Abstract
The present review describes the current status of synthetic five and six-membered cyclic peroxides such as 1,2-dioxolanes, 1,2,4-trioxolanes (ozonides), 1,2-dioxanes, 1,2-dioxenes, 1,2,4-trioxanes, and 1,2,4,5-tetraoxanes. The literature from 2000 onwards is surveyed to provide an update on synthesis of cyclic peroxides. The indicated period of time is, on the whole, characterized by the development of new efficient and scale-up methods for the preparation of these cyclic compounds. It was shown that cyclic peroxides remain unchanged throughout the course of a wide range of fundamental organic reactions. Due to these properties, the molecular structures can be greatly modified to give peroxide ring-retaining products. The chemistry of cyclic peroxides has attracted considerable attention, because these compounds are used in medicine for the design of antimalarial, antihelminthic, and antitumor agents.
Graphical Abstract
Introduction
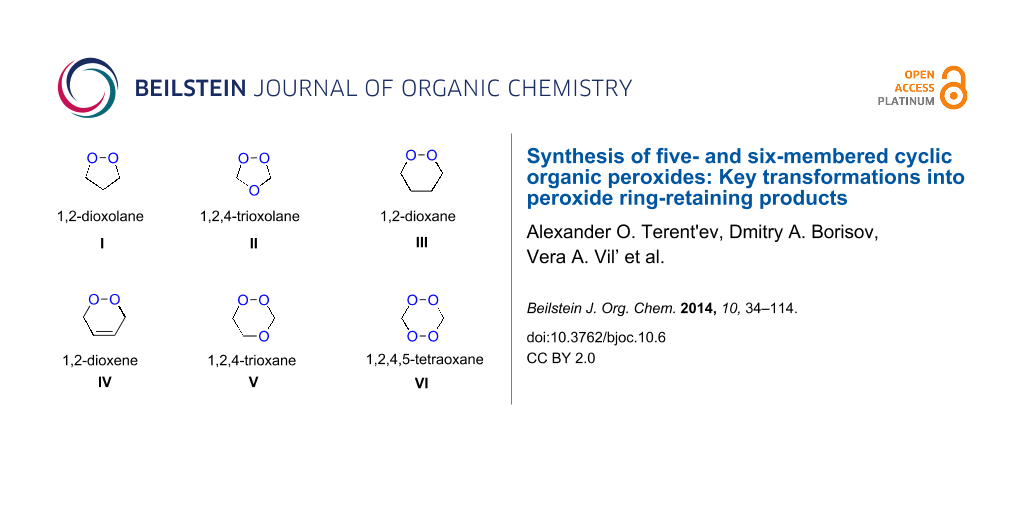
Approaches to the synthesis of five and six-membered cyclic peroxides, such as 1,2-dioxolanes I, 1,2,4-trioxolanes (ozonides) II, 1,2-dioxanes III, 1,2-dioxenes IV, 1,2,4-trioxanes V, and 1,2,4,5-tetraoxanes VI, published from 2000 to present are reviewed. These compounds are widely used in synthetic and medicinal chemistry (Figure 1).
Figure 1: Five and six-membered cyclic peroxides.
Figure 1: Five and six-membered cyclic peroxides.
In the last decade, two reviews on this rapidly progressing field were published by McCullough and Nojima [1] and Korshin and Bachi [2] covering earlier studies. There are several review articles on medicinal chemistry of peroxides, where the problems of their synthesis are briefly considered. In addition to these reviews other publications dealing with this subject appeared: Tang et al. [3], O’Neill, Posner and colleagues [4,5], Masuyama et al. [6], Van Ornum et al. [7], Jefford [8,9], Dembitsky et al. [10-15], Opsenica and Šolaja [16], Muraleedharan and Avery [17], and other [18-27] including dissertations [28-32].
Reviews published earlier on the chemistry of ozone [33-36] and on the chemistry and biological activity of natural peroxides, and cyclic peroxides [37-46] are closely related to this review. Generally speaking, state-of-the-art approaches to the synthesis of cyclic peroxides are based on three key reagents: oxygen, ozone, and hydrogen peroxide. These reagents and their derivatives are used in the main methods for the introduction of the peroxide group, such as the singlet-oxygen ene reaction with alkenes, the [4 + 2]-cycloaddition of singlet oxygen to dienes, the Mukaiyama–Isayama peroxysilylation of unsaturated compounds, the Kobayashi cyclization, the nucleophilic addition of hydrogen peroxide to carbonyl compounds, the ozonolysis, and reactions with the involvement of peroxycarbenium ions.
Each part of the review deals with a particular class of the above-mentioned peroxides in accordance with an increase in the number of oxygen atoms and the ring size. In the individual sections, the data are arranged mainly according to the common key step in the synthesis of the cyclic peroxides. Examples of the synthesis of peroxide derivatives via modifications of functional groups, with the peroxide bond remaining unbroken, are given in the end of each chapter. In most cases, the syntheses of compounds having high biological activity are considered.
Currently, the rapid progress in chemistry of organic peroxides is to a large degree determined by their high biological activity. In medicinal chemistry of peroxides, particular emphasis is given to the design of compounds having activity against causative agents of malaria and helminth infections. The World Health Organization (WHO) considers malaria as one of the most dangerous social diseases. Worldwide, 300–500 million cases of malaria occur each year, and 2 million people die from it [47,48].
Due to a high degree of resistance in malaria to traditional drugs as quinine, chloroquine, and mefloquine, an active search for other classes of new drugs is performed. In this respect, organic peroxides play a considerable role. In medicinal chemistry of peroxides, artemisinin a natural peroxide exhibiting high antimalarial activity, is the most important drug in use for approximately 30 years. Artemisinin was isolated in 1971 from leaves of annual wormwood (Artemesia annua) [49-51]; the 1,2,4-trioxane ring V is the key pharmacophore of these drugs. A series of semi-synthetic derivatives of artemisinin were synthesized: artesunate, artemether, and artemisone (Figure 2). Currently, drugs based on these compounds are considered as the most efficacious for the treatment of malaria [52-76].
Figure 2: Artemisinin and semi-synthetic derivatives.
Figure 2: Artemisinin and semi-synthetic derivatives.
The discovery of arterolane, a synthetic 1,2,4-trioxolane, is a considerable success in the search for easily available synthetic peroxides capable of replacing artemisinin and its derivatives in medical practice. Currently, this compound is currently in phase III clinical trials [77-81].
The mechanism of antimalarial action of peroxides is unusual for pharmaceutical chemistry. According to the commonly accepted mechanism, peroxides diffuse into Plasmodium-infected erythrocytes, and the heme iron ion of the latter reduces the peroxide bond to form a separated oxygen-centered radical anion, which rearranges to the C-centered radical having a toxic effect on Plasmodium [82-87].
In the course of the large-scale search for synthetically accessible and cheap antimalarial peroxides (compared with natural and semi-synthetic structures), it was found that structures containing 1,2-dioxolane [88-90], 1,2,4-trioxolane [91-101], 1,2-dioxane [102-112], 1,2-dioxene [113-119], 1,2,4-trioxane [120-127] or 1,2,4,5-tetraoxane rings [128-146] exhibit pronounced activity, and in some cases, even superior to that of artemisinin.
Another important field of medicinal chemistry of organic peroxides includes the search for antihelminthic drugs. For example, compounds containing 1,2-dioxolane [147], 1,2,4-trioxolane [148-152], 1,2,4-trioxane [153-158] or bridged 1,2,4,5-tetraoxane [159] moieties show activity against Schistosoma. Schistosomiasis is one of the most widespread helminthic diseases; 800 million people are at risk of acquiring this infection [160-174].
Additionally, based on synthetic peroxides, several compounds exhibiting antitumor activity were synthesized. These compounds contain 1,2-dioxolane [10-15,175-178], 1,2-dioxane [10-15,112,178-181], 1,2-dioxene [114,182-185] or 1,2,4-trioxane [10-15,175,176] rings. More than 300 peroxides are known to have a toxic effect on cancer cells [10-15,73,186-206].
Synthetic peroxides exhibit also other activities. For example, compounds containing the 1,2,4-trioxane ring are active against Trichomonas [207], compounds with the 1,2-dioxane ring show antitrypanosomal and antileishmanial activities [208-212], and compounds containing the 1,2-dioxene ring possess fungicidal [210,213-224] and antimycobacterial activities [128-131,225-228]. The present review covers literature relating to 5- and 6-membered cyclic peroxide chemistry published between 2000 and 2013.
Review
1. Synthesis of 1,2-dioxolanes
The modern approaches to the synthesis of 1,2-dioxolanes are based on the use of oxygen and ozone for the formation of the peroxide moiety, the Isayama–Mukaiyama peroxysilylation, and reactions involving peroxycarbenium ions. Syntheses employing hydrogen peroxide and the intramolecular Kobayashi cyclization are less frequently used.
1.1. Use of oxygen for the peroxide ring formation
The singlet-oxygen ene reaction with alkenes provides an efficient tool for introducing the hydroperoxide function. The reaction starts with the coordination of oxygen to the double bond followed by the formation of hydroperoxides presumably by a stepwise or concerted mechanism [229,230]. The oxidation of α,β-unsaturated ketones 1a–c by singlet oxygen affords 3-hydroxy-1,2-dioxolanes 3a–c via the formation of β-hydroperoxy ketones 2a–c (Scheme 1) [231].
Scheme 1: Synthesis of 3-hydroxy-1,2-dioxolanes 3a–c.
Scheme 1: Synthesis of 3-hydroxy-1,2-dioxolanes 3a–c.
Dioxolane 6 was synthesized in 36% yield by the reaction of oxygen with hydroperoxide 4 in the presence of di-tert-butyl peroxalate (DTBPO) followed by the treatment of the reaction mixture with acetic anhydride and pyridine at room temperature (Scheme 2).
It should be emphasized that a mixture of dioxolanes 5 and 6 in a ratio of 7:3 is formed already in the first step [232].
The photooxygenation of oxazolidines 7a–d through the formation of hydroperoxides 8a–d gives spiro-fused oxazolidine-containing dioxolanes 9a–d in low yields (12–30%) (Scheme 3) [233].
Scheme 3: Photooxygenation of oxazolidines 7a–d with formation of spiro-fused oxazolidine-containing dioxolanes 9a–d.
Scheme 3: Photooxygenation of oxazolidines 7a–d with formation of spiro-fused oxazolidine-containing dioxolan...
The reaction was performed in a temperature range from −10 to −5 °С. The conversion of oxazolidines 7 and the yields of dioxolanes 9 were determined by 1H NMR spectroscopy.
An efficient method for the synthesis of 1,2-dioxolanes is based on the oxidation of cyclopropanes by oxygen in the presence of transition-metal salts as the catalysts. The reactions of bicycloalkanols 10a–e with singlet oxygen in the presence of catalytic amounts of Fe(III) acetylacetonate produce peroxides 12a–e, which can also be synthesized starting from silylated bicycloalkanols 11a–e with the use of Cu(II) acetylacetonate (Scheme 4, Table 1) [234].
Scheme 4: Oxidation of cyclopropanes 10a–e and 11a–e with preparation of 1,2-dioxolanes 12a–e.
Scheme 4: Oxidation of cyclopropanes 10a–e and 11a–e with preparation of 1,2-dioxolanes 12a–e.
Table 1: Structures and yields of dioxolanes 12a–e.
|
Bicycloalkanol 10a–e,
silylated bicycloalkanol 11a–e |
1,2-Dioxolane 12a–e | |||||
|---|---|---|---|---|---|---|
| Method Aa | Method Bb | |||||
| R | n | Reaction time, h | Yield, % | Reaction time, h | Yield, % | |
| a | CH3 | 1 | 3 | 35 | 5 | 54 |
| b | C4H9 | 1 | 3 | 55 | 3.5 | 84 |
| c | C6H13 | 1 | 3 | 68 | – | – |
| d | CH2Ph | 1 | 3 | 50 | 5 | 78 |
| e | CH3 | 2 | 36 | 54 | 6 | 80 |
aEt2O, O2, hν, silica gel, Fe(acac)3 (4 mol %).
bEtOH, O2, hν, Cu(acac)2 (4 mol %).
Similarly, the reactions of silylated bicycloalkanols 13a–c with oxygen in the presence of the catalyst VO(acac)2 yielded dioxolanes 14a–c, which made it possible to perform the oxidation without irradiation (Scheme 5, Table 2) [235].
Scheme 5: VO(acac)2-catalyzed oxidation of silylated bicycloalkanols 13a–c.
Scheme 5: VO(acac)2-catalyzed oxidation of silylated bicycloalkanols 13a–c.
This reaction gives β-hydroxyketones as by-products that are formed as a result of the decomposition of dioxolanes 14.
Cyclopropanols 15a–g are readily oxidized by molecular oxygen in the presence of Mn(II) abietate or acetylacetonate (Scheme 6) [236].
Scheme 6: Mn(II)-catalyzed oxidation of cyclopropanols 15a–g.
Scheme 6: Mn(II)-catalyzed oxidation of cyclopropanols 15a–g.
Presumably, the reaction proceeds via the intermediate formation of О- and С-centered radicals 16a–g and 17a–g, respectively. According to this method, dioxolanes 18a–g (exist in equilibrium with the open form 19a–g) were synthesized in 60–80% yields.
Like hydroxycyclopropanes, aminocyclopropanes are transformed into 1,2-dioxolanes. For example, N-cyclopropyl-N-phenylamines 20a–c form dioxolanes 21a–c in the presence of atmospheric oxygen (Table 3). It was found that the reaction rate substantially increases in the presence of catalytic amounts of [(phen)3Fe(III)(PF6)3] or equimolar amounts of benzoyl peroxide or di-tert-butyl peroxide. The possible mechanism of the oxidation is shown in Scheme 7 [237].
Scheme 7: Oxidation of aminocyclopropanes 20a–c.
Scheme 7: Oxidation of aminocyclopropanes 20a–c.
Table 3: Peroxidation of N-cyclopropyl-N-phenylamines 20a–c to form 3-(1,2-dioxalanyl)-N-phenylamines 21a–c.
| Dioxolane 21a–c | |||
|---|---|---|---|
| R1 | R2 | Reaction conditions | |
| a | H | H |
1. (BzO)2 (1 mol/1 mol 20a), CHCl3, dark, −20 °C, 3 days.
2. (t-BuO)2 (1 mol/1 mol 20a), CHCl3, UV (254 nm), ambient temperature, aerobic, 2 h. 3. [(phen)3Fe(III)(PF6)3] (0.6 % mol), CHCl3, ambient temperature, aerobic, 1 h. |
| b | Me | H | 1. (t-BuO)2 (1 mol/1 mol 20b), CHCl3, UV (254 nm), ambient temperature, aerobic, 2 h. |
| c | H | Me |
1. (t-BuO)2 (1 mol/1 mol 20c), CHCl3, UV (254 nm), ambient temperature, aerobic, 2 h.
2. [(phen)3Fe(III)(PF6)3] (0.6 % mol), CHCl3, ambient temperature, aerobic, 1 h. |
According to the 1H NMR data, dioxolanes 21a–c are formed under the above-mentioned conditions in almost quantitative yields; the yields based on the isolated product were not higher than 80% [237].
Structurally similar 3-ethyl-6a-methyl-6-(4-phenoxyphenyl)hexahydro[1,2]dioxolo[3,4-b]pyrroles 24a and 24b were synthesized from (Z)-N-(hex-3-enyl)-N-(4-phenoxyphenyl)acetamide (22). It was suggested that aminocyclopropane 23 is formed in situ, which is subsequently oxidized in air on silica gel (Scheme 8) [238]. The total yield of both isomers 24 was 31%.
Scheme 8: Synthesis of aminodioxolanes 24.
Scheme 8: Synthesis of aminodioxolanes 24.
Trifluoromethyl-containing dioxolane 25 (Figure 3) was synthesized according to this method in 40% yield [239].
Figure 3: Trifluoromethyl-containing dioxolane 25.
Figure 3: Trifluoromethyl-containing dioxolane 25.
A series of 1,2-dioxolanes 27a–e containing various functional groups R were prepared by the oxidation of cyclopropanes 26a–e (Scheme 9, Table 4).
Scheme 9: Synthesis of 1,2-dioxolanes 27a–e by the oxidation of cyclopropanes 26a–e.
Scheme 9: Synthesis of 1,2-dioxolanes 27a–e by the oxidation of cyclopropanes 26a–e.
The reaction was performed in the presence of Ph2Se2 (10 mol %) and azobisisobutyronitrile (AIBN, 8 mol %) in air under irradiation for two days. The product was purified by flash chromatography to obtain a mixture of cis and trans isomers, whose ratio depends primarily on the nature of the substituent in cyclopropanes 26a–e [240].
The oxidation of methylenecyclopropanes 28a and 28b under photoinduced electron-transfer conditions is described by a similar scheme (Scheme 10).
Scheme 10: Photoinduced oxidation of methylenecyclopropanes 28.
Scheme 10: Photoinduced oxidation of methylenecyclopropanes 28.
The reaction was performed in acetonitrile or in a mixture of toluene and acetonitrile with the use of 9,10-dicyanoanthracene (DCA), 1,2,4,5-tetracyanobenzene (TCNB), or N-methyl-quinolinium tetrafluoroborate (NMQ+BF4−) as sensitizers. Under these conditions, dioxolane 29a was obtained in quantitative yield (1H NMR data), the yield of 29b was not reported [241].
Under irradiation in the presence of oxygen, 1,5-bis(4-methoxyphenyl)bicyclo[3.1.0]hexane (30) and 1,5-bis(4-methoxyphenyl)-6,7-diazabicyclo[3.2.1]oct-6-ene (31) were transformed into bicyclic dioxolane 33. It was suggested that both reactions proceed via the formation of 1,3-radical cation 32 (Scheme 11).
Scheme 11: Irradiation-mediated oxidation.
Scheme 11: Irradiation-mediated oxidation.
Dioxolane 33 was synthesized in the highest yields (91% from 30 and 100% from 31) in acetonitrile with the use of 9,10-dicyanoanthracene (DCA) as the sensitizer [242].
After irradiation of diazene 34 in an argon matrix at 10 K, biradical 35 was detected by IR spectroscopy and the reaction of the latter with oxygen at 10 K proceeded regioselectively to give dioxolane 36 (Scheme 12) [243].
Scheme 12: Application of diazene 34 for dioxolane synthesis.
Scheme 12: Application of diazene 34 for dioxolane synthesis.
Bicyclic peroxide 2-heptyl-3,4-dioxabicyclo[3.3.0]oct-1(8)-ene was prepared by a similar process [244].
The oxidation of arylacetylenes 37a–h with atmospheric oxygen in the presence of catalytic amounts of Mn(OAc)3 in an excess of acetylacetone afforded dioxolanes 38a–h in moderate yields (34–64%) (Scheme 13, Table 5) [245].
Scheme 13: Mn(OAc)3-catalyzed cooxidation of arylacetylenes 37a–h and acetylacetone with atmospheric oxygen.
Scheme 13: Mn(OAc)3-catalyzed cooxidation of arylacetylenes 37a–h and acetylacetone with atmospheric oxygen.
The reaction was performed at 23 °С in glacial acetic acid in air; the 37/acetylacetone/Mn(OAc)3 molar ratio was 1/10/10. The reaction gave oxiranes 39 as by-products, which can also be synthesized in quantitative yields by the treatment of dioxolanes 38 with silica gel in methanol [245].
1.2. Peroxidation of alkenes with the Co(II)/Et3SiH/O2 system (Isayama–Mukaiyama reaction)
Peroxysilylation of alkenes with molecular oxygen in the presence of triethylsilane catalyzed by cobalt(II) diketonates was described for the first time by S. Isayama and T. Mukaiyama in 1989 [246,247]. Currently, this approach is one of the main methods for the preparation of peroxides from alkenes.
Compounds (oxidized by the Isayama–Mukaiyama reaction) containing a reaction center that can be subjected to the attack by a peroxide radical, are able to undergo intramolecular cyclization to form the 1,2-dioxolane ring. For example, the Co(modp)2-catalyzed peroxysilylation (modp = 1-morpholino-5,5-dimethyl-1,2,4-hexanetrionate) of (2-vinylcyclopropyl)benzene (40) affords triethyl(1-(5-phenyl-1,2-dioxolan-3-yl)ethylperoxy)silane (41) in 37% yield (Scheme 14).
Scheme 14: Peroxidation of (2-vinylcyclopropyl)benzene (40).
Scheme 14: Peroxidation of (2-vinylcyclopropyl)benzene (40).
The reaction was carried out in 1,2-dichloroethane at room temperature, and the reaction products were separated by column chromatography. 1-Hydroxy-1-phenylpentan-3-one (42) was isolated as a by-product in 16% yield [248].
The peroxidation of 1,4-dienes 43a,b with the Co(modp)2/Et3SiH/O2 system according to a similar reaction scheme gave dioxolanes 44a,b. Acetophenone (45) was obtained as the by-product (Scheme 15, Table 6) [249].
Scheme 15: Peroxidation of 1,4-dienes 43a,b.
Scheme 15: Peroxidation of 1,4-dienes 43a,b.
The desilylation of the initially formed silicon peroxide followed by cyclization of the hydroperoxide accompanied by the attack on the electrophilic center is another example of the use of the Isayama–Mukaiyama reaction for the synthesis of cyclic peroxides. In some cases, the reaction with 1,5-dienes 46a–d produces, along with 1,2-dioxanes 51 (desilylation products of the corresponding 1,2-dioxanes 48), 1,2-dioxolanes (52b,d) as a result of cyclization of the corresponding peroxysilyl epoxides 49. In these reactions, unsaturated triethylsilyl peroxides 47 are formed as by-products, which are desilylated during hydrolysis to give the unsaturated hydroperoxides 50 (Scheme 16, Table 7) [249].
Scheme 16: Peroxidation of 1,5-dienes 46.
Scheme 16: Peroxidation of 1,5-dienes 46.
Table 7: Peroxidation of 1,5-dienes 46.
| Diene 46 | R1 | R2 | Reaction time, h | Conversion, % | Yielda, % | ||||
|---|---|---|---|---|---|---|---|---|---|
| 45 | 49 | 50 | 51 | 52 | |||||
| a | H | H | 6 | 82 | 4 | – | 13 | 31 | – |
| b | H | Me | 2.5 | 83 | 36 | – | 12 | 13 | 33 |
| c | H | Ph | 3.5 | 75 | 57 | 38 | 7 | 27 | – |
| d | Me | Me | 3 | 84 | 51 | – | – | 31 | 26 |
aThe yields are given based on the converted dienes 46a–d.
1,2-Dioxolanes can be produced from oxetanes 53a,b containing a double bond in the side chain according to a similar scheme. The first step afforded peroxysilanes 54a,b, which upon treatment with aqueous HF gave the target dioxolanes 55a,b (Scheme 17) [250].
Scheme 17: Peroxidation of oxetanes 53a,b.
Scheme 17: Peroxidation of oxetanes 53a,b.
A similar way to 1,2-dioxolanes used an oxirane cycle for the stages of ring opening followed by 1,2-dioxolane ring closing [251].
The synthesis of spirodioxolane 59 involved the peroxysilylation of 1,3-dicyclohexenylpropan-2-yl acetate (56) catalyzed by cobalt complexed with 2,2,6,6-tetramethylheptane-3,5-dione (Co(THD)2) as the first step giving 1,3-bis(1-(triethylsilylperoxy)cyclohexyl)propan-2-yl acetate (57) that was subsequently transformed into the carbonyl-containing diperoxide (1,3-bis(1-(triethylsilylperoxy)cyclohexyl)propan-2-one) (58) in two steps. The latter was treated with p-TsOH to give the target peroxide 59 (Scheme 18) [252].
1.3. The use of ozone. Peroxycarbenium ions in the 1,2-dioxolanes synthesis
The ozonolysis of unsaturated compounds is a reliable and facile method for the introduction of the peroxide functional group. As in the above-considered studies, the intramolecular cyclization of ozonolysis products can be performed with the use of the hydroperoxide group provided that there is an appropriate electrophilic center.
The reaction of oxetanes 60a,b with ozone in methanol produced 3-alkoxy-1,2-dioxolanes 62a,b. The analysis of the reaction mixture (TLC, NMR) confirmed that cyclic peroxides are formed immediately in the reaction mixture rather than in the course of the treatment or purification of the reaction products. It was suggested that the reaction proceeds via the formation of hydroperoxy acetals 61a,b (Scheme 19) [250].
Scheme 19: Synthesis of 3-alkoxy-1,2-dioxolanes 62a,b.
Scheme 19: Synthesis of 3-alkoxy-1,2-dioxolanes 62a,b.
The ozonolysis of 9-methyleneheptadecane-7,11-diyl-bis(methanesulfonate) (63) gave 9-oxoheptadecane-7,11-diyl-bis(methanesulfonate) (64). The latter reacted with H2O2 in the presence of sulfuric acid (or iodine) as the catalyst to form 9,9-dihydroperoxyheptadecane-7,11-diyl-bis(methanesulfonate) 65, and the replacement of the mesyl groups in the latter compound afforded 3,8-dihexyl-1,2,6,7-tetraoxaspiro[4.4]nonane (66, Scheme 20). The yield of dioxolane 66 was 36% based on 63 [252].
Scheme 20: Synthesis of spiro-bis(1,2-dioxolane) 66.
Scheme 20: Synthesis of spiro-bis(1,2-dioxolane) 66.
The treatment of 3,3'-(cyclohexa-3,6-diene-1,3-diyl)dipropan-1-ol (67) and 4,4'-(cyclohexa-3,6-diene-1,3-diyl)dibutan-2-ol (69) with ozone in MeOH/CH2Cl2 followed by the addition of a catalytic amount of p-TsOH lead to the intramolecular peroxyketalization that proceeds through the formation of the peroxycarbenium ion (shown in Scheme 21 for the ozonolysis of 67 as an example) to give finally dispiro-1,2-dioxolanes: 1,8,12,13-tetra-oxadispiro-[4.1.4.2]tridecane 68 (yield 67%) and two isomers of 2,9-dimethyl-1,8,12,13-tetraoxadispiro[4.1.4.2]tridecane 70 and 71 (combined yield 72%) (Scheme 21) [253].
Scheme 21: Synthesis of dispiro-1,2-dioxolanes 68, 70, 71.
Scheme 21: Synthesis of dispiro-1,2-dioxolanes 68, 70, 71.
The spirohydroperoxydioxolanes, 5-hydroperoxy-2',3'-dihydrospiro[[1,2]dioxolane-3,1'-indene] (75a) and 5-hydroperoxy-3',4'-dihydro-2'H-spiro[[1,2]dioxolane-3,1'-naphthalene] (75b), were synthesized by the ozonolysis of 1-allyl-1-hydroperoxy-2,3-dihydro-1H-indene (72a) and 1-allyl-1-hydroperoxy-1,2,3,4-tetrahydronaphthalene (72b), respectively, in an Et2O/CF3CH2OH system (2:1). The reaction proceeds via the formation of ozonide 73 followed by elimination of formaldehyde to give peroxycarbenium ion 74 that undergoes cyclization via the attack of the hydroperoxide group on the carbon center of peroxycarbenium ion 74 (Scheme 22) [254].
Scheme 22: Synthesis of spirohydroperoxydioxolanes 75a,b.
Scheme 22: Synthesis of spirohydroperoxydioxolanes 75a,b.
Spirohydroperoxydioxolane 75a (n = 1) was obtained in 71% yield (the diastereoisomeric ratio was 1:1); the yield of 75b (n = 2) was 21% (the diastereoisomeric ratio was 1:1).
5'-Hydroperoxyspiro[chromane-2,3'-[1,2]dioxolane] (77, yield 18%) and (3S,5S)-3,5-dihydroperoxy-3-(3-phenylpropyl)-1,2-dioxolane (79, yield 22%) (Scheme 23) were synthesized in a similar way starting from 2-allyl-2-hydroperoxychromane (76) and (4,4-dihydroperoxyhept-6-enyl)benzene (78), respectively [254].
Scheme 23: Synthesis of spirohydroperoxydioxolane 77 and dihydroperoxydioxolane 79.
Scheme 23: Synthesis of spirohydroperoxydioxolane 77 and dihydroperoxydioxolane 79.
An oxidative rearrangement takes place in the reaction of azepino[4,5-b]indole 80 with ozone. The addition of ozone to the endocyclic double bond (molozonide 81) and the formation of the Criegee intermediate are followed by a 1,3-dipolar interaction of the peroxycarbenium ion with the double bond (82) to form dioxolane 83. The yield was not lower than 48% but no exact yield was reported (Scheme 24) [255].
Scheme 24: Ozonolysis of azepino[4,5-b]indole 80.
Scheme 24: Ozonolysis of azepino[4,5-b]indole 80.
The peroxycarbenium ions produced by the decomposition of 1,2,4-trioxolanes can be trapped with allyltrimethylsilane. For example, the SnCl4-mediated fragmentation of ozonides 84a–l in the presence of allyltrimethylsilane in dichloromethane gives a complex mixture of products 85–94, including dioxolanes 86a–i, 87i, 90j–l, and 91j (Scheme 25, Table 8) [256].
Scheme 25: SnCl4-mediated fragmentation of ozonides 84a–l in the presence of allyltrimethylsilane.
Scheme 25: SnCl4-mediated fragmentation of ozonides 84a–l in the presence of allyltrimethylsilane.
Table 8: The SnCl4-mediated fragmentation of ozonides 84a–l in the presence of allyltrimethylsilane.
| Yield, % | ||||||||
|---|---|---|---|---|---|---|---|---|
| Ozonide 84 | R1 | R2 | T, °С | Lactone 85 | Dioxolane 86 | Dioxolane 87 | Ketone 88 | Alcohol 89 |
| a | -(CH2)4- | −78 to 0 | 11 | 50 | – | 88a (traces) | – | |
| b | -(CH2)5- | −78 to 0 | 17 | 57 | – | 88b (traces) | – | |
| c | -(CH2)6- | −78 to 0 | 39 | 24 | – | 88c (traces) | – | |
| d | CH3 | Ph | −78 to 0 | 25 | 61 | – | 88d (93%) | – |
| e | C4H9 | C4H9 | −78 to 0 | 40 | 14 | – | 88e (70%) | 75 |
| f | H | C8H17 | −78 | – | 56 | – | – | 50 |
| g | H | Ph | −78 | – | 79 | – | – | 13 |
| h | H | H | −78 | – | 10 | – | – | – |
| i | CH3 | C(CH3)3 | −78 to 0 | 31 | 21 | 9 (cis) | – | – |
| Ozonide 84 | R1 | R2 | T, °С | Dioxolane 90 (cis:trans) | Dioxolane 91 | Carbonyl compound 92 | Alcohol 93 | Alkene 94 |
| j | H | C3H7 | −78 | 15 (1:1) | 7 | 39 | 93j (20%) | – |
| k | H | H | −78 | 15 (1:1) | – | 22 | 93j (24%) | – |
| l | CH3 | H | −78 | 9 (1:1) | – | 43 | – | 2.5 |
Treatment of the bicyclic ozonide 1-methyl-6,7,8-trioxabicyclo[3.2.1]octane 84m, with SnCl4 in the presence of allyltrimethylsilane produces a mixture of two cis diastereomers and two trans diastereomers (in a ratio of 35:35:15:15) of 7-(3-methyl-5-((trimethylsilyl)methyl)-1,2-dioxolan-3-yl)hept-1-en-4-ol 95 in a total yield of 48% (Scheme 26) [256].
Scheme 26: SnCl4-mediated fragmentation of bicyclic ozonide 84m in the presence of allyltrimethylsilane.
Scheme 26: SnCl4-mediated fragmentation of bicyclic ozonide 84m in the presence of allyltrimethylsilane.
These syntheses of dioxolanes involve the formation of the peroxycarbenium ion as the key step. The reaction of the latter with allyltrimethylsilane followed by the intramolecular cyclization finally leads to the dioxolane ring.
Dioxolanes 99–102 are produced from alkoxyhydroperoxides 96a–g (ozonolysis products of alkenes) in a similar way. The first step results in the formation of peroxycarbenium ions 97, which are trapped with allyltrimethylsilane under the formation of intermediate hydroperoxides 98. Then either cyclic dioxolanes 99–102 or unsaturated compounds 103–107 are formed as the major reaction products depending on the nature of the substituents and the Lewis acid (Scheme 27, Table 9) [257].
Scheme 27: MCl4-mediated fragmentation of alkoxyhydroperoxides 96 in the presence of allyltrimethylsilane.
Scheme 27: MCl4-mediated fragmentation of alkoxyhydroperoxides 96 in the presence of allyltrimethylsilane.
Table 9: Synthesis of 1,2-dioxolanes 99–102.
| Hydroperoxide 96 | R1 | R2 | R3 | M | Dioxolane 99–102 (yield, %) | Alkene 103–107 (X, yield, %) |
|---|---|---|---|---|---|---|
| a | Me | Me | Me | Ti | 99 (31) | – |
| b | Me | Me | (CH2)2OMe | Sn | 99 (56) | – |
| b | Me | Me | (CH2)2OMe | Ti | 99 (12) | 103 (–OOH, 23) |
| с |
4-tert-butyl-
cyclohexylidene |
Me | Ti | – | 104 (–O2–, 31) | |
| c |
4-tert-butyl-
cyclohexylidene |
Me | Sn | 100 (42) | – | |
| d |
4-tert-butyl-
cyclohexylidene |
(CH2)2OMe | Sn | 100 (59) | – | |
| e | Me | BnOCH2 | Me | Ti | 101 (12) | 105 (=O, 62) |
| f | Bu | H | Me | Ti | 102 (7) | 106 (OMe, 63) |
| g | Bu | H | (CH2)2OMe | Ti | 102 (15) |
107 (O(CH2)2OMe,
the yield was not determined) |
The reaction of trialkylsilylperoxyacetals with alkenes in the presence of Lewis acids also proceeds through the formation of peroxycarbenium ions. For example, the reaction of methyl 2-(4-methoxy-4-(triethylsilylperoxy)cyclohexyl)acetate (108) with 2-methyleneadamantane (109) produced adamantane-2-spiro-3’,8’-methoxycarbonylmethyl-1’,2’-dioxaspiro[4.5]decane (110) in 40% yield (Scheme 28) [258].
Scheme 28: SnCl4-catalyzed reaction of monotriethylsilylperoxyacetal 108 with alkene 109.
Scheme 28: SnCl4-catalyzed reaction of monotriethylsilylperoxyacetal 108 with alkene 109.
The use of easily accessible triethylsilylperoxyacetals 111 as the starting materials for the generation of silylperoxycarbenium ions 112 enabled the synthesis of 1,2-dioxolanes containing various functional groups 113–130 in good yields by the reactions with alkenes (Scheme 29, Table 10) [88,90,259].
Scheme 29: SnCl4-catalyzed reaction of triethylsilylperoxyacetals 111 with alkenes.
Scheme 29: SnCl4-catalyzed reaction of triethylsilylperoxyacetals 111 with alkenes.
Table 10: Structures and yields of 1,2-dioxolanes 113–130.
| Product | Structure | Yielda, % | Product | Structure | Yielda, % |
|---|---|---|---|---|---|
| 113 |
|
80 | 122 |
|
92 |
| 114 |
|
72 | 123 |
|
47 |
| 115 |
|
57 | 124 |
|
72 |
| 116 |
|
67 | 125 |
|
28 |
| 117 |
|
57 | 126 |
|
48 |
| 118 |
|
34 | 127 |
|
59 |
| 119 |
|
46 | 128 |
|
51 |
| 120 |
|
74 | 129 |
|
68 |
| 121 |
|
94 | 130 |
|
90 |
aReagents and conditions: SnCl4 (1.0–2.0 equiv), alkene (1.0–3.0 equiv), CH2Cl2, from −78 °С to 25 °С, 2–24 h.
1.4. Methods for the synthesis of 1,2-dioxolanes from hydrogen peroxide and hydroperoxides
This section deals with reactions, in which hydrogen peroxide or hydroperoxides are used for the construction of the five-membered peroxide ring. In all syntheses, the final (key) step involves the intramolecular cyclization of hydroperoxide with the attack on the electrophilic center (an activated double bond or a carbon atom of a keto or ester group).
The desilylation of tert-butyldimethylsilylperoxy ketones 131a,b with HF followed by cyclization and subsequent reaction with monomethylethylene glycol afforded dioxolanes 132a,b in 75 and 88% yield, respectively. The intermediate hydroxydioxolanes 131'a,b were used in the second step without isolation (Scheme 30) [260]. A series of analogues of plakinic acids were synthesized by the modification of the peroxyketal moiety of dioxolanes 132a and 132b [260].
Scheme 30: Desilylation of tert-butyldimethylsilylperoxy ketones 131a,b followed by cyclization.
Scheme 30: Desilylation of tert-butyldimethylsilylperoxy ketones 131a,b followed by cyclization.
The monoperoxy ketal moiety of 4-(2-methoxypropan-2-ylperoxy)nonan-2-one (133) was used for the generation of the hydroperoxide group. The intramolecular cyclization afforded 3-methyl-5-pentyl-1,2-dioxolan-3-ol (134), which could be easily reacted with monomethylethylene glycol to form 3-(2-methoxyethoxy)-3-methyl-5-pentyl-1,2-dioxolane (135). Allylation of the latter produced 3-allyl-3-methyl-5-pentyl-1,2-dioxolane (136) in 47% yield (Scheme 31) [261].
Scheme 31: Deprotection of peroxide 133 followed by cyclization.
Scheme 31: Deprotection of peroxide 133 followed by cyclization.
The asymmetric peroxidation of methyl vinyl ketones 137a–e with 9-amino-9-deoxyepiquinine 138 and CCl3COOH afforded hydroxydioxolanes 139a–e with high enantiomeric excess (ee 94–95%) (Scheme 32) [262].
Scheme 32: Asymmetric peroxidation of methyl vinyl ketones 137a–e.
Scheme 32: Asymmetric peroxidation of methyl vinyl ketones 137a–e.
The Kobayashi synthesis of 1,2-dioxolanes represents an intramolecular version of the Michael reaction, in which the hydroperoxide group acts as the nucleophile. Generally, the reaction is performed in fluorinated alcohols (CF3CH2OH or (CF3)2CHOH) in the presence of diethylamine or, in some cases, of cesium hydroxide. Initially, the method was proposed for the synthesis of the 1,2-dioxane moiety (examples are considered in the corresponding section) [263]. However, it was shown that this method is also applicable to the preparation of structurally complex 1,2-dioxolanes, such as methyl 2-(5-(5-methylfuran-2-yl)-1,2-dioxolan-3-yl)acetate (141) from the furan derivative (E)-methyl 5-hydroperoxy-5-(5-methylfuran-2-yl)pent-2-enoate (140) (Scheme 33) [264].
Scheme 33: Et2NH-catalyzed intramolecular cyclization.
Scheme 33: Et2NH-catalyzed intramolecular cyclization.
A simple method was developed for the synthesis of cyclopropane-containing oxodioxolanes 143a–j and is based on the hydroperoxidation of tertiary alcohols 142a–j in an acidic medium followed by cyclization of the intermediate hydroperoxides through the ester group (Scheme 34) [265].
Scheme 34: Synthesis of oxodioxolanes 143a–j.
Scheme 34: Synthesis of oxodioxolanes 143a–j.
This method allows for the use of a nonhazardous 30% hydrogen peroxide solution. However, the authors mentioned that structurally similar tertiary alcohols, without a cyclopropane substituent, are inert under the reported conditions.
Haloperoxidation reaction that is accompanied by intramolecular ring closure represents another version of the cyclization reaction. For example, the reaction of bromine with unsaturated hydroperoxide 146 (produced by reaction of 1,4,5,8-tetrahydronaphthalene (144) with singlet oxygen via the formation of 4a-hydroperoxy-1,4,4a,5-tetrahydronapthalene (145) gives hydroperoxide-containing bromonium cation 147 as the intermediate, which undergoes cyclization to form 1,2-dioxolane-containing 7-bromo-4,5,10,11-tetraoxatetracyclo[7.2.2.13,6.03,9]tetradec-12-ene (148) (Scheme 35).
Scheme 35: Haloperoxidation accompanied by intramolecular ring closure.
Scheme 35: Haloperoxidation accompanied by intramolecular ring closure.
The cyclization occurs selectively because the hydroperoxide group in intermediate 147 attacks only one of two possible electrophilic carbon centers [266].
1.5. 1,2-Dioxolane ring formation through oxidation of the allylic position
1,2-Dioxolane-containing compounds 150a–d were synthesized by the oxidation of triterpenes 149a–d with Na2Cr2O7/N-hydroxysuccinimide (Scheme 36). The resulting compounds exhibit antitumor activity comparable with that of betulinic acid [175-177].
Scheme 36: Oxidation of triterpenes 149a–d with Na2Cr2O7/N-hydroxysuccinimide.
Scheme 36: Oxidation of triterpenes 149a–d with Na2Cr2O7/N-hydroxysuccinimide.
1.6. Structural modifications, in which the 1,2-dioxolane ring remains intact
The possibility of performing the Curtius and Wolff rearrangements to form 1,2-dioxolane ring-retaining products was exemplified by the synthesis of ethyl (3,5,5-trimethyl-1,2-dioxolan-3-yl)methylcarbamate (152) and methyl 3-(3,5,5-trimethyl-1,2-dioxolan-3-yl)propanoate (154) (through formation of stable diazodioxolane 153) from 2-(3,5,5-trimethyl-1,2-dioxolan-3-yl)acetic acid (151) (Scheme 37) [267].
Scheme 37: Curtius and Wolff rearrangements to form 1,2-dioxolane ring-retaining products.
Scheme 37: Curtius and Wolff rearrangements to form 1,2-dioxolane ring-retaining products.
Dioxolane 155 that contains a free hydroxy group was synthesized by the oxidative desilylation of silicon-containing peroxide 124 with n-Bu4NF and H2O2 (Scheme 38) [259].
Scheme 38: Oxidative desilylation of peroxide 124.
Scheme 38: Oxidative desilylation of peroxide 124.
Dioxolane 158 with the aminoquinoline antimalarial pharmacophore was synthesized in two steps by the oxidation of alcohol 156 with H5IO6/RuCl3 followed by amidation of the acid 157 (Scheme 39) [88]. It was shown that compound 158 exhibits antimalarial activity comparable with that of artemisinin [88].
Scheme 39: Synthesis of dioxolane 158, a compound containing the aminoquinoline antimalarial pharmacophore.
Scheme 39: Synthesis of dioxolane 158, a compound containing the aminoquinoline antimalarial pharmacophore.
Plakinic acids belong to a large family of natural products, which were shown to be highly cytotoxic toward cancer cells and fungi. Diastereomers of plakinic acid A, 162a and 162b were synthesized starting from dioxolane ((R)-3-((2R,3E,6S,7E)-2,6-dimethyl-8-phenylocta-3,7-dienyl)-5-(2-methoxyethoxy)-3,5-dimethyl-1,2-dioxolane) (159) [260]. In the first step, dioxolane 159 was treated with (1-(ethylthio)vinyloxy)-trimethylsilane in the presence of TiCl4 to obtain S-ethyl 2-((R)-5-((2R,3E,6S,7E)-2,6-dimethyl-8-phenylocta-3,7-dienyl)-3,5-dimethyl-1,2-dioxolan-3-yl)-ethanethioate (160). The subsequent reaction with sodium methoxide in methanol produced the corresponding esters 161a and 161b, which were hydrolyzed to prepare the target plakinic acids (Scheme 40).
Scheme 40: Diastereomers of plakinic acid A, 162a and 162b.
Scheme 40: Diastereomers of plakinic acid A, 162a and 162b.
2. Synthesis of 1,2,4-trioxolanes (ozonides)
The currently most widely used methods for the synthesis of 1,2,4-trioxolanes are based on reactions of ozone with unsaturated compounds, such as the ozonolysis of alkenes, the cross-ozonolysis of alkenes with carbonyl compounds, and the cross-ozonolysis of О-alkylated oximes in the presence of carbonyl compounds (Griesbaum coozonolysis).
2.1. Ozonolysis of alkenes
According to the mechanism proposed by R. Criegee [268,269] the ozonolysis of alkenes 163 involves several steps: the 1,3-dipolar cycloaddition of ozone to the double bond to form unstable 1,2,3-trioxolane 164 (so-called molozonide) that is followed by its decomposition to a peroxycarbenium ion and a carbonyl compound (Criegee intermediates). The 1,3-dipolar cycloaddition of the intermediates with each other form the 1,2,4-trioxolane 165 (Scheme 41, Table 11). Generally, the ozonolysis is performed in aprotic solvents at low temperatures and in some cases, on polymeric substrates. Since various compounds containing a С=С group are easily available, a wide range of functionalized 1,2,4-trioxolanes can be synthesized in moderate to high yields.
Table 11: Examples of 1,2,4-trioxolanes produced by the ozonolysis of alkenes.
| Alkene 163 | Ozonolysis conditions | 1,2,4-Trioxolane 165 | Yield, % | Reference |
|---|---|---|---|---|
|
|
Et2O, −70 °C |
|
24 | [270] |
|
|
Et2O, −70 °C |
|
27 | [270] |
|
|
hexane, −78 °C |
|
78 | [256] |
|
|
hexane, −78 °C |
|
73 | [256] |
|
|
hexane, −78 °C |
|
77 | [256] |
|
|
hexane, −78 °C |
|
61 | [256] |
|
|
isooctane/CCl4, −78 °C, 1 h |
|
>82 | [271] |
|
|
CH2Cl2, −78 °C |
|
95 | [272] |
|
|
CH2Cl2, −78 °C |
|
90 | [272] |
|
|
CH2Cl2, −78 °C |
|
92 | [272] |
|
|
CH2Cl2, −78 °C |
|
93 | [272] |
|
|
CH2Cl2, −78 °C |
|
93 | [272] |
|
|
CH2Cl2, −78 °C |
|
94 | [272] |
|
|
pentane,
−78 °C |
|
63 | [272] |
|
|
freon-113, 15–20 °C, 2 h |
|
The yield was not determined | [273,274] |
|
|
freon-113, 15–20 °C, 2 h |
|
The yield was not determined | [273] |
|
|
CH2Cl2, −78 °C |
|
96 | [275] |
|
|
polymer-based, −78 °C, 8 h |
|
23 | [276] |
|
|
polymer-based, −78 °C, 3 h |
|
38 | [276] |
|
|
CH2Cl2, −70 °C |
|
48 | [277] |
|
|
without solvent, −133 to −43 °C |
|
100 | [278] |
|
|
1) CH2Cl2,
−78 °C, 15 min. 2) Me2S, rt, 6 h |
|
71 | [279] |
|
|
hexane,
−78 °C, 30 min |
|
6 | [280] |
|
|
CH2Cl2, −78 °C, 20 min |
|
The yield was not determined | [281] |
|
|
CH2Cl2, −78 °C, 2 h |
|
>97 | [282] |
|
|
CDCl3, −65 °C |
|
88 | [283] |
|
|
CFCl3, −70 °C |
|
100 | [283] |
|
|
CH2Cl2, −78 °C, 1 h |
|
85 | [284] |
|
|
CH2Cl2, −78 °C, 1 h |
|
70 | [285] |
|
|
pentane,
−78 °C |
|
10-30 | [148-152] |
|
|
Et2O/CH3OH, −78 °C |
|
12 | [254] |
|
|
CH2Cl2, −78 °C |
|
92 | [286] |
|
|
CH2Cl2, 0 °C |
|
95 | [287,288] |
|
|
H2O/CH2Cl2,
0 °C |
|
72 | [289] |
|
|
1) SiO2,
−78 °C; 2) Me2S, MeOH |
|
30 | [290] |
2.2. Cross-ozonolysis of alkenes with carbonyl compounds
The peroxycarbenium ion produced by the decomposition of 1,2,3-trioxolane (molozonide) can react with externally introduced carbonyl compounds to form the corresponding 1,2,4-trioxolanes. The pathway of decomposition of 1,2,3-trioxolanes is determined by the structure of the starting alkene 166. In some cases, a high selectivity of the formation of cross-ozonolysis products 1,2,4-trioxolanes (ozonides) 167, can be achieved (Scheme 42, Table 12).
Scheme 42: Cross-ozonolysis of alkenes 166 with carbonyl compounds.
Scheme 42: Cross-ozonolysis of alkenes 166 with carbonyl compounds.
Table 12: Examples of 1,2,4-trioxolanes synthesized by the cross-ozonolysis of alkenes in the presence of carbonyl compounds.
| Alkene 166 | Carbonyl compound | Ozonolysis conditions | 1,2,4-Trioxolane 167 | Yield, % | Reference |
|---|---|---|---|---|---|
|
|
|
CH2Cl2,
−78 °C |
|
17–74 | [291] |
|
|
|
CH2Cl2,
−78 °C |
|
9–57 | [291] |
|
|
|
CH2Cl2,
−78 °C |
|
50
42 |
[291] |
|
|
|
CH2Cl2,
−78 °C |
|
38
32 |
[291] |
|
|
|
CH2Cl2,
−78 °C |
|
18–48 | [291] |
|
|
|
CH2Cl2,
−78 °C |
|
25–37 | [291] |
|
|
|
CH2Cl2,
−78 °C |
|
63–80 | [291] |
|
|
|
CH2Cl2,
−78 °C |
|
55–77 | [291] |
|
|
|
CH2Cl2,
−78 °C |
|
49–74 | [291] |
|
|
|
CH2Cl2,
−78 °C |
|
63–66 | [291] |
|
|
|
CH2Cl2,
−78 °C |
|
52
62 |
[291] |
|
|
|
CH2Cl2,
−78 °C |
|
41
46 |
[291] |
|
|
|
CH2Cl2,
−78 °C |
|
82 | [291] |
|
|
|
CH2Cl2,
−78 °C |
|
26 | [291] |
|
|
|
CH2Cl2,
−78 °C |
|
58
53 |
[292] |
|
|
21
21 |
[292] | |||
|
|
|
CH2Cl2,
−78 °C |
|
23
25 |
[292] |
|
|
68
60 |
[292] | |||
|
|
|
CH2Cl2,
−78 °C |
|
30
25 |
[292] |
|
|
15
14 |
[292] | |||
|
|
25
27 |
[292] | |||
|
|
|
Et2O,
−70 °C |
|
60
56 |
[293] |
|
|
|
Et2O,
−70 °C |
|
59
65 |
[293] |
|
|
|
Et2O,
−70 °C |
|
59 | [293] |
For the ozonolysis of the bicyclic cyclohexenone, 2,3,4,5,6,7,8,9,10,11,12,13,14,15-tetradecahydro-1H-benzo[13]annulen-1-one (168), two reaction pathways can be proposed through intermediate 169 to form ozonides 170 and 171. It appeared that the reaction gave only 16,17,18-trioxatricyclo[10.3.2.11,12]octadecan-2-one 171 as two isomers, with the anti isomer in 60% and the syn isomer in 10% yield (Scheme 43) [294]. The structures of these compounds were established by X-ray diffraction [294].
Scheme 43: Ozonolysis of the bicyclic cyclohexenone 168.
Scheme 43: Ozonolysis of the bicyclic cyclohexenone 168.
The cross-ozonolysis of enol ethers 172a,b with cyclohexanone enabled the synthesis of 1,2,4-trioxolanes 173a,b containing the easily oxidizable C–H fragment in the third position (Scheme 44) [256].
Scheme 44: Cross-ozonolysis of enol ethers 172a,b with cyclohexanone.
Scheme 44: Cross-ozonolysis of enol ethers 172a,b with cyclohexanone.
2.3. Cross-ozonolysis of O-alkyl oximes in the presence of carbonyl compounds (Griesbaum co-ozonolysis)
In 1995, K. Griesbaum and co-workers reported a new type of cross-ozonolysis [295]. This method enables the synthesis of tetrasubstituted ozonides 176 by an ozone-mediated reaction of О-alkyl oximes 174 with ketones 175 (Scheme 45, Table 13). The selective synthesis of ozonides has attracted great interest because it allows the preparation of compounds exhibiting high antiparasitic activity.
Table 13: Examples of ozonides (1,2,4-trioxolanes) synthesized by the Griesbaum method.
| Oxime 174 | Ketone 175 | Ozonolysis conditions | 1,2,4-Trioxolane 176 | Yield, % | Ref. |
|---|---|---|---|---|---|
|
|
|
hexane, −78 °C |
|
47–67 | [256] |
|
|
|
pentane, CH2Cl2, 0 °C |
|
54 | [91] |
|
|
|
pentane, CH2Cl2, 0 °C |
|
10–75 |
[91,94]
[95,296] |
|
|
|
pentane, CH2Cl2, 0 °C |
|
23–50 | [91-93] |
|
|
|
pentane, 0 °C |
|
48 | [92,93] |
|
|
|
pentane, CH2Cl2, 0 °C |
|
32–58 | [91-93] |
|
|
|
pentane, CH2Cl2, 0 °C |
|
20–70 |
[91-93,96]
[97,297] |
|
|
|
pentane, 0 °C |
|
38 | [91] |
|
|
|
pentane, 0 °C |
|
41 | [91] |
|
|
|
pentane, CH2Cl2, 0 °C |
|
33 | [91] |
|
|
|
hexane, CH2Cl2,
0 °C |
|
17 | [91] |
|
|
|
pentane, CH2Cl2, 0 °C |
|
27 | [91] |
|
|
|
pentane, CH2Cl2, 0 °C |
|
53 | [92,93] |
|
|
|
pentane, CH2Cl2, 0 °C |
|
n.d.a | [96,97] |
|
|
|
cyclohexane CH2Cl2, 0 °C |
|
30 | [298] |
|
|
|
cyclohexane CH2Cl2, 0 °C |
|
54 | [298] |
|
|
|
cyclohexane, CH2Cl2, 0 °C |
|
78 | [258] |
aYield was not determined
The Griesbaum method is widely applicable and allows the selective synthesis of symmetrical and unsymmetrical 1,2,4-trioxolanes, which are not accessible by direct ozonolysis of alkenes or the cross-ozonolysis of alkenes or enol ethers in the presence of carbonyl compounds. In addition, this method does not need tetrasubstituted alkenes or enol ethers as starting materials, which are difficult to prepare. Taking into account a wide range of commercially available ketones, it can be concluded that this is the most universal method for the synthesis of 1,2,4-trioxolanes in terms of selectivity and structural diversity of the final products.
2.4. Other methods for the synthesis of 1,2,4-trioxolanes
The reactions of aryloxiranes 177a,b with oxygen in the presence of 9,10-dicyanoanthracene (DCA) and biphenyl (BiP) under irradiation produced 1,2,4-trioxolanes 178a and 178b (Scheme 46). It should be noted that the oxirane moiety is oxidized rather than the double bond in these reactions [299].
Scheme 46: Reactions of aryloxiranes 177a,b with oxygen.
Scheme 46: Reactions of aryloxiranes 177a,b with oxygen.
This unusual result was obtained upon treatment of the hydroxydioxepane, 3-methoxy-3-methyloctahydro-3H-benzo[c][1,2]dioxepin-9a-ol (179) with TMSOTf/Et3SiH. Thus, the peroxide moiety was not reduced with Et3SiH, and the reaction produced the bicyclic peroxide, 1-methyl-10,11,12-trioxatricyclo[7.2.1.04,9]dodecane (180) containing the 1,2,4-trioxolane moiety, as the major product (Scheme 47) [270].
Scheme 47: Intramolecular formation of 1,2,4-trioxolane 180.
Scheme 47: Intramolecular formation of 1,2,4-trioxolane 180.
The same bicyclic peroxide 180 was synthesized in good yield by the reaction of 2-(2-(2-methyl-1,3-dioxolan-2-yl)ethyl)cyclohexanone (181) with hydrogen peroxide in the presence of phosphomolybdic acid (PMA) (Scheme 48) [300].
Scheme 48: Formation of 1,2,4-trioxolane 180 by the reaction of 1,5-ketoacetal 181 with H2O2.
Scheme 48: Formation of 1,2,4-trioxolane 180 by the reaction of 1,5-ketoacetal 181 with H2O2.
2.5. Structural modifications, in which 1,2,4-trioxolane ring remains intact
Scheme 49 shows possible modifications of substituents at the ozonide ring by the reduction of the ester group in cis-adamantane-2-spiro-3’-8’-ethoxycarbonyl-1’,2’,4’-trioxa-spiro[4.5]decane 182 to form the alcohol cis-adamantane-2-spiro-3’-8’-hydroxymethyl-1’,2’,4’-trioxaspiro[4.5]decane 183. The latter was mesylated to 184 (cis-adamantane-2-spiro-3’-8’-methanesulfonylmethyl-1’,2’,4’-trioxa-spiro[4.5]decane), and used in the reaction with sodium 1-methyl-1H-tetrazole-5-thiolate 185 for the synthesis of cis-adamantane-2-spiro-3’-8’-[[(1’-methyl-1’H-tetrazol-5’-yl)thio]methyl]-1’,2’,4’-trioxaspiro[4.5]decane 186 through nucleophilic substitution of the mesyl group by the thio group of tetrazole 185 (Scheme 49) [297].
Scheme 49: 1,2,4-Trioxolane 186 with tetrazole fragment.
Scheme 49: 1,2,4-Trioxolane 186 with tetrazole fragment.
Ozonide 188 was synthesized by Mitsunobu reaction of alcohol 183 with pyridin-4-ol (187) (Scheme 50) [93]. It should be emphasized that this method can be applied in spite of the use of triphenylphosphine, which is a strong reducing agent for peroxides.
Scheme 50: 1,2,4-Trioxolane 188 with a pyridine fragment.
Scheme 50: 1,2,4-Trioxolane 188 with a pyridine fragment.
The alkylation of the sodium salt of alcohol 183 with 2-chloropyrimidine in dimethylformamide gave ozonide 189 (Scheme 51). In this reaction, neither sodium hydride nor sodium salt 183 cleave the ozonide ring to a substantial degree. The resulting 1,2,4-trioxolanes 188 and 189 exhibit high in vitro antimalarial activity comparable with that of artemisinin and in vivo even higher activity than that of artemisinin [93].
Scheme 51: 1,2,4-Trioxolane 189 with pyrimidine fragment.
Scheme 51: 1,2,4-Trioxolane 189 with pyrimidine fragment.
Aminoquinoline-containing 1,2,4-trioxalane 191 was synthesized by reductive amination of adamantane-2-spiro-3’-8’-oxo-1’,2’,4’-trioxaspiro[4.5]-decane 190 (Scheme 52). Ozonide 191 is an example of a combination of two known antiparasitic pharmacophores, viz. a peroxide and an aminoquinoline moiety [296].
Scheme 52: Synthesis of aminoquinoline-containing 1,2,4-trioxalane 191.
Scheme 52: Synthesis of aminoquinoline-containing 1,2,4-trioxalane 191.
Arterolane is a fully synthetic 1,2,4-trioxalane, also known as OZ277. It has high antimalarial activity and is currently in the final stage of clinical trials. As drug, this compound is used in combination with piperaquine. The synthesis of arterolane is based on the Griesbaum coozonolysis of a mixture of adamantan-2-one O-methyloxime (192) and 4-carbomethoxycyclohexanone 193 to form cis-adamantane-2-spiro-3’-8’-methoxycarbonylmethyl-1’,2’,4’-trioxaspiro[4.5]decane 194. The latter is hydrolyzed to cis-adamantane-2-spiro-3’-8’-carboxymethyl-1’,2’,4’-trioxaspiro[4.5]decane 195, followed by mild amidation with the formation of the intermediate ozonide 196 that on treatment with 2-methylpropane-1,2-diamine finally gives the target compound (Scheme 53). The in vitro and in vivo studies showed that arterolane is more active against causative agents of malaria than artemisinin, chloroquine, and mefloquine [77,78,81].
3. Synthesis of 1,2-dioxanes
Modern approaches to the synthesis of 1,2-dioxanes are based on reactions with singlet oxygen, the oxidative coupling of carbonyl compounds and alkenes in the presence of manganese and cerium salts, the co-oxidation of alkenes and thiols with oxygen, the Isayama–Mukaiyama peroxidation, the Kobayashi cyclization of hydroperoxides, the reaction of 1,4-diketones with hydrogen peroxide, the intramolecular nucleophilic substitution by the hydroperoxide group, the cyclization with participation of halogenonium ion donors, acid-mediated rearrangements of peroxides, the palladium-catalyzed cyclization of compounds with С=С and –О–О– groups, and reactions with the participation of peroxycarbenium ions.
3.1. Methods for the synthesis of 1,2-dioxanes using singlet oxygen
The oxidation of diarylheptadienes 197a–c with singlet oxygen in acetonitrile afforded bicyclic peroxides 198a–c in 33–58% yields. 2,4,6-Triphenylpyrylium tetrafluoroborate was used as the sensitizer for singlet oxygen generation (Scheme 54) [301].
Scheme 54: Oxidation of diarylheptadienes 197a–c with singlet oxygen.
Scheme 54: Oxidation of diarylheptadienes 197a–c with singlet oxygen.
It was found that tris(bipyrazyl)ruthenium(II) [(Ru(bpz)3(PF6)2] is an excellent photocatalyst for the synthesis of 1,2-dioxanes by aerobic photooxygenation of α,ω-dienes [302].
The addition of singlet oxygen to substrate 199 occurs in the last step of the synthesis of natural hexacyclinol peroxide 200 (Scheme 55) [303].
Scheme 55: Synthesis of hexacyclinol peroxide 200.
Scheme 55: Synthesis of hexacyclinol peroxide 200.
The reactions of 6-methylhept-5-en-2-one (201) and 5-methylhex-4-enenitrile (203) with singlet oxygen produced 1,2-dioxanes, 3-methyl-6-(prop-1-en-2-yl)-1,2-dioxan-3-ol (202) and 6-(prop-1-en-2-yl)-1,2-dioxane-3-imine (204), containing the hydroxy and imine groups, respectively (Scheme 56) [304].
Scheme 56: Oxidation of enone 201 and enenitrile 203 with singlet oxygen.
Scheme 56: Oxidation of enone 201 and enenitrile 203 with singlet oxygen.
3.2. Oxidative coupling of carbonyl compounds and alkenes in the presence of manganese or cerium salts
The synthesis of 1,2-dioxanes 207 is based on the addition of alkene 205 and oxygen to carbonyl compound 206 via the intermediate formation of carbon-centered peroxide radicals. The reaction occurs in the presence of catalytic amounts of manganese or cerium salts, which are involved in a redox cycle. It is assumed that the oxidation of β-dicarbonyl compounds proceeds through a formation of an enol-containing complex with a metal ion (Scheme 57, Table 14).
Scheme 57: Synthesis of 1,2-dioxanes 207 by oxidative coupling of carbonyl compounds 206 and alkenes 205.
Scheme 57: Synthesis of 1,2-dioxanes 207 by oxidative coupling of carbonyl compounds 206 and alkenes 205.
Table 14: Examples of 1,2-dioxanes 207 synthesized by oxidative coupling of carbonyl compounds 206 and alkenes 205.
| Alkene 205 | Carbonyl compound 206 | Reaction conditions | 1,2-Dioxane 207 | Yield, % | Ref. |
|---|---|---|---|---|---|
|
|
|
Mn(OAc)2, O2, AcOH, 80 °C,
10 h |
|
67 | [305] |
|
|
|
Mn(OAc)3, air, AcOH, 23 °C, 0.5–24 h |
|
20–84 | [306] |
|
|
|
CeCl3×7H2O, air, iPrOH, rt, 16 h |
|
42–87 | [307] |
|
|
|
CeCl3×7H2O, air, iPrOH, rt, 16 h |
|
5–73 | [307] |
|
|
|
CeCl3×7H2O, air, iPrOH, rt,
14–16 h |
|
19 (n = 1),
33 (n = 2) |
[308] |
|
|
|
CeCl3×7H2O, air, iPrOH, rt,
14–16 h |
|
18 | [308] |
|
|
|
Mn(OAc)3, air, AcOH, 23 °C, 15–18 h |
|
22–89 | [309] |
|
|
|
Mn(OAc)3, air, AcOH, 23 °C, 9–18 h |
|
17–99 | [310] |
|
|
|
Mn(OAc)3, air, AcOH, reflux, 10 min |
|
9–11 | [311] |
|
|
|
Mn(OAc)3, air, AcOH, rt, 3.5–7.5 h |
|
8–75 | [312] |
3.3. Oxidation of 1,5-dienes in the presence of thiols
The co-oxidation of 1,4-dienes and thiols (thiol–olefin co-oxygenation, TOCO reaction) was described for the first time by Beckwith and Wagner as a method for the synthesis of sulfur-containing 1,2-dioxolanes [313,314]. More recently, it has been shown that under similar conditions, the oxidation of 1,5-dienes 208 affords the corresponding sulfur-containing 1,2-dioxanes 209. The reaction proceeds under oxygen atmosphere in the presence of azobisisobutyronitrile (AIBN) or ditert-butyl peroxalate (DBPO) as radical initiators. The resulting unstable hydroperoxides are reduced with triphenylphosphine to hydroxy derivatives 209 (Scheme 58, Table 15).
Scheme 58: 1,2-Dioxanes 209 synthesis by co-oxidation of 1,5-dienes 208 and thiols.
Scheme 58: 1,2-Dioxanes 209 synthesis by co-oxidation of 1,5-dienes 208 and thiols.
Table 15: Examples of 1,2-dioxanes synthesized by co-oxidation of 1,5-dienes and thiols.
| Diene 208 | Thiol | Reaction conditions | 1,2-Dioxane 209 | Yield, % | Reference |
|---|---|---|---|---|---|
|
|
R3SH
R3 = Ph, 4-FC6H4, n-Bu, t-Bu, MeO2CCH2, cyclohexyl, Ph3C |
1) O2, DBPO, benzene/heptane, rt, 10 h or O2, AIBN, hν, MeCN, 4 °C,
10 h 2) Ph3P, CH2Cl2, 0–5 °C, 2 h, rt, 1 h |
|
6–54 | [107,179,315] |
|
|
PhSH
4-ClC6H4SH |
1) O2, AIBN, hν, MeCN, 0 °C, 2 h
2) Ph3P, CH2Cl2, 0 °C to rt |
|
70 (Ar = Ph),
21 (Ar = 4-ClC6H4) |
[102,316] |
The oxidation of acetophenones 210 produces bicyclic 1,2-dioxanes 212 (Scheme 59). It is hypothesized that the reaction gives hydroperoxide 211 as the intermediate, that undergoes rapid cyclization to form the target 1,2-dioxane 212 [317].
Scheme 59: Synthesis of bicyclic 1,2-dioxanes 212 with aryl substituents.
Scheme 59: Synthesis of bicyclic 1,2-dioxanes 212 with aryl substituents.
3.4. Synthesis of 1,2-dioxanes by the Isayama–Mukaiyama method
The Isayama–Mukaiyama peroxysilylation of 1,5-dienes 213 followed by desilylation under acidic conditions gives hydroperoxide-containing 1,2-dioxanes 214 (Scheme 60, Table 16).
Scheme 60: Isayama–Mukaiyama peroxysilylation of 1,5-dienes 213 followed by desilylation under acidic conditions.
Scheme 60: Isayama–Mukaiyama peroxysilylation of 1,5-dienes 213 followed by desilylation under acidic conditio...
Table 16: Synthesis of 1,2-dioxanes by the Isayama–Mukaiyama method.
amodp = 1-morpholino-5,5-dimethyl-1,2,4-hexanetrionate.
The oxidation of (Z)-ethyl 2-(3-(prop-1-en-2-yl)cyclohexylidene)acetate (215) gives ethyl 2-(4,4-dimethyl-2,3-dioxabicyclo[3.3.1]nonan-1-yl)-2-hydroxyacetate (218) in 29% yield. The oxidative reaction proceeds presumably with formation of an O-centered radical 216, then a C-centered radical 217 and the latter adds oxygen and is reduced to the hydroxy derivative of 1,2-dioxane 218 (Scheme 61) [318].
Scheme 61: Synthesis of bicycle 218 with an 1,2-dioxane ring.
Scheme 61: Synthesis of bicycle 218 with an 1,2-dioxane ring.
An alternative synthesis of a 1,2-dioxane by the Isayama–Mukaiyama method includes the following sequence of reactions: peroxysilylation, desilylation, and recyclization accompanied by a ring opening of oxirane or oxetane (Scheme 62 and Scheme 63).
Scheme 62: Intramolecular cyclization with an oxirane-ring opening.
Scheme 62: Intramolecular cyclization with an oxirane-ring opening.
Scheme 63: Inramolecular cyclization with the oxetane-ring opening.
Scheme 63: Inramolecular cyclization with the oxetane-ring opening.
Cobalt(II) acetylacetonate (acac) or bis-2,2,6,6-tetramethylheptane-3,5-dienoate (thd) were used as the catalyst for the peroxidation of 219. The cyclization of the intermediate peroxide 220 was performed with Amberlyst-15 ion-exchange resin. This approach was used in the multistep synthesis of the natural endoperoxide 9,10-dihydroplakortin, which exhibits antimalarial and anticancer activities as do its structural analogues [320,321].
2-(3,6,6-Trimethyl-1,2-dioxan-3-yl)ethanol (224) was synthesized in a similar way starting with the peroxidation of 2-methyl-2-(3-methylbut-3-enyl)oxetane (222), followed by oxetane-ring opening in triethyl(2-methyl-4-(2-methyloxetan-2-yl)butan-2-ylperoxy)silane (223) (Scheme 63) [250].
Dioxanes can also be synthesized by inramolecular cyclizations with the attack on a keto group. The peroxysilylation of the unsaturated ketone 1,5-dicyclohexenylpentan-3-one (225), with the Co(thd)2/Et3SiH/O2 system produced 1,5-bis(1-(triethylsilylperoxy)cyclohexyl)pentan-3-one (226), which underwent cyclization in the presence of p-toluenesulfonic acid to give the spiro-fused 7,8,10,11-tetraoxatrispiro[5.2.2.5.2.2]henicosane 227 (Scheme 64) [252].
Scheme 64: Intramolecular cyclization with the attack on a keto group.
Scheme 64: Intramolecular cyclization with the attack on a keto group.
3.5. Synthesis of 1,2-dioxanes by the Kobayashi method
The synthesis is based on the peroxidation of the carbonyl group of unsaturated ketones 228 with the urea–hydrogen peroxide complex followed by a Michael cyclization of the hydroperoxy acetals 229 under basic conditions. This method is suitable for the efficient synthesis of functionalized 1,2-dioxanes 230 in moderate to high yields (Scheme 65, Table 17). In early studies, scandium(III) triflate was used as the catalyst for the hydroperoxidation of ketones with the H2O2–H2NCONH2 complex. More recently, it was shown that in some cases, cheaper catalysts such as p-toluenesulfonic and 10-camphorsulfonic acid can be used for this purpose (Table 17).
Scheme 65: Peroxidation of the carbonyl group in unsaturated ketones 228 followed by cyclization of hydroperoxy acetals 229.
Scheme 65: Peroxidation of the carbonyl group in unsaturated ketones 228 followed by cyclization of hydroperox...
Table 17: Examples of 1,2-dioxanes synthesized by the Kobayashi method.
| Unsaturated ketone 228 | Reaction conditions | 1,2-Dioxane 230 | Yield, % | Reference |
|---|---|---|---|---|
|
|
1) H2O2.H2NCONH2, Sc(OTf)3, MeOH
2) Et2NH, CF3CH2OH, 0 °C, 2 d |
|
1) 67–83
2) 60–72 |
[322,323] |
|
|
1) H2O2.H2NCONH2, Sc(OTf)3, MeOH
2) Et2NH, CF3CH2OH 3) HF, pyridine, THF |
|
1) 52
2) 87 3) 100 |
[324] |
|
|
1) H2O2.H2NCONH2, Sc(OTf)3, MeOH
2) Et2NH, CF3CH2OH |
|
8–38 | [104-106] |
|
|
1) H2O2.H2NCONH2, Sc(OTf)3, MeOH
2) Et2NH, CF3CH2OH |
|
28–58 | [104-106] |
|
Intermediate product 229 has the following structure: |
1) H2O2.H2NCONH2, p-TsOH, MeOH, rt,
20 h 2) Et2NH, CF3CH2OH, rt, 24 h |
|
1) 54–82
2) 52 |
[325-327] |
|
|
H2O2–H2NCONH2, p-TsOH, 1,2-dimethoxyethane, rt, 11 h |
|
35 | [325-327] |
|
Intermediate product 229 has the following structure: |
1) H2O2.H2NCONH2, 10-camphorsulfonic acid, 1,2-dimethoxyethane, rt, 18 h
2) Et2NH, CF3CH2OH, rt, 2 h |
|
1) 86
2) 54 |
[325-327] |
|
|
1) H2O2.H2NCONH2, p-TsOH, EtOH, rt,
10 h 2) Et2NH, CF3CH2OH, rt, 12 h |
|
1) 72–80
2) 40–52 |
[328] |
|
|
1) H2O2.H2NCONH2, p-TsOH, EtOH, rt,
12 h 2) Et2NH, CF3CH2OH, rt |
|
1) 70–93
2) 42–65 |
[328] |
It was found that cesium hydroxide can be used as a base for the cyclization to give 232 and 234. Compared to Scheme 65, the method is suitable for the cyclization of hydroperoxides 231 and 233, which are no ketone derivatives (Scheme 66) [264]. Et3N in MeOH can also be used as catalyst for this type of cyclization [263].
Scheme 66: CsOH and Et2NH-catalyzed cyclization.
Scheme 66: CsOH and Et2NH-catalyzed cyclization.
The synthesis of peroxyplakoric acid methyl ethers A and D 238a and 238b, which are natural peroxides isolated from marine sponges exhibiting fungicidal and antitumor activities [329,330] is an interesting example of the synthesis of complex structures. The polyunsaturated compound (E)-methyl 6-methyleneundec-2-en-10-ynoate (235) was subjected to ozonolysis to obtain methoxyhydroperoxide, (E)-methyl 6-hydroperoxy-6-methoxyundec-2-en-10-ynoate (236), whose cyclization afforded methyl 2-(6-methoxy-6-(pent-4-inyl)-1,2-dioxan-3-yl)acetate (237), in which the triple bond is easily modified by palladium-catalyzed cross-coupling reactions to form the target 1,2-dioxanes 238a,b (Scheme 67).
Scheme 67: Preparation of peroxyplakoric acid methyl ethers A and D.
Scheme 67: Preparation of peroxyplakoric acid methyl ethers A and D.
Initially, an attempt was made to synthesize diethyl 2,2'-(1,2,7,8-tetraoxaspiro[5.5]undecane-3,9-diyl)diacetate (241) by cyclization of (2E,9E)-diethyl 6,6-dihydroperoxyundeca-2,9-dienedioate bis(hydroperoxide) (240) (the bishydroperoxidation product of (2E,9E)-diethyl 6,6-dimethoxyundeca-2,9-dienedioate (239)) with Et2NH in CF3CH2OH. However, these attempts failed. Spiroperoxide 241 was prepared in satisfactory yield by reaction of 240 with the use of mercury (II) acetate (Scheme 68) [331]. The intermediate mercury-containing peroxide produced by the cyclization of bis(hydroperoxide) 240 was reduced with NaBH4 in an alkaline medium [331].
Scheme 68: Hg(OAc)2 in 1,2-dioxane synthesis.
Scheme 68: Hg(OAc)2 in 1,2-dioxane synthesis.
3.6. Synthesis of 1,2-dioxanes from 1,4-dicarbonyl compounds
The reaction of 1,4-diketones 242 (cyclohexanone derivatives) with hydrogen peroxide in a neutral medium produced 3,6-dihydroxydioxanes 243 albeit without reported yields (Scheme 69). The resulting compounds exhibit a broad spectrum of antiparasitic activities against causative agents of malaria, trypanosomiasis, and leishmaniasis [208-212].
Scheme 69: Reaction of 1,4-diketones 242 with hydrogen peroxide.
Scheme 69: Reaction of 1,4-diketones 242 with hydrogen peroxide.
3.7. Methods for the synthesis of 1,2-dioxanes from hydroperoxides
Compounds containing a С=С group and an oxygen-containing ring are convenient starting materials for the synthesis of cyclic peroxides [250-252,332]. For example, the ozonolysis of the double bond in the oxetane-containing compound, 2-methyl-2-(3-methylbut-3-enyl)oxetane (244) afforded 2-(3-hydroperoxy-3-methoxybutyl)-2-methyloxetane (245), which underwent recyclization in the presence of ytterbium triflate to give 2-(6-methoxy-3,6-dimethyl-1,2-dioxan-3-yl)ethanol (246) along with the seven-membered compound 2-hydroperoxy-5-methoxy-2,5-dimethyloxepane (247) (Scheme 70) [250].
Scheme 70: Inramolecular cyclization with oxetane-ring opening.
Scheme 70: Inramolecular cyclization with oxetane-ring opening.
Spirodioxane 227, whose synthesis by the Isayama–Mukaiyama method was described above (Scheme 64), could also be synthesized via the ozonolysis of alkene 248 in the presence of hydrogen peroxide followed by the cyclization of bis(hydroperoxide) 249 with potassium tert-butoxide (Scheme 71) [252].
Scheme 71: Inramolecular cyclization with MsO fragment substitution.
Scheme 71: Inramolecular cyclization with MsO fragment substitution.
An approach to the cyclization based on an intramolecular nucleophilic substitution was used also for the synthesis of diastereomers of dioxanes 252a,b containing triple bonds. Hydroperoxides 251a,b that were synthesized by the ozonolysis of 250 were treated with potassium tert-butoxide. One of the diastereomers, 252a, was then modified first via the stereoselective hydrozirconation and iodination to 253a and then by the Negishi cross coupling to produce silylated product 254a, which was desilylated to obtain alcohol 255a (Scheme 72). 1,2-Dioxane 255a is structurally similar to natural peroxyplakoric acids having fungicidal and antimalarial activities [332].
Scheme 72: Synthesis of 1,2-dioxane 255a, a structurally similar compound to natural peroxyplakoric acids.
Scheme 72: Synthesis of 1,2-dioxane 255a, a structurally similar compound to natural peroxyplakoric acids.
3.8. Use of halonium ions in the cyclization
This approach to the synthesis of 1,2-dioxane rings is based on the intramolecular cyclization of hydroperoxides containing a C=C group. In the first step, the addition of a halonium ion to the double bond results in the formation of a carbocation, which is subjected to the intramolecular attack of the hydroperoxide group.
The treatment of unsaturated monoperoxyketals 257, 260, and 263 (prepared by ozonolysis of 256, 259, and 262 in methanol, respectively) with such donors of halonium ions such as N-iodosuccinimide (NIS), I2/t-BuOK, or bis(sym-collidine)iodonium hexafluorophosphate gave iodine-containing 1,2-dioxanes 258, 261, and 264, in moderated yields (Scheme 73) [333]. It should be noted that attempts to synthesize related peroxides with N-bromosuccinimide failed [333].
Scheme 73: Synthesis of 1,2-dioxanes based on the intramolecular cyclization of hydroperoxides containing C=C groups.
Scheme 73: Synthesis of 1,2-dioxanes based on the intramolecular cyclization of hydroperoxides containing C=C ...
In the studies [334,335] iodine-containing 1,2-dioxanes 266a–c, 268, and 270a,b were synthesized from the corresponding hydroperoxyalkenes 265a–c, 267, and 269a,b with bis(sym-collidine)iodonium hexaflulorophosphate (BCIH) in the cyclization step (Scheme 74).
Scheme 74: Use of BCIH in the intramolecular cyclization.
Scheme 74: Use of BCIH in the intramolecular cyclization.
3.9. Pd(II)-catalyzed cyclization
The palladium-catalyzed cyclization of δ-unsaturated hydroperoxides 271 represents a new route to 1,2-dioxane cyclic compounds 272 (Scheme 75). The cyclization was performed in toluene, 1,4-dioxane, or 1,2-dichloroethane at 80 °C for 3 h in the presence of p-benzoquinone or silver carbonate as the oxidizing agent for Pd(0) that was formed in the catalytic cycle. To the best of our knowledge, this method is the first example of a palladium acetate-catalyzed synthesis of cyclic peroxides [336].
Scheme 75: Palladium-catalyzed cyclization of δ-unsaturated hydroperoxides 271a–e.
Scheme 75: Palladium-catalyzed cyclization of δ-unsaturated hydroperoxides 271a–e.
3.10. Acid-mediated cyclizations of peroxides
The intramolecular cyclization of unsaturated peroxyacetals 273a–d in the presence of TiCl4 or SnCl4 occurs via formation of peroxycarbenium ions to give methoxy- and chlorine-containing dioxanes 274a–d as the reaction products (Scheme 76) [257].
Scheme 76: Intramolecular cyclization of unsaturated peroxyacetals 273a–d.
Scheme 76: Intramolecular cyclization of unsaturated peroxyacetals 273a–d.
The treatment of endoperoxides 275a–d with allyltrimethylsilane in the presence of catalytic amounts of trimethylsilyl triflate or SnCl4 gave bicyclic 1,2-dioxanes 276a–d (Scheme 77) [337].
Scheme 77: Allyltrimethylsilane in the synthesis of 1,2-dioxanes 276a–d.
Scheme 77: Allyltrimethylsilane in the synthesis of 1,2-dioxanes 276a–d.
The electrophilic center of the peroxycarbenium ion produced by the decomposition of molozonide can be trapped by the hydroperoxide group of the molecule. This type of cyclization was used as the basis for the synthesis of hydroperoxide-containing 1,2-dioxanes. The ozonolysis of 1-hydroperoxy-1-methoxy-2-methyl-5-(prop-1-en-2-yl)cyclohexane (277) in a trifluoroethanol/dichloromethane mixture through formation of molozonide 278 and peroxycarbenium ion 279 finally afforded (6S)-6-hydroperoxy-1-methoxy-2,6-dimethyl-7,8-dioxabicyclo[3.3.1]nonane (280) (Scheme 78) [334]. The intramolecular cyclization of intermediate 279 is only possible if the hydroperoxide group is in a particular spatial arrangement [334].
Scheme 78: Intramolecular cyclization using the electrophilic center of the peroxycarbenium ion 279.
Scheme 78: Intramolecular cyclization using the electrophilic center of the peroxycarbenium ion 279.
Under these conditions, ethyl 2-(3-(2-hydroperoxypropan-2-yl)cyclohexylidene)acetate hydroperoxide (281) and ethyl 2-(3-(1-hydroperoxy-1-methoxyethyl)cyclohexylidene)acetate hydroperoxide (283) react to form dioxanes, (1S,5S)-1-hydroperoxy-4,4-dimethyl-2,3-dioxabicyclo[3.3.1]nonane (282), (1S,4S,5S)-1-hydroperoxy-4-methoxy-4-methyl-2,3-dioxabicyclo[3.3.1]nonane (284a), and (1S,4R,5S)-1-hydroperoxy-4-methoxy-4-methyl-2,3-dioxabicyclo[3.3.1]nonane (284b) (Scheme 79) [338].
Scheme 79: Synthesis of bicyclic 1,2-dioxanes.
Scheme 79: Synthesis of bicyclic 1,2-dioxanes.
Under similar conditions, the reaction of 5-hydroperoxy-5-(2-methoxyethoxy)-2-methylhex-1-ene (285) in AcOH/CH2Cl2 produced 3-hydroperoxy-6-(2-methoxyethoxy)-3,6-dimethyl-1,2-dioxane (286) (Scheme 80) [270].
Scheme 80: Preparation of 1,2-dioxane 286.
Scheme 80: Preparation of 1,2-dioxane 286.
3.11. Other methods for the synthesis of 1,2-dioxanes
The di(tert-butyl)peroxalate-initiated radical cyclization of unsaturated 2-(3-hydroperoxypropyl)-6,6-dimethylbicyclo[3.1.1]hept-2-ene hydroperoxide (287) in the presence of oxygen gave 1,2-dioxane (289). The reaction proceeds through formation of compound 288 containing a hydroperoxide group, which is transformed into a carbonyl group by treatment with Ac2O/pyridine (Scheme 81) [232]. The yield of 289 was 14% based on 287.
Scheme 81: Di(tert-butyl)peroxalate-initiated radical cyclization of unsaturated hydroperoxide 287.
Scheme 81: Di(tert-butyl)peroxalate-initiated radical cyclization of unsaturated hydroperoxide 287.
The original cyclization occurs during the oxidation of 1,4-betaines 291a–d prepared from dienones 290a–d containing an azide group in the side chain. The reaction yields peroxide-bridged indolizinediones 292a–d (Scheme 82) [339].
Scheme 82: Oxidation of 1,4-betaines 291a–d.
Scheme 82: Oxidation of 1,4-betaines 291a–d.
3.12. Structural modifications, in which 1,2-dioxane ring remains intact
This section deals with syntheses of compounds exhibiting high antimalarial activity that is comparable with or higher than that of artemisinin.
N-(2-(7-Chloroquinolin-4-ylamino)ethyl)-2-((S)-6,6-dimethyl-1,2-dioxan-3-yl)propanamide (294) containing the aminoquinoline moiety that is characteristic for antiparasitic compounds was synthesized by the following series of steps: reduction of the double bond in the presence of the peroxide group (transformation of ethyl 2-(6,6-dimethyl-1,2-dioxan-3-yl)acrylate (272d) into ethyl 2-((S)-6,6-dimethyl-1,2-dioxan-3-yl)propanoate (293)), alkaline hydrolysis, and amidation (Scheme 83) [336].
Scheme 83: Synthesis of aminoquinoline-containing 1,2-dioxane 294.
Scheme 83: Synthesis of aminoquinoline-containing 1,2-dioxane 294.
The synthesis of the sulfonyl-containing 1,2-dioxane 2-(benzyloxy)-2,6-dimethyl-6-(phenylsulfonylmethyl)-7,8-dioxabicyclo[3.3.1]nonane )297a), included the following steps: oxidation of the sulfide group in 2,6-dimethyl-6-(phenylthiomethyl)-7,8-dioxabicyclo[3.3.1]nonan-2-ol (295) to form 2,6-dimethyl-6-(phenylsulfonylmethyl)-7,8-dioxabicyclo[3.3.1]nonan-2-ol (296) followed by the isolation of the isomer (6R)-2,6-dimethyl-6-(phenylsulfonylmethyl)-7,8-dioxabicyclo[3.3.1]nonan-2-ol (296a) and benzylation of the latter to obtain the target peroxide 297a (Scheme 84) [107].
Scheme 84: Synthesis of the sulfonyl-containing 1,2-dioxane.
Scheme 84: Synthesis of the sulfonyl-containing 1,2-dioxane.
Methyl 2-(6-methoxy-6-pentyl-1,2-dioxan-3-yl)acetate (298) was enzymatically hydrolyzed to 2-(6-methoxy-6-pentyl-1,2-dioxan-3-yl)acetic acid (299). The next step in the synthesis of target compound 301 involved the two-step amidation via the intermediate formation of perfluorophenyl 2-(6-methoxy-6-pentyl-1,2-dioxan-3-yl)acetate (300) (Scheme 85) [110].
The enzymatic hydrolysis step was necessary because attempts to hydrolize ester 298 under alkaline conditions (LiOH in dimethyl sulfoxide) failed and led to peroxide ring-opening [110].
Scheme 85: Synthesis of the amido-containing 1,2-dioxane 301.
Scheme 85: Synthesis of the amido-containing 1,2-dioxane 301.
4. Synthesis of 1,2-dioxenes
4.1. Reaction of 1,3-dienes with singlet oxygen
The reaction of singlet oxygen with the 1,3-diene system can proceed by the following pathways: the [4 + 2]-cycloaddition, the singlet-oxygen–ene reaction, and the [2 + 2]-cycloaddition to form dioxetanes. The reaction pathway depends on the nature of the solvent, and on electronic and steric factors. However, the [4 + 2]-cycloaddition (302 + 1O2) occurs in most cases, and this reaction is frequently used for the synthesis of the 1,2-dioxene system 303 (Scheme 86). Table 18 gives examples of 1,2-dioxenes synthesized by the reaction of singlet oxygen with 1,3-diene systems.
Scheme 86: Reaction of singlet oxygen with the 1,3-diene system 302.
Scheme 86: Reaction of singlet oxygen with the 1,3-diene system 302.
Table 18: Examples of the use of 1O2 in the synthesis of 1,2-dioxenes.
| Alkene 302 | Reaction conditions | 1,2-Dioxene 303 | Yield, % | Reference |
|---|---|---|---|---|
|
|
O2, hν, tetraphenylporphyrin, CH2Cl2, −78 °C, 1 h |
|
100 | [340] |
|
|
O2, hν, tetraphenylporphyrin, CH2Cl2, −78 °C, 2 h |
|
85 | [341] |
|
|
O2, hν, fullerene C60, CDCl3, 0 °C, 2 h |
|
93 | [342] |
|
|
O2, hν, tetraphenylporphyrin, CCl4 |
|
75 | [343] |
|
|
O2, hν, tetraphenylporphyrin, CCl4, rt, 30 min |
|
90 | [344] |
|
|
O2, hν, tetraphenylporphyrin, CCl4, rt, 18–20 h |
|
74
95 |
[345] |
|
|
O2, hν, tetraphenylporphyrin, CCl4, 10 °C, 1.5 h |
|
73 | [346] |
|
|
O2, hν, tetraphenylporphyrin, CHCl3, 10 °C, 45 min |
|
94 | [346] |
|
|
O2, hν, tetraphenylporphyrin, CCl4, rt, 24 h to 9 d |
|
94
36 |
[347] |
|
|
O2, hν, tetraphenylporphyrin, CH2Cl2, −10 °C, 6 h |
|
54 | [348] |
|
|
1) O2, hν, Rose Bengal, MeOH/CH2Cl2 (1/19), 0 °C, 8 h
2) CH2N2 |
|
40 | [182,183] |
|
|
1) O2, hν, methylene blue, CH2Cl2, 15 °C, 30 min
2) PPh3, acetone, rt, 40 min |
|
50 | [349] |
|
|
O2, hν, Rose Bengal, MeCN, 0 °C, 6–16 h |
|
54–82 | [350] |
|
|
O2, hν, Rose Bengal, MeOH/CH2Cl2 (1/19), 0 °C, 6 h |
|
42
66 |
[351] |
|
|
O2, hν, Rose Bengal, CH2Cl2, 6 h |
|
23–70 | [352] |
|
|
O2, hν, tetraphenylporphyrin, benzene, 18 h |
|
28 | [353] |
|
|
O2, hν, Rose Bengal, EtOH, 10 °C, 70 min |
|
56 | [186-190] |
|
|
O2, hν, tetraphenylporphyrin, CH2Cl2 |
|
13–95 | [191-195] |
|
|
O2, hν, Rose Bengal, CH2Cl2 |
|
89 | [354] |
|
|
O2, hν, tetraphenylporphyrin, CHCl3 or Rose Bengal, MeOH, −15 to 0 °C, 1–4 h |
|
70–92 | [355] |
|
|
O2, hν, Rose Bengal, CH2Cl2, 6 h |
|
91 | [356] |
|
|
air, sunlight, CHCl3, rt, 5 d or O2, hν, CH2Cl2, CuSO4, TsOH, 1 h |
|
53–80 | [357,358] |
|
|
air, sunlight, CHCl3, rt, 3 d |
|
60–85 |
[113-116]
[359,360] |
|
|
air, sunlight, CHCl3, rt, 3 d |
|
23 | [119] |
|
|
air, sunlight, CHCl3, rt, 6 d |
|
15 | [119] |
|
|
air, sunlight, CCl4, rt, 160 h |
|
55–80 | [361] |
(+)-Premnalane A is a natural compound of plant origin exhibiting pronounced antimicrobial activity. The synthesis of this compound includes the following steps: oxidation of the furan ring of compound 304, the singlet-oxygen–ene reaction of the double bond-containing bicyclic compound 305, and acid-induced ketalization (Scheme 87) [362].
Scheme 87: Synthesis of (+)-premnalane А and 8-epi-premnalane A.
Scheme 87: Synthesis of (+)-premnalane А and 8-epi-premnalane A.
This synthesis produced a 1:1 mixture of diastereomeric (+)-premnalane А and 8-epi-premnalane A in 24% combined yield and diastereomeric 1,2-dioxolanes 306 in 49% yield. Pure (+)-premnalane A was isolated by column chromatography.
4.2. Structural modifications, in which 1,2-dioxene ring remains intact
Diazo-containing 1,2-dioxenes 309a–e were synthesized starting from the corresponding acids 307a–e, which were transformed into acid chlorides 308a–e and then subjected to diazotization (Scheme 88) [363]. The 1,2-dioxenes 309a–e were used for the intramolecular insertion of carbenes, that were produced by decomposition of the diazo group, into the –О–О–bond [363].
Scheme 88: Synthesis of the diazo group containing 1,2-dioxenes 309a–e.
Scheme 88: Synthesis of the diazo group containing 1,2-dioxenes 309a–e.
6-Epiplakortolide Е is a bicyclic peroxylactone that was isolated in low yield (0.0003%) from the marine sponge Plakortis sp. The structurally related plakortolide Е (Figure 4) exhibits high cytotoxicity against cancer cells and shows also activity against Toxoplasma gondii, which is the causative agent of toxoplasmosis [184,185].
6-Epiplakortolide Е was synthesized by the multistep synthesis involving the oxidation of diene 310 with singlet oxygen to give two isomeric 1,2-dioxenes 311a,b, the isolation of dioxene 311a, its silyl deprotection to form alcohol 312, the oxidation of the latter to 1,2-dioxenic acid 313, the I+-induced lactonization to produce 314, and the deiodination to obtain the target product (Scheme 89) [184,185]. It should be noted that the cyclic peroxide compound 314 remains intact under the reductive conditions in the presence of tributylstannane; this step occurs in good yield (68%) [184,185].
Scheme 89: Synthesis of 6-epiplakortolide Е.
Scheme 89: Synthesis of 6-epiplakortolide Е.
More recently, a similar approach was used for the preparation of tetrahydrofuran-containing bicyclic peroxides 318a,b. It involves the synthesis of 1,2-dioxenes 316 from dienes 315, the cation-initiated cyclization to give bicyclic compounds 317, and the reduction with Bu3SnH. N-Bromo- and iodosuccinimides (NBS and NIS, respectively) were used as donors of halogenide ions. Additionallay, the cyclization was successfully performed with the use of phenylselenyl chloride as the donor of PhSe+ cation (Scheme 90) [364].
Scheme 90: Application of Bu3SnH for the preparation of tetrahydrofuran-containing bicyclic peroxides 318a,b.
Scheme 90: Application of Bu3SnH for the preparation of tetrahydrofuran-containing bicyclic peroxides 318a,b.
Acids 307a and 307b were synthesized by oxidation of the corresponding alcohols with the bis(acetoxy)iodobenzene/2,2,6,6-tetramethyl-1-piperidinyl oxyl (BAIB/TEMPO) system. The cyclization to bicyclic peroxides 319a–f containing the lactone ring was performed with the use of N-bromo- and iodosuccinimides and PhSeCl (Scheme 91) [364]. As in the above-considered case, the peroxide ring remains unchanged upon the reduction of the С–X bond in compounds 319a–f with Bu3SnH [364].
Scheme 91: Application of Bu3SnH for the preparation of lactone-containing bicyclic peroxides 320a–f.
Scheme 91: Application of Bu3SnH for the preparation of lactone-containing bicyclic peroxides 320a–f.
The double bond in the 1,2-dioxene ring of 321 was subjected to dihydroxylation with osmium tetroxide (Scheme 92) [354,365]. The reaction was performed in aqueous tert-butanol, acetone, or acetonitrile at room temperature. Several methods were used for the oxidation. For example, the commercially available AD-mix, a mixture consisting of K2OsO2(OH)4 (catalytic amounts, a source of OsO4) and K3Fe(CN)6 (oxidizer), was employed for this purpose. In this reaction, K2OsO4 (0.5 mol %) combined with oxidizers (K3Fe(CN)6, N-methylmorpholine N-oxide, citric acid, or KClO3) was also used [354,365].
Scheme 92: Dihydroxylation of the double bond in the 1,2-dioxene ring 321 with OsO4.
Scheme 92: Dihydroxylation of the double bond in the 1,2-dioxene ring 321 with OsO4.
The epoxidation of 1,2-dioxenes 324 produced by the addition of singlet oxygen to dienes 323 was performed by treatment with m-chlorobenzoic acid (Scheme 93). It was shown that epoxidized dioxanes 325 and 326, as well as dioxenes 324, have inhibiting activity against the causative agents of candidiasis infections Candida albicans, Candida krusei, and Candida tropicalis, that are in some cases comparable with the activity of the currently used amphotericin B, ketonazole, and nystatin [218-228]. In addition, these compounds exhibit pronounced antimalarial activity, although lower than that of artemisinin [366,367].
Scheme 93: Epoxidation of 1,2-dioxenes 324.
Scheme 93: Epoxidation of 1,2-dioxenes 324.
The cyclopropanation of the double bond in endoperoxides 327 was performed by the reaction with diazomethane in the presence of Pd(OAc)2 to produce 328a,b (Scheme 94) [368].
Scheme 94: Cyclopropanation of the double bond in endoperoxides 327.
Scheme 94: Cyclopropanation of the double bond in endoperoxides 327.
Pyridazine-containing bicyclic endoperoxides 334a–c were synthesized by the inverse-electron-demand Diels–Alder cycloaddition of dimethyl 1,2,4,5-tetrazine-3,6-dicarboxylate (329) to 1,2-dioxenes 330 followed by the elimination of dinitrogen from 331a–c to give 332a–c, the isomerization to 333a–c, and the oxidation (Scheme 95) [369].
Scheme 95: Preparation of pyridazine-containing bicyclic endoperoxides 334a–c.
Scheme 95: Preparation of pyridazine-containing bicyclic endoperoxides 334a–c.
5. Synthesis of 1,2,4-trioxanes
This part is devoted to methods for the synthesis of the 1,2,4-trioxane ring by the singlet-oxygen ene reaction with unsaturated alcohols, the photooxidation of enol ethers and vinyl sulfides, the [4+2]-cycloaddition of singlet oxygen to the pyran system, the Isayama-Mukaiyama peroxysilylation of unsaturated alcohols, reactions with hydrogen peroxide, and the intramolecular Kobayashi cyclization.
5.1. Use of singlet oxygen in the synthesis of 1,2,4-trioxane
One of the widely used approaches to the synthesis of the 1,2,4-trioxane ring 337 is based on the hydroperoxidation of unsaturated alcohols 335 with singlet oxygen (the singlet-oxygen ene reaction) and the acid-catalyzed condensation of the resulting vicinal hydroxy hydroperoxides 336 with ketones or aldehydes (acetals, orthoesters) (Scheme 96, Table 19).
The method was described for the first time by Singh in 1990 [370]. Due to a wide structural series of prepared 1,2,4-trioxane systems and the use of readily available inexpensive reagents, this is an efficient method for their synthesis.
Scheme 96: Synthesis of 1,2,4-trioxanes 337 by the hydroperoxidation of unsaturated alcohols 335 with 1O2 and the condensation of the hydroxy hydroperoxides 336 with carbonyl compounds.
Scheme 96: Synthesis of 1,2,4-trioxanes 337 by the hydroperoxidation of unsaturated alcohols 335 with 1O2 and ...
Table 19: Examples of 1,2,4-trioxanes synthesized by the singlet oxygen ene reaction.
| Alkene 335 | Carbonyl compound | Reaction conditionsa | Product 337 |
Yield
i) 336, % ii) 337, % |
Reference |
|---|---|---|---|---|---|
|
|
|
1) O2, hν, TPP
2) BF3·Et2O, CH2Cl2 |
|
i) 88–94
ii) 63–78 |
[371] |
|
|
|
1) O2, hν, methylene blue, MeCN,
0 °C, 4–6 h 2) TsOH, CH2Cl2, rt, 1 h |
|
i) 30–45
ii) 50–74 |
[372] |
|
|
|
1) O2, hν, methylene blue, MeCN, −10 °C
2) acid 3) NaBH4, MeOH |
|
The yield was not determined | [373] |
|
|
|
1) O2, hν, methylene blue, MeCN,
0 °C, 4–6 h 2) HCl, 5°C, 18 h |
|
22–51 | [374-379] |
|
|
|
1) O2, hν, methylene blue, MeCN,
0 °C, 4–6 h 2) HCl, rt, 1 h |
|
45–70 | [380] |
|
|
|
1) O2, hν, methylene blue, MeCN,
0 °C, 6 h 2) TsOH, CH2Cl2, rt, 1 h |
|
i) 43
ii) 65–76 |
[381] |
|
|
|
1) O2, hν, methylene blue, MeCN,
0 °C, 6 h 2) TsOH, CH2Cl2, rt, 1 h |
|
i) 37
ii) 46–59 |
[381] |
|
|
|
1) O2, hν, methylene blue, MeCN,
0 °C, 18 h 2) HCl, CH2Cl2, 0 °C, 3–6 h |
|
i) 22–35
ii) 12–37 |
[382] |
|
|
|
1) O2, hν, methylene blue, MeCN,
0 °C, 6 h 2) TsOH, CH2Cl2, rt, 2 h |
|
i) 50
ii) 31–75 |
[383] |
|
|
|
1) O2, hν, methylene blue, MeCN,
0 °C, 6 h; 2) TsOH, CH2Cl2, rt, 2 h |
|
i) 35
ii) 57–83 |
[383] |
|
|
|
1) O2, hν, TPP
2) BF3·Et2O, Et2O, 0 °C |
|
i) 54–97
ii) 8–78 |
[384-387] |
|
|
|
1) O2, hν, TTP,
polystyrene 2) BF3·Et2O, CH2Cl2 |
|
20–86 | [385,387,388] |
|
|
|
1) O2, hν, TTP or TPP PS-DVB-based
2) BF3·Et2O, CH2Cl2 |
|
20 | [389] |
|
|
|
1) O2, hν, methylene blue, MeCN, 0 °C,
4–5 h 2) HCl, CH2Cl2, rt, 1 h |
|
38–54 | [390] |
|
|
|
1) O2, hν, methylene blue, MeCN, 0 °C, 5 h
2) HCl, MeCN, rt, 3 h |
|
23-69 | [125-127] |
|
|
|
1) O2, hν, methylene blue, MeCN,
0 °C, 5 h 2) HCl, MeCN, rt, 3 h |
|
40–87 | [125-127] |
|
|
|
1) O2, hν, methylene blue, MeCN,
0 °C, 5 h 2) HCl, MeCN, rt, 3 h |
|
35–55 | [125-127] |
|
|
|
1) O2, hν, TTP or TPP PS-DVB-based
2) BF3·Et2O, CH2Cl2 |
|
4–19 | [389] |
|
|
Intramolecular process |
1) O2, hν, TTP or TPP PS-DVB-based
2) BF3·Et2O, CH2Cl2 |
|
i) 84–91
ii) 12–19 |
[391] |
|
|
R1C(OMe)3
R1 = Me, Et, n-Pr, Ph |
1) O2, hν, TPP, CCl4, 10 °C
2) PPTS, CH2Cl2 |
|
i) 94–99
ii) 21–30 |
[391] |
|
|
MeC(OMe)3 |
1) O2, hν, TPP, CCl4, 10 °C
2) PPTS, CH2Cl2 |
|
i) 83
ii) 19 |
[391] |
|
|
RC(OMe)3
R = Me, Et, n-Pr |
1) O2, hν, TPP, CCl4, rt
2) TsOH, CH2Cl2, rt |
|
i) 83
ii) 18–55 |
[392] |
aTPP is tetraphenylporphyrin; TTP is tetratolylporphyrin; PPTS is pyridinium para-toluenesulfonate.
A similar approach based on the co-oxidation of hydroxyalkenes 338 and thiols (TOCO-reaction, thiol–olefin co-oxygenation) was applied to the synthesis of sulfur-containing 1,2,4-trioxanes 339 (Scheme 97).
Scheme 97: Synthesis of sulfur-containing 1,2,4-trioxanes 339.
Scheme 97: Synthesis of sulfur-containing 1,2,4-trioxanes 339.
Azobisisobutyronitrile (AIBN) was used as the initiator of the radical reaction. In the second step (condensation), cyclopentanone, cyclohexanone, tert-butylcyclohexanone, 1,4-cyclohexanedione, cyclododecanone, and adamantanone were employed. 1,2,4-Trioxanes 339 were prepared in 25–68% yields in two steps [120,393].
The formation of peroxyketals 342a–g from vicinal hydroxyhydroperoxides 341 (oxidation products of unsaturated alcohols 340) in the presence of boron trifluoride is a convenient approach to the synthesis of the 1,2,4-trioxane ring (Scheme 98) [385].
Scheme 98: BF3·Et2O-catalyzed synthesis of the 1,2,4-trioxanes 342a–g.
Scheme 98: BF3·Et2O-catalyzed synthesis of the 1,2,4-trioxanes 342a–g.
The approach to the synthesis of 1,2,4-trioxanes proposed by Jefford and co-workers in 1993 [394] is based on the photooxidation of enol ethers or vinyl sulfides 343 with oxygen followed by the rearrangement of the resulting 1,2-dioxetanes in the presence of trialkylsilyl triflates. The resulting bicyclic compound 344 is structurally similar to artemisinin. Another version of this synthesis is based on the use of the ozone/triphenylphosphite in the oxidation step 1) (Scheme 99, Table 20).
Scheme 99: Photooxidation of enol ethers or vinyl sulfides 343.
Scheme 99: Photooxidation of enol ethers or vinyl sulfides 343.
Table 20: Examples of 1,2,4-trioxanes synthesized by oxidation of enol ethers or vinyl sulfides.
| Enol ether or vinyl sulfide 343 | Reaction conditions | Product 344 | Yield, % | Reference |
|---|---|---|---|---|
|
|
1) O2, hν, methylene blue, CH2Cl2, −78 °C
2) TBDMSOTf, CH2Cl2, −78 °C 3) Et3N, CH2Cl2, −78 °C to −15 °C |
|
47 (12-α)
47 (12-β) |
[395] |
|
|
1) O3, (PhO)3P, CH2Cl2,
−78 °C 2) Et3SiOTf, CH2Cl2, −78 °C 3) Et3N, CH2Cl2, −78 °C to rt |
|
30–38 | [395] |
|
|
1) O3, (PhO)3P, CH2Cl2,
−78 °C 2) Me3SiOTf, CH2Cl2, −78 °C 3) Et3N, CH2Cl2, −78 °C to rt |
|
7 | [395] |
|
|
1) O3, (PhO)3P, CH2Cl2,
−78 °C 2) Me3SiOTf, CH2Cl2, −78 °C 3) Et3N, CH2Cl2, −78 °C to rt |
|
3 (12-α)
11 (12-β) |
[395] |
|
|
1) Air, hν, methylene blue, CH2Cl2, −90 °C
2) Me3SiOTf, CH2Cl2, −90 °C, 1 h 3) 1- ethylpiperidine, CH2Cl2, −78 °C to rt |
|
36 (12-α)
15 (12-β) |
[396] |
|
|
1) Air, hν, methylene blue, CH2Cl2, −78 °C, 1 h
2) Me3SiOTf, CH2Cl2, −78 °C, 2 h 3) 1-ethylpiperidine, CH2Cl2, −78 °C to rt |
|
33 | [397] |
This method was applied to the synthesis of tricyclic peroxide 346 (containing one carbon atom less in the mono-oxygen ring compared to structures 344) from the enol ether, 1-(2-(methoxymethylene)cyclohexyl)-3-phenylpropan-2-one (345) (Scheme 100) [207].
Scheme 100: Synthesis of tricyclic peroxide 346.
Scheme 100: Synthesis of tricyclic peroxide 346.
The reaction of endoperoxides 348a,b derived from cyclohexadienes 347a,b with 1,4-cyclohexanedione produced trioxanes 349a,b containing a keto group which is useful for further transformations (Scheme 101) [398].
Scheme 101: Reaction of endoperoxides 348a,b derived from cyclohexadienes 347a,b with 1,4-cyclohexanedione.
Scheme 101: Reaction of endoperoxides 348a,b derived from cyclohexadienes 347a,b with 1,4-cyclohexanedione.
Unsaturated bicyclic trioxanes 351 are [4 + 2]-cycloaddition products of singlet oxygen to the pyran moiety in 350 (Scheme 102, Table 21).
Scheme 102: [4 + 2]-Cycloaddition of singlet oxygen to 2Н-pyrans 350.
Scheme 102: [4 + 2]-Cycloaddition of singlet oxygen to 2Н-pyrans 350.
It was shown that in this reaction the starting pyran can serve as the sensitizer for the formation of singlet oxygen [402].
5.2. Synthesis of 1,2,4-trioxanes by the Isayama–Mukaiyama method
The Isayama–Mukaiyama peroxysilylation of unsaturated alcohols 352 is a new route to hydroxy silyl peroxides 353, whose condensation with ketones in an acidic medium affords 1,2,4-trioxanes 354 (Scheme 103, Table 22).
Scheme 103: Synthesis of 1,2,4-trioxanes 354 using peroxysilylation stage.
Scheme 103: Synthesis of 1,2,4-trioxanes 354 using peroxysilylation stage.
Table 22: Examples of 1,2,4-trioxanes synthesized through the Isayama–Mukaiyama peroxysilylation.
| Unsaturated alcohol 352 | Carbonyl compound | Reaction conditionsa | 1,2,4-Trioxane 354 | Yield, % | Reference |
|---|---|---|---|---|---|
|
|
|
1) Co(acac)2, Et3SiH, O2, rt
2) TsOH |
|
1) 60
2) 40–90 |
[403] |
|
|
|
1) Co(acac)2, Et3SiH, O2, rt
2) TsOH |
|
42
54 |
[403] |
|
|
|
1) Co(thd)2, Et3SiH, O2, rt
2) TsOH |
|
40–85 | [404] |
|
|
|
1) Co(acac)2, Et3SiH, O2, EtOH, 4 h
2) TsOH, CHCl3, 45 min to 3.5 h |
|
1) 36
2) 57–100 |
[153-158] |
a(Co(II)(thd)2) is bis(2,2,6,6-tetramethyl-3,5-heptanedionato)cobalt (II).
5.3. Use of epoxides as starting reagents in the synthesis of 1,2,4-trioxanes
An important approach to the synthesis of 1,2,4-trioxanes 357 is based on the epoxide-ring opening in 355 with hydrogen peroxide in the presence of a catalyst followed by the condensation of the vicinal hydroxy hydroperoxides 356 with ketones (Scheme 104, Table 23). The drawbacks of this method are generally low yields of 356 in the step of the epoxide–ring opening and difficulties of their isolation from the reaction mixture.
Scheme 104: Epoxide-ring opening in 355 with H2O2 followed by the condensation of hydroxy hydroperoxides 356 with ketones.
Scheme 104: Epoxide-ring opening in 355 with H2O2 followed by the condensation of hydroxy hydroperoxides 356 wi...
Table 23: Examples of 1,2,4-trioxanes 357 synthesized based on epoxides 355.
| Epoxide 355 | Carbonyl compound | Reaction conditions | 1,2,4-Trioxane 357 |
Yield
i) 356 ii) 357, % |
Ref. |
|---|---|---|---|---|---|
|
|
|
1) MoO2(acac)2, H2O2 , Et2O, MgSO4
2) TsOH, CH2Cl2, rt |
|
i) 59
ii) 95 |
[405] |
|
|
|
i) 59
ii) 69 |
[405] | ||
|
|
|
1) MoO2(acac)2, H2O2, THF, MgSO4
2) 10-camphor- sulfonic acid, CH2Cl2, rt |
|
i) 29
ii) 46 |
[405] |
|
|
|
1) H2O2, Et2O,
0 °C, 4 h 2) H2SO4, CH2Cl2, rt, 4 d |
|
i) 8
ii) 28 |
[406] |
|
|
|
1) MoO2(acac)2, H2O2, Et2O, MgSO4, rt, 22 h
2) TsOH, CH2Cl2, rt, 5 h |
|
i) 98
ii) 92 |
[407] |
|
|
|
1) MoO2(acac)2, H2O2, Et2O
2) 10-camphor- sulfonic acid, CH2Cl2 |
|
i) 25
ii) 39 |
[407] |
|
|
|
1) MoO2(acac)2, H2O2 , Et2O, MgSO4
2) BF3·Et2O, CH2Cl2, −78 °C to 0 °C, 5 h |
|
i) -
ii) 27–35 |
[175]
[176] |
The reaction of unsaturated ketones 358 with H2O2/CF3COOH/H2SO4 in dichloromethane produced 1,2,4-trioxanes 359 in 25–95% yields (Scheme 105). It is assumed that in the first step, the hydroperoxidation of the keto group in 358 and the epoxidation of the double bond occur followed by the acid-induced intramolecular cyclization to form bicyclic compound 359 [408].
Scheme 105: Peroxidation of unsaturated ketones 358 with the H2O2/CF3COOH/H2SO4 system.
Scheme 105: Peroxidation of unsaturated ketones 358 with the H2O2/CF3COOH/H2SO4 system.
5.4. Synthesis of 1,2,4-trioxanes by the Kobayashi method
A convenient method for the synthesis of bicyclic trioxanes 362 was developed based on the hydroperoxidation of polyfunctional compounds 360 with the urea–hydrogen peroxide complex followed by the base-mediated intramolecular cyclization of 361 (Scheme 106). The yield of hydroperoxides 361 was 86–90%. In the second step, the intramolecular cyclization was performed in the presence of a catalytic amount of diethylamine. The yields of trioxanes 362 are in the range of 10–35% [409,410].
Scheme 106: Synthesis of 1,2,4-trioxanes 362 through Et2NH-catalyzed intramolecular cyclization.
Scheme 106: Synthesis of 1,2,4-trioxanes 362 through Et2NH-catalyzed intramolecular cyclization.
5.5. Structural modifications, in which 1,2,4-trioxane ring remains intact
The possibility of the reduction of the double bond in tricyclic peroxides 363 by hydrogen with the use of the mixed platinum–rhodium catalyst to form products, in which the 1,2,4-trioxane moiety remains intact, was exemplified by the synthesis of peroxides 364 (Scheme 107) [411].
Scheme 107: Reduction of the double bond in tricyclic peroxides 363.
Scheme 107: Reduction of the double bond in tricyclic peroxides 363.
1,2,4-Trioxane esters 366 were synthesized in high yield from 1,2,4-trioxane ketones 365 by the Horner–Wadsworth–Emmons reaction in the presence of sodium hydride as the base (Scheme 108) [375]. Compounds 366 exhibit antimalarial activity comparable with that of artemisinin.
Scheme 108: Horner–Wadsworth–Emmons reaction in the presence of peroxide group.
Scheme 108: Horner–Wadsworth–Emmons reaction in the presence of peroxide group.
Peroxide dyad 369 consisting of 1,2,4-trioxane moieties of different types was synthesized by the esterification of artesunic acid with 2-((3S,6R)-1-methyl-6-(prop-1-en-2-yl)-7,8,9-trioxabicyclo[3.3.1]nonan-3-yl)ethanol (368) (obtained by the reduction of ethyl 2-((3S,6R)-1-methyl-6-(prop-1-en-2-yl)-7,8,9-trioxabicyclo[3.3.1]nonan-3-yl)acetate (367)) in the presence of N,N’-dicyclohexylcarbodiimide (DCC) (Scheme 109) [392]. The particular structural feature of compound 369 is that it contains a natural peroxide moiety (artesunic acid) combined with the synthetic 1,2,4-trioxane moiety.
Scheme 109: Reduction of ester group by LiBH4 in the presence of 1,2,4-trioxane moiety.
Scheme 109: Reduction of ester group by LiBH4 in the presence of 1,2,4-trioxane moiety.
Trioxaquines are hybrid compounds containing the 1,2,4-trioxane and aminoquinoline moieties. They attracted interest because of a dual mode of action on Plasmodium. One of these compounds, PA1103/SAR116242, was selected as a drug candidate. The final step of its synthesis involves the reductive amination of keto-containing 1,2,4-trioxane 370 with N1-(7-chloroquin-4-yl)cyclohexane-1,4-diamine (371) (Scheme 110) [86].
Scheme 110: Reductive amination of keto-containing 1,2,4-trioxane 370.
Scheme 110: Reductive amination of keto-containing 1,2,4-trioxane 370.
Trioxaferroquines, ferrocene-containing compounds, belong to a new type of hybrid molecules exhibiting high antimalarial activity. The last step of the synthesis of one of these compounds (373) based on the reductive amination of ketone 370 with amine 372 is shown in Scheme 111. The unusual fact is that compound 373 bearing the peroxide bond and a Fe-containing moiety is stable [412].
Scheme 111: Reductive amination of keto-containing 1,2,4-trioxane and a Fe-containing moiety.
Scheme 111: Reductive amination of keto-containing 1,2,4-trioxane and a Fe-containing moiety.
6. Synthesis fo 1,2,4,5-tetraoxanes
The most widely used approaches to the synthesis of 1,2,4,5-tetraoxanes are based on the reaction of ketones and aldehydes with hydrogen peroxide or gem-bishydroperoxides catalyzed by protic or aprotic acids, MeReO3, Re2O7, and iodine. These methods were used for the synthesis of a wide range of symmetrical and unsymmetrical 1,2,4,5-tetraoxanes.
6.1. Acid-catalyzed cyclocondensation of ketones and aldehydes with hydrogen peroxide
This cyclocondensation is the simplest route to some symmetrical (containing identical substituents in positions 3 and 6) 1,2,4,5-tetraoxanes 375 starting from ketones 374 (Scheme 112, Table 24). The acid-catalyzed reactions of hydrogen peroxide with dialkyl ketones, cycloalkanones, and substituted medium-size cycloalkanones produce symmetrical 1,2,4,5-tetraoxanes in moderate to high yields. The drawback of this method is the high sensitivity of the yields of the target peroxides to the structure of the starting carbonyl compounds.
Scheme 112: Acid-catalyzed reactions of Н2О2 with ketones and aldehydes 374.
Scheme 112: Acid-catalyzed reactions of Н2О2 with ketones and aldehydes 374.
Table 24: Examples of symmetrical 1,2,4,5-tetraoxanes 375 synthesized by the acid-catalyzed cyclocondensation of ketones and aldehydes with Н2О2.
| Tetraoxane 375; R1, R2 | Reaction conditions | Yield, % | Reference |
|---|---|---|---|
| –CH(Et)(CH2)4– | H2O2, MeCN, H2SO4, −20 °С, 48 h | 24 | [413] |
| –CH(Pr)(CH2)4– | 7 | ||
| –CH2CH(Me)(CH2)3– | 32 | ||
| –CH(Me)CH(Me)(CH2)3– | 4 | ||
| –CH(Me)CH2CH(Me)(CH2)2– | 20 | ||
| –CH(Me)(CH2)2CH(Me)CH2– | 26 | ||
| –(CH2)2C(Me)2(CH2)2– | 29 | ||
| –CH(iPr)(CH2)2CH(Me)CH2– | 20 | ||
| –CH(Me)(CH2)2CH(iPr)CH2– | 26 | ||
| –CH(Me)CH2C(Me)2(CH2)2– | 34 | ||
| –CH2C(Me)2CH2C(Me)2CH2– | 68 | ||
| –C(Me)2(CH2)4– | 26 | [414] | |
| –CH(Me)(CH2)3CH(Me)– | H2O2, EtOH/H2O, H2SO4, 0 °С | 18 | |
| –(CH2)5– | 90 | ||
| –(CH2)5– | H2O2, (CF3)2CHOH | ≈100 | [415] |
| H, –(CH2)3CHO | H2O2, EtOH/H2O, H2SO4, −10°C, 1 h | 80 | [416] |
6.2. Use of the bis(trimethylsilyl)peroxide/trimethylsilyltrifluoromethanesulfonate system in the cyclocondensation of carbonyl compounds
The cyclocondensation of carbonyl compounds 376a–d with Me3SiOOSiMe3/CF3SO3SiMe3 afforded steroidal tetraoxanes 377a–d (Scheme 113) [417,418]. The cyclocondensation of ketones 376 was performed at 0 °С in acetonitrile using a 1.5-fold molar excess of Me3SiOOSiMe3 and CF3SO3SiMe3 with respect to ketone 376 [417,418].
Scheme 113: Cyclocondensation of carbonyl compounds 376a–d using Me3SiOOSiMe3/CF3SO3SiMe3.
Scheme 113: Cyclocondensation of carbonyl compounds 376a–d using Me3SiOOSiMe3/CF3SO3SiMe3.
6.3. MeReO3-catalyzed peroxidation of ketones
1,1-Dihydroperoxy-4-methylcyclohexane (379) and symmetrical tetraoxane 380 were selectively synthesized in high yields from 4-methylcyclohexanone (378) with the use of the 30% Н2О2/MeReO3/fluorinated alcohol system (Scheme 114) [419].
Scheme 114: Peroxidation of 4-methylcyclohexanone (378).
Scheme 114: Peroxidation of 4-methylcyclohexanone (378).
The use of fluorinated alcohols as the solvent results in an increase in the selectivity of the synthesis. Under similar conditions, symmetrical 3,6-diphenyl- and 3,6-di-(n-heptyl)-1,2,4,5-tetraoxanes 382a,b were synthesized from benzaldehyde (381a) and n-octanal (381b), respectively (Scheme 115) [419].
Scheme 115: Synthesis of symmetrical tetraoxanes 382a,b from aldehydes 381a,b.
Scheme 115: Synthesis of symmetrical tetraoxanes 382a,b from aldehydes 381a,b.
Unsymmetrical tetraoxanes 383a–d were prepared from 4-methylcyclohexanone (378) by the reaction with ketones (R1COR2) using of 1 equiv of HBF4, 2 equiv of Н2О2, and 0.1 mol % MeReO3 in CF3CH2OH (TFE) at room temperature. The unsymmetrical tetraoxane, 3,3-dibutyl-6-heptyl-1,2,4,5-tetraoxane (384), was synthesized from octanal (381b) with the use of CH3CHOHCF3 (HFIP) (Scheme 116) [419].
Scheme 116: Synthesis of unsymmetrical tetraoxanes using of MeReO3.
Scheme 116: Synthesis of unsymmetrical tetraoxanes using of MeReO3.
This method was applied to the synthesis of 3,3,6,6-tetraalkyl-1,2,4,5-tetraoxanes 386a–c and 388a–i from cyclic 385a–c and acyclic ketones 387a–i (Scheme 117) [420], as well as of dispiro-1,2,4,5-tetraoxanes 390a–c from 4-substituted cyclohexanones 389a–c (Scheme 118) [421].
Scheme 117: Synthesis of symmetrical tetraoxanes using of MeReO3.
Scheme 117: Synthesis of symmetrical tetraoxanes using of MeReO3.
Scheme 118: Synthesis of symmetrical tetraoxanes using of MeReO3.
Scheme 118: Synthesis of symmetrical tetraoxanes using of MeReO3.
The use of the 30% Н2О2/MeReO3 (МТО)/fluorinated alcohol system enabled the synthesis of symmetrical compounds 392 from aldehydes 391 and unsymmetrical tetraoxanes 393 containing aryl (peroxide-destabilizing) substituents from aldehydes 391 (and cycloalkanones) (Scheme 119) [422].
Scheme 119: MeReO3 in the synthesis of symmetrical tetraoxanes with the use of aldehydes.
Scheme 119: MeReO3 in the synthesis of symmetrical tetraoxanes with the use of aldehydes.
Unsymmmetrical 1,2,4,5-tetraoxanes containing adamantane (395a–i) and cyclodecane moieties (396a–d) exhibiting high antimalarial activity (Scheme 120) were prepared from sulfonylpiperidones 394 [138].
Scheme 120: Preparation of unsymmmetrical 1,2,4,5-tetraoxanes with high antimalarial activity.
Scheme 120: Preparation of unsymmmetrical 1,2,4,5-tetraoxanes with high antimalarial activity.
6.4. Re2O7-Catalyzed cyclocondensation of gem-bishydroperoxides with ketones
Re2O7 is an efficient catalyst for the addition of hydroperoxide groups to ketones and aldehydes. Due to these properties, Re2O7 can be used in the one-pot synthesis of unsymmetrical 1,2,4,5-tetraoxanes 398 from ketones 397 in good yields (Scheme 121, Table 25) [423].
Scheme 121: Re2O7-Catalyzed synthesis of tetraoxanes 398.
Scheme 121: Re2O7-Catalyzed synthesis of tetraoxanes 398.
Table 25: Re2O7-Catalyzed synthesis of tetraoxanes 398.
| Ketone A, 397 | Ketone B | Reaction conditions | Tetraoxane 398 | Yield, % |
|---|---|---|---|---|
|
|
|
1) Н2О2 (2 equiv), 0.5 h, 0 °С
2) Ketone B (2 equiv), CH2Cl2, 1 h, rt |
|
67 |
|
|
|
1) Н2О2 (4 equiv), 0.5 h, rt
2) Ketone B (4 equiv), 2,2,2-trifluoroethanol, Re2O7, 0.5 h, rt |
|
69 |
|
|
|
1) Н2О2 (4 equiv), 6 h, rt
2) Ketone B (4 equiv), 2,2,2-trifluoroethanol, Re2O7, 2 h, rt |
|
49 |
6.5. Protic acid-catalyzed cyclocondensation of gem-bishydroperoxides with ketones
Unsymmetrical steroidal tetraoxanes 401 were synthesized by the hydroperoxidation of methyl 3-oxo-7α,12α-diacetoxy-5β-cholan-24-oate (399) in the presence of HCl followed by the condensation of bishydroperoxide 400 with the corresponding ketone in the presence of H2SO4 (Scheme 122) [128,132,141,142].
Scheme 122: H2SO4-Catalyzed synthesis of steroidal tetraoxanes 401.
Scheme 122: H2SO4-Catalyzed synthesis of steroidal tetraoxanes 401.
Structurally more simple ketones, for example, acetone, are also involved in the cyclocondensation with bishydroperoxide 400 [141].
The synthesis of keto-containing tetraoxane 403 was also performed in two steps [144]. Thus the intermediate 1,1-dihydroperoxycyclohexane 402 was prepared from cyclohexanone in a neutral medium, and its condensation with 1,4-cyclohexanedione was carried out in the presence of HBF4 (Scheme 123).
Scheme 123: HBF4-Catalyzed condensation of bishydroperoxide 402 with 1,4-cyclohexanedione.
Scheme 123: HBF4-Catalyzed condensation of bishydroperoxide 402 with 1,4-cyclohexanedione.
6.6. Cyclocondensation of bishydroperoxides with acetals and enol ethers
The method for the synthesis of 1,2,4,5-tetraoxanes 407 and 408 is based on the boron trifluoride etherate-catalyzed reaction of gem-bishydroperoxides 404 with enol ethers 405 and acetals 406 under mild conditions. More than two dozens of tetraoxanes were synthesized in yields from 45 to 95% according to this method (Scheme 124). The advantage of this method is the use of readily available starting compounds, such as acetals, enol ethers, and boron trifluoride etherate [424,425].
Scheme 124: BF3·Et2O-Catalyzed reaction of gem-bishydroperoxides 404 with enol ethers 405 and acetals 406.
Scheme 124: BF3·Et2O-Catalyzed reaction of gem-bishydroperoxides 404 with enol ethers 405 and acetals 406.
The bishydroperoxidation of 1,3-dioxolane 409 was carried out in the presence of H2WO4. The following HBF4-catalyzed condensation of bishydroperoxide 410 with ketones gave 1,2,4,5-tetraoxanes 411a–c containing the ester group (Scheme 125) [144].
Scheme 125: HBF4-Catalyzed cyclocondensation of bishydroperoxide 410 with ketones.
Scheme 125: HBF4-Catalyzed cyclocondensation of bishydroperoxide 410 with ketones.
6.7. Iodine-catalyzed one-pot synthesis of symmetrical and unsymmetrical tetraoxanes
The reaction of substituted benzaldehyde 412 with hydrogen peroxide in the presence of the Lewis acid I2 produced geminal bishydroperoxide, whose condensation with the starting or another substituted benzaldehyde gave tetraoxane 413 (Scheme 126, Table 26) [426,427].
Scheme 126: Synthesis of symmetrical and unsymmetrical tetraoxanes 413 from benzaldehydes 412.
Scheme 126: Synthesis of symmetrical and unsymmetrical tetraoxanes 413 from benzaldehydes 412.
Table 26: Iodine-catalyzed one-pot synthesis of tetraoxanes 413.
| Tetraoxane 413 | Yield, % | Tetraoxane 413 | Yield, % | ||
|---|---|---|---|---|---|
| R1 | R2 | R1 | R2 | ||
| o-Me | o-Me | 42 | p-(t-Bu) | p-(n-Pr) | 32 |
| o-Me | m-Me | 33 | p-(t-Bu) | p-(iPr) | 38 |
| p-Me | p-Me | 54 | p-(n-Pr) | p-(iPr) | 28 |
| p-Me | p-(iPr) | 33 | p-(t-Bu) | p-OMe | 22 |
| p-Me | p-(t-Bu) | 46 | p-(iPr) | p-OMe | 24 |
| p-Me | p-OMe | 25 | p-Et | p-Me | 41 |
| p-Me | p-CO2Me | 37 | p-Et | m-Me | 39 |
| p-Me | o-Me | 25 | p-Et | p-(iPr) | 37 |
| p-Me | m-Me | 38 | p-Et | p-(t-Bu) | 25 |
| p-Me | p-(n-Pr) | 37 | p-(n-Pr) | p-OMe | 24 |
| p-Me | H | 43 | p-Cl | p-Cl | 25 |
| p-Me | p-CHO | 31 | p-Br | p-Br | 22 |
| p-(n-Bu) | p-(n-Bu) | 40 | p-F | p-F | 29 |
| p-(t-Bu) | p-(t-Bu) | 53 | p-OMe | p-OMe | 27 |
| m-Me | m-Me | 51 | p-Et | p-Et | 44 |
| m-Me | H | 30 | p-(n-Pr) | p-(n-Pr) | 38 |
| m-Me | p-OMe | 29 | p- (iPr) | p-(iPr) | 41 |
The iodine-catalyzed one-pot synthesis of symmetrical and unsymmetrical tetraoxanes from substituted benzaldehydes has some advantages over other methods. Thus, it can be performed with the use of mild reagents (which do not decompose peroxide) and it does not need an excess of hydrogen peroxide and substituted benzaldehyde.
6.8. Acid-catalyzed condensation of β-diketones with hydrogen peroxide
The acid-catalyzed condensation of β-diketones 414a–l with hydrogen peroxide is a simple and facile method for the synthesis of bridged 1,2,4,5-tetraoxanes 415a–l. This method enables the synthesis of these compounds on the multigram scale in 47–77% yields (Scheme 127). The high concentration of a strong acid, such as H2SO4, HBF4, or HClO4 (2 g of the acid per 5 mL of the solvent) is the key factor determining the yield and selectivity of the synthesis of 1,2,4,5-tetraoxanes. Under these conditions, the targeted compounds are produced selectively even in the presence of an excess of hydrogen peroxide [428]. Unlike many compounds with the O–O bond, which are rearranged in acidic media, the resulting cyclic peroxides are fairly stable under these reaction conditions.
Scheme 127: Synthesis of bridged 1,2,4,5-tetraoxanes 415a–l from β-diketones 414a–l and H2O2.
Scheme 127: Synthesis of bridged 1,2,4,5-tetraoxanes 415a–l from β-diketones 414a–l and H2O2.
It was found that phosphomolybdic acid and phosphotungstic acid efficiently catalyze the addition of H2O2 to β-diketones resulting in the selective formation of bridged 1,2,4,5-tetraoxanes. The use of these catalysts made it possible to obtain bridged tetraoxanes from easily oxidizable benzoylacetone derivatives and α-unsubstituted β-diketones [429].
6.9. Synthesis of symmetrical 1,2,4,5-tetraoxanes by the ozonolysis of unsaturated compounds
The dimerization of zwitterions 417 produced by decomposition of ozonides 416 affords symmetrical tetraoxanes 418 (Scheme 128).
Scheme 128: Dimerization of zwitterions 417.
Scheme 128: Dimerization of zwitterions 417.
For example, the ozonolysis of verbenone 419 via the formation of zwitterioninc structures 420 and 421 gives a mixture of two symmetrical 1,2,4,5-tetraoxanes 422 and 423 (Scheme 129) [430]. Peroxides 422 and 423 are unstable due to the presence of carbonyl groups adjacent to the O–O group, and they almost completely decompose as the temperature is raised.
Scheme 129: Ozonolysis of verbenone 419.
Scheme 129: Ozonolysis of verbenone 419.
3,3,6,6-Tetrapentyl-1,2,4,5-tetraoxane (425) was synthesized in a similar way by the ozonolysis of undecan-6-one O-methyl oxime (424) (Scheme 130) [431,432]. It should be noted that this approach is not widely used because of a limited number of appropriate structures and low yields of the target products.
Scheme 130: Ozonolysis of O-methyl oxime 424.
Scheme 130: Ozonolysis of O-methyl oxime 424.
6.10. Other methods for the synthesis of 1,2,4,5-tetraoxanes
The peroxidation of 1,1,1-trifluorododecan-2-one (426) with oxone afforded the symmetrical tetraoxane, 3,6-didecyl-3,6-bis(trifluoromethyl)-1,2,4,5-tetraoxane (427) (Scheme 131) [433].
Scheme 131: Peroxidation of 1,1,1-trifluorododecan-2-one 426 with oxone.
Scheme 131: Peroxidation of 1,1,1-trifluorododecan-2-one 426 with oxone.
The synthesis of unsymmetrical steroidal tetraoxane 429 in 19% yield was performed by the intramolecular cyclization of dialdehyde 428 with hydrogen peroxide under acidic conditions (Scheme 132) [434].
Scheme 132: Intramolecular cyclization of dialdehyde 428 with H2O2.
Scheme 132: Intramolecular cyclization of dialdehyde 428 with H2O2.
In the synthesis of geminal bishydroperoxides by BF3·Et2O or BF3·MeOH-catalyzed reactions of ketals 430–432 with hydrogen peroxide in Et2O tetraoxanes 433–435 (Scheme 133) are obtained as by-products in 12%, 6%, and 19% yields, respectively [435].
Scheme 133: Tetraoxanes 433–435 as by-products in peroxidation of ketals 430–432.
Scheme 133: Tetraoxanes 433–435 as by-products in peroxidation of ketals 430–432.
Scheme 134 shows the synthesis of 3,3,6,6-tetramethyl-1,2,4,5-tetraoxane (437) in 90% yield by the transformation of the intermediate 3,3,6,6,9,9-hexamethyl-1,2,4,5,7,8-hexaoxononane (436) in acetone [436]. This method is suitable for the preparation of the target product in amounts of only several hundred milligrams.
Scheme 134: Transformation of triperoxide 436 in diperoxide 437.
Scheme 134: Transformation of triperoxide 436 in diperoxide 437.
6.11. Structural modifications, in which 1,2,4,5-tetraoxane ring remains intact
In the last two decades, 1,2,4,5-tetraoxanes were considered as the most promising compounds for the design of antiparasitic drugs. This is due, first, to the high activity of their derivatives and, second, to a wide scope of structural modifications, in which the tetraoxane ring remains intact.
Amides 440a,b and amines 444a,b, and 446 active against various strains of P.falciparum were synthesized from methyl 7,8,15,16-tetraoxadispiro[5.2.5.2]hexadecane-3-carboxylate (438) containing the ester group (Scheme 135) [135,437]. To prepare aminoquinoline derivatives 440a,b, ester 438 was hydrolyzed to 7,8,15,16-tetraoxadispiro[5.2.5.2]hexadecane-3-carboxylic acid (439) followed by the amidation of the latter. The synthesis of products 444a,b and 446 was performed with a wide range of classical reagents for organic synthesis with the intermediate formation of compounds containing such groups as hydroxy 441, azide 442, amino 443, and aldehyde 445.
Scheme 135: Preparation and structural modifications of tetraoxanes.
Scheme 135: Preparation and structural modifications of tetraoxanes.
An interesting feature of the synthesis according to Scheme 135 is the use of such strong reducing agents as LiAlH4 and NaBH(OAc)3, with the products retaining the peroxide ring.
Steroidal tetraoxane 448, which is approximately six times more active that Artelinic acid and 2.4 times as active as arteether against P. falciparum, was also synthesized by the alkaline hydrolysis of ester 401g followed by the amidation of acid 447 (Scheme 136) [128].
Scheme 136: Structural modifications of steroidal tetraoxanes.
Scheme 136: Structural modifications of steroidal tetraoxanes.
Compounds containing a fluorescent moiety are of interest in terms of the mechanism of antiparasitic action of peroxides. For example, 1,2,4,5-tetraoxane 454 containing the 4-chloro-7-methylbenzo[c][1,2,5]oxadiazole moiety was synthesized according to Scheme 137. In the first step, ketone 449 was transformed in tetraoxane 450, whose ester group was subjected to the alkaline hydrolysis to form acid 451 followed by the amidation to give 452 and the hydrolysis to obtain hydrochloride 453. Then the reaction of the latter with 4-chloro-7-nitrobenzo[c][1,2,5]oxadiazole afforded the target compound 454 [138].
Scheme 137: Synthesis of 1,2,4,5-tetraoxane 454 containing the fluorescent moiety.
Scheme 137: Synthesis of 1,2,4,5-tetraoxane 454 containing the fluorescent moiety.
The synthesis of tetraoxane 458 (RKA182) exhibiting the in vitro and in vivo activity comparable with that of artemisinin was performed on the kilogram scale according to Scheme 138. This compound is a promising malaria drug candidate [82,83]. The key steps in this synthesis are the preparation of adamantane-containing tetraoxane 456 from ethyl 2-(4-oxocyclohexyl)acetate (455), the hydrolysis of 456, and the purification to obtain acid 457. The amidation of the latter affords target product 458.
Scheme 138: Synthesis of tetraoxane 458 (RKA182).
Scheme 138: Synthesis of tetraoxane 458 (RKA182).
Conclusion
The review summarizes and generalizes studies on the synthesis of five- and six-membered cyclic peroxides published last decade (since 2000 to present). Most of the currently established methods for the synthesis of these compounds are based on the use of such key oxidizing agents as oxygen, ozone, and hydrogen peroxide. The Isayama–Mukaiyama and Kobayashi methods are widely used in the synthesis of 1,2-dioxolanes, 1,2-dioxanes, and 1,2,4-trioxanes. The reactions with the participation of peroxycarbenium ions play an important role in the synthesis of peroxides.
The Griesbaum coozonolysis of ketones and O-alkyl oximes is the most flexible and efficient method for the synthesis of unsymmetrical 1,2,4-trioxolanes. The [4 + 2]-cycloaddition of oxygen to a 1,3-diene system is, in fact, the only route to 1,2-dioxenes.
Methods for the synthesis of 1,2,4,5-tetraoxanes are based on reactions of ketones, aldehydes, and their dialkyloxy derivatives with hydrogen peroxide or gem-bishydroperoxides catalyzed by protic and aprotic acids, such as MeReO3, Re2O7, and iodine.
Modifications of functional groups to form peroxide ring-retaining products are applicable to the synthesis of cyclic peroxides of various structural types. This approach can be used to prepare complex peroxides exhibiting antiparasitic and antitumor activities.
Carbonyl compound are generally employed as the starting reagents in the synthesis of cyclic peroxides. These methods can be used for the selective peroxidation of monocarbonyl compounds. In the case of dicarbonyl compounds, there are a limited number of efficient procedures for the synthesis of cyclic peroxides.
References
-
McCullough, K. J.; Nojima, M. Curr. Org. Chem. 2001, 5, 601. doi:10.2174/1385272013375346
Return to citation in text: [1] -
Korshin, E. E.; Bachi, M. D. Synthesis of cyclic peroxides. In The Chemistry of Peroxides; Rappoport, Z., Ed.; John Wiley & Sons, Ltd: Chichester, 2006; Vol. 2, Part 1, pp 189 ff. doi:10.1002/0470862769.ch5
Return to citation in text: [1] -
Tang, Y.; Dong, Y.; Vennerstrom, J. L. Med. Res. Rev. 2004, 24, 425. doi:10.1002/med.10066
Return to citation in text: [1] -
O’Neill, P. M.; Posner, G. H. J. Med. Chem. 2004, 47, 2945. doi:10.1021/jm030571c
Return to citation in text: [1] -
Slack, R. D.; Jacobine, A. M.; Posner, G. H. Med. Chem. Commun. 2012, 3, 281. doi:10.1039/c2md00277a
Return to citation in text: [1] -
Masuyama, A.; Wu, J.-M.; Nojima, M.; Kim, H.-S.; Wataya, Y. Mini-Rev. Med. Chem. 2005, 5, 1035. doi:10.2174/138955705774575255
Return to citation in text: [1] -
Van Ornum, S. G.; Champeau, R. M.; Pariza, R. Chem. Rev. 2006, 106, 2990. doi:10.1021/cr040682z
Return to citation in text: [1] -
Jefford, C. W. Drug Discovery Today 2007, 12, 487. doi:10.1016/j.drudis.2007.04.009
Return to citation in text: [1] -
Jefford, C. W. Curr. Top. Med. Chem. 2012, 12, 373. doi:10.2174/156802612799362940
Return to citation in text: [1] -
Dembitsky, V. M. Eur. J. Med. Chem. 2008, 43, 223. doi:10.1016/j.ejmech.2007.04.019
Return to citation in text: [1] [2] [3] [4] [5] -
Siddiq, A.; Dembitsky, V. Anti-Cancer Agents Med. Chem. 2008, 8, 132. doi:10.2174/187152008783497073
Return to citation in text: [1] [2] [3] [4] [5] -
Dembitsky, V. M.; Levitsky, D. O. Nat. Prod. Commun. 2006, 1, 405.
Return to citation in text: [1] [2] [3] [4] [5] -
Dembitsky, V. M. Lipids 2006, 41, 883. doi:10.1007/s11745-006-5044-3
Return to citation in text: [1] [2] [3] [4] [5] -
Dembitsky, V. M. Lipids 2005, 40, 869. doi:10.1007/s11745-005-1449-2
Return to citation in text: [1] [2] [3] [4] [5] -
Dembitsky, V. M. Chem. Biodiversity 2004, 1, 673. doi:10.1002/cbdv.200490060
Return to citation in text: [1] [2] [3] [4] [5] -
Opsenica, D. M.; Šolaja, B. A. J. Serb. Chem. Soc. 2009, 74, 1155. doi:10.2298/JSC0911155O
Return to citation in text: [1] -
Muraleedharan, K. M.; Avery, M. A. Drug Discovery Today 2009, 14, 793. doi:10.1016/j.drudis.2009.05.008
Return to citation in text: [1] -
Kim, B. J.; Sasaki, T. Org. Prep. Proced. Int. 2006, 38, 1. doi:10.1080/00304940609355981
Return to citation in text: [1] -
Baby, J.; Sujatha, S. J. Nat. Prod. (Gorakhpur, India) 2011, 4, 5.
Return to citation in text: [1] -
Hepworth, J. D.; Heron, B. M. Prog. Heterocycl. Chem. 1996, 8, 277. doi:10.1016/S0959-6380(96)80017-1
Return to citation in text: [1] -
Kabal'nova, N. N.; Khursan, S. L.; Shereshovets, V. V.; Tolstikov, G. A. Kinet. Catal. 1999, 40, 207.
Return to citation in text: [1] -
Terent'ev, A. O.; Platonov, M. M.; Levitsky, D. O.; Dembitsky, V. M. Russ. Chem. Rev. 2011, 80, 807. doi:10.1070/RC2011v080n09ABEH004189
Return to citation in text: [1] -
Baader, W. J.; Bastos, E. L. Sci. Synth. 2008, 38, 345–378.
Return to citation in text: [1] -
Baader, W. J.; Bastos, E. L. Sci. Synth. 2008, 38, 379–395.
Return to citation in text: [1] -
Baader, W. J.; Bastos, E. L. Sci. Synth. 2008, 38, 397–420.
Return to citation in text: [1] -
Kumar, N.; Singh, R.; Rawat, D. S. Med. Res. Rev. 2012, 32, 581–610. doi:10.1002/med.20223
Return to citation in text: [1] -
Zmitek, K.; Zupan, M.; Iskra, J. Org. Biomol. Chem. 2007, 5, 3895. doi:10.1039/b711647k
Return to citation in text: [1] -
Trullinger, T. K. Ph.D. Thesis, University of Nebraska, Lincoln, NE, USA, 2002; pp 195 ff.
Diss. Abstr. Int., B 2003, 63, 5855.
Return to citation in text: [1] -
Dai, P. Ph.D. Thesis, University of Nebraska, Lincoln, NE, USA, 2004; pp 186 ff.
Diss. Abstr. Int., B 2005, 66, 281.
Return to citation in text: [1] -
Ramirez, A. P. Ph.D. Thesis, University of California, Irvine, CA, USA, 2007; pp 224 ff.
Diss. Abstr. Int., B 2007, 67, 6412.
Return to citation in text: [1] -
Pena-Quevedo, A. J. Ph.D. Thesis, University of Puerto Rico, Mayaguez, P. R., 2009; pp 159 ff.
Diss. Abstr. Int., B 2010, 70, 4186.
Return to citation in text: [1] -
Yao, G. Ph.D. Thesis, Boston University, Boston, MA, USA, 1999; pp 243 ff.
Diss. Abstr. Int., B 1999, 60, 193.
Return to citation in text: [1] -
Morgan, N. N. Int. J. Phys. Sci. 2009, 4, 885.
Return to citation in text: [1] -
Odinokov, V. N.; Tolstikov, G. A. Russ. Chem. Rev. 1981, 50, 636. doi:10.1070/RC1981v050n07ABEH002658
Return to citation in text: [1] -
Razumovskii, S. D.; Zaikov, G. E. Russ. Chem. Rev. 1980, 49, 1163. doi:10.1070/RC1980v049n12ABEH002535
Return to citation in text: [1] -
Schmidt-Szalowski, K. Przem. Chem. 2000, 79, 115.
Return to citation in text: [1] -
Capon, R. J. Stud. Nat. Prod. Chem. 1991, 9, 15.
Return to citation in text: [1] -
Cafferata, L. F. R. Trends Org. Chem. 1993, 4, 773.
Return to citation in text: [1] -
Tolstikov, G. A.; Tolstikov, A. G.; Tolstikova, O. V. Russ. Chem. Rev. 1996, 65, 769. doi:10.1070/RC1996v065n09ABEH000240
Return to citation in text: [1] -
McCullough, K. J. Contemp. Org. Synth. 1995, 2, 225. doi:10.1039/co9950200225
Return to citation in text: [1] -
Adam, W. Acc. Chem. Res. 1979, 12, 390. doi:10.1021/ar50143a002
Return to citation in text: [1] -
Opsenica, D. M.; Šolaja, B. A. Maced. J. Chem. Chem. Eng. 2012, 31, 137.
Return to citation in text: [1] -
Sawwan, N.; Greer, A. Chem. Rev. 2007, 107, 3247. doi:10.1021/cr0400717
Return to citation in text: [1] -
Clennan, E. L.; Foote, C. In Organic Peroxides; Ando, E., Ed.; Wiley: New York, 1992.
Return to citation in text: [1] -
Balci, M. Chem. Rev. 1981, 81, 91. doi:10.1021/cr00041a005
Return to citation in text: [1] -
Clennan, E. L. Tetrahedron 1991, 47, 1343. doi:10.1016/S0040-4020(01)86413-9
Return to citation in text: [1] -
Doerig, C. D. Nat. Chem. Biol. 2008, 4, 334. doi:10.1038/nchembio0608-334
Return to citation in text: [1] -
Vangapandu, S.; Jain, M.; Kaur, K.; Patil, P.; Patel, S. R.; Jain, R. Med. Res. Rev. 2007, 27, 65. doi:10.1002/med.20062
Return to citation in text: [1] -
Tu, Y. Nat. Med. (N. Y., NY, U. S.) 2011, 17, 1217. doi:10.1038/nm.2471
Return to citation in text: [1] -
Kumar, N.; Sharma, M.; Rawat, D. S. Curr. Med. Chem. 2011, 18, 3889. doi:10.2174/092986711803414340
Return to citation in text: [1] -
Liao, F. Molecules 2009, 14, 5362. doi:10.3390/molecules14125362
Return to citation in text: [1] -
Morris, C. A.; Duparc, S.; Borghini-Fuhrer, I.; Jung, D.; Shin, C.-S.; Fleckenstein, L. Malar. J. 2011, 10, 263. doi:10.1186/1475-2875-10-263
Return to citation in text: [1] -
Robert, A.; Dechy-Cabaret, O.; Cazalles, J.; Meunier, B. Acc. Chem. Res. 2002, 35, 167–174. doi:10.1021/ar990164o
Return to citation in text: [1] -
Hombhanje, F. W.; Huang, Q. Pharmaceuticals 2010, 3, 3581. doi:10.3390/ph3123581
Return to citation in text: [1] -
Kano, S. J. Infect. Chemother. 2010, 16, 375. doi:10.1007/s10156-010-0077-1
Return to citation in text: [1] -
Li, Q.; Weina, P. Pharmaceuticals 2010, 3, 2322. doi:10.3390/ph3072322
Return to citation in text: [1] -
Wright, C. W. Nat. Prod. Rep. 2010, 27, 961. doi:10.1039/c002196m
Return to citation in text: [1] -
Kaur, H.; Green, M. D.; Hostetler, D. M.; Fernandez, F. M.; Newton, P. N. Therapy 2010, 7, 49. doi:10.2217/thy.09.84
Return to citation in text: [1] -
Medhi, B.; Patyar, S.; Rao, R. S.; Byrav, D. S. P.; Prakash, A. Pharmacology 2009, 84, 323. doi:10.1159/000252658
Return to citation in text: [1] -
Eastman, R. T.; Fidock, D. A. Nat. Rev. Microbiol. 2009, 7, 864. doi:10.1038/nrmicro2239
Return to citation in text: [1] -
Ehrhardt, S.; Meyer, C. G. Ther. Clin. Risk Manage. 2009, 5, 805. doi:10.2147/TCRM.S5375
Return to citation in text: [1] -
Gautam, A.; Ahmed, T.; Paliwal, J.; Batra, V. Curr. Drug Metab. 2009, 10, 289. doi:10.2174/138920009787846323
Return to citation in text: [1] -
Bathurst, I.; Hentschel, C. Trends Parasitol. 2006, 22, 301. doi:10.1016/j.pt.2006.05.011
Return to citation in text: [1] -
White, N. J. Science 2008, 320, 330. doi:10.1126/science.1155165
Return to citation in text: [1] -
Nakase, I.; Lai, H.; Singh, N. P.; Sasaki, T. Int. J. Pharm. 2008, 354, 28. doi:10.1016/j.ijpharm.2007.09.003
Return to citation in text: [1] -
Nosten, F.; White, N. J. Am. J. Trop. Med. Hyg. 2007, 77 (Suppl. 6), 181.
Return to citation in text: [1] -
Stocks, P. A.; Bray, P. G.; Barton, V. E.; Al-Helal, M.; Jones, M.; Araujo, N. C.; Gibbons, P.; Ward, S. A.; Hughes, R. H.; Biagini, G. A.; Davies, J.; Amewu, R.; Mercer, A. E.; Ellis, G.; ONeill, P. M. Angew. Chem., Int. Ed. 2007, 46, 6278. doi:10.1002/anie.200604697
Return to citation in text: [1] -
Checkley, A. M.; Whitty, C. J. M. Expert Rev. Anti-Infect. Ther. 2007, 5, 199. doi:10.1586/14787210.5.2.199
Return to citation in text: [1] -
Tilley, L.; Davis, T. M. E.; Bray, P. G. Future Microbiol. 2006, 1, 127. doi:10.2217/17460913.1.1.127
Return to citation in text: [1] -
Gelb, M. H. Curr. Opin. Chem. Biol. 2007, 11, 440. doi:10.1016/j.cbpa.2007.05.038
Return to citation in text: [1] -
Namdeo, A. G.; Mahadik, K. R.; Kadam, S. S. Pharm. Mag. 2006, 2, 106.
Return to citation in text: [1] -
Haynes, R. K. Curr. Top. Med. Chem. (Sharjah, United Arab Emirates) 2006, 6, 509. doi:10.2174/156802606776743129
Return to citation in text: [1] -
Begue, J.-P.; Bonnet-Delpon, D. Drugs Future 2005, 30, 509. doi:10.1358/dof.2005.030.05.901987
Return to citation in text: [1] [2] -
Rosenthal, A. S.; Chen, X.; Liu, J. O.; West, D. C.; Hergenrother, P. J.; Shapiro, T. A.; Posner, G. H. J. Med. Chem. 2009, 52, 1198. doi:10.1021/jm801484v
Return to citation in text: [1] -
Nagelschmitz, J.; Voith, B.; Wensing, G.; Roemer, A.; Fugmann, B.; Haynes, R. K.; Kotecka, B. M.; Rieckmann, K. H.; Edstein, M. D. Antimicrob. Agents Chemother. 2008, 52, 3085. doi:10.1128/AAC.01585-07
Return to citation in text: [1] -
Van der Meersch, H. J. Pharm. Belg. 2005, 60, 23.
Return to citation in text: [1] -
Vennerstrom, J. L.; Arbe-Barnes, S.; Brun, R.; Charman, S. A.; Chiu, F. C. K.; Chollet, J.; Dong, Y.; Dorn, A.; Hunziker, D.; Matile, H.; McIntosh, K.; Padmanilayam, M.; Santo Tomas, J.; Scheurer, C.; Scorneaux, B.; Tang, Y.; Urwyler, H.; Wittlin, S.; Charman, W. N. Nature 2004, 430, 900. doi:10.1038/nature02779
Return to citation in text: [1] [2] -
Valecha, N.; Looareesuwan, S.; Martensson, A.; Abdulla, S. M.; Krudsood, S.; Tangpukdee, N.; Mohanty, S.; Mishra, S. K.; Tyagi, P. K.; Sharma, S. K.; Moehrle, J.; Gautam, A.; Roy, A.; Paliwal, J. K.; Kothari, M.; Saha, N.; Dash, A. P.; Bjorkman, A. Clin. Infect. Dis. 2010, 51, 684. doi:10.1086/655831
Return to citation in text: [1] [2] -
Uhlemann, A. C.; Wittlin, S.; Matile, H.; Bustamante, L. Y.; Krishna, S. Antimicrob. Agents Chemother. 2007, 51, 667. doi:10.1128/AAC.01064-06
Return to citation in text: [1] -
Gupta, A.; Singh, Y.; Srinivas, K. S.; Jain, G.; Sreekumar, V. B.; Semwal, V. P. J. Pharm. BioAllied Sci. 2010, 2, 32. doi:10.4103/0975-7406.62706
Return to citation in text: [1] -
Dong, Y.; Wittlin, S.; Sriraghavan, K.; Chollet, J.; Charman, S. A.; Charman, W. N.; Scheurer, C.; Urwyler, H.; Santo Tomas, J.; Snyder, C.; Creek, D. L.; Morizzi, J.; Koltun, M.; Matile, H.; Wang, X.; Padmanilayam, M.; Tang, Y.; Dorn, A.; Brun, R.; Vennerstrom, J. L. J. Med. Chem. 2010, 53, 481. doi:10.1021/jm901473s
Return to citation in text: [1] [2] -
O’Neill, P. M.; Amewu, R. K.; Nixon, G. L.; ElGarah, F. B.; Mungthin, M.; Chadwick, J.; Shone, A. E.; Vivas, L.; Lander, H.; Barton, V.; Muangnoicharoen, S.; Bray, P. G.; Davies, J.; Park, B. K.; Wittlin, S.; Brun, R.; Preschel, M.; Zhang, K.; Ward, S. A. Angew. Chem., Int. Ed. 2010, 49, 5693. doi:10.1002/anie.201001026
Return to citation in text: [1] [2] -
Bousejra-El Garah, F.; Wong, M. H.-L.; Amewu, R. K.; Muangnoicharoen, S.; Maggs, J. L.; Stigliani, J.-L.; Park, B. K.; Chadwick, J.; Ward, S. A.; O'Neill, P. M. J. Med. Chem. 2011, 54, 6443. doi:10.1021/jm200768h
Return to citation in text: [1] [2] -
Posner, G. H.; O’Neill, P. M. Acc. Chem. Res. 2004, 37, 397. doi:10.1021/ar020227u
Return to citation in text: [1] -
Sonnet, P.; Mullié, C. Exp. Parasitol. 2011, 128, 26. doi:10.1016/j.exppara.2011.01.018
Return to citation in text: [1] -
Coslédan, F.; Fraisse, L.; Pellet, A.; Guillou, G.; Mordmüller, M.; Kremsner, P.; Moreno, A.; Mazier, D.; Maffrand, J.-P.; Meunier, B. Proc. Natl. Acad. Sci. U. S. A. 2008, 105, 17579. doi:10.1073/pnas.0804338105
Return to citation in text: [1] [2] -
Meunier, B.; Robert, A. Acc. Chem. Res. 2010, 43, 1444. doi:10.1021/ar100070k
Return to citation in text: [1] -
Martyn, D. C.; Ramirez, A. P.; Beattie, M. J.; Cortese, J. F.; Patel, V.; Rush, M. A.; Woerpel, K. A.; Clardy, J. Bioorg. Med. Chem. Lett. 2008, 18, 6521. doi:10.1016/j.bmcl.2008.10.083
Return to citation in text: [1] [2] [3] [4] -
Schiaffo, C. E.; Rottman, M.; Wittlin, S.; Dussault, P. H. ACS Med. Chem. Lett. 2011, 2, 316.
Return to citation in text: [1] -
Wang, X.; Dong, Y.; Wittlin, S.; Creek, D.; Chollet, J.; Charman, S. A.; Santo Tomas, J.; Scheurer, C.; Snyder, C.; Vennerstrom, J. L. J. Med. Chem. 2007, 50, 5840. doi:10.1021/jm0707673
Return to citation in text: [1] [2] -
Hartwig, C. L.; Lauterwasser, E. M. W.; Mahajan, S. S.; Hoke, J. M.; Cooper, R. A.; Renslo, A. R. J. Med. Chem. 2011, 54, 8207. doi:10.1021/jm2012003
Return to citation in text: [1] [2] [3] [4] [5] [6] [7] [8] [9] [10] [11] -
Dong, Y.; Chollet, J.; Matile, H.; Charman, S. A.; Chiu, F. C. K.; Charman, W. N.; Scorneaux, B.; Urwyler, H.; Santo Tomas, J.; Scheurer, C.; Snyder, C.; Dorn, A.; Wang, X.; Karle, J. M.; Tang, Y.; Wittlin, S.; Brun, R.; Vennerstrom, J. L. J. Med. Chem. 2005, 48, 4953. doi:10.1021/jm049040u
Return to citation in text: [1] [2] [3] [4] [5] [6] -
Dong, Y.; Tan, Y.; Chollet, J.; Matile, H.; Wittlin, S.; Charman, S. A.; Charman, W. N.; Tomas, J. S.; Scheurer, C.; Snyder, C.; Scorneaux, B.; Bajpai, S.; Alexander, S. A.; Wang, X.; Padmanilayam, M.; Cheruku, S. R.; Brun, R.; Vennerstrom, J. L. Bioorg. Med. Chem. 2006, 14, 6368. doi:10.1016/j.bmc.2006.05.041
Return to citation in text: [1] [2] [3] [4] [5] [6] [7] [8] -
Padmanilayam, M.; Scorneaux, B.; Dong, Y.; Chollet, J.; Matile, H.; Charman, S. A.; Creek, D. J.; Charman, W. N.; Tomas, J. S.; Scheurer, C.; Wittlin, S.; Brun, R.; Vennerstrom, J. L. Bioorg. Med. Chem. Lett. 2006, 16, 5542. doi:10.1016/j.bmcl.2006.08.046
Return to citation in text: [1] [2] -
Yadav, G. C.; Dorwal, H. N.; Tanwar, P.; Gahlot, U. B. S. A process for the preparation of 1, 2, 4-trioxolane antimalarials. PCT Int. Appl. WO2010119425 A1, Oct 21, 2010.
Return to citation in text: [1] [2] -
Tang, Y.; Dong, Y.; Wittlin, S.; Charman, S. A.; Chollet, J.; Chiu, F. C. K.; Charman, W. N.; Matile, H.; Urwyler, H.; Dorn, A.; Bajpai, S.; Wang, X.; Padmanilayam, M.; Karle, J. M.; Brun, R.; Vennerstrom, J. L. Bioorg. Med. Chem. Lett. 2007, 17, 1260. doi:10.1016/j.bmcl.2006.12.007
Return to citation in text: [1] [2] [3] -
Tang, Y.; Wittlin, S.; Charman, S. A.; Chollet, J.; Chiu, F. C. K.; Morizzi, J.; Johnson, L. M.; Santo Tomas, J.; Scheurer, C.; Snyder, C.; Zhou, L.; Dong, Y.; Charman, W. N.; Matile, H.; Urwyler, H.; Dorn, A.; Vennerstrom, J. L. Bioorg. Med. Chem. Lett. 2010, 20, 563. doi:10.1016/j.bmcl.2009.11.088
Return to citation in text: [1] [2] [3] -
Arora, V. K.; Madan, S.; Trehan, A.; Tyagi, P. Stable dosage forms of spiro and dispiro 1,2,4-trioxolane antimalarials. PCT Int. Appl. WO 2006123314 A2, Nov 23, 2006.
Return to citation in text: [1] -
Vennerstrom, J. L.; Dong, Y.; Chollet, J.; Matile, H.; Wang, X.; Sriraghavan, K.; Charman, W. N. Spiro and dispiro 1,2,4-trioxolane antimalarials. U. S. Pat. Appl. US 20050256185 A1, Nov 17, 2005.
Return to citation in text: [1] -
Cazelles, J.; Cosledan, F.; Meunier, B.; Pellet, A. Dual molecules containing peroxy derivative, the synthesis and therapeutic applications thereof. Patent Fr. Demande FR 2862304 A1, May 20, 2005.
Return to citation in text: [1] -
Vennerstrom, J. L.; Dong, Y.; Chollet, J.; Matile, H.; Padmanilayam, H.; Tang, Y.; Charman, N. W. Spiro and dispiro 1,2,4-trioxolane antimalarials. U.S. Pat. Appl. US 20040186168 A1, Sept 23, 2004.
Return to citation in text: [1] -
O’Neill, P. M.; Stocks, P. A.; Pugh, M. D.; Araujo, N. C.; Korshin, E. E.; Bickley, J. F.; Ward, S. A.; Bray, P. G.; Pasini, E.; Davies, J.; Verissimo, E.; Bachi, M. D. Angew. Chem., Int. Ed. 2004, 43, 4193. doi:10.1002/anie.200453859
Return to citation in text: [1] [2] -
Fattorusso, C.; Persico, M.; Calcinai, B.; Cerrano, C.; Parapini, S.; Taramelli, D.; Novellino, E.; Romano, A.; Scala, F.; Fattorusso, E.; Taglialatela-Scafati, O. J. Nat. Prod. 2010, 73, 1138. doi:10.1021/np100196b
Return to citation in text: [1] -
Kawanishi, M.; Kotoku, N.; Itagaki, S.; Horii, T.; Kobayashi, M. Bioorg. Med. Chem. 2004, 12, 5297. doi:10.1016/j.bmc.2004.04.051
Return to citation in text: [1] [2] [3] -
Taglialatela-Scafati, O.; Fattorusso, E.; Romano, A.; Scala, F.; Barone, V.; Cimino, P.; Stendardo, E.; Catalanotti, B.; Persico, M.; Fattorusso, C. Org. Biomol. Chem. 2010, 8, 846. doi:10.1039/b918600j
Return to citation in text: [1] [2] [3] -
Christoffers, J.; Werner, T.; Roessle, M. Catal. Today 2007, 121, 22. doi:10.1016/j.cattod.2006.11.008
Return to citation in text: [1] [2] [3] -
Bachi, M. D.; Korshin, E. E.; Hoos, R.; Szpilman, A. M.; Ploypradith, P.; Xie, S.; Shapiro, T. A.; Posner, G. H. J. Med. Chem. 2003, 46, 2516. doi:10.1021/jm020584a
Return to citation in text: [1] [2] [3] -
Fattorusso, C.; Campiani, G.; Catalanotti, B.; Persico, M.; Basilico, N.; Parapini, S.; Taramelli, D.; Campagnuolo, C.; Fattorusso, E.; Romano, A. J. Med. Chem. 2006, 49, 7088. doi:10.1021/jm060899g
Return to citation in text: [1] -
Murakami, N.; Kawanishi, M.; Itagaki, S.; Horii, T.; Kobayashi, M. Bioorg. Med. Chem. Lett. 2002, 12, 69. doi:10.1016/S0960-894X(01)00673-4
Return to citation in text: [1] -
Murakami, N.; Kawanishi, M.; Mostaqul, H. M.; Li, J.; Itagaki, S.; Horii, T.; Kobayashi, M. Bioorg. Med. Chem. Lett. 2003, 13, 4081. doi:10.1016/j.bmcl.2003.08.073
Return to citation in text: [1] [2] [3] -
Liu, H.-H.; Zhang, Q.; Jin, H.-X.; Shen, X.; Wu, Y.-K. Chin. J. Chem. 2006, 24, 1180. doi:10.1002/cjoc.200690221
Return to citation in text: [1] -
Liu, H.-H.; Jin, H.-X.; Zhang, Q.; Wu, Y.-K.; Kim, H.-S.; Wataya, Y. Chin. J. Chem. 2005, 23, 1469. doi:10.1002/cjoc.200591469
Return to citation in text: [1] [2] -
Givelet, C.; Bernat, V.; Danel, M.; André-Barrès, C.; Vial, H. Eur. J. Org. Chem. 2007, 3095. doi:10.1002/ejoc.200700086
Return to citation in text: [1] [2] -
Rudi, A.; Talpir, R.; Kashman, Y.; Benayahu, Y.; Schleyer, M. J. Nat. Prod. 1993, 56, 2178. doi:10.1021/np50102a023
Return to citation in text: [1] [2] [3] -
Ruiz, J.; Tuccio, B.; Lauricella, R.; Maynadier, M.; Vial, H.; André-Barrès, C. Tetrahedron 2013, 69, 6709. doi:10.1016/j.tet.2013.05.099
Return to citation in text: [1] [2] -
Kamata, M.; Hagiwara, J.-i.; Hokari, T.; Suzuki, C.; Fujino, R.; Kobayashi, S.; Kim, H.-S.; Wataya, Y. Res. Chem. Intermed. 2013, 39, 127. doi:10.1007/s11164-012-0637-3
Return to citation in text: [1] [2] -
Najjar, F.; Gorrichon, L.; Baltas, M.; André-Barrès, C.; Vial, H. Org. Biomol. Chem. 2005, 3, 1612. doi:10.1039/b503402g
Return to citation in text: [1] -
Kondo, K.; Matsumoto, M.; Hatsutani, M. Patent Jpn. Kokai Tokkyo Koho JP 49061179 A, June 13, 1974.
Return to citation in text: [1] -
Bernat, V.; Saffon, N.; Maynadier, M.; Vial, H.; André-Barrès, C. Tetrahedron 2009, 65, 7372. doi:10.1016/j.tet.2009.07.030
Return to citation in text: [1] [2] [3] -
Amewu, R.; Gibbons, P.; Mukhtar, A.; Stachulski, A. V.; Ward, S. A.; Hall, C.; Rimmer, K.; Davies, J.; Vivas, L.; Bacsa, J.; Mercer, A. E.; Nixon, G.; Stocks, P. A.; O’Neill, P. M. Org. Biomol. Chem. 2010, 8, 2068. doi:10.1039/b924319d
Return to citation in text: [1] [2] -
Chauhan, S. S.; Sharma, M.; Chauhan, P. M. S. Drug News Perspect. 2010, 23, 632. doi:10.1358/dnp.2010.23.10.1468390
Return to citation in text: [1] -
Fernandez, I.; Robert, A. Org. Biomol. Chem. 2011, 9, 4098. doi:10.1039/c1ob05088e
Return to citation in text: [1] -
Singh, C.; Verma, V. P.; Naikade, N. K.; Singh, A. S.; Hassam, M.; Puri, S. K. J. Med. Chem. 2008, 51, 7581. doi:10.1021/jm801006v
Return to citation in text: [1] -
Brown, G. D. Molecules 2010, 15, 7603. doi:10.3390/molecules15117603
Return to citation in text: [1] -
Singh, C.; Sharma, U.; Saxena, G.; Puri, S. Bioorg. Med. Chem. Lett. 2007, 17, 4097. doi:10.1016/j.bmcl.2007.05.055
Return to citation in text: [1] [2] [3] [4] -
Singh, C.; Singh, A. S.; Naikade, N. K.; Verma, V. P.; Hassam, M.; Gupta, N.; Pandey, S. Synthesis 2010, 1014. doi:10.1055/s-0029-1218639
Return to citation in text: [1] [2] [3] [4] -
Hencken, C. P.; Kalinda, A. S.; Gaetano D'Angelo, J. Annu. Rep. Med. Chem. 2009, 44, 359. doi:10.1016/S0065-7743(09)04418-2
Return to citation in text: [1] [2] [3] [4] -
Šolaja, B. A.; Terzić, N.; Pocsfalvi, G.; Gerena, L.; Tinant, B.; Opsenica, D.; Milhous, W. K. J. Med. Chem. 2002, 45, 3331. doi:10.1021/jm020891g
Return to citation in text: [1] [2] [3] [4] -
Kirchhofer, C.; Vargas, M.; Braissant, O.; Dong, Y.; Wang, X.; Vennerstrom, J. L.; Keiser, J. Acta Trop. 2011, 118, 56. doi:10.1016/j.actatropica.2011.02.003
Return to citation in text: [1] [2] -
Vennerstrom, J. L.; Fu, H. N.; Ellis, W. Y.; Ager, A. L., Jr.; Wood, J. K.; Andersen, S. L.; Gerena, L.; Milhous, W. K. J. Med. Chem. 1992, 35, 3023. doi:10.1021/jm00094a015
Return to citation in text: [1] [2] -
Hamann, H.-J.; Hecht, M.; Bunge, A.; Gogol, M.; Liebscher, J. Tetrahedron Lett. 2011, 52, 107. doi:10.1016/j.tetlet.2010.10.151
Return to citation in text: [1] [2] -
Opsenica, I.; Terzić, N.; Opsenica, D.; Angelovski, G.; Lehnig, L.; Eilbracht, P.; Tinant, B.; Juranić, Z.; Smith, K. S.; Yang, Y. S.; Diaz, D. S.; Smith, P. L.; Milhous, W. K.; Doković, D.; Šolaja, B. A. J. Med. Chem. 2006, 49, 3790. doi:10.1021/jm050966r
Return to citation in text: [1] [2] -
Dong, Y.; McCullough, K. J.; Wittlin, S.; Chollet, J.; Vennerstrom, J. L. Bioorg. Med. Chem. Lett. 2010, 20, 6359. doi:10.1016/j.bmcl.2010.09.113
Return to citation in text: [1] -
Cvijetic, I. N.; Zizak, Z. P.; Stanojkovic, T. P.; Juranic, Z. D.; Terzic, N.; Opsenica, I. M.; Opsenica, D. M.; Juranic, I. O.; Drakulic, B. J. Eur. J. Med. Chem. 2010, 45, 4570. doi:10.1016/j.ejmech.2010.07.019
Return to citation in text: [1] -
Opsenica, I.; Opsenica, D.; Lanteri, C. A.; Anova, L.; Milhous, W. K.; Smith, K. S.; Solaja, B. A. J. Med. Chem. 2008, 51, 6216. doi:10.1021/jm8006905
Return to citation in text: [1] [2] -
Bhattacharjee, A. K.; Carvalho, K. A.; Opsenica, D.; Solaja, B. A. J. Serb. Chem. Soc. 2005, 70, 329. doi:10.2298/JSC0503329B
Return to citation in text: [1] -
Tonmunphean, S.; Wijitkosoom, A.; Tantirungrotechai, Y. Bioorg. Med. Chem. 2004, 12, 2005. doi:10.1016/j.bmc.2004.03.003
Return to citation in text: [1] -
Ellis, G. L.; Amewu, R.; Sabbani, S.; Stocks, P. A.; Shone, A.; Stanford, D.; Gibbons, P.; Davies, J.; Vivas, L.; Charnaud, S.; Bongard, E.; Hall, C.; Rimmer, K.; Lozanom, S.; Jesús, M.; Gargallo, D.; Ward, S. A.; O’Neill, P. M. J. Med. Chem. 2008, 51, 2170. doi:10.1021/jm701435h
Return to citation in text: [1] [2] [3] -
Liu, H.-H.; Wu, Y.-K.; Shen, X. Chin. J. Chem. 2003, 21, 875. doi:10.1002/cjoc.20030210731
Return to citation in text: [1] -
Dong, Y. Mini-Rev. Med. Chem. 2002, 2, 113.
Return to citation in text: [1] -
Opsenica, D. M.; Terzić, N.; Smith, P. L.; Yang, Y.; Anova, L.; Smith, K. S.; Solaja, B. A. Bioorg. Med. Chem. 2008, 16, 7039. doi:10.1016/j.bmc.2008.05.017
Return to citation in text: [1] [2] [3] -
Terzić, N.; Opsenica, D.; Milić, D.; Tinan, B.; Smith, K. S.; Milhous, W. K.; Šolaja, B. A. J. Med. Chem. 2007, 50, 5118. doi:10.1021/jm070684m
Return to citation in text: [1] [2] -
Pis Diez, R.; Jubert, A. H. J. Mol. Struct.: THEOCHEM 2000, 499, 85. doi:10.1016/S0166-1280(99)00281-X
Return to citation in text: [1] -
Amewu, R.; Stachulski, A. V.; Ward, S. A.; Berry, N. G.; Bray, P. G.; Davies, J.; Labat, G.; Vivas, L.; O’Neill, P. M. Org. Biomol. Chem. 2006, 4, 4431. doi:10.1039/b613565j
Return to citation in text: [1] [2] [3] -
Dong, Y.; Matile, H.; Chollet, J.; Kaminsky, R.; Wood, J. K.; Vennerstrom, J. L. J. Med. Chem. 1999, 42, 1477. doi:10.1021/jm980698f
Return to citation in text: [1] -
Kumura, N.; Furukawa, H.; Kobayashi, M.; Onyango, A. N.; Izumi, M.; Nakajima, S.; Kim, H.-S.; Wataya, Y.; Baba, N. Biosci., Biotechnol., Biochem. 2009, 73, 217. doi:10.1271/bbb.80571
Return to citation in text: [1] -
Ingram, K.; Schiaffo, C. E.; Sittiwong, W.; Benner, E.; Dussault, P. H.; Keiser, J. J. Antimicrob. Chemother. 2012, 67, 1979. doi:10.1093/jac/dks141
Return to citation in text: [1] -
Dong, Y.; Chollet, J.; Vargas, M.; Mansour, N. R.; Bickle, Q.; Alnouti, Y.; Huang, J.; Keiser, J.; Vennerstrom, J. L. Bioorg. Med. Chem. Lett. 2010, 20, 2481. doi:10.1016/j.bmcl.2010.03.001
Return to citation in text: [1] [2] -
Mohamed, A. E.-H. H.; El-Sayed, M. A.; Hegazy, M. E.; Helaly, S. E.; Esmail, A. M.; Mohamed, N. S. Rec. Nat. Prod. 2010, 4, 1.
Return to citation in text: [1] [2] -
Yang, Z.-S.; Wu, W.-M.; Li, Y.; Wu, Y.-L. Helv. Chim. Acta 2005, 88, 2865. doi:10.1002/hlca.200590229
Return to citation in text: [1] [2] -
Keiser, J.; Utzinger, J. Adv. Parasitol. 2010, 73, 197. doi:10.1016/S0065-308X(10)73008-6
Return to citation in text: [1] [2] -
Dembitsky, V.; Shkrob, I.; Hanus, L. O. Biomed. Pap. 2008, 152, 209. doi:10.5507/bp.2008.032
Return to citation in text: [1] [2] -
Laurent, S. A.-L.; Boissier, J.; Coslédan, F.; Gornitzka, H.; Robert, A.; Meunier, B. Eur. J. Org. Chem. 2008, 895. doi:10.1002/ejoc.200700975
Return to citation in text: [1] [2] -
Jung, M.; Lee, K.; Kim, H.; Park, M. Curr. Med. Chem. 2004, 11, 1265. doi:10.2174/0929867043365233
Return to citation in text: [1] [2] -
Xiao, S.-h.; Mei, J.-y.; Jiao, P.-y. Parasitol. Res. 2011, 108, 431. doi:10.1007/s00436-010-2084-7
Return to citation in text: [1] [2] -
Boissier, J.; Coslédan, F.; Robert, A.; Meunier, B. Antimicrob. Agents Chemother. 2009, 53, 4903. doi:10.1128/AAC.00640-09
Return to citation in text: [1] [2] -
Pradines, V.; Portela, J.; Boissier, J.; Cosledan, F.; Meunier, B.; Robert, A. Antimicrob. Agents Chemother. 2011, 55, 2403. doi:10.1128/AAC.00082-11
Return to citation in text: [1] [2] -
Maurya, R.; Soni, A.; Anand, D.; Ravi, M.; Raju, K. S. R.; Taneja, I.; Naikade, N. K.; Puri, S. K.; Wahajuddin, K.; Anojiya, S.; Yadav, P. P. Med. Chem. Lett. 2013, 4, 165. doi:10.1021/ml300188t
Return to citation in text: [1] [2] -
Ingram, K.; Yaremenko, I. A.; Krylov, I.; Hofer, L.; Terent'ev, A. O.; Keiser, J. J. Med. Chem. 2012, 55, 8700. doi:10.1021/jm3009184
Return to citation in text: [1] -
Keiser, J.; Brun, R.; Fried, B.; Utzinger, J. Antimicrob. Agents Chemother. 2006, 50, 803. doi:10.1128/AAC.50.2.803-805.2006
Return to citation in text: [1] -
Vennervald, B. J.; Polman, K. Parasite Immunol. 2009, 31, 686. doi:10.1111/j.1365-3024.2009.01163.x
Return to citation in text: [1] -
Yajima, A.; Gabrielli, A. F.; Montresor, A.; Engels, D. Trans. R. Soc. Trop. Med. Hyg. 2011, 105, 68. doi:10.1016/j.trstmh.2010.11.003
Return to citation in text: [1] -
Keiser, J.; Utzinger, J.; Tanner, M.; Dong, Y.; Vennerstrom, J. L. J. Antimicrob. Chemother. 2006, 58, 1193. doi:10.1093/jac/dkl408
Return to citation in text: [1] -
Cooper, P. J.; Ayre, G.; Martin, C.; Rizzo, J. A.; Ponte, E. V.; Cruz, A. A. Allergy (Oxford, U. K.) 2008, 63, 409.
Return to citation in text: [1] -
Vandemark, L. M.; Jia, T.-W.; Zhou, X.-N. Adv. Parasitol. 2010, 73, 137. doi:10.1016/S0065-308X(10)73006-2
Return to citation in text: [1] -
Keiser, J.; Utzinger, J. Trends Parasitol. 2007, 23, 555. doi:10.1016/j.pt.2007.07.012
Return to citation in text: [1] -
Brooker, S.; Akhwale, W.; Pullan, R.; Estambale, B.; Clarke, S. E.; Snow, R. W.; Hotez, P. J. Am. J. Trop. Med. Hyg. 2007, 77 (Suppl. 6), 88.
Return to citation in text: [1] -
Lv, S.; Zhang, Y.; Steinmann, P.; Zhou, X.-N.; Utzinger, J. Adv. Parasitol. 2010, 72, 351. doi:10.1016/S0065-308X(10)72012-1
Return to citation in text: [1] -
Xiao, S.-H.; Keiser, J.; Chollet, J.; Utzinger, J.; Dong, Y.; Endriss, Y.; Vennerstrom, J. L.; Tanner, M. Antimicrob. Agents Chemother. 2007, 51, 1440. doi:10.1128/AAC.01537-06
Return to citation in text: [1] -
Cooper, P. J.; Nutman, T. B. Lung Biol. Health Dis. 2002, 164, 409.
Return to citation in text: [1] -
Brooker, S. Int. J. Parasitol. 2010, 40, 1137. doi:10.1016/j.ijpara.2010.04.004
Return to citation in text: [1] -
Keiser, J.; Utzinger, J. Curr. Opin. Infect. Dis. 2007, 20, 605. doi:10.1097/QCO.0b013e3282f19ec4
Return to citation in text: [1] -
Feldmeier, H.; Chitsulo, L. Arzneim. Forsch. 1999, 49, 557.
Return to citation in text: [1] -
Thun, M. J.; Henley, S. J.; Gansler, T. Novartis Found. Symp. 2004, 256, 6. doi:10.1002/0470856734.ch2
Return to citation in text: [1] -
Niesen, A.; Barthel, A.; Kluge, R.; Köwitzsch, A.; Ströhl, D.; Schwarz, S.; Csuk, R. Arch. Pharm. Chem. Life Sci. 2009, 342, 569. doi:10.1002/ardp.200900051
Return to citation in text: [1] [2] [3] [4] -
Sawant, S. S.; Youssef, D. T. A.; Sylvester, P. W.; Wali, V.; El Sayed, K. A. Nat. Prod. Commun. 2007, 2, 117.
Return to citation in text: [1] [2] [3] [4] -
Csuk, R.; Niesen-Barthel, A.; Barthel, A.; Kluge, R.; Ströhl, D. Eur. J. Med. Chem. 2010, 45, 3840. doi:10.1016/j.ejmech.2010.05.036
Return to citation in text: [1] [2] -
Rudi, A.; Afanii, R.; Gravalos, L. G.; Aknin, M.; Gaydou, E.; Vacelet, J.; Kashman, Y. J. Nat. Prod. 2003, 66, 682. doi:10.1021/np020589a
Return to citation in text: [1] [2] -
Szpilman, A. M.; Korshin, E. E.; Rozenberg, H.; Bachi, M. D. J. Org. Chem. 2005, 70, 3618. doi:10.1021/jo050074z
Return to citation in text: [1] [2] -
Berrue, F.; Thomas, O. P.; Funel-Le Bon, C.; Reyes, F.; Amade, P. Tetrahedron 2005, 61, 11843. doi:10.1016/j.tet.2005.09.077
Return to citation in text: [1] -
Rudi, A.; Kashman, Y. J. Nat. Prod. 1993, 56, 1827. doi:10.1021/np50100a027
Return to citation in text: [1] -
Yao, G.; Steliou, K. Org. Lett. 2002, 4, 485. doi:10.1021/ol016943y
Return to citation in text: [1] [2] -
Jung, M.; Ham, J.; Song, J. Org. Lett. 2002, 4, 2763. doi:10.1021/ol026285x
Return to citation in text: [1] [2] -
Wijeratne, E. M. K.; Liu, M. X.; Kantipudi, N. B.; Brochini, C. B.; Gunatilaka, A. A. L.; Canfield, L. M. Bioorg. Med. Chem. 2006, 14, 7875. doi:10.1016/j.bmc.2006.07.057
Return to citation in text: [1] [2] [3] [4] -
Aldeco-Pérez, E.; Rudler, H.; Parlier, A.; Alvarez, C.; Apan, M. T.; Herson, P.; Toscano, A. Tetrahedron Lett. 2006, 47, 9053. doi:10.1016/j.tetlet.2006.10.093
Return to citation in text: [1] [2] [3] [4] -
Dembitsky, V. M.; Gloriozova, T. A.; Poroikov, V. V. Mini-Rev. Med. Chem. 2007, 7, 571. doi:10.2174/138955707780859396
Return to citation in text: [1] [2] -
Hang, T. T. N.; Hu'o'ng, L. M.; Ha, T. T. H.; Ha, L. M. Tap Chi Duoc Hoc 2009, 49, 47.
Return to citation in text: [1] [2] -
Ma, Y.-M.; Feng, C.-L. Youji Huaxue 2008, 28, 1697.
Return to citation in text: [1] [2] -
Dai, J.; Liu, Y.; Zhou, Y.-D.; Nagle, D. G. J. Nat. Prod. 2007, 70, 130. doi:10.1021/np0604883
Return to citation in text: [1] [2] -
Rubush, D. M.; Morges, M. A.; Rose, B. J.; Thamm, D. H.; Rovis, T. J. Am. Chem. Soc. 2012, 134, 13554. doi:10.1021/ja3052427
Return to citation in text: [1] [2] -
Dembitsky, V. M.; Gloriozova, T. A.; Poroikov, V. V. Mini-Rev. Med. Chem. 2005, 5, 319. doi:10.2174/1389557053175362
Return to citation in text: [1] [2] -
Matsugo, S.; Kayamori, N.; Konishi, T. Niigata Yakka Daigaku Kenkyu Hokoku 1986, 6, 23.
Return to citation in text: [1] [2] -
Ryden, A.-M.; Kayser, O. Top. Heterocycl. Chem. 2007, 9, 1. doi:10.1007/7081_2007_085
Return to citation in text: [1] [2] -
Roshchina, V. V. Uspekhi Sovrem. Biol. 2006, 126, 366.
Return to citation in text: [1] [2] -
Bradley, D. Drug Discovery Today 2000, 5, 44. doi:10.1016/S1359-6446(99)01453-1
Return to citation in text: [1] [2] -
Jiménez, M. S.; Garznm, S. P.; Rodríguez, A. D. J. Nat. Prod. 2003, 66, 655.
Return to citation in text: [1] -
Casteel, D. A. Nat. Prod. Rep. 1999, 16, 55. doi:10.1039/a705725c
Return to citation in text: [1] -
Casteel, D. A. Nat. Prod. Rep. 1992, 9, 289. doi:10.1039/np9920900289
Return to citation in text: [1] -
Lee, K. R. Saengyak Hakhoechi 1991, 22, 145.
Return to citation in text: [1] -
Kasozi, D. M.; Rahlfs, S.; Becker, K. Drug Discovery Infect. Dis. 2011, 2, 413.
Return to citation in text: [1] -
Denisov, E. T.; Solodova, S. L.; Denisova, T. G. Russ. Chem. Rev. 2010, 79, 981. doi:10.1070/RC2010v079n11ABEH004143
Return to citation in text: [1] -
Davies-Coleman, M. T. Stud. Nat. Prod. Chem. 2005, 32, 61. doi:10.1016/S1572-5995(05)80054-7
Return to citation in text: [1] -
Mancini, I.; Guella, G.; Defant, A. Mini-Rev. Med. Chem. 2008, 8, 1265. doi:10.2174/138955708786141007
Return to citation in text: [1] -
Lupulescu, A. Prostaglandins, Leukotrienes Essent. Fatty Acids 1996, 54, 83. doi:10.1016/S0952-3278(96)90064-2
Return to citation in text: [1] -
Ridley, R. G.; Hudson, A. T. Curr. Opin. Infect. Dis. 1998, 11, 691. doi:10.1097/00001432-199812000-00008
Return to citation in text: [1] -
Mohammed, R.; Peng, J.; Kelly, M.; Yousaf, M.; Winn, E.; Odde, S.; Bie, Z.; Xie, A.; Doerksen, R. J.; Hamann, M. T. Aust. J. Chem. 2010, 63, 877. doi:10.1071/CH09665
Return to citation in text: [1] -
Camuzat-Dedenis, B.; Provot, O.; Cointeaux, L.; Perroux, V.; Berrien, J.-F.; Bories, C.; Loiseau, P. M.; Mayrargue, J. Eur. J. Med. Chem. 2001, 36, 837. doi:10.1016/S0223-5234(01)01278-8
Return to citation in text: [1] [2] -
Howarth, J.; Wilson, D. Bioorg. Med. Chem. Lett. 2003, 13, 2013. doi:10.1016/S0960-894X(03)00326-3
Return to citation in text: [1] [2] -
Van Assche, T.; Deschacht, M.; Inocencio da Luz, R. A.; Maes, L.; Cos, P. Free Radical Biol. Med. 2011, 51, 337. doi:10.1016/j.freeradbiomed.2011.05.011
Return to citation in text: [1] [2] -
Lim, C.-W.; Kim, Y.-K.; Jang, M. S.; Park, J.-I.; Park, H.-Y. J. Fish. Sci. Technol. 2006, 9, 175.
Return to citation in text: [1] [2] [3] -
Oguri, H.; Hiruma, T.; Yamagishi, Y.; Oikawa, H.; Ishiyama, A.; Otoguro, K.; Yamada, H.; Omura, S. J. Am. Chem. Soc. 2011, 133, 7096. doi:10.1021/ja200374q
Return to citation in text: [1] [2] -
Feng, Y.-J.; Davis, R. A.; Sykes, M.; Avery, V. M.; Camp, D.; Quinn, R. J. J. Nat. Prod. 2010, 73, 716. doi:10.1021/np900535z
Return to citation in text: [1] [2] -
Macreadie, P.; Avery, T.; Greatrex, B.; Taylor, D.; Macreadie, I. Bioorg. Med. Chem. Lett. 2006, 16, 920. doi:10.1016/j.bmcl.2005.10.101
Return to citation in text: [1] -
Manzo, E.; Ciavatta, M. L.; Melck, D.; Schupp, P.; de Voogd, N.; Gavagnin, M. J. Nat. Prod. 2009, 72, 1547. doi:10.1021/np900310j
Return to citation in text: [1] -
Dembitsky, V. M. Tetrahedron 2003, 59, 4701. doi:10.1016/S0040-4020(03)00701-4
Return to citation in text: [1] -
Dembitsky, V. M.; Srebnik, M. Eurasian Chem.–Technol. J. 2002, 4, 221.
Return to citation in text: [1] -
Chen, Y.; Chilson, K.; Killday, K. B.; Harmody, D.; McCarthy, P. J.; Pomponi,, S. A.; Schimoler, R.; Selitrennikoff, C.; Wright, A. E. Cyclic peroxides as novel antifungal agents. U. S. Patent US 6521661 B1, Feb 18, 2003.
Return to citation in text: [1] -
Avery, T. D.; Macreadie, P. I.; Greatrex, B. W.; Robinson, T. V.; Taylor, D. K.; Macreadie, I. G. Bioorg. Med. Chem. 2007, 17, 36. doi:10.1016/j.bmc.2006.10.021
Return to citation in text: [1] [2] -
Dembitsky, V. M.; Srebnik, M. Prog. Lipid Res. 2002, 41, 315. doi:10.1016/S0163-7827(02)00003-6
Return to citation in text: [1] [2] -
Fattorusso, E.; Parapini, S.; Campagnuolo, C.; Basilico, N.; Taglialatela-Scafati, O.; Taramelli, D. J. Antimicrob. Chemother. 2002, 50, 883. doi:10.1093/jac/dkg008
Return to citation in text: [1] [2] -
Macreadie, I. G.; Avery, T. D.; Robinson, T. V.; Macreadie, P.; Barraclough, M.; Taylor, D. K.; Tiekink, E. R. T. Tetrahedron 2008, 64, 1225. doi:10.1016/j.tet.2007.11.071
Return to citation in text: [1] [2] -
Kishi, M.; Maeno, K.; Natsuhara, M.; Kususe, M. Mold removal agent composition. Patent Jpn. Kokai Tokkyo Koho JP 11166196 A, June 22, 1999.
Return to citation in text: [1] [2] -
Gunasekera, S. P.; Gunasekera, M.; Gunawardana, G. P.; McCarthy, P.; Burres, N. J. Nat. Prod. 1990, 53, 669. doi:10.1021/np50069a021
Return to citation in text: [1] [2] -
Phillipson, D. W.; Rinehart, K. L., Jr. J. Am. Chem. Soc. 1983, 105, 7735. doi:10.1021/ja00364a045
Return to citation in text: [1] [2] -
Rugutt, J. K.; Rugutt, K. J. Nat. Prod. Lett. 2002, 16, 107. doi:10.1080/10575630290020000
Return to citation in text: [1] [2] -
Modi, I. A.; Ghosh, P. K.; Bhardwaj, D.; Desai, N. M.; Khamar, B. M. P38 inhibitors. PCT Int. Appl. WO 2008114119 A2, Sept 25, 2008.
Return to citation in text: [1] [2] -
Kyle, D. E.; Milhous, W.; Opsenica, D. M.; Pocsfalvi, G.; Solaja, B. Mixed steroidal 1,2,4,5-tetraoxane compounds and methods of making and using thereof. PCT Int. Appl. WO 2003068736 A2, Aug 21, 2003.
Return to citation in text: [1] [2] -
Chen, L.-W.; Cheng, M.-J.; Peng, C.-F.; Chen, I.-S. Chem. Biodiversity 2010, 7, 1814. doi:10.1002/cbdv.200900227
Return to citation in text: [1] [2] -
Stratakis, M.; Orfanopoulos, M. Tetrahedron 2000, 56, 1595. doi:10.1016/S0040-4020(99)00950-3
Return to citation in text: [1] -
Clennan, E. L.; Pace, A. Tetrahedron 2005, 61, 6665. doi:10.1016/j.tet.2005.04.017
Return to citation in text: [1] -
Frimer, A. A.; Afri, M.; Baumel, S. D.; Gilinsky-Sharon, P.; Rosenthal, Z.; Gottlieb, H. E. J. Org. Chem. 2000, 65, 1807. doi:10.1021/jo991803b
Return to citation in text: [1] -
Cointeaux, L.; Berrien, J.-F.; Mayrargue, J. Tetrahedron Lett. 2002, 43, 6275. doi:10.1016/S0040-4039(02)01122-X
Return to citation in text: [1] [2] -
Adam, W.; Peters, K.; Peters, E.-M.; Schambony, S. J. Am. Chem. Soc. 2000, 122, 7610. doi:10.1021/ja001113l
Return to citation in text: [1] -
Barnier, J.-P.; Morisson, V.; Blanco, L. Synth. Commun. 2001, 31, 349. doi:10.1081/SCC-100000523
Return to citation in text: [1] -
Kirihara, M.; Kakuda, H.; Ichinose, M.; Ochiai, Y.; Takizawa, S.; Mokuya, A.; Okubo, K.; Hatano, A.; Shiro, M. Tetrahedron 2005, 61, 4831. doi:10.1016/j.tet.2005.03.033
Return to citation in text: [1] -
Kulinkovich, O. G.; Astashko, D. A.; Tyvorskii, V. I.; Ilyina, N. A. Synthesis 2001, 1453. doi:10.1055/s-2001-16089
Return to citation in text: [1] -
Wimalasena, K.; Wickman, H. B.; Mahindaratne, M. P. D. Eur. J. Org. Chem. 2001, 3811. doi:10.1002/1099-0690(200110)2001:20<3811::AID-EJOC3811>3.0.CO;2-6
Return to citation in text: [1] [2] -
Ouhamou, N.; Six, Y. Org. Biomol. Chem. 2003, 1, 3007. doi:10.1039/b306719j
Return to citation in text: [1] -
Madelaine, C.; Buzas, A. K.; Kowalska-Six, J. A.; Six, Y.; Crousse, B. Tetrahedron Lett. 2009, 50, 5367. doi:10.1016/j.tetlet.2009.07.031
Return to citation in text: [1] -
Zhao, Q.; Wong, H. N. C. Tetrahedron 2007, 63, 6296. doi:10.1016/j.tet.2007.02.121
Return to citation in text: [1] -
Ikeda, H.; Akiyama, K.; Takahashi, Y.; Nakamura, T.; Ishizaki, S.; Shiratori, Y.; Ohaku, H.; Goodman, J. L.; Houmam, A.; Wayner, D. D. M.; Tero-Kubota, S.; Miyashi, T. J. Am. Chem. Soc. 2003, 125, 9147. doi:10.1021/ja0277982
Return to citation in text: [1] -
Ikeda, H.; Hoshi, Y.; Miyashi, T. Tetrahedron Lett. 2001, 42, 8485. doi:10.1016/S0040-4039(01)01786-5
Return to citation in text: [1] -
Abe, M.; Kawanami, S.; Masuyama, A.; Hayashi, T. J. Org. Chem. 2006, 71, 6607. doi:10.1021/jo060587l
Return to citation in text: [1] -
Gbur, R. K.; Little, R. D. J. Org. Chem. 2012, 77, 2134–2141. doi:10.1021/jo300297u
Return to citation in text: [1] -
Tsubusaki, T.; Nishino, H. Tetrahedron 2009, 65, 3745. doi:10.1016/j.tet.2009.02.045
Return to citation in text: [1] [2] -
Isayama, S.; Mukaiyama, T. Chem. Lett. 1989, 573. doi:10.1246/cl.1989.573
Return to citation in text: [1] -
Isayama, S. Bull. Chem. Soc. Jpn. 1990, 63, 1305. doi:10.1246/bcsj.63.1305
Return to citation in text: [1] -
Tokuyasu, T.; Kunikawa, S.; Masuyama, A.; Nojima, M. Org. Lett. 2002, 4, 3595. doi:10.1021/ol0201299
Return to citation in text: [1] -
Tokuyasu, T.; Kunikawa, S.; McCullough, K. J.; Masuyama, A.; Nojima, M. J. Org. Chem. 2005, 70, 251. doi:10.1021/jo048359j
Return to citation in text: [1] [2] [3] -
Dai, P.; Dussault, P. H. Org. Lett. 2005, 7, 4333. doi:10.1021/ol051407h
Return to citation in text: [1] [2] [3] [4] [5] -
Barnych, B.; Vatèle, J.-M. Tetrahedron 2012, 68, 3717. doi:10.1016/j.tet.2012.03.024
Return to citation in text: [1] [2] -
Ghorai, P.; Dussault, P. H.; Hu, C. Org. Lett. 2008, 10, 2401. doi:10.1021/ol800657m
Return to citation in text: [1] [2] [3] [4] [5] -
Kumar, D. N.; Sudhakar, N.; Rao, B. V.; Kishore, K. H.; Murty, U. S. Tetrahedron Lett. 2006, 47, 771. doi:10.1016/j.tetlet.2005.11.107
Return to citation in text: [1] -
Hamann, H.-J.; Wlosnewski, A.; Greco, T.; Liebscher, J. Eur. J. Org. Chem. 2006, 2174. doi:10.1002/ejoc.200500803
Return to citation in text: [1] [2] [3] -
Stewart, S. G.; Ghisalberti, E. L.; Skelton, B. W.; Heath, C. H. Org. Biomol. Chem. 2010, 8, 3563. doi:10.1039/c003742g
Return to citation in text: [1] -
Dussault, P. H.; Lee, H.-J.; Liu, X. J. Chem. Soc., Perkin Trans. 1 2000, 3006. doi:10.1039/B001391I
Return to citation in text: [1] [2] [3] [4] [5] [6] [7] [8] -
Dussault, P. H.; Lee, I. Q.; Lee, H.-J.; Lee, R. J.; Niu, Q. J.; Schultz, J. A.; Zope, U. R. J. Org. Chem. 2000, 65, 8407. doi:10.1021/jo991714z
Return to citation in text: [1] [2] -
Zhao, Q.; Vargas, M.; Dong, Y.; Zhou, L.; Wang, X.; Sriraghavan, K.; Keiser, J.; Vennerstrom, J. L. J. Med. Chem. 2010, 53, 4223. doi:10.1021/jm100226t
Return to citation in text: [1] [2] -
Ramirez, A.; Woerpel, K. A. Org. Lett. 2005, 7, 4617. doi:10.1021/ol051703u
Return to citation in text: [1] [2] -
Dai, P.; Trullinger, T. K.; Liu, X.; Dussault, P. H. J. Org. Chem. 2006, 71, 2283. doi:10.1021/jo0522254
Return to citation in text: [1] [2] [3] -
Lu, X.; Liu, Y.; Sun, B.; Cindric, B.; Deng, L. J. Am. Chem. Soc. 2008, 130, 8134. doi:10.1021/ja802982h
Return to citation in text: [1] -
Reisinger, C. M.; Wang, X.; List, B. Angew. Chem., Int. Ed. 2008, 47, 8112. doi:10.1002/anie.200803238
Return to citation in text: [1] -
Silva, E. M. P.; Pye, R. J.; Brown, G. D.; Harwood, L. M. Eur. J. Org. Chem. 2012, 1209. doi:10.1002/ejoc.201101477
Return to citation in text: [1] [2] -
Guerra, F. M.; Zubia, E.; Ortega, M. J.; Moreno-Dorado, F. J.; Massanet, G. M. Tetrahedron 2010, 66, 157. doi:10.1016/j.tet.2009.11.026
Return to citation in text: [1] [2] -
Singh, C.; Srivastav, N. C.; Srivastava, N.; Puri, S. K. Tetrahedron Lett. 2005, 46, 2757. doi:10.1016/j.tetlet.2005.02.158
Return to citation in text: [1] -
Kishali, N.; Sahin, E.; Kara, Y. Org. Lett. 2006, 8, 1791. doi:10.1021/ol060272s
Return to citation in text: [1] -
Dussault, P. H.; Xu, C. Tetrahedron Lett. 2004, 45, 7455. doi:10.1016/j.tetlet.2004.08.059
Return to citation in text: [1] -
Criegee, R. Angew. Chem., Int. Ed. Engl. 1975, 87, 745. doi:10.1002/anie.197507451
Return to citation in text: [1] -
Geletneky, C.; Berger, S. Eur. J. Org. Chem. 1998, 1625. doi:10.1002/(SICI)1099-0690(199808)1998:8<1625::AID-EJOC1625>3.0.CO;2-L
Return to citation in text: [1] -
Tokuyasu, T.; Ito, T.; Masuyama, A.; Nojima, M. Heterocycles 2000, 53, 1293. doi:10.3987/COM-00-8874
Return to citation in text: [1] [2] [3] [4] -
Nicolaou, K. C.; Rodriguez, R. M.; Mitchell, H. J.; Suzuki, H.; Fylaktakidou, K. C.; Baudoin, O.; van Delft, F. L. Chem.–Eur. J. 2000, 6, 3095. doi:10.1002/1521-3765(20000901)6:17<3095::AID-CHEM3095>3.0.CO;2-4
Return to citation in text: [1] -
Hon, Y.-S.; Lu, L.; Chang, R.-C.; Lin, S.-W.; Sun, P.-P.; Lee, C.-F. Tetrahedron 2000, 56, 9269. doi:10.1016/S0040-4020(00)00903-0
Return to citation in text: [1] [2] [3] [4] [5] [6] [7] -
Odinokov, V. N.; Akhmetova, V. R.; Savchenko, R. G.; Bazunova, M. V.; Paramonov, E. A.; Khalilov, L. M. Russ. Chem. Bull. 2000, 49, 1103. doi:10.1007/BF02494903
Return to citation in text: [1] [2] -
Odinokov, V. N.; Akhmetova, V. R.; Bazunova, M. V.; Savchenko, R. G.; Paramonov, E. A.; Khalilov, L. M. Russ. J. Org. Chem. 2001, 37, 321. doi:10.1023/A:1012311903712
Return to citation in text: [1] -
Dussault, P. H.; Raible, J. M. Org. Lett. 2000, 2, 3377. doi:10.1021/ol006478p
Return to citation in text: [1] -
Jung, I. C. Eur. J. Org. Chem. 2001, 1899. doi:10.1002/1099-0690(200105)2001:10<1899::AID-EJOC1899>3.0.CO;2-L
Return to citation in text: [1] [2] -
Caronna, T.; Gabbiadini, S.; Mele, A.; Recupero, F. Helv. Chim. Acta 2002, 85, 1. doi:10.1002/1522-2675(200201)85:1<1::AID-HLCA1>3.0.CO;2-C
Return to citation in text: [1] -
Kiryukhin, D. P.; Barkalov, I. M.; Ismoilov, I. L. Russ. J. Appl. Chem. 2001, 74, 1740. doi:10.1023/A:1014829924249
Return to citation in text: [1] -
Chen, L.; Wiemer, D. F. J. Org. Chem. 2002, 67, 7561. doi:10.1021/jo020362k
Return to citation in text: [1] -
Rücker, G.; Manns, D.; Schenkel, E. P.; Hartmann, R.; Heinzmann, B. M. Arch. Pharm. Pharm. Med. Chem. 2003, 336, 205. doi:10.1002/ardp.200300740
Return to citation in text: [1] -
Hitchcock, P. B.; Papadopoulos, K.; Young, D. W. Org. Biomol. Chem. 2003, 1, 2670. doi:10.1039/b304607a
Return to citation in text: [1] -
Wattanasereekul, S.; Maier, M. E. Adv. Synth. Catal. 2004, 346, 855. doi:10.1002/adsc.200404004
Return to citation in text: [1] -
Schank, K.; Beck, H.; Pistorius, S. Helv. Chim. Acta 2004, 87, 2025. doi:10.1002/hlca.200490182
Return to citation in text: [1] [2] -
Laventine, D. M.; Davies, M.; Evinson, E. L.; Jenkins, P. R.; Cullis, P. M.; Fawcett, J. Tetrahedron Lett. 2005, 46, 307. doi:10.1016/j.tetlet.2004.11.032
Return to citation in text: [1] -
Laventine, D. M.; Davies, M.; Evinson, E. L.; Jenkins, P. R.; Cullis, P. M.; Garcia, M. D. Tetrahedron 2009, 65, 4766. doi:10.1016/j.tet.2009.04.011
Return to citation in text: [1] -
Wang, C.; Liu, J.; Ji, Y.; Zhao, J.; Li, L.; Zhang, H. Org. Lett. 2006, 8, 2479. doi:10.1021/ol060382z
Return to citation in text: [1] -
Schwartz, C.; Raible, J.; Mott, K.; Dussault, P. H. Org. Lett. 2006, 8, 3199. doi:10.1021/ol061001k
Return to citation in text: [1] -
Schwartz, C.; Raible, J.; Mott, K.; Dussault, P. H. Tetrahedron 2006, 62, 10747. doi:10.1016/j.tet.2006.08.092
Return to citation in text: [1] -
Schiaffo, C. E.; Dussault, P. H. J. Org. Chem. 2008, 73, 4688. doi:10.1021/jo800323x
Return to citation in text: [1] -
Percy, J. M.; Roig, R.; Singh, K. Eur. J. Org. Chem. 2009, 1058. doi:10.1002/ejoc.200801130
Return to citation in text: [1] -
Shin, H. S.; Lee, C.; Lee, J. Y.; Huh, T. S. Eur. J. Org. Chem. 2000, 335. doi:10.1002/(SICI)1099-0690(200001)2000:2<335::AID-EJOC335>3.0.CO;2-2
Return to citation in text: [1] [2] [3] [4] [5] [6] [7] [8] [9] [10] [11] [12] [13] [14] -
Park, S. H.; Lee, J. Y.; Huh, T. S. Eur. J. Org. Chem. 2001, 3083. doi:10.1002/1099-0690(200108)2001:16<3083::AID-EJOC3083>3.0.CO;2-K
Return to citation in text: [1] [2] [3] [4] [5] [6] [7] -
Kawamura, S.-i.; Yamakoshi, H.; Abe, M.; Masuyama, A.; Nojima, M. Tetrahedron 2002, 58, 891. doi:10.1016/S0040-4020(01)01183-8
Return to citation in text: [1] [2] [3] -
Griesbaum, K.; Frank, A.; McCullough, K. J. Eur. J. Org. Chem. 2006, 1978. doi:10.1002/ejoc.200500806
Return to citation in text: [1] [2] -
Griesbaum, K.; Bikem, Ö.; Huh, T. S.; Dong, Y. Liebigs Ann. 1995, 1571. doi:10.1002/jlac.1995199508217
Return to citation in text: [1] -
Araújo, N. C. P.; Barton, V.; Jones, M.; Stocks, P. A.; Ward, S. A.; Davies, J.; Bray, P. G.; Shone, A. E.; Cristiano, M. L. S.; O’Neill, P. M. Bioorg. Med. Chem. Lett. 2009, 19, 2038. doi:10.1016/j.bmcl.2009.02.013
Return to citation in text: [1] [2] -
Tang, Y.; Dong, Y.; Karle, J. M.; DiTusa, C. A.; Vennerstrom, J. L. J. Org. Chem. 2004, 69, 6470. doi:10.1021/jo040171c
Return to citation in text: [1] [2] -
Zhou, L.; Alker, A.; Ruf, A.; Wang, X.; Chiu, F. C. K.; Morizzi, J.; Charman, S. A.; Charman, W. N.; Scheurer, C.; Wittlin, S.; Dong, Y.; Hunziker, D.; Vennerstrom, J. L. Bioorg. Med. Chem. Lett. 2008, 18, 1555. doi:10.1016/j.bmcl.2008.01.087
Return to citation in text: [1] [2] -
Kamata, M.; Komatsu, K.-i.; Akaba, R. Tetrahedron Lett. 2001, 42, 9203. doi:10.1016/S0040-4039(01)01972-4
Return to citation in text: [1] -
Li, Y.; Hao, H.-D.; Zhang, Q.; Wu, Y. Org. Lett. 2009, 11, 1615. doi:10.1021/ol900262t
Return to citation in text: [1] -
Kamata, M.; Ohta, M.; Komatsu, K.-i.; Kim, H.-S.; Wataya, Y. Tetrahedron Lett. 2002, 43, 2063. doi:10.1016/S0040-4039(02)00166-1
Return to citation in text: [1] -
Parrish, J. D.; Ischay, M. A.; Lu, Z.; Guo, S.; Peters, N. R.; Yoon, T. P. Org. Lett. 2012, 14, 1640. doi:10.1021/ol300428q
Return to citation in text: [1] -
La Clair, J. J. Angew. Chem., Int. Ed. 2006, 45, 2769. doi:10.1002/anie.200504033
Return to citation in text: [1] -
Griesbeck, A. G.; Cho, M. Tetrahedron Lett. 2009, 50, 121. doi:10.1016/j.tetlet.2008.10.094
Return to citation in text: [1] -
Iwahama, T.; Sakaguchi, S.; Ishii, Y. Chem. Commun. 2000, 2317. doi:10.1039/b007182j
Return to citation in text: [1] -
Kumabe, R.; Nishino, H.; Yasutake, M.; Nguyen, V.-H.; Kurosawa, K. Tetrahedron Lett. 2001, 42, 69. doi:10.1016/S0040-4039(00)01884-0
Return to citation in text: [1] -
Christoffers, J.; Werner, T.; Frey, W.; Baro, A. Eur. J. Org. Chem. 2003, 4879. doi:10.1002/ejoc.200300439
Return to citation in text: [1] [2] -
Rössle, M.; Werner, T.; Frey, W.; Christoffers, J. Eur. J. Org. Chem. 2005, 5031. doi:10.1002/ejoc.200500487
Return to citation in text: [1] [2] -
Kumabe, R.; Nishino, H. Tetrahedron Lett. 2004, 45, 703. doi:10.1016/j.tetlet.2003.11.054
Return to citation in text: [1] -
Asahi, K.; Nishino, H. Tetrahedron 2005, 61, 11107. doi:10.1016/j.tet.2005.09.029
Return to citation in text: [1] -
Asahi, K.; Nishino, H. Tetrahedron 2008, 64, 1620. doi:10.1016/j.tet.2007.12.017
Return to citation in text: [1] -
Asahi, K.; Nishino, H. Eur. J. Org. Chem. 2008, 2404. doi:10.1002/ejoc.200701232
Return to citation in text: [1] -
Beckwith, A. L. J.; Wagner, R. D. J. Am. Chem. Soc. 1979, 101, 7099. doi:10.1021/ja00517a069
Return to citation in text: [1] -
Beckwith, A. L. J.; Wagner, R. D. J. Chem. Soc., Chem. Commun. 1980, 485. doi:10.1039/C39800000485
Return to citation in text: [1] -
Korshin, E. E.; Hoos, R.; Szpilman, A. M.; Konstantinovski, L.; Posner, G. H.; Bachi, M. D. Tetrahedron 2002, 58, 2449. doi:10.1016/S0040-4020(02)00126-6
Return to citation in text: [1] -
O’Neill, P. M.; Verissimo, E.; Ward, S. A.; Davies, J.; Korshin, E. E.; Araujo, N.; Pugh, M. D.; Cristiano, M. L. S.; Stocks, P. A.; Bachi, M. D. Bioorg. Med. Chem. Lett. 2006, 16, 2991. doi:10.1016/j.bmcl.2006.02.059
Return to citation in text: [1] -
Kim, J.; Li, H. B.; Rosenthal, A. S.; Sang, D.; Shapiro, T. A.; Bachi, M. D.; Posner, G. H. Tetrahedron 2006, 62, 4120. doi:10.1016/j.tet.2006.02.008
Return to citation in text: [1] -
Tokuyasu, T.; Kunikawa, S.; Abe, M.; Masuyama, A.; Nojima, M.; Kim, H.-S.; Begum, K.; Wataya, Y. J. Org. Chem. 2003, 68, 7361. doi:10.1021/jo030107f
Return to citation in text: [1] [2] -
Wu, J.-M.; Kunikawa, S.; Tokuyasu, T.; Masuyama, A.; Nojima, M.; Kim, H.-S.; Wataya, Y. Tetrahedron 2005, 61, 9961. doi:10.1016/j.tet.2005.08.025
Return to citation in text: [1] -
Gemma, S.; Martí, F.; Gabellieri, E.; Campiani, G.; Novellino, E.; Butini, S. Tetrahedron Lett. 2009, 50, 5719. doi:10.1016/j.tetlet.2009.07.137
Return to citation in text: [1] -
Gemma, S.; Gabellieri, E.; Coccone, S. S.; Martí, F.; Taglialatela-Scafati, O.; Novellino, E.; Campiani, G.; Butini, S. J. Org. Chem. 2010, 75, 2333. doi:10.1021/jo1001559
Return to citation in text: [1] -
Murakami, N.; Kawanishi, M.; Itagaki, S.; Horii, T.; Kobayashi, M. Tetrahedron Lett. 2001, 42, 7281. doi:10.1016/S0040-4039(01)01493-9
Return to citation in text: [1] -
Murakami, N.; Kawanishi, M.; Itagaki, S.; Horii, T.; Kobayashi, M. Bioorg. Med. Chem. Lett. 2002, 12, 69. doi:10.1016/S0960-894X(01)00673-4
Return to citation in text: [1] -
Murakami, N.; Kawanishi, M.; Itagaki, S.; Horii, T.; Kobayashi, M. Bioorg. Med. Chem. Lett. 2004, 14, 3513. doi:10.1016/j.bmcl.2004.04.086
Return to citation in text: [1] -
Jin, H.-X.; Liu, H.-H.; Zhang, Q.; Wu, Y. Tetrahedron Lett. 2005, 46, 5767. doi:10.1016/j.tetlet.2005.06.110
Return to citation in text: [1] [2] [3] -
Jin, H.-X.; Zhang, Q.; Kim, H.-S.; Wataya, Y.; Liu, H.-H.; Wu, Y. Tetrahedron 2006, 62, 7699. doi:10.1016/j.tet.2006.05.065
Return to citation in text: [1] [2] [3] -
Jin, H.-X.; Liu, H.-H.; Zhang, Q.; Wu, Y. J. Org. Chem. 2005, 70, 4240. doi:10.1021/jo050139y
Return to citation in text: [1] [2] [3] -
Li, Y.; Zhang, Q.; Wittlin, S.; Jin, H.-X.; Wu, Y. Tetrahedron 2009, 65, 6972. doi:10.1016/j.tet.2009.06.050
Return to citation in text: [1] [2] -
Xu, C.; Raible, J. M.; Dussault, P. H. Org. Lett. 2005, 7, 2509. doi:10.1021/ol050291m
Return to citation in text: [1] -
Fontana, A.; d'Ippolito, G.; D'Souza, L.; Mollo, E.; Parameswaram, P. S.; Cimino, G. J. Nat. Prod. 2001, 64, 131. doi:10.1021/np0002435
Return to citation in text: [1] -
Zhang, Q.; Li, Y.; Wu, Y.-K. Chin. J. Chem. 2007, 25, 1304. doi:10.1002/cjoc.200790242
Return to citation in text: [1] [2] -
Xu, C.; Schwartz, C.; Raible, J.; Dussault, P. H. Tetrahedron 2009, 65, 9680. doi:10.1016/j.tet.2009.09.068
Return to citation in text: [1] [2] -
Tokuyasu, T.; Masuyama, A.; Nojima, M.; McCullough, K. J. J. Org. Chem. 2000, 65, 1069. doi:10.1021/jo991499m
Return to citation in text: [1] [2] -
Tokuyasu, T.; Masuyama, A.; Nojima, M.; Kim, H.-S.; Wataya, Y. Tetrahedron Lett. 2000, 41, 3145. doi:10.1016/S0040-4039(00)00372-5
Return to citation in text: [1] [2] [3] -
Kim, H.-S.; Begum, K.; Ogura, N.; Wataya, Y.; Tokuyasu, T.; Masuyama, A.; Nojima, M.; McCullough, K. J. J. Med. Chem. 2002, 45, 4732. doi:10.1021/jm020208q
Return to citation in text: [1] -
Harris, J. R.; Waetzig, S. R.; Woerpel, K. A. Org. Lett. 2009, 11, 3290. doi:10.1021/ol901046z
Return to citation in text: [1] [2] -
O’Neill, P. M.; Rawe, S. L.; Storr, R. C.; Ward, S. A.; Posner, G. H. Tetrahedron Lett. 2005, 46, 3029. doi:10.1016/j.tetlet.2005.03.022
Return to citation in text: [1] -
Tokuyasu, T.; Masuyama, A.; Nojima, M.; McCullough, K. J.; Kim, H.-S.; Wataya, Y. Tetrahedron 2001, 57, 5979. doi:10.1016/S0040-4020(01)00557-9
Return to citation in text: [1] -
Rostami, A.; Wang, Y.; Arif, A. M.; McDonald, R.; West, F. G. Org. Lett. 2007, 9, 703. doi:10.1021/ol070053m
Return to citation in text: [1] -
Matsumoto, M.; Nasu, S.; Takeda, M.; Murakami, H.; Watanabe, N. Chem. Commun. 2000, 821. doi:10.1039/b002161j
Return to citation in text: [1] -
López, D.; Quiñoá, E.; Riguera, R. J. Org. Chem. 2000, 65, 4671. doi:10.1021/jo000288a
Return to citation in text: [1] -
Takabatake, T.; Miyazawa, T.; Hasegawa, M.; Foote, C. S. Tetrahedron Lett. 2001, 42, 987. doi:10.1016/S0040-4039(00)02216-4
Return to citation in text: [1] -
Özen, R.; Kormalı, F.; Balci, M.; Atasoy, B. Tetrahedron 2001, 57, 7529. doi:10.1016/S0040-4020(01)00702-5
Return to citation in text: [1] -
Daştan, A.; Saracoglu, N.; Balci, M. Eur. J. Org. Chem. 2001, 3519. doi:10.1002/1099-0690(200109)2001:18<3519::AID-EJOC3519>3.0.CO;2-2
Return to citation in text: [1] -
Güney, M.; Daştan, A.; Balci, M. Helv. Chim. Acta 2005, 88, 830. doi:10.1002/hlca.200590061
Return to citation in text: [1] -
Daştan, A.; Balci, M. Tetrahedron 2006, 62, 4003. doi:10.1016/j.tet.2006.02.026
Return to citation in text: [1] [2] -
Güney, M.; Ceylan, Z. C.; Daştan, A.; Balci, M. Can. J. Chem. 2005, 83, 227. doi:10.1139/v05-046
Return to citation in text: [1] -
Gu, X.; Zhang, W.; Salomon, R. G. J. Am. Chem. Soc. 2007, 129, 6088. doi:10.1021/ja0689785
Return to citation in text: [1] -
Blay, G.; Garcia, B.; Molina, E.; Pedro, J. R. Org. Lett. 2005, 7, 3291. doi:10.1021/ol0511023
Return to citation in text: [1] -
Yang, Y.-K.; Lee, S.; Tae, J. Bull. Korean Chem. Soc. 2004, 25, 1307. doi:10.5012/bkcs.2004.25.9.1307
Return to citation in text: [1] -
Yang, Y.-K.; Choi, J.-H.; Tae, J. J. Org. Chem. 2005, 70, 6995. doi:10.1021/jo050957q
Return to citation in text: [1] -
Avery, T. D.; Caiazza, D.; Culbert, J. A.; Taylor, D. K.; Tiekink, E. R. T. J. Org. Chem. 2005, 70, 8344. doi:10.1021/jo050806n
Return to citation in text: [1] -
Van der Westhuyzen, C. W.; Parkinson, C. J. S. Afr. J. Chem. 2005, 58, 41.
Return to citation in text: [1] -
Valente, P.; Avery, T. D.; Taylor, D. K.; Tiekink, E. R. T. J. Org. Chem. 2009, 74, 274. doi:10.1021/jo8020506
Return to citation in text: [1] [2] [3] -
Kao, T.-C.; Chuang, G. J.; Liao, C.-C. Angew. Chem., Int. Ed. 2008, 120, 7435–7437. doi:10.1002/anie.200802130
Return to citation in text: [1] -
Robinson, T. V.; Pedersen, D. S.; Taylor, D. K.; Tiekink, E. R. T. J. Org. Chem. 2009, 74, 5093–5096. doi:10.1021/jo900669u
Return to citation in text: [1] -
Schobert, R.; Siegfried, S.; Weingärtner, J.; Nieuwenhuyzen, M. J. Chem. Soc., Perkin Trans. 1 2001, 2009. doi:10.1039/B104227K
Return to citation in text: [1] -
Schobert, R.; Stehle, R.; Milius, W. J. Org. Chem. 2003, 68, 9827. doi:10.1021/jo030270a
Return to citation in text: [1] -
Gavrilan, M.; André-Barrès, C.; Baltas, M.; Tzedakis, T.; Gorrichon, L. Tetrahedron Lett. 2001, 42, 2465. doi:10.1016/S0040-4039(01)00199-X
Return to citation in text: [1] -
Najjar, F.; Baltas, M.; Gorrichon, L.; Moreno, Y.; Tzedakis, T.; Vial, H.; André-Barrès, C. Eur. J. Org. Chem. 2003, 3335. doi:10.1002/ejoc.200300165
Return to citation in text: [1] -
Çetin, F.; Yenil, N.; Yüceer, L. Carbohydr. Res. 2005, 340, 2583. doi:10.1016/j.carres.2005.09.006
Return to citation in text: [1] -
Margaros, I.; Montagnon, T.; Vassilikogiannakis, G. Org. Lett. 2007, 9, 5585. doi:10.1021/ol702575a
Return to citation in text: [1] -
Zvarec, O.; Avery, T. D.; Taylor, D. K. J. Org. Chem. 2010, 75, 450. doi:10.1021/jo902290g
Return to citation in text: [1] [2] -
Zvarec, O.; Avery, T. D.; Taylor, D. K.; Tiekink, E. R. T. Tetrahedron 2010, 66, 1007. doi:10.1016/j.tet.2009.11.068
Return to citation in text: [1] [2] [3] -
Robinson, T. V.; Taylor, D. K.; Tiekink, E. R. T. J. Org. Chem. 2006, 71, 7236. doi:10.1021/jo060949p
Return to citation in text: [1] [2] -
Taylor, D. K.; Avery, T. D.; Greatrex, B. W.; Tiekink, E. R. T.; Macreadie, I. G.; Macreadie, P. I.; Humphries, A. D.; Kalkanidis, M.; Fox, E. N.; Klonis, N.; Tilley, L. J. Med. Chem. 2004, 47, 1833. doi:10.1021/jm0305319
Return to citation in text: [1] -
del Pilar Crespo, M.; Avery, T. D.; Hanssen, E.; Fox, E.; Robinson, T. V.; Valente, P.; Taylor, D. K.; Tilley, L. Antimicrob. Agents Chemother. 2008, 52, 98. doi:10.1128/AAC.00609-07
Return to citation in text: [1] -
Emerzian, M. A.; Davenport, W.; Song, J.; Li, J.; Erden, I. Adv. Synth. Catal. 2009, 351, 999. doi:10.1002/adsc.200800804
Return to citation in text: [1] -
Özer, G.; Saraçoğlu, N.; Balci, M. J. Org. Chem. 2003, 68, 7009. doi:10.1021/jo0345300
Return to citation in text: [1] -
Singh, C. Tetrahedron Lett. 1990, 31, 6901. doi:10.1016/S0040-4039(00)97202-2
Return to citation in text: [1] -
Griesbeck, A. G.; El-Idreesy, T. T.; Fiege, M.; Brun, R. Org. Lett. 2002, 4, 4193. doi:10.1021/ol026916n
Return to citation in text: [1] -
Singh, C.; Gupta, N.; Puri, S. K. Bioorg. Med. Chem. Lett. 2003, 13, 3447. doi:10.1016/S0960-894X(03)00782-0
Return to citation in text: [1] -
Singh, C.; Gupta, N.; Puri, S. K. Bioorg. Med. Chem. Lett. 2002, 12, 1913. doi:10.1016/S0960-894X(02)00320-7
Return to citation in text: [1] -
Singh, C.; Malik, H.; Puri, S. K. Bioorg. Med. Chem. Lett. 2004, 14, 459. doi:10.1016/j.bmcl.2003.10.051
Return to citation in text: [1] -
Singh, C.; Malik, H.; Puri, S. K. Bioorg. Med. Chem. Lett. 2005, 15, 4484. doi:10.1016/j.bmcl.2005.07.013
Return to citation in text: [1] [2] -
Singh, C.; Malik, H. Org. Lett. 2005, 7, 5673. doi:10.1021/ol052378d
Return to citation in text: [1] -
Singh, C.; Malik, H.; Puri, S. K. Bioorg. Med. Chem. 2004, 12, 1177. doi:10.1016/j.bmc.2003.11.021
Return to citation in text: [1] -
Singh, C.; Kanchan, R.; Srivastava, N. C.; Puri, S. K. Bioorg. Med. Chem. Lett. 2006, 16, 584. doi:10.1016/j.bmcl.2005.10.044
Return to citation in text: [1] -
Singh, C.; Malik, H.; Puri, S. K. J. Med. Chem. 2006, 49, 2794. doi:10.1021/jm051130r
Return to citation in text: [1] -
Singh, C.; Verma, V. P.; Naikade, N. K.; Singh, A. S.; Hassam, M.; Puri, S. K. Bioorg. Med. Chem. Lett. 2010, 20, 4459. doi:10.1016/j.bmcl.2010.06.045
Return to citation in text: [1] -
Singh, C.; Gupta, N.; Puri, S. K. Bioorg. Med. Chem. 2004, 12, 5553. doi:10.1016/j.bmc.2004.08.005
Return to citation in text: [1] [2] -
Singh, C.; Gupta, N.; Puri, S. K. Tetrahedron Lett. 2005, 46, 205. doi:10.1016/j.tetlet.2004.11.078
Return to citation in text: [1] -
Singh, C.; Srivastava, N. C.; Puri, S. K. Bioorg. Med. Chem. 2004, 12, 5745. doi:10.1016/j.bmc.2004.08.042
Return to citation in text: [1] [2] -
Griesbeck, A. G.; El-Idreesy, T. T.; Höinck, L.-O.; Lex, J.; Brun, R. Bioorg. Med. Chem. Lett. 2005, 15, 595. doi:10.1016/j.bmcl.2004.11.043
Return to citation in text: [1] -
Bartoschek, A.; El-Idreesy, T. T.; Griesbeck, A. G.; Höinck, L.-O.; Lex, J.; Miara, C.; Neudörfl, J. M. Synthesis 2005, 2433. doi:10.1055/s-2005-872103
Return to citation in text: [1] [2] [3] -
Griesbeck, A. G.; El-Idreesy, T. T.; Lex, J. Tetrahedron 2006, 62, 10615. doi:10.1016/j.tet.2006.05.093
Return to citation in text: [1] -
Sabbani, S.; La Pensée, L.; Bacsa, J.; Hedenström, E.; O’Neill, P. M. Tetrahedron 2009, 65, 8531. doi:10.1016/j.tet.2009.08.020
Return to citation in text: [1] [2] -
Griesbeck, A. G.; Höinck, L.-O.; Neudörfl, J. M. Beilstein J. Org. Chem. 2010, 6, No. 61. doi:10.3762/bjoc.6.61
Return to citation in text: [1] -
Griesbeck, A. G.; Höinck, L.-O.; Lex, J.; Neudörfl, J.; Blunk, D.; El-Idreesy, T. T. Molecules 2008, 13, 1743. doi:10.3390/molecules13081743
Return to citation in text: [1] [2] -
Singh, C.; Kanchan, R.; Sharma, U.; Puri, S. K. J. Med. Chem. 2007, 50, 521. doi:10.1021/jm0610043
Return to citation in text: [1] -
Griesbeck, A. G.; Blunk, D.; El-Idreesy, T. T.; Raabe, A. Angew. Chem., Int. Ed. 2007, 46, 8883. doi:10.1002/anie.200701397
Return to citation in text: [1] [2] [3] -
Griesbeck, A. G.; Raabe, A. Synlett 2009, 1514. doi:10.1055/s-0029-1216740
Return to citation in text: [1] [2] -
O’Neill, P. M.; Mukhtar, A.; Ward, S. A.; Bickley, J. F.; Davies, J.; Bachi, M. D.; Stocks, P. A. Org. Lett. 2004, 6, 3035. doi:10.1021/ol0492142
Return to citation in text: [1] -
Jefford, C. W.; Velarde, J. A.; Bernardinelli, G.; Bray, D. H.; Warhurst, D. C.; Milhous, W. K. Helv. Chim. Acta 1993, 76, 2775. doi:10.1002/hlca.19930760804
Return to citation in text: [1] -
Posner, G. H.; Maxwell, J. P.; O'Dowd, H.; Krasavin, M.; Xie, S.; Shapiro, T. A. Bioorg. Med. Chem. 2000, 8, 1361. doi:10.1016/S0968-0896(00)00079-1
Return to citation in text: [1] [2] [3] [4] -
Posner, G. H.; Jeon, H. B.; Parker, M. H.; Krasavin, M.; Paik, I.-H.; Shapiro, T. A. J. Med. Chem. 2001, 44, 3054. doi:10.1021/jm0102396
Return to citation in text: [1] -
Posner, G. H.; Jeon, H. B.; Ploypradith, P.; Paik, I.-H.; Borstnik, K.; Xie, S.; Shapiro, T. A. J. Med. Chem. 2002, 45, 3824. doi:10.1021/jm020210h
Return to citation in text: [1] -
Dechy-Cabaret, O.; Benoit-Vical, F.; Loup, C.; Robert, A.; Gornitzka, H.; Bonhoure, A.; Vial, H.; Magnaval, J.-F.; Séguéla, J.-P.; Meunier, B. Chem.–Eur. J. 2004, 10, 1625. doi:10.1002/chem.200305576
Return to citation in text: [1] -
Singh, R.; Ishar, M. P. S. Tetrahedron Lett. 2003, 44, 1943. doi:10.1016/S0040-4039(03)00086-8
Return to citation in text: [1] -
Cole, K. P.; Hsung, R. P. Chem. Commun. 2005, 5784. doi:10.1039/b511338e
Return to citation in text: [1] -
Tang, Y.; Cole, K. P.; Buchanan, G. S.; Li, G.; Hsung, R. P. Org. Lett. 2009, 11, 1591. doi:10.1021/ol900237e
Return to citation in text: [1] -
Borsarelli, C. D.; Mischne, M.; La Venia, A.; Morán Vieyra, F. E. Photochem. Photobiol. 2007, 83, 1313. doi:10.1111/j.1751-1097.2007.00147.x
Return to citation in text: [1] -
O’Neil, P. M.; Pugh, M.; Davies, J.; Ward, S. A.; Park, B. K. Tetrahedron Lett. 2001, 42, 4569. doi:10.1016/S0040-4039(01)00791-2
Return to citation in text: [1] [2] -
O’Neill, P. M.; Hindley, S.; Pugh, M. D.; Davies, J.; Bray, P. G.; Park, B. K.; Kapu, D. S.; Ward, S. A.; Stocks, P. A. Tetrahedron Lett. 2003, 44, 8135. doi:10.1016/j.tetlet.2003.09.033
Return to citation in text: [1] -
Tang, Y.; Dong, Y.; Wang, X.; Sriraghavan, K.; Wood, J. K.; Vennerstrom, J. L. J. Org. Chem. 2005, 70, 5103. doi:10.1021/jo050385+
Return to citation in text: [1] [2] [3] -
Erhardt, S.; Macgregor, S. A.; McCullough, K. J.; Savill, K.; Taylor, B. J. Org. Lett. 2007, 9, 5569. doi:10.1021/ol702534d
Return to citation in text: [1] -
Sabbani, S.; Stocks, P. A.; Ellis, G. L.; Davies, J.; Hedenstrom, E.; Ward, S. A.; O’Neill, P. M. Bioorg. Med. Chem. Lett. 2008, 18, 5804. doi:10.1016/j.bmcl.2008.09.052
Return to citation in text: [1] [2] -
Ramirez, A. P.; Thomas, A. M.; Woerpel, K. A. Org. Lett. 2009, 11, 507. doi:10.1021/ol8022853
Return to citation in text: [1] -
Zhang, Q.; Jin, H.-X.; Wu, Y. Tetrahedron 2006, 62, 11627. doi:10.1016/j.tet.2006.09.061
Return to citation in text: [1] -
Zhang, Q.; Wu, Y. Tetrahedron 2007, 63, 10189. doi:10.1016/j.tet.2007.07.090
Return to citation in text: [1] -
Riveira, M. J.; La-Venia, A.; Mischne, M. P. Tetrahedron Lett. 2010, 51, 804. doi:10.1016/j.tetlet.2009.11.135
Return to citation in text: [1] -
Bellot, F.; Coslédan, F.; Vendier, L.; Brocard, J.; Meunier, B.; Robert, A. J. Med. Chem. 2010, 53, 4103. doi:10.1021/jm100117e
Return to citation in text: [1] -
Vennerstrom, J. L.; Dong, Y.; Andersen, S. L.; Ager, A. L.; Fu, H.; Miller, R. E.; Wesche, D. L.; Kyle, D. E.; Gerena, L.; Walters, S. M.; Wood, J. K.; Edwards, G.; Holme, A. D.; McLean, W. G.; Milhous, W. K. J. Med. Chem. 2000, 43, 2753. doi:10.1021/jm0000766
Return to citation in text: [1] -
McCullough, K. J.; Wood, J. K.; Bhattacharjee, A. K.; Dong, Y.; Kyle, D. E.; Milhous, W. K.; Vennerstrom, J. L. J. Med. Chem. 2000, 43, 1246. doi:10.1021/jm990530+
Return to citation in text: [1] -
Berkessel, A.; Andreae, M. R. M.; Schmickler, H.; Lex, J. Angew. Chem., Int. Ed. 2002, 41, 4481. doi:10.1002/1521-3773(20021202)41:23<4481::AID-ANIE4481>3.0.CO;2-7
Return to citation in text: [1] -
Ayala, D. A.; Romero, J. M.; Jorge, N. L.; Gómez-Vara, M. E.; Jubert, A. H.; Castro, E. A. Spectrochim. Acta, Part A: Mol. Biomol. Spectrosc. 2006, 64, 717. doi:10.1016/j.saa.2005.06.046
Return to citation in text: [1] -
Opsenica, D.; Pocsfalvi, G.; Juranić, Z.; Tinant, B.; Declercq, J.-P.; Kyle, D. E.; Milhous, W. K.; Šolaja, B. A. J. Med. Chem. 2000, 43, 3274. doi:10.1021/jm000952f
Return to citation in text: [1] [2] -
Opsenica, D.; Angelovski, G.; Pocsfalvi, G.; Juranić, Z.; Žižak, Ž.; Kyle, D.; Milhous, W. K.; Šolaja, B. A. Bioorg. Med. Chem. 2003, 11, 2761. doi:10.1016/S0968-0896(03)00224-4
Return to citation in text: [1] [2] -
Iskra, J.; Bonnet-Delpon, D.; Bégué, J. P. Tetrahedron Lett. 2003, 44, 6309. doi:10.1016/S0040-4039(03)01472-2
Return to citation in text: [1] [2] [3] -
Žmitek, K.; Stavber, S.; Zupan, M.; Bonnet-Delpon, D.; Charneau, S.; Grellier, P.; Iskra, J. Bioorg. Med. Chem. 2006, 14, 7790. doi:10.1016/j.bmc.2006.07.069
Return to citation in text: [1] -
Žmitek, K.; Stavber, S.; Zupan, M.; Bonnet-Delpon, D.; Iskra, J. Tetrahedron 2006, 62, 1479. doi:10.1016/j.tet.2005.11.022
Return to citation in text: [1] -
Atheaya, H.; Khan, S. I.; Mamgain, R.; Rawat, D. S. Bioorg. Med. Chem. Lett. 2008, 18, 1446. doi:10.1016/j.bmcl.2007.12.069
Return to citation in text: [1] -
Ghorai, P.; Dussault, P. H. Org. Lett. 2009, 11, 213. doi:10.1021/ol8023874
Return to citation in text: [1] -
Terent'ev, A. O.; Kutkin, A. V.; Starikova, Z. A.; Antipin, M. Y.; Ogibin, Y. N.; Nikishin, G. I. Synthesis 2004, 2356. doi:10.1055/s-2004-831171
Return to citation in text: [1] -
Terent'ev, A. O.; Kutkin, A. V.; Platonov, M. M.; Vorontsov, I. I.; Antipin, M. Y.; Ogibin, Y. N.; Nikishin, G. I. Russ. Chem. Bull. 2004, 53, 681. doi:10.1023/B:RUCB.0000035657.58776.cc
Return to citation in text: [1] -
Kumar, N.; Khan, S. I.; Sharma, M.; Atheaya, H.; Rawat, D. S. Bioorg. Med. Chem. Lett. 2009, 19, 1675. doi:10.1016/j.bmcl.2009.01.103
Return to citation in text: [1] -
Kumar, N.; Khan, S. I.; Beena, R.; Rajalakshmi, G.; Kumaradhas, P.; Rawat, D. S. Bioorg. Med. Chem. 2009, 17, 5632. doi:10.1016/j.bmc.2009.06.020
Return to citation in text: [1] -
Terent'ev, A. O.; Borisov, D. A.; Chernyshev, V. V.; Nikishin, G. I. J. Org. Chem. 2009, 74, 3335. doi:10.1021/jo900226b
Return to citation in text: [1] -
Terent'ev, A. O.; Yaremenko, I. A.; Vil’, V. A.; Moiseev, I. K.; Kon’kov, S. A.; Dembitsky, V. M.; Levitsky, D. O.; Nikishin, G. I. Org. Biomol. Chem. 2013, 11, 2613–2623. doi:10.1039/c3ob27239g
Return to citation in text: [1] -
Kukovinets, O. S.; Zvereva, T. I.; Kabalnova, N. N.; Kasradze, V. G.; Salimova, E. V.; Khalitova, L. R.; Abdullin, M. I.; Spirikhin, L. V. Mendeleev Commun. 2009, 19, 106. doi:10.1016/j.mencom.2009.03.019
Return to citation in text: [1] -
Dong, Y.; Vennerstrom, J. L. J. Heterocycl. Chem. 2001, 38, 463. doi:10.1002/jhet.5570380224
Return to citation in text: [1] -
Dong, Y.; Vennerstrom, J. L. J. Org. Chem. 1998, 63, 8582. doi:10.1021/jo981261i
Return to citation in text: [1] -
Song, C. E.; Lim, J. S.; Kim, S. C.; Lee, K.-J.; Chi, D. Y. Chem. Commun. 2000, 2415. doi:10.1039/b005604i
Return to citation in text: [1] -
Opsenica, D.; Pocsfalvi, G.; Milhous, W. K.; Šolaja, B. A. J. Serb. Chem. Soc. 2002, 67, 465. doi:10.2298/JSC0207465O
Return to citation in text: [1] -
Terent’ev, A. O.; Kutkin, A. V.; Platonov, M. M.; Ogibin, Y. N.; Nikishin, G. I. Tetrahedron Lett. 2003, 44, 7359. doi:10.1016/S0040-4039(03)01844-6
Return to citation in text: [1] -
Dubnikova, F.; Kosloff, R.; Almog, J.; Zeiri, Y.; Boese, R.; Itzhaky, H.; Alt, A.; Keinan, E. J. Am. Chem. Soc. 2005, 127, 1146. doi:10.1021/ja0464903
Return to citation in text: [1] -
Opsenica, I.; Opsenica, D.; Smith, K. S.; Milhous, W. K.; Šolaja, B. A. J. Med. Chem. 2008, 51, 2261. doi:10.1021/jm701417a
Return to citation in text: [1]
| 382. | Singh, C.; Gupta, N.; Puri, S. K. Tetrahedron Lett. 2005, 46, 205. doi:10.1016/j.tetlet.2004.11.078 |
| 257. | Dussault, P. H.; Lee, I. Q.; Lee, H.-J.; Lee, R. J.; Niu, Q. J.; Schultz, J. A.; Zope, U. R. J. Org. Chem. 2000, 65, 8407. doi:10.1021/jo991714z |
| 258. | Zhao, Q.; Vargas, M.; Dong, Y.; Zhou, L.; Wang, X.; Sriraghavan, K.; Keiser, J.; Vennerstrom, J. L. J. Med. Chem. 2010, 53, 4223. doi:10.1021/jm100226t |
| 256. | Dussault, P. H.; Lee, H.-J.; Liu, X. J. Chem. Soc., Perkin Trans. 1 2000, 3006. doi:10.1039/B001391I |
| 256. | Dussault, P. H.; Lee, H.-J.; Liu, X. J. Chem. Soc., Perkin Trans. 1 2000, 3006. doi:10.1039/B001391I |
| 296. | Araújo, N. C. P.; Barton, V.; Jones, M.; Stocks, P. A.; Ward, S. A.; Davies, J.; Bray, P. G.; Shone, A. E.; Cristiano, M. L. S.; O’Neill, P. M. Bioorg. Med. Chem. Lett. 2009, 19, 2038. doi:10.1016/j.bmcl.2009.02.013 |
| 255. | Stewart, S. G.; Ghisalberti, E. L.; Skelton, B. W.; Heath, C. H. Org. Biomol. Chem. 2010, 8, 3563. doi:10.1039/c003742g |
| 93. | Dong, Y.; Tan, Y.; Chollet, J.; Matile, H.; Wittlin, S.; Charman, S. A.; Charman, W. N.; Tomas, J. S.; Scheurer, C.; Snyder, C.; Scorneaux, B.; Bajpai, S.; Alexander, S. A.; Wang, X.; Padmanilayam, M.; Cheruku, S. R.; Brun, R.; Vennerstrom, J. L. Bioorg. Med. Chem. 2006, 14, 6368. doi:10.1016/j.bmc.2006.05.041 |
| 125. | Singh, C.; Sharma, U.; Saxena, G.; Puri, S. Bioorg. Med. Chem. Lett. 2007, 17, 4097. doi:10.1016/j.bmcl.2007.05.055 |
| 126. | Singh, C.; Singh, A. S.; Naikade, N. K.; Verma, V. P.; Hassam, M.; Gupta, N.; Pandey, S. Synthesis 2010, 1014. doi:10.1055/s-0029-1218639 |
| 127. | Hencken, C. P.; Kalinda, A. S.; Gaetano D'Angelo, J. Annu. Rep. Med. Chem. 2009, 44, 359. doi:10.1016/S0065-7743(09)04418-2 |
| 300. | Li, Y.; Hao, H.-D.; Zhang, Q.; Wu, Y. Org. Lett. 2009, 11, 1615. doi:10.1021/ol900262t |
| 390. | Singh, C.; Kanchan, R.; Sharma, U.; Puri, S. K. J. Med. Chem. 2007, 50, 521. doi:10.1021/jm0610043 |
| 270. | Tokuyasu, T.; Ito, T.; Masuyama, A.; Nojima, M. Heterocycles 2000, 53, 1293. doi:10.3987/COM-00-8874 |
| 389. | Griesbeck, A. G.; Höinck, L.-O.; Lex, J.; Neudörfl, J.; Blunk, D.; El-Idreesy, T. T. Molecules 2008, 13, 1743. doi:10.3390/molecules13081743 |
| 262. | Reisinger, C. M.; Wang, X.; List, B. Angew. Chem., Int. Ed. 2008, 47, 8112. doi:10.1002/anie.200803238 |
| 93. | Dong, Y.; Tan, Y.; Chollet, J.; Matile, H.; Wittlin, S.; Charman, S. A.; Charman, W. N.; Tomas, J. S.; Scheurer, C.; Snyder, C.; Scorneaux, B.; Bajpai, S.; Alexander, S. A.; Wang, X.; Padmanilayam, M.; Cheruku, S. R.; Brun, R.; Vennerstrom, J. L. Bioorg. Med. Chem. 2006, 14, 6368. doi:10.1016/j.bmc.2006.05.041 |
| 125. | Singh, C.; Sharma, U.; Saxena, G.; Puri, S. Bioorg. Med. Chem. Lett. 2007, 17, 4097. doi:10.1016/j.bmcl.2007.05.055 |
| 126. | Singh, C.; Singh, A. S.; Naikade, N. K.; Verma, V. P.; Hassam, M.; Gupta, N.; Pandey, S. Synthesis 2010, 1014. doi:10.1055/s-0029-1218639 |
| 127. | Hencken, C. P.; Kalinda, A. S.; Gaetano D'Angelo, J. Annu. Rep. Med. Chem. 2009, 44, 359. doi:10.1016/S0065-7743(09)04418-2 |
| 297. | Tang, Y.; Dong, Y.; Karle, J. M.; DiTusa, C. A.; Vennerstrom, J. L. J. Org. Chem. 2004, 69, 6470. doi:10.1021/jo040171c |
| 125. | Singh, C.; Sharma, U.; Saxena, G.; Puri, S. Bioorg. Med. Chem. Lett. 2007, 17, 4097. doi:10.1016/j.bmcl.2007.05.055 |
| 126. | Singh, C.; Singh, A. S.; Naikade, N. K.; Verma, V. P.; Hassam, M.; Gupta, N.; Pandey, S. Synthesis 2010, 1014. doi:10.1055/s-0029-1218639 |
| 127. | Hencken, C. P.; Kalinda, A. S.; Gaetano D'Angelo, J. Annu. Rep. Med. Chem. 2009, 44, 359. doi:10.1016/S0065-7743(09)04418-2 |
| 260. | Dai, P.; Trullinger, T. K.; Liu, X.; Dussault, P. H. J. Org. Chem. 2006, 71, 2283. doi:10.1021/jo0522254 |
| 298. | Zhou, L.; Alker, A.; Ruf, A.; Wang, X.; Chiu, F. C. K.; Morizzi, J.; Charman, S. A.; Charman, W. N.; Scheurer, C.; Wittlin, S.; Dong, Y.; Hunziker, D.; Vennerstrom, J. L. Bioorg. Med. Chem. Lett. 2008, 18, 1555. doi:10.1016/j.bmcl.2008.01.087 |
| 383. | Singh, C.; Srivastava, N. C.; Puri, S. K. Bioorg. Med. Chem. 2004, 12, 5745. doi:10.1016/j.bmc.2004.08.042 |
| 261. | Lu, X.; Liu, Y.; Sun, B.; Cindric, B.; Deng, L. J. Am. Chem. Soc. 2008, 130, 8134. doi:10.1021/ja802982h |
| 298. | Zhou, L.; Alker, A.; Ruf, A.; Wang, X.; Chiu, F. C. K.; Morizzi, J.; Charman, S. A.; Charman, W. N.; Scheurer, C.; Wittlin, S.; Dong, Y.; Hunziker, D.; Vennerstrom, J. L. Bioorg. Med. Chem. Lett. 2008, 18, 1555. doi:10.1016/j.bmcl.2008.01.087 |
| 383. | Singh, C.; Srivastava, N. C.; Puri, S. K. Bioorg. Med. Chem. 2004, 12, 5745. doi:10.1016/j.bmc.2004.08.042 |
| 88. | Martyn, D. C.; Ramirez, A. P.; Beattie, M. J.; Cortese, J. F.; Patel, V.; Rush, M. A.; Woerpel, K. A.; Clardy, J. Bioorg. Med. Chem. Lett. 2008, 18, 6521. doi:10.1016/j.bmcl.2008.10.083 |
| 90. | Wang, X.; Dong, Y.; Wittlin, S.; Creek, D.; Chollet, J.; Charman, S. A.; Santo Tomas, J.; Scheurer, C.; Snyder, C.; Vennerstrom, J. L. J. Med. Chem. 2007, 50, 5840. doi:10.1021/jm0707673 |
| 259. | Ramirez, A.; Woerpel, K. A. Org. Lett. 2005, 7, 4617. doi:10.1021/ol051703u |
| 299. | Kamata, M.; Komatsu, K.-i.; Akaba, R. Tetrahedron Lett. 2001, 42, 9203. doi:10.1016/S0040-4039(01)01972-4 |
| 385. | Bartoschek, A.; El-Idreesy, T. T.; Griesbeck, A. G.; Höinck, L.-O.; Lex, J.; Miara, C.; Neudörfl, J. M. Synthesis 2005, 2433. doi:10.1055/s-2005-872103 |
| 387. | Sabbani, S.; La Pensée, L.; Bacsa, J.; Hedenström, E.; O’Neill, P. M. Tetrahedron 2009, 65, 8531. doi:10.1016/j.tet.2009.08.020 |
| 388. | Griesbeck, A. G.; Höinck, L.-O.; Neudörfl, J. M. Beilstein J. Org. Chem. 2010, 6, No. 61. doi:10.3762/bjoc.6.61 |
| 260. | Dai, P.; Trullinger, T. K.; Liu, X.; Dussault, P. H. J. Org. Chem. 2006, 71, 2283. doi:10.1021/jo0522254 |
| 258. | Zhao, Q.; Vargas, M.; Dong, Y.; Zhou, L.; Wang, X.; Sriraghavan, K.; Keiser, J.; Vennerstrom, J. L. J. Med. Chem. 2010, 53, 4223. doi:10.1021/jm100226t |
| 384. | Griesbeck, A. G.; El-Idreesy, T. T.; Höinck, L.-O.; Lex, J.; Brun, R. Bioorg. Med. Chem. Lett. 2005, 15, 595. doi:10.1016/j.bmcl.2004.11.043 |
| 385. | Bartoschek, A.; El-Idreesy, T. T.; Griesbeck, A. G.; Höinck, L.-O.; Lex, J.; Miara, C.; Neudörfl, J. M. Synthesis 2005, 2433. doi:10.1055/s-2005-872103 |
| 386. | Griesbeck, A. G.; El-Idreesy, T. T.; Lex, J. Tetrahedron 2006, 62, 10615. doi:10.1016/j.tet.2006.05.093 |
| 387. | Sabbani, S.; La Pensée, L.; Bacsa, J.; Hedenström, E.; O’Neill, P. M. Tetrahedron 2009, 65, 8531. doi:10.1016/j.tet.2009.08.020 |
| 77. | Vennerstrom, J. L.; Arbe-Barnes, S.; Brun, R.; Charman, S. A.; Chiu, F. C. K.; Chollet, J.; Dong, Y.; Dorn, A.; Hunziker, D.; Matile, H.; McIntosh, K.; Padmanilayam, M.; Santo Tomas, J.; Scheurer, C.; Scorneaux, B.; Tang, Y.; Urwyler, H.; Wittlin, S.; Charman, W. N. Nature 2004, 430, 900. doi:10.1038/nature02779 |
| 78. | Valecha, N.; Looareesuwan, S.; Martensson, A.; Abdulla, S. M.; Krudsood, S.; Tangpukdee, N.; Mohanty, S.; Mishra, S. K.; Tyagi, P. K.; Sharma, S. K.; Moehrle, J.; Gautam, A.; Roy, A.; Paliwal, J. K.; Kothari, M.; Saha, N.; Dash, A. P.; Bjorkman, A. Clin. Infect. Dis. 2010, 51, 684. doi:10.1086/655831 |
| 81. | Dong, Y.; Wittlin, S.; Sriraghavan, K.; Chollet, J.; Charman, S. A.; Charman, W. N.; Scheurer, C.; Urwyler, H.; Santo Tomas, J.; Snyder, C.; Creek, D. L.; Morizzi, J.; Koltun, M.; Matile, H.; Wang, X.; Padmanilayam, M.; Tang, Y.; Dorn, A.; Brun, R.; Vennerstrom, J. L. J. Med. Chem. 2010, 53, 481. doi:10.1021/jm901473s |
| 175. | Niesen, A.; Barthel, A.; Kluge, R.; Köwitzsch, A.; Ströhl, D.; Schwarz, S.; Csuk, R. Arch. Pharm. Chem. Life Sci. 2009, 342, 569. doi:10.1002/ardp.200900051 |
| 176. | Sawant, S. S.; Youssef, D. T. A.; Sylvester, P. W.; Wali, V.; El Sayed, K. A. Nat. Prod. Commun. 2007, 2, 117. |
| 177. | Csuk, R.; Niesen-Barthel, A.; Barthel, A.; Kluge, R.; Ströhl, D. Eur. J. Med. Chem. 2010, 45, 3840. doi:10.1016/j.ejmech.2010.05.036 |
| 267. | Dussault, P. H.; Xu, C. Tetrahedron Lett. 2004, 45, 7455. doi:10.1016/j.tetlet.2004.08.059 |
| 265. | Singh, C.; Srivastav, N. C.; Srivastava, N.; Puri, S. K. Tetrahedron Lett. 2005, 46, 2757. doi:10.1016/j.tetlet.2005.02.158 |
| 266. | Kishali, N.; Sahin, E.; Kara, Y. Org. Lett. 2006, 8, 1791. doi:10.1021/ol060272s |
| 263. | Silva, E. M. P.; Pye, R. J.; Brown, G. D.; Harwood, L. M. Eur. J. Org. Chem. 2012, 1209. doi:10.1002/ejoc.201101477 |
| 264. | Guerra, F. M.; Zubia, E.; Ortega, M. J.; Moreno-Dorado, F. J.; Massanet, G. M. Tetrahedron 2010, 66, 157. doi:10.1016/j.tet.2009.11.026 |
| 308. | Rössle, M.; Werner, T.; Frey, W.; Christoffers, J. Eur. J. Org. Chem. 2005, 5031. doi:10.1002/ejoc.200500487 |
| 306. | Kumabe, R.; Nishino, H.; Yasutake, M.; Nguyen, V.-H.; Kurosawa, K. Tetrahedron Lett. 2001, 42, 69. doi:10.1016/S0040-4039(00)01884-0 |
| 305. | Iwahama, T.; Sakaguchi, S.; Ishii, Y. Chem. Commun. 2000, 2317. doi:10.1039/b007182j |
| 307. | Christoffers, J.; Werner, T.; Frey, W.; Baro, A. Eur. J. Org. Chem. 2003, 4879. doi:10.1002/ejoc.200300439 |
| 307. | Christoffers, J.; Werner, T.; Frey, W.; Baro, A. Eur. J. Org. Chem. 2003, 4879. doi:10.1002/ejoc.200300439 |
| 88. | Martyn, D. C.; Ramirez, A. P.; Beattie, M. J.; Cortese, J. F.; Patel, V.; Rush, M. A.; Woerpel, K. A.; Clardy, J. Bioorg. Med. Chem. Lett. 2008, 18, 6521. doi:10.1016/j.bmcl.2008.10.083 |
| 302. | Parrish, J. D.; Ischay, M. A.; Lu, Z.; Guo, S.; Peters, N. R.; Yoon, T. P. Org. Lett. 2012, 14, 1640. doi:10.1021/ol300428q |
| 260. | Dai, P.; Trullinger, T. K.; Liu, X.; Dussault, P. H. J. Org. Chem. 2006, 71, 2283. doi:10.1021/jo0522254 |
| 301. | Kamata, M.; Ohta, M.; Komatsu, K.-i.; Kim, H.-S.; Wataya, Y. Tetrahedron Lett. 2002, 43, 2063. doi:10.1016/S0040-4039(02)00166-1 |
| 259. | Ramirez, A.; Woerpel, K. A. Org. Lett. 2005, 7, 4617. doi:10.1021/ol051703u |
| 304. | Griesbeck, A. G.; Cho, M. Tetrahedron Lett. 2009, 50, 121. doi:10.1016/j.tetlet.2008.10.094 |
| 88. | Martyn, D. C.; Ramirez, A. P.; Beattie, M. J.; Cortese, J. F.; Patel, V.; Rush, M. A.; Woerpel, K. A.; Clardy, J. Bioorg. Med. Chem. Lett. 2008, 18, 6521. doi:10.1016/j.bmcl.2008.10.083 |
| 303. | La Clair, J. J. Angew. Chem., Int. Ed. 2006, 45, 2769. doi:10.1002/anie.200504033 |
| 309. | Kumabe, R.; Nishino, H. Tetrahedron Lett. 2004, 45, 703. doi:10.1016/j.tetlet.2003.11.054 |
| 308. | Rössle, M.; Werner, T.; Frey, W.; Christoffers, J. Eur. J. Org. Chem. 2005, 5031. doi:10.1002/ejoc.200500487 |
| 240. | Zhao, Q.; Wong, H. N. C. Tetrahedron 2007, 63, 6296. doi:10.1016/j.tet.2007.02.121 |
| 241. | Ikeda, H.; Akiyama, K.; Takahashi, Y.; Nakamura, T.; Ishizaki, S.; Shiratori, Y.; Ohaku, H.; Goodman, J. L.; Houmam, A.; Wayner, D. D. M.; Tero-Kubota, S.; Miyashi, T. J. Am. Chem. Soc. 2003, 125, 9147. doi:10.1021/ja0277982 |
| 239. | Madelaine, C.; Buzas, A. K.; Kowalska-Six, J. A.; Six, Y.; Crousse, B. Tetrahedron Lett. 2009, 50, 5367. doi:10.1016/j.tetlet.2009.07.031 |
| 248. | Tokuyasu, T.; Kunikawa, S.; Masuyama, A.; Nojima, M. Org. Lett. 2002, 4, 3595. doi:10.1021/ol0201299 |
| 102. | O’Neill, P. M.; Stocks, P. A.; Pugh, M. D.; Araujo, N. C.; Korshin, E. E.; Bickley, J. F.; Ward, S. A.; Bray, P. G.; Pasini, E.; Davies, J.; Verissimo, E.; Bachi, M. D. Angew. Chem., Int. Ed. 2004, 43, 4193. doi:10.1002/anie.200453859 |
| 316. | O’Neill, P. M.; Verissimo, E.; Ward, S. A.; Davies, J.; Korshin, E. E.; Araujo, N.; Pugh, M. D.; Cristiano, M. L. S.; Stocks, P. A.; Bachi, M. D. Bioorg. Med. Chem. Lett. 2006, 16, 2991. doi:10.1016/j.bmcl.2006.02.059 |
| 107. | Bachi, M. D.; Korshin, E. E.; Hoos, R.; Szpilman, A. M.; Ploypradith, P.; Xie, S.; Shapiro, T. A.; Posner, G. H. J. Med. Chem. 2003, 46, 2516. doi:10.1021/jm020584a |
| 179. | Szpilman, A. M.; Korshin, E. E.; Rozenberg, H.; Bachi, M. D. J. Org. Chem. 2005, 70, 3618. doi:10.1021/jo050074z |
| 315. | Korshin, E. E.; Hoos, R.; Szpilman, A. M.; Konstantinovski, L.; Posner, G. H.; Bachi, M. D. Tetrahedron 2002, 58, 2449. doi:10.1016/S0040-4020(02)00126-6 |
| 245. | Tsubusaki, T.; Nishino, H. Tetrahedron 2009, 65, 3745. doi:10.1016/j.tet.2009.02.045 |
| 249. | Tokuyasu, T.; Kunikawa, S.; McCullough, K. J.; Masuyama, A.; Nojima, M. J. Org. Chem. 2005, 70, 251. doi:10.1021/jo048359j |
| 246. | Isayama, S.; Mukaiyama, T. Chem. Lett. 1989, 573. doi:10.1246/cl.1989.573 |
| 247. | Isayama, S. Bull. Chem. Soc. Jpn. 1990, 63, 1305. doi:10.1246/bcsj.63.1305 |
| 317. | Kim, J.; Li, H. B.; Rosenthal, A. S.; Sang, D.; Shapiro, T. A.; Bachi, M. D.; Posner, G. H. Tetrahedron 2006, 62, 4120. doi:10.1016/j.tet.2006.02.008 |
| 244. | Gbur, R. K.; Little, R. D. J. Org. Chem. 2012, 77, 2134–2141. doi:10.1021/jo300297u |
| 311. | Asahi, K.; Nishino, H. Tetrahedron 2008, 64, 1620. doi:10.1016/j.tet.2007.12.017 |
| 245. | Tsubusaki, T.; Nishino, H. Tetrahedron 2009, 65, 3745. doi:10.1016/j.tet.2009.02.045 |
| 310. | Asahi, K.; Nishino, H. Tetrahedron 2005, 61, 11107. doi:10.1016/j.tet.2005.09.029 |
| 242. | Ikeda, H.; Hoshi, Y.; Miyashi, T. Tetrahedron Lett. 2001, 42, 8485. doi:10.1016/S0040-4039(01)01786-5 |
| 313. | Beckwith, A. L. J.; Wagner, R. D. J. Am. Chem. Soc. 1979, 101, 7099. doi:10.1021/ja00517a069 |
| 314. | Beckwith, A. L. J.; Wagner, R. D. J. Chem. Soc., Chem. Commun. 1980, 485. doi:10.1039/C39800000485 |
| 243. | Abe, M.; Kawanami, S.; Masuyama, A.; Hayashi, T. J. Org. Chem. 2006, 71, 6607. doi:10.1021/jo060587l |
| 312. | Asahi, K.; Nishino, H. Eur. J. Org. Chem. 2008, 2404. doi:10.1002/ejoc.200701232 |
| 251. | Barnych, B.; Vatèle, J.-M. Tetrahedron 2012, 68, 3717. doi:10.1016/j.tet.2012.03.024 |
| 249. | Tokuyasu, T.; Kunikawa, S.; McCullough, K. J.; Masuyama, A.; Nojima, M. J. Org. Chem. 2005, 70, 251. doi:10.1021/jo048359j |
| 249. | Tokuyasu, T.; Kunikawa, S.; McCullough, K. J.; Masuyama, A.; Nojima, M. J. Org. Chem. 2005, 70, 251. doi:10.1021/jo048359j |
| 254. | Hamann, H.-J.; Wlosnewski, A.; Greco, T.; Liebscher, J. Eur. J. Org. Chem. 2006, 2174. doi:10.1002/ejoc.200500803 |
| 254. | Hamann, H.-J.; Wlosnewski, A.; Greco, T.; Liebscher, J. Eur. J. Org. Chem. 2006, 2174. doi:10.1002/ejoc.200500803 |
| 252. | Ghorai, P.; Dussault, P. H.; Hu, C. Org. Lett. 2008, 10, 2401. doi:10.1021/ol800657m |
| 253. | Kumar, D. N.; Sudhakar, N.; Rao, B. V.; Kishore, K. H.; Murty, U. S. Tetrahedron Lett. 2006, 47, 771. doi:10.1016/j.tetlet.2005.11.107 |
| 252. | Ghorai, P.; Dussault, P. H.; Hu, C. Org. Lett. 2008, 10, 2401. doi:10.1021/ol800657m |
| 291. | Shin, H. S.; Lee, C.; Lee, J. Y.; Huh, T. S. Eur. J. Org. Chem. 2000, 335. doi:10.1002/(SICI)1099-0690(200001)2000:2<335::AID-EJOC335>3.0.CO;2-2 |
| 291. | Shin, H. S.; Lee, C.; Lee, J. Y.; Huh, T. S. Eur. J. Org. Chem. 2000, 335. doi:10.1002/(SICI)1099-0690(200001)2000:2<335::AID-EJOC335>3.0.CO;2-2 |
| 355. | Kao, T.-C.; Chuang, G. J.; Liao, C.-C. Angew. Chem., Int. Ed. 2008, 120, 7435–7437. doi:10.1002/anie.200802130 |
| 291. | Shin, H. S.; Lee, C.; Lee, J. Y.; Huh, T. S. Eur. J. Org. Chem. 2000, 335. doi:10.1002/(SICI)1099-0690(200001)2000:2<335::AID-EJOC335>3.0.CO;2-2 |
| 186. | Dembitsky, V. M.; Gloriozova, T. A.; Poroikov, V. V. Mini-Rev. Med. Chem. 2007, 7, 571. doi:10.2174/138955707780859396 |
| 187. | Hang, T. T. N.; Hu'o'ng, L. M.; Ha, T. T. H.; Ha, L. M. Tap Chi Duoc Hoc 2009, 49, 47. |
| 188. | Ma, Y.-M.; Feng, C.-L. Youji Huaxue 2008, 28, 1697. |
| 189. | Dai, J.; Liu, Y.; Zhou, Y.-D.; Nagle, D. G. J. Nat. Prod. 2007, 70, 130. doi:10.1021/np0604883 |
| 190. | Rubush, D. M.; Morges, M. A.; Rose, B. J.; Thamm, D. H.; Rovis, T. J. Am. Chem. Soc. 2012, 134, 13554. doi:10.1021/ja3052427 |
| 291. | Shin, H. S.; Lee, C.; Lee, J. Y.; Huh, T. S. Eur. J. Org. Chem. 2000, 335. doi:10.1002/(SICI)1099-0690(200001)2000:2<335::AID-EJOC335>3.0.CO;2-2 |
| 291. | Shin, H. S.; Lee, C.; Lee, J. Y.; Huh, T. S. Eur. J. Org. Chem. 2000, 335. doi:10.1002/(SICI)1099-0690(200001)2000:2<335::AID-EJOC335>3.0.CO;2-2 |
| 354. | Valente, P.; Avery, T. D.; Taylor, D. K.; Tiekink, E. R. T. J. Org. Chem. 2009, 74, 274. doi:10.1021/jo8020506 |
| 291. | Shin, H. S.; Lee, C.; Lee, J. Y.; Huh, T. S. Eur. J. Org. Chem. 2000, 335. doi:10.1002/(SICI)1099-0690(200001)2000:2<335::AID-EJOC335>3.0.CO;2-2 |
| 191. | Dembitsky, V. M.; Gloriozova, T. A.; Poroikov, V. V. Mini-Rev. Med. Chem. 2005, 5, 319. doi:10.2174/1389557053175362 |
| 192. | Matsugo, S.; Kayamori, N.; Konishi, T. Niigata Yakka Daigaku Kenkyu Hokoku 1986, 6, 23. |
| 193. | Ryden, A.-M.; Kayser, O. Top. Heterocycl. Chem. 2007, 9, 1. doi:10.1007/7081_2007_085 |
| 194. | Roshchina, V. V. Uspekhi Sovrem. Biol. 2006, 126, 366. |
| 195. | Bradley, D. Drug Discovery Today 2000, 5, 44. doi:10.1016/S1359-6446(99)01453-1 |
| 291. | Shin, H. S.; Lee, C.; Lee, J. Y.; Huh, T. S. Eur. J. Org. Chem. 2000, 335. doi:10.1002/(SICI)1099-0690(200001)2000:2<335::AID-EJOC335>3.0.CO;2-2 |
| 350. | Yang, Y.-K.; Lee, S.; Tae, J. Bull. Korean Chem. Soc. 2004, 25, 1307. doi:10.5012/bkcs.2004.25.9.1307 |
| 291. | Shin, H. S.; Lee, C.; Lee, J. Y.; Huh, T. S. Eur. J. Org. Chem. 2000, 335. doi:10.1002/(SICI)1099-0690(200001)2000:2<335::AID-EJOC335>3.0.CO;2-2 |
| 349. | Blay, G.; Garcia, B.; Molina, E.; Pedro, J. R. Org. Lett. 2005, 7, 3291. doi:10.1021/ol0511023 |
| 291. | Shin, H. S.; Lee, C.; Lee, J. Y.; Huh, T. S. Eur. J. Org. Chem. 2000, 335. doi:10.1002/(SICI)1099-0690(200001)2000:2<335::AID-EJOC335>3.0.CO;2-2 |
| 352. | Avery, T. D.; Caiazza, D.; Culbert, J. A.; Taylor, D. K.; Tiekink, E. R. T. J. Org. Chem. 2005, 70, 8344. doi:10.1021/jo050806n |
| 291. | Shin, H. S.; Lee, C.; Lee, J. Y.; Huh, T. S. Eur. J. Org. Chem. 2000, 335. doi:10.1002/(SICI)1099-0690(200001)2000:2<335::AID-EJOC335>3.0.CO;2-2 |
| 351. | Yang, Y.-K.; Choi, J.-H.; Tae, J. J. Org. Chem. 2005, 70, 6995. doi:10.1021/jo050957q |
| 182. | Yao, G.; Steliou, K. Org. Lett. 2002, 4, 485. doi:10.1021/ol016943y |
| 183. | Jung, M.; Ham, J.; Song, J. Org. Lett. 2002, 4, 2763. doi:10.1021/ol026285x |
| 82. | O’Neill, P. M.; Amewu, R. K.; Nixon, G. L.; ElGarah, F. B.; Mungthin, M.; Chadwick, J.; Shone, A. E.; Vivas, L.; Lander, H.; Barton, V.; Muangnoicharoen, S.; Bray, P. G.; Davies, J.; Park, B. K.; Wittlin, S.; Brun, R.; Preschel, M.; Zhang, K.; Ward, S. A. Angew. Chem., Int. Ed. 2010, 49, 5693. doi:10.1002/anie.201001026 |
| 83. | Bousejra-El Garah, F.; Wong, M. H.-L.; Amewu, R. K.; Muangnoicharoen, S.; Maggs, J. L.; Stigliani, J.-L.; Park, B. K.; Chadwick, J.; Ward, S. A.; O'Neill, P. M. J. Med. Chem. 2011, 54, 6443. doi:10.1021/jm200768h |
| 293. | Kawamura, S.-i.; Yamakoshi, H.; Abe, M.; Masuyama, A.; Nojima, M. Tetrahedron 2002, 58, 891. doi:10.1016/S0040-4020(01)01183-8 |
| 292. | Park, S. H.; Lee, J. Y.; Huh, T. S. Eur. J. Org. Chem. 2001, 3083. doi:10.1002/1099-0690(200108)2001:16<3083::AID-EJOC3083>3.0.CO;2-K |
| 362. | Margaros, I.; Montagnon, T.; Vassilikogiannakis, G. Org. Lett. 2007, 9, 5585. doi:10.1021/ol702575a |
| 292. | Park, S. H.; Lee, J. Y.; Huh, T. S. Eur. J. Org. Chem. 2001, 3083. doi:10.1002/1099-0690(200108)2001:16<3083::AID-EJOC3083>3.0.CO;2-K |
| 361. | Çetin, F.; Yenil, N.; Yüceer, L. Carbohydr. Res. 2005, 340, 2583. doi:10.1016/j.carres.2005.09.006 |
| 293. | Kawamura, S.-i.; Yamakoshi, H.; Abe, M.; Masuyama, A.; Nojima, M. Tetrahedron 2002, 58, 891. doi:10.1016/S0040-4020(01)01183-8 |
| 363. | Zvarec, O.; Avery, T. D.; Taylor, D. K. J. Org. Chem. 2010, 75, 450. doi:10.1021/jo902290g |
| 293. | Kawamura, S.-i.; Yamakoshi, H.; Abe, M.; Masuyama, A.; Nojima, M. Tetrahedron 2002, 58, 891. doi:10.1016/S0040-4020(01)01183-8 |
| 363. | Zvarec, O.; Avery, T. D.; Taylor, D. K. J. Org. Chem. 2010, 75, 450. doi:10.1021/jo902290g |
| 292. | Park, S. H.; Lee, J. Y.; Huh, T. S. Eur. J. Org. Chem. 2001, 3083. doi:10.1002/1099-0690(200108)2001:16<3083::AID-EJOC3083>3.0.CO;2-K |
| 359. | Gavrilan, M.; André-Barrès, C.; Baltas, M.; Tzedakis, T.; Gorrichon, L. Tetrahedron Lett. 2001, 42, 2465. doi:10.1016/S0040-4039(01)00199-X |
| 360. | Najjar, F.; Baltas, M.; Gorrichon, L.; Moreno, Y.; Tzedakis, T.; Vial, H.; André-Barrès, C. Eur. J. Org. Chem. 2003, 3335. doi:10.1002/ejoc.200300165 |
| 292. | Park, S. H.; Lee, J. Y.; Huh, T. S. Eur. J. Org. Chem. 2001, 3083. doi:10.1002/1099-0690(200108)2001:16<3083::AID-EJOC3083>3.0.CO;2-K |
| 113. | Givelet, C.; Bernat, V.; Danel, M.; André-Barrès, C.; Vial, H. Eur. J. Org. Chem. 2007, 3095. doi:10.1002/ejoc.200700086 |
| 114. | Rudi, A.; Talpir, R.; Kashman, Y.; Benayahu, Y.; Schleyer, M. J. Nat. Prod. 1993, 56, 2178. doi:10.1021/np50102a023 |
| 115. | Ruiz, J.; Tuccio, B.; Lauricella, R.; Maynadier, M.; Vial, H.; André-Barrès, C. Tetrahedron 2013, 69, 6709. doi:10.1016/j.tet.2013.05.099 |
| 116. | Kamata, M.; Hagiwara, J.-i.; Hokari, T.; Suzuki, C.; Fujino, R.; Kobayashi, S.; Kim, H.-S.; Wataya, Y. Res. Chem. Intermed. 2013, 39, 127. doi:10.1007/s11164-012-0637-3 |
| 292. | Park, S. H.; Lee, J. Y.; Huh, T. S. Eur. J. Org. Chem. 2001, 3083. doi:10.1002/1099-0690(200108)2001:16<3083::AID-EJOC3083>3.0.CO;2-K |
| 119. | Bernat, V.; Saffon, N.; Maynadier, M.; Vial, H.; André-Barrès, C. Tetrahedron 2009, 65, 7372. doi:10.1016/j.tet.2009.07.030 |
| 292. | Park, S. H.; Lee, J. Y.; Huh, T. S. Eur. J. Org. Chem. 2001, 3083. doi:10.1002/1099-0690(200108)2001:16<3083::AID-EJOC3083>3.0.CO;2-K |
| 119. | Bernat, V.; Saffon, N.; Maynadier, M.; Vial, H.; André-Barrès, C. Tetrahedron 2009, 65, 7372. doi:10.1016/j.tet.2009.07.030 |
| 292. | Park, S. H.; Lee, J. Y.; Huh, T. S. Eur. J. Org. Chem. 2001, 3083. doi:10.1002/1099-0690(200108)2001:16<3083::AID-EJOC3083>3.0.CO;2-K |
| 357. | Schobert, R.; Siegfried, S.; Weingärtner, J.; Nieuwenhuyzen, M. J. Chem. Soc., Perkin Trans. 1 2001, 2009. doi:10.1039/B104227K |
| 358. | Schobert, R.; Stehle, R.; Milius, W. J. Org. Chem. 2003, 68, 9827. doi:10.1021/jo030270a |
| 356. | Robinson, T. V.; Pedersen, D. S.; Taylor, D. K.; Tiekink, E. R. T. J. Org. Chem. 2009, 74, 5093–5096. doi:10.1021/jo900669u |
| 256. | Dussault, P. H.; Lee, H.-J.; Liu, X. J. Chem. Soc., Perkin Trans. 1 2000, 3006. doi:10.1039/B001391I |
| 256. | Dussault, P. H.; Lee, H.-J.; Liu, X. J. Chem. Soc., Perkin Trans. 1 2000, 3006. doi:10.1039/B001391I |
| 256. | Dussault, P. H.; Lee, H.-J.; Liu, X. J. Chem. Soc., Perkin Trans. 1 2000, 3006. doi:10.1039/B001391I |
| 256. | Dussault, P. H.; Lee, H.-J.; Liu, X. J. Chem. Soc., Perkin Trans. 1 2000, 3006. doi:10.1039/B001391I |
| 270. | Tokuyasu, T.; Ito, T.; Masuyama, A.; Nojima, M. Heterocycles 2000, 53, 1293. doi:10.3987/COM-00-8874 |
| 95. | Yadav, G. C.; Dorwal, H. N.; Tanwar, P.; Gahlot, U. B. S. A process for the preparation of 1, 2, 4-trioxolane antimalarials. PCT Int. Appl. WO2010119425 A1, Oct 21, 2010. |
| 296. | Araújo, N. C. P.; Barton, V.; Jones, M.; Stocks, P. A.; Ward, S. A.; Davies, J.; Bray, P. G.; Shone, A. E.; Cristiano, M. L. S.; O’Neill, P. M. Bioorg. Med. Chem. Lett. 2009, 19, 2038. doi:10.1016/j.bmcl.2009.02.013 |
| 218. | Avery, T. D.; Macreadie, P. I.; Greatrex, B. W.; Robinson, T. V.; Taylor, D. K.; Macreadie, I. G. Bioorg. Med. Chem. 2007, 17, 36. doi:10.1016/j.bmc.2006.10.021 |
| 219. | Dembitsky, V. M.; Srebnik, M. Prog. Lipid Res. 2002, 41, 315. doi:10.1016/S0163-7827(02)00003-6 |
| 220. | Fattorusso, E.; Parapini, S.; Campagnuolo, C.; Basilico, N.; Taglialatela-Scafati, O.; Taramelli, D. J. Antimicrob. Chemother. 2002, 50, 883. doi:10.1093/jac/dkg008 |
| 221. | Macreadie, I. G.; Avery, T. D.; Robinson, T. V.; Macreadie, P.; Barraclough, M.; Taylor, D. K.; Tiekink, E. R. T. Tetrahedron 2008, 64, 1225. doi:10.1016/j.tet.2007.11.071 |
| 222. | Kishi, M.; Maeno, K.; Natsuhara, M.; Kususe, M. Mold removal agent composition. Patent Jpn. Kokai Tokkyo Koho JP 11166196 A, June 22, 1999. |
| 223. | Gunasekera, S. P.; Gunasekera, M.; Gunawardana, G. P.; McCarthy, P.; Burres, N. J. Nat. Prod. 1990, 53, 669. doi:10.1021/np50069a021 |
| 224. | Phillipson, D. W.; Rinehart, K. L., Jr. J. Am. Chem. Soc. 1983, 105, 7735. doi:10.1021/ja00364a045 |
| 225. | Rugutt, J. K.; Rugutt, K. J. Nat. Prod. Lett. 2002, 16, 107. doi:10.1080/10575630290020000 |
| 226. | Modi, I. A.; Ghosh, P. K.; Bhardwaj, D.; Desai, N. M.; Khamar, B. M. P38 inhibitors. PCT Int. Appl. WO 2008114119 A2, Sept 25, 2008. |
| 227. | Kyle, D. E.; Milhous, W.; Opsenica, D. M.; Pocsfalvi, G.; Solaja, B. Mixed steroidal 1,2,4,5-tetraoxane compounds and methods of making and using thereof. PCT Int. Appl. WO 2003068736 A2, Aug 21, 2003. |
| 228. | Chen, L.-W.; Cheng, M.-J.; Peng, C.-F.; Chen, I.-S. Chem. Biodiversity 2010, 7, 1814. doi:10.1002/cbdv.200900227 |
| 270. | Tokuyasu, T.; Ito, T.; Masuyama, A.; Nojima, M. Heterocycles 2000, 53, 1293. doi:10.3987/COM-00-8874 |
| 91. | Hartwig, C. L.; Lauterwasser, E. M. W.; Mahajan, S. S.; Hoke, J. M.; Cooper, R. A.; Renslo, A. R. J. Med. Chem. 2011, 54, 8207. doi:10.1021/jm2012003 |
| 94. | Padmanilayam, M.; Scorneaux, B.; Dong, Y.; Chollet, J.; Matile, H.; Charman, S. A.; Creek, D. J.; Charman, W. N.; Tomas, J. S.; Scheurer, C.; Wittlin, S.; Brun, R.; Vennerstrom, J. L. Bioorg. Med. Chem. Lett. 2006, 16, 5542. doi:10.1016/j.bmcl.2006.08.046 |
| 354. | Valente, P.; Avery, T. D.; Taylor, D. K.; Tiekink, E. R. T. J. Org. Chem. 2009, 74, 274. doi:10.1021/jo8020506 |
| 365. | Robinson, T. V.; Taylor, D. K.; Tiekink, E. R. T. J. Org. Chem. 2006, 71, 7236. doi:10.1021/jo060949p |
| 92. | Dong, Y.; Chollet, J.; Matile, H.; Charman, S. A.; Chiu, F. C. K.; Charman, W. N.; Scorneaux, B.; Urwyler, H.; Santo Tomas, J.; Scheurer, C.; Snyder, C.; Dorn, A.; Wang, X.; Karle, J. M.; Tang, Y.; Wittlin, S.; Brun, R.; Vennerstrom, J. L. J. Med. Chem. 2005, 48, 4953. doi:10.1021/jm049040u |
| 93. | Dong, Y.; Tan, Y.; Chollet, J.; Matile, H.; Wittlin, S.; Charman, S. A.; Charman, W. N.; Tomas, J. S.; Scheurer, C.; Snyder, C.; Scorneaux, B.; Bajpai, S.; Alexander, S. A.; Wang, X.; Padmanilayam, M.; Cheruku, S. R.; Brun, R.; Vennerstrom, J. L. Bioorg. Med. Chem. 2006, 14, 6368. doi:10.1016/j.bmc.2006.05.041 |
| 268. | Criegee, R. Angew. Chem., Int. Ed. Engl. 1975, 87, 745. doi:10.1002/anie.197507451 |
| 269. | Geletneky, C.; Berger, S. Eur. J. Org. Chem. 1998, 1625. doi:10.1002/(SICI)1099-0690(199808)1998:8<1625::AID-EJOC1625>3.0.CO;2-L |
| 91. | Hartwig, C. L.; Lauterwasser, E. M. W.; Mahajan, S. S.; Hoke, J. M.; Cooper, R. A.; Renslo, A. R. J. Med. Chem. 2011, 54, 8207. doi:10.1021/jm2012003 |
| 92. | Dong, Y.; Chollet, J.; Matile, H.; Charman, S. A.; Chiu, F. C. K.; Charman, W. N.; Scorneaux, B.; Urwyler, H.; Santo Tomas, J.; Scheurer, C.; Snyder, C.; Dorn, A.; Wang, X.; Karle, J. M.; Tang, Y.; Wittlin, S.; Brun, R.; Vennerstrom, J. L. J. Med. Chem. 2005, 48, 4953. doi:10.1021/jm049040u |
| 93. | Dong, Y.; Tan, Y.; Chollet, J.; Matile, H.; Wittlin, S.; Charman, S. A.; Charman, W. N.; Tomas, J. S.; Scheurer, C.; Snyder, C.; Scorneaux, B.; Bajpai, S.; Alexander, S. A.; Wang, X.; Padmanilayam, M.; Cheruku, S. R.; Brun, R.; Vennerstrom, J. L. Bioorg. Med. Chem. 2006, 14, 6368. doi:10.1016/j.bmc.2006.05.041 |
| 366. | Taylor, D. K.; Avery, T. D.; Greatrex, B. W.; Tiekink, E. R. T.; Macreadie, I. G.; Macreadie, P. I.; Humphries, A. D.; Kalkanidis, M.; Fox, E. N.; Klonis, N.; Tilley, L. J. Med. Chem. 2004, 47, 1833. doi:10.1021/jm0305319 |
| 367. | del Pilar Crespo, M.; Avery, T. D.; Hanssen, E.; Fox, E.; Robinson, T. V.; Valente, P.; Taylor, D. K.; Tilley, L. Antimicrob. Agents Chemother. 2008, 52, 98. doi:10.1128/AAC.00609-07 |
| 295. | Griesbaum, K.; Bikem, Ö.; Huh, T. S.; Dong, Y. Liebigs Ann. 1995, 1571. doi:10.1002/jlac.1995199508217 |
| 364. | Zvarec, O.; Avery, T. D.; Taylor, D. K.; Tiekink, E. R. T. Tetrahedron 2010, 66, 1007. doi:10.1016/j.tet.2009.11.068 |
| 256. | Dussault, P. H.; Lee, H.-J.; Liu, X. J. Chem. Soc., Perkin Trans. 1 2000, 3006. doi:10.1039/B001391I |
| 364. | Zvarec, O.; Avery, T. D.; Taylor, D. K.; Tiekink, E. R. T. Tetrahedron 2010, 66, 1007. doi:10.1016/j.tet.2009.11.068 |
| 91. | Hartwig, C. L.; Lauterwasser, E. M. W.; Mahajan, S. S.; Hoke, J. M.; Cooper, R. A.; Renslo, A. R. J. Med. Chem. 2011, 54, 8207. doi:10.1021/jm2012003 |
| 354. | Valente, P.; Avery, T. D.; Taylor, D. K.; Tiekink, E. R. T. J. Org. Chem. 2009, 74, 274. doi:10.1021/jo8020506 |
| 365. | Robinson, T. V.; Taylor, D. K.; Tiekink, E. R. T. J. Org. Chem. 2006, 71, 7236. doi:10.1021/jo060949p |
| 256. | Dussault, P. H.; Lee, H.-J.; Liu, X. J. Chem. Soc., Perkin Trans. 1 2000, 3006. doi:10.1039/B001391I |
| 364. | Zvarec, O.; Avery, T. D.; Taylor, D. K.; Tiekink, E. R. T. Tetrahedron 2010, 66, 1007. doi:10.1016/j.tet.2009.11.068 |
| 272. | Hon, Y.-S.; Lu, L.; Chang, R.-C.; Lin, S.-W.; Sun, P.-P.; Lee, C.-F. Tetrahedron 2000, 56, 9269. doi:10.1016/S0040-4020(00)00903-0 |
| 184. | Wijeratne, E. M. K.; Liu, M. X.; Kantipudi, N. B.; Brochini, C. B.; Gunatilaka, A. A. L.; Canfield, L. M. Bioorg. Med. Chem. 2006, 14, 7875. doi:10.1016/j.bmc.2006.07.057 |
| 185. | Aldeco-Pérez, E.; Rudler, H.; Parlier, A.; Alvarez, C.; Apan, M. T.; Herson, P.; Toscano, A. Tetrahedron Lett. 2006, 47, 9053. doi:10.1016/j.tetlet.2006.10.093 |
| 271. | Nicolaou, K. C.; Rodriguez, R. M.; Mitchell, H. J.; Suzuki, H.; Fylaktakidou, K. C.; Baudoin, O.; van Delft, F. L. Chem.–Eur. J. 2000, 6, 3095. doi:10.1002/1521-3765(20000901)6:17<3095::AID-CHEM3095>3.0.CO;2-4 |
| 294. | Griesbaum, K.; Frank, A.; McCullough, K. J. Eur. J. Org. Chem. 2006, 1978. doi:10.1002/ejoc.200500806 |
| 184. | Wijeratne, E. M. K.; Liu, M. X.; Kantipudi, N. B.; Brochini, C. B.; Gunatilaka, A. A. L.; Canfield, L. M. Bioorg. Med. Chem. 2006, 14, 7875. doi:10.1016/j.bmc.2006.07.057 |
| 185. | Aldeco-Pérez, E.; Rudler, H.; Parlier, A.; Alvarez, C.; Apan, M. T.; Herson, P.; Toscano, A. Tetrahedron Lett. 2006, 47, 9053. doi:10.1016/j.tetlet.2006.10.093 |
| 272. | Hon, Y.-S.; Lu, L.; Chang, R.-C.; Lin, S.-W.; Sun, P.-P.; Lee, C.-F. Tetrahedron 2000, 56, 9269. doi:10.1016/S0040-4020(00)00903-0 |
| 294. | Griesbaum, K.; Frank, A.; McCullough, K. J. Eur. J. Org. Chem. 2006, 1978. doi:10.1002/ejoc.200500806 |
| 184. | Wijeratne, E. M. K.; Liu, M. X.; Kantipudi, N. B.; Brochini, C. B.; Gunatilaka, A. A. L.; Canfield, L. M. Bioorg. Med. Chem. 2006, 14, 7875. doi:10.1016/j.bmc.2006.07.057 |
| 185. | Aldeco-Pérez, E.; Rudler, H.; Parlier, A.; Alvarez, C.; Apan, M. T.; Herson, P.; Toscano, A. Tetrahedron Lett. 2006, 47, 9053. doi:10.1016/j.tetlet.2006.10.093 |
| 92. | Dong, Y.; Chollet, J.; Matile, H.; Charman, S. A.; Chiu, F. C. K.; Charman, W. N.; Scorneaux, B.; Urwyler, H.; Santo Tomas, J.; Scheurer, C.; Snyder, C.; Dorn, A.; Wang, X.; Karle, J. M.; Tang, Y.; Wittlin, S.; Brun, R.; Vennerstrom, J. L. J. Med. Chem. 2005, 48, 4953. doi:10.1021/jm049040u |
| 93. | Dong, Y.; Tan, Y.; Chollet, J.; Matile, H.; Wittlin, S.; Charman, S. A.; Charman, W. N.; Tomas, J. S.; Scheurer, C.; Snyder, C.; Scorneaux, B.; Bajpai, S.; Alexander, S. A.; Wang, X.; Padmanilayam, M.; Cheruku, S. R.; Brun, R.; Vennerstrom, J. L. Bioorg. Med. Chem. 2006, 14, 6368. doi:10.1016/j.bmc.2006.05.041 |
| 381. | Singh, C.; Gupta, N.; Puri, S. K. Bioorg. Med. Chem. 2004, 12, 5553. doi:10.1016/j.bmc.2004.08.005 |
| 91. | Hartwig, C. L.; Lauterwasser, E. M. W.; Mahajan, S. S.; Hoke, J. M.; Cooper, R. A.; Renslo, A. R. J. Med. Chem. 2011, 54, 8207. doi:10.1021/jm2012003 |
| 381. | Singh, C.; Gupta, N.; Puri, S. K. Bioorg. Med. Chem. 2004, 12, 5553. doi:10.1016/j.bmc.2004.08.005 |
| 96. | Tang, Y.; Dong, Y.; Wittlin, S.; Charman, S. A.; Chollet, J.; Chiu, F. C. K.; Charman, W. N.; Matile, H.; Urwyler, H.; Dorn, A.; Bajpai, S.; Wang, X.; Padmanilayam, M.; Karle, J. M.; Brun, R.; Vennerstrom, J. L. Bioorg. Med. Chem. Lett. 2007, 17, 1260. doi:10.1016/j.bmcl.2006.12.007 |
| 97. | Tang, Y.; Wittlin, S.; Charman, S. A.; Chollet, J.; Chiu, F. C. K.; Morizzi, J.; Johnson, L. M.; Santo Tomas, J.; Scheurer, C.; Snyder, C.; Zhou, L.; Dong, Y.; Charman, W. N.; Matile, H.; Urwyler, H.; Dorn, A.; Vennerstrom, J. L. Bioorg. Med. Chem. Lett. 2010, 20, 563. doi:10.1016/j.bmcl.2009.11.088 |
| 91. | Hartwig, C. L.; Lauterwasser, E. M. W.; Mahajan, S. S.; Hoke, J. M.; Cooper, R. A.; Renslo, A. R. J. Med. Chem. 2011, 54, 8207. doi:10.1021/jm2012003 |
| 373. | Singh, C.; Gupta, N.; Puri, S. K. Bioorg. Med. Chem. Lett. 2002, 12, 1913. doi:10.1016/S0960-894X(02)00320-7 |
| 91. | Hartwig, C. L.; Lauterwasser, E. M. W.; Mahajan, S. S.; Hoke, J. M.; Cooper, R. A.; Renslo, A. R. J. Med. Chem. 2011, 54, 8207. doi:10.1021/jm2012003 |
| 372. | Singh, C.; Gupta, N.; Puri, S. K. Bioorg. Med. Chem. Lett. 2003, 13, 3447. doi:10.1016/S0960-894X(03)00782-0 |
| 91. | Hartwig, C. L.; Lauterwasser, E. M. W.; Mahajan, S. S.; Hoke, J. M.; Cooper, R. A.; Renslo, A. R. J. Med. Chem. 2011, 54, 8207. doi:10.1021/jm2012003 |
| 380. | Singh, C.; Verma, V. P.; Naikade, N. K.; Singh, A. S.; Hassam, M.; Puri, S. K. Bioorg. Med. Chem. Lett. 2010, 20, 4459. doi:10.1016/j.bmcl.2010.06.045 |
| 91. | Hartwig, C. L.; Lauterwasser, E. M. W.; Mahajan, S. S.; Hoke, J. M.; Cooper, R. A.; Renslo, A. R. J. Med. Chem. 2011, 54, 8207. doi:10.1021/jm2012003 |
| 374. | Singh, C.; Malik, H.; Puri, S. K. Bioorg. Med. Chem. Lett. 2004, 14, 459. doi:10.1016/j.bmcl.2003.10.051 |
| 375. | Singh, C.; Malik, H.; Puri, S. K. Bioorg. Med. Chem. Lett. 2005, 15, 4484. doi:10.1016/j.bmcl.2005.07.013 |
| 376. | Singh, C.; Malik, H. Org. Lett. 2005, 7, 5673. doi:10.1021/ol052378d |
| 377. | Singh, C.; Malik, H.; Puri, S. K. Bioorg. Med. Chem. 2004, 12, 1177. doi:10.1016/j.bmc.2003.11.021 |
| 378. | Singh, C.; Kanchan, R.; Srivastava, N. C.; Puri, S. K. Bioorg. Med. Chem. Lett. 2006, 16, 584. doi:10.1016/j.bmcl.2005.10.044 |
| 379. | Singh, C.; Malik, H.; Puri, S. K. J. Med. Chem. 2006, 49, 2794. doi:10.1021/jm051130r |
| 91. | Hartwig, C. L.; Lauterwasser, E. M. W.; Mahajan, S. S.; Hoke, J. M.; Cooper, R. A.; Renslo, A. R. J. Med. Chem. 2011, 54, 8207. doi:10.1021/jm2012003 |
| 92. | Dong, Y.; Chollet, J.; Matile, H.; Charman, S. A.; Chiu, F. C. K.; Charman, W. N.; Scorneaux, B.; Urwyler, H.; Santo Tomas, J.; Scheurer, C.; Snyder, C.; Dorn, A.; Wang, X.; Karle, J. M.; Tang, Y.; Wittlin, S.; Brun, R.; Vennerstrom, J. L. J. Med. Chem. 2005, 48, 4953. doi:10.1021/jm049040u |
| 93. | Dong, Y.; Tan, Y.; Chollet, J.; Matile, H.; Wittlin, S.; Charman, S. A.; Charman, W. N.; Tomas, J. S.; Scheurer, C.; Snyder, C.; Scorneaux, B.; Bajpai, S.; Alexander, S. A.; Wang, X.; Padmanilayam, M.; Cheruku, S. R.; Brun, R.; Vennerstrom, J. L. Bioorg. Med. Chem. 2006, 14, 6368. doi:10.1016/j.bmc.2006.05.041 |
| 369. | Özer, G.; Saraçoğlu, N.; Balci, M. J. Org. Chem. 2003, 68, 7009. doi:10.1021/jo0345300 |
| 368. | Emerzian, M. A.; Davenport, W.; Song, J.; Li, J.; Erden, I. Adv. Synth. Catal. 2009, 351, 999. doi:10.1002/adsc.200800804 |
| 97. | Tang, Y.; Wittlin, S.; Charman, S. A.; Chollet, J.; Chiu, F. C. K.; Morizzi, J.; Johnson, L. M.; Santo Tomas, J.; Scheurer, C.; Snyder, C.; Zhou, L.; Dong, Y.; Charman, W. N.; Matile, H.; Urwyler, H.; Dorn, A.; Vennerstrom, J. L. Bioorg. Med. Chem. Lett. 2010, 20, 563. doi:10.1016/j.bmcl.2009.11.088 |
| 297. | Tang, Y.; Dong, Y.; Karle, J. M.; DiTusa, C. A.; Vennerstrom, J. L. J. Org. Chem. 2004, 69, 6470. doi:10.1021/jo040171c |
| 371. | Griesbeck, A. G.; El-Idreesy, T. T.; Fiege, M.; Brun, R. Org. Lett. 2002, 4, 4193. doi:10.1021/ol026916n |
| 91. | Hartwig, C. L.; Lauterwasser, E. M. W.; Mahajan, S. S.; Hoke, J. M.; Cooper, R. A.; Renslo, A. R. J. Med. Chem. 2011, 54, 8207. doi:10.1021/jm2012003 |
| 92. | Dong, Y.; Chollet, J.; Matile, H.; Charman, S. A.; Chiu, F. C. K.; Charman, W. N.; Scorneaux, B.; Urwyler, H.; Santo Tomas, J.; Scheurer, C.; Snyder, C.; Dorn, A.; Wang, X.; Karle, J. M.; Tang, Y.; Wittlin, S.; Brun, R.; Vennerstrom, J. L. J. Med. Chem. 2005, 48, 4953. doi:10.1021/jm049040u |
| 93. | Dong, Y.; Tan, Y.; Chollet, J.; Matile, H.; Wittlin, S.; Charman, S. A.; Charman, W. N.; Tomas, J. S.; Scheurer, C.; Snyder, C.; Scorneaux, B.; Bajpai, S.; Alexander, S. A.; Wang, X.; Padmanilayam, M.; Cheruku, S. R.; Brun, R.; Vennerstrom, J. L. Bioorg. Med. Chem. 2006, 14, 6368. doi:10.1016/j.bmc.2006.05.041 |
| 96. | Tang, Y.; Dong, Y.; Wittlin, S.; Charman, S. A.; Chollet, J.; Chiu, F. C. K.; Charman, W. N.; Matile, H.; Urwyler, H.; Dorn, A.; Bajpai, S.; Wang, X.; Padmanilayam, M.; Karle, J. M.; Brun, R.; Vennerstrom, J. L. Bioorg. Med. Chem. Lett. 2007, 17, 1260. doi:10.1016/j.bmcl.2006.12.007 |
| 370. | Singh, C. Tetrahedron Lett. 1990, 31, 6901. doi:10.1016/S0040-4039(00)97202-2 |
| 325. | Jin, H.-X.; Liu, H.-H.; Zhang, Q.; Wu, Y. Tetrahedron Lett. 2005, 46, 5767. doi:10.1016/j.tetlet.2005.06.110 |
| 326. | Jin, H.-X.; Zhang, Q.; Kim, H.-S.; Wataya, Y.; Liu, H.-H.; Wu, Y. Tetrahedron 2006, 62, 7699. doi:10.1016/j.tet.2006.05.065 |
| 327. | Jin, H.-X.; Liu, H.-H.; Zhang, Q.; Wu, Y. J. Org. Chem. 2005, 70, 4240. doi:10.1021/jo050139y |
| 104. | Kawanishi, M.; Kotoku, N.; Itagaki, S.; Horii, T.; Kobayashi, M. Bioorg. Med. Chem. 2004, 12, 5297. doi:10.1016/j.bmc.2004.04.051 |
| 105. | Taglialatela-Scafati, O.; Fattorusso, E.; Romano, A.; Scala, F.; Barone, V.; Cimino, P.; Stendardo, E.; Catalanotti, B.; Persico, M.; Fattorusso, C. Org. Biomol. Chem. 2010, 8, 846. doi:10.1039/b918600j |
| 106. | Christoffers, J.; Werner, T.; Roessle, M. Catal. Today 2007, 121, 22. doi:10.1016/j.cattod.2006.11.008 |
| 407. | Sabbani, S.; Stocks, P. A.; Ellis, G. L.; Davies, J.; Hedenstrom, E.; Ward, S. A.; O’Neill, P. M. Bioorg. Med. Chem. Lett. 2008, 18, 5804. doi:10.1016/j.bmcl.2008.09.052 |
| 329. | Xu, C.; Raible, J. M.; Dussault, P. H. Org. Lett. 2005, 7, 2509. doi:10.1021/ol050291m |
| 330. | Fontana, A.; d'Ippolito, G.; D'Souza, L.; Mollo, E.; Parameswaram, P. S.; Cimino, G. J. Nat. Prod. 2001, 64, 131. doi:10.1021/np0002435 |
| 86. | Coslédan, F.; Fraisse, L.; Pellet, A.; Guillou, G.; Mordmüller, M.; Kremsner, P.; Moreno, A.; Mazier, D.; Maffrand, J.-P.; Meunier, B. Proc. Natl. Acad. Sci. U. S. A. 2008, 105, 17579. doi:10.1073/pnas.0804338105 |
| 263. | Silva, E. M. P.; Pye, R. J.; Brown, G. D.; Harwood, L. M. Eur. J. Org. Chem. 2012, 1209. doi:10.1002/ejoc.201101477 |
| 331. | Zhang, Q.; Li, Y.; Wu, Y.-K. Chin. J. Chem. 2007, 25, 1304. doi:10.1002/cjoc.200790242 |
| 413. | Vennerstrom, J. L.; Dong, Y.; Andersen, S. L.; Ager, A. L.; Fu, H.; Miller, R. E.; Wesche, D. L.; Kyle, D. E.; Gerena, L.; Walters, S. M.; Wood, J. K.; Edwards, G.; Holme, A. D.; McLean, W. G.; Milhous, W. K. J. Med. Chem. 2000, 43, 2753. doi:10.1021/jm0000766 |
| 331. | Zhang, Q.; Li, Y.; Wu, Y.-K. Chin. J. Chem. 2007, 25, 1304. doi:10.1002/cjoc.200790242 |
| 412. | Bellot, F.; Coslédan, F.; Vendier, L.; Brocard, J.; Meunier, B.; Robert, A. J. Med. Chem. 2010, 53, 4103. doi:10.1021/jm100117e |
| 328. | Li, Y.; Zhang, Q.; Wittlin, S.; Jin, H.-X.; Wu, Y. Tetrahedron 2009, 65, 6972. doi:10.1016/j.tet.2009.06.050 |
| 409. | Zhang, Q.; Jin, H.-X.; Wu, Y. Tetrahedron 2006, 62, 11627. doi:10.1016/j.tet.2006.09.061 |
| 410. | Zhang, Q.; Wu, Y. Tetrahedron 2007, 63, 10189. doi:10.1016/j.tet.2007.07.090 |
| 325. | Jin, H.-X.; Liu, H.-H.; Zhang, Q.; Wu, Y. Tetrahedron Lett. 2005, 46, 5767. doi:10.1016/j.tetlet.2005.06.110 |
| 326. | Jin, H.-X.; Zhang, Q.; Kim, H.-S.; Wataya, Y.; Liu, H.-H.; Wu, Y. Tetrahedron 2006, 62, 7699. doi:10.1016/j.tet.2006.05.065 |
| 327. | Jin, H.-X.; Liu, H.-H.; Zhang, Q.; Wu, Y. J. Org. Chem. 2005, 70, 4240. doi:10.1021/jo050139y |
| 408. | Ramirez, A. P.; Thomas, A. M.; Woerpel, K. A. Org. Lett. 2009, 11, 507. doi:10.1021/ol8022853 |
| 264. | Guerra, F. M.; Zubia, E.; Ortega, M. J.; Moreno-Dorado, F. J.; Massanet, G. M. Tetrahedron 2010, 66, 157. doi:10.1016/j.tet.2009.11.026 |
| 375. | Singh, C.; Malik, H.; Puri, S. K. Bioorg. Med. Chem. Lett. 2005, 15, 4484. doi:10.1016/j.bmcl.2005.07.013 |
| 328. | Li, Y.; Zhang, Q.; Wittlin, S.; Jin, H.-X.; Wu, Y. Tetrahedron 2009, 65, 6972. doi:10.1016/j.tet.2009.06.050 |
| 411. | Riveira, M. J.; La-Venia, A.; Mischne, M. P. Tetrahedron Lett. 2010, 51, 804. doi:10.1016/j.tetlet.2009.11.135 |
| 325. | Jin, H.-X.; Liu, H.-H.; Zhang, Q.; Wu, Y. Tetrahedron Lett. 2005, 46, 5767. doi:10.1016/j.tetlet.2005.06.110 |
| 326. | Jin, H.-X.; Zhang, Q.; Kim, H.-S.; Wataya, Y.; Liu, H.-H.; Wu, Y. Tetrahedron 2006, 62, 7699. doi:10.1016/j.tet.2006.05.065 |
| 327. | Jin, H.-X.; Liu, H.-H.; Zhang, Q.; Wu, Y. J. Org. Chem. 2005, 70, 4240. doi:10.1021/jo050139y |
| 176. | Sawant, S. S.; Youssef, D. T. A.; Sylvester, P. W.; Wali, V.; El Sayed, K. A. Nat. Prod. Commun. 2007, 2, 117. |
| 175. | Niesen, A.; Barthel, A.; Kluge, R.; Köwitzsch, A.; Ströhl, D.; Schwarz, S.; Csuk, R. Arch. Pharm. Chem. Life Sci. 2009, 342, 569. doi:10.1002/ardp.200900051 |
| 208. | Howarth, J.; Wilson, D. Bioorg. Med. Chem. Lett. 2003, 13, 2013. doi:10.1016/S0960-894X(03)00326-3 |
| 209. | Van Assche, T.; Deschacht, M.; Inocencio da Luz, R. A.; Maes, L.; Cos, P. Free Radical Biol. Med. 2011, 51, 337. doi:10.1016/j.freeradbiomed.2011.05.011 |
| 210. | Lim, C.-W.; Kim, Y.-K.; Jang, M. S.; Park, J.-I.; Park, H.-Y. J. Fish. Sci. Technol. 2006, 9, 175. |
| 211. | Oguri, H.; Hiruma, T.; Yamagishi, Y.; Oikawa, H.; Ishiyama, A.; Otoguro, K.; Yamada, H.; Omura, S. J. Am. Chem. Soc. 2011, 133, 7096. doi:10.1021/ja200374q |
| 212. | Feng, Y.-J.; Davis, R. A.; Sykes, M.; Avery, V. M.; Camp, D.; Quinn, R. J. J. Nat. Prod. 2010, 73, 716. doi:10.1021/np900535z |
| 273. | Odinokov, V. N.; Akhmetova, V. R.; Savchenko, R. G.; Bazunova, M. V.; Paramonov, E. A.; Khalilov, L. M. Russ. Chem. Bull. 2000, 49, 1103. doi:10.1007/BF02494903 |
| 336. | Harris, J. R.; Waetzig, S. R.; Woerpel, K. A. Org. Lett. 2009, 11, 3290. doi:10.1021/ol901046z |
| 420. | Žmitek, K.; Stavber, S.; Zupan, M.; Bonnet-Delpon, D.; Charneau, S.; Grellier, P.; Iskra, J. Bioorg. Med. Chem. 2006, 14, 7790. doi:10.1016/j.bmc.2006.07.069 |
| 273. | Odinokov, V. N.; Akhmetova, V. R.; Savchenko, R. G.; Bazunova, M. V.; Paramonov, E. A.; Khalilov, L. M. Russ. Chem. Bull. 2000, 49, 1103. doi:10.1007/BF02494903 |
| 274. | Odinokov, V. N.; Akhmetova, V. R.; Bazunova, M. V.; Savchenko, R. G.; Paramonov, E. A.; Khalilov, L. M. Russ. J. Org. Chem. 2001, 37, 321. doi:10.1023/A:1012311903712 |
| 334. | Tokuyasu, T.; Masuyama, A.; Nojima, M.; Kim, H.-S.; Wataya, Y. Tetrahedron Lett. 2000, 41, 3145. doi:10.1016/S0040-4039(00)00372-5 |
| 335. | Kim, H.-S.; Begum, K.; Ogura, N.; Wataya, Y.; Tokuyasu, T.; Masuyama, A.; Nojima, M.; McCullough, K. J. J. Med. Chem. 2002, 45, 4732. doi:10.1021/jm020208q |
| 419. | Iskra, J.; Bonnet-Delpon, D.; Bégué, J. P. Tetrahedron Lett. 2003, 44, 6309. doi:10.1016/S0040-4039(03)01472-2 |
| 276. | Jung, I. C. Eur. J. Org. Chem. 2001, 1899. doi:10.1002/1099-0690(200105)2001:10<1899::AID-EJOC1899>3.0.CO;2-L |
| 337. | O’Neill, P. M.; Rawe, S. L.; Storr, R. C.; Ward, S. A.; Posner, G. H. Tetrahedron Lett. 2005, 46, 3029. doi:10.1016/j.tetlet.2005.03.022 |
| 275. | Dussault, P. H.; Raible, J. M. Org. Lett. 2000, 2, 3377. doi:10.1021/ol006478p |
| 257. | Dussault, P. H.; Lee, I. Q.; Lee, H.-J.; Lee, R. J.; Niu, Q. J.; Schultz, J. A.; Zope, U. R. J. Org. Chem. 2000, 65, 8407. doi:10.1021/jo991714z |
| 421. | Žmitek, K.; Stavber, S.; Zupan, M.; Bonnet-Delpon, D.; Iskra, J. Tetrahedron 2006, 62, 1479. doi:10.1016/j.tet.2005.11.022 |
| 272. | Hon, Y.-S.; Lu, L.; Chang, R.-C.; Lin, S.-W.; Sun, P.-P.; Lee, C.-F. Tetrahedron 2000, 56, 9269. doi:10.1016/S0040-4020(00)00903-0 |
| 332. | Xu, C.; Schwartz, C.; Raible, J.; Dussault, P. H. Tetrahedron 2009, 65, 9680. doi:10.1016/j.tet.2009.09.068 |
| 417. | Opsenica, D.; Pocsfalvi, G.; Juranić, Z.; Tinant, B.; Declercq, J.-P.; Kyle, D. E.; Milhous, W. K.; Šolaja, B. A. J. Med. Chem. 2000, 43, 3274. doi:10.1021/jm000952f |
| 418. | Opsenica, D.; Angelovski, G.; Pocsfalvi, G.; Juranić, Z.; Žižak, Ž.; Kyle, D.; Milhous, W. K.; Šolaja, B. A. Bioorg. Med. Chem. 2003, 11, 2761. doi:10.1016/S0968-0896(03)00224-4 |
| 272. | Hon, Y.-S.; Lu, L.; Chang, R.-C.; Lin, S.-W.; Sun, P.-P.; Lee, C.-F. Tetrahedron 2000, 56, 9269. doi:10.1016/S0040-4020(00)00903-0 |
| 252. | Ghorai, P.; Dussault, P. H.; Hu, C. Org. Lett. 2008, 10, 2401. doi:10.1021/ol800657m |
| 417. | Opsenica, D.; Pocsfalvi, G.; Juranić, Z.; Tinant, B.; Declercq, J.-P.; Kyle, D. E.; Milhous, W. K.; Šolaja, B. A. J. Med. Chem. 2000, 43, 3274. doi:10.1021/jm000952f |
| 418. | Opsenica, D.; Angelovski, G.; Pocsfalvi, G.; Juranić, Z.; Žižak, Ž.; Kyle, D.; Milhous, W. K.; Šolaja, B. A. Bioorg. Med. Chem. 2003, 11, 2761. doi:10.1016/S0968-0896(03)00224-4 |
| 272. | Hon, Y.-S.; Lu, L.; Chang, R.-C.; Lin, S.-W.; Sun, P.-P.; Lee, C.-F. Tetrahedron 2000, 56, 9269. doi:10.1016/S0040-4020(00)00903-0 |
| 333. | Tokuyasu, T.; Masuyama, A.; Nojima, M.; McCullough, K. J. J. Org. Chem. 2000, 65, 1069. doi:10.1021/jo991499m |
| 419. | Iskra, J.; Bonnet-Delpon, D.; Bégué, J. P. Tetrahedron Lett. 2003, 44, 6309. doi:10.1016/S0040-4039(03)01472-2 |
| 272. | Hon, Y.-S.; Lu, L.; Chang, R.-C.; Lin, S.-W.; Sun, P.-P.; Lee, C.-F. Tetrahedron 2000, 56, 9269. doi:10.1016/S0040-4020(00)00903-0 |
| 333. | Tokuyasu, T.; Masuyama, A.; Nojima, M.; McCullough, K. J. J. Org. Chem. 2000, 65, 1069. doi:10.1021/jo991499m |
| 419. | Iskra, J.; Bonnet-Delpon, D.; Bégué, J. P. Tetrahedron Lett. 2003, 44, 6309. doi:10.1016/S0040-4039(03)01472-2 |
| 414. | McCullough, K. J.; Wood, J. K.; Bhattacharjee, A. K.; Dong, Y.; Kyle, D. E.; Milhous, W. K.; Vennerstrom, J. L. J. Med. Chem. 2000, 43, 1246. doi:10.1021/jm990530+ |
| 272. | Hon, Y.-S.; Lu, L.; Chang, R.-C.; Lin, S.-W.; Sun, P.-P.; Lee, C.-F. Tetrahedron 2000, 56, 9269. doi:10.1016/S0040-4020(00)00903-0 |
| 416. | Ayala, D. A.; Romero, J. M.; Jorge, N. L.; Gómez-Vara, M. E.; Jubert, A. H.; Castro, E. A. Spectrochim. Acta, Part A: Mol. Biomol. Spectrosc. 2006, 64, 717. doi:10.1016/j.saa.2005.06.046 |
| 250. | Dai, P.; Dussault, P. H. Org. Lett. 2005, 7, 4333. doi:10.1021/ol051407h |
| 251. | Barnych, B.; Vatèle, J.-M. Tetrahedron 2012, 68, 3717. doi:10.1016/j.tet.2012.03.024 |
| 252. | Ghorai, P.; Dussault, P. H.; Hu, C. Org. Lett. 2008, 10, 2401. doi:10.1021/ol800657m |
| 332. | Xu, C.; Schwartz, C.; Raible, J.; Dussault, P. H. Tetrahedron 2009, 65, 9680. doi:10.1016/j.tet.2009.09.068 |
| 415. | Berkessel, A.; Andreae, M. R. M.; Schmickler, H.; Lex, J. Angew. Chem., Int. Ed. 2002, 41, 4481. doi:10.1002/1521-3773(20021202)41:23<4481::AID-ANIE4481>3.0.CO;2-7 |
| 276. | Jung, I. C. Eur. J. Org. Chem. 2001, 1899. doi:10.1002/1099-0690(200105)2001:10<1899::AID-EJOC1899>3.0.CO;2-L |
| 283. | Schank, K.; Beck, H.; Pistorius, S. Helv. Chim. Acta 2004, 87, 2025. doi:10.1002/hlca.200490182 |
| 110. | Murakami, N.; Kawanishi, M.; Mostaqul, H. M.; Li, J.; Itagaki, S.; Horii, T.; Kobayashi, M. Bioorg. Med. Chem. Lett. 2003, 13, 4081. doi:10.1016/j.bmcl.2003.08.073 |
| 428. | Terent'ev, A. O.; Borisov, D. A.; Chernyshev, V. V.; Nikishin, G. I. J. Org. Chem. 2009, 74, 3335. doi:10.1021/jo900226b |
| 283. | Schank, K.; Beck, H.; Pistorius, S. Helv. Chim. Acta 2004, 87, 2025. doi:10.1002/hlca.200490182 |
| 107. | Bachi, M. D.; Korshin, E. E.; Hoos, R.; Szpilman, A. M.; Ploypradith, P.; Xie, S.; Shapiro, T. A.; Posner, G. H. J. Med. Chem. 2003, 46, 2516. doi:10.1021/jm020584a |
| 426. | Kumar, N.; Khan, S. I.; Sharma, M.; Atheaya, H.; Rawat, D. S. Bioorg. Med. Chem. Lett. 2009, 19, 1675. doi:10.1016/j.bmcl.2009.01.103 |
| 427. | Kumar, N.; Khan, S. I.; Beena, R.; Rajalakshmi, G.; Kumaradhas, P.; Rawat, D. S. Bioorg. Med. Chem. 2009, 17, 5632. doi:10.1016/j.bmc.2009.06.020 |
| 285. | Laventine, D. M.; Davies, M.; Evinson, E. L.; Jenkins, P. R.; Cullis, P. M.; Garcia, M. D. Tetrahedron 2009, 65, 4766. doi:10.1016/j.tet.2009.04.011 |
| 284. | Laventine, D. M.; Davies, M.; Evinson, E. L.; Jenkins, P. R.; Cullis, P. M.; Fawcett, J. Tetrahedron Lett. 2005, 46, 307. doi:10.1016/j.tetlet.2004.11.032 |
| 110. | Murakami, N.; Kawanishi, M.; Mostaqul, H. M.; Li, J.; Itagaki, S.; Horii, T.; Kobayashi, M. Bioorg. Med. Chem. Lett. 2003, 13, 4081. doi:10.1016/j.bmcl.2003.08.073 |
| 1. | McCullough, K. J.; Nojima, M. Curr. Org. Chem. 2001, 5, 601. doi:10.2174/1385272013375346 |
| 280. | Rücker, G.; Manns, D.; Schenkel, E. P.; Hartmann, R.; Heinzmann, B. M. Arch. Pharm. Pharm. Med. Chem. 2003, 336, 205. doi:10.1002/ardp.200300740 |
| 232. | Cointeaux, L.; Berrien, J.-F.; Mayrargue, J. Tetrahedron Lett. 2002, 43, 6275. doi:10.1016/S0040-4039(02)01122-X |
| 144. | Amewu, R.; Stachulski, A. V.; Ward, S. A.; Berry, N. G.; Bray, P. G.; Davies, J.; Labat, G.; Vivas, L.; O’Neill, P. M. Org. Biomol. Chem. 2006, 4, 4431. doi:10.1039/b613565j |
| 279. | Chen, L.; Wiemer, D. F. J. Org. Chem. 2002, 67, 7561. doi:10.1021/jo020362k |
| 270. | Tokuyasu, T.; Ito, T.; Masuyama, A.; Nojima, M. Heterocycles 2000, 53, 1293. doi:10.3987/COM-00-8874 |
| 141. | Opsenica, D. M.; Terzić, N.; Smith, P. L.; Yang, Y.; Anova, L.; Smith, K. S.; Solaja, B. A. Bioorg. Med. Chem. 2008, 16, 7039. doi:10.1016/j.bmc.2008.05.017 |
| 282. | Wattanasereekul, S.; Maier, M. E. Adv. Synth. Catal. 2004, 346, 855. doi:10.1002/adsc.200404004 |
| 336. | Harris, J. R.; Waetzig, S. R.; Woerpel, K. A. Org. Lett. 2009, 11, 3290. doi:10.1021/ol901046z |
| 144. | Amewu, R.; Stachulski, A. V.; Ward, S. A.; Berry, N. G.; Bray, P. G.; Davies, J.; Labat, G.; Vivas, L.; O’Neill, P. M. Org. Biomol. Chem. 2006, 4, 4431. doi:10.1039/b613565j |
| 281. | Hitchcock, P. B.; Papadopoulos, K.; Young, D. W. Org. Biomol. Chem. 2003, 1, 2670. doi:10.1039/b304607a |
| 339. | Rostami, A.; Wang, Y.; Arif, A. M.; McDonald, R.; West, F. G. Org. Lett. 2007, 9, 703. doi:10.1021/ol070053m |
| 424. | Terent'ev, A. O.; Kutkin, A. V.; Starikova, Z. A.; Antipin, M. Y.; Ogibin, Y. N.; Nikishin, G. I. Synthesis 2004, 2356. doi:10.1055/s-2004-831171 |
| 425. | Terent'ev, A. O.; Kutkin, A. V.; Platonov, M. M.; Vorontsov, I. I.; Antipin, M. Y.; Ogibin, Y. N.; Nikishin, G. I. Russ. Chem. Bull. 2004, 53, 681. doi:10.1023/B:RUCB.0000035657.58776.cc |
| 6. | Masuyama, A.; Wu, J.-M.; Nojima, M.; Kim, H.-S.; Wataya, Y. Mini-Rev. Med. Chem. 2005, 5, 1035. doi:10.2174/138955705774575255 |
| 334. | Tokuyasu, T.; Masuyama, A.; Nojima, M.; Kim, H.-S.; Wataya, Y. Tetrahedron Lett. 2000, 41, 3145. doi:10.1016/S0040-4039(00)00372-5 |
| 138. | Ellis, G. L.; Amewu, R.; Sabbani, S.; Stocks, P. A.; Shone, A.; Stanford, D.; Gibbons, P.; Davies, J.; Vivas, L.; Charnaud, S.; Bongard, E.; Hall, C.; Rimmer, K.; Lozanom, S.; Jesús, M.; Gargallo, D.; Ward, S. A.; O’Neill, P. M. J. Med. Chem. 2008, 51, 2170. doi:10.1021/jm701435h |
| 4. | O’Neill, P. M.; Posner, G. H. J. Med. Chem. 2004, 47, 2945. doi:10.1021/jm030571c |
| 5. | Slack, R. D.; Jacobine, A. M.; Posner, G. H. Med. Chem. Commun. 2012, 3, 281. doi:10.1039/c2md00277a |
| 422. | Atheaya, H.; Khan, S. I.; Mamgain, R.; Rawat, D. S. Bioorg. Med. Chem. Lett. 2008, 18, 1446. doi:10.1016/j.bmcl.2007.12.069 |
| 3. | Tang, Y.; Dong, Y.; Vennerstrom, J. L. Med. Res. Rev. 2004, 24, 425. doi:10.1002/med.10066 |
| 278. | Kiryukhin, D. P.; Barkalov, I. M.; Ismoilov, I. L. Russ. J. Appl. Chem. 2001, 74, 1740. doi:10.1023/A:1014829924249 |
| 338. | Tokuyasu, T.; Masuyama, A.; Nojima, M.; McCullough, K. J.; Kim, H.-S.; Wataya, Y. Tetrahedron 2001, 57, 5979. doi:10.1016/S0040-4020(01)00557-9 |
| 128. | Šolaja, B. A.; Terzić, N.; Pocsfalvi, G.; Gerena, L.; Tinant, B.; Opsenica, D.; Milhous, W. K. J. Med. Chem. 2002, 45, 3331. doi:10.1021/jm020891g |
| 132. | Opsenica, I.; Terzić, N.; Opsenica, D.; Angelovski, G.; Lehnig, L.; Eilbracht, P.; Tinant, B.; Juranić, Z.; Smith, K. S.; Yang, Y. S.; Diaz, D. S.; Smith, P. L.; Milhous, W. K.; Doković, D.; Šolaja, B. A. J. Med. Chem. 2006, 49, 3790. doi:10.1021/jm050966r |
| 141. | Opsenica, D. M.; Terzić, N.; Smith, P. L.; Yang, Y.; Anova, L.; Smith, K. S.; Solaja, B. A. Bioorg. Med. Chem. 2008, 16, 7039. doi:10.1016/j.bmc.2008.05.017 |
| 142. | Terzić, N.; Opsenica, D.; Milić, D.; Tinan, B.; Smith, K. S.; Milhous, W. K.; Šolaja, B. A. J. Med. Chem. 2007, 50, 5118. doi:10.1021/jm070684m |
| 2. | Korshin, E. E.; Bachi, M. D. Synthesis of cyclic peroxides. In The Chemistry of Peroxides; Rappoport, Z., Ed.; John Wiley & Sons, Ltd: Chichester, 2006; Vol. 2, Part 1, pp 189 ff. doi:10.1002/0470862769.ch5 |
| 277. | Caronna, T.; Gabbiadini, S.; Mele, A.; Recupero, F. Helv. Chim. Acta 2002, 85, 1. doi:10.1002/1522-2675(200201)85:1<1::AID-HLCA1>3.0.CO;2-C |
| 334. | Tokuyasu, T.; Masuyama, A.; Nojima, M.; Kim, H.-S.; Wataya, Y. Tetrahedron Lett. 2000, 41, 3145. doi:10.1016/S0040-4039(00)00372-5 |
| 423. | Ghorai, P.; Dussault, P. H. Org. Lett. 2009, 11, 213. doi:10.1021/ol8023874 |
| 16. | Opsenica, D. M.; Šolaja, B. A. J. Serb. Chem. Soc. 2009, 74, 1155. doi:10.2298/JSC0911155O |
| 10. | Dembitsky, V. M. Eur. J. Med. Chem. 2008, 43, 223. doi:10.1016/j.ejmech.2007.04.019 |
| 11. | Siddiq, A.; Dembitsky, V. Anti-Cancer Agents Med. Chem. 2008, 8, 132. doi:10.2174/187152008783497073 |
| 12. | Dembitsky, V. M.; Levitsky, D. O. Nat. Prod. Commun. 2006, 1, 405. |
| 13. | Dembitsky, V. M. Lipids 2006, 41, 883. doi:10.1007/s11745-006-5044-3 |
| 14. | Dembitsky, V. M. Lipids 2005, 40, 869. doi:10.1007/s11745-005-1449-2 |
| 15. | Dembitsky, V. M. Chem. Biodiversity 2004, 1, 673. doi:10.1002/cbdv.200490060 |
| 8. | Jefford, C. W. Drug Discovery Today 2007, 12, 487. doi:10.1016/j.drudis.2007.04.009 |
| 9. | Jefford, C. W. Curr. Top. Med. Chem. 2012, 12, 373. doi:10.2174/156802612799362940 |
| 7. | Van Ornum, S. G.; Champeau, R. M.; Pariza, R. Chem. Rev. 2006, 106, 2990. doi:10.1021/cr040682z |
| 291. | Shin, H. S.; Lee, C.; Lee, J. Y.; Huh, T. S. Eur. J. Org. Chem. 2000, 335. doi:10.1002/(SICI)1099-0690(200001)2000:2<335::AID-EJOC335>3.0.CO;2-2 |
| 348. | Gu, X.; Zhang, W.; Salomon, R. G. J. Am. Chem. Soc. 2007, 129, 6088. doi:10.1021/ja0689785 |
| 291. | Shin, H. S.; Lee, C.; Lee, J. Y.; Huh, T. S. Eur. J. Org. Chem. 2000, 335. doi:10.1002/(SICI)1099-0690(200001)2000:2<335::AID-EJOC335>3.0.CO;2-2 |
| 347. | Güney, M.; Ceylan, Z. C.; Daştan, A.; Balci, M. Can. J. Chem. 2005, 83, 227. doi:10.1139/v05-046 |
| 138. | Ellis, G. L.; Amewu, R.; Sabbani, S.; Stocks, P. A.; Shone, A.; Stanford, D.; Gibbons, P.; Davies, J.; Vivas, L.; Charnaud, S.; Bongard, E.; Hall, C.; Rimmer, K.; Lozanom, S.; Jesús, M.; Gargallo, D.; Ward, S. A.; O’Neill, P. M. J. Med. Chem. 2008, 51, 2170. doi:10.1021/jm701435h |
| 291. | Shin, H. S.; Lee, C.; Lee, J. Y.; Huh, T. S. Eur. J. Org. Chem. 2000, 335. doi:10.1002/(SICI)1099-0690(200001)2000:2<335::AID-EJOC335>3.0.CO;2-2 |
| 289. | Schiaffo, C. E.; Dussault, P. H. J. Org. Chem. 2008, 73, 4688. doi:10.1021/jo800323x |
| 345. | Güney, M.; Daştan, A.; Balci, M. Helv. Chim. Acta 2005, 88, 830. doi:10.1002/hlca.200590061 |
| 436. | Dubnikova, F.; Kosloff, R.; Almog, J.; Zeiri, Y.; Boese, R.; Itzhaky, H.; Alt, A.; Keinan, E. J. Am. Chem. Soc. 2005, 127, 1146. doi:10.1021/ja0464903 |
| 287. | Schwartz, C.; Raible, J.; Mott, K.; Dussault, P. H. Org. Lett. 2006, 8, 3199. doi:10.1021/ol061001k |
| 288. | Schwartz, C.; Raible, J.; Mott, K.; Dussault, P. H. Tetrahedron 2006, 62, 10747. doi:10.1016/j.tet.2006.08.092 |
| 344. | Daştan, A.; Saracoglu, N.; Balci, M. Eur. J. Org. Chem. 2001, 3519. doi:10.1002/1099-0690(200109)2001:18<3519::AID-EJOC3519>3.0.CO;2-2 |
| 435. | Terent’ev, A. O.; Kutkin, A. V.; Platonov, M. M.; Ogibin, Y. N.; Nikishin, G. I. Tetrahedron Lett. 2003, 44, 7359. doi:10.1016/S0040-4039(03)01844-6 |
| 291. | Shin, H. S.; Lee, C.; Lee, J. Y.; Huh, T. S. Eur. J. Org. Chem. 2000, 335. doi:10.1002/(SICI)1099-0690(200001)2000:2<335::AID-EJOC335>3.0.CO;2-2 |
| 346. | Daştan, A.; Balci, M. Tetrahedron 2006, 62, 4003. doi:10.1016/j.tet.2006.02.026 |
| 128. | Šolaja, B. A.; Terzić, N.; Pocsfalvi, G.; Gerena, L.; Tinant, B.; Opsenica, D.; Milhous, W. K. J. Med. Chem. 2002, 45, 3331. doi:10.1021/jm020891g |
| 290. | Percy, J. M.; Roig, R.; Singh, K. Eur. J. Org. Chem. 2009, 1058. doi:10.1002/ejoc.200801130 |
| 346. | Daştan, A.; Balci, M. Tetrahedron 2006, 62, 4003. doi:10.1016/j.tet.2006.02.026 |
| 135. | Opsenica, I.; Opsenica, D.; Lanteri, C. A.; Anova, L.; Milhous, W. K.; Smith, K. S.; Solaja, B. A. J. Med. Chem. 2008, 51, 6216. doi:10.1021/jm8006905 |
| 437. | Opsenica, I.; Opsenica, D.; Smith, K. S.; Milhous, W. K.; Šolaja, B. A. J. Med. Chem. 2008, 51, 2261. doi:10.1021/jm701417a |
| 148. | Dong, Y.; Chollet, J.; Vargas, M.; Mansour, N. R.; Bickle, Q.; Alnouti, Y.; Huang, J.; Keiser, J.; Vennerstrom, J. L. Bioorg. Med. Chem. Lett. 2010, 20, 2481. doi:10.1016/j.bmcl.2010.03.001 |
| 149. | Mohamed, A. E.-H. H.; El-Sayed, M. A.; Hegazy, M. E.; Helaly, S. E.; Esmail, A. M.; Mohamed, N. S. Rec. Nat. Prod. 2010, 4, 1. |
| 150. | Yang, Z.-S.; Wu, W.-M.; Li, Y.; Wu, Y.-L. Helv. Chim. Acta 2005, 88, 2865. doi:10.1002/hlca.200590229 |
| 151. | Keiser, J.; Utzinger, J. Adv. Parasitol. 2010, 73, 197. doi:10.1016/S0065-308X(10)73008-6 |
| 152. | Dembitsky, V.; Shkrob, I.; Hanus, L. O. Biomed. Pap. 2008, 152, 209. doi:10.5507/bp.2008.032 |
| 341. | López, D.; Quiñoá, E.; Riguera, R. J. Org. Chem. 2000, 65, 4671. doi:10.1021/jo000288a |
| 431. | Dong, Y.; Vennerstrom, J. L. J. Heterocycl. Chem. 2001, 38, 463. doi:10.1002/jhet.5570380224 |
| 432. | Dong, Y.; Vennerstrom, J. L. J. Org. Chem. 1998, 63, 8582. doi:10.1021/jo981261i |
| 340. | Matsumoto, M.; Nasu, S.; Takeda, M.; Murakami, H.; Watanabe, N. Chem. Commun. 2000, 821. doi:10.1039/b002161j |
| 430. | Kukovinets, O. S.; Zvereva, T. I.; Kabalnova, N. N.; Kasradze, V. G.; Salimova, E. V.; Khalitova, L. R.; Abdullin, M. I.; Spirikhin, L. V. Mendeleev Commun. 2009, 19, 106. doi:10.1016/j.mencom.2009.03.019 |
| 286. | Wang, C.; Liu, J.; Ji, Y.; Zhao, J.; Li, L.; Zhang, H. Org. Lett. 2006, 8, 2479. doi:10.1021/ol060382z |
| 343. | Özen, R.; Kormalı, F.; Balci, M.; Atasoy, B. Tetrahedron 2001, 57, 7529. doi:10.1016/S0040-4020(01)00702-5 |
| 434. | Opsenica, D.; Pocsfalvi, G.; Milhous, W. K.; Šolaja, B. A. J. Serb. Chem. Soc. 2002, 67, 465. doi:10.2298/JSC0207465O |
| 254. | Hamann, H.-J.; Wlosnewski, A.; Greco, T.; Liebscher, J. Eur. J. Org. Chem. 2006, 2174. doi:10.1002/ejoc.200500803 |
| 342. | Takabatake, T.; Miyazawa, T.; Hasegawa, M.; Foote, C. S. Tetrahedron Lett. 2001, 42, 987. doi:10.1016/S0040-4039(00)02216-4 |
| 433. | Song, C. E.; Lim, J. S.; Kim, S. C.; Lee, K.-J.; Chi, D. Y. Chem. Commun. 2000, 2415. doi:10.1039/b005604i |
| 429. | Terent'ev, A. O.; Yaremenko, I. A.; Vil’, V. A.; Moiseev, I. K.; Kon’kov, S. A.; Dembitsky, V. M.; Levitsky, D. O.; Nikishin, G. I. Org. Biomol. Chem. 2013, 11, 2613–2623. doi:10.1039/c3ob27239g |
| 153. | Laurent, S. A.-L.; Boissier, J.; Coslédan, F.; Gornitzka, H.; Robert, A.; Meunier, B. Eur. J. Org. Chem. 2008, 895. doi:10.1002/ejoc.200700975 |
| 154. | Jung, M.; Lee, K.; Kim, H.; Park, M. Curr. Med. Chem. 2004, 11, 1265. doi:10.2174/0929867043365233 |
| 155. | Xiao, S.-h.; Mei, J.-y.; Jiao, P.-y. Parasitol. Res. 2011, 108, 431. doi:10.1007/s00436-010-2084-7 |
| 156. | Boissier, J.; Coslédan, F.; Robert, A.; Meunier, B. Antimicrob. Agents Chemother. 2009, 53, 4903. doi:10.1128/AAC.00640-09 |
| 157. | Pradines, V.; Portela, J.; Boissier, J.; Cosledan, F.; Meunier, B.; Robert, A. Antimicrob. Agents Chemother. 2011, 55, 2403. doi:10.1128/AAC.00082-11 |
| 158. | Maurya, R.; Soni, A.; Anand, D.; Ravi, M.; Raju, K. S. R.; Taneja, I.; Naikade, N. K.; Puri, S. K.; Wahajuddin, K.; Anojiya, S.; Yadav, P. P. Med. Chem. Lett. 2013, 4, 165. doi:10.1021/ml300188t |
| 159. | Ingram, K.; Yaremenko, I. A.; Krylov, I.; Hofer, L.; Terent'ev, A. O.; Keiser, J. J. Med. Chem. 2012, 55, 8700. doi:10.1021/jm3009184 |
| 160. | Keiser, J.; Brun, R.; Fried, B.; Utzinger, J. Antimicrob. Agents Chemother. 2006, 50, 803. doi:10.1128/AAC.50.2.803-805.2006 |
| 161. | Vennervald, B. J.; Polman, K. Parasite Immunol. 2009, 31, 686. doi:10.1111/j.1365-3024.2009.01163.x |
| 162. | Yajima, A.; Gabrielli, A. F.; Montresor, A.; Engels, D. Trans. R. Soc. Trop. Med. Hyg. 2011, 105, 68. doi:10.1016/j.trstmh.2010.11.003 |
| 163. | Keiser, J.; Utzinger, J.; Tanner, M.; Dong, Y.; Vennerstrom, J. L. J. Antimicrob. Chemother. 2006, 58, 1193. doi:10.1093/jac/dkl408 |
| 164. | Cooper, P. J.; Ayre, G.; Martin, C.; Rizzo, J. A.; Ponte, E. V.; Cruz, A. A. Allergy (Oxford, U. K.) 2008, 63, 409. |
| 165. | Vandemark, L. M.; Jia, T.-W.; Zhou, X.-N. Adv. Parasitol. 2010, 73, 137. doi:10.1016/S0065-308X(10)73006-2 |
| 166. | Keiser, J.; Utzinger, J. Trends Parasitol. 2007, 23, 555. doi:10.1016/j.pt.2007.07.012 |
| 167. | Brooker, S.; Akhwale, W.; Pullan, R.; Estambale, B.; Clarke, S. E.; Snow, R. W.; Hotez, P. J. Am. J. Trop. Med. Hyg. 2007, 77 (Suppl. 6), 88. |
| 168. | Lv, S.; Zhang, Y.; Steinmann, P.; Zhou, X.-N.; Utzinger, J. Adv. Parasitol. 2010, 72, 351. doi:10.1016/S0065-308X(10)72012-1 |
| 169. | Xiao, S.-H.; Keiser, J.; Chollet, J.; Utzinger, J.; Dong, Y.; Endriss, Y.; Vennerstrom, J. L.; Tanner, M. Antimicrob. Agents Chemother. 2007, 51, 1440. doi:10.1128/AAC.01537-06 |
| 170. | Cooper, P. J.; Nutman, T. B. Lung Biol. Health Dis. 2002, 164, 409. |
| 171. | Brooker, S. Int. J. Parasitol. 2010, 40, 1137. doi:10.1016/j.ijpara.2010.04.004 |
| 172. | Keiser, J.; Utzinger, J. Curr. Opin. Infect. Dis. 2007, 20, 605. doi:10.1097/QCO.0b013e3282f19ec4 |
| 173. | Feldmeier, H.; Chitsulo, L. Arzneim. Forsch. 1999, 49, 557. |
| 174. | Thun, M. J.; Henley, S. J.; Gansler, T. Novartis Found. Symp. 2004, 256, 6. doi:10.1002/0470856734.ch2 |
| 208. | Howarth, J.; Wilson, D. Bioorg. Med. Chem. Lett. 2003, 13, 2013. doi:10.1016/S0960-894X(03)00326-3 |
| 209. | Van Assche, T.; Deschacht, M.; Inocencio da Luz, R. A.; Maes, L.; Cos, P. Free Radical Biol. Med. 2011, 51, 337. doi:10.1016/j.freeradbiomed.2011.05.011 |
| 210. | Lim, C.-W.; Kim, Y.-K.; Jang, M. S.; Park, J.-I.; Park, H.-Y. J. Fish. Sci. Technol. 2006, 9, 175. |
| 211. | Oguri, H.; Hiruma, T.; Yamagishi, Y.; Oikawa, H.; Ishiyama, A.; Otoguro, K.; Yamada, H.; Omura, S. J. Am. Chem. Soc. 2011, 133, 7096. doi:10.1021/ja200374q |
| 212. | Feng, Y.-J.; Davis, R. A.; Sykes, M.; Avery, V. M.; Camp, D.; Quinn, R. J. J. Nat. Prod. 2010, 73, 716. doi:10.1021/np900535z |
| 210. | Lim, C.-W.; Kim, Y.-K.; Jang, M. S.; Park, J.-I.; Park, H.-Y. J. Fish. Sci. Technol. 2006, 9, 175. |
| 213. | Macreadie, P.; Avery, T.; Greatrex, B.; Taylor, D.; Macreadie, I. Bioorg. Med. Chem. Lett. 2006, 16, 920. doi:10.1016/j.bmcl.2005.10.101 |
| 214. | Manzo, E.; Ciavatta, M. L.; Melck, D.; Schupp, P.; de Voogd, N.; Gavagnin, M. J. Nat. Prod. 2009, 72, 1547. doi:10.1021/np900310j |
| 215. | Dembitsky, V. M. Tetrahedron 2003, 59, 4701. doi:10.1016/S0040-4020(03)00701-4 |
| 216. | Dembitsky, V. M.; Srebnik, M. Eurasian Chem.–Technol. J. 2002, 4, 221. |
| 217. | Chen, Y.; Chilson, K.; Killday, K. B.; Harmody, D.; McCarthy, P. J.; Pomponi,, S. A.; Schimoler, R.; Selitrennikoff, C.; Wright, A. E. Cyclic peroxides as novel antifungal agents. U. S. Patent US 6521661 B1, Feb 18, 2003. |
| 218. | Avery, T. D.; Macreadie, P. I.; Greatrex, B. W.; Robinson, T. V.; Taylor, D. K.; Macreadie, I. G. Bioorg. Med. Chem. 2007, 17, 36. doi:10.1016/j.bmc.2006.10.021 |
| 219. | Dembitsky, V. M.; Srebnik, M. Prog. Lipid Res. 2002, 41, 315. doi:10.1016/S0163-7827(02)00003-6 |
| 220. | Fattorusso, E.; Parapini, S.; Campagnuolo, C.; Basilico, N.; Taglialatela-Scafati, O.; Taramelli, D. J. Antimicrob. Chemother. 2002, 50, 883. doi:10.1093/jac/dkg008 |
| 221. | Macreadie, I. G.; Avery, T. D.; Robinson, T. V.; Macreadie, P.; Barraclough, M.; Taylor, D. K.; Tiekink, E. R. T. Tetrahedron 2008, 64, 1225. doi:10.1016/j.tet.2007.11.071 |
| 222. | Kishi, M.; Maeno, K.; Natsuhara, M.; Kususe, M. Mold removal agent composition. Patent Jpn. Kokai Tokkyo Koho JP 11166196 A, June 22, 1999. |
| 223. | Gunasekera, S. P.; Gunasekera, M.; Gunawardana, G. P.; McCarthy, P.; Burres, N. J. Nat. Prod. 1990, 53, 669. doi:10.1021/np50069a021 |
| 224. | Phillipson, D. W.; Rinehart, K. L., Jr. J. Am. Chem. Soc. 1983, 105, 7735. doi:10.1021/ja00364a045 |
| 10. | Dembitsky, V. M. Eur. J. Med. Chem. 2008, 43, 223. doi:10.1016/j.ejmech.2007.04.019 |
| 11. | Siddiq, A.; Dembitsky, V. Anti-Cancer Agents Med. Chem. 2008, 8, 132. doi:10.2174/187152008783497073 |
| 12. | Dembitsky, V. M.; Levitsky, D. O. Nat. Prod. Commun. 2006, 1, 405. |
| 13. | Dembitsky, V. M. Lipids 2006, 41, 883. doi:10.1007/s11745-006-5044-3 |
| 14. | Dembitsky, V. M. Lipids 2005, 40, 869. doi:10.1007/s11745-005-1449-2 |
| 15. | Dembitsky, V. M. Chem. Biodiversity 2004, 1, 673. doi:10.1002/cbdv.200490060 |
| 73. | Begue, J.-P.; Bonnet-Delpon, D. Drugs Future 2005, 30, 509. doi:10.1358/dof.2005.030.05.901987 |
| 186. | Dembitsky, V. M.; Gloriozova, T. A.; Poroikov, V. V. Mini-Rev. Med. Chem. 2007, 7, 571. doi:10.2174/138955707780859396 |
| 187. | Hang, T. T. N.; Hu'o'ng, L. M.; Ha, T. T. H.; Ha, L. M. Tap Chi Duoc Hoc 2009, 49, 47. |
| 188. | Ma, Y.-M.; Feng, C.-L. Youji Huaxue 2008, 28, 1697. |
| 189. | Dai, J.; Liu, Y.; Zhou, Y.-D.; Nagle, D. G. J. Nat. Prod. 2007, 70, 130. doi:10.1021/np0604883 |
| 190. | Rubush, D. M.; Morges, M. A.; Rose, B. J.; Thamm, D. H.; Rovis, T. J. Am. Chem. Soc. 2012, 134, 13554. doi:10.1021/ja3052427 |
| 191. | Dembitsky, V. M.; Gloriozova, T. A.; Poroikov, V. V. Mini-Rev. Med. Chem. 2005, 5, 319. doi:10.2174/1389557053175362 |
| 192. | Matsugo, S.; Kayamori, N.; Konishi, T. Niigata Yakka Daigaku Kenkyu Hokoku 1986, 6, 23. |
| 193. | Ryden, A.-M.; Kayser, O. Top. Heterocycl. Chem. 2007, 9, 1. doi:10.1007/7081_2007_085 |
| 194. | Roshchina, V. V. Uspekhi Sovrem. Biol. 2006, 126, 366. |
| 195. | Bradley, D. Drug Discovery Today 2000, 5, 44. doi:10.1016/S1359-6446(99)01453-1 |
| 196. | Jiménez, M. S.; Garznm, S. P.; Rodríguez, A. D. J. Nat. Prod. 2003, 66, 655. |
| 197. | Casteel, D. A. Nat. Prod. Rep. 1999, 16, 55. doi:10.1039/a705725c |
| 198. | Casteel, D. A. Nat. Prod. Rep. 1992, 9, 289. doi:10.1039/np9920900289 |
| 199. | Lee, K. R. Saengyak Hakhoechi 1991, 22, 145. |
| 200. | Kasozi, D. M.; Rahlfs, S.; Becker, K. Drug Discovery Infect. Dis. 2011, 2, 413. |
| 201. | Denisov, E. T.; Solodova, S. L.; Denisova, T. G. Russ. Chem. Rev. 2010, 79, 981. doi:10.1070/RC2010v079n11ABEH004143 |
| 202. | Davies-Coleman, M. T. Stud. Nat. Prod. Chem. 2005, 32, 61. doi:10.1016/S1572-5995(05)80054-7 |
| 203. | Mancini, I.; Guella, G.; Defant, A. Mini-Rev. Med. Chem. 2008, 8, 1265. doi:10.2174/138955708786141007 |
| 204. | Lupulescu, A. Prostaglandins, Leukotrienes Essent. Fatty Acids 1996, 54, 83. doi:10.1016/S0952-3278(96)90064-2 |
| 205. | Ridley, R. G.; Hudson, A. T. Curr. Opin. Infect. Dis. 1998, 11, 691. doi:10.1097/00001432-199812000-00008 |
| 206. | Mohammed, R.; Peng, J.; Kelly, M.; Yousaf, M.; Winn, E.; Odde, S.; Bie, Z.; Xie, A.; Doerksen, R. J.; Hamann, M. T. Aust. J. Chem. 2010, 63, 877. doi:10.1071/CH09665 |
| 207. | Camuzat-Dedenis, B.; Provot, O.; Cointeaux, L.; Perroux, V.; Berrien, J.-F.; Bories, C.; Loiseau, P. M.; Mayrargue, J. Eur. J. Med. Chem. 2001, 36, 837. doi:10.1016/S0223-5234(01)01278-8 |
| 114. | Rudi, A.; Talpir, R.; Kashman, Y.; Benayahu, Y.; Schleyer, M. J. Nat. Prod. 1993, 56, 2178. doi:10.1021/np50102a023 |
| 182. | Yao, G.; Steliou, K. Org. Lett. 2002, 4, 485. doi:10.1021/ol016943y |
| 183. | Jung, M.; Ham, J.; Song, J. Org. Lett. 2002, 4, 2763. doi:10.1021/ol026285x |
| 184. | Wijeratne, E. M. K.; Liu, M. X.; Kantipudi, N. B.; Brochini, C. B.; Gunatilaka, A. A. L.; Canfield, L. M. Bioorg. Med. Chem. 2006, 14, 7875. doi:10.1016/j.bmc.2006.07.057 |
| 185. | Aldeco-Pérez, E.; Rudler, H.; Parlier, A.; Alvarez, C.; Apan, M. T.; Herson, P.; Toscano, A. Tetrahedron Lett. 2006, 47, 9053. doi:10.1016/j.tetlet.2006.10.093 |
| 10. | Dembitsky, V. M. Eur. J. Med. Chem. 2008, 43, 223. doi:10.1016/j.ejmech.2007.04.019 |
| 11. | Siddiq, A.; Dembitsky, V. Anti-Cancer Agents Med. Chem. 2008, 8, 132. doi:10.2174/187152008783497073 |
| 12. | Dembitsky, V. M.; Levitsky, D. O. Nat. Prod. Commun. 2006, 1, 405. |
| 13. | Dembitsky, V. M. Lipids 2006, 41, 883. doi:10.1007/s11745-006-5044-3 |
| 14. | Dembitsky, V. M. Lipids 2005, 40, 869. doi:10.1007/s11745-005-1449-2 |
| 15. | Dembitsky, V. M. Chem. Biodiversity 2004, 1, 673. doi:10.1002/cbdv.200490060 |
| 175. | Niesen, A.; Barthel, A.; Kluge, R.; Köwitzsch, A.; Ströhl, D.; Schwarz, S.; Csuk, R. Arch. Pharm. Chem. Life Sci. 2009, 342, 569. doi:10.1002/ardp.200900051 |
| 176. | Sawant, S. S.; Youssef, D. T. A.; Sylvester, P. W.; Wali, V.; El Sayed, K. A. Nat. Prod. Commun. 2007, 2, 117. |
| 10. | Dembitsky, V. M. Eur. J. Med. Chem. 2008, 43, 223. doi:10.1016/j.ejmech.2007.04.019 |
| 11. | Siddiq, A.; Dembitsky, V. Anti-Cancer Agents Med. Chem. 2008, 8, 132. doi:10.2174/187152008783497073 |
| 12. | Dembitsky, V. M.; Levitsky, D. O. Nat. Prod. Commun. 2006, 1, 405. |
| 13. | Dembitsky, V. M. Lipids 2006, 41, 883. doi:10.1007/s11745-006-5044-3 |
| 14. | Dembitsky, V. M. Lipids 2005, 40, 869. doi:10.1007/s11745-005-1449-2 |
| 15. | Dembitsky, V. M. Chem. Biodiversity 2004, 1, 673. doi:10.1002/cbdv.200490060 |
| 175. | Niesen, A.; Barthel, A.; Kluge, R.; Köwitzsch, A.; Ströhl, D.; Schwarz, S.; Csuk, R. Arch. Pharm. Chem. Life Sci. 2009, 342, 569. doi:10.1002/ardp.200900051 |
| 176. | Sawant, S. S.; Youssef, D. T. A.; Sylvester, P. W.; Wali, V.; El Sayed, K. A. Nat. Prod. Commun. 2007, 2, 117. |
| 177. | Csuk, R.; Niesen-Barthel, A.; Barthel, A.; Kluge, R.; Ströhl, D. Eur. J. Med. Chem. 2010, 45, 3840. doi:10.1016/j.ejmech.2010.05.036 |
| 178. | Rudi, A.; Afanii, R.; Gravalos, L. G.; Aknin, M.; Gaydou, E.; Vacelet, J.; Kashman, Y. J. Nat. Prod. 2003, 66, 682. doi:10.1021/np020589a |
| 10. | Dembitsky, V. M. Eur. J. Med. Chem. 2008, 43, 223. doi:10.1016/j.ejmech.2007.04.019 |
| 11. | Siddiq, A.; Dembitsky, V. Anti-Cancer Agents Med. Chem. 2008, 8, 132. doi:10.2174/187152008783497073 |
| 12. | Dembitsky, V. M.; Levitsky, D. O. Nat. Prod. Commun. 2006, 1, 405. |
| 13. | Dembitsky, V. M. Lipids 2006, 41, 883. doi:10.1007/s11745-006-5044-3 |
| 14. | Dembitsky, V. M. Lipids 2005, 40, 869. doi:10.1007/s11745-005-1449-2 |
| 15. | Dembitsky, V. M. Chem. Biodiversity 2004, 1, 673. doi:10.1002/cbdv.200490060 |
| 112. | Liu, H.-H.; Jin, H.-X.; Zhang, Q.; Wu, Y.-K.; Kim, H.-S.; Wataya, Y. Chin. J. Chem. 2005, 23, 1469. doi:10.1002/cjoc.200591469 |
| 178. | Rudi, A.; Afanii, R.; Gravalos, L. G.; Aknin, M.; Gaydou, E.; Vacelet, J.; Kashman, Y. J. Nat. Prod. 2003, 66, 682. doi:10.1021/np020589a |
| 179. | Szpilman, A. M.; Korshin, E. E.; Rozenberg, H.; Bachi, M. D. J. Org. Chem. 2005, 70, 3618. doi:10.1021/jo050074z |
| 180. | Berrue, F.; Thomas, O. P.; Funel-Le Bon, C.; Reyes, F.; Amade, P. Tetrahedron 2005, 61, 11843. doi:10.1016/j.tet.2005.09.077 |
| 181. | Rudi, A.; Kashman, Y. J. Nat. Prod. 1993, 56, 1827. doi:10.1021/np50100a027 |
| 128. | Šolaja, B. A.; Terzić, N.; Pocsfalvi, G.; Gerena, L.; Tinant, B.; Opsenica, D.; Milhous, W. K. J. Med. Chem. 2002, 45, 3331. doi:10.1021/jm020891g |
| 129. | Kirchhofer, C.; Vargas, M.; Braissant, O.; Dong, Y.; Wang, X.; Vennerstrom, J. L.; Keiser, J. Acta Trop. 2011, 118, 56. doi:10.1016/j.actatropica.2011.02.003 |
| 130. | Vennerstrom, J. L.; Fu, H. N.; Ellis, W. Y.; Ager, A. L., Jr.; Wood, J. K.; Andersen, S. L.; Gerena, L.; Milhous, W. K. J. Med. Chem. 1992, 35, 3023. doi:10.1021/jm00094a015 |
| 131. | Hamann, H.-J.; Hecht, M.; Bunge, A.; Gogol, M.; Liebscher, J. Tetrahedron Lett. 2011, 52, 107. doi:10.1016/j.tetlet.2010.10.151 |
| 225. | Rugutt, J. K.; Rugutt, K. J. Nat. Prod. Lett. 2002, 16, 107. doi:10.1080/10575630290020000 |
| 226. | Modi, I. A.; Ghosh, P. K.; Bhardwaj, D.; Desai, N. M.; Khamar, B. M. P38 inhibitors. PCT Int. Appl. WO 2008114119 A2, Sept 25, 2008. |
| 227. | Kyle, D. E.; Milhous, W.; Opsenica, D. M.; Pocsfalvi, G.; Solaja, B. Mixed steroidal 1,2,4,5-tetraoxane compounds and methods of making and using thereof. PCT Int. Appl. WO 2003068736 A2, Aug 21, 2003. |
| 228. | Chen, L.-W.; Cheng, M.-J.; Peng, C.-F.; Chen, I.-S. Chem. Biodiversity 2010, 7, 1814. doi:10.1002/cbdv.200900227 |
| 229. | Stratakis, M.; Orfanopoulos, M. Tetrahedron 2000, 56, 1595. doi:10.1016/S0040-4020(99)00950-3 |
| 230. | Clennan, E. L.; Pace, A. Tetrahedron 2005, 61, 6665. doi:10.1016/j.tet.2005.04.017 |
| 231. | Frimer, A. A.; Afri, M.; Baumel, S. D.; Gilinsky-Sharon, P.; Rosenthal, Z.; Gottlieb, H. E. J. Org. Chem. 2000, 65, 1807. doi:10.1021/jo991803b |
| 391. | Griesbeck, A. G.; Blunk, D.; El-Idreesy, T. T.; Raabe, A. Angew. Chem., Int. Ed. 2007, 46, 8883. doi:10.1002/anie.200701397 |
| 385. | Bartoschek, A.; El-Idreesy, T. T.; Griesbeck, A. G.; Höinck, L.-O.; Lex, J.; Miara, C.; Neudörfl, J. M. Synthesis 2005, 2433. doi:10.1055/s-2005-872103 |
| 120. | Amewu, R.; Gibbons, P.; Mukhtar, A.; Stachulski, A. V.; Ward, S. A.; Hall, C.; Rimmer, K.; Davies, J.; Vivas, L.; Bacsa, J.; Mercer, A. E.; Nixon, G.; Stocks, P. A.; O’Neill, P. M. Org. Biomol. Chem. 2010, 8, 2068. doi:10.1039/b924319d |
| 393. | O’Neill, P. M.; Mukhtar, A.; Ward, S. A.; Bickley, J. F.; Davies, J.; Bachi, M. D.; Stocks, P. A. Org. Lett. 2004, 6, 3035. doi:10.1021/ol0492142 |
| 389. | Griesbeck, A. G.; Höinck, L.-O.; Lex, J.; Neudörfl, J.; Blunk, D.; El-Idreesy, T. T. Molecules 2008, 13, 1743. doi:10.3390/molecules13081743 |
| 391. | Griesbeck, A. G.; Blunk, D.; El-Idreesy, T. T.; Raabe, A. Angew. Chem., Int. Ed. 2007, 46, 8883. doi:10.1002/anie.200701397 |
| 391. | Griesbeck, A. G.; Blunk, D.; El-Idreesy, T. T.; Raabe, A. Angew. Chem., Int. Ed. 2007, 46, 8883. doi:10.1002/anie.200701397 |
| 237. | Wimalasena, K.; Wickman, H. B.; Mahindaratne, M. P. D. Eur. J. Org. Chem. 2001, 3811. doi:10.1002/1099-0690(200110)2001:20<3811::AID-EJOC3811>3.0.CO;2-6 |
| 238. | Ouhamou, N.; Six, Y. Org. Biomol. Chem. 2003, 1, 3007. doi:10.1039/b306719j |
| 236. | Kulinkovich, O. G.; Astashko, D. A.; Tyvorskii, V. I.; Ilyina, N. A. Synthesis 2001, 1453. doi:10.1055/s-2001-16089 |
| 237. | Wimalasena, K.; Wickman, H. B.; Mahindaratne, M. P. D. Eur. J. Org. Chem. 2001, 3811. doi:10.1002/1099-0690(200110)2001:20<3811::AID-EJOC3811>3.0.CO;2-6 |
| 234. | Barnier, J.-P.; Morisson, V.; Blanco, L. Synth. Commun. 2001, 31, 349. doi:10.1081/SCC-100000523 |
| 235. | Kirihara, M.; Kakuda, H.; Ichinose, M.; Ochiai, Y.; Takizawa, S.; Mokuya, A.; Okubo, K.; Hatano, A.; Shiro, M. Tetrahedron 2005, 61, 4831. doi:10.1016/j.tet.2005.03.033 |
| 232. | Cointeaux, L.; Berrien, J.-F.; Mayrargue, J. Tetrahedron Lett. 2002, 43, 6275. doi:10.1016/S0040-4039(02)01122-X |
| 233. | Adam, W.; Peters, K.; Peters, E.-M.; Schambony, S. J. Am. Chem. Soc. 2000, 122, 7610. doi:10.1021/ja001113l |
| 395. | Posner, G. H.; Maxwell, J. P.; O'Dowd, H.; Krasavin, M.; Xie, S.; Shapiro, T. A. Bioorg. Med. Chem. 2000, 8, 1361. doi:10.1016/S0968-0896(00)00079-1 |
| 394. | Jefford, C. W.; Velarde, J. A.; Bernardinelli, G.; Bray, D. H.; Warhurst, D. C.; Milhous, W. K. Helv. Chim. Acta 1993, 76, 2775. doi:10.1002/hlca.19930760804 |
| 395. | Posner, G. H.; Maxwell, J. P.; O'Dowd, H.; Krasavin, M.; Xie, S.; Shapiro, T. A. Bioorg. Med. Chem. 2000, 8, 1361. doi:10.1016/S0968-0896(00)00079-1 |
| 398. | Dechy-Cabaret, O.; Benoit-Vical, F.; Loup, C.; Robert, A.; Gornitzka, H.; Bonhoure, A.; Vial, H.; Magnaval, J.-F.; Séguéla, J.-P.; Meunier, B. Chem.–Eur. J. 2004, 10, 1625. doi:10.1002/chem.200305576 |
| 207. | Camuzat-Dedenis, B.; Provot, O.; Cointeaux, L.; Perroux, V.; Berrien, J.-F.; Bories, C.; Loiseau, P. M.; Mayrargue, J. Eur. J. Med. Chem. 2001, 36, 837. doi:10.1016/S0223-5234(01)01278-8 |
| 399. | Singh, R.; Ishar, M. P. S. Tetrahedron Lett. 2003, 44, 1943. doi:10.1016/S0040-4039(03)00086-8 |
| 395. | Posner, G. H.; Maxwell, J. P.; O'Dowd, H.; Krasavin, M.; Xie, S.; Shapiro, T. A. Bioorg. Med. Chem. 2000, 8, 1361. doi:10.1016/S0968-0896(00)00079-1 |
| 395. | Posner, G. H.; Maxwell, J. P.; O'Dowd, H.; Krasavin, M.; Xie, S.; Shapiro, T. A. Bioorg. Med. Chem. 2000, 8, 1361. doi:10.1016/S0968-0896(00)00079-1 |
| 397. | Posner, G. H.; Jeon, H. B.; Ploypradith, P.; Paik, I.-H.; Borstnik, K.; Xie, S.; Shapiro, T. A. J. Med. Chem. 2002, 45, 3824. doi:10.1021/jm020210h |
| 396. | Posner, G. H.; Jeon, H. B.; Parker, M. H.; Krasavin, M.; Paik, I.-H.; Shapiro, T. A. J. Med. Chem. 2001, 44, 3054. doi:10.1021/jm0102396 |
| 47. | Doerig, C. D. Nat. Chem. Biol. 2008, 4, 334. doi:10.1038/nchembio0608-334 |
| 48. | Vangapandu, S.; Jain, M.; Kaur, K.; Patil, P.; Patel, S. R.; Jain, R. Med. Res. Rev. 2007, 27, 65. doi:10.1002/med.20062 |
| 49. | Tu, Y. Nat. Med. (N. Y., NY, U. S.) 2011, 17, 1217. doi:10.1038/nm.2471 |
| 50. | Kumar, N.; Sharma, M.; Rawat, D. S. Curr. Med. Chem. 2011, 18, 3889. doi:10.2174/092986711803414340 |
| 51. | Liao, F. Molecules 2009, 14, 5362. doi:10.3390/molecules14125362 |
| 33. | Morgan, N. N. Int. J. Phys. Sci. 2009, 4, 885. |
| 34. | Odinokov, V. N.; Tolstikov, G. A. Russ. Chem. Rev. 1981, 50, 636. doi:10.1070/RC1981v050n07ABEH002658 |
| 35. | Razumovskii, S. D.; Zaikov, G. E. Russ. Chem. Rev. 1980, 49, 1163. doi:10.1070/RC1980v049n12ABEH002535 |
| 36. | Schmidt-Szalowski, K. Przem. Chem. 2000, 79, 115. |
| 37. | Capon, R. J. Stud. Nat. Prod. Chem. 1991, 9, 15. |
| 38. | Cafferata, L. F. R. Trends Org. Chem. 1993, 4, 773. |
| 39. | Tolstikov, G. A.; Tolstikov, A. G.; Tolstikova, O. V. Russ. Chem. Rev. 1996, 65, 769. doi:10.1070/RC1996v065n09ABEH000240 |
| 40. | McCullough, K. J. Contemp. Org. Synth. 1995, 2, 225. doi:10.1039/co9950200225 |
| 41. | Adam, W. Acc. Chem. Res. 1979, 12, 390. doi:10.1021/ar50143a002 |
| 42. | Opsenica, D. M.; Šolaja, B. A. Maced. J. Chem. Chem. Eng. 2012, 31, 137. |
| 43. | Sawwan, N.; Greer, A. Chem. Rev. 2007, 107, 3247. doi:10.1021/cr0400717 |
| 44. | Clennan, E. L.; Foote, C. In Organic Peroxides; Ando, E., Ed.; Wiley: New York, 1992. |
| 45. | Balci, M. Chem. Rev. 1981, 81, 91. doi:10.1021/cr00041a005 |
| 46. | Clennan, E. L. Tetrahedron 1991, 47, 1343. doi:10.1016/S0040-4020(01)86413-9 |
| 18. | Kim, B. J.; Sasaki, T. Org. Prep. Proced. Int. 2006, 38, 1. doi:10.1080/00304940609355981 |
| 19. | Baby, J.; Sujatha, S. J. Nat. Prod. (Gorakhpur, India) 2011, 4, 5. |
| 20. | Hepworth, J. D.; Heron, B. M. Prog. Heterocycl. Chem. 1996, 8, 277. doi:10.1016/S0959-6380(96)80017-1 |
| 21. | Kabal'nova, N. N.; Khursan, S. L.; Shereshovets, V. V.; Tolstikov, G. A. Kinet. Catal. 1999, 40, 207. |
| 22. | Terent'ev, A. O.; Platonov, M. M.; Levitsky, D. O.; Dembitsky, V. M. Russ. Chem. Rev. 2011, 80, 807. doi:10.1070/RC2011v080n09ABEH004189 |
| 23. | Baader, W. J.; Bastos, E. L. Sci. Synth. 2008, 38, 345–378. |
| 24. | Baader, W. J.; Bastos, E. L. Sci. Synth. 2008, 38, 379–395. |
| 25. | Baader, W. J.; Bastos, E. L. Sci. Synth. 2008, 38, 397–420. |
| 26. | Kumar, N.; Singh, R.; Rawat, D. S. Med. Res. Rev. 2012, 32, 581–610. doi:10.1002/med.20223 |
| 27. | Zmitek, K.; Zupan, M.; Iskra, J. Org. Biomol. Chem. 2007, 5, 3895. doi:10.1039/b711647k |
| 28. |
Trullinger, T. K. Ph.D. Thesis, University of Nebraska, Lincoln, NE, USA, 2002; pp 195 ff.
Diss. Abstr. Int., B 2003, 63, 5855. |
| 29. |
Dai, P. Ph.D. Thesis, University of Nebraska, Lincoln, NE, USA, 2004; pp 186 ff.
Diss. Abstr. Int., B 2005, 66, 281. |
| 30. |
Ramirez, A. P. Ph.D. Thesis, University of California, Irvine, CA, USA, 2007; pp 224 ff.
Diss. Abstr. Int., B 2007, 67, 6412. |
| 31. |
Pena-Quevedo, A. J. Ph.D. Thesis, University of Puerto Rico, Mayaguez, P. R., 2009; pp 159 ff.
Diss. Abstr. Int., B 2010, 70, 4186. |
| 32. |
Yao, G. Ph.D. Thesis, Boston University, Boston, MA, USA, 1999; pp 243 ff.
Diss. Abstr. Int., B 1999, 60, 193. |
| 17. | Muraleedharan, K. M.; Avery, M. A. Drug Discovery Today 2009, 14, 793. doi:10.1016/j.drudis.2009.05.008 |
| 82. | O’Neill, P. M.; Amewu, R. K.; Nixon, G. L.; ElGarah, F. B.; Mungthin, M.; Chadwick, J.; Shone, A. E.; Vivas, L.; Lander, H.; Barton, V.; Muangnoicharoen, S.; Bray, P. G.; Davies, J.; Park, B. K.; Wittlin, S.; Brun, R.; Preschel, M.; Zhang, K.; Ward, S. A. Angew. Chem., Int. Ed. 2010, 49, 5693. doi:10.1002/anie.201001026 |
| 83. | Bousejra-El Garah, F.; Wong, M. H.-L.; Amewu, R. K.; Muangnoicharoen, S.; Maggs, J. L.; Stigliani, J.-L.; Park, B. K.; Chadwick, J.; Ward, S. A.; O'Neill, P. M. J. Med. Chem. 2011, 54, 6443. doi:10.1021/jm200768h |
| 84. | Posner, G. H.; O’Neill, P. M. Acc. Chem. Res. 2004, 37, 397. doi:10.1021/ar020227u |
| 85. | Sonnet, P.; Mullié, C. Exp. Parasitol. 2011, 128, 26. doi:10.1016/j.exppara.2011.01.018 |
| 86. | Coslédan, F.; Fraisse, L.; Pellet, A.; Guillou, G.; Mordmüller, M.; Kremsner, P.; Moreno, A.; Mazier, D.; Maffrand, J.-P.; Meunier, B. Proc. Natl. Acad. Sci. U. S. A. 2008, 105, 17579. doi:10.1073/pnas.0804338105 |
| 87. | Meunier, B.; Robert, A. Acc. Chem. Res. 2010, 43, 1444. doi:10.1021/ar100070k |
| 52. | Morris, C. A.; Duparc, S.; Borghini-Fuhrer, I.; Jung, D.; Shin, C.-S.; Fleckenstein, L. Malar. J. 2011, 10, 263. doi:10.1186/1475-2875-10-263 |
| 53. | Robert, A.; Dechy-Cabaret, O.; Cazalles, J.; Meunier, B. Acc. Chem. Res. 2002, 35, 167–174. doi:10.1021/ar990164o |
| 54. | Hombhanje, F. W.; Huang, Q. Pharmaceuticals 2010, 3, 3581. doi:10.3390/ph3123581 |
| 55. | Kano, S. J. Infect. Chemother. 2010, 16, 375. doi:10.1007/s10156-010-0077-1 |
| 56. | Li, Q.; Weina, P. Pharmaceuticals 2010, 3, 2322. doi:10.3390/ph3072322 |
| 57. | Wright, C. W. Nat. Prod. Rep. 2010, 27, 961. doi:10.1039/c002196m |
| 58. | Kaur, H.; Green, M. D.; Hostetler, D. M.; Fernandez, F. M.; Newton, P. N. Therapy 2010, 7, 49. doi:10.2217/thy.09.84 |
| 59. | Medhi, B.; Patyar, S.; Rao, R. S.; Byrav, D. S. P.; Prakash, A. Pharmacology 2009, 84, 323. doi:10.1159/000252658 |
| 60. | Eastman, R. T.; Fidock, D. A. Nat. Rev. Microbiol. 2009, 7, 864. doi:10.1038/nrmicro2239 |
| 61. | Ehrhardt, S.; Meyer, C. G. Ther. Clin. Risk Manage. 2009, 5, 805. doi:10.2147/TCRM.S5375 |
| 62. | Gautam, A.; Ahmed, T.; Paliwal, J.; Batra, V. Curr. Drug Metab. 2009, 10, 289. doi:10.2174/138920009787846323 |
| 63. | Bathurst, I.; Hentschel, C. Trends Parasitol. 2006, 22, 301. doi:10.1016/j.pt.2006.05.011 |
| 64. | White, N. J. Science 2008, 320, 330. doi:10.1126/science.1155165 |
| 65. | Nakase, I.; Lai, H.; Singh, N. P.; Sasaki, T. Int. J. Pharm. 2008, 354, 28. doi:10.1016/j.ijpharm.2007.09.003 |
| 66. | Nosten, F.; White, N. J. Am. J. Trop. Med. Hyg. 2007, 77 (Suppl. 6), 181. |
| 67. | Stocks, P. A.; Bray, P. G.; Barton, V. E.; Al-Helal, M.; Jones, M.; Araujo, N. C.; Gibbons, P.; Ward, S. A.; Hughes, R. H.; Biagini, G. A.; Davies, J.; Amewu, R.; Mercer, A. E.; Ellis, G.; ONeill, P. M. Angew. Chem., Int. Ed. 2007, 46, 6278. doi:10.1002/anie.200604697 |
| 68. | Checkley, A. M.; Whitty, C. J. M. Expert Rev. Anti-Infect. Ther. 2007, 5, 199. doi:10.1586/14787210.5.2.199 |
| 69. | Tilley, L.; Davis, T. M. E.; Bray, P. G. Future Microbiol. 2006, 1, 127. doi:10.2217/17460913.1.1.127 |
| 70. | Gelb, M. H. Curr. Opin. Chem. Biol. 2007, 11, 440. doi:10.1016/j.cbpa.2007.05.038 |
| 71. | Namdeo, A. G.; Mahadik, K. R.; Kadam, S. S. Pharm. Mag. 2006, 2, 106. |
| 72. | Haynes, R. K. Curr. Top. Med. Chem. (Sharjah, United Arab Emirates) 2006, 6, 509. doi:10.2174/156802606776743129 |
| 73. | Begue, J.-P.; Bonnet-Delpon, D. Drugs Future 2005, 30, 509. doi:10.1358/dof.2005.030.05.901987 |
| 74. | Rosenthal, A. S.; Chen, X.; Liu, J. O.; West, D. C.; Hergenrother, P. J.; Shapiro, T. A.; Posner, G. H. J. Med. Chem. 2009, 52, 1198. doi:10.1021/jm801484v |
| 75. | Nagelschmitz, J.; Voith, B.; Wensing, G.; Roemer, A.; Fugmann, B.; Haynes, R. K.; Kotecka, B. M.; Rieckmann, K. H.; Edstein, M. D. Antimicrob. Agents Chemother. 2008, 52, 3085. doi:10.1128/AAC.01585-07 |
| 76. | Van der Meersch, H. J. Pharm. Belg. 2005, 60, 23. |
| 77. | Vennerstrom, J. L.; Arbe-Barnes, S.; Brun, R.; Charman, S. A.; Chiu, F. C. K.; Chollet, J.; Dong, Y.; Dorn, A.; Hunziker, D.; Matile, H.; McIntosh, K.; Padmanilayam, M.; Santo Tomas, J.; Scheurer, C.; Scorneaux, B.; Tang, Y.; Urwyler, H.; Wittlin, S.; Charman, W. N. Nature 2004, 430, 900. doi:10.1038/nature02779 |
| 78. | Valecha, N.; Looareesuwan, S.; Martensson, A.; Abdulla, S. M.; Krudsood, S.; Tangpukdee, N.; Mohanty, S.; Mishra, S. K.; Tyagi, P. K.; Sharma, S. K.; Moehrle, J.; Gautam, A.; Roy, A.; Paliwal, J. K.; Kothari, M.; Saha, N.; Dash, A. P.; Bjorkman, A. Clin. Infect. Dis. 2010, 51, 684. doi:10.1086/655831 |
| 79. | Uhlemann, A. C.; Wittlin, S.; Matile, H.; Bustamante, L. Y.; Krishna, S. Antimicrob. Agents Chemother. 2007, 51, 667. doi:10.1128/AAC.01064-06 |
| 80. | Gupta, A.; Singh, Y.; Srinivas, K. S.; Jain, G.; Sreekumar, V. B.; Semwal, V. P. J. Pharm. BioAllied Sci. 2010, 2, 32. doi:10.4103/0975-7406.62706 |
| 81. | Dong, Y.; Wittlin, S.; Sriraghavan, K.; Chollet, J.; Charman, S. A.; Charman, W. N.; Scheurer, C.; Urwyler, H.; Santo Tomas, J.; Snyder, C.; Creek, D. L.; Morizzi, J.; Koltun, M.; Matile, H.; Wang, X.; Padmanilayam, M.; Tang, Y.; Dorn, A.; Brun, R.; Vennerstrom, J. L. J. Med. Chem. 2010, 53, 481. doi:10.1021/jm901473s |
| 402. | Borsarelli, C. D.; Mischne, M.; La Venia, A.; Morán Vieyra, F. E. Photochem. Photobiol. 2007, 83, 1313. doi:10.1111/j.1751-1097.2007.00147.x |
| 401. | Tang, Y.; Cole, K. P.; Buchanan, G. S.; Li, G.; Hsung, R. P. Org. Lett. 2009, 11, 1591. doi:10.1021/ol900237e |
| 322. | Murakami, N.; Kawanishi, M.; Itagaki, S.; Horii, T.; Kobayashi, M. Tetrahedron Lett. 2001, 42, 7281. doi:10.1016/S0040-4039(01)01493-9 |
| 323. | Murakami, N.; Kawanishi, M.; Itagaki, S.; Horii, T.; Kobayashi, M. Bioorg. Med. Chem. Lett. 2002, 12, 69. doi:10.1016/S0960-894X(01)00673-4 |
| 405. | Tang, Y.; Dong, Y.; Wang, X.; Sriraghavan, K.; Wood, J. K.; Vennerstrom, J. L. J. Org. Chem. 2005, 70, 5103. doi:10.1021/jo050385+ |
| 252. | Ghorai, P.; Dussault, P. H.; Hu, C. Org. Lett. 2008, 10, 2401. doi:10.1021/ol800657m |
| 405. | Tang, Y.; Dong, Y.; Wang, X.; Sriraghavan, K.; Wood, J. K.; Vennerstrom, J. L. J. Org. Chem. 2005, 70, 5103. doi:10.1021/jo050385+ |
| 104. | Kawanishi, M.; Kotoku, N.; Itagaki, S.; Horii, T.; Kobayashi, M. Bioorg. Med. Chem. 2004, 12, 5297. doi:10.1016/j.bmc.2004.04.051 |
| 105. | Taglialatela-Scafati, O.; Fattorusso, E.; Romano, A.; Scala, F.; Barone, V.; Cimino, P.; Stendardo, E.; Catalanotti, B.; Persico, M.; Fattorusso, C. Org. Biomol. Chem. 2010, 8, 846. doi:10.1039/b918600j |
| 106. | Christoffers, J.; Werner, T.; Roessle, M. Catal. Today 2007, 121, 22. doi:10.1016/j.cattod.2006.11.008 |
| 407. | Sabbani, S.; Stocks, P. A.; Ellis, G. L.; Davies, J.; Hedenstrom, E.; Ward, S. A.; O’Neill, P. M. Bioorg. Med. Chem. Lett. 2008, 18, 5804. doi:10.1016/j.bmcl.2008.09.052 |
| 324. | Murakami, N.; Kawanishi, M.; Itagaki, S.; Horii, T.; Kobayashi, M. Bioorg. Med. Chem. Lett. 2004, 14, 3513. doi:10.1016/j.bmcl.2004.04.086 |
| 406. | Erhardt, S.; Macgregor, S. A.; McCullough, K. J.; Savill, K.; Taylor, B. J. Org. Lett. 2007, 9, 5569. doi:10.1021/ol702534d |
| 318. | Tokuyasu, T.; Kunikawa, S.; Abe, M.; Masuyama, A.; Nojima, M.; Kim, H.-S.; Begum, K.; Wataya, Y. J. Org. Chem. 2003, 68, 7361. doi:10.1021/jo030107f |
| 404. | O’Neill, P. M.; Hindley, S.; Pugh, M. D.; Davies, J.; Bray, P. G.; Park, B. K.; Kapu, D. S.; Ward, S. A.; Stocks, P. A. Tetrahedron Lett. 2003, 44, 8135. doi:10.1016/j.tetlet.2003.09.033 |
| 318. | Tokuyasu, T.; Kunikawa, S.; Abe, M.; Masuyama, A.; Nojima, M.; Kim, H.-S.; Begum, K.; Wataya, Y. J. Org. Chem. 2003, 68, 7361. doi:10.1021/jo030107f |
| 319. | Wu, J.-M.; Kunikawa, S.; Tokuyasu, T.; Masuyama, A.; Nojima, M.; Kim, H.-S.; Wataya, Y. Tetrahedron 2005, 61, 9961. doi:10.1016/j.tet.2005.08.025 |
| 403. | O’Neil, P. M.; Pugh, M.; Davies, J.; Ward, S. A.; Park, B. K. Tetrahedron Lett. 2001, 42, 4569. doi:10.1016/S0040-4039(01)00791-2 |
| 405. | Tang, Y.; Dong, Y.; Wang, X.; Sriraghavan, K.; Wood, J. K.; Vennerstrom, J. L. J. Org. Chem. 2005, 70, 5103. doi:10.1021/jo050385+ |
| 320. | Gemma, S.; Martí, F.; Gabellieri, E.; Campiani, G.; Novellino, E.; Butini, S. Tetrahedron Lett. 2009, 50, 5719. doi:10.1016/j.tetlet.2009.07.137 |
| 321. | Gemma, S.; Gabellieri, E.; Coccone, S. S.; Martí, F.; Taglialatela-Scafati, O.; Novellino, E.; Campiani, G.; Butini, S. J. Org. Chem. 2010, 75, 2333. doi:10.1021/jo1001559 |
| 153. | Laurent, S. A.-L.; Boissier, J.; Coslédan, F.; Gornitzka, H.; Robert, A.; Meunier, B. Eur. J. Org. Chem. 2008, 895. doi:10.1002/ejoc.200700975 |
| 154. | Jung, M.; Lee, K.; Kim, H.; Park, M. Curr. Med. Chem. 2004, 11, 1265. doi:10.2174/0929867043365233 |
| 155. | Xiao, S.-h.; Mei, J.-y.; Jiao, P.-y. Parasitol. Res. 2011, 108, 431. doi:10.1007/s00436-010-2084-7 |
| 156. | Boissier, J.; Coslédan, F.; Robert, A.; Meunier, B. Antimicrob. Agents Chemother. 2009, 53, 4903. doi:10.1128/AAC.00640-09 |
| 157. | Pradines, V.; Portela, J.; Boissier, J.; Cosledan, F.; Meunier, B.; Robert, A. Antimicrob. Agents Chemother. 2011, 55, 2403. doi:10.1128/AAC.00082-11 |
| 158. | Maurya, R.; Soni, A.; Anand, D.; Ravi, M.; Raju, K. S. R.; Taneja, I.; Naikade, N. K.; Puri, S. K.; Wahajuddin, K.; Anojiya, S.; Yadav, P. P. Med. Chem. Lett. 2013, 4, 165. doi:10.1021/ml300188t |
| 147. | Ingram, K.; Schiaffo, C. E.; Sittiwong, W.; Benner, E.; Dussault, P. H.; Keiser, J. J. Antimicrob. Chemother. 2012, 67, 1979. doi:10.1093/jac/dks141 |
| 148. | Dong, Y.; Chollet, J.; Vargas, M.; Mansour, N. R.; Bickle, Q.; Alnouti, Y.; Huang, J.; Keiser, J.; Vennerstrom, J. L. Bioorg. Med. Chem. Lett. 2010, 20, 2481. doi:10.1016/j.bmcl.2010.03.001 |
| 149. | Mohamed, A. E.-H. H.; El-Sayed, M. A.; Hegazy, M. E.; Helaly, S. E.; Esmail, A. M.; Mohamed, N. S. Rec. Nat. Prod. 2010, 4, 1. |
| 150. | Yang, Z.-S.; Wu, W.-M.; Li, Y.; Wu, Y.-L. Helv. Chim. Acta 2005, 88, 2865. doi:10.1002/hlca.200590229 |
| 151. | Keiser, J.; Utzinger, J. Adv. Parasitol. 2010, 73, 197. doi:10.1016/S0065-308X(10)73008-6 |
| 152. | Dembitsky, V.; Shkrob, I.; Hanus, L. O. Biomed. Pap. 2008, 152, 209. doi:10.5507/bp.2008.032 |
| 120. | Amewu, R.; Gibbons, P.; Mukhtar, A.; Stachulski, A. V.; Ward, S. A.; Hall, C.; Rimmer, K.; Davies, J.; Vivas, L.; Bacsa, J.; Mercer, A. E.; Nixon, G.; Stocks, P. A.; O’Neill, P. M. Org. Biomol. Chem. 2010, 8, 2068. doi:10.1039/b924319d |
| 121. | Chauhan, S. S.; Sharma, M.; Chauhan, P. M. S. Drug News Perspect. 2010, 23, 632. doi:10.1358/dnp.2010.23.10.1468390 |
| 122. | Fernandez, I.; Robert, A. Org. Biomol. Chem. 2011, 9, 4098. doi:10.1039/c1ob05088e |
| 123. | Singh, C.; Verma, V. P.; Naikade, N. K.; Singh, A. S.; Hassam, M.; Puri, S. K. J. Med. Chem. 2008, 51, 7581. doi:10.1021/jm801006v |
| 124. | Brown, G. D. Molecules 2010, 15, 7603. doi:10.3390/molecules15117603 |
| 125. | Singh, C.; Sharma, U.; Saxena, G.; Puri, S. Bioorg. Med. Chem. Lett. 2007, 17, 4097. doi:10.1016/j.bmcl.2007.05.055 |
| 126. | Singh, C.; Singh, A. S.; Naikade, N. K.; Verma, V. P.; Hassam, M.; Gupta, N.; Pandey, S. Synthesis 2010, 1014. doi:10.1055/s-0029-1218639 |
| 127. | Hencken, C. P.; Kalinda, A. S.; Gaetano D'Angelo, J. Annu. Rep. Med. Chem. 2009, 44, 359. doi:10.1016/S0065-7743(09)04418-2 |
| 403. | O’Neil, P. M.; Pugh, M.; Davies, J.; Ward, S. A.; Park, B. K. Tetrahedron Lett. 2001, 42, 4569. doi:10.1016/S0040-4039(01)00791-2 |
| 128. | Šolaja, B. A.; Terzić, N.; Pocsfalvi, G.; Gerena, L.; Tinant, B.; Opsenica, D.; Milhous, W. K. J. Med. Chem. 2002, 45, 3331. doi:10.1021/jm020891g |
| 129. | Kirchhofer, C.; Vargas, M.; Braissant, O.; Dong, Y.; Wang, X.; Vennerstrom, J. L.; Keiser, J. Acta Trop. 2011, 118, 56. doi:10.1016/j.actatropica.2011.02.003 |
| 130. | Vennerstrom, J. L.; Fu, H. N.; Ellis, W. Y.; Ager, A. L., Jr.; Wood, J. K.; Andersen, S. L.; Gerena, L.; Milhous, W. K. J. Med. Chem. 1992, 35, 3023. doi:10.1021/jm00094a015 |
| 131. | Hamann, H.-J.; Hecht, M.; Bunge, A.; Gogol, M.; Liebscher, J. Tetrahedron Lett. 2011, 52, 107. doi:10.1016/j.tetlet.2010.10.151 |
| 132. | Opsenica, I.; Terzić, N.; Opsenica, D.; Angelovski, G.; Lehnig, L.; Eilbracht, P.; Tinant, B.; Juranić, Z.; Smith, K. S.; Yang, Y. S.; Diaz, D. S.; Smith, P. L.; Milhous, W. K.; Doković, D.; Šolaja, B. A. J. Med. Chem. 2006, 49, 3790. doi:10.1021/jm050966r |
| 133. | Dong, Y.; McCullough, K. J.; Wittlin, S.; Chollet, J.; Vennerstrom, J. L. Bioorg. Med. Chem. Lett. 2010, 20, 6359. doi:10.1016/j.bmcl.2010.09.113 |
| 134. | Cvijetic, I. N.; Zizak, Z. P.; Stanojkovic, T. P.; Juranic, Z. D.; Terzic, N.; Opsenica, I. M.; Opsenica, D. M.; Juranic, I. O.; Drakulic, B. J. Eur. J. Med. Chem. 2010, 45, 4570. doi:10.1016/j.ejmech.2010.07.019 |
| 135. | Opsenica, I.; Opsenica, D.; Lanteri, C. A.; Anova, L.; Milhous, W. K.; Smith, K. S.; Solaja, B. A. J. Med. Chem. 2008, 51, 6216. doi:10.1021/jm8006905 |
| 136. | Bhattacharjee, A. K.; Carvalho, K. A.; Opsenica, D.; Solaja, B. A. J. Serb. Chem. Soc. 2005, 70, 329. doi:10.2298/JSC0503329B |
| 137. | Tonmunphean, S.; Wijitkosoom, A.; Tantirungrotechai, Y. Bioorg. Med. Chem. 2004, 12, 2005. doi:10.1016/j.bmc.2004.03.003 |
| 138. | Ellis, G. L.; Amewu, R.; Sabbani, S.; Stocks, P. A.; Shone, A.; Stanford, D.; Gibbons, P.; Davies, J.; Vivas, L.; Charnaud, S.; Bongard, E.; Hall, C.; Rimmer, K.; Lozanom, S.; Jesús, M.; Gargallo, D.; Ward, S. A.; O’Neill, P. M. J. Med. Chem. 2008, 51, 2170. doi:10.1021/jm701435h |
| 139. | Liu, H.-H.; Wu, Y.-K.; Shen, X. Chin. J. Chem. 2003, 21, 875. doi:10.1002/cjoc.20030210731 |
| 140. | Dong, Y. Mini-Rev. Med. Chem. 2002, 2, 113. |
| 141. | Opsenica, D. M.; Terzić, N.; Smith, P. L.; Yang, Y.; Anova, L.; Smith, K. S.; Solaja, B. A. Bioorg. Med. Chem. 2008, 16, 7039. doi:10.1016/j.bmc.2008.05.017 |
| 142. | Terzić, N.; Opsenica, D.; Milić, D.; Tinan, B.; Smith, K. S.; Milhous, W. K.; Šolaja, B. A. J. Med. Chem. 2007, 50, 5118. doi:10.1021/jm070684m |
| 143. | Pis Diez, R.; Jubert, A. H. J. Mol. Struct.: THEOCHEM 2000, 499, 85. doi:10.1016/S0166-1280(99)00281-X |
| 144. | Amewu, R.; Stachulski, A. V.; Ward, S. A.; Berry, N. G.; Bray, P. G.; Davies, J.; Labat, G.; Vivas, L.; O’Neill, P. M. Org. Biomol. Chem. 2006, 4, 4431. doi:10.1039/b613565j |
| 145. | Dong, Y.; Matile, H.; Chollet, J.; Kaminsky, R.; Wood, J. K.; Vennerstrom, J. L. J. Med. Chem. 1999, 42, 1477. doi:10.1021/jm980698f |
| 146. | Kumura, N.; Furukawa, H.; Kobayashi, M.; Onyango, A. N.; Izumi, M.; Nakajima, S.; Kim, H.-S.; Wataya, Y.; Baba, N. Biosci., Biotechnol., Biochem. 2009, 73, 217. doi:10.1271/bbb.80571 |
| 102. | O’Neill, P. M.; Stocks, P. A.; Pugh, M. D.; Araujo, N. C.; Korshin, E. E.; Bickley, J. F.; Ward, S. A.; Bray, P. G.; Pasini, E.; Davies, J.; Verissimo, E.; Bachi, M. D. Angew. Chem., Int. Ed. 2004, 43, 4193. doi:10.1002/anie.200453859 |
| 103. | Fattorusso, C.; Persico, M.; Calcinai, B.; Cerrano, C.; Parapini, S.; Taramelli, D.; Novellino, E.; Romano, A.; Scala, F.; Fattorusso, E.; Taglialatela-Scafati, O. J. Nat. Prod. 2010, 73, 1138. doi:10.1021/np100196b |
| 104. | Kawanishi, M.; Kotoku, N.; Itagaki, S.; Horii, T.; Kobayashi, M. Bioorg. Med. Chem. 2004, 12, 5297. doi:10.1016/j.bmc.2004.04.051 |
| 105. | Taglialatela-Scafati, O.; Fattorusso, E.; Romano, A.; Scala, F.; Barone, V.; Cimino, P.; Stendardo, E.; Catalanotti, B.; Persico, M.; Fattorusso, C. Org. Biomol. Chem. 2010, 8, 846. doi:10.1039/b918600j |
| 106. | Christoffers, J.; Werner, T.; Roessle, M. Catal. Today 2007, 121, 22. doi:10.1016/j.cattod.2006.11.008 |
| 107. | Bachi, M. D.; Korshin, E. E.; Hoos, R.; Szpilman, A. M.; Ploypradith, P.; Xie, S.; Shapiro, T. A.; Posner, G. H. J. Med. Chem. 2003, 46, 2516. doi:10.1021/jm020584a |
| 108. | Fattorusso, C.; Campiani, G.; Catalanotti, B.; Persico, M.; Basilico, N.; Parapini, S.; Taramelli, D.; Campagnuolo, C.; Fattorusso, E.; Romano, A. J. Med. Chem. 2006, 49, 7088. doi:10.1021/jm060899g |
| 109. | Murakami, N.; Kawanishi, M.; Itagaki, S.; Horii, T.; Kobayashi, M. Bioorg. Med. Chem. Lett. 2002, 12, 69. doi:10.1016/S0960-894X(01)00673-4 |
| 110. | Murakami, N.; Kawanishi, M.; Mostaqul, H. M.; Li, J.; Itagaki, S.; Horii, T.; Kobayashi, M. Bioorg. Med. Chem. Lett. 2003, 13, 4081. doi:10.1016/j.bmcl.2003.08.073 |
| 111. | Liu, H.-H.; Zhang, Q.; Jin, H.-X.; Shen, X.; Wu, Y.-K. Chin. J. Chem. 2006, 24, 1180. doi:10.1002/cjoc.200690221 |
| 112. | Liu, H.-H.; Jin, H.-X.; Zhang, Q.; Wu, Y.-K.; Kim, H.-S.; Wataya, Y. Chin. J. Chem. 2005, 23, 1469. doi:10.1002/cjoc.200591469 |
| 113. | Givelet, C.; Bernat, V.; Danel, M.; André-Barrès, C.; Vial, H. Eur. J. Org. Chem. 2007, 3095. doi:10.1002/ejoc.200700086 |
| 114. | Rudi, A.; Talpir, R.; Kashman, Y.; Benayahu, Y.; Schleyer, M. J. Nat. Prod. 1993, 56, 2178. doi:10.1021/np50102a023 |
| 115. | Ruiz, J.; Tuccio, B.; Lauricella, R.; Maynadier, M.; Vial, H.; André-Barrès, C. Tetrahedron 2013, 69, 6709. doi:10.1016/j.tet.2013.05.099 |
| 116. | Kamata, M.; Hagiwara, J.-i.; Hokari, T.; Suzuki, C.; Fujino, R.; Kobayashi, S.; Kim, H.-S.; Wataya, Y. Res. Chem. Intermed. 2013, 39, 127. doi:10.1007/s11164-012-0637-3 |
| 117. | Najjar, F.; Gorrichon, L.; Baltas, M.; André-Barrès, C.; Vial, H. Org. Biomol. Chem. 2005, 3, 1612. doi:10.1039/b503402g |
| 118. | Kondo, K.; Matsumoto, M.; Hatsutani, M. Patent Jpn. Kokai Tokkyo Koho JP 49061179 A, June 13, 1974. |
| 119. | Bernat, V.; Saffon, N.; Maynadier, M.; Vial, H.; André-Barrès, C. Tetrahedron 2009, 65, 7372. doi:10.1016/j.tet.2009.07.030 |
| 88. | Martyn, D. C.; Ramirez, A. P.; Beattie, M. J.; Cortese, J. F.; Patel, V.; Rush, M. A.; Woerpel, K. A.; Clardy, J. Bioorg. Med. Chem. Lett. 2008, 18, 6521. doi:10.1016/j.bmcl.2008.10.083 |
| 89. | Schiaffo, C. E.; Rottman, M.; Wittlin, S.; Dussault, P. H. ACS Med. Chem. Lett. 2011, 2, 316. |
| 90. | Wang, X.; Dong, Y.; Wittlin, S.; Creek, D.; Chollet, J.; Charman, S. A.; Santo Tomas, J.; Scheurer, C.; Snyder, C.; Vennerstrom, J. L. J. Med. Chem. 2007, 50, 5840. doi:10.1021/jm0707673 |
| 91. | Hartwig, C. L.; Lauterwasser, E. M. W.; Mahajan, S. S.; Hoke, J. M.; Cooper, R. A.; Renslo, A. R. J. Med. Chem. 2011, 54, 8207. doi:10.1021/jm2012003 |
| 92. | Dong, Y.; Chollet, J.; Matile, H.; Charman, S. A.; Chiu, F. C. K.; Charman, W. N.; Scorneaux, B.; Urwyler, H.; Santo Tomas, J.; Scheurer, C.; Snyder, C.; Dorn, A.; Wang, X.; Karle, J. M.; Tang, Y.; Wittlin, S.; Brun, R.; Vennerstrom, J. L. J. Med. Chem. 2005, 48, 4953. doi:10.1021/jm049040u |
| 93. | Dong, Y.; Tan, Y.; Chollet, J.; Matile, H.; Wittlin, S.; Charman, S. A.; Charman, W. N.; Tomas, J. S.; Scheurer, C.; Snyder, C.; Scorneaux, B.; Bajpai, S.; Alexander, S. A.; Wang, X.; Padmanilayam, M.; Cheruku, S. R.; Brun, R.; Vennerstrom, J. L. Bioorg. Med. Chem. 2006, 14, 6368. doi:10.1016/j.bmc.2006.05.041 |
| 94. | Padmanilayam, M.; Scorneaux, B.; Dong, Y.; Chollet, J.; Matile, H.; Charman, S. A.; Creek, D. J.; Charman, W. N.; Tomas, J. S.; Scheurer, C.; Wittlin, S.; Brun, R.; Vennerstrom, J. L. Bioorg. Med. Chem. Lett. 2006, 16, 5542. doi:10.1016/j.bmcl.2006.08.046 |
| 95. | Yadav, G. C.; Dorwal, H. N.; Tanwar, P.; Gahlot, U. B. S. A process for the preparation of 1, 2, 4-trioxolane antimalarials. PCT Int. Appl. WO2010119425 A1, Oct 21, 2010. |
| 96. | Tang, Y.; Dong, Y.; Wittlin, S.; Charman, S. A.; Chollet, J.; Chiu, F. C. K.; Charman, W. N.; Matile, H.; Urwyler, H.; Dorn, A.; Bajpai, S.; Wang, X.; Padmanilayam, M.; Karle, J. M.; Brun, R.; Vennerstrom, J. L. Bioorg. Med. Chem. Lett. 2007, 17, 1260. doi:10.1016/j.bmcl.2006.12.007 |
| 97. | Tang, Y.; Wittlin, S.; Charman, S. A.; Chollet, J.; Chiu, F. C. K.; Morizzi, J.; Johnson, L. M.; Santo Tomas, J.; Scheurer, C.; Snyder, C.; Zhou, L.; Dong, Y.; Charman, W. N.; Matile, H.; Urwyler, H.; Dorn, A.; Vennerstrom, J. L. Bioorg. Med. Chem. Lett. 2010, 20, 563. doi:10.1016/j.bmcl.2009.11.088 |
| 98. | Arora, V. K.; Madan, S.; Trehan, A.; Tyagi, P. Stable dosage forms of spiro and dispiro 1,2,4-trioxolane antimalarials. PCT Int. Appl. WO 2006123314 A2, Nov 23, 2006. |
| 99. | Vennerstrom, J. L.; Dong, Y.; Chollet, J.; Matile, H.; Wang, X.; Sriraghavan, K.; Charman, W. N. Spiro and dispiro 1,2,4-trioxolane antimalarials. U. S. Pat. Appl. US 20050256185 A1, Nov 17, 2005. |
| 100. | Cazelles, J.; Cosledan, F.; Meunier, B.; Pellet, A. Dual molecules containing peroxy derivative, the synthesis and therapeutic applications thereof. Patent Fr. Demande FR 2862304 A1, May 20, 2005. |
| 101. | Vennerstrom, J. L.; Dong, Y.; Chollet, J.; Matile, H.; Padmanilayam, H.; Tang, Y.; Charman, N. W. Spiro and dispiro 1,2,4-trioxolane antimalarials. U.S. Pat. Appl. US 20040186168 A1, Sept 23, 2004. |
© 2014 Terent'ev et al; licensee Beilstein-Institut.
This is an Open Access article under the terms of the Creative Commons Attribution License (http://creativecommons.org/licenses/by/2.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
The license is subject to the Beilstein Journal of Organic Chemistry terms and conditions: (http://www.beilstein-journals.org/bjoc)