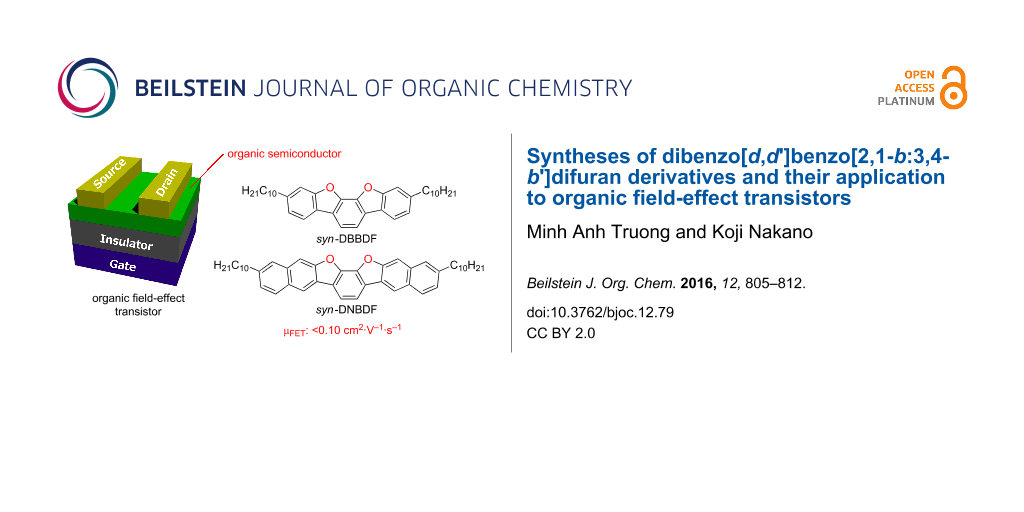
Abstract
Ladder-type π-conjugated compounds containing a benzo[2,1-b:3,4-b']difuran skeleton, such as dibenzo[d,d']benzo[2,1-b:3,4-b']difuran (syn-DBBDF) and dinaphtho[2,3-d:2',3'-d']benzo[2,1-b:3,4-b']difuran (syn-DNBDF) were synthesized. Their photophysical and electrochemical properties were revealed by UV–vis absorption and photoluminescence spectroscopy and cyclic voltammetry. Organic field-effect transistors (OFETs) were fabricated with these compounds as organic semiconductors, and their semiconducting properties were evaluated. OFETs with syn-DBBDF and syn-DNBDF showed typical p-type characteristics with hole mobilities of <1.5 × 10−3 cm2·V−1·s−1 and <1.0 × 10−1 cm2·V−1·s−1, respectively.
Graphical Abstract
Introduction
Organic semiconductors have significantly been developed in the past two decades by virtue of their advantages, such as low weight, flexibility, large-area processability, which are different features from conventional silicon-based semiconductors. Organic semiconducting materials can be used as active layers in organic field-effect transistors (OFETs) [1-7], organic light-emitting diodes (OLEDs) [8-10], and organic photovoltaics (OPVs) [11,12]. Among many organic semiconducting materials so far reported, thiophene-fused π-conjugated compounds have been widely studied as organic semiconducting materials and found to exhibit high semiconducting performances [5,13-16].
Furan-containing π-conjugated compounds have attracted less attention until recently [17-27]. The oxygen atom possesses a smaller van der Waals radius than a sulfur atom. Accordingly, furan-containing π-conjugated compounds should be expected to form a denser packing structure in the solid state, which is one of the main requirements for high semiconducting properties [28-31]. In 2007, Nakamura and co-workers reported the synthesis of furan-fused ladder-type π-conjugated compounds, benzo[1,2-b:4,5-b']difurans (BDFs) 1 and their application to OLEDs as hole-transporting materials (Figure 1) [32]. They also synthesized a series of isomeric BDFs (benzo[1,2-b:5,4-b']difurans and benzo[1,2-b:6,5-b']difurans) and studied their structure–property relationship [33,34]. Furthermore, naphthodifurans with a fused-naphthalene between two furan rings have been developed as organic semiconductors for OFETs [19,20]. In particular, the naphtho[2,1-b:6,5-b']difuran derivative 2 has been reported to demonstrate an excellent OFET mobility of 3.6 cm2·V−1·s−1 [19]. Previously, we have reported the synthesis of dibenzo[d,d']benzo[1,2-b:4,5-b']difurans (anti-DBBDFs), which is also a π-extended homologue of BDF [35]. The OFET devices with an anti-DBBDF skeleton exhibited p-type semiconducting properties [36,37]. For example, dialkyl-substituted anti-DBBDF 3 showed a hole mobility of 0.042 cm2·V−1·s−1 [38]. Recently, we have also found that dinaphtho[2,3-d:2',3'-d']benzo[1,2-b:4,5-b']difuran (anti-DNBDF 4) with a more extended π-conjugation afforded higher hole mobility of 0.33 cm2·V−1·s−1 [39-41]. These studies clearly demonstrate that furan-fused π-conjugated compounds are promising candidates as organic semiconducting materials, and it is highly desirable to investigate the structure−property relationship thoroughly for further development of furan-containing semiconducting materials.
Figure 1: Structures of furan-fused ladder-type π-conjugated compounds.
Figure 1: Structures of furan-fused ladder-type π-conjugated compounds.
Herein we report the synthesis of ladder-type π-conjugated compounds containing a benzo[2,1-b:3,4-b']difuran skeleton, such as dibenzo[d,d']benzo[2,1-b:3,4-b']difuran (syn-DBBDF 5) and dinaphtho[2,3-d:2',3'-d']benzo[2,1-b:3,4-b']difuran (syn-DNBDF 6, Figure 1) [42-46]. The physical and electrochemical properties of the synthesized compounds are also discussed. OFETs with these compounds as semiconducting layers were found to exhibit relatively high hole mobility of <1.0 × 10−1 cm2·V−1·s−1.
Results and Discussion
Synthesis
The synthetic routes to syn-DBBDF 5 and syn-DNBDF 6 are described in Scheme 1 and Scheme 2. 3-Decylanisole was first synthesized from commercially available 3-bromoanisole via iron-catalyzed cross-coupling reaction with decylmagnesium bromide in 71% yield [47]. Lithiation of the obtained 3-decylanisole with s-BuLi and the following treatment with 2-isopropoxy-4,4,5,5-tetramethyl-1,3,2-dioxaborolane (iPrO-Bpin) gave boronate ester 7 in 57% yield. Then, terphenyl 9 was synthesized via palladium-catalyzed Suzuki–Miyaura cross coupling of boronate ester 7 with 2,3-difluoro-1,4-diiodobenzene (96% yield) and subsequent demethylation (95% yield). Finally, the desired syn-DBBDF 5 was successfully synthesized via the double intramolecular cyclization under basic conditions at high temperature (92% yield) [37,43]. The same synthetic strategy was applied to the synthesis of syn-DNBDF (Scheme 2). 2-Decyl-7-methoxynaphthalene was prepared from 7-methoxynaphthalen-2-ol in two steps according to the literature [23,48], and used for the synthesis of boronate ester 10 (45% yield). The following cross coupling (80% yield), demethylation (85% yield), and the double cyclization (87% yield) gave the target syn-DNBDF 6. The obtained syn-DBBDF 5 is soluble in common organic solvents and can be purified by column chromatography. In contrast, because of low solubility in common organic solvents, the crude product of syn-DNBDF 6 was purified by washing several times with water and the subsequent sublimation.
Thermal properties
The phase-transition properties and thermal stability of syn-DBBDF 5 and syn-DNBDF 6 were evaluated by differential scanning calorimetry (DSC) and thermogravimetric analysis (TGA), respectively. The DSC scans of syn-DBBDF 5 and syn-DNBDF 6 showed some transition peaks with the first phase-transition temperature at 20 °C and 45 °C, respectively, in the heating process (Figure 2a). Such phase-transition temperatures are >50 °C lower than those of their anti-isomers 3 and 4 [38,39]. These results indicate that syn-DBBDF 5 and syn-DNBDF 6 form weaker intermolecular interactions in the solid state than their corresponding anti-isomers. The mesophase of syn-DBBDF 5 was converted to the isotropic phase at 115 °C, while syn-DNBDF 6 did not melt below 250 °C. From the TG measurement, the temperatures of 5% weight loss (Td5) of syn-DBBDF 5 and syn-DNBDF 6 were estimated to be 272 °C and 423 °C, respectively (Figure 2b).
![[1860-5397-12-79-2]](/bjoc/content/figures/1860-5397-12-79-2.png?scale=2.0&max-width=1024&background=FFFFFF)
Figure 2: (a) DSC and (b) TG curves of syn-DBBDF 5 and syn-DNBDF 6.
Figure 2: (a) DSC and (b) TG curves of syn-DBBDF 5 and syn-DNBDF 6.
Photophysical properties
The UV–vis spectrum of syn-DBBDF 5 in chloroform showed the strongest absorption maximum at 324 nm, while syn-DNBDF 6 showed a red-shifted absorption spectrum with the strongest absorption maximum at 365 nm (Figure 3a and Table 1). Since syn-DNBDF 6 contains one more benzene ring at each terminal of the π-conjugated skeleton than syn-DBBDF 5, it should possess an extended π-conjugation length, resulting in a red-shifted absorption spectrum. The HOMO–LUMO energy gaps estimated from the absorption edges were 3.72 eV and 3.32 eV for syn-DBBDF 5 and syn-DNBDF 6, respectively. Their photoluminescence spectra as shown in Figure 3b exhibited mirror images of their absorption spectra with small Stokes shifts (376 cm–1 for syn-DBBDF 5; 370 cm–1 for syn-DNBDF 6), which reflect their high rigidity. Similar to its absorption spectra, syn-DNBDF 6 showed a red-shifted emission band with a relatively high quantum yield (Φ = 61% in CHCl3 solution).
![[1860-5397-12-79-3]](/bjoc/content/figures/1860-5397-12-79-3.png?scale=2.0&max-width=1024&background=FFFFFF)
Figure 3: (a) UV–vis absorption spectra of syn-DBBDF 5 (blue line) and syn-DNBDF 6 (red line) in CHCl3 (1.0 × 10−5 M) and (b) normalized photoluminescence spectra of syn-DBBDF 5 (blue line) and syn-DNBDF 6 (red line) in CHCl3 (1.0 × 10−7 M).
Figure 3: (a) UV–vis absorption spectra of syn-DBBDF 5 (blue line) and syn-DNBDF 6 (red line) in CHCl3 (1.0 ×...
Table 1: Photophysical and electrochemical properties of syn/anti-DBBDFs and DNBDFs.
| Compound | λabs (nm)a | λem (nm)b | Φ (%)c | Stokes shift (cm–1) | Eg (eV)d | Eoxonset (V)e | EHOMO (eV)f |
|---|---|---|---|---|---|---|---|
| syn-DBBDF 5 | 324 | 328 | 18 | 376 | 3.72 | 0.84 | −5.64 |
| syn-DNBDF 6 | 365 | 370 | 61 | 370 | 3.32 | 0.56 | −5.36 |
| anti-DBBDF 3 | 342 | – | – | – | 3.51 | – | – |
| anti-DNBDF 4 | 394 | – | – | – | 3.15 | – | – |
aIn CHCl3 (1.0 × 10−5 M). bIn CHCl3 (1.0 × 10−7 M). Excitation at 310 nm. cAbsolute quantum yield determined by a calibrated integrating sphere system. Excitation at 275 nm for syn-DBBDF 5 and syn-DNBDF 6. dOptical band gaps estimated from the onset position of the UV–vis absorption spectra in solution. eOnset potentials (vs Fc/Fc+) of the first oxidation wave determined by cyclic voltammetry: 1.0 mM solution in CH2Cl2 (syn-DBBDF 5) or Cl2CHCHCl2 (syn-DNBDF 6) with 0.1 M Bu4NClO4, Pt as working and counter electrodes, scan rate = 50 mV·s−1. fCalculated according to EHOMO = −(Eox + 4.80) eV (Fc/Fc+ redox couple: 4.8 eV below the vacuum level).
To investigate the structure–property relationship of DBBDFs and DNBDFs, the optical properties of syn-DBBDF 5 and syn-DNBDF 6 were compared with those of anti-DBBDF 3 and anti-DNBDF 4. The UV–vis spectra of anti-DBBDF 3 and anti-DNBDF 4 were reported to show absorption maxima (342 nm for anti-DBBDF 3; 394 nm for anti-DNBDF 4) and absorption edges [353 nm (3.51 eV) for anti-DBBDF 3; 410 nm (3.15 eV) for anti-DNBDF 4] at longer wavelengths than syn-DBBDF 5 and syn-DNBDF 6, respectively [38,39]. Accordingly, syn-isomers are indicated to possess shorter π-conjugation lengths than anti-isomers.
Electrochemical properties
Cyclic voltammograms of syn-DBBDF 5 and syn-DNBDF 6 are shown in Figure 4 [1.0 mM solution in CH2Cl2 (syn-DBBDF 5) or Cl2CHCHCl2 (syn-DNBDF 6) with 0.10 M Bu4NClO4], and the electrochemical properties were summarized in Table 1. syn-DBBDF 5 exhibited two oxidation waves, and an onset potential of the first oxidation wave was determined to be 0.84 V (vs Fc/Fc+). Accordingly, the HOMO energy level was estimated to be −5.64 eV under the premise that the energy level of Fc/Fc+ is 4.8 eV below the vacuum level [49-51]. In contrast, syn-DNBDF 6 showed one oxidation wave with an onset potential of 0.56 eV (vs Fc/Fc+, HOMO = −5.36 eV). The lower oxidation potential and higher HOMO energy level of syn-DNBDF 6 should reflect its longer π-conjugation length than syn-DBBDF 5. Based on their HOMO energy levels and HOMO−LUMO energy gaps, syn-DBBDF 5 and syn-DNBDF 6 are expected to work as stable semiconducting materials under ambient conditions.
![[1860-5397-12-79-4]](/bjoc/content/figures/1860-5397-12-79-4.png?scale=2.0&max-width=1024&background=FFFFFF)
Figure 4: Cyclic voltammograms of syn-DBBDF 5 and syn-DNBDF 6 (measurement conditions: 1.0 mM in CH2Cl2 for syn-DBBDF 5 or Cl2CHCHCl2 for syn-DNBDF 6 with 0.1 M Bu4NClO4; Pt as working and counter electrodes; scan rate = 50 mV·s−1).
Figure 4: Cyclic voltammograms of syn-DBBDF 5 and syn-DNBDF 6 (measurement conditions: 1.0 mM in CH2Cl2 for s...
Fabrication of OFETs with syn-DBBDF- and syn-DNBDF-based thin films and evaluation of semiconducting properties
To study the semiconducting properties of syn-DBBDF 5 and syn-DNBDF 6, bottom-gate/top-contact OTFTs were utilized as a device structure. Thin films of syn-DBBDF 5 and syn-DNBDF 6 were deposited by sublimation under high vacuum (p < 10−5 Pa) at a rate of ca. 1 Å·s−1 for syn-DBBDF and ca. 0.4 Å·s−1 for syn-DNBDF onto the Si/SiO2 substrates. The substrate temperature (Tsub) during deposition has been known to have a great impact on the OTFT performance by affecting the nucleation and growth of the organic molecules [52,53]. Accordingly, the thin films were fabricated at different substrate temperatures. In addition to the bare Si/SiO2 substrates, the HMDS (hexamethyldisilazane)-treated substrates were used to evaluate the effect of the substrate structure on the device performance. The gold source/drain electrodes were deposited on the thin films. The channel width and length were 500 μm and 50 μm, respectively.
Both syn-DBBDF- and syn-DNBDF-based OFETs demonstrated typical p-type semiconducting characteristics. The extracted FET parameters and the transfer/output characteristics are summarized in Table 2, Figure 5, and Figure S21 (Supporting Information File 1). The syn-DBBDF-based OFETs fabricated on bare Si/SiO2 substrates at Tsub = 30 °C showed a field-effect mobility μFET of 5.0 × 10−5 cm2·V−1·s−1 and an Ion/Ioff ratio of 101, while those with HMDS-treated substrates demonstrated higher mobility of 1.5 × 10−3 cm2·V−1·s−1 with an Ion/Ioff ratio of 103. The deposition of syn-DBBDF 5 at Tsub = 60 °C did not give a thin film, which should be caused by re-sublimation of syn-DBBDF 5 from the surface. The more π-extended syn-DNBDF 6 afforded higher performances than syn-DBBDF 5. OFETs fabricated on the bare and HMDS-treated Si/SiO2 substrates at Tsub = 30 °C showed a field-effect mobility of 2.3 × 10−2 cm2·V−1·s−1 (Ion/Ioff = 103) and 2.0 × 10−2 cm2·V−1·s−1 (Ion/Ioff = 103), respectively. The FET performance also depends on the substrate temperature during thin-film fabrication. Thus, the highest hole mobility of 1.0 × 10−1 cm2·V−1·s−1 was obtained for the syn-DNBDF-based device fabricated on the HMDS-treated substrate at Tsub = 90 °C, while it was lower than that fabricated with anti-DNBDF derivatives [39].
![[1860-5397-12-79-5]](/bjoc/content/figures/1860-5397-12-79-5.png?scale=2.0&max-width=1024&background=FFFFFF)
Figure 5: Output and transfer characteristics of the representative OFETs with a thin film of (a) syn-DBBDF 5 (Tsub = 30 °C) and (b) syn-DNBDF 6 (Tsub = 90 °C) on HMDS-treated Si/SiO2 substrates.
Figure 5: Output and transfer characteristics of the representative OFETs with a thin film of (a) syn-DBBDF 5...
Analysis of thin films
The vapor-deposited thin films of syn-DBBDF 5 and syn-DNBDF 6 were analyzed by X-ray diffraction (XRD) and atomic force microscopy (AFM). Figure 6 shows the out-of-plane XRD pattern and an AFM image of the thin film of syn-DNBDF 6 on the HMDS-treated Si/SiO2 substrate (Tsub = 90 °C), which demonstrated the highest mobility in this study. The layer structure was confirmed with a monolayer thickness (d-spacing) of 3.94 nm (2θ = 2.24°). Molecular lengths with extended linear alkyl chains are expected to be ca. 4.2 nm. Accordingly, syn-DNBDF 6 should be arranged on the substrate with its molecular long axis almost perpendicular to the substrate. Such a layer structure was also confirmed by AFM. As shown in Figure 6b,c, the thin film of syn-DNBDF 6 forms relatively large grains (ca. 0.5 μm in size) with a layer structure (step heights ca. 4.0 nm) along with heterogeneous protrusions. The molecular arrangement indicated by these observations is advantageous for the in-plane charge transfer of OFETs. Based on XRD patterns and AFM images, the substrate treatment and the substrate temperature seem to have a limited impact on the molecular arrangement (Figures S22 and S23, Supporting Information File 1). The similar layer structure was also confirmed for syn-DBBDF 5 (Figures S22 and S23, Supporting Information File 1).
![[1860-5397-12-79-6]](/bjoc/content/figures/1860-5397-12-79-6.png?scale=2.0&max-width=1024&background=FFFFFF)
Figure 6: (a) XRD pattern, (b) AFM image (2 × 2 μm), and (c) cross-section height of a thin film of syn-DNBDF 6 on HMDS-treated Si/SiO2 substrates (Tsub = 90 °C).
Figure 6: (a) XRD pattern, (b) AFM image (2 × 2 μm), and (c) cross-section height of a thin film of syn-DNBDF ...
Conclusion
In summary, we investigated the synthesis and properties of ladder-type π-conjugated compounds, dibenzo[d,d']benzo[2,1-b:3,4-b']difuran (syn-DBBDF 5) and dinaphtho[2,3-d:2',3'-d']benzo[2,1-b:3,4-b']difuran (syn-DNBDF 6). Based on the photophysical and electrochemical data, both compounds are expected to possess good air stability as organic semiconducting materials. The comparison with their anti-isomers revealed that the π-conjugation in syn-DBBDF 5 and syn-DNBDF 6 is less effective than those of their anti-isomers. OFETs based on these compounds were fabricated as bottom-gate top-contact devices, and their semiconducting properties were evaluated. All devices showed typical p-type transistor characteristics. The highest hole mobility of 1.0 × 10−1 cm2·V−1·s−1 was achieved when using syn-DNBDF-based OFET device.
Supporting Information
| Supporting Information File 1: General experimental procedures, synthetic procedures/characterization data of compounds 5–12, device fabrication/evaluation procedures, OFET characteristics, XRD patterns, and AFM images. | ||
| Format: PDF | Size: 1.7 MB | Download |
Acknowledgements
This work was partially supported by MEXT KAKENHI Grant Number 26104510. A part of this work (XRD) was conducted in Nagoya University, supported by the Nanotechnology Platform Program (Molecule and Material Synthesis) of the Ministry of Education, Culture, Sports, Science and Technology (MEXT), Japan. We are grateful to Prof. Shusaku Nagano at Nagoya University for XRD analysis of thin films.
References
-
Anthony, J. E. Chem. Rev. 2006, 106, 5028–5048. doi:10.1021/cr050966z
Return to citation in text: [1] -
Bao, Z.; Locklin, J. Organic Field-Effect Transistors; CRC Press: Boca Raton, 2007. doi:10.1201/9781420008012
Return to citation in text: [1] -
Murphy, A. R.; Fréchet, J. M. J. Chem. Rev. 2007, 107, 1066–1096. doi:10.1021/cr0501386
Return to citation in text: [1] -
Allard, S.; Forster, M.; Souharce, B.; Thiem, H.; Scherf, U. Angew. Chem., Int. Ed. 2008, 47, 4070–4098. doi:10.1002/anie.200701920
Return to citation in text: [1] -
Takimiya, K.; Shinamura, S.; Osaka, I.; Miyazaki, E. Adv. Mater. 2011, 23, 4347–4370. doi:10.1002/adma.201102007
Return to citation in text: [1] [2] -
Wang, C.; Dong, H.; Hu, W.; Liu, Y.; Zhu, D. Chem. Rev. 2012, 112, 2208–2267. doi:10.1021/cr100380z
Return to citation in text: [1] -
Mei, J.; Diao, Y.; Appleton, A. L.; Fang, L.; Bao, Z. J. Am. Chem. Soc. 2013, 135, 6724–6746. doi:10.1021/ja400881n
Return to citation in text: [1] -
Kraft, A.; Grimsdale, A. C.; Holmes, A. B. Angew. Chem., Int. Ed. 1998, 37, 402–428. doi:10.1002/(SICI)1521-3773(19980302)37:4<402::AID-ANIE402>3.0.CO;2-9
Return to citation in text: [1] -
Kafafi, Z. Organic Electroluminescence; CRC Press: Boca Raton, 2005. doi:10.1201/9781420028201
Return to citation in text: [1] -
Müllen, K.; Scherf, U. Organic Light-Emitting Devices: Synthesis, Properties and Applications; Wiley-VCH Verlag GmbH & Co. KGaA: Weinheim, 2006. doi:10.1002/3527607986.fmatter
Return to citation in text: [1] -
Sun, S.-S.; Sariciftci, N. S. Organic Photovoltaics: Mechanism, Materials, and Devices; CRC Press: Boca Raton, 2005.
Return to citation in text: [1] -
Scharber, M. C.; Mühlbacher, D.; Koppe, M.; Denk, P.; Waldauf, C.; Heeger, A. J.; Brabec, C. J. Adv. Mater. 2006, 18, 789–794. doi:10.1002/adma.200501717
Return to citation in text: [1] -
Perepichka, I. F.; Perepichka, D. F. Handbook of Thiophene-Based Materials: Applications in Organic Electronics and Photonics; John Wiley & Sons Ltd.: Chichester, U.K., 2009. doi:10.1002/9780470745533
Return to citation in text: [1] -
Wu, W.; Liu, Y.; Zhu, D. Chem. Soc. Rev. 2010, 39, 1489–1502. doi:10.1039/B813123F
Return to citation in text: [1] -
Youn, J.; Kewalramani, S.; Emery, J. D.; Shi, Y.; Zhang, S.; Chang, H. C.; Liang, Y. J.; Yeh, C. M.; Feng, C. Y.; Huang, H.; Stern, C.; Chen, L. H.; Ho, J. C.; Chen, M. C.; Bedzyk, M. J.; Facchetti, A.; Marks, T. J. Adv. Funct. Mater. 2013, 23, 3850–3865. doi:10.1002/adfm.201203439
Return to citation in text: [1] -
Cinar, M. E.; Ozturk, T. Chem. Rev. 2015, 115, 3036–3140. doi:10.1021/cr500271a
Return to citation in text: [1] -
Li, H.; Jiang, P.; Yi, C.; Li, C.; Liu, S.-X.; Tan, S.; Zhao, B.; Braun, J.; Meier, W.; Wandlowski, T.; Decurtins, S. Macromolecules 2010, 43, 8058–8062. doi:10.1021/ma101693d
Return to citation in text: [1] -
Kobilka, B. M.; Dubrovskiy, A. V.; Ewan, M. D.; Tomlinson, A. L.; Larock, R. C.; Chaudhary, S.; Jeffries-El, M. Chem. Commun. 2012, 48, 8919–8921. doi:10.1039/c2cc34070d
Return to citation in text: [1] -
Mitsui, C.; Soeda, J.; Miwa, K.; Tsuji, H.; Takeya, J.; Nakamura, E. J. Am. Chem. Soc. 2012, 134, 5448–5451. doi:10.1021/ja2120635
Return to citation in text: [1] [2] [3] -
Nakano, M.; Mori, H.; Shinamura, S.; Takimiya, K. Chem. Mater. 2012, 24, 190–198. doi:10.1021/cm202853b
Return to citation in text: [1] [2] -
Watanabe, M.; Su, W.-T.; Chang, Y. J.; Chao, T.-H.; Wen, Y.-S.; Chow, T. J. Chem. – Asian J. 2013, 8, 60–64. doi:10.1002/asia.201200834
Return to citation in text: [1] -
Mitsui, C.; Okamoto, T.; Matsui, H.; Yamagishi, M.; Matsushita, T.; Soeda, J.; Miwa, K.; Sato, H.; Yamano, A.; Uemura, T.; Takeya, J. Chem. Mater. 2013, 25, 3952–3956. doi:10.1021/cm303376g
Return to citation in text: [1] -
Nakahara, K.; Mitsui, C.; Okamoto, T.; Yamagishi, M.; Matsui, H.; Ueno, T.; Tanaka, Y.; Yano, M.; Matsushita, T.; Soeda, J.; Hirose, Y.; Sato, H.; Yamano, A.; Takeya, J. Chem. Commun. 2014, 50, 5342–5344. doi:10.1039/C3CC47577H
Return to citation in text: [1] [2] -
Shi, S.; Xie, X.; Gao, C.; Shi, K.; Chen, S.; Yu, G.; Guo, L.; Li, X.; Wang, H. Macromolecules 2014, 47, 616–625. doi:10.1021/ma402107n
Return to citation in text: [1] -
Moussalem, C.; Segut, O.; Gohier, F.; Allain, M.; Frère, P. ACS Sustainable Chem. Eng. 2014, 2, 1043–1048. doi:10.1021/sc500042u
Return to citation in text: [1] -
Du, Z.; Chen, Y.; Chen, W.; Qiao, S.; Wen, S.; Liu, Q.; Zhu, D.; Sun, M.; Yang, R. Chem. – Asian J. 2014, 9, 2621–2627. doi:10.1002/asia.201402467
Return to citation in text: [1] -
Mitsui, C.; Soeda, J.; Miwa, K.; Shoyama, K.; Ota, Y.; Tsuji, H.; Takeya, J.; Nakamura, E. Bull. Chem. Soc. Jpn. 2015, 88, 776–783. doi:10.1246/bcsj.20150033
Return to citation in text: [1] -
Hutchison, G. R.; Ratner, M. A.; Marks, T. J. J. Am. Chem. Soc. 2005, 127, 16866–16881. doi:10.1021/ja0533996
Return to citation in text: [1] -
Gidron, O.; Diskin-Posner, Y.; Bendikov, M. J. Am. Chem. Soc. 2010, 132, 2148–2150. doi:10.1021/ja9093346
Return to citation in text: [1] -
Huang, J.-D.; Wen, S.-H.; Deng, W.-Q.; Han, K.-L. J. Phys. Chem. B 2011, 115, 2140–2147. doi:10.1021/jp108125q
Return to citation in text: [1] -
Gidron, O.; Dadvand, A.; Sheynin, Y.; Bendikov, M.; Perepichka, D. F. Chem. Commun. 2011, 47, 1976–1978. doi:10.1039/C0CC04699J
Return to citation in text: [1] -
Tsuji, H.; Mitsui, C.; Ilies, L.; Sato, Y.; Nakamura, E. J. Am. Chem. Soc. 2007, 129, 11902–11903. doi:10.1021/ja074365w
Return to citation in text: [1] -
Tsuji, H.; Mitsui, C.; Sato, Y.; Nakamura, E. Heteroat. Chem. 2011, 22, 316–324. doi:10.1002/hc.20682
Return to citation in text: [1] -
Mitsui, C.; Tanaka, H.; Tsuji, H.; Nakamura, E. Chem. – Asian J. 2011, 6, 2296–2300. doi:10.1002/asia.201100326
Return to citation in text: [1] -
Kawaguchi, K.; Nakano, K.; Nozaki, K. J. Org. Chem. 2007, 72, 5119–5128. doi:10.1021/jo070427p
Return to citation in text: [1] -
Kawaguchi, K. Synthesis of Heteroacenes and Their Applications to Organic Functional Materials. Ph.D. Thesis, The University of Tokyo, Tokyo, 2008.
Return to citation in text: [1] -
Kitamura, T.; Takaku, K.; Sotoyama, W. Organic thin film transistor having benzobisbenzofuran derivative-containing semiconductor active layer, organic semiconductor thin film, and organic semiconductor material. JP 2014045099, March 13, 2014.
Return to citation in text: [1] [2] -
Nakano, K.; Chayama, N.; Truong, M. A.; Kawaguchi, K.; Nozaki, K. Polym. Prepr., Jpn. 2011, 60, 4144–4145.
Return to citation in text: [1] [2] [3] -
Yamagata, Y.; Nakano, K. Abstracts of Papers. The 94th annual Meeting of The Chemical Society of Japan, Nagoya, March 27–30, 2014; The Chemical Society of Japan: Tokyo, 2014; 4A7-35.
Return to citation in text: [1] [2] [3] [4] -
Okamoto, T.; Mitsui, C.; Yamagishi, M.; Nakamura, K.-I.; Nakahara, K.; Soeda, J.; Hirose, Y.; Sato, H.; Yamano, A.; Takeya, J. Abstracts of Papers. The 61st JSAP Spring Meeting, Sagamihara, March 17–20, 2014; The Japan Society of Applied Physics, 2014; pp 12–260.
Return to citation in text: [1] -
Takeya, J.; Okamoto, T.; Mitsui, C.; Matsushita, T. Preparation of chalcogen-containing organic compounds as semiconductor materials. WO 2014136827, Sept 12, 2014.
Return to citation in text: [1] -
Solution-processed OFETs with syn-DBBDFs and syn-DNBDFs have been reported in the patent. However, the detailed description was not given for their synthesis, characterization, and physical properties. See reference [43].
Return to citation in text: [1] -
Kitamura, T.; Takaku, K.; Toyama, W. Organic thin-film transistor, benzobisbenzofuran compound, coating solution for non-emitting organic semiconductor device, organic semiconductor thin film, and organic semiconductor material. JP 2014082248, May 8, 2014.
Return to citation in text: [1] [2] [3] -
For π-conjugated molecules with a syn-DBBDF or syn-DNBDF skeleton, see references [45,46].
Return to citation in text: [1] -
Nakanishi, K.; Sasamori, T.; Kuramochi, K.; Tokitoh, N.; Kawabata, T.; Tsubaki, K. J. Org. Chem. 2014, 79, 2625–2631. doi:10.1021/jo500085a
Return to citation in text: [1] [2] -
Nakanishi, K.; Fukatsu, D.; Takaishi, K.; Tsuji, T.; Uenaka, K.; Kuramochi, K.; Kawabata, T.; Tsubaki, K. J. Am. Chem. Soc. 2014, 136, 7101–7109. doi:10.1021/ja502209w
Return to citation in text: [1] [2] -
Nakamura, M.; Matsuo, K.; Ito, S.; Nakamura, E. J. Am. Chem. Soc. 2004, 126, 3686–3687. doi:10.1021/ja049744t
Return to citation in text: [1] -
Mewshaw, R. E.; Edsall, R. J.; Yang, C.; Manas, E. S.; Xu, Z. B.; Henderson, R. A.; Keith, J. C., Jr.; Harris, H. A. J. Med. Chem. 2005, 48, 3953–3979. doi:10.1021/jm058173s
Return to citation in text: [1] -
Pommerehne, J.; Vestweber, H.; Guss, W.; Mahrt, R. F.; Bassler, H.; Porsch, M.; Daub, J. Adv. Mater. 1995, 7, 551–554. doi:10.1002/adma.19950070608
Return to citation in text: [1] -
Johansson, T.; Mammo, W.; Svensson, M.; Andersson, M. R.; Inganäs, O. J. Mater. Chem. 2003, 13, 1316–1323. doi:10.1039/B301403G
Return to citation in text: [1] -
Cardona, C. M.; Li, W.; Kaifer, A. E.; Stockdale, D.; Bazan, G. C. Adv. Mater. 2011, 23, 2367–2371. doi:10.1002/adma.201004554
Return to citation in text: [1] -
Kymissis, I. Organic Field Effect Transistors: Theory, Fabrication and Characterization; Springer Science+Business Media, LLC: New York, 2009.
Return to citation in text: [1] -
Ling, M. M.; Bao, Z. Chem. Mater. 2004, 16, 4824–4840. doi:10.1021/cm0496117
Return to citation in text: [1]
| 1. | Anthony, J. E. Chem. Rev. 2006, 106, 5028–5048. doi:10.1021/cr050966z |
| 2. | Bao, Z.; Locklin, J. Organic Field-Effect Transistors; CRC Press: Boca Raton, 2007. doi:10.1201/9781420008012 |
| 3. | Murphy, A. R.; Fréchet, J. M. J. Chem. Rev. 2007, 107, 1066–1096. doi:10.1021/cr0501386 |
| 4. | Allard, S.; Forster, M.; Souharce, B.; Thiem, H.; Scherf, U. Angew. Chem., Int. Ed. 2008, 47, 4070–4098. doi:10.1002/anie.200701920 |
| 5. | Takimiya, K.; Shinamura, S.; Osaka, I.; Miyazaki, E. Adv. Mater. 2011, 23, 4347–4370. doi:10.1002/adma.201102007 |
| 6. | Wang, C.; Dong, H.; Hu, W.; Liu, Y.; Zhu, D. Chem. Rev. 2012, 112, 2208–2267. doi:10.1021/cr100380z |
| 7. | Mei, J.; Diao, Y.; Appleton, A. L.; Fang, L.; Bao, Z. J. Am. Chem. Soc. 2013, 135, 6724–6746. doi:10.1021/ja400881n |
| 17. | Li, H.; Jiang, P.; Yi, C.; Li, C.; Liu, S.-X.; Tan, S.; Zhao, B.; Braun, J.; Meier, W.; Wandlowski, T.; Decurtins, S. Macromolecules 2010, 43, 8058–8062. doi:10.1021/ma101693d |
| 18. | Kobilka, B. M.; Dubrovskiy, A. V.; Ewan, M. D.; Tomlinson, A. L.; Larock, R. C.; Chaudhary, S.; Jeffries-El, M. Chem. Commun. 2012, 48, 8919–8921. doi:10.1039/c2cc34070d |
| 19. | Mitsui, C.; Soeda, J.; Miwa, K.; Tsuji, H.; Takeya, J.; Nakamura, E. J. Am. Chem. Soc. 2012, 134, 5448–5451. doi:10.1021/ja2120635 |
| 20. | Nakano, M.; Mori, H.; Shinamura, S.; Takimiya, K. Chem. Mater. 2012, 24, 190–198. doi:10.1021/cm202853b |
| 21. | Watanabe, M.; Su, W.-T.; Chang, Y. J.; Chao, T.-H.; Wen, Y.-S.; Chow, T. J. Chem. – Asian J. 2013, 8, 60–64. doi:10.1002/asia.201200834 |
| 22. | Mitsui, C.; Okamoto, T.; Matsui, H.; Yamagishi, M.; Matsushita, T.; Soeda, J.; Miwa, K.; Sato, H.; Yamano, A.; Uemura, T.; Takeya, J. Chem. Mater. 2013, 25, 3952–3956. doi:10.1021/cm303376g |
| 23. | Nakahara, K.; Mitsui, C.; Okamoto, T.; Yamagishi, M.; Matsui, H.; Ueno, T.; Tanaka, Y.; Yano, M.; Matsushita, T.; Soeda, J.; Hirose, Y.; Sato, H.; Yamano, A.; Takeya, J. Chem. Commun. 2014, 50, 5342–5344. doi:10.1039/C3CC47577H |
| 24. | Shi, S.; Xie, X.; Gao, C.; Shi, K.; Chen, S.; Yu, G.; Guo, L.; Li, X.; Wang, H. Macromolecules 2014, 47, 616–625. doi:10.1021/ma402107n |
| 25. | Moussalem, C.; Segut, O.; Gohier, F.; Allain, M.; Frère, P. ACS Sustainable Chem. Eng. 2014, 2, 1043–1048. doi:10.1021/sc500042u |
| 26. | Du, Z.; Chen, Y.; Chen, W.; Qiao, S.; Wen, S.; Liu, Q.; Zhu, D.; Sun, M.; Yang, R. Chem. – Asian J. 2014, 9, 2621–2627. doi:10.1002/asia.201402467 |
| 27. | Mitsui, C.; Soeda, J.; Miwa, K.; Shoyama, K.; Ota, Y.; Tsuji, H.; Takeya, J.; Nakamura, E. Bull. Chem. Soc. Jpn. 2015, 88, 776–783. doi:10.1246/bcsj.20150033 |
| 42. | Solution-processed OFETs with syn-DBBDFs and syn-DNBDFs have been reported in the patent. However, the detailed description was not given for their synthesis, characterization, and physical properties. See reference [43]. |
| 43. | Kitamura, T.; Takaku, K.; Toyama, W. Organic thin-film transistor, benzobisbenzofuran compound, coating solution for non-emitting organic semiconductor device, organic semiconductor thin film, and organic semiconductor material. JP 2014082248, May 8, 2014. |
| 44. | For π-conjugated molecules with a syn-DBBDF or syn-DNBDF skeleton, see references [45,46]. |
| 45. | Nakanishi, K.; Sasamori, T.; Kuramochi, K.; Tokitoh, N.; Kawabata, T.; Tsubaki, K. J. Org. Chem. 2014, 79, 2625–2631. doi:10.1021/jo500085a |
| 46. | Nakanishi, K.; Fukatsu, D.; Takaishi, K.; Tsuji, T.; Uenaka, K.; Kuramochi, K.; Kawabata, T.; Tsubaki, K. J. Am. Chem. Soc. 2014, 136, 7101–7109. doi:10.1021/ja502209w |
| 5. | Takimiya, K.; Shinamura, S.; Osaka, I.; Miyazaki, E. Adv. Mater. 2011, 23, 4347–4370. doi:10.1002/adma.201102007 |
| 13. | Perepichka, I. F.; Perepichka, D. F. Handbook of Thiophene-Based Materials: Applications in Organic Electronics and Photonics; John Wiley & Sons Ltd.: Chichester, U.K., 2009. doi:10.1002/9780470745533 |
| 14. | Wu, W.; Liu, Y.; Zhu, D. Chem. Soc. Rev. 2010, 39, 1489–1502. doi:10.1039/B813123F |
| 15. | Youn, J.; Kewalramani, S.; Emery, J. D.; Shi, Y.; Zhang, S.; Chang, H. C.; Liang, Y. J.; Yeh, C. M.; Feng, C. Y.; Huang, H.; Stern, C.; Chen, L. H.; Ho, J. C.; Chen, M. C.; Bedzyk, M. J.; Facchetti, A.; Marks, T. J. Adv. Funct. Mater. 2013, 23, 3850–3865. doi:10.1002/adfm.201203439 |
| 16. | Cinar, M. E.; Ozturk, T. Chem. Rev. 2015, 115, 3036–3140. doi:10.1021/cr500271a |
| 47. | Nakamura, M.; Matsuo, K.; Ito, S.; Nakamura, E. J. Am. Chem. Soc. 2004, 126, 3686–3687. doi:10.1021/ja049744t |
| 11. | Sun, S.-S.; Sariciftci, N. S. Organic Photovoltaics: Mechanism, Materials, and Devices; CRC Press: Boca Raton, 2005. |
| 12. | Scharber, M. C.; Mühlbacher, D.; Koppe, M.; Denk, P.; Waldauf, C.; Heeger, A. J.; Brabec, C. J. Adv. Mater. 2006, 18, 789–794. doi:10.1002/adma.200501717 |
| 38. | Nakano, K.; Chayama, N.; Truong, M. A.; Kawaguchi, K.; Nozaki, K. Polym. Prepr., Jpn. 2011, 60, 4144–4145. |
| 8. | Kraft, A.; Grimsdale, A. C.; Holmes, A. B. Angew. Chem., Int. Ed. 1998, 37, 402–428. doi:10.1002/(SICI)1521-3773(19980302)37:4<402::AID-ANIE402>3.0.CO;2-9 |
| 9. | Kafafi, Z. Organic Electroluminescence; CRC Press: Boca Raton, 2005. doi:10.1201/9781420028201 |
| 10. | Müllen, K.; Scherf, U. Organic Light-Emitting Devices: Synthesis, Properties and Applications; Wiley-VCH Verlag GmbH & Co. KGaA: Weinheim, 2006. doi:10.1002/3527607986.fmatter |
| 39. | Yamagata, Y.; Nakano, K. Abstracts of Papers. The 94th annual Meeting of The Chemical Society of Japan, Nagoya, March 27–30, 2014; The Chemical Society of Japan: Tokyo, 2014; 4A7-35. |
| 40. | Okamoto, T.; Mitsui, C.; Yamagishi, M.; Nakamura, K.-I.; Nakahara, K.; Soeda, J.; Hirose, Y.; Sato, H.; Yamano, A.; Takeya, J. Abstracts of Papers. The 61st JSAP Spring Meeting, Sagamihara, March 17–20, 2014; The Japan Society of Applied Physics, 2014; pp 12–260. |
| 41. | Takeya, J.; Okamoto, T.; Mitsui, C.; Matsushita, T. Preparation of chalcogen-containing organic compounds as semiconductor materials. WO 2014136827, Sept 12, 2014. |
| 19. | Mitsui, C.; Soeda, J.; Miwa, K.; Tsuji, H.; Takeya, J.; Nakamura, E. J. Am. Chem. Soc. 2012, 134, 5448–5451. doi:10.1021/ja2120635 |
| 20. | Nakano, M.; Mori, H.; Shinamura, S.; Takimiya, K. Chem. Mater. 2012, 24, 190–198. doi:10.1021/cm202853b |
| 35. | Kawaguchi, K.; Nakano, K.; Nozaki, K. J. Org. Chem. 2007, 72, 5119–5128. doi:10.1021/jo070427p |
| 33. | Tsuji, H.; Mitsui, C.; Sato, Y.; Nakamura, E. Heteroat. Chem. 2011, 22, 316–324. doi:10.1002/hc.20682 |
| 34. | Mitsui, C.; Tanaka, H.; Tsuji, H.; Nakamura, E. Chem. – Asian J. 2011, 6, 2296–2300. doi:10.1002/asia.201100326 |
| 36. | Kawaguchi, K. Synthesis of Heteroacenes and Their Applications to Organic Functional Materials. Ph.D. Thesis, The University of Tokyo, Tokyo, 2008. |
| 37. | Kitamura, T.; Takaku, K.; Sotoyama, W. Organic thin film transistor having benzobisbenzofuran derivative-containing semiconductor active layer, organic semiconductor thin film, and organic semiconductor material. JP 2014045099, March 13, 2014. |
| 32. | Tsuji, H.; Mitsui, C.; Ilies, L.; Sato, Y.; Nakamura, E. J. Am. Chem. Soc. 2007, 129, 11902–11903. doi:10.1021/ja074365w |
| 28. | Hutchison, G. R.; Ratner, M. A.; Marks, T. J. J. Am. Chem. Soc. 2005, 127, 16866–16881. doi:10.1021/ja0533996 |
| 29. | Gidron, O.; Diskin-Posner, Y.; Bendikov, M. J. Am. Chem. Soc. 2010, 132, 2148–2150. doi:10.1021/ja9093346 |
| 30. | Huang, J.-D.; Wen, S.-H.; Deng, W.-Q.; Han, K.-L. J. Phys. Chem. B 2011, 115, 2140–2147. doi:10.1021/jp108125q |
| 31. | Gidron, O.; Dadvand, A.; Sheynin, Y.; Bendikov, M.; Perepichka, D. F. Chem. Commun. 2011, 47, 1976–1978. doi:10.1039/C0CC04699J |
| 19. | Mitsui, C.; Soeda, J.; Miwa, K.; Tsuji, H.; Takeya, J.; Nakamura, E. J. Am. Chem. Soc. 2012, 134, 5448–5451. doi:10.1021/ja2120635 |
| 38. | Nakano, K.; Chayama, N.; Truong, M. A.; Kawaguchi, K.; Nozaki, K. Polym. Prepr., Jpn. 2011, 60, 4144–4145. |
| 39. | Yamagata, Y.; Nakano, K. Abstracts of Papers. The 94th annual Meeting of The Chemical Society of Japan, Nagoya, March 27–30, 2014; The Chemical Society of Japan: Tokyo, 2014; 4A7-35. |
| 37. | Kitamura, T.; Takaku, K.; Sotoyama, W. Organic thin film transistor having benzobisbenzofuran derivative-containing semiconductor active layer, organic semiconductor thin film, and organic semiconductor material. JP 2014045099, March 13, 2014. |
| 43. | Kitamura, T.; Takaku, K.; Toyama, W. Organic thin-film transistor, benzobisbenzofuran compound, coating solution for non-emitting organic semiconductor device, organic semiconductor thin film, and organic semiconductor material. JP 2014082248, May 8, 2014. |
| 23. | Nakahara, K.; Mitsui, C.; Okamoto, T.; Yamagishi, M.; Matsui, H.; Ueno, T.; Tanaka, Y.; Yano, M.; Matsushita, T.; Soeda, J.; Hirose, Y.; Sato, H.; Yamano, A.; Takeya, J. Chem. Commun. 2014, 50, 5342–5344. doi:10.1039/C3CC47577H |
| 48. | Mewshaw, R. E.; Edsall, R. J.; Yang, C.; Manas, E. S.; Xu, Z. B.; Henderson, R. A.; Keith, J. C., Jr.; Harris, H. A. J. Med. Chem. 2005, 48, 3953–3979. doi:10.1021/jm058173s |
| 43. | Kitamura, T.; Takaku, K.; Toyama, W. Organic thin-film transistor, benzobisbenzofuran compound, coating solution for non-emitting organic semiconductor device, organic semiconductor thin film, and organic semiconductor material. JP 2014082248, May 8, 2014. |
| 45. | Nakanishi, K.; Sasamori, T.; Kuramochi, K.; Tokitoh, N.; Kawabata, T.; Tsubaki, K. J. Org. Chem. 2014, 79, 2625–2631. doi:10.1021/jo500085a |
| 46. | Nakanishi, K.; Fukatsu, D.; Takaishi, K.; Tsuji, T.; Uenaka, K.; Kuramochi, K.; Kawabata, T.; Tsubaki, K. J. Am. Chem. Soc. 2014, 136, 7101–7109. doi:10.1021/ja502209w |
| 52. | Kymissis, I. Organic Field Effect Transistors: Theory, Fabrication and Characterization; Springer Science+Business Media, LLC: New York, 2009. |
| 53. | Ling, M. M.; Bao, Z. Chem. Mater. 2004, 16, 4824–4840. doi:10.1021/cm0496117 |
| 39. | Yamagata, Y.; Nakano, K. Abstracts of Papers. The 94th annual Meeting of The Chemical Society of Japan, Nagoya, March 27–30, 2014; The Chemical Society of Japan: Tokyo, 2014; 4A7-35. |
| 38. | Nakano, K.; Chayama, N.; Truong, M. A.; Kawaguchi, K.; Nozaki, K. Polym. Prepr., Jpn. 2011, 60, 4144–4145. |
| 39. | Yamagata, Y.; Nakano, K. Abstracts of Papers. The 94th annual Meeting of The Chemical Society of Japan, Nagoya, March 27–30, 2014; The Chemical Society of Japan: Tokyo, 2014; 4A7-35. |
| 49. | Pommerehne, J.; Vestweber, H.; Guss, W.; Mahrt, R. F.; Bassler, H.; Porsch, M.; Daub, J. Adv. Mater. 1995, 7, 551–554. doi:10.1002/adma.19950070608 |
| 50. | Johansson, T.; Mammo, W.; Svensson, M.; Andersson, M. R.; Inganäs, O. J. Mater. Chem. 2003, 13, 1316–1323. doi:10.1039/B301403G |
| 51. | Cardona, C. M.; Li, W.; Kaifer, A. E.; Stockdale, D.; Bazan, G. C. Adv. Mater. 2011, 23, 2367–2371. doi:10.1002/adma.201004554 |
© 2016 Truong and Nakano; licensee Beilstein-Institut.
This is an Open Access article under the terms of the Creative Commons Attribution License (http://creativecommons.org/licenses/by/2.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
The license is subject to the Beilstein Journal of Organic Chemistry terms and conditions: (http://www.beilstein-journals.org/bjoc)