Search results
Search for "Bisamidines" in Full Text gives 2 result(s) in Beilstein Journal of Organic Chemistry.
An overview of recent advances in duplex DNA recognition by small molecules
Beilstein J. Org. Chem. 2018, 14, 1051–1086, doi:10.3762/bjoc.14.93
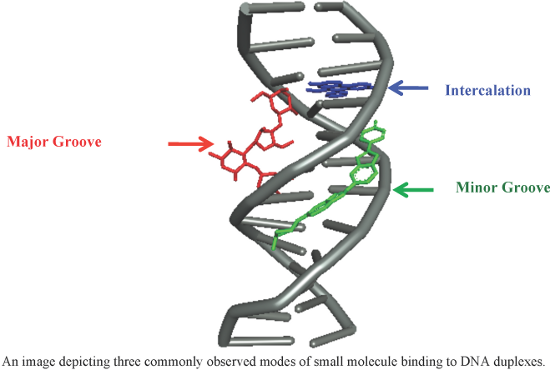
- could not bind into the minor groove due to change in their conformation, thereby showing negligible DNA binding. Thus, their antibacterial activity is not attributed to DNA binding affinity due to lack of DNA stabilization by these conjugates. 2.3. Bisamidines One of the oldest known clinically
C2- symmetric bisamidines: Chiral Brønsted bases catalysing the Diels- Alder reaction of anthrones
Beilstein J. Org. Chem. 2008, 4, No. 28, doi:10.3762/bjoc.4.28
- Deniz Akalay Gerd Durner Jan W. Bats Michael W. Gobel Johann Wolfgang Goethe University Frankfurt, Institute of Organic Chemistry and Chemical Biology, Max-von-Laue-Str. 7, D-60438 Frankfurt am Main, Germany. 10.3762/bjoc.4.28 Abstract C2-symmetric bisamidines 8 have been tested as chiral
- a cycloaddition step stereoselectively controlled by the chiral ion pair. Keywords: Asymmetric Catalysis; Bisamidines; Brønsted base; Diels-Alder reaction; Organocatalysis; Introduction The cycloadditions of anthrones 1 and N-substituted maleimides 2 are prominent examples of asymmetric catalysis
- , in spite of their limited Brønsted-basicity [5] (Scheme 1). Our study was motivated by the structural similarity of bisoxazolines 7 and bisamidines 8. Bisamidines 8, readily accessible from malonodinitrile in two steps, prefer the conjugated tautomeric form (enamine-imine) in the monoprotonated state



































