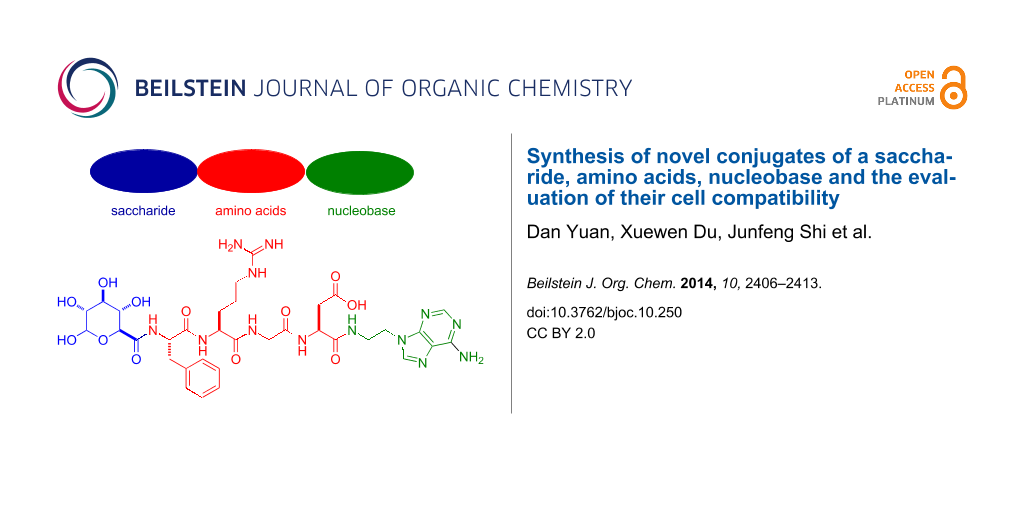
Abstract
This article reports the synthesis of a novel type of conjugate of three fundamental biological build blocks (i.e., saccharide, amino acids, and nucleobase) and their cell compatibility. The facile synthesis starts with the synthesis of nucleobase and saccharide derivatives, then uses solid-phase peptide synthesis (SPPS) to build the peptide segment (Phe-Arg-Gly-Asp or naphthAla-Phe-Arg-Gly-Asp with fully protected groups), and later, an amidation reaction in liquid phase connects these three parts together. The overall yield of these multiple step synthesis is about 34%. Besides exhibiting excellent solubility, these conjugates of saccharide–amino acids–nucleobase (SAN), like the previously reported conjugates of nucleobase–amino acids–saccharide (NAS) and nucleobase–saccharide–amino acids (NSA), are mammalian cell compatible.
Graphical Abstract
Introduction
As a result of evolution, nature selects saccharides, peptides, and nucleobases as the fundamental building blocks for the creation of biomacromolecules, which lay the molecular foundations of life. This simple fact and the self-assembly of small molecules in water have inspired us to explore the conjugates of those three types of building blocks for generating supramolecular nanofibers in water [1-15]. Recently, we have demonstrated that not only the conjugates of nucleobase–amino acids–saccharide (NAS) [16-18] but also the conjugates of nucleobase–saccharide–amino acids (NSA) [19] are able to self-assemble to form supramolecular hydrogels and exhibit promising biological functions, such as promoting the development of zygotes [20]. Moreover, both NAS- and NSA-type conjugates exhibit excellent cell compatibility. Except for a few known motifs (naphthalene [21,22], Fmoc [23,24], lipid [25]), it is still challenging to judge which molecules could self-assemble to form ordered nanostructures. This challenge requires the molecular engineering of the conjugates and intense study of their properties to provide a molecular basis for the understanding of the fundamental correlation between molecular structure and self-assembly.
Here we investigate new molecular conjugates made of basic building blocks by connecting a saccharide segment to amino acids, and nucleobase (i.e., saccharide–amino acids–nucleobase, we term it SAN-type). As shown in Figure 1, in the conjugates 1–4, the saccharide is glucuronic acid, the amino acid segment consists of Phe-Arg-Gly-Asp or naphthAla (3-(2-naphthyl)-alanine)-Phe-Arg-Gly-Asp, and the nucleobase is thymine or adenine. Our results show that the conjugates synthesized and examined in this work exhibit excellent cell compatibility but are unable to self-assemble in water to form hydrogels. Their excellent solubility in water implies that these molecules may find applications for breaking up molecular aggregates in water, a much-needed property for warranting genuine monomeric ligand–receptor interactions. The conjugates 5–8, without a saccharide segment, have good solubility at acidic conditions, also present great cell compatibility and dissolve well in water. These results imply that glucuronic acid is unlikely to be the key factor for cell compatibility of SAN-type conjugates.
Results and Discussion
Molecular design
Figure 1 shows the structures of the designed molecules based on the mutation of the sequence of the connection of nucleobase, amino acids, and saccharide. To explore the properties of the conjugates dependent on the relative positions of the building blocks we synthesized conjugates having the following order: saccharide–amino acids–nucleobase (i.e., SAN-type, 1–4). Although oligosaccharide moieties serve as constituents of glycoproteins in a broad range of cell–cell and cell–matrix recognition events, the introduction of glucuronic acid into peptides at the N-terminus (Figure 1) is rare and worth the exploration [26]. As an amino acid, Phe or naphthAla increases molecular aromatic–aromatic interactions [19,21,27]. Arg-Gly-Asp, which is a well-established tripeptidic epitope, that modulates mammalian cell adhesion through binding with integrins on the cell membrane [28,29]. Thymine or adenine, as a unique types of heteroatom aromatics, not only promote self-assembly [30], but also have the capability for DNA delivery [16]. To examine the role of the saccharide, we also designed and synthesized the respective conjugates of amino acids and nucleobase without the glucuronic moiety (5–8) for comparison.
Figure 1: Chemical structures of the saccharide–amino acids–nucleobase conjugates (SAN, 1–4) and amino acids–nucleobase (AN, 5–8).
Figure 1: Chemical structures of the saccharide–amino acids–nucleobase conjugates (SAN, 1–4) and amino acids–...
Synthesis
Schemes 1–4 show the syntheses of the SAN conjugates formed by the reaction of the amino acid segment with the nucleobase and the saccharide derivative. The key steps include N-alkylation, acetylation, solid-phase peptide synthesis (SPPS) and N-hydroxysuccinimide (NHS)/N,N-diisopropylcarbodiimide (DIC)-catalyzed amidation reaction. As demonstrated by the example of the synthesis of 3, the use of reported methods [31-35] (Scheme 1) affords the nucleobase and saccharide derivatives 12, 16, and 19. We used SPPS [36] (Scheme 2) to synthesize the fully protected naphthAla-Phe-Arg-Gly-Asp (20). After loading the first amino acid (Fmoc-Asp(Ot-Bu)-OH) on the 2-chlorotrityl chloride resin, we blocked the resin by dichloromethane (DCM)/methanol (MeOH)/N,N-diisopropylethylamine (DIEA) (8:1.5:0.5), next removed the Fmoc protecting group by 20% piperidine in N,N-dimethylformamide (DMF), then used the coupling reagent N,N,N’,N’-tetramethyl-O-(1H-benzotriazol-1-yl)uronium hexafluorophosphate (HBTU)/DIEA to connect the following N-Fmoc amino acid (i.e., Fmoc-Gly-OH, Fmoc-Arg(pbf)-OH, Fmoc-Phe-OH, Fmoc-3-(2-naphthyl)alanine) by repeating the deprotection and coupling steps. At last, we cleaved the pentapeptide 20 with all protected groups from the resin by using trifluoroethanol (TFE)/DCM (2/8, 2 × 1 hour). The reaction of 20 and NHS/DIC activates the carboxyl group on 20 to couple with 12 at pH 8.5, which results in 21. The subsequent removal of the Fmoc group provides the protected nucleopeptide 22 [30] (Scheme 3). The activation of 19 to react with 22 and the subsequent removal of Fmoc results in 23. Finally, treatment of 23 with 95% trifluoroacetic acid (TFA) for 1 hour and triethylamine (Et3N)/MeOH/H2O 1:4:5) for 2 hours affords conjugate 3 (Scheme 4). By changing the nucleobase and using the similar reaction procedures, we obtained the rest of the conjugates shown in Figure 1.
Scheme 1: Synthesis of nucleobase (thymine and adenine) and saccharide (glucuronic acid) derivatives 12, 16, and 19.
Scheme 1: Synthesis of nucleobase (thymine and adenine) and saccharide (glucuronic acid) derivatives 12, 16, ...
Scheme 2: Solid-phase peptide synthesis of peptide segment Fmoc-naphthAla-Phe-Arg(pbf)-Gly-Asn(Ot-Bu)-OH (20).
Scheme 2: Solid-phase peptide synthesis of peptide segment Fmoc-naphthAla-Phe-Arg(pbf)-Gly-Asn(Ot-Bu)-OH (20)....
Scheme 3: Synthesis of H-naphthAla-Phe-Arg(pbf)-Gly-Asn(Ot-Bu)-adenine (22).
Scheme 3: Synthesis of H-naphthAla-Phe-Arg(pbf)-Gly-Asn(Ot-Bu)-adenine (22).
Scheme 4: Synthesis of saccharide–amino acids–nucleobase conjugate 3.
Scheme 4: Synthesis of saccharide–amino acids–nucleobase conjugate 3.
Inability of hydrogelation
Unlike the NAS- and NSA-type conjugates, none of the SAN-type conjugates form hydrogels at the conditions tested. As shown in Supporting Information File 1, Figure S1A, B, 1 or 2 form a viscous aqueous solution at a concentration of 3 wt % (pH 4.0) after one week aging. Although our previous study shows that the incorporation of naphthAla, a naphthalene-containing unnatural amino acid, into NSA is able to induce hydrogelation [19], the introduction of naphthAla to 1 or 2, which makes conjugate 3 or 4, however, is unable to result in hydrogelation (Supporting Information File 1, Figure S2A and S2B). The corresponding TEM images showed that conjugates 1–4 all form aggregates (upon drying) without ordered structures. Meanwhile, the removal of the saccharide from the SAN-type conjugates, which forms compound 5–8, results in no hydrogel (Supporting Information File 1, Figure S1C, D and Figure S2C, D). These results suggest that the sequences of the three types of fundamental biological building blocks in the conjugates are critical for their self-assembly.
Cell compatibility
To investigate the cell compatibility of the conjugates, we incubated compounds 1–8 with HeLa cells [37] for 3 days at a concentration range from 20 μM to 500 μM. We compared the cytotoxicity of conjugates 1, 2, 5, and 6 in Figure 2. Compounds 5 and 6 serve as the control for compounds 1 and 2 since they do not have a saccharide part. The viability of HeLa cells incubated with 1, 2, 5, and 6 is around 100% at the study concentrations for 3 days. Also conjugates 3, 4, 7, and 8 exhibit little toxicity to HeLa cells (Figure 3). Particularly, the cytotoxicities of conjugates 1, 3–5, and 8 are slightly higher than 100% at 72 h, it is likely originate from the metabolic activity increase in the HeLa cells treated by 1, 3–5, and 8 [38]. These results are consistent with our previous reports that NAS- and NSA-type conjugates are cell compatible, indicating that the sequences of the three types of building blocks have slight influences to their cell responses. Based on these results, we speculate that the cell compatibility of this type of conjugate is not only due to the natural building blocks, but also may originate from their inability to form aggregates at low concentrations.
![[1860-5397-10-250-2]](/bjoc/content/figures/1860-5397-10-250-2.png?scale=2.4&max-width=1024&background=FFFFFF)
Figure 2: Cell viability of HeLa cells incubated with (A) 1, (B) 2, (c) 5, (D) 6 at different concentrations for 3 days.
Figure 2: Cell viability of HeLa cells incubated with (A) 1, (B) 2, (c) 5, (D) 6 at different concentrations ...
![[1860-5397-10-250-3]](/bjoc/content/figures/1860-5397-10-250-3.png?scale=2.4&max-width=1024&background=FFFFFF)
Figure 3: Cell viability of HeLa cells incubated with (A) 3, (B) 4, (C) 7, (D) 8 at different concentrations for 3 days.
Figure 3: Cell viability of HeLa cells incubated with (A) 3, (B) 4, (C) 7, (D) 8 at different concentrations ...
Conclusion
In summary, this article reports the synthesis of novel conjugates containing three fundamental biological build blocks (saccharide, amino acids, and nucleobase) and their cell compatibility. The attachment of the saccharide to the N-terminal of the peptide or the nucleobase to the C-terminal of the peptide apparently reduces the self-assembly ability of these conjugates in water. Although the atomistic details remain to be elucidated, the observation itself is intriguing. These results provide a new understanding of the self-assembly of the conjugates consisting of fundamental building blocks of biomacromolecules. One potential application of these SAN-type conjugates is to minimize unwanted aggregates of drug candidates for increasing the efficacy of drugs, which is a direction worth exploring.
Supporting Information
| Supporting Information File 1: General section, experimental section, TEM images of solutions of 1–8. | ||
| Format: PDF | Size: 279.0 KB | Download |
References
-
Izawa, H.; Kawakami, K.; Sumita, M.; Tateyama, Y.; Hill, J. P.; Ariga, K. J. Mater. Chem. B 2013, 1, 2155–2161. doi:10.1039/c3tb00503h
Return to citation in text: [1] -
Ellis-Behnke, R. G.; Liang, Y.-X.; You, S.-W.; Tay, D. K. C.; Zhang, S.; So, K.-F.; Schneider, G. E. Proc. Natl. Acad. Sci. U. S. A. 2006, 103, 5054–5059. doi:10.1073/pnas.0600559103
Return to citation in text: [1] -
Horii, A.; Wang, X.; Gelain, F.; Zhang, S. PLoS One 2007, 2, e190. doi:10.1371/journal.pone.0000190
Return to citation in text: [1] -
Tian, Y.; Wang, H.; Liu, Y.; Mao, L.; Chen, W.; Zhu, Z.; Liu, W.; Zheng, W.; Zhao, Y.; Kong, D.; Yang, Z.; Zhang, W.; Shao, Y.; Jiang, X. Nano Lett. 2014, 14, 1439–1445. doi:10.1021/nl404560v
Return to citation in text: [1] -
He, M.; Li, J.; Tan, S.; Wang, R.; Zhang, Y. J. Am. Chem. Soc. 2013, 135, 18718–18721. doi:10.1021/ja409000b
Return to citation in text: [1] -
Qin, L.; Duan, P.; Xie, F.; Zhang, L.; Liu, M. Chem. Commun. 2013, 49, 10823–10825. doi:10.1039/c3cc47004k
Return to citation in text: [1] -
Ochi, R.; Kurotani, K.; Ikeda, M.; Kiyonaka, S.; Hamachi, I. Chem. Commun. 2013, 49, 2115–2117. doi:10.1039/c2cc37908b
Return to citation in text: [1] -
Yamamichi, S.; Jinno, Y.; Haraya, N.; Oyoshi, T.; Tomitori, H.; Kashiwagi, K.; Yamanaka, M. Chem. Commun. 2011, 47, 10344–10346. doi:10.1039/c1cc13826j
Return to citation in text: [1] -
Kiyonaka, S.; Sada, K.; Yoshimura, I.; Shinkai, S.; Kato, N.; Hamachi, I. Nat. Mater. 2004, 3, 58–64. doi:10.1038/nmat1034
Return to citation in text: [1] -
Tamesue, S.; Takashima, Y.; Yamaguchi, H.; Shinkai, S.; Harada, A. Angew. Chem., Int. Ed. 2010, 49, 7461–7464. doi:10.1002/anie.201003567
Return to citation in text: [1] -
Araki, K.; Yoshikawa, I. Nucleobase-containing gelators. Low Molecular Mass Gelator; Topics in Current Chemistry, Vol. 256; Springer: Berlin, Heidelberg, 2005; pp 133–165. doi:10.1007/b107173
Return to citation in text: [1] -
Babu, S. S.; Praveen, V. K.; Ajayaghosh, A. Chem. Rev. 2014, 114, 1973–2129. doi:10.1021/cr400195e
Return to citation in text: [1] -
Shome, A.; Debnath, S.; Das, P. K. Langmuir 2008, 24, 4280–4288. doi:10.1021/la704024p
Return to citation in text: [1] -
Zhou, M.; Smith, A. M.; Das, A. K.; Hodson, N. W.; Collins, R. F.; Ulijn, R. V.; Gough, J. E. Biomaterials 2009, 30, 2523–2530. doi:10.1016/j.biomaterials.2009.01.010
Return to citation in text: [1] -
Tan, H.; Xiao, C.; Sun, J.; Xiong, D.; Hu, X. Chem. Commun. 2012, 48, 10289–10291. doi:10.1039/c2cc35449g
Return to citation in text: [1] -
Li, X.; Kuang, Y.; Shi, J.; Gao, Y.; Lin, H.-C.; Xu, B. J. Am. Chem. Soc. 2011, 133, 17513–17518. doi:10.1021/ja208456k
Return to citation in text: [1] [2] -
Li, X.; Du, X.; Gao, Y.; Shi, J.; Kuang, Y.; Xu, B. Soft Matter 2012, 8, 7402–7407. doi:10.1039/c2sm25725d
Return to citation in text: [1] -
Li, X.; Kuang, Y.; Xu, B. Soft Matter 2012, 8, 2801–2806. doi:10.1039/c2sm06920b
Return to citation in text: [1] -
Wu, D.; Zhou, J.; Shi, J.; Du, X.; Xu, B. Chem. Commun. 2014, 50, 1992–1994. doi:10.1039/c3cc48946a
Return to citation in text: [1] [2] [3] -
Du, X.; Zhou, J.; Guvench, O.; Sangiorgi, F. O.; Li, X.; Zhou, N.; Xu, B. Bioconjugate Chem. 2014, 25, 1031–1035. doi:10.1021/bc500187m
Return to citation in text: [1] -
Zhang, Y.; Kuang, Y.; Gao, Y.; Xu, B. Langmuir 2011, 27, 529–537. doi:10.1021/la1020324
Return to citation in text: [1] [2] -
Chen, L.; Morris, K.; Laybourn, A.; Elias, D.; Hicks, M. R.; Rodger, A.; Serpell, L.; Adams, D. J. Langmuir 2010, 26, 5232–5242. doi:10.1021/la903694a
Return to citation in text: [1] -
Smith, A. M.; Williams, R. J.; Tang, C.; Coppo, P.; Collins, R. F.; Turner, M. L.; Saiani, A.; Ulijn, R. V. Adv. Mater. 2008, 20, 37–41. doi:10.1002/adma.200701221
Return to citation in text: [1] -
Fleming, S.; Debnath, S.; Frederix, P. W. J. M.; Tuttle, T.; Ulijn, R. V. Chem. Commun. 2013, 49, 10587–10589. doi:10.1039/c3cc45822a
Return to citation in text: [1] -
Palmer, L. C.; Stupp, S. I. Acc. Chem. Res. 2008, 41, 1674–1684. doi:10.1021/ar8000926
Return to citation in text: [1] -
Venkateswara Rao, B.; Dhokale, S.; Rajamohanan, P. R.; Hotha, S. Chem. Commun. 2013, 49, 10808–10810. doi:10.1039/c3cc46634e
Return to citation in text: [1] -
Shi, J.; Gao, Y.; Yang, Z.; Xu, B. Beilstein J. Org. Chem. 2011, 7, 167–172. doi:10.3762/bjoc.7.23
Return to citation in text: [1] -
Ruoslahti, E. Annu. Rev. Cell Dev. Biol. 1996, 12, 697–715. doi:10.1146/annurev.cellbio.12.1.697
Return to citation in text: [1] -
Ojima, I.; Chakravarty, S.; Dong, Q. Bioorg. Med. Chem. 1995, 3, 337–360. doi:10.1016/0968-0896(95)00036-G
Return to citation in text: [1] -
Li, X.; Kuang, Y.; Lin, H.-C.; Gao, Y.; Shi, J.; Xu, B. Angew. Chem., Int. Ed. 2011, 50, 9365–9369. doi:10.1002/anie.201103641
Return to citation in text: [1] [2] -
Pandey, M. D.; Mishra, A. K.; Chandrasekhar, V.; Verma, S. Inorg. Chem. 2010, 49, 2020–2022. doi:10.1021/ic9022008
Return to citation in text: [1] -
Elidrisi, I.; Negin, S.; Bhatt, P. V.; Govender, T.; Kruger, H. G.; Gokel, G. W.; Maguire, G. E. M. Org. Biomol. Chem. 2011, 9, 4498–4506. doi:10.1039/c0ob01236j
Return to citation in text: [1] -
Das, B. K.; Shibata, N.; Takeuchi, Y. J. Chem. Soc., Perkin Trans. 1 2002, 197–206. doi:10.1039/B108760F
Return to citation in text: [1] -
Tosin, M.; Murphy, P. V. Org. Lett. 2002, 4, 3675–3678. doi:10.1021/ol026629j
Return to citation in text: [1] -
Cagnoni, A. J.; Varela, O.; Gouin, S. G.; Kovensky, J.; Uhrig, M. L. J. Org. Chem. 2011, 76, 3064–3077. doi:10.1021/jo102421e
Return to citation in text: [1] -
Chan, W. C.; White, P. D. Basic Procedures. In Fmoc Solid Phase Peptide Synthesis: A Practical Approach; Chan, W. C.; White, P. D., Eds.; Oxford University Press Inc.: New York, 2000; pp 41–73.
Return to citation in text: [1] -
Masters, J. R. Nat. Rev. Cancer 2002, 2, 315–319. doi:10.1038/nrc775
Return to citation in text: [1] -
Wang, P.; Henning, S. M.; Heber, D. PLoS One 2010, 5, e10202. doi:10.1371/journal.pone.0010202
Return to citation in text: [1]
| 38. | Wang, P.; Henning, S. M.; Heber, D. PLoS One 2010, 5, e10202. doi:10.1371/journal.pone.0010202 |
| 1. | Izawa, H.; Kawakami, K.; Sumita, M.; Tateyama, Y.; Hill, J. P.; Ariga, K. J. Mater. Chem. B 2013, 1, 2155–2161. doi:10.1039/c3tb00503h |
| 2. | Ellis-Behnke, R. G.; Liang, Y.-X.; You, S.-W.; Tay, D. K. C.; Zhang, S.; So, K.-F.; Schneider, G. E. Proc. Natl. Acad. Sci. U. S. A. 2006, 103, 5054–5059. doi:10.1073/pnas.0600559103 |
| 3. | Horii, A.; Wang, X.; Gelain, F.; Zhang, S. PLoS One 2007, 2, e190. doi:10.1371/journal.pone.0000190 |
| 4. | Tian, Y.; Wang, H.; Liu, Y.; Mao, L.; Chen, W.; Zhu, Z.; Liu, W.; Zheng, W.; Zhao, Y.; Kong, D.; Yang, Z.; Zhang, W.; Shao, Y.; Jiang, X. Nano Lett. 2014, 14, 1439–1445. doi:10.1021/nl404560v |
| 5. | He, M.; Li, J.; Tan, S.; Wang, R.; Zhang, Y. J. Am. Chem. Soc. 2013, 135, 18718–18721. doi:10.1021/ja409000b |
| 6. | Qin, L.; Duan, P.; Xie, F.; Zhang, L.; Liu, M. Chem. Commun. 2013, 49, 10823–10825. doi:10.1039/c3cc47004k |
| 7. | Ochi, R.; Kurotani, K.; Ikeda, M.; Kiyonaka, S.; Hamachi, I. Chem. Commun. 2013, 49, 2115–2117. doi:10.1039/c2cc37908b |
| 8. | Yamamichi, S.; Jinno, Y.; Haraya, N.; Oyoshi, T.; Tomitori, H.; Kashiwagi, K.; Yamanaka, M. Chem. Commun. 2011, 47, 10344–10346. doi:10.1039/c1cc13826j |
| 9. | Kiyonaka, S.; Sada, K.; Yoshimura, I.; Shinkai, S.; Kato, N.; Hamachi, I. Nat. Mater. 2004, 3, 58–64. doi:10.1038/nmat1034 |
| 10. | Tamesue, S.; Takashima, Y.; Yamaguchi, H.; Shinkai, S.; Harada, A. Angew. Chem., Int. Ed. 2010, 49, 7461–7464. doi:10.1002/anie.201003567 |
| 11. | Araki, K.; Yoshikawa, I. Nucleobase-containing gelators. Low Molecular Mass Gelator; Topics in Current Chemistry, Vol. 256; Springer: Berlin, Heidelberg, 2005; pp 133–165. doi:10.1007/b107173 |
| 12. | Babu, S. S.; Praveen, V. K.; Ajayaghosh, A. Chem. Rev. 2014, 114, 1973–2129. doi:10.1021/cr400195e |
| 13. | Shome, A.; Debnath, S.; Das, P. K. Langmuir 2008, 24, 4280–4288. doi:10.1021/la704024p |
| 14. | Zhou, M.; Smith, A. M.; Das, A. K.; Hodson, N. W.; Collins, R. F.; Ulijn, R. V.; Gough, J. E. Biomaterials 2009, 30, 2523–2530. doi:10.1016/j.biomaterials.2009.01.010 |
| 15. | Tan, H.; Xiao, C.; Sun, J.; Xiong, D.; Hu, X. Chem. Commun. 2012, 48, 10289–10291. doi:10.1039/c2cc35449g |
| 21. | Zhang, Y.; Kuang, Y.; Gao, Y.; Xu, B. Langmuir 2011, 27, 529–537. doi:10.1021/la1020324 |
| 22. | Chen, L.; Morris, K.; Laybourn, A.; Elias, D.; Hicks, M. R.; Rodger, A.; Serpell, L.; Adams, D. J. Langmuir 2010, 26, 5232–5242. doi:10.1021/la903694a |
| 30. | Li, X.; Kuang, Y.; Lin, H.-C.; Gao, Y.; Shi, J.; Xu, B. Angew. Chem., Int. Ed. 2011, 50, 9365–9369. doi:10.1002/anie.201103641 |
| 20. | Du, X.; Zhou, J.; Guvench, O.; Sangiorgi, F. O.; Li, X.; Zhou, N.; Xu, B. Bioconjugate Chem. 2014, 25, 1031–1035. doi:10.1021/bc500187m |
| 19. | Wu, D.; Zhou, J.; Shi, J.; Du, X.; Xu, B. Chem. Commun. 2014, 50, 1992–1994. doi:10.1039/c3cc48946a |
| 19. | Wu, D.; Zhou, J.; Shi, J.; Du, X.; Xu, B. Chem. Commun. 2014, 50, 1992–1994. doi:10.1039/c3cc48946a |
| 31. | Pandey, M. D.; Mishra, A. K.; Chandrasekhar, V.; Verma, S. Inorg. Chem. 2010, 49, 2020–2022. doi:10.1021/ic9022008 |
| 32. | Elidrisi, I.; Negin, S.; Bhatt, P. V.; Govender, T.; Kruger, H. G.; Gokel, G. W.; Maguire, G. E. M. Org. Biomol. Chem. 2011, 9, 4498–4506. doi:10.1039/c0ob01236j |
| 33. | Das, B. K.; Shibata, N.; Takeuchi, Y. J. Chem. Soc., Perkin Trans. 1 2002, 197–206. doi:10.1039/B108760F |
| 34. | Tosin, M.; Murphy, P. V. Org. Lett. 2002, 4, 3675–3678. doi:10.1021/ol026629j |
| 35. | Cagnoni, A. J.; Varela, O.; Gouin, S. G.; Kovensky, J.; Uhrig, M. L. J. Org. Chem. 2011, 76, 3064–3077. doi:10.1021/jo102421e |
| 16. | Li, X.; Kuang, Y.; Shi, J.; Gao, Y.; Lin, H.-C.; Xu, B. J. Am. Chem. Soc. 2011, 133, 17513–17518. doi:10.1021/ja208456k |
| 17. | Li, X.; Du, X.; Gao, Y.; Shi, J.; Kuang, Y.; Xu, B. Soft Matter 2012, 8, 7402–7407. doi:10.1039/c2sm25725d |
| 18. | Li, X.; Kuang, Y.; Xu, B. Soft Matter 2012, 8, 2801–2806. doi:10.1039/c2sm06920b |
| 36. | Chan, W. C.; White, P. D. Basic Procedures. In Fmoc Solid Phase Peptide Synthesis: A Practical Approach; Chan, W. C.; White, P. D., Eds.; Oxford University Press Inc.: New York, 2000; pp 41–73. |
| 19. | Wu, D.; Zhou, J.; Shi, J.; Du, X.; Xu, B. Chem. Commun. 2014, 50, 1992–1994. doi:10.1039/c3cc48946a |
| 21. | Zhang, Y.; Kuang, Y.; Gao, Y.; Xu, B. Langmuir 2011, 27, 529–537. doi:10.1021/la1020324 |
| 27. | Shi, J.; Gao, Y.; Yang, Z.; Xu, B. Beilstein J. Org. Chem. 2011, 7, 167–172. doi:10.3762/bjoc.7.23 |
| 30. | Li, X.; Kuang, Y.; Lin, H.-C.; Gao, Y.; Shi, J.; Xu, B. Angew. Chem., Int. Ed. 2011, 50, 9365–9369. doi:10.1002/anie.201103641 |
| 26. | Venkateswara Rao, B.; Dhokale, S.; Rajamohanan, P. R.; Hotha, S. Chem. Commun. 2013, 49, 10808–10810. doi:10.1039/c3cc46634e |
| 16. | Li, X.; Kuang, Y.; Shi, J.; Gao, Y.; Lin, H.-C.; Xu, B. J. Am. Chem. Soc. 2011, 133, 17513–17518. doi:10.1021/ja208456k |
| 25. | Palmer, L. C.; Stupp, S. I. Acc. Chem. Res. 2008, 41, 1674–1684. doi:10.1021/ar8000926 |
| 23. | Smith, A. M.; Williams, R. J.; Tang, C.; Coppo, P.; Collins, R. F.; Turner, M. L.; Saiani, A.; Ulijn, R. V. Adv. Mater. 2008, 20, 37–41. doi:10.1002/adma.200701221 |
| 24. | Fleming, S.; Debnath, S.; Frederix, P. W. J. M.; Tuttle, T.; Ulijn, R. V. Chem. Commun. 2013, 49, 10587–10589. doi:10.1039/c3cc45822a |
| 28. | Ruoslahti, E. Annu. Rev. Cell Dev. Biol. 1996, 12, 697–715. doi:10.1146/annurev.cellbio.12.1.697 |
| 29. | Ojima, I.; Chakravarty, S.; Dong, Q. Bioorg. Med. Chem. 1995, 3, 337–360. doi:10.1016/0968-0896(95)00036-G |
© 2014 Yuan et al; licensee Beilstein-Institut.
This is an Open Access article under the terms of the Creative Commons Attribution License (http://creativecommons.org/licenses/by/2.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
The license is subject to the Beilstein Journal of Organic Chemistry terms and conditions: (http://www.beilstein-journals.org/bjoc)