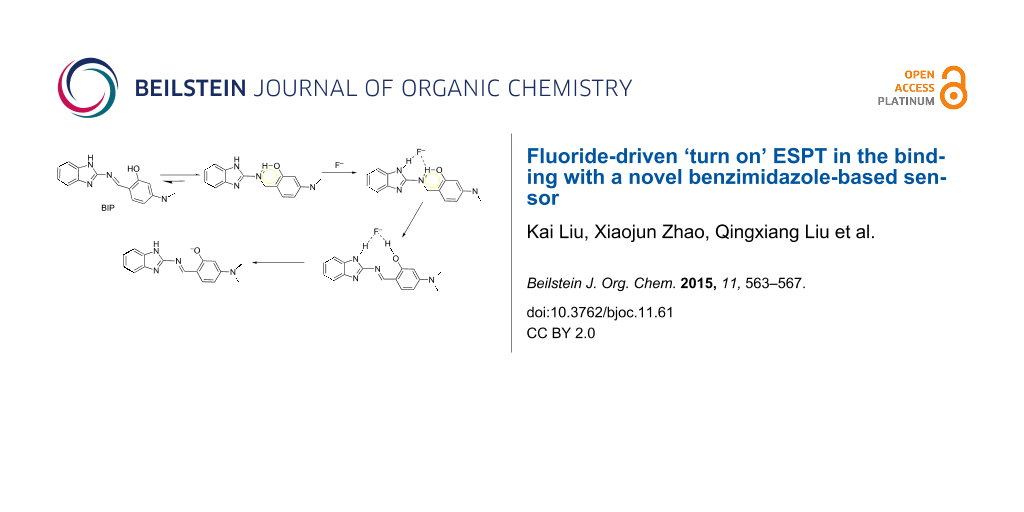
Abstract
A novel fluorescence sensor (BIP) bearing NH and OH subunits displayed a highly selective and sensitive recognition property for fluoride over other anions. Fluoride-driven ESPT, poorly used in anion recognition and sensing, was suggested to be responsible for the fluorescence enhancement with a blue shift of 35 nm in the emission spectrum.
Graphical Abstract
Introduction
Design and synthesis of selective and efficient sensors for various anions involved in biological, industrial and environmental processes have drawn a lot of attention [1-3]. In recent years, much effort has been devoted to the development of anion fluorescent sensors [4]. Of particular interest concerning anion recognition and sensing was fluoride [5-7], as it is important to human health (for example, dental care, osteoporosis and osteosarcoma). To realize the high selectivity and sensitivity to fluoride, many anion fluorescent sensors have been designed based upon numerous signal mechanisms [4,8-10]. However, excited-state intra/intermolecular proton transfer (ESPT), as an extensively exploited mechanism in many biological and chemical processes, has been employed poorly in anion recognition and sensing [2,11-16]. In the ESPT molecules, a five or six-membered intramolecular hydrogen-bonded ring formed, and a proton/hydrogen atom is transferred to an electronegative atom at the excited state [2,17,18]. By regulating the proton acidity of the H-bond donor which plays a vital role in the ESPT process, the biologically and chemically important anion would be distinguished by the different optical outputs for the interaction between sensor and anion [2,14,15,19].
During the last few years, various types of artificial sensors have been developed for anion recognition and sensing [1,4,14,15,19,20]. Among these, the hydrogen bond, especially NH [21-24], was particularly useful and effective [1,3,14,15,19,20]. However, to OH-based anion sensors it has been paid less attention [25,26]. In fact, we have succeeded in designing and synthesizing the anion fluorescent probes, based on ESPT [15,16], containing phenolic OH as H-bond donor. In these systems, the probe displayed a narrower linear range for fluoride. Furthermore, to the best of our knowledge, there were few reports on the anion sensors containing both NH and OH binding sites [14,25,27]. On the foundation of the above-mentioned background, herein we report the synthesis and H-bond donor ability of the novel anion sensor (E)-2-(((1H-benzo[d]imidazol-2-yl)imino)methyl)-5-(dimethylamino)phenol (BIP) made up of benzimidazole and phenol units with a C=N linker. Although BIP is weakly fluorescent, its anion-binding adduct is expected to be highly fluorescent by opening up ESPT channel [2,14,15,19]. In this paper, we demonstrated that BIP displayed selectively fluorescence enhancement for fluoride by ESPT. Importantly, BIP displayed a fluoride-selective fluorescence response with a wider linear range from 0.04 to 1.2 μM.
Results and Discussion
BIP was synthesized by the condensation of 2-aminobenzimidazole and 4-(dimethylamino)salicylaldehyde according to the literature [28,29] with little modification, as displayed in Scheme 1. It was well characterized by 1H NMR, mass spectrometry and elemental analysis before its practical application.
The anion binding and sensing properties of BIP were first evaluated by UV–vis studies. There were three characteristic bands centered at 410, 276 and 228 nm in CH3CN. Among these, the band in the region around 276 and 228 nm is attributed to the excitation of π electrons localized at the C=N bond and at the aromatic system [30], and the peak at 410 nm was ascribed to the intramolecular charge transition between the phenol and benzimidazole [2,30]. Upon addition of F− (as tetrabutylammonium salts, TBA salts), as shown in Figure 1, the intensity of the band at 410 nm decreased and broadened, and a new shoulder around 466 nm formed, meanwhile, the shoulder peak at 228 nm increased. However, no noticeable spectral changes were found for other anions (Cl−, Br−, I−, H2PO4−, NO3−, AcO−, added as their TBA salts). This indicated the poor interaction between BIP and these anions.
![[1860-5397-11-61-1]](/bjoc/content/figures/1860-5397-11-61-1.png?scale=2.0&max-width=1024&background=FFFFFF)
Figure 1: Spectral changes of BIP (4 × 10−5 M) in CH3CN after the addition of 50 equiv of anions (as their tetrabutylammonium salts).
Figure 1: Spectral changes of BIP (4 × 10−5 M) in CH3CN after the addition of 50 equiv of anions (as their te...
The interaction of BIP with fluoride was further investigated by UV–vis spectroscopic titration. As seen from Figure 2, the spectral changes with two well-defined isosbestic points (344, 270 nm) indicated the formation of a single complex between BIP and fluoride [2]. Evidence for 1:1 complex formation was also validated by non-linear least-square analysis [31] for BIP with F−. The association constant, calculated from the UV−vis plot at 410 nm as the function of anion concentration [15], was 1.25 × 104 (R = 0.9926) for F− (see Figure S1 in Supporting Information File 1). Due to the little spectral changes of BIP upon other anions (Cl−, Br−, I−, NO3−, H2PO4−, AcO−, added as TBA salts), as displayed in Figure 1, the binding constants cannot be determined. Furthermore, the similar absorption was found for BIP with F− and OH− (see Figure S4 in Supporting Information File 1), which suggested the occurrence of deprotonation during the titration process.
![[1860-5397-11-61-2]](/bjoc/content/figures/1860-5397-11-61-2.png?scale=2.0&max-width=1024&background=FFFFFF)
Figure 2: UV−vis absorption spectra of BIP (4 × 10−5 M) in CH3CN after the addition of 0–50 equiv of TBAF. Insert: Non-linear curve fit of absorbance changes at 410 nm as a function of fluoride concentration.
Figure 2: UV−vis absorption spectra of BIP (4 × 10−5 M) in CH3CN after the addition of 0–50 equiv of TBAF. In...
The mechanism of the UV–vis response for F− was further assessed by 1H NMR studies, which were conducted in DMSO-d6. The characteristic peak of BIP at 12.85 ppm suggested the formation of an intramolecular hydrogen bond in BIP itself between the imine nitrogen and the phenolic OH proton [32-34], that progressively disappeared with the successive addition of F− (see Figure S5 and Figure S6 in Supporting Information File 1). This indicated that the proton transfer from BIP to F− [1]. Meanwhile, the NH signal of BIP at 12.41 ppm was shifted downfield, broadened and weakened, which was ascribed to the formation of an intermolecular hydrogen bond between F− and the above-mentioned proton in BIP.
To further explore the photophysical sensing properties of BIP with various anions (F−, Cl−, Br−, I−, H2PO4−, NO3−, AcO−, added as TBA salts), fluorescence measurements were carried out. The weak fluorescence emission at 411 nm, corresponding to the enol form of a Schiff base [32-34], suggested the formation of the geometrically restricted six-membered intramolecular hydrogen bond ring in the BIP at transition state [32-34]. As displayed in Figure 3, with gradual addition of F− to the CH3CN solution of BIP, the fluorescence intensity at 411 nm increased slowly. This indicated the occurrence of hydrogen-bonding interaction between BIP and F−. Moreover, the acidity of the OH group of BIP was expected to be enhanced by anion binding, and the ESPT channel would be open [8-10,15]. The subsequent spectral behavior proved our assumption. The new peak at 376 nm occurred and reached a limiting value after the addition of 20 equiv F−. The fluoride-driven ESPT displayed a fluorescence enhancement and a dramatically blue-shift with 35 nm in the emission spectrum.
![[1860-5397-11-61-3]](/bjoc/content/figures/1860-5397-11-61-3.png?scale=2.0&max-width=1024&background=FFFFFF)
Figure 3: Emission spectral changes of BIP (4 × 10−7 M) in CH3CN upon addition of 0–20 equiv TBAF. Insert: Linear plot of emission intensity at 376 nm against fluoride concentration.
Figure 3: Emission spectral changes of BIP (4 × 10−7 M) in CH3CN upon addition of 0–20 equiv TBAF. Insert: Li...
Analogous investigations were carried out with Cl−, Br−, I−, NO3−, H2PO4− and AcO−. As shown in Figure 4, the fluorescence emission hardly experienced any perturbation on adding a similar amount of anions as done with F−. Also, the detection of F− displayed no interference in presence of other competitive anions (See Figure S7 in Supporting Information File 1). This demonstrated BIP has a high selectivity for F−.
![[1860-5397-11-61-4]](/bjoc/content/figures/1860-5397-11-61-4.png?scale=2.0&max-width=1024&background=FFFFFF)
Figure 4: Emission spectral changes of BIP (4 × 10−7 M) in CH3CN after the addition of 20 equiv of anions (added as their TBA salts).
Figure 4: Emission spectral changes of BIP (4 × 10−7 M) in CH3CN after the addition of 20 equiv of anions (ad...
The binding constant of BIP with F− was also determined by nonlinear regression based on spectrofluorometric titration. Good nonlinear curve fits (R = 0.9907) also indicated the formation of an 1:1 stoichiometric complex of BIP with F− (see Figure S2 in Supporting Information File 1). The association constant was 8.79 × 105 M−1, which was bigger than that obtained from UV–vis spectroscopy. This suggested the enhancement of the phenolic OH acidity by the photoexcitation [2,11-15]. Moreover, fluoride-dependent fluorescent intensity at 376 nm displayed an excellent linearity (R = 0.9880) in the concentration range of 0.04–1.2 μM (insert Figure 3 and Figure S3 in Supporting Information File 1). The limit of detection for F− was found to be 0.021 μM, which is lower than 0.10 μM set by the US Environmental Protection Agency. This suggested that BIP had a good sensitivity for fluoride and has the potential utility to detect fluoride.
Based on the aforementioned UV–vis, spectrofluorimetric and 1H NMR studies, the possible mechanism of F− recognition by BIP was proposed and illustrated in Scheme 2. On binding of F−, the isomerization of BIP was restricted, which enhanced the intramolecular charge transfer interaction between the assembly of binding sites with F− to the chromophore [1]. With further addition of F−, intermolecular hydrogen bond formation disturbed the intramolecular hydrogen bonding and destroyed the geometrically restricted conformation [2], at the same time, the new geometrically restricted intermolecular H-bonding ring formed. Subsequent the fluoride-triggered ESPT occurred, which displayed ‘turn-on’ fluorescence.
Scheme 2: Possible binding mode between BIP and fluoride.
Scheme 2: Possible binding mode between BIP and fluoride.
Conclusion
In conclusion, a new benzimidazole-based BIP has been developed which is selective for the recognition of F− also in the presence of anions with similar basicity and surface charge density such as AcO− and H2PO4−. Fluorescence spectral changes were driven by original hydrogen bonding interaction between BIP and F−, and the subsequent ESPT which resulted in the ‘turn-on’ of fluorescence. The strategy for the design of simple sensors to detect the important neutral and anionic guest may contribute to design more efficient anion chemosensors.
Experimental
Synthesis of compound 1: 4-(Dimethylamino)salicylaldehyde was synthesized according to the literature [15,16] with little modification. POCl3 (4.0 mL, 43.2 mmol) was added dropwise to dry DMF (21.0 mL, 271.8 mmol) containing 3-(dimethylamino)phenol (3.10 g, 22.6 mmol) at 0 °C, and the mixture was stirred for 10 min, slowly warmed to room temperature and stirred for another 30 min. The reaction mixture was heated at 80 °C overnight. After cooling to room temperature, the mixture was poured into ice cold water. The solution was neutralized with saturated Na2CO3. The precipitate was washed several times with distilled water, and dried under vacuum to yield 3.30 g of 1. 1H NMR (400 MHz, CDCl3) δ 3.09 (s, 6H, CH3), 6.10–7.29 (m, 3H, Ar-H), 9.53 (s, 1H, OH), 11.61 (s, 1H, CHO). This was consistent with the literature.
Synthesis of compound BIP: 2-Aminobenzimidazole (0.80 g, 6 mmol) and compound 1 (0.99 g, 6 mmol) were dissolved in 25 mL ethanol. The resulting solution was heated at reflux for 4 h. The solvent was removed by a rotary evaporator at reduced pressure. The crude product was purified by silica gel column chromatography (ethyl acetate/n-hexane 1:3) and recrystallized with ethanol to yield yellow solid. Yield: 1.03 g (61.2%); 1H NMR (400 MHz, DMSO-d6) δ 3.05 (s, 6H, CH3), 6.16–7.53 (m, 7H, Ar-H), 9.34 (s, 1H, CH=N), 12.41 (s, 1H, NH), 12.85 (s, 1H, OH); ESIMS: 280.1408 (M + H+).
Supporting Information
| Supporting Information File 1: Data analysis of QP after the addition of fluoride. | ||
| Format: PDF | Size: 664.6 KB | Download |
References
-
Mao, S.; Liu, K.; Lu, F.; Du, L. Mini-Rev. Org. Chem. 2010, 7, 221–229. doi:10.2174/157019310791384137
Return to citation in text: [1] [2] [3] [4] [5] -
Liu, K.; Huo, J.; Zhu, B.; Huo, R. J. Fluoresc. 2012, 22, 1231–1236. doi:10.1007/s10895-012-1063-z
Return to citation in text: [1] [2] [3] [4] [5] [6] [7] [8] [9] -
Amendola, V.; Fabbrizzi, L.; Mosca, L. Chem. Soc. Rev. 2010, 39, 3889–3915. doi:10.1039/b822552b
Return to citation in text: [1] [2] -
Wu, J.; Liu, W.; Ge, J.; Zhang, H.; Wang, P. Chem. Soc. Rev. 2011, 40, 3483–3495. doi:10.1039/C0CS00224K
Return to citation in text: [1] [2] [3] -
Jung, H. S.; Kim, H. J.; Vicens, J.; Kim, J. S. Tetrahedron Lett. 2009, 50, 983–987. doi:10.1016/j.tetlet.2008.12.026
Return to citation in text: [1] -
Lee, M. H.; Cao, Q.-Y.; Kim, S. K.; Sessler, J. L.; Kim, J. S. J. Org. Chem. 2011, 76, 870–874. doi:10.1021/jo1021713
Return to citation in text: [1] -
Zhang, J. F.; Lim, C. S.; Bhuniya, S.; Cho, B. R.; Kim, J. S. Org. Lett. 2011, 13, 1190–1193. doi:10.1021/ol200072e
Return to citation in text: [1] -
Sahoo, S. K.; Bera, R. K.; Kanungo, B. K.; Baral, M. Spectrochim. Acta, Part A: Mol. Biomol. Spectrosc. 2012, 89, 322–328. doi:10.1016/j.saa.2011.12.049
Return to citation in text: [1] [2] -
Sharma, D.; Sahoo, S. K.; Chaudhary, S.; Bera, R. K.; Callan, J. F. Analyst 2013, 138, 3646–3650. doi:10.1039/C3AN00199G
Return to citation in text: [1] [2] -
Yadav, U. N.; Pant, P.; Sharma, D.; Sahoo, S. K.; Shankarling, G. S. Sens. Actuators, B 2014, 197, 73–80. doi:10.1016/j.snb.2014.02.075
Return to citation in text: [1] [2] -
Solntsev, K. M.; Bartolo, E.-A.; Pan, G.; Muller, G.; Bommireddy, S.; Huppert, D.; Tolbert, L. M. Isr. J. Chem. 2009, 49, 227–233. doi:10.1560/ijc.49.2.227
Return to citation in text: [1] [2] -
Song, P.; Ding, J.-X.; Chu, T.-S. Spectrochim. Acta, Part A: Mol. Biomol. Spectrosc. 2012, 97, 746–752. doi:10.1016/j.saa.2012.07.010
Return to citation in text: [1] [2] -
Zhao, Y.-q.; Zhang, B.-q.; Duan, C.-y.; Lin, Z.-h.; Meng, Q.-j. New J. Chem. 2006, 30, 1207–1213. doi:10.1039/b605087e
Return to citation in text: [1] [2] -
Kanagaraj, K.; Pitchumani, K. Chem. – Asian J. 2014, 9, 146–152. doi:10.1002/asia.201300816
Return to citation in text: [1] [2] [3] [4] [5] [6] [7] -
Liu, K.; Zhao, X.; Liu, Q.; Huo, J.; Fu, H.; Wang, Y. J. Photochem. Photobiol., B: Biol. 2014, 138, 75–79. doi:10.1016/j.jphotobiol.2014.05.004
Return to citation in text: [1] [2] [3] [4] [5] [6] [7] [8] [9] [10] -
Liu, K.; Zhao, X.; Liu, Q.; Huo, J.; Wang, X.; Wu, Y. Spectrochim. Acta, Part A: Mol. Biomol. Spectrosc. 2015, 140, 198–201. doi:10.1016/j.saa.2014.12.092
Return to citation in text: [1] [2] [3] -
Wu, F.; Ma, L.; Zhang, S.; Wang, Z.; Cheng, X. Mater. Lett. 2014, 116, 231–234. doi:10.1016/j.matlet.2013.11.017
Return to citation in text: [1] -
Chipem, F. A. S.; Krishnamoorthy, G. J. Phys. Chem. B 2013, 117, 14079–14088. doi:10.1021/jp405804c
Return to citation in text: [1] -
Goswami, S.; Manna, A.; Paul, S.; Das, A. K.; Nandi, P. K.; Maity, A. K.; Saha, P. Tetrahedron Lett. 2014, 55, 490–494. doi:10.1016/j.tetlet.2013.11.055
Return to citation in text: [1] [2] [3] [4] -
Duke, R. M.; Veale, E. B.; Pfeffer, F. M.; Kruger, P. E.; Gunnlaugsson, T. Chem. Soc. Rev. 2010, 39, 3936–3953. doi:10.1039/B910560N
Return to citation in text: [1] [2] -
Aydogan, A.; Coady, D. J.; Kim, S. K.; Akar, A.; Bielawski, C. W.; Marquez, M.; Sessler, J. L. Angew. Chem., Int. Ed. 2008, 47, 9648–9652. doi:10.1002/anie.200803970
Return to citation in text: [1] -
Aydogan, A.; Coady, D. J.; Lynch, V. M.; Akar, A.; Marquez, M.; Bielawski, C. W.; Sessler, J. L. Chem. Commun. 2008, 1455–1457. doi:10.1039/b718021g
Return to citation in text: [1] -
Silver, E. S.; Rambo, B. M.; Bielawski, C. W.; Sessler, J. L. Supramol. Chem. 2012, 24, 101–105. doi:10.1080/10610278.2011.628392
Return to citation in text: [1] -
Silver, E. S.; Rambo, B. M.; Bielawski, C. W.; Sessler, J. L. J. Am. Chem. Soc. 2014, 136, 2252–2255. doi:10.1021/ja4123895
Return to citation in text: [1] -
Bao, X.; Zhou, Y. Sens. Actuators, B 2010, 147, 434–441. doi:10.1016/j.snb.2010.03.068
Return to citation in text: [1] [2] -
Erdemir, S.; Kocyigit, O.; Alici, O.; Malkondu, S. Tetrahedron Lett. 2013, 54, 613–617. doi:10.1016/j.tetlet.2012.11.138
Return to citation in text: [1] -
Saluja, P.; Kaur, N.; Kang, J.; Singh, N.; Jang, D. O. Tetrahedron 2013, 69, 9001–9006. doi:10.1016/j.tet.2013.08.043
Return to citation in text: [1] -
Hai, Y.; Chen, J.-J.; Zhao, P.; Lv, H.; Yu, Y.; Xu, P.; Zhang, J.-L. Chem. Commun. 2011, 47, 2435–2437. doi:10.1039/C0CC04113K
Return to citation in text: [1] -
Takakura, H.; Sasakura, K.; Ueno, T.; Urano, Y.; Terai, T.; Hanaoka, K.; Tsuboi, T.; Nagano, T. Chem. – Asian J. 2010, 5, 2053–2061. doi:10.1002/asia.201000219
Return to citation in text: [1] -
Hammud, H. H.; Ghannoum, A.; Masoud, M. S. Spectrochim. Acta, Part A: Mol. Biomol. Spectrosc. 2006, 63, 255–265. doi:10.1016/j.saa.2005.04.020
Return to citation in text: [1] [2] -
Arnaut, L. G.; Formosinho, S. J. J. Photochem. Photobiol., A: Chem. 1993, 75, 1–20. doi:10.1016/1010-6030(93)80157-5
Return to citation in text: [1] -
Cohen, M. D.; Schmidt, G. M. J. J. Phys. Chem. 1962, 66, 2442–2446. doi:10.1021/j100818a030
Return to citation in text: [1] [2] [3] -
Richey, W. F.; Becker, R. S. J. Chem. Phys. 1968, 49, 2092–2100. doi:10.1063/1.1670370
Return to citation in text: [1] [2] [3] -
Naeimi, H.; Moradian, M. J. Coord. Chem. 2010, 63, 156–162. doi:10.1080/00958970903225866
Return to citation in text: [1] [2] [3]
| 2. | Liu, K.; Huo, J.; Zhu, B.; Huo, R. J. Fluoresc. 2012, 22, 1231–1236. doi:10.1007/s10895-012-1063-z |
| 15. | Liu, K.; Zhao, X.; Liu, Q.; Huo, J.; Fu, H.; Wang, Y. J. Photochem. Photobiol., B: Biol. 2014, 138, 75–79. doi:10.1016/j.jphotobiol.2014.05.004 |
| 16. | Liu, K.; Zhao, X.; Liu, Q.; Huo, J.; Wang, X.; Wu, Y. Spectrochim. Acta, Part A: Mol. Biomol. Spectrosc. 2015, 140, 198–201. doi:10.1016/j.saa.2014.12.092 |
| 1. | Mao, S.; Liu, K.; Lu, F.; Du, L. Mini-Rev. Org. Chem. 2010, 7, 221–229. doi:10.2174/157019310791384137 |
| 2. | Liu, K.; Huo, J.; Zhu, B.; Huo, R. J. Fluoresc. 2012, 22, 1231–1236. doi:10.1007/s10895-012-1063-z |
| 3. | Amendola, V.; Fabbrizzi, L.; Mosca, L. Chem. Soc. Rev. 2010, 39, 3889–3915. doi:10.1039/b822552b |
| 2. | Liu, K.; Huo, J.; Zhu, B.; Huo, R. J. Fluoresc. 2012, 22, 1231–1236. doi:10.1007/s10895-012-1063-z |
| 11. | Solntsev, K. M.; Bartolo, E.-A.; Pan, G.; Muller, G.; Bommireddy, S.; Huppert, D.; Tolbert, L. M. Isr. J. Chem. 2009, 49, 227–233. doi:10.1560/ijc.49.2.227 |
| 12. | Song, P.; Ding, J.-X.; Chu, T.-S. Spectrochim. Acta, Part A: Mol. Biomol. Spectrosc. 2012, 97, 746–752. doi:10.1016/j.saa.2012.07.010 |
| 13. | Zhao, Y.-q.; Zhang, B.-q.; Duan, C.-y.; Lin, Z.-h.; Meng, Q.-j. New J. Chem. 2006, 30, 1207–1213. doi:10.1039/b605087e |
| 14. | Kanagaraj, K.; Pitchumani, K. Chem. – Asian J. 2014, 9, 146–152. doi:10.1002/asia.201300816 |
| 15. | Liu, K.; Zhao, X.; Liu, Q.; Huo, J.; Fu, H.; Wang, Y. J. Photochem. Photobiol., B: Biol. 2014, 138, 75–79. doi:10.1016/j.jphotobiol.2014.05.004 |
| 16. | Liu, K.; Zhao, X.; Liu, Q.; Huo, J.; Wang, X.; Wu, Y. Spectrochim. Acta, Part A: Mol. Biomol. Spectrosc. 2015, 140, 198–201. doi:10.1016/j.saa.2014.12.092 |
| 28. | Hai, Y.; Chen, J.-J.; Zhao, P.; Lv, H.; Yu, Y.; Xu, P.; Zhang, J.-L. Chem. Commun. 2011, 47, 2435–2437. doi:10.1039/C0CC04113K |
| 29. | Takakura, H.; Sasakura, K.; Ueno, T.; Urano, Y.; Terai, T.; Hanaoka, K.; Tsuboi, T.; Nagano, T. Chem. – Asian J. 2010, 5, 2053–2061. doi:10.1002/asia.201000219 |
| 4. | Wu, J.; Liu, W.; Ge, J.; Zhang, H.; Wang, P. Chem. Soc. Rev. 2011, 40, 3483–3495. doi:10.1039/C0CS00224K |
| 8. | Sahoo, S. K.; Bera, R. K.; Kanungo, B. K.; Baral, M. Spectrochim. Acta, Part A: Mol. Biomol. Spectrosc. 2012, 89, 322–328. doi:10.1016/j.saa.2011.12.049 |
| 9. | Sharma, D.; Sahoo, S. K.; Chaudhary, S.; Bera, R. K.; Callan, J. F. Analyst 2013, 138, 3646–3650. doi:10.1039/C3AN00199G |
| 10. | Yadav, U. N.; Pant, P.; Sharma, D.; Sahoo, S. K.; Shankarling, G. S. Sens. Actuators, B 2014, 197, 73–80. doi:10.1016/j.snb.2014.02.075 |
| 30. | Hammud, H. H.; Ghannoum, A.; Masoud, M. S. Spectrochim. Acta, Part A: Mol. Biomol. Spectrosc. 2006, 63, 255–265. doi:10.1016/j.saa.2005.04.020 |
| 5. | Jung, H. S.; Kim, H. J.; Vicens, J.; Kim, J. S. Tetrahedron Lett. 2009, 50, 983–987. doi:10.1016/j.tetlet.2008.12.026 |
| 6. | Lee, M. H.; Cao, Q.-Y.; Kim, S. K.; Sessler, J. L.; Kim, J. S. J. Org. Chem. 2011, 76, 870–874. doi:10.1021/jo1021713 |
| 7. | Zhang, J. F.; Lim, C. S.; Bhuniya, S.; Cho, B. R.; Kim, J. S. Org. Lett. 2011, 13, 1190–1193. doi:10.1021/ol200072e |
| 14. | Kanagaraj, K.; Pitchumani, K. Chem. – Asian J. 2014, 9, 146–152. doi:10.1002/asia.201300816 |
| 25. | Bao, X.; Zhou, Y. Sens. Actuators, B 2010, 147, 434–441. doi:10.1016/j.snb.2010.03.068 |
| 27. | Saluja, P.; Kaur, N.; Kang, J.; Singh, N.; Jang, D. O. Tetrahedron 2013, 69, 9001–9006. doi:10.1016/j.tet.2013.08.043 |
| 4. | Wu, J.; Liu, W.; Ge, J.; Zhang, H.; Wang, P. Chem. Soc. Rev. 2011, 40, 3483–3495. doi:10.1039/C0CS00224K |
| 2. | Liu, K.; Huo, J.; Zhu, B.; Huo, R. J. Fluoresc. 2012, 22, 1231–1236. doi:10.1007/s10895-012-1063-z |
| 14. | Kanagaraj, K.; Pitchumani, K. Chem. – Asian J. 2014, 9, 146–152. doi:10.1002/asia.201300816 |
| 15. | Liu, K.; Zhao, X.; Liu, Q.; Huo, J.; Fu, H.; Wang, Y. J. Photochem. Photobiol., B: Biol. 2014, 138, 75–79. doi:10.1016/j.jphotobiol.2014.05.004 |
| 19. | Goswami, S.; Manna, A.; Paul, S.; Das, A. K.; Nandi, P. K.; Maity, A. K.; Saha, P. Tetrahedron Lett. 2014, 55, 490–494. doi:10.1016/j.tetlet.2013.11.055 |
| 21. | Aydogan, A.; Coady, D. J.; Kim, S. K.; Akar, A.; Bielawski, C. W.; Marquez, M.; Sessler, J. L. Angew. Chem., Int. Ed. 2008, 47, 9648–9652. doi:10.1002/anie.200803970 |
| 22. | Aydogan, A.; Coady, D. J.; Lynch, V. M.; Akar, A.; Marquez, M.; Bielawski, C. W.; Sessler, J. L. Chem. Commun. 2008, 1455–1457. doi:10.1039/b718021g |
| 23. | Silver, E. S.; Rambo, B. M.; Bielawski, C. W.; Sessler, J. L. Supramol. Chem. 2012, 24, 101–105. doi:10.1080/10610278.2011.628392 |
| 24. | Silver, E. S.; Rambo, B. M.; Bielawski, C. W.; Sessler, J. L. J. Am. Chem. Soc. 2014, 136, 2252–2255. doi:10.1021/ja4123895 |
| 25. | Bao, X.; Zhou, Y. Sens. Actuators, B 2010, 147, 434–441. doi:10.1016/j.snb.2010.03.068 |
| 26. | Erdemir, S.; Kocyigit, O.; Alici, O.; Malkondu, S. Tetrahedron Lett. 2013, 54, 613–617. doi:10.1016/j.tetlet.2012.11.138 |
| 1. | Mao, S.; Liu, K.; Lu, F.; Du, L. Mini-Rev. Org. Chem. 2010, 7, 221–229. doi:10.2174/157019310791384137 |
| 4. | Wu, J.; Liu, W.; Ge, J.; Zhang, H.; Wang, P. Chem. Soc. Rev. 2011, 40, 3483–3495. doi:10.1039/C0CS00224K |
| 14. | Kanagaraj, K.; Pitchumani, K. Chem. – Asian J. 2014, 9, 146–152. doi:10.1002/asia.201300816 |
| 15. | Liu, K.; Zhao, X.; Liu, Q.; Huo, J.; Fu, H.; Wang, Y. J. Photochem. Photobiol., B: Biol. 2014, 138, 75–79. doi:10.1016/j.jphotobiol.2014.05.004 |
| 19. | Goswami, S.; Manna, A.; Paul, S.; Das, A. K.; Nandi, P. K.; Maity, A. K.; Saha, P. Tetrahedron Lett. 2014, 55, 490–494. doi:10.1016/j.tetlet.2013.11.055 |
| 20. | Duke, R. M.; Veale, E. B.; Pfeffer, F. M.; Kruger, P. E.; Gunnlaugsson, T. Chem. Soc. Rev. 2010, 39, 3936–3953. doi:10.1039/B910560N |
| 15. | Liu, K.; Zhao, X.; Liu, Q.; Huo, J.; Fu, H.; Wang, Y. J. Photochem. Photobiol., B: Biol. 2014, 138, 75–79. doi:10.1016/j.jphotobiol.2014.05.004 |
| 16. | Liu, K.; Zhao, X.; Liu, Q.; Huo, J.; Wang, X.; Wu, Y. Spectrochim. Acta, Part A: Mol. Biomol. Spectrosc. 2015, 140, 198–201. doi:10.1016/j.saa.2014.12.092 |
| 2. | Liu, K.; Huo, J.; Zhu, B.; Huo, R. J. Fluoresc. 2012, 22, 1231–1236. doi:10.1007/s10895-012-1063-z |
| 14. | Kanagaraj, K.; Pitchumani, K. Chem. – Asian J. 2014, 9, 146–152. doi:10.1002/asia.201300816 |
| 15. | Liu, K.; Zhao, X.; Liu, Q.; Huo, J.; Fu, H.; Wang, Y. J. Photochem. Photobiol., B: Biol. 2014, 138, 75–79. doi:10.1016/j.jphotobiol.2014.05.004 |
| 19. | Goswami, S.; Manna, A.; Paul, S.; Das, A. K.; Nandi, P. K.; Maity, A. K.; Saha, P. Tetrahedron Lett. 2014, 55, 490–494. doi:10.1016/j.tetlet.2013.11.055 |
| 2. | Liu, K.; Huo, J.; Zhu, B.; Huo, R. J. Fluoresc. 2012, 22, 1231–1236. doi:10.1007/s10895-012-1063-z |
| 17. | Wu, F.; Ma, L.; Zhang, S.; Wang, Z.; Cheng, X. Mater. Lett. 2014, 116, 231–234. doi:10.1016/j.matlet.2013.11.017 |
| 18. | Chipem, F. A. S.; Krishnamoorthy, G. J. Phys. Chem. B 2013, 117, 14079–14088. doi:10.1021/jp405804c |
| 1. | Mao, S.; Liu, K.; Lu, F.; Du, L. Mini-Rev. Org. Chem. 2010, 7, 221–229. doi:10.2174/157019310791384137 |
| 3. | Amendola, V.; Fabbrizzi, L.; Mosca, L. Chem. Soc. Rev. 2010, 39, 3889–3915. doi:10.1039/b822552b |
| 14. | Kanagaraj, K.; Pitchumani, K. Chem. – Asian J. 2014, 9, 146–152. doi:10.1002/asia.201300816 |
| 15. | Liu, K.; Zhao, X.; Liu, Q.; Huo, J.; Fu, H.; Wang, Y. J. Photochem. Photobiol., B: Biol. 2014, 138, 75–79. doi:10.1016/j.jphotobiol.2014.05.004 |
| 19. | Goswami, S.; Manna, A.; Paul, S.; Das, A. K.; Nandi, P. K.; Maity, A. K.; Saha, P. Tetrahedron Lett. 2014, 55, 490–494. doi:10.1016/j.tetlet.2013.11.055 |
| 20. | Duke, R. M.; Veale, E. B.; Pfeffer, F. M.; Kruger, P. E.; Gunnlaugsson, T. Chem. Soc. Rev. 2010, 39, 3936–3953. doi:10.1039/B910560N |
| 31. | Arnaut, L. G.; Formosinho, S. J. J. Photochem. Photobiol., A: Chem. 1993, 75, 1–20. doi:10.1016/1010-6030(93)80157-5 |
| 2. | Liu, K.; Huo, J.; Zhu, B.; Huo, R. J. Fluoresc. 2012, 22, 1231–1236. doi:10.1007/s10895-012-1063-z |
| 30. | Hammud, H. H.; Ghannoum, A.; Masoud, M. S. Spectrochim. Acta, Part A: Mol. Biomol. Spectrosc. 2006, 63, 255–265. doi:10.1016/j.saa.2005.04.020 |
| 2. | Liu, K.; Huo, J.; Zhu, B.; Huo, R. J. Fluoresc. 2012, 22, 1231–1236. doi:10.1007/s10895-012-1063-z |
| 2. | Liu, K.; Huo, J.; Zhu, B.; Huo, R. J. Fluoresc. 2012, 22, 1231–1236. doi:10.1007/s10895-012-1063-z |
| 11. | Solntsev, K. M.; Bartolo, E.-A.; Pan, G.; Muller, G.; Bommireddy, S.; Huppert, D.; Tolbert, L. M. Isr. J. Chem. 2009, 49, 227–233. doi:10.1560/ijc.49.2.227 |
| 12. | Song, P.; Ding, J.-X.; Chu, T.-S. Spectrochim. Acta, Part A: Mol. Biomol. Spectrosc. 2012, 97, 746–752. doi:10.1016/j.saa.2012.07.010 |
| 13. | Zhao, Y.-q.; Zhang, B.-q.; Duan, C.-y.; Lin, Z.-h.; Meng, Q.-j. New J. Chem. 2006, 30, 1207–1213. doi:10.1039/b605087e |
| 14. | Kanagaraj, K.; Pitchumani, K. Chem. – Asian J. 2014, 9, 146–152. doi:10.1002/asia.201300816 |
| 15. | Liu, K.; Zhao, X.; Liu, Q.; Huo, J.; Fu, H.; Wang, Y. J. Photochem. Photobiol., B: Biol. 2014, 138, 75–79. doi:10.1016/j.jphotobiol.2014.05.004 |
| 1. | Mao, S.; Liu, K.; Lu, F.; Du, L. Mini-Rev. Org. Chem. 2010, 7, 221–229. doi:10.2174/157019310791384137 |
| 32. | Cohen, M. D.; Schmidt, G. M. J. J. Phys. Chem. 1962, 66, 2442–2446. doi:10.1021/j100818a030 |
| 33. | Richey, W. F.; Becker, R. S. J. Chem. Phys. 1968, 49, 2092–2100. doi:10.1063/1.1670370 |
| 34. | Naeimi, H.; Moradian, M. J. Coord. Chem. 2010, 63, 156–162. doi:10.1080/00958970903225866 |
| 8. | Sahoo, S. K.; Bera, R. K.; Kanungo, B. K.; Baral, M. Spectrochim. Acta, Part A: Mol. Biomol. Spectrosc. 2012, 89, 322–328. doi:10.1016/j.saa.2011.12.049 |
| 9. | Sharma, D.; Sahoo, S. K.; Chaudhary, S.; Bera, R. K.; Callan, J. F. Analyst 2013, 138, 3646–3650. doi:10.1039/C3AN00199G |
| 10. | Yadav, U. N.; Pant, P.; Sharma, D.; Sahoo, S. K.; Shankarling, G. S. Sens. Actuators, B 2014, 197, 73–80. doi:10.1016/j.snb.2014.02.075 |
| 15. | Liu, K.; Zhao, X.; Liu, Q.; Huo, J.; Fu, H.; Wang, Y. J. Photochem. Photobiol., B: Biol. 2014, 138, 75–79. doi:10.1016/j.jphotobiol.2014.05.004 |
| 1. | Mao, S.; Liu, K.; Lu, F.; Du, L. Mini-Rev. Org. Chem. 2010, 7, 221–229. doi:10.2174/157019310791384137 |
| 32. | Cohen, M. D.; Schmidt, G. M. J. J. Phys. Chem. 1962, 66, 2442–2446. doi:10.1021/j100818a030 |
| 33. | Richey, W. F.; Becker, R. S. J. Chem. Phys. 1968, 49, 2092–2100. doi:10.1063/1.1670370 |
| 34. | Naeimi, H.; Moradian, M. J. Coord. Chem. 2010, 63, 156–162. doi:10.1080/00958970903225866 |
| 15. | Liu, K.; Zhao, X.; Liu, Q.; Huo, J.; Fu, H.; Wang, Y. J. Photochem. Photobiol., B: Biol. 2014, 138, 75–79. doi:10.1016/j.jphotobiol.2014.05.004 |
| 32. | Cohen, M. D.; Schmidt, G. M. J. J. Phys. Chem. 1962, 66, 2442–2446. doi:10.1021/j100818a030 |
| 33. | Richey, W. F.; Becker, R. S. J. Chem. Phys. 1968, 49, 2092–2100. doi:10.1063/1.1670370 |
| 34. | Naeimi, H.; Moradian, M. J. Coord. Chem. 2010, 63, 156–162. doi:10.1080/00958970903225866 |
© 2015 Liu et al; licensee Beilstein-Institut.
This is an Open Access article under the terms of the Creative Commons Attribution License (http://creativecommons.org/licenses/by/2.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
The license is subject to the Beilstein Journal of Organic Chemistry terms and conditions: (http://www.beilstein-journals.org/bjoc)