Search results
Search for "4-aminoantipyrine" in Full Text gives 3 result(s) in Beilstein Journal of Organic Chemistry.
Syntheses and medicinal chemistry of spiro heterocyclic steroids
Beilstein J. Org. Chem. 2024, 20, 1713–1745, doi:10.3762/bjoc.20.152
- from 17α-hydroxyandrost-4-en-3-one (150) in a two-step sequence [64]. An initial condensation between 4-aminoantipyrine and the carbonyl group of 150 led to the imine 151. Then, treatment of the latter with hydrazine provoked an addition to the imine carbon, followed by a dehydrative cyclization. To
An overview of recent advances in duplex DNA recognition by small molecules
Beilstein J. Org. Chem. 2018, 14, 1051–1086, doi:10.3762/bjoc.14.93
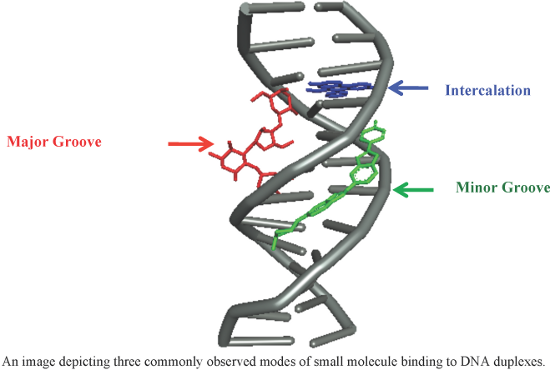
- exceptionally stable duplexes (ΔTm = +44.8 °C) and high selectivity [92]. Six novel 4-aminoantipyrine derived Schiff bases and their metal complexes with Cu(II), Ni(II), Zn(II) ions (conjugates 26–31) were synthesized and characterized and binding of these complexes with ct-DNA were analyzed by electronic
A novel 4-aminoantipyrine-Pd(II) complex catalyzes Suzuki–Miyaura cross-coupling reactions of aryl halides
Beilstein J. Org. Chem. 2014, 10, 2821–2826, doi:10.3762/bjoc.10.299
- 58030, tel.: +52 443 326 5790; fax: +52 443 326 5788 10.3762/bjoc.10.299 Abstract A simple and efficient catalytic system based on a Pd complex of 4-aminoantipyrine, 4-AAP–Pd(II), was found to be highly active for Suzuki–Miyaura cross-coupling of aryl iodides and bromides with phenylboronic acids under
- are tolerated. Keywords: 4-aminoantipyrine; arylboronic acids; biaryls; cross-coupling; palladium(II) complex; Introduction The sp2–sp2 carbon–carbon bond formation through cross-coupling reactions catalyzed by metal complexes has emerged as a powerful tool in organic synthesis [1][2][3][4][5][6
- -free catalysts for SM coupling reactions. Transition metal complexes that have shown a wide range of biological activity are those containing the pyrazolone derivative 4-aminoantipyrine (4-amino-1,5-dimethyl-2-phenyl-1H-pyrazol-3(2H)-one, or simply “4-AAP”) [18]. Pyrazoles, in general, are one of the
















































































