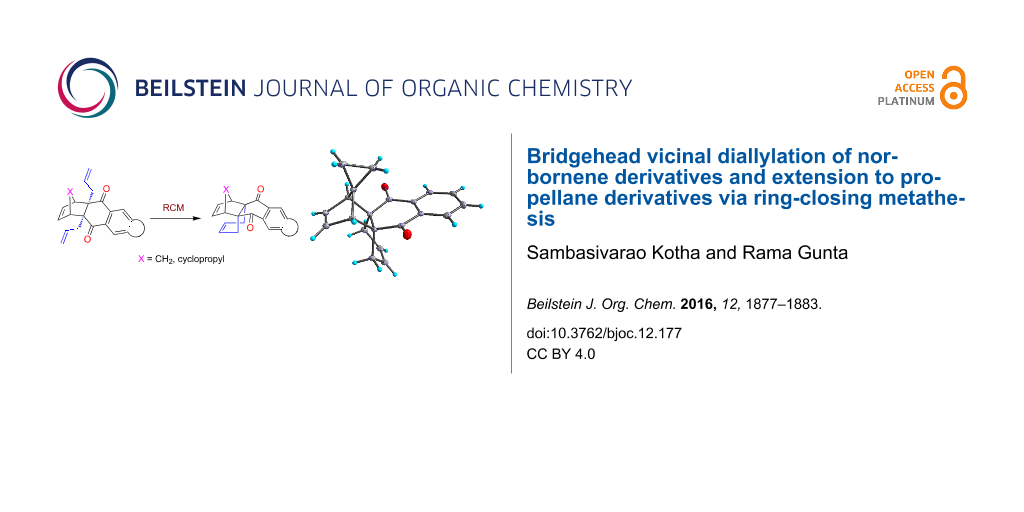
Abstract
Here, we report a simple synthetic strategy to the bridgehead vicinal diallylation of norbornene derivatives. These substrates are useful to generate propellanes via ring-closing metathesis. Single-crystal X-ray diffraction analysis of four compounds led to the realization of configurational correction of earlier reported molecules.
Graphical Abstract
Introduction
The norbornene moiety is a useful template and also a versatile synthon in organic synthesis [1]. The double bond present in the norbornene frame is strained and therefore participates in cycloaddition sequences as a C2-synthon [2,3]. It was reported that the norbornene system is as strained as cyclopropane or cyclobutane (norbornene, 100 kJ/mol; cyclopropane, 115 kJ/mol; cyclobutane, 110 kJ/mol) [4,5]. Some of the annulated norbornene derivatives undergo retro Diels–Alder (rDA) reactions at ambient temperature in the presence of methylaluminium dichloride and a reactive dienophile [6-8]. Cage compounds with interesting applications have been assembled by a cyclization reaction starting with suitably functionalized norbornene derivatives [9-11]. Moreover, the norbornene unit induces a hairpin-like architecture when it is incorporated into a peptide chain. This property is useful to design norbornene-based ionophores [12]. Due to the strained nature of norbornene systems they are useful precursors for ring-rearrangement metathesis (RRM) [13-21] to generate intricate polycyclics involving non-traditional retrosynthetic routes. Recently, functionalization of unactivated aromatic C–H bonds was achieved by using palladium catalysts and norbornene (Catellani reaction) [22,23]. In view of these applications, the design and synthesis of vicinal diallylnorbornene derivatives is a worthwhile exercise. The double bond present in the allyl group can be further converted into various other useful functionalities for further synthetic manipulation by adopting the appropriate functional group transformations.
Strategy
Our approach to various propellane derivatives is shown in Figure 1. The target propellane 1 could be assembled from diallyl compound 2 via ring-closing metathesis (RCM) [24-32]. Whereas, the diallyl derivative 2 can be derived from a readily available Diels–Alder (DA) adduct 3 through an allylation sequence.
Figure 1: Retrosynthetic approach to propellane derivatives.
Figure 1: Retrosynthetic approach to propellane derivatives.
Results and Discussion
Installation of two C–C bonds to generate quaternary centers in a stereocontrolled manner in a single step is not a trivial exercise. Generally, it was accomplished by radical three-component coupling reactions or Michael-type additions of organocopper reagents starting with conjugated carbonyl compounds [33,34]. But, the resulting alkyl groups are in trans orientation. Our journey to propellane 1 synthesis (Figure 1) was commenced with the preparation of known DA adducts 3a, 3b, 3aa' and 3bb' [35-37]. In this regard, DA adduct 3a was treated with allyl bromide in the presence of NaH to obtain the corresponding O-allylated compound (70%) and C-allylated compound 2a (28%) by using our earlier reported method [38]. Next, diallyl compound 2a on RCM using Grubbs first generation (G-I) catalyst in CH2Cl2 at room temperature (rt) gave the desired propellane derivative 1a (61%) along with a minor amount of quinone derivative 4 (17%) (Scheme 1). The formation of quinone 4 can be explained on the basis that compound 2a underwent rDA and RCM in one-pot. Here, the compound 2a didn’t undergo RRM because a metallacyclobutane cannot be formed between the allyl and norbornene double bonds due to structural constraint [39] and moreover, we didn’t observe any ring-opening metathesis (ROM) product during RCM reaction. This may be due to the fact that sparging with an inert gas (N2 or Ar) during RCM process helps to accelerate the loss of ethylene and thus, prevents ROM [39].
Scheme 1: Synthesis of the propellane derivative 1a via RCM.
Scheme 1: Synthesis of the propellane derivative 1a via RCM.
Garratt and Hollowood reported that bridgehead functionalization of norbornene derivatives such as endo-5-norbornene-2,3-dicarboximide 5 gave bridgehead alkylated compound 6 with retention of configuration (Scheme 2) [40]. Based on this report, we expected the allyl groups introduced via alkylation sequence will occupy the exo position (see 2ab) because the bridgehead hydrogens in DA adduct 3a are in exo configuration. Thus, in the final compound 1a the newly formed 6-membered ring during RCM is supposed to be in the exo configuration.
Scheme 2: Garratts work on alkylation of norbornene with retention of configuration.
Scheme 2: Garratts work on alkylation of norbornene with retention of configuration.
To our surprise, single-crystal X-ray analysis of 1a revealed that the 6-membered ring (C28–C30–C31–C32–C33–C27) formed via RCM is in endo configuration as depicted in Figure 2.
![[1860-5397-12-177-2]](/bjoc/content/figures/1860-5397-12-177-2.png?scale=2.0&max-width=1024&background=FFFFFF)
Figure 2: The molecular structure of 1a, with displacement ellipsoids drawn at the 50% probability level.
Figure 2: The molecular structure of 1a, with displacement ellipsoids drawn at the 50% probability level.
At this point, we turned our attention to understand the configurational origin of the allyl groups in 2a. To understand whether compound 2a was formed by Claisen rearrangement (CR) of the corresponding O-allyl compound or by carbanion mediated C-allylation of the DA adduct 3a, we carried out the alkylation of compound 3a with n-propyl bromide in refluxing THF for 2 h. Here, di-O-propyl compound 7 was obtained in 36% yield along with a dehydrogenated compound 8 (20%, Scheme 3). Surprisingly, no C-alkylation product was observed from 3a. This result suggests that the C-allyl compound 2a was formed from the corresponding O-allyl compound via CR.
Scheme 3: Control experiment carried out to probe the configuration of 2a.
Scheme 3: Control experiment carried out to probe the configuration of 2a.
Based on the X-ray structure of 1a and the above observations, it is clear that the allyl groups in 2a are in endo configuration which can be explained as follows. Since the stereocenters are unaffected during the RCM sequence it is evident that the allyl groups present in 2a should be in endo configuration. To confirm the configuration of the allyl groups, the X-ray structure of previously reported oxa-bowl/propellane hybrid (15) [38] was also recorded and it is in agreement with the above findings (Figure 3). These results suggested the revision of earlier reported configuration of allyl groups. More specifically, various compounds (2ab, 2aa'b and 9a–15a) reported in our previous report [38] need configurational correction and the revised structures (2aa' and 9–15) are included in Table 1.
![[1860-5397-12-177-3]](/bjoc/content/figures/1860-5397-12-177-3.png?scale=2.0&max-width=1024&background=FFFFFF)
Figure 3: Crystal structure of compound 15 showing 50% displacement ellipsoids.
Figure 3: Crystal structure of compound 15 showing 50% displacement ellipsoids.
When the other previously prepared diallyl compound 2aa' [38] was subjected to RCM using G-I catalyst under similar reaction conditions the propellane derivative 1aa' was obtained in 79% yield (Scheme 4).
To expand the scope of this strategy, cyclopropyl containing diallyl products 2b and 2bb' were also prepared along similar lines starting with the corresponding DA adducts 3b and 3bb' [41]. Initially, the diallyl compound 2b was reacted with G-I catalyst to afford the desired propellane 1b in 86% yield (Scheme 5). Its structure has been established on the basis of spectroscopic data (1H NMR, 13C NMR and DEPT-135) and was further supported by HRMS data.
Scheme 5: RCM approach to the propellane derivative 1b.
Scheme 5: RCM approach to the propellane derivative 1b.
In addition, the configuration of 1b and 2b were unambiguously determined via single-crystal X-ray diffraction analysis (Figure 4). Based on this data it is clear that the bridgehead allyl groups in the RCM precursor 2b are in endo configuration. Subsequent RCM of diallyl compound 2b gave the ring-closing product 1b with retention of the configuration.
![[1860-5397-12-177-4]](/bjoc/content/figures/1860-5397-12-177-4.png?scale=2.0&max-width=1024&background=FFFFFF)
Figure 4: The molecular structures of the compounds 2b (left) and 1b (right) showing 30% displacement ellipsoids.
Figure 4: The molecular structures of the compounds 2b (left) and 1b (right) showing 30% displacement ellipso...
Similarly, staring with substrate 2bb', another propellane derivative 1bb' was synthesized using the same catalyst (i.e., G-I) in CH2Cl2 at rt. Here, along with the desired propellane 1bb' (79%) a minor amount of quinone derivative 16 (13%) was also generated due to a one-pot RCM-rDA sequence of 2bb' which is similar to the substrate 2a (Scheme 6). Compound 1bb' was characterized based on the 1H and 13C NMR, DEPT-135 and further supported by HRMS data. However, spectroscopic data of quinone 16 were identical with the literature values [41].
Scheme 6: Construction of the propellane derivative 1bb' using RCM.
Scheme 6: Construction of the propellane derivative 1bb' using RCM.
Conclusion
This methodology was found to be useful to synthesize various propellane derivatives containing a norbornene moiety by employing RCM sequence. Moreover, we have firmly established the configuration of allyl groups at bridgehead position of norbornene derivatives by single-crystal X-ray diffraction analysis. A control experiment with propyl bromide provided an insight into the reaction mechanism that the bridgehead allylation proceeds through enolization, O-allylation followed by CR and not via carbanion chemistry. This alternative strategy is useful to introduce vicinal diallyl groups in a cis orientation to generate propellane derivatives, which is a different protocol from previously reported methods where the two vicinal alkyl groups are introduced in trans orientation. In this study, we have also revised the configuration of our earlier reported molecules containing allyl groups and oxa-bowl/propellane hybrids. Since non-flattened molecules are implicated in biological systems, our results would be useful in drug design [42].
Supporting Information
Crystallographic data have been deposited with the Cambridge Crystallographic Data Centre (CCDC) as supplementary publication numbers CCDC 1475412 (1a), 1475453 (1b), 1475403 (2b) and 1451438 (15). Copies of the data can be obtained free of charge on application to the Director at CCDC, 12 Union Road, Cambridge CB2 1EZ, UK (FAX: (+44) 1223-336-033; email: deposit@ccdc.cam.ac.uk).
| Supporting Information File 1: Experimental procedures, characterization data, copies of 1H & 13C NMR for all new compounds and X-ray data of the compounds 1a, 1b, 2b and 15. | ||
| Format: PDF | Size: 1.1 MB | Download |
| Supporting Information File 2: Crystallographic information files of compounds 1a (CCDC 1475412), 1b (CCDC 1475453), 2b (CCDC 1475403) and 15 (CCDC 1451438). | ||
| Format: ZIP | Size: 489.0 KB | Download |
Acknowledgements
We thank the Department of Science and Technology (DST), New Delhi for the financial support and also thank Darshan Mhatre for his help in collecting the X-ray data and structure refinement. S. K. thanks the Department of Science and Technology for the award of a J. C. Bose fellowship and Praj industries for Chair Professorship (Green Chemistry). R. G. thanks the University Grants Commission (UGC), New Delhi for the award of a research fellowship.
References
-
Ghosh, S.; Banerjee, S. ARKIVOC 2002, No. vii, 8–20.
Return to citation in text: [1] -
Thansandote, P.; Hulcoop, D. G.; Langer, M.; Lautens, M. J. Org. Chem. 2009, 74, 1673–1678. doi:10.1021/jo802604g
Return to citation in text: [1] -
Parham, M. E.; Frazer, M. G.; Bradsher, C. K. J. Org. Chem. 1972, 37, 358–362. doi:10.1021/jo00968a005
Return to citation in text: [1] -
Engler, E. M.; Andose, J. D.; Schleyer, P. V. R. J. Am. Chem. Soc. 1973, 95, 8005–8025. doi:10.1021/ja00805a012
Return to citation in text: [1] -
North, M. ROMP of Norbornene Derivatives of Amino Esters and Amino Acids. In Ring Opening Metathesis Polymerisation and Related Chemistry: State of the Art and Visions for the New Century; Khosravi, E.; Szymanska-Buzar, T., Eds.; Kluwer Academic: Dordrecht, Netherlands, 2002; pp 157–166.
Return to citation in text: [1] -
Eddolls, J. P.; Iqbal, M.; Roberts, S. M.; Santoro, M. G. Tetrahedron 2004, 60, 2539–2550. doi:10.1016/j.tet.2004.01.047
Return to citation in text: [1] -
Grieco, P. A.; Abood, N. J. Org. Chem. 1989, 54, 6008–6010. doi:10.1021/jo00287a005
Return to citation in text: [1] -
Kotha, S.; Banerjee, S. RSC Adv. 2013, 3, 7642–7666. doi:10.1039/c3ra22762f
Return to citation in text: [1] -
Wu, H.-J.; Tsai, S.-H.; Chern, J.-H.; Lin, H.-C. J. Org. Chem. 1997, 62, 6367–6373. doi:10.1021/jo970348l
Return to citation in text: [1] -
Masusai, C.; Soorukram, D.; Kuhakarn, C.; Tuchinda, P.; Pakawatchai, C.; Saithong, S.; Reutrakul, V.; Pohmakotr, M. J. Org. Chem. 2015, 80, 1577–1592. doi:10.1021/jo502501v
Return to citation in text: [1] -
Kotha, S.; Dipak, M. K. Chem. – Eur. J. 2006, 12, 4446–4450. doi:10.1002/chem.200501366
Return to citation in text: [1] -
Ranganathan, D.; Haridas, V.; Kurur, S.; Nagaraj, R.; Bikshapathy, E.; Kunwar, A. C.; Sarma, A. V. S.; Vairamani, M. J. Org. Chem. 2000, 65, 365–374. doi:10.1021/jo9912045
Return to citation in text: [1] -
Holub, N.; Blechert, S. Chem. – Asian J. 2007, 2, 1064–1082. doi:10.1002/asia.200700072
Return to citation in text: [1] -
Kotha, S.; Meshram, M.; Khedkar, P.; Banerjee, S.; Deodhar, D. Beilstein J. Org. Chem. 2015, 11, 1833–1864. doi:10.3762/bjoc.11.199
Return to citation in text: [1] -
Malik, C. K.; Yadav, R. N.; Drew, M. G. B.; Ghosh, S. J. Org. Chem. 2009, 74, 1957–1963. doi:10.1021/jo802077t
Return to citation in text: [1] -
Kotha, S.; Gunta, R. Beilstein J. Org. Chem. 2015, 11, 1373–1378. doi:10.3762/bjoc.11.148
Return to citation in text: [1] -
Kotha, S.; Ravikumar, O. Beilstein J. Org. Chem. 2015, 11, 1259–1264. doi:10.3762/bjoc.11.140
Return to citation in text: [1] -
Kotha, S.; Ravikumar, O. Eur. J. Org. Chem. 2014, 5582–5590. doi:10.1002/ejoc.201402273
Return to citation in text: [1] -
Funel, J.-A.; Prunet, J. Synlett 2005, 235–238. doi:10.1055/s-2004-837200
Return to citation in text: [1] -
Kiss, L.; Kardos, M.; Forró, E.; Fülöp, F. Eur. J. Org. Chem. 2015, 2015, 1283–1289. doi:10.1002/ejoc.201403493
Return to citation in text: [1] -
Bose, S.; Ghosh, M.; Ghosh, S. J. Org. Chem. 2012, 77, 6345–6350. doi:10.1021/jo300945b
Return to citation in text: [1] -
Zhu, H.; Ye, C.; Chen, Z. Chin. J. Org. Chem. 2015, 35, 2291–2300. doi:10.6023/cjoc201505035
Return to citation in text: [1] -
Han, J.; Zhang, L.; Zhu, Y.; Zheng, Y.; Chen, X.; Huang, Z.-B.; Shi, D.-Q.; Zhao, Y. Chem. Commun. 2016, 52, 6903–6906. doi:10.1039/C6CC02384C
Return to citation in text: [1] -
Grubbs, R. H.; O'Leary, D. J., Eds. Handbook of Metathesis: Applications in Organic Synthesis, 2nd ed.; Wiley-VCH: Weinheim, Germany, 2015; Vol. 2.
Return to citation in text: [1] -
Cossy, J.; Arseniyadis, S.; Meyer, C., Eds. Metathesis in Natural Product Synthesis: Strategies, Substrates and Catalysts; Wiley-VCH: Weinheim, Germany, 2010.
Return to citation in text: [1] -
Grela, K., Ed. Olefin Metathesis: Theory and Practice; John Wiley & Sons: Hoboken, NJ, U.S.A., 2014.
Return to citation in text: [1] -
Kotha, S.; Krishna, N. G.; Halder, S.; Misra, S. Org. Biomol. Chem. 2011, 9, 5597–5624. doi:10.1039/c1ob05413a
Return to citation in text: [1] -
Kotha, S.; Mandal, K.; Deb, A. C.; Banerjee, S. Tetrahedron Lett. 2004, 45, 9603–9605. doi:10.1016/j.tetlet.2004.11.012
Return to citation in text: [1] -
Kotha, S.; Mandal, K.; Tiwari, A.; Mobin, S. M. Chem. – Eur. J. 2006, 12, 8024–8038. doi:10.1002/chem.200600540
Return to citation in text: [1] -
Kotha, S.; Mandal, K. Tetrahedron Lett. 2004, 45, 1391–1394. doi:10.1016/j.tetlet.2003.12.075
Return to citation in text: [1] -
Prunet, J. Eur. J. Org. Chem. 2011, 3634–3647. doi:10.1002/ejoc.201100442
Return to citation in text: [1] -
Ghosh, S.; Ghosh, S.; Sarkar, N. J. Chem. Sci. 2006, 118, 223–235. doi:10.1007/Bf02708281
Return to citation in text: [1] -
Keck, G. E.; Kordik, C. P. Tetrahedron Lett. 1993, 34, 6875–6876. doi:10.1016/S0040-4039(00)91818-5
Return to citation in text: [1] -
Perlmutter, P. Conjugate Addition Reactions in Organic Synthesis; Pergamon Press: Oxford, United Kingdom, 1992.
Return to citation in text: [1] -
Valderrama, J. A.; Espinoza, O.; González, M. F.; Tapia, R. A.; Rodríguez, J. A.; Theoduloz, C.; Schmeda-Hirschmann, G. Tetrahedron 2006, 62, 2631–2638. doi:10.1016/j.tet.2005.12.038
Return to citation in text: [1] -
Patney, H. K.; Paddon-Row, M. N. Synthesis 1986, 1986, 326–328. doi:10.1055/s-1986-31602
Return to citation in text: [1] -
Singh, V.; Raju, B. N. S. Indian J. Chem. 1996, 35B, 303–311.
Return to citation in text: [1] -
Kotha, S.; Gunta, R. Beilstein J. Org. Chem. 2015, 11, 1727–1731. doi:10.3762/bjoc.11.188
Return to citation in text: [1] [2] [3] [4] [5] [6] [7] -
van Lierop, B. J.; Lummiss, J. A. M.; Fogg, D. E. In Olefin Metathesis: Theory and Practice; Grela, K., Ed.; John Wiley & Sons: Hoboken, NJ, U.S.A., 2014; pp 85–152. doi:10.1002/9781118711613.ch3
Return to citation in text: [1] [2] -
Garratt, P. J.; Hollowood, F. J. Org. Chem. 1982, 47, 68–72. doi:10.1021/jo00340a014
Return to citation in text: [1] -
Kotha, S.; Gunta, R. Tetrahedron Lett. 2016, 57, 3021–3023. doi:10.1016/j.tetlet.2016.05.101
Return to citation in text: [1] [2] -
Torres, E.; Leiva, R.; Gazzarrini, S.; Rey-Carrizo, M.; Frigolé-Vivas, M.; Moroni, A.; Naesens, L.; Vázquez, S. ACS Med. Chem. Lett. 2014, 5, 831–836. doi:10.1021/ml500108s
Return to citation in text: [1]
| 38. | Kotha, S.; Gunta, R. Beilstein J. Org. Chem. 2015, 11, 1727–1731. doi:10.3762/bjoc.11.188 |
| 38. | Kotha, S.; Gunta, R. Beilstein J. Org. Chem. 2015, 11, 1727–1731. doi:10.3762/bjoc.11.188 |
| 38. | Kotha, S.; Gunta, R. Beilstein J. Org. Chem. 2015, 11, 1727–1731. doi:10.3762/bjoc.11.188 |
| 9. | Wu, H.-J.; Tsai, S.-H.; Chern, J.-H.; Lin, H.-C. J. Org. Chem. 1997, 62, 6367–6373. doi:10.1021/jo970348l |
| 10. | Masusai, C.; Soorukram, D.; Kuhakarn, C.; Tuchinda, P.; Pakawatchai, C.; Saithong, S.; Reutrakul, V.; Pohmakotr, M. J. Org. Chem. 2015, 80, 1577–1592. doi:10.1021/jo502501v |
| 11. | Kotha, S.; Dipak, M. K. Chem. – Eur. J. 2006, 12, 4446–4450. doi:10.1002/chem.200501366 |
| 40. | Garratt, P. J.; Hollowood, F. J. Org. Chem. 1982, 47, 68–72. doi:10.1021/jo00340a014 |
| 6. | Eddolls, J. P.; Iqbal, M.; Roberts, S. M.; Santoro, M. G. Tetrahedron 2004, 60, 2539–2550. doi:10.1016/j.tet.2004.01.047 |
| 7. | Grieco, P. A.; Abood, N. J. Org. Chem. 1989, 54, 6008–6010. doi:10.1021/jo00287a005 |
| 8. | Kotha, S.; Banerjee, S. RSC Adv. 2013, 3, 7642–7666. doi:10.1039/c3ra22762f |
| 38. | Kotha, S.; Gunta, R. Beilstein J. Org. Chem. 2015, 11, 1727–1731. doi:10.3762/bjoc.11.188 |
| 4. | Engler, E. M.; Andose, J. D.; Schleyer, P. V. R. J. Am. Chem. Soc. 1973, 95, 8005–8025. doi:10.1021/ja00805a012 |
| 5. | North, M. ROMP of Norbornene Derivatives of Amino Esters and Amino Acids. In Ring Opening Metathesis Polymerisation and Related Chemistry: State of the Art and Visions for the New Century; Khosravi, E.; Szymanska-Buzar, T., Eds.; Kluwer Academic: Dordrecht, Netherlands, 2002; pp 157–166. |
| 39. | van Lierop, B. J.; Lummiss, J. A. M.; Fogg, D. E. In Olefin Metathesis: Theory and Practice; Grela, K., Ed.; John Wiley & Sons: Hoboken, NJ, U.S.A., 2014; pp 85–152. doi:10.1002/9781118711613.ch3 |
| 42. | Torres, E.; Leiva, R.; Gazzarrini, S.; Rey-Carrizo, M.; Frigolé-Vivas, M.; Moroni, A.; Naesens, L.; Vázquez, S. ACS Med. Chem. Lett. 2014, 5, 831–836. doi:10.1021/ml500108s |
| 2. | Thansandote, P.; Hulcoop, D. G.; Langer, M.; Lautens, M. J. Org. Chem. 2009, 74, 1673–1678. doi:10.1021/jo802604g |
| 3. | Parham, M. E.; Frazer, M. G.; Bradsher, C. K. J. Org. Chem. 1972, 37, 358–362. doi:10.1021/jo00968a005 |
| 39. | van Lierop, B. J.; Lummiss, J. A. M.; Fogg, D. E. In Olefin Metathesis: Theory and Practice; Grela, K., Ed.; John Wiley & Sons: Hoboken, NJ, U.S.A., 2014; pp 85–152. doi:10.1002/9781118711613.ch3 |
| 24. | Grubbs, R. H.; O'Leary, D. J., Eds. Handbook of Metathesis: Applications in Organic Synthesis, 2nd ed.; Wiley-VCH: Weinheim, Germany, 2015; Vol. 2. |
| 25. | Cossy, J.; Arseniyadis, S.; Meyer, C., Eds. Metathesis in Natural Product Synthesis: Strategies, Substrates and Catalysts; Wiley-VCH: Weinheim, Germany, 2010. |
| 26. | Grela, K., Ed. Olefin Metathesis: Theory and Practice; John Wiley & Sons: Hoboken, NJ, U.S.A., 2014. |
| 27. | Kotha, S.; Krishna, N. G.; Halder, S.; Misra, S. Org. Biomol. Chem. 2011, 9, 5597–5624. doi:10.1039/c1ob05413a |
| 28. | Kotha, S.; Mandal, K.; Deb, A. C.; Banerjee, S. Tetrahedron Lett. 2004, 45, 9603–9605. doi:10.1016/j.tetlet.2004.11.012 |
| 29. | Kotha, S.; Mandal, K.; Tiwari, A.; Mobin, S. M. Chem. – Eur. J. 2006, 12, 8024–8038. doi:10.1002/chem.200600540 |
| 30. | Kotha, S.; Mandal, K. Tetrahedron Lett. 2004, 45, 1391–1394. doi:10.1016/j.tetlet.2003.12.075 |
| 31. | Prunet, J. Eur. J. Org. Chem. 2011, 3634–3647. doi:10.1002/ejoc.201100442 |
| 32. | Ghosh, S.; Ghosh, S.; Sarkar, N. J. Chem. Sci. 2006, 118, 223–235. doi:10.1007/Bf02708281 |
| 35. | Valderrama, J. A.; Espinoza, O.; González, M. F.; Tapia, R. A.; Rodríguez, J. A.; Theoduloz, C.; Schmeda-Hirschmann, G. Tetrahedron 2006, 62, 2631–2638. doi:10.1016/j.tet.2005.12.038 |
| 36. | Patney, H. K.; Paddon-Row, M. N. Synthesis 1986, 1986, 326–328. doi:10.1055/s-1986-31602 |
| 37. | Singh, V.; Raju, B. N. S. Indian J. Chem. 1996, 35B, 303–311. |
| 41. | Kotha, S.; Gunta, R. Tetrahedron Lett. 2016, 57, 3021–3023. doi:10.1016/j.tetlet.2016.05.101 |
| 22. | Zhu, H.; Ye, C.; Chen, Z. Chin. J. Org. Chem. 2015, 35, 2291–2300. doi:10.6023/cjoc201505035 |
| 23. | Han, J.; Zhang, L.; Zhu, Y.; Zheng, Y.; Chen, X.; Huang, Z.-B.; Shi, D.-Q.; Zhao, Y. Chem. Commun. 2016, 52, 6903–6906. doi:10.1039/C6CC02384C |
| 38. | Kotha, S.; Gunta, R. Beilstein J. Org. Chem. 2015, 11, 1727–1731. doi:10.3762/bjoc.11.188 |
| 41. | Kotha, S.; Gunta, R. Tetrahedron Lett. 2016, 57, 3021–3023. doi:10.1016/j.tetlet.2016.05.101 |
| 13. | Holub, N.; Blechert, S. Chem. – Asian J. 2007, 2, 1064–1082. doi:10.1002/asia.200700072 |
| 14. | Kotha, S.; Meshram, M.; Khedkar, P.; Banerjee, S.; Deodhar, D. Beilstein J. Org. Chem. 2015, 11, 1833–1864. doi:10.3762/bjoc.11.199 |
| 15. | Malik, C. K.; Yadav, R. N.; Drew, M. G. B.; Ghosh, S. J. Org. Chem. 2009, 74, 1957–1963. doi:10.1021/jo802077t |
| 16. | Kotha, S.; Gunta, R. Beilstein J. Org. Chem. 2015, 11, 1373–1378. doi:10.3762/bjoc.11.148 |
| 17. | Kotha, S.; Ravikumar, O. Beilstein J. Org. Chem. 2015, 11, 1259–1264. doi:10.3762/bjoc.11.140 |
| 18. | Kotha, S.; Ravikumar, O. Eur. J. Org. Chem. 2014, 5582–5590. doi:10.1002/ejoc.201402273 |
| 19. | Funel, J.-A.; Prunet, J. Synlett 2005, 235–238. doi:10.1055/s-2004-837200 |
| 20. | Kiss, L.; Kardos, M.; Forró, E.; Fülöp, F. Eur. J. Org. Chem. 2015, 2015, 1283–1289. doi:10.1002/ejoc.201403493 |
| 21. | Bose, S.; Ghosh, M.; Ghosh, S. J. Org. Chem. 2012, 77, 6345–6350. doi:10.1021/jo300945b |
| 38. | Kotha, S.; Gunta, R. Beilstein J. Org. Chem. 2015, 11, 1727–1731. doi:10.3762/bjoc.11.188 |
| 12. | Ranganathan, D.; Haridas, V.; Kurur, S.; Nagaraj, R.; Bikshapathy, E.; Kunwar, A. C.; Sarma, A. V. S.; Vairamani, M. J. Org. Chem. 2000, 65, 365–374. doi:10.1021/jo9912045 |
| 33. | Keck, G. E.; Kordik, C. P. Tetrahedron Lett. 1993, 34, 6875–6876. doi:10.1016/S0040-4039(00)91818-5 |
| 34. | Perlmutter, P. Conjugate Addition Reactions in Organic Synthesis; Pergamon Press: Oxford, United Kingdom, 1992. |
| 38. | Kotha, S.; Gunta, R. Beilstein J. Org. Chem. 2015, 11, 1727–1731. doi:10.3762/bjoc.11.188 |
© 2016 Kotha and Gunta; licensee Beilstein-Institut.
This is an Open Access article under the terms of the Creative Commons Attribution License (http://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
The license is subject to the Beilstein Journal of Organic Chemistry terms and conditions: (http://www.beilstein-journals.org/bjoc)