Search results
Search for "gold(I)" in Full Text gives 88 result(s) in Beilstein Journal of Organic Chemistry.
A laterally-fused N-heterocyclic carbene framework from polysubstituted aminoimidazo[5,1-b]oxazol-6-ium salts
Beilstein J. Org. Chem. 2024, 20, 621–627, doi:10.3762/bjoc.20.54
- aminide 7 in good yield on a gram scale (Scheme 1b). With the novel 3-aminoimidazo[5,1-b]oxazol-6-ium salt in hand, we examined its use as an NHC precursor for the preparation of late transition metal complexes. Treating compound 9a with triethylamine and either dimethyl sulfide gold(I) chloride or copper
Ligand effects, solvent cooperation, and large kinetic solvent deuterium isotope effects in gold(I)-catalyzed intramolecular alkene hydroamination
Beilstein J. Org. Chem. 2024, 20, 479–496, doi:10.3762/bjoc.20.43
- Since the seminal 1998 report by Teles et al. on the gold(I)-catalyzed addition of alcohols to alkynes [1], a multitude of gold-catalyzed reactions have been reported. Great successes in mechanistic analysis and synthetic methods have been achieved for allene and alkyne activation, while the activation
- hydroamination to 3a, commercially available ((acetonitrile)[(2-biphenyl)di-tert-butylphosphine]gold(I) hexafluoroantimonate (5) was used as catalyst (Figure 1), where gold is supported by the ligand commonly known as “Johnphos” or, henceforth, “JPhos”. Interestingly, there are minor differences in observed rate
- /mol and ΔS‡ −42.1 ± 3.9 cal/mol·K. Non-linear behavior in the presence of methanol indicated a more complex rate law (less than first order in methanol and catalyst), precluding determination of activation parameters under those conditions at this time. Discussion The first reports of gold(I
1,4-Dithianes: attractive C2-building blocks for the synthesis of complex molecular architectures
Beilstein J. Org. Chem. 2023, 19, 115–132, doi:10.3762/bjoc.19.12
- use in intermolecular cycloadditions, as the putative gold(I)-coordinated intermediates like 112 are indeed quite similar to those expected to arise from dihydrodithiin alcohol 90 (see Scheme 19) [112]. However, our results with the simple 2-ethynyl-1,3-dithiolane (116) immediately showed that the
- expected allyl cation (3 + 2) cycloaddition reactivity is not operating under gold(I) catalysis, but instead it behaved as a reliable and quite stereoselective vinylgold carbenoid species, affording exclusively cyclopropanation products with a wide range of olefin substrates (Scheme 18c). The carbene-type
- of dithiin-fused allyl alcohols and similar non-cyclic sulfur-substituted allyl alcohols. Applications of dihydrodithiins in the rapid assembly of polycyclic terpenoid scaffolds [108][109]. Dihydrodithiin-mediated allyl cation and vinyl carbene cycloadditions via a gold(I)-catalyzed 1,2-sulfur
Total synthesis of grayanane natural products
Beilstein J. Org. Chem. 2022, 18, 1707–1719, doi:10.3762/bjoc.18.181
- Conia-ene-type cyclization, classically considered as a reliable method for the construction of bridged bicyclic structures [41], was envisaged. However, using a gold(I) catalyst the desired 5-exo-dig cyclization failed and only a 6-endo-dig cyclization was observed. Thus, Hong et al. explored a Mn(III
Formal total synthesis of macarpine via a Au(I)-catalyzed 6-endo-dig cycloisomerization strategy
Beilstein J. Org. Chem. 2022, 18, 1589–1595, doi:10.3762/bjoc.18.169
- building blocks 5 and 8 in hand, ketone 9 was prepared via a palladium-catalyzed Sonogashira coupling reaction in a yield of 95%. The precursor 10 for the gold(I)-catalyzed [19][20][21][22][23][24] cycloisomerization was then synthesized by treating ketone 9 with sodium bis(trimethylsilyl)amide (NaHMDS
- ) and tert-butyldimethylsilyl chloride (TBSCl) (Scheme 5). To find the best cycloisomerization conditions, the 1,5-enyne substrate 10 was subjected to different reaction conditions as listed in Table 1. It was observed that [1,3-bis(2,6-diisopropylphenyl)imidazol-2-ylidene]gold(I) chloride (IPrAuCl
- ). The Au(I)-catalyzed cycloisomerization reaction of substrate 10 occurred under the catalysis of 5 mol % [1,3-bis(2,6-diisopropylphenyl)imidazol-2-ylidene]gold(I) chloride (IPrAuCl) and 5 mol % silver hexafluoroantimonate (AgSbF6) [25][26] in anhydrous DCM at room temperature for 2 h forming a benzene
Supramolecular approaches to mediate chemical reactivity
Beilstein J. Org. Chem. 2022, 18, 1463–1465, doi:10.3762/bjoc.18.152

- showcasing innovative research regarding supramolecular catalysis using macrocyclic and acyclic hosts, as well as other molecular architectures such as self-assembled capsules and metallocages. In their contribution, Secchi and Cera [15] reported the synthesis of diphosphine gold(I) calix[6]arene complexes
- whose geometry, in low-polarity solvents, is controlled by the 1,2,3-alternate conformation of the calix[6]arene skeleton. These catalysts can tune the selectivity of the catalytic cycloisomerization of 1,6-enynes in response to the relative orientation of the coordinated gold(I) atom with respect to
Heteroleptic metallosupramolecular aggregates/complexation for supramolecular catalysis
Beilstein J. Org. Chem. 2022, 18, 597–630, doi:10.3762/bjoc.18.62

- molecular host to modulate the reactivity. Recently, Reek and co-workers have constructed an M12L24 nanosphere by treating the bispyridyl 120° ligand 30 with a Pd(II) precursor [72]. Here, the ligand 30 is optimally functionalized with a phosphine gold(I) chloride moiety so that the metal catalyst will
Regioselectivity of the SEAr-based cyclizations and SEAr-terminated annulations of 3,5-unsubstituted, 4-substituted indoles
Beilstein J. Org. Chem. 2022, 18, 293–302, doi:10.3762/bjoc.18.33
- -cd]indoles 21 from the intramolecular cyclization of Ugi adducts 20 in moderate to good yields and excellent chemo-, regio-, and diastereoselectivity (Scheme 7) [17]. Mechanistically, the reaction involves a tandem gold(I)-catalyzed dearomatization/ipso-cyclization/Michael addition sequence to
Diametric calix[6]arene-based phosphine gold(I) cavitands
Beilstein J. Org. Chem. 2022, 18, 190–196, doi:10.3762/bjoc.18.21
- polarity solvents, of a novel class of diametric phosphine gold(I) cavitands characterized by a 1,2,3-alternate geometry. Preliminary catalytic studies were performed on a model cycloisomerization of 1,6-enynes as a function of the relative orientation of the bonded gold(I) nuclei with respect to the
- macrocyclic cavity. Keywords: 1,2,3-alternate conformation; calix[6]arenes; gold(I) catalysis; phosphines; Introduction One of the latest challenges in supramolecular chemistry is the design and development of novel macrocyclic-based entities able to influence the catalytic activities of the metal center [1
- , calix[4]- [9][10][11][12][13] and resorcin[4]arene [14][15][16][17] are the most exploited cavitands due to their inherent limited flexibility and already proved their ability to control the catalytic activity of late-transition metals and particularly gold(I) catalysts [18][19][20][21][22][23][24][25
Methodologies for the synthesis of quaternary carbon centers via hydroalkylation of unactivated olefins: twenty years of advances
Beilstein J. Org. Chem. 2021, 17, 1565–1590, doi:10.3762/bjoc.17.112
- further demonstrated by the hydroalkylation of the anti-inflammatory drug phenylbutazone (7j). Gold(I)/gold(III) and silver(I) catalysis in olefin hydroalkylation The early limitations associated with Pd catalysis in olefin hydroalkylation reactions prompted the search for alternative metals for olefin
- . Cationic gold(I) complexes are also suitable catalysts for olefin functionalization, and their use has become more popular than gold(III) catalysis [39]. In 2007, Che and Zhou reported the olefin intramolecular hydroalkylation of N-alkenyl β-ketoamides 13 using the gold(I)–phosphine complex Au[P(t-Bu)2(o
- , suggesting that the coordination and proton-shuttle ability of the anionic counterion were the main factors responsible for the observed effects. The proposed mechanism (Scheme 10) begins with the activation of the gold(I) pre-catalyst by the exchange of a chloride anion with a non-coordinated one (Y−) to
The preparation and properties of 1,1-difluorocyclopropane derivatives
Beilstein J. Org. Chem. 2021, 17, 245–272, doi:10.3762/bjoc.17.25
- (Scheme 23) [64]. Fürstner et al. [67] showed that (trifluoromethyl)gold(I)triphenylphosphine in dichloromethane can be used for the production of difluorocyclopropanes at low temperatures. The advantage of the method is its stereoselectivity. The disadvantages include the stoichiometric use of gold, low
Au(III) complexes with tetradentate-cyclam-based ligands
Beilstein J. Org. Chem. 2021, 17, 186–192, doi:10.3762/bjoc.17.18
- range of other transition metals. The utilization of gold in synthetic organic chemistry has become a topic of interest during the last decades, as evidenced by the increasing number of review articles published in this period [1][2][3][4][5][6][7][8]. Whereas both gold(I) and gold(III) are proven to be
- catalytic active forms of gold, gold(I) has so far, received main attention, likely due to the higher stability, as demonstrated by the development of a high number of gold(I)-catalyzed transformations and ligated gold(I) complexes, along with improved mechanistic understanding [9][10][11][12][13][14][15
- ]. In contrast, gold(III) catalysis was for a long time mostly based on inorganic salts, such as AuCl3, AuBr3, or pyridine–AuCl3 and Pic–AuCl2. However, Au(III) complexes with various coordinated ligands are about to become more explored. Different from the linear coordination mode of gold(I), gold(III
Pentannulation of N-heterocycles by a tandem gold-catalyzed [3,3]-rearrangement/Nazarov reaction of propargyl ester derivatives: a computational study on the crucial role of the nitrogen atom
Beilstein J. Org. Chem. 2020, 16, 3059–3068, doi:10.3762/bjoc.16.255
- Studi di Firenze, Via della Lastruccia 13, 50019 Sesto Fiorentino, Florence, Italy 10.3762/bjoc.16.255 Abstract The tandem gold(I)-catalyzed rearrangement/Nazarov reaction of enynyl acetates in which the double bond is embedded in a piperidine ring was computationally and experimentally studied. The
- first step [1][9][10][11][12]. In the framework of our studies on gold(I)-catalyzed reactions of propargyl alcohol derivatives [13][14][15], we have recently reported that the pentannulation of N-heterocycles [16] can be efficiently achieved by a cascade gold-catalyzed [3,3]-rearrangement/Nazarov
- cyclopenta-fused heterocyclic system with an alternate position of the C=O group on the five-membered ring when treated with gold(I) (see 7, Figure 1c). In this context, the Nazarov cyclization has been profusely studied, and it was found that it is very sensitive to the electronic features of the substrates
Using multiple self-sorting for switching functions in discrete multicomponent systems
Beilstein J. Org. Chem. 2020, 16, 2831–2853, doi:10.3762/bjoc.16.233

- backbones in the presence of Ag2O provided exclusively the three homomeric cylinders [Ag3(13)2]3+, [Ag3(14)2]3+, and [Ag3(15)2]3+ (state: SelfSORT-I in Figure 7). Upon the addition of gold(I) ions, a one-pot transmetalation triggered an exchange of the Ag+ ions for Au+ in the tris-NHC ligand-based cylinders
Reactions of 3-aryl-1-(trifluoromethyl)prop-2-yn-1-iminium salts with 1,3-dienes and styrenes
Beilstein J. Org. Chem. 2020, 16, 2064–2072, doi:10.3762/bjoc.16.173
- presumably renders an ionic [2 + 2] cycloaddition pathway competitive with the Diels–Alder reaction. The few reported examples of cyclobutene formation from alkynes and unactivated 1,3-dienes include the sensitized photocycloaddition of phenylacetylene and DMBD [30] and the gold(I)-catalyzed reaction of
Regioselectively α- and β-alkynylated BODIPY dyes via gold(I)-catalyzed direct C–H functionalization and their photophysical properties
Beilstein J. Org. Chem. 2020, 16, 587–595, doi:10.3762/bjoc.16.53

- , Japan 10.3762/bjoc.16.53 Abstract A series of α- and β-ethynyl-substituted BODIPY derivatives (3a, 4a, 5a, 5b, 6a, 6b) were synthesized by gold(I)-catalyzed direct C–H alkynylation reactions of dipyrromethane and BODIPY, respectively, with ethynylbenziodoxolone (EBX) in a regioselective manner
- -materials. Keywords: alkynylation; BODIPY; direct C–H functionalization; gold(I); Introduction Boron-dipyrromethene (BODIPY, 1) and its derivatives are representative families of fluorophores that have been widely used in applications for bioimaging [1][2][3][4][5][6], photodynamic therapy [7][8][9][10
- BODIPY core has not yet been achieved. Inspired by the works of Waser and co-workers showing the gold(I)-catalyzed C–H electrophilic alkynylation of various heterocycles (e.g., pyrroles, indoles, etc.) with ethynylbenziodoxolone (EBX) as an activated ethynyl synthon [38][39][40][41][42], we investigated
Iodine-mediated hydration of alkynes on keto-functionalized scaffolds: mechanistic insight and the regiospecific hydration of internal alkynes
Beilstein J. Org. Chem. 2019, 15, 2747–2752, doi:10.3762/bjoc.15.265
- (II) [7], silver(I) [8], and gold(I)/(III) [9][10][11][12]. While less toxic than mercury, such catalysts are still environmental hazards and tend to be costlier [13][14]. The iodine-mediated hydration described herein is a viable solution to these issues [15][16]. This metal-free method produces
Self-assembled coordination thioether silver(I) macrocyclic complexes for homogeneous catalysis
Beilstein J. Org. Chem. 2019, 15, 2465–2472, doi:10.3762/bjoc.15.239
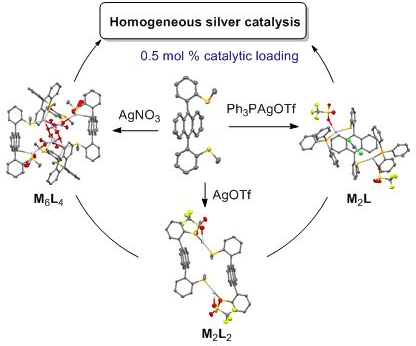
- for gold(III) chloride whose photoreduction to gold(I) was fast and controlled [44][45]. These gold complexes at different oxidation states showed efficient catalytic properties, that were highlighted in a one-pot cascade synthesis of 4H-benzoquinolizin-4-one. Now, we propose to investigate a rigid
Synthesis of 2H-furo[2,3-c]pyrazole ring systems through silver(I) ion-mediated ring-closure reaction
Beilstein J. Org. Chem. 2019, 15, 679–684, doi:10.3762/bjoc.15.62
- desired 2-phenyl-2H-furo[2,3-c]pyrazole was obtained in a significantly higher yield (47%, Table 1, entry 3). Some of the most effective catalysts for the electrophilic activation of alkynes under homogeneous conditions are gold(I) [35] and silver(I) [36] salts or complexes, and a broad range of versatile
- synthetic methods has been developed for the construction of carbon–heteroatom bonds using these types of catalysts. For example, the gold(I) catalyst [(Ph3PAu)3O]BF4 was applied in the regioselective intramolecular cyclization of alkynols to construct bicyclic ethers [37], while the silver(I) catalyst
- AgOTf efficiently catalyzed the intramolecular cyclization of phenoxyethynyl diols into 2,3-unsaturated lactones [38]. In our case, the addition of 10 mol % chloro(triphenylphosphine)gold(I) improved the yield of product 4a to 69% (Table 1, entry 4). However the best results were obtained when AgOTf was
A chemoenzymatic synthesis of ceramide trafficking inhibitor HPA-12
Beilstein J. Org. Chem. 2019, 15, 490–496, doi:10.3762/bjoc.15.42
- reaction [20], tandem approach from (S)-Wynberg lactone [21], chiral ruthenium-catalyzed N-demethylative rearrangement of 1,2-isoxazolidines [22], gold(I)-catalyzed cyclization of a propargylic N-hydroxylamine [23], from β-sulfinamido ketones derived from chiral sulfinimines [24], and a Kornblum–DeLaMare
Synthesis of unnatural α-amino esters using ethyl nitroacetate and condensation or cycloaddition reactions
Beilstein J. Org. Chem. 2018, 14, 2846–2852, doi:10.3762/bjoc.14.263
- -amino esters was achieved using gold(I) chemistry. Keywords: α-amino ester; α-nitro esters; cerium ammonium nitrate; cycloaddition; gold(I) cyclization; Introduction In the course of our work on an original synthesis of imidazo[1,2-a]pyrazin-3(7H)-one luciferins [1], a large variety of racemic α-amino
- 16. A second [2 + 3] cycloaddition-based approach is described in Scheme 4. It started with the preparation of the methylene-bearing dipolarophile 25 from propargylamide 24 using gold(I) chemistry, which turned out to be tolerant to a wide variety of dry solvents (dichloromethane, tetrahydrofuran
- in Scheme 5, the oxazole-bearing α-amino ester 43 was prepared from the aspartic acid derivative 37 through the propargylamide 38 followed by a gold(I)-catalyzed cyclization to form the oxazoline derivative 40. Concerning the amidation step, propargylamide 38 was obtained in 78% yield provided that
Rhodium-catalyzed C–H functionalization of heteroarenes using indoleBX hypervalent iodine reagents
Beilstein J. Org. Chem. 2018, 14, 1208–1214, doi:10.3762/bjoc.14.102
- nitrogen and a transition metal catalyst (reaction 1, Scheme 1A) [11][12][13][14][15][16][17][18][19]. In particular, Li and co-workers have used ethynylbenziodoxolone (EBX) hypervalent iodine reagents to achieve a regiodivergent alkynylation of the pyridinone core employing either a gold(I) or a rhodium
AuBr3-catalyzed azidation of per-O-acetylated and per-O-benzoylated sugars
Beilstein J. Org. Chem. 2018, 14, 682–687, doi:10.3762/bjoc.14.56
- their operationally simple, safe and neutral reaction conditions, had widely contributed to the development of new glycosylation methods. Gold(I) and gold(III) complexes are usually alkynophilic [2], carbophilic and oxophilic because of their affinity towards the alkynes’ and C–O π systems [3][4][5][6
- ]. Thus, various research groups employed either a remote alkyne group possessing versatile glycosyl donors [7][8][9][10][11][12][13][14][15][16] or used glycals [17] for effective O-, C-, and S-glycosylation reactions using gold(I) and gold(III) catalysts. Among the gold-catalyzed activation of non
Is the tungsten(IV) complex (NEt4)2[WO(mnt)2] a functional analogue of acetylene hydratase?
Beilstein J. Org. Chem. 2017, 13, 2332–2339, doi:10.3762/bjoc.13.230
- . More specifically, gold complex AuCl(PPh3) is not usually considered an alkyne hydration catalyst itself, but turns into a catalytically active gold(I) cation after activation with silver salt or Lewis acid [31][32][33][34]. Under the forcing microwave reaction conditions in aqueous methanol
Glyco-gold nanoparticles: synthesis and applications
Beilstein J. Org. Chem. 2017, 13, 1008–1021, doi:10.3762/bjoc.13.100

- metal-based anticancer drugs need to be developed. Recently, it has been reported the interesting synthesis of AuNPs coated with glyco-polymers and functionalized with gold(I) triphenylphosphine (Figure 7) [88]. This work showed the potentiality of these structures as novel cancer therapeutic drugs. The
































































































































































































































































































































































