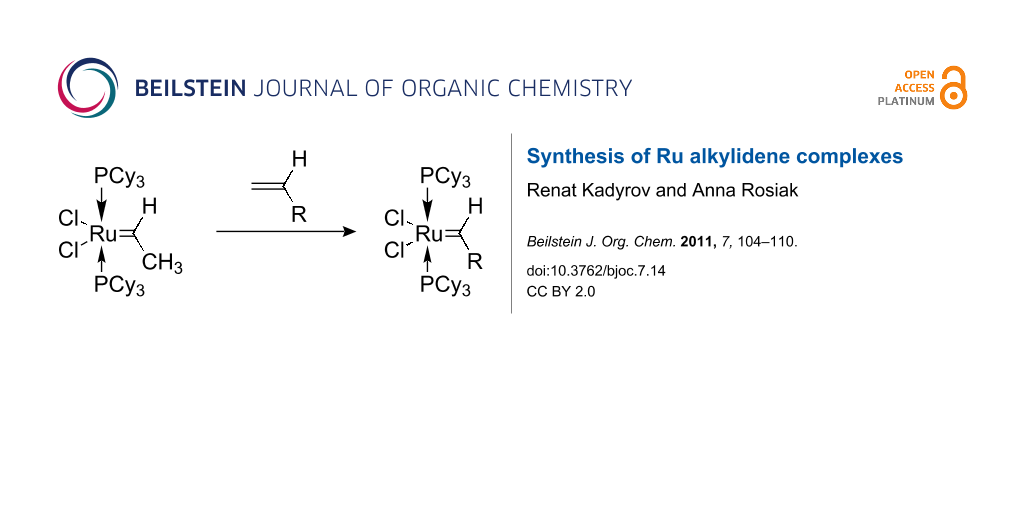
Abstract
The present work describes the robust synthesis of Ru alkylidene complexes (PCy3)2Cl2Ru=CHR – precursors for metathesis catalysts. Moreover, the dynamic behavior of complexes where R = 2-naphthyl and 2-thienyl was studied. 1H NMR techniques were employed to establish the preferred conformations in solution for both complexes and the energy barrier for rotation around single (Ru=)CH–C(thienyl) bond was estimated (ΔG≠303K = 12.6 kcal/mol).
Graphical Abstract
Introduction
The key to active ruthenium metathesis initiators is the accessibility of the ruthenium precursor containing the alkylidene moiety. The most commonly used precursors for the “second generation” catalysts bearing NHC ligands are the alkylidene ruthenium complexes coordinated with two phosphines [1]. For recent reviews see [2-4]. There are several routes for accessing five-coordinated ruthenium(II) alkylidene complexes such as diazo-transfer [5] and the reaction of vinyl or propargyl halides with hydrido(dihydrogen)-Ru-complexes generated from [Ru(COD)Cl2] and PCy3 under hydrogen pressure [6]. It should also be noted that the method for the generation of such highly reactive hydrido(dihydrogen)-Ru-complexes was first described by Werner and co-workers who employed two equivalents of iPr3P in 2-butanol and hydrogen [7]. This last attractive one-pot procedure without the use of hydrogen was improved by the Ciba-group [8,9]. Werner and co-workers also published a one-pot synthesis of the complex (PCy3)2Cl2Ru=CHMe (1a) by direct reduction of RuCl3 with Mg/ClCH2CH2Cl in THF in the presence of excess PCy3 and hydrogen followed by subsequent reaction with acetylene [10].
We report herein on an improved protocol for the synthesis of the ethylidene complex (PCy3)2Cl2Ru=CHMe (1a) under mild conditions which is an efficient precursor for the preparation of wide variety of other alkylidene complexes.
Results and Discussion
Van der Schaaf and co-workers published in 2000 a simple one-pot procedure for the synthesis of the ruthenium benzylidene complex (iPr3P)2Cl2Ru=CHPh [8]. It was mentioned that also (PCy3)2Cl2Ru=CHPh could be similarly prepared. To our surprise, by following exactly the given protocol using DBU as base, a mixture of the desired benzylidene complex (PCy3)2Cl2Ru=CHPh together with the vinylidene complex (PCy3)2Cl2Ru=C=CHPh was obtained. Obviously, the last complex originated from reaction of an intermediate hydride species with phenyl acetylene along with formation of the benzylmethylidene complex (PCy3)2Cl2Ru=CHCH2Ph as described previously by Werner [7]. We have found that the use of trimethylsilylacetylene afforded the ethylidene complex 1a as the sole product in very good isolated yield (see Scheme 1).
In sharp contrast, the use of 1-phenyl-2-trimethylsilylacetylene or 1-trimethylsilyl-1-hexyne gave the vinylidene complexes 2 and 3 in only moderate isolated yields (see Scheme 2).
Scheme 2: Synthesis of complexes 2 and 3.
Scheme 2: Synthesis of complexes 2 and 3.
Compound 1a is remarkably stable below room temperature and readily exchanges the ethylidene moiety with other alkenes. Thus, compound 1a is an ideal precursor for a variety of other ruthenium alkylidene complexes. Compounds 1b–i (Scheme 3) were readily isolated and characterized spectroscopically. It is noteworthy, that with the exception of 1e and 1g, all isolated complexes decompose slowly in chlorinated organic solvents. Therefore, cross metatheses in toluene in general led to alkylidene complexes with higher isolated yields.
The NMR spectra of compounds 1b,c,e–i displayed more or less broad signals at ambient temperature. In particular, lowering the temperature of solutions of 1e and 1g in CD2Cl2 caused further broadening of the NMR resonances which only become properly resolved for the aromatic and methylidene signals at −80 °C. The slow exchange resonances of compound 1g are better resolved due to the lower concentration of the minor isomer. A 1H,1H-COSY experiment at −80 °C enabled the identification of the aromatic resonances in the low temperature spectrum (Figure 1). The singlet at 8.49 ppm is assigned to H1 and the doublet at 9.01 ppm to H3 on the basis of the observed weak coupling 4J(H1H3). The strong coupling of 3J(H3H4) = 8.2 Hz with doublet at 9.01 ppm allows the assignment of H4 (7.78 ppm). Other coupling patterns are consistent with the resonances of the residual protons H5 (8.12, d, J = 8.2 Hz), H6 (7.50, t, J = 7.0 Hz) and H7/H8 (7.67-7.75, m).
Figure 1: Naphthyl-group region of 1H,1H-COSY NMR for 1g in CD2Cl2 at −80 °C.
Figure 1: Naphthyl-group region of 1H,1H-COSY NMR for 1g in CD2Cl2 at −80 °C.
Strong NOE enhancement of H1 upon saturation of the carbene proton at 19.75 ppm (see Figure 2) is consistent with preferred conformer 1g in which the naphthyl moiety is directed away from the phosphine ligand (see Scheme 4).
Figure 2: 1H NMR (top) and NOE difference spectrum (bottom) of 1g in CD2Cl2 at −80 °C, saturating the methylidene H signal at δ = 19.75 ppm.
Figure 2: 1H NMR (top) and NOE difference spectrum (bottom) of 1g in CD2Cl2 at −80 °C, saturating the methyli...
Scheme 4: Conformational isomerism in complex 1g.
Scheme 4: Conformational isomerism in complex 1g.
At low temperature both isomers of 1e are visible in the NMR spectrum due to comparable concentrations (obtained enthalpy difference ΔH = 1.3 kcal/mol, see Supporting Information File 1). A number of NOE experiments at −40 °C allowed the assignment of the resonances of both isomers 1e and 1e’. Saturation of the carbene proton at 18.9 ppm led to strong NOE enhancement of the singlet at 7.68 ppm (Figure 3) and allowed the assignment of this signal to the H3 proton of the thienyl moiety and was consistent with the s-trans isomer 1e being the preferred conformer (see Scheme 5). The EXSY effect made it possible to assign the signal at 8.80 ppm to H3’ of the minor s-cis conformer. Enhancement of the signal at 6.99 ppm (Figure 4) by saturation of the signal at 8.07 ppm and EXSY inversion of the resonance at 7.79 ppm allowed the assignment of the signals for H5 (8.07 ppm), H4 (6.99 ppm), H5’ (7.79 ppm) and H4’ (7.03 ppm).
Figure 3: Olefin and alkylidene-proton region of the 1H NMR (top) and NOE difference spectrum (bottom) of 1e in CD2Cl2 at −40 °C, saturating the methylidene H signal at δ = 18.9 ppm.
Figure 3: Olefin and alkylidene-proton region of the 1H NMR (top) and NOE difference spectrum (bottom) of 1e ...
Scheme 5: Conformational isomerism in complex 1e.
Scheme 5: Conformational isomerism in complex 1e.
Figure 4: Olefin and alkyl group region of the 1H NMR (top) and NOE difference spectrum (bottom) of 1e in CD2Cl2 at −40 °C, saturating the thienyl-H5 signal at δ = 8.07 ppm.
Figure 4: Olefin and alkyl group region of the 1H NMR (top) and NOE difference spectrum (bottom) of 1e in CD2...
The thiophene region of the 1H NMR spectrum of 1e was simulated and iteratively fitted to the experimental spectra in order to evaluate the rate constants at various temperatures (Supporting Information File 1). Linear regression analysis of these data gave activation enthalpy ΔH≠= 13.7 kcal/mol. From the rate constant at 303 K the value of free energy of activation (ΔG≠303K = 12.6 kcal/mol) was also calculated. This is substantially higher than several calculated (Ea = 4.4 kcal/mol) [11,12] and experimentally estimated (Ea = 5.7 kcal/mol) [13] internal rotation barriers of styrene, 2-vinylthiophene (Ea = 4.8 kcal/mol) [14] and is comparable with rotation barrier of the aryl ring in chromium carbene complexes (ΔG≠298K = 13.0–16.2 kcal/mol) [15].
Experimental
Routine, 2D-correlation spectra (1H,1H-COSY) and SELNOE experiments were recorded on a Bruker Avance-400 (BPFO-Plus with Z gradient) spectrometers. 1H NMR chemical shifts are reported in ppm relative to TMS at 0 ppm. IR spectra were recorded on a Tensor 27 FT-IR Spectrometer (Bruker) with MKII Golden Gate Single Reflection Diamond ATR System. For ESI-MS spectra, a Q-TOF Premier (Waters) was used. All solvents used were anhydrous grade purchased from Aldrich. Commercially available compounds were used without further purification. 2-Vinylthiophene [16], 2-vinylfuran [17], 1-vinylcyclohexene [18], 1-vinylcyclopentene [18] and nopadiene [19] were prepared according to known procedures.
2-Vinylindene was prepared by a slightly modified literature procedure [18]: A solution of 2-indanone (5 g, 38 mmol) in dry THF (10 mL) was added over 10 min to a cooled (ice bath) and stirred solution of vinylmagnesium chloride (1.6 M in THF, 36 mL, 57 mmol). The mixture was stirred at 60 °C for a further 30 min and then cooled, quenched with saturated NH4Cl solution, and finally extracted thoroughly with ether. The combined organic extracts were washed with brine, dried, and concentrated at reduced pressure. The residue was dissolved in pyridine (30 mL). POCl3 (4.5 mL, 45 mmol) was slowly added to this solution at 4 °C under an argon atmosphere. The resulting mixture was stirred for further 10 h in an ice bath and then slowly allowed to warm to ambient temperature overnight. The resulting dark brown mixture was poured into ice water and the product extracted with ether. The extracts were washed successively with 2N HCl and then brine. After drying and filtration through a short pad of silica gel, the crude product was purified by distillation to yield 2-vinylindene (2.59 g, 18.2 mmol, 48%) as a colorless liquid, bp 93–94 °C/200 mbar. 1H NMR (CDCl3): δ = 7.38 (d, J = 7.1 Hz, 1H), 7.30 (d, J = 7.4 Hz, 1H), 7.21 (dd, J = 8.0 Hz, J = 7.5 Hz, 1H), 7.21 (dt, J = 1.1 Hz, J = 7.4 Hz, 1H), 6.74 (dd, J = 17.5 Hz, J = 10.6 Hz, 1H), 6.71 (s, 1H), 5.41 (d, J = 17.4 Hz, 1H), 7.30 (d, J = 10.6 Hz, 1H), 3.52 (s, 2H) ppm.
Dichlorobis(tricyclohexylphosphine)(ethylidene)ruthenium(II) (1a): 1,8-Diazabicyclo[5.4.0]undec-7-ene (3.3 mL, 22 mmol) and tricyclohexylphosphine (6.17 g, 22 mmol) were added under an argon atmosphere to a suspension of dichloro(1,5-cyclooctadiene)ruthenium(II) (2.8 g, 10 mmol) in isopropanol (100 mL). The resulting mixture was heated at reflux for 2 h. THF (150 mL) was added to the resulting brick-red suspension which was allowed to cool to 15 °C prior to the addition of 2M HCl in ether (12 mL). After stirring for 5 min, trimethylsilylacetylene (4.2 mL, 30 mmol) was added and the resulting purple colored mixture stirred in an ice bath for 3 h. THF was then evaporated at 4 °C in order to complete the precipitation. The solid product was filtered by suction, washed thoroughly with chilled methanol and vacuum dried at 0–5 °C to give 6.85 g (90%) of purple crystals. 31P NMR (CDCl3): δ = 35.8 ppm; 1H NMR (CDCl3): δ = 19.30 (q, J = 5.6 Hz, 1H), 2.60 (d, J = 5.5 Hz, 3H), 2.60–2.52 (m, 6H), 1.88–1.22 (m, 60H) ppm.
General procedure A for the synthesis of alkylidene complexes: (PCy3)Cl2Ru=CHMe (1a) (1 mmol) was added to a stirred and cooled (ice bath) solution containing a four-fold excess of the respective olefin in degassed CH2Cl2 (25 mL). Argon was bubbled through the resulting dark violet solution for 2 h at 4 °C and then for a further 30 min at room temperature. The reaction mixture was again chilled in ice bath. Degassed methanol (20 mL) was added and the CH2Cl2 removed in vacuo at 0–5 °C. To complete the precipitation another portion of degassed chilled methanol (10 mL) was added and the precipitated product was filtered by suction. The resulting solid was washed thoroughly with chilled methanol, sucked as dry as possible, washed with hexane and dried under vacuum.
General procedure B for the synthesis of alkylidene complexes: (PCy3)Cl2Ru=CHMe (1a) (1 mmol) was added to a stirred and cooled (ice bath) solution containing a four-fold excess of the respective olefin in degassed toluene (25 mL). Argon was bubbled through the resulting dark violet solution for 2 h at 4 °C and then for a further 30 min at room temperature. Toluene was removed in vacuum at 20 °C and the residue triturated with chilled methanol (20 mL). The precipitated product was filtered by suction, washed thoroughly with chilled methanol and dried under vacuum.
Dichlorobis(tricyclohexylphosphine)(cyclopenten-1-ylmethylidene)ruthenium(II) (1b): The product (violet solid) was prepared according to general procedure B in 80% yield. 31P NMR (CDCl3): δ = 37.26 ppm; 1H NMR (CDCl3): δ = 19.30 (s, 1H), 6.97 (s, 1H), 3.14 (m, 2H), 2.60 (m, 6H), 1.95–1.11 (m, 64H) ppm. 13C NMR: δ = 285.83, 164.61, 139.83, 36.97, 34.80; 31.95 (t, J = 9.1), 29.63, 27.91 (t, J = 5.0), 26.64, 25.15.
Dichlorobis(tricyclohexylphosphine)(cyclohexen-1-ylmethylidene)ruthenium(II) (1c): The product as a toluene adduct (intensive violet solid) was prepared according to general procedure B in 46% yield. 31P NMR (C6D6): δ = 36.53 ppm; 1H NMR (C6D6): δ =19.08 (s, 1H), 7.21 (s, 1H), 2.87 (m, 2H), 2.60 (m, 6H), 1.95–1.11 (m, 66H) ppm. 13C NMR: δ = 296.40 (d, J = 113.4), 157.46, 140.27, 32.08 (t, J = 9.1); 30.28, 29.99, 29.70; 27.93 (t, J = 5.0), 27.93, 26.67, 22.97, 21.45. Toluene 137.82, 129.05, 128.24, 125.31, 21.41.
Dichlorobis(tricyclohexylphosphine)(benzylidene)ruthenium(II) (1d): The product (violet solid) was prepared according to general procedure A in 81% yield. The NMR spectra were in agreement with the spectra reported in the literature [5].
Dichlorobis(tricyclohexylphosphine)(thien-2-ylmethylidene)ruthenium(II) (1e): The product (dark violet solid) was prepared according to general procedure A in 71% yield. 31P NMR (CDCl3): δ = 35.96 ppm; 1H NMR (CD2Cl2): δ = 19.05 (s, 1H), 8.09 (s, br., 1H), 7.84 (d, J = 4.1 Hz, 1H), 6.90 (t, J = 4.3 Hz, 1H), 2.55 (m, 6H), 1.75–1.60 (m, 30H), 1.39–1.35 (m, 12H), 1.20–1.12 (m, 18H) ppm. 13C NMR (CDCl3): δ = 269.11, 163.84 (br.), 133.09 (br.), 129.22, 32.26 (t, J 0 9.1), 29.68, 27.85 (t, J = 5.0), 26.55. IR (ATR): λ−1 = 2919 (vs), 2848 (s), 2169 (w), 2051 (w), 1936 (w), 1901 (w), 1443 (m), 1403 (m), 1353 (m), 1263 (m), 1005 (m), 734 (vs) cm−1. MS(ESI): m/z (%) = 828 (21) [M+], 793 (9), 281 (100).
Dichlorobis(tricyclohexylphosphine)(fur-2-ylmethylidene)ruthenium(II) (1f): The product (dark violet solid) was prepared according to general procedure A in 56% yield. 31P NMR (CDCl3): δ = 37.04 ppm; 1H NMR (CDCl3): δ = 18.79 (s, 1H), 8.12 (s, br., 1H), 7.74 (s, br., 1H), 6.43 (dd, J = 3.6 Hz, J = 1.7, 1H), 2.64 (m, 6H), 1.81–1.67 (m, 30H), 1.48–1.41 (m, 12H), 1.27–1.14 (m, 18H) ppm. 13C NMR: δ = 259.90 (d, J = 105.1), 172.34 (br.), 141.71 (br.), 121.54 (br.), 115,44, 32,11 (t, J = 9.0), 29.62, 27.85 (t, J = 5.1), 26.56.
Dichlorobis(tricyclohexylphosphine)(naphth-2-ylmethylidene)ruthenium(II) (1g): The product (dark violet solid) was prepared according to general procedure A in 56% yield. 31P NMR (CDCl3): δ = 37.43 ppm; 1H NMR (CDCl3): δ = 20.12 (s, 1H), 8.82 (s, br., 1H), 8.77 (d, J = 8.5 Hz, 1H), 8.06 (d, J = 8.1 Hz, 1H), 7.74 (d, J = 8.1 Hz, 1H), 7.71 (d, J = 8.4 Hz, 1H), 7.67–7.63 (m, 1H), 7.46–7.42 (m, 1H), 2.63 (m, 6H), 1.90–1.60 (m, 30H), 1.46–1.37 (m, 12H), 1.30–1.10 (m, 18H) ppm. 13C NMR: δ = 292.71, 150.48, 133.98, 133.11, 130.56, 129.77, 129.04, 128.35, 128.05, 127.23, 126.86, 32.19 (t, J = 9.1), 29.70, 27.85 (t, J = 5.1), 26.54. IR (ATR): λ−1 = 2922 (vs), 2848 (s), 2358 (w), 2003 (w), 1443 (m), 1265 (m), 1004 (m), 733 (vs) cm−1. MS (ESI): m/z (%) = 872 (2) [M+], 333 (100).
Dichlorobis(tricyclohexylphosphine)(inden-2-ylmethylidene)ruthenium(II) (1h): The product (brick-red solid) was prepared according to general procedure A in 37% yield. 31P NMR (CDCl3): δ = 36.93 ppm; 1H NMR (CDCl3): δ = 19.64 (s, 1H), 7.94 (s, br., 1H), 7.73 (d, J = 7.7 Hz, 1H), 7.45 (m, 3H), 4.23 (s, 2H), 2.63 (m, 6H), 1.81 (d, J = 12.0 Hz, 18H), 1.70 (dd, J = 23.7, J = 11.9, 12H), 1.47 (dd, J = 23.7 Hz, J = 11.9 Hz, 12H), 1.28–1.17 (m, 18H) ppm.
Dichlorobis(tricyclohexylphosphine)(norpinanylmethylidene)ruthenium(II) (1i): The product (violet solid) was prepared according to general procedure B in 43 % yield. 31P NMR (CDCl3): δ = 36.54 ppm; 1H NMR (CDCl3): δ = 19.12 (s, 1H), 7.65 (s, 1H), 3.02 (m, 1H), 2.48 (m, 6H), 2.30 (m, 1H), 2.09–1.12 (m, 67H), 0.70 (s, 3H) ppm. 13C NMR: δ = 291.46, 163.88, 134.87, 49.14, 39.66, 38.84, 34.77, 31.98 (t, J = 9.0), 31.84, 29.72 (d, J = 11.0), 27.93 (t, J = 4.7), 26.60, 26.45, 20.77 ppm.
Dichlorobis(tricyclohexylphosphine)(2-phenylvinylylidene)ruthenium(II) (2): 1,8-Diazabicyclo[5.4.0]undec-7-ene (0.75 mL, 5.2 mmol) and a 20% solution of tricyclohexylphosphine in toluene (7.7 mL, 5.9 mmol) were added under an argon atmosphere to a suspension of dichloro(1,5-cyclooctadiene)ruthenium(II) (660 mg, 2.35 mmol) in isopropanol (20 mL). The mixture was heated at reflux under an argon atmosphere for 1 h. Toluene (24 mL) was added to the resulting brick-red suspension and the mixture heated for further 30 min at reflux and then allowed to cool to 5–10 °C. 1-Phenyl-2-trimethylsilylacetylene (1.4 ml, 7 mmol) was added followed 10 min later by HCl in ether (2M, 2.4 mL, 4.8 mmol). The resulting purple colored mixture was stirred at ambient temperature for 2h and then concentrated. The residue was treated with 40 mL of chilled methanol and the precipitated product was filtered by suction. The solid was washed thoroughly with chilled methanol and dried under vacuum at 0–5 °C to yield 826 mg (42%) of a violet solid. 31P NMR (CDCl3): δ = 22.54 ppm; 1H NMR (CDCl3): δ = 7.13 (t, J = 7.6 Hz, 2H), 6.89 (d, J = 7.5 Hz, 2H), 6.84 (t, J = 7.2 Hz, 1H), 4.35 (t, J = 3.3 Hz, 1H), 2.62 (m, 6H), 2.06 (d, J = 12.3 Hz, 12H), 1.66–1.73 (m, 18H), 1.59 (dd, J = 23.6 Hz, J = 11.9 Hz, 12H), 1.16–1.26 (m, 18H) ppm. These data are in agreement with the literature [20].
Dichlorobis(tricyclohexylphosphine)(2-butylvinylidene)ruthenium(II) (3): 1,8-Diazabicyclo[5.4.0]undec-7-ene (0.75 mL, 52 mmol) and 20% solution of tricyclohexylphosphine in toluene (7.7 mL, 5.9 mmol) were added under an argon atmosphere to a suspension of dichloro(1,5-cyclooctadiene)ruthenium(II) (660 mg, 2.35 mmol) in isopropanol (20 mL). The mixture was then heated at reflux under an argon atmosphere for 1 h. Toluene (24 mL) was added to the resulting brick-red suspension and the mixture heated for further 30 min at reflux and then allowed to cool to 5–10 °C. 1-Trimethylsilyl-1-hexyne (1.4 mL, 7 mmol) was added followed 10 min later by HCl in ether (2M, 2.4 mL, 4.8 mmol) and the resulting purple colored mixture stirred at ambient temperature for 2 h and then concentrated. The residue was treated with 40 mL of chilled methanol and the precipitated product was filtered by suction. The solid was washed thoroughly with chilled methanol and dried under vacuum to give 720 mg (38%) of a red-brown solid. 31P NMR (CDCl3): δ = 25.34 ppm; 1H NMR (CDCl3): δ = 3.41 (tt, J = 7.3 Hz, J = 1.7 Hz, 1H), 2.59 (m, 6H), 2.36 (dd, J = 14.0 Hz, J = 7.1 Hz, 2H), 2.06 (d, J = 12.1 Hz, 12H), 1.72–1.81 (m, 20H), 1.59 (dd, J = 22.7 Hz, J = 11.5 Hz, 12H), 1.16–1.26 (m, 22H), 0.87 (t, J = 6.8 Hz, 3H) ppm.
Supporting Information
Features variable-temperature and simulated 1H NMR spectra of various compounds, Arrhenius plot of the equilibrium constants for 1e and Eyring plot of the rate constants for 1e interconversion.
| Supporting Information File 1: Detailed experimental data. | ||
| Format: PDF | Size: 692.8 KB | Download |
References
-
Weskamp, T.; Schattenmann, W. C.; Spiegler, M.; Herrmann, W. A. Angew. Chem. 1998, 110, 2631–2633. doi:10.1002/(SICI)1521-3757(19980918)110:18<2631::AID-ANGE2631>3.0.CO;2-J
Angew. Chem. Int. Ed. 1998, 37, 2490–2493.
Return to citation in text: [1] -
Diesendruck, C. E.; Tzur, E.; Lemcoff, N. G. Eur. J. Inorg. Chem. 2009, 4185–4203. doi:10.1002/ejic.200900526
Return to citation in text: [1] -
Samojłowicz, C.; Bieniek, M.; Grela, K. Chem. Rev. 2009, 109, 3708–3742. doi:10.1021/cr800524f
Return to citation in text: [1] -
Vougioukalakis, G. C.; Grubbs, R. H. Chem. Rev. 2010, 110, 1746–1787. doi:10.1021/cr9002424
Return to citation in text: [1] -
Schwab, P.; Grubbs, R. H.; Ziller, J. W. J. Am. Chem. Soc. 1996, 118, 100–110. doi:10.1021/ja952676d
Return to citation in text: [1] [2] -
Wilhelm, T. E.; Belderrain, T. R.; Brown, S. N.; Grubbs, R. H. Organometallics 1997, 16, 3867–3869. doi:10.1021/om9705259
Return to citation in text: [1] -
Grünwald, C.; Gevert, O.; Wolf, J.; González-Herrero, P.; Werner, H. Organometallics 1996, 15, 1960–1962. doi:10.1021/om9509554
Return to citation in text: [1] [2] -
van der Schaaf, P. A.; Kolly, R.; Hafner, A. Chem. Commun. 2000, 1045–1046. doi:10.1039/b002298p
Return to citation in text: [1] [2] -
van der Schaaf, P. A.; Kolly, R.; Kirner, H.-J.; Rime, F.; Mühlebach, A.; Hafner, A. J. Organomet. Chem. 2000, 606, 65–74. doi:10.1016/S0022-328X(00)00289-8
Return to citation in text: [1] -
Wolf, J.; Stüer, W.; Grünwald, C.; Werner, H.; Schwab, P.; Schulz, M. Angew. Chem., Int. Ed. 1998, 37, 1124–1126. doi:10.1002/(SICI)1521-3773(19980504)37:8<1124::AID-ANIE1124>3.0.CO;2-C
Angew. Chem. 1998, 110, 1165–1167.
Return to citation in text: [1] -
Sebbar, N.; Bockhorn, H.; Bozzelli, J. W. J. Phys. Chem. A 2005, 109, 2233–2253. doi:10.1021/jp046285+
Return to citation in text: [1] -
Head-Gordon, M.; Pople, J. A. J. Phys. Chem. 1993, 97, 1147–1151. doi:10.1021/j100108a008
Return to citation in text: [1] -
Grajcar, L.; Baudet, J. J. Mol. Struct. 1977, 38, 121–134. doi:10.1016/0022-2860(77)87084-1
Return to citation in text: [1] -
Parr, W. J. E.; Wasylishen, R. E.; Schaefer, T. Can. J. Chem. 1976, 54, 3216–3223. doi:10.1139/v76-459
Return to citation in text: [1] -
Tobrman, T.; Meca, L.; Dvořáková, H.; Černý, J.; Dvořák, D. Organometallics 2006, 25, 5540–5548. doi:10.1021/om0605837
Return to citation in text: [1] -
Emerson, W. S.; Patrick, T. M., Jr. Org. Synth. 1963, Coll. Vol. 4, 980–983.
Return to citation in text: [1] -
Schmidt, U.; Werner, J. Synthesis 1986, 1986, 986–992. doi:10.1055/s-1986-31846
Return to citation in text: [1] -
Herz, W.; Juo, R.-R. J. Org. Chem. 1985, 50, 618–627. doi:10.1021/jo00205a013
Return to citation in text: [1] [2] [3] -
Paquette, L. A.; Gugelchuk, M.; McLaughlin, M. L. J. Org. Chem. 1987, 52, 4732–4740. doi:10.1021/jo00230a015
Return to citation in text: [1] -
Katayama, H.; Ozawa, F. Organometallics 1998, 17, 5190–5196. doi:10.1021/om980582h
Return to citation in text: [1]
| 18. | Herz, W.; Juo, R.-R. J. Org. Chem. 1985, 50, 618–627. doi:10.1021/jo00205a013 |
| 18. | Herz, W.; Juo, R.-R. J. Org. Chem. 1985, 50, 618–627. doi:10.1021/jo00205a013 |
| 19. | Paquette, L. A.; Gugelchuk, M.; McLaughlin, M. L. J. Org. Chem. 1987, 52, 4732–4740. doi:10.1021/jo00230a015 |
| 1. |
Weskamp, T.; Schattenmann, W. C.; Spiegler, M.; Herrmann, W. A. Angew. Chem. 1998, 110, 2631–2633. doi:10.1002/(SICI)1521-3757(19980918)110:18<2631::AID-ANGE2631>3.0.CO;2-J
Angew. Chem. Int. Ed. 1998, 37, 2490–2493. |
| 7. | Grünwald, C.; Gevert, O.; Wolf, J.; González-Herrero, P.; Werner, H. Organometallics 1996, 15, 1960–1962. doi:10.1021/om9509554 |
| 17. | Schmidt, U.; Werner, J. Synthesis 1986, 1986, 986–992. doi:10.1055/s-1986-31846 |
| 6. | Wilhelm, T. E.; Belderrain, T. R.; Brown, S. N.; Grubbs, R. H. Organometallics 1997, 16, 3867–3869. doi:10.1021/om9705259 |
| 18. | Herz, W.; Juo, R.-R. J. Org. Chem. 1985, 50, 618–627. doi:10.1021/jo00205a013 |
| 5. | Schwab, P.; Grubbs, R. H.; Ziller, J. W. J. Am. Chem. Soc. 1996, 118, 100–110. doi:10.1021/ja952676d |
| 15. | Tobrman, T.; Meca, L.; Dvořáková, H.; Černý, J.; Dvořák, D. Organometallics 2006, 25, 5540–5548. doi:10.1021/om0605837 |
| 2. | Diesendruck, C. E.; Tzur, E.; Lemcoff, N. G. Eur. J. Inorg. Chem. 2009, 4185–4203. doi:10.1002/ejic.200900526 |
| 3. | Samojłowicz, C.; Bieniek, M.; Grela, K. Chem. Rev. 2009, 109, 3708–3742. doi:10.1021/cr800524f |
| 4. | Vougioukalakis, G. C.; Grubbs, R. H. Chem. Rev. 2010, 110, 1746–1787. doi:10.1021/cr9002424 |
| 7. | Grünwald, C.; Gevert, O.; Wolf, J.; González-Herrero, P.; Werner, H. Organometallics 1996, 15, 1960–1962. doi:10.1021/om9509554 |
| 13. | Grajcar, L.; Baudet, J. J. Mol. Struct. 1977, 38, 121–134. doi:10.1016/0022-2860(77)87084-1 |
| 8. | van der Schaaf, P. A.; Kolly, R.; Hafner, A. Chem. Commun. 2000, 1045–1046. doi:10.1039/b002298p |
| 14. | Parr, W. J. E.; Wasylishen, R. E.; Schaefer, T. Can. J. Chem. 1976, 54, 3216–3223. doi:10.1139/v76-459 |
| 10. |
Wolf, J.; Stüer, W.; Grünwald, C.; Werner, H.; Schwab, P.; Schulz, M. Angew. Chem., Int. Ed. 1998, 37, 1124–1126. doi:10.1002/(SICI)1521-3773(19980504)37:8<1124::AID-ANIE1124>3.0.CO;2-C
Angew. Chem. 1998, 110, 1165–1167. |
| 5. | Schwab, P.; Grubbs, R. H.; Ziller, J. W. J. Am. Chem. Soc. 1996, 118, 100–110. doi:10.1021/ja952676d |
| 8. | van der Schaaf, P. A.; Kolly, R.; Hafner, A. Chem. Commun. 2000, 1045–1046. doi:10.1039/b002298p |
| 9. | van der Schaaf, P. A.; Kolly, R.; Kirner, H.-J.; Rime, F.; Mühlebach, A.; Hafner, A. J. Organomet. Chem. 2000, 606, 65–74. doi:10.1016/S0022-328X(00)00289-8 |
| 11. | Sebbar, N.; Bockhorn, H.; Bozzelli, J. W. J. Phys. Chem. A 2005, 109, 2233–2253. doi:10.1021/jp046285+ |
| 12. | Head-Gordon, M.; Pople, J. A. J. Phys. Chem. 1993, 97, 1147–1151. doi:10.1021/j100108a008 |
| 20. | Katayama, H.; Ozawa, F. Organometallics 1998, 17, 5190–5196. doi:10.1021/om980582h |
© 2011 Kadyrov and Rosiak; licensee Beilstein-Institut.
This is an Open Access article under the terms of the Creative Commons Attribution License (http://creativecommons.org/licenses/by/2.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
The license is subject to the Beilstein Journal of Organic Chemistry terms and conditions: (http://www.beilstein-journals.org/bjoc)