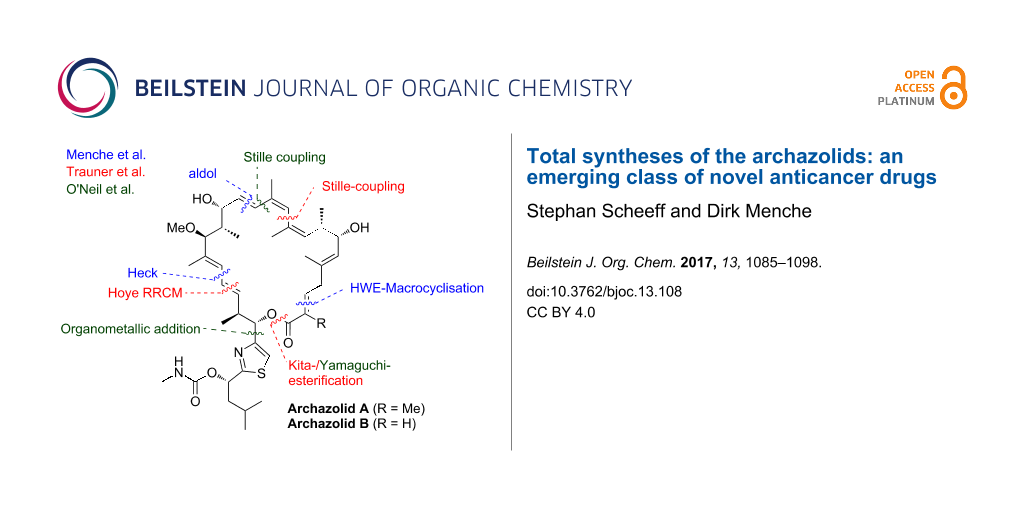
Abstract
V-ATPase has recently emerged as a promising novel anticancer target based on extensive in vitro and in vivo studies with the archazolids, complex polyketide macrolides which present the most potent V-ATPase inhibitors known to date, rendering these macrolides important lead structures for the development of novel anticancer agents. The limited natural supply of these metabolites from their myxobacterial source renders total synthesis of vital importance for the further preclinical development. This review describes in detail the various tactics and strategies employed so far in archazolid syntheses that culminated in three total syntheses and discusses the future synthetic challenges that have to be addressed.
Graphical Abstract
Introduction
The complex structures of polyketides continues to be a great challenge for synthetic chemists and has also been a key driver for the development of new methodologies [1-9]. In many cases, total synthesis is of critical importance to enhance the supply of these often scarce metabolites and even complex polyketides have been prepared on an industrial scale [10,11]. These natural products are also valuable molecular probes for the discovery and evaluation of novel biological targets and pathways [12,13]. Vacuolar-type ATPases (V-ATPases) are heteromultimeric proton translocating proteins that are localized in a multitude of eukaryotic membranes [14-16] that have been known as key mediators in a variety of biochemical pathways. They are responsible for a variety of important cellular functions, including pH-control [17,18], membrane trafficking, protein degradation, release of neurotransmitters [18], urinary acidification [19], bone resorption [20], sperm maturation [21], cholesterol biosynthesis [22] and cytokine secretion [23]. In recent years, a key role of these multimeric enzymes also in cancer development and progression was discovered and has been increasingly unraveled. The archazolids, polyketide macrolides from the myxobacterium Archangium gephyra, have played a key role in these studies. They present the most potent V-ATPase inhibitors known to date with activities in the low nanomolar range [24,25], by binding to the functional transmembrane subunit c [26,27], and display highly potent growth-inhibitory activities against a range of cancer cell lines, both in vitro and in vivo [23,28-34]. In detail, archazolid inhibition of V-ATPase abrogates tumor metastasis via repression of endocytic activation [28], leads to impaired cathepsin B activation in vivo [30], modulates anoikis resistance and metastasis of cancer cells [31], overcomes trastuzumab resistance of breast cancers [32], blocks iron metabolism and thereby mediates therapeutic effects in breast cancers [33], and plays a role in tumor sensitizing in the context of the MDM2 antagonist nutlin-3a [34]. Based on these studies V-ATPases have been increasingly emerging as an extremely promising novel anticancer target [26,27,35-37] and the archazolids have become important lead structures for the development of novel anticancer agents.
As shown in Scheme 1 for the most prominent representatives archazolid A (1) and B (2) [38-40], their unique architectures are characterized by a 24-membered macrolactone ring with seven alkenes, including a characteristic (Z,Z,E)-triene, a thiazole side chain and a characteristic sequence of eight methyl and hydroxy-bearing stereocenters.
Scheme 1: Molecular structures of the archazolids.
Scheme 1: Molecular structures of the archazolids.
Synthetic chemistry is of key importance to enhance the supply of these scarce polyketides to fully evaluate the biological potential and develop them as potential drug candidates. The important biological properties in combination with their limited natural supply as well as their intriguing molecular architectures and initially unknown stereochemistry, have rendered the archazolids as highly attractive synthetic targets and so far, three total syntheses have been reported by the groups of Menche and Trauner [41-43]. Furthermore several fragment syntheses as well as methodological studies to access key structural elements have been published in the last years [44-54]. Recently, the focus has been increasingly shifted towards analog development and SAR studies [49,55-58] and the group of O’Neil has been targeting dihydroarchazolid B (3) as a potentially equipotent structurally simplified derivative [45,46,48]. This review covers the various tactics and strategies, employed by the Menche, Trauner and O’Neil group in archazolid syntheses and discusses the challenges for the development of a scalable route.
Review
The first total synthesis of archazolids A and B were independently developed by the Menche group and the group of Trauner in 2007 by completely independent routes. In 2009, a second total synthesis of archazolid B has been described again by the Menche group following a sequence that was related to their archazolid A synthesis.
Menche’s retrosynthetic analysis and strategy
As a prelude to initiating a synthetic campaign directed towards the archazolids the Menche group first elucidated the full stereochemistry and three dimensional conformation of the archazolids by NMR methods, molecular modelling and chemical derivatizations [59,60]. During these studies, they became aware that C2–C5 diene of acyclic analogs would be very labile towards isomerization. However, such processes would be suppressed in the macrolide natural products, presumably due to conformational factors. In contrast, the Z,Z,E-triene system at northern part of the target molecule (i.e., C9 to C14) was stable, also in an acyclic state, presumably due to distortion of the conjugated system due to constraints exerted by the methyl groups at C10 and C12. Based on this analysis, their synthetic plan was to first build the triene, while the C2–C5 fragment should be constructed directly before or during ring closure as shown in Scheme 2. Consequently, they first planned to connect the north-eastern fragment 5 with the propionate unit 4 by a suitable olefination strategy. Subsequently, for connecting the resulting fragment to the thiazole subunit 6 a Heck reaction was envisioned as part of studies advancing this type of Palladium-catalyzed coupling strategies in complex target synthesis [61-66]. Finally, a HWE-macrocyclization was planned which would likewise set the labile C2–C5 diene and thus concomitantly stabilize this functionality by macrocyclic constraints.
Scheme 2: Retrosynthetic analysis of archazolid A by the Menche group.
Scheme 2: Retrosynthetic analysis of archazolid A by the Menche group.
Menche’s total synthesis of archazolid A
Synthesis of the north-eastern fragment
As shown in Scheme 3, the construction of the north-eastern fragment 5 relied on a boron-mediated anti-aldol reaction [67] of lactate-derived ethyl ketone 13 with aldehyde 12, which in turn was available from aldehyde 9 by HWE olefination. This Paterson aldol reaction and related aldol reactions, which have been amply used by the Menche group [68-70], proceeded with excellent yield and selectivity. The resulting β-hydoxyketone 14 was then protected as TBS ether and converted to aldehyde 15 by reductive removal of the benzoate group and periodate cleavage. Notably, depending on the choice of protection group, deprotection and further oxidation with NaIO4 may be observed, a procedure that was further studied by the group [71]. The two vicinal Z-alkenes were then installed by two consecutive Still–Gennari olefinations [72] with aldehydes 15 and 18. In both cases coupling with the Still–Gennari reagent 16 gave 17 and after reduction the final building block 5 was formed in high yields and selectivity. While the overall sequence towards 5 consequently required twice a two-step adaption of the oxidation state which renders this route quite lengthy, the authors argue that the robustness, facile conduction and scalability of each step was very high and made them decide to stick to this sequence as compared to likewise tested alternatives.
Scheme 3: Synthesis of north-eastern fragment 5 through a Paterson anti-aldol addition and multiple Still–Gennari olefinations.
Scheme 3: Synthesis of north-eastern fragment 5 through a Paterson anti-aldol addition and multiple Still–Gen...
Synthesis of the north-western fragment
For the construction of the north-western fragment 4 the Menche group opted to first install the terminal E-configured vinyl iodide. While appearing to be a simple problem, quite some efforts had to be invested, before acid 21 could be efficiently obtained as shown in Scheme 4. Finally, after optimization of a reported procedure [73] the successful route employed a one-pot process involving a sodium hydride-mediated coupling of methyl malonate 19 with iodoform (20) followed by a stereoselective elimination and decarboxylation in situ. The corresponding aldehyde 22 was then homologated by an Abiko–Masamune anti-aldol addition [74] with ephedrine-derived ester 23, which proceeded with excellent yield and stereoselectivity. However, the subsequent removal of the sterically hindered chiral auxiliary proved demanding. The Menche group realized that a transformation to a Weinreb amide may be realized in an effective manner by an in situ activation of 24 with iPrMgCl [75], followed by a methylation of the free hydroxy group and introduction of the methyl ketone. This procedure proved superior to an original sequence involving a reductive cleavage of the auxiliary.
Scheme 4: Synthesis of 4 through an Abiko–Masamune anti-aldol addition.
Scheme 4: Synthesis of 4 through an Abiko–Masamune anti-aldol addition.
Synthesis of the southern fragment
The same southern fragment 6 was independently chosen by the Menche group in their total synthesis of archazolid A and by the Trauner group in their total synthesis of archazolid B. Both groups also pursued identical routes to this subunit. As shown in Scheme 5, this sequence started from L-leucine (26) which was first converted with nitrous acid to the hydroxy acid 27, which proceeds with retention of the configuration due to intermediate lactone formation after generation of the diazonium-intermediate [76]. After conversion to amide 28 and thioamide 29 the thiazole 31 was obtained by condensation with bromoester 30. The carbamate was then introduced by activation of the deprotected hydroxy group with carbonyldiimidazole and treatment with methylamine, before the ester was selectively reduced to the aldehyde 32 with DIBALH. Finally, a Brown crotylation [77] set the two vicinal stereogenic centers of 6 with high stereoselectivity. In total this route enabled an efficient and reliable access to this key fragment. However, one drawback of this sequence was a tendency of epimerisation at C1’ during thiazole formation on large scale, requiring an oxidation–reduction sequence (90%) in this case.
Scheme 5: Thiazol construction and synthesis of the southern fragment 6.
Scheme 5: Thiazol construction and synthesis of the southern fragment 6.
Completion of the total synthesis
After evaluation of several strategies, the assembly of the two northern fragments 4 and 5 could be realized by an aldol condensation, involving a boron-mediated aldol coupling, acetate protection of the newly generated hydroxy group and DBU-mediated elimination. This three-step sequence proceeded as shown in Scheme 6 with excellent yields (94%) giving the triene 33 as a single diastereomer, which demonstrates the usefulness of aldol condensations in complex target synthesis, also on highly elaborate substrates. Considerable efforts were invested before the challenging Heck coupling with the thiazole fragment 6 could be effected with useful selectivities. Besides the desired E,E-diene 34 formation of other double bond isomers both in the southern and northern part of the molecule could not be suppressed and required a tedious HPLC separation at this stage. After attachment of the phosphonate 7, aldehyde 35 was obtained by removal of the PMB group and oxidation of the primary alcohol. The moderate yields of this sequence are mainly due to side reactions in the deprotection step. The Menche group then had considerable difficulties in closing the macrocyclic ring using an HWE reaction. Finally, the macrocylization could be realised by using NaH as a strong non-nucleophilic base. It proved essential to perform this reaction in the presence of molecular sieves to remove any traces of water which were shown to lead to unfavourable isomerization pathways. Finally, an oxazaborolidine-assisted borane reduction [78] set the stereogenic center at C15 and global deprotection gave synthetic archazolid A, which proved to be fully identical with a natural sample, thus unambiguously confirming the stereochemistry of this macrolide [59].
Scheme 6: Completion of the total synthesis of archazolid A.
Scheme 6: Completion of the total synthesis of archazolid A.
Menche’s total synthesis of archazolid B
One the methodological incentives of the synthetic campaign of the Menche group directed towards the archazolids were the further development and application of the Heck reaction in complex target synthesis. Accordingly, they applied a Heck macrocyclization strategy for the total synthesis of archazolid B. As shown in Scheme 7, this strategy could be successfully implemented and the macrocyclic core of the target compound was obtained in useful yields by coupling of 38, which in turn was accessible by an intermolecular HWE reaction of 36 with 37 using the procedure evaluated above. Final stereoselective CBS reduction and global deprotection liberated archazolid B in 41% yield over 3 steps [42]. This accomplishment presented the second total synthesis of this macrolide while the first total synthesis was enabled by the group of Trauner and will be discussed below.
Scheme 7: Synthesis of archazolid B (2) by a ring closing Heck reaction of 38.
Scheme 7: Synthesis of archazolid B (2) by a ring closing Heck reaction of 38.
Trauner’s retrosynthetic analysis and strategy
Shortly after the total synthesis of archazolid A (1) by Menche et al. [41] the total synthesis of archazolid B 2 was reported by Trauner and co-workers [43]. As shown in Scheme 8, they could successfully couple the three main fragments 39, 40 and 6 by first a Stille reaction, followed by a Kita esterification. Notably, this esterification was critical to avoid unfavorable isomerizations. For closing of the macrolide core they planned a challenging Hoye relay ring closing metathesis (RRCM) which would lead directly after deprotection to archazolid B (2).
Scheme 8: Retrosynthetic analysis of archazolid B by the Trauner group.
Scheme 8: Retrosynthetic analysis of archazolid B by the Trauner group.
Trauner’s total synthesis of archazolid B
Synthesis of the north-eastern fragment
The synthesis of fragment 40 started with the literature-known protocol for generation of ynone 42 derived from (S)-Roche ester 41 [79] as shown in Scheme 9. After reduction the alcohol was protected with TIPS and the TBS ether was cleaved by acetic acid to get to the primary alcohol 43. The reduction with (S)-alpine borane was highly diastereoselective (dr > 20:1). Following this sequence over 6 steps the two stereogenic centers at C7 and C8 were successfully built up. The primary alcohol of fragment 43 was then oxidized by the Dess–Martin reagent (DMP) and then treated with CBr4 and PPh3 to generate the dibromoalkene 44 in high yield of 75% over 2 steps. The group now installed the vinyliodide for the Stille coupling by treating the alkene with lithium dimethylcuprate. In comparison with the likewise attempted Stork–Zhao olefination this protocol by Tanino and Miyashita was superior in yield and stereoselectivity [80]. To complete the fragment synthesis the [Ru]-catalyzed Trost–Alder-ene reaction [81] generated the desired primary alcohol which was oxidized in 2 steps with DMP and NaClO2/NaH2PO4 to the free acid 40. The high regioselectivity of the Alder-Ene reaction is remarkable and was argued to be derived by a coordinating effect of the carbonate. Also, the overall high yield for synthesis of this elaborate vinyl iodide is impressive.
Scheme 9: Synthesis of acid 40 from Roche ester 41 involving a highly efficient Trost–Alder ene reaction.
Scheme 9: Synthesis of acid 40 from Roche ester 41 involving a highly efficient Trost–Alder ene reaction.
Synthesis of the north-western fragment
As shown in Scheme 10, the synthesis of stannane 39 started with aldehyde 48 which was derived from propargyl alcohol in five steps [82]. After DMP oxidation the generated aldehyde 48 underwent a syn-selective Evans aldol addition with oxazolidinone 47 to obtain the alcohol 49 in 76% over 2 steps [83]. Before TBS protection the Weinreb amide was generated and then conversed into the phosphonate 50 as a precursor for a Horner–Wadsworth–Emmons reaction. The olefination led to unsaturated ketone 52 in 79% yield. For the final fragment synthesis the ketone was reduced with sodium borohydride to generate all three required stereogenic centers for this fragment with excellent diasteroselectivity. Final methylation of the free alcohol was followed by conversion of the vinyl iodide into the desired stannane to get fragment 39 which was used directly for coupling.
Scheme 10: Synthesis of precursor 39 for the projected relay RCM reaction.
Scheme 10: Synthesis of precursor 39 for the projected relay RCM reaction.
Completion of the total synthesis
Final assembly of the fragments as shown in Scheme 11 began with an unusual [Ru]-catalyzed Kita esterification due to the instability of fragment 40 towards basic esterification approaches [84]. The following Stille coupling with stannane 39 was then accomplished by CuTC co-catalysis to get the final fragment 54 in 32% yield [85,86]. The envisioned Hoye relay RCM was catalyzed by Grubbs’ second generation catalyst to close the macrocycle in 27% yield. The final acid-mediated deprotection liberated finally archazolid B (2). Notably, no cyclization was observed in an analogous RCM reaction with a substrate without the relay tether, which underscores the usefulness of this relay tactic.
Scheme 11: Final steps of Trauner’s total synthesis of archazolid B.
Scheme 11: Final steps of Trauner’s total synthesis of archazolid B.
It is important to mention, that the three main fragments were coupled in only four steps, showing the highly modular approach from Trauner and co-workers. With this strategy it was possible to synthesize archazolid B (2) in only 19 steps from (S)-Roche ester 41 (longest linear sequence).
O'Neil’s retrosynthetic analysis and strategy
As discussed above one of the main difficulties of any archazolid synthesis involves the labile C1 to C5-dienoate system, which is prone to isomerization. However, the similar biological potency of archazolids A and B as well as the archazolid B isomer archazolid F, which bears a 3,4- instead of the 2,3-alkene, suggest that the C2-olefin may not be essential for the biological potency. Accordingly, the group of O‘Neil and co-workers has been targeted dihydroarchazolid B (3). They assumed a similar biological potency of the derivative with simultaneous simplification of the synthesis. While so far, they have not been able to finish this synthesis, they have reported several very instructive and efficient fragment syntheses, including the three main fragments 55, 56 and 57 as shown in Scheme 12 [45,46,48]. The challenging ring-closing metathesis between C13 and C14 could not be established mainly due a competing backbiting process of the corresponding western fragment [46].
Scheme 12: Overview of the different retrosynthetic approaches for the synthesis of dihydroarchazolid B (3) reported by the O'Neil group.
Scheme 12: Overview of the different retrosynthetic approaches for the synthesis of dihydroarchazolid B (3) re...
O'Neil’s syntheses of advanced dihydroarchazolid B fragments
Synthesis of the macrocyclic skeleton
Based on this unsuccessful approach O‘Neil and co-workers published a new synthetic route towards 2,3-dihydroarchazolid B (3) [48]. As shown in Scheme 13 they were able to synthesize the macrocyclic skeleton 68 by a Stille coupling between stannane 56 and iodide 55 as the key step. Notably, they had to switch the halide/organometallic functionality of each building block after an unsuccessful coupling between the stannane synthesized by reduction and methylation of ketone 67 and the iodine derived from fragment 62. They assume that the steric hindrance of the methyl group in C10 position possibly lowers the reactivity of the iodine in the oxidative addition step in the catalytic cycle.
Scheme 13: Fragment synthesis of 69 towards the total synthesis of 3.
Scheme 13: Fragment synthesis of 69 towards the total synthesis of 3.
For the synthesis of stannane 56 the authors could benefit from the previous fragment synthesis. In 2010 they first published an approach to an eastern building block through an allylation-elimination sequence to form the triene system C9 to C14 [44]. In a second generation fragment synthesis building block 62 was firstly synthesized in 2014 [46]. Starting with the TBS-protected alkyne 58 [87] a zirconium-mediated carboalumination reaction [88] and subsequent coupling with aldehyde 59 gave alcohol 60. In this Δ5,6-trans-selective reaction the desired anti-diastereomer could be isolated in 85% with a dr of 6:1, which presumably arises from a chelation-controlled stereoselectivity [89]. The aldehyde 59 itself can be prepared in two steps from the corresponding Roche ester [90]. After protection of the free alcohol and deprotection of the primary PMB-protected alcohol with DDQ, the resulting alcohol was oxidized to the corresponding aldehyde. The crude aldehyde was then directly transformed into the (Z)-α,β-unsaturated ester 62 as a single stereoisomer by a Still–Gennari [72] olefination with phosphonate 61 in an excellent yield of 93% over 4 steps. After reduction of ester 62 to the corresponding aldehyde (by a method that was not specified by the authors) phosphorane 63 was used to generate the respective Z-vinyl iodide in 85% yield as an 8:1 (Z,Z):(Z,E) mixture [91] which was later switched to the stannane 56 by lithium–halogen exchange and further treatment with Bu3SnCl [92] in 90% yield.
The synthesis of the coupling partner 55 started with known Weinreb amide 64 which underwent a syn-selective palladium-catalyzed hydrostannylation and was then transformed to phosphonate 65 in good yield. For coupling with known aldehyde 66 [93] the O’Neil group chose Ba(OH)2 as base for the HWE-type reaction [94] to generate the α,β-unsaturated ketone 67 in 75% yield as a 10:1 mixture of isomers. Similar to the earlier discussed synthesis of Trauner and co-workers [43] reduction with sodium borohydride delivered the desired alcohol in a 10:1 diastereoselectivity. The alcohol was methylated by a protocol involving methyl iodide and LiHMDS, that had been previously used by the group [45]. The stannane was finally converted to the iodide 55 by iododestannylation [95] to complete the fragment synthesis in 44% yield over 3 steps.
For the final step the authors decided to follow a Stille coupling protocol established by Fürstner et al. [96] with CuTC as co-catalyst and [Ph2PO2][NBu4] as tin scavenger. Subsequently, the triene 68 could be synthesized in excellent 82% yield. For biological studies the final fragment 68 was globally deprotected to the alcohol 69.
To this end, the O’Neil group successfully established a route to the dihydroarchazolid B fragment 68 in only 9 steps (longest linear synthesis). This route also proved that a retrosynthetic disconnection between C12 and C13 can be useful for new approaches to the 24-membered macrolide core. For the completion of the synthesis of 3 the side chain would have to be introduced, followed by an oxidation to the acid and a ring closing esterification.
Concise synthesis of the thiazole fragment
In an earlier synthesis the O’Neil group had already coupled a similar fragment 70 with the southern fragment 57 by an organometallic addition, however, with a lack of stereoselectivity in C23 position [45]. As shown in Scheme 14, they started with the deprotection of the primary TBS ether 70 and DMP oxidation to the aldehyde 71. The bromide 57, derived in two steps from literature-known ketone 72 [49], was converted to an organolithium compound which attacked the aldehyde to give the free alcohol 73 in 1:2.5 diasteroselectivity in favor of the undesired R-isomer of 73, which can be explained by the Felkin–Ahn model. For generation of only (S)-73 the both epimers were oxidized to the ketone by DMP followed by reduction with L-selectride. Protection of the free alcohol with acetate and deprotection of the TES group was then required to install the carbamate with CDI and MeNH2. After deprotection, fragment 75 was synthesized in 67% yield over 2 steps.
Scheme 14: Organometallic addition of the side chain to access free alcohol 75.
Scheme 14: Organometallic addition of the side chain to access free alcohol 75.
Having these tactics in hand it will be interesting to follow the completion of the first synthesis of dihydroarchazolid B (3) by the O’Neil group and the biological data of this compound.
Conclusion
The discovery of the archazolids led to important advances in the understanding of the role of V-ATPases in cancer development and progression. Based on extensive studies with these macrolides as chemical tools V-ATPases have emerged as a completely novel and highly promising novel class of anticancer targets. Along these lines synthetic chemistry has played a pivotal role, not only by providing these scarce natural products for biological evaluation, but also in supplying novel analogues with tailored functional properties to decipher the target inhibitor interactions at a molecular level. Finally, the total syntheses of the Menche and Trauner group were also of key importance to assign the full stereochemistry in the first place. The various approaches discussed within this manuscript show the various tactics and strategies that may be employed in complex polyketide synthesis. Notable features of the total synthesis by the Menche group include the robustness of boron mediated aldol reactions to set both the characteristic assemblies of neighbored methyl and hydroxy group bearing stereogenic centers. In addition, an aldol condensation was shown to enable an efficient route for construction of a delicate triene system. The final E-selective Heck coupling on a highly elaborate substrate and the subsequent HWE macrocyclization are remarkable. The Trauner group in turn effectively employed various ruthenium-catalyzed reactions, including a relay ring-closing metathesis, which demonstrates the powerfulness of such a tactic even for highly elaborate substrates with several initiation positions. However, despite these advances and impressive total syntheses the design and development of a truly reliable and scalable route that will enable an access to gram amounts of thee scarce metabolites continues to present a key scientific challenge and the O’Neil group has already demonstrated that a more concise route may be possible. Efforts are now being directed in the design and development of truly practicable and scalable routes to more stable archazolids to enhance the further preclinical development of these novel anticancer agents. Particular importance will be the development of a truly reliable and high yielding macrocyclization method, while efficient methods for fragment syntheses have been established. It will also be interesting to follow whether synthetic chemists will be successful to establish a scalable route that will enable the synthesis of gram quantities of the authentic natural products or novel archazolids with likewise potent or even improved pharmacological and pharmacokinetic properties to fully exploit the extremely promising biological profile of these polyketide macrolides.
Acknowledgements
Financial support by the Deutsche Forschungsgemeinschaft (Forschergruppe 1406), the Volkswagenstiftung and the Fonds der Chemischen Industrie is gratefully acknowledged. We greatly appreciate the dedicated work of those members of our group who have developed this field of research and the stimulating cooperations with our scientific partners – their names can be seen from the literature cited.
References
-
Yeung, K.-S.; Paterson, I. Chem. Rev. 2005, 105, 4237–4313. doi:10.1021/cr040614c
Return to citation in text: [1] -
Schetter, B.; Mahrwald, R. Angew. Chem., Int. Ed. 2006, 45, 7506–7525. doi:10.1002/anie.200602780
Return to citation in text: [1] -
Dalby, S. M.; Paterson, I. Curr. Opin. Drug Discovery Dev. 2010, 13, 777–794.
Return to citation in text: [1] -
Brodmann, T.; Lorenz, M.; Schäckel, R.; Simsek, S.; Kalesse, M. Synlett 2009, 174–192. doi:10.1055/s-0028-1087520
Return to citation in text: [1] -
Li, J.; Menche, D. Synthesis 2009, 2293–2315. doi:10.1055/s-0029-1216881
Return to citation in text: [1] -
Dieckmann, M.; Menche, D. Org. Lett. 2013, 15, 228–231. doi:10.1021/ol3033303
Return to citation in text: [1] -
Dechert-Schmitt, A.-M. R.; Schmitt, D. C.; Gao, X.; Itoh, T.; Krische, M. J. Nat. Prod. Rep. 2014, 31, 504–513. doi:10.1039/c3np70076c
Return to citation in text: [1] -
Seiple, I. B.; Zhang, Z.; Jakubec, P.; Langlois-Mercier, A.; Wright, P. M.; Hog, D. T.; Yabu, K.; Allu, S. R.; Fukuzaki, T.; Carlsen, P. N.; Kitamura, Y.; Zhou, X.; Condakes, M. L.; Szczypiński, F. T.; Green, W. D.; Myers, A. G. Nature 2016, 533, 338–345. doi:10.1038/nature17967
Return to citation in text: [1] -
Bredenkamp, A.; Wegener, M.; Hummel, S.; Häring, A. P.; Kirsch, S. F. Chem. Commun. 2016, 52, 1875–1878. doi:10.1039/C5CC09328G
Return to citation in text: [1] -
Mickel, S. J. Curr. Opin. Drug Discovery Dev. 2004, 7, 869–881.
Return to citation in text: [1] -
Klar, U.; Buchmann, B.; Schwede, W.; Skuballa, W.; Hoffmann, J.; Lichtner, R. B. Angew. Chem., Int. Ed. 2006, 45, 7942–7948. doi:10.1002/anie.200602785
Return to citation in text: [1] -
Cragg, G. M.; Grothaus, P. G.; Newman, D. J. Chem. Rev. 2009, 109, 3012–3043. doi:10.1021/cr900019j
Return to citation in text: [1] -
Weissman, K. J.; Müller, R. Nat. Prod. Rep. 2010, 27, 1276–1295. doi:10.1039/c001260m
Return to citation in text: [1] -
Nishi, T.; Forgac, M. Nat. Rev. Mol. Cell Biol. 2002, 3, 94–103. doi:10.1038/nrm729
Return to citation in text: [1] -
Jefferies, K. C.; Cipriano, D. J.; Forgac, M. Arch. Biochem. Biophys. 2008, 476, 33–42. doi:10.1016/j.abb.2008.03.025
Return to citation in text: [1] -
Beyenbach, K. W.; Wieczorek, H. J. Exp. Biol. 2006, 209, 577–589. doi:10.1242/jeb.02014
Return to citation in text: [1] -
Maxfield, F. R.; McGraw, T. E. Nat. Rev. Mol. Cell Biol. 2004, 5, 121–132. doi:10.1038/nrm1315
Return to citation in text: [1] -
Hiesinger, P. R.; Fayyazuddin, A.; Mehta, S. Q.; Rosenmund, T.; Schulze, K. L.; Zhai, R. G.; Verstreken, P.; Cao, Y.; Zhou, Y.; Kunz, J.; Bellen, H. J. Cell 2005, 121, 607–620. doi:10.1016/j.cell.2005.03.012
Return to citation in text: [1] [2] -
Brown, D.; Paunescu, T. G.; Breton, S.; Marshansky, V. J. Exp. Biol. 2009, 212, 1762–1772. doi:10.1242/jeb.028803
Return to citation in text: [1] -
Toyomura, T.; Murata, Y.; Yamamoto, A.; Oka, T.; Sun-Wada, G.-H.; Wada, Y.; Futai, M. J. Biol. Chem. 2003, 278, 22023–22030. doi:10.1074/jbc.M302436200
Return to citation in text: [1] -
Pietrement, C.; Sun-Wada, G.-H.; Da Silva, N.; McKee, M.; Marshansky, V.; Brown, D.; Futai, M.; Breton, S. Biol. Reprod. 2006, 74, 185–194. doi:10.1095/biolreprod.105.043752
Return to citation in text: [1] -
Hamm, R.; Chen, Y.-R.; Seo, E.-J.; Zeino, M.; Wu, C.-F.; Müller, R.; Yang, N.-S.; Efferth, T. Biochem. Pharmacol. 2014, 91, 18–30. doi:10.1016/j.bcp.2014.06.018
Return to citation in text: [1] -
Scherer, O.; Steinmetz, H.; Kaether, C.; Weinigel, C.; Barz, D.; Kleinert, H.; Menche, D.; Müller, R.; Pergola, C.; Werz, O. Biochem. Pharmacol. 2014, 91, 490–500. doi:10.1016/j.bcp.2014.07.028
Return to citation in text: [1] [2] -
Sasse, F.; Steinmetz, H.; Höfle, G.; Reichenbach, H. J. Antibiot. 2003, 56, 520–525. doi:10.7164/antibiotics.56.520
Return to citation in text: [1] -
Huss, M.; Sasse, F.; Kunze, B.; Jansen, R.; Steinmetz, H.; Ingenhorst, G.; Zeeck, A.; Wieczorek, H. BMC Biochem. 2005, 6, No. 13. doi:10.1186/1471-2091-6-13
Return to citation in text: [1] -
Bockelmann, S.; Menche, D.; Rudolph, S.; Bender, T.; Grond, S.; von Zezschwitz, P.; Muench, S. P.; Wieczorek, H.; Huss, M. J. Biol. Chem. 2010, 285, 38304–38314. doi:10.1074/jbc.M110.137539
Return to citation in text: [1] [2] -
Gölz, J. P.; Bockelmann, S.; Mayer, K.; Steinhoff, H.-J.; Wieczorek, H.; Huss, M.; Klare, J. P.; Menche, D. ChemMedChem 2016, 11, 420–428. doi:10.1002/cmdc.201500500
Return to citation in text: [1] [2] -
Wiedmann, R. M.; von Schwarzenberg, K.; Palamidessi, A.; Schreiner, L.; Kubisch, R.; Liebl, J.; Schempp, C.; Trauner, D.; Vereb, G.; Zahler, S.; Wagner, E.; Müller, R.; Scita, G.; Vollmar, A. M. Cancer Res. 2012, 72, 5976–5987. doi:10.1158/0008-5472.CAN-12-1772
Return to citation in text: [1] [2] -
von Schwarzenberg, K.; Wiedmann, R. M.; Oak, P.; Schulz, S.; Zischka, H.; Wanner, G.; Efferth, T.; Trauner, D.; Vollmar, A. M. J. Biol. Chem. 2013, 288, 1385–1396. doi:10.1074/jbc.M112.412007
Return to citation in text: [1] -
Kubisch, R.; Fröhlich, T.; Arnold, G. J.; Schreiner, L.; von Schwarzenberg, K.; Roidl, A.; Vollmar, A. M.; Wagner, E. Int. J. Cancer 2014, 134, 2478–2488. doi:10.1002/ijc.28562
Return to citation in text: [1] [2] -
Schempp, C. M.; von Schwarzenberg, K.; Schreiner, L.; Kubisch, R.; Müller, R.; Wagner, E.; Vollmar, A. M. Mol. Cancer Ther. 2014, 13, 926–937. doi:10.1158/1535-7163.MCT-13-0484
Return to citation in text: [1] [2] -
von Schwarzenberg, K.; Lajtos, T.; Simon, L.; Müller, R.; Vereb, G.; Vollmar, A. M. Mol. Oncol. 2014, 8, 9–19. doi:10.1016/j.molonc.2013.08.011
Return to citation in text: [1] [2] -
Schneider, L. S.; von Schwarzenberg, K.; Lehr, T.; Ulrich, M.; Kubisch-Dohmen, R.; Liebl, J.; Trauner, D.; Menche, D.; Vollmar, A. M. Cancer Res. 2015, 75, 2863–2874. doi:10.1158/0008-5472.CAN-14-2097
Return to citation in text: [1] [2] -
Schneider, L. S.; Ulrich, M.; Lehr, T.; Menche, D.; Müller, R.; von Schwarzenberg, K. Mol. Oncol. 2016, 10, 1054–1062. doi:10.1016/j.molonc.2016.04.005
Return to citation in text: [1] [2] -
Pérez-Sayáns, M.; Somoza-Martin, J. M.; Barros-Angueira, F.; Rey, J. M. G.; García-García, A. Cancer Treat. Rev. 2009, 35, 707–713. doi:10.1016/j.ctrv.2009.08.003
Return to citation in text: [1] -
Hernandez, A.; Serrano-Bueno, G.; Perez-Castineira, J. R.; Serrano, A. Curr. Pharm. Des. 2012, 18, 1383–1394. doi:10.2174/138161212799504821
Return to citation in text: [1] -
Cotter, K.; Capecci, J.; Sennoune, S.; Huss, M.; Maier, M.; Martinez-Zaguilan, R.; Forgac, M. J. Biol. Chem. 2015, 290, 3680–3692. doi:10.1074/jbc.M114.611210
Return to citation in text: [1] -
Menche, D.; Hassfeld, J.; Steinmetz, H.; Huss, M.; Wieczorek, H.; Sasse, F. J. Antibiot. 2007, 60, 328–331. doi:10.1038/ja.2007.43
Return to citation in text: [1] -
Menche, D.; Hassfeld, J.; Steinmetz, H.; Huss, M.; Wieczorek, H.; Sasse, F. Eur. J. Org. Chem. 2007, 1196–1202. doi:10.1002/ejoc.200600912
Return to citation in text: [1] -
Horstmann, N.; Essig, S.; Bockelmann, S.; Wieczorek, H.; Huss, M.; Sasse, F.; Menche, D. J. Nat. Prod. 2011, 74, 1100–1105. doi:10.1021/np200036v
Return to citation in text: [1] -
Menche, D.; Hassfeld, J.; Li, J.; Rudolph, S. J. Am. Chem. Soc. 2007, 129, 6100–6101. doi:10.1021/ja071461o
Return to citation in text: [1] [2] -
Menche, D.; Hassfeld, J.; Li, J.; Mayer, K.; Rudolph, S. J. Org. Chem. 2009, 74, 7220–7229. doi:10.1021/jo901565n
Return to citation in text: [1] [2] -
Roethle, P. A.; Chen, I. T.; Trauner, D. J. Am. Chem. Soc. 2007, 129, 8960–8961. doi:10.1021/ja0733033
Return to citation in text: [1] [2] [3] -
O'Neil, G. W.; Black, M. J. Synlett 2010, 107–110. doi:10.1055/s-0029-1218537
Return to citation in text: [1] [2] -
Tran, A. B.; Melly, G. C.; Doucette, R.; Ashcraft, B.; Sebren, L. J.; Havko, N.; Young, J. C.; O'Neil, G. W. Org. Biomol. Chem. 2011, 9, 7671–7674. doi:10.1039/c1ob06446k
Return to citation in text: [1] [2] [3] [4] [5] -
King, B. R.; Swick, S. M.; Schaefer, S. L.; Welch, J. R.; Hunter, E. F.; O'Neil, G. W. Synthesis 2014, 46, 2927–2936. doi:10.1055/s-0034-1379003
Return to citation in text: [1] [2] [3] [4] [5] -
Swick, S. M.; Schaefer, S. L.; O'Neil, G. W. Tetrahedron Lett. 2015, 56, 4039–4042. doi:10.1016/j.tetlet.2015.05.014
Return to citation in text: [1] -
O'Neil, G. W.; Craig, A. M.; Williams, J. R.; Young, J. C.; Spiegel, P. C. Synlett 2017, 28, 1101–1105. doi:10.1055/s-0036-1588413
Return to citation in text: [1] [2] [3] [4] -
Moulin, E.; Nevado, C.; Gagnepain, J.; Kelter, G.; Fiebig, H.-H.; Fürstner, A. Tetrahedron 2010, 66, 6421–6428. doi:10.1016/j.tet.2010.05.043
Return to citation in text: [1] [2] [3] -
Xu, S.; Negishi, E.-i. Heterocycles 2014, 88, 845–877. doi:10.3987/REV-13-SR(S)5
Return to citation in text: [1] -
Pop, L.; Lassalas, P.; Bencze, L. C.; Toşa, M. I.; Nagy, B.; Irimie, F. D.; Hoarau, C. Tetrahedron: Asymmetry 2012, 23, 474–481. doi:10.1016/j.tetasy.2012.03.014
Return to citation in text: [1] -
de Paolis, M.; Chataigner, I.; Maddaluno, J. Top. Curr. Chem. 2012, 327, 87–146. doi:10.1007/128_2012_320
Return to citation in text: [1] -
Negishi, E.-i.; Wang, G.; Rao, H.; Xu, Z. J. Org. Chem. 2010, 75, 3151–3182. doi:10.1021/jo1003218
Return to citation in text: [1] -
Huang, Z.; Negishi, E.-i. J. Am. Chem. Soc. 2007, 129, 14788–14792. doi:10.1021/ja0772039
Return to citation in text: [1] -
Menche, D.; Hassfeld, J.; Sasse, F.; Huss, M.; Wieczorek, H. Bioorg. Med. Chem. Lett. 2007, 17, 1732–1735. doi:10.1016/j.bmcl.2006.12.073
Return to citation in text: [1] -
Persch, E.; Basile, T.; Bockelmann, S.; Huss, M.; Wieczorek, H.; Carlomagno, T.; Menche, D. Bioorg. Med. Chem. Lett. 2012, 22, 7735–7738. doi:10.1016/j.bmcl.2012.09.081
Return to citation in text: [1] -
Dreisigacker, S.; Latek, D.; Bockelmann, S.; Huss, M.; Wieczorek, H.; Filipek, S.; Gohlke, H.; Menche, D.; Carlomagno, T. J. Chem. Inf. Model. 2012, 52, 2265–2272. doi:10.1021/ci300242d
Return to citation in text: [1] -
Reker, D.; Perna, A. M.; Rodrigues, T.; Schneider, P.; Reutlinger, M.; Mönch, B.; Koeberle, A.; Lamers, C.; Gabler, M.; Steinmetz, H.; Müller, R.; Schubert-Zsilavecz, M.; Werz, O.; Schneider, G. Nat. Chem. 2014, 6, 1072–1078. doi:10.1038/nchem.2095
Return to citation in text: [1] -
Hassfeld, J.; Farès, C.; Steinmetz, H.; Carlomagno, T.; Menche, D. Org. Lett. 2006, 8, 4751–4754. doi:10.1021/ol061831y
Return to citation in text: [1] [2] -
Farès, C.; Hassfeld, J.; Menche, D.; Carlomagno, T. Angew. Chem., Int. Ed. 2008, 47, 3722–3726. doi:10.1002/anie.200800225
Return to citation in text: [1] -
Dieckmann, M.; Rudolph, S.; Dreisigacker, S.; Menche, D. J. Org. Chem. 2012, 77, 10782–10788. doi:10.1021/jo302134y
Return to citation in text: [1] -
Kretschmer, M.; Dieckmann, M.; Li, P.; Rudolph, S.; Herkommer, D.; Troendlin, J.; Menche, D. Chem. – Eur. J. 2013, 19, 15993–16018. doi:10.1002/chem.201302197
Return to citation in text: [1] -
Symkenberg, G.; Kalesse, M. Angew. Chem., Int. Ed. 2014, 53, 1795–1798. doi:10.1002/anie.201309386
Return to citation in text: [1] -
Li, P.; Li, J.; Arikan, F.; Ahlbrecht, W.; Dieckmann, M.; Menche, D. J. Am. Chem. Soc. 2009, 131, 11678–11679. doi:10.1021/ja9056163
Return to citation in text: [1] -
Li, P.; Li, J.; Arikan, F.; Ahlbrecht, W.; Dieckmann, M.; Menche, D. J. Org. Chem. 2010, 75, 2429–2444. doi:10.1021/jo100201f
Return to citation in text: [1] -
Yang, Z.; Xu, X.; Yang, C.-H.; Tian, Y.; Chen, X.; Lian, L.; Pan, W.; Su, X.; Zhang, W.; Chen, Y. Org. Lett. 2016, 18, 5768–5770. doi:10.1021/acs.orglett.6b02729
Return to citation in text: [1] -
Cowden, C. J.; Paterson, I. Org. React. 1997, 51, 1–200. doi:10.1002/0471264180.or051.01
Return to citation in text: [1] -
Arikan, F.; Li, J.; Menche, D. Org. Lett. 2008, 10, 3521–3524. doi:10.1021/ol801292t
Return to citation in text: [1] -
Kretschmer, M.; Menche, D. Synlett 2010, 2989–3007. doi:10.1055/s-0030-1259070
Return to citation in text: [1] -
Essig, S.; Menche, D. Pure Appl. Chem. 2013, 85, 1103–1120. doi:10.1351/PAC-CON-12-09-12
Return to citation in text: [1] -
Li, J.; Menche, D. Synthesis 2009, 1904–1908. doi:10.1055/s-0028-1088062
Return to citation in text: [1] -
Still, W. C.; Gennari, C. Tetrahedron Lett. 1983, 24, 4405–4408. doi:10.1016/S0040-4039(00)85909-2
Return to citation in text: [1] [2] -
Baker, R.; Castro, J. L. J. Chem. Soc., Perkin Trans. 1 1990, 47–65. doi:10.1039/p19900000047
Return to citation in text: [1] -
Inoue, T.; Liu, J.-F.; Buske, D. C.; Abiko, A. J. Org. Chem. 2002, 67, 5250–5256. doi:10.1021/jo0257896
Return to citation in text: [1] -
Li, J.; Li, P.; Menche, D. Synlett 2009, 2417–2420. doi:10.1055/s-0029-1217819
Return to citation in text: [1] -
Pitt, N.; Gani, D. Tetrahedron Lett. 1999, 40, 3811–3814. doi:10.1016/S0040-4039(99)00534-1
Return to citation in text: [1] -
Brown, H. C.; Bhat, K. S.; Randad, R. S. J. Org. Chem. 1989, 54, 1570–1576. doi:10.1021/jo00268a017
Return to citation in text: [1] -
Corey, E. J.; Helal, C. J. Angew. Chem., Int. Ed. 1998, 37, 1986–2012. doi:10.1002/(SICI)1521-3773(19980817)37:15<1986::AID-ANIE1986>3.0.CO;2-Z
Return to citation in text: [1] -
Trost, B. M.; Gunzner, J. L. J. Am. Chem. Soc. 2001, 123, 9449–9450. doi:10.1021/ja011424b
Return to citation in text: [1] -
Tanino, K.; Arakawa, K.; Satoh, M.; Iwata, Y.; Miyashita, M. Tetrahedron Lett. 2006, 47, 861–864. doi:10.1016/j.tetlet.2005.12.002
Return to citation in text: [1] -
Trost, B. M.; Dean Toste, F. Tetrahedron Lett. 1999, 40, 7739–7743. doi:10.1016/S0040-4039(99)01639-1
Return to citation in text: [1] -
Beaudry, C. M.; Trauner, D. Org. Lett. 2002, 4, 2221–2224. doi:10.1021/ol026069o
Return to citation in text: [1] -
Evans, D. A.; Black, W. C. J. Am. Chem. Soc. 1993, 115, 4497–4513. doi:10.1021/ja00064a011
Return to citation in text: [1] -
Kita, Y.; Maeda, H.; Omori, K.; Okuno, T.; Tamura, Y. Synlett 1993, 273–274. doi:10.1055/s-1993-22428
Return to citation in text: [1] -
Farina, V.; Kapadia, S.; Krishnan, B.; Wang, C.; Liebeskind, L. S. J. Org. Chem. 1994, 59, 5905–5911. doi:10.1021/jo00099a018
Return to citation in text: [1] -
Allred, G. D.; Liebeskind, L. S. J. Am. Chem. Soc. 1996, 118, 2748–2749. doi:10.1021/ja9541239
Return to citation in text: [1] -
Molander, G. A.; Fumagalli, T. J. Org. Chem. 2006, 71, 5743–5747. doi:10.1021/jo0608366
Return to citation in text: [1] -
van Horn, D. E.; Negishi, E.-i. J. Am. Chem. Soc. 1978, 100, 2252–2254. doi:10.1021/ja00475a058
Return to citation in text: [1] -
Reetz, M. T. Acc. Chem. Res. 1993, 26, 462–468. doi:10.1021/ar00033a002
Return to citation in text: [1] -
Chen, T.; Altmann, K.-H. Chemistry 2015, 21, 8403–8407. doi:10.1002/chem.201501252
Return to citation in text: [1] -
Loiseleur, O.; Koch, G.; Cercus, J.; Schürch, F. Org. Process Res. Dev. 2005, 9, 259–271. doi:10.1021/op049807s
Return to citation in text: [1] -
Dineen, T. A.; Roush, W. R. Org. Lett. 2004, 6, 2043–2046. doi:10.1021/ol049331x
Return to citation in text: [1] -
Mandal, A. K.; Schneekloth, J. S., Jr.; Kuramochi, K.; Crews, C. M. Org. Lett. 2006, 8, 427–430. doi:10.1021/ol052620g
Return to citation in text: [1] -
Paterson, I.; Yeung, K.-S.; Smaill, J. B. Synlett 1993, 774–776. doi:10.1055/s-1993-22605
Return to citation in text: [1] -
Börding, S.; Bach, T. Chem. Commun. 2014, 50, 4901–4903. doi:10.1039/C4CC01338G
Return to citation in text: [1] -
Fürstner, A.; Funel, J.-A.; Tremblay, M.; Bouchez, L. C.; Nevado, C.; Waser, M.; Ackerstaff, J.; Stimson, C. C. Chem. Commun. 2008, 2873–2875. doi:10.1039/B805299A
Return to citation in text: [1]
| 61. | Dieckmann, M.; Rudolph, S.; Dreisigacker, S.; Menche, D. J. Org. Chem. 2012, 77, 10782–10788. doi:10.1021/jo302134y |
| 62. | Kretschmer, M.; Dieckmann, M.; Li, P.; Rudolph, S.; Herkommer, D.; Troendlin, J.; Menche, D. Chem. – Eur. J. 2013, 19, 15993–16018. doi:10.1002/chem.201302197 |
| 63. | Symkenberg, G.; Kalesse, M. Angew. Chem., Int. Ed. 2014, 53, 1795–1798. doi:10.1002/anie.201309386 |
| 64. | Li, P.; Li, J.; Arikan, F.; Ahlbrecht, W.; Dieckmann, M.; Menche, D. J. Am. Chem. Soc. 2009, 131, 11678–11679. doi:10.1021/ja9056163 |
| 65. | Li, P.; Li, J.; Arikan, F.; Ahlbrecht, W.; Dieckmann, M.; Menche, D. J. Org. Chem. 2010, 75, 2429–2444. doi:10.1021/jo100201f |
| 66. | Yang, Z.; Xu, X.; Yang, C.-H.; Tian, Y.; Chen, X.; Lian, L.; Pan, W.; Su, X.; Zhang, W.; Chen, Y. Org. Lett. 2016, 18, 5768–5770. doi:10.1021/acs.orglett.6b02729 |
| 67. | Cowden, C. J.; Paterson, I. Org. React. 1997, 51, 1–200. doi:10.1002/0471264180.or051.01 |
| 68. | Arikan, F.; Li, J.; Menche, D. Org. Lett. 2008, 10, 3521–3524. doi:10.1021/ol801292t |
| 69. | Kretschmer, M.; Menche, D. Synlett 2010, 2989–3007. doi:10.1055/s-0030-1259070 |
| 70. | Essig, S.; Menche, D. Pure Appl. Chem. 2013, 85, 1103–1120. doi:10.1351/PAC-CON-12-09-12 |
| 77. | Brown, H. C.; Bhat, K. S.; Randad, R. S. J. Org. Chem. 1989, 54, 1570–1576. doi:10.1021/jo00268a017 |
| 78. | Corey, E. J.; Helal, C. J. Angew. Chem., Int. Ed. 1998, 37, 1986–2012. doi:10.1002/(SICI)1521-3773(19980817)37:15<1986::AID-ANIE1986>3.0.CO;2-Z |
| 75. | Li, J.; Li, P.; Menche, D. Synlett 2009, 2417–2420. doi:10.1055/s-0029-1217819 |
| 76. | Pitt, N.; Gani, D. Tetrahedron Lett. 1999, 40, 3811–3814. doi:10.1016/S0040-4039(99)00534-1 |
| 73. | Baker, R.; Castro, J. L. J. Chem. Soc., Perkin Trans. 1 1990, 47–65. doi:10.1039/p19900000047 |
| 74. | Inoue, T.; Liu, J.-F.; Buske, D. C.; Abiko, A. J. Org. Chem. 2002, 67, 5250–5256. doi:10.1021/jo0257896 |
| 72. | Still, W. C.; Gennari, C. Tetrahedron Lett. 1983, 24, 4405–4408. doi:10.1016/S0040-4039(00)85909-2 |
| 59. | Hassfeld, J.; Farès, C.; Steinmetz, H.; Carlomagno, T.; Menche, D. Org. Lett. 2006, 8, 4751–4754. doi:10.1021/ol061831y |
| 42. | Menche, D.; Hassfeld, J.; Li, J.; Mayer, K.; Rudolph, S. J. Org. Chem. 2009, 74, 7220–7229. doi:10.1021/jo901565n |
| 41. | Menche, D.; Hassfeld, J.; Li, J.; Rudolph, S. J. Am. Chem. Soc. 2007, 129, 6100–6101. doi:10.1021/ja071461o |
| 84. | Kita, Y.; Maeda, H.; Omori, K.; Okuno, T.; Tamura, Y. Synlett 1993, 273–274. doi:10.1055/s-1993-22428 |
| 85. | Farina, V.; Kapadia, S.; Krishnan, B.; Wang, C.; Liebeskind, L. S. J. Org. Chem. 1994, 59, 5905–5911. doi:10.1021/jo00099a018 |
| 86. | Allred, G. D.; Liebeskind, L. S. J. Am. Chem. Soc. 1996, 118, 2748–2749. doi:10.1021/ja9541239 |
| 82. | Beaudry, C. M.; Trauner, D. Org. Lett. 2002, 4, 2221–2224. doi:10.1021/ol026069o |
| 83. | Evans, D. A.; Black, W. C. J. Am. Chem. Soc. 1993, 115, 4497–4513. doi:10.1021/ja00064a011 |
| 80. | Tanino, K.; Arakawa, K.; Satoh, M.; Iwata, Y.; Miyashita, M. Tetrahedron Lett. 2006, 47, 861–864. doi:10.1016/j.tetlet.2005.12.002 |
| 81. | Trost, B. M.; Dean Toste, F. Tetrahedron Lett. 1999, 40, 7739–7743. doi:10.1016/S0040-4039(99)01639-1 |
| 43. | Roethle, P. A.; Chen, I. T.; Trauner, D. J. Am. Chem. Soc. 2007, 129, 8960–8961. doi:10.1021/ja0733033 |
| 79. | Trost, B. M.; Gunzner, J. L. J. Am. Chem. Soc. 2001, 123, 9449–9450. doi:10.1021/ja011424b |
| 46. | King, B. R.; Swick, S. M.; Schaefer, S. L.; Welch, J. R.; Hunter, E. F.; O'Neil, G. W. Synthesis 2014, 46, 2927–2936. doi:10.1055/s-0034-1379003 |
| 48. | O'Neil, G. W.; Craig, A. M.; Williams, J. R.; Young, J. C.; Spiegel, P. C. Synlett 2017, 28, 1101–1105. doi:10.1055/s-0036-1588413 |
| 45. | Tran, A. B.; Melly, G. C.; Doucette, R.; Ashcraft, B.; Sebren, L. J.; Havko, N.; Young, J. C.; O'Neil, G. W. Org. Biomol. Chem. 2011, 9, 7671–7674. doi:10.1039/c1ob06446k |
| 46. | King, B. R.; Swick, S. M.; Schaefer, S. L.; Welch, J. R.; Hunter, E. F.; O'Neil, G. W. Synthesis 2014, 46, 2927–2936. doi:10.1055/s-0034-1379003 |
| 48. | O'Neil, G. W.; Craig, A. M.; Williams, J. R.; Young, J. C.; Spiegel, P. C. Synlett 2017, 28, 1101–1105. doi:10.1055/s-0036-1588413 |
| 1. | Yeung, K.-S.; Paterson, I. Chem. Rev. 2005, 105, 4237–4313. doi:10.1021/cr040614c |
| 2. | Schetter, B.; Mahrwald, R. Angew. Chem., Int. Ed. 2006, 45, 7506–7525. doi:10.1002/anie.200602780 |
| 3. | Dalby, S. M.; Paterson, I. Curr. Opin. Drug Discovery Dev. 2010, 13, 777–794. |
| 4. | Brodmann, T.; Lorenz, M.; Schäckel, R.; Simsek, S.; Kalesse, M. Synlett 2009, 174–192. doi:10.1055/s-0028-1087520 |
| 5. | Li, J.; Menche, D. Synthesis 2009, 2293–2315. doi:10.1055/s-0029-1216881 |
| 6. | Dieckmann, M.; Menche, D. Org. Lett. 2013, 15, 228–231. doi:10.1021/ol3033303 |
| 7. | Dechert-Schmitt, A.-M. R.; Schmitt, D. C.; Gao, X.; Itoh, T.; Krische, M. J. Nat. Prod. Rep. 2014, 31, 504–513. doi:10.1039/c3np70076c |
| 8. | Seiple, I. B.; Zhang, Z.; Jakubec, P.; Langlois-Mercier, A.; Wright, P. M.; Hog, D. T.; Yabu, K.; Allu, S. R.; Fukuzaki, T.; Carlsen, P. N.; Kitamura, Y.; Zhou, X.; Condakes, M. L.; Szczypiński, F. T.; Green, W. D.; Myers, A. G. Nature 2016, 533, 338–345. doi:10.1038/nature17967 |
| 9. | Bredenkamp, A.; Wegener, M.; Hummel, S.; Häring, A. P.; Kirsch, S. F. Chem. Commun. 2016, 52, 1875–1878. doi:10.1039/C5CC09328G |
| 17. | Maxfield, F. R.; McGraw, T. E. Nat. Rev. Mol. Cell Biol. 2004, 5, 121–132. doi:10.1038/nrm1315 |
| 18. | Hiesinger, P. R.; Fayyazuddin, A.; Mehta, S. Q.; Rosenmund, T.; Schulze, K. L.; Zhai, R. G.; Verstreken, P.; Cao, Y.; Zhou, Y.; Kunz, J.; Bellen, H. J. Cell 2005, 121, 607–620. doi:10.1016/j.cell.2005.03.012 |
| 28. | Wiedmann, R. M.; von Schwarzenberg, K.; Palamidessi, A.; Schreiner, L.; Kubisch, R.; Liebl, J.; Schempp, C.; Trauner, D.; Vereb, G.; Zahler, S.; Wagner, E.; Müller, R.; Scita, G.; Vollmar, A. M. Cancer Res. 2012, 72, 5976–5987. doi:10.1158/0008-5472.CAN-12-1772 |
| 72. | Still, W. C.; Gennari, C. Tetrahedron Lett. 1983, 24, 4405–4408. doi:10.1016/S0040-4039(00)85909-2 |
| 14. | Nishi, T.; Forgac, M. Nat. Rev. Mol. Cell Biol. 2002, 3, 94–103. doi:10.1038/nrm729 |
| 15. | Jefferies, K. C.; Cipriano, D. J.; Forgac, M. Arch. Biochem. Biophys. 2008, 476, 33–42. doi:10.1016/j.abb.2008.03.025 |
| 16. | Beyenbach, K. W.; Wieczorek, H. J. Exp. Biol. 2006, 209, 577–589. doi:10.1242/jeb.02014 |
| 30. | Kubisch, R.; Fröhlich, T.; Arnold, G. J.; Schreiner, L.; von Schwarzenberg, K.; Roidl, A.; Vollmar, A. M.; Wagner, E. Int. J. Cancer 2014, 134, 2478–2488. doi:10.1002/ijc.28562 |
| 12. | Cragg, G. M.; Grothaus, P. G.; Newman, D. J. Chem. Rev. 2009, 109, 3012–3043. doi:10.1021/cr900019j |
| 13. | Weissman, K. J.; Müller, R. Nat. Prod. Rep. 2010, 27, 1276–1295. doi:10.1039/c001260m |
| 26. | Bockelmann, S.; Menche, D.; Rudolph, S.; Bender, T.; Grond, S.; von Zezschwitz, P.; Muench, S. P.; Wieczorek, H.; Huss, M. J. Biol. Chem. 2010, 285, 38304–38314. doi:10.1074/jbc.M110.137539 |
| 27. | Gölz, J. P.; Bockelmann, S.; Mayer, K.; Steinhoff, H.-J.; Wieczorek, H.; Huss, M.; Klare, J. P.; Menche, D. ChemMedChem 2016, 11, 420–428. doi:10.1002/cmdc.201500500 |
| 10. | Mickel, S. J. Curr. Opin. Drug Discovery Dev. 2004, 7, 869–881. |
| 11. | Klar, U.; Buchmann, B.; Schwede, W.; Skuballa, W.; Hoffmann, J.; Lichtner, R. B. Angew. Chem., Int. Ed. 2006, 45, 7942–7948. doi:10.1002/anie.200602785 |
| 23. | Scherer, O.; Steinmetz, H.; Kaether, C.; Weinigel, C.; Barz, D.; Kleinert, H.; Menche, D.; Müller, R.; Pergola, C.; Werz, O. Biochem. Pharmacol. 2014, 91, 490–500. doi:10.1016/j.bcp.2014.07.028 |
| 28. | Wiedmann, R. M.; von Schwarzenberg, K.; Palamidessi, A.; Schreiner, L.; Kubisch, R.; Liebl, J.; Schempp, C.; Trauner, D.; Vereb, G.; Zahler, S.; Wagner, E.; Müller, R.; Scita, G.; Vollmar, A. M. Cancer Res. 2012, 72, 5976–5987. doi:10.1158/0008-5472.CAN-12-1772 |
| 29. | von Schwarzenberg, K.; Wiedmann, R. M.; Oak, P.; Schulz, S.; Zischka, H.; Wanner, G.; Efferth, T.; Trauner, D.; Vollmar, A. M. J. Biol. Chem. 2013, 288, 1385–1396. doi:10.1074/jbc.M112.412007 |
| 30. | Kubisch, R.; Fröhlich, T.; Arnold, G. J.; Schreiner, L.; von Schwarzenberg, K.; Roidl, A.; Vollmar, A. M.; Wagner, E. Int. J. Cancer 2014, 134, 2478–2488. doi:10.1002/ijc.28562 |
| 31. | Schempp, C. M.; von Schwarzenberg, K.; Schreiner, L.; Kubisch, R.; Müller, R.; Wagner, E.; Vollmar, A. M. Mol. Cancer Ther. 2014, 13, 926–937. doi:10.1158/1535-7163.MCT-13-0484 |
| 32. | von Schwarzenberg, K.; Lajtos, T.; Simon, L.; Müller, R.; Vereb, G.; Vollmar, A. M. Mol. Oncol. 2014, 8, 9–19. doi:10.1016/j.molonc.2013.08.011 |
| 33. | Schneider, L. S.; von Schwarzenberg, K.; Lehr, T.; Ulrich, M.; Kubisch-Dohmen, R.; Liebl, J.; Trauner, D.; Menche, D.; Vollmar, A. M. Cancer Res. 2015, 75, 2863–2874. doi:10.1158/0008-5472.CAN-14-2097 |
| 34. | Schneider, L. S.; Ulrich, M.; Lehr, T.; Menche, D.; Müller, R.; von Schwarzenberg, K. Mol. Oncol. 2016, 10, 1054–1062. doi:10.1016/j.molonc.2016.04.005 |
| 90. | Chen, T.; Altmann, K.-H. Chemistry 2015, 21, 8403–8407. doi:10.1002/chem.201501252 |
| 21. | Pietrement, C.; Sun-Wada, G.-H.; Da Silva, N.; McKee, M.; Marshansky, V.; Brown, D.; Futai, M.; Breton, S. Biol. Reprod. 2006, 74, 185–194. doi:10.1095/biolreprod.105.043752 |
| 23. | Scherer, O.; Steinmetz, H.; Kaether, C.; Weinigel, C.; Barz, D.; Kleinert, H.; Menche, D.; Müller, R.; Pergola, C.; Werz, O. Biochem. Pharmacol. 2014, 91, 490–500. doi:10.1016/j.bcp.2014.07.028 |
| 87. | Molander, G. A.; Fumagalli, T. J. Org. Chem. 2006, 71, 5743–5747. doi:10.1021/jo0608366 |
| 20. | Toyomura, T.; Murata, Y.; Yamamoto, A.; Oka, T.; Sun-Wada, G.-H.; Wada, Y.; Futai, M. J. Biol. Chem. 2003, 278, 22023–22030. doi:10.1074/jbc.M302436200 |
| 24. | Sasse, F.; Steinmetz, H.; Höfle, G.; Reichenbach, H. J. Antibiot. 2003, 56, 520–525. doi:10.7164/antibiotics.56.520 |
| 25. | Huss, M.; Sasse, F.; Kunze, B.; Jansen, R.; Steinmetz, H.; Ingenhorst, G.; Zeeck, A.; Wieczorek, H. BMC Biochem. 2005, 6, No. 13. doi:10.1186/1471-2091-6-13 |
| 88. | van Horn, D. E.; Negishi, E.-i. J. Am. Chem. Soc. 1978, 100, 2252–2254. doi:10.1021/ja00475a058 |
| 19. | Brown, D.; Paunescu, T. G.; Breton, S.; Marshansky, V. J. Exp. Biol. 2009, 212, 1762–1772. doi:10.1242/jeb.028803 |
| 44. | O'Neil, G. W.; Black, M. J. Synlett 2010, 107–110. doi:10.1055/s-0029-1218537 |
| 18. | Hiesinger, P. R.; Fayyazuddin, A.; Mehta, S. Q.; Rosenmund, T.; Schulze, K. L.; Zhai, R. G.; Verstreken, P.; Cao, Y.; Zhou, Y.; Kunz, J.; Bellen, H. J. Cell 2005, 121, 607–620. doi:10.1016/j.cell.2005.03.012 |
| 22. | Hamm, R.; Chen, Y.-R.; Seo, E.-J.; Zeino, M.; Wu, C.-F.; Müller, R.; Yang, N.-S.; Efferth, T. Biochem. Pharmacol. 2014, 91, 18–30. doi:10.1016/j.bcp.2014.06.018 |
| 46. | King, B. R.; Swick, S. M.; Schaefer, S. L.; Welch, J. R.; Hunter, E. F.; O'Neil, G. W. Synthesis 2014, 46, 2927–2936. doi:10.1055/s-0034-1379003 |
| 33. | Schneider, L. S.; von Schwarzenberg, K.; Lehr, T.; Ulrich, M.; Kubisch-Dohmen, R.; Liebl, J.; Trauner, D.; Menche, D.; Vollmar, A. M. Cancer Res. 2015, 75, 2863–2874. doi:10.1158/0008-5472.CAN-14-2097 |
| 31. | Schempp, C. M.; von Schwarzenberg, K.; Schreiner, L.; Kubisch, R.; Müller, R.; Wagner, E.; Vollmar, A. M. Mol. Cancer Ther. 2014, 13, 926–937. doi:10.1158/1535-7163.MCT-13-0484 |
| 32. | von Schwarzenberg, K.; Lajtos, T.; Simon, L.; Müller, R.; Vereb, G.; Vollmar, A. M. Mol. Oncol. 2014, 8, 9–19. doi:10.1016/j.molonc.2013.08.011 |
| 93. | Mandal, A. K.; Schneekloth, J. S., Jr.; Kuramochi, K.; Crews, C. M. Org. Lett. 2006, 8, 427–430. doi:10.1021/ol052620g |
| 94. | Paterson, I.; Yeung, K.-S.; Smaill, J. B. Synlett 1993, 774–776. doi:10.1055/s-1993-22605 |
| 91. | Loiseleur, O.; Koch, G.; Cercus, J.; Schürch, F. Org. Process Res. Dev. 2005, 9, 259–271. doi:10.1021/op049807s |
| 92. | Dineen, T. A.; Roush, W. R. Org. Lett. 2004, 6, 2043–2046. doi:10.1021/ol049331x |
| 45. | Tran, A. B.; Melly, G. C.; Doucette, R.; Ashcraft, B.; Sebren, L. J.; Havko, N.; Young, J. C.; O'Neil, G. W. Org. Biomol. Chem. 2011, 9, 7671–7674. doi:10.1039/c1ob06446k |
| 46. | King, B. R.; Swick, S. M.; Schaefer, S. L.; Welch, J. R.; Hunter, E. F.; O'Neil, G. W. Synthesis 2014, 46, 2927–2936. doi:10.1055/s-0034-1379003 |
| 48. | O'Neil, G. W.; Craig, A. M.; Williams, J. R.; Young, J. C.; Spiegel, P. C. Synlett 2017, 28, 1101–1105. doi:10.1055/s-0036-1588413 |
| 59. | Hassfeld, J.; Farès, C.; Steinmetz, H.; Carlomagno, T.; Menche, D. Org. Lett. 2006, 8, 4751–4754. doi:10.1021/ol061831y |
| 60. | Farès, C.; Hassfeld, J.; Menche, D.; Carlomagno, T. Angew. Chem., Int. Ed. 2008, 47, 3722–3726. doi:10.1002/anie.200800225 |
| 44. | O'Neil, G. W.; Black, M. J. Synlett 2010, 107–110. doi:10.1055/s-0029-1218537 |
| 45. | Tran, A. B.; Melly, G. C.; Doucette, R.; Ashcraft, B.; Sebren, L. J.; Havko, N.; Young, J. C.; O'Neil, G. W. Org. Biomol. Chem. 2011, 9, 7671–7674. doi:10.1039/c1ob06446k |
| 46. | King, B. R.; Swick, S. M.; Schaefer, S. L.; Welch, J. R.; Hunter, E. F.; O'Neil, G. W. Synthesis 2014, 46, 2927–2936. doi:10.1055/s-0034-1379003 |
| 47. | Swick, S. M.; Schaefer, S. L.; O'Neil, G. W. Tetrahedron Lett. 2015, 56, 4039–4042. doi:10.1016/j.tetlet.2015.05.014 |
| 48. | O'Neil, G. W.; Craig, A. M.; Williams, J. R.; Young, J. C.; Spiegel, P. C. Synlett 2017, 28, 1101–1105. doi:10.1055/s-0036-1588413 |
| 49. | Moulin, E.; Nevado, C.; Gagnepain, J.; Kelter, G.; Fiebig, H.-H.; Fürstner, A. Tetrahedron 2010, 66, 6421–6428. doi:10.1016/j.tet.2010.05.043 |
| 50. | Xu, S.; Negishi, E.-i. Heterocycles 2014, 88, 845–877. doi:10.3987/REV-13-SR(S)5 |
| 51. | Pop, L.; Lassalas, P.; Bencze, L. C.; Toşa, M. I.; Nagy, B.; Irimie, F. D.; Hoarau, C. Tetrahedron: Asymmetry 2012, 23, 474–481. doi:10.1016/j.tetasy.2012.03.014 |
| 52. | de Paolis, M.; Chataigner, I.; Maddaluno, J. Top. Curr. Chem. 2012, 327, 87–146. doi:10.1007/128_2012_320 |
| 53. | Negishi, E.-i.; Wang, G.; Rao, H.; Xu, Z. J. Org. Chem. 2010, 75, 3151–3182. doi:10.1021/jo1003218 |
| 54. | Huang, Z.; Negishi, E.-i. J. Am. Chem. Soc. 2007, 129, 14788–14792. doi:10.1021/ja0772039 |
| 45. | Tran, A. B.; Melly, G. C.; Doucette, R.; Ashcraft, B.; Sebren, L. J.; Havko, N.; Young, J. C.; O'Neil, G. W. Org. Biomol. Chem. 2011, 9, 7671–7674. doi:10.1039/c1ob06446k |
| 49. | Moulin, E.; Nevado, C.; Gagnepain, J.; Kelter, G.; Fiebig, H.-H.; Fürstner, A. Tetrahedron 2010, 66, 6421–6428. doi:10.1016/j.tet.2010.05.043 |
| 55. | Menche, D.; Hassfeld, J.; Sasse, F.; Huss, M.; Wieczorek, H. Bioorg. Med. Chem. Lett. 2007, 17, 1732–1735. doi:10.1016/j.bmcl.2006.12.073 |
| 56. | Persch, E.; Basile, T.; Bockelmann, S.; Huss, M.; Wieczorek, H.; Carlomagno, T.; Menche, D. Bioorg. Med. Chem. Lett. 2012, 22, 7735–7738. doi:10.1016/j.bmcl.2012.09.081 |
| 57. | Dreisigacker, S.; Latek, D.; Bockelmann, S.; Huss, M.; Wieczorek, H.; Filipek, S.; Gohlke, H.; Menche, D.; Carlomagno, T. J. Chem. Inf. Model. 2012, 52, 2265–2272. doi:10.1021/ci300242d |
| 58. | Reker, D.; Perna, A. M.; Rodrigues, T.; Schneider, P.; Reutlinger, M.; Mönch, B.; Koeberle, A.; Lamers, C.; Gabler, M.; Steinmetz, H.; Müller, R.; Schubert-Zsilavecz, M.; Werz, O.; Schneider, G. Nat. Chem. 2014, 6, 1072–1078. doi:10.1038/nchem.2095 |
| 49. | Moulin, E.; Nevado, C.; Gagnepain, J.; Kelter, G.; Fiebig, H.-H.; Fürstner, A. Tetrahedron 2010, 66, 6421–6428. doi:10.1016/j.tet.2010.05.043 |
| 38. | Menche, D.; Hassfeld, J.; Steinmetz, H.; Huss, M.; Wieczorek, H.; Sasse, F. J. Antibiot. 2007, 60, 328–331. doi:10.1038/ja.2007.43 |
| 39. | Menche, D.; Hassfeld, J.; Steinmetz, H.; Huss, M.; Wieczorek, H.; Sasse, F. Eur. J. Org. Chem. 2007, 1196–1202. doi:10.1002/ejoc.200600912 |
| 40. | Horstmann, N.; Essig, S.; Bockelmann, S.; Wieczorek, H.; Huss, M.; Sasse, F.; Menche, D. J. Nat. Prod. 2011, 74, 1100–1105. doi:10.1021/np200036v |
| 95. | Börding, S.; Bach, T. Chem. Commun. 2014, 50, 4901–4903. doi:10.1039/C4CC01338G |
| 41. | Menche, D.; Hassfeld, J.; Li, J.; Rudolph, S. J. Am. Chem. Soc. 2007, 129, 6100–6101. doi:10.1021/ja071461o |
| 42. | Menche, D.; Hassfeld, J.; Li, J.; Mayer, K.; Rudolph, S. J. Org. Chem. 2009, 74, 7220–7229. doi:10.1021/jo901565n |
| 43. | Roethle, P. A.; Chen, I. T.; Trauner, D. J. Am. Chem. Soc. 2007, 129, 8960–8961. doi:10.1021/ja0733033 |
| 96. | Fürstner, A.; Funel, J.-A.; Tremblay, M.; Bouchez, L. C.; Nevado, C.; Waser, M.; Ackerstaff, J.; Stimson, C. C. Chem. Commun. 2008, 2873–2875. doi:10.1039/B805299A |
| 34. | Schneider, L. S.; Ulrich, M.; Lehr, T.; Menche, D.; Müller, R.; von Schwarzenberg, K. Mol. Oncol. 2016, 10, 1054–1062. doi:10.1016/j.molonc.2016.04.005 |
| 43. | Roethle, P. A.; Chen, I. T.; Trauner, D. J. Am. Chem. Soc. 2007, 129, 8960–8961. doi:10.1021/ja0733033 |
| 26. | Bockelmann, S.; Menche, D.; Rudolph, S.; Bender, T.; Grond, S.; von Zezschwitz, P.; Muench, S. P.; Wieczorek, H.; Huss, M. J. Biol. Chem. 2010, 285, 38304–38314. doi:10.1074/jbc.M110.137539 |
| 27. | Gölz, J. P.; Bockelmann, S.; Mayer, K.; Steinhoff, H.-J.; Wieczorek, H.; Huss, M.; Klare, J. P.; Menche, D. ChemMedChem 2016, 11, 420–428. doi:10.1002/cmdc.201500500 |
| 35. | Pérez-Sayáns, M.; Somoza-Martin, J. M.; Barros-Angueira, F.; Rey, J. M. G.; García-García, A. Cancer Treat. Rev. 2009, 35, 707–713. doi:10.1016/j.ctrv.2009.08.003 |
| 36. | Hernandez, A.; Serrano-Bueno, G.; Perez-Castineira, J. R.; Serrano, A. Curr. Pharm. Des. 2012, 18, 1383–1394. doi:10.2174/138161212799504821 |
| 37. | Cotter, K.; Capecci, J.; Sennoune, S.; Huss, M.; Maier, M.; Martinez-Zaguilan, R.; Forgac, M. J. Biol. Chem. 2015, 290, 3680–3692. doi:10.1074/jbc.M114.611210 |
| 45. | Tran, A. B.; Melly, G. C.; Doucette, R.; Ashcraft, B.; Sebren, L. J.; Havko, N.; Young, J. C.; O'Neil, G. W. Org. Biomol. Chem. 2011, 9, 7671–7674. doi:10.1039/c1ob06446k |
© 2017 Scheeff and Menche; licensee Beilstein-Institut.
This is an Open Access article under the terms of the Creative Commons Attribution License (http://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
The license is subject to the Beilstein Journal of Organic Chemistry terms and conditions: (http://www.beilstein-journals.org/bjoc)