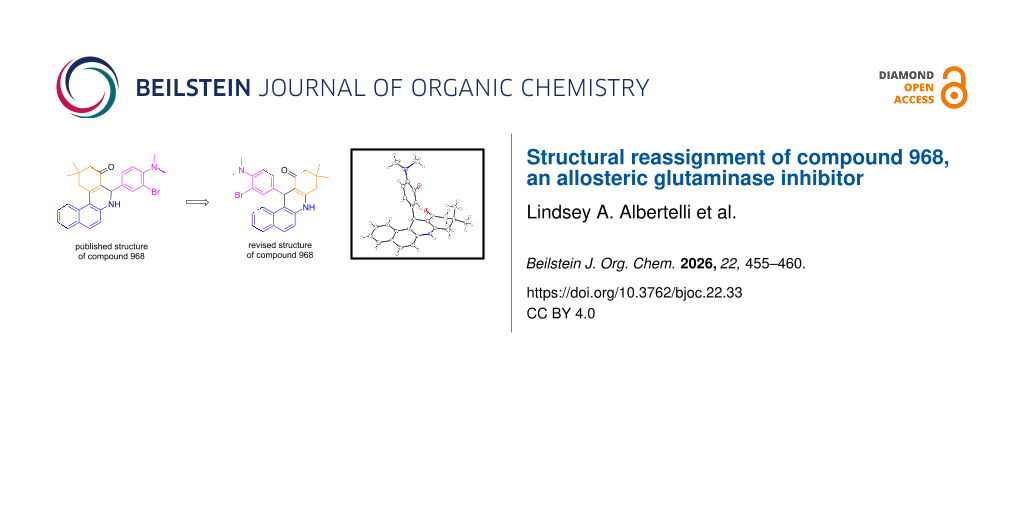
Abstract
Many cancer cells require extracellular glutamine to meet the energetic, biosynthetic, and redox demands of the proliferative state. Glutaminases catalyze the hydrolysis of glutamine to glutamate, which supports the biosynthesis of amino acids, lipids, and glutathione and can also be oxidatively deaminated to α-ketoglutarate and enter the citric acid cycle. The “glutamine addiction” of cancer cells has made glutaminase an attractive anticancer drug target. Compound 968 is a glutaminase inhibitor that is widely used to probe cancer cells’ dependence on glutaminase activity. Here, we show by NMR spectroscopy and X-ray crystallography that the reported benzo[c]phenanthridine structure of compound 968 is incorrect; its true structure is the isomeric benzo[c]acridine. The structural reassignment of compound 968 will aid the medicinal chemistry development of this important compound.
Graphical Abstract
Introduction
Cancer cells often show a strong reliance on glutamine uptake and metabolism [1,2]. Glutamine serves as a nitrogen donor for the biosynthesis of asparagine, amino sugars, and nucleotides. Glutamine is also hydrolyzed to glutamate by glutaminase (GLS) enzymes, which are often overexpressed in cancer cells. The resulting glutamate supports the biosynthesis of glutathione and can be oxidatively deaminated to the TCA cycle intermediate α-ketoglutarate for energy production and additional biosynthetic pathways. There are two glutaminase isozymes in humans: KGA is encoded by the GLS1 gene and is expressed mainly in the kidney and brain and LGA encoded by the GLS2 gene is expressed mainly in the liver. GAC is a highly active splice variant of KGA often expressed in cancer cells [3]. GLS1 and GLS2 enzymes share similar sequences, tetrameric structures, and enzymatic stimulation by phosphate [4-6].
The reliance of cancer cells on glutamine to meet the energetic, biosynthetic, and redox demands of the proliferative state creates an opportunity to selectively target cancer cells [1,7]. Chemical inhibition of GLS has emerged as an attractive anticancer strategy, and several classes of GLS inhibitors have been discovered [1,2]. DON is a diazo-containing electrophilic glutamine analog that inhibits glutaminase by covalently labeling the catalytic serine [8]. DON inhibits other glutamine-utilizing enzymes such as asparagine synthetase and has activity against a wide range of cancer cell lines [9,10]. BPTES is a potent allosteric inhibitor of GAC/KGA that binds at the dimer–dimer interface and stabilizes a catalytically inactive form of the enzyme [11,12]. The BPTES scaffold has undergone significant medicinal chemistry development resulting in CB-839/telaglenastat, which is being evaluated in clinical trials in combination with other anticancer agents [13]. Compound 968 is a third class of glutaminase inhibitor discovered in a screen for compounds able to revert the transformed phenotype of cells harboring oncogenic Dbl, the nucleotide exchange factor of the small GTPase Rho [14]. Compound 968 is a dual GLS1/GLS2 inhibitor and has found wide use to probe the effects of glutaminase inhibition on several cancer cell lines (Figure 1) [15,16]. The anticancer effects of compound 968 have been tested in combination with other drugs such as paclitaxel [17], erlotinib [18], apigenin [19], metformin [20], and inhibitors of tissue transglutaminase [21]. Compound 968 was recently found to suppress the growth of a luminal breast cancer cell line that expresses GLS2 and is insensitive to GLS1-specific inhibitors such as BPTES and CB-839 [22].
Figure 1: The glutaminase enzymes GLS1 and GLS2 catalyze the hydrolysis of glutamine to glutamate and ammonia. Compound 968 is a dual inhibitor of GLS1 and GLS2.
Figure 1: The glutaminase enzymes GLS1 and GLS2 catalyze the hydrolysis of glutamine to glutamate and ammonia...
After initiating a project to synthesize derivatives of compound 968, we searched the literature for synthetic routes towards its scaffold. Compound 968 is a benzo[c]phenanthridine, made by a three-component cyclocondensation reaction between the 1,3 diketone dimedone, the aryl aldehyde 3-bromo-4-dimethylaminobenzaldehyde, and 2-naphthylamine. This reaction producing the benzo[c]phenanthridine core was first reported in the late 1960s [23,24] and the substrate scope of diketone, arylamine, and arylaldehyde was widened over the subsequent decades [25-27]. However, later reports by Martinez et al. [28] using X-ray crystallography and Kozlov et al. [29,30] using NOESY NMR showed that this three-component cyclocondensation reaction does not produce benzo[c]phenanthridine 1 as product, but instead yields the isomeric benzo[c]acridine 2 (Figure 2).
Figure 2: The cycloaddition reaction between an arylaldehyde (shown here is the specific aldehyde that produces compound 968), dimedone, and 2-naphthylamine was originally reported to produce benzo[c]phenanthridine 1, of which the commonly accepted structure of 968 is a member. The structure of the product of this reaction was subsequently reported to be the isomeric benzo[c]acridine 2.
Figure 2: The cycloaddition reaction between an arylaldehyde (shown here is the specific aldehyde that produc...
Upon learning of these results, we became concerned that the accepted structure of compound 968 is incorrect. We then sought to determine whether compound 968 is a benzo[c]phenanthridine 1 or benzo[c]acridine 2. Clarifying this issue would benefit the community of cancer biologists who use compound 968, enable medicinal chemistry around the compound 968 scaffold, as well as correct the structure displayed by vendors of this compound.
Results and Discussion
To determine whether the structure of compound 968 is a benzo[c]phenanthridine 1 or benzo[c]acridine 2 we synthesized it by the reported cyclocondensation reaction, which consists of heating an equimolar mixture of 3-bromo-4-dimethylaminobenzaldehyde, 2-naphthylamine, and dimedone to reflux in 1-butanol. We also purchased compound 968 from Sigma (catalog # 352010; CAS # 311795-38-7) which is advertised as the benzo[c]phenanthridine isomer 1. We also purchased material advertised as benzo[c]acridine isomer 2, which is available from ChemDiv (catalog # 8012-8736; CAS # 442660-67-5). Based on the results of Martinez and Kozlov reassigning the structure of the cycloaddition reaction product from benzo[c]phenanthridine 1 to benzo[c]acridine 2, we predicted that all three compounds are the benzo[c]acridine isomer 2.
The 1H NMR spectra of all three compounds are identical, indicating that they share the same structure (Figure 3A). We assayed each compound for inhibitory activity against the GAC isoform of glutaminase and found that the three compounds inhibited it with equal potency, further suggesting that the compounds are identical (Figure 3B). To unambiguously determine the structure of this compound, we grew crystals of our synthetic material and solved the structure by X-ray crystallography. The structure that fits the diffraction data is the benzo[c]acridine isomer 2 (Figure 3C). From this, we conclude that the structure of compound 968 is not the commonly accepted benzo[c]phenanthridine 1 but is instead the benzo[c]acridine isomer 2 in agreement with the reports of Martinez and Koslov. Since the only reaction reported to give the benzo[c]phenanthridine isomer 1 is the three-component reaction that instead produces the isomeric benzo[c]acridine 2, we believe that the synthesis of benzo[c]phenanthridine isomer 1 remains unrealized.
Figure 3: A) 1H NMR spectra of compound 968 purchased from Sigma-Aldrich (top), the same compound synthesized using the published cyclocondensation reaction (middle), and the isomeric benzo[c]acridine compound purchased from ChemDiv. B) Dose–response curves of the three compounds against the glutaminase isoform GAC. C) ORTEP diagram of the X-ray crystal structure of the material synthesized according to the published cyclocondensation reaction.
Figure 3: A) 1H NMR spectra of compound 968 purchased from Sigma-Aldrich (top), the same compound synthesized...
This result places the cyclocondensation reaction that generates compound 968 within a family of three-component reactions of 1,3 dicarbonyl compounds, an aryl aldehyde, and various 1,3 di-nucleophiles (Figure 4). These reactions proceed by an aldol condensation between the dicarbonyl and aldehyde, followed by conjugate addition and cyclo-condensation with a 1,3 dinucleophile. Members of this reaction family are variations on the Biginelli and Hantzch reactions, where the dinucleophile is urea and the ammonia adduct of a second equivalent of the dicarbonyl, respectively [31]. A similar reaction with 2-naphthol as the dinucleophile is also known [32]. In all cases, the regiochemistry of cyclization is selective and no isomeric products are formed.
Figure 4: The cyclocondensation reaction that produces compound 968 and related compounds.
Figure 4: The cyclocondensation reaction that produces compound 968 and related compounds.
Conclusion
In this report, we confirm the finding by Martinez [28] and Kozlov [29,30] that the three-component reaction originally claimed to generate benzo[c]phenanthridines [23,24] instead generates the isomeric benzo[c]acridines and show that this correction applies to compound 968, a prominent member of this class. The compound library that 968 originated from may have been structurally annotated before the reports of Martinez and Koslov were published or without knowledge of these results. The structure reassignment of compound 968 should aid in medicinal chemistry efforts around this scaffold with increased potency and improved pharmacological properties.
Experimental
General
X-ray data were obtained on a Bruker Smart Breeze CCD diffractometer. Nuclear magnetic resonance spectra were recorded on a JEOL ECX400 spectrometer. Glutaminase assays were measured on a Tecan Infinite M nano spectrophotometer. Reagents and solvents for chemical synthesis were used without additional purification.
Chemical synthesis
3-Bromo-4-dimethylaminobenzaldehyde
4-Dimethylaminobenzaldehyde (4.02 g, 26.9 mmol) was dissolved in 1,4-dioxane (52 mL) and NBS (5.01 g, 28.2 mmol) was added in small portions with stirring over 5 minutes. The reaction mixture was stirred at room temperature for 20 minutes then poured into 50 mL water. The mixture was diluted with EtOAc (70 mL), which was separated and washed with water (3 × 50 mL). The organic layer was dried with anhydrous MgSO4, filtered, and the solvent removed by rotary evaporation to yield 3-bromo-4-dimethylaminobenzaldehyde as a thick gold oil (5.49 g, 89%). 1H NMR (400 MHz, CDCl3) δ 9.78 (s, 1H), 8.01 (m, 1H), 7.71 (dd, J = 8.7, 2.3 Hz, 1H), 7.04 (d, J = 8.7 Hz, 1H), 2.93 (s, 6H); 13C NMR (100 MHz, CDCl3) δ 189.7, 156.9, 136.1, 131.1, 130.0, 119.5, 116.7, 43.6.
12-(3-Bromo-4-(dimethylamino)phenyl)-8,9,10,12-tetrahydro-9,9-dimethylbenzo[a]acridin-11(7H)-one (2); suggested revised structure for compound 968
3-Bromo-4-dimethylaminobenzaldehyde (2.71 g, 11.8 mmol), 2-naphthylamine (1.70 g, 11.8 mmol), and dimedone (1.66 g, 11.8 mmol) were dissolved in 1-butanol (40 mL) and heated to reflux for 3 hours, during which time a precipitate formed. This precipitate was collected by filtration and washed with ethanol to yield the title compound as a light gray powder (4.05 g, 72%). 1H NMR (400 MHz, DMSO-d6) δ 9.72 (s, 1H), 7.91 (d, J = 8.7 Hz, 2H), 7.76 (t, J = 6.9 Hz, 2H), 7.41 (t, J = 7.3 Hz, 1H), 7.35 (d, J = 1.8 Hz, 1H), 7.29 (m, 2H), 7.71 (dd, J = 8.3, 1.8 Hz, 1H), 6.90 (d, J = 8.2 Hz, 1H), 5.70 (s, 1H), 2.51 (s, 6H), 2.39 (d, J = 16.5 H, 1Hz), 2.18 (d, J = 16.0 Hz, 1H), 2.02 (d, J = 16.0 Hz, 1H), 0.99 (s, 3H), 0.85 (s, 3H). One diastereotopic methylene hydrogen is obscured by the residual solvent peak at 2.47 ppm; 13C NMR (100 MHz, DMSO-d6) δ 193.72, 151.37, 149.50, 143.73, 134.80, 132.80, 131.69, 130.92, 129.02, 128.68, 128.12, 127.49, 124.25, 122.93, 120.88, 118.45, 117.59, 116.53, 107.43, 50.77, 44.22, 35.30, 32.80, 29.56, 27.06.
X-ray crystallography
Crystals of compound 2 were grown by slow evaporation from a saturated acetone solution. A yellow, multi-faceted block of suitable size (0.362 × 0.124 × 0.064 mm3) and quality was selected from a representative sample of crystals of the same habit using an optical microscope, mounted onto a Mitegen MicroLoopsTM (MiTeGen, LLC., Ithaca, NY) and placed in a cold nitrogen stream of nitrogen. Low temperature (100 K) X-ray data were obtained on a Bruker Smart Breeze CCD diffractometer (Mo sealed X-ray tube, Kα = 0.71073 Å). All diffractometer manipulations, including data collection, integration and scaling were carried out using the Bruker APEXII software. The structure was solved using SHELXS [33] and was refined using SHELXL [34]. Olex2 was employed for the final data presentation and structure plots [35].
The crystal data have been deposited in the Cambridge Crystallographic Data Centre, deposition number CCDC 2426481.
Enzymatic assay
Expression and purification of the GAC isoform of GLS1
Overnight cultures of BL21(DE3) strains carrying GAC cloned into pQE80 were grown at 37 °C overnight in LB supplemented with 100 µg/mL ampicillin. These cultures were diluted 1:100 into 2 L of LB media supplemented with 100 µg/mL ampicillin and grown at 37 °C with shaking at 225 rpm until the OD600 reached 0.6. IPTG was added to a final concentration of 0.5 mM and the cultures were shaken at 17 °C for 18 hours. The cells were harvested by centrifugation and stored at −80 °C. The cell pellets were lysed in 20 mL B-PER complete (ThermoFisher) supplemented with an EDTA-free protease inhibitor tablet (Roche) according to the manufacturer’s instructions. The supernatant was applied to 2 mL of Ni:NTA resin (Thermo Scientific) equilibrated with wash buffer (50 mM Tris (pH 8.0), 500 mM NaCl) and rocked at 4 °C for 30 minutes. The beads were transferred to a disposable column and drained. The bead bed was washed at 4 °C with 30 mL of wash buffer and 20 mL of wash buffer supplemented with 10 mM imidazole. Protein was eluted into 1 mL fractions at 4 °C with wash buffer supplemented with 300 mM imidazole. A 20 µL aliquot of each fraction was analyzed by SDS-PAGE. Fractions with pure protein were pooled and the buffer was changed to 20 mM Tris (pH 8.5), 120 mM NaCl using a PD-10 desalting column (Cytiva). The eluent was divided into aliquots, snap frozen in liquid nitrogen, and stored at −80 °C.
Glutaminase enzymatic assay
The GAC isoform of glutaminase (79 µL, 50 nM) in Tris acetate (65 mM, pH 8.6) was added to flat, clear-bottomed 96 well plates. A solution of the test compound in DMSO or DMSO itself was added (1 µL), mixed by gently pipetting up and down, then incubated for seven minutes at room temperature. The glutaminase reaction was initiated by addition of 20 µL of a solution of glutamine (100 mM) and K2HPO4 (500 mM), then mixed by gently pipetting up and down and incubated at room temperature for seven minutes. The reactions were quenched by addition of cold 3 M HCl (10 µL). An aliquot (10 µL) of each quenched GAC reaction was added to 190 µL of a glutamate dehydrogenase reaction which consisted of a solution containing Tris-HCl (100 mM, pH 9.4), NAD+ (2 mM), glutamate dehydrogenase (1 µL of a 50% glycerol solution, 15 mg/mL), and hydrazine (0.5 µL) and incubated at room temperature for 40 min. The absorbance at 340 nm was measured and converted to glutamate concentration using the extinction coefficient for NADH of 6220 M−1 cm−1.
Data Availability Statement
All data that supports the findings of this study is available in the published article and/or the supporting information of this article. The crystal data have been deposited in the Cambridge Crystallographic Data Centre (https://www.ccdc.cam.ac.uk/) deposition number CCDC 2426481.
References
-
Hensley, C. T.; Wasti, A. T.; DeBerardinis, R. J. J. Clin. Invest. 2013, 123, 3678–3684. doi:10.1172/jci69600
Return to citation in text: [1] [2] [3] -
Yang, W.-H.; Qiu, Y.; Stamatatos, O.; Janowitz, T.; Lukey, M. J. Trends Cancer 2021, 7, 790–804. doi:10.1016/j.trecan.2021.04.003
Return to citation in text: [1] [2] -
van den Heuvel, A. P. J.; Jing, J.; Wooster, R. F.; Bachman, K. E. Cancer Biol. Ther. 2012, 13, 1185–1194. doi:10.4161/cbt.21348
Return to citation in text: [1] -
Li, Y.; Erickson, J. W.; Stalnecker, C. A.; Katt, W. P.; Huang, Q.; Cerione, R. A.; Ramachandran, S. J. Biol. Chem. 2016, 291, 20900–20910. doi:10.1074/jbc.m116.720268
Return to citation in text: [1] -
Ferreira, I. M.; Quesñay, J. E. N.; Bastos, A. C. S.; Rodrigues, C. T.; Vollmar, M.; Krojer, T.; Strain-Damerell, C.; Burgess-Brown, N. A.; von Delft, F.; Yue, W. W.; Dias, S. M. G.; Ambrosio, A. L. B. Biochimie 2021, 185, 96–104. doi:10.1016/j.biochi.2021.03.009
Return to citation in text: [1] -
Nguyen, T.-T. T.; Ramachandran, S.; Hill, M. J.; Cerione, R. A. J. Biol. Chem. 2022, 298, 101564. doi:10.1016/j.jbc.2022.101564
Return to citation in text: [1] -
Jin, J.; Byun, J.-K.; Choi, Y.-K.; Park, K.-G. Exp. Mol. Med. 2023, 55, 706–715. doi:10.1038/s12276-023-00971-9
Return to citation in text: [1] -
Thangavelu, K.; Chong, Q. Y.; Low, B. C.; Sivaraman, J. Sci. Rep. 2014, 4, 3827. doi:10.1038/srep03827
Return to citation in text: [1] -
Novotná, K.; Tenora, L.; Slusher, B. S.; Rais, R. Adv. Pharmacol. (San Diego, CA, U. S.) 2024, 100, 157–180. doi:10.1016/bs.apha.2024.04.003
Return to citation in text: [1] -
Recouvreux, M. V.; Grenier, S. F.; Zhang, Y.; Esparza, E.; Lambies, G.; Galapate, C. M.; Maganti, S.; Duong-Polk, K.; Bhullar, D.; Naeem, R.; Scott, D. A.; Lowy, A. M.; Tiriac, H.; Commisso, C. Nat. Cancer 2023, 5, 100–113. doi:10.1038/s43018-023-00649-1
Return to citation in text: [1] -
Thangavelu, K.; Pan, C. Q.; Karlberg, T.; Balaji, G.; Uttamchandani, M.; Suresh, V.; Schüler, H.; Low, B. C.; Sivaraman, J. Proc. Natl. Acad. Sci. U. S. A. 2012, 109, 7705–7710. doi:10.1073/pnas.1116573109
Return to citation in text: [1] -
Robinson, M. M.; Mcbryant, S. J.; Tsukamoto, T.; Rojas, C.; Ferraris, D. V.; Hamilton, S. K.; Hansen, J. C.; Curthoys, N. P. Biochem. J. 2007, 406, 407–414. doi:10.1042/bj20070039
Return to citation in text: [1] -
Telaglenastat, CB-839·HCl; ClinicalTrials.gov; National Center for Biotechnology Information; National Library of Medicine. https://clinicaltrials.gov/search?intr=Telaglenastat (accessed March 2, 2026).
Return to citation in text: [1] -
Wang, J.-B.; Erickson, J. W.; Fuji, R.; Ramachandran, S.; Gao, P.; Dinavahi, R.; Wilson, K. F.; Ambrosio, A. L. B.; Dias, S. M. G.; Dang, C. V.; Cerione, R. A. Cancer Cell 2010, 18, 207–219. doi:10.1016/j.ccr.2010.08.009
Return to citation in text: [1] -
Xi, J.; Sun, Y.; Zhang, M.; Fa, Z.; Wan, Y.; Min, Z.; Xu, H.; Xu, C.; Tang, J. Exp. Cell Res. 2019, 381, 1–9. doi:10.1016/j.yexcr.2019.04.005
Return to citation in text: [1] -
Han, T.; Guo, M.; Zhang, T.; Gan, M.; Xie, C.; Wang, J.-B. Oncotarget 2017, 8, 28063–28073. doi:10.18632/oncotarget.14188
Return to citation in text: [1] -
Yuan, L.; Sheng, X.; Clark, L. H.; Zhang, L.; Guo, H.; Jones, H. M.; Willson, A. K.; Gehrig, P. A.; Zhou, C.; Bae-Jump, V. L. Am. J. Transl. Res. 2016, 8, 4265–4277.
Return to citation in text: [1] -
Xie, C.; Jin, J.; Bao, X.; Zhan, W.-H.; Han, T.-Y.; Gan, M.; Zhang, C.; Wang, J. Oncotarget 2016, 7, 610–621. doi:10.18632/oncotarget.6311
Return to citation in text: [1] -
Lee, Y.-M.; Lee, G.; Oh, T.-I.; Kim, B. M.; Shim, D.-W.; Lee, K.-H.; Kim, Y. J.; Lim, B. O.; Lim, J.-H. Int. J. Oncol. 2016, 48, 399–408. doi:10.3892/ijo.2015.3243
Return to citation in text: [1] -
Kim, J. H.; Lee, K. J.; Seo, Y.; Kwon, J.-H.; Yoon, J. P.; Kang, J. Y.; Lee, H. J.; Park, S. J.; Hong, S. P.; Cheon, J. H.; Kim, W. H.; Il Kim, T. Sci. Rep. 2018, 8, 409. doi:10.1038/s41598-017-18762-4
Return to citation in text: [1] -
Katt, W. P.; Antonyak, M. A.; Cerione, R. A. Mol. Pharmaceutics 2015, 12, 46–55. doi:10.1021/mp500405h
Return to citation in text: [1] -
Lukey, M. J.; Cluntun, A. A.; Katt, W. P.; Lin, M.-c. J.; Druso, J. E.; Ramachandran, S.; Erickson, J. W.; Le, H. H.; Wang, Z.-E.; Blank, B.; Greene, K. S.; Cerione, R. A. Cell Rep. 2019, 29, 76–88.e7. doi:10.1016/j.celrep.2019.08.076
Return to citation in text: [1] -
Lielbriedis, I. E.; Chirkova, V. V.; Gudrinietse, E. Izv. Akad. Nauk Latv. SSR, Ser. Khim. 1969, 193–196.
Return to citation in text: [1] [2] -
Kozlov, N. S.; Nugumanov, Z. Z. Izv. Akad. Nauk BSSR, Ser. Khim. Nauk 1968, 67–72.
Return to citation in text: [1] [2] -
Gusak, K. N.; Tereshko, A. B.; Kozlov, N. G.; Shakailo, N. I. Russ. J. Gen. Chem. 2000, 70, 1793–1800.
Return to citation in text: [1] -
Kozlov, N. G.; Gusak, K. N. Russ. J. Org. Chem. 1999, 35, 402–414.
Return to citation in text: [1] -
Kozlov, N. G.; Gusak, K. N.; Bezborodov, V. S. Russ. J. Org. Chem. 2000, 36, 88–92.
Return to citation in text: [1] -
Martínez, R.; Cortés, E.; Toscano, R. A.; Linzaga, I. J. Heterocycl. Chem. 1990, 27, 363–366. doi:10.1002/jhet.5570270248
Return to citation in text: [1] [2] -
Kozlov, N. G.; Gusak, K. N.; Tkachev, A. V. Chem. Heterocycl. Compd. 2007, 43, 740–747. doi:10.1007/s10593-007-0120-z
Return to citation in text: [1] [2] -
Kozlov, N. G.; Gusak, K. N. Russ. J. Gen. Chem. 2006, 76, 278–281. doi:10.1134/s1070363206020204
Return to citation in text: [1] [2] -
Nikoofar, K.; Yielzoleh, F. M. J. Saudi Chem. Soc. 2018, 22, 715–741. doi:10.1016/j.jscs.2017.12.005
Return to citation in text: [1] -
Das, B.; Laxminarayana, K.; Krishnaiah, M.; Srinivas, Y. Synlett 2007, 3107–3112. doi:10.1055/s-2007-990922
Return to citation in text: [1] -
Sheldrick, G. M. Acta Crystallogr., Sect. A: Found. Crystallogr. 2008, 64, 112–122. doi:10.1107/s0108767307043930
Return to citation in text: [1] -
Sheldrick, G. M. Acta Crystallogr., Sect. C: Struct. Chem. 2015, 71, 3–8. doi:10.1107/s2053229614024218
Return to citation in text: [1] -
Dolomanov, O. V.; Bourhis, L. J.; Gildea, R. J.; Howard, J. A. K.; Puschmann, H. J. Appl. Crystallogr. 2009, 42, 339–341. doi:10.1107/s0021889808042726
Return to citation in text: [1]
| 34. | Sheldrick, G. M. Acta Crystallogr., Sect. C: Struct. Chem. 2015, 71, 3–8. doi:10.1107/s2053229614024218 |
| 35. | Dolomanov, O. V.; Bourhis, L. J.; Gildea, R. J.; Howard, J. A. K.; Puschmann, H. J. Appl. Crystallogr. 2009, 42, 339–341. doi:10.1107/s0021889808042726 |
| 1. | Hensley, C. T.; Wasti, A. T.; DeBerardinis, R. J. J. Clin. Invest. 2013, 123, 3678–3684. doi:10.1172/jci69600 |
| 2. | Yang, W.-H.; Qiu, Y.; Stamatatos, O.; Janowitz, T.; Lukey, M. J. Trends Cancer 2021, 7, 790–804. doi:10.1016/j.trecan.2021.04.003 |
| 1. | Hensley, C. T.; Wasti, A. T.; DeBerardinis, R. J. J. Clin. Invest. 2013, 123, 3678–3684. doi:10.1172/jci69600 |
| 2. | Yang, W.-H.; Qiu, Y.; Stamatatos, O.; Janowitz, T.; Lukey, M. J. Trends Cancer 2021, 7, 790–804. doi:10.1016/j.trecan.2021.04.003 |
| 20. | Kim, J. H.; Lee, K. J.; Seo, Y.; Kwon, J.-H.; Yoon, J. P.; Kang, J. Y.; Lee, H. J.; Park, S. J.; Hong, S. P.; Cheon, J. H.; Kim, W. H.; Il Kim, T. Sci. Rep. 2018, 8, 409. doi:10.1038/s41598-017-18762-4 |
| 1. | Hensley, C. T.; Wasti, A. T.; DeBerardinis, R. J. J. Clin. Invest. 2013, 123, 3678–3684. doi:10.1172/jci69600 |
| 7. | Jin, J.; Byun, J.-K.; Choi, Y.-K.; Park, K.-G. Exp. Mol. Med. 2023, 55, 706–715. doi:10.1038/s12276-023-00971-9 |
| 21. | Katt, W. P.; Antonyak, M. A.; Cerione, R. A. Mol. Pharmaceutics 2015, 12, 46–55. doi:10.1021/mp500405h |
| 4. | Li, Y.; Erickson, J. W.; Stalnecker, C. A.; Katt, W. P.; Huang, Q.; Cerione, R. A.; Ramachandran, S. J. Biol. Chem. 2016, 291, 20900–20910. doi:10.1074/jbc.m116.720268 |
| 5. | Ferreira, I. M.; Quesñay, J. E. N.; Bastos, A. C. S.; Rodrigues, C. T.; Vollmar, M.; Krojer, T.; Strain-Damerell, C.; Burgess-Brown, N. A.; von Delft, F.; Yue, W. W.; Dias, S. M. G.; Ambrosio, A. L. B. Biochimie 2021, 185, 96–104. doi:10.1016/j.biochi.2021.03.009 |
| 6. | Nguyen, T.-T. T.; Ramachandran, S.; Hill, M. J.; Cerione, R. A. J. Biol. Chem. 2022, 298, 101564. doi:10.1016/j.jbc.2022.101564 |
| 18. | Xie, C.; Jin, J.; Bao, X.; Zhan, W.-H.; Han, T.-Y.; Gan, M.; Zhang, C.; Wang, J. Oncotarget 2016, 7, 610–621. doi:10.18632/oncotarget.6311 |
| 3. | van den Heuvel, A. P. J.; Jing, J.; Wooster, R. F.; Bachman, K. E. Cancer Biol. Ther. 2012, 13, 1185–1194. doi:10.4161/cbt.21348 |
| 19. | Lee, Y.-M.; Lee, G.; Oh, T.-I.; Kim, B. M.; Shim, D.-W.; Lee, K.-H.; Kim, Y. J.; Lim, B. O.; Lim, J.-H. Int. J. Oncol. 2016, 48, 399–408. doi:10.3892/ijo.2015.3243 |
| 13. | Telaglenastat, CB-839·HCl; ClinicalTrials.gov; National Center for Biotechnology Information; National Library of Medicine. https://clinicaltrials.gov/search?intr=Telaglenastat (accessed March 2, 2026). |
| 15. | Xi, J.; Sun, Y.; Zhang, M.; Fa, Z.; Wan, Y.; Min, Z.; Xu, H.; Xu, C.; Tang, J. Exp. Cell Res. 2019, 381, 1–9. doi:10.1016/j.yexcr.2019.04.005 |
| 16. | Han, T.; Guo, M.; Zhang, T.; Gan, M.; Xie, C.; Wang, J.-B. Oncotarget 2017, 8, 28063–28073. doi:10.18632/oncotarget.14188 |
| 11. | Thangavelu, K.; Pan, C. Q.; Karlberg, T.; Balaji, G.; Uttamchandani, M.; Suresh, V.; Schüler, H.; Low, B. C.; Sivaraman, J. Proc. Natl. Acad. Sci. U. S. A. 2012, 109, 7705–7710. doi:10.1073/pnas.1116573109 |
| 12. | Robinson, M. M.; Mcbryant, S. J.; Tsukamoto, T.; Rojas, C.; Ferraris, D. V.; Hamilton, S. K.; Hansen, J. C.; Curthoys, N. P. Biochem. J. 2007, 406, 407–414. doi:10.1042/bj20070039 |
| 17. | Yuan, L.; Sheng, X.; Clark, L. H.; Zhang, L.; Guo, H.; Jones, H. M.; Willson, A. K.; Gehrig, P. A.; Zhou, C.; Bae-Jump, V. L. Am. J. Transl. Res. 2016, 8, 4265–4277. |
| 9. | Novotná, K.; Tenora, L.; Slusher, B. S.; Rais, R. Adv. Pharmacol. (San Diego, CA, U. S.) 2024, 100, 157–180. doi:10.1016/bs.apha.2024.04.003 |
| 10. | Recouvreux, M. V.; Grenier, S. F.; Zhang, Y.; Esparza, E.; Lambies, G.; Galapate, C. M.; Maganti, S.; Duong-Polk, K.; Bhullar, D.; Naeem, R.; Scott, D. A.; Lowy, A. M.; Tiriac, H.; Commisso, C. Nat. Cancer 2023, 5, 100–113. doi:10.1038/s43018-023-00649-1 |
| 8. | Thangavelu, K.; Chong, Q. Y.; Low, B. C.; Sivaraman, J. Sci. Rep. 2014, 4, 3827. doi:10.1038/srep03827 |
| 14. | Wang, J.-B.; Erickson, J. W.; Fuji, R.; Ramachandran, S.; Gao, P.; Dinavahi, R.; Wilson, K. F.; Ambrosio, A. L. B.; Dias, S. M. G.; Dang, C. V.; Cerione, R. A. Cancer Cell 2010, 18, 207–219. doi:10.1016/j.ccr.2010.08.009 |
| 25. | Gusak, K. N.; Tereshko, A. B.; Kozlov, N. G.; Shakailo, N. I. Russ. J. Gen. Chem. 2000, 70, 1793–1800. |
| 26. | Kozlov, N. G.; Gusak, K. N. Russ. J. Org. Chem. 1999, 35, 402–414. |
| 27. | Kozlov, N. G.; Gusak, K. N.; Bezborodov, V. S. Russ. J. Org. Chem. 2000, 36, 88–92. |
| 22. | Lukey, M. J.; Cluntun, A. A.; Katt, W. P.; Lin, M.-c. J.; Druso, J. E.; Ramachandran, S.; Erickson, J. W.; Le, H. H.; Wang, Z.-E.; Blank, B.; Greene, K. S.; Cerione, R. A. Cell Rep. 2019, 29, 76–88.e7. doi:10.1016/j.celrep.2019.08.076 |
| 23. | Lielbriedis, I. E.; Chirkova, V. V.; Gudrinietse, E. Izv. Akad. Nauk Latv. SSR, Ser. Khim. 1969, 193–196. |
| 24. | Kozlov, N. S.; Nugumanov, Z. Z. Izv. Akad. Nauk BSSR, Ser. Khim. Nauk 1968, 67–72. |
| 23. | Lielbriedis, I. E.; Chirkova, V. V.; Gudrinietse, E. Izv. Akad. Nauk Latv. SSR, Ser. Khim. 1969, 193–196. |
| 24. | Kozlov, N. S.; Nugumanov, Z. Z. Izv. Akad. Nauk BSSR, Ser. Khim. Nauk 1968, 67–72. |
| 33. | Sheldrick, G. M. Acta Crystallogr., Sect. A: Found. Crystallogr. 2008, 64, 112–122. doi:10.1107/s0108767307043930 |
| 28. | Martínez, R.; Cortés, E.; Toscano, R. A.; Linzaga, I. J. Heterocycl. Chem. 1990, 27, 363–366. doi:10.1002/jhet.5570270248 |
| 29. | Kozlov, N. G.; Gusak, K. N.; Tkachev, A. V. Chem. Heterocycl. Compd. 2007, 43, 740–747. doi:10.1007/s10593-007-0120-z |
| 30. | Kozlov, N. G.; Gusak, K. N. Russ. J. Gen. Chem. 2006, 76, 278–281. doi:10.1134/s1070363206020204 |
| 31. | Nikoofar, K.; Yielzoleh, F. M. J. Saudi Chem. Soc. 2018, 22, 715–741. doi:10.1016/j.jscs.2017.12.005 |
| 32. | Das, B.; Laxminarayana, K.; Krishnaiah, M.; Srinivas, Y. Synlett 2007, 3107–3112. doi:10.1055/s-2007-990922 |
| 28. | Martínez, R.; Cortés, E.; Toscano, R. A.; Linzaga, I. J. Heterocycl. Chem. 1990, 27, 363–366. doi:10.1002/jhet.5570270248 |
| 29. | Kozlov, N. G.; Gusak, K. N.; Tkachev, A. V. Chem. Heterocycl. Compd. 2007, 43, 740–747. doi:10.1007/s10593-007-0120-z |
| 30. | Kozlov, N. G.; Gusak, K. N. Russ. J. Gen. Chem. 2006, 76, 278–281. doi:10.1134/s1070363206020204 |
© 2026 Albertelli et al.; licensee Beilstein-Institut.
This is an open access article licensed under the terms of the Beilstein-Institut Open Access License Agreement (https://www.beilstein-journals.org/bjoc/terms), which is identical to the Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0). The reuse of material under this license requires that the author(s), source and license are credited. Third-party material in this article could be subject to other licenses (typically indicated in the credit line), and in this case, users are required to obtain permission from the license holder to reuse the material.