Abstract
Conversion of a myo-inositol derivative into a scyllo-inositol-derived scaffold with C3v symmetry bearing three axial pyridyl appendages is presented. This pre-organized hexadentate ligand allows complexation of silver(I). The crystal structure of the complex was established.
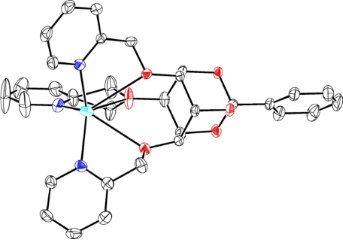
Graphical Abstract
Introduction
Despite its recognized potential for nuclear medicine applications [1], the β− emitter 111Ag (t1/2 = 7.47 d), which can be produced carrier-free and with high specific activity [2,3], has not yet found widespread uses [4,5]. This is due in part to the lack of suitable Ag complexing agents and in this work the synthesis of an cyclitol-derived scaffold designed to sequester Ag is presented. Interaction of myo-inositol-derived podands [6] and crown ethers [7] with Ag salts has been shown to depend on the relative spatial orientation of the binding sites moreover, in mono-orthoesters of scyllo-inositol the three axial hydroxy groups can be used to link substituents in a pre-organized manner [8-10]. Thus the introduction of pyridine groups (known to bind Ag(I) efficiently [11-14]) on a scyllo-inositol orthoester was considered, which led to the design of scaffold 1 (Figure 1). Indeed, upon complexation with participation of three oxygen atoms in the binding, no substantial rearrangement of this semi-rigid structure is to be expected. In addition, reductive cleavage of one of the ortho-ester C–H benzylic bonds, which is known to yield a free hydroxy group [15,16], would allow ligation of this scaffold.
Figure 1: spatial representation of structure 1.
Figure 1: spatial representation of structure 1.
Results and Discussion
Chemistry
The synthesis of 1 was accomplished in 6 steps from the readily available myo-inositol orthobenzoate (2) [17] (Figure 2). As scyllo-inositol derivatives are derived from myo-inositols by an oxidation/reduction sequence [8,18,19], selective protection (tert-butyldimethylsilyl chloride, 2,6-lutidine, DMF, 80 °C, 2 days) of the equatorial hydroxy group of 2 was first performed, to give the silyl ether 3 (70%) and subsequent picolylation (NaH, 2-picolyl chloride, DMF) of the remaining free hydroxy groups gave 4 (65%). The equatorial hydroxy group then was deblocked (tetrabutylammonium fluoride in THF) to afford 5 (86%). Inversion of configuration was accomplished via Swern oxidation [20] to furnish ketone 6 (95%), which was then reduced (NaBH4, CH3OH) to 7 (99%). Although a configurational assignment did not prove possible from 1H NMR data (in epimers 5 and 7 the dihedral angles of the proton geminal to the hydroxy group with vicinal protons are similar), the 1H- and 13C NMR spectra of 7 clearly differ from those of its epimer. Compounds 5 and 7 were also acetylated (to 8 and 9, respectively) and the chemical shift of the acetyl methyl group of 9 (δ = 1.7 ppm) displayed an upfield shift compared to that of 8 (δ = 2.1 ppm) due to ring-current effects from through-space interactions with the pyridyl groups. That a scyllo-inositol derivative had been obtained was confirmed after the introduction of the third pyridyl group (NaH, 2-picolyl chloride). In the resulting tri-picolyl derivative 1 (71%) a C3v axis of symmetry is now present, which leads to simplification of NMR spectra. The structure of 1 was subsequently confirmed (see below) by X-ray diffraction of a single crystal of its Ag-complex.
Complexation
Incremental addition of silver(I) trifluoromethanesulfonate to a solution of 1 in 1:1 CD3OD/CDCl3 was monitored by NMR. A single set of resonances was observed at all concentrations. That the pyridine rings and the "oxygenated cavity" were involved in silver binding was shown by the relevant shifts in both the 1H- and 13C-NMR spectra (see Supporting Information File 1). However, in the absence of characteristic signals for Ag(I)-complex (even at low temperature: −50 °C) NMR methods cannot be used for the determination of the complex stability constant. The electrospray mass spectrum (from the very solution contained in the NMR tube) revealed a 1:1 complex, with the expected 107Ag/109Ag isotopic ratio. The stability constant of the complex could be determined by spectrophotometric titration, which was carried out in 1:1 aqueous methanol (the complex is not water soluble) and its value (log K1·Ag = 3.8 ± 0.2) is in agreement with that of a previously described Ag(I) complex in a N3O3 environment [13].
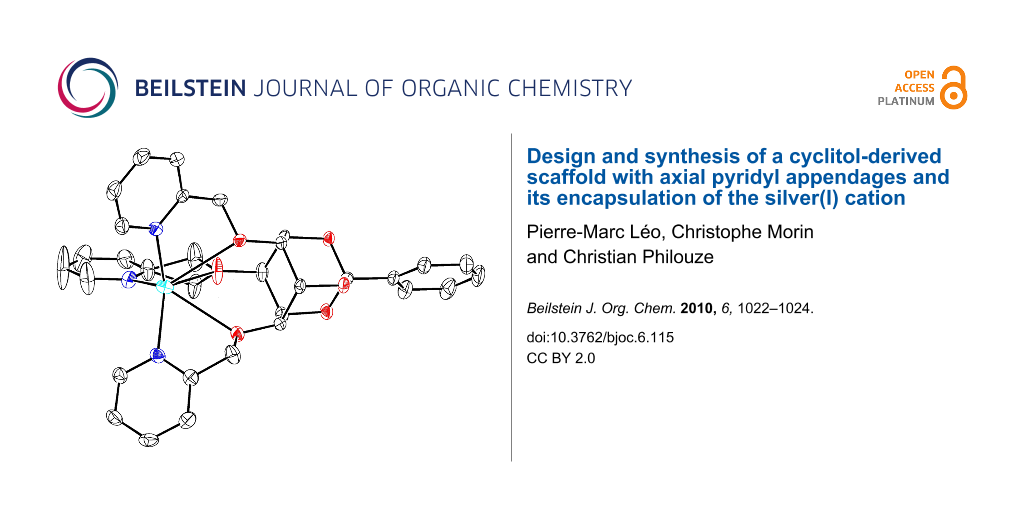
Study of the molecular structure of the complex was made possible as a single crystal could be grown by slow evaporation of a methanol/ethanol solution of stoichiometric amounts of 1 and silver trifluoromethanesulfonate. The complex crystallises in the triclinic system, with a pseudo-ternary axis of symmetry. Its coordination sphere involves all 3 nitrogen atoms as well as the 3 oxygen atoms which are implied in the linkage of the pyridines (Figure 3). Bond distances and angles (see Supporting Information File 1) are in accord with those of known silver complexes which are hexa-coordinated with pyridyl groups and oxygen atoms [13,14].
![[1860-5397-6-115-3]](/bjoc/content/figures/1860-5397-6-115-3.png?scale=1.2&max-width=1024&background=FFFFFF)
Figure 3: ORTEP drawing for the 1·Ag(I) cation complex (ellipsoids are drawn at the 50% probability level and H atoms are omitted for clarity).
Figure 3: ORTEP drawing for the 1·Ag(I) cation complex (ellipsoids are drawn at the 50% probability level and...
Conclusion
A rigid scaffold with C3v symmetry bearing three appendages in a pre-organized manner, 1, could be synthesized in 6 steps from myo-inositol orthobenzoate and in 25% overall yield. Noteworthy is that the synthetic scheme may allow variation of heterocyclic substituents, which need not be the same since they are introduced at different stages of the synthesis. The complex formed with silver cation (log K1·Ag = 3.8 ± 0.2) was shown to involve N3O3 coordination with the Ag(I) ion lying within the cage. However, the complex is not water soluble (which precludes its use for biomedical applications) although the grafting of hydrophilic appendages to aromatic residues could increase its hydrophilicity.
Supporting Information
Supporting Information features experimental procedures, 1H NMR and 13C NMR spectra; electrospray mass spectrum and the crystal structure of the Ag complex.
| Supporting Information File 1: Synthesis and characterisation data for compounds 1–9 and 1·Ag(I). | ||
| Format: PDF | Size: 1.1 MB | Download |
References
-
Alberto, R.; Schubiger, P. A. Recent Res. Dev. Inorg. Chem. 1998, 1, 73–89.
Return to citation in text: [1] -
Alberto, R.; Bläuenstein, P.; Novak-Hofer, I.; Smith, A.; Schubiger, P. A. Int. J. Radiat. Appl. Instrum., Part A 1992, 43, 869–872. doi:10.1016/0883-2889(92)90148-8
Return to citation in text: [1] -
Das, N. R.; Banerjee, S.; Chatterjee, K.; Lahiri, S. Appl. Radiat. Isot. 1999, 50, 643–647. doi:10.1016/S0969-8043(98)00115-8
Return to citation in text: [1] -
Kaden, T. A. Dalton Trans. 2006, 3617–3623. doi:10.1039/b606410h
Return to citation in text: [1] -
Chatopadhyay, S.; Vilmanath, K. V.; Saha, S.; Korde, A.; Sarma, H. D.; Pal, S.; Das, M. K. Appl. Radiat. Isot. 2008, 66, 334–339. doi:10.1016/j.apradiso.2007.09.003
Return to citation in text: [1] -
Sureshan, K. M.; Shashidhar, M. S.; Varma, A. J. J. Chem. Soc., Perkin Trans. 2 2001, 2298–2302. doi:10.1039/b106717f
Return to citation in text: [1] -
Sureshan, K. M.; Shashidhar, M. S.; Varma, A. J. J. Org. Chem. 2002, 6884–6888. doi:10.1021/jo025783g
Return to citation in text: [1] -
Lee, H. W.; Kishi, Y. J. Org. Chem. 1985, 50, 4402–4404. doi:10.1021/jo00222a046
Return to citation in text: [1] [2] -
Tse, B.; Kishi, Y. J. Am. Chem. Soc. 1993, 115, 7892–7893. doi:10.1021/ja00070a051
Return to citation in text: [1] -
Lee, N.-Y.; Jang, W.-J.; Yu, S.-H.; Im, J.; Chung, S.-K. Tetrahedron Lett. 2005, 46, 6063–6066. doi:10.1016/j.tetlet.2005.07.001
Return to citation in text: [1] -
Inokuma, S.; Sakai, S.; Katoh, R.; Nishimura, J. Bull. Chem. Soc. Jpn. 1994, 67, 1462–1467. doi:10.1246/bcsj.67.1462
Return to citation in text: [1] -
Lamb, J. D.; Nazarenko, A. Y.; Uenishi, J.; Tsukube, H. Anal. Chim. Acta 1998, 373, 167–173. doi:10.1016/S0003-2670(98)00360-2
Return to citation in text: [1] -
Danil de Namor, A. F.; Piro, O. E.; Pulcha Salazar, L. E.; Aguilar-Cornejo, A. F.; Al-Rawi, N.; Castellano, E. E.; Sueros Velarde, F. J. J. Chem. Soc., Faraday Trans. 1998, 94, 3097–3104. doi:10.1039/a805430d
Return to citation in text: [1] [2] [3] -
Bowmaker, G. A.; Effendy; Lim, K. C.; Skelton, B. W.; Sukarianingsih, D.; White, A. H. Inorg. Chim. Acta 2005, 358, 4342–4370. doi:10.1016/j.ica.2005.04.008
Return to citation in text: [1] [2] -
Honda, T.; Endo, K.; Ono, S. Chem. Pharm. Bull. 2000, 48, 1545–1548.
Return to citation in text: [1] -
Murali, C.; Shashidhar, M. S.; Gopinath, C. S. Tetrahedron 2007, 63, 4149–4155. doi:10.1016/j.tet.2007.02.096
Return to citation in text: [1] -
Bhosekar, G.; Murali, C.; Gonnade, R. G.; Shashidhar, M. S.; Bhadbhade, M. M. Cryst. Growth Des. 2005, 5, 1977–1982. doi:10.1021/cg050272j
Return to citation in text: [1] -
Shashidhar, M. S. ARKIVOC 2002, vii, 63–75.
Return to citation in text: [1] -
Sureshan, K. M.; Shashidhar, M. S.; Praveen, T.; Das, T. Chem. Rev. 2003, 103, 4477–4504. doi:10.1021/cr0200724
Return to citation in text: [1] -
Tidwell, T. T. Org. React. 1990, 39, 297–572.
Return to citation in text: [1]
| 7. | Sureshan, K. M.; Shashidhar, M. S.; Varma, A. J. J. Org. Chem. 2002, 6884–6888. doi:10.1021/jo025783g |
| 6. | Sureshan, K. M.; Shashidhar, M. S.; Varma, A. J. J. Chem. Soc., Perkin Trans. 2 2001, 2298–2302. doi:10.1039/b106717f |
| 4. | Kaden, T. A. Dalton Trans. 2006, 3617–3623. doi:10.1039/b606410h |
| 5. | Chatopadhyay, S.; Vilmanath, K. V.; Saha, S.; Korde, A.; Sarma, H. D.; Pal, S.; Das, M. K. Appl. Radiat. Isot. 2008, 66, 334–339. doi:10.1016/j.apradiso.2007.09.003 |
| 13. | Danil de Namor, A. F.; Piro, O. E.; Pulcha Salazar, L. E.; Aguilar-Cornejo, A. F.; Al-Rawi, N.; Castellano, E. E.; Sueros Velarde, F. J. J. Chem. Soc., Faraday Trans. 1998, 94, 3097–3104. doi:10.1039/a805430d |
| 14. | Bowmaker, G. A.; Effendy; Lim, K. C.; Skelton, B. W.; Sukarianingsih, D.; White, A. H. Inorg. Chim. Acta 2005, 358, 4342–4370. doi:10.1016/j.ica.2005.04.008 |
| 2. | Alberto, R.; Bläuenstein, P.; Novak-Hofer, I.; Smith, A.; Schubiger, P. A. Int. J. Radiat. Appl. Instrum., Part A 1992, 43, 869–872. doi:10.1016/0883-2889(92)90148-8 |
| 3. | Das, N. R.; Banerjee, S.; Chatterjee, K.; Lahiri, S. Appl. Radiat. Isot. 1999, 50, 643–647. doi:10.1016/S0969-8043(98)00115-8 |
| 17. | Bhosekar, G.; Murali, C.; Gonnade, R. G.; Shashidhar, M. S.; Bhadbhade, M. M. Cryst. Growth Des. 2005, 5, 1977–1982. doi:10.1021/cg050272j |
| 15. | Honda, T.; Endo, K.; Ono, S. Chem. Pharm. Bull. 2000, 48, 1545–1548. |
| 16. | Murali, C.; Shashidhar, M. S.; Gopinath, C. S. Tetrahedron 2007, 63, 4149–4155. doi:10.1016/j.tet.2007.02.096 |
| 13. | Danil de Namor, A. F.; Piro, O. E.; Pulcha Salazar, L. E.; Aguilar-Cornejo, A. F.; Al-Rawi, N.; Castellano, E. E.; Sueros Velarde, F. J. J. Chem. Soc., Faraday Trans. 1998, 94, 3097–3104. doi:10.1039/a805430d |
| 11. | Inokuma, S.; Sakai, S.; Katoh, R.; Nishimura, J. Bull. Chem. Soc. Jpn. 1994, 67, 1462–1467. doi:10.1246/bcsj.67.1462 |
| 12. | Lamb, J. D.; Nazarenko, A. Y.; Uenishi, J.; Tsukube, H. Anal. Chim. Acta 1998, 373, 167–173. doi:10.1016/S0003-2670(98)00360-2 |
| 13. | Danil de Namor, A. F.; Piro, O. E.; Pulcha Salazar, L. E.; Aguilar-Cornejo, A. F.; Al-Rawi, N.; Castellano, E. E.; Sueros Velarde, F. J. J. Chem. Soc., Faraday Trans. 1998, 94, 3097–3104. doi:10.1039/a805430d |
| 14. | Bowmaker, G. A.; Effendy; Lim, K. C.; Skelton, B. W.; Sukarianingsih, D.; White, A. H. Inorg. Chim. Acta 2005, 358, 4342–4370. doi:10.1016/j.ica.2005.04.008 |
| 8. | Lee, H. W.; Kishi, Y. J. Org. Chem. 1985, 50, 4402–4404. doi:10.1021/jo00222a046 |
| 9. | Tse, B.; Kishi, Y. J. Am. Chem. Soc. 1993, 115, 7892–7893. doi:10.1021/ja00070a051 |
| 10. | Lee, N.-Y.; Jang, W.-J.; Yu, S.-H.; Im, J.; Chung, S.-K. Tetrahedron Lett. 2005, 46, 6063–6066. doi:10.1016/j.tetlet.2005.07.001 |
| 8. | Lee, H. W.; Kishi, Y. J. Org. Chem. 1985, 50, 4402–4404. doi:10.1021/jo00222a046 |
| 18. | Shashidhar, M. S. ARKIVOC 2002, vii, 63–75. |
| 19. | Sureshan, K. M.; Shashidhar, M. S.; Praveen, T.; Das, T. Chem. Rev. 2003, 103, 4477–4504. doi:10.1021/cr0200724 |
© 2010 Léo et al; licensee Beilstein-Institut.
This is an Open Access article under the terms of the Creative Commons Attribution License (http://creativecommons.org/licenses/by/2.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
The license is subject to the Beilstein Journal of Organic Chemistry terms and conditions: (http://www.beilstein-journals.org/bjoc)