Abstract
Reports of the gas sensing properties of ZnSe are few, presumably because of the decomposition and oxidation of ZnSe at high temperatures. In this study, ZnSe nanowires were synthesized by the thermal evaporation of ZnSe powders and the sensing performance of multiple-networked ZnSe nanowire sensors toward NO2 gas was examined. The results showed that ZnSe might be a promising gas sensor material if it is used at room temperature. The response of the ZnSe nanowires to 50 ppb–5 ppm NO2 at room temperature under dark and UV illumination conditions were 101–102% and 113–234%, respectively. The responses of the ZnSe nanowires to 5 ppm NO2 increased from 102 to 234% with increasing UV illumination intensity from 0 to 1.2 mW/cm2. The response of the ZnSe nanowires was stronger than or comparable to that of typical metal oxide semiconductors reported in the literature, which require higher NO2 concentrations and operate at higher temperatures. The origin of the enhanced response of the ZnSe nanowires towards NO2 under UV illumination is also discussed.
Introduction
ZnSe has been widely used in fabricating short-wave optoelectronic devices [1] including blue–green laser diodes [2], tunable mid-IR laser diodes for remote sensing [3], white-light LEDs [4], continuous wave ZnSe-based laser diodes [5] and UV photodetectors [6]. On the other hand, there are almost no reports on the gas sensing properties of ZnSe. This might be due to the decomposition and oxidation of ZnSe at temperatures above 200 °C [7] and a lack of good sensing performance at room temperature.
In recent years, one-dimensionally (1D) nanostructured, metal oxide semiconductor sensors have been studied extensively because of the associated higher sensitivity due to the high surface-to-volume ratios as compared to thin film gas sensors [8-13]. Most metal oxides exhibit some sensitivity to many gases at high temperatures because gas sensitivity tends to increase with increasing temperature. On the other hand, the development of highly sensitive and selective sensors at room temperature is still a challenge. Several techniques including the doping [12,14,15] or surface functionalization [16-18] of metal catalysts, core–shell structure formation [19-21] and UV irradiation [22-24] have been developed to improve the sensing performance, detection limit and selectivity of 1D nanostructure sensors at room temperature. Among these techniques, the UV illumination method was used in the present study to enhance the sensing performance of ZnSe, 1D nanostructure-based sensors at room temperature. In this study, multiple-networked ZnSe nanowire sensors were fabricated and examined for their room-temperature, NO2-gas sensing properties under UV illumination. Unlike individual 1D nanostructure sensors, multiple-networked 1D-nanostructured sensors have the benefits of low sensor fabrication cost (because there is no need for precise techniques to connect the nanostructures), as well as outstanding sensing performance.
Results and Discussion
Analysis of the structure of ZnSe nanowires
Figure 1a shows a SEM image of the ZnSe, 1D nanostructures. The 1D nanostructures exhibited a wire- or fiber-like morphology with widths ranging from 30 to 100 nm and lengths ranging up to ≈300 μm. Figure 1b shows the corresponding XRD pattern of the ZnSe nanowires. The XRD pattern of the ZnSe nanowires showed six sharp reflection peaks assigned to wurtzite-structured ZnSe with lattice constants of a = 0.3996 nm and c = 0.6626 nm (JCPDS No. 89-2940), suggesting that the nanowires were crystalline.
![[2190-4286-5-194-1]](/bjnano/content/figures/2190-4286-5-194-1.png?scale=1.6&max-width=1024&background=FFFFFF)
Figure 1: (a) SEM image of ZnSe nanowires. (b) XRD pattern of the ZnSe nanowires.
Figure 1: (a) SEM image of ZnSe nanowires. (b) XRD pattern of the ZnSe nanowires.
The low-magnification TEM image in Figure 2a revealed a typical ZnSe nanowire with an extremely uniform diameter of approximately 80 nm. The HRTEM image in Figure 2b confirmed that the core region of the nanowire was perfectly crystalline, whereas the edge region showed twinning along the axis of the nanowire. Fringes with spacings of 0.346 and 0.331 nm corresponding to the interplanar distances of the {100} and {002} lattice planes, respectively, were clearly observed in the core region. The corresponding selected area in the electron diffraction pattern (Figure 2c) exhibited two types of reflection spots assigned to wurtzite-structured ZnSe: a round reflection from the core region and an elongated reflection from the edge region.
![[2190-4286-5-194-2]](/bjnano/content/figures/2190-4286-5-194-2.png?scale=2.0&max-width=1024&background=FFFFFF)
Figure 2: (a) Low-magnification TEM image of a typical ZnSe nanowire, (b) high-resolution TEM image of a typical ZnSe nanowire and (c) corresponding SAED pattern.
Figure 2: (a) Low-magnification TEM image of a typical ZnSe nanowire, (b) high-resolution TEM image of a typi...
The corresponding selected area electron diffraction pattern (Figure 2c) exhibited two types of reflection spots assigned to wurtzite-structured ZnSe: round one from the core region and elongated one from the edge region.
Performance of nanowire gas sensors
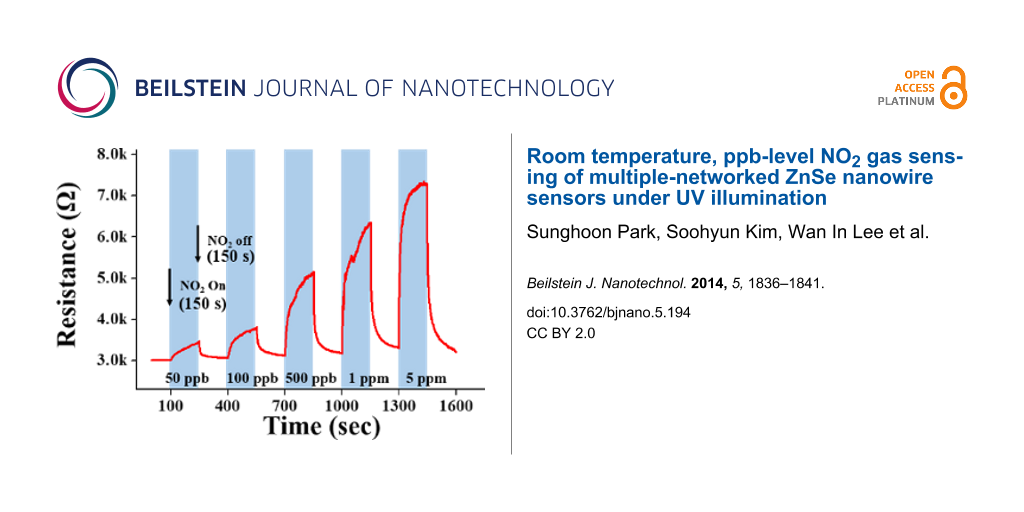
Figure 3a and Figure 3b show the dynamic response of the ZnSe nanowires towards NO2 gas at room temperature in the dark and under UV illumination, respectively. The maximum resistance was reached immediately upon exposure to NO2 and recovered completely to the initial value after the removal of NO2. The resistance increased with increasing NO2 concentration. The resistance showed good reversibility during the introduction and exhaust cycles of NO2. The ZnSe nanowires showed responses to 50 ppb–5 ppm NO2 ranging from ≈101% to ≈102% and from ≈113% to ≈234% in the dark and under UV (365 nm) illumination, respectively. In other words, UV (365 nm) irradiation increased the response of the ZnSe nanowires to 50 ppb–5 ppm NO2 by 1.1–2.3 times.
![[2190-4286-5-194-3]](/bjnano/content/figures/2190-4286-5-194-3.png?scale=1.8&max-width=1024&background=FFFFFF)
Figure 3: Electrical responses of the gas sensors fabricated from ZnSe nanowires to 50 ppb, 100 ppb, 500 ppb, 1 ppm and 5 ppm NO2 gas at room temperature (a) in the dark and (b) under UV (365 nm) illumination at 1.2 mW/cm2.
Figure 3: Electrical responses of the gas sensors fabricated from ZnSe nanowires to 50 ppb, 100 ppb, 500 ppb,...
Figure 4a–c show the responses, response times and recovery times of the ZnSe nanowires towards NO2 gas at room temperature in the dark and under UV illumination, respectively. This information was determined from that of Figure 3a and Figure 3b. The ZnSe nanowires showed a sufficiently strong response to NO2 gas, even at 50 ppb. The response of the ZnSe nanowires to NO2 gas tended to increase more rapidly with increasing NO2 concentrations under UV illumination than in the dark. Regarding the sensing time, both the response times and recovery times were shorter under UV illumination than in the dark. In particular, recovery times were more than 35 s shorter under UV illumination than in the dark at a NO2 gas concentration range from 50 ppb to 5 ppm. Figure 4a–c show the dependence of the response, response time and recovery times of the ZnSe nanowires to 5 ppm NO2 gas at room temperature on the illumination intensity of UV light used to illuminate the gas sensors. The response of the nanowires was 102% in the dark. The responses of the nanowires increased from ≈102 to ≈234% with increasing UV illumination intensity from 0 to 1.2 mW/cm2 (Figure 4a). Figure 5a shows a strong dependence of the electrical response of the ZnSe nanowires on the UV illumination intensity towards 5 ppm NO2 gas at room temperature. The response increased rapidly with increasing UV illumination intensity. On the other hand, Figure 5a and Figure 5b show that both the response time and recovery time of the ZnSe nanowires at room temperature towards 5 ppm NO2 gas tend to decrease with the UV illumination intensity. These high responses at room temperature highlight the strong influence of UV irradiation on the response of the nanosensor to NO2 gas.
![[2190-4286-5-194-4]](/bjnano/content/figures/2190-4286-5-194-4.png?scale=2.0&max-width=1024&background=FFFFFF)
Figure 4: (a) Response, (b) response times and (c) recovery times of the multiple-networked ZnSe nanowire gas sensor to NO2 gas in the dark and under 365 nm UV illumination at 1.2 mW/cm2.
Figure 4: (a) Response, (b) response times and (c) recovery times of the multiple-networked ZnSe nanowire gas...
![[2190-4286-5-194-5]](/bjnano/content/figures/2190-4286-5-194-5.png?scale=2.0&max-width=1024&background=FFFFFF)
Figure 5: (a) Electrical response and (b) response and recovery times of ZnSe nanowire gas sensors under UV (365 nm) illumination for different UV illumination intensities.
Figure 5: (a) Electrical response and (b) response and recovery times of ZnSe nanowire gas sensors under UV (...
Table 1 compares the responses of the ZnSe nanowires towards NO2 synthesized in this study with those of metal oxide semiconductor, 1D nanostructures reported in the literature. The response of the ZnSe nanowires to NO2 gas with a lower concentration obtained at room temperature in the dark in this study was stronger than or comparable to those of typical metal oxide, 1D nanostructures, such as ZnO, SnO2, In2O3, and MoO3 at higher temperatures and higher NO2 concentrations [25-30]. This suggests that the ZnSe nanowires are also a promising candidate as a NO2 gas sensor material.
Table 1: Responses of various nanomaterial gas sensors to NO2 gas.
| Nanomaterial |
Temperature
(°C) |
NO2 Conc.
(ppm) |
Response
(%) |
Ref. |
|---|---|---|---|---|
| ZnSe NWs (dark) | 25 | 0.05 | 101 | Present work |
| ZnSe NWs (UV) | 25 | 0.05 | 113 | Present work |
| ZnO nanorods | 300 | 0.1 | 35 | [24] |
| ZnO fibers | 100 | 0.4 | 50 | [25] |
| SnO2 NWs | 300 | 10 | 1.01 | [26] |
| SnO2 nanobelts | 300 | 10 | 1.9 | [27] |
| In2O3 NWs (multi-NW) | 200 | 0.5 | 2.1 | [28] |
| In2O3 NWs (single-NW) | 200 | 0.5 | 2.6 | [28] |
| MoO3 lamellae | 225 | 10 | 118 | [29] |
Conclusion
ZnSe nanowires exhibited responses towards 50 ppb–5 ppm NO2 ranging from ≈101% to ≈102% and from ≈113% to ≈234% at room temperature in the dark and under UV (365 nm) illumination, respectively. These responses of ZnSe nanowires were stronger than or comparable to those of typical metal oxide semiconductors reported in the literature, such as ZnO, SnO2, In2O3, and MoO3, at higher temperatures and higher NO2 concentrations. The ZnSe nanowire sensors cannot be used at high temperatures, such as 300 °C, because of the oxidation of ZnSe, but their sensing performance could be enhanced when used at room temperature under UV illumination. The response of the ZnSe nanowires increased from 0 to ≈234% with increasing UV illumination intensity from 0 to 1.2 mW/cm2 and the response time and recovery time of the ZnSe nanowires tended to decrease with increasing UV illumination intensity from 0 to 1.2 mW/cm2. The results show that ZnSe nanowires are also a promising nanomaterial for the fabrication of NO2 gas sensors when used at room temperature. In addition, the enhanced response of the ZnSe nanowires under UV illumination to NO2 gas might be due to (1) modulation of the depletion layer width and (2) the UV-activated adsorption, and desorption of NO2 species.
Experimental
Synthesis of ZnSe nanowires
Similar to that previously described [31], ZnSe nanowires were synthesized on 3 nm-thick gold (Au) layer-coated, c-plane sapphire (Al2O3(0001)) substrates by the thermal evaporation of ZnSe powders. A quartz tube was mounted inside a horizontal tube furnace. The quartz tube consisted of two temperature zones: zone A at 850 °C and zone B at 700 °C. An alumina boat loaded with pure ZnSe powder was located in zone A, whereas the Au-coated Al2O3 substrate was placed in zone B. The nitrogen (N2) gas flow rate and chamber pressure were 100 cm3/min and 1 Torr, respectively. The synthesis process time was 1 h.
Characterization of the structure of the nanowires
The morphology and structure of the collected nanowire samples were examined by scanning electron microscopy (SEM, Hitachi S-4200) and transmission electron microscopy (TEM, Philips CM-200), respectively. The crystallographic structures of the samples were determined by glancing angle X-ray diffraction (XRD, Philips X’pert MRD diffractometer) using Cu Kα radiation (λ = 0.15406 nm) at a scan rate of 4°/min, and a 0.5° glancing angle with a rotating detector.
Preparation of sensors and gas sensing tests
ZnSe nanowire samples were dispersed ultrasonically in a mixture of deionized water (5 mL) and isopropyl alcohol (5 mL), and dried at 90 °C for 30 min. A slurry droplet containing the nanowires (10 µL) was placed onto the SiO2-coated Si substrates equipped with a pair of interdigitated (IDE) Ni (≈200 nm)/Au (≈50 nm) electrodes with a gap of 20 μm. The flow-through technique was used to test the gas sensing properties. All measurements were performed in a temperature-stabilized, sealed chamber with a constant flow rate of 200 cm3/min at 25 °C under 50% RH. The NO2 concentration was controlled by mixing NO2 gas with synthetic air at different ratios. The detailed procedures for the sensor fabrication and sensing test are reported elsewhere [32]. The electrical resistance of the gas sensors was determined in the dark and under UV light (λ = 365 nm) illumination at intensities ranging from 0.35 to 1.2 mW/cm2 by measuring the electric current between the Ni/Au IDEs at 1 V and at room temperature. The response was defined as (Rg/Ra) × 100% for NO2 gas, where Rg and Ra are the electrical resistances of the sensors in the target gas and air, respectively.
References
-
Park, S.; An, S.; Ko, H.; Lee, C. Mater. Chem. Phys. 2014, 143, 735–739. doi:10.1016/j.matchemphys.2013.10.007
Return to citation in text: [1] -
Ma, R.; Bando, Y. Chem. Phys. Lett. 2003, 374, 358–361. doi:10.1016/S0009-2614(03)00776-0
Return to citation in text: [1] -
Mirov, S. B.; Fedorov, V. V.; Graham, K.; Moskalev, I. S.; Badikov, V. V.; Panyutin, V. Opt. Lett. 2002, 27, 909–911. doi:10.1364/OL.27.000909
Return to citation in text: [1] -
Katayama, K.; Matsubara, H.; Nakanishi, F.; Nakamura, T.; Doi, H.; Saegusa, A.; Mitsui, T.; Matsuoka, T.; Irikusa, M.; Takebe, T.; Nishine, S.; Shirakawa, T. J. Cryst. Growth 2000, 214, 1064–1070. doi:10.1016/S0022-0248(00)00275-X
Return to citation in text: [1] -
Okuyama, H. Trans. Inst. Electron., Inf. Commun. Eng., Sect. E 2000, E83-C, 536–545.
Return to citation in text: [1] -
Chang, S. J.; Su, Y. K.; Chen, W. R.; Chen, J. F.; Lan, W. H.; Lin, W. J.; Cherng, Y. T.; Liu, C. H.; Liaw, U. H. IEEE Photonics Technol. Lett. 2002, 14, 188–190. doi:10.1109/68.980508
Return to citation in text: [1] -
Dow Chemical Corporate Website - The Dow Chemical Company. http://www.dow.com/assets/attachments/business/gt/infrared_materials/cvd_zinc_selenide/tds/cvd_zinc_selenide.pdf (accessed Oct 8, 2014).
Return to citation in text: [1] -
Kim, H.; Jin, C.; An, S.; Lee, C. Ceram. Int. 2012, 38, 3563–3567. doi:10.1016/j.ceramint.2011.12.072
Return to citation in text: [1] -
Kolmakov, A.; Zhang, Y.; Cheng, G.; Moskovits, M. Adv. Mater. 2003, 15, 997–1000. doi:10.1002/adma.200304889
Return to citation in text: [1] -
Liu, Y.; Koep, E.; Liu, M. Chem. Mater. 2005, 17, 3997–4000. doi:10.1021/cm050451o
Return to citation in text: [1] -
Law, M.; Kind, H.; Messer, B.; Kim, F.; Yang, P. Angew. Chem. 2002, 114, 2511–2514. doi:10.1002/1521-3757(20020703)114:13<2511::AID-ANGE2511>3.0.CO;2-N
Return to citation in text: [1] -
Lin, Y.-H.; Huang, M.-W.; Liu, C.-K.; Chen, J.-R.; Wu, J.-M.; Shih, H.-C. J. Electrochem. Soc. 2009, 156, K196–K199. doi:10.1149/1.3223984
Return to citation in text: [1] [2] -
Kim, H. S.; Jin, C. H.; Park, S. H.; Kim, S. I.; Lee, C. Sens. Actuators, B 2012, 161, 594–599. doi:10.1016/j.snb.2011.11.006
Return to citation in text: [1] -
Ramgir, N. S.; Mulla, I. S.; Vijayamohanan, K. P. Sens. Actuators, B 2005, 107, 708–715. doi:10.1016/j.snb.2004.12.073
Return to citation in text: [1] -
Wan, Q.; Wang, T. H. Chem. Commun. 2005, 1, 3841–3843. doi:10.1039/b504094a
Return to citation in text: [1] -
Kolmakov, A.; Klenov, D. O.; Lilach, Y.; Stemmer, S.; Moskovits, M. Nano Lett. 2005, 5, 667–673. doi:10.1021/nl050082v
Return to citation in text: [1] -
Kuang, Q.; Lao, C.-S.; Li, Z.; Liu, Y.-Z.; Xie, Z.-X.; Zheng, L.-S.; Wang, Z. L. J. Phys. Chem. C 2008, 112, 11539–11544. doi:10.1021/jp802880c
Return to citation in text: [1] -
Wright, J. S.; Lim, W.; Gila, B. P.; Pearton, S. J.; Johnson, J. L.; Ural, A.; Ren, F. Sens. Actuators, B 2009, 140, 196–199. doi:10.1016/j.snb.2009.04.009
Return to citation in text: [1] -
Tamaki, J.; Shimanoe, K.; Yamada, Y.; Yamamoto, Y.; Miura, N.; Yamazoe, N. Sens. Actuators, B 1998, 49, 121–125. doi:10.1016/S0925-4005(98)00144-0
Return to citation in text: [1] -
Park, S.; Ko, H.; Kim, S.; Lee, C. ACS Appl. Mater. Interfaces 2014, 6, 9595–9596. doi:10.1021/am501975v
Return to citation in text: [1] -
Jin, C.; Park, S.; Kim, H.; Lee, C. Sens. Actuators, B 2012, 161, 223–228. doi:10.1016/j.snb.2011.10.023
Return to citation in text: [1] -
Comini, E.; Cristalli, A.; Faglia, G.; Sberveglieri, G. Sens. Actuators, B 2000, 65, 260–263. doi:10.1016/S0925-4005(99)00350-0
Return to citation in text: [1] -
Gong, J.; Li, Y.; Chai, X.; Hu, Z.; Deng, Y. J. Phys. Chem. C 2010, 114, 1293–1298. doi:10.1021/jp906043k
Return to citation in text: [1] -
Lu, G.; Xu, J.; Sun, J.; Yu, Y.; Zhang, Y.; Liu, F. Sens. Actuators, B 2012, 162, 82–88. doi:10.1016/j.snb.2011.12.039
Return to citation in text: [1] [2] -
Park, S. H.; An, S. Y.; Ko, H. S.; Jin, C. H.; Lee, C. ACS Appl. Mater. Interfaces 2012, 4, 3650–3656. doi:10.1021/am300741r
Return to citation in text: [1] [2] -
Baratto, C.; Sberveglieri, G.; Onischuk, A.; Caruso, B.; di Stasio, S. Sens. Actuators, B 2004, 100, 261–265. doi:10.1016/j.snb.2003.12.045
Return to citation in text: [1] [2] -
Kim, H.; An, S.; Jin, C.; Lee, C. Curr. Appl. Phys. 2012, 12, 1125–1130. doi:10.1016/j.cap.2012.02.006
Return to citation in text: [1] [2] -
Law, M.; Kind, H.; Messer, B.; Kim, F.; Yang, P. Angew. Chem., Int. Ed. 2002, 41, 2405–2408. doi:10.1002/1521-3773(20020703)41:13<2405::AID-ANIE2405>3.0.CO;2-3
Return to citation in text: [1] [2] [3] -
Moon, S. E.; Kim, E.-K.; Lee, H.-Y.; Lee, J.-W.; Park, J.; Park, S.-J.; Kwak, J.-H.; Park, K.-H.; Kim, J.; Jo, G.-H.; Lee, T.-H. J. Korean Phys. Soc. 2009, 54, 830–834. doi:10.3938/jkps.54.830
Return to citation in text: [1] [2] -
Rahmani, M. B.; Keshmiri, S. H.; Yu, J.; Sadek, A. Z.; Al-Mashat, L.; Moafi, A.; Latham, K.; Li, Y. X.; Wlodarski, W.; Kalantar-zadeh, K. Sens. Actuators, B 2010, 145, 13–19. doi:10.1016/j.snb.2009.11.007
Return to citation in text: [1] -
Lee, C.; Jin, C.; Kim, H.; Kim, H. W. Curr. Appl. Phys. 2010, 10, 1017–1021. doi:10.1016/j.cap.2009.12.032
Return to citation in text: [1] -
Oh, E.; Choi, H.-Y.; Jung, S.-H.; Cho, S.; Kim, J. C.; Lee, K.-H.; Kang, S.-W.; Kim, J.; Yun, J.-Y.; Jeong, S.-H. Sens. Actuators, B 2009, 141, 239–243. doi:10.1016/j.snb.2009.06.031
Return to citation in text: [1]
| 28. | Law, M.; Kind, H.; Messer, B.; Kim, F.; Yang, P. Angew. Chem., Int. Ed. 2002, 41, 2405–2408. doi:10.1002/1521-3773(20020703)41:13<2405::AID-ANIE2405>3.0.CO;2-3 |
| 27. | Kim, H.; An, S.; Jin, C.; Lee, C. Curr. Appl. Phys. 2012, 12, 1125–1130. doi:10.1016/j.cap.2012.02.006 |
| 28. | Law, M.; Kind, H.; Messer, B.; Kim, F.; Yang, P. Angew. Chem., Int. Ed. 2002, 41, 2405–2408. doi:10.1002/1521-3773(20020703)41:13<2405::AID-ANIE2405>3.0.CO;2-3 |
| 1. | Park, S.; An, S.; Ko, H.; Lee, C. Mater. Chem. Phys. 2014, 143, 735–739. doi:10.1016/j.matchemphys.2013.10.007 |
| 5. | Okuyama, H. Trans. Inst. Electron., Inf. Commun. Eng., Sect. E 2000, E83-C, 536–545. |
| 25. | Park, S. H.; An, S. Y.; Ko, H. S.; Jin, C. H.; Lee, C. ACS Appl. Mater. Interfaces 2012, 4, 3650–3656. doi:10.1021/am300741r |
| 4. | Katayama, K.; Matsubara, H.; Nakanishi, F.; Nakamura, T.; Doi, H.; Saegusa, A.; Mitsui, T.; Matsuoka, T.; Irikusa, M.; Takebe, T.; Nishine, S.; Shirakawa, T. J. Cryst. Growth 2000, 214, 1064–1070. doi:10.1016/S0022-0248(00)00275-X |
| 26. | Baratto, C.; Sberveglieri, G.; Onischuk, A.; Caruso, B.; di Stasio, S. Sens. Actuators, B 2004, 100, 261–265. doi:10.1016/j.snb.2003.12.045 |
| 3. | Mirov, S. B.; Fedorov, V. V.; Graham, K.; Moskalev, I. S.; Badikov, V. V.; Panyutin, V. Opt. Lett. 2002, 27, 909–911. doi:10.1364/OL.27.000909 |
| 25. | Park, S. H.; An, S. Y.; Ko, H. S.; Jin, C. H.; Lee, C. ACS Appl. Mater. Interfaces 2012, 4, 3650–3656. doi:10.1021/am300741r |
| 26. | Baratto, C.; Sberveglieri, G.; Onischuk, A.; Caruso, B.; di Stasio, S. Sens. Actuators, B 2004, 100, 261–265. doi:10.1016/j.snb.2003.12.045 |
| 27. | Kim, H.; An, S.; Jin, C.; Lee, C. Curr. Appl. Phys. 2012, 12, 1125–1130. doi:10.1016/j.cap.2012.02.006 |
| 28. | Law, M.; Kind, H.; Messer, B.; Kim, F.; Yang, P. Angew. Chem., Int. Ed. 2002, 41, 2405–2408. doi:10.1002/1521-3773(20020703)41:13<2405::AID-ANIE2405>3.0.CO;2-3 |
| 29. | Moon, S. E.; Kim, E.-K.; Lee, H.-Y.; Lee, J.-W.; Park, J.; Park, S.-J.; Kwak, J.-H.; Park, K.-H.; Kim, J.; Jo, G.-H.; Lee, T.-H. J. Korean Phys. Soc. 2009, 54, 830–834. doi:10.3938/jkps.54.830 |
| 30. | Rahmani, M. B.; Keshmiri, S. H.; Yu, J.; Sadek, A. Z.; Al-Mashat, L.; Moafi, A.; Latham, K.; Li, Y. X.; Wlodarski, W.; Kalantar-zadeh, K. Sens. Actuators, B 2010, 145, 13–19. doi:10.1016/j.snb.2009.11.007 |
| 2. | Ma, R.; Bando, Y. Chem. Phys. Lett. 2003, 374, 358–361. doi:10.1016/S0009-2614(03)00776-0 |
| 24. | Lu, G.; Xu, J.; Sun, J.; Yu, Y.; Zhang, Y.; Liu, F. Sens. Actuators, B 2012, 162, 82–88. doi:10.1016/j.snb.2011.12.039 |
| 12. | Lin, Y.-H.; Huang, M.-W.; Liu, C.-K.; Chen, J.-R.; Wu, J.-M.; Shih, H.-C. J. Electrochem. Soc. 2009, 156, K196–K199. doi:10.1149/1.3223984 |
| 14. | Ramgir, N. S.; Mulla, I. S.; Vijayamohanan, K. P. Sens. Actuators, B 2005, 107, 708–715. doi:10.1016/j.snb.2004.12.073 |
| 15. | Wan, Q.; Wang, T. H. Chem. Commun. 2005, 1, 3841–3843. doi:10.1039/b504094a |
| 19. | Tamaki, J.; Shimanoe, K.; Yamada, Y.; Yamamoto, Y.; Miura, N.; Yamazoe, N. Sens. Actuators, B 1998, 49, 121–125. doi:10.1016/S0925-4005(98)00144-0 |
| 20. | Park, S.; Ko, H.; Kim, S.; Lee, C. ACS Appl. Mater. Interfaces 2014, 6, 9595–9596. doi:10.1021/am501975v |
| 21. | Jin, C.; Park, S.; Kim, H.; Lee, C. Sens. Actuators, B 2012, 161, 223–228. doi:10.1016/j.snb.2011.10.023 |
| 32. | Oh, E.; Choi, H.-Y.; Jung, S.-H.; Cho, S.; Kim, J. C.; Lee, K.-H.; Kang, S.-W.; Kim, J.; Yun, J.-Y.; Jeong, S.-H. Sens. Actuators, B 2009, 141, 239–243. doi:10.1016/j.snb.2009.06.031 |
| 8. | Kim, H.; Jin, C.; An, S.; Lee, C. Ceram. Int. 2012, 38, 3563–3567. doi:10.1016/j.ceramint.2011.12.072 |
| 9. | Kolmakov, A.; Zhang, Y.; Cheng, G.; Moskovits, M. Adv. Mater. 2003, 15, 997–1000. doi:10.1002/adma.200304889 |
| 10. | Liu, Y.; Koep, E.; Liu, M. Chem. Mater. 2005, 17, 3997–4000. doi:10.1021/cm050451o |
| 11. | Law, M.; Kind, H.; Messer, B.; Kim, F.; Yang, P. Angew. Chem. 2002, 114, 2511–2514. doi:10.1002/1521-3757(20020703)114:13<2511::AID-ANGE2511>3.0.CO;2-N |
| 12. | Lin, Y.-H.; Huang, M.-W.; Liu, C.-K.; Chen, J.-R.; Wu, J.-M.; Shih, H.-C. J. Electrochem. Soc. 2009, 156, K196–K199. doi:10.1149/1.3223984 |
| 13. | Kim, H. S.; Jin, C. H.; Park, S. H.; Kim, S. I.; Lee, C. Sens. Actuators, B 2012, 161, 594–599. doi:10.1016/j.snb.2011.11.006 |
| 22. | Comini, E.; Cristalli, A.; Faglia, G.; Sberveglieri, G. Sens. Actuators, B 2000, 65, 260–263. doi:10.1016/S0925-4005(99)00350-0 |
| 23. | Gong, J.; Li, Y.; Chai, X.; Hu, Z.; Deng, Y. J. Phys. Chem. C 2010, 114, 1293–1298. doi:10.1021/jp906043k |
| 24. | Lu, G.; Xu, J.; Sun, J.; Yu, Y.; Zhang, Y.; Liu, F. Sens. Actuators, B 2012, 162, 82–88. doi:10.1016/j.snb.2011.12.039 |
| 7. | Dow Chemical Corporate Website - The Dow Chemical Company. http://www.dow.com/assets/attachments/business/gt/infrared_materials/cvd_zinc_selenide/tds/cvd_zinc_selenide.pdf (accessed Oct 8, 2014). |
| 29. | Moon, S. E.; Kim, E.-K.; Lee, H.-Y.; Lee, J.-W.; Park, J.; Park, S.-J.; Kwak, J.-H.; Park, K.-H.; Kim, J.; Jo, G.-H.; Lee, T.-H. J. Korean Phys. Soc. 2009, 54, 830–834. doi:10.3938/jkps.54.830 |
| 6. | Chang, S. J.; Su, Y. K.; Chen, W. R.; Chen, J. F.; Lan, W. H.; Lin, W. J.; Cherng, Y. T.; Liu, C. H.; Liaw, U. H. IEEE Photonics Technol. Lett. 2002, 14, 188–190. doi:10.1109/68.980508 |
| 16. | Kolmakov, A.; Klenov, D. O.; Lilach, Y.; Stemmer, S.; Moskovits, M. Nano Lett. 2005, 5, 667–673. doi:10.1021/nl050082v |
| 17. | Kuang, Q.; Lao, C.-S.; Li, Z.; Liu, Y.-Z.; Xie, Z.-X.; Zheng, L.-S.; Wang, Z. L. J. Phys. Chem. C 2008, 112, 11539–11544. doi:10.1021/jp802880c |
| 18. | Wright, J. S.; Lim, W.; Gila, B. P.; Pearton, S. J.; Johnson, J. L.; Ural, A.; Ren, F. Sens. Actuators, B 2009, 140, 196–199. doi:10.1016/j.snb.2009.04.009 |
| 31. | Lee, C.; Jin, C.; Kim, H.; Kim, H. W. Curr. Appl. Phys. 2010, 10, 1017–1021. doi:10.1016/j.cap.2009.12.032 |
© 2014 Park et al; licensee Beilstein-Institut.
This is an Open Access article under the terms of the Creative Commons Attribution License (http://creativecommons.org/licenses/by/2.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
The license is subject to the Beilstein Journal of Nanotechnology terms and conditions: (http://www.beilstein-journals.org/bjnano)