Abstract
The use of FeCl3 resulted in a fast homocoupling of aryllithiums, and this enabled its integration with the halogen–lithium exchange reaction of aryl halides in a flow microreactor. This system allows the homocoupling of two aryl halides bearing electrophilic functional groups, such as CN and NO2, in under a minute.
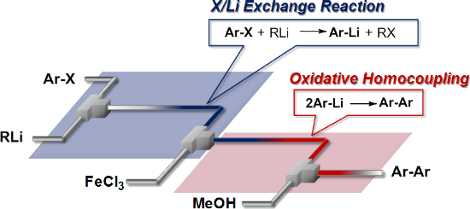
Graphical Abstract
Introduction
Biaryl structures often occur in various organic compounds including natural products, bioactive compounds, functional polymers, ligands in catalysts and theoretically interesting molecules, and the oxidative homocoupling of arylmetals is one of the most useful methods for the construction of biaryl frameworks [1]. Stoichiometric amounts of transition metal salts such as TiCl4 [2], TlCl [3], VO(OEt)Cl2 [4], CoCl2 [5], CuCl2 [6] and Pd(OAc)2 [7] have been used for homocoupling of arylmetals. In some cases catalytic processes in the presence of a reoxidant, such as oxygen or other organic oxidants, are effective. Recently, iron salts have been also used because of their low costs and lack of toxicity [8-18]. For example, Hayashi et al. reported the iron-catalyzed oxidative homocoupling of Grignard reagents, using 1,2-dihalogenoethanes as an oxidant [19]. Cahiez et al. have also reported the FeCl3-catalyzed homocoupling reaction of Grignard reagents bearing functional groups, using atmospheric oxygen [20]. The use of aryllithium compounds instead of Grignard reagents is very interesting, because they are easily generated by halogen–lithium exchange under homogeneous conditions, thus enabling the generation in a flow. However, to the best of our knowledge, oxidative homocoupling of aryllithiums using iron salts has not been reported so far. One of the major reasons for this seems to be the instability of aryllithiums, especially of those bearing electrophilic functional groups such as cyano and nitro groups [21], making the subsequent homocoupling difficult or impossible.
Recently, we have reported that flow microreactor systems [22-85] are quite effective for the generation and reaction of highly reactive organolithiums such as functionalized aryllithiums, oxiranyllithums, aziridinyllithiums, and allenyllithiums [86-98]. Herein we report that integration [99,100] of the generation of aryllithiums, especially those bearing electrophilic functional groups, by halogen–lithium exchange and FeCl3 promoted homocoupling has been effectively accomplished in an integrated flow microreactor system.
Results and Discussion
First, we focused on the generation of p-methoxyphenyllithium from p-bromoanisole (Scheme 1). A flow microreactor system, consisting of two T-shaped micromixers (M1 and M2) and two microtube reactors (R1 and R2) shown in Figure 1, was used. A solution of p-bromoanisole (Ar–X) (0.10 M in THF, flow rate: 6.0 mL/min) and a solution of n-butyllithium (0.40 M in hexane, flow rate: 1.5 mL/min) were introduced to M1 ( = 250 μm) by syringe pumps. The resulting mixture was passed through R1 to conduct the bromine–lithium exchange reaction. Methanol (neat, flow rate: 3.0 mL/min) was added in M2 (
= 500 μm) and the mixture was passed through R2 (
= 1000 μm, L = 50 cm) to protonate p-methoxyphenyllithium. The reactions were carried out with varying residence time in R1 (tR1: 0.2–6.3 s) and varying temperature (T: −78 to 24 °C). The temperature (T) was controlled by adjusting the bath temperature. The residence time (tR1) was adjusted by changing the inner diameter and the length in the microtube reactor R1 with a fixed flow rate. After a steady state was reached, the product solution was collected for 30 s. As shown in Figure 2, the yield of the protonated product, anisole, depends on both T and tR1. The reaction at low temperatures (T < −48 °C) with short residence times (tR1 < 0.79 s) resulted in very low yields, because the Br–Li exchange reaction was not complete. The increase in T and tR1 caused an increase in the yield, and high yields (>85%) were obtained through the appropriate choice of T and tR1.
Scheme 1: Halogen–lithium exchange of p-bromoanisole followed by reaction with methanol.
Scheme 1: Halogen–lithium exchange of p-bromoanisole followed by reaction with methanol.
![[1860-5397-7-122-1]](/bjoc/content/figures/1860-5397-7-122-1.png?scale=2.0&max-width=1024&background=FFFFFF)
Figure 1:
Flow microreactor system for halogen–lithium exchange of aryl halide followed by reaction with methanol. T-shaped micromixer: M1 (inner diameter: 250 μm), and M2 (inner diameter: 500 μm), microtube reactor: R1 and R2 ( = 1000 μm, length = 50 cm), a solution of aryl halides: 0.10 M in THF (6.0 mL/min), a solution of lithium reagent: 0.40 M or 0.42 M in hexane (n-BuLi) or Et2O (PhLi) (1.5 mL/min), a solution of methanol: Neat (3.0 mL/min).
Figure 1: Flow microreactor system for halogen–lithium exchange of aryl halide followed by reaction with meth...
![[1860-5397-7-122-2]](/bjoc/content/figures/1860-5397-7-122-2.png?scale=2.0&max-width=1024&background=FFFFFF)
Figure 2: Effects of the temperature (T) and the residence time in R1 (tR1) on the yield of anisole in the Br–Li exchange reaction of p-bromoanisole followed by reaction with methanol in the flow microreactor system. The contour plot with scatter overlay shows the yields of anisole (%), which are indicated by small circles.
Figure 2: Effects of the temperature (T) and the residence time in R1 (tR1) on the yield of anisole in the Br...
Next, we examined the integration of the halogen–lithium exchange reaction with FeCl3 promoted homocoupling (Scheme 2). Integrated flow microreactor systems consisting of three micromixers (M1, M2, and M3) and three microtube reactors (R1, R2, and R3) were used, as shown in Figure 3. A solution of p-bromoanisole (Ar–X) (0.10 M in THF, flow rate: 6.0 mL/min) and a solution of n-butyllithium (0.42 M in hexane, flow rate: 1.5 mL/min) were introduced to M1 ( = 250 μm) by syringe pumps. The resulting mixture was passed through R1 (tR1 = 13 s (−78 °C), tR1 = 13 s (−48 °C), tR1 = 3.1 s (−28 °C), tR1 = 3.1 s (0 °C), tR1 = 3.1 s (24 °C)) at the corresponding temperatures and was mixed with a solution of FeCl3 (0.10 M in THF, flow rate: 6.0 mL/min) in M2 (
= 500 μm). The resulting mixture was passed through R2 and was then mixed with methanol (neat, flow rate: 1.5 mL/min) in M3 (
= 500 μm) to protonate the unchanged p-methoxyphenyllithium. The resulting solution was passed through R3 (
= 1000 μm, L = 50 cm). The temperature (T) was controlled by adjusting the bath temperature, and the residence time in R2 (tR2) by changing the inner diameter and the length in R2 with the fixed flow rate. After a steady state was reached, the product solution was collected for 30 s. The results obtained from varying tR2 and T are summarized in Figure 4, in which the yield of 4,4'-dimethoxybiphenyl is plotted against T and tR2 as a contour map with scattered overlay (see Supporting Information File 1 for details). The yield depends on both T and tR2. At −78 °C, the yield increased with tR2 because of the progress of the homocoupling. At 0 °C, the homocoupling product was obtained in reasonable yields for a wide range of tR2. The productivity of the present system is acceptable for large scale laboratory synthesis (6.2 g/h). It is noteworthy that the integrated reactions were complete within the overall residence time of 14.7 s, even at low temperatures such as −48 °C. Thus, we envisaged that the reaction could also be applied to less stable aryllithium compounds that decompose very quickly.
Scheme 2: Halogen-lithium exchange of p-bromoanisole followed by oxidative homocoupling with FeCl3.
Scheme 2: Halogen-lithium exchange of p-bromoanisole followed by oxidative homocoupling with FeCl3.
![[1860-5397-7-122-3]](/bjoc/content/figures/1860-5397-7-122-3.png?scale=2.0&max-width=1024&background=FFFFFF)
Figure 3:
Integrated flow microreactor system for oxidative homocoupling reaction of aryllithium with FeCl3. T-shaped micromixer: M1 (inner diameter: 250 μm), M2 (inner diameter: 500 μm), and M3 (inner diameter: 500 μm), microtube reactor: R1, R2 and R3 ( = 1000 μm, length = 50 cm), a solution of aryl halides: 0.10 M in THF (6.0 mL/min), a solution of lithium reagent: 0.42 M in hexane (n-BuLi) or Et2O (PhLi) (1.5 mL/min), a solution of FeCl3: 0.10 M in THF (6 mL/min), a solution of methanol: Neat (1.5 mL/min).
Figure 3: Integrated flow microreactor system for oxidative homocoupling reaction of aryllithium with FeCl3. ...
![[1860-5397-7-122-4]](/bjoc/content/figures/1860-5397-7-122-4.png?scale=2.0&max-width=1024&background=FFFFFF)
Figure 4: Effects of the temperature (T) and the residence time in R2 (tR2) on the yield of 4,4'-dimethoxybiphenyl in the oxidative homocoupling of p-methoxyphenyllithium with FeCl3 in the integrated flow microreactor system. Contour plot with scatter overlay of the yields of 4,4'-dimethoxybiphenyl (%), which are indicated by small circles.
Figure 4: Effects of the temperature (T) and the residence time in R2 (tR2) on the yield of 4,4'-dimethoxybip...
One of the major benefits of flow microreactor synthesis is the ability to use highly unstable reactive intermediates. Such intermediates can be rapidly generated and transferred to another location to be used in a subsequent reaction before they decompose. We have already reported the generation and reactions of unstable aryllithium species such as o-bromophenyllithiums, and aryllithiums bearing alkoxycarbonyl, cyano, nitro, and ketone carbonyl groups [86,87,93,95,96,98], which are difficult to use in conventional macro batch reactors. As shown in Table 1, reactions of aryllithiums bearing cyano and nitro groups proceeded successfully to give the corresponding homocoupling products, where in contrast it is very difficult to achieve such reactions using conventional batch reactors. A mechanism involving transmetalation of the aryl group from lithium to iron followed by reductive elimination of the homo-coupling product seems to be plausible, while a similar mechanism is proposed for homo-coupling of organomagnesium compounds with FeCl3 [19,20]. The regiospecificity of the coupling is consistent with this mechanism. Radical coupling seems to be less likely.
Conclusion
In conclusion, we found that the use of FeCl3 results in fast oxidative homocoupling of aryllithiums, which enables its integration with the halogen–lithium exchange of aryl halides. Various aryl halides, including those bearing electrophilic functional groups, can be used for this transformation in the integrated flow microreactor system. Hence, the method greatly enhances the synthetic utility of aryllithium compounds and adds a new dimension to the chemistry of coupling reactions.
Supporting Information
Supporting Information features experimental procedures and full spectroscopic data for all new compounds.
| Supporting Information File 1: Experimental details. | ||
| Format: PDF | Size: 270.3 KB | Download |
References
-
Cepanec, I. Synthesis of Biaryls; Elsevier: Amsterdam, Boston, 2004.
Return to citation in text: [1] -
Inoue, A.; Kitagawa, K.; Shinokubo, H.; Oshima, K. Tetrahedron 2000, 56, 9601–9605. doi:10.1016/S0040-4020(00)00929-7
Return to citation in text: [1] -
McKillop, A.; Elsom, L. F.; Taylor, E. C. J. Am. Chem. Soc. 1968, 90, 2423–2424. doi:10.1021/ja01011a041
Return to citation in text: [1] -
Ishikawa, T.; Ogawa, A.; Hirao, T. Organometallics 1998, 17, 5713–5716. doi:10.1021/om980607c
Return to citation in text: [1] -
Kharasch, M. S.; Fields, E. K. J. Am. Chem. Soc. 1941, 63, 2316–2320. doi:10.1021/ja01854a006
Return to citation in text: [1] -
Sakellarios, E.; Kyrimis, T. Ber. Dtsch. Chem. Ges. 1924, 57, 322–326. doi:10.1002/cber.19240570233
Return to citation in text: [1] -
Lei, A.; Srivastava, M.; Zhang, X. J. Org. Chem. 2002, 67, 1969–1971. doi:10.1021/jo011098i
Return to citation in text: [1] -
Cahiez, G.; Marquais, S. Pure Appl. Chem. 1996, 68, 53–60. doi:10.1351/pac199668010053
Return to citation in text: [1] -
Cahiez, G.; Marquais, S. Tetrahedron Lett. 1996, 37, 1773–1776. doi:10.1016/0040-4039(96)00116-5
Return to citation in text: [1] -
Cahiez, G.; Avedissian, H. Synthesis 1998, 1199–1205. doi:10.1055/s-1998-2135
Return to citation in text: [1] -
Dohle, W.; Kopp, F.; Cahiez, G.; Knochel, P. Synlett 2001, 1901–1904. doi:10.1055/s-2001-18748
Return to citation in text: [1] -
Fürstner, A.; Leitner, A.; Méndez, M.; Krause, H. J. Am. Chem. Soc. 2002, 124, 13856–13863. doi:10.1021/ja027190t
Return to citation in text: [1] -
Fürstner, A.; Leitner, A. Angew. Chem., Int. Ed. 2002, 41, 609–612. doi:10.1002/1521-3773(20020215)41:4<609::AID-ANIE609>3.0.CO;2-M
Return to citation in text: [1] -
Quintin, J.; Franck, X.; Hocquemiller, R.; Figadère, B. Tetrahedron Lett. 2002, 43, 3547–3549. doi:10.1016/S0040-4039(02)00568-3
Return to citation in text: [1] -
Martin, R.; Fürstner, A. Angew. Chem., Int. Ed. 2004, 43, 3955–3957. doi:10.1002/anie.200460504
Return to citation in text: [1] -
Scheiper, B.; Bonnekessel, M.; Krause, H.; Fürstner, A. J. Org. Chem. 2004, 69, 3943–3949. doi:10.1021/jo0498866
Return to citation in text: [1] -
Nakamura, M.; Matsuo, K.; Ito, S.; Nakamura, E. J. Am. Chem. Soc. 2004, 126, 3686–3687. doi:10.1021/ja049744t
Return to citation in text: [1] -
Nagano, T.; Hayashi, T. Org. Lett. 2004, 6, 1297–1299. doi:10.1021/ol049779y
Return to citation in text: [1] -
Nagano, T.; Hayashi, T. Org. Lett. 2005, 7, 491–493. doi:10.1021/ol047509+
Return to citation in text: [1] [2] -
Cahiez, G.; Chaboche, C.; Mahuteau-Betzer, F.; Ahr, M. Org. Lett. 2005, 7, 1943–1946. doi:10.1021/ol050340v
Return to citation in text: [1] [2] -
Knochel, P., Ed. Handbook of Functionalized Organometallics; Wiley-VCH: Weinheim, Germany, 2005. doi:10.1002/9783527619467
Return to citation in text: [1] -
Yoshida, J. Flash Chemistry: Fast Organic Synthesis in Microsystems; Wiley: Hoboken, N.J., 2008.
Return to citation in text: [1] -
Fletcher, P. D. I.; Haswell, S. J.; Pombo-Villar, E.; Warrington, B. H.; Watts, P.; Wong, S. Y. F.; Zhang, X. Tetrahedron 2002, 58, 4735–4757. doi:10.1016/S0040-4020(02)00432-5
Return to citation in text: [1] -
Jähnisch, K.; Hessel, V.; Löwe, H.; Baerns, M. Angew. Chem., Int. Ed. 2004, 43, 406–446. doi:10.1002/anie.200300577
Return to citation in text: [1] -
Yoshida, J. Chem. Commun. 2005, 4509–4516. doi:10.1039/b508341a
Return to citation in text: [1] -
Doku, G. N.; Verboom, W.; Reinhoudt, D. N.; van den Berg, A. Tetrahedron 2005, 61, 2733–2742. doi:10.1016/j.tet.2005.01.028
Return to citation in text: [1] -
Yoshida, J.; Nagaki, A.; Iwasaki, T.; Suga, S. Chem. Eng. Technol. 2005, 28, 259–266. doi:10.1002/ceat.200407127
Return to citation in text: [1] -
Geyer, K.; Codee, J. D. C.; Seeberger, P. H. Chem.–Eur. J. 2006, 12, 8434–8442. doi:10.1002/chem.200600596
Return to citation in text: [1] -
Whitesides, G. Nature 2006, 442, 368–373. doi:10.1038/nature05058
Return to citation in text: [1] -
deMello, A. J. Nature 2006, 442, 394–402. doi:10.1038/nature05062
Return to citation in text: [1] -
Song, H.; Chen, D. L.; Ismagilov, R. F. Angew. Chem., Int. Ed. 2006, 45, 7336–7356. doi:10.1002/anie.200601554
Return to citation in text: [1] -
Kobayashi, J.; Mori, Y.; Kobayashi, S. Chem.–Asian J. 2006, 1, 22–35. doi:10.1002/asia.200600058
Return to citation in text: [1] -
Mason, B. P.; Price, K. E.; Steinbacher, J. L.; Bogdan, A. R.; McQuade, D. T. Chem. Rev. 2007, 107, 2300–2318. doi:10.1021/cr050944c
Return to citation in text: [1] -
Ahmed-Omer, B.; Brandtand, J. C.; Wirth, T. Org. Biomol. Chem. 2007, 5, 733–740. doi:10.1039/b615072a
Return to citation in text: [1] -
Watts, P.; Wiles, C. Chem. Commun. 2007, 443–467. doi:10.1039/b609428g
Return to citation in text: [1] -
Yoshida, J.; Nagaki, A.; Yamada, T. Chem.–Eur. J. 2008, 14, 7450–7459. doi:10.1002/chem.200800582
Return to citation in text: [1] -
Fukuyama, T.; Rahman, M. T.; Sato, M.; Ryu, I. Synlett 2008, 151–163. doi:10.1055/s-2007-1000884
Return to citation in text: [1] -
McMullen, J. P.; Jensen, K. F. Annu. Rev. Anal. Chem. 2010, 3, 19–42. doi:10.1146/annurev.anchem.111808.073718
Return to citation in text: [1] -
Yoshida, J. Chem. Rec. 2010, 10, 332–341. doi:10.1002/tcr.201000020
Return to citation in text: [1] -
Yoshida, J.; Kim, H.; Nagaki, A. ChemSusChem 2011, 4, 331–340. doi:10.1002/cssc.201000271
Return to citation in text: [1] -
Watts, P.; Wiles, C.; Haswell, S. J.; Pombo-Villar, E.; Styring, P. Chem. Commun. 2001, 990–991. doi:10.1039/b102125g
Return to citation in text: [1] -
Suga, S.; Okajima, M.; Fujiwara, K.; Yoshida, J. J. Am. Chem. Soc. 2001, 123, 7941–7942. doi:10.1021/ja015823i
Return to citation in text: [1] -
Fukuyama, T.; Shinmen, M.; Nishitani, S.; Sato, M.; Ryu, I. Org. Lett. 2002, 4, 1691–1694. doi:10.1021/ol0257732
Return to citation in text: [1] -
Ueno, M.; Hisamoto, H.; Kitamori, T.; Kobayashi, S. Chem. Commun. 2003, 936–937. doi:10.1039/b301638b
Return to citation in text: [1] -
Garcia-Egido, E.; Spikmans, V.; Wong, S. Y. F.; Warrington, B. H. Lab Chip 2003, 3, 73–76. doi:10.1039/b302381h
Return to citation in text: [1] -
Lai, S. M.; Martin-Aranda, R.; Yeung, K. L. Chem. Commun. 2003, 218–219. doi:10.1039/b209297b
Return to citation in text: [1] -
Mikami, K.; Yamanaka, M.; Islam, M. N.; Kudo, K.; Seino, N.; Shinoda, M. Tetrahedron Lett. 2003, 44, 7545–7548. doi:10.1016/S0040-4039(03)01835-5
Return to citation in text: [1] -
Kobayashi, J.; Mori, Y.; Okamoto, K.; Akiyama, R.; Ueno, M.; Kitamori, T.; Kobayashi, S. Science 2004, 304, 1305–1308. doi:10.1126/science.1096956
Return to citation in text: [1] -
Nagaki, A.; Kawamura, K.; Suga, S.; Ando, T.; Sawamoto, M.; Yoshida, J. J. Am. Chem. Soc. 2004, 126, 14702–14703. doi:10.1021/ja044879k
Return to citation in text: [1] -
Lee, C.-C.; Sui, G.; Elizarov, A.; Shu, C. J.; Shin, Y.-S.; Dooley, A. N.; Huang, J.; Daridon, A.; Wyatt, P.; Stout, D.; Kolb, H. C.; Witte, O. N.; Satyamurthy, N.; Heath, J. R.; Phelps, M. E.; Quake, S. R.; Tseng, H.-R. Science 2005, 310, 1793–1796. doi:10.1126/science.1118919
Return to citation in text: [1] -
Horcajada, R.; Okajima, M.; Suga, S.; Yoshida, J. Chem. Commun. 2005, 1303–1305. doi:10.1039/b417388k
Return to citation in text: [1] -
Kawaguchi, T.; Miyata, H.; Ataka, K.; Mae, K.; Yoshida, J. Angew. Chem., Int. Ed. 2005, 44, 2413–2416. doi:10.1002/anie.200462466
Return to citation in text: [1] -
Ducry, L.; Roberge, D. M. Angew. Chem., Int. Ed. 2005, 44, 7972–7975. doi:10.1002/anie.200502387
Return to citation in text: [1] -
Nagaki, A.; Togai, M.; Suga, S.; Aoki, N.; Mae, K.; Yoshida, J. J. Am. Chem. Soc. 2005, 127, 11666–11675. doi:10.1021/ja0527424
Return to citation in text: [1] -
He, P.; Watts, P.; Marken, F.; Haswell, S. J. Angew. Chem., Int. Ed. 2006, 45, 4146–4149. doi:10.1002/anie.200600951
Return to citation in text: [1] -
Uozumi, Y.; Yamada, Y.; Beppu, T.; Fukuyama, N.; Ueno, M.; Kitamori, T. J. Am. Chem. Soc. 2006, 128, 15994–15995. doi:10.1021/ja066697r
Return to citation in text: [1] -
Tanaka, K.; Motomatsu, S.; Koyama, K.; Tanaka, S.; Fukase, K. Org. Lett. 2007, 9, 299–302. doi:10.1021/ol062777o
Return to citation in text: [1] -
Sahoo, H. R.; Kralj, J. G.; Jensen, K. F. Angew. Chem., Int. Ed. 2007, 46, 5704–5708. doi:10.1002/anie.200701434
Return to citation in text: [1] -
Hornung, C. H.; Mackley, M. R.; Baxendale, I. R.; Ley, S. V. Org. Process Res. Dev. 2007, 11, 399–405. doi:10.1021/op700015f
Return to citation in text: [1] -
Iwasaki, T.; Nagaki, A.; Yoshida, J. Chem. Commun. 2007, 1263–1265. doi:10.1039/b615159k
Return to citation in text: [1] -
Nagaki, A.; Iwasaki, T.; Kawamura, K.; Yamada, D.; Suga, S.; Ando, T.; Sawamoto, M.; Yoshida, J. Chem.–Asian J. 2008, 3, 1558–1567. doi:10.1002/asia.200800081
Return to citation in text: [1] -
Nagaki, A.; Tomida, Y.; Yoshida, J. Macromolecules 2008, 41, 6322–6330. doi:10.1021/ma800769n
Return to citation in text: [1] -
Fukuyama, T.; Kobayashi, M.; Rahman, M. T.; Kamata, N.; Ryu, I. Org. Lett. 2008, 10, 533–536. doi:10.1021/ol702718z
Return to citation in text: [1] -
Nagaki, A.; Tomida, Y.; Miyazaki, A.; Yoshida, J. Macromolecules 2009, 42, 4384–4387. doi:10.1021/ma900551a
Return to citation in text: [1] -
Tricotet, T.; O’Shea, D. F. Chem.–Eur. J. 2010, 16, 6678–6686. doi:10.1002/chem.200903284
Return to citation in text: [1] -
Greenway, G. M. S.; Haswell, J.; Morgan, D. O.; Skelton, V.; Styring, P. Sens. Actuators, B 2000, 63, 153–158. doi:10.1016/S0925-4005(00)00352-X
Return to citation in text: [1] -
Haswell, S. J.; O'Sullivan, B.; Styring, P. Lab Chip 2001, 1, 164–166. doi:10.1039/b104035a
Return to citation in text: [1] -
Niwa, S.; Eswaramoorthy, M.; Nair, J.; Raj, A.; Itoh, N.; Shoji, H.; Namba, T.; Mizukami, F. Science 2002, 295, 105–107. doi:10.1126/science.1066527
Return to citation in text: [1] -
Jas, G.; Kirschning, A. Chem.–Eur. J. 2003, 9, 5708–5723. doi:10.1002/chem.200305212
Return to citation in text: [1] -
Basheer, C.; Hussain, F. S. J.; Lee, H. K.; Valiyaveettil, S. Tetrahedron Lett. 2004, 45, 7297–7300. doi:10.1016/j.tetlet.2004.08.017
Return to citation in text: [1] -
Liu, S.; Fukuyama, T.; Sato, M.; Ryu, I. Org. Process Res. Dev. 2004, 8, 477–481. doi:10.1021/op034200h
Return to citation in text: [1] -
Kunz, U.; Schönfeld, H.; Solodenko, W.; Jas, G.; Kirschning, A. Ind. Eng. Chem. Res. 2005, 44, 8458–8467. doi:10.1021/ie048891x
Return to citation in text: [1] -
Comer, E.; Organ, M. G. J. Am. Chem. Soc. 2005, 127, 8160–8167. doi:10.1021/ja0512069
Return to citation in text: [1] -
Lee, C. K. Y.; Holmes, A. B.; Ley, S. V.; McConvey, I. F.; Al-Duri, B.; Leeke, G. A.; Santos, R. C. D.; Sevilled, J. P. K. Chem. Commun. 2005, 2175–2177. doi:10.1039/b418669a
Return to citation in text: [1] -
Mauger, C.; Buisine, O.; Caravieilhes, S.; Mignani, G. J. Organomet. Chem. 2005, 690, 3627–3629. doi:10.1016/j.jorganchem.2005.03.071
Return to citation in text: [1] -
Kirschning, A.; Solodenko, W.; Mennecke, K. Chem.–Eur. J. 2006, 12, 5972–5990. doi:10.1002/chem.200600236
Return to citation in text: [1] -
Shore, G.; Morin, S.; Organ, M. G. Angew. Chem., Int. Ed. 2006, 45, 2761–2766. doi:10.1002/anie.200503600
Return to citation in text: [1] -
Shi, G.; Hong, F.; Liang, Q.; Fang, H.; Nelson, S.; Weber, S. G. Anal. Chem. 2006, 78, 1972–1979. doi:10.1021/ac051844+
Return to citation in text: [1] -
Rahman, M. T.; Fukuyama, T.; Kamata, N.; Sato, M.; Ryu, I. Chem. Commun. 2006, 2236–2238. doi:10.1039/b600970k
Return to citation in text: [1] -
Murphy, E. R.; Martinelli, J. R.; Zaborenko, N.; Buchwald, S. L.; Jensen, K. F. Angew. Chem., Int. Ed. 2007, 46, 1734–1737. doi:10.1002/anie.200604175
Return to citation in text: [1] -
Kawanami, H.; Matsushima, K.; Sato, M.; Ikushima, Y. Angew. Chem., Int. Ed. 2007, 46, 5129–5132. doi:10.1002/anie.200700611
Return to citation in text: [1] -
Yamada, Y. M. A.; Watanabe, T.; Torii, K.; Uozumi, Y. Chem. Commun. 2009, 5594–5596. doi:10.1039/b912696a
Return to citation in text: [1] -
Jin, J.; Cai, M.-M.; Li, J. Synlett 2009, 2534–2538. doi:10.1055/s-0029-1217730
Return to citation in text: [1] -
Ahmed-Omer, B.; Barrow, D. A.; Wirth, T. Tetrahedron Lett. 2009, 50, 3352–3355. doi:10.1016/j.tetlet.2009.02.133
Return to citation in text: [1] -
McMullen, J. P.; Stone, M. T.; Buchwald, S. L.; Jensen, K. F. Angew. Chem., Int. Ed. 2010, 49, 7076–7080. doi:10.1002/anie.201002590
Return to citation in text: [1] -
Usutani, H.; Tomida, Y.; Nagaki, A.; Okamoto, H.; Nokami, T.; Yoshida, J. J. Am. Chem. Soc. 2007, 129, 3046–3047. doi:10.1021/ja068330s
Return to citation in text: [1] [2] -
Nagaki, A.; Tomida, Y.; Usutani, H.; Kim, H.; Takabayashi, N.; Nokami, T.; Okamoto, H.; Yoshida, J. Chem.–Asian J. 2007, 2, 1513–1523. doi:10.1002/asia.200700231
Return to citation in text: [1] [2] -
Nagaki, A.; Takabayashi, N.; Tomida, Y.; Yoshida, J. Org. Lett. 2008, 10, 3937–3940. doi:10.1021/ol8015572
Return to citation in text: [1] -
Nagaki, A.; Kim, H.; Yoshida, J. Angew. Chem., Int. Ed. 2008, 47, 7833–7836. doi:10.1002/anie.200803205
Return to citation in text: [1] -
Nagaki, A.; Takizawa, E.; Yoshida, J. J. Am. Chem. Soc. 2009, 131, 1654–1655. doi:10.1021/ja809325a
Return to citation in text: [1] -
Nagaki, A.; Takabayashi, N.; Tomida, Y.; Yoshida, J. Beilstein J. Org. Chem. 2009, 5, No. 16. doi:10.3762/bjoc.5.16
Return to citation in text: [1] -
Tomida, Y.; Nagaki, A.; Yoshida, J. Org. Lett. 2009, 11, 3614–3617. doi:10.1021/ol901352t
Return to citation in text: [1] -
Nagaki, A.; Kim, H.; Yoshida, J. Angew. Chem., Int. Ed. 2009, 48, 8063–8065. doi:10.1002/anie.200904316
Return to citation in text: [1] [2] -
Nagaki, A.; Takizawa, E.; Yoshida, J. Chem.–Eur. J. 2010, 16, 14149–14158. doi:10.1002/chem.201000815
Return to citation in text: [1] -
Nagaki, A.; Kim, H.; Moriwaki, Y.; Matsuo, C.; Yoshida, J. Chem.–Eur. J. 2010, 16, 11167–11177. doi:10.1002/chem.201000876
Return to citation in text: [1] [2] -
Nagaki, A.; Kim, H.; Matsuo, C.; Yoshida, J. Org. Biomol. Chem. 2010, 8, 1212–1217. doi:10.1039/b919325c
Return to citation in text: [1] [2] -
Tomida, Y.; Nagaki, A.; Yoshida, J. J. Am. Chem. Soc. 2011, 133, 3744–3747. doi:10.1021/ja110898s
Return to citation in text: [1] -
Kim, H.; Nagaki, A.; Yoshida, J. Nat. Commun. 2011, 2, No. 264. doi:10.1038/ncomms1264
Return to citation in text: [1] [2] -
Suga, S.; Yamada, D.; Yoshida, J. Chem. Lett. 2010, 39, 404–406. doi:10.1246/cl.2010.404
Return to citation in text: [1] -
Nagaki, A.; Kenmoku, A.; Moriwaki, Y.; Hayashi, A.; Yoshida, J. Angew. Chem., Int. Ed. 2010, 49, 7543–7547. doi:10.1002/anie.201002763
Return to citation in text: [1]
| 5. | Kharasch, M. S.; Fields, E. K. J. Am. Chem. Soc. 1941, 63, 2316–2320. doi:10.1021/ja01854a006 |
| 86. | Usutani, H.; Tomida, Y.; Nagaki, A.; Okamoto, H.; Nokami, T.; Yoshida, J. J. Am. Chem. Soc. 2007, 129, 3046–3047. doi:10.1021/ja068330s |
| 87. | Nagaki, A.; Tomida, Y.; Usutani, H.; Kim, H.; Takabayashi, N.; Nokami, T.; Okamoto, H.; Yoshida, J. Chem.–Asian J. 2007, 2, 1513–1523. doi:10.1002/asia.200700231 |
| 93. | Nagaki, A.; Kim, H.; Yoshida, J. Angew. Chem., Int. Ed. 2009, 48, 8063–8065. doi:10.1002/anie.200904316 |
| 95. | Nagaki, A.; Kim, H.; Moriwaki, Y.; Matsuo, C.; Yoshida, J. Chem.–Eur. J. 2010, 16, 11167–11177. doi:10.1002/chem.201000876 |
| 96. | Nagaki, A.; Kim, H.; Matsuo, C.; Yoshida, J. Org. Biomol. Chem. 2010, 8, 1212–1217. doi:10.1039/b919325c |
| 98. | Kim, H.; Nagaki, A.; Yoshida, J. Nat. Commun. 2011, 2, No. 264. doi:10.1038/ncomms1264 |
| 4. | Ishikawa, T.; Ogawa, A.; Hirao, T. Organometallics 1998, 17, 5713–5716. doi:10.1021/om980607c |
| 19. | Nagano, T.; Hayashi, T. Org. Lett. 2005, 7, 491–493. doi:10.1021/ol047509+ |
| 20. | Cahiez, G.; Chaboche, C.; Mahuteau-Betzer, F.; Ahr, M. Org. Lett. 2005, 7, 1943–1946. doi:10.1021/ol050340v |
| 3. | McKillop, A.; Elsom, L. F.; Taylor, E. C. J. Am. Chem. Soc. 1968, 90, 2423–2424. doi:10.1021/ja01011a041 |
| 86. | Usutani, H.; Tomida, Y.; Nagaki, A.; Okamoto, H.; Nokami, T.; Yoshida, J. J. Am. Chem. Soc. 2007, 129, 3046–3047. doi:10.1021/ja068330s |
| 87. | Nagaki, A.; Tomida, Y.; Usutani, H.; Kim, H.; Takabayashi, N.; Nokami, T.; Okamoto, H.; Yoshida, J. Chem.–Asian J. 2007, 2, 1513–1523. doi:10.1002/asia.200700231 |
| 88. | Nagaki, A.; Takabayashi, N.; Tomida, Y.; Yoshida, J. Org. Lett. 2008, 10, 3937–3940. doi:10.1021/ol8015572 |
| 89. | Nagaki, A.; Kim, H.; Yoshida, J. Angew. Chem., Int. Ed. 2008, 47, 7833–7836. doi:10.1002/anie.200803205 |
| 90. | Nagaki, A.; Takizawa, E.; Yoshida, J. J. Am. Chem. Soc. 2009, 131, 1654–1655. doi:10.1021/ja809325a |
| 91. | Nagaki, A.; Takabayashi, N.; Tomida, Y.; Yoshida, J. Beilstein J. Org. Chem. 2009, 5, No. 16. doi:10.3762/bjoc.5.16 |
| 92. | Tomida, Y.; Nagaki, A.; Yoshida, J. Org. Lett. 2009, 11, 3614–3617. doi:10.1021/ol901352t |
| 93. | Nagaki, A.; Kim, H.; Yoshida, J. Angew. Chem., Int. Ed. 2009, 48, 8063–8065. doi:10.1002/anie.200904316 |
| 94. | Nagaki, A.; Takizawa, E.; Yoshida, J. Chem.–Eur. J. 2010, 16, 14149–14158. doi:10.1002/chem.201000815 |
| 95. | Nagaki, A.; Kim, H.; Moriwaki, Y.; Matsuo, C.; Yoshida, J. Chem.–Eur. J. 2010, 16, 11167–11177. doi:10.1002/chem.201000876 |
| 96. | Nagaki, A.; Kim, H.; Matsuo, C.; Yoshida, J. Org. Biomol. Chem. 2010, 8, 1212–1217. doi:10.1039/b919325c |
| 97. | Tomida, Y.; Nagaki, A.; Yoshida, J. J. Am. Chem. Soc. 2011, 133, 3744–3747. doi:10.1021/ja110898s |
| 98. | Kim, H.; Nagaki, A.; Yoshida, J. Nat. Commun. 2011, 2, No. 264. doi:10.1038/ncomms1264 |
| 2. | Inoue, A.; Kitagawa, K.; Shinokubo, H.; Oshima, K. Tetrahedron 2000, 56, 9601–9605. doi:10.1016/S0040-4020(00)00929-7 |
| 99. | Suga, S.; Yamada, D.; Yoshida, J. Chem. Lett. 2010, 39, 404–406. doi:10.1246/cl.2010.404 |
| 100. | Nagaki, A.; Kenmoku, A.; Moriwaki, Y.; Hayashi, A.; Yoshida, J. Angew. Chem., Int. Ed. 2010, 49, 7543–7547. doi:10.1002/anie.201002763 |
| 21. | Knochel, P., Ed. Handbook of Functionalized Organometallics; Wiley-VCH: Weinheim, Germany, 2005. doi:10.1002/9783527619467 |
| 8. | Cahiez, G.; Marquais, S. Pure Appl. Chem. 1996, 68, 53–60. doi:10.1351/pac199668010053 |
| 9. | Cahiez, G.; Marquais, S. Tetrahedron Lett. 1996, 37, 1773–1776. doi:10.1016/0040-4039(96)00116-5 |
| 10. | Cahiez, G.; Avedissian, H. Synthesis 1998, 1199–1205. doi:10.1055/s-1998-2135 |
| 11. | Dohle, W.; Kopp, F.; Cahiez, G.; Knochel, P. Synlett 2001, 1901–1904. doi:10.1055/s-2001-18748 |
| 12. | Fürstner, A.; Leitner, A.; Méndez, M.; Krause, H. J. Am. Chem. Soc. 2002, 124, 13856–13863. doi:10.1021/ja027190t |
| 13. | Fürstner, A.; Leitner, A. Angew. Chem., Int. Ed. 2002, 41, 609–612. doi:10.1002/1521-3773(20020215)41:4<609::AID-ANIE609>3.0.CO;2-M |
| 14. | Quintin, J.; Franck, X.; Hocquemiller, R.; Figadère, B. Tetrahedron Lett. 2002, 43, 3547–3549. doi:10.1016/S0040-4039(02)00568-3 |
| 15. | Martin, R.; Fürstner, A. Angew. Chem., Int. Ed. 2004, 43, 3955–3957. doi:10.1002/anie.200460504 |
| 16. | Scheiper, B.; Bonnekessel, M.; Krause, H.; Fürstner, A. J. Org. Chem. 2004, 69, 3943–3949. doi:10.1021/jo0498866 |
| 17. | Nakamura, M.; Matsuo, K.; Ito, S.; Nakamura, E. J. Am. Chem. Soc. 2004, 126, 3686–3687. doi:10.1021/ja049744t |
| 18. | Nagano, T.; Hayashi, T. Org. Lett. 2004, 6, 1297–1299. doi:10.1021/ol049779y |
| 22. | Yoshida, J. Flash Chemistry: Fast Organic Synthesis in Microsystems; Wiley: Hoboken, N.J., 2008. |
| 23. | Fletcher, P. D. I.; Haswell, S. J.; Pombo-Villar, E.; Warrington, B. H.; Watts, P.; Wong, S. Y. F.; Zhang, X. Tetrahedron 2002, 58, 4735–4757. doi:10.1016/S0040-4020(02)00432-5 |
| 24. | Jähnisch, K.; Hessel, V.; Löwe, H.; Baerns, M. Angew. Chem., Int. Ed. 2004, 43, 406–446. doi:10.1002/anie.200300577 |
| 25. | Yoshida, J. Chem. Commun. 2005, 4509–4516. doi:10.1039/b508341a |
| 26. | Doku, G. N.; Verboom, W.; Reinhoudt, D. N.; van den Berg, A. Tetrahedron 2005, 61, 2733–2742. doi:10.1016/j.tet.2005.01.028 |
| 27. | Yoshida, J.; Nagaki, A.; Iwasaki, T.; Suga, S. Chem. Eng. Technol. 2005, 28, 259–266. doi:10.1002/ceat.200407127 |
| 28. | Geyer, K.; Codee, J. D. C.; Seeberger, P. H. Chem.–Eur. J. 2006, 12, 8434–8442. doi:10.1002/chem.200600596 |
| 29. | Whitesides, G. Nature 2006, 442, 368–373. doi:10.1038/nature05058 |
| 30. | deMello, A. J. Nature 2006, 442, 394–402. doi:10.1038/nature05062 |
| 31. | Song, H.; Chen, D. L.; Ismagilov, R. F. Angew. Chem., Int. Ed. 2006, 45, 7336–7356. doi:10.1002/anie.200601554 |
| 32. | Kobayashi, J.; Mori, Y.; Kobayashi, S. Chem.–Asian J. 2006, 1, 22–35. doi:10.1002/asia.200600058 |
| 33. | Mason, B. P.; Price, K. E.; Steinbacher, J. L.; Bogdan, A. R.; McQuade, D. T. Chem. Rev. 2007, 107, 2300–2318. doi:10.1021/cr050944c |
| 34. | Ahmed-Omer, B.; Brandtand, J. C.; Wirth, T. Org. Biomol. Chem. 2007, 5, 733–740. doi:10.1039/b615072a |
| 35. | Watts, P.; Wiles, C. Chem. Commun. 2007, 443–467. doi:10.1039/b609428g |
| 36. | Yoshida, J.; Nagaki, A.; Yamada, T. Chem.–Eur. J. 2008, 14, 7450–7459. doi:10.1002/chem.200800582 |
| 37. | Fukuyama, T.; Rahman, M. T.; Sato, M.; Ryu, I. Synlett 2008, 151–163. doi:10.1055/s-2007-1000884 |
| 38. | McMullen, J. P.; Jensen, K. F. Annu. Rev. Anal. Chem. 2010, 3, 19–42. doi:10.1146/annurev.anchem.111808.073718 |
| 39. | Yoshida, J. Chem. Rec. 2010, 10, 332–341. doi:10.1002/tcr.201000020 |
| 40. | Yoshida, J.; Kim, H.; Nagaki, A. ChemSusChem 2011, 4, 331–340. doi:10.1002/cssc.201000271 |
| 41. | Watts, P.; Wiles, C.; Haswell, S. J.; Pombo-Villar, E.; Styring, P. Chem. Commun. 2001, 990–991. doi:10.1039/b102125g |
| 42. | Suga, S.; Okajima, M.; Fujiwara, K.; Yoshida, J. J. Am. Chem. Soc. 2001, 123, 7941–7942. doi:10.1021/ja015823i |
| 43. | Fukuyama, T.; Shinmen, M.; Nishitani, S.; Sato, M.; Ryu, I. Org. Lett. 2002, 4, 1691–1694. doi:10.1021/ol0257732 |
| 44. | Ueno, M.; Hisamoto, H.; Kitamori, T.; Kobayashi, S. Chem. Commun. 2003, 936–937. doi:10.1039/b301638b |
| 45. | Garcia-Egido, E.; Spikmans, V.; Wong, S. Y. F.; Warrington, B. H. Lab Chip 2003, 3, 73–76. doi:10.1039/b302381h |
| 46. | Lai, S. M.; Martin-Aranda, R.; Yeung, K. L. Chem. Commun. 2003, 218–219. doi:10.1039/b209297b |
| 47. | Mikami, K.; Yamanaka, M.; Islam, M. N.; Kudo, K.; Seino, N.; Shinoda, M. Tetrahedron Lett. 2003, 44, 7545–7548. doi:10.1016/S0040-4039(03)01835-5 |
| 48. | Kobayashi, J.; Mori, Y.; Okamoto, K.; Akiyama, R.; Ueno, M.; Kitamori, T.; Kobayashi, S. Science 2004, 304, 1305–1308. doi:10.1126/science.1096956 |
| 49. | Nagaki, A.; Kawamura, K.; Suga, S.; Ando, T.; Sawamoto, M.; Yoshida, J. J. Am. Chem. Soc. 2004, 126, 14702–14703. doi:10.1021/ja044879k |
| 50. | Lee, C.-C.; Sui, G.; Elizarov, A.; Shu, C. J.; Shin, Y.-S.; Dooley, A. N.; Huang, J.; Daridon, A.; Wyatt, P.; Stout, D.; Kolb, H. C.; Witte, O. N.; Satyamurthy, N.; Heath, J. R.; Phelps, M. E.; Quake, S. R.; Tseng, H.-R. Science 2005, 310, 1793–1796. doi:10.1126/science.1118919 |
| 51. | Horcajada, R.; Okajima, M.; Suga, S.; Yoshida, J. Chem. Commun. 2005, 1303–1305. doi:10.1039/b417388k |
| 52. | Kawaguchi, T.; Miyata, H.; Ataka, K.; Mae, K.; Yoshida, J. Angew. Chem., Int. Ed. 2005, 44, 2413–2416. doi:10.1002/anie.200462466 |
| 53. | Ducry, L.; Roberge, D. M. Angew. Chem., Int. Ed. 2005, 44, 7972–7975. doi:10.1002/anie.200502387 |
| 54. | Nagaki, A.; Togai, M.; Suga, S.; Aoki, N.; Mae, K.; Yoshida, J. J. Am. Chem. Soc. 2005, 127, 11666–11675. doi:10.1021/ja0527424 |
| 55. | He, P.; Watts, P.; Marken, F.; Haswell, S. J. Angew. Chem., Int. Ed. 2006, 45, 4146–4149. doi:10.1002/anie.200600951 |
| 56. | Uozumi, Y.; Yamada, Y.; Beppu, T.; Fukuyama, N.; Ueno, M.; Kitamori, T. J. Am. Chem. Soc. 2006, 128, 15994–15995. doi:10.1021/ja066697r |
| 57. | Tanaka, K.; Motomatsu, S.; Koyama, K.; Tanaka, S.; Fukase, K. Org. Lett. 2007, 9, 299–302. doi:10.1021/ol062777o |
| 58. | Sahoo, H. R.; Kralj, J. G.; Jensen, K. F. Angew. Chem., Int. Ed. 2007, 46, 5704–5708. doi:10.1002/anie.200701434 |
| 59. | Hornung, C. H.; Mackley, M. R.; Baxendale, I. R.; Ley, S. V. Org. Process Res. Dev. 2007, 11, 399–405. doi:10.1021/op700015f |
| 60. | Iwasaki, T.; Nagaki, A.; Yoshida, J. Chem. Commun. 2007, 1263–1265. doi:10.1039/b615159k |
| 61. | Nagaki, A.; Iwasaki, T.; Kawamura, K.; Yamada, D.; Suga, S.; Ando, T.; Sawamoto, M.; Yoshida, J. Chem.–Asian J. 2008, 3, 1558–1567. doi:10.1002/asia.200800081 |
| 62. | Nagaki, A.; Tomida, Y.; Yoshida, J. Macromolecules 2008, 41, 6322–6330. doi:10.1021/ma800769n |
| 63. | Fukuyama, T.; Kobayashi, M.; Rahman, M. T.; Kamata, N.; Ryu, I. Org. Lett. 2008, 10, 533–536. doi:10.1021/ol702718z |
| 64. | Nagaki, A.; Tomida, Y.; Miyazaki, A.; Yoshida, J. Macromolecules 2009, 42, 4384–4387. doi:10.1021/ma900551a |
| 65. | Tricotet, T.; O’Shea, D. F. Chem.–Eur. J. 2010, 16, 6678–6686. doi:10.1002/chem.200903284 |
| 66. | Greenway, G. M. S.; Haswell, J.; Morgan, D. O.; Skelton, V.; Styring, P. Sens. Actuators, B 2000, 63, 153–158. doi:10.1016/S0925-4005(00)00352-X |
| 67. | Haswell, S. J.; O'Sullivan, B.; Styring, P. Lab Chip 2001, 1, 164–166. doi:10.1039/b104035a |
| 68. | Niwa, S.; Eswaramoorthy, M.; Nair, J.; Raj, A.; Itoh, N.; Shoji, H.; Namba, T.; Mizukami, F. Science 2002, 295, 105–107. doi:10.1126/science.1066527 |
| 69. | Jas, G.; Kirschning, A. Chem.–Eur. J. 2003, 9, 5708–5723. doi:10.1002/chem.200305212 |
| 70. | Basheer, C.; Hussain, F. S. J.; Lee, H. K.; Valiyaveettil, S. Tetrahedron Lett. 2004, 45, 7297–7300. doi:10.1016/j.tetlet.2004.08.017 |
| 71. | Liu, S.; Fukuyama, T.; Sato, M.; Ryu, I. Org. Process Res. Dev. 2004, 8, 477–481. doi:10.1021/op034200h |
| 72. | Kunz, U.; Schönfeld, H.; Solodenko, W.; Jas, G.; Kirschning, A. Ind. Eng. Chem. Res. 2005, 44, 8458–8467. doi:10.1021/ie048891x |
| 73. | Comer, E.; Organ, M. G. J. Am. Chem. Soc. 2005, 127, 8160–8167. doi:10.1021/ja0512069 |
| 74. | Lee, C. K. Y.; Holmes, A. B.; Ley, S. V.; McConvey, I. F.; Al-Duri, B.; Leeke, G. A.; Santos, R. C. D.; Sevilled, J. P. K. Chem. Commun. 2005, 2175–2177. doi:10.1039/b418669a |
| 75. | Mauger, C.; Buisine, O.; Caravieilhes, S.; Mignani, G. J. Organomet. Chem. 2005, 690, 3627–3629. doi:10.1016/j.jorganchem.2005.03.071 |
| 76. | Kirschning, A.; Solodenko, W.; Mennecke, K. Chem.–Eur. J. 2006, 12, 5972–5990. doi:10.1002/chem.200600236 |
| 77. | Shore, G.; Morin, S.; Organ, M. G. Angew. Chem., Int. Ed. 2006, 45, 2761–2766. doi:10.1002/anie.200503600 |
| 78. | Shi, G.; Hong, F.; Liang, Q.; Fang, H.; Nelson, S.; Weber, S. G. Anal. Chem. 2006, 78, 1972–1979. doi:10.1021/ac051844+ |
| 79. | Rahman, M. T.; Fukuyama, T.; Kamata, N.; Sato, M.; Ryu, I. Chem. Commun. 2006, 2236–2238. doi:10.1039/b600970k |
| 80. | Murphy, E. R.; Martinelli, J. R.; Zaborenko, N.; Buchwald, S. L.; Jensen, K. F. Angew. Chem., Int. Ed. 2007, 46, 1734–1737. doi:10.1002/anie.200604175 |
| 81. | Kawanami, H.; Matsushima, K.; Sato, M.; Ikushima, Y. Angew. Chem., Int. Ed. 2007, 46, 5129–5132. doi:10.1002/anie.200700611 |
| 82. | Yamada, Y. M. A.; Watanabe, T.; Torii, K.; Uozumi, Y. Chem. Commun. 2009, 5594–5596. doi:10.1039/b912696a |
| 83. | Jin, J.; Cai, M.-M.; Li, J. Synlett 2009, 2534–2538. doi:10.1055/s-0029-1217730 |
| 84. | Ahmed-Omer, B.; Barrow, D. A.; Wirth, T. Tetrahedron Lett. 2009, 50, 3352–3355. doi:10.1016/j.tetlet.2009.02.133 |
| 85. | McMullen, J. P.; Stone, M. T.; Buchwald, S. L.; Jensen, K. F. Angew. Chem., Int. Ed. 2010, 49, 7076–7080. doi:10.1002/anie.201002590 |
| 7. | Lei, A.; Srivastava, M.; Zhang, X. J. Org. Chem. 2002, 67, 1969–1971. doi:10.1021/jo011098i |
| 6. | Sakellarios, E.; Kyrimis, T. Ber. Dtsch. Chem. Ges. 1924, 57, 322–326. doi:10.1002/cber.19240570233 |
| 20. | Cahiez, G.; Chaboche, C.; Mahuteau-Betzer, F.; Ahr, M. Org. Lett. 2005, 7, 1943–1946. doi:10.1021/ol050340v |
© 2011 Nagaki et al; licensee Beilstein-Institut.
This is an Open Access article under the terms of the Creative Commons Attribution License (http://creativecommons.org/licenses/by/2.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
The license is subject to the Beilstein Journal of Organic Chemistry terms and conditions: (http://www.beilstein-journals.org/bjoc)