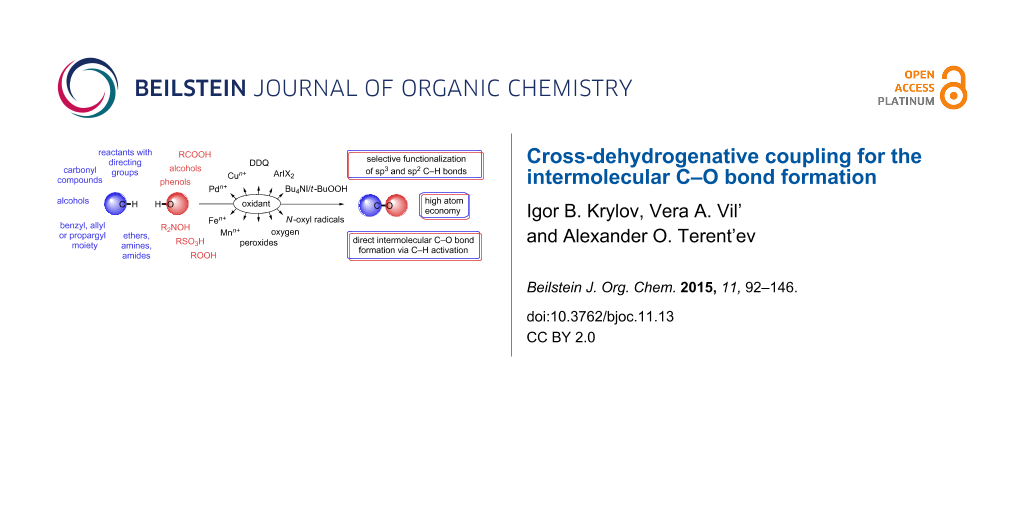
Abstract
The present review summarizes primary publications on the cross-dehydrogenative C–O coupling, with special emphasis on the studies published after 2000. The starting compound, which donates a carbon atom for the formation of a new C–O bond, is called the CH-reagent or the C-reagent, and the compound, an oxygen atom of which is involved in the new bond, is called the OH-reagent or the O-reagent. Alcohols and carboxylic acids are most commonly used as O-reagents; hydroxylamine derivatives, hydroperoxides, and sulfonic acids are employed less often. The cross-dehydrogenative C–O coupling reactions are carried out using different C-reagents, such as compounds containing directing functional groups (amide, heteroaromatic, oxime, and so on) and compounds with activated C–H bonds (aldehydes, alcohols, ketones, ethers, amines, amides, compounds containing the benzyl, allyl, or propargyl moiety). An analysis of the published data showed that the principles at the basis of a particular cross-dehydrogenative C–O coupling reaction are dictated mainly by the nature of the C-reagent. Hence, in the present review the data are classified according to the structures of C-reagents, and, in the second place, according to the type of oxidative systems. Besides the typical cross-dehydrogenative coupling reactions of CH- and OH-reagents, closely related C–H activation processes involving intermolecular C–O bond formation are discussed: acyloxylation reactions with ArI(O2CR)2 reagents and generation of O-reagents in situ from C-reagents (methylarenes, aldehydes, etc.).
Graphical Abstract
Introduction
The development of methods for the cross-dehydrogenative coupling (CDC; or oxidative cross coupling) is an important field of modern organic chemistry. These terms commonly refer to reactions, in which two different molecules are linked by a new bond accompanied by the elimination of a hydrogen atom from each molecule (Scheme 1) [1-15]; however, these terms are also applied to a large number of various reactions with oxidants, which involve the intermolecular formation of new bonds between the starting molecules. For instance, such reactions involve the oxidation of several C–H bonds, the elimination not only of hydrogen atoms but also of other moieties from the starting molecules, the addition at C–C multiple bonds, and so on.
Scheme 1: Cross-dehydrogenative coupling.
Scheme 1: Cross-dehydrogenative coupling.
The cross-dehydrogenative coupling can be employed to form a new bond with high atomic efficiency and does not require additional synthetic steps for the introduction of functional groups (for example, such as -Hal, -OTf, -BR2, -SnR3, -SiR3, -ZnHal, -MgHal) into molecules necessary in other cross-coupling reactions. Therefore, the cross-dehydrogenative coupling is a promising approach to the minimization of byproduct formation and the reduction of the number of steps of the organic synthesis [1-15].
Studies of the cross-dehydrogenative coupling are not only of practical but also of fundamental interest because new aspects of the reactivity of organic compounds have to be found for the performance of these reactions. The prediction of the conditions necessary for the efficient cross-dehydrogenative coupling is an important problem that requires an understanding of the mechanisms of these processes.
Among cross-dehydrogenative coupling reactions, C–C coupling reactions have been studied in most detail [1-14], whereas the C–O coupling is less well-known (Scheme 2). We present the first systematic review of the main approaches to the cross-dehydrogenative C–O coupling. The starting compound, which donates a carbon atom for the formation of a new C–O bond, is called the CH-reagent or the C-reagent, and the compound, an oxygen atom of which is involved in the new bond, is called the OH-reagent or the O-reagent.
Scheme 2: Cross-dehydrogenative C–O coupling.
Scheme 2: Cross-dehydrogenative C–O coupling.
Alcohols and carboxylic acids are most commonly used as O-reagents; hydroxylamine derivatives, hydroperoxides, and sulfonic acids are employed less frequently. In the case of O-reagent PhI(O2CR)2, dehydrogenated carboxylic acid RCO2H is preliminary included in the oxidant, so C–H acyloxylation with PhI(O2CR)2 can be considered as the second stage in a two-step cross-dehydrogenative C–O coupling. The formation of a new C–O bond generally takes place with the involvement of an O-nucleophile, an O-radical, or an O-electrophile. In the oxidative coupling with O-reagents as nucleophiles, electrophilic intermediates that are generated from C-reagents are prone to side transformations. Therefore, O-reagents are often used in excess amounts. The C–O coupling reactions involving O-centered radicals are generally performed under severe conditions. In addition, O-radicals are highly reactive and unstable, and the reactions with these radicals are often non-selective and are accompanied by the formation of alcohols, carbonyl compounds, and fragmentation products. The examples of the C–O bond formation between two molecules using O-electrophiles are rare; electron-deficient peroxides with a specific structure can act as O-electrophiles [16-18]. These processes are not consistent with general Scheme 2 of the cross-dehydrogenative C–O coupling and are not considered in the present review.
The cross-dehydrogenative C–O coupling reactions are carried out using different C-reagents, such as compounds containing directing functional groups (amide, heteroaromatic, oxime, and so on) and compounds with activated C–H bonds (aldehydes, alcohols, ketones, ethers, amines, amides, compounds containing a benzyl, allyl, or propargyl moiety). An analysis of the published data showed that the principles at the basis of a particular cross-dehydrogenative C–O coupling reaction are dictated mainly by the nature of the C-reagent. Hence, in the present review the data are classified according to the structures of C-reagents, and, in the second place, according to the type of oxidative systems. Since structurally different OH-reagents are often involved in C–O coupling reactions of the same type, the classification according to the structures of O-reagents is inconvenient and was not applied.
Some cross-dehydrogenative C–O coupling reactions are cursorily described in reviews on the oxidative C–heteroatom bond formation without the use of metal compounds [15], the Pd(II)-catalyzed oxidative C–C, C–O, and C–N bond formation [3], the transition metal-catalyzed etherification of unactivated C–H bonds [19], the Pd(II)-catalyzed oxidative functionalization at the allylic position of alkenes [20,21], the oxidative functionalization catalyzed by copper compounds to form C–C, C–N, C–O, C–Hal, C–P, and N–N bonds [10], the Bu4NI/t-BuOOH oxidative system [22], selective functionalization of molecules [23], the oxidative esterification and oxidative amidation of aldehydes [24], and the transition metal-catalyzed radical oxidative cross-couplings [13].
The present review summarizes primary publications on the cross-dehydrogenative C–O coupling, with special emphasis on the studies published after 2000. The focus is on the reactions described by general Scheme 2.
Review
1 C-Reagents containing directing groups in cross-dehydrogenative C–O coupling
Nitrogen-containing moieties (amide, pyridine, oxime, etc.) are most commonly used as directing groups, which are responsible for the regioselectivity of the C–O coupling. Most transformations of this type are catalyzed by Pd(II) compounds. Examples of the use of copper and ruthenium compounds as catalysts were also reported. It is commonly assumed that the reaction proceeds via the C–metal bond formation accompanied by the C–H bond cleavage assisted by the directing group of the substrate, which forms a complex with the metal ion. The mechanism of this type of reactions was considered in detail in the publications [25-32].
1.1 Reactions involving C(sp2)–H bonds of aromatic C-reagents
In one of the first publications on the preparative introduction of the –OR group into CH-reagents containing directing groups, 8-methylquinoline, 2-arylpyridines, N-phenylpyrazole, azobenzene, and benzylideneaniline were subjected to the acetoxylation using the Pd(OAc)2/PhI(OAc)2 system [33]. More recently, reactions involving the same and some other directing groups were studied in more detail. In most of the studies, Pd(OAc)2 was used as the catalyst, and PhI(OAc)2 or peroxides served as the oxidants.
The regioselectivity of the ortho-acetoxylation of meta-substituted arylpyridines and N-arylamides 1 was studied [34]. The acetoxylation occurs mainly at the sterically more accessible para-position relative to the substituent R to form product 2. The lowest regioselectivity (2:3 = 6:1) was observed in the case of R = F (Scheme 3).
Scheme 3: Regioselective ortho-acetoxylation of meta-substituted arylpyridines and N-arylamides.
Scheme 3: Regioselective ortho-acetoxylation of meta-substituted arylpyridines and N-arylamides.
The pyridine moiety served as the directing group in many works to accomplish the ortho-acyloxylation of arenes 4 giving products 5 (Table 1). The reactions were catalyzed by copper, palladium, or rhodium salts. Carboxylic acids or their salts, as well as aldehydes, methylarenes, arylethylenes, and arylacetylenes were used as precursors of the acyloxy fragment.
Table 1: ortho-Acyloxylation of arenes 4 directed by pyridine moiety.
|
|
|||
| Conditions | RX | Yield of 5, % | Ref. |
|---|---|---|---|
|
[Rh(cod)Cl]2 (5 mol %)
P(Cyclohexyl)3·HBF4 (7.5 mol %) CuI (40 mol %) phenanthroline (10 mol %) N-methylpyrrolidone, 130 °C, 36 h |
RCOOH (0.5 or 2 equiv); R = Ar, CH=CHPh, Me | 43–85 | [35] |
|
Pd(OAc)2 (10 mol %)
CuI (1 equiv) Ag2CO3 (1 equiv) O2 dichloroethane, 80 °C |
RCOOH; R = Ar, Me | 53–78 | [36] |
|
Cu(OTf)2
K2S2O8 toluene, 130 °C, 24 h |
RCOONa; R = Ar | 35–86 | [37] |
|
Cu(OAc)2 (10 mol %)
t-BuOOH (2–4 equiv) PhCl or without a solvent, 135 °C, 24–40 h |
RCHO or ArCH3; R = Ar, n-Bu, n-Pr | 20–56 | [38] |
|
Cu(OAc)2 (20 mol %)
t-BuOOH (10 moles per mole of arene) PhCl, 120 °C, 10–22 h |
ArCH=CH2 or ArC≡CH (2 equiv) | 48–81 | [39] |
The cross-dehydrogenative C–O coupling with 2-arylpyridines 4 proceeds also in the presence of the Cu(OAc)2/O2 system [40] and under electrochemical oxidation in the presence of Pd(II) salts [41].
The pyrimidine (acetoxylation of 6 to form 7) [42], benzoxazole (acetoxylation of 8 to form 9) [43], benzimidazole (alkoxylation of 10 to form 11) [44], and triazole (acyloxylation of 12 to form 13 [45], alkoxylation of 14 to form 15 [46]) moieties were also used as directing groups for the ortho-acyloxylation and alkoxylation of arenes (Scheme 4).
Scheme 4: ortho-Acyloxylation and alkoxylation of arenes directed by pyrimidine, benzoxazole, benzimidazole and triazole groups.
Scheme 4: ortho-Acyloxylation and alkoxylation of arenes directed by pyrimidine, benzoxazole, benzimidazole a...
The pyridine, pyrimidine, or pyrazole moiety serves as the directing group in the oxidative ortho-alkoxylation of arenes 16 with the Cu(OAc)2/AgOTf/O2 system giving coupling products 17 (Scheme 5) [47]. It is supposed that copper is inserted into the C–H bond of arene, the resulting Cu(II) complex is oxidized by silver(I) ions to Cu(III) complex 18, and the C–O bond is formed via reductive elimination. The drawbacks of this method are the use of large amounts of silver triflate and alcohol and the high temperature of the reaction.
Scheme 5: Cu(OAc)2/AgOTf/O2 oxidative system in the ortho-alkoxylation of arenes.
Scheme 5: Cu(OAc)2/AgOTf/O2 oxidative system in the ortho-alkoxylation of arenes.
The Pd(OAc)2/persulfate system was used in the ortho-alkoxylation of arylnitriles 19–20 [48], N-methoxybenzamides 21 [49], and acetanilides 22 [50] and in the ortho-acetoxylation of acetanilides 22 [51] and sulfoximines 23 [52] to prepare cross-dehydrogenative C–O coupling products 24–30 (Scheme 6). The alkoxylation of 1-naphthonitrile 20 occurs not at the ortho-position but at the 8-position of the aromatic system to give product 26. The acetoxylation takes place under more severe conditions compared with the alkoxylation. The acetoxylation employing the S-methyl-S-2-pyridylsulfoximine moiety as the bidentate directing group can be performed at lower temperature (50 °C instead of 100 °C, as in the case of CH-reagents 22 and 23) [53].
Scheme 6: Pd(OAc)2/persulfate oxidative system in the ortho-alkoxylation and acetoxylation of arenes with nitrile, amide, and sulfoximine directing groups.
Scheme 6: Pd(OAc)2/persulfate oxidative system in the ortho-alkoxylation and acetoxylation of arenes with nit...
The ortho-acetoxylation and methoxylation of O-methyl aryl oximes 31 with Pd(OAc)2 combined with such oxidants as oxone, potassium persulfate, and (diacetoxyiodo)benzene (Scheme 7, coupling products 32 and 33) occur under similar conditions [54]. N-Phenylpyrrolidin-2-one (34) and (3-benzyl-4,5-dihydroisoxazol-5-yl)methyl acetate (35) react in a similar fashion to afford products 36–38. Related ortho-acetoxylation reactions of the aryl group of methoxyimino-2-aryl acetates [55], 2-methoxyimino-2-arylacetamides [55], and O-acetyl aryl oximes [56] in the presence of the Pd(OAc)2/PhI(OAc)2 system were described.
Scheme 7: ortho-Acetoxylation and methoxylation of O-methyl aryl oximes, N-phenylpyrrolidin-2-one, and (3-benzyl-4,5-dihydroisoxazol-5-yl)methyl acetate.
Scheme 7: ortho-Acetoxylation and methoxylation of O-methyl aryl oximes, N-phenylpyrrolidin-2-one, and (3-ben...
The ruthenium-catalyzed ortho-acyloxylation of acetanilides 39 with carboxylic acids in the presence of AgSbF6 and ammonium persulfate afforded products 40 (Scheme 8) [57]. This method can be used for the selective replacement of one of the two hydrogen atoms in the ortho-position of acetanilide; the molar ratio of the C- and O-reagents is close to stoichiometric.
Scheme 8: Ruthenium-catalyzed ortho-acyloxylation of acetanilides.
Scheme 8: Ruthenium-catalyzed ortho-acyloxylation of acetanilides.
The acetoxylation (product 42) and methoxylation (product 43) of N-(2-benzoylphenyl)benzamides 41 at the ortho-position of the benzamide moiety of the substrate were performed using Pd(OAc)2 combined with PhI(OAc)2 as the oxidant (Scheme 9) [58]. The alkoxylation of N-tosylbenzamides 44 in the presence of the same oxidative system takes place at room temperature and gives products 45 [59]. The reactions of benzamides containing the nitro group at the ortho-position are most difficult to perform [58,59].
Scheme 9: Acetoxylation and alkoxylation of arenes with amide directing group using Pd(OAc)2/PhI(OAc)2 oxidative system.
Scheme 9: Acetoxylation and alkoxylation of arenes with amide directing group using Pd(OAc)2/PhI(OAc)2 oxidat...
The Pd(OAc)2/PhI(OAc)2 system was also employed to accomplish the ortho-alkoxylation of azoarenes 46 [60], 2-aryloxypyridines 47 [61], picolinamides 48 [62], and N-(1-methyl-1-(pyridin-2-yl)ethyl)amides 49 [63], resulting in the formation of products 50–54 (Scheme 10).
Scheme 10: Alkoxylation of azoarenes, 2-aryloxypyridines, picolinamides, and N-(1-methyl-1-(pyridin-2-yl)ethyl)amides using the Pd(OAc)2/PhI(OAc)2 oxidative system.
Scheme 10: Alkoxylation of azoarenes, 2-aryloxypyridines, picolinamides, and N-(1-methyl-1-(pyridin-2-yl)ethyl...
The ortho-acetoxylation of compounds containing picolinamide and quinoline-8-amine moieties (55 and 56, respectively) with the Pd(OAc)2/PhI(OAc)2 system in a AcOH/Ac2O mixture, resulting in the formation of products 57, 58, was performed at higher temperature (150 °C, Scheme 11) compared with the alkoxylation of structures 46–49 [64]. Under similar conditions, aryl phosphates and benzyl phosphonic monoacids were subjected to ortho-acetoxylation in the presence of the Pd(OAc)2/PhI(OAc)2 system; (diacetoxyiodo)benzene served as the source of the acetoxy group [65].
Scheme 11: Acetoxylation of compounds containing picolinamide and quinoline-8-amine moieties using the Pd(OAc)2/PhI(OAc)2 system.
Scheme 11: Acetoxylation of compounds containing picolinamide and quinoline-8-amine moieties using the Pd(OAc)2...
The quinoline-8-amine directing moiety was used for copper-catalyzed aerobic ortho-aryloxylation and alkoxylation of arenes 59 with electron donating or electron withdrawing substituents to afford products 60 [66] (Scheme 12). The method is applicable to a wide range of OH-reagents; the molar ratio OH-reagent/CH-reagent was 1:1 for aromatic OH-reagents and 5:1 for aliphatic alcohols. Different organic and inorganic bases were used depending on the structures of substrates.
Scheme 12: (CuOH)2CO3 catalyzed oxidative ortho-etherification using air as oxidant.
Scheme 12: (CuOH)2CO3 catalyzed oxidative ortho-etherification using air as oxidant.
The cross-dehydrogenative coupling of phenols and arenes 61 with a directing group containing a pyridine N-oxide moiety occurs in the presence of Cu(OAc)2 under air atmosphere to form coupling products with one (product 62) or two (product 63) equivalents of phenol (Scheme 13) [67]. The same directing group was used for aerobic ortho-alkoxylation of arenes 64 in the presence of CuCl and K2CO3 to afford monoalkoxylated products 65 [68].
Scheme 13: Copper-catalyzed aerobic alkoxylation and aryloxylation of arenes containing pyridine-N-oxide moiety.
Scheme 13: Copper-catalyzed aerobic alkoxylation and aryloxylation of arenes containing pyridine-N-oxide moiet...
The 2-aminopyridine-1-oxide directing group was used in a rare example of a cobalt-catalyzed oxidative alkoxylation of arenes 66 and alkenes 67 to afford products 68 and 69 under mild contitions [69] (Scheme 14). The directing group can be removed to obtain the corresponding benzoic acid 71 from the cross-dehydrogenative coupling product 70 [69].
Scheme 14: Cobalt-catalyzed aerobic alkoxylation of arenes and alkenes containing pyridine N-oxide moiety.
Scheme 14: Cobalt-catalyzed aerobic alkoxylation of arenes and alkenes containing pyridine N-oxide moiety.
The majority of above mentioned directing groups cannot be easily removed or modified, thus limiting the scope of possible target products. To overcome these limitations, 2-pyridyldiisopropylsilyl (PyDipSi) [70,71] and 2-pyrimidyldiisopropylsilyl (PyrDipSi) [72,73] directing groups were proposed by the research group of Prof. V. Gevorgyan. The PyDipSi group was used for selective monoacetoxylation or pivaloyloxylation of the arene ortho-position [70,71], whereas the PyDipSi group allowed to achieve one-pot sequential acyloxylation of both ortho-positions affording, in particular, orthogonally protected resorcinol derivatives 73 in one step starting from PyrDipSi arenes 72 [72] (Scheme 15).
Scheme 15: Non-symmetric double-fold C–H ortho-acyloxylation.
Scheme 15: Non-symmetric double-fold C–H ortho-acyloxylation.
Pd(OAc)2 was used as catalyst with addition of AgOAc or LiOAc; PhI(OAc)2 or PhI(OPiv)2 were used as oxidants and sources of OAc and OPiv fragments, respectively, the syntheses were performed in dichloroethane at 80 °C for 1–168 h. After ortho-acyloxylation step silyl directing group can be removed or substituted by a desirable moiety (-Aryl, -OH, -B(pin), -I, etc.).
Another example of an easily modifiable directing group is an N-nitroso moiety that can be easily reduced to amine employing Fe/NH4Cl [74]. N-Nitroso directed C–H alkoxylation of arenes 74 was realized using the Pd(MeCN)2Cl2/PhI(OAc)2 oxidative system to obtain products 75 [74] (Scheme 16).
Scheme 16: N-nitroso directed ortho-alkoxylation of arenes.
Scheme 16: N-nitroso directed ortho-alkoxylation of arenes.
1.2 Reactions involving C(sp3)-H bonds of C-reagents with alkyl groups
In some studies, directing groups were used to accomplish the C–O coupling involving sp3-carbon atoms of C-reagents. Scheme 17 presents the alkoxylation of CH-reagents 76, 77 and the acetoxylation of CH-reagents 78. In these examples, the reaction occurs at the methylene group of the molecule (with the sp3-carbon atom) rather than at the ortho-position of the aromatic system, which is also adjacent to the directing group. The Pd(OAc)2/PhI(OAc)2 oxidative system was employed in the methoxylation of dimethylcarbamoyltetrahydrocarbazoles 76 [75] and the acetoxylation of compounds containing the picolinamide directing group 78 [76,77]; the butoxylation of the methylene group of 2-benzylpyridine 77 took place in the presence of the Cu(OAc)2/AgOTf/O2 system [47]. Cross-dehydrogenative C–O coupling products 79–81 were obtained, despite the potential possibility of the more profound oxidation of the methylene group to the keto group.
Scheme 17: Selective alkoxylation and acetoxylation of alkyl groups.
Scheme 17: Selective alkoxylation and acetoxylation of alkyl groups.
The benzylic position of 2-alkylpyridines and related compounds 82 was acetoxylated using the Pd(OAc)2/CuI catalytic system in acetic acid under an oxygen pressure of 8 atm to prepare products 83 (Scheme 18) [78].
Scheme 18: Acetoxylation of 2-alkylpyridines and related compounds.
Scheme 18: Acetoxylation of 2-alkylpyridines and related compounds.
The acyloxylation of methyl or methylene groups of 8-methylquinoline and its derivatives in the presence of the Pd(OAc)2/PhI(OAc)2 [79] and Pd(OAc)2/ligand/O2 [80] oxidative systems was also described.
In the above considered examples, the oxidative C–O coupling occurs with the involvement of methyl or methylene groups directly bonded to the aromatic ring. The C–O coupling reactions involving unactivated alkyl groups of C-reagents are considered below.
The amide moiety was most commonly employed as the directing group. The alkoxylation of alkyl groups of N-(quinolin-8-yl)amides 84 [81], picolinamides 85 [62], and N-(2-pyridin-2-yl)propan-2-yl)amides 86 [63], the trifluoroacetoxylation of amides 87 [82], and the acyloxylation of compounds containing the S-methyl-S-pyridylsulfoximine moiety 88 [83] were accomplished to prepare coupling products 90–95 (Scheme 19). In some cases, the latter reaction proceeds efficiently even at room temperature. Iodine(III) compounds 89, (diacetoxyiodo)benzene, or potassium persulfate served as oxidants. The former three reactions are applicable to a broad range of substrates and alcohols. The drawback is that a large excess of alcohols is required. Unlike the alkoxylation of 84–86, the trifluoroacetoxylation of 87 was performed with structurally simple amides. This method is applicable only to α-disubstituted amides. The reactions with amides containing hydrogen in the α-position give coupling products 93 in substantially lower yields [82]. Similar limitations are encountered when performing the acyloxylation of S-methyl-S-pyridylsulfoximines 88.
Scheme 19: Acyloxylation and alkoxylation of alkyl fragments of substrates containing amide or sulfoximine directing groups.
Scheme 19: Acyloxylation and alkoxylation of alkyl fragments of substrates containing amide or sulfoximine dir...
Using the N-(quinolin-8-yl)amide directing group Pd(OAc)2 catalyzed the double sp3 C–H alkoxylation of 96 for the synthesis of symmetric acetals 97 and unsymmetric acetals [84] (Scheme 20). The method demonstrates good functional group tolerance and the applicability in the synthesis of products containing α-hydrogen next to an amide moiety.
Scheme 20: Palladium-catalyzed double sp3 C–H alkoxylation of N-(quinolin-8-yl)amides for the synthesis of symmetric and unsymmetric acetals.
Scheme 20: Palladium-catalyzed double sp3 C–H alkoxylation of N-(quinolin-8-yl)amides for the synthesis of sym...
Copper-catalyzed acyloxylation of methyl groups of N-(quinolin-8-yl)amides 99 was achieved [85] (Scheme 21, product 100); the reaction requires higher temperatures (170 °C) then Pd(OAc)2 catalyzed alkoxylation of analogous substrates in the presence of iodine(III) oxidants (50–130 °C, Scheme 19 and Scheme 20). Under similar conditions methyl groups of N-(quinolin-8-yl)amides were acetoxylated using the Cu(OAc)2 catalyst (50 mol %) and AgOAc (3 equiv) as acetoxylating agent.
Scheme 21: Copper-catalyzed acyloxylation of methyl groups of N-(quinolin-8-yl)amides.
Scheme 21: Copper-catalyzed acyloxylation of methyl groups of N-(quinolin-8-yl)amides.
The acetoxylation of alkyl groups of O-acetyl oximes 102 taking place in the presence of the Pd(OAc)2/PhI(OAc)2 system in a AcOH/Ac2O mixture affords products 103 (Scheme 22) [56]. The acylation of oxime 101 and the C–H acetoxylation of 102 are performed as a one-pot operation. In the acetoxylation of alkyl groups, the methyl group is more reactive than the methylene one.
Scheme 22: One-pot acylation and sp3 C–H acetoxylation of oximes.
Scheme 22: One-pot acylation and sp3 C–H acetoxylation of oximes.
The O-methyl oxime [54,86] or pyridine moieties [86] were employed as directing groups in the acetoxylation of alkyl groups using the Pd(OAc)2/PhI(OAc)2 system. The synthesis was carried out at 80–100 °C during a period of time from 5 min to 12 h in acetic acid, a 1:1 AcOH/Ac2O mixture, or dichloromethane. More recently, a Pd(OAc)2/NaNO3/O2 catalytic system was developed for acetoxylation of unactivated sp3-C–H bonds using the same directing groups [87]. Air or oxygen (1 atm) played a role of terminal oxidant and sodium nitrate was a redox co-catalyst; the reactions were performed in a AcOH/Ac2O solvent mixture at 100–110 °C for 18 h.
The oxazole moiety also acts as the directing group in the acetoxylation of alkyl groups with Pd(OAc)2/AcOOt-Bu or Pd(OAc)2/lauroyl peroxide oxidative systems; in these reactions acetic anhydride served as the source of the acetoxy groups [88]. Recently, Cu(OAc)2-mediated аcetoxylation of unactivated methyl fragments using the N-(quinolin-8-yl)amide directing group and AgOAc as oxidant was achieved without a Pd catalyst [89].
2 Aldehydes and alcohols as C-reagents in cross-dehydrogenative C–O coupling
There are numerous reactions, in which aldehydes are involved in the oxidative C–O coupling with alcohols under the action of oxidizing agents to form esters. In some cases, primary alcohols are used instead of aldehydes. It is commonly assumed that, under these reaction conditions, primary alcohols are oxidized to aldehydes followed by cross-dehydrogenative C–O coupling. These processes with aldehydes or primary alcohols as C-reagents giving esters are often referred to as oxidative esterification.
2.1 Transition metal salt-catalyzed reactions using compounds with C=C, C=O, and C–Hal bonds as oxidants
One of the types of the oxidative esterification is based on the hydrogen transfer catalyzed by transition metal complexes. In these reactions, compounds with a double bond or a C–Hal bond act as oxidants (hydrogen acceptors). Examples of these reactions with aldehydes or primary alcohols as C–H reagents are given in Table 2.
Table 2: Transition metal salt-catalyzed oxidative coupling of primary alcohols or aldehydes with alcohols using compounds with C=C, C=O, and C–Hal bonds as oxidants.
|
|
|||||
| CH-reagent | R2 (amount of R2OH) | Catalyst | Oxidant | Conditions; yields | Ref. |
|---|---|---|---|---|---|
| R1CH=O, R1CH2OH |
Me
(excess) |
RuH2(CO)(PPh3)3/4,5-bis(diphenylphosphino)-9,9-dimethylxanthene |
|
MeOH/PhMe (1:1), 4–48 h, 110 °C; 74–95% | [90,91] |
| R1CH2OH |
Me
(excess) |
[CpIrCl2]2/2-methylaminoethanol/
Cs2CO3 |
|
acetone, 24 h, room temperature; 23–92% | [92] |
| R1CH=O |
Me, Et, iPr,
s-Bu, CH2CF3 (excess) |
Pd(OAc)2/2-dicyclohexylphosphino-2',4',6'-triisopropylbiphenyl/K2CO3 |
|
acetone/R2OH, 2–24 h, room temperature–50 °C; 7–99% | [93] |
| R1CH=O |
Et
(excess) |
Pd(PPh3)4/K2CO3 | BnCl or BnBr | EtOH, MW, 30 min, 90 °C; 65–93% | [94] |
| R1CH=O | n-alkyl, sec-alkyl, Bn, etc. (1 equiv) | PdCl2(PPh3)2/K2CO3 | BnCl | THF, 20 h, 50 °C; 72–99% | [95] |
Crotononitrile, acetone, and benzyl halides were used as oxidants. Ruthenium, iridium, and palladium complexes acted as catalysts. In most cases, structurally simple alcohols, which are taken in a large excess relative to the CH-reagent, served as OH-reagents. The exception is a study [95], in which the coupling was accomplished using an equivalent amount of alcohol.
2.2 Oxidative systems based on noble metals and oxygen
The oxidative coupling of benzyl alcohols with aliphatic alcohols in the presence of Pd(II) salt/Ag(I) salt/base/oxygen system was proposed [96,97]. In the study [97], phosphine ligands were additionally employed. It is suggested that the coupling occurs through oxidation of benzyl alcohol to benzaldehyde. Alcohols (OH-reagents) are taken in a twofold molar excess [96] or are used as solvents [96,97]; the reaction time is 20–40 h at 45–80 °C. The aerobic oxidative coupling of aldehydes or primary alcohols as CH-reagents with low-molecular-weight alcohols was performed in the presence of heterogeneous catalysts, such as Au/TiO2 [98-100], Au/β-Ga2O3 [101], Au/polymer [102], and AuNiOx/SiO2-Al2O3-MgO [103]. In all the above-mentioned processes in the presence of heterogeneous catalysts, low-molecular-weight alcohols are used as the solvents or are taken in a large excess relative to the CH-reagent.
2.3 Reactions catalyzed by N-heterocyclic carbenes
N-heterocyclic nucleophilic carbenes 105 have found use for the oxidative esterification. Scheme 23 shows, in a simplified way, the proposed mechanism of this type of cross-dehydrogenative C–O coupling [104-113]. The aldehyde is subjected to the attack of N-heterocyclic nucleophilic carbene 105, which can be generated from the corresponding azolium salt 104. The resulting intermediate 106 is oxidized to 107 followed by the nucleophilic attack by the alcohol to form ester 108.
Scheme 23: Possible mechanism of oxidative esterification catalyzed by N-heterocyclic nucleophilic carbene.
Scheme 23: Possible mechanism of oxidative esterification catalyzed by N-heterocyclic nucleophilic carbene.
The mechanism of the aerobic oxidative coupling of benzaldehyde with methanol in the presence of the 4-ethyl-1-methyl-1H-1,2,4-triazolium iodide/DBU system was studied in detail [114]. It was shown that the reaction proceeds via another mechanism, involving the formation of 2-hydroxy-1,2-diphenylethanone from benzaldehyde followed by the oxidation of this intermediate to 1,2-diphenylethanedione.
Scheme 24 shows an example of the oxidative coupling of aldehydes and alcohols, in which CH- and OH-reagents are taken in equivalent amounts [104]. Thiazolium salt 109 combined with triethylamine acted as the catalyst, and azobenzene 110 served as the oxidizing agent. Esters 111 were prepared in 16–97% yield.
Scheme 24: Oxidative esterification employing stoichiometric amounts of aldehydes and alcohols.
Scheme 24: Oxidative esterification employing stoichiometric amounts of aldehydes and alcohols.
The selective oxidative coupling of aldehydes 112 and alcohols 113 in the presence of amines 114 using 1,4-dimethyltriazolium iodide (115), DBU and quinone 116 afforded esters 117. This method was also applied to the synthesis of esters 117a–c from amino alcohols (Scheme 25) [115,116].
Scheme 25: Selective oxidative coupling of aldehydes with alcohols in the presence of amines.
Scheme 25: Selective oxidative coupling of aldehydes with alcohols in the presence of amines.
Different conditions were proposed for the cross-dehydrogenative C–O coupling of aldehydes with alcohols and phenols catalyzed by N-heterocyclic carbenes (118, 129) or their precursors, azolium salts (119–128), combined with bases (Table 3; the publications are summarized in chronological order). In most cases, alcohols were taken in an excess.
Table 3: Oxidative esterification of aldehydes with alcohols.
|
|
||||
| R1 | R2 | Oxidative system | Conditions; yield | Ref. |
|---|---|---|---|---|
| Ar | n-alkyl, iPr |
118 (1 mol per mol of aldehyde), generated in situ |
R2OH, bp; 17–75% | [117] |
| n-alkyl, sec-alkyl, t-Bu, etc. | Me, n-Pr, cyclohexyl, CH2CH2SiMe3, CH2CCl3, CH(Me)COOMe |
119 (10 mol %) DBU (1.1 mol per mol of aldehyde) MnO2 (5 mol per mol of aldehyde) |
R2OH (5 mol per mol of aldehyde)
CH2Cl2, room temperature; 56–99% |
[105,106] |
| Ar | Me, iPr, Ph, etc. |
120 Cs2CO3 (1.5 equiv), air |
R2OH (3 equiv)
cyclohexane, 25 °C, 10 h; 34–80% |
[118] |
| Ar, PhCH=CH, cyclohexyl | Ar’ |
121 (5 mol %) Pd(OAc)2 (5 mol %) Na2CO3 (4 equiv) air |
aldehyde/phenol (3:2)
xylene, 100 °C, 24–48 h; 25–99% |
[119] |
| Ar, PhCH=CH, cyclohexyl | Ar’ |
121 (20 mol %)
Fe(OTf)2(20 mol %) t-BuOK (1 equiv) air |
aldehyde/phenol (1:1)
dioxane, 90 °C, 24 h; 15–89% |
[107] |
| Ar, PhCH=CH, cyclohexyl | Me, Et, Bn, 2-PhEt, s-Bu, allyl, propargyl, 4-pentinyl, etc. |
122 (10 mol %) DBU (1 equiv) electrochemical oxidation, Bu4NBr (30 mol %) |
aldehyde/alcohol (1:1.1)
MeCN, room temperature; 60–97% |
[108] |
| Ar | Me, Et, Pr, iPr |
123 (3 mol per mol of aldehyde) |
aldehyde/alcohol (1:3)
50–60 °C; 50–90% |
[120] |
| PhCH2CH2 | Bn, allyl |
124 (5 mol %) NHEt2 (1.1 equiv) Et3N (1.2 equiv) N-chlorosuccinimide (1 equiv) |
aldehyde/alcohol (1:2)
CH2Cl2, room temperature; 83–87% |
[121] |
| Ar | Me, Et, iPr, allyl, propargyl, Bn |
125 (10 mol %) DBU (20 mol %) oxygen |
aldehyde/alcohol (1:1.2)
THF, 25 °C; 63–82% |
[109] |
| Ar, PhCH=CH, cyclohexyl | Me, Et, n-Pr, Bn, allyl, propargyl |
126 (2.5 mol %) [Ru(2,2’-bipyrazine)3](PF6)2 (5 mol %), air |
aldehyde/alcohol (1:10)
MeCN, room temperature; 15–81% |
[110] |
| Ar, PhCH2CH2 | Me, Bn, allyl, CH2CCl3 |
127 (15 mol %) DBU (110 mol %) air |
THF/ROH (1:1) or 3 equiv ROH in THF, room temperature;
15–94% |
[114,122] |
| Ar, n-alkyl | n-alkyl, Bn, CH2CH2NEt2 |
128 (solvent) DBU (1 equiv), Cs2CO3 (3 equiv), MnO2 (3 equiv) |
aldehyde:alcohol (1:3)
25 °C, 24 h; 25–91% |
[111] |
| Ar, CH=CHPh, CH=CH(2-C6H4OMe) | Bn, CH=CHPh, Et, Ph, (CH2)3Ph |
129 (5 mol %) TEMPO (2 equiv) |
aldehyde:alcohol (1:1.5)
toluene, 100 °C, 4–6 h 60–86% |
[112] |
The oxidative esterification in the presence of azolium salts 119, 121, and 125 was performed with primary alcohols instead of aldehydes (Table 4). It is supposed that under the reaction conditions primary alcohol is initially oxidized to aldehyde without the participation of N-heterocyclic carbene [106,113,123,124].
Table 4: Oxidative esterification using primary alcohols as C-reagents.
|
|
||||
| R1 | R2 | Oxidative system | Conditions; yield | Ref. |
|---|---|---|---|---|
| substituted vinyl, alkynyl, Ar, Bn, etc. | Me, Bu, iPr, CH2CCl3, CH2CH2OMe, CH2CH2OTMS, etc. |
125 (10–50 mol %), DBU (10–50 mol %), MnO2 (15 equiv) |
R2OH or R2OH (5 equiv) in toluene, 23 °C; 65–95% | [113] |
| RCH=CH, PhCH=CMe, PhC≡C, Ar | Me, n-Bu, iPr |
119 (10 mol %), DBU (10–110 mol %), MnO2 (15 mol per mol of R1OH) |
R2OH or R2OH (3–5 equiv) in toluene, 23 °C; 73–95% | [106] |
| ArCH2, allyl | Ar |
121 (10 mol %), Pd(OAc)2 (5 mol %), Na2CO3 (0.5 mol per mol of phenol), oxygen |
R1OH/R2OH (3:2)
xylene, 130 °C, 36 h; 35–95% |
[123] |
| ArCH2, allyl, Bu | Ar | 121 (10 mol %), [RuCl2(p-cymene)]2 (5 mol %), Cs2CO3 (10 mol %), oxygen |
R1OH/R2OH (3:2)
xylene, 130 °C, 24 h; 50–95% |
[124] |
The oxidative esterification of aldehydes with racemic mixtures of alcohols catalyzed by chiral N-heterocyclic carbenes was used for the kinetic separation of enantiomers of alcohols [125,126].
2.4 Reactions using halogen-containing oxidative systems
This section considers cross-dehydrogenative C–O coupling reactions with aldehydes and primary alcohols as C-reagents in which halogens and their compounds, for example, molecular iodine, the Bu4NI/t-BuOOH system, organic iodine(III or V) compounds, bromides combined with oxidants, hypochlorite, and so on, acted as oxidants.
The oxidative coupling of primary alcohols 130 with methanol or trifluoroethanol and the oxidative coupling of aldehydes 132 with structurally diverse alcohols 133 were performed using molecular iodine in the presence of potassium carbonate (Scheme 26) [127]. In the former case, methanol or trifluoroethanol served as the solvent. In the latter case, the reaction was carried out in tert-butanol; the amount alcohol was nearly equivalent relative to aldehyde. It is suggested that the reaction proceeds through the formation of hemiacetal from alcohol and aldehyde, which is oxidized by iodine to ester 131 or 134. In the coupling of two alcohols, one of them is initially oxidized to aldehyde by iodine.
Scheme 26: Iodine mediated oxidative esterification.
Scheme 26: Iodine mediated oxidative esterification.
In the oxidative coupling of aldehydes or primary alcohols, the second reagent (alcohol) usually served as the solvent. The oxidation was performed with halogen-containing reactants: I2/KOH [128], KIcat/t-BuOOH [129], I2cat/PhI(OAc)2 [130], I2/NaNO2 [131], NaBr/PhI(OAc)2 [132], LiBr/NaIO4/H2SO4 [133], Bu4NBr/NaOCl [134], NaOCl/AcOH [135], Py·HBr3 [136], N-bromosuccinimide/pyridine [137], N-iodosuccinimide/K2CO3 [138], and N,N'-diiodo-N,N'-1,2-ethanediylbis(p-toluenesulfonamide) [139].
The oxidative C–O coupling of benzyl alcohols 135 with alkylarenes 136 took place under the action of the Bu4NI/t-BuOOH system in the presence of NaH2PO4 [140]. It was proposed that under the reaction conditions benzyl alcohol 135 is oxidized to carboxylic acid 138, while alkylarene gives iodide 139; the nucleophilic substitution between the carboxylate anion and benzyl iodide affords coupling product 137 [140] (Scheme 27).
Scheme 27: Oxidative C–O coupling of benzyl alcohols with methylarenes under the action of Bu4NI/t-BuOOH system in the presence of NaH2PO4.
Scheme 27: Oxidative C–O coupling of benzyl alcohols with methylarenes under the action of Bu4NI/t-BuOOH syste...
This mechanism differs from that proposed in the study [141]. According to the latter mechanism, the benzylic carbocation rather than benzyl iodide is generated and this carbocation undergoes nucleophilic attack by carboxylic acid. This mechanism is confirmed by the fact that iodide is inert under the reaction conditions. The conditions of the oxidative coupling used in the study [140] differ from the conditions described in the work [141]. However, it should be noted that the formation of benzyl iodide was not experimentally confirmed in the study [140], and the presence of this species was proposed based on the literature data.
The coupling of methyl- and ethylarenes 141 with aromatic aldehydes 140 was performed with the Bu4NI/t-BuOOH system in the presence of an excess of either alkylarene 141 or aldehyde 140 [142]. It was supposed that the coupling occurs through the generation of tert-butoxyl radicals, which abstract a hydrogen atom from the benzylic position of the C-reagent to form the C-radical, which is oxidized to the carbocation; in turn, the aldehyde is oxidized to acid, which reacts with the carbocation to give the target coupling product 142 (Scheme 28).
Scheme 28: Oxidative coupling of methyl- and ethylarenes with aromatic aldehydes under the action of Bu4NI/t-BuOOH system.
Scheme 28: Oxidative coupling of methyl- and ethylarenes with aromatic aldehydes under the action of Bu4NI/t-B...
It was shown that under the reaction conditions, tert-butyl perester is generated as an important intermediate from aldehyde and t-BuOOH, and this intermediate can serve as the source of acid and tert-butoxyl radicals [142].
tert-Butyl peresters 144 were synthesized on a preparative scale by the cross-dehydrogenative C–O coupling of aldehydes 143 with t-BuOOH in the presence of Bu4NI [143]. It is proposed that tert-butyl peresters 144 are produced as a result of the recombination of acyl radicals 145 and tert-butyl peroxide radicals. The radical reaction mechanism was confirmed by the experiment, in which acyl radicals generated from aldehyde 146 were trapped by the stable radical TEMPO, and trapping product 147 was obtained in an almost quantitative yield (Scheme 29).
Scheme 29: Cross-dehydrogenative C–O coupling of aldehydes with t-BuOOH in the presence of Bu4NI.
Scheme 29: Cross-dehydrogenative C–O coupling of aldehydes with t-BuOOH in the presence of Bu4NI.
The reaction of aldehydes 148a with ethers 149 in the presence of Bu4NI and t-BuOOH generated corresponding α-acyloxy ethers 150. Reactions between (hetero)aromatic aldehydes or cyclohexanecarbaldehyde 148b with arylalkyl ketones 151 under similar conditions resulted in α-acyloxy ketones 152 [144] (Scheme 30). The plausible mechanism includes the formation of tert-butyl peresters from the aldehydes.
Scheme 30: Bu4NI-catalyzed α-acyloxylation reaction of ethers and ketones with aldehydes and t-BuOOH.
Scheme 30: Bu4NI-catalyzed α-acyloxylation reaction of ethers and ketones with aldehydes and t-BuOOH.
N-Hydroxyimides 154 are efficient OH-reagents in the coupling with aldehydes and primary alcohols. Their oxidative coupling products, so-called activated esters 155, readily react with nucleophiles, alcohols or amines, due to which they are employed for the preparation of esters and amides. This type of coupling was accomplished using iodine-containing oxidants. The proposed reaction mechanism involves the nucleophilic addition of N-hydroxyimides to aldehydes followed by the oxidation of the resulting adduct to form the activated ester. For example, the C–O coupling of aldehydes 153 with N-hydroxyimides 154 was carried out in the presence of the Bu4NHal/t-BuOOH system (Hal = I or Br) [145]. This method is applicable to the oxidative coupling of aldehydes 156 with hexafluoroisopropanol giving esters 157. One of the coupling components is added in a twofold excess (Scheme 31).
Scheme 31: Oxidative coupling of aldehydes with N-hydroxyimides and hexafluoroisopropanol.
Scheme 31: Oxidative coupling of aldehydes with N-hydroxyimides and hexafluoroisopropanol.
A similar oxidative coupling reaction was performed with primary alcohols 158 as CH-reagents and N-hydroxyimides 154a,b [146]. The coupling reaction with N-hydroxyphthalimide (154b) was carried out using the NaI/aqueous t-BuOOH/KOH system instead of Bu4NI/t-BuOOH in decane [146]. The resulting activated esters 159 and 160 were isolated or used in the one-pot reaction with amines to prepare amides (Scheme 32).
Scheme 32: Oxidative coupling of alcohols with N-hydroxyimides.
Scheme 32: Oxidative coupling of alcohols with N-hydroxyimides.
The oxidative C–O coupling of alcohols and aldehydes 161–164 with N-hydroxysuccinimide (154a) was performed in the presence of (diacetoxyiodo)benzene [147] or iodoxybenzoic acid (IBX) [148] as the oxidant. The authors hypothesized that the reaction proceeds via the nucleophilic addition of N-hydroxysuccinimide (154a) to aldehyde followed by the oxidation of the adduct that formed with iodine(III) [147] or iodine(V) [148] compounds to form products 165, 166 (Scheme 33).
Scheme 33: Oxidative coupling of aldehydes and primary alcohols with N-hydroxyimides using (diacetoxyiodo)benzene or iodoxybenzoic acid (IBX) as oxidants.
Scheme 33: Oxidative coupling of aldehydes and primary alcohols with N-hydroxyimides using (diacetoxyiodo)benz...
In the study [149], activated esters were synthesized by the oxidative coupling of aldehydes and N-hydroxysuccinimide in the presence of (diacetoxyiodo)benzene, and these compounds were subjected, without isolation, in the reaction with amines to prepare amides. The reaction was performed at room temperature with N-hydroxysuccinimide (154a) taken in an equivalent amount or a small excess relative to aldehyde. Iodoxybenzoic acid (IBX) or the Co(OAc)2·4H2O/O2 system proved to be less efficient in the coupling reactions compared with (diacetoxyiodo)benzene. According to the proposed radical mechanism, N-hydroxysuccinimide (154a) adds to aldehyde to form intermediate 167 followed by the hydrogen atom abstraction from 167 by succinimide-N-oxyl radical 168 to give radical 169 and the oxidation of the latter resulted in the formation of coupling product 170 (Scheme 34). Radical intermediates 168 and 169 were detected by ESR spectroscopy [149].
Scheme 34: Proposed mechanism of the oxidative coupling of aldehydes and N-hydroxysuccinimide under action of (diacetoxyiodo)benzene.
Scheme 34: Proposed mechanism of the oxidative coupling of aldehydes and N-hydroxysuccinimide under action of ...
The oxidative coupling of aldehydes 171 with pivalic acid (172) was performed using the TEMPOcat/t-BuOCl system; the coupling products, unsymmetrical anhydrides 173, were employed in the synthesis of esters and amides 174 (Scheme 35) [150].
Scheme 35: Oxidative coupling of aldehydes with pivalic acid (172).
Scheme 35: Oxidative coupling of aldehydes with pivalic acid (172).
It is suggested that pivalic acid 172 adds to aldehyde 171 followed by the oxidation of the intermediate that formed with the oxoammonium salt, which is generated from TEMPO and t-BuOCl, to yield anhydride 173.
2.5 Oxidative systems based on transition metal salts and peroxides
Esters 177 were synthesized by the oxidative C–O coupling of aldehydes 175 with alkylarenes 176 using the Cu(OAc)2/t-BuOOH system (Scheme 36) [151]. The coupling was performed with toluene, xylenes, 1,3,5-trimethylbenzene, 2,4-dichlorotoluene, and ethylbenzene.
Scheme 36: Oxidative C–O coupling of aldehydes with alkylarenes using the Cu(OAc)2/t-BuOOH system.
Scheme 36: Oxidative C–O coupling of aldehydes with alkylarenes using the Cu(OAc)2/t-BuOOH system.
α-Acyloxy ethers 180 were synthesized by the oxidative coupling of benzyl alcohols 178 with ethers 179 (dioxane, tetrahydropyran, tetrahydrofuran, 1,2-dimethoxyethane) using Cu(OAc)2/t-BuOOH system [152] (Scheme 37).
Scheme 37: Copper-catalyzed acyloxylation of C(sp3)-H bond adjacent to oxygen in ethers using benzyl alcohols.
Scheme 37: Copper-catalyzed acyloxylation of C(sp3)-H bond adjacent to oxygen in ethers using benzyl alcohols.
In a series of works, the oxidative coupling of alcohols, aldehydes, or formamides with 1,3-dicarbonyl compounds or phenols was accomplished in the presence of tert-butyl hydroperoxide and copper salts (Table 5). In most cases, the range of phenols applicable to the coupling is limited to 2-acylphenols. However, 2-(benzothiazol-2-yl)phenol was used along with 2-acylphenols as the OH-reagent in the study [153].
Table 5: Oxidative coupling of alcohols, aldehydes, or formamides with 1,3-dicarbonyl compounds or phenols in the presence of tert-butyl hydroperoxide and copper salts.
|
|
|||
| C-reagent | O-reagent | Molar ratio C-reagent/O-reagent; conditions; yields | Ref. |
|---|---|---|---|
|
R1 = Ar, n-pentyl, cyclohexyl, diethylmethyl |
R2 = Me, Et, CH2Cl, Ph R3 = Me, Et, OMe, OEt |
1:1.1;
CuBr (2.5 mol %), t-BuOOH (5.5 M in decane, 1.5 equiv), 80 °C, 16 h; 57–89% |
[154] |
|
R12 = di-Me, di-Et, di-iPr, -(CH2)5- |
R2, R4 = alkyl, Ph, -(CH2)4-; R4 may be H; R3 = alkoxy, BnO R5 = Me, OMe, Ph, NHPh R6 = H, OMe, Br |
Formamide as the solvent;
CuBr2 or Cu(OAc)2 (5 mol %), t-BuOOH (70% aq, 1.5 equiv), 80 °C, 3 h; 62–86% |
[155] |
|
R12 = di-Me, di-Et, di-iPr, -(CH2)5- |
R2, R4 = alkyl, Ph, -(CH2)4-; R4 may be H; R3 = alkoxy, BnO R5 = Me, OMe, Ph, NHPh R6 = H, OMe, Hal |
Formamide as the solvent;
CuCl (1 mol %), t-BuOOH (70% aq, 6 equiv), 70 °C, 15–30 min; 61–99% |
[156] |
|
R1 = Ar, n-alkyl, cyclohexyl, CH2CH2Ph, |
R4 = Me, OMe, OEt, OBn, Ph |
2:1;
Cu(OAc)2 (5 mol %), t-BuOOH (70% aq, 4 equiv), DMSO, 80 °C, 20 h; 35–88% |
[153] |
|
R12 = di-Me, di-Et, di-iPr, -(CH2)5-, -(CH2)2O(CH2)2- |
R5 = H, Me, t-Bu, OMe, NEt2, Cl, Br, etc. |
Formamide as the solvent;
CuCl (1–2 mol %), t-BuOOH (70% aq, 6 equiv), 80 °C, 15–90 min; 26–99% |
[157] |
|
R12 = di-Me, di-Et |
R2 = Me, Ph, n-Pr, etc. R3 = Me, OMe, OEt, Bn, allyl, etc. |
Formamide as the solvent;
CuO/α-Fe2O3/carbon nanotubes, t-BuOOH (70% aq, 1.5 equiv), 80 °C, 4 h; 40–80% |
[158] |
|
|
|
1:1;
CuO/α-Fe2O3/carbon nanotubes, t-BuOOH (70% aq, 1.5 equiv), DMSO, 80 °C, 10 h; 20–82% |
[158] |
In the coupling with aldehydes, the components are taken in a nearly stoichiometric molar ratio. In the coupling with formamides, the latter served as the solvent. In the studies [155,156], 1,3-cyclohexanedione was used as the OH-reagent along with 1,3-ketoesters. However, the reaction with 1,3-cyclohexanedione gave products in low yields (19–26%). The coupling of a number of substituted salicylaldehydes with formamides was performed; the aldehyde group, which is prone to oxidation, remaining intact [157].
The coupling of aldehydes and formamides with 2-substituted phenols was carried out in the presence of heterogeneous catalysts, such as CuO on α-Fe2O3-modified carbon nanotubes (a magnetically separable catalyst) [158] and the metal-organic framework Cu2(4,4’-biphenyldicarboxylate)2(4,4’-bipyridine) [159].
Aldehydes were oxidized to esters in alcohols with V2O5/H2O2aq/HClO4 [160], V2O5/percarbonate, sodium perborate/HClO4 [161], VO(acac)2/H2O2aq [162], Cu(ClO4)2/t-BuOOH in decane/InBr3 [163], Fe(ClO4)3/H2O2aq [164], γ-Fe2O3-SiO2-supported heteropoly acids combined with H2O2aq [165], silica gel-immobilized manganese phthalocyanine/H2O2aq [166], a Ni(II) complex/H2O2aq [167], and ZnBr2/H2O2 [168].
The oxidative C–O coupling of aromatic aldehydes 181 with cycloalkanes 182 was accomplished in the presence of the Cu(OAc)2/t-BuOOH system to prepare products 183. This reaction is unusual in that it involves the cleavage of four C–H bonds, including unactivated C–H bonds of cycloalkane, and the formation of two C–O bonds and one C=C double bond [169]. The yields of products 183 were not higher than 53%. However, the transformation is a rare example of the oxidative coupling with the participation of inert CH-reagents, cycloalkanes, to form a product, which can be subjected to higher oxidation under the conditions of oxidative coupling (Scheme 38).
Scheme 38: Oxidative C–O coupling of aromatic aldehydes with cycloalkanes.
Scheme 38: Oxidative C–O coupling of aromatic aldehydes with cycloalkanes.
Related oxidative C–O coupling reactions of alkanes with methylarenes and carboxylic acids are discussed in sections 4.1 and 6, respectively. Different radical mechanisms were proposed, the common feature is the generation of alkene via the radical dehydrogenation of alkane.
2.6 Other oxidative systems
Esters were synthesized from aldehydes and methanol (6 equiv excess relative to aldehyde) using pyridinium dichromate in DMF [170]. Methyl esters were synthesized also from aromatic aldehydes, α,β-unsaturated aldehydes, or allylic alcohols in the presence of the DDQ (2,3-dichloro-5,6-dicyanobenzoquinone)/amberlyst-15 system in a methanol/toluene mixture under microwave irradiation [171]. Methyl, ethyl, and isopropyl benzoates were prepared from benzaldehyde under irradiation of its alcoholic solutions with a mercury lamp in an oxygen atmosphere; esters were obtained in higher yields in the presence of catalytic amounts of HCl [172]. The formation of primary and secondary alcohol esters was observed after the ozonolysis of a mixture of aldehyde and alcohol in a basic medium [173]. The oxidative esterification of aldehydes was performed using peroxides in the presence of Lewis acids or in the absence of the latter; for example, using oxone in the presence of In(OTf)3 [174,175], oxone [174,176], Caro’s acid [177], H2O2aq (30%) in the presence of CaCl2 or MgCl2 [178], H2O2aq (50%) [179].
The oxidative coupling of structurally diverse aldehydes (aromatic, α,β-unsaturated, etc.) with hexafluoroisopropanol was carried out in the presence of the oxoammonium salt (4-acetylamino-2,2,6,6-tetramethylpiperidine-1-oxoammonium tetrafluoroborate) and pyridine [180]. The drawback of this method is that it requires a rather complicated and expensive oxidant.
An efficient aerobic cross-dehydrogenative coupling of alcohols and α-carbonyl aldehydes was achieved employing a CuBr/pyridine catalytic system in toluene, only 1.5-fold excess of alcohols over aldehydes was used [181]. The reaction time is 18 h at 90 °C, the yields of α-ketoesters vary from 42 to 88%.
The unusual C–O cross-coupling of primary alcohols 184 with secondary alcohols 185 in the absence of oxidants was performed in the presence of ruthenium complex 186 as the catalyst; the reaction afforded molecular hydrogen and unsymmetrical ester 187 (Scheme 39) [182].
Scheme 39: Ruthenium catalyzed cross-dehydrogenative coupling of primary and secondary alcohols.
Scheme 39: Ruthenium catalyzed cross-dehydrogenative coupling of primary and secondary alcohols.
Cross-coupling products 187 were obtained in high yields; the expected homocoupling of primary alcohols giving symmetrical esters and the dehydrogenation of secondary alcohols yielding ketones were avoided.
3 Ketones and 1,3-dicarbonyl compounds as C-reagents in cross-dehydrogenative C–O coupling
Most of cross-dehydrogenative C–O coupling reactions involving the α-position of carbonyl compounds (acetoxylation, alkoxylation, sulfonyloxylation) are based on the use of iodine-containing oxidizing agents. Transition metal salts, such as copper and manganese salts, were less often employed for this purpose.
3.1 Oxidative systems based on iodine compounds
Iodine(III) organic compounds, including those generated in situ from aryl iodides and peracids (for example, m-chloroperbenzoic acid (MCPBA) and peracetic acid) are most commonly employed in the oxidative coupling of OH-reagents with carbonyl compounds. Methods were developed for the sulfonyloxylation of ketones, in which iodoarene is generated in situ by the iodination of arene with molecular iodine [183] or NH4I [184] in the presence of m-chloroperbenzoic acid.
In the presence of p-(difluoroiodo)toluene (188), β-dicarbonyl compounds 189 undergo oxidative coupling with various OH-reagents (Scheme 40), such as sulfonic acids (coupling products 190), acetic acid (products 191), diphenyl phosphate (products 192), and alcohols (products 193) [185]. The reactions with alcohols are most slow. It is suggested that the oxidative C–O coupling at the α-position of carbonyl (or β-dicarbonyl) compounds and various OH-reagents in the presence of iodine(III) compounds proceeds through an ionic mechanism. Thus, the electrophilic iodine atom attacks the enol of carbonyl compound 194 followed by the replacement of the iodine-containing moiety in intermediate 195 by the O-nucleophile to form C–O coupling product 196 (Scheme 40).
Scheme 40: Cross-dehydrogenative C–O coupling reactions of β-dicarbonyl compounds with sulfonic acids, acetic acid, diphenyl phosphate, and alcohols using p-(difluoroiodo)toluene.
Scheme 40: Cross-dehydrogenative C–O coupling reactions of β-dicarbonyl compounds with sulfonic acids, acetic ...
Table 6 presents other examples of C–O coupling reactions at the α-position of carbonyl compounds based on the oxidation with iodine(III) compounds or iodoarenes in the presence of peroxides. Chiral iodoarenes, such as 197, served as the catalysts in the asymmetric C–O coupling of sulfonic acids with ketones. The enantiomeric excess of the product was not higher than 58% due, in particular, to instability of the configuration of the products, α-sulfonyloxy ketones, under the reaction conditions, resulting in the partial racemization [186].
Table 6: C–O coupling reactions at the α-position of carbonyl compounds mediated by iodine(III) compounds or iodoarenes in the presence of peroxides.
|
|
||||
| C-reagent | O-reagent | Oxidative system | Conditions; yields | Ref. |
|---|---|---|---|---|
| β-diketones, β-keto esters | MeSO3H (1 equiv) | PhIO |
CHCl3, reflux, 2 h;
37–83% |
[187] |
| β-diketones, β-keto esters | MeOH or EtOH (as the solvents) | PhIO, BF3·Et2O |
room temperature, 5 h;
59–67% |
[187] |
|
|
ROH, R = iBu, CMe2Et, (CH2)2CF3, (CH2)3OBn, etc. | PhIO, BF3·Et2O |
CHCl3,
room temperature, 5 h; yields were not reported |
[188] |
|
|
acetoxylation with PhI(OAc)2 (1.2 equiv) | PhI(OAc)2, Bu4NBr, KOH | dioxane, room temperature, 1 h; 74–87% | [189] |
|
R1 = Ar, alkyl R2 = H, alkyl, COOMe |
RSO3H (1.1–5 equiv) | PhI or poly(4-iodostyrene) (cat), MCPBA; KBrcat or TEMPOcat was added to oxidize alcohols | MeCN or CHCl3, 50 °C, 5 h; 25–81% | [190] |
|
R1 = Ar, alkyl, R2 = H, alkyl, CO2Et |
p-TsOH (1.1–5 equiv.) | polymer-immobilized iodobenzene, MCPBA (1.1–2.5 equiv), p-TsOH | 50 °C, 9–16 h; 51–88% | [191] |
|
R1 = Ar, Et, t-Bu, etc. R2 = H, alkyl, etc. |
acetoxylation
with Ac2O |
PhI, 30% aq H2O2, Ac2O, BF3·Et2O | 30 °C, 7 h; 32–86% | [192] |
|
R1 = Ar, alkyl, R2 = H, alkyl |
p-TsOH (3–5 equiv) | p-MeC6H4I (1 equiv), oxone (1.5 equiv), |
MeCN, 60 °C;
32–100% |
[193] |
|
R1 = Ar, alkyl, R2 = H, alkyl |
RSO3H (1.5 equiv), R = Ar, alkyl, etc. |
197 (0.1 equiv) MCPBA (1.5 equiv) |
EtOAc, room temperature;
8–41% |
[186] |
Good results were achieved in the oxidative C–O coupling of ketones, aldehydes and β-dicarbonyl compounds 198 with carboxylic acids 199 in the presence of the Bu4NI/t-BuOOH system [194] (Scheme 41). The coupling can be accomplished with a wide range of substrates and gives products 200 in high yields, the C-component and the O-component being taken in a ratio of 1:1. The reactions of aldehydes are similar to the reactions of ketones, the aldehyde moiety remaining unchanged. The authors hypothesized that the reaction proceeds through a radical mechanism [194].
Scheme 41: Acyloxylation of ketones, aldehydes and β-dicarbonyl compounds using carboxylic acids and Bu4NI/t-BuOOH system.
Scheme 41: Acyloxylation of ketones, aldehydes and β-dicarbonyl compounds using carboxylic acids and Bu4NI/t-B...
The Bu4NI/t-BuOOH system was employed also in the oxidative coupling of carboxylic acids with β-keto esters [195].
The cross-dehydrogenative C–O coupling of alcohols 201 and ketones 202 in the presence of the Bu4NI/t-BuOOH system was accomplished to prepare α-acyloxy ketones 203 [196]. Coupling products 203 were synthesized mainly from benzyl alcohols and propiophenone (Scheme 42).
Scheme 42: Acyloxylation of ketones using Bu4NI/t-BuOOH system.
Scheme 42: Acyloxylation of ketones using Bu4NI/t-BuOOH system.
The authors proposed two radical reaction pathways, including the formation of C-radicals from ketones. One of these pathways involves the formation of tert-butyl peresters from alcohols. This pathway is confirmed by the fact that, in the presence of Bu4NI, tert-butyl per(1-naphthylate) gives products 203 in the reaction with propiophenone.
3.2 Oxidative systems based on transition metal compounds
In addition to iodine compounds, transition metal salts, such as copper and manganese salts, were used for the oxidative functionalization at the α-position of carbonyl compounds.
N-Hydroxyimides and N-hydroxyamides 204 are involved in the oxidative C–O coupling with 1,3-dicarbonyl compounds and their hetero analogues, such as 2-substituted malononitriles and cyanoacetic esters, 205 in the presence of oxidants based on manganese, cobalt, and cerium [197]. The best results were achieved with the use of Mn(OAc)3 and the Co(OAc)2(cat)/KMnO4 system (Scheme 43). The yields of products 206 were as high as 94%. It is supposed that the oxidant serves two functions: the generation of N-oxyl radicals 207 from N-hydroxyimides or N-hydroxyamides 204 and the one-electron oxidation of 1,3-dicarbonyl compounds via the formation of complex 208. Apparently, the oxidative coupling of 1,3-dicarbonyl compounds 209 with oximes 210 occurs via a similar mechanism [198]. The reaction takes place in the presence of various oxidants, such as KMnO4, Mn(OAc)2/KMnO4, Mn(OAc)3, MnO2, Mn(acac)3, Fe(ClO4)3, Cu(ClO4)2, Cu(NO3)2, and (NH4)2Ce(NO3)6. Twenty coupling products 211 were prepared in yields of 27–92% using KMnO4, Mn(OAc)3·2H2O, or the Mn(OAc)2/KMnO4 system. The syntheses do not require excess amounts of reagents. The formation of N-oxyl radicals 212 and 207 from oximes and N-hydroxyimides was confirmed by ESR spectroscopy [197,198].
Scheme 43: Cross-dehydrogenative C–O coupling of β-dicarbonyl compounds and their heteroanalogues with N-hydroxyimides, N-hydroxyamides, and oximes.
Scheme 43: Cross-dehydrogenative C–O coupling of β-dicarbonyl compounds and their heteroanalogues with N-hydro...
The oxidative coupling of 1,3-dicarbonyl compounds [199] and their hetero analogues [200] 213 with tert-butyl hydroperoxides catalyzed by transition metal salts (Cu, Fe, Co, Mn) was achieved. tert-Butyl hydroperoxide acts both as the oxidizing agent and the O-component in the coupling. The best results were obtained in the reaction catalyzed by Cu(ClO4)2·6H2O. It was hypothesized that copper serves for the formation of the reactive complex with 1,3-dicarbonyl compounds or their hetero analogues, as well as for the generation of tert-butyl peroxide radicals, which react with this complex to give coupling products 214 (Scheme 44).
Scheme 44: Cross-dehydrogenative C–O coupling of β-dicarbonyl compounds and their heteroanalogues with t-BuOOH.
Scheme 44: Cross-dehydrogenative C–O coupling of β-dicarbonyl compounds and their heteroanalogues with t-BuOOH....
The related peroxidation reactions with hydroperoxides (t-BuOOH, PhMe2COOH) in the presence of transition metal salts (cobalt, manganese, or copper) were performed with cyclohexanone, 2-methylcyclohexanone, cyclohexene, 1-octene, cumene, xylene, dimethylaniline, and dioxane [201].
The enantioselective oxidative coupling of 2,6-dialkylphenyl-β-keto esters and thioesters 215 with tert-butyl hydroxycarbamate 216 was performed using the Cu(OTf)2/chiral ligand 217/MnO2 system (Scheme 45) [202]. Apparently, product 218 is generated via an ionic mechanism involving the generation of electrophilic nitrosocarbonyl intermediate 219 [202].
Scheme 45: Oxidative C–O coupling of 2,6-dialkylphenyl-β-keto esters and thioesters with tert-butyl hydroxycarbamate.
Scheme 45: Oxidative C–O coupling of 2,6-dialkylphenyl-β-keto esters and thioesters with tert-butyl hydroxycar...
The improved version of that oxidative coupling method is based on CuCl/Cu(OAc)2/ligand/air catalytic system; different β-keto esters and hydroxamic acid derivatives can be used [203].
The acetoxylation at the α’-position of α,β-unsaturated ketones with Mn(OAc)3 was studied in detail. It is suggested that manganese(III) acetate causes the generation of C-radicals from ketones followed by the acetoxylation of these radicals. In this reaction, Mn(OAc)3 [204-210] or acetic acid [209,210], which was used as the co-solvent, can serve as the source of the acetoxy group. The synthesis is usually performed in benzene. In some cases, the α’-phenylation-α’-acetoxylation was observed due, apparently, to the addition of the C-radical generated from ketone to benzene [204]. It was shown [209] that the α’-acetoxylation of enones occurs with good selectivity in other solvents, such as cyclohexane and acetonitrile, as well.
The acyloxylation of enones and aryl ketones 220 with carboxylic acids occurs in benzene in the presence of KMnO4 to form coupling products 221 (Scheme 46) [211]. Acids were present in a large excess relative to ketones.
Scheme 46: α’-Acyloxylation of α,β-unsaturated ketones using KMnO4.
Scheme 46: α’-Acyloxylation of α,β-unsaturated ketones using KMnO4.
The synthesis was successfully accomplished, in particular, with readily oxidizable formic acid; the corresponding formates (for example, 221b) were obtained in 61–85% yield [211]. This method was modified by using not carboxylic acids but their mixtures with the corresponding anhydrides and was applied to the α’-acyloxylation of α,β-unsaturated ketones with complex structures with the aim of preparing analogues of natural compounds [212].
4 Compounds with an allyl, propargyl, or benzyl group as C-reagents in cross-dehydrogenative C–O coupling
4.1 Palladium- and copper-based oxidative systems
Studies on the acyloxylation at the allylic position of alkenes with palladium(II) complexes started in the 1960s [213]. This type of reactions was considered in more detail in reviews on the palladium complex-catalyzed functionalization of allyl-containing compounds [20,21].
It is suggested that the reaction proceeds through the cleavage of the allylic C–H bond in 222 to form π-allyl–palladium complex 223 followed by the nucleophilic attack of acetate to give C–O coupling product 224a (Scheme 47) [214-217]. The alternative mechanism involves the acetoxypalladation of the double bond in 222 resulting in the formation of intermediate 225 followed by the elimination of HPdOAc to give product 224b [213]. The first mechanism of the acetoxylation was confirmed by the data on the acetoxylation of 1,2-dideuteriocyclohexene [215], as well as by the detection of the π-allyl–palladium intermediate [217]. However, it cannot be ruled out that the acetoxylation can occur via the second mechanism under certain conditions (Scheme 47) [21,213].
Scheme 47: Possible mechanisms of the acetoxylation at the allylic position of alkenes by Pd(OAc)2.
Scheme 47: Possible mechanisms of the acetoxylation at the allylic position of alkenes by Pd(OAc)2.
The palladium-complex-catalyzed acetoxylation of terminal alkenes 226 under non-optimized conditions can afford a large number of products: vinyl acetate 227 and methyl ketone 228 (Wacker reaction), E and Z isomers of linear allyl ethers 229 and 230, and branched allyl ether 231 (Scheme 48) [218].
Scheme 48: Products of the oxidation of terminal alkenes by Pd(II)/AcOH/oxidant system.
Scheme 48: Products of the oxidation of terminal alkenes by Pd(II)/AcOH/oxidant system.
It was found that the selectivity of the reaction can be controlled by changing the polarity of the solvent. The reactions of terminal alkens with the Pd(OAc)2/benzoquinone system in a mixture of DMSO and AcOH as the solvent selectively produced linear E-allyl acetates in 50–65% yield [218]. The reaction in acetic acid affords methyl ketone and vinyl acetate; branched allyl ether is formed as the major product in the CH2Cl2/AcOH system using the 1,2-bis (benzylsulfinyl)ethane ligand [218].
The acyloxylation of terminal alkenes 232 with carboxylic acids 233 in the presence of 1,4-benzoquinone (BQ) as the oxidant, vinyl phenyl sulfoxide ligand 234, and Pd(OAc)2 resulted in the selective formation of branched allyl esters 235 [217]. This reaction afforded linear esters 236 as byproducts. Apparently, ligand 234 serves for the formation of the π-allyl–palladium intermediate, and benzoquinone mediates the subsequent reductive elimination to give product 235 (Scheme 49) [217].
Scheme 49: Acyloxylation of terminal alkenes with carboxylic acids.
Scheme 49: Acyloxylation of terminal alkenes with carboxylic acids.
The Pd(OAc)2·[1,2-bis(phenylsulfinyl)ethane]-catalyzed enantioselective acetoxylation of terminal alkenes was accomplished in the presence of a chiral Lewis acid; ee = 45–63%; the reaction was performed with a small excess of AcOH (1.1 equiv) in ethyl acetate using benzoquinone as the oxidant [219].
The Pd(OAc)2-catalyzed acetoxylation of terminal alkenes employing 4,5-diazafluorenone as the ligand can be performed in the presence of oxygen as the oxidizing agent (1 atm). The reaction gave linear allyl esters. The authors reported that this ligand facilitates the reductive elimination of the allyl ester from the π-allyl–palladium intermediate [220]. Linear E-allyl acetates were synthesized also by the acetoxylation of terminal alkenes with the PdCl2/NaOAc/AcOH/O2 system (5 atm) in N,N-dimethylacetamide [221]. The Wacker reaction giving methyl ketones occurs when using water instead of sodium acetate and acetic acid, all other conditions being the same [221].
The coupling of terminal alkenes 237 with carboxylic acids having complex structures 238 resulted in the selective formation of linear E-allyl esters 239 (Scheme 50) [222]. These reactions afford a Z isomer of linear ester and branched allyl ester as byproducts.
Scheme 50: Synthesis of linear E-allyl esters by cross-dehydrogenative coupling of terminal alkenes wih carboxylic acids.
Scheme 50: Synthesis of linear E-allyl esters by cross-dehydrogenative coupling of terminal alkenes wih carbox...
Similar results were obtained in the coupling of terminal alkenes in the presence of lithium hydroxide as the base in carboxylic acid acting as the OH-reagent or in a carboxylic acid/acetonitrile mixture; the coupling was performed with acetic, propionic, isobutyric, and pivalic acids [223].
It was shown that the stereoselectivity of the Pd(OAc)2-catalyzed acetoxylation of Z-vinyl(triethylsilanes) 240 can be controlled using benzoquinone or (diacetoxyiodo)benzene as the oxidant [224]. The reaction with benzoquinone affords the E isomer of C–O coupling product 241, whereas Z isomer 242 is formed as the major product in the case of (diacetoxyiodo)benzene (Scheme 51).
Scheme 51: Pd(OAc)2-catalyzed acetoxylation of Z-vinyl(triethylsilanes).
Scheme 51: Pd(OAc)2-catalyzed acetoxylation of Z-vinyl(triethylsilanes).
The α’-acetoxylation of α-acetoxyalkenes 243 with copper(II) chloride in acetic acid was reported [225] (Scheme 52). Alkenes free of the α-acetoxy group undergo trans-chlorination of the double bond in the presence of CuCl2. α,α’-Diacetoxyalkenes 244 were synthesized from alkenes with the additional use of the PdCl2/NaOAc/DDQ or Pd(OAc)2/benzoquinone/MnO2 reagents.
Scheme 52: α’-Acetoxylation of α-acetoxyalkenes with copper(II) chloride in acetic acid.
Scheme 52: α’-Acetoxylation of α-acetoxyalkenes with copper(II) chloride in acetic acid.
The oxidative acyloxylation at the allylic position of alkenes and at the benzylic position of some alkylarenes 245 with carboxylic acids 246 was accomplished in the presence of tert-butyl hydroperoxide and mixed copper aluminum oxide as the heterogeneous catalyst (Scheme 53) [226]. The catalyst was prepared from CuCl2 and AlCl3·6H2O by the co-precipitation from an aqueous solution in the presence of NaOH and Na2CO3. Allyl esters 247 with various structures were synthesized. The reactions were carried out taking alkene and carboxylic acid in a ratio of 1:1 or in the presence of an excess of alkene.
Scheme 53: Oxidative acyloxylation at the allylic position of alkenes and at the benzylic position of alkylarenes with carboxylic acids.
Scheme 53: Oxidative acyloxylation at the allylic position of alkenes and at the benzylic position of alkylare...
The intermolecular alkoxylation of methylheterocyclic compounds 248, 249 using the CuBr/5,6-dimethyl-1,10-phenanthroline (5,6-Me2phen)/(t-BuO)2 oxidative system was shown by a few examples [227]. The yields of coupling products 250, 251 were not higher than 37% (Scheme 54).
Scheme 54: Copper-catalyzed alkoxylation of methylheterocyclic compounds using di-tert-butylperoxide as oxidant.
Scheme 54: Copper-catalyzed alkoxylation of methylheterocyclic compounds using di-tert-butylperoxide as oxidan...
Methylarenes 252 were introduced into the oxidative C–O coupling with β-dicarbonyl compounds 253 and phenols 254 [228]. The coupling afforded structures 255 and 256. The method is applicable to phenols containing an aldehyde group, which was not oxidized in the course of the reaction (Scheme 55).
Scheme 55: Oxidative C–O coupling of methylarenes with β-dicarbonyl compounds or phenols.
Scheme 55: Oxidative C–O coupling of methylarenes with β-dicarbonyl compounds or phenols.
Oxidative C–O coupling of methylarenes 257 with cyclic ethers 258 and cycloalkanes 259 afforded α-acyloxy ethers 260 and allyl esters 261, respectively. The reactions were accomplished using the Cu(OAc)2/t-BuOOH oxidative system at low conversions of methylarenes in the corresponding ether or cycloalkane as solvent [229] (Scheme 56).
Scheme 56: Copper-catalyzed esterification of methylbenzenes with cyclic ethers and cycloalkanes.
Scheme 56: Copper-catalyzed esterification of methylbenzenes with cyclic ethers and cycloalkanes.
The proposed mechanism includes the copper-mediated generation of tert-butyl peresters from t-BuOOH and methylarenes and the formation of final products via intermolecular coupling of O-centered and C-centered radicals. Related oxidative C–O coupling reactions of alkanes with aromatic aldehydes [169] and carboxylic acids are discussed in sections 2.5 and 6, respectively.
The 4-methyl group of 2,4,6-trimethylphenol was selectively methoxylated in methanol in the presence of a copper(II) complex and hydrogen peroxide [230]; the similar alkoxylation reaction was performed with a stoichiometric amount of copper(II) chloride [231].
The benzylation of aryl-, alkyl-, and cycloalkylcarboxylic acids 262 in toluene was accomplished in the presence of the Pd(OAc)2/CF3SO3H/dimethylacetamide/O2 system (Scheme 57) [232]. Apparently, the reaction proceeds through an ionic mechanism involving the cleavage of the C–H bond of toluene with Pd(II). The product is formed either via the nucleophilic attack of carboxylic acid on Pd(II) complex 264 or as a result of the reductive elimination of product 263 from complex 265. It is supposed that dimethylacetamide promotes the reoxidation of Pd(0) to Pd(II) with oxygen and suppresses the aggregation of Pd(0), while trifluoromethanesulfonic acid facilitates the cleavage of the C–H bond of toluene through the formation of cationic Pd(II) compounds [232].
Scheme 57: Oxidative C–O coupling of carboxylic acids with toluene catalyzed by Pd(OAc)2.
Scheme 57: Oxidative C–O coupling of carboxylic acids with toluene catalyzed by Pd(OAc)2.
The gas-phase aerobic acetoxylation of toluene with acetic acid catalyzed by TiO2-, γ-Al2O3-, SiO2-, and ZrO2-supported Pd–Sb particles was also reported [233-236].
4.2 Reactions catalyzed by tetrabutylammonium iodide
The oxidative acyloxylation at the allylic position of alkenes 266 with carboxylic acids 267 was performed using the Bu4NI/t-BuOOH system to prepare esters 268 [237]. Apparently, the reaction proceeds through a radical mechanism, involving the hydrogen atom abstraction from the allylic position of alkene 266 with the tert-butyl peroxyl or tert-butoxyl radical; the authors suggested that the new C–O bond is formed as a result of the cross-recombination of the allyl and carboxyl radicals (Scheme 58) [237].
Scheme 58: Oxidative acyloxylation at the allylic position of alkenes with carboxylic acids using the Bu4NI/t-BuOOH system.
Scheme 58: Oxidative acyloxylation at the allylic position of alkenes with carboxylic acids using the Bu4NI/t-...
The coupling of carboxylic acids 269 with alkylarenes 270 occurs analogously to the coupling with alkenes (Scheme 59) [141,237]. It is supposed that [Bu4N]+IO− or [Bu4N]+[IO2]− generated in the Bu4NI/t-BuOOH system act as active oxidizing agents; the fact that the reaction proceeds via the hydrogen atom abstraction from the benzylic position to form the benzyl radical is confirmed by a number of experiments, including the trapping of the benzyl radical by the TEMPO radical [141]. The authors hypothesized that the benzyl radical is oxidized to the benzyl cation, which undergoes nucleophilic attack by the carboxylate anion to give cross-dehydrogenative C–O coupling product 271. It was shown that 1-iodo-1-phenylethane does not react with carboxylic acid under these reaction conditions [141].
Scheme 59: Cross-dehydrogenative C–O coupling of carboxylic acids with alkylarenes using the Bu4NI/t-BuOOH system.
Scheme 59: Cross-dehydrogenative C–O coupling of carboxylic acids with alkylarenes using the Bu4NI/t-BuOOH sys...
The oxidative C–O coupling of alkylarenes with aromatic benzyl alcohols [140] and aldehydes [142] in the presence of the Bu4NI/t-BuOOH system is considered in section 2.4 (Scheme 27 and Scheme 28).
The synthesis of symmetrical esters from methylarenes and of unsymmetrical esters 274 from methylarenes 272 and ethylarenes 273 in the presence of the Bu4NI/t-BuOOH system was documented (Scheme 60). The reaction was accomplished at low conversions of alkylarenes [238]. It was suggested that methylarene is oxidized to carboxylic acid and reacts with the benzylic carbocation generated from the second methylarene molecule or from ethylarene through the hydrogen atom abstraction followed by the oxidation.
Scheme 60: Oxidative C–O cross-coupling of methylarenes with ethyl or isopropylarenes.
Scheme 60: Oxidative C–O cross-coupling of methylarenes with ethyl or isopropylarenes.
The achievement of the cited study is that the authors succeeded in performing the selective oxidative C–O cross-coupling of methylarenes 272 with ethylarenes 273 and excluded side processes, in which symmetrical esters could be generated from methylarenes 272 (the formation of only small amounts of symmetrical esters was observed; 2–5% yield), as well as the oxidation of ethylarene 273 to aryl methyl ketone.
Phosphorylation of benzyl C–H bonds of alkylarenes 275 using the Bu4NI/t-BuOOH oxidative system was documented to afford products 277 (Scheme 61) [239]. In this process two bonds (C–H and P–H) are cleaved and two new bonds (C–O and P–O) are formed. The proposed mechanism includes benzyl radical formation, oxidation of the latter to the benzylic cation, and nucleophilic attack of phosphate which is formed by the oxidation of the P–H bond in 276.
Scheme 61: Phosphorylation of benzyl C–H bonds using the Bu4NI/t-BuOOH oxidative system.
Scheme 61: Phosphorylation of benzyl C–H bonds using the Bu4NI/t-BuOOH oxidative system.
2,3-Disubstituted indoles 278 are acetoxylated in the presence of the Bu4NI/PhI(OAc)2 system in a CH2Cl2/AcOH solvent mixture. In most cases, the reaction resulted in the regioselective introduction of the acetoxy group at one of the two possible positions resulting in the formation of product 279 or 280 (Scheme 62) [240].
Scheme 62: Selective C–H acetoxylation of 2,3-disubstituted indoles.
Scheme 62: Selective C–H acetoxylation of 2,3-disubstituted indoles.
The authors supposed that indoles undergo iodination (as exemplified by indoles 278a,b) with AcOI, which is generated from PhI(OAc)2 and the iodide anion, through the formation of intermediates 281a,b to give one of the two allyl iodides (282a or 282b); the nucleophilic attack of acetic acid or the acetate anion (SN’ nucleophilic substitution) affords products 280a or 279a.
4.3 Reactions using 2,3-dichloro-5,6-dicyano-1,4-benzoquinone (DDQ)
The common feature of the cross-dehydrogenative C–O coupling reactions that take place in the presence of DDQ is that C-reagents should contain conjugated systems without strong electron-withdrawing groups.
For example, the acetoxylation of alkylarenes 283 with DDQ in acetic acid under ultrasound irradiation produced acetates 284 (Scheme 63) [241].
Scheme 63: Acetoxylation of benzylic position of alkylarenes using DDQ as oxidant.
Scheme 63: Acetoxylation of benzylic position of alkylarenes using DDQ as oxidant.
The ultrasound irradiation results in a considerable increase in the selectivity of the reaction. The proposed mechanism involves the generation of acetoxyl radicals and DDQ-H radicals in the reaction of DDQ with acetic acid, and the latter radicals abstract a hydrogen atom from the benzylic position of alkylarene. The target product is formed as a result of the recombination of benzyl and acetoxyl radicals. The reactions with methylarenes were not performed; alkylarenes containing electron-withdrawing substituents (Br, NO2) were not involved in the oxidative coupling.
The oxidative coupling of diarylmethanes 285 with carboxylic acids 286 takes place in the presence of the DDQcat/MnO2 system (Scheme 64) [242]. It is suggested that DDQ oxidizes diarylmethanes to diarylmethyl cations, which react with carboxylic acids to form esters 287. Manganese dioxide serves for the oxidation of the reduced form of the catalyst DDQH2 to DDQ. The drawback of this method is that the reaction center of the C-component should contain two aryl groups and that a fourfold excess of carboxylic acids (O-components) is required. In the cited study, it was also reported that the coupling of carboxylic acids 289 with 3-phenyl-2-propen-1-yl acetate (288) in the presence of DDQ gives allyl esters 290. DDQ was used also for the acyloxylation at the benzylic position of dimethoxyarene 291 with carboxylic acids 292 to prepare esters 293 (Scheme 64) [243].
Scheme 64: C–H acyloxylation of diarylmethanes, 3-phenyl-2-propen-1-yl acetate and dimethoxyarene using DDQ.
Scheme 64: C–H acyloxylation of diarylmethanes, 3-phenyl-2-propen-1-yl acetate and dimethoxyarene using DDQ.
The benzylic position of β-phenylethylbenzamides was acetoxylated with DDQ in acetic acid at 80 °C; the reaction readily proceeds if the phenyl ring contains activating methoxy groups; in the case of unsubstituted β-phenylethylbenzamide, the conversion was incomplete [244]. (p-Isopropoxyphenyl)acetic acid esters were stereoselectively acetoxylated with DDQ despite the presence of an electron-withdrawing ester group in the vicinity of the reaction center [245].
The oxidative C–O coupling of 1,3-diarylpropylenes 294 with alcohols 295 was accomplished in the presence of DDQ [246]; the reaction was completed at room temperature in a period of time shorter than one hour; a mixture of two isomeric coupling products 296 was obtained in the case of different substituents R1 and R2. Under similar conditions, the oxidative coupling of 1,3-diarylpropynes 297 with alcohols, phenols, and carboxylic acids 298 afforded coupling products 299 (Scheme 65) [247].
Scheme 65: Cross-dehydrogenative C–O coupling of 1,3-diarylpropylenes and 1,3-diarylpropynes with alcohols.
Scheme 65: Cross-dehydrogenative C–O coupling of 1,3-diarylpropylenes and 1,3-diarylpropynes with alcohols.
A method was developed for the one-pot synthesis of 3-acyloxy-3-azido-1-aryl-1-propynes 302 from 3-chloro-1-arylpropynes 300 involving the acyloxylation of the methylene group with carboxylic acids 301 under the action of the FeCl2/DDQ system (Scheme 66) [248]. In the absence of iron salts, the reaction is less efficient.
Scheme 66: One-pot azidation and C–H acyloxylation of 3-chloro-1-arylpropynes.
Scheme 66: One-pot azidation and C–H acyloxylation of 3-chloro-1-arylpropynes.
The cross-dehydrogenative C–O coupling reactions with the use of oximes as O-reagents in the presence of DDQ were documented. 1,3-Diarylpropylenes 303, as well as (E)-1-phenyl-2-isopropylethylene (306) are involved in the oxidative coupling with oximes 304 and 307 in the presence of DDQ [249]. Under similar conditions, oximes 310 are involved in the coupling with isochromanes 309 [250] (Scheme 67).
Scheme 67: Cross-dehydrogenative C–O coupling of 1,3-diarylpropylenes, (E)-1-phenyl-2-isopropylethylene and isochromanes with oximes.
Scheme 67: Cross-dehydrogenative C–O coupling of 1,3-diarylpropylenes, (E)-1-phenyl-2-isopropylethylene and is...
It was suggested that carbocations are generated from C-reagents under the action of DDQ, and these carbocations undergo nucleophilic attack by oxime anions to form coupling products 305, 308, and 311 [249,250]. N-Hydroxyphthalimide was also employed as the O-reagent, the yield of the coupling product with isochromane was 62% [250]. DDQ was also used in the acetoxylation at the benzylic position of p-alkoxyalkylarenes and p-alkylphenols in acetic acid [251].
4.4 Reactions with N-hydroxyphthalimide
The oxidative C–O coupling of alkylarenes and related compounds 312 with N-hydroxyphthalimide (154b) was performed using CuCl/PhI(OAc)2 [252] or (NH4)2Ce(NO3)6 [253] as the oxidizing agent (Scheme 68). The reaction in the presence of the CuCl/PhI(OAc)2 system gives somewhat higher yields of coupling products 313 compared with (NH4)2Ce(NO3)6, but it requires higher temperature, a longer reaction time, and relatively larger excesses of alkylarene. Apparently, the C–O bond is formed as a result of the recombination of benzyl and phthalimide-N-oxyl radicals. Alkylarenes should be taken in an excess amount to achieve high yields. The coupling of N-hydroxyphthalimide with ethers and alkenes proceeds in a similar fashion [252].
Scheme 68: Cross-dehydrogenative C–O coupling of alkylarenes and related compounds with N-hydroxyphthalimide.
Scheme 68: Cross-dehydrogenative C–O coupling of alkylarenes and related compounds with N-hydroxyphthalimide.
It was reported that such oxidative coupling reactions with NHPI and alkylarenes can proceed through the generation of phthalimide-N-oxyl radicals from NHPI in the presence of Pb(OAc)4; however, the yields of the products were not published [254]. Later on, it was shown that cross-dehydrogenative coupling of NHPI with toluene proceeds in the presence of a variety of oxidants, (NH4)2Ce(NO3)6, PhI(OAc)2, KMnO4, Mn(OAc)3·2H2O, Pb(OAc)4, or Co(OAc)2/O2 to afford N-benzyloxyphthalimide in yields of 11–75% [255]. The oxidative coupling of NHPI with cyclohexene and cyclooctene using NaIO4 in the CH2Cl2/H2O mixture in the presence of silica gel was documented [256]; the coupling products were only partially characterized (the NMR spectra of the crude compounds were reported). In another study, the oxidative coupling of NHPI with cyclic and acyclic alkenes was performed in the presence of (NH4)2Ce(NO3)6, Pb(OAc)4, or anthraquinone; in the case of metal-containing oxidizing agents, the radical reaction mechanism is suggested along with the competitive ionic mechanism; the reaction products and procedures for their synthesis were not completely characterized [257].
The acetoxylation at the benzylic position of alkylarenes 314 in acetic acid was performed using the N-hydroxyphthalimide/iodine/nitric acid system; in some experiments, Co(OAc)2 was employed as the cocatalyst (Scheme 69) [258]. Oxygen or nitric acid acts as the oxidant.
Scheme 69: Acetoxylation at the benzylic position of alkylarenes mediated by N-hydroxyphthalimide.
Scheme 69: Acetoxylation at the benzylic position of alkylarenes mediated by N-hydroxyphthalimide.
In the presence of oxidizing agents, phthalimide-N-oxyl radicals are generated from NHPI, and these radicals abstract a hydrogen atom from the benzylic position of alkylarene 314; the resulting benzyl radical is trapped by iodine to form iodide followed by the replacement of iodine by acetic acid to give product 315. The proposed mechanism is confirmed by the fact that the reactions of alkylarenes containing electron-withdrawing substituents in the benzene ring afford benzyl iodides rather than the corresponding acetates.
4.5 Other reactions
The C–O coupling of methylarenes 316 with aromatic carboxylic acids 317 in the presence of the NaBrO3/NaHSO3 system was described [259]. The synthesis was performed at room temperature with aromatic carboxylic acids 317 containing both electron-donating and -withdrawing groups; methylarene 316 (toluene or 3-ethoxytoluene), and carboxylic acid 317 were taken in a ratio of 1:1. It was suggested that benzyl bromides are formed in situ followed by the generation of products 318 through the nucleophilic attack by carboxylic acid (Scheme 70).
Scheme 70: C–O coupling of methylarenes with aromatic carboxylic acids employing the NaBrO3/NaHSO3 system.
Scheme 70: C–O coupling of methylarenes with aromatic carboxylic acids employing the NaBrO3/NaHSO3 system.
The tert-butyl peroxide group was introduced into allyl, propargyl and benzyl ethers 319 in the presence of the Fe(acac)3/t-BuOOH system [260]. It was suggested that a hydrogen atom is abstracted from the CH-reagent by tert-butylperoxyl and tert-butoxyl radicals generated from tert-butyl hydroperoxide in the presence of Fe(acac)3; the resulting C-radical is oxidized with Fe(III) to the carbocation, and the latter is subjected to nucleophilic attack by tert-butyl hydroperoxide to give product 320 (Scheme 71).
Scheme 71: tert-Butyl peroxidation of allyl, propargyl and benzyl ethers catalyzed by Fe(acac)3.
Scheme 71: tert-Butyl peroxidation of allyl, propargyl and benzyl ethers catalyzed by Fe(acac)3.
The tert-butyl peroxidation at the allylic and benzylic positions of cyclohexene and ethylbenzene with tert-butyl hydroperoxide catalyzed by nanosized CeO2 proceeds with low yields [261].
The acetoxylation of alkylarenes in acetic acid was performed using the CeO2/NaBrO3 [262], LiBr/NaIO4 [263], and H5PV2Mo10O40/NaNO3 systems [264]. The methyl group of 1,2,3-trimethoxy-5-methylbenzene in acetic acid was acetoxylated with 65% nitric acid [265]. N-Benzylphthalimides were acetoxylated at the benzylic position with the N-bromosuccinimide/NaOAc/HOAc system by heating under reflux in chlorobenzene for 12 h [266].
The alkoxylation [267,268] or acyloxylation [267,269] at the allylic position of alkenes occurs in the presence of selenium-based oxidizing agents, such as ArSeSeArcat./S2O82− [267,268] or SeO2 [269]. It is suggested that the reactions proceed through electrophilic attack of selenium on the C=C double bond [268,269].
5 Ethers, amines, and amides as C-reagents in cross-dehydrogenative C–O coupling
5.1 Reactions catalyzed by tetrabutylammonium iodide
The Bu4NI/t-BuOOH system proved to be efficient in the oxidative C–O coupling of ethers 322 with carboxylic acids 321 (Scheme 72) [270]. The coupling was performed with ethers taken in a 20-fold excess relative to carboxylic acids. It is supposed that the tert-butoxyl radical, which is generated in the Bu4NI/t-BuOOH system, abstracts a hydrogen atom from the α-position of the ether, and the resulting C-radical is oxidized to the carbocation, which reacts with the carboxylate anion to form coupling product 323.
Scheme 72: Cross-dehydrogenative C–O coupling of ethers with carboxylic acids mediated by Bu4NI/t-BuOOH system.
Scheme 72: Cross-dehydrogenative C–O coupling of ethers with carboxylic acids mediated by Bu4NI/t-BuOOH system....
The Bu4NI/t-BuOOH system was applied to perform the oxidative acyloxylation of dimethylamides 325 and dioxane with 2-aryl-2-oxoacetic acids 324 accompanied by the decarboxylation [271]. It was shown that, in the presence of the Bu4NI/t-BuOOH system, 2-oxo-2-phenylacetic acid (324a) is transformed into benzoic acid (328) followed by the coupling of the latter with dimethylformamide to give product 326a. Hence, it is supposed that arylcarboxylic acids are intermediates in the synthesis of compounds 326 and 327 (Scheme 73).
Scheme 73: Oxidative acyloxylation of dimethylamides and dioxane with 2-aryl-2-oxoacetic acids accompanied by the decarboxylation.
Scheme 73: Oxidative acyloxylation of dimethylamides and dioxane with 2-aryl-2-oxoacetic acids accompanied by ...
The peroxidation of N-benzylamides and N-allylbenzamide 329 with the Bu4NI/t-BuOOH system (Scheme 74) was documented [272]. The tert-butyl peroxide group in products 330 can be replaced with an aryl, benzyl, or alkyl group by the reaction with the corresponding Grignard reagent to afford products 331.
Scheme 74: tert-Butyl peroxidation of N-benzylamides and N-allylbenzamide using the Bu4NI/t-BuOOH system.
Scheme 74: tert-Butyl peroxidation of N-benzylamides and N-allylbenzamide using the Bu4NI/t-BuOOH system.
5.2 Reactions catalyzed by transition metal salts
The oxidative coupling of aromatic carboxylic acids 332 with ethers 333, which served as the solvents, was accomplished using the Fe(acac)3/di-tert-butyl peroxide system to prepare α-acyloxy ethers 334 (Scheme 75) [273].
Scheme 75: Cross-dehydrogenative C–O coupling of aromatic carboxylic acids with ethers using Fe(acac)3 as catalyst and di-tert-butyl peroxide as oxidant.
Scheme 75: Cross-dehydrogenative C–O coupling of aromatic carboxylic acids with ethers using Fe(acac)3 as cata...
In addition to aromatic carboxylic acids 332, the coupling was performed with phenylacetic acid. The reaction takes place both with ethers and cyclohexene. In almost all syntheses, one of the following two C-reagents was used: 1,4-dioxane or 1,3-dioxolane. It is interesting that the oxidative coupling of carboxylic acids with 1,3-dioxolane occurs at the 4-poisition of dioxolane (with one adjacent oxygen atom) rather than at the 2-position (with two adjacent oxygen atoms), which is usually considered to be more activated for the hydrogen atom abstraction.
The oxidative coupling of 2-hydroxybenzaldehydes 335 with cyclic ethers 336 (1,4-dioxane, tetrahydrofuran) was performed in the presence of the iron carbonyl/t-BuOOH system; in these reactions, the ethers served as the solvents (Scheme 76) [274]. The reactions afforded products 337, leaving the aldehyde group unchanged.
Scheme 76: Cross-dehydrogenative C–O coupling of cyclic ethers with 2-hydroxybenzaldehydes using iron carbonyl as catalyst and tert-butyl hydroperoxide as oxidant.
Scheme 76: Cross-dehydrogenative C–O coupling of cyclic ethers with 2-hydroxybenzaldehydes using iron carbonyl...
Ethers 338 were subjected to oxidative coupling with 1,3-dicarbonyl compounds 339 and o-acylphenols 340 using the copper salt/t-BuOOH system to prepare esters 341 and 342 (Scheme 77) [275].
Scheme 77: Cross-dehydrogenative C–O coupling of ethers with β-dicarbonyl compounds and phenols using copper catalysts and tert-butyl hydroperoxide as oxidant.
Scheme 77: Cross-dehydrogenative C–O coupling of ethers with β-dicarbonyl compounds and phenols using copper c...
In the proposed mechanism (Scheme 77) the reactive benzoyl radical 344 derived from ether 343 by TBHP and a catalytic amount of copper catalyst reacts with complex 345 by single electron transfer to produce Cu(III) complex 346. Finally, reductive elimination of intermediate 346 affords the desired product 347 and the Cu(I) complex, which can be reoxidized to Cu(II) by TBHP.
Dioxane undergoes C–O oxidative coupling with 2-hydroxybenzaldehyde 348 in the presence of tert-butyl hydroperoxide and the metal-organic framework (MOF) Cu2(BPDC)2(BPY) (BPY = 4,4’-bipyridine, BPDC = 4,4’-biphenyldicarboxylate) as the heterogeneous catalyst [276] (Scheme 78). The yield of product 349 and the reactivity of related CH- and OH-reagents under these reaction conditions were not discussed.
Scheme 78: Cross-dehydrogenative C–O coupling of 2-hydroxybenzaldehyde with dioxane catalyzed by Cu2(BPDC)2(BPY).
Scheme 78: Cross-dehydrogenative C–O coupling of 2-hydroxybenzaldehyde with dioxane catalyzed by Cu2(BPDC)2(BP...
The ruthenium chloride-catalyzed acyloxylation of β-lactams 350 with an excess of acids as O-reagents was accomplished using oxygen in the presence of acetaldehyde (Scheme 79). It was hypothesized that the reaction proceeds through the generation of the Ru(V) oxo intermediate, which oxidizes the β-lactam to the carbocation, and the latter is subjected to nucleophilic attack by carboxylic acid to form substituted β-lactam 351 [277].
Scheme 79: Ruthenium chloride-catalyzed acyloxylation of β-lactams.
Scheme 79: Ruthenium chloride-catalyzed acyloxylation of β-lactams.
The tert-butyl peroxidation (product 354) or acetoxylation (product 355) of the methylene groups of amides 352 and 353 adjacent to the nitrogen atom was performed in the presence of a ruthenium-based catalyst using tert-butyl hydroperoxide or peracetic acid, respectively [278] (Scheme 80).
Scheme 80: Ruthenium-catalyzed tert-butyl peroxydation amides and acetoxylation of β-lactams.
Scheme 80: Ruthenium-catalyzed tert-butyl peroxydation amides and acetoxylation of β-lactams.
The α,β-diacetoxylation of N-arylpiperidines 356 with PhI(OAc)2 affords products 357 in 20–46% yield. Apparently, the reaction proceeds through the oxidation of amine to enamine [279] (Scheme 81).
Scheme 81: PhI(OAc)2-mediated α,β-diacetoxylation of tertiary amines.
Scheme 81: PhI(OAc)2-mediated α,β-diacetoxylation of tertiary amines.
Examples of the alkoxylation of tertiary amines 358 and 359 by their electrooxidation in alcohols were reported in the literature, examples are given in Scheme 82 [280] (the ratio of products 360–363 was determined by gas chromatography).
Scheme 82: Electrochemical oxidative methoxylation of tertiary amines.
Scheme 82: Electrochemical oxidative methoxylation of tertiary amines.
The coupling of N-hydroxyphthalimide with tetrahydrofuran (70% yield) and isochromane (71% yield) takes place in the presence of the CuCl/PhI(OAc)2 system; CH-reagents are taken in a 10-fold excess (see section 4.4, Scheme 68) [252].
6 Other cross-dehydrogenative C–O coupling reactions
Ketene dithioacetals 364 were subjected to acyloxylation in the presence of the Pd(OAc)2/PhI(OAc)2 system and an excess of carboxylic acids that served as the co-solvents to prepare products 365 (Scheme 83) [281]. The range of carboxylic acids used in this reaction is limited to lower members of the series.
Scheme 83: Cross-dehydrogenative C–O coupling of ketene dithioacetals with carboxylic acids in the presence of the Pd(OAc)2/PhI(OAc)2 system.
Scheme 83: Cross-dehydrogenative C–O coupling of ketene dithioacetals with carboxylic acids in the presence of...
The oxidative coupling of enamines containing an electron-withdrawing substituent 366 with carboxylic acids 367 occurs efficiently in the presence of iodosobenzene (Scheme 84); oxidative coupling products 368 were employed in the synthesis of oxazoles [282].
Scheme 84: Cross-dehydrogenative C–O coupling of enamides with carboxylic acids using iodosobenzene as oxidant.
Scheme 84: Cross-dehydrogenative C–O coupling of enamides with carboxylic acids using iodosobenzene as oxidant....
The oxidative alkoxylation [283], acetoxylation [283], and tosyloxylation [284] of anilides 369 at the para-position were accomplished using PhI(OOCCF3)2 in the presence of boron trifluoride to prepare products 370–372, respectively (Scheme 85); OH-reagents were taken in an excess.
Scheme 85: Oxidative alkoxylation, acetoxylation, and tosyloxylation of acylanilides using PhI(O(O)CCF3)2 in the presence of boron trifluoride.
Scheme 85: Oxidative alkoxylation, acetoxylation, and tosyloxylation of acylanilides using PhI(O(O)CCF3)2 in t...
The following mechanism was proposed. Anilide 373 is subjected to electrophilic attack by (di(trifluoroacetoxy)iodo)benzene to form intermediate 374 followed by the elimination of phenyl iodide from 374 to give cation 375, which is subjected to nucleophilic attack (ROH, AcOH, or TsOH) affording coupling product 376 (Scheme 86).
Scheme 86: Proposed mechanism of the oxidative C–O coupling of actetanilide with O-nucleophiles in the presence of (di(trifluoroacetoxy)iodo)benzene.
Scheme 86: Proposed mechanism of the oxidative C–O coupling of actetanilide with O-nucleophiles in the presenc...
In the presence of PhI(OAc)2, aldehydes 377, anilines 378 and alcohols undergo three-component coupling to produce alkoxyl-substituted N-arylimines 379 (Scheme 87). The preliminary mechanism investigations revealed that the transformation proceeds via imines as intermediates. Reactions were performed in the medium of alcohol (OH-reagent) at room temperature [285].
Scheme 87: Three-component coupling of aldehydes, anilines and alcohols involving oxidative intermolecular C–O bond formation.
Scheme 87: Three-component coupling of aldehydes, anilines and alcohols involving oxidative intermolecular C–O...
Various oxidants, such as (diacetoxyiodo)benzene [286-288], PbCl2/HClO4, NaIO3/HClO4, Br2/N-iodosuccinimide, or I2/30% aq H2O2 [289], were employed in the oxidative coupling of phenols 380 with alcohols serving as the solvents (Scheme 88) to prepare non-aromatic products 381 or 382.
Scheme 88: Oxidative coupling of phenols with alcohols.
Scheme 88: Oxidative coupling of phenols with alcohols.
The 2-acyloxylation of quinoline N-oxides 383 with arylaldehydes 384 in the presence of the CuOTf/t-BuOOH system afforded products 385 (Scheme 89) [290]. Benzaldehyde and aromatic aldehydes containing electron-donating groups were employed in the coupling. In addition to aromatic aldehydes, the coupling was performed with cyclohexanecarbaldehyde. It is suggested that the reaction proceeds through a radical mechanism.
Scheme 89: 2-Acyloxylation of quinoline N-oxides with arylaldehydes in the presence of the CuOTf/t-BuOOH system.
Scheme 89: 2-Acyloxylation of quinoline N-oxides with arylaldehydes in the presence of the CuOTf/t-BuOOH syste...
The oxidative coupling of primary alcohols 387 with azoles 386 was performed in the presence of the CuCl/(t-BuO)2 system (Scheme 90) to prepare products 388 in 16–57% yield [291]. It was hypothesized that the C–O bond is formed as a result of the reductive elimination of coupling product 388 from the copper(III) complex.
Scheme 90: Cross-dehydrogenative C–O coupling of azoles with primary alcohols.
Scheme 90: Cross-dehydrogenative C–O coupling of azoles with primary alcohols.
Alkoxylated heterocycles 391 were obtained in an excess of alcohol in the presence of Na3Co(NO2)6 from dipyrrins 390, which were generated from dipyrroles 389 (Scheme 91) [292].
Scheme 91: Oxidation of dipyrroles to dipyrrins and subsequent oxidative alkoxylation in the presence of Na3Co(NO2)6.
Scheme 91: Oxidation of dipyrroles to dipyrrins and subsequent oxidative alkoxylation in the presence of Na3Co...
1,2- and 1,4-naphthoquinones undergo alkoxylation in an alcoholic medium in the presence of transition metal salts [293], I2/CeCl3 [294], and HgO [295].
Alkanes are rarely used in the cross-dehydrogenative C–O coupling because of low reactivity of C–H bonds. An example is the trifluoroacetoxylation of cyclohexane, cycloheptane, cyclooctane, and adamantane in trifluoroacetic acid in the presence of peracetic acid [296] or hydrogen peroxide [297-299] with the addition of transition metal salts (Rh, Ru, Pd, Pt, Fe) [296,297] or in the absence of metal compounds [298,299]; the reactions were usually accomplished at room temperature for a few hours.
Copper-catalyzed oxidative dehydrogenative carboxylation of unactivated alkanes 393 with various aromatic acids 392 to produce the corresponding allylic esters 394 was reported recently [300] (Scheme 92). Mechanistic studies allowed proposing a mechanism involving the generation of an allyl radical via the formation of alkene from the starting alkane. Related oxidative C–O coupling of aromatic aldehydes [169] and methylarenes [229] with cycloalkanes using a Cu(OAc)2/t-BuOOH system were considered in sections 2.5 (Scheme 38) and 4.1 (Scheme 56), respectively.
Scheme 92: Oxidative dehydrogenative carboxylation of alkanes and cycloalkanes to allylic esters.
Scheme 92: Oxidative dehydrogenative carboxylation of alkanes and cycloalkanes to allylic esters.
Acetoxylation of benzene was achieved employing Pd(OAc)2/K2S2O8 oxidative system in AcOH/Ac2O mixture in the presence of a pyridinium-substituted pyridine ligand [301] (Scheme 93). It was proposed that a key role for this cationic ligand is to serve as a phase transfer catalyst to bring poorly soluble S2O82− into contact with the Pd catalyst.
Scheme 93: Pd-catalyzed acetoxylation of benzene.
Scheme 93: Pd-catalyzed acetoxylation of benzene.
The electrochemical cross-dehydrogenative C–O coupling, in which the O-reagent serves as the solvent, was described in the literature. For example, the methoxylation of methylarenes, amides, phenols, and other compounds was performed in methanol using bases immobilized on a solid support [302].
Conclusion
The analysis of the published data shows that oxidative C–O coupling reactions attract an increasing interest of organic chemists in recent years. In the past two decades, a considerable body of experimental data has been accumulated. Compared with other types of coupling, the oxidative C–O coupling is less well-studied despite the fact that the C–O–R group is rather commonly present in various classes of organic compounds, and a large number of diverse O-reagents are available for the coupling.
The drawback of the majority of the available methods for the cross-dehydrogenative C–O coupling, which limits their application to the coupling of two expensive complex compounds, is the use of an excess of one of the starting components, the C- or O-reagent. Besides, the synthesis often requires high temperature and long time to be completed.
The main goals in the development of the cross-dehydrogenative C–O coupling are as follows: (1) a search for new reactions involving various oxidative systems, C- and O-reagents; (2) the development of methods, which do not require an excess of the C- or O-reagent; (3) the development of methods based on available, convenient, and safe oxidants; (4) a decrease in the reaction temperature and time; (5) investigations of the mechanisms of the oxidative coupling with the aim of predicting conditions necessary for the efficient synthesis.
References
-
Li, C.-J.; Li, Z. Pure Appl. Chem. 2006, 78, 935–945. doi:10.1351/pac200678050935
Return to citation in text: [1] [2] [3] -
Li, Z.; Bohle, D. S.; Li, C.-J. Proc. Natl. Acad. Sci. U. S. A. 2006, 103, 8928–8933. doi:10.1073/pnas.0601687103
Return to citation in text: [1] [2] [3] -
Beccalli, E. M.; Broggini, G.; Martinelli, M.; Sottocornola, S. Chem. Rev. 2007, 107, 5318–5365. doi:10.1021/cr068006f
Return to citation in text: [1] [2] [3] [4] -
Li, C.-J. Acc. Chem. Res. 2009, 42, 335–344. doi:10.1021/ar800164n
Return to citation in text: [1] [2] [3] -
Scheuermann, C. J. Chem. – Asian J. 2010, 5, 436–451. doi:10.1002/asia.200900487
Return to citation in text: [1] [2] [3] -
Yoo, W.-J.; Li, C.-J. Top. Curr. Chem. 2010, 292, 281–302. doi:10.1007/128_2009_17
Return to citation in text: [1] [2] [3] -
Yeung, C. S.; Dong, V. M. Chem. Rev. 2011, 111, 1215–1292. doi:10.1021/cr100280d
Return to citation in text: [1] [2] [3] -
Cho, S. H.; Kim, J. Y.; Kwak, J.; Chang, S. Chem. Soc. Rev. 2011, 40, 5068–5083. doi:10.1039/c1cs15082k
Return to citation in text: [1] [2] [3] -
Sun, C.-L.; Li, B.-J.; Shi, Z.-J. Chem. Rev. 2011, 111, 1293–1314. doi:10.1021/cr100198w
Return to citation in text: [1] [2] [3] -
Zhang, C.; Tang, C.; Jiao, N. Chem. Soc. Rev. 2012, 41, 3464–3484. doi:10.1039/C2CS15323H
Return to citation in text: [1] [2] [3] [4] -
Song, G.; Wang, F.; Li, X. Chem. Soc. Rev. 2012, 41, 3651–3678. doi:10.1039/c2cs15281a
Return to citation in text: [1] [2] [3] -
Hirano, K.; Miura, M. Chem. Commun. 2012, 48, 10704–10714. doi:10.1039/C2CC34659A
Return to citation in text: [1] [2] [3] -
Liu, C.; Liu, D.; Lei, A. Acc. Chem. Res. 2014, 47, 3459–3470. doi:10.1021/ar5002044
Return to citation in text: [1] [2] [3] [4] -
Girard, S. A.; Knauber, T.; Li, C.-J. Angew. Chem., Int. Ed. 2014, 53, 74–100. doi:10.1002/anie.201304268
Return to citation in text: [1] [2] [3] -
Samanta, R.; Matcha, K.; Antonchick, A. P. Eur. J. Org. Chem. 2013, 5769–5804. doi:10.1002/ejoc.201300286
Return to citation in text: [1] [2] [3] -
Hoffman, R. V.; Wilson, A. L.; Kim, H. O. J. Org. Chem. 1990, 55, 1267–1270. doi:10.1021/jo00291a030
Return to citation in text: [1] -
Lifchits, O.; Demoulin, N.; List, B. Angew. Chem., Int. Ed. 2011, 50, 9680–9683. doi:10.1002/anie.201104244
Return to citation in text: [1] -
Jones, K. M.; Tomkinson, N. C. O. J. Org. Chem. 2012, 77, 921–928. doi:10.1021/jo202084w
Return to citation in text: [1] -
Liu, B.; Shi, B.-F. Tetrahedron Lett. 2015, 56, 15–22. doi:10.1016/j.tetlet.2014.11.039
Return to citation in text: [1] -
Liu, G.; Wu, Y. Top. Curr. Chem. 2010, 292, 195–209. doi:10.1007/128_2009_16
Return to citation in text: [1] [2] -
Liron, F.; Oble, J.; Lorion, M. M.; Poli, G. Eur. J. Org. Chem. 2014, 5863–5883. doi:10.1002/ejoc.201402049
Return to citation in text: [1] [2] [3] -
Wu, X.-F.; Gong, J.-L.; Qi, X. Org. Biomol. Chem. 2014, 12, 5807–5817. doi:10.1039/C4OB00276H
Return to citation in text: [1] -
Ananikov, V. P.; Khemchyan, L. L.; Ivanova, Yu. V.; Bukhtiyarov, V. I.; Sorokin, A. M.; Prosvirin, I. P.; Vatsadze, S. Z.; Medved'ko, A. V.; Nuriev, V. N.; Dilman, A. D.; Levin, V. V.; Koptyug, I. V.; Kovtunov, K. V.; Zhivonitko, V. V.; Likholobov, V. A.; Romanenko, A. V.; Simonov, P. A.; Nenajdenko, V. G.; Shmatova, O. I.; Muzalevskiy, V. M.; Nechaev, M. S.; Asachenko, A. F.; Morozov, O. S.; Dzhevakov, P. B.; Osipov, S. N.; Vorobyeva, D. V.; Topchiy, M. A.; Zotova, M. A.; Ponomarenko, S. A.; Borshchev, O. V.; Luponosov, Y. N.; Rempel, A. A.; Valeeva, A. A.; Stakheev, A. Yu.; Turova, O. V.; Mashkovsky, I. S.; Sysolyatin, S. V.; Malykhin, V. V.; Bukhtiyarova, G. A.; Terent'ev, A. O.; Krylov, I. B. Russ. Chem. Rev. 2014, 83, 885–985. doi:10.1070/RC2014v083n10ABEH004471
Return to citation in text: [1] -
Ekoue-Kovi, K.; Wolf, C. Chem. – Eur. J. 2008, 14, 6302–6315. doi:10.1002/chem.200800353
Return to citation in text: [1] -
Stowers, K. J.; Sanford, M. S. Org. Lett. 2009, 11, 4584–4587. doi:10.1021/ol901820w
Return to citation in text: [1] -
Anand, M.; Sunoj, R. B. Org. Lett. 2011, 13, 4802–4805. doi:10.1021/ol201830r
Return to citation in text: [1] -
Zhu, W.; Wang, H.; Peng, H.; Liu, G.; Guo, Y. Chin. J. Chem. 2013, 31, 371–376. doi:10.1002/cjoc.201201167
Return to citation in text: [1] -
Desai, L. V.; Stowers, K. J.; Sanford, M. S. J. Am. Chem. Soc. 2008, 130, 13285–13293. doi:10.1021/ja8045519
Return to citation in text: [1] -
Racowski, J. M.; Dick, A. R.; Sanford, M. S. J. Am. Chem. Soc. 2009, 131, 10974–10983. doi:10.1021/ja9014474
Return to citation in text: [1] -
Powers, D. C.; Ritter, T. Acc. Chem. Res. 2012, 45, 840–850. doi:10.1021/ar2001974
Return to citation in text: [1] -
Camasso, N. M.; Pérez-Temprano, M. H.; Sanford, M. S. J. Am. Chem. Soc. 2014, 136, 12771–12775. doi:10.1021/ja507056u
Return to citation in text: [1] -
Dick, A. R.; Kampf, J. W.; Sanford, M. S. J. Am. Chem. Soc. 2005, 127, 12790–12791. doi:10.1021/ja0541940
Return to citation in text: [1] -
Dick, A. R.; Hull, K. L.; Sanford, M. S. J. Am. Chem. Soc. 2004, 126, 2300–2301. doi:10.1021/ja031543m
Return to citation in text: [1] -
Kalyani, D.; Sanford, M. S. Org. Lett. 2005, 7, 4149–4152. doi:10.1021/ol051486x
Return to citation in text: [1] -
Ye, Z.; Wang, W.; Luo, F.; Zhang, S.; Cheng, J. Org. Lett. 2009, 11, 3974–3977. doi:10.1021/ol901609t
Return to citation in text: [1] -
Hu, C.-J.; Zhang, X.-H.; Ding, Q.-P.; Lv, T.; Ge, S.-P.; Zhong, P. Tetrahedron Lett. 2012, 53, 2465–2468. doi:10.1016/j.tetlet.2012.03.022
Return to citation in text: [1] -
Li, L.; Yu, P.; Cheng, J.; Chen, F.; Pan, C. Chem. Lett. 2012, 41, 600–602. doi:10.1246/cl.2012.600
Return to citation in text: [1] -
Bian, Y.-J.; Xiang, C.-B.; Chen, Z.-M.; Huang, Z.-Z. Synlett 2011, 2407–2409. doi:10.1055/s-0030-1261224
Return to citation in text: [1] -
Rout, S. K.; Guin, S.; Gogoi, A.; Majji, G.; Patel, B. K. Org. Lett. 2014, 16, 1614–1617. doi:10.1021/ol500224e
Return to citation in text: [1] -
Chen, X.; Hao, X.-S.; Goodhue, C. E.; Yu, J.-Q. J. Am. Chem. Soc. 2006, 128, 6790–6791. doi:10.1021/ja061715q
Return to citation in text: [1] -
Dudkina, Y. B.; Mikhaylov, D. Y.; Gryaznova, T. V.; Tufatullin, A. I.; Kataeva, O. N.; Vicic, D. A.; Budnikova, Y. H. Organometallics 2013, 32, 4785–4792. doi:10.1021/om400492g
Return to citation in text: [1] -
Zheng, X.; Song, B.; Xu, B. Eur. J. Org. Chem. 2010, 4376–4380. doi:10.1002/ejoc.201000631
Return to citation in text: [1] -
Leng, Y.; Yang, F.; Zhu, W.; Wu, Y.; Li, X. Org. Biomol. Chem. 2011, 9, 5288–5296. doi:10.1039/C1OB05223C
Return to citation in text: [1] -
Kamal, A.; Srinivasulu, V.; Sathish, M.; Tangella, Y.; Nayak, V. L.; Rao, M. P. N.; Shankaraiah, N.; Nagesh, N. Asian J. Org. Chem. 2014, 3, 68–76. doi:10.1002/ajoc.201300214
Return to citation in text: [1] -
Wang, Z.; Kuang, C. Adv. Synth. Catal. 2014, 356, 1549–1554. doi:10.1002/adsc.201400096
Return to citation in text: [1] -
Shi, S.; Kuang, C. J. Org. Chem. 2014, 79, 6105–6112. doi:10.1021/jo5008306
Return to citation in text: [1] -
Bhadra, S.; Matheis, C.; Katayev, D.; Gooßen, L. J. Angew. Chem., Int. Ed. 2013, 52, 9279–9283. doi:10.1002/anie.201303702
Return to citation in text: [1] [2] -
Li, W.; Sun, P. J. Org. Chem. 2012, 77, 8362–8366. doi:10.1021/jo301384r
Return to citation in text: [1] -
Wang, G.-W.; Yuan, T.-T. J. Org. Chem. 2010, 75, 476–479. doi:10.1021/jo902139b
Return to citation in text: [1] -
Jiang, T.-S.; Wang, G.-W. J. Org. Chem. 2012, 77, 9504–9509. doi:10.1021/jo301964m
Return to citation in text: [1] -
Wang, G.-W.; Yuan, T.-T.; Wu, X.-L. J. Org. Chem. 2008, 73, 4717–4720. doi:10.1021/jo8003088
Return to citation in text: [1] -
Yadav, M. R.; Rit, R. K.; Sahoo, A. K. Chem. – Eur. J. 2012, 18, 5541–5545. doi:10.1002/chem.201200092
Return to citation in text: [1] -
Rit, R. K.; Yadav, M. R.; Sahoo, A. K. Org. Lett. 2014, 16, 968–971. doi:10.1021/ol403699d
Return to citation in text: [1] -
Desai, L. V.; Malik, H. A.; Sanford, M. S. Org. Lett. 2006, 8, 1141–1144. doi:10.1021/ol0530272
Return to citation in text: [1] [2] -
Wang, L.; Xia, X.-D.; Guo, W.; Chen, J.-R.; Xiao, W.-J. Org. Biomol. Chem. 2011, 9, 6895–6898. doi:10.1039/C1OB05887H
Return to citation in text: [1] [2] -
Neufeldt, S. R.; Sanford, M. S. Org. Lett. 2010, 12, 532–535. doi:10.1021/ol902720d
Return to citation in text: [1] [2] -
Padala, K.; Jeganmohan, M. Chem. Commun. 2013, 49, 9651–9653. doi:10.1039/C3CC45350B
Return to citation in text: [1] -
Reddy, B. V. S.; Revathi, G.; Reddy, A. S.; Yadav, J. S. Tetrahedron Lett. 2011, 52, 5926–5929. doi:10.1016/j.tetlet.2011.08.098
Return to citation in text: [1] [2] -
Péron, F.; Fossey, C.; Sopkova-de Oliveira Santos, J.; Cailly, T.; Fabis, F. Chem. – Eur. J. 2014, 20, 7507–7513. doi:10.1002/chem.201303923
Return to citation in text: [1] [2] -
Yin, Z.; Jiang, X.; Sun, P. J. Org. Chem. 2013, 78, 10002–10007. doi:10.1021/jo401623j
Return to citation in text: [1] -
Zhang, C.; Sun, P. J. Org. Chem. 2014, 79, 8457–8461. doi:10.1021/jo5014146
Return to citation in text: [1] -
Zhang, S.-Y.; He, G.; Zhao, Y.; Wright, K.; Nack, W. A.; Chen, G. J. Am. Chem. Soc. 2012, 134, 7313–7316. doi:10.1021/ja3023972
Return to citation in text: [1] [2] -
Chen, F.-J.; Zhao, S.; Hu, F.; Chen, K.; Zhang, Q.; Zhang, S.-Q.; Shi, B.-F. Chem. Sci. 2013, 4, 4187–4192. doi:10.1039/C3SC51993G
Return to citation in text: [1] [2] -
Gou, F.-R.; Wang, X.-C.; Huo, P.-F.; Bi, H.-P.; Guan, Z.-H.; Liang, Y.-M. Org. Lett. 2009, 11, 5726–5729. doi:10.1021/ol902497k
Return to citation in text: [1] -
Chan, L. Y.; Meng, X.; Kim, S. J. Org. Chem. 2013, 78, 8826–8832. doi:10.1021/jo4011188
Return to citation in text: [1] -
Roane, J.; Daugulis, O. Org. Lett. 2013, 15, 5842–5845. doi:10.1021/ol402904d
Return to citation in text: [1] -
Hao, X.-Q.; Chen, L.-J.; Ren, B.; Li, L.-Y.; Yang, X.-Y.; Gong, J.-F.; Niu, J.-L.; Song, M.-P. Org. Lett. 2014, 16, 1104–1107. doi:10.1021/ol500166d
Return to citation in text: [1] -
Zhang, L.-B.; Hao, X.-Q.; Zhang, S.-K.; Liu, K.; Ren, B.; Gong, J.-F.; Niu, J.-L.; Song, M.-P. J. Org. Chem. 2014, 79, 10399–10409. doi:10.1021/jo502005j
Return to citation in text: [1] -
Zhang, L.-B.; Hao, X.-Q.; Zhang, S.-K.; Liu, Z.-J.; Zheng, X.-X.; Gong, J.-F.; Niu, J.-L.; Song, M.-P. Angew. Chem., Int. Ed. 2015, 54, 272–275. doi:10.1002/anie.201409751
Return to citation in text: [1] [2] -
Chernyak, N.; Dudnik, A. S.; Huang, C.; Gevorgyan, V. J. Am. Chem. Soc. 2010, 132, 8270–8272. doi:10.1021/ja1033167
Return to citation in text: [1] [2] -
Huang, C.; Chernyak, N.; Dudnik, A. S.; Gevorgyan, V. Adv. Synth. Catal. 2011, 353, 1285–1305. doi:10.1002/adsc.201000975
Return to citation in text: [1] [2] -
Gulevich, A. V.; Melkonyan, F. S.; Sarkar, D.; Gevorgyan, V. J. Am. Chem. Soc. 2012, 134, 5528–5531. doi:10.1021/ja3010545
Return to citation in text: [1] [2] -
Sarkar, D.; Melkonyan, F. S.; Gulevich, A. V.; Gevorgyan, V. Angew. Chem., Int. Ed. 2013, 52, 10800–10804. doi:10.1002/anie.201304884
Return to citation in text: [1] -
Gao, T.; Sun, P. J. Org. Chem. 2014, 79, 9888–9893. doi:10.1021/jo501902d
Return to citation in text: [1] [2] -
Nakano, Y.; Lupton, D. W. Chem. Commun. 2014, 50, 1757–1760. doi:10.1039/C3CC48545E
Return to citation in text: [1] -
Ju, L.; Yao, J.; Wu, Z.; Liu, Z.; Zhang, Y. J. Org. Chem. 2013, 78, 10821–10831. doi:10.1021/jo401830k
Return to citation in text: [1] -
Cheng, T.; Yin, W.; Zhang, Y.; Zhang, Y.; Huang, Y. Org. Biomol. Chem. 2014, 12, 1405–1411. doi:10.1039/C3OB42196A
Return to citation in text: [1] -
Jiang, H.; Chen, H.; Wang, A.; Liu, X. Chem. Commun. 2010, 46, 7259–7261. doi:10.1039/C0CC00841A
Return to citation in text: [1] -
Zhang, S.; Luo, F.; Wang, W.; Jia, X.; Hu, M.; Cheng, J. Tetrahedron Lett. 2010, 51, 3317–3319. doi:10.1016/j.tetlet.2010.04.075
Return to citation in text: [1] -
Wang, D.; Zavalij, P. Y.; Vedernikov, A. N. Organometallics 2013, 32, 4882–4891. doi:10.1021/om400618n
Return to citation in text: [1] -
Shan, G.; Yang, X.; Zong, Y.; Rao, Y. Angew. Chem., Int. Ed. 2013, 52, 13606–13610. doi:10.1002/anie.201307090
Return to citation in text: [1] -
Zhou, L.; Lu, W. Org. Lett. 2014, 16, 508–511. doi:10.1021/ol403393w
Return to citation in text: [1] [2] -
Rit, R. K.; Yadav, M. R.; Sahoo, A. K. Org. Lett. 2012, 14, 3724–3727. doi:10.1021/ol301579q
Return to citation in text: [1] -
Zong, Y.; Rao, Y. Org. Lett. 2014, 16, 5278–5281. doi:10.1021/ol502377x
Return to citation in text: [1] -
Wu, X.; Zhao, Y.; Ge, H. Chem. – Asian J. 2014, 9, 2736–2739. doi:10.1002/asia.201402510
Return to citation in text: [1] -
Desai, L. V.; Hull, K. L.; Sanford, M. S. J. Am. Chem. Soc. 2004, 126, 9542–9543. doi:10.1021/ja046831c
Return to citation in text: [1] [2] -
Stowers, K. J.; Kubota, A.; Sanford, M. S. Chem. Sci. 2012, 3, 3192–3195. doi:10.1039/C2SC20800H
Return to citation in text: [1] -
Giri, R.; Liang, J.; Lei, J.-G.; Li, J.-J.; Wang, D.-H.; Chen, X.; Naggar, I. C.; Guo, C.; Foxman, B. M.; Yu, J.-Q. Angew. Chem., Int. Ed. 2005, 44, 7420–7424. doi:10.1002/anie.200502767
Return to citation in text: [1] -
Wang, Z.; Kuninobu, Y.; Kanai, M. Org. Lett. 2014, 16, 4790–4793. doi:10.1021/ol5022542
Return to citation in text: [1] -
Owston, N. A.; Parker, A. J.; Williams, J. M. J. Chem. Commun. 2008, 624–625. doi:10.1039/B717073D
Return to citation in text: [1] -
Owston, N. A.; Nixon, T. D.; Parker, A. J.; Whittlesey, M. K.; Williams, J. M. J. Synthesis 2009, 1578–1581. doi:10.1055/s-0028-1088026
Return to citation in text: [1] -
Yamamoto, N.; Obora, Y.; Ishii, Y. J. Org. Chem. 2011, 76, 2937–2941. doi:10.1021/jo2003264
Return to citation in text: [1] -
Tschaen, B. A.; Schmink, J. R.; Molander, G. A. Org. Lett. 2013, 15, 500–503. doi:10.1021/ol303298g
Return to citation in text: [1] -
Heropoulos, G. A.; Villalonga-Barber, C. Tetrahedron Lett. 2011, 52, 5319–5322. doi:10.1016/j.tetlet.2011.08.021
Return to citation in text: [1] -
Liu, C.; Tang, S.; Zheng, L.; Liu, D.; Zhang, H.; Lei, A. Angew. Chem., Int. Ed. 2012, 51, 5662–5666. doi:10.1002/anie.201201960
Return to citation in text: [1] [2] -
Liu, C.; Wang, J.; Meng, L.; Deng, Y.; Li, Y.; Lei, A. Angew. Chem., Int. Ed. 2011, 50, 5144–5148. doi:10.1002/anie.201008073
Return to citation in text: [1] [2] [3] -
Gowrisankar, S.; Neumann, H.; Beller, M. Angew. Chem., Int. Ed. 2011, 50, 5139–5143. doi:10.1002/anie.201008035
Return to citation in text: [1] [2] [3] -
Nielsen, I. S.; Taarning, E.; Egeblad, K.; Madsen, R.; Christensen, C. H. Catal. Lett. 2007, 116, 35–40. doi:10.1007/s10562-007-9086-9
Return to citation in text: [1] -
Marsden, C.; Taarning, E.; Hansen, D.; Johansen, L.; Klitgaard, S. K.; Egeblad, K.; Christensen, C. H. Green Chem. 2008, 10, 168–170. doi:10.1039/B712171G
Return to citation in text: [1] -
Fristrup, P.; Johansen, L. B.; Christensen, C. H. Chem. Commun. 2008, 2750–2752. doi:10.1039/B803270J
Return to citation in text: [1] -
Su, F.-Z.; Ni, J.; Sun, H.; Cao, Y.; He, H.-Y.; Fa, K.-N. Chem. – Eur. J. 2008, 14, 7131–7135. doi:10.1002/chem.200800982
Return to citation in text: [1] -
Yasukawa, T.; Miyamura, H.; Kobayashi, S. Chem. – Asian J. 2011, 6, 621–627. doi:10.1002/asia.201000624
Return to citation in text: [1] -
Suzuki, K.; Yamaguchi, T.; Matsushita, K.; Iitsuka, C.; Miura, J.; Akaogi, T.; Ishida, H. ACS Catal. 2013, 3, 1845–1849. doi:10.1021/cs4004084
Return to citation in text: [1] -
Noonan, C.; Baragwanath, L.; Connon, S. J. Tetrahedron Lett. 2008, 49, 4003–4006. doi:10.1016/j.tetlet.2008.04.097
Return to citation in text: [1] [2] -
Maki, B. E.; Scheidt, K. A. Org. Lett. 2008, 10, 4331–4334. doi:10.1021/ol8018488
Return to citation in text: [1] [2] -
Maki, B. E.; Chan, A.; Phillips, E. M.; Scheidt, K. A. Tetrahedron 2009, 65, 3102–3109. doi:10.1016/j.tet.2008.10.033
Return to citation in text: [1] [2] [3] [4] -
Reddy, R. S.; Rosa, J. N.; Veiros, L. F.; Caddick, S.; Gois, P. M. P. Org. Biomol. Chem. 2011, 9, 3126–3129. doi:10.1039/C1OB05151B
Return to citation in text: [1] [2] -
Finney, E. E.; Ogawa, K. A.; Boydston, A. J. J. Am. Chem. Soc. 2012, 134, 12374–12377. doi:10.1021/ja304716r
Return to citation in text: [1] [2] -
Kiran, I. N. C.; Lalwani, K.; Sudalai, A. RSC Adv. 2013, 3, 1695–1698. doi:10.1039/C2RA22718E
Return to citation in text: [1] [2] -
Zhao, J.; Mück-Lichtenfeld, C.; Studer, A. Adv. Synth. Catal. 2013, 355, 1098–1106. doi:10.1002/adsc.201300034
Return to citation in text: [1] [2] -
Chiarotto, I.; Feroci, M.; Sotgiu, G.; Inesi, A. Tetrahedron 2013, 69, 8088–8095. doi:10.1016/j.tet.2013.06.014
Return to citation in text: [1] [2] -
Ji, M.; Wang, X.; Lim, Y. N.; Kang, Y.-W.; Jang, H.-Y. Eur. J. Org. Chem. 2013, 7881–7885. doi:10.1002/ejoc.201301337
Return to citation in text: [1] [2] -
Maki, B. E.; Chan, A.; Phillips, E. M.; Scheidt, K. A. Org. Lett. 2007, 9, 371–374. doi:10.1021/ol062940f
Return to citation in text: [1] [2] [3] -
Delany, E. G.; Fagan, C.-L.; Gundala, S.; Zeitler, K.; Connon, S. J. Chem. Commun. 2013, 49, 6513–6515. doi:10.1039/C3CC42597E
Return to citation in text: [1] [2] -
De Sarkar, S.; Grimme, S.; Studer, A. J. Am. Chem. Soc. 2010, 132, 1190–1191. doi:10.1021/ja910540j
Return to citation in text: [1] -
Samanta, R. C.; De Sarkar, S.; Fröhlich, R.; Grimme, S.; Studer, A. Chem. Sci. 2013, 4, 2177–2184. doi:10.1039/C3SC00099K
Return to citation in text: [1] -
Schmidt, A.; Habeck, T.; Snovydovych, B.; Eisfeld, W. Org. Lett. 2007, 9, 3515–3518. doi:10.1021/ol0713739
Return to citation in text: [1] -
Cheng, S.; Chen, J.; Gao, W.; Jin, H.; Ding, J.; Wu, H. J. Chem. Res. 2010, 34, 130–132. doi:10.3184/030823410X12670951969185
Return to citation in text: [1] -
Zhang, M.; Zhang, S.; Zhang, G.; Chen, F.; Cheng, J. Tetrahedron Lett. 2011, 52, 2480–2483. doi:10.1016/j.tetlet.2011.03.017
Return to citation in text: [1] -
Valizadeh, H.; Ahmadi, M. C. R. Chim. 2012, 15, 1077–1080. doi:10.1016/j.crci.2012.08.004
Return to citation in text: [1] -
Kuwano, S.; Harada, S.; Oriez, R.; Yamada, K. Chem. Commun. 2012, 48, 145–147. doi:10.1039/C1CC15539C
Return to citation in text: [1] -
Delany, E. G.; Fagan, C.-L.; Gundala, S.; Mari, A.; Broja, T.; Zeitler, K.; Connon, S. J. Chem. Commun. 2013, 49, 6510–6512. doi:10.1039/C3CC42596G
Return to citation in text: [1] -
Luo, F.; Pan, C.; Cheng, J.; Chen, F. Tetrahedron 2011, 67, 5878–5882. doi:10.1016/j.tet.2011.06.060
Return to citation in text: [1] [2] -
Zhang, D.; Pan, C. Catal. Commun. 2012, 20, 41–45. doi:10.1016/j.catcom.2011.12.041
Return to citation in text: [1] [2] -
Iwahana, S.; Iida, H.; Yashima, E. Chem. – Eur. J. 2011, 17, 8009–8013. doi:10.1002/chem.201100737
Return to citation in text: [1] -
De Sarkar, S.; Biswas, A.; Song, C. H.; Studer, A. Synthesis 2011, 1974–1983. doi:10.1055/s-0030-1260030
Return to citation in text: [1] -
Mori, N.; Togo, H. Tetrahedron 2005, 61, 5915–5925. doi:10.1016/j.tet.2005.03.097
Return to citation in text: [1] -
Yamada, S.; Morizono, D.; Yamamoto, K. Tetrahedron Lett. 1992, 33, 4329–4332. doi:10.1016/S0040-4039(00)74252-3
Return to citation in text: [1] -
Reddy, K. R.; Venkateshwar, M.; Maheswari, C. U.; Prashanthi, S. Synth. Commun. 2009, 40, 186–195. doi:10.1080/00397910902838920
Return to citation in text: [1] -
Karade, N. N.; Budhewar, V. H.; Katkar, A. N.; Tiwari, G. B. ARKIVOC 2006, No. xi, 162–167.
Return to citation in text: [1] -
Kiran, Y. B.; Ikeda, R.; Sakai, N.; Konakahara, T. Synthesis 2010, 276–282. doi:10.1055/s-0029-1217121
Return to citation in text: [1] -
Karade, N. N.; Shirodkar, S. G.; Dhoot, B. M.; Waghmare, P. B. J. Chem. Res. 2005, 274–276. doi:10.3184/0308234054213357
Return to citation in text: [1] -
Shaikh, T. M. A.; Emmanuvel, L.; Sudalai, A. Synth. Commun. 2007, 37, 2641–2646. doi:10.1080/00397910701465032
Return to citation in text: [1] -
Leduc, A. B.; Jamison, T. F. Org. Process Res. Dev. 2012, 16, 1082–1089. doi:10.1021/op200118h
Return to citation in text: [1] -
Stevens, R. V.; Chapman, K. T.; Stubbs, C. A.; Tam, W. W.; Albizati, K. F. Tetrahedron Lett. 1982, 23, 4647–4650. doi:10.1016/S0040-4039(00)85677-4
Return to citation in text: [1] -
Sayama, S. Synlett 2004, 2739–2745. doi:10.1055/s-2004-835630
Return to citation in text: [1] -
Agrawal, M. K.; Adimurthy, S.; Ghosh, P. K. Synth. Commun. 2012, 42, 2931–2936. doi:10.1080/00397911.2011.572219
Return to citation in text: [1] -
McDonald, C.; Holcomb, H.; Kennedy, K.; Kirkpatrick, E.; Leathers, T.; Vanemon, P. J. Org. Chem. 1989, 54, 1213–1215. doi:10.1021/jo00266a046
Return to citation in text: [1] -
Ghorbani-Vaghei, R.; Shahbazee, E.; Veisi, H. Mendeleev Commun. 2005, 15, 207–208. doi:10.1070/MC2005v015n05ABEH002091
Return to citation in text: [1] -
Liu, L.; Yun, L.; Wang, Z.; Fu, X.; Yan, C. Tetrahedron Lett. 2013, 54, 5383–5386. doi:10.1016/j.tetlet.2013.07.114
Return to citation in text: [1] [2] [3] [4] [5] -
Feng, J.; Liang, S.; Chen, S.-Y.; Zhang, J.; Fu, S.-S.; Yu, X.-Q. Adv. Synth. Catal. 2012, 354, 1287–1292. doi:10.1002/adsc.201100920
Return to citation in text: [1] [2] [3] [4] [5] -
Huang, J.; Li, L.-T.; Li, H.-Y.; Husan, E.; Wang, P.; Wang, B. Chem. Commun. 2012, 48, 10204–10206. doi:10.1039/C2CC35450K
Return to citation in text: [1] [2] [3] -
Wei, W.; Zhang, C.; Xu, Y.; Wan, X. Chem. Commun. 2011, 47, 10827–10829. doi:10.1039/C1CC14602E
Return to citation in text: [1] -
Zhu, F.; Wang, Z.-X. Tetrahedron 2014, 70, 9819–9827. doi:10.1016/j.tet.2014.11.002
Return to citation in text: [1] -
Tan, B.; Toda, N.; Barbas, C. F., III. Angew. Chem., Int. Ed. 2012, 51, 12538–12541. doi:10.1002/anie.201205921
Return to citation in text: [1] -
Wang, G.; Yu, Q.-Y.; Wang, J.; Wang, S.; Chen, S.-Y.; Yu, X.-Q. RSC Adv. 2013, 3, 21306–21310. doi:10.1039/C3RA43799J
Return to citation in text: [1] [2] -
Wang, N.; Liu, R.; Xu, Q.; Liang, X. Chem. Lett. 2006, 35, 566–567. doi:10.1246/cl.2006.566
Return to citation in text: [1] [2] -
Schulze, A.; Giannis, A. Adv. Synth. Catal. 2004, 346, 252–256. doi:10.1002/adsc.200303195
Return to citation in text: [1] [2] -
Yao, H.; Tang, Y.; Yamamoto, K. Tetrahedron Lett. 2012, 53, 5094–5098. doi:10.1016/j.tetlet.2012.07.024
Return to citation in text: [1] [2] -
Toledo, H.; Pisarevsky, E.; Abramovich, A.; Szpilman, A. M. Chem. Commun. 2013, 49, 4367–4369. doi:10.1039/C2CC35220F
Return to citation in text: [1] -
Rout, S. K.; Guin, S.; Ghara, K. K.; Banerjee, A.; Patel, B. K. Org. Lett. 2012, 14, 3982–3985. doi:10.1021/ol301756y
Return to citation in text: [1] -
Raju, K. B.; Kumar, B. N.; Nagaiah, K. RSC Adv. 2014, 4, 50795–50800. doi:10.1039/C4RA06233G
Return to citation in text: [1] -
Sharma, S.; Park, J.; Kim, M.; Kwak, J. H.; Jung, Y. H.; Kim, I. S. Tetrahedron 2013, 69, 9391–9397. doi:10.1016/j.tet.2013.08.079
Return to citation in text: [1] [2] -
Yoo, W.-J.; Li, C.-J. J. Org. Chem. 2006, 71, 6266–6268. doi:10.1021/jo0606103
Return to citation in text: [1] -
Kumar, G. S.; Maheswari, C. U.; Kumar, R. A.; Kantam, M. L.; Reddy, K. R. Angew. Chem., Int. Ed. 2011, 50, 11748–11751. doi:10.1002/anie.201105020
Return to citation in text: [1] [2] -
Barve, B. D.; Wu, Y.-C.; El-Shazly, M.; Chuang, D.-W.; Chung, Y.-M.; Tsai, Y.-H.; Wu, S.-F.; Korinek, M.; Du, Y.-C.; Hsieh, C.-T.; Wang, J.-J.; Chang, F.-R. Eur. J. Org. Chem. 2012, 6760–6766. doi:10.1002/ejoc.201201160
Return to citation in text: [1] [2] -
Barve, B. D.; Wu, Y.-C.; El-Shazly, M.; Chuang, D.-W.; Cheng, Y.-B.; Wang, J.-J.; Chang, F.-R. J. Org. Chem. 2014, 79, 3206–3214. doi:10.1021/jo402798k
Return to citation in text: [1] [2] -
Saberi, D.; Heydari, A. Tetrahedron Lett. 2013, 54, 4178–4180. doi:10.1016/j.tetlet.2013.05.113
Return to citation in text: [1] [2] [3] -
Phan, N. T. S.; Nguyen, T. T.; Vu, P. H. L. ChemCatChem 2013, 5, 3068–3077. doi:10.1002/cctc.201300400
Return to citation in text: [1] -
Gopinath, R.; Patel, B. K. Org. Lett. 2000, 2, 577–579. doi:10.1021/ol990383+
Return to citation in text: [1] -
Gopinath, R.; Barkakaty, B.; Talukdar, B.; Patel, B. K. J. Org. Chem. 2003, 68, 2944–2947. doi:10.1021/jo0266902
Return to citation in text: [1] -
Talukdar, D.; Sharma, K.; Bharadwaj, S. K.; Thakur, A. J. Synlett 2013, 24, 963–966. doi:10.1055/s-0032-1316914
Return to citation in text: [1] -
Yoo, W.-J.; Li, C.-J. Tetrahedron Lett. 2007, 48, 1033–1035. doi:10.1016/j.tetlet.2006.11.169
Return to citation in text: [1] -
Wu, X.-F.; Darcel, C. Eur. J. Org. Chem. 2009, 1144–1147. doi:10.1002/ejoc.200801176
Return to citation in text: [1] -
Rafiee, E.; Eavani, S. J. Mol. Catal. A: Chem. 2013, 373, 30–37. doi:10.1016/j.molcata.2013.02.024
Return to citation in text: [1] -
Sharma, R. K.; Gulati, S. J. Mol. Catal. A: Chem. 2012, 363–364, 291–303. doi:10.1016/j.molcata.2012.07.004
Return to citation in text: [1] -
Esfandiari, H.; Jameh-bozorghi, S.; Esmaielzadeh, S.; Shafiee, M. R. M.; Ghashang, M. Res. Chem. Intermed. 2013, 39, 3319–3325. doi:10.1007/s11164-012-0844-y
Return to citation in text: [1] -
Wu, X.-F. Tetrahedron Lett. 2012, 53, 3397–3399. doi:10.1016/j.tetlet.2012.04.111
Return to citation in text: [1] -
Zhao, J.; Fang, H.; Han, J.; Pan, Y. Org. Lett. 2014, 16, 2530–2533. doi:10.1021/ol5009119
Return to citation in text: [1] [2] [3] -
O'Connor, B.; Just, G. Tetrahedron Lett. 1987, 28, 3235–3236. doi:10.1016/S0040-4039(00)95480-7
Return to citation in text: [1] -
Sinha, A. K.; Sharma, A.; Swaroop, A.; Kumar, V. Tetrahedron 2007, 63, 1000–1007. doi:10.1016/j.tet.2006.11.011
Return to citation in text: [1] -
Sakuragi, H.; Tokumaru, K. Chem. Lett. 1974, 3, 475–476. doi:10.1246/cl.1974.475
Return to citation in text: [1] -
Sundararaman, P.; Walker, E. C.; Djerassi, C. Tetrahedron Lett. 1978, 19, 1627–1628. doi:10.1016/S0040-4039(01)94623-4
Return to citation in text: [1] -
Mineno, T.; Sakai, M.; Ubukata, A.; Nakahara, K.; Yoshimitsu, H.; Kansui, H. Chem. Pharm. Bull. 2013, 61, 870–872. doi:10.1248/cpb.c13-00072
Return to citation in text: [1] [2] -
Mineno, T.; Yoshino, S.; Ubukata, A. Green Sustainable Chem. 2014, 4, 20–23. doi:10.4236/gsc.2014.41004
Return to citation in text: [1] -
Travis, B. R.; Sivakumar, M.; Hollist, G. O.; Borhan, B. Org. Lett. 2003, 5, 1031–1034. doi:10.1021/ol0340078
Return to citation in text: [1] -
Nishihara, A.; Kubota, I. J. Org. Chem. 1968, 33, 2525–2526. doi:10.1021/jo01270a082
Return to citation in text: [1] -
Feng, J.-B.; Gong, J.-L.; Li, Q.; Wu, X.-F. Tetrahedron Lett. 2014, 55, 1657–1659. doi:10.1016/j.tetlet.2014.01.111
Return to citation in text: [1] -
Tank, R.; Pathak, U.; Vimal, M.; Bhattacharyya, S.; Pandey, L. K. Green Chem. 2011, 13, 3350–3354. doi:10.1039/C1GC16041A
Return to citation in text: [1] -
Kelly, C. B.; Mercadante, M. A.; Wiles, R. J.; Leadbeater, N. E. Org. Lett. 2013, 15, 2222–2225. doi:10.1021/ol400785d
Return to citation in text: [1] -
Zhang, C.; Jiao, N. Org. Chem. Front. 2014, 1, 109–112. doi:10.1039/C3QO00041A
Return to citation in text: [1] -
Srimani, D.; Balaraman, E.; Gnanaprakasam, B.; Ben-David, Y.; Milstein, D. Adv. Synth. Catal. 2012, 354, 2403–2406. doi:10.1002/adsc.201200438
Return to citation in text: [1] -
Tanaka, A.; Moriyama, K.; Togo, H. Synlett 2011, 1853–1858. doi:10.1055/s-0030-1260948
Return to citation in text: [1] -
Hu, J.; Zhu, M.; Xu, Y.; Yan, J. Synthesis 2012, 1226–1232. doi:10.1055/s-0031-1289750
Return to citation in text: [1] -
Yu, J.; Tian, J.; Zhang, C. Adv. Synth. Catal. 2010, 352, 531–546. doi:10.1002/adsc.200900737
Return to citation in text: [1] -
Yu, J.; Cui, J.; Hou, X.-S.; Liu, S.-S.; Gao, W.-C.; Jiang, S.; Tian, J.; Zhang, C. Tetrahedron: Asymmetry 2011, 22, 2039–2055. doi:10.1016/j.tetasy.2011.12.003
Return to citation in text: [1] [2] -
Moriarty, R. M.; Vaid, R. K.; Ravikumar, V. T.; Vaid, B. K.; Hopkins, T. E. Tetrahedron 1988, 44, 1603–1607. doi:10.1016/S0040-4020(01)86720-X
Return to citation in text: [1] [2] -
Price, D. A.; Gayton, S.; Stupple, P. A. Synlett 2002, 1170–1172. doi:10.1055/s-2002-32577
Return to citation in text: [1] -
Liu, W.-B.; Chen, C.; Zhang, Q.; Zhu, Z.-B. Bull. Korean Chem. Soc. 2012, 33, 1823–1824. doi:10.5012/bkcs.2012.33.6.1823
Return to citation in text: [1] -
Yamamoto, Y.; Kawano, Y.; Toy, P. H.; Togo, H. Tetrahedron 2007, 63, 4680–4687. doi:10.1016/j.tet.2007.03.091
Return to citation in text: [1] -
Suzuki, Y.; Togo, H. Synthesis 2010, 2355–2360. doi:10.1055/s-0029-1218795
Return to citation in text: [1] -
Sheng, J.; Li, X.; Tang, M.; Gao, B.; Huang, G. Synthesis 2007, 1165–1168. doi:10.1055/s-2007-965984
Return to citation in text: [1] -
Tanaka, A.; Togo, H. Synlett 2009, 3360–3364. doi:10.1055/s-0029-1218370
Return to citation in text: [1] -
Uyanik, M.; Suzuki, D.; Yasui, T.; Ishihara, K. Angew. Chem., Int. Ed. 2011, 50, 5331–5334. doi:10.1002/anie.201101522
Return to citation in text: [1] [2] -
Li, X.; Zhou, C.; Xu, X. ARKIVOC 2012, No. ix, 150–158.
Return to citation in text: [1] -
Guo, S.; Yu, J.-T.; Dai, Q.; Yang, H.; Cheng, J. Chem. Commun. 2014, 50, 6240–6242. doi:10.1039/C4CC01652A
Return to citation in text: [1] -
Terent’ev, A. O.; Krylov, I. B.; Timofeev, V. P.; Starikova, Z. A.; Merkulova, V. M.; Ilovaisky, A. I.; Nikishin, G. I. Adv. Synth. Catal. 2013, 355, 2375–2390. doi:10.1002/adsc.201300341
Return to citation in text: [1] [2] -
Krylov, I. B.; Terent’ev, A. O.; Timofeev, V. P.; Shelimov, B. N.; Novikov, R. A.; Merkulova, V. M.; Nikishin, G. I. Adv. Synth. Catal. 2014, 356, 2266–2280. doi:10.1002/adsc.201400143
Return to citation in text: [1] [2] -
Terent'ev, A. O.; Borisov, D. A.; Yaremenko, I. A.; Chernyshev, V. V.; Nikishin, G. I. J. Org. Chem. 2010, 75, 5065–5071. doi:10.1021/jo100793j
Return to citation in text: [1] -
Terent'ev, A. O.; Borisov, D. A.; Semenov, V. V.; Chernyshev, V. V.; Dembitsky, V. M.; Nikishin, G. I. Synthesis 2011, 2091–2100. doi:10.1055/s-0030-1260027
Return to citation in text: [1] -
Kharasch, M. S.; Fono, A. J. Org. Chem. 1959, 24, 72–78. doi:10.1021/jo01083a022
Return to citation in text: [1] -
Baidya, M.; Griffin, K. A.; Yamamoto, H. J. Am. Chem. Soc. 2012, 134, 18566–18569. doi:10.1021/ja309311z
Return to citation in text: [1] [2] -
Frazier, C. P.; Sandoval, D.; Palmer, L. I.; de Alaniz, J. R. Chem. Sci. 2013, 4, 3857–3862. doi:10.1039/C3SC51658J
Return to citation in text: [1] -
Tanyeli, C.; Sezen, B. Tetrahedron Lett. 2000, 41, 7973–7976. doi:10.1016/S0040-4039(00)01374-5
Return to citation in text: [1] [2] -
Tanyeli, C.; Sezen, B.; İyigün, Ç.; Elmalı, O. Tetrahedron Lett. 2001, 42, 6397–6399. doi:10.1016/S0040-4039(01)01262-X
Return to citation in text: [1] -
Tanyeli, C.; Özdemirhan, D.; Sezen, B. Tetrahedron 2002, 58, 9983–9988. doi:10.1016/S0040-4020(02)01350-9
Return to citation in text: [1] -
Tanyeli, C.; Tosun, A.; Turkut, E.; Sezen, B. Tetrahedron 2003, 59, 1055–1058. doi:10.1016/S0040-4020(02)01634-4
Return to citation in text: [1] -
Tanyeli, C.; Iyigün, Ç. Tetrahedron 2003, 59, 7135–7139. doi:10.1016/S0040-4020(03)01094-9
Return to citation in text: [1] -
Demir, A. S.; Reis, Ö.; Igdir, A. C. Tetrahedron 2004, 60, 3427–3432. doi:10.1016/j.tet.2004.02.039
Return to citation in text: [1] [2] [3] -
Demir, A. S.; Caliskan, Z.; Aydin, A. E.; Bicer, I. Tetrahedron: Asymmetry 2006, 17, 786–791. doi:10.1016/j.tetasy.2006.01.025
Return to citation in text: [1] [2] -
Demir, A. S.; Findik, H. Tetrahedron 2008, 64, 6196–6201. doi:10.1016/j.tet.2008.05.004
Return to citation in text: [1] [2] -
Marín-Barrios, R.; García-Cabeza, A. L.; Moreno-Dorado, F. J.; Guerra, F. M.; Massanet, G. M. J. Org. Chem. 2014, 79, 6501–6509. doi:10.1021/jo500915r
Return to citation in text: [1] -
Kitching, W.; Rappoport, Z.; Winstein, S.; Young, W. G. J. Am. Chem. Soc. 1966, 88, 2054–2055. doi:10.1021/ja00961a043
Return to citation in text: [1] [2] [3] -
Engelin, C.; Jensen, T.; Rodriguez-Rodriguez, S.; Fristrup, P. ACS Catal. 2013, 3, 294–302. doi:10.1021/cs3007878
Return to citation in text: [1] -
Grennberg, H.; Bäckvall, J.-E. Chem. – Eur. J. 1998, 4, 1083–1089. doi:10.1002/(SICI)1521-3765(19980615)4:6<1083::AID-CHEM1083>3.0.CO;2-F
Return to citation in text: [1] [2] -
Diao, T.; Stahl, S. S. Polyhedron 2014, 84, 96–102. doi:10.1016/j.poly.2014.06.038
Return to citation in text: [1] -
Chen, M. S.; Prabagaran, N.; Labenz, N. A.; White, M. C. J. Am. Chem. Soc. 2005, 127, 6970–6971. doi:10.1021/ja0500198
Return to citation in text: [1] [2] [3] [4] -
Chen, M. S.; White, M. C. J. Am. Chem. Soc. 2004, 126, 1346–1347. doi:10.1021/ja039107n
Return to citation in text: [1] [2] [3] -
Covell, D. J.; White, M. C. Angew. Chem., Int. Ed. 2008, 47, 6448–6451. doi:10.1002/anie.200802106
Return to citation in text: [1] -
Campbell, A. N.; White, P. B.; Guzei, I. A.; Stahl, S. S. J. Am. Chem. Soc. 2010, 132, 15116–15119. doi:10.1021/ja105829t
Return to citation in text: [1] -
Mitsudome, T.; Umetani, T.; Nosaka, N.; Mori, K.; Mizugaki, T.; Ebitani, K.; Kaneda, K. Angew. Chem., Int. Ed. 2006, 45, 481–485. doi:10.1002/anie.200502886
Return to citation in text: [1] [2] -
Vermeulen, N. A.; Delcamp, J. H.; White, M. C. J. Am. Chem. Soc. 2010, 132, 11323–11328. doi:10.1021/ja104826g
Return to citation in text: [1] -
Thiery, E.; Aouf, C.; Belloy, J.; Harakat, D.; Le Bras, J.; Muzart, J. J. Org. Chem. 2010, 75, 1771–1774. doi:10.1021/jo902587u
Return to citation in text: [1] -
Check, C. T.; Henderson, W. H.; Wray, B. C.; Vanden Eynden, M. J.; Stambuli, J. P. J. Am. Chem. Soc. 2011, 133, 18503–18505. doi:10.1021/ja2089102
Return to citation in text: [1] -
Macsári, I.; Szabó, K. J. Tetrahedron Lett. 1998, 39, 6345–6348. doi:10.1016/S0040-4039(98)01304-5
Return to citation in text: [1] -
García-Cabeza, A. L.; Marín-Barrios, R.; Moreno-Dorado, F. J.; Ortega, M. J.; Massanet, G. M.; Guerra, F. M. Org. Lett. 2014, 16, 1598–1601. doi:10.1021/ol500198c
Return to citation in text: [1] -
Takemura, N.; Kuninobu, Y.; Kanai, M. Org. Biomol. Chem. 2014, 12, 2528–2532. doi:10.1039/C4OB00215F
Return to citation in text: [1] -
Rout, S. K.; Guin, S.; Banerjee, A.; Khatun, N.; Gogoi, A.; Patel, B. K. Org. Lett. 2013, 15, 4106–4109. doi:10.1021/ol401682a
Return to citation in text: [1] -
Rout, S. K.; Guin, S.; Ali, W.; Gogoi, A.; Patel, B. K. Org. Lett. 2014, 16, 3086–3089. doi:10.1021/ol5011906
Return to citation in text: [1] [2] -
Boldron, C.; Gamez, P.; Tooke, D. M.; Spek, A. L.; Reedijk, J. Angew. Chem., Int. Ed. 2005, 44, 3585–3587. doi:10.1002/anie.200462946
Return to citation in text: [1] -
Boldron, C.; Özalp-Yaman, Ş.; Gamez, P.; Tooke, D. M.; Spek, A. L.; Reedijk, J. Dalton Trans. 2005, 3535–3541. doi:10.1039/B507199B
Return to citation in text: [1] -
Liu, H.; Shi, G.; Pan, S.; Jiang, Y.; Zhang, Y. Org. Lett. 2013, 15, 4098–4101. doi:10.1021/ol401687f
Return to citation in text: [1] [2] -
Benhmid, A.; Narayana, K. V.; Martin, A.; Lücke, B. Chem. Commun. 2004, 2118–2119. doi:10.1039/B406396A
Return to citation in text: [1] -
Kalevaru, V. N.; Benhmid, A.; Radnik, J.; Pohl, M.-M.; Lücke, B.; Martin, A. Catal. Today 2009, 141, 317–324. doi:10.1016/j.cattod.2008.06.004
Return to citation in text: [1] -
Gatla, S.; Madaan, N.; Radnik, J.; Kalevaru, V. N.; Pohl, M.-M.; Lücke, B.; Martin, A.; Brückner, A. Appl. Catal., A 2011, 398, 104–112. doi:10.1016/j.apcata.2011.03.025
Return to citation in text: [1] -
Madaan, N.; Gatla, S.; Kalevaru, V. N.; Radnik, J.; Pohl, M.-M.; Lücke, B.; Brückner, A.; Martin, A. ChemCatChem 2013, 5, 185–191. doi:10.1002/cctc.201200522
Return to citation in text: [1] -
Shi, E.; Shao, Y.; Chen, S.; Hu, H.; Liu, Z.; Zhang, J.; Wan, X. Org. Lett. 2012, 14, 3384–3387. doi:10.1021/ol3013606
Return to citation in text: [1] [2] [3] -
Majji, G.; Guin, S.; Gogoi, A.; Rout, S. K.; Patel, B. K. Chem. Commun. 2013, 49, 3031–3033. doi:10.1039/C3CC40832A
Return to citation in text: [1] -
Xu, J.; Zhang, P.; Li, X.; Gao, Y.; Wu, J.; Tang, G.; Zhao, Y. Adv. Synth. Catal. 2014, 356, 3331–3335. doi:10.1002/adsc.201400436
Return to citation in text: [1] -
Zaimoku, H.; Hatta, T.; Taniguchi, T.; Ishibashi, H. Org. Lett. 2012, 14, 6088–6091. doi:10.1021/ol302983t
Return to citation in text: [1] -
Kumar, V.; Sharma, A.; Sharma, M.; Sharma, U. K.; Sinha, A. K. Tetrahedron 2007, 63, 9718–9723. doi:10.1016/j.tet.2007.07.018
Return to citation in text: [1] -
Yi, H.; Liu, Q.; Liu, J.; Zeng, Z.; Yang, Y.; Lei, A. ChemSusChem 2012, 5, 2143–2146. doi:10.1002/cssc.201200458
Return to citation in text: [1] -
Aubry, S.; Pellet-Rostaing, S.; Lemaire, M. Eur. J. Org. Chem. 2007, 5212–5225. doi:10.1002/ejoc.200700366
Return to citation in text: [1] -
Estévez, J. C.; Villaverde, M. C.; Estévez, R. J.; Seijas, J. A.; Castedo, L. Synth. Commun. 1990, 20, 503–507. doi:10.1080/00397919008244897
Return to citation in text: [1] -
Guy, A.; Lemor, A.; Imbert, D.; Lemair, M. Tetrahedron Lett. 1989, 30, 327–330. doi:10.1016/S0040-4039(00)95192-X
Return to citation in text: [1] -
Li, Y.; Bao, W. Adv. Synth. Catal. 2009, 351, 865–868. doi:10.1002/adsc.200800810
Return to citation in text: [1] -
Mo, H.; Bao, W. Tetrahedron 2011, 67, 4793–4799. doi:10.1016/j.tet.2011.05.030
Return to citation in text: [1] -
Wang, T.; Zhou, W.; Yin, H.; Ma, J.-A.; Jiao, N. Angew. Chem., Int. Ed. 2012, 51, 10823–10826. doi:10.1002/anie.201205779
Return to citation in text: [1] -
Jin, J.; Li, Y.; Wang, Z.-j.; Qian, W.-x.; Bao, W.-l. Eur. J. Org. Chem. 2010, 1235–1238. doi:10.1002/ejoc.200901321
Return to citation in text: [1] [2] -
He, H.-F.; Wang, K.; Xing, B.; Sheng, G.; Ma, T.; Bao, W. Synlett 2013, 24, 211–214. doi:10.1055/s-0032-1317960
Return to citation in text: [1] [2] [3] -
Bouquet, M.; Guy, A.; Lemaire, M.; Guetté, J. P. Synth. Commun. 1985, 15, 1153–1157. doi:10.1080/00397918508077258
Return to citation in text: [1] -
Lee, J. M.; Park, E. J.; Cho, S. H.; Chang, S. J. Am. Chem. Soc. 2008, 130, 7824–7825. doi:10.1021/ja8031218
Return to citation in text: [1] [2] [3] -
Terent’ev, A. O.; Krylov, I. B.; Sharipov, M. Y.; Kazanskaya, Z. M.; Nikishin, G. I. Tetrahedron 2012, 68, 10263–10271. doi:10.1016/j.tet.2012.10.018
Return to citation in text: [1] -
Koshino, N.; Saha, B.; Espenson, J. H. J. Org. Chem. 2003, 68, 9364–9370. doi:10.1021/jo0348017
Return to citation in text: [1] -
Terent’ev, A. O.; Krylov, I. B.; Lipatnikov, A. D. Russ. J. Gen. Chem. 2014, 84, 2084–2087. doi:10.1134/S1070363214110061
Zhurnal Obschei Khimii 2014, 84, 1786-1789.
Return to citation in text: [1] -
Coseri, S. Eur. J. Org. Chem. 2007, 1725–1729. doi:10.1002/ejoc.200601072
Return to citation in text: [1] -
Coseri, S. J. Phys. Org. Chem. 2009, 22, 397–402. doi:10.1002/poc.1466
Return to citation in text: [1] -
Minisci, F.; Recupero, F.; Gambarotti, C.; Punta, C.; Paganelli, R. Tetrahedron Lett. 2003, 44, 6919–6922. doi:10.1016/S0040-4039(03)01687-3
Return to citation in text: [1] -
Khan, K. M.; Maharvi, G. M.; Hayat, S.; Zia-Ullah; Choudhary, M. I.; Atta-ur-Rahman. Tetrahedron 2003, 59, 5549–5554. doi:10.1016/S0040-4020(03)00812-3
Return to citation in text: [1] -
Iwata, S.; Hata, T.; Urabe, H. Adv. Synth. Catal. 2012, 354, 3480–3484. doi:10.1002/adsc.201200410
Return to citation in text: [1] -
Macedo, A. G.; Fernandes, S. E. M.; Valente, A. A.; Ferreira, R. A. S.; Carlos, L. D.; Rocha, J. Molecules 2010, 15, 747–765. doi:10.3390/molecules15020747
Return to citation in text: [1] -
Akhlaghinia, B.; Ebrahimabadi, H.; Goharshadi, E. K.; Samiee, S.; Rezazadeh, S. J. Mol. Catal. A: Chem. 2012, 357, 67–72. doi:10.1016/j.molcata.2012.01.020
Return to citation in text: [1] -
Shaikh, T. M.; Sudalai, A. Tetrahedron Lett. 2005, 46, 5589–5592. doi:10.1016/j.tetlet.2005.06.033
Return to citation in text: [1] -
Khenkin, A. M.; Neumann, R. J. Am. Chem. Soc. 2004, 126, 6356–6362. doi:10.1021/ja031710i
Return to citation in text: [1] -
Occhipinti, G.; Liguori, L.; Tsoukala, A.; Bjørsvik, H.-R. Org. Process Res. Dev. 2010, 14, 1379–1384. doi:10.1021/op100185x
Return to citation in text: [1] -
Cho, S.-D.; Kim, H.-J.; Ahn, C.; Falck, J. R.; Shin, D.-S. Tetrahedron Lett. 1999, 40, 8215–8217. doi:10.1016/S0040-4039(99)01713-X
Return to citation in text: [1] -
Iwaoka, M.; Tomoda, S. J. Chem. Soc., Chem. Commun. 1992, 1165–1167. doi:10.1039/c39920001165
Return to citation in text: [1] [2] [3] -
Tiecco, M.; Testaferri, L.; Tingoli, M.; Bagnoli, L.; Santi, C. J. Chem. Soc., Chem. Commun. 1993, 637–639. doi:10.1039/C39930000637
Return to citation in text: [1] [2] [3] -
Shibuya, K. Synth. Commun. 1994, 24, 2923–2941. doi:10.1080/00397919408010614
Return to citation in text: [1] [2] [3] -
Chen, L.; Shi, E.; Liu, Z.; Chen, S.; Wei, W.; Li, H.; Xu, K.; Wan, X. Chem. – Eur. J. 2011, 17, 4085–4089. doi:10.1002/chem.201100192
Return to citation in text: [1] -
Zhang, S.; Guo, L.-N.; Wang, H.; Duan, X.-H. Org. Biomol. Chem. 2013, 11, 4308–4311. doi:10.1039/C3OB40748A
Return to citation in text: [1] -
Yu, H.; Shen, J. Org. Lett. 2014, 16, 3204–3207. doi:10.1021/ol5012168
Return to citation in text: [1] -
Zhao, J.; Fang, H.; Zhou, W.; Han, J.; Pan, Y. J. Org. Chem. 2014, 79, 3847–3855. doi:10.1021/jo500192h
Return to citation in text: [1] -
Barve, B. D.; Wu, Y.-C.; El-Shazly, M.; Korinek, M.; Cheng, Y.-B.; Wang, J.-J.; Chang, F.-R. Org. Lett. 2014, 16, 1912–1915. doi:10.1021/ol5004115
Return to citation in text: [1] -
Park, J.; Han, S. H.; Sharma, S.; Han, S.; Shin, Y.; Mishra, N. K.; Kwak, J. H.; Lee, C. H.; Lee, J.; Kim, I. S. J. Org. Chem. 2014, 79, 4735–4742. doi:10.1021/jo500576x
Return to citation in text: [1] -
Phan, N. T. S.; Vu, P. H. L.; Nguyen, T. T. J. Catal. 2013, 306, 38–46. doi:10.1016/j.jcat.2013.06.006
Return to citation in text: [1] -
Murahash, S.-I.; Saito, T.; Naota, T.; Kumobayashi, H.; Akutagawa, S. Tetrahedron Lett. 1991, 32, 5991–5994. doi:10.1016/S0040-4039(00)79446-9
Return to citation in text: [1] -
Murahashi, S.; Naota, T.; Kuwabara, T.; Saito, T.; Kumobayashi, H.; Akutagawa, S. J. Am. Chem. Soc. 1990, 112, 7820–7822. doi:10.1021/ja00177a067
Return to citation in text: [1] -
Shu, X.-Z.; Xia, X.-F.; Yang, Y.-F.; Ji, K.-G.; Liu, X.-Y.; Liang, Y.-M. J. Org. Chem. 2009, 74, 7464–7469. doi:10.1021/jo901583r
Return to citation in text: [1] -
Weinberg, N. L.; Brown, E. A. J. Org. Chem. 1966, 31, 4058–4061. doi:10.1021/jo01350a041
Return to citation in text: [1] -
Liang, D.; Wang, M.; Dong, Y.; Guo, Y.; Liu, Q. RSC Adv. 2014, 4, 6564–6567. doi:10.1039/C3RA47282E
Return to citation in text: [1] -
Liu, X.; Cheng, R.; Zhao, F.; Zhang-Negrerie, D.; Du, Y.; Zhao, K. Org. Lett. 2012, 14, 5480–5483. doi:10.1021/ol3025583
Return to citation in text: [1] -
Liu, H.; Wang, X.; Gu, Y. Org. Biomol. Chem. 2011, 9, 1614–1620. doi:10.1039/C0OB00749H
Return to citation in text: [1] [2] -
Liu, H.; Xie, Y.; Gu, Y. Tetrahedron Lett. 2011, 52, 4324–4326. doi:10.1016/j.tetlet.2011.06.041
Return to citation in text: [1] -
Jiang, Q.; Wang, J.-Y.; Guo, C. J. Org. Chem. 2014, 79, 8768–8773. doi:10.1021/jo501601u
Return to citation in text: [1] -
Pelter, A.; Elgendy, S. M. A. J. Chem. Soc., Perkin Trans. 1 1993, 1891–1896. doi:10.1039/P19930001891
Return to citation in text: [1] -
Fleck, A. E.; Hobart, J. A.; Morrow, G. W. Synth. Commun. 1992, 22, 179–187. doi:10.1080/00397919208021090
Return to citation in text: [1] -
Mitchell, A. S.; Russell, R. A. Tetrahedron Lett. 1993, 34, 545–548. doi:10.1016/0040-4039(93)85123-E
Return to citation in text: [1] -
Omura, K. Synthesis 2010, 208–210. doi:10.1055/s-0029-1217127
Return to citation in text: [1] -
Chen, X.; Zhu, C.; Cui, X.; Wu, Y. Chem. Commun. 2013, 49, 6900–6902. doi:10.1039/c3cc43947j
Return to citation in text: [1] -
Takemura, N.; Kuninobu, Y.; Kanai, M. Org. Lett. 2013, 15, 844–847. doi:10.1021/ol303533z
Return to citation in text: [1] -
Halper, S. R.; Stork, J. R.; Cohen, S. M. Dalton Trans. 2007, 1067–1074. doi:10.1039/B615801C
Return to citation in text: [1] -
Takuwa, A.; Soga, O.; Iwamoto, H.; Maruyama, K. Bull. Chem. Soc. Jpn. 1986, 59, 2959–2961. doi:10.1246/bcsj.59.2959
Return to citation in text: [1] -
Kidwai, M.; Kumar, P.; Kohli, S. J. Chem. Res., Synop. 1997, 24–25. doi:10.1039/A601810F
Return to citation in text: [1] -
Bansal, V.; Sharma, J.; Khanna, R. N. J. Chem. Res., Synop. 1998, 720–721. doi:10.1039/A803513J
Return to citation in text: [1] -
Komiya, N.; Noji, S.; Murahashi, S.-I. Chem. Commun. 2001, 65–66. doi:10.1039/B006869L
Return to citation in text: [1] [2] -
Nomura, K.; Uemura, S. J. Chem. Soc., Chem. Commun. 1994, 129–130. doi:10.1039/C39940000129
Return to citation in text: [1] [2] -
Deno, N. C.; Messer, L. A. J. Chem. Soc., Chem. Commun. 1976, 1051a. doi:10.1039/C3976001051A
Return to citation in text: [1] [2] -
Moody, C. J.; O’Connell, J. L. Chem. Commun. 2000, 1311–1312. doi:10.1039/B003074K
Return to citation in text: [1] [2] -
Tran, B. L.; Driess, M.; Hartwig, J. F. J. Am. Chem. Soc. 2014, 136, 17292–17301. doi:10.1021/ja510093x
Return to citation in text: [1] -
Gary, J. B.; Cook, A. K.; Sanford, M. S. ACS Catal. 2013, 3, 700–703. doi:10.1021/cs300786j
Return to citation in text: [1] -
Tajima, T.; Fuchigami, T. Chem. – Eur. J. 2005, 11, 6192–6196. doi:10.1002/chem.200500340
Return to citation in text: [1]
| 267. | Iwaoka, M.; Tomoda, S. J. Chem. Soc., Chem. Commun. 1992, 1165–1167. doi:10.1039/c39920001165 |
| 269. | Shibuya, K. Synth. Commun. 1994, 24, 2923–2941. doi:10.1080/00397919408010614 |
| 85. | Wu, X.; Zhao, Y.; Ge, H. Chem. – Asian J. 2014, 9, 2736–2739. doi:10.1002/asia.201402510 |
| 83. | Rit, R. K.; Yadav, M. R.; Sahoo, A. K. Org. Lett. 2012, 14, 3724–3727. doi:10.1021/ol301579q |
| 174. | Mineno, T.; Sakai, M.; Ubukata, A.; Nakahara, K.; Yoshimitsu, H.; Kansui, H. Chem. Pharm. Bull. 2013, 61, 870–872. doi:10.1248/cpb.c13-00072 |
| 175. | Mineno, T.; Yoshino, S.; Ubukata, A. Green Sustainable Chem. 2014, 4, 20–23. doi:10.4236/gsc.2014.41004 |
| 173. | Sundararaman, P.; Walker, E. C.; Djerassi, C. Tetrahedron Lett. 1978, 19, 1627–1628. doi:10.1016/S0040-4039(01)94623-4 |
| 275. | Park, J.; Han, S. H.; Sharma, S.; Han, S.; Shin, Y.; Mishra, N. K.; Kwak, J. H.; Lee, C. H.; Lee, J.; Kim, I. S. J. Org. Chem. 2014, 79, 4735–4742. doi:10.1021/jo500576x |
| 170. | O'Connor, B.; Just, G. Tetrahedron Lett. 1987, 28, 3235–3236. doi:10.1016/S0040-4039(00)95480-7 |
| 169. | Zhao, J.; Fang, H.; Han, J.; Pan, Y. Org. Lett. 2014, 16, 2530–2533. doi:10.1021/ol5009119 |
| 271. | Zhang, S.; Guo, L.-N.; Wang, H.; Duan, X.-H. Org. Biomol. Chem. 2013, 11, 4308–4311. doi:10.1039/C3OB40748A |
| 88. | Giri, R.; Liang, J.; Lei, J.-G.; Li, J.-J.; Wang, D.-H.; Chen, X.; Naggar, I. C.; Guo, C.; Foxman, B. M.; Yu, J.-Q. Angew. Chem., Int. Ed. 2005, 44, 7420–7424. doi:10.1002/anie.200502767 |
| 172. | Sakuragi, H.; Tokumaru, K. Chem. Lett. 1974, 3, 475–476. doi:10.1246/cl.1974.475 |
| 274. | Barve, B. D.; Wu, Y.-C.; El-Shazly, M.; Korinek, M.; Cheng, Y.-B.; Wang, J.-J.; Chang, F.-R. Org. Lett. 2014, 16, 1912–1915. doi:10.1021/ol5004115 |
| 171. | Sinha, A. K.; Sharma, A.; Swaroop, A.; Kumar, V. Tetrahedron 2007, 63, 1000–1007. doi:10.1016/j.tet.2006.11.011 |
| 273. | Zhao, J.; Fang, H.; Zhou, W.; Han, J.; Pan, Y. J. Org. Chem. 2014, 79, 3847–3855. doi:10.1021/jo500192h |
| 86. | Desai, L. V.; Hull, K. L.; Sanford, M. S. J. Am. Chem. Soc. 2004, 126, 9542–9543. doi:10.1021/ja046831c |
| 166. | Sharma, R. K.; Gulati, S. J. Mol. Catal. A: Chem. 2012, 363–364, 291–303. doi:10.1016/j.molcata.2012.07.004 |
| 269. | Shibuya, K. Synth. Commun. 1994, 24, 2923–2941. doi:10.1080/00397919408010614 |
| 87. | Stowers, K. J.; Kubota, A.; Sanford, M. S. Chem. Sci. 2012, 3, 3192–3195. doi:10.1039/C2SC20800H |
| 165. | Rafiee, E.; Eavani, S. J. Mol. Catal. A: Chem. 2013, 373, 30–37. doi:10.1016/j.molcata.2013.02.024 |
| 267. | Iwaoka, M.; Tomoda, S. J. Chem. Soc., Chem. Commun. 1992, 1165–1167. doi:10.1039/c39920001165 |
| 268. | Tiecco, M.; Testaferri, L.; Tingoli, M.; Bagnoli, L.; Santi, C. J. Chem. Soc., Chem. Commun. 1993, 637–639. doi:10.1039/C39930000637 |
| 56. | Neufeldt, S. R.; Sanford, M. S. Org. Lett. 2010, 12, 532–535. doi:10.1021/ol902720d |
| 168. | Wu, X.-F. Tetrahedron Lett. 2012, 53, 3397–3399. doi:10.1016/j.tetlet.2012.04.111 |
| 270. | Chen, L.; Shi, E.; Liu, Z.; Chen, S.; Wei, W.; Li, H.; Xu, K.; Wan, X. Chem. – Eur. J. 2011, 17, 4085–4089. doi:10.1002/chem.201100192 |
| 54. | Desai, L. V.; Malik, H. A.; Sanford, M. S. Org. Lett. 2006, 8, 1141–1144. doi:10.1021/ol0530272 |
| 86. | Desai, L. V.; Hull, K. L.; Sanford, M. S. J. Am. Chem. Soc. 2004, 126, 9542–9543. doi:10.1021/ja046831c |
| 167. | Esfandiari, H.; Jameh-bozorghi, S.; Esmaielzadeh, S.; Shafiee, M. R. M.; Ghashang, M. Res. Chem. Intermed. 2013, 39, 3319–3325. doi:10.1007/s11164-012-0844-y |
| 268. | Tiecco, M.; Testaferri, L.; Tingoli, M.; Bagnoli, L.; Santi, C. J. Chem. Soc., Chem. Commun. 1993, 637–639. doi:10.1039/C39930000637 |
| 269. | Shibuya, K. Synth. Commun. 1994, 24, 2923–2941. doi:10.1080/00397919408010614 |
| 174. | Mineno, T.; Sakai, M.; Ubukata, A.; Nakahara, K.; Yoshimitsu, H.; Kansui, H. Chem. Pharm. Bull. 2013, 61, 870–872. doi:10.1248/cpb.c13-00072 |
| 176. | Travis, B. R.; Sivakumar, M.; Hollist, G. O.; Borhan, B. Org. Lett. 2003, 5, 1031–1034. doi:10.1021/ol0340078 |
| 94. | Heropoulos, G. A.; Villalonga-Barber, C. Tetrahedron Lett. 2011, 52, 5319–5322. doi:10.1016/j.tetlet.2011.08.021 |
| 95. | Liu, C.; Tang, S.; Zheng, L.; Liu, D.; Zhang, H.; Lei, A. Angew. Chem., Int. Ed. 2012, 51, 5662–5666. doi:10.1002/anie.201201960 |
| 92. | Yamamoto, N.; Obora, Y.; Ishii, Y. J. Org. Chem. 2011, 76, 2937–2941. doi:10.1021/jo2003264 |
| 93. | Tschaen, B. A.; Schmink, J. R.; Molander, G. A. Org. Lett. 2013, 15, 500–503. doi:10.1021/ol303298g |
| 89. | Wang, Z.; Kuninobu, Y.; Kanai, M. Org. Lett. 2014, 16, 4790–4793. doi:10.1021/ol5022542 |
| 90. | Owston, N. A.; Parker, A. J.; Williams, J. M. J. Chem. Commun. 2008, 624–625. doi:10.1039/B717073D |
| 91. | Owston, N. A.; Nixon, T. D.; Parker, A. J.; Whittlesey, M. K.; Williams, J. M. J. Synthesis 2009, 1578–1581. doi:10.1055/s-0028-1088026 |
| 185. | Yu, J.; Tian, J.; Zhang, C. Adv. Synth. Catal. 2010, 352, 531–546. doi:10.1002/adsc.200900737 |
| 182. | Srimani, D.; Balaraman, E.; Gnanaprakasam, B.; Ben-David, Y.; Milstein, D. Adv. Synth. Catal. 2012, 354, 2403–2406. doi:10.1002/adsc.201200438 |
| 181. | Zhang, C.; Jiao, N. Org. Chem. Front. 2014, 1, 109–112. doi:10.1039/C3QO00041A |
| 184. | Hu, J.; Zhu, M.; Xu, Y.; Yan, J. Synthesis 2012, 1226–1232. doi:10.1055/s-0031-1289750 |
| 183. | Tanaka, A.; Moriyama, K.; Togo, H. Synlett 2011, 1853–1858. doi:10.1055/s-0030-1260948 |
| 97. | Gowrisankar, S.; Neumann, H.; Beller, M. Angew. Chem., Int. Ed. 2011, 50, 5139–5143. doi:10.1002/anie.201008035 |
| 178. | Feng, J.-B.; Gong, J.-L.; Li, Q.; Wu, X.-F. Tetrahedron Lett. 2014, 55, 1657–1659. doi:10.1016/j.tetlet.2014.01.111 |
| 96. | Liu, C.; Wang, J.; Meng, L.; Deng, Y.; Li, Y.; Lei, A. Angew. Chem., Int. Ed. 2011, 50, 5144–5148. doi:10.1002/anie.201008073 |
| 177. | Nishihara, A.; Kubota, I. J. Org. Chem. 1968, 33, 2525–2526. doi:10.1021/jo01270a082 |
| 95. | Liu, C.; Tang, S.; Zheng, L.; Liu, D.; Zhang, H.; Lei, A. Angew. Chem., Int. Ed. 2012, 51, 5662–5666. doi:10.1002/anie.201201960 |
| 180. | Kelly, C. B.; Mercadante, M. A.; Wiles, R. J.; Leadbeater, N. E. Org. Lett. 2013, 15, 2222–2225. doi:10.1021/ol400785d |
| 96. | Liu, C.; Wang, J.; Meng, L.; Deng, Y.; Li, Y.; Lei, A. Angew. Chem., Int. Ed. 2011, 50, 5144–5148. doi:10.1002/anie.201008073 |
| 97. | Gowrisankar, S.; Neumann, H.; Beller, M. Angew. Chem., Int. Ed. 2011, 50, 5139–5143. doi:10.1002/anie.201008035 |
| 179. | Tank, R.; Pathak, U.; Vimal, M.; Bhattacharyya, S.; Pandey, L. K. Green Chem. 2011, 13, 3350–3354. doi:10.1039/C1GC16041A |
| 187. | Moriarty, R. M.; Vaid, R. K.; Ravikumar, V. T.; Vaid, B. K.; Hopkins, T. E. Tetrahedron 1988, 44, 1603–1607. doi:10.1016/S0040-4020(01)86720-X |
| 186. | Yu, J.; Cui, J.; Hou, X.-S.; Liu, S.-S.; Gao, W.-C.; Jiang, S.; Tian, J.; Zhang, C. Tetrahedron: Asymmetry 2011, 22, 2039–2055. doi:10.1016/j.tetasy.2011.12.003 |
| 67. | Hao, X.-Q.; Chen, L.-J.; Ren, B.; Li, L.-Y.; Yang, X.-Y.; Gong, J.-F.; Niu, J.-L.; Song, M.-P. Org. Lett. 2014, 16, 1104–1107. doi:10.1021/ol500166d |
| 68. | Zhang, L.-B.; Hao, X.-Q.; Zhang, S.-K.; Liu, K.; Ren, B.; Gong, J.-F.; Niu, J.-L.; Song, M.-P. J. Org. Chem. 2014, 79, 10399–10409. doi:10.1021/jo502005j |
| 66. | Roane, J.; Daugulis, O. Org. Lett. 2013, 15, 5842–5845. doi:10.1021/ol402904d |
| 192. | Sheng, J.; Li, X.; Tang, M.; Gao, B.; Huang, G. Synthesis 2007, 1165–1168. doi:10.1055/s-2007-965984 |
| 70. | Chernyak, N.; Dudnik, A. S.; Huang, C.; Gevorgyan, V. J. Am. Chem. Soc. 2010, 132, 8270–8272. doi:10.1021/ja1033167 |
| 71. | Huang, C.; Chernyak, N.; Dudnik, A. S.; Gevorgyan, V. Adv. Synth. Catal. 2011, 353, 1285–1305. doi:10.1002/adsc.201000975 |
| 186. | Yu, J.; Cui, J.; Hou, X.-S.; Liu, S.-S.; Gao, W.-C.; Jiang, S.; Tian, J.; Zhang, C. Tetrahedron: Asymmetry 2011, 22, 2039–2055. doi:10.1016/j.tetasy.2011.12.003 |
| 72. | Gulevich, A. V.; Melkonyan, F. S.; Sarkar, D.; Gevorgyan, V. J. Am. Chem. Soc. 2012, 134, 5528–5531. doi:10.1021/ja3010545 |
| 70. | Chernyak, N.; Dudnik, A. S.; Huang, C.; Gevorgyan, V. J. Am. Chem. Soc. 2010, 132, 8270–8272. doi:10.1021/ja1033167 |
| 71. | Huang, C.; Chernyak, N.; Dudnik, A. S.; Gevorgyan, V. Adv. Synth. Catal. 2011, 353, 1285–1305. doi:10.1002/adsc.201000975 |
| 188. | Price, D. A.; Gayton, S.; Stupple, P. A. Synlett 2002, 1170–1172. doi:10.1055/s-2002-32577 |
| 72. | Gulevich, A. V.; Melkonyan, F. S.; Sarkar, D.; Gevorgyan, V. J. Am. Chem. Soc. 2012, 134, 5528–5531. doi:10.1021/ja3010545 |
| 73. | Sarkar, D.; Melkonyan, F. S.; Gulevich, A. V.; Gevorgyan, V. Angew. Chem., Int. Ed. 2013, 52, 10800–10804. doi:10.1002/anie.201304884 |
| 187. | Moriarty, R. M.; Vaid, R. K.; Ravikumar, V. T.; Vaid, B. K.; Hopkins, T. E. Tetrahedron 1988, 44, 1603–1607. doi:10.1016/S0040-4020(01)86720-X |
| 69. | Zhang, L.-B.; Hao, X.-Q.; Zhang, S.-K.; Liu, Z.-J.; Zheng, X.-X.; Gong, J.-F.; Niu, J.-L.; Song, M.-P. Angew. Chem., Int. Ed. 2015, 54, 272–275. doi:10.1002/anie.201409751 |
| 190. | Yamamoto, Y.; Kawano, Y.; Toy, P. H.; Togo, H. Tetrahedron 2007, 63, 4680–4687. doi:10.1016/j.tet.2007.03.091 |
| 69. | Zhang, L.-B.; Hao, X.-Q.; Zhang, S.-K.; Liu, Z.-J.; Zheng, X.-X.; Gong, J.-F.; Niu, J.-L.; Song, M.-P. Angew. Chem., Int. Ed. 2015, 54, 272–275. doi:10.1002/anie.201409751 |
| 189. | Liu, W.-B.; Chen, C.; Zhang, Q.; Zhu, Z.-B. Bull. Korean Chem. Soc. 2012, 33, 1823–1824. doi:10.5012/bkcs.2012.33.6.1823 |
| 76. | Ju, L.; Yao, J.; Wu, Z.; Liu, Z.; Zhang, Y. J. Org. Chem. 2013, 78, 10821–10831. doi:10.1021/jo401830k |
| 77. | Cheng, T.; Yin, W.; Zhang, Y.; Zhang, Y.; Huang, Y. Org. Biomol. Chem. 2014, 12, 1405–1411. doi:10.1039/C3OB42196A |
| 47. | Bhadra, S.; Matheis, C.; Katayev, D.; Gooßen, L. J. Angew. Chem., Int. Ed. 2013, 52, 9279–9283. doi:10.1002/anie.201303702 |
| 75. | Nakano, Y.; Lupton, D. W. Chem. Commun. 2014, 50, 1757–1760. doi:10.1039/C3CC48545E |
| 62. | Zhang, S.-Y.; He, G.; Zhao, Y.; Wright, K.; Nack, W. A.; Chen, G. J. Am. Chem. Soc. 2012, 134, 7313–7316. doi:10.1021/ja3023972 |
| 63. | Chen, F.-J.; Zhao, S.; Hu, F.; Chen, K.; Zhang, Q.; Zhang, S.-Q.; Shi, B.-F. Chem. Sci. 2013, 4, 4187–4192. doi:10.1039/C3SC51993G |
| 80. | Wang, D.; Zavalij, P. Y.; Vedernikov, A. N. Organometallics 2013, 32, 4882–4891. doi:10.1021/om400618n |
| 81. | Shan, G.; Yang, X.; Zong, Y.; Rao, Y. Angew. Chem., Int. Ed. 2013, 52, 13606–13610. doi:10.1002/anie.201307090 |
| 78. | Jiang, H.; Chen, H.; Wang, A.; Liu, X. Chem. Commun. 2010, 46, 7259–7261. doi:10.1039/C0CC00841A |
| 79. | Zhang, S.; Luo, F.; Wang, W.; Jia, X.; Hu, M.; Cheng, J. Tetrahedron Lett. 2010, 51, 3317–3319. doi:10.1016/j.tetlet.2010.04.075 |
| 143. | Wei, W.; Zhang, C.; Xu, Y.; Wan, X. Chem. Commun. 2011, 47, 10827–10829. doi:10.1039/C1CC14602E |
| 142. | Huang, J.; Li, L.-T.; Li, H.-Y.; Husan, E.; Wang, P.; Wang, B. Chem. Commun. 2012, 48, 10204–10206. doi:10.1039/C2CC35450K |
| 240. | Zaimoku, H.; Hatta, T.; Taniguchi, T.; Ishibashi, H. Org. Lett. 2012, 14, 6088–6091. doi:10.1021/ol302983t |
| 141. | Feng, J.; Liang, S.; Chen, S.-Y.; Zhang, J.; Fu, S.-S.; Yu, X.-Q. Adv. Synth. Catal. 2012, 354, 1287–1292. doi:10.1002/adsc.201100920 |
| 142. | Huang, J.; Li, L.-T.; Li, H.-Y.; Husan, E.; Wang, P.; Wang, B. Chem. Commun. 2012, 48, 10204–10206. doi:10.1039/C2CC35450K |
| 140. | Liu, L.; Yun, L.; Wang, Z.; Fu, X.; Yan, C. Tetrahedron Lett. 2013, 54, 5383–5386. doi:10.1016/j.tetlet.2013.07.114 |
| 140. | Liu, L.; Yun, L.; Wang, Z.; Fu, X.; Yan, C. Tetrahedron Lett. 2013, 54, 5383–5386. doi:10.1016/j.tetlet.2013.07.114 |
| 142. | Huang, J.; Li, L.-T.; Li, H.-Y.; Husan, E.; Wang, P.; Wang, B. Chem. Commun. 2012, 48, 10204–10206. doi:10.1039/C2CC35450K |
| 239. | Xu, J.; Zhang, P.; Li, X.; Gao, Y.; Wu, J.; Tang, G.; Zhao, Y. Adv. Synth. Catal. 2014, 356, 3331–3335. doi:10.1002/adsc.201400436 |
| 140. | Liu, L.; Yun, L.; Wang, Z.; Fu, X.; Yan, C. Tetrahedron Lett. 2013, 54, 5383–5386. doi:10.1016/j.tetlet.2013.07.114 |
| 238. | Majji, G.; Guin, S.; Gogoi, A.; Rout, S. K.; Patel, B. K. Chem. Commun. 2013, 49, 3031–3033. doi:10.1039/C3CC40832A |
| 140. | Liu, L.; Yun, L.; Wang, Z.; Fu, X.; Yan, C. Tetrahedron Lett. 2013, 54, 5383–5386. doi:10.1016/j.tetlet.2013.07.114 |
| 141. | Feng, J.; Liang, S.; Chen, S.-Y.; Zhang, J.; Fu, S.-S.; Yu, X.-Q. Adv. Synth. Catal. 2012, 354, 1287–1292. doi:10.1002/adsc.201100920 |
| 237. | Shi, E.; Shao, Y.; Chen, S.; Hu, H.; Liu, Z.; Zhang, J.; Wan, X. Org. Lett. 2012, 14, 3384–3387. doi:10.1021/ol3013606 |
| 139. | Ghorbani-Vaghei, R.; Shahbazee, E.; Veisi, H. Mendeleev Commun. 2005, 15, 207–208. doi:10.1070/MC2005v015n05ABEH002091 |
| 237. | Shi, E.; Shao, Y.; Chen, S.; Hu, H.; Liu, Z.; Zhang, J.; Wan, X. Org. Lett. 2012, 14, 3384–3387. doi:10.1021/ol3013606 |
| 141. | Feng, J.; Liang, S.; Chen, S.-Y.; Zhang, J.; Fu, S.-S.; Yu, X.-Q. Adv. Synth. Catal. 2012, 354, 1287–1292. doi:10.1002/adsc.201100920 |
| 141. | Feng, J.; Liang, S.; Chen, S.-Y.; Zhang, J.; Fu, S.-S.; Yu, X.-Q. Adv. Synth. Catal. 2012, 354, 1287–1292. doi:10.1002/adsc.201100920 |
| 140. | Liu, L.; Yun, L.; Wang, Z.; Fu, X.; Yan, C. Tetrahedron Lett. 2013, 54, 5383–5386. doi:10.1016/j.tetlet.2013.07.114 |
| 141. | Feng, J.; Liang, S.; Chen, S.-Y.; Zhang, J.; Fu, S.-S.; Yu, X.-Q. Adv. Synth. Catal. 2012, 354, 1287–1292. doi:10.1002/adsc.201100920 |
| 237. | Shi, E.; Shao, Y.; Chen, S.; Hu, H.; Liu, Z.; Zhang, J.; Wan, X. Org. Lett. 2012, 14, 3384–3387. doi:10.1021/ol3013606 |
| 149. | Yao, H.; Tang, Y.; Yamamoto, K. Tetrahedron Lett. 2012, 53, 5094–5098. doi:10.1016/j.tetlet.2012.07.024 |
| 147. | Wang, N.; Liu, R.; Xu, Q.; Liang, X. Chem. Lett. 2006, 35, 566–567. doi:10.1246/cl.2006.566 |
| 248. | Wang, T.; Zhou, W.; Yin, H.; Ma, J.-A.; Jiao, N. Angew. Chem., Int. Ed. 2012, 51, 10823–10826. doi:10.1002/anie.201205779 |
| 148. | Schulze, A.; Giannis, A. Adv. Synth. Catal. 2004, 346, 252–256. doi:10.1002/adsc.200303195 |
| 247. | Mo, H.; Bao, W. Tetrahedron 2011, 67, 4793–4799. doi:10.1016/j.tet.2011.05.030 |
| 149. | Yao, H.; Tang, Y.; Yamamoto, K. Tetrahedron Lett. 2012, 53, 5094–5098. doi:10.1016/j.tetlet.2012.07.024 |
| 250. | He, H.-F.; Wang, K.; Xing, B.; Sheng, G.; Ma, T.; Bao, W. Synlett 2013, 24, 211–214. doi:10.1055/s-0032-1317960 |
| 148. | Schulze, A.; Giannis, A. Adv. Synth. Catal. 2004, 346, 252–256. doi:10.1002/adsc.200303195 |
| 249. | Jin, J.; Li, Y.; Wang, Z.-j.; Qian, W.-x.; Bao, W.-l. Eur. J. Org. Chem. 2010, 1235–1238. doi:10.1002/ejoc.200901321 |
| 146. | Wang, G.; Yu, Q.-Y.; Wang, J.; Wang, S.; Chen, S.-Y.; Yu, X.-Q. RSC Adv. 2013, 3, 21306–21310. doi:10.1039/C3RA43799J |
| 244. | Estévez, J. C.; Villaverde, M. C.; Estévez, R. J.; Seijas, J. A.; Castedo, L. Synth. Commun. 1990, 20, 503–507. doi:10.1080/00397919008244897 |
| 145. | Tan, B.; Toda, N.; Barbas, C. F., III. Angew. Chem., Int. Ed. 2012, 51, 12538–12541. doi:10.1002/anie.201205921 |
| 243. | Aubry, S.; Pellet-Rostaing, S.; Lemaire, M. Eur. J. Org. Chem. 2007, 5212–5225. doi:10.1002/ejoc.200700366 |
| 147. | Wang, N.; Liu, R.; Xu, Q.; Liang, X. Chem. Lett. 2006, 35, 566–567. doi:10.1246/cl.2006.566 |
| 246. | Li, Y.; Bao, W. Adv. Synth. Catal. 2009, 351, 865–868. doi:10.1002/adsc.200800810 |
| 146. | Wang, G.; Yu, Q.-Y.; Wang, J.; Wang, S.; Chen, S.-Y.; Yu, X.-Q. RSC Adv. 2013, 3, 21306–21310. doi:10.1039/C3RA43799J |
| 245. | Guy, A.; Lemor, A.; Imbert, D.; Lemair, M. Tetrahedron Lett. 1989, 30, 327–330. doi:10.1016/S0040-4039(00)95192-X |
| 144. | Zhu, F.; Wang, Z.-X. Tetrahedron 2014, 70, 9819–9827. doi:10.1016/j.tet.2014.11.002 |
| 242. | Yi, H.; Liu, Q.; Liu, J.; Zeng, Z.; Yang, Y.; Lei, A. ChemSusChem 2012, 5, 2143–2146. doi:10.1002/cssc.201200458 |
| 241. | Kumar, V.; Sharma, A.; Sharma, M.; Sharma, U. K.; Sinha, A. K. Tetrahedron 2007, 63, 9718–9723. doi:10.1016/j.tet.2007.07.018 |
| 104. | Noonan, C.; Baragwanath, L.; Connon, S. J. Tetrahedron Lett. 2008, 49, 4003–4006. doi:10.1016/j.tetlet.2008.04.097 |
| 105. | Maki, B. E.; Scheidt, K. A. Org. Lett. 2008, 10, 4331–4334. doi:10.1021/ol8018488 |
| 106. | Maki, B. E.; Chan, A.; Phillips, E. M.; Scheidt, K. A. Tetrahedron 2009, 65, 3102–3109. doi:10.1016/j.tet.2008.10.033 |
| 107. | Reddy, R. S.; Rosa, J. N.; Veiros, L. F.; Caddick, S.; Gois, P. M. P. Org. Biomol. Chem. 2011, 9, 3126–3129. doi:10.1039/C1OB05151B |
| 108. | Finney, E. E.; Ogawa, K. A.; Boydston, A. J. J. Am. Chem. Soc. 2012, 134, 12374–12377. doi:10.1021/ja304716r |
| 109. | Kiran, I. N. C.; Lalwani, K.; Sudalai, A. RSC Adv. 2013, 3, 1695–1698. doi:10.1039/C2RA22718E |
| 110. | Zhao, J.; Mück-Lichtenfeld, C.; Studer, A. Adv. Synth. Catal. 2013, 355, 1098–1106. doi:10.1002/adsc.201300034 |
| 111. | Chiarotto, I.; Feroci, M.; Sotgiu, G.; Inesi, A. Tetrahedron 2013, 69, 8088–8095. doi:10.1016/j.tet.2013.06.014 |
| 112. | Ji, M.; Wang, X.; Lim, Y. N.; Kang, Y.-W.; Jang, H.-Y. Eur. J. Org. Chem. 2013, 7881–7885. doi:10.1002/ejoc.201301337 |
| 113. | Maki, B. E.; Chan, A.; Phillips, E. M.; Scheidt, K. A. Org. Lett. 2007, 9, 371–374. doi:10.1021/ol062940f |
| 114. | Delany, E. G.; Fagan, C.-L.; Gundala, S.; Zeitler, K.; Connon, S. J. Chem. Commun. 2013, 49, 6513–6515. doi:10.1039/C3CC42597E |
| 102. | Yasukawa, T.; Miyamura, H.; Kobayashi, S. Chem. – Asian J. 2011, 6, 621–627. doi:10.1002/asia.201000624 |
| 103. | Suzuki, K.; Yamaguchi, T.; Matsushita, K.; Iitsuka, C.; Miura, J.; Akaogi, T.; Ishida, H. ACS Catal. 2013, 3, 1845–1849. doi:10.1021/cs4004084 |
| 98. | Nielsen, I. S.; Taarning, E.; Egeblad, K.; Madsen, R.; Christensen, C. H. Catal. Lett. 2007, 116, 35–40. doi:10.1007/s10562-007-9086-9 |
| 99. | Marsden, C.; Taarning, E.; Hansen, D.; Johansen, L.; Klitgaard, S. K.; Egeblad, K.; Christensen, C. H. Green Chem. 2008, 10, 168–170. doi:10.1039/B712171G |
| 100. | Fristrup, P.; Johansen, L. B.; Christensen, C. H. Chem. Commun. 2008, 2750–2752. doi:10.1039/B803270J |
| 153. | Sharma, S.; Park, J.; Kim, M.; Kwak, J. H.; Jung, Y. H.; Kim, I. S. Tetrahedron 2013, 69, 9391–9397. doi:10.1016/j.tet.2013.08.079 |
| 101. | Su, F.-Z.; Ni, J.; Sun, H.; Cao, Y.; He, H.-Y.; Fa, K.-N. Chem. – Eur. J. 2008, 14, 7131–7135. doi:10.1002/chem.200800982 |
| 156. | Barve, B. D.; Wu, Y.-C.; El-Shazly, M.; Chuang, D.-W.; Chung, Y.-M.; Tsai, Y.-H.; Wu, S.-F.; Korinek, M.; Du, Y.-C.; Hsieh, C.-T.; Wang, J.-J.; Chang, F.-R. Eur. J. Org. Chem. 2012, 6760–6766. doi:10.1002/ejoc.201201160 |
| 255. |
Terent’ev, A. O.; Krylov, I. B.; Lipatnikov, A. D. Russ. J. Gen. Chem. 2014, 84, 2084–2087. doi:10.1134/S1070363214110061
Zhurnal Obschei Khimii 2014, 84, 1786-1789. |
| 158. | Saberi, D.; Heydari, A. Tetrahedron Lett. 2013, 54, 4178–4180. doi:10.1016/j.tetlet.2013.05.113 |
| 96. | Liu, C.; Wang, J.; Meng, L.; Deng, Y.; Li, Y.; Lei, A. Angew. Chem., Int. Ed. 2011, 50, 5144–5148. doi:10.1002/anie.201008073 |
| 97. | Gowrisankar, S.; Neumann, H.; Beller, M. Angew. Chem., Int. Ed. 2011, 50, 5139–5143. doi:10.1002/anie.201008035 |
| 157. | Barve, B. D.; Wu, Y.-C.; El-Shazly, M.; Chuang, D.-W.; Cheng, Y.-B.; Wang, J.-J.; Chang, F.-R. J. Org. Chem. 2014, 79, 3206–3214. doi:10.1021/jo402798k |
| 153. | Sharma, S.; Park, J.; Kim, M.; Kwak, J. H.; Jung, Y. H.; Kim, I. S. Tetrahedron 2013, 69, 9391–9397. doi:10.1016/j.tet.2013.08.079 |
| 253. | Terent’ev, A. O.; Krylov, I. B.; Sharipov, M. Y.; Kazanskaya, Z. M.; Nikishin, G. I. Tetrahedron 2012, 68, 10263–10271. doi:10.1016/j.tet.2012.10.018 |
| 152. | Raju, K. B.; Kumar, B. N.; Nagaiah, K. RSC Adv. 2014, 4, 50795–50800. doi:10.1039/C4RA06233G |
| 252. | Lee, J. M.; Park, E. J.; Cho, S. H.; Chang, S. J. Am. Chem. Soc. 2008, 130, 7824–7825. doi:10.1021/ja8031218 |
| 155. | Kumar, G. S.; Maheswari, C. U.; Kumar, R. A.; Kantam, M. L.; Reddy, K. R. Angew. Chem., Int. Ed. 2011, 50, 11748–11751. doi:10.1002/anie.201105020 |
| 254. | Koshino, N.; Saha, B.; Espenson, J. H. J. Org. Chem. 2003, 68, 9364–9370. doi:10.1021/jo0348017 |
| 154. | Yoo, W.-J.; Li, C.-J. J. Org. Chem. 2006, 71, 6266–6268. doi:10.1021/jo0606103 |
| 252. | Lee, J. M.; Park, E. J.; Cho, S. H.; Chang, S. J. Am. Chem. Soc. 2008, 130, 7824–7825. doi:10.1021/ja8031218 |
| 117. | Schmidt, A.; Habeck, T.; Snovydovych, B.; Eisfeld, W. Org. Lett. 2007, 9, 3515–3518. doi:10.1021/ol0713739 |
| 249. | Jin, J.; Li, Y.; Wang, Z.-j.; Qian, W.-x.; Bao, W.-l. Eur. J. Org. Chem. 2010, 1235–1238. doi:10.1002/ejoc.200901321 |
| 250. | He, H.-F.; Wang, K.; Xing, B.; Sheng, G.; Ma, T.; Bao, W. Synlett 2013, 24, 211–214. doi:10.1055/s-0032-1317960 |
| 104. | Noonan, C.; Baragwanath, L.; Connon, S. J. Tetrahedron Lett. 2008, 49, 4003–4006. doi:10.1016/j.tetlet.2008.04.097 |
| 151. | Rout, S. K.; Guin, S.; Ghara, K. K.; Banerjee, A.; Patel, B. K. Org. Lett. 2012, 14, 3982–3985. doi:10.1021/ol301756y |
| 251. | Bouquet, M.; Guy, A.; Lemaire, M.; Guetté, J. P. Synth. Commun. 1985, 15, 1153–1157. doi:10.1080/00397918508077258 |
| 115. | De Sarkar, S.; Grimme, S.; Studer, A. J. Am. Chem. Soc. 2010, 132, 1190–1191. doi:10.1021/ja910540j |
| 116. | Samanta, R. C.; De Sarkar, S.; Fröhlich, R.; Grimme, S.; Studer, A. Chem. Sci. 2013, 4, 2177–2184. doi:10.1039/C3SC00099K |
| 150. | Toledo, H.; Pisarevsky, E.; Abramovich, A.; Szpilman, A. M. Chem. Commun. 2013, 49, 4367–4369. doi:10.1039/C2CC35220F |
| 250. | He, H.-F.; Wang, K.; Xing, B.; Sheng, G.; Ma, T.; Bao, W. Synlett 2013, 24, 211–214. doi:10.1055/s-0032-1317960 |
| 163. | Yoo, W.-J.; Li, C.-J. Tetrahedron Lett. 2007, 48, 1033–1035. doi:10.1016/j.tetlet.2006.11.169 |
| 267. | Iwaoka, M.; Tomoda, S. J. Chem. Soc., Chem. Commun. 1992, 1165–1167. doi:10.1039/c39920001165 |
| 268. | Tiecco, M.; Testaferri, L.; Tingoli, M.; Bagnoli, L.; Santi, C. J. Chem. Soc., Chem. Commun. 1993, 637–639. doi:10.1039/C39930000637 |
| 162. | Talukdar, D.; Sharma, K.; Bharadwaj, S. K.; Thakur, A. J. Synlett 2013, 24, 963–966. doi:10.1055/s-0032-1316914 |
| 266. | Cho, S.-D.; Kim, H.-J.; Ahn, C.; Falck, J. R.; Shin, D.-S. Tetrahedron Lett. 1999, 40, 8215–8217. doi:10.1016/S0040-4039(99)01713-X |
| 164. | Wu, X.-F.; Darcel, C. Eur. J. Org. Chem. 2009, 1144–1147. doi:10.1002/ejoc.200801176 |
| 159. | Phan, N. T. S.; Nguyen, T. T.; Vu, P. H. L. ChemCatChem 2013, 5, 3068–3077. doi:10.1002/cctc.201300400 |
| 263. | Shaikh, T. M.; Sudalai, A. Tetrahedron Lett. 2005, 46, 5589–5592. doi:10.1016/j.tetlet.2005.06.033 |
| 158. | Saberi, D.; Heydari, A. Tetrahedron Lett. 2013, 54, 4178–4180. doi:10.1016/j.tetlet.2013.05.113 |
| 262. | Akhlaghinia, B.; Ebrahimabadi, H.; Goharshadi, E. K.; Samiee, S.; Rezazadeh, S. J. Mol. Catal. A: Chem. 2012, 357, 67–72. doi:10.1016/j.molcata.2012.01.020 |
| 161. | Gopinath, R.; Barkakaty, B.; Talukdar, B.; Patel, B. K. J. Org. Chem. 2003, 68, 2944–2947. doi:10.1021/jo0266902 |
| 265. | Occhipinti, G.; Liguori, L.; Tsoukala, A.; Bjørsvik, H.-R. Org. Process Res. Dev. 2010, 14, 1379–1384. doi:10.1021/op100185x |
| 160. | Gopinath, R.; Patel, B. K. Org. Lett. 2000, 2, 577–579. doi:10.1021/ol990383+ |
| 264. | Khenkin, A. M.; Neumann, R. J. Am. Chem. Soc. 2004, 126, 6356–6362. doi:10.1021/ja031710i |
| 158. | Saberi, D.; Heydari, A. Tetrahedron Lett. 2013, 54, 4178–4180. doi:10.1016/j.tetlet.2013.05.113 |
| 259. | Khan, K. M.; Maharvi, G. M.; Hayat, S.; Zia-Ullah; Choudhary, M. I.; Atta-ur-Rahman. Tetrahedron 2003, 59, 5549–5554. doi:10.1016/S0040-4020(03)00812-3 |
| 258. | Minisci, F.; Recupero, F.; Gambarotti, C.; Punta, C.; Paganelli, R. Tetrahedron Lett. 2003, 44, 6919–6922. doi:10.1016/S0040-4039(03)01687-3 |
| 157. | Barve, B. D.; Wu, Y.-C.; El-Shazly, M.; Chuang, D.-W.; Cheng, Y.-B.; Wang, J.-J.; Chang, F.-R. J. Org. Chem. 2014, 79, 3206–3214. doi:10.1021/jo402798k |
| 261. | Macedo, A. G.; Fernandes, S. E. M.; Valente, A. A.; Ferreira, R. A. S.; Carlos, L. D.; Rocha, J. Molecules 2010, 15, 747–765. doi:10.3390/molecules15020747 |
| 155. | Kumar, G. S.; Maheswari, C. U.; Kumar, R. A.; Kantam, M. L.; Reddy, K. R. Angew. Chem., Int. Ed. 2011, 50, 11748–11751. doi:10.1002/anie.201105020 |
| 156. | Barve, B. D.; Wu, Y.-C.; El-Shazly, M.; Chuang, D.-W.; Chung, Y.-M.; Tsai, Y.-H.; Wu, S.-F.; Korinek, M.; Du, Y.-C.; Hsieh, C.-T.; Wang, J.-J.; Chang, F.-R. Eur. J. Org. Chem. 2012, 6760–6766. doi:10.1002/ejoc.201201160 |
| 260. | Iwata, S.; Hata, T.; Urabe, H. Adv. Synth. Catal. 2012, 354, 3480–3484. doi:10.1002/adsc.201200410 |
| 201. | Kharasch, M. S.; Fono, A. J. Org. Chem. 1959, 24, 72–78. doi:10.1021/jo01083a022 |
| 200. | Terent'ev, A. O.; Borisov, D. A.; Semenov, V. V.; Chernyshev, V. V.; Dembitsky, V. M.; Nikishin, G. I. Synthesis 2011, 2091–2100. doi:10.1055/s-0030-1260027 |
| 209. | Demir, A. S.; Reis, Ö.; Igdir, A. C. Tetrahedron 2004, 60, 3427–3432. doi:10.1016/j.tet.2004.02.039 |
| 204. | Tanyeli, C.; Sezen, B. Tetrahedron Lett. 2000, 41, 7973–7976. doi:10.1016/S0040-4039(00)01374-5 |
| 211. | Demir, A. S.; Findik, H. Tetrahedron 2008, 64, 6196–6201. doi:10.1016/j.tet.2008.05.004 |
| 211. | Demir, A. S.; Findik, H. Tetrahedron 2008, 64, 6196–6201. doi:10.1016/j.tet.2008.05.004 |
| 203. | Frazier, C. P.; Sandoval, D.; Palmer, L. I.; de Alaniz, J. R. Chem. Sci. 2013, 4, 3857–3862. doi:10.1039/C3SC51658J |
| 202. | Baidya, M.; Griffin, K. A.; Yamamoto, H. J. Am. Chem. Soc. 2012, 134, 18566–18569. doi:10.1021/ja309311z |
| 209. | Demir, A. S.; Reis, Ö.; Igdir, A. C. Tetrahedron 2004, 60, 3427–3432. doi:10.1016/j.tet.2004.02.039 |
| 210. | Demir, A. S.; Caliskan, Z.; Aydin, A. E.; Bicer, I. Tetrahedron: Asymmetry 2006, 17, 786–791. doi:10.1016/j.tetasy.2006.01.025 |
| 204. | Tanyeli, C.; Sezen, B. Tetrahedron Lett. 2000, 41, 7973–7976. doi:10.1016/S0040-4039(00)01374-5 |
| 205. | Tanyeli, C.; Sezen, B.; İyigün, Ç.; Elmalı, O. Tetrahedron Lett. 2001, 42, 6397–6399. doi:10.1016/S0040-4039(01)01262-X |
| 206. | Tanyeli, C.; Özdemirhan, D.; Sezen, B. Tetrahedron 2002, 58, 9983–9988. doi:10.1016/S0040-4020(02)01350-9 |
| 207. | Tanyeli, C.; Tosun, A.; Turkut, E.; Sezen, B. Tetrahedron 2003, 59, 1055–1058. doi:10.1016/S0040-4020(02)01634-4 |
| 208. | Tanyeli, C.; Iyigün, Ç. Tetrahedron 2003, 59, 7135–7139. doi:10.1016/S0040-4020(03)01094-9 |
| 209. | Demir, A. S.; Reis, Ö.; Igdir, A. C. Tetrahedron 2004, 60, 3427–3432. doi:10.1016/j.tet.2004.02.039 |
| 210. | Demir, A. S.; Caliskan, Z.; Aydin, A. E.; Bicer, I. Tetrahedron: Asymmetry 2006, 17, 786–791. doi:10.1016/j.tetasy.2006.01.025 |
| 202. | Baidya, M.; Griffin, K. A.; Yamamoto, H. J. Am. Chem. Soc. 2012, 134, 18566–18569. doi:10.1021/ja309311z |
| 212. | Marín-Barrios, R.; García-Cabeza, A. L.; Moreno-Dorado, F. J.; Guerra, F. M.; Massanet, G. M. J. Org. Chem. 2014, 79, 6501–6509. doi:10.1021/jo500915r |
| 121. | Kuwano, S.; Harada, S.; Oriez, R.; Yamada, K. Chem. Commun. 2012, 48, 145–147. doi:10.1039/C1CC15539C |
| 218. | Chen, M. S.; White, M. C. J. Am. Chem. Soc. 2004, 126, 1346–1347. doi:10.1021/ja039107n |
| 120. | Valizadeh, H.; Ahmadi, M. C. R. Chim. 2012, 15, 1077–1080. doi:10.1016/j.crci.2012.08.004 |
| 21. | Liron, F.; Oble, J.; Lorion, M. M.; Poli, G. Eur. J. Org. Chem. 2014, 5863–5883. doi:10.1002/ejoc.201402049 |
| 213. | Kitching, W.; Rappoport, Z.; Winstein, S.; Young, W. G. J. Am. Chem. Soc. 1966, 88, 2054–2055. doi:10.1021/ja00961a043 |
| 110. | Zhao, J.; Mück-Lichtenfeld, C.; Studer, A. Adv. Synth. Catal. 2013, 355, 1098–1106. doi:10.1002/adsc.201300034 |
| 218. | Chen, M. S.; White, M. C. J. Am. Chem. Soc. 2004, 126, 1346–1347. doi:10.1021/ja039107n |
| 109. | Kiran, I. N. C.; Lalwani, K.; Sudalai, A. RSC Adv. 2013, 3, 1695–1698. doi:10.1039/C2RA22718E |
| 218. | Chen, M. S.; White, M. C. J. Am. Chem. Soc. 2004, 126, 1346–1347. doi:10.1021/ja039107n |
| 119. | Zhang, M.; Zhang, S.; Zhang, G.; Chen, F.; Cheng, J. Tetrahedron Lett. 2011, 52, 2480–2483. doi:10.1016/j.tetlet.2011.03.017 |
| 213. | Kitching, W.; Rappoport, Z.; Winstein, S.; Young, W. G. J. Am. Chem. Soc. 1966, 88, 2054–2055. doi:10.1021/ja00961a043 |
| 118. | Cheng, S.; Chen, J.; Gao, W.; Jin, H.; Ding, J.; Wu, H. J. Chem. Res. 2010, 34, 130–132. doi:10.3184/030823410X12670951969185 |
| 214. | Engelin, C.; Jensen, T.; Rodriguez-Rodriguez, S.; Fristrup, P. ACS Catal. 2013, 3, 294–302. doi:10.1021/cs3007878 |
| 215. | Grennberg, H.; Bäckvall, J.-E. Chem. – Eur. J. 1998, 4, 1083–1089. doi:10.1002/(SICI)1521-3765(19980615)4:6<1083::AID-CHEM1083>3.0.CO;2-F |
| 216. | Diao, T.; Stahl, S. S. Polyhedron 2014, 84, 96–102. doi:10.1016/j.poly.2014.06.038 |
| 217. | Chen, M. S.; Prabagaran, N.; Labenz, N. A.; White, M. C. J. Am. Chem. Soc. 2005, 127, 6970–6971. doi:10.1021/ja0500198 |
| 108. | Finney, E. E.; Ogawa, K. A.; Boydston, A. J. J. Am. Chem. Soc. 2012, 134, 12374–12377. doi:10.1021/ja304716r |
| 217. | Chen, M. S.; Prabagaran, N.; Labenz, N. A.; White, M. C. J. Am. Chem. Soc. 2005, 127, 6970–6971. doi:10.1021/ja0500198 |
| 107. | Reddy, R. S.; Rosa, J. N.; Veiros, L. F.; Caddick, S.; Gois, P. M. P. Org. Biomol. Chem. 2011, 9, 3126–3129. doi:10.1039/C1OB05151B |
| 215. | Grennberg, H.; Bäckvall, J.-E. Chem. – Eur. J. 1998, 4, 1083–1089. doi:10.1002/(SICI)1521-3765(19980615)4:6<1083::AID-CHEM1083>3.0.CO;2-F |
| 105. | Maki, B. E.; Scheidt, K. A. Org. Lett. 2008, 10, 4331–4334. doi:10.1021/ol8018488 |
| 106. | Maki, B. E.; Chan, A.; Phillips, E. M.; Scheidt, K. A. Tetrahedron 2009, 65, 3102–3109. doi:10.1016/j.tet.2008.10.033 |
| 20. | Liu, G.; Wu, Y. Top. Curr. Chem. 2010, 292, 195–209. doi:10.1007/128_2009_16 |
| 21. | Liron, F.; Oble, J.; Lorion, M. M.; Poli, G. Eur. J. Org. Chem. 2014, 5863–5883. doi:10.1002/ejoc.201402049 |
| 213. | Kitching, W.; Rappoport, Z.; Winstein, S.; Young, W. G. J. Am. Chem. Soc. 1966, 88, 2054–2055. doi:10.1021/ja00961a043 |
| 114. | Delany, E. G.; Fagan, C.-L.; Gundala, S.; Zeitler, K.; Connon, S. J. Chem. Commun. 2013, 49, 6513–6515. doi:10.1039/C3CC42597E |
| 122. | Delany, E. G.; Fagan, C.-L.; Gundala, S.; Mari, A.; Broja, T.; Zeitler, K.; Connon, S. J. Chem. Commun. 2013, 49, 6510–6512. doi:10.1039/C3CC42596G |
| 125. | Iwahana, S.; Iida, H.; Yashima, E. Chem. – Eur. J. 2011, 17, 8009–8013. doi:10.1002/chem.201100737 |
| 126. | De Sarkar, S.; Biswas, A.; Song, C. H.; Studer, A. Synthesis 2011, 1974–1983. doi:10.1055/s-0030-1260030 |
| 224. | Check, C. T.; Henderson, W. H.; Wray, B. C.; Vanden Eynden, M. J.; Stambuli, J. P. J. Am. Chem. Soc. 2011, 133, 18503–18505. doi:10.1021/ja2089102 |
| 124. | Zhang, D.; Pan, C. Catal. Commun. 2012, 20, 41–45. doi:10.1016/j.catcom.2011.12.041 |
| 223. | Thiery, E.; Aouf, C.; Belloy, J.; Harakat, D.; Le Bras, J.; Muzart, J. J. Org. Chem. 2010, 75, 1771–1774. doi:10.1021/jo902587u |
| 128. | Yamada, S.; Morizono, D.; Yamamoto, K. Tetrahedron Lett. 1992, 33, 4329–4332. doi:10.1016/S0040-4039(00)74252-3 |
| 127. | Mori, N.; Togo, H. Tetrahedron 2005, 61, 5915–5925. doi:10.1016/j.tet.2005.03.097 |
| 225. | Macsári, I.; Szabó, K. J. Tetrahedron Lett. 1998, 39, 6345–6348. doi:10.1016/S0040-4039(98)01304-5 |
| 1. | Li, C.-J.; Li, Z. Pure Appl. Chem. 2006, 78, 935–945. doi:10.1351/pac200678050935 |
| 2. | Li, Z.; Bohle, D. S.; Li, C.-J. Proc. Natl. Acad. Sci. U. S. A. 2006, 103, 8928–8933. doi:10.1073/pnas.0601687103 |
| 3. | Beccalli, E. M.; Broggini, G.; Martinelli, M.; Sottocornola, S. Chem. Rev. 2007, 107, 5318–5365. doi:10.1021/cr068006f |
| 4. | Li, C.-J. Acc. Chem. Res. 2009, 42, 335–344. doi:10.1021/ar800164n |
| 5. | Scheuermann, C. J. Chem. – Asian J. 2010, 5, 436–451. doi:10.1002/asia.200900487 |
| 6. | Yoo, W.-J.; Li, C.-J. Top. Curr. Chem. 2010, 292, 281–302. doi:10.1007/128_2009_17 |
| 7. | Yeung, C. S.; Dong, V. M. Chem. Rev. 2011, 111, 1215–1292. doi:10.1021/cr100280d |
| 8. | Cho, S. H.; Kim, J. Y.; Kwak, J.; Chang, S. Chem. Soc. Rev. 2011, 40, 5068–5083. doi:10.1039/c1cs15082k |
| 9. | Sun, C.-L.; Li, B.-J.; Shi, Z.-J. Chem. Rev. 2011, 111, 1293–1314. doi:10.1021/cr100198w |
| 10. | Zhang, C.; Tang, C.; Jiao, N. Chem. Soc. Rev. 2012, 41, 3464–3484. doi:10.1039/C2CS15323H |
| 11. | Song, G.; Wang, F.; Li, X. Chem. Soc. Rev. 2012, 41, 3651–3678. doi:10.1039/c2cs15281a |
| 12. | Hirano, K.; Miura, M. Chem. Commun. 2012, 48, 10704–10714. doi:10.1039/C2CC34659A |
| 13. | Liu, C.; Liu, D.; Lei, A. Acc. Chem. Res. 2014, 47, 3459–3470. doi:10.1021/ar5002044 |
| 14. | Girard, S. A.; Knauber, T.; Li, C.-J. Angew. Chem., Int. Ed. 2014, 53, 74–100. doi:10.1002/anie.201304268 |
| 15. | Samanta, R.; Matcha, K.; Antonchick, A. P. Eur. J. Org. Chem. 2013, 5769–5804. doi:10.1002/ejoc.201300286 |
| 113. | Maki, B. E.; Chan, A.; Phillips, E. M.; Scheidt, K. A. Org. Lett. 2007, 9, 371–374. doi:10.1021/ol062940f |
| 221. | Mitsudome, T.; Umetani, T.; Nosaka, N.; Mori, K.; Mizugaki, T.; Ebitani, K.; Kaneda, K. Angew. Chem., Int. Ed. 2006, 45, 481–485. doi:10.1002/anie.200502886 |
| 106. | Maki, B. E.; Chan, A.; Phillips, E. M.; Scheidt, K. A. Tetrahedron 2009, 65, 3102–3109. doi:10.1016/j.tet.2008.10.033 |
| 113. | Maki, B. E.; Chan, A.; Phillips, E. M.; Scheidt, K. A. Org. Lett. 2007, 9, 371–374. doi:10.1021/ol062940f |
| 123. | Luo, F.; Pan, C.; Cheng, J.; Chen, F. Tetrahedron 2011, 67, 5878–5882. doi:10.1016/j.tet.2011.06.060 |
| 124. | Zhang, D.; Pan, C. Catal. Commun. 2012, 20, 41–45. doi:10.1016/j.catcom.2011.12.041 |
| 220. | Campbell, A. N.; White, P. B.; Guzei, I. A.; Stahl, S. S. J. Am. Chem. Soc. 2010, 132, 15116–15119. doi:10.1021/ja105829t |
| 123. | Luo, F.; Pan, C.; Cheng, J.; Chen, F. Tetrahedron 2011, 67, 5878–5882. doi:10.1016/j.tet.2011.06.060 |
| 222. | Vermeulen, N. A.; Delcamp, J. H.; White, M. C. J. Am. Chem. Soc. 2010, 132, 11323–11328. doi:10.1021/ja104826g |
| 106. | Maki, B. E.; Chan, A.; Phillips, E. M.; Scheidt, K. A. Tetrahedron 2009, 65, 3102–3109. doi:10.1016/j.tet.2008.10.033 |
| 221. | Mitsudome, T.; Umetani, T.; Nosaka, N.; Mori, K.; Mizugaki, T.; Ebitani, K.; Kaneda, K. Angew. Chem., Int. Ed. 2006, 45, 481–485. doi:10.1002/anie.200502886 |
| 15. | Samanta, R.; Matcha, K.; Antonchick, A. P. Eur. J. Org. Chem. 2013, 5769–5804. doi:10.1002/ejoc.201300286 |
| 217. | Chen, M. S.; Prabagaran, N.; Labenz, N. A.; White, M. C. J. Am. Chem. Soc. 2005, 127, 6970–6971. doi:10.1021/ja0500198 |
| 16. | Hoffman, R. V.; Wilson, A. L.; Kim, H. O. J. Org. Chem. 1990, 55, 1267–1270. doi:10.1021/jo00291a030 |
| 17. | Lifchits, O.; Demoulin, N.; List, B. Angew. Chem., Int. Ed. 2011, 50, 9680–9683. doi:10.1002/anie.201104244 |
| 18. | Jones, K. M.; Tomkinson, N. C. O. J. Org. Chem. 2012, 77, 921–928. doi:10.1021/jo202084w |
| 1. | Li, C.-J.; Li, Z. Pure Appl. Chem. 2006, 78, 935–945. doi:10.1351/pac200678050935 |
| 2. | Li, Z.; Bohle, D. S.; Li, C.-J. Proc. Natl. Acad. Sci. U. S. A. 2006, 103, 8928–8933. doi:10.1073/pnas.0601687103 |
| 3. | Beccalli, E. M.; Broggini, G.; Martinelli, M.; Sottocornola, S. Chem. Rev. 2007, 107, 5318–5365. doi:10.1021/cr068006f |
| 4. | Li, C.-J. Acc. Chem. Res. 2009, 42, 335–344. doi:10.1021/ar800164n |
| 5. | Scheuermann, C. J. Chem. – Asian J. 2010, 5, 436–451. doi:10.1002/asia.200900487 |
| 6. | Yoo, W.-J.; Li, C.-J. Top. Curr. Chem. 2010, 292, 281–302. doi:10.1007/128_2009_17 |
| 7. | Yeung, C. S.; Dong, V. M. Chem. Rev. 2011, 111, 1215–1292. doi:10.1021/cr100280d |
| 8. | Cho, S. H.; Kim, J. Y.; Kwak, J.; Chang, S. Chem. Soc. Rev. 2011, 40, 5068–5083. doi:10.1039/c1cs15082k |
| 9. | Sun, C.-L.; Li, B.-J.; Shi, Z.-J. Chem. Rev. 2011, 111, 1293–1314. doi:10.1021/cr100198w |
| 10. | Zhang, C.; Tang, C.; Jiao, N. Chem. Soc. Rev. 2012, 41, 3464–3484. doi:10.1039/C2CS15323H |
| 11. | Song, G.; Wang, F.; Li, X. Chem. Soc. Rev. 2012, 41, 3651–3678. doi:10.1039/c2cs15281a |
| 12. | Hirano, K.; Miura, M. Chem. Commun. 2012, 48, 10704–10714. doi:10.1039/C2CC34659A |
| 13. | Liu, C.; Liu, D.; Lei, A. Acc. Chem. Res. 2014, 47, 3459–3470. doi:10.1021/ar5002044 |
| 14. | Girard, S. A.; Knauber, T.; Li, C.-J. Angew. Chem., Int. Ed. 2014, 53, 74–100. doi:10.1002/anie.201304268 |
| 112. | Ji, M.; Wang, X.; Lim, Y. N.; Kang, Y.-W.; Jang, H.-Y. Eur. J. Org. Chem. 2013, 7881–7885. doi:10.1002/ejoc.201301337 |
| 219. | Covell, D. J.; White, M. C. Angew. Chem., Int. Ed. 2008, 47, 6448–6451. doi:10.1002/anie.200802106 |
| 1. | Li, C.-J.; Li, Z. Pure Appl. Chem. 2006, 78, 935–945. doi:10.1351/pac200678050935 |
| 2. | Li, Z.; Bohle, D. S.; Li, C.-J. Proc. Natl. Acad. Sci. U. S. A. 2006, 103, 8928–8933. doi:10.1073/pnas.0601687103 |
| 3. | Beccalli, E. M.; Broggini, G.; Martinelli, M.; Sottocornola, S. Chem. Rev. 2007, 107, 5318–5365. doi:10.1021/cr068006f |
| 4. | Li, C.-J. Acc. Chem. Res. 2009, 42, 335–344. doi:10.1021/ar800164n |
| 5. | Scheuermann, C. J. Chem. – Asian J. 2010, 5, 436–451. doi:10.1002/asia.200900487 |
| 6. | Yoo, W.-J.; Li, C.-J. Top. Curr. Chem. 2010, 292, 281–302. doi:10.1007/128_2009_17 |
| 7. | Yeung, C. S.; Dong, V. M. Chem. Rev. 2011, 111, 1215–1292. doi:10.1021/cr100280d |
| 8. | Cho, S. H.; Kim, J. Y.; Kwak, J.; Chang, S. Chem. Soc. Rev. 2011, 40, 5068–5083. doi:10.1039/c1cs15082k |
| 9. | Sun, C.-L.; Li, B.-J.; Shi, Z.-J. Chem. Rev. 2011, 111, 1293–1314. doi:10.1021/cr100198w |
| 10. | Zhang, C.; Tang, C.; Jiao, N. Chem. Soc. Rev. 2012, 41, 3464–3484. doi:10.1039/C2CS15323H |
| 11. | Song, G.; Wang, F.; Li, X. Chem. Soc. Rev. 2012, 41, 3651–3678. doi:10.1039/c2cs15281a |
| 12. | Hirano, K.; Miura, M. Chem. Commun. 2012, 48, 10704–10714. doi:10.1039/C2CC34659A |
| 13. | Liu, C.; Liu, D.; Lei, A. Acc. Chem. Res. 2014, 47, 3459–3470. doi:10.1021/ar5002044 |
| 14. | Girard, S. A.; Knauber, T.; Li, C.-J. Angew. Chem., Int. Ed. 2014, 53, 74–100. doi:10.1002/anie.201304268 |
| 15. | Samanta, R.; Matcha, K.; Antonchick, A. P. Eur. J. Org. Chem. 2013, 5769–5804. doi:10.1002/ejoc.201300286 |
| 111. | Chiarotto, I.; Feroci, M.; Sotgiu, G.; Inesi, A. Tetrahedron 2013, 69, 8088–8095. doi:10.1016/j.tet.2013.06.014 |
| 217. | Chen, M. S.; Prabagaran, N.; Labenz, N. A.; White, M. C. J. Am. Chem. Soc. 2005, 127, 6970–6971. doi:10.1021/ja0500198 |
| 10. | Zhang, C.; Tang, C.; Jiao, N. Chem. Soc. Rev. 2012, 41, 3464–3484. doi:10.1039/C2CS15323H |
| 20. | Liu, G.; Wu, Y. Top. Curr. Chem. 2010, 292, 195–209. doi:10.1007/128_2009_16 |
| 21. | Liron, F.; Oble, J.; Lorion, M. M.; Poli, G. Eur. J. Org. Chem. 2014, 5863–5883. doi:10.1002/ejoc.201402049 |
| 19. | Liu, B.; Shi, B.-F. Tetrahedron Lett. 2015, 56, 15–22. doi:10.1016/j.tetlet.2014.11.039 |
| 3. | Beccalli, E. M.; Broggini, G.; Martinelli, M.; Sottocornola, S. Chem. Rev. 2007, 107, 5318–5365. doi:10.1021/cr068006f |
| 137. | Agrawal, M. K.; Adimurthy, S.; Ghosh, P. K. Synth. Commun. 2012, 42, 2931–2936. doi:10.1080/00397911.2011.572219 |
| 233. | Benhmid, A.; Narayana, K. V.; Martin, A.; Lücke, B. Chem. Commun. 2004, 2118–2119. doi:10.1039/B406396A |
| 234. | Kalevaru, V. N.; Benhmid, A.; Radnik, J.; Pohl, M.-M.; Lücke, B.; Martin, A. Catal. Today 2009, 141, 317–324. doi:10.1016/j.cattod.2008.06.004 |
| 235. | Gatla, S.; Madaan, N.; Radnik, J.; Kalevaru, V. N.; Pohl, M.-M.; Lücke, B.; Martin, A.; Brückner, A. Appl. Catal., A 2011, 398, 104–112. doi:10.1016/j.apcata.2011.03.025 |
| 236. | Madaan, N.; Gatla, S.; Kalevaru, V. N.; Radnik, J.; Pohl, M.-M.; Lücke, B.; Brückner, A.; Martin, A. ChemCatChem 2013, 5, 185–191. doi:10.1002/cctc.201200522 |
| 232. | Liu, H.; Shi, G.; Pan, S.; Jiang, Y.; Zhang, Y. Org. Lett. 2013, 15, 4098–4101. doi:10.1021/ol401687f |
| 138. | McDonald, C.; Holcomb, H.; Kennedy, K.; Kirkpatrick, E.; Leathers, T.; Vanemon, P. J. Org. Chem. 1989, 54, 1213–1215. doi:10.1021/jo00266a046 |
| 133. | Shaikh, T. M. A.; Emmanuvel, L.; Sudalai, A. Synth. Commun. 2007, 37, 2641–2646. doi:10.1080/00397910701465032 |
| 230. | Boldron, C.; Gamez, P.; Tooke, D. M.; Spek, A. L.; Reedijk, J. Angew. Chem., Int. Ed. 2005, 44, 3585–3587. doi:10.1002/anie.200462946 |
| 132. | Karade, N. N.; Shirodkar, S. G.; Dhoot, B. M.; Waghmare, P. B. J. Chem. Res. 2005, 274–276. doi:10.3184/0308234054213357 |
| 169. | Zhao, J.; Fang, H.; Han, J.; Pan, Y. Org. Lett. 2014, 16, 2530–2533. doi:10.1021/ol5009119 |
| 135. | Stevens, R. V.; Chapman, K. T.; Stubbs, C. A.; Tam, W. W.; Albizati, K. F. Tetrahedron Lett. 1982, 23, 4647–4650. doi:10.1016/S0040-4039(00)85677-4 |
| 232. | Liu, H.; Shi, G.; Pan, S.; Jiang, Y.; Zhang, Y. Org. Lett. 2013, 15, 4098–4101. doi:10.1021/ol401687f |
| 134. | Leduc, A. B.; Jamison, T. F. Org. Process Res. Dev. 2012, 16, 1082–1089. doi:10.1021/op200118h |
| 231. | Boldron, C.; Özalp-Yaman, Ş.; Gamez, P.; Tooke, D. M.; Spek, A. L.; Reedijk, J. Dalton Trans. 2005, 3535–3541. doi:10.1039/B507199B |
| 129. | Reddy, K. R.; Venkateshwar, M.; Maheswari, C. U.; Prashanthi, S. Synth. Commun. 2009, 40, 186–195. doi:10.1080/00397910902838920 |
| 227. | Takemura, N.; Kuninobu, Y.; Kanai, M. Org. Biomol. Chem. 2014, 12, 2528–2532. doi:10.1039/C4OB00215F |
| 226. | García-Cabeza, A. L.; Marín-Barrios, R.; Moreno-Dorado, F. J.; Ortega, M. J.; Massanet, G. M.; Guerra, F. M. Org. Lett. 2014, 16, 1598–1601. doi:10.1021/ol500198c |
| 131. | Kiran, Y. B.; Ikeda, R.; Sakai, N.; Konakahara, T. Synthesis 2010, 276–282. doi:10.1055/s-0029-1217121 |
| 229. | Rout, S. K.; Guin, S.; Ali, W.; Gogoi, A.; Patel, B. K. Org. Lett. 2014, 16, 3086–3089. doi:10.1021/ol5011906 |
| 130. | Karade, N. N.; Budhewar, V. H.; Katkar, A. N.; Tiwari, G. B. ARKIVOC 2006, No. xi, 162–167. |
| 228. | Rout, S. K.; Guin, S.; Banerjee, A.; Khatun, N.; Gogoi, A.; Patel, B. K. Org. Lett. 2013, 15, 4106–4109. doi:10.1021/ol401682a |
| 47. | Bhadra, S.; Matheis, C.; Katayev, D.; Gooßen, L. J. Angew. Chem., Int. Ed. 2013, 52, 9279–9283. doi:10.1002/anie.201303702 |
| 55. | Wang, L.; Xia, X.-D.; Guo, W.; Chen, J.-R.; Xiao, W.-J. Org. Biomol. Chem. 2011, 9, 6895–6898. doi:10.1039/C1OB05887H |
| 55. | Wang, L.; Xia, X.-D.; Guo, W.; Chen, J.-R.; Xiao, W.-J. Org. Biomol. Chem. 2011, 9, 6895–6898. doi:10.1039/C1OB05887H |
| 53. | Rit, R. K.; Yadav, M. R.; Sahoo, A. K. Org. Lett. 2014, 16, 968–971. doi:10.1021/ol403699d |
| 54. | Desai, L. V.; Malik, H. A.; Sanford, M. S. Org. Lett. 2006, 8, 1141–1144. doi:10.1021/ol0530272 |
| 51. | Wang, G.-W.; Yuan, T.-T.; Wu, X.-L. J. Org. Chem. 2008, 73, 4717–4720. doi:10.1021/jo8003088 |
| 52. | Yadav, M. R.; Rit, R. K.; Sahoo, A. K. Chem. – Eur. J. 2012, 18, 5541–5545. doi:10.1002/chem.201200092 |
| 49. | Wang, G.-W.; Yuan, T.-T. J. Org. Chem. 2010, 75, 476–479. doi:10.1021/jo902139b |
| 50. | Jiang, T.-S.; Wang, G.-W. J. Org. Chem. 2012, 77, 9504–9509. doi:10.1021/jo301964m |
| 56. | Neufeldt, S. R.; Sanford, M. S. Org. Lett. 2010, 12, 532–535. doi:10.1021/ol902720d |
| 57. | Padala, K.; Jeganmohan, M. Chem. Commun. 2013, 49, 9651–9653. doi:10.1039/C3CC45350B |
| 280. | Weinberg, N. L.; Brown, E. A. J. Org. Chem. 1966, 31, 4058–4061. doi:10.1021/jo01350a041 |
| 58. | Reddy, B. V. S.; Revathi, G.; Reddy, A. S.; Yadav, J. S. Tetrahedron Lett. 2011, 52, 5926–5929. doi:10.1016/j.tetlet.2011.08.098 |
| 279. | Shu, X.-Z.; Xia, X.-F.; Yang, Y.-F.; Ji, K.-G.; Liu, X.-Y.; Liang, Y.-M. J. Org. Chem. 2009, 74, 7464–7469. doi:10.1021/jo901583r |
| 281. | Liang, D.; Wang, M.; Dong, Y.; Guo, Y.; Liu, Q. RSC Adv. 2014, 4, 6564–6567. doi:10.1039/C3RA47282E |
| 252. | Lee, J. M.; Park, E. J.; Cho, S. H.; Chang, S. J. Am. Chem. Soc. 2008, 130, 7824–7825. doi:10.1021/ja8031218 |
| 276. | Phan, N. T. S.; Vu, P. H. L.; Nguyen, T. T. J. Catal. 2013, 306, 38–46. doi:10.1016/j.jcat.2013.06.006 |
| 278. | Murahashi, S.; Naota, T.; Kuwabara, T.; Saito, T.; Kumobayashi, H.; Akutagawa, S. J. Am. Chem. Soc. 1990, 112, 7820–7822. doi:10.1021/ja00177a067 |
| 277. | Murahash, S.-I.; Saito, T.; Naota, T.; Kumobayashi, H.; Akutagawa, S. Tetrahedron Lett. 1991, 32, 5991–5994. doi:10.1016/S0040-4039(00)79446-9 |
| 64. | Gou, F.-R.; Wang, X.-C.; Huo, P.-F.; Bi, H.-P.; Guan, Z.-H.; Liang, Y.-M. Org. Lett. 2009, 11, 5726–5729. doi:10.1021/ol902497k |
| 65. | Chan, L. Y.; Meng, X.; Kim, S. J. Org. Chem. 2013, 78, 8826–8832. doi:10.1021/jo4011188 |
| 62. | Zhang, S.-Y.; He, G.; Zhao, Y.; Wright, K.; Nack, W. A.; Chen, G. J. Am. Chem. Soc. 2012, 134, 7313–7316. doi:10.1021/ja3023972 |
| 63. | Chen, F.-J.; Zhao, S.; Hu, F.; Chen, K.; Zhang, Q.; Zhang, S.-Q.; Shi, B.-F. Chem. Sci. 2013, 4, 4187–4192. doi:10.1039/C3SC51993G |
| 60. | Yin, Z.; Jiang, X.; Sun, P. J. Org. Chem. 2013, 78, 10002–10007. doi:10.1021/jo401623j |
| 59. | Péron, F.; Fossey, C.; Sopkova-de Oliveira Santos, J.; Cailly, T.; Fabis, F. Chem. – Eur. J. 2014, 20, 7507–7513. doi:10.1002/chem.201303923 |
| 58. | Reddy, B. V. S.; Revathi, G.; Reddy, A. S.; Yadav, J. S. Tetrahedron Lett. 2011, 52, 5926–5929. doi:10.1016/j.tetlet.2011.08.098 |
| 59. | Péron, F.; Fossey, C.; Sopkova-de Oliveira Santos, J.; Cailly, T.; Fabis, F. Chem. – Eur. J. 2014, 20, 7507–7513. doi:10.1002/chem.201303923 |
| 283. | Liu, H.; Wang, X.; Gu, Y. Org. Biomol. Chem. 2011, 9, 1614–1620. doi:10.1039/C0OB00749H |
| 282. | Liu, X.; Cheng, R.; Zhao, F.; Zhang-Negrerie, D.; Du, Y.; Zhao, K. Org. Lett. 2012, 14, 5480–5483. doi:10.1021/ol3025583 |
| 283. | Liu, H.; Wang, X.; Gu, Y. Org. Biomol. Chem. 2011, 9, 1614–1620. doi:10.1039/C0OB00749H |
| 291. | Takemura, N.; Kuninobu, Y.; Kanai, M. Org. Lett. 2013, 15, 844–847. doi:10.1021/ol303533z |
| 290. | Chen, X.; Zhu, C.; Cui, X.; Wu, Y. Chem. Commun. 2013, 49, 6900–6902. doi:10.1039/c3cc43947j |
| 293. | Takuwa, A.; Soga, O.; Iwamoto, H.; Maruyama, K. Bull. Chem. Soc. Jpn. 1986, 59, 2959–2961. doi:10.1246/bcsj.59.2959 |
| 292. | Halper, S. R.; Stork, J. R.; Cohen, S. M. Dalton Trans. 2007, 1067–1074. doi:10.1039/B615801C |
| 285. | Jiang, Q.; Wang, J.-Y.; Guo, C. J. Org. Chem. 2014, 79, 8768–8773. doi:10.1021/jo501601u |
| 284. | Liu, H.; Xie, Y.; Gu, Y. Tetrahedron Lett. 2011, 52, 4324–4326. doi:10.1016/j.tetlet.2011.06.041 |
| 286. | Pelter, A.; Elgendy, S. M. A. J. Chem. Soc., Perkin Trans. 1 1993, 1891–1896. doi:10.1039/P19930001891 |
| 287. | Fleck, A. E.; Hobart, J. A.; Morrow, G. W. Synth. Commun. 1992, 22, 179–187. doi:10.1080/00397919208021090 |
| 288. | Mitchell, A. S.; Russell, R. A. Tetrahedron Lett. 1993, 34, 545–548. doi:10.1016/0040-4039(93)85123-E |
| 33. | Dick, A. R.; Hull, K. L.; Sanford, M. S. J. Am. Chem. Soc. 2004, 126, 2300–2301. doi:10.1021/ja031543m |
| 34. | Kalyani, D.; Sanford, M. S. Org. Lett. 2005, 7, 4149–4152. doi:10.1021/ol051486x |
| 13. | Liu, C.; Liu, D.; Lei, A. Acc. Chem. Res. 2014, 47, 3459–3470. doi:10.1021/ar5002044 |
| 25. | Stowers, K. J.; Sanford, M. S. Org. Lett. 2009, 11, 4584–4587. doi:10.1021/ol901820w |
| 26. | Anand, M.; Sunoj, R. B. Org. Lett. 2011, 13, 4802–4805. doi:10.1021/ol201830r |
| 27. | Zhu, W.; Wang, H.; Peng, H.; Liu, G.; Guo, Y. Chin. J. Chem. 2013, 31, 371–376. doi:10.1002/cjoc.201201167 |
| 28. | Desai, L. V.; Stowers, K. J.; Sanford, M. S. J. Am. Chem. Soc. 2008, 130, 13285–13293. doi:10.1021/ja8045519 |
| 29. | Racowski, J. M.; Dick, A. R.; Sanford, M. S. J. Am. Chem. Soc. 2009, 131, 10974–10983. doi:10.1021/ja9014474 |
| 30. | Powers, D. C.; Ritter, T. Acc. Chem. Res. 2012, 45, 840–850. doi:10.1021/ar2001974 |
| 31. | Camasso, N. M.; Pérez-Temprano, M. H.; Sanford, M. S. J. Am. Chem. Soc. 2014, 136, 12771–12775. doi:10.1021/ja507056u |
| 32. | Dick, A. R.; Kampf, J. W.; Sanford, M. S. J. Am. Chem. Soc. 2005, 127, 12790–12791. doi:10.1021/ja0541940 |
| 23. | Ananikov, V. P.; Khemchyan, L. L.; Ivanova, Yu. V.; Bukhtiyarov, V. I.; Sorokin, A. M.; Prosvirin, I. P.; Vatsadze, S. Z.; Medved'ko, A. V.; Nuriev, V. N.; Dilman, A. D.; Levin, V. V.; Koptyug, I. V.; Kovtunov, K. V.; Zhivonitko, V. V.; Likholobov, V. A.; Romanenko, A. V.; Simonov, P. A.; Nenajdenko, V. G.; Shmatova, O. I.; Muzalevskiy, V. M.; Nechaev, M. S.; Asachenko, A. F.; Morozov, O. S.; Dzhevakov, P. B.; Osipov, S. N.; Vorobyeva, D. V.; Topchiy, M. A.; Zotova, M. A.; Ponomarenko, S. A.; Borshchev, O. V.; Luponosov, Y. N.; Rempel, A. A.; Valeeva, A. A.; Stakheev, A. Yu.; Turova, O. V.; Mashkovsky, I. S.; Sysolyatin, S. V.; Malykhin, V. V.; Bukhtiyarova, G. A.; Terent'ev, A. O.; Krylov, I. B. Russ. Chem. Rev. 2014, 83, 885–985. doi:10.1070/RC2014v083n10ABEH004471 |
| 24. | Ekoue-Kovi, K.; Wolf, C. Chem. – Eur. J. 2008, 14, 6302–6315. doi:10.1002/chem.200800353 |
| 22. | Wu, X.-F.; Gong, J.-L.; Qi, X. Org. Biomol. Chem. 2014, 12, 5807–5817. doi:10.1039/C4OB00276H |
| 37. | Li, L.; Yu, P.; Cheng, J.; Chen, F.; Pan, C. Chem. Lett. 2012, 41, 600–602. doi:10.1246/cl.2012.600 |
| 35. | Ye, Z.; Wang, W.; Luo, F.; Zhang, S.; Cheng, J. Org. Lett. 2009, 11, 3974–3977. doi:10.1021/ol901609t |
| 36. | Hu, C.-J.; Zhang, X.-H.; Ding, Q.-P.; Lv, T.; Ge, S.-P.; Zhong, P. Tetrahedron Lett. 2012, 53, 2465–2468. doi:10.1016/j.tetlet.2012.03.022 |
| 295. | Bansal, V.; Sharma, J.; Khanna, R. N. J. Chem. Res., Synop. 1998, 720–721. doi:10.1039/A803513J |
| 294. | Kidwai, M.; Kumar, P.; Kohli, S. J. Chem. Res., Synop. 1997, 24–25. doi:10.1039/A601810F |
| 198. | Krylov, I. B.; Terent’ev, A. O.; Timofeev, V. P.; Shelimov, B. N.; Novikov, R. A.; Merkulova, V. M.; Nikishin, G. I. Adv. Synth. Catal. 2014, 356, 2266–2280. doi:10.1002/adsc.201400143 |
| 229. | Rout, S. K.; Guin, S.; Ali, W.; Gogoi, A.; Patel, B. K. Org. Lett. 2014, 16, 3086–3089. doi:10.1021/ol5011906 |
| 197. | Terent’ev, A. O.; Krylov, I. B.; Timofeev, V. P.; Starikova, Z. A.; Merkulova, V. M.; Ilovaisky, A. I.; Nikishin, G. I. Adv. Synth. Catal. 2013, 355, 2375–2390. doi:10.1002/adsc.201300341 |
| 169. | Zhao, J.; Fang, H.; Han, J.; Pan, Y. Org. Lett. 2014, 16, 2530–2533. doi:10.1021/ol5009119 |
| 199. | Terent'ev, A. O.; Borisov, D. A.; Yaremenko, I. A.; Chernyshev, V. V.; Nikishin, G. I. J. Org. Chem. 2010, 75, 5065–5071. doi:10.1021/jo100793j |
| 302. | Tajima, T.; Fuchigami, T. Chem. – Eur. J. 2005, 11, 6192–6196. doi:10.1002/chem.200500340 |
| 197. | Terent’ev, A. O.; Krylov, I. B.; Timofeev, V. P.; Starikova, Z. A.; Merkulova, V. M.; Ilovaisky, A. I.; Nikishin, G. I. Adv. Synth. Catal. 2013, 355, 2375–2390. doi:10.1002/adsc.201300341 |
| 198. | Krylov, I. B.; Terent’ev, A. O.; Timofeev, V. P.; Shelimov, B. N.; Novikov, R. A.; Merkulova, V. M.; Nikishin, G. I. Adv. Synth. Catal. 2014, 356, 2266–2280. doi:10.1002/adsc.201400143 |
| 301. | Gary, J. B.; Cook, A. K.; Sanford, M. S. ACS Catal. 2013, 3, 700–703. doi:10.1021/cs300786j |
| 194. | Uyanik, M.; Suzuki, D.; Yasui, T.; Ishihara, K. Angew. Chem., Int. Ed. 2011, 50, 5331–5334. doi:10.1002/anie.201101522 |
| 296. | Komiya, N.; Noji, S.; Murahashi, S.-I. Chem. Commun. 2001, 65–66. doi:10.1039/B006869L |
| 297. | Nomura, K.; Uemura, S. J. Chem. Soc., Chem. Commun. 1994, 129–130. doi:10.1039/C39940000129 |
| 194. | Uyanik, M.; Suzuki, D.; Yasui, T.; Ishihara, K. Angew. Chem., Int. Ed. 2011, 50, 5331–5334. doi:10.1002/anie.201101522 |
| 297. | Nomura, K.; Uemura, S. J. Chem. Soc., Chem. Commun. 1994, 129–130. doi:10.1039/C39940000129 |
| 298. | Deno, N. C.; Messer, L. A. J. Chem. Soc., Chem. Commun. 1976, 1051a. doi:10.1039/C3976001051A |
| 299. | Moody, C. J.; O’Connell, J. L. Chem. Commun. 2000, 1311–1312. doi:10.1039/B003074K |
| 196. | Guo, S.; Yu, J.-T.; Dai, Q.; Yang, H.; Cheng, J. Chem. Commun. 2014, 50, 6240–6242. doi:10.1039/C4CC01652A |
| 300. | Tran, B. L.; Driess, M.; Hartwig, J. F. J. Am. Chem. Soc. 2014, 136, 17292–17301. doi:10.1021/ja510093x |
| 298. | Deno, N. C.; Messer, L. A. J. Chem. Soc., Chem. Commun. 1976, 1051a. doi:10.1039/C3976001051A |
| 299. | Moody, C. J.; O’Connell, J. L. Chem. Commun. 2000, 1311–1312. doi:10.1039/B003074K |
| 44. | Kamal, A.; Srinivasulu, V.; Sathish, M.; Tangella, Y.; Nayak, V. L.; Rao, M. P. N.; Shankaraiah, N.; Nagesh, N. Asian J. Org. Chem. 2014, 3, 68–76. doi:10.1002/ajoc.201300214 |
| 45. | Wang, Z.; Kuang, C. Adv. Synth. Catal. 2014, 356, 1549–1554. doi:10.1002/adsc.201400096 |
| 42. | Zheng, X.; Song, B.; Xu, B. Eur. J. Org. Chem. 2010, 4376–4380. doi:10.1002/ejoc.201000631 |
| 296. | Komiya, N.; Noji, S.; Murahashi, S.-I. Chem. Commun. 2001, 65–66. doi:10.1039/B006869L |
| 43. | Leng, Y.; Yang, F.; Zhu, W.; Wu, Y.; Li, X. Org. Biomol. Chem. 2011, 9, 5288–5296. doi:10.1039/C1OB05223C |
| 40. | Chen, X.; Hao, X.-S.; Goodhue, C. E.; Yu, J.-Q. J. Am. Chem. Soc. 2006, 128, 6790–6791. doi:10.1021/ja061715q |
| 41. | Dudkina, Y. B.; Mikhaylov, D. Y.; Gryaznova, T. V.; Tufatullin, A. I.; Kataeva, O. N.; Vicic, D. A.; Budnikova, Y. H. Organometallics 2013, 32, 4785–4792. doi:10.1021/om400492g |
| 38. | Bian, Y.-J.; Xiang, C.-B.; Chen, Z.-M.; Huang, Z.-Z. Synlett 2011, 2407–2409. doi:10.1055/s-0030-1261224 |
| 39. | Rout, S. K.; Guin, S.; Gogoi, A.; Majji, G.; Patel, B. K. Org. Lett. 2014, 16, 1614–1617. doi:10.1021/ol500224e |
© 2015 Krylov et al; licensee Beilstein-Institut.
This is an Open Access article under the terms of the Creative Commons Attribution License (http://creativecommons.org/licenses/by/2.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
The license is subject to the Beilstein Journal of Organic Chemistry terms and conditions: (http://www.beilstein-journals.org/bjoc)