Search results
Search for "1,2-addition" in Full Text gives 51 result(s) in Beilstein Journal of Organic Chemistry.
Palladium-catalyzed three-component radical-polar crossover carboamination of 1,3-dienes or allenes with diazo esters and amines
- Geng-Xin Liu,
- Xiao-Ting Jie,
- Ge-Jun Niu,
- Li-Sheng Yang,
- Xing-Lin Li,
- Jian Luo and
- Wen-Hao Hu
Beilstein J. Org. Chem. 2024, 20, 661–671, doi:10.3762/bjoc.20.59
- temperature (rt), the desired unsaturated ε-AA derivative 4a was obtained in 75% isolated yield (Table 1, entry 1). Isolation and NMR analysis demonstrated that this model reaction provided amino acid 4a with good E-selectivity and excellent regioselectivity (E/Z = 91:9, 1,4-/1,2-addition >20:1). Control
- -substituted dienes 2b and 2c were suitable for this MCR, affording the 1,4-addition products 4l and 4m albeit with moderate regioselectivity (1,4-/1,2-addition = 2:1). To our delight, the reactions with 2,3-disubstituted diene 2d and 1,4-disubstituted diene 2e also readily provided products 4n and 4o. In the
- case of 1,3-cyclohexadiene 2e, the amine was expected to attack the π-allyl palladium from the exo side. Considering that substituent effects might affect the regioselectivity in this MCR, we further investigated the 1,4-/1,2-addition selectivity with 1-phenyl-substituted 1,3-dienes 2f–i. Interestingly

Graphical Abstract

Scheme 1: Background (a and b) and proposed carboamination MCR with diazo esters (c). a) Selected bioactive γ...

Scheme 2: Substrate scope of diazo compounds, 1,3-dienes and amines. aReactions (1/2/3/Pd(OAc)2/Xantphos = 0....

Scheme 3: Substrate scope of diazo compounds, allenes and amines. aReactions (1/5/3/Pd(OAc)2/Xantphos = 0.3.0...

Scheme 4: Mechanistic experiments. a) Radical trapping experiments with TEMPO. b) Exclusion of possible inter...

Scheme 5: Proposed mechanisms for the carboamination of 1,3-dienes or allenes with diazo esters and amines.

Scheme 6: Scale-up reactions and synthetic transformations. Reaction conditions: a) LiAlH4, THF, 0 °C; b) MeM...
Trifluoromethylated hydrazones and acylhydrazones as potent nitrogen-containing fluorinated building blocks
- Zhang Dongxu
Beilstein J. Org. Chem. 2023, 19, 1741–1754, doi:10.3762/bjoc.19.127
- -triazolines and their derivatives via tandem 1,2-addition/cyclization reactions between trifluoromethyl acylhydrazones and cyanamide [105] (Scheme 17b). Afterwards, Hu et al. developed a method for the N-arylation and N-alkylation of trifluoromethyl acylhydrazones with diaryliodonium salts and alkyl halides

Graphical Abstract

Scheme 1: Synthesis of trifluoromethylpyrazoles from trifluoroacetaldehyde hydrazones.

Scheme 2: Synthesis of polysubstituted pyrazolidines and pyrazolines.

Scheme 3: Asymmetric synthesis of 3-trifluoromethyl-1,4-dihydropyridazines reported by Rueping et al. [39].

Scheme 4: Synthesis of 3-trifluoromethyl-1,4-dihydropyridazine with Brønsted acid-assisted Lewis base catalys...

Scheme 5: Synthesis of CF3-pyrazoles and CF3-1,6-dihydropyridazines.

Scheme 6: Asymmetric reactions of trifluoromethylimines with organometallic reagents.

Scheme 7: Mannich-type reaction of trifluoroacetaldehyde hydrazones.

Scheme 8: Synthesis of trifluoromethylated hydrazonoyl halides.

Scheme 9: Early work of trifluoromethylated hydrazonoyl halides.

Scheme 10: [3 + 2]/[3 + 3] Cycloadditions of trifluoromethylated hydrazonoyl halides.

Scheme 11: Substrate scope for [3 + 2] cycloadditions with trifluoroacetonitrile imines reported by Jasiński’s...

Scheme 12: Synthesis of trifluoromethylated 1,2,4-triazole and 1,2,4-triazine derivatives.

Scheme 13: [3 + 2] Cycloadditions of difluoromethylated hydrazonoyl halides.

Scheme 14: Preparation and early applications of trifluoromethylated acylhydrazones.

Scheme 15: 1,2-Nucleophilic addition reactions of trifluoromethylated acylhydrazones.

Scheme 16: Cascade oxidation/cyclization reactions of trifluoromethylated homoallylic acylhydrazines.

Scheme 17: Synthesis of trifluoromethylated cyanohydrazines and 3-trifluoromethyl-1,2,4-triazolines.

Scheme 18: N-Arylation and N-alkylation of trifluoromethyl acylhydrazones.

Scheme 19: [3 + 2]-Cycladditions of trifluoromethyl acylhydrazones.
Aromatic C–H bond functionalization through organocatalyzed asymmetric intermolecular aza-Friedel–Crafts reaction: a recent update
- Anup Biswas
Beilstein J. Org. Chem. 2023, 19, 956–981, doi:10.3762/bjoc.19.72
- dehydration to generate isoxazolium cation 80 paired with a phosphate anion. This chiral phosphate is engaged in H-bonding with the free NH of the heteroarene ring to ease the stereoselective 1,2-addition to in situ generate the cationic heterocyclic scaffold 81. The reaction proceeded faster with pyrroles
- Brønsted acid to generate (N-acyl)(propargyl)imine 90 as intermediate which added to the deprotonated phosphoric acid to form phosphate ester 91 as the next intermediate through an equilibrium process. Then, 1,2-addition by the C3 position of the heteroarene ring to the acylimine intermediate afforded the

Graphical Abstract

Scheme 1: First organocatalyzed asymmetric aza-Friedel–Crafts reaction.

Scheme 2: Aza-Friedel–Crafts reaction between indoles and cyclic ketimines.

Scheme 3: Aza-Friedel–Crafts reaction utilizing trifluoromethyldihydrobenzoazepinoindoles as electrophiles.

Scheme 4: Aza-Friedel–Crafts reaction utilizing cyclic N-sulfimines as electrophiles.

Scheme 5: Aza-Friedel–Crafts reaction involving N-unprotected imino ester as electrophile.

Scheme 6: Aza-Friedel–Crafts and lactonization cascade.

Scheme 7: One-pot oxidation and aza-Friedel–Crafts reaction.

Scheme 8: C1 and C2-symmetric phosphoric acids as catalysts.

Scheme 9: Aza-Friedel–Crafts reaction using Nps-iminophosphonates as electrophiles.

Scheme 10: Aza-Friedel–Crafts reaction between indole and α-iminophosphonate.

Scheme 11: [2.2]-Paracyclophane-derived chiral phosphoric acids as catalyst.

Scheme 12: Aza-Friedel–Crafts reaction through ring opening of sulfamidates.

Scheme 13: Isoquinoline-1,3(2H,4H)-dione scaffolds as electrophiles.

Scheme 14: Functionalization of the carbocyclic ring of substituted indoles.

Scheme 15: Aza-Friedel–Crafts reaction between unprotected imines and aza-heterocycles.

Scheme 16: Anilines and α-naphthols as potential nucleophiles.

Scheme 17: Solvent-controlled regioselective aza-Friedel–Crafts reaction.

Scheme 18: Generating central and axial chirality via aza-Friedel–Crafts reaction.

Scheme 19: Reaction between indoles and racemic 2,3-dihydroisoxazol-3-ol derivatives.

Scheme 20: Exploiting 5-aminoisoxazoles as nucleophiles.

Scheme 21: Reaction between unsubstituted indoles and 3-alkynylated 3-hydroxy-1-oxoisoindolines.

Scheme 22: Synthesis of unnatural amino acids bearing an aza-quaternary stereocenter.

Scheme 23: Atroposelective aza-Friedel–Crafts reaction.

Scheme 24: Coupling of 5-aminopyrazole and 3H-indol-3-ones.

Scheme 25: Pyrophosphoric acid-catalyzed aza-Friedel–Crafts reaction on phenols.

Scheme 26: Squaramide-assisted aza-Friedel–Crafts reaction.

Scheme 27: Thiourea-catalyzed aza-Friedel–Crafts reaction.

Scheme 28: Squaramide-catalyzed reaction between β-naphthols and benzothiazolimines.

Scheme 29: Thiourea-catalyzed reaction between β-naphthol and isatin-derived ketamine.

Scheme 30: Quinine-derived molecule as catalyst.

Scheme 31: Cinchona alkaloid as catalyst.

Scheme 32: aza-Friedel–Crafts reaction by phase transfer catalyst.

Scheme 33: Disulfonamide-catalyzed reaction.

Scheme 34: Heterogenous thiourea-catalyzed aza-Friedel–Crafts reaction.

Scheme 35: Total synthesis of (+)-gracilamine.

Scheme 36: Total synthesis of (−)-fumimycin.
Enolates ambushed – asymmetric tandem conjugate addition and subsequent enolate trapping with conventional and less traditional electrophiles
- Péter Kisszékelyi and
- Radovan Šebesta
Beilstein J. Org. Chem. 2023, 19, 593–634, doi:10.3762/bjoc.19.44
- diastereomers were detected (Scheme 9A). Furthermore, the authors have also demonstrated a four-component coupling reaction: by simply increasing the amount of the organolithium reagent (2.05 equiv) used for the activation of the Zn enolate, β-hydroxyketones 40 were gained via 1,2-addition of the zincate

Graphical Abstract

Scheme 1: General scheme depicting tandem reactions based on an asymmetric conjugate addition followed by an ...

Scheme 2: Cu-catalyzed tandem conjugate addition of R2Zn/aldol reaction with chiral acetals.

Scheme 3: Cu-catalyzed asymmetric desymmetrization of cyclopentene-1,3-diones using a tandem conjugate additi...

Scheme 4: Stereocontrolled assembly of dialkylzincs, cyclic enones, and sulfinylimines utilizing a Cu-catalyz...

Scheme 5: Cu-catalyzed tandem conjugate addition/Mannich reaction (A). Access to chiral isoindolinones and tr...

Scheme 6: Cu-catalyzed tandem conjugate addition/nitro-Mannich reaction (A) with syn–anti or syn–syn selectiv...

Figure 1: Various chiral ligands utilized for the tandem conjugate addition/Michael reaction sequences.

Scheme 7: Cu-catalyzed tandem conjugate addition/Michael reaction: side-product formation with chalcone (A) a...

Scheme 8: Zn enolate trapping using allyl iodides (A), Stork–Jung vinylsilane reagents (B), and allyl bromide...

Scheme 9: Cu-catalyzed tandem conjugate addition/acylation through Li R2Zn enolate (A). A four-component coup...

Scheme 10: Selected examples for the Cu-catalyzed tandem conjugate addition/trifluoromethylthiolation sequence....

Scheme 11: Zn enolates trapped by vinyloxiranes: synthesis of allylic alcohols.

Scheme 12: Stereoselective cyclopropanation of Mg enolates formed by ACA of Grignard reagents to chlorocrotona...

Scheme 13: Domino aldol reactions of Mg enolates formed from coumarin and chromone.

Scheme 14: Oxidative coupling of ACA-produced Mg enolates.

Scheme 15: Tandem ACA of Grignard reagents to enones and Mannich reaction.

Scheme 16: Diastereodivergent Mannich reaction of Mg enolates with differently N-protected imines.

Scheme 17: Tandem Grignard–ACA–Mannich using Taddol-based phosphine-phosphite ligands.

Scheme 18: Tandem reaction of Mg enolates with aminomethylating reagents.

Scheme 19: Tandem reaction composed of Grignard ACA to alkynyl enones.

Scheme 20: Rh/Cu-catalyzed tandem reaction of diazo enoates leading to cyclobutanes.

Scheme 21: Tandem Grignard-ACA of cyclopentenones and alkylation of enolates.

Scheme 22: Tandem ACA of Grignard reagents followed by enolate trapping reaction with onium compounds.

Scheme 23: Mg enolates generated from unsaturated lactones in reaction with activated alkenes.

Scheme 24: Lewis acid mediated ACA to amides and SN2 cyclization of a Br-appended enolate.

Scheme 25: Trapping reactions of aza-enolates with Michael acceptors.

Scheme 26: Si enolates generated by TMSOTf-mediated ACA of Grignard reagents and enolate trapping reaction wit...

Scheme 27: Trapping reactions of enolates generated from alkenyl heterocycles (A) and carboxylic acids (B) wit...

Scheme 28: Reactions of heterocyclic Mg enolates with onium compounds.

Scheme 29: Synthetic transformations of cycloheptatrienyl and benzodithiolyl substituents.

Scheme 30: Aminomethylation of Al enolates generated by ACA of trialkylaluminum reagents.

Scheme 31: Trapping reactions of enolates with activated alkenes.

Scheme 32: Alkynylation of racemic aluminum or magnesium enolates.

Scheme 33: Trapping reactions of Zr enolates generated by Cu-ACA of organozirconium reagents.

Scheme 34: Chloromethylation of Zr enolates using the Vilsmeier–Haack reagent.

Scheme 35: Tandem conjugate borylation with subsequent protonation or enolate trapping by an electrophile.

Scheme 36: Tandem conjugate borylation/aldol reaction of cyclohexenones.

Scheme 37: Selected examples for the tandem asymmetric borylation/intramolecular aldol reaction; synthesis of ...

Scheme 38: Cu-catalyzed tandem methylborylation of α,β-unsaturated phosphine oxide in the presence of (R,Sp)-J...

Scheme 39: Cu-catalyzed tandem transannular conjugated borylation/aldol cyclization of macrocycles containing ...

Scheme 40: Stereoselective tandem conjugate borylation/Mannich cyclization: selected examples (A) and a multi-...

Scheme 41: Some examples of Cu-catalyzed asymmetric tandem borylation/aldol cyclization (A). Application to di...

Scheme 42: Atropisomeric P,N-ligands used in tandem conjugate borylation/aldol cyclization sequence.

Scheme 43: Selected examples for the enantioselective Cu-catalyzed borylation/intramolecular Michael addition ...

Scheme 44: Selected examples for the preparation of enantioenriched spiroindanes using a Cu-catalyzed tandem c...

Scheme 45: Enantioselective conjugate borylation of cyclobutene-1-carboxylic acid diphenylmethyl ester 175 wit...

Scheme 46: Cu-catalyzed enantioselective tandem conjugate silylation of α,β-unsaturated ketones with subsequen...

Scheme 47: Cu-catalyzed enantioselective tandem conjugate silylation of α,β-unsaturated ketones with subsequen...

Scheme 48: Cu-catalyzed tandem conjugate silylation/aldol condensation. The diastereoselectivity is controlled...

Scheme 49: Chiral Ru-catalyzed three-component coupling reaction.

Scheme 50: Rh-Phebox complex-catalyzed reductive cyclization and subsequent reaction with Michael acceptors th...

Scheme 51: Rh-catalyzed tandem asymmetric conjugate alkynylation/aldol reaction (A) and subsequent spiro-cycli...

Scheme 52: Rh-bod complex-catalyzed tandem asymmetric conjugate arylation/intramolecular aldol addition (A). S...

Scheme 53: Co-catalyzed C–H-bond activation/asymmetric conjugate addition/aldol reaction.

Scheme 54: (Diisopinocampheyl)borane-promoted 1,4-hydroboration of α,β-unsaturated morpholine carboxamides and...

Figure 2: Some examples of total syntheses that have been recently reviewed.

Scheme 55: Stereoselective synthesis of antimalarial prodrug (+)-artemisinin utilizing a tandem conjugate addi...

Scheme 56: Amphilectane and serrulatane diterpenoids: preparation of chiral starting material via asymmetric t...

Scheme 57: Various asymmetric syntheses of pleuromutilin and related compounds based on a tandem conjugate add...

Scheme 58: Total synthesis of glaucocalyxin A utilizing a tandem conjugate addition/acylation reaction sequenc...

Scheme 59: Installation of the exocyclic double bond using a tandem conjugate addition/aminomethylation sequen...

Scheme 60: Synthesis of the taxol core using a tandem conjugate addition/enolate trapping sequence with Vilsme...

Scheme 61: Synthesis of the tricyclic core of 12-epi-JBIR-23/24 utilizing a Rh-catalyzed asymmetric conjugate ...

Scheme 62: Total synthesis of (−)-peyssonoside A utilizing a Cu-catalyzed enantioselective tandem conjugate ad...
Synthetic study toward tridachiapyrone B
- Morgan Cormier,
- Florian Hernvann and
- Michaël De Paolis
Beilstein J. Org. Chem. 2022, 18, 1741–1748, doi:10.3762/bjoc.18.183
- oxidative anionic oxy-Cope rearrangement of the tertiary alcohol arising from the 1,2-addition of a 1,3-dimethylallyl reagent to 2,5-cyclohexadienone connected to the α’-methoxy-γ-pyrone motif. Keywords: α’-methoxy-γ-pyrone; 2,5-cyclohexadienone; oxy-Cope; quaternary carbon; Robinson-type annulation
- was not the case, the process was poorly reproducible. On the other hand, the 1,2-addition of Grignard reagents to 5 was observed, providing thus an alternative way of grafting a side chain. As summarized in Scheme 7a, this was envisaged through a sequence encompassing the 1,2-addition of the 1,3
- 1,2-addition of a rather hindered nucleophile to a carbonyl electrophile with low reactivity. Precedents were noted though, as Carreira used this strategy for the synthesis of indoxamycine B exploiting the reactivity of 1,3-dimethylallyltitanocene species [39]. The 1,2-addition of various

Graphical Abstract

Scheme 1: Routes to crispatene, photodeoxytridachione, aureothin, and tridachiapyrone B.

Scheme 2: Desymmetrization of 2.

Scheme 3: Addition of lithiocyclopentadiene to pyrone 2.

Scheme 4: Plan to reach 2,5-cyclohexadienone 5.

Scheme 5: Preparation of 2,5-cyclohexadienone 5.

Scheme 6: Attempts to perform the conjugate addition.

Scheme 7: Updated route to tridachiapyrone B.
A recent overview on the synthesis of 1,4,5-trisubstituted 1,2,3-triazoles
- Pezhman Shiri,
- Ali Mohammad Amani and
- Thomas Mayer-Gall
Beilstein J. Org. Chem. 2021, 17, 1600–1628, doi:10.3762/bjoc.17.114
- process displayed good regioselectivity (Scheme 10) [23]. The authors proposed that the reaction may proceed through the mechanism illustrated in Scheme 11. Initially, EDAM 24 is treated with an azide source and subsequently, the intermediate 26 is formed through 1,2-addition reaction. The intermediate 26

Graphical Abstract

Figure 1: Some significant triazole derivatives [8,23-27].

Scheme 1: A general comparison between synthetic routes for disubstituted 1,2,3-triazole derivatives and full...

Scheme 2: Synthesis of formyltriazoles 3 from the treatment of α-bromoacroleins 1 with azides 2.

Scheme 3: A probable mechanism for the synthesis of formyltriazoles 5 from the treatment of α-bromoacroleins 1...

Scheme 4: Synthesis of 1,4,5-trisubstituted 1,2,3-triazoles 8 from the reaction of aryl azides 7 with enamino...

Scheme 5: Proposed mechanism for the synthesis of 1,4,5-trisubstituted 1,2,3-triazoles from the reaction of a...

Scheme 6: Synthesis of 1,4,5-trisubstituted 1,2,3-triazoles 11 from the reaction of primary amines 10 with 1,...

Scheme 7: The proposed mechanism for the synthesis of 1,4,5-trisubstituted 1,2,3-triazoles 11 from the reacti...

Scheme 8: Synthesis of fully decorated 1,2,3-triazoles 19 containing a sulfur-based side chain.

Scheme 9: Mechanism for the formation of fully decorated 1,2,3-triazoles 19 containing a sulfur-based side ch...

Scheme 10: Synthesis of fully decorated 1,2,3-triazole compounds 25 through the regioselective addition and cy...

Scheme 11: A reasonable mechanism for the synthesis of fully decorated 1,2,3-triazole compounds 25 through the...

Scheme 12: Synthesis of 1,4,5-trisubstituted glycosyl-containing 1,2,3-triazole derivatives 30 from the reacti...

Scheme 13: Synthesis of 1,4,5-trisubstituted 1,2,3-triazoles 34 via intramolecular cyclization reaction of ket...

Scheme 14: Synthesis of fully decorated 1,2,3-triazoles 38 from the reaction of aldehydes 35, amines 36, and α...

Scheme 15: A reasonable mechanism for the synthesis of fully decorated 1,2,3-triazoles 38 from the reaction of...

Scheme 16: Synthesis of functionally rich double C- and N-vinylated 1,2,3-triazoles 45 and 47.

Scheme 17: Synthesis of disubstituted 4-chloro-, 4-bromo-, and 4-iodo-1,2,3-triazoles 50.

Scheme 18: a) A general route for SPAAC in polymer chemistry and b) synthesis of a novel pH-sensitive polymeri...

Scheme 19: Synthesis of 5-allenyl-1,2,3-triazoles 60 by the treatment of alkynes 57, azides 58, and propargyli...

Scheme 20: A reasonable mechanism for the synthesis of 5-allenyl-1,2,3-triazoles 60 by the treatment of alkyne...

Scheme 21: Synthesis of 5‐alkynyl-1,2,3-triazoles 69.

Scheme 22: A reasonable mechanism for the synthesis of 5‐alkynyl-1,2,3-triazoles 69.

Scheme 23: Synthesis of sulfur-cycle-fused 1,2,3-triazoles 75 and 77.

Scheme 24: A reasonable mechanism for the synthesis of sulfur-cycle-fused 1,2,3‐triazoles 75 and 77.

Scheme 25: Synthesis of 5-selanyltriazoles 85 from the reaction of ethynylstibanes 82, organic azides 83, and ...

Scheme 26: A mechanism for the synthesis of 5-selanyltriazoles 85 from the reaction of ethynylstibanes 82, org...

Scheme 27: Synthesis of trisubstituted triazoles containing an Sb substituent at position C5 in 93 and 5-unsub...

Scheme 28: Synthesis of asymmetric triazole disulfides 98 from disulfide-containing tert-butyltosyl disulfide 97...

Scheme 29: A mechanism for the synthesis of asymmetric triazole disulfides 98 from disulfide-containing tert-bu...

Scheme 30: Synthesis of triazole-fused sultams 104.

Scheme 31: Synthesis of 1,2,3-triazole-fused tricyclic heterocycles 106.

Scheme 32: A reasonable mechanism for the synthesis of 1,2,3-triazole-fused tricyclic heterocycles 106.

Scheme 33: Synthesis of 5-aryl-substituted 1,2,3-triazole derivatives 112.

Scheme 34: A reasonable mechanism for the synthesis of 5-aryl-substituted 1,2,3-triazole derivatives 112.

Scheme 35: Synthesis of 1,4,5-trisubstituted 1,2,3-triazole-5-carboxamides 119.

Scheme 36: A probable mechanism for the synthesis of 1,4,5-trisubstituted 1,2,3-triazole-5-carboxamides 119.

Scheme 37: Synthesis of fully decorated triazoles 125 via the Pd/C-catalyzed arylation of disubstituted triazo...

Scheme 38: Synthesis of triazolo[1,5-a]indolones 131.

Scheme 39: Synthesis of unsymmetrically substituted triazole-fused enediyne systems 135 and 5-aryl-4-ethynyltr...

Scheme 40: Synthesis of Pd/Cu-BNP 139 and application of 139 in the synthesis of polycyclic triazoles 142.

Scheme 41: A probable mechanism for the synthesis of polycyclic triazoles 142.

Scheme 42: Synthesis of highly functionalized 1,2,3-triazole-fused 5-, 6-, and 7-membered rings 152–154.

Scheme 43: A probable mechanism for the synthesis of highly functionalized 1,2,3-triazole-fused 5-, 6-, and 7-...

Scheme 44: Synthesis of fully functionalized 1,2,3-triazolo-fused chromenes 162, 164, and 166 via the intramol...

Scheme 45: Ru-catalyzed synthesis of fully decorated triazoles 172.

Scheme 46: Synthesis of 4-cyano-1,2,3-triazoles 175.

Scheme 47: Synthesis of functionalized triazoles from the reaction of 1-alkyltriazenes 176 and azides 177 and ...

Scheme 48: Mechanism for the synthesis of functionalized triazoles from the reaction of 1-alkyltriazenes 176 a...
N-tert-Butanesulfinyl imines in the asymmetric synthesis of nitrogen-containing heterocycles
- Joseane A. Mendes,
- Paulo R. R. Costa,
- Miguel Yus,
- Francisco Foubelo and
- Camilla D. Buarque
Beilstein J. Org. Chem. 2021, 17, 1096–1140, doi:10.3762/bjoc.17.86

- solvent, metal and additives in 1,2-addition reactions to N-tert-butanesulfinyl imines of organometallic compounds, different transition models have been proposed to explain the stereochemical outcomes. The cyclic model justified by the Zimmermann–Traxler transition state [40][41][42] is the typical

Graphical Abstract

Scheme 1: General strategy for the enantioselective synthesis of N-containing heterocycles from N-tert-butane...

Scheme 2: Methodologies for condensation of aldehydes and ketones with tert-butanesulfinamides (1).

Scheme 3: Transition models for cis-aziridines and trans-aziridines.

Scheme 4: Mechanism for the reduction of N-tert-butanesulfinyl imines.

Scheme 5: Transition models for the addition of organomagnesium and organolithium compounds to N-tert-butanes...

Scheme 6: Synthesis of 2,2-dibromoaziridines 15 from aldimines 14 and bromoform, and proposed non-chelation-c...

Scheme 7: Diastereoselective synthesis of aziridines from tert-butanesulfinyl imines.

Scheme 8: Synthesis of vinylaziridines 22 from aldimines 14 and 1,3-dibromopropene 23, and proposed chelation...

Scheme 9: Synthesis of vinylaziridines 27 from aldimines 14 and α-bromoesters 26, and proposed transition sta...

Scheme 10: Synthesis of 2-chloroaziridines 28 from aldimines 14 and dichloromethane, and proposed transition s...

Scheme 11: Synthesis of cis-vinylaziridines 30 and 31 from aldimines 14 and bromomethylbutenolide 29.

Scheme 12: Synthesis of 2-chloro-2-aroylaziridines 36 and 32 from aldimines 14, arylnitriles 34, and silyldich...

Scheme 13: Synthesis of trifluoromethylaziridines 39 and proposed transition state of the aziridination.

Scheme 14: Synthesis of aziridines 42 and proposed state transition.

Scheme 15: Synthesis of 1-substituted 2-azaspiro[3.3]heptanes, 1-phenyl-2-azaspiro[3.4]octane and 1-phenyl-2-a...

Scheme 16: Synthesis of 1-substituted 2,6-diazaspiro[3.3]heptanes 48 from chiral imines 14 and 1-Boc-azetidine...

Scheme 17: Synthesis of β-lactams 52 from chiral imines 14 and dimethyl malonate (49).

Scheme 18: Synthesis of spiro-β-lactam 57 from chiral (RS)-N-tert-butanesulfinyl isatin ketimine 53 and ethyl ...

Scheme 19: Synthesis of β-lactam 60, a precursor of (−)-batzelladine D (61) and (−)-13-epi-batzelladine D (62)...

Scheme 20: Rhodium-catalyzed asymmetric synthesis of 3-substituted pyrrolidines 66 from chiral imine (RS)-63 a...

Scheme 21: Asymmetric synthesis of 1,3-disubstituted isoindolines 69 and 70 from chiral imine 67.

Scheme 22: Asymmetric synthesis of cis-2,5-disubstituted pyrrolidines 73 from chiral imine (RS)-71.

Scheme 23: Asymmetric synthesis of 3-hydroxy-5-substituted pyrrolidin-2-ones 77 from chiral imine (RS)-74.

Scheme 24: Asymmetric synthesis of 4-hydroxy-5-substituted pyrrolidin-2-ones 80 from chiral imines 79.

Scheme 25: Asymmetric synthesis of 3-pyrrolines 82 from chiral imines 14 and ethyl 4-bromocrotonate (81).

Scheme 26: Asymmetric synthesis of γ-amino esters 84, and tetramic acid derivative 86 from chiral imines (RS)-...

Scheme 27: Asymmetric synthesis of α-methylene-γ-butyrolactams 90 from chiral imines (Z,SS)-87 and ethyl 2-bro...

Scheme 28: Asymmetric synthesis of methylenepyrrolidines 92 from chiral imines (RS)-14 and 2-(trimethysilylmet...

Scheme 29: Synthesis of dibenzoazaspirodecanes from cyclic N-tert-butanesulfinyl imines.

Scheme 30: Stereoselective synthesis of cyclopenta[c]proline derivatives 103 from β,γ-unsaturated α-amino acid...

Scheme 31: Stereoselective synthesis of alkaloids (−)-angustureine (107) and (−)-cuspareine (108).

Scheme 32: Stereoselective synthesis of alkaloids (−)-pelletierine (112) and (+)-coniine (117).

Scheme 33: Synthesis of piperidine alkaloids (+)-dihydropinidine (122a), (+)-isosolenopsin (122b) and (+)-isos...

Scheme 34: Stereoselective synthesis of the alkaloids(+)-sedamine (125) from chiral imine (SS)-119.

Scheme 35: Stereoselective synthesis of trans-5-hydroxy-6-substituted-2-piperidinones 127 and 129 from chiral ...

Scheme 36: Stereoselective synthesis of trans-5-hydroxy-6-substituted ethanone-2-piperidinones 132 from chiral...

Scheme 37: Stereoselective synthesis of trans-3-benzyl-5-hydroxy-6-substituted-2-piperidinones 136 from chiral...

Scheme 38: Stereoselective synthesis of trans-5-hydroxy-6-substituted 2-piperidinones 139 from chiral imine 138...

Scheme 39: Stereoselective synthesis of ʟ-hydroxypipecolic acid 145 from chiral imine 144.

Scheme 40: Synthesis of 1-substituted isoquinolones 147, 149 and 151.

Scheme 41: Stereoselective synthesis of 3-substituted dihydrobenzo[de]isoquinolinones 154.

Scheme 42: Enantioselective synthesis of alkaloids (S)-1-benzyl-6,7-dimethoxy-1,2,3,4-tetrahydroisoquinoline (...

Scheme 43: Enantioselective synthesis of alkaloids (−)-cermizine B (171) and (+)-serratezomine E (172) develop...

Scheme 44: Stereoselective synthesis of (+)-isosolepnosin (177) and (+)-solepnosin (178) from homoallylamine d...

Scheme 45: Stereoselective synthesis of tetrahydroquinoline derivatives 184, 185 and 187 from chiral imines (RS...

Scheme 46: Stereoselective synthesis of pyridobenzofuran and pyridoindole derivatives 193 from homopropargylam...

Scheme 47: Stereoselective synthesis of 2-substituted 1,2,5,6-tetrahydropyridines 196 from chiral imines (RS)-...

Scheme 48: Stereoselective synthesis of 2-substituted trans-2,6-disubstituted piperidine 199 from chiral imine...

Scheme 49: Stereoselective synthesis of cis-2,6-disubstituted piperidines 200, and alkaloid (+)-241D, from chi...

Scheme 50: Stereoselective synthesis of 6-substituted piperidines-2,5-diones 206 and 1,7-diazaspiro[4.5]decane...

Scheme 51: Stereoselective synthesis of spirocyclic oxindoles 210 from chiral imines (RS)-53.

Scheme 52: Stereoselective synthesis of azaspiro compound 213 from chiral imine 211.

Scheme 53: Stereoselective synthesis of tetrahydroisoquinoline derivatives from chiral imines (RS)-214.

Scheme 54: Stereoselective synthesis of (−)-crispine A 223 from chiral imine (RS)-214.

Scheme 55: Synthesis of (−)-harmicine (228) using tert-butanesulfinamide through haloamide cyclization.

Scheme 56: Stereoselective synthesis of tetraponerines T1–T8.

Scheme 57: Stereoselective synthesis of phenanthroindolizidines 246a and (−)-tylophorine (246b), and phenanthr...

Scheme 58: Stereoselective synthesis of indoline, tetrahydroquinoline and tetrahydrobenzazepine derivatives 253...

Scheme 59: Stereoselective synthesis of (+)-epohelmin A (258) and (+)-epohelmin B (260) from aldimine (RS)-79.

Scheme 60: Stereoselective synthesis of (−)-epiquinamide (266) from chiral aldimine (SS)-261.

Scheme 61: Synthesis synthesis of (–)-hippodamine (273) and (+)-epi-hippodamine (272) using chiral sulfinyl am...

Scheme 62: Stereoselective synthesis of (+)-grandisine D (279) and (+)-amabiline (283).

Scheme 63: Stereoselective synthesis of (−)-epiquinamide (266) and (+)-swaisonine (291) from aldimine (SS)-126....

Scheme 64: Stereoselective synthesis of (+)-C(9a)-epi-epiquinamide (294).

Scheme 65: Stereoselective synthesis of (+)-lasubine II (298) from chiral aldimine (SS)-109.

Scheme 66: Stereoselective synthesis of (−)-epimyrtine (300a) and (−)-lasubine II (ent-302) from β-amino keton...

Scheme 67: Stereoselective synthesis of (−)-tabersonine (310), (−)-vincadifformine (311), and (−)-aspidospermi...

Scheme 68: Stereoselective synthesis of (+)-epohelmin A (258) and (+)-epohelmin B (260) from aldehyde 313 and ...

Scheme 69: Total synthesis of (+)-lysergic acid (323) from N-tert-butanesulfinamide (RS)-1.
Synthetic approaches to bowl-shaped π-conjugated sumanene and its congeners
- Shakeel Alvi and
- Rashid Ali
Beilstein J. Org. Chem. 2020, 16, 2212–2259, doi:10.3762/bjoc.16.186

- parent sumanene (2). Later on, they transformed trione 40 into the corresponding exo-trimethyl derivative 44 by means of a Grignard reaction with MeMgBr via 1,2-addition (Scheme 7). The imination of monoketosumanene 38 as well as triketosumanene 40 has also been reported by these authors to obtain the

Graphical Abstract

Figure 1: Representation of corannulene (1) and sumanene (2), the subunits of fullerene (C60).

Scheme 1: Mehta’s unsuccessful effort for the synthesis of sumanene scaffold 2.

Scheme 2: First synthesis of sumanene 2 by Sakurai et al. from norbornadiene 10.

Scheme 3: Synthesis of trimethylsumanene 28 from easily accessible norbornadiene (10).

Scheme 4: Generation of anions 29–31 and the preparation of tris(trimethylsilyl)sumanene 32.

Scheme 5: Synthesis of tri- and hexa-substituted sumanene derivatives.

Scheme 6: Synthesis of bowl-shaped π-extended sumanene derivatives 37a–f.

Scheme 7: Synthesis of monooxasumanene 38, trioxosumanene 40 along with imination of them.

Scheme 8: Synthesis of trimethylsumanenetrione 46 and exo-functionalized products 45a,b.

Scheme 9: Synthesis of bisumanenylidene 47 and sumanene dimer 48 from 2.

Scheme 10: The mono-substitution of 2 to generate diverse mono-sumanene derivatives 49a–d.

Scheme 11: Synthesis of sumanene building block 53 useful for further extension.

Scheme 12: Synthesis of hexafluorosumanene derivative 55 by Sakurai and co-workers.

Scheme 13: Preparation of sumanene-based carbene 60 and its reaction with cyclohexane.

Scheme 14: Barton–Kellogg reaction for the synthesis of sterically hindered alkenes.

Scheme 15: Synthesis of hydroxysumanene 68 by employing Baeyer–Villiger oxidation.

Scheme 16: Synthesis of sumanene derivatives having functionality at an internal carbon.

Scheme 17: Mechanism for nucleophilic substitution reaction at the internal carbon.

Scheme 18: Synthesis of diverse monosubstituted sumanene derivatives.

Scheme 19: Synthesis of di- and trisubstituted sumanene derivatives from sumanene (2).

Scheme 20: Preparation of monochlorosumanene 88 and hydrogenation of sumanene (2).

Scheme 21: The dimer 90 and bissumanenyl 92 achieved from halosumannes.

Scheme 22: Pyrenylsumanene 93 involving the Suzuki-coupling as a key transformation.

Scheme 23: Synthesis of various hexaarylsumanene derivatives using the Suzuki-coupling reaction.

Scheme 24: Synthesis of hexasubstituted sumanene derivatives 96 and 97.

Scheme 25: Synthesis of thioalkylsumanenes via an aromatic nucleophilic substitution reaction.

Scheme 26: Synthesis of tris(ethoxycarbonylethenyl)sumanene derivative 108.

Scheme 27: Synthesis of ferrocenyl-based sumanene derivatives.

Scheme 28: Synthesis of sumanenylferrocene architectures 118 and 119 via Negishi coupling.

Scheme 29: Diosmylation and the synthesis of phenylboronate ester 121 of sumanene.

Scheme 30: Synthesis of the iron-complex of sumanene.

Scheme 31: Synthesis of tri- and mononuclear sumanenyl zirconocene complexes.

Scheme 32: Synthesis of [CpRu(η6-sumanene)]PF6.

Scheme 33: Preparation of sumanene-based porous coordination networks 127 (spherical tetramer units) and 128 (...

Scheme 34: Synthesis of sumanenylhafnocene complexes 129 and 130.

Scheme 35: Synthesis of 134 and 135 along with PdII coordination complex 136.

Scheme 36: Synthesis of alkali metals sumanene complex K7(C21H102−)2(C21H93−)·8THF (137) containing di- and tr...

Scheme 37: The encapsulation of a Cs+ ion between two sumanenyl anions.

Scheme 38: Synthesis of monothiasumanene 140 and dithiasumanene 141 from 139.

Scheme 39: Synthesis of trithiasumanene 151 by Otsubo and his co-workers.

Scheme 40: Synthesis of trithiasumanene derivatives 155 and 156.

Scheme 41: Synthetic route towards hexathiolated trithiasumanenes 158.

Scheme 42: Synthesis of triselenasumanene 160 by Shao and teammates.

Scheme 43: Synthesis of tritellurasumanene derivatives from triphenylene skeletons.

Scheme 44: Synthesis of pyrazine-fused sumanene architectures through condensation reaction.

Scheme 45: Treatment of the trichalcogenasumanenes with diverse oxidative reagents.

Scheme 46: Ring-opening reaction with H2O2 and oxone of heterasumanenes 178 and 179.

Scheme 47: Synthesis of polycyclic compounds from sumanene derivatives.

Scheme 48: Synthesis of diimide-based heterocycles reported by Shao’s and co-workers.

Scheme 49: Synthesis of pristine trichalcogenasumanenes, 151, 205, and 206.

Scheme 50: Synthesis of trichalcogenasumanenes via hexaiodotriphenylene precursor 208.

Scheme 51: Synthesis of trisilasumanenes 214 and 215.

Scheme 52: Synthesis of trisilasumanene derivatives 218 and 219.

Scheme 53: Synthesis of novel trigermasumanene derivative 223.

Scheme 54: An attempt towards the synthesis of tristannasumanene derivative 228.

Scheme 55: Synthesis of triphosphasumanene trisulfide 232 from commercially available 229.

Scheme 56: The doping of sumanene derivatives with chalcogens (S, Se, Te) and phosphorus.

Scheme 57: Synthesis of heterasumanene containing three different heteroatoms.

Scheme 58: Synthesis of trichalcogenasumanene derivatives 240 and 179.

Scheme 59: Preparation of trichalcogenasumanenes 245 and 248.

Scheme 60: Design and synthesis of trichalcogenasumanene derivatives 252 and 178.

Scheme 61: Synthesis of spirosumanenes 264–269 and non-spiroheterasumanenes 258–263.

Scheme 62: Synthesis of sumanene-type hetero polycyclic compounds.

Scheme 63: Synthesis of triazasumanenes 288 and its sulfone congener 287.

Scheme 64: Synthesis of C3-symmetric chiral triaryltriazasumanenes via cross-coupling reaction.

Scheme 65: Synthesis of mononaphthosumanene 293 using Suzuki coupling as a key step.

Scheme 66: Synthesis of di- and trinaphthosumanene derivatives 302–304.

Scheme 67: Synthesis of hemifullerene skeletons by Hirao’s group.

Scheme 68: Design and construction of C70 fragment from a C60 sumanene fragment.
Tuneable access to indole, indolone, and cinnoline derivatives from a common 1,4-diketone Michael acceptor
- Dalel El-Marrouki,
- Sabrina Touchet,
- Abderrahmen Abdelli,
- Hédi M’Rabet,
- Mohamed Lotfi Efrit and
- Philippe C. Gros
Beilstein J. Org. Chem. 2020, 16, 1722–1731, doi:10.3762/bjoc.16.144
- % yield. We assume that the tertiary amine would interact with the protonated intermediate, and thus promoting the 1,2-addition (Scheme 6). However, for the pyridine substituent (compounds 6k and 6l), another intermediate may be involved as the shape of this diamine does not allow enough flexibility to
- , only the indolone derivative is obtained in those conditions as well (compound 7i, Scheme 7). Based on these results, we found it important to check whether the indole 6b resulted from a 1,2-addition and not from a degradation of the indolone 7b. For this purpose, the indolone 7b was refluxed overnight
- with acetic acid in toluene, under these conditions producing mainly the indole (Table 1, entry 5). The indolone 7b was found unchanged, with no trace of the indole 6b being detected (see Supporting Information File 1, chapter I), indicating that the indole was formed intramolecularly by a 1,2-addition

Graphical Abstract

Figure 1: Examples of bioactive nitrogen-containing heterocycles (indole [9], indolone [10], and cinnoline [11] derivati...

Scheme 1: General strategy to access indole, indolone, and cinnoline derivatives from 1,4-diketones.

Scheme 2: Synthesis of the 1,4-diketones 5a–k via the Nef reaction or the Wittig reaction. i) HCHO (aq), DMAP...

Scheme 3: Mechanism of the formation of indole and indolone derivatives.

Scheme 4: Synthesis of the indoles 6a–f and the corresponding side product indolones 7a–f.

Scheme 5: Reaction of 5b with a diamine.

Scheme 6: Synthesis of the indoles 6h–l.

Scheme 7: Synthesis of the indolone derivatives 7b, 7d, and 7g–k.

Scheme 8: Synthesis of the cinnoline derivatives 8a–k.

Scheme 9: Proposed mechanism for the preparation of the compounds 6, 7, and 8.
In silico rationalisation of selectivity and reactivity in Pd-catalysed C–H activation reactions
- Liwei Cao,
- Mikhail Kabeshov,
- Steven V. Ley and
- Alexei A. Lapkin
Beilstein J. Org. Chem. 2020, 16, 1465–1475, doi:10.3762/bjoc.16.122
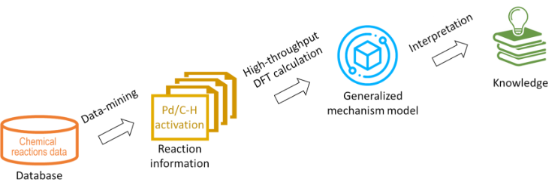
- , explaining the experimental observations for C–H activation reactions, depending on the nature of a ligand (Ln) and transition metal (M) in the catalytically active species (LnM). These mechanisms include four elementary steps: oxidative addition, σ-bond metathesis, electrophilic substitution and 1,2
- -addition, respectively [15]. Even though the mechanisms are inherently different, three most important aspects should be primarily taken into account when classifying and rationalising C–H activation reactions: the proximity of C–H bond to the transition metal; the energy of C–H bond cleavage within the

Graphical Abstract

Figure 1: An approximate energy map for the electrophilic aromatic substitution mechanism.

Scheme 1: Schematic representation of the two mechanisms of Pd-catalysed C–H activation reaction considered i...
Copper-catalysed alkylation of heterocyclic acceptors with organometallic reagents
- Yafei Guo and
- Syuzanna R. Harutyunyan
Beilstein J. Org. Chem. 2020, 16, 1006–1021, doi:10.3762/bjoc.16.90
- aluminium reagents and commercial alkylaluminium reagents were examined in this methodology, providing the corresponding products with a moderate yield but high enantioselectivity (Scheme 2B). In 2009, Feringa and co-workers presented the first highly enantioselective 1,2-addition of dialkylzinc reagents to

Graphical Abstract

Scheme 1: Copper-catalysed ACA of organometallics to piperidones. A) addition of organozinc reagents; B) addi...

Scheme 2: Copper-catalysed ACA of alkenylalanes to N-substituted-2,3-dehydro-4-piperidones.

Scheme 3: Copper-catalysed asymmetric addition of dialkylzinc reagents to N-acyl-4-methoxypyridinium salts fo...

Scheme 4: Copper-catalysed ACA of organozirconium reagents to N-substituted 2,3-dehydro-4-piperidones and lac...

Scheme 5: Copper-catalysed ACA of Grignard reagents to chromones and coumarins and further derivatisation of ...

Scheme 6: Copper-catalysed ACA of Grignard reagents to N-protected quinolones.

Scheme 7: Copper-catalysed ACAs of organometallics to conjugated unsaturated lactams.

Scheme 8: Copper-catalysed ACA of Et2Zn to 5,6-dihydro-2-pyranone.

Scheme 9: Copper-catalysed ACA of Grignard reagents to pyranone and 5,6-dihydro-2-pyranone.

Scheme 10: Copper-catalysed AAA of an organozirconium reagent to heterocyclic acceptors.

Scheme 11: Copper-catalysed ring opening of an oxygen-bridged substrate with trialkylaluminium reagents.

Scheme 12: Copper-catalysed ring opening of oxabicyclic substrates with organolithium reagents (selected examp...

Scheme 13: Copper-catalysed ring opening of polycyclic meso hydrazines.

Scheme 14: Copper-catalysed ACA of Grignard reagents to alkenyl-substituted aromatic N-heterocycles.

Scheme 15: Copper-catalysed ACA of Grignard reagents to β-substituted alkenylpyridines.

Scheme 16: Copper-catalysed ACA of organozinc reagents to alkylidene Meldrum’s acids.
Recent advances in Cu-catalyzed C(sp3)–Si and C(sp3)–B bond formation
- Balaram S. Takale,
- Ruchita R. Thakore,
- Elham Etemadi-Davan and
- Bruce H. Lipshutz
Beilstein J. Org. Chem. 2020, 16, 691–737, doi:10.3762/bjoc.16.67

- 46). The product featuring the PhMe2Si residue at the β-location 287, however, arises by way of a 1,2-addition to an imine, formed from the same Pd(II) intermediate via elimination [84]. Oestrich and co-workers have recently demonstrated non-directed, asymmetric syn-addition-silylations of 3,3
- an appropriate electrophile led to C–C bond formation, ultimately delivering chiral tertiary alcohols. Mechanistic studies and DFT calculations showed that an in situ-formed borylcopper(I) species is responsible for the 1,2-addition (Scheme 73) [136]. C,O-Diboration of ketones 464 was explored using

Graphical Abstract

Scheme 1: Pharmaceuticals possessing a silicon or boron atom.

Scheme 2: The first Cu-catalyzed C(sp3)–Si bond formation.

Scheme 3: Conversion of benzylic phosphate 6 to the corresponding silane.

Scheme 4: Conversion of alkyl triflates to alkylsilanes.

Scheme 5: Conversion of secondary alkyl triflates to alkylsilanes.

Scheme 6: Conversion of alkyl iodides to alkylsilanes.

Scheme 7: Trapping of intermediate radical through cascade reaction.

Scheme 8: Radical pathway for conversion of alkyl iodides to alkylsilanes.

Scheme 9: Conversion of alkyl ester of N-hydroxyphthalimide to alkylsilanes.

Scheme 10: Conversion of gem-dibromides to bis-silylalkanes.

Scheme 11: Conversion of imines to α-silylated amines (A) and the reaction pathway (B).

Scheme 12: Conversion of N-tosylimines to α-silylated amines.

Scheme 13: Screening of diamine ligands.

Scheme 14: Conversion of N-tert-butylsulfonylimines to α-silylated amines.

Scheme 15: Conversion of aldimines to nonracemic α-silylated amines.

Scheme 16: Conversion of N-tosylimines to α-silylated amines.

Scheme 17: Reaction pathway [A] and conversion of aldehydes to α-silylated alcohols [B].

Scheme 18: Conversion of aldehydes to benzhydryl silyl ethers.

Scheme 19: Conversion of ketones to 1,2-diols (A) and conversion of imines to 1,2-amino alcohols (B).

Scheme 20: Ligand screening (A) and conversion of aldehydes to α-silylated alcohols (B).

Scheme 21: Conversion of aldehydes to α-silylated alcohols.

Scheme 22: 1,4-Additions to α,β-unsaturated ketones.

Scheme 23: 1,4-Additions to unsaturated ketones to give β-silylated derivatives.

Scheme 24: Additions onto α,β-unsaturated lactones to give β-silylated lactones.

Scheme 25: Conversion of α,β-unsaturated to β-silylated lactams.

Scheme 26: Conversion of N-arylacrylamides to silylated oxindoles.

Scheme 27: Conversion of α,β-unsaturated carbonyl compounds to silylated tert-butylperoxides.

Scheme 28: Catalytic cycle for Cu(I) catalyzed α,β-unsaturated compounds.

Scheme 29: Conversion of p-quinone methides to benzylic silanes.

Scheme 30: Conversion of α,β-unsaturated ketimines to regio- and stereocontrolled allylic silanes.

Scheme 31: Conversion of α,β-unsaturated ketimines to enantioenriched allylic silanes.

Scheme 32: Regioselective conversion of dienedioates to allylic silanes.

Scheme 33: Conversion of alkenyl-substituted azaarenes to β-silylated adducts.

Scheme 34: Conversion of conjugated benzoxazoles to enantioenriched β-silylated adducts.

Scheme 35: Conversion of α,β-unsaturated carbonyl indoles to α-silylated N-alkylated indoles.

Scheme 36: Conversion of β-amidoacrylates to α-aminosilanes.

Scheme 37: Conversion of α,β-unsaturated ketones to enantioenriched β-silylated ketones, nitriles, and nitro d...

Scheme 38: Regio-divergent silacarboxylation of allenes.

Scheme 39: Silylation of diazocarbonyl compounds, (A) asymmetric and (B) racemic.

Scheme 40: Enantioselective hydrosilylation of alkenes.

Scheme 41: Conversion of 3-acylindoles to indolino-silanes.

Scheme 42: Proposed mechanism for the silylation of 3-acylindoles.

Scheme 43: Silyation of N-chlorosulfonamides.

Scheme 44: Conversion of acyl silanes to α-silyl alcohols.

Scheme 45: Conversion of N-tosylaziridines to β-silylated N-tosylamines.

Scheme 46: Conversion of N-tosylaziridines to silylated N-tosylamines.

Scheme 47: Conversion of 3,3-disubstituted cyclopropenes to silylated cyclopropanes.

Scheme 48: Conversion of conjugated enynes to 1,3-bis(silyl)propenes.

Scheme 49: Proposed sequence for the Cu-catalyzed borylation of substituted alkenes.

Scheme 50: Cu-catalyzed synthesis of nonracemic allylic boronates.

Scheme 51: Cu–NHC catalyzed synthesis of α-substituted allylboronates.

Scheme 52: Synthesis of α-chiral (γ-alkoxyallyl)boronates.

Scheme 53: Cu-mediated formation of nonracemic cis- or trans- 2-substituted cyclopropylboronates.

Scheme 54: Cu-catalyzed synthesis of γ,γ-gem-difluoroallylboronates.

Scheme 55: Cu-catalyzed hydrofunctionalization of internal alkenes and vinylarenes.

Scheme 56: Cu-catalyzed Markovnikov and anti-Markovnikov borylation of alkenes.

Scheme 57: Cu-catalyzed borylation/ortho-cyanation/Cope rearrangement.

Scheme 58: Borylfluoromethylation of alkenes.

Scheme 59: Cu-catalyzed synthesis of tertiary nonracemic alcohols.

Scheme 60: Synthesis of densely functionalized and synthetically versatile 1,2- or 4,3-borocyanated 1,3-butadi...

Scheme 61: Cu-catalyzed trifunctionalization of allenes.

Scheme 62: Cu-catalyzed selective arylborylation of arenes.

Scheme 63: Asymmetric borylative coupling between styrenes and imines.

Scheme 64: Regio-divergent aminoboration of unactivated terminal alkenes.

Scheme 65: Cu-catalyzed 1,4-borylation of α,β-unsaturated ketones.

Scheme 66: Cu-catalyzed protodeboronation of α,β-unsaturated ketones.

Scheme 67: Cu-catalyzed β-borylation of α,β-unsaturated imines.

Scheme 68: Cu-catalyzed synthesis of β-trifluoroborato carbonyl compounds.

Scheme 69: Asymmetric 1,4-borylation of α,β-unsaturated carbonyl compounds.

Scheme 70: Cu-catalyzed ACB and ACA reactions of α,β-unsaturated 2-acyl-N-methylimidazoles.

Scheme 71: Cu-catalyzed diborylation of aldehydes.

Scheme 72: Umpolung pathway for chiral, nonracemic tertiary alcohol synthesis (top) and proposed mechanism for...

Scheme 73: Cu-catalyzed synthesis of α-hydroxyboronates.

Scheme 74: Cu-catalyzed borylation of ketones.

Scheme 75: Cu-catalyzed borylation of unactivated alkyl halides.

Scheme 76: Cu-catalyzed borylation of allylic difluorides.

Scheme 77: Cu-catalyzed borylation of cyclic and acyclic alkyl halides.

Scheme 78: Cu-catalyzed borylation of unactivated alkyl chlorides and bromides.

Scheme 79: Cu-catalyzed decarboxylative borylation of carboxylic acids.

Scheme 80: Cu-catalyzed borylation of benzylic, allylic, and propargylic alcohols.
Copper-catalyzed enantioselective conjugate addition of organometallic reagents to challenging Michael acceptors
- Delphine Pichon,
- Jennifer Morvan,
- Christophe Crévisy and
- Marc Mauduit
Beilstein J. Org. Chem. 2020, 16, 212–232, doi:10.3762/bjoc.16.24
- . However, as depicted in Scheme 1, due to their stronger reactivity than that of usual esters or ketones, a competitive 1,2-addition to the carbonyl function of enals could occur, leading to the corresponding alcohol as a byproduct. Moreover, even if the 1,4-addition is favored, thanks to the copper/ligand

Graphical Abstract

Scheme 1: Competitive side reactions in the Cu ECA of organometallic reagents to α,β-unsaturated aldehydes.

Scheme 2: Cu-catalyzed ECA of α,β-unsaturated aldehydes with phosphoramidite- (a) and phosphine-based ligands...

Scheme 3: One-pot Cu-catalyzed ECA/organocatalyzed α-substitution of enals.

Scheme 4: Combination of copper and amino catalysis for enantioselective β-functionalizations of enals.

Scheme 5: Optimized conditions for the Cu ECAs of R2Zn, RMgBr, and AlMe3 with α,β-unsaturated aldehydes.

Scheme 6: CuECA of Grignard reagents to α,β-unsaturated thioesters and their application in the asymmetric to...

Scheme 7: Improved Cu ECA of Grignard reagents to α,β-unsaturated thioesters, and their application in the as...

Scheme 8: Catalytic enantioselective synthesis of vicinal dialkyl arrays via Cu ECA of Grignard reagents to γ...

Scheme 9: 1,6-Cu ECA of MeMgBr to α,β,γ,δ-bisunsaturated thioesters: an iterative approach to deoxypropionate...

Scheme 10: Tandem Cu ECA/intramolecular enolate trapping involving 4-chloro-α,β-unsaturated thioester 22.

Scheme 11: Cu ECA of Grignard reagents to 3-boronyl α,β-unsaturated thioesters.

Scheme 12: Cu ECA of alkylzirconium reagents to α,β-unsaturated thioesters.

Scheme 13: Conversion of acylimidazoles into aldehydes, ketones, acids, esters, amides, and amines.

Scheme 14: Cu ECA of dimethyl malonate to α,β-unsaturated acylimidazole 31 with triazacyclophane-based ligand ...

Scheme 15: Cu/L13-catalyzed ECA of alkylboranes to α,β-unsaturated acylimidazoles.

Scheme 16: Cu/hydroxyalkyl-NHC-catalyzed ECA of dimethylzinc to α,β-unsaturated acylimidazoles.

Scheme 17: Stereocontrolled synthesis of 3,5,7-all-syn and anti,anti-stereotriads via iterative Cu ECAs.

Scheme 18: Stereocontrolled synthesis of anti,syn- and anti,anti-3,5,7-(Me,OR,Me) units via iterative Cu ECA/B...

Scheme 19: Cu-catalyzed ECA of dialkylzinc reagents to α,β-unsaturated N-acyloxazolidinones.

Scheme 20: Cu/phosphoramidite L16-catalyzed ECA of dialkylzincs to α,β-unsaturated N-acyl-2-pyrrolidinones.

Scheme 21: Cu/(R,S)-Josiphos (L9)-catalyzed ECA of Grignard reagents to α,β-unsaturated amides.

Scheme 22: Cu/Josiphos (L9)-catalyzed ECA of Grignard reagents to polyunsaturated amides.

Scheme 23: Cu-catalyzed ECA of trimethylaluminium to N-acylpyrrole derivatives.
Allylic cross-coupling using aromatic aldehydes as α-alkoxyalkyl anions
- Akihiro Yuasa,
- Kazunori Nagao and
- Hirohisa Ohmiya
Beilstein J. Org. Chem. 2020, 16, 185–189, doi:10.3762/bjoc.16.21

- aromatic aldehydes can be used as α-alkoxyalkyl anions for catalytic carbon–carbon bond formations [7][8][9]. For example, a nucleophilic α-silyloxybenzylcopper(I) species can be generated catalytically from aromatic aldehydes through the 1,2-addition of a silylcopper(I) species followed by [1,2]-Brook
- ) species B. The 1,2-addition of silylcopper(I) B to the aromatic aldehyde 1 [15][16][17][18][19] and the subsequent [1,2]-Brook rearrangement from the obtained α-silyl-substituted copper(I) alkoxide C forms the key intermediate, an α-silyloxybenzylcopper(I) species D. The transmetallation between D and an

Graphical Abstract

Scheme 1: Our strategy.

Scheme 2: Allylic cross-coupling using aldehydes as α-alkoxyalkyl anions.

Scheme 3: Substrate scope and reaction conditions. a) reactions were carried out with 1 (0.4 mmol), 2 (0.2 mm...

Scheme 4: Stoichiometric reaction.

Scheme 5: Possible pathway.
Efficient method for propargylation of aldehydes promoted by allenylboron compounds under microwave irradiation
- Jucleiton J. R. Freitas,
- Queila P. S. B. Freitas,
- Silvia R. C. P. Andrade,
- Juliano C. R. Freitas,
- Roberta A. Oliveira and
- Paulo H. Menezes
Beilstein J. Org. Chem. 2020, 16, 168–174, doi:10.3762/bjoc.16.19
- results indicated that the substituent nature, whether electron-donating or electron-withdrawing, has no dramatic influence on the product yields. When the α,β-unsaturated aldehyde, cinnamaldehyde was used, the corresponding 1,2-addition product 2i was obtained exclusively. The chemoselectivity of the

Graphical Abstract

Scheme 1: Scope of the propargylation reaction. Reactions were performed with the appropriate aldehyde (1 mmo...

Scheme 2: Synthesis of potassium allenyltrifluoroborate (4).

Scheme 3: Propargylation of aldehydes using potassium allenyltrifluoroborate (4).
Synthesis of 3-alkenylindoles through regioselective C–H alkenylation of indoles by a ruthenium nanocatalyst
- Abhijit Paul,
- Debnath Chatterjee,
- Srirupa Banerjee and
- Somnath Yadav
Beilstein J. Org. Chem. 2020, 16, 140–148, doi:10.3762/bjoc.16.16
- following three categories: (i) by Wittig or Doebner reaction of indoles bearing a 3-aldehyde group; (ii) by 1,4- or 1,2-addition of α,β-enones or carbonyl compounds, followed by oxidation or elimination, respectively; (iii) by Pd-catalysed oxidative coupling of indoles with activated alkenes. Several

Graphical Abstract

Figure 1: Biologically and medicinally important 3-alkenylindoles.

Scheme 1: a) Previous and b) present work related to the synthesis of 3-alkenylindoles.

Scheme 2: Substrate scope for the C–H alkenylation of the indoles 1. Reaction conditions: 1 (1 mmol), 2 (2 mm...

Scheme 3: a) Three-phase test to determine a homogeneous or heterogeneous catalytic mechanism of action for t...

Scheme 4: Probable catalytic mechanism for the transformation of 1a by the RuNC.
Regioselective addition of Grignard reagents to N-acylpyrazinium salts: synthesis of substituted 1,2-dihydropyrazines and Δ5-2-oxopiperazines
- Valentine R. St. Hilaire,
- William E. Hopkins,
- Yenteeo S. Miller,
- Srinivasa R. Dandepally and
- Alfred L. Williams
Beilstein J. Org. Chem. 2019, 15, 72–78, doi:10.3762/bjoc.15.8
- Grignard reagent to add regioselectively to give 1,2-dihydropyrazine 3a. DFT calculations support the observations that the isolated regioisomer we obtained was the result of a thermodynamically favored 1,2-addition over a 1,6-addition [9]. It has also been shown that TMS-ketene acetals add selectively to
- the two vinyl protons at C5 and C6, respectively. This result is in agreement with our observation that nucleophiles favored 1,2-addition over a 1,6-addition [9]. A formation of 1,6-dihydropyrazine was not observed. With THF identified as the optimal solvent to use, we sought to expand the scope of
- . Based on our results from the monosubstituted substrates, a 1,2-addition of the Grignard was expected to occur. When an aryl group was present on the ring, trisubstituted dihydropyrazines in good yields ranging from 78–100% were produced (Figure 1, compounds 6, 7a,b and 12). An alkyl substitution with

Graphical Abstract

Figure 1: Regioselective addition of Grignard reagents to mono- and disubstituted pyrazinium salts (yields re...
Molecular iodine-catalyzed one-pot multicomponent synthesis of 5-amino-4-(arylselanyl)-1H-pyrazoles
- Camila S. Pires,
- Daniela H. de Oliveira,
- Maria R. B. Pontel,
- Jean C. Kazmierczak,
- Roberta Cargnelutti,
- Diego Alves,
- Raquel G. Jacob and
- Ricardo F. Schumacher
Beilstein J. Org. Chem. 2018, 14, 2789–2798, doi:10.3762/bjoc.14.256
- could be obtained in 90% yield as depicted in Scheme 3. On the basis of previous reports [21][25] a mechanism can be proposed for this reaction as depicted in Scheme 4. Initially, we believe that arylhydrazine 2 reacts with benzoylacetonitrile 1 by a 1,2-addition reaction in the presence of iodine as

Graphical Abstract

Scheme 1: Synthesis of selanyl-pyrazoles and their derivatives previously described.

Scheme 2: Multicomponent reaction proposed in this work.

Scheme 3: Direct selanylation reaction of 5-amino-pyrazole 5a with diphenyl diselenide (3a) under the optimiz...

Scheme 4: Proposed reaction mechanism.

Scheme 5: Synthesis of diazo pyrazole derivative 6.

Figure 1: Molecular structure of compound 6. The hydrogen atoms are omitted for clarity [27].
Some mechanistic aspects regarding the Suzuki–Miyaura reaction between selected ortho-substituted phenylboronic acids and 3,4,5-tribromo-2,6-dimethylpyridine
- Piotr Pomarański,
- Piotr Roszkowski,
- Jan K. Maurin,
- Armand Budzianowski and
- Zbigniew Czarnocki
Beilstein J. Org. Chem. 2018, 14, 2384–2393, doi:10.3762/bjoc.14.214
- with ortho-substituted phenylboronic acid for triaryl products. Summary of results for coupling with ortho-substituted phenylboronic acid for diaryl products. Proposed intermediates for the 1,2-addition of 5 with methoxy group. A) Oxidative addition step. B) Transmetalation step. Proposed intermediates
- for the 1,3-addition with methoxy group. A) Oxidative addition step. B) Transmetalation step. Proposed intermediates for the 1,2-addition with chlorine atom. A) Oxidative addition step. B) Transmetalation step. Proposed intermediates for the 1,3-addition with chlorine atom. A) Oxidative addition step

Graphical Abstract

Figure 1: Structures of stereoisomers of 3,4,5-tris(2-methoxyphenyl)-2,6-dimethylpyridines determined by X-ra...

Figure 2: Graphical representation of kinetic, time-dependent 1H NMR analysis of (syn)-7 (100 °C).

Figure 3: Graphical representation of kinetic, time-dependent 1H NMR analysis of (syn)-10 (120 °C).

Figure 4: HT-NMR (300 MHz, DMSO-d6) spectra of A) (syn)-7. B) (syn)-10. Only the upfield (ca. 3.4–4 ppm) regi...

Figure 5: Summary of the results for coupling with ortho-substituted phenylboronic acid for triaryl products.

Figure 6: Summary of results for coupling with ortho-substituted phenylboronic acid for diaryl products.

Figure 7: Proposed intermediates for the 1,2-addition of 5 with methoxy group. A) Oxidative addition step. B)...

Figure 8: Proposed intermediates for the 1,3-addition with methoxy group. A) Oxidative addition step. B) Tran...

Figure 9: Proposed intermediates for the 1,2-addition with chlorine atom. A) Oxidative addition step. B) Tran...

Figure 10: Proposed intermediates for the 1,3-addition with chlorine atom. A) Oxidative addition step. B) Tran...
Bi-mediated allylation of aldehydes in [bmim][Br]: a mechanistic investigation
- Mrunesh Koli,
- Sucheta Chatterjee,
- Subrata Chattopadhyay and
- Dibakar Goswami
Beilstein J. Org. Chem. 2018, 14, 2198–2203, doi:10.3762/bjoc.14.193
- , respectively, in >86% yields (Table 2, entries 8 and 9). Allylation of the conjugated aldehyde 1k furnished the 1,2-addition product 2k exclusively, establishing chemoselectivity of the protocol (Table 2, entry 10). With the chiral substrate (R)-2,3-O-cyclohexylideneglyceraldehyde (1l), the anti-homoallylic

Graphical Abstract

Scheme 1: Bi-mediated allylation of aldehydes.

Figure 1: Partial 1H NMR spectra (recorded at two temperatures) of the reaction mixture of allyl bromide and ...

Figure 2: Structures of all the possible allylbismuth species.

Scheme 2: Probable reaction mechanism.
Hypervalent organoiodine compounds: from reagents to valuable building blocks in synthesis
- Gwendal Grelier,
- Benjamin Darses and
- Philippe Dauban
Beilstein J. Org. Chem. 2018, 14, 1508–1528, doi:10.3762/bjoc.14.128
- the conversion of allenes 20. A regioselective 1,2-addition on the internal π-bond was observed to afford the products 21, because of the presence of a heteroatom substituent that can stabilize the radical or cationic intermediate by coordination with the copper complex (Scheme 9) [42]. However, the
- complexes has led the authors to demonstrate the superior ability of cheap FeCl2 to mediate the 1,2-addition thereby allowing the formation of the trans-products 43 with complete regio- and diastereocontrol (Scheme 14). The group of Hamashima has reported that various 1,2-difunctionalizations of alkenes can

Graphical Abstract

Scheme 1: Strategies to address the issue of sustainability with polyvalent organoiodine reagents.

Scheme 2: Functionalization of ketones and alkenes with IBX.

Scheme 3: Functionalization of pyrroles with DMP.

Scheme 4: Catalytic benzoyloxy-trifluoromethylation reported by Szabó.

Scheme 5: Catalytic benzoyloxy-trifluoromethylation reported by Mideoka.

Scheme 6: Catalytic 1,4-benzoyloxy-trifluoromethylation of dienes.

Scheme 7: Catalytic benzoyloxy-trifluoromethylation of allylamines.

Scheme 8: Catalytic benzoyloxy-trifluoromethylation of enynes.

Scheme 9: Catalytic benzoyloxy-trifluoromethylation of allenes.

Scheme 10: Alkynylation of N-(aryl)imines with EBX for the formation of furans.

Scheme 11: Catalytic benzoyloxy-alkynylation of diazo compounds.

Scheme 12: Catalytic asymmetric benzoyloxy-alkynylation of diazo compounds.

Scheme 13: Catalytic 1,2-benzoyloxy-azidation of alkenes.

Scheme 14: Catalytic 1,2-benzoyloxy-azidation of enamides.

Scheme 15: Catalytic 1,2-benzoyloxy-iodination of alkenes.

Scheme 16: Seminal study with cyclic diaryl-λ3-iodane.

Scheme 17: Synthesis of alkylidenefluorenes from cyclic diaryl-λ3-iodanes.

Scheme 18: Synthesis of alkyne-substituted alkylidenefluorenes.

Scheme 19: Synthesis of phenanthrenes from cyclic diaryl-λ3-iodanes.

Scheme 20: Synthesis of dibenzocarbazoles from cyclic diaryl-λ3-iodanes.

Scheme 21: Synthesis of triazolophenantridines from cyclic diaryl-λ3-iodanes.

Scheme 22: Synthesis of functionalized benzoxazoles from cyclic diaryl-λ3-iodanes.

Scheme 23: Sequential difunctionalization of cyclic diaryl-λ3-iodanes.

Scheme 24: Double Suzuki–Miyaura coupling reaction of cyclic diaryl-λ3-iodanes.

Scheme 25: Synthesis of a δ-carboline from cyclic diaryl-λ3-iodane.

Scheme 26: Synthesis of N-(aryl)carbazoles from cyclic diaryl-λ3-iodanes.

Scheme 27: Synthesis of carbazoles from cyclic diaryl-λ3-iodanes.

Scheme 28: Synthesis of carbazoles and acridines from cyclic diaryl-λ3-iodanes.

Scheme 29: Synthesis of dibenzothiophenes from cyclic diaryl-λ3-iodanes.

Scheme 30: Synthesis of various sulfur heterocycles from cyclic diaryl-λ3-iodanes.

Scheme 31: Synthesis of dibenzothioheterocycles from cyclic diaryl-λ3-iodanes.

Scheme 32: Synthesis of dibenzosulfides and dibenzoselenides from cyclic diaryl-λ3-iodanes.

Scheme 33: Synthesis of dibenzosulfones from cyclic diaryl-λ3-iodanes.

Scheme 34: Seminal study with linear diaryl-λ3-iodanes.

Scheme 35: N-Arylation of benzotriazole with symmetrical diaryl-λ3-iodanes.

Scheme 36: Tandem catalytic C–H/N–H arylation of indoles with diaryl-λ3-iodanes.

Scheme 37: Tandem N-arylation/C(sp2)–H arylation with diaryl-λ3-iodanes.

Scheme 38: Catalytic intermolecular diarylation of anilines with diaryl-λ3-iodanes.

Scheme 39: Catalytic synthesis of diarylsulfides with diaryl-λ3-iodanes.

Scheme 40: α-Arylation of enolates using [bis(trifluoroacetoxy)iodo]arenes.

Scheme 41: Mechanism of the α-arylation using [bis(trifluoroacetoxy)iodo]arene.

Scheme 42: Catalytic nitrene additions mediated by [bis(acyloxy)iodo]arenes.

Scheme 43: Tandem of C(sp3)–H amination/sila-Sonogashira–Hagihara coupling.

Scheme 44: Tandem reaction using a λ3-iodane as an oxidant, a substrate and a coupling partner.

Scheme 45: Synthesis of 1,2-diarylated acrylamidines with ArI(OAc)2.
One hundred years of benzotropone chemistry
- Arif Dastan,
- Haydar Kilic and
- Nurullah Saracoglu
Beilstein J. Org. Chem. 2018, 14, 1120–1180, doi:10.3762/bjoc.14.98
- oxidized to 1-ethynylbenzotropone 150 in situ, the etheric compound 152 was obtained from the reaction of 1,2-addition product 151 with HCl (Scheme 25). c. Decarbonylation of 4,5-benzotropone (11): The mechanism for the neutral and radical-cationic decarbonylation of tropone and benzannulated tropones was

Graphical Abstract

Scheme 1: Tropone (1), tropolone (2) and their resonance structures.

Figure 1: Natural products containing a tropone nucleus.

Figure 2: Possible isomers 11–13 of benzotropone.

Scheme 2: Synthesis of benzotropones 11 and 12.

Scheme 3: Oxidation products of benzotropylium fluoroborate (16).

Scheme 4: Oxidation of 7-bromo-5H-benzo[7]annulene (22).

Scheme 5: Synthesis of 4,5-benzotropone (11) using o-phthalaldehyde (27).

Scheme 6: Synthesis of 4,5-benzotropone (11) starting from oxobenzonorbornadiene 31.

Scheme 7: Acid-catalyzed cleavage of oxo-bridge of 34.

Scheme 8: Synthesis of 4,5-benzotropone (11) from o-xylylene dibromide (38).

Scheme 9: Synthesis of 4,5-benzotropone (11) via the carbene adduct 41.

Scheme 10: Heck coupling strategy for the synthesis of 11.

Scheme 11: Synthesis of benzofulvalenes via carbonyl group of 4,5-benzotropone (11).

Figure 3: Some cycloheptatrienylium cations.

Scheme 12: Synthesis of condensation product 63 and its subsequent oxidative cyclization products.

Figure 4: A novel series of benzo[7]annulenes prepared from 4,5-benzotropone (11).

Scheme 13: Preparation of substituted benzo[7]annulene 72 using the Mukaiyama-Michael reaction.

Figure 5: Possible benzo[7]annulenylidenes 73–75.

Scheme 14: Thermal and photochemical decomposition of 7-diazo-7H-benzo[7]annulene (76) and the trapping of int...

Scheme 15: Synthesis of benzoheptafulvalene 86.

Scheme 16: Synthesis of 7-(diphenylmethylene)-7H-benzo[7]annulene (89).

Scheme 17: Reaction of 4,5-benzotropone (11) with dimethyl diazomethane.

Scheme 18: Synthesis of dihydrobenzomethoxyazocine 103.

Scheme 19: Synthesis and reducibility of benzo-homo-2-methoxyazocines.

Scheme 20: Synthesis of 4,5-benzohomotropones 104 and 115 from 4,5-benzotropones 11 and 113.

Scheme 21: A catalytic deuterogenation of 4,5-benzotropone (11) and synthesis of 5-monosubstituted benzo[7]ann...

Scheme 22: Synthesis of methyl benzo[7]annulenes 131 and 132.

Scheme 23: Ambident reactivity of halobenzo[7]annulenylium cations 133a/b.

Scheme 24: Preparation of benzo[7]annulenylidene–iron complexes 147.

Scheme 25: Synthesis of 1-ethynylbenzotropone (150) and the etheric compound 152 from 4,5-benzotropone (11) wi...

Scheme 26: Thermal decomposition of 4,5-benzotropone (11).

Scheme 27: Reaction of 4,5-benzotropone (11) with 1,2-ethanediol and 1,2-ethanedithiol.

Scheme 28: Conversions of 1-benzosuberone (162) to 2,3-benzotropone (12).

Scheme 29: Synthesis strategies for 2,3-bezotropone (12) using 1-benzosuberones.

Scheme 30: Oxidation-based synthesis of 2,3-benzotropone (12) via 1-benzosuberone (162).

Scheme 31: Synthesis of 2,3-benzotropone (12) from α-tetralone (171) via ring-expansion.

Scheme 32: Preparation of 2,3-benzotropone (12) by using of benzotropolone 174.

Figure 6: Benzoheptafulvenes as condensation products of 2,3-benzotropone (12).

Scheme 33: Conversion of 2,3-benzotropone (12) to tosylhydrazone salt 182 and gem-dichloride 187.

Figure 7: Benzohomoazocines 191–193 and benzoazocines 194–197.

Scheme 34: From 2,3-benzotropone (12) to carbonium ions 198–201.

Scheme 35: Cycloaddition reactions of 2,3-benzotropone (12).

Scheme 36: Reaction of 2,3-benzotropone (12) with various reagents and compounds.

Figure 8: 3,4-Benzotropone (13) and its resonance structure.

Scheme 37: Synthesis of 6,7-benzobicyclo[3.2.0]hepta-3,6-dien-2-one (230).

Figure 9: Photolysis and thermolysis products of 230.

Figure 10: Benzotropolones and their tautomeric structures.

Scheme 38: Synthesis strategies of 4,5-benzotropolone (238).

Scheme 39: Synthesis protocol for 2-hydroxy-4,5-benzotropone (238) using oxazole-benzo[7]annulene 247.

Figure 11: Some quinoxaline and pyrazine derivatives 254–256 prepared from 4,5-benzotropolone (238).

Scheme 40: Nitration product of 4,5-benzotropolone (238) and its isomerization to 1-nitro-naphthoic acid (259)....

Scheme 41: Synthesis protocol for 6-hydroxy-2,3-benzotropone (239) from benzosuberone (162).

Scheme 42: Various reactions via 6-hydroxy-2,3-benzotropone (239).

Scheme 43: Photoreaction of 6-hydroxy-2,3-benzotropone (239).

Scheme 44: Synthesis of 7-hydroxy-2,3-benzotropone (241) from benzosuberone (162).

Scheme 45: Synthesis strategy for 7-hydroxy-2,3-benzotropone (241) from ketone 276.

Scheme 46: Synthesis of 7-hydroxy-2,3-benzotropone (241) from β-naphthoquinone (280).

Scheme 47: Synthesis of 7-hydroxy-2,3-benzotropone (241) from bicyclic endoperoxide 213.

Scheme 48: Synthesis of 7-hydroxy-2,3-benzotropone (241) by ring-closing metathesis.

Figure 12: Various monosubstitution products 289–291 of 7-hydroxy-2,3-benzotropone (241).

Scheme 49: Reaction of 7-hydroxy-2,3-benzotropone (241) with various reagents.

Scheme 50: Synthesis of 4-hydroxy-2,3-benzotropones 174 and 304 from diketones 300/301.

Scheme 51: Catalytic hydrogenation of diketones 300 and 174.

Scheme 52: Synthesis of halo-benzotropones from alkoxy-naphthalenes 306, 307 and 310.

Figure 13: Unexpected byproducts 313–315 during synthesis of chlorobenzotropone 309.

Figure 14: Some halobenzotropones and their cycloadducts.

Scheme 53: Multisep synthesis of 2-chlorobenzotropone 309.

Scheme 54: A multistep synthesis of 2-bromo-benzotropone 26.

Scheme 55: A multistep synthesis of bromo-2,3-benzotropones 311 and 316.

Scheme 56: Oxidation reactions of 8-bromo-5H-benzo[7]annulene (329) with some oxidants.

Scheme 57: Synthesis of 2-bromo-4,5-benzotropone (26).

Scheme 58: Synthesis of 6-chloro-2,3-benzotropone (335) using LiCl and proposed intermediate 336.

Scheme 59: Reaction of 7-bromo-2,3-benzotropone (316) with methylamine.

Scheme 60: Reactions of bromo-2,3-benzotropones 26 and 311 with dimethylamine.

Scheme 61: Reactions of bromobenzotropones 311 and 26 with NaOMe.

Scheme 62: Reactions of bromobenzotropones 26 and 312 with t-BuOK in the presence of DPIBF.

Scheme 63: Cobalt-catalyzed reductive cross-couplings of 7-bromo-2,3-benzotropone (316) with cyclic α-bromo en...

Figure 15: Cycloadduct 357 and its di-π-methane rearrangement product 358.

Scheme 64: Catalytic hydrogenation of 2-chloro-4,5-benzotropone (311).

Scheme 65: Synthesis of dibromo-benzotropones from benzotropones.

Scheme 66: Bromination/dehydrobromination of benzosuberone (162).

Scheme 67: Some transformations of isomeric dibromo-benzotropones 261A/B.

Scheme 68: Transformations of benzotropolone 239B to halobenzotropolones 369–371.

Figure 16: Bromobenzotropolones 372–376 and 290 prepared via bromination/dehydrobromination strategy.

Scheme 69: Synthesis of some halobenzotropolones 289, 377 and 378.

Figure 17: Bromo-chloro-derivatives 379–381 prepared via chlorination.

Scheme 70: Synthesis of 7-iodo-3,4-benzotropolone (382).

Scheme 71: Hydrogenation of bromobenzotropolones 369 and 370.

Scheme 72: Debromination reactions of mono- and dibromides 290 and 375.

Figure 18: Nitratation and oxidation products of some halobenzotropolenes.

Scheme 73: Azo-coupling reactions of some halobenzotropolones 294, 375 and 378.

Figure 19: Four possible isomers of dibenzotropones 396–399.

Figure 20: Resonance structures of tribenzotropone (400).

Scheme 74: Two synthetic pathways for tribenzotropone (400).

Scheme 75: Synthesis of tribenzotropone (400) from dibenzotropone 399.

Scheme 76: Synthesis of tribenzotropone (400) from 9,10-phenanthraquinone (406).

Scheme 77: Synthesis of tribenzotropone (400) from trifluoromethyl-substituted arene 411.

Figure 21: Dibenzosuberone (414).

Figure 22: Reduction products 415 and 416 of tribenzotropone (400).

Figure 23: Structures of tribenzotropone dimethyl ketal 417 and 4-phenylfluorenone (412) and proposed intermed...

Figure 24: Structures of benzylidene- and methylene-9H-tribenzo[a,c,e][7]annulenes 419 and 420 and chiral phos...

Figure 25: Structures of tetracyclic alcohol 422, p-quinone methide 423 and cation 424.

Figure 26: Structures of host molecules 425–427.

Scheme 78: Synthesis of non-helical overcrowded derivatives syn/anti-431.

Figure 27: Hexabenzooctalene 432.

Figure 28: Structures of possible eight isomers 433–440 of naphthotropone.

Scheme 79: Synthesis of naphthotropone 437 starting from 1-phenylcycloheptene (441).

Scheme 80: Synthesis of 10-hydroxy-11H-cyclohepta[a]naphthalen-11-one (448) from diester 445.

Scheme 81: Synthesis of naphthotropone 433.

Scheme 82: Synthesis of naphthotropones 433 and 434 via cycloaddition reaction.

Scheme 83: Synthesis of naphthotropone 434 starting from 452.

Figure 29: Structures of tricarbonyl(tropone)irons 458, and possible cycloadducts 459.

Scheme 84: Synthesis of naphthotropone 436.

Scheme 85: Synthesis of precursor 465 for naphthotropone 435.

Scheme 86: Generation of naphthotropone 435 from 465.

Figure 30: Structures of tropylium cations 469 and 470.

Figure 31: Structures of tropylium ions 471+.BF4−, 472+.BF4−, and 473+.BF4−.

Scheme 87: Synthesis of tropylium ions 471+.BF4− and 479+.ClO4−.

Scheme 88: Synthesis of 1- and 2-methylanthracene (481 and 482) via carbene–carbene rearrangement.

Figure 32: Trapping products 488–490.

Scheme 89: Generation and chemistry of a naphthoannelated cycloheptatrienylidene-cycloheptatetraene intermedia...

Scheme 90: Proposed intermediates and reaction pathways for adduct 498.

Scheme 91: Exited-state intramolecular proton transfer of 505.

Figure 33: Benzoditropones 506 and 507.

Scheme 92: Synthesis of benzoditropone 506e.

Scheme 93: Synthetic approaches for dibenzotropone 507 via tropone (1).

Scheme 94: Formation mechanisms of benzoditropone 507 and 516 via 515.

Scheme 95: Synthesis of benzoditropones 525 and 526 from pyromellitic dianhydride (527).

Figure 34: Possible three benzocyclobutatropones 534–536.

Scheme 96: Synthesis of benzocyclobutatropones 534 and 539.

Scheme 97: Synthesis attempts for benzocyclobutatropone 545.

Scheme 98: Generation and trapping of symmetric benzocyclobutatropone 536.

Scheme 99: Synthesis of chloro-benzocyclobutatropone 552 and proposed mechanism of fluorenone derivatives.

Scheme 100: Synthesis of tropolone analogue 559.

Scheme 101: Synthesis of tropolones 561 and 562.

Figure 35: o/p-Tropoquinone rings (563 and 564) and benzotropoquinones (565–567).

Scheme 102: Synthesis of benzotropoquinone 566.

Scheme 103: Synthesis of benzotropoquinone 567 via a Diels–Alder reaction.

Figure 36: Products 575–577 through 1,2,3-benzotropoquinone hydrate 569.

Scheme 104: Structures 578–582 prepared from tropoquinone 567.

Figure 37: Two possible structures 583 and 584 for dibenzotropoquinone, and precursor compound 585 for 583.

Scheme 105: Synthesis of saddle-shaped ketone 592 using dibenzotropoquinone 584.
Gram-scale preparation of negative-type liquid crystals with a CF2CF2-carbocycle unit via an improved short-step synthetic protocol
- Tatsuya Kumon,
- Shohei Hashishita,
- Takumi Kida,
- Shigeyuki Yamada,
- Takashi Ishihara and
- Tsutomu Konno
Beilstein J. Org. Chem. 2018, 14, 148–154, doi:10.3762/bjoc.14.10
- the 1,2-addition reaction of Int-I proceeds in preference to the conjugate addition reaction, the desired octa-1,7-diene 5a should be produced in higher yield. However, no significant improvement was observed after various attempts such as employing a more nucleophilic lithium reagent instead of the

Graphical Abstract

Figure 1: Typical examples of previously reported negative-type liquid crystals containing a CF2CF2-carbocycl...

Scheme 1: Improved short-step synthetic protocol for multicyclic mesogens 1 and 2.

Scheme 2: Short-step approach to CF2CF2-containing carbocycles.

Figure 2: (a) Expected products of over-reaction in the Grignard reaction of dimethyl tetrafluorosuccinate (7...

Scheme 3: Mechanism for the reaction of γ-keto ester 6 with vinyl Grignard reagents.

Scheme 4: First multigram-scale preparation of CF2CF2-containing multicyclic mesogens.

Scheme 5: Stereochemical assignment of the ring-closing metathesis products.
Quinone-catalyzed oxidative deformylation: synthesis of imines from amino alcohols
- Xinyun Liu,
- Johnny H. Phan,
- Benjamin J. Haugeberg,
- Shrikant S. Londhe and
- Michael D. Clift
Beilstein J. Org. Chem. 2017, 13, 2895–2901, doi:10.3762/bjoc.13.282
- resulting imines (7r−t) are hydrolyzed under the current reaction conditions. Notably, imines 7n [42][43][44][45] and 7t [46][47][48] are useful imines for diastereoselective 1,2-addition reactions. Following these substrate scope studies, we next examined the quinone-catalyzed C–C bond cleavage of

Graphical Abstract

Scheme 1: Established methods for the preparation of imines vs this work.

Scheme 2: Proposed catalytic cycle for quinone-catalyzed deformylation.

Scheme 3: Studies of quinone-catalyzed C−C bond cleavage in related substrates.

Scheme 4: Sequential oxidative deformylation/Mukaiyama−Mannich addition using phenylglycinol.
Electron-deficient pyridinium salts/thiourea cooperative catalyzed O-glycosylation via activation of O-glycosyl trichloroacetimidate donors
- Mukta Shaw,
- Yogesh Kumar,
- Rima Thakur and
- Amit Kumar
Beilstein J. Org. Chem. 2017, 13, 2385–2395, doi:10.3762/bjoc.13.236
- trichloroacetimidate donors using a synergistic catalytic system of electron-deficient pyridinium salts/aryl thiourea derivatives at room temperature is demonstrated. The acidity of the adduct formed by the 1,2-addition of alcohol to the electron-deficient pyridinium salt is increased in the presence of an aryl
- to lower acidity of the ammonium salt formed by 1,2-addition of the acceptor to the pyridinium salt. The conjugate base formed after the release of a proton from the ammonium salt may be quite stable to impart a negative charge to the acceptor oxygen. The acidity of the ammonium salt may be enhanced
- -deficient pyridinium salt/thiourea cocatalyzed glycosidation is outlined in Figure 4. It is presumed that at first electron-deficient pyridinium salt 3a undergoes 1,2-addition with the acceptor to produce ammonium salt X. The addition of the thiourea derivative as hydrogen-bonding cocatalyst could activate

Graphical Abstract

Scheme 1: Mechanistic hypothesis for work.

Figure 1: 1H NMR (a) glycosyl donor 1α and (b) a mixture of 1α and 10 mol % 3a in CD2Cl2 at room temperature.

Figure 2: 1H NMR (a) glycosyl acceptor 2a, (b) pyridinium salt 3a (in DMSO-d6) and (c) a mixture of 2a and 3a...

Figure 3: 1H NMR (a) glycosyl acceptor 2a, (b) pyridinium salt 3a (in DMSO-d6), (c) aryl thiourea and (d) a m...

Scheme 2: Synergistic electron-deficient pyridinium salt/aryl thiourea-catalyzed regioselective O-glycosylati...

Figure 4: Plausible reaction mechanism.






