R. J. B. H. N.; Bonger, K. M.; Donker-Koopman, W. E.; Ghisaidoobe, A.; van der Marel, G. A.; Strijland, A.; Aerts, J. M. F. G.; Overkleeft, H. S. Tetrahedron: Asymmetry 2009, 20, 836–846. doi:10.1016/j.tetasy.2009.02.043 |
| 15. | Marra, A.; Scherrmann, M.-C.; Dondoni, A.; Ungaro, R.; Casnati, A.; Minari, P. Angew. Chem., Int. Ed. Engl. 1995, 33, 2479–2481. doi:10.1002/anie.199424791 |
| 3. | Compain, P.; Martin, O. R. Iminosugars: From Synthesis to Therapeutic Applications; John Wiley & Sons, Ltd: Chichester, U. K., 2007. |
| 15. | Marra, A.; Scherrmann, M.-C.; Dondoni, A.; Ungaro, R.; Casnati, A.; Minari, P. Angew. Chem., Int. Ed. Engl. 1995, 33, 2479–2481. doi:10.1002/anie.199424791 |
| 3. | Compain, P.; Martin, O. R. Iminosugars: From Synthesis to Therapeutic Applications; John Wiley & Sons, Ltd: Chichester, U. K., 2007. |
| 16. | Dondoni, A.; Marra, A. Chem. Rev. 2010, 110, 4949–4977. doi:10.1021/cr100027b |
| 17. | Sansone, F.; Rispoli, G.; Casnati, A.; Ungaro, R. Multivalent Glycocalixarenes. In Synthesis and Biological Applications of Glycoconjugates; Renaudet, O.; Spinelli, N., Eds.; Bentham Science Publishers: eBook, 2011; pp 36–63. doi:10.2174/978160805277611101010036 |
| 9. | Decroocq, C.; Rodríguez-Lucena, D.; Russo, V.; Mena Barragán, T.; Ortiz Mellet, C.; Compain, P. Chem.–Eur. J. 2011, 17, 13825–13831. doi:10.1002/chem.201102266 |
| 12. | Ungaro, R.; Arduini, A.; Casnati, A.; Pochini, A.; Ugozzoli, F. Pure Appl. Chem. 1996, 68, 1213–1218. doi:10.1351/pac199668061213 |
| 13. | Casnati, A.; Sansone, F.; Ungaro, R. Calixarene Receptors in Ion Recognition and Sensing. In Advances in Supramolecular Chemistry; Gokel, G. W., Ed.; Cerberus Press Inc.: Miami, FL, U. S. A., 2003; Vol. 9, pp 165–218. |
| 8. | Compain, P.; Decroocq, C.; Iehl, J.; Holler, M.; Hazelard, D.; Mena Barragán, T.; Ortiz Mellet, C.; Nierengarten, J.-F. Angew. Chem., Int. Ed. 2010, 49, 5753–5756. doi:10.1002/anie.201002802 |
| 14. | Sansone, F.; Baldini, L.; Casnati, A.; Ungaro, R. New J. Chem. 2010, 34, 2715–2728. doi:10.1039/c0nj00285b |
| 10. | Kiessling, L. L.; Gestwicki, J. E.; Strong, L. E. Angew. Chem., Int. Ed. 2006, 45, 2348–2368. doi:10.1002/anie.200502794 |
| 38. | Casolari, S.; Cozzi, P. G.; Oriolo, P.; Tagliavini, E.; Umani-Ronchi, A. J. Chem. Soc., Chem. Commun. 1997, 2123–2124. doi:10.1039/a705045c |
| 39. | Pinkhassik, E.; Stibor, I.; Casnati, A.; Ungaro, R. J. Org. Chem. 1997, 62, 8654–8659. doi:10.1021/jo9706990 |
| 8. | Compain, P.; Decroocq, C.; Iehl, J.; Holler, M.; Hazelard, D.; Mena Barragán, T.; Ortiz Mellet, C.; Nierengarten, J.-F. Angew. Chem., Int. Ed. 2010, 49, 5753–5756. doi:10.1002/anie.201002802 |
| 9. | Decroocq, C.; Rodríguez-Lucena, D.; Russo, V.; Mena Barragán, T.; Ortiz Mellet, C.; Compain, P. Chem.–Eur. J. 2011, 17, 13825–13831. doi:10.1002/chem.201102266 |
| 11. | Gutsche, C. D. Calixarenes: An Introduction; Royal Society of Chemistry: Cambridge, U. K., 2008. |
| 24. | Chabre, Y. M.; Roy, R. Adv. Carbohydr. Chem. Biochem. 2010, 63, 165–393. doi:10.1016/S0065-2318(10)63006-5 |
| 25. | Turnbull, W. B.; Stoddart, J. F. Rev. Mol. Biotechnol. 2002, 90, 231–255. doi:10.1016/S1389-0352(01)00062-9 |
| 26. | Röckendorf, N.; Lindhorst, T. K. Glycodendrimers. In Dendrimers IV. Metal Coordination, Self Assembly, Catalysis; Vögtle, F.; Schalley, C. A., Eds.; Topics in Current Chemistry, Vol. 217; Springer: Berlin, 2001; pp 201–238. |
| 21. | André, S.; Sansone, F.; Kaltner, H.; Casnati, A.; Kopitz, J.; Gabius, H.-J.; Ungaro, R. ChemBioChem 2008, 9, 1649–1661. doi:10.1002/cbic.200800035 |
| 22. | André, S.; Grandjean, C.; Gautier, F.-M.; Bernardi, S.; Sansone, F.; Gabius, H.-J.; Ungaro, R. Chem. Commun. 2011, 47, 6126–6128. doi:10.1039/c1cc11163a |
| 23. | Cecioni, S.; Lalor, R.; Blanchard, B.; Praly, J.-P.; Imberty, A.; Matthews, S. E.; Vidal, S. Chem.–Eur. J. 2009, 15, 13232–13240. doi:10.1002/chem.200901799 |
| 32. | Arduini, A.; Ghidini, E.; Pochini, A.; Ungaro, R.; Andreetti, G. D.; Calestani, G.; Ugozzoli, F. J. Inclusion Phenom. Mol. Recognit. Chem. 1988, 6, 119–134. doi:10.1007/BF00663045 |
| 33. | Arnaud-Neu, F.; Barboso, S.; Berny, F.; Casnati, A.; Muzet, N.; Pinalli, A.; Ungaro, R.; Schwing-Weill, M.-J.; Wipff, G. J. Chem. Soc., Perkin Trans. 2 1999, 1727–1738. doi:10.1039/a902001b |
| 34. | Arduini, A.; Fabbi, M.; Mantovani, M.; Mirone, L.; Pochini, A.; Secchi, A.; Ungaro, R. J. Org. Chem. 1995, 60, 1454–1457. doi:10.1021/jo00110a055 |
| 31. | Arakawa, Y.; Yoshifuji, S. Chem. Pharm. Bull. 1991, 39, 2219–2224. doi:10.1248/cpb.39.2219 |
| 31. | Arakawa, Y.; Yoshifuji, S. Chem. Pharm. Bull. 1991, 39, 2219–2224. doi:10.1248/cpb.39.2219 |
| 29. | Cicchi, S.; Goti, A.; Rosini, C.; Brandi, A. Eur. J. Org. Chem. 1998, 2591–2597. doi:10.1002/(SICI)1099-0690(199811)1998:11<2591::AID-EJOC2591>3.0.CO;2-Q |
| 30. | Baklouti, L.; Cheriaa, N.; Mahouachi, M.; Abidi, R.; Kom, J. S.; Kim, Y.; Vicens, J. J. Inclusion Phenom. Macrocyclic Chem. 2006, 54, 1–7. doi:10.1007/s10847-005-6994-4 |
| 27. | Roy, R.; Kim, J. M. Angew. Chem., Int. Ed. 1999, 38, 369–372. doi:10.1002/(SICI)1521-3773(19990201)38:3<369::AID-ANIE369>3.0.CO;2-1 |
| 28. | Nagel, U.; Kinzel, E.; Andrade, J.; Prescher, G. Chem. Ber. 1986, 119, 3326–3342. doi:10.1002/cber.19861191112 |
© 2012 Marradi et al; licensee Beilstein-Institut.
This is an Open Access article under the terms of the Creative Commons Attribution License (http://creativecommons.org/licenses/by/2.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
The license is subject to the Beilstein Journal of Organic Chemistry terms and conditions: (http://www.beilstein-journals.org/bjoc)