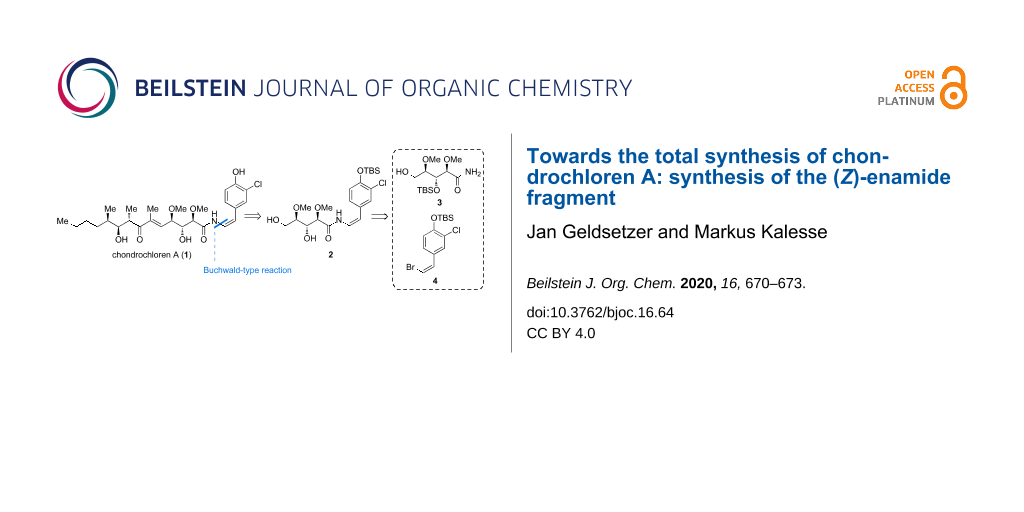
Abstract
The stereoselective synthesis of the (Z)-enamide fragment of chondrochloren (1) is described. A Buchwald-type coupling between amide 3 and (Z)-bromide 4 was used to generate the required fragment. The employed amide 3 comprising three chiral centers was obtained through a seven-step sequence starting from ᴅ-ribonic acid-1,4-lactone. The (Z)-vinyl bromide 4 is accessible in four steps from 4-hydroxybenzaldehyde. The pivotal cross coupling between both fragments was achieved after extensive experimentation with copper(I) iodide, K2CO3 and N,N′-dimethylethane-1,2-diamine.
Graphical Abstract
Introduction
In the course of our program to provide synthetic access to biologically active natural products we targeted complex polyketides and depsipetides [1-10]. One particular group of compounds of particular focus in our research activities are natural products with enamide moieties [11]. Among these, chondrochloren having a (Z)-enamide moiety features a rare structural motif. The myxobacterial metabolite chondrochloren A (1) was isolated from Chondromyces crocatus (Cmc5) by the groups of Höfle and Reichenbach in 2003 [12]. This PKS/NRPS-derived natural product shows only weak antibiotic effects in agar diffusion tests against Micrococcus luteus, Schizosaccharomyces pombe, Bacillus subtilis and Staphylococcus aureus [12]. The relative and absolute stereochemistry of the molecule was elucidated by a combination of NMR, UV and IR spectroscopy and molecular dynamics calculations (MD, MM2) [12]. However, its (Z)-enamide motif and the polyoxygenated middle segment are synthetically challenging.
Results and Discussion
Synthesis of amide 3
Here we report our investigations on the construction of the segments 3 and 4 as well as our results on the cross coupling between both fragments. Our retrosynthetic approach (Figure 1) divides chondrochloren A (1) into three fragments of similar complexity: amide 2, (Z)-bromide 4 and amide 3. For the coupling of amide 3 and (Z)-bromide 4 we decided to use a (Z)-selective Buchwald-type reaction encouraged by the previous works of the Buchwald group on producing (E)-enamide coupling products [13-15].
Figure 1: Retrosynthetic analysis of chondrochlorene A (1).
Figure 1: Retrosynthetic analysis of chondrochlorene A (1).
The synthesis of the amide 3 started with TIPDS-protection of commercially available ᴅ-ribonic acid-1,4-lactone (5) (Scheme 1). A subsequent transesterification under mild conditions with Bu2SnO provided dihydroxy ester 7 in 72% yield. The 1,3-diol in 7 was methylated with an excess of the Meerwein reagent and TIPDS-removal afforded ester 9 in good yields. A double TBS-protection and liberation of the primary alcohol provided alcohol 11 in an excellent yield which was subjected to aminolysis to provide amide 3 in seven steps and an overall yield of 16% [16-20].
Scheme 1: Synthesis of amide 3 [16-20]. TIPDSCl2 = 1,3-dichloro-1,1,3,3-tetraisopropyldisiloxane, TBSOTf = tert-butyldimethylsilyl trifluoromethanesulfonate, proton sponge = 1,8-bis(N,N-dimethylamino)naphthalene.
Scheme 1: Synthesis of amide 3 [16-20]. TIPDSCl2 = 1,3-dichloro-1,1,3,3-tetraisopropyldisiloxane, TBSOTf = tert-buty...
Synthesis of (Z)-bromide 4
For the synthesis of (Z)-bromide 4 we chose a palladium-catalyzed, stereoselective dehalogenation as the key step (Scheme 2). Therefore, 4-hydroxybenzaldehyde (12) was chlorinated and phenol 13 was protected as TBS ether to afford aldehyde 14 which was then converted into dibromoolefine 15 in good yields using the Corey–Fuchs protocol. Uenishi et al. [21] published an effective way of defunctionalizing dihalogenated double bonds into the corresponding (Z)-monohalogenated derivatives using palladium(II) acetate, triphenylphosphine and tributyltin hydride. Following this procedure, we were able to obtain (Z)-bromide 4 in four steps and an overall yield of 39% [21-24].
Scheme 2: Synthesis of (Z)-bromide 4 using a palladium-catalyzed, stereoselective dehalogenation [21-24]. TBSOTf = tert-butyldimethylsilyl trifluoromethanesulfonate, NCS = N-chlorosuccinimide, 2,6-lutidine = 2,6-dimethylpyridine.
Scheme 2: Synthesis of (Z)-bromide 4 using a palladium-catalyzed, stereoselective dehalogenation [21-24]. TBSOTf = t...
Cross coupling of fragments 3 and 4
The formation of the (Z)-enamide should occur in a copper-catalyzed Buchwald-type reaction (Scheme 3). Based on a previous work of Buchwald and his group [13], we decided to use copper(I) iodide and N,N′-dimethylethylenediamine (DMEDA) as the catalytic system in THF, which was reported to be the solvent of choice in this type of coupling reaction. Potassium carbonate was chosen due to the sensitivity of the amide 3 towards harsh basic conditions. With these conditions we were able to couple (Z)-bromide 4 with amide 3 selectively to yield (Z)-enamide 16. The obtained double bond geometry was confirmed by the indicative NMR coupling constants of 9.6 Hz. Moreover, we observed a concentration dependent formation of the undesired desilylated (Z)-enamide 17 (Table 1). The best results were achieved using a 65 mM solution of the amide 3. Using dry potassium carbonate, purified copper(I) iodide provided the best results for the cross-coupling reaction.
Scheme 3: Cross coupling of amide 3 and (Z)-bromide 4 (see Table 1 for conditions).
Scheme 3: Cross coupling of amide 3 and (Z)-bromide 4 (see Table 1 for conditions).
Table 1: Concentration studies of amide 3 and (Z)-bromide 4 in the Buchwald-type reaction.a
| entry | c (amide 3, mM) | 16 | 17 | 3 |
| 1 | 20 | 33 | – | 31 |
| 2 | 65 | 42 | 11 | – |
| 3 | 136 | 3 | 11 | 2 |
| 4 | 140 | 17 | 27 | – |
aConditions: amide 3 (1.0 equiv), (Z)-bromide 4 (1.0 equiv), DMEDA (0.60 equiv), CuI (0.30 equiv), THF, 65 °C. DMEDA = N,N′-dimethylethylenediamine.
Conclusion
In summary, we established a (Z)-selective Buchwald-type coupling reaction as a key step in the synthesis of an advanced fragment of chondrochloren A (1). The required amide 3 can be synthesized in seven steps with a 16% overall yield [16-20], whereas the (Z)-bromide 4 can be generated in a four-step sequence with a 39% overall yield, including a palladium-catalyzed, stereoselective dehalogenation [21-25].
Supporting Information
| Supporting Information File 1: Experimental procedures and spectral data of the synthesized compounds. | ||
| Format: PDF | Size: 3.0 MB | Download |
References
-
Kalesse, M.; Chary, K. P.; Quitschalle, M.; Burzlaff, A.; Kasper, C.; Scheper, T. Chem. – Eur. J. 2003, 9, 1129–1136. doi:10.1002/chem.200390130
Return to citation in text: [1] -
Ehrlich, G.; Hassfeld, J.; Eggert, U.; Kalesse, M. J. Am. Chem. Soc. 2006, 128, 14038–14039. doi:10.1021/ja0659572
Return to citation in text: [1] -
Ehrlich, G.; Hassfeld, J.; Eggert, U.; Kalesse, M. Chem. – Eur. J. 2008, 14, 2232–2247. doi:10.1002/chem.200701529
Return to citation in text: [1] -
Brodmann, T.; Janssen, D.; Kalesse, M. J. Am. Chem. Soc. 2010, 132, 13610–13611. doi:10.1021/ja107290s
Return to citation in text: [1] -
Hartmann, O.; Kalesse, M. Org. Lett. 2012, 14, 3064–3067. doi:10.1021/ol3011387
Return to citation in text: [1] -
Hartmann, O.; Kalesse, M. Angew. Chem., Int. Ed. 2014, 53, 7335–7338. doi:10.1002/anie.201402259
Return to citation in text: [1] -
Gieseler, M. T.; Kalesse, M. Org. Lett. 2014, 16, 548–551. doi:10.1021/ol403423r
Return to citation in text: [1] -
Gerstmann, L.; Kalesse, M. Chem. – Eur. J. 2016, 22, 11210–11212. doi:10.1002/chem.201602682
Return to citation in text: [1] -
Poock, C.; Kalesse, M. Org. Lett. 2017, 19, 4536–4539. doi:10.1021/acs.orglett.7b02112
Return to citation in text: [1] -
Lücke, D.; Linne, Y.; Hempel, K.; Kalesse, M. Org. Lett. 2018, 20, 4475–4477. doi:10.1021/acs.orglett.8b01768
Return to citation in text: [1] -
Rentsch, A.; Kalesse, M. Angew. Chem., Int. Ed. 2012, 51, 11381–11384. doi:10.1002/anie.201206560
Return to citation in text: [1] -
Jansen, R.; Kunze, B.; Reichenbach, H.; Höfle, G. Eur. J. Org. Chem. 2003, 2684–2689. doi:10.1002/ejoc.200200699
Return to citation in text: [1] [2] [3] -
Jiang, L.; Job, G. E.; Klapars, A.; Buchwald, S. L. Org. Lett. 2003, 5, 3667–3669. doi:10.1021/ol035355c
Return to citation in text: [1] [2] -
Martín, R.; Cuenca, A.; Buchwald, S. L. Org. Lett. 2007, 9, 5521–5524. doi:10.1021/ol7024718
Return to citation in text: [1] -
Kuranaga, T.; Sesoko, Y.; Inoue, M. Nat. Prod. Rep. 2014, 31, 514. doi:10.1039/c3np70103d
See for Cu-mediated enamide formations in the total synthesis of complex peptide natural products.
Return to citation in text: [1] -
Cen, Y.; Sauve, A. A. J. Am. Chem. Soc. 2010, 132, 12286–12298. doi:10.1021/ja910342d
Return to citation in text: [1] [2] [3] -
Hildbrand, S.; Blaser, A.; Parel, S. P.; Leumann, C. J. J. Am. Chem. Soc. 1997, 119, 5499–5511. doi:10.1021/ja9704904
Return to citation in text: [1] [2] [3] -
Baumhof, P.; Mazitschek, R.; Giannis, A. Angew. Chem., Int. Ed. 2001, 40, 3672. doi:10.1002/1521-3773(20011001)40:19<3672::aid-anie3672>3.0.co;2-y
Return to citation in text: [1] [2] [3] -
Schläger, N.; Kirschning, A. Org. Biomol. Chem. 2012, 10, 7721. doi:10.1039/c2ob26185e
Return to citation in text: [1] [2] [3] -
Uenishi, J.; Kawahama, R.; Yonemitsu, O.; Tsuji, J. J. Org. Chem. 1998, 63, 8965–8975. doi:10.1021/jo9812781
Return to citation in text: [1] [2] [3] -
Rama Rao, A. V.; Chakraborty, T. K.; Laxma Reddy, K.; Srinivasa Rao, A. Tetrahedron Lett. 1994, 35, 5043–5046. doi:10.1016/s0040-4039(00)73315-6
Return to citation in text: [1] [2] [3] [4] -
Symkenberg, G.; Kalesse, M. Angew. Chem., Int. Ed. 2014, 53, 1795–1798. doi:10.1002/anie.201309386
Return to citation in text: [1] [2] [3] -
Oppolzer, W.; Birkinshaw, T. N.; Bernardinelli, G. Tetrahedron Lett. 1990, 31, 6995–6998. doi:10.1016/s0040-4039(00)97225-3
Return to citation in text: [1] [2] [3] -
Shen, R.; Porco, J. A. Org. Lett. 2000, 2, 1333–1336. doi:10.1021/ol005800t
Return to citation in text: [1] [2] [3] -
Yang, X.; Liu, H.; Fu, H.; Qiao, R.; Jiang, Y.; Zhao, Y. Synlett 2010, 101–106. doi:10.1055/s-0029-1218530
Return to citation in text: [1]
| 1. | Kalesse, M.; Chary, K. P.; Quitschalle, M.; Burzlaff, A.; Kasper, C.; Scheper, T. Chem. – Eur. J. 2003, 9, 1129–1136. doi:10.1002/chem.200390130 |
| 2. | Ehrlich, G.; Hassfeld, J.; Eggert, U.; Kalesse, M. J. Am. Chem. Soc. 2006, 128, 14038–14039. doi:10.1021/ja0659572 |
| 3. | Ehrlich, G.; Hassfeld, J.; Eggert, U.; Kalesse, M. Chem. – Eur. J. 2008, 14, 2232–2247. doi:10.1002/chem.200701529 |
| 4. | Brodmann, T.; Janssen, D.; Kalesse, M. J. Am. Chem. Soc. 2010, 132, 13610–13611. doi:10.1021/ja107290s |
| 5. | Hartmann, O.; Kalesse, M. Org. Lett. 2012, 14, 3064–3067. doi:10.1021/ol3011387 |
| 6. | Hartmann, O.; Kalesse, M. Angew. Chem., Int. Ed. 2014, 53, 7335–7338. doi:10.1002/anie.201402259 |
| 7. | Gieseler, M. T.; Kalesse, M. Org. Lett. 2014, 16, 548–551. doi:10.1021/ol403423r |
| 8. | Gerstmann, L.; Kalesse, M. Chem. – Eur. J. 2016, 22, 11210–11212. doi:10.1002/chem.201602682 |
| 9. | Poock, C.; Kalesse, M. Org. Lett. 2017, 19, 4536–4539. doi:10.1021/acs.orglett.7b02112 |
| 10. | Lücke, D.; Linne, Y.; Hempel, K.; Kalesse, M. Org. Lett. 2018, 20, 4475–4477. doi:10.1021/acs.orglett.8b01768 |
| 12. | Jansen, R.; Kunze, B.; Reichenbach, H.; Höfle, G. Eur. J. Org. Chem. 2003, 2684–2689. doi:10.1002/ejoc.200200699 |
| 12. | Jansen, R.; Kunze, B.; Reichenbach, H.; Höfle, G. Eur. J. Org. Chem. 2003, 2684–2689. doi:10.1002/ejoc.200200699 |
| 12. | Jansen, R.; Kunze, B.; Reichenbach, H.; Höfle, G. Eur. J. Org. Chem. 2003, 2684–2689. doi:10.1002/ejoc.200200699 |
| 16. | Cen, Y.; Sauve, A. A. J. Am. Chem. Soc. 2010, 132, 12286–12298. doi:10.1021/ja910342d |
| 17. | Hildbrand, S.; Blaser, A.; Parel, S. P.; Leumann, C. J. J. Am. Chem. Soc. 1997, 119, 5499–5511. doi:10.1021/ja9704904 |
| 18. | Baumhof, P.; Mazitschek, R.; Giannis, A. Angew. Chem., Int. Ed. 2001, 40, 3672. doi:10.1002/1521-3773(20011001)40:19<3672::aid-anie3672>3.0.co;2-y |
| 19. | Schläger, N.; Kirschning, A. Org. Biomol. Chem. 2012, 10, 7721. doi:10.1039/c2ob26185e |
| 20. | Uenishi, J.; Kawahama, R.; Yonemitsu, O.; Tsuji, J. J. Org. Chem. 1998, 63, 8965–8975. doi:10.1021/jo9812781 |
| 11. | Rentsch, A.; Kalesse, M. Angew. Chem., Int. Ed. 2012, 51, 11381–11384. doi:10.1002/anie.201206560 |
| 21. | Rama Rao, A. V.; Chakraborty, T. K.; Laxma Reddy, K.; Srinivasa Rao, A. Tetrahedron Lett. 1994, 35, 5043–5046. doi:10.1016/s0040-4039(00)73315-6 |
| 22. | Symkenberg, G.; Kalesse, M. Angew. Chem., Int. Ed. 2014, 53, 1795–1798. doi:10.1002/anie.201309386 |
| 23. | Oppolzer, W.; Birkinshaw, T. N.; Bernardinelli, G. Tetrahedron Lett. 1990, 31, 6995–6998. doi:10.1016/s0040-4039(00)97225-3 |
| 24. | Shen, R.; Porco, J. A. Org. Lett. 2000, 2, 1333–1336. doi:10.1021/ol005800t |
| 25. | Yang, X.; Liu, H.; Fu, H.; Qiao, R.; Jiang, Y.; Zhao, Y. Synlett 2010, 101–106. doi:10.1055/s-0029-1218530 |
| 21. | Rama Rao, A. V.; Chakraborty, T. K.; Laxma Reddy, K.; Srinivasa Rao, A. Tetrahedron Lett. 1994, 35, 5043–5046. doi:10.1016/s0040-4039(00)73315-6 |
| 21. | Rama Rao, A. V.; Chakraborty, T. K.; Laxma Reddy, K.; Srinivasa Rao, A. Tetrahedron Lett. 1994, 35, 5043–5046. doi:10.1016/s0040-4039(00)73315-6 |
| 22. | Symkenberg, G.; Kalesse, M. Angew. Chem., Int. Ed. 2014, 53, 1795–1798. doi:10.1002/anie.201309386 |
| 23. | Oppolzer, W.; Birkinshaw, T. N.; Bernardinelli, G. Tetrahedron Lett. 1990, 31, 6995–6998. doi:10.1016/s0040-4039(00)97225-3 |
| 24. | Shen, R.; Porco, J. A. Org. Lett. 2000, 2, 1333–1336. doi:10.1021/ol005800t |
| 16. | Cen, Y.; Sauve, A. A. J. Am. Chem. Soc. 2010, 132, 12286–12298. doi:10.1021/ja910342d |
| 17. | Hildbrand, S.; Blaser, A.; Parel, S. P.; Leumann, C. J. J. Am. Chem. Soc. 1997, 119, 5499–5511. doi:10.1021/ja9704904 |
| 18. | Baumhof, P.; Mazitschek, R.; Giannis, A. Angew. Chem., Int. Ed. 2001, 40, 3672. doi:10.1002/1521-3773(20011001)40:19<3672::aid-anie3672>3.0.co;2-y |
| 19. | Schläger, N.; Kirschning, A. Org. Biomol. Chem. 2012, 10, 7721. doi:10.1039/c2ob26185e |
| 20. | Uenishi, J.; Kawahama, R.; Yonemitsu, O.; Tsuji, J. J. Org. Chem. 1998, 63, 8965–8975. doi:10.1021/jo9812781 |
| 13. | Jiang, L.; Job, G. E.; Klapars, A.; Buchwald, S. L. Org. Lett. 2003, 5, 3667–3669. doi:10.1021/ol035355c |
| 16. | Cen, Y.; Sauve, A. A. J. Am. Chem. Soc. 2010, 132, 12286–12298. doi:10.1021/ja910342d |
| 17. | Hildbrand, S.; Blaser, A.; Parel, S. P.; Leumann, C. J. J. Am. Chem. Soc. 1997, 119, 5499–5511. doi:10.1021/ja9704904 |
| 18. | Baumhof, P.; Mazitschek, R.; Giannis, A. Angew. Chem., Int. Ed. 2001, 40, 3672. doi:10.1002/1521-3773(20011001)40:19<3672::aid-anie3672>3.0.co;2-y |
| 19. | Schläger, N.; Kirschning, A. Org. Biomol. Chem. 2012, 10, 7721. doi:10.1039/c2ob26185e |
| 20. | Uenishi, J.; Kawahama, R.; Yonemitsu, O.; Tsuji, J. J. Org. Chem. 1998, 63, 8965–8975. doi:10.1021/jo9812781 |
| 13. | Jiang, L.; Job, G. E.; Klapars, A.; Buchwald, S. L. Org. Lett. 2003, 5, 3667–3669. doi:10.1021/ol035355c |
| 14. | Martín, R.; Cuenca, A.; Buchwald, S. L. Org. Lett. 2007, 9, 5521–5524. doi:10.1021/ol7024718 |
| 15. |
Kuranaga, T.; Sesoko, Y.; Inoue, M. Nat. Prod. Rep. 2014, 31, 514. doi:10.1039/c3np70103d
See for Cu-mediated enamide formations in the total synthesis of complex peptide natural products. |
| 21. | Rama Rao, A. V.; Chakraborty, T. K.; Laxma Reddy, K.; Srinivasa Rao, A. Tetrahedron Lett. 1994, 35, 5043–5046. doi:10.1016/s0040-4039(00)73315-6 |
| 22. | Symkenberg, G.; Kalesse, M. Angew. Chem., Int. Ed. 2014, 53, 1795–1798. doi:10.1002/anie.201309386 |
| 23. | Oppolzer, W.; Birkinshaw, T. N.; Bernardinelli, G. Tetrahedron Lett. 1990, 31, 6995–6998. doi:10.1016/s0040-4039(00)97225-3 |
| 24. | Shen, R.; Porco, J. A. Org. Lett. 2000, 2, 1333–1336. doi:10.1021/ol005800t |
© 2020 Geldsetzer and Kalesse; licensee Beilstein-Institut.
This is an Open Access article under the terms of the Creative Commons Attribution License (http://creativecommons.org/licenses/by/4.0). Please note that the reuse, redistribution and reproduction in particular requires that the authors and source are credited.
The license is subject to the Beilstein Journal of Organic Chemistry terms and conditions: (https://www.beilstein-journals.org/bjoc)