Abstract
This review is the first to collate and summarize main data on named and unnamed rearrangement reactions of peroxides. It should be noted, that in the chemistry of peroxides two types of processes are considered under the term rearrangements. These are conventional rearrangements occurring with the retention of the molecular weight and transformations of one of the peroxide moieties after O–O-bond cleavage. Detailed information about the Baeyer−Villiger, Criegee, Hock, Kornblum−DeLaMare, Dakin, Elbs, Schenck, Smith, Wieland, and Story reactions is given. Unnamed rearrangements of organic peroxides and related processes are also analyzed. The rearrangements and related processes of important natural and synthetic peroxides are discussed separately.
Graphical Abstract
Introduction
The chemistry of organic peroxides has more than a hundred-year history. Currently, organic peroxides are widely used as oxidizing agents and initiators for free-radical reactions both in industry and in laboratory. These compounds are produced and involved in various natural and biological processes and were explored extensively as antimalarial agents, anthelmintics, and anticancer drugs.
Organic peroxides, such as alkyl hydroperoxides, aryl hydroperoxides, ketone peroxides, dialkyl peroxides, diacyl peroxides, peroxy esters, peroxydicarbonates, peroxyacetals, and inorganic peroxides are the most important radical initiators that are widely used in industrial processes in the manufacture of polymers from unsaturated monomers [1-9].
Nowadays, the progress in the chemistry of organic peroxides is mainly a result of their biological activity and pharmaceutical application. The search of effective antimalarial and antihelminthic drugs is the main challenge of medicinal chemistry of peroxides. According to the World Health Organization (WHO) malaria is a widely distributed illness. About 3.2 billion people remain at risk of malaria and in 2015 214 million cases of malaria and 438 thousands deaths from it have been registered [10]. Compounds with high antimalarial [11-23], antihelminthic [24-28], and antitumor activities [29-34] were found among natural, semisynthetic, and synthetic peroxides. The main biologically active frame of these compounds includes five-membered 1,2-dioxolane [35-37], 1,2,4-trioxolane [38,39], and six-membered 1,2-dioxane [40-42], 1,2-dioxene [43], 1,2,4-trioxane [22,44,45] cycles. The naturally occuring peroxide artemisinin and its semisynthetic derivatives, artemether, arteether, and artesunate, are applied in large scale for malaria treatment [46,47].
Organic peroxides, their rearrangements and related processes play an important role in the chemistry of oxidation processes. Thus, the key reagent in the Sharpless epoxidation of allylic alcohols [48] and in the manufacture of propylene oxide via the Prilezhaev reaction [49-51] is tert-butyl hydroperoxide. In industry, phenol and acetone are mainly produced by the Hock process, which is based on the rearrangement of cumene hydroperoxide. In 2003, phenol was produced to more than 95% by this oxidation process [52-54]. Another important application of organic peroxides is the synthesis of lactones from cyclic ketones via the Baeyer−Villiger oxidation and it is one of the methods for the synthesis of commercially important caprolactone from cyclohexanone with peracetic acid [55,56].
Autoxidation processes with formation of hydroperoxides and their subsequent free-radical transformations with generation of carbon- and oxygen-centered radicals are key reactions in the drying process of oil-based and alkyd paints containing double bonds [57-61].
Organic peroxides and their transformation play an important role not only in industrial but also in biological processes. Thus, the firefly luciferase-catalyzed oxidation of luciferin yields the peroxy compound 1,2-dioxetane. This four-membered peroxide cycle is unstable and spontaneously decays to carbon dioxide and excited ketones, which release excess energy through light emission (bioluminescence) [62-65]. The in vivo oxidation of cholesterol by singlet oxygen produces the hydroperoxide cholesterol-5α-OOH, which undergoes a Hock oxidation to form atheronal A. The latter possesses proatherogenic effects and triggers the development of cardiovascular diseases [66-71].
The development of the chemistry of organic peroxides is closely related to the application and preparation of unsaturated compounds, such as epoxides, aldehydes, ketones, carboxylic acids, and their derivatives [72-113]. Organic peroxides are widely used as oxidants in oxidative coupling processes [114-120].
Industrial-scale production of readily available and efficient initiators of free radical polymerization and effective biologically active compounds promotes the search for new synthetic methods for peroxides starting from carbonyl compounds, hydrogen peroxide, and hydroperoxides [121-182].
In many cases, rearrangements and related reactions of peroxides are key pathways in laboratory, industrial, and biological processes. The rearrangements of organic peroxides are covered in the literature in hundreds of publications and in several specialized and partial reviews [183-188]. The present review is the first to combine the key data on both, name rearrangements and less well-known rearrangements and related oxidative processes, and to summarize systematically related and different features of these reactions, compares their mechanisms, and assesses the prospects of their application.
By definition, a rearrangement is a migration of an atom or a group of atoms from one atom to another within the same molecule [189]. In contrast, a rearrangement of organic peroxides means a change in the structure of the starting molecule to form an isomeric compound without a peroxy group [183]. The terminology of rearrangements of organic peroxides and related processes encountered in the literature shows that this definition is not generally applicable as rearrangements of peroxides can give both isomeric and non-isomeric compounds either containing a peroxy group or without the latter. In most cases, a rearrangement involves the migration or cleavage of the peroxide group in an intermediate molecule, and the stability of the latter is responsible for the further pathway of the process.
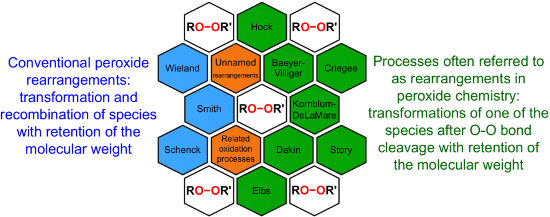
The review covers main studies published over the last 15–20 years with a brief excursion to the history of the development of various reactions and transformations. The review consists of three parts: the first part considers named transformations of organic peroxides (Figure 1), the second one deals with unnamed reactions, and the third part covers transformations of some important natural and synthetic peroxides. Since the term “rearrangements”, as applied to transformations of peroxides, is not clearly defined all parts of the review include processes related to rearrangements.
Figure 1: The named transformations considered in this review.
Figure 1: The named transformations considered in this review.
Review
1 Named rearrangements of organic peroxides
Rearrangements of organic peroxides are the key steps in many well-known processes such as the Baeyer–Villiger (BV), the Criegee and Hock reactions, the Kornblum–DeLaMare rearrangement, Dakin, and Elbs oxidation.
The BV oxidation is widely used in organic synthesis for the preparation of esters and lactones and the Criegee reaction is applied to transform tertiary alcohols into ketones and aldehydes. The Hock rearrangement is a key step in the cumene (cumene–phenol) process and the Kornblum–DeLaMare is an important tool in the synthesis of functionalized ketones and alcohols, including γ-hydroxy enones. The Dakin oxidation finds application for the synthesis of phenols from arylaldehydes or aryl ketones and the Elbs persulfate oxidation allows the preparation of hydroxyphenols from phenols. Finally, the Schenck and Smith rearrangements are of interest in allyl hydroperoxide transformations.
1.1 Baeyer–Villiger oxidation
The BV reaction is the oxidation of ketones or aldehydes A under the action of hydrogen peroxide, hydroperoxides, Caro’s acid (H2SO5), or organic peracids to yield esters, lactones, or carboxylic acids B (Scheme 1) [190,191].
Baeyer and Villiger accomplished the oxidation of ketones to esters for the first time in 1899 while they attempted the reaction of Caro’s acid (H2SO5) with menthone, tetrahydrocarvone, and camphor to transform these compounds into the corresponding lactones [192-194].
Since that time, this reaction has shown to be of general applicability and it has gained wide application for the oxidation of carbonyl compounds of different structures. In this reaction, cyclic ketones are transformed into lactones, acyclic ketones, into esters and aldehydes into carboxylic acids. The BV oxidation is one of the most important reactions in organic chemistry because it produces lactones, which are useful synthetic products in polymer, agrochemical, and pharmaceutical industry.
m-Chloroperbenzoic, peracetic, and perfluoroacetic acids, as well as hydrogen peroxide/protic acid, hydrogen peroxide/Lewis acid, and hydrogen peroxide/base systems are widely employed in the Baeyer–Villiger oxidation [185,194,195].
The general mechanism of the peracid-promoted Baeyer–Villiger oxidation involves two main steps. In the first step, the oxygen atom of the peroxide moiety of the peracid 2 binds to the carbonyl group of ketone 1 to form the tetrahedral intermediate 3 which is referred to as the Criegee intermediate. The next step involves the concerted migration of the R2 group to the peroxide oxygen atom, resulting in the formation of ester 4 and carboxylic acid 5 (Scheme 2).
Scheme 2: The general mechanism of the peracid-promoted Baeyer–Villiger oxidation.
Scheme 2: The general mechanism of the peracid-promoted Baeyer–Villiger oxidation.
The ability of peracids to oxidize cyclic and acyclic ketones and aldehydes to the corresponding lactones, esters, and carboxylic acids decreases in the series peroxotrifluoroacetic acid > monopermaleic acid > mono-o-perphthalic acid > 3,5-dinitroperbenzoic acid > p-nitroperbenzoic acid > MCPBA ≈ performic acid > perbenzoic acid > peracetic acid >> H2O2 > t-BuOOH [196].
The migratory ability of substituents in the Criegee intermediate decreases in the following series: tertiary alkyl > cyclohexyl > secondary alkyl > benzyl > phenyl > primary alkyl > cyclopentyl, cyclopropyl > methyl. In some cases, stereoelectronical effects strongly influence the regioselectivity of the reaction, specifically the ability of the migrating C–C to align with the back of the breaking O–O bond, and the presence or absence of strain in cyclic ketone substrates [197,198]. The strongest electron-donating group migrates in unsymmetrical ketones [199].
There are thousands of publications on the Baeyer–Villiger reaction. In the latest reviews published by Krow [195] in 1993 and by Renz and Meunier [185] in 1999, the field of application, the reactivity of substrates, and the reaction kinetics and mechanisms are considered in detail. In the review by Strukul, special emphasis was placed on metal-catalyzed Baeyer–Villiger oxidations [196]. Green approaches in the Baeyer–Villiger reaction were highlighted by another review [200].
The present review covers a more modern aspect of this reaction, viz., the performance of the process using hydrogen peroxide. Oxidizing systems containing hydrogen peroxide as the oxidizing agent allow the usual and asymmetric oxidation of the substrate to the target product with high conversion and yield. In recent years, the inexpensive, commercially available, and environmentally friendly H2O2 was utilized in the Baeyer–Villiger reaction with increasing frequency. Various catalysts that activate hydrogen peroxide, such as heterogeneous catalysts based on solid acids [201], zeolites [202,203], Se [204], As [205], Co [206], sulfonated organic ion exchange resins [203,207], and homogeneous catalysts based on Pt [208], Zr [209], Re [210,211], Se [212,213], As [205], Mo [214], Co [215], Brønsted [216], and Lewis acids [217] are described in the literature. The general mechanism of a Lewis acid-catalyzed Baeyer–Villiger rearrangement is presented in Scheme 3 [200,218].
Scheme 3: General mechanism of the Lewis acid-catalyzed Baeyer–Villiger rearrangement.
Scheme 3: General mechanism of the Lewis acid-catalyzed Baeyer–Villiger rearrangement.
Scheme 4 shows the theoretically studied mechanism of the oxidation reaction promoted by H2O2 and the Lewis acid BF3 [217,219]. In the first step, the hydrogen peroxide–boron trifluoride complex 8 reacts with ketone 9 to form adduct 10. The latter intermediate rearranges through transition state 11 into the tetrahedral peroxyacetal intermediate 12. Then BF3 migrates to another oxygen atom through transition state 13 to give the second Criegee intermediate 14. The decomposition of intermediate 14 finally produces 15, hydrogen fluoride (16) and ester 17.
Scheme 4: The theoretically studied mechanism of the BV oxidation reaction promoted by H2O2 and the Lewis acid BF3.
Scheme 4: The theoretically studied mechanism of the BV oxidation reaction promoted by H2O2 and the Lewis aci...
Despite the fact that the Baeyer–Villiger reaction is known since 1899, the mechanism of this reaction is still not fully understood. The nature of the acid catalyst [220] and the type of O–O-bond cleavage in the Criegee intermediate [221] were found to play an important role in this reaction. Probably the hydrogen bonds in Baeyer–Villiger reactions play an important role [222]. The tetramolecular transition states TS1 and TS2 are considered to be the two key steps determining the course of the oxidation: the nucleophilic addition of a peroxy acid molecule to ketone (TS1) and the migration of R and cleavage of O–O bond (TS2). Thus, electrophilic substrates favor TS1 and nucleophilic migrating groups prefer TS2 (Scheme 5).
Scheme 5: Proton movements in the transition states of the Baeyer–Villiger oxidation.
Scheme 5: Proton movements in the transition states of the Baeyer–Villiger oxidation.
The dependence of the course of the Baeyer–Villiger oxidation on the type of O−O-bond cleavage in the Criegee intermediate was studied in the oxidation reaction of 1,2-quinone 18 with perbenzoic acid [221]. The reaction gave two oxidation products – anhydride 20 and the seven-membered α-ketolactone 21. The investigation of the reaction mechanism demonstrated that the formation of the seven-membered α-ketolactone 21 proceeds through the heterolytic O–O-bond cleavage in Criegee intermediate 19, whereas the homolytic O–O cleavage affords anhydride 20 (Scheme 6).
Scheme 6: The dependence of the course of the Baeyer–Villiger oxidation on the type of O–O-bond cleavage in the Criegee intermediate.
Scheme 6: The dependence of the course of the Baeyer–Villiger oxidation on the type of O–O-bond cleavage in t...
The acid-catalyzed Baeyer–Villiger oxidation of cyclic epoxy ketones 22 produces lactones of type 23, which convert into carbenium ions 24 in the presence of the acid. Subsequently, these ions can be transformed with participation of H2O2 through three different pathways into dihydroperoxides 25, dicarboxylic acids 28, carboxylic acids 26, and keto carboxylic acids 27 (Scheme 7, Table 1) [223].
Scheme 7: The acid-catalyzed Baeyer–Villiger oxidation of cyclic epoxy ketones 22.
Scheme 7: The acid-catalyzed Baeyer–Villiger oxidation of cyclic epoxy ketones 22.
Table 1: Oxidation of cyclic epoxy ketones 22a–c by H2O2.
| Epoxy ketone | R | 25, % | 26, % | 27, % | 28, % |
|---|---|---|---|---|---|
| 22a | Me | 25a, 12 | 26a, 6 | 27a, 15 | 28a, 53 |
| 22b | Et | 25b, 19 | a | a | a |
| 22c | Ph | 25c, 19 | – | 27c, 35 | 28c, 18 |
aThe aqueous phase consisted of a complex mixture and could not be analyzed.
The oxidation of isophorone oxide (29) is an industrial process for the production of dimethylglutaric acid 30 (Scheme 8) [223].
Scheme 8: Oxidation of isophorone oxide 29.
Scheme 8: Oxidation of isophorone oxide 29.
Acyl phosphate 32 can be synthesized from acyl phosphonate 31 in high yield by oxidation with H2O2 (Scheme 9) [224].
Scheme 9: Synthesis of acyl phosphate 32 from acyl phosphonate 31.
Scheme 9: Synthesis of acyl phosphate 32 from acyl phosphonate 31.
The Baeyer–Villiger oxidation provides a valuable tool for the synthesis of oxygenated natural products [218,225,226] as exemplified by the synthesis of aflatoxin B2 (36, Scheme 10) [227].
Scheme 10: Synthesis of aflatoxin B2 (36).
Scheme 10: Synthesis of aflatoxin B2 (36).
The Baeyer–Villiger reaction is also a key step in the multistep synthesis of cannabinergic lactones from dimethylheptylresorcinol. Two regioisomeric cannabinergic lactones were obtained, one of which possessed pronounced affinity towards the CB1 receptor and lower affinities for mCB2 and hCB2 receptors [228].
Oxidation with H2O2–acid systems: With in situ generated peracids from carbodiimide, hydrogen peroxide, and carboxylic acids as catalysts ketones 37 are rearranged to lactones 38 (Scheme 11) [229].
Scheme 11: The Baeyer–Villiger rearrangement of ketones 37 to lactones 38.
Scheme 11: The Baeyer–Villiger rearrangement of ketones 37 to lactones 38.
3,4-Dimethoxybenzoic acid (40) was prepared with 78% yield by a Baeyer–Villiger reaction of substrate 39 with 30% H2O2, HCOOH and 1,2-dichloroethane at 50 °C for 24 h (Scheme 12) [230].
Scheme 12: Synthesis of 3,4-dimethoxybenzoic acid (40) via Baeyer–Villiger oxidation.
Scheme 12: Synthesis of 3,4-dimethoxybenzoic acid (40) via Baeyer–Villiger oxidation.
Oxone is a convenient reagent for the transformation of α,β-unsaturated ketones 43 of determined stereochemistry into vinyl acetates 44 via the Baeyer–Villiger reaction in dry DMF for 7–39 h (Scheme 13) [231].
Scheme 13: Oxone transforms α,β-unsaturated ketones 43 into vinyl acetates 44.
Scheme 13: Oxone transforms α,β-unsaturated ketones 43 into vinyl acetates 44.
Oxidation with H2O2–heteroorganic catalyst systems: The activity of oxidizing systems such as H2O2/aryl benzyl selenoxide and H2O2/diaryl diselenide is similar to that of m-chloroperbenzoic acid [212,232,233]. The main advantage of these selenium-containing systems is that the catalysts are regenerated and can therefore be used at low loadings [234-236]. Some results of the oxidation of ketones and aldehydes 45a–c to the corresponding esters 46a–c using the H2O2/aryl benzyl selenoxide system are collected in Table 2 [232].
The oxidation results of ketones 47a,b and aldehydes 47c–e to lactones 48a,b and carboxylic acids 49a–c promoted by the H2O2/diaryl diselenide system is presented in Table 3 [212].
Table 3: Baeyer–Villiger oxidation of ketones 47a,b and aldehydes 47c–e promoted by the H2O2/diaryl diselenide system.
|
|
||||
| Ketone | Time, h | Product | Conversion, %a | Selectivity (BV product), %a |
|---|---|---|---|---|
|
47a |
1 |
48a |
99 | 90 |
|
47b |
8 |
48b |
95 | 94 |
|
47cb |
2 |
49a |
98 | 98 |
|
47db |
3 |
49b |
88 | 96 |
|
47eb |
3 |
49c |
>90 | 99 |
aDetermined by GC; b60 °C.
In the first step of the catalytic cycle of the Baeyer–Villiger oxidation using diaryl diselenide 50 and hydrogen peroxide seleninic acid 51 is generated, which is then oxidized to perseleninic acid 52. Oxidation of the ketone 45 by perseleninic acid 52 involves the intermediate peroxide 53 (Scheme 14) [235].
Scheme 14: The Baeyer–Villiger oxidation of ketones 45 using diaryl diselenide and hydrogen peroxide.
Scheme 14: The Baeyer–Villiger oxidation of ketones 45 using diaryl diselenide and hydrogen peroxide.
Similarly, the versatile 4-methylenebutanolides 55 can be prepared from (E)-2-methylenecyclobutanones 54 in the presence of (PhSe)2/H2O2 at room temperature (Scheme 15). Likely the Baeyer–Villiger reaction proceeds through the formation of benzeneseleninoperoxoic anhydride [PhSe(O)O]2O in the first step, which then transforms to the active oxidant benzeneseleninoperoxoic acid PhSe(O)OOH [233].
Scheme 15: Baeyer–Villiger oxidation of (E)-2-methylenecyclobutanones.
Scheme 15: Baeyer–Villiger oxidation of (E)-2-methylenecyclobutanones.
The Baeyer–Villiger oxidation of (E)-α,β-unsaturated ketones to (E)-vinyl esters was performed with hydrogen peroxide and dibenzyl diselenide as pre-catalyst at room temperature [236]. Catalyzed by the dibenzyl diselenide, β-ionone (56) was oxidized by H2O2 with formation of (E)-2-(2,6,6-trimethylcyclohex-1-en-1-yl)vinyl acetate (57) with 91% yield (Scheme 16) [237].
Scheme 16: Oxidation of β-ionone (56) by H2O2/(BnSe)2 with formation of (E)-2-(2,6,6-trimethylcyclohex-1-en-1-yl)vinyl acetate (57).
Scheme 16: Oxidation of β-ionone (56) by H2O2/(BnSe)2 with formation of (E)-2-(2,6,6-trimethylcyclohex-1-en-1-...
The Baeyer–Villiger oxidation of ketones 58a–f to form esters 59a–f can be accomplished in good yields in the presence of H2O2 and arsenic-containing ion exchange resins on polystyrene as the catalyst (Table 4) [203,205].
The mechanism of the oxidation of ketones 58a–f by hydrogen peroxide in the presence of arsonated polystyrene 60 as the catalyst is shown in Scheme 17. First, hydrogen peroxide reacts with the arsonic acid 60 to form peroxyarsonic acid 61 or it adds to ketones 58a–f to form vicinal hydroperoxyalkanols 63. In the second step the peroxyarsonic acid 61 adds to ketones 58a–f or the vicinal hydroperoxyalkanols 63 interact with arsonated polystyrene 60 under formation of perester 62. Finally, the decomposition of 62 gives esters 59a–f.
Scheme 17: The mechanism of oxidation of ketones 58a–f by hydrogen peroxide in the presence of arsonated polystyrene 60.
Scheme 17: The mechanism of oxidation of ketones 58a–f by hydrogen peroxide in the presence of arsonated polys...
A number of other modern oxidizing systems are based on transition metal-peroxo complexes. The use of transition metal complexes were also used as catalysts for the Baeyer–Villiger reaction and the first example was documented in 1978 [196,214]. For example, Mo(VI) peroxo complexes 64 and 65 were employed as the catalysts and 90% H2O2 served as the oxidizer (Table 5).
The results obtained from the reactions using molybdenum systems have stimulated the search for new catalysts based on transition metal complexes. The usage of the platimum complex [(dppe)Pt(CF3(CH2Cl2)]BF4 (66·BF4) allowed the oxidation of 2-methylcyclohexanone (58b) in the presence of 32% H2O2 at room temperature to form 6-methylcaprolactone (59b) in 22% yield (Scheme 18) [238].
Scheme 18: Oxidation of ketone (58b) by H2O2 to 6-methylcaprolactone (59b) catalyzed by Pt complex 66·BF4.
Scheme 18: Oxidation of ketone (58b) by H2O2 to 6-methylcaprolactone (59b) catalyzed by Pt complex 66·BF4.
Acyclic ketones 67 could be oxidized to the corresponding esters 68 in the presence of the catalyst [(dppb}Pt(µ-OH)]22+, where dppb is butane-1,4-diylbis(diphenylphosphane) (Scheme 19) [208].
Scheme 19: Oxidation of ketones 67 with H2O2 in the presence of [(dppb}Pt(µ-OH)]22+.
Scheme 19: Oxidation of ketones 67 with H2O2 in the presence of [(dppb}Pt(µ-OH)]22+.
The oxidation mechanism of ketones 67 is displayed in Scheme 20.
Scheme 20: The mechanism of oxidation of ketones 67 in the presence of [(dppb}Pt(µ-OH)]22+ and H2O2.
Scheme 20: The mechanism of oxidation of ketones 67 in the presence of [(dppb}Pt(µ-OH)]22+ and H2O2.
The use of variable-valence metal complexes opened up a new field of application of the Baeyer–Villiger oxidation and there are now dozens of studies on this topic [196,239-246].
Hydroxylated and methoxylated benzaldehydes 69 (Scheme 21) and acetophenones 72 (Scheme 22) can be oxidized to the corresponding phenols 70a–d, and 73 in good yields in the presence of the H2O2/MeReO3 system in ionic liquids [bmim]BF4 or [bmim]PF6 [247]. Benzoic acids 71a–d, 74 and phenyl esters 75a–d were reported as oxidation byproducts.
Scheme 21: Oxidation of benzaldehydes 69 in the presence of the H2O2/MeReO3 system.
Scheme 21: Oxidation of benzaldehydes 69 in the presence of the H2O2/MeReO3 system.
Scheme 22: Oxidation of acetophenones 72 in the presence of the H2O2/MeReO3 system.
Scheme 22: Oxidation of acetophenones 72 in the presence of the H2O2/MeReO3 system.
Sn-containing mesoporous silica nanospheres (Sn-MSNSs) with uniform crater-like mesopores exhibited high activities in the Baeyer–Villiger oxidation of 2-adamantanone (45c) (Scheme 23) [248].
Scheme 23: Baeyer–Villiger oxidation of 2-adamantanone (45c) in the presence of Sn-containing mesoporous silica nanospheres (Sn-MSNSs).
Scheme 23: Baeyer–Villiger oxidation of 2-adamantanone (45c) in the presence of Sn-containing mesoporous silic...
The Baeyer–Villiger rearrangement of 2-adamantanone (45с) was performed using hydrogen peroxide (H2O2) and stannosilicate zeolites with nanosheet morphology and MFI topology (Sn-MFI-ns) as highly efficient catalysts [249]. The Sn-beta zeolites prepared by a steam-assisted conversion method are efficient catalysts for the Baeyer–Villiger reaction of cyclohexanone to ε-caprolactone [250]. A mesoporous Mg–Al-mixed oxide showed good catalytic efficiency in the Baeyer–Villiger oxidation of a series of ketones to the corresponding lactones and esters in the presence of diluted aqueous H2O2 and benzonitrile [251].
The Baeyer–Villiger oxidation of ketones 76 under the action of oxygen to the related esters 77 was performed using metal-free carbon (Ketjen Black) as a solid catalyst and benzaldehyde as the sacrificing agent. This metal-free carbon catalyst showed excellent catalytic activity and can be recycled after the reaction under oxygen atmosphere at 50 °C (Scheme 24) [252].
Scheme 24: Aerobic Baeyer–Villiger oxidation of ketones 76 using metal-free carbon.
Scheme 24: Aerobic Baeyer–Villiger oxidation of ketones 76 using metal-free carbon.
The boron-containing catalysts LiB(C6F5)4 or Ca[B(C6F5)4]2 were developed for the Baeyer–Villiger oxidation of ketones with aqueous H2O2 to give the lactones in high yields [253,254].
A regioselective Baeyer–Villiger oxidation of functionalized cyclohexenones 78 lead to dihydrooxepine structures 79. Here, the combination of SnCl4 and bis(trimethylsilyl)peroxide (BTSP), in the presence of trans-1,2-diaminocyclohexane as the ligand, generated the desired products 79 in high yields (Scheme 25) [255].
Scheme 25: A regioselective Baeyer-Villiger oxidation of functionalized cyclohexenones 78 into a dihydrooxepine structures 79.
Scheme 25: A regioselective Baeyer-Villiger oxidation of functionalized cyclohexenones 78 into a dihydrooxepin...
The Co4HP2Mo15V3O62-catalyzed oxidation of aldehydes and ketones 80 by hydrogen peroxide in ionic liquid [TEBSA][BF4] resulted in carboxylic acids and esters 81 in good to high yields (Scheme 26) [256].
Scheme 26: The oxidation of aldehydes and ketones 80 by H2O2 catalyzed by Co4HP2Mo15V3O62.
Scheme 26: The oxidation of aldehydes and ketones 80 by H2O2 catalyzed by Co4HP2Mo15V3O62.
Oxidation with H2O2–base systems: The oxidative cleavage of ketones 82 with hydrogen peroxide in alkaline solution yielded carboxylic acids 84. The authors suggested that the reaction of a ketone with the hydroperoxide anion resulted in the intermediate esters 83, which hydrolyzed in the basic reaction medium with formation of acids 84 (Scheme 27) [257].
Scheme 27: The cleavage of ketones 82 with hydrogen peroxide in alkaline solution.
Scheme 27: The cleavage of ketones 82 with hydrogen peroxide in alkaline solution.
The use of hydrotalcites in the Baeyer–Villiger oxidation of various ketones resulted in high yields of the corresponding lactones or esters [258-260]. The esters 86 were synthesized by the reaction of ketones 85 with H2O2 and benzonitrile under basic reaction conditions (KHCO3) with the intermediate generation of peroxyimidic acids. This oxidation can be successfully applied to alkyl-containing ketones to give the target products in yields of 30–91% and good regioselectivity 7:1 to 20:1 (Scheme 28) [261].
Scheme 28: Oxidation of ketones 85 to esters 86 with H2O2–urea in the presence of KHCO3.
Scheme 28: Oxidation of ketones 85 to esters 86 with H2O2–urea in the presence of KHCO3.
Asymmetric oxidation: Asymmetric Baeyer–Villiger oxidation reactions can be performed using chiral acetals, organic hydroperoxides, chiral metal complexes and organocatalysts [262,263]. There are also Green chemistry approaches for Baeyer−Villiger oxidations based on enzyme-mediated processes, which are used for the preparation of chiral lactones. This type of biocatalysis is useful in synthetic chemistry and either isolated enzymes or living whole cells are applied for the oxidative production of valuable intermediates [264-269].
The asymmetric oxidation of 3-substituted cyclopentane-1,2-diones 87a–f is an efficient tool in organic synthesis for the preparation of unsymmetrical γ-lactone acids 88a–f with high optical purity and good yields (Table 6). These γ-lactone acids are valuable substrates for the synthesis of compounds with potentially useful pharmacological properties, such as homocitrates, alkyl- and aryl-substituted nucleosides [270-272].
The reaction starts with an asymmetric epoxidation of the substituted cyclopentane-1,2-dione 87a to form epoxide 89a. The second step involves the Baeyer–Villiger oxidation of epoxide 89a to peroxide 90a followed by the rearrangement into intermediate 91a. The latter is hydrolyzed by H2O to form dicarboxylic acid 92a, which is cyclized under the acidic conditions to γ-lactone acid 88a (Scheme 29) [270].
Scheme 29: Mechanism of the asymmetric oxidation of cyclopentane-1,2-dione 87a with the Ti(OiPr)4/(+)DET/t-BuOOH system.
Scheme 29: Mechanism of the asymmetric oxidation of cyclopentane-1,2-dione 87a with the Ti(OiPr)4/(+)DET/t-BuO...
In most cases, the Baeyer–Villiger oxidation is a stereospecific and regioselective process with retention of the configuration. The oxidation of cis-4-tert-butyl-2-fluorocyclohexanone (93) with m-chloroperbenzoic acid in the presence of NaHCO3 affords fluorolactones 94 and 95 in 91% and 9% yields, respectively (Scheme 30) [273].
Scheme 30: The oxidation of cis-4-tert-butyl-2-fluorocyclohexanone (93) with m-chloroperbenzoic acid.
Scheme 30: The oxidation of cis-4-tert-butyl-2-fluorocyclohexanone (93) with m-chloroperbenzoic acid.
However, in order to perform the asymmetric oxidation of 3-substituted cyclobutanones 96a–f to the corresponding lactones 97a–f (Table 7) [274], it is necessary to employ chiral Brønsted acids [274-277], organocatalysts [278,279] or enzymes [280-282] as the catalyst. The obtained asymmetric oxidation products can be used in the multistep synthesis of new biologically active compounds.
Possible mechanisms for the asymmetric oxidation of 3-substituted cyclobutanone 96a with H2O2 catalyzed by chiral phosphoric acid are presented in Scheme 31 [275].
Scheme 31: The mechanism of the asymmetric oxidation of 3-substituted cyclobutanone 96a in the presence of chiral phosphoric acid.
Scheme 31: The mechanism of the asymmetric oxidation of 3-substituted cyclobutanone 96a in the presence of chi...
A number of optically active ε- and γ-lactones 99, 100 was prepared by the enantioselective Baeyer–Villiger oxidation of racemic cyclic ketones 98 in up to 99% yield and 95% ee using the chiral N,N′-dioxide–Sc(III) complex as catalyst (Scheme 32) [283].
Scheme 32: Enantioselective Baeyer–Villiger oxidation of cyclic ketones 98.
Scheme 32: Enantioselective Baeyer–Villiger oxidation of cyclic ketones 98.
In another work, a chiral N,N′-dioxide–Sc(III) complex promoted Baeyer–Villiger oxidation was applied as instrument for a kinetic resolution of racemic 2-substituted cyclopentanones with formation of the 6-substituted δ-lactones in up to 98% ee and >95% regioselectivity [284].
A highly regio- and enantioselective Baeyer–Villiger oxidation of cyclic ketones 101 bearing amido, ureido, or sulfonamido functional groups to lactones 102 and 103 was carried out using the peptide-based catalyst 104. Hydrogen-bonding interactions are responsible for both types of selectivity. Notably, a reversal of the typically seen selectivity was observed with the peptide catalyst (Scheme 33) [285].
Scheme 33: Regio- and enantioselective Baeyer–Villiger oxidation of cyclic ketones 101.
Scheme 33: Regio- and enantioselective Baeyer–Villiger oxidation of cyclic ketones 101.
Versatility of the Baeyer–Villiger reaction with respect to starting reactants: The Baeyer–Villiger reaction cannot only be performed with ketones but also with acetals and aldimines as the starting substrates. The oxidation of cycloalkanone acetals 105a–g with performic acid generated in situ provides a new route to dicarboxylic acids 106a–g and hydroxycarboxylic acids 107a–g (Table 8) [286].
The proposed mechanism of the oxidation of acetal 105f is shown in Scheme 34.
Scheme 34: The proposed mechanism of the Baeyer–Villiger oxidation of acetal 105f.
Scheme 34: The proposed mechanism of the Baeyer–Villiger oxidation of acetal 105f.
In the first step of the reaction, the elimination of methanol from 105f and formation of 108 takes place. Probably perester 109 is formed alongside of 108. After formation of 109, the reaction proceeds by two different routes A and B (second stage). The first route A leads to formation of epoxide 110, whereas the second route (B) proceeds through the Baeyer–Villiger reaction with formation of lactone 111 and subsequent acid hydrolysis to give 107f. At the third stage (route A), ether 112 is formed from 110 and subsequently rearranged by a Baeyer–Villiger reaction into 113, which is oxidized to form 106f.
This method can be applied to the synthesis of dodecanedioic acid, which is used in anticorrosive composites, polyester and polyamide threads, and lubricants, for the synthesis of tridecanedioic acid, and as a component of perfume formulations.
Scheme 35 presents the synthesis of hydroxy-10H-acridin-9-one 117 starting from tetramethoxyanthracene 114 through the formation of peroxide 115, which rearranges through an acid-catalyzed Baeyer–Villiger-type rearrangement into 116. Hydroxy-10H-acridin-9-ones 117 proved to be promising antipsoriatic agents [287].
Scheme 35: Synthesis of hydroxy-10H-acridin-9-one 117 from tetramethoxyanthracene 114.
Scheme 35: Synthesis of hydroxy-10H-acridin-9-one 117 from tetramethoxyanthracene 114.
The oxidation of aldimines 118a–f with m-chloroperbenzoic acid in the presence of boron trifluoride etherate produces amides 119a–f in good yields (Table 9). The products of this transformation are strongly dependent on the electronic properties of the aromatic substituents at the carbon atom of the aldimines [288]. In the case of electron-donating substituents on the aryl fragment (Ar), formamides 119a–c are obtained as the result of imine oxidation and aryl migration. On the other hand, electron-withdrawing substituents on the aryl group (Ar) promote the formation of amides 119d–f as result of hydride migration.
Table 9: Oxidation of aldimines 118a-f to amides by m-CPBA-BF3·Et2O system.
|
|
|||
| Compound | Imine | Product | Yield, % |
|---|---|---|---|
| 118a | C6H5CH=NC6H5 | HCON(C6H5)2 | 82 |
| 118b | p-Me-C6H4CH=NC6H5 | HCONC6H5 p-Me-C6H4 | 90 |
| 118c | p-MeO-C6H4CH=NC6H5 | HCONC6H5 p-MeO-C6H4 | 91 |
| 118d | p-NO2-C6H4CH=NC6H5 | p-NO2-C6H4CONHC6H5 | 71 |
| 118e | p-NC-C6H4CH=NC6H5 | p-NC-C6H4CONHC6H5 | 79 |
| 118f | p-F3C-C6H4CH=NC6H5 | p-F3C-C6H4CONHC6H5 | 75 |
The sterically hindered and fully substituted pyrrole 120 underwent a Baeyer–Villiger reaction to yield a 4,5-dihydro-1H-ketopyrrole 121 (Scheme 36) [289].
Scheme 36: The Baeyer–Villiger oxidation of the fully substituted pyrrole 120.
Scheme 36: The Baeyer–Villiger oxidation of the fully substituted pyrrole 120.
1.2 Criegee rearrangement
The Criegee rearrangement involves the transformation of a peroxide, mainly peroxyesters B, into carbonates, esters, or ketones C and alcohols D through an oxygen insertion or consecutive oxygen insertions. The peroxyester B is initially prepared from a tertiary alcohol A and a peracid. In addition, the peroxy ester can also be prepared via the reaction of a ketone and a peracid (i.e., through a Baeyer–Villiger oxidation); the additional product of peracid to ketone is often referred to as the Criegee intermediate. From this point of view, the Baeyer–Villiger oxidation is a subset of the Criegee rearrangement (Scheme 37) [290].
As mentioned above the Criegee reaction and the Baeyer–Villiger oxidation are related processes and both reactions involve the formation of the Criegee intermediate. The distinguishing feature of the Criegee rearrangement is that the Criegee intermediate rearranges into a carbocation. The mechanism of the Criegee reaction is presented in Scheme 38.
Scheme 38: The mechanism of the Criegee reaction of a peracid with a tertiary alcohol 122.
Scheme 38: The mechanism of the Criegee reaction of a peracid with a tertiary alcohol 122.
Initially the reaction of the peracid with the tertiary alcohol 122 produces perester (Criegee intermediate) 123. One alkyl substituent migrates from the carbon atom to the adjacent oxygen atom and replaces the carboxylic acid moiety to form carbocation 124. Then, the addition of water to carbocation 124 affords ketone 125 and alcohol 126. p-Nitroperbenzoic acid is usually used to oxidize tertiary alcohols because the anion of this acid is a good leaving group.
The Criegee rearrangement was discovered in 1944 in the reaction of decaline ethylperoxoate 127 that rearranged into isomeric ester ketal 128 (Scheme 39) [291].
Scheme 39: Criegee rearrangement of decaline ethylperoxoate 127 into ketal 128.
Scheme 39: Criegee rearrangement of decaline ethylperoxoate 127 into ketal 128.
The mechanism of the Criegee rearrangement was studied using 2-alkoxy-2-propyl per-4-nitrobenzoates [292]. It was shown that the ionic cleavage of 2-methoxy-2-propyl perester 129 to p-nitrobenzoic acid (132), methyl acetate (133) and dimethyl ether (134) occurred through transition state 130 with generation of dimethoxycarbonium ion 131 (Scheme 40).
Scheme 40: The ionic cleavage of 2-methoxy-2-propyl perester 129.
Scheme 40: The ionic cleavage of 2-methoxy-2-propyl perester 129.
Investigations using aromatic peroxy esters 129 demonstrated that the migratory ability of the migrating group R decreases in the series t-Bu > C6H5 > iPr > OEt > OMe > Et > Me [293,294].
The Criegee rearrangement of α-methoxy hydroperoxide 136 obtained from (+)-trans-dihydrocarvone 135 produces trans-5-acetoxy-2-methylcyclohexanone 137 and intermediate peroxyacetate 138 (Scheme 41) [295].
Scheme 41: The Criegee rearrangement of α-methoxy hydroperoxide 136.
Scheme 41: The Criegee rearrangement of α-methoxy hydroperoxide 136.
Later on, the Criegee rearrangement was extended [296] to peroxides 139, 142, and 145 which made it possible to selectively synthesize both cyclic 140, 141, 144 and acyclic enol esters 146 and acetal 143 (Scheme 42).
Scheme 42: Synthesis of enol esters and acetals via the Criegee rearrangement.
Scheme 42: Synthesis of enol esters and acetals via the Criegee rearrangement.
The Criegee rearrangement of 1-hydroperoxy-2-oxabicycloalkanes 147a–d in formic or acetic acid containing catalytic amounts of sulfuric acid affords ω-alkoxy-(ω-3)-hydroxyalkanoic acid lactones 148a–d and 149a–d (Table 10) [297].
Table 10: Synthesis of ω-alkoxy-(ω-3)-hydroxyalkanoic acid lactones 148a–d and 149a–d from 1-hydroperoxy-2-oxabicycloalkanones 147a–d.
|
|
||||
| Substrate | RCOOH | Time (min) | Lactone | Yield, % |
|---|---|---|---|---|
| 147a | HCOOH | 20 | 148a | 64 |
| 147a | AcOH | 20 | 149a | 65 |
| 147b | HCOOH | 20 | 148b | 68 |
| 147b | AcOH | 20 | 149b | 70 |
| 147c | HCOOH | 30 | 148c | 57 |
| 147c | AcOH | 30 | 149c | 65 |
| 147d | HCOOH | 90 | 148d | 68 |
| 147d | AcOH | 90 | 149d | 53 |
The transformation of 1-hydroperoxy-2-oxabicycloalkanones 147a–d into ω-alkoxy-(ω-3)-hydroxyalkanoic acid lactones 148a–d and 149a–d is proposed to occur through intermediate peroxy ester 150 (Scheme 43).
Scheme 43: Proposed mechanism of the transformation of 1-hydroperoxy-2-oxabicycloalkanones 147a–d.
Scheme 43: Proposed mechanism of the transformation of 1-hydroperoxy-2-oxabicycloalkanones 147a–d.
1,2-Dioxolanes and related cyclic systems have attracted considerable attention from synthetic chemists as they may be used for the preparation of biologically active compounds. Under acidic conditions, 3-hydroxy-1,2-dioxolanes 151 are rearranged similarly to the Criegee mechanism into diketone derivatives 152 (Scheme 44) [298].
Scheme 44: Transformation of 3-hydroxy-1,2-dioxolanes 151 into diketone derivatives 152.
Scheme 44: Transformation of 3-hydroxy-1,2-dioxolanes 151 into diketone derivatives 152.
Unlike the Baeyer–Villiger rearrangement, in which only mono-O-insertion can take place, the Criegee rearrangement of peroxide 153 in an acidic medium and under solvent-free conditions does not have such limitations. Thus, the latter reaction can proceed sequentially through the mono-, di-, and tri-O-insertion steps with formation of ketone 154, ester 155 and carbonate ester 156 (Scheme 45) [299,300].
Scheme 45: Criegee rearrangement of peroxide 153 with the mono-, di-, and tri-O-insertion.
Scheme 45: Criegee rearrangement of peroxide 153 with the mono-, di-, and tri-O-insertion.
The selective double Criegee rearrangement next to a tertiary carbon was shown in the oxidative fragmentation at the bridgehead position of adamantanes 157a,b. The reaction employed the trifluoroperacetic acid (TFPAA)/trifluoroacetic acid (TFAA) system and afforded compounds 158a,b in high yields (Scheme 46) [300].
Scheme 46: The sequential Criegee rearrangements of adamantanes 157a,b.
Scheme 46: The sequential Criegee rearrangements of adamantanes 157a,b.
This method for the insertion of an oxygen atom was applied to the oxidation of triarylmethanols 159a–d [299]. The successive insertion of oxygen atoms gave rise to diaryl carbonates 160a–d in good yields (Scheme 47).
Scheme 47: Synthesis of diaryl carbonates 160a–d from triarylmethanols 159a–d through successive oxygen insertion.
Scheme 47: Synthesis of diaryl carbonates 160a–d from triarylmethanols 159a–d through successive oxygen insert...
In the last years, new enantiospecific approaches for the synthesis of sesquiterpenes 162 from ketone 161 were developed [301-307]. In these methods, the Criegee rearrangement represents one key step and one example is presented in Scheme 48 [303].
Scheme 48: The synthesis of sesquiterpenes 162 from ketone 161 with a Criegee rearrangement as one key step.
Scheme 48: The synthesis of sesquiterpenes 162 from ketone 161 with a Criegee rearrangement as one key step.
A method for the large-scale synthesis of a trans-hydrindan derivatives 164, 165 related to vitamin D, based on the Criegee rearrangement of alkene 163 was realized (Scheme 49) [308].
Scheme 49: Synthesis of trans-hydrindan derivatives 164, 165.
Scheme 49: Synthesis of trans-hydrindan derivatives 164, 165.
Carbonyl oxides (Criegee intermediates) are one of the most important compounds in tropospheric chemistry [309]. Direct investigations of formaldehyde oxide (CH2OO) or acetaldehyde oxide (CH3CHOO) reactions with water vapor, SO2, NO2 were carried out [310-312].
1.3 Hock rearrangement
The Hock rearrangement is a protic or Lewis acid-promoted rearrangement of hydroperoxides A resulting in a C–C bond cleavage to form alcohol B and carbonyl compound C (Scheme 50) [313].
The Hock rearrangement is a key step in the cumene process, which is used for the industrial production of phenol (170) and acetone (171) from benzene (166) and propylene (167) in the presence of air and radical initiators. The cumene process was described by Udris and Sergeev in 1947 [314,315] and independently by Hock in 1944 [316,317]. The general scheme of the cumene process, involving the formation of cumene hydroperoxide is shown in Scheme 51.
Scheme 51: The general scheme of the cumene process.
Scheme 51: The general scheme of the cumene process.
The cumene process involves the acid-catalyzed rearrangement of cumene hydroperoxide (168) as a key step. The reaction starts with the protonation of the terminal oxygen atom of cumene hydroperoxide (168) followed by the migration of the phenyl group from the benzylic carbon atom to the peroxide oxygen atom and the elimination of a water molecule to form carbocation 169. The carbocation 169 is attacked by a water molecule, a proton is transferred to the oxygen atom attached to the phenyl group, and finally the cleavage of the adduct yields phenol (170) and acetone (171).
The Hock rearrangement of aliphatic hydroperoxides proceeds quite readily in concentrated H2SO4 [318] or superacids [319] (Scheme 52). This is associated with higher resistance of these compounds toward acid-catalyzed rearrangements compared with benzylic or allylic hydroperoxides. For example, aliphatic hydroperoxides are not cleaved in 5–50% aqueous H2SO4 but on the contrary, these compounds are produced under these conditions. More efficient catalysts are the compounds Sn(OTf)2 and La(OTf)3 which can be used for the transformation of 2-hydroperoxy-2,4,4-trimethylpentane (172) into neopentyl alcohol (173) and acetone (171). The Sn(OTf)2 and La(OTf)3-catalyzed reaction afforded neopentyl alcohol (173) in 62 and 70% yield, respectively [320].
Scheme 52: The Hock rearrangement of aliphatic hydroperoxides.
Scheme 52: The Hock rearrangement of aliphatic hydroperoxides.
The hydrogen peroxide promoted ring expansion for the synthesis of oxabicycles 176a–c was described for the first time in 1985 [321]. The reaction involved the solvolysis of homoallylic brosylates 174a–c or spiro cyclopropyl carbinols 175a–c in the THF/H2O2 system, resulting in the increase in the ring size by two atoms and the formation of hydroperoxy oxabicyclo derivatives 176a–c (Table 11).
The mechanism of the solvolysis of 174 or 175 in the THF/H2O2 system involves the formation of solvolytically generated cyclobutyl hydroperoxides 177 followed by the rearrangement of the latter into oxa-bridged, hydroperoxyhemiketals 176 (Scheme 53).
Scheme 53: The mechanism of solvolysis of brosylates 174a–c and spiro cyclopropyl carbinols 175a–c in THF/H2O2.
Scheme 53: The mechanism of solvolysis of brosylates 174a–c and spiro cyclopropyl carbinols 175a–c in THF/H2O2....
The fragmentation of hydroperoxy acetals 178a–e in the presence of Ca(OCl)2 or t-BuOCl as the catalysts in CH3CN generating esters 179a–e proceeds through the Hock-like rearrangement mechanism (Table 12) [322].
Table 12: Fragmentation of hydroperoxy acetals 178a–e catalyzed by Ca(OCl)2 or t-BuOCl.
|
|
|||||||
| Substrate | Product | Ca(OCl)2 (equiv) | Time (min) | Yield, % | t-BuOCl (equiv) | Time (min) | Yield, % |
|---|---|---|---|---|---|---|---|
| 178a | 179a | 1.3 | 10 | 75 | 0.25 | 15 | 78 |
| 178b | 179b | 1.3 | 10 | 86 | 1.2 | 10 | 85 |
| 178c | 179c | 1.3 | 10 | 83 | 1.2 | 10 | 84 |
| 178d | 179d | 1.3 | 10 | 85 | 0.25 | 15 | 85 |
| 178e | 179e | 1.3 | 10 | 82 | 1.2 | 10 | 84 |
The fragmentation of hydroperoxy acetals 178 to esters 179 involves the formation and heterolytic fragmentation of intermediate secondary chloroperoxides 180. The possible mechanism of the process is presented in Scheme 54.
Scheme 54: The fragmentation mechanism of hydroperoxy acetals 178 to esters 179.
Scheme 54: The fragmentation mechanism of hydroperoxy acetals 178 to esters 179.
The acid-catalyzed rearrangement of phenylcyclopentyl hydroperoxide 181, involving the Hock reaction, is accompanied by the formation of a series of products: 1-phenylcyclopentene (182), phenol (170), cyclopentanone (183), and 5-acetoxyvalerophenone (184) (Scheme 55) [323].
Scheme 55: The acid-catalyzed rearrangement of phenylcyclopentyl hydroperoxide 181.
Scheme 55: The acid-catalyzed rearrangement of phenylcyclopentyl hydroperoxide 181.
An attempt was made [324] to synthesize hydroperoxides through the peroxidation of tertiary alcohols in the presence of a catalytic amount of acid. The treatment of 185 with H2O2 in the presence of a catalytic amount of H2SO4 for 72 hours did not lead to the formation of products via the Hock rearrangement of hydroperoxides, bicyclic hydroperoxides and о-hydroxyphenyl alkyl ketones. Instead, cyclic 2-methylchroman-2-yl hydroperoxide 188, geminal bishydroperoxides 190, and condensation products of peroxides such as 191 were isolated (Scheme 56).
Scheme 56: The peroxidation of tertiary alcohols in the presence of a catalytic amount of acid.
Scheme 56: The peroxidation of tertiary alcohols in the presence of a catalytic amount of acid.
The reaction mechanism presumably involves the following steps: the replacement of the hydroxy group by hydrogen peroxide to form tertiary hydroperoxides 186, the acid-catalyzed rearrangement of compounds 186 into cyclic phenoxycarbenium ions 187, and the addition of the second hydrogen peroxide molecule to 187 resulting in the formation of cyclic phenoxy hydroperoxide 188. The latter was isolated as the major product in the case of the six-membered ring (n = 1). In the case of the seven-membered ring (n = 2), geminal dihydroperoxide 190 and bridged bis(hydroxy)dialkyl peroxide 191 were obtained instead of 188. In case of the eight-membered ring (n = 3) an exclusive transformation into geminal dihydroperoxide 190 was observed (Table 13).
The formation of geminal dihydroperoxides 195 was also observed in the acid-catalyzed reaction of bicyclic secondary alcohols 192 with hydrogen peroxide. This reaction starts with the formation of bicyclic hydroperoxides 193 followed by the acid-catalyzed rearrangement with intermediate formation of peroxy hemiacetal 194. The latter is finally transformed into primary geminal bishydroperoxides 195 (Scheme 57) [325].
Scheme 57: The acid-catalyzed reaction of bicyclic secondary alcohols 192 with hydrogen peroxide.
Scheme 57: The acid-catalyzed reaction of bicyclic secondary alcohols 192 with hydrogen peroxide.
The photooxidation of 5,6-disubstituted 3,4-dihydro-2H-pyrans 196 generates the stable hydroperoxide 197 as the major product, which rearranges into dioxetane 198 at 28 °C in CCl4 within 13 h. Compounds 198 can be further transformed into keto esters 199 by treatment for 24 h with triphenylphosphine in CCl4 or concentrated HCl in CCl4. When compound 197 is heated at 70 °C its rearrangement into 199 occurs very rapidly and dioxetane 198 was not detected (Scheme 58) [326,327].
Scheme 58: The photooxidation of 5,6-disubstituted 3,4-dihydro-2H-pyrans 196.
Scheme 58: The photooxidation of 5,6-disubstituted 3,4-dihydro-2H-pyrans 196.
The oxidation of tertiary alcohols 200a–g, 203a,b, and 206, involving the rearrangement of hydroperoxides 201a–g, 204a,b, and 207, occurs in good yields in the presence of such systems as NaBO3·4H2O/BF3·Et2O [328], H2O2/BF3·Et2O, and H2O2/p-TsOH [329] (Scheme 59). The Hock rearrangement can be used to prepare alcohols 202a–g, 205, and 208 containing electron-donating substituents.
Scheme 59: The oxidation of tertiary alcohols 200a–g, 203a,b, and 206.
Scheme 59: The oxidation of tertiary alcohols 200a–g, 203a,b, and 206.
The intramolecular capture of the cationic intermediate derived from the Hock rearrangement of peroxyketone 209 provides a direct and efficient one-step synthesis of 2,3-disubstituted furans 210 (Scheme 60) [330].
Scheme 60: Transformation of functional peroxide 209 leading to 2,3-disubstitued furans 210 in one step.
Scheme 60: Transformation of functional peroxide 209 leading to 2,3-disubstitued furans 210 in one step.
The benzannulation of indoles 211 can be performed with γ-carbonyl tert-butyl peroxides 212 catalyzed by trifluoromethanesulfonic acid to give carbazoles 213. The key step of this approach is based on the acid-catalyzed rearrangement of tert-butyl peroxides (Scheme 61) [331].
Scheme 61: The synthesis of carbazoles 213 via peroxide rearrangement.
Scheme 61: The synthesis of carbazoles 213 via peroxide rearrangement.
The direct dehydrogenative construction of C–N bonds between unprotected phenols 215 and a series of 10H-phenoxazines and 10H-phenothiazines 214 with formation of 216 was carried out using a Hock-like activation with O2 followed by amine oxidation (Scheme 62) [332].
Scheme 62: The construction of C–N bonds using the Hock rearrangement.
Scheme 62: The construction of C–N bonds using the Hock rearrangement.
The Hock rearrangement plays an important role not only in fine organic synthesis but also in biological processes. Scheme 63 shows the proposed mechanism for the biosynthetic conversion of 217 to 218, which is an important component of the structural skeleton of the antitumor–antibiotic CC-1065 [333].
Scheme 63: The synthesis of moiety 218 from 217 which is a structural motif in the antitumor–antibiotic of CC-1065.
Scheme 63: The synthesis of moiety 218 from 217 which is a structural motif in the antitumor–antibiotic of CC-...
The synthetic model of the in vivo oxidation of cholesterol (219) by singlet oxygen produces cholesterol-5α-OOH 220, which is subjected to a Hock reaction to form the aldolization product 221 and keto aldehyde (atheronal A, 222) (Scheme 64) [67].
Scheme 64: The in vivo oxidation steps of cholesterol (219) by singlet oxygen.
Scheme 64: The in vivo oxidation steps of cholesterol (219) by singlet oxygen.
Keto aldehyde (atheronal A, 222) exhibits proatherogenic activity and plays a causal role in the development of cardiovascular diseases [66]. The proposed mechanism of the rearrangement of cholesterol-5α-OOH 220 is presented in Scheme 65.
Scheme 65: The proposed mechanism of the rearrangement of cholesterol-5α-OOH 220.
Scheme 65: The proposed mechanism of the rearrangement of cholesterol-5α-OOH 220.
Therefore, the acid-catalyzed Hock rearrangement of hydroperoxide 220 is a key step in the oxidation of cholesterol (219).
In a photochemical route developed for the synthesis of artemisinin the Hock rearrangement of hydroperoxide 223 selectively affords enol 224. This reactive intermediate 224 is then finally oxidized into artemisinin (Scheme 66) [334].
Scheme 66: Photochemical route to artemisinin via Hock rearrangement of 223.
Scheme 66: Photochemical route to artemisinin via Hock rearrangement of 223.
1.4 Kornblum−DeLaMare rearrangement
The Kornblum−DeLaMare rearrangement (KDLM) is a rearrangement of organic peroxides A containing a primary or secondary carbon atom into ketones B and alcohols C mainly under base-catalyzed reaction conditions (Scheme 67) [335].
Scheme 67: The Kornblum–DeLaMare rearrangement.
Scheme 67: The Kornblum–DeLaMare rearrangement.
In 1951, Kornblum and DeLaMare observed that the treatment of 1-phenylethyl tert-butyl peroxide (225) with KOH, NaOEt, or pyridine resulted in the decomposition of 225 to give acetophenone (227) and tert-butanol (228). A three-step mechanism for this reaction was proposed (Scheme 68) [336,337].
Scheme 68: Kornblum–DeLaMare transformation of 1-phenylethyl tert-butyl peroxide (225).
Scheme 68: Kornblum–DeLaMare transformation of 1-phenylethyl tert-butyl peroxide (225).
The reaction commences with a base-mediated α-proton abstraction from 225 to form carbanion 226 and the latter decomposes to yield the tert-butoxide anion and acetophenone (227). These steps occur presumably in a concerted manner. Finally, the protonation of the tert-butoxide anion results in the formation of tert-butanol (228). As alternative bases Et3N [338,339], phosphorus ylides [340] and LiOH [341,342] can be used and the Kornblum–DeLaMare rearrangement proceeds also on SiO2 [343].
The Kornblum–DeLaMare rearrangement is a convenient tool in organic chemistry for the conversion of monocyclic endoperoxides. These compounds are discussed in this review in the order of increasing ring size and the number of the starting substrates.
The treatment of unsubstituted bicyclic endoperoxides 229 by bases affords 4-hydroxyenones 230 [344] which are useful precursors in asymmetric organic syntheses. Alternative synthetic methods towards this class of compounds normally require a metal-catalyzed or biocatalyzed oxidation of diols 231 in an additional reaction step [345] (Scheme 69).
Scheme 69: The synthesis 4-hydroxyenones 230 from peroxide 229.
Scheme 69: The synthesis 4-hydroxyenones 230 from peroxide 229.
The treatment of endoperoxide 232 with triethylamine in ethanol at room temperature results in the O–O-bond cleavage to form 5-hydroxytropolone (233) (Scheme 70) [346].
Scheme 70: The Kornblum–DeLaMare rearrangement of peroxide 232.
Scheme 70: The Kornblum–DeLaMare rearrangement of peroxide 232.
It is interesting to note, that a reduction of the bicyclic endoperoxide 234 with thiourea in methanol at 10 °C produces similar to KDLM product tropolone 235 in 94% yield (Scheme 71) [347].
Scheme 71: The reduction of peroxide 234.
Scheme 71: The reduction of peroxide 234.
The Kornblum–DeLaMare reaction of the endoperoxide 236 with triethylamine in chloroform at −30 °C affords tropolone 237 in 97% yield (Scheme 72) [347].
Scheme 72: The Kornblum–DeLaMare rearrangement of endoperoxide 236.
Scheme 72: The Kornblum–DeLaMare rearrangement of endoperoxide 236.
Tropolones exhibit a broad spectrum of biological activities, including antibacterial, antiviral, antifungal, anti-allergic, anti-oxidant, and anti-inflammatory [348,349].
The treatment of endoperoxide 238 with Et3N gave 1,4-diketone 240 in quantitative yield instead of expected hydroxy ketone 239 (Scheme 73) [350-352].
Scheme 73: The rearrangement of peroxide 238 under Kornblum–DeLaMare conditions.
Scheme 73: The rearrangement of peroxide 238 under Kornblum–DeLaMare conditions.
The endoperoxide 238 is presumably converted into hemiketal 241, which is rearranged in several steps into diketone 240 (Scheme 74) [351].
Scheme 74: The proposed mechanism of rearrangement of peroxide 238.
Scheme 74: The proposed mechanism of rearrangement of peroxide 238.
The reaction of endoperoxide 242a containing an electron-donating substituent at the double bond with bases results in the rearrangement product diketone 243. Under the same conditions, the base-catalyzed rearrangement of endoperoxide 242b containing an electron-withdrawing substituent leads to a product mixture of hydroxy ketone 244, and diketones 245 and 246 (Scheme 75) [353].
Scheme 75: The Kornblum–DeLaMare rearrangement of peroxides 242a,b.
Scheme 75: The Kornblum–DeLaMare rearrangement of peroxides 242a,b.
A further study [352] on the base-catalyzed rearrangements of substituted bicyclic endoperoxides showed that the pathway of the rearrangement is largely determined by the position of the substituent. The rearrangement of endoperoxides 247a,b containing an electron-withdrawing substituent in the seven-membered ring occurs mainly via a retro-aldol cleavage giving rise to formyl benzoates 248a,b (Scheme 76).
Scheme 76: The base-catalyzed rearrangements of bicyclic endoperoxides having electron-withdrawing substituents.
Scheme 76: The base-catalyzed rearrangements of bicyclic endoperoxides having electron-withdrawing substituent...
On the other hand, endoperoxides 249a,b bearing electron-withdrawing groups (ester, acetyl) attached to the seven-membered ring are isomerized to diketones 250a,b (Scheme 77) [345].
Scheme 77: The base-catalyzed rearrangements of bicyclic endoperoxides 249a,b having electron-donating substituents.
Scheme 77: The base-catalyzed rearrangements of bicyclic endoperoxides 249a,b having electron-donating substit...
The Kornblum–DeLaMare reaction of endoperoxide 251a containing an electron-withdrawing substituent at the bridge head atom lead to the 1,2-dicarbonyl compound 252a whereas the ester 251b polymerized upon treatment with triethylamine (Scheme 78).
Scheme 78: The base-catalyzed rearrangements of bridge-head substituted bicyclic endoperoxides 251a,b.
Scheme 78: The base-catalyzed rearrangements of bridge-head substituted bicyclic endoperoxides 251a,b.
The disproportionation of endoperoxide 253 promoted by triethylamine affords β- and γ-hydroxy hydroperoxides 254 and 256. Under these conditions, the reaction afforded oxodiol 255 and diketone 257, which cyclized to hemiketal 258 as the products (Scheme 79) [354].
Scheme 79: The Kornblum–DeLaMare rearrangement of hydroperoxide 253.
Scheme 79: The Kornblum–DeLaMare rearrangement of hydroperoxide 253.
As the above reaction did not allow the isolation of hydroperoxide 254, an alternative strategy towards this compound was developed. The introduction of a protecting group into endoperoxide 253 using 2-methoxypropene gave protected peroxide 259. The subsequent triethylamine-catalyzed rearrangement of 259 leads to protected intermediate 260 the treatment of which under acidic conditions afforded hydroperoxide 254 in 70% yield (Scheme 80).
Scheme 80: Synthesis of β-hydroxy hydroperoxide 254 from endoperoxide 253.
Scheme 80: Synthesis of β-hydroxy hydroperoxide 254 from endoperoxide 253.
One approach to the enantioselective synthesis of 4-hydroxyenones 262 is based on the Kornblum–DeLaMare rearrangement of meso-endoperoxides 261 catalyzed by a chiral base [345] (Table 14).
Table 14: Enantioselective rearrangement of meso-endoperoxides 261a–f into 4-hydroxy enones 262a–f.
| No | Endoperoxide | Reaction conditionsa | No | Product | Yield,% | eе, % | |
|---|---|---|---|---|---|---|---|
| 261a |
|
R = H | 5 mol % cat., rt, 6 h | 262a |
|
97 | 99 |
| 261b | R,R = OC(Me)2O | 5 mol % cat., rt, 10 h | 262b | 99 | 99 | ||
| 261c |
|
R = H |
5 mol % cat.,
0 °C, 24 h |
262c |
|
99 | 87 |
| 261d | R = TBS | 5 mol % cat., rt, 12 h | 262d | 83 | 99 | ||
| 261e |
|
R = Bn | 10 mol % cat., rt, 24 h | 262e |
|
90 | 96 |
| 261f | R = -C(Me)2- | 10 mol % cat., rt, 36 h | 262f | 76 | 89 | ||
acat. =
The amine-catalyzed rearrangement of bicyclic endoperoxide 263 produced (S)-(+)-4-hydroxycyclohept-2-en-1-one (264), which was oxidized to bicyclic ketone 265. The synthetic value of chiral bicyclic ketone 265 was demonstrated by the transformation of this compound into (+)-sundiversifolide (266) (Scheme 81) [355].
Scheme 81: The amine-catalyzed rearrangement of bicyclic endoperoxide 263.
Scheme 81: The amine-catalyzed rearrangement of bicyclic endoperoxide 263.
The photooxidation of diene 267 followed by the base-catalyzed rearrangement of meso-endoperoxide 268 lead to (±)-trans,cis-4-hydroxy-5,6-di-O-isopropylidenecyclohex-2-en-1-one (269). The protection of the hydroxy group in compound 269 provides an efficient route to functionalized 4-hydroxy-2-cyclohexene-1-ones 270 (Scheme 82) [356].
Scheme 82: The base-catalyzed rearrangement of meso-endoperoxide 268 into 269.
Scheme 82: The base-catalyzed rearrangement of meso-endoperoxide 268 into 269.
The photooxidation of 271 in the presence of tetraphenylporphyrin produces endoperoxide 272, which undergoes a Kornblum–DeLaMare transformation when treated with triethylamine. The obtained product 4-hydroxycyclohexen-2-one 273 releases benzoic acid through β-elimination under the basic conditions to give cyclohexadienone 274 (Scheme 83) [357].
Scheme 83: The photooxidation of 271 and subsequent Kornblum–DeLaMare reaction.
Scheme 83: The photooxidation of 271 and subsequent Kornblum–DeLaMare reaction.
The base-catalyzed isomerization of bicyclic saturated fulvene endoperoxides 275 is employed as one approach to the preparation of 2-alkenylcyclopentanones 276 and cyclopentenones 277 [358]. Thus, the treatment of a solution of endoperoxides 275 in CH2Cl2 with triethylamine while increasing the temperature from 0 °C to room temperature affords hydroxyketone 276. The use of the stronger base DBU results in the formation of 2-vinyl-2-cyclopentenones 277 in high yield (Table 15).
In the case of acyclic enamine 278, the initial dioxetane product from the photochemical oxidation of 279 rearranged to amide 280. The reactions using cyclic enamines 281 involve the Kornblum–DeLaMare rearrangement of dioxetanes 282 into 1,2-diketones 283 (Scheme 84) [359,360].
Scheme 84: The Kornblum–DeLaMare rearrangement as one step in the oxidation reaction of enamines.
Scheme 84: The Kornblum–DeLaMare rearrangement as one step in the oxidation reaction of enamines.
The Kornblum–DeLaMare rearrangement of 1,2-dioxenes 284 [361], 1,2-dioxanes 286 [362], and tert-butyl peroxides 288 [330,363] produces 1,4-dicarbonyl compounds 285, 287, and 289, respectively (Scheme 85). These compounds are versatile starting substrates for the synthesis of various heterocyclic systems, such as furan, thiophene, and pyrrole derivatives.
Scheme 85: The Kornblum–DeLaMare rearrangement of 3,5-dihydro-1,2-dioxenes 284, 1,2-dioxanes 286, and tert-butyl peroxides 288.
Scheme 85: The Kornblum–DeLaMare rearrangement of 3,5-dihydro-1,2-dioxenes 284, 1,2-dioxanes 286, and tert-but...
The reaction of unsymmetrical epoxy dioxanes 290a–d with triethylamine is accompanied by the 1,2-dioxane-ring opening to form 4-hydroxy-2,3-epoxy ketones 291a–d in high yields. The base catalysis involves the abstraction of the most acidic α-proton in the vicinity of the O–O bond followed by the rearrangement accompanied by the O–O-bond cleavage to form 4-hydroxy-2,3-epoxy ketones (Scheme 86) [364].
Scheme 86: The Kornblum–DeLaMare rearrangement of epoxy dioxanes 290a–d.
Scheme 86: The Kornblum–DeLaMare rearrangement of epoxy dioxanes 290a–d.
The Kornblum–DeLaMare rearrangement is of special synthetic value in view of the synthesis of biologically active compounds. For instance, prostaglandin H2 (292) containing the bicyclic [2.2.1]endoperoxide moiety is rearranged in situ into prostaglandin Е2 (293) (Scheme 87) [365,366].
Scheme 87: Rearrangement of prostaglandin H2 292.
Scheme 87: Rearrangement of prostaglandin H2 292.
Nicolaou et al. [367] described the synthesis of epicoccin G (297) and related diketopiperazines 296 through the photooxidation of 294 and the Kornblum–DeLaMare rearrangement of peroxide 295 (Scheme 88).
Scheme 88: The synthesis of epicoccin G (297).
Scheme 88: The synthesis of epicoccin G (297).
The base-catalyzed transformation of organic peroxide 298 was used to synthesize compound 299, a precursor for the synthesis of the natural compound phomactin A (300). Phomactin A is a representative of a new class of platelet-activating factor (PAF) antagonists (Scheme 89) [368].
Scheme 89: The Kornblum–DeLaMare rearrangement used in the synthesis of phomactin A.
Scheme 89: The Kornblum–DeLaMare rearrangement used in the synthesis of phomactin A.
In another study [369], the transformation of peroxide 302, produced from 301, was applied to prepare compounds such as 3H-quinazolin-4-one 303, which is a core subunit of some important quinazolinone-based drugs (Scheme 90).
Scheme 90: The Kornblum–DeLaMare rearrangement in the synthesis of 3H-quinazolin-4-one 303.
Scheme 90: The Kornblum–DeLaMare rearrangement in the synthesis of 3H-quinazolin-4-one 303.
The Kornblum–DeLaMare rearrangement is one of the steps in the synthesis of the natural compound angelone from Nauclea, a plant species widely acclaimed for its anti-inflammatory and antibacterial utilities in traditional Chinese herbal medical formulations [370]. A Kornblum–DeLaMare enantiomeric resolution was also used to obtain both fragments of the polypropionate metabolite dolabriferol from a common precursor. The endoperoxide 304 was converted into ketone 305 with the help of the pseudo-enantiomeric quinine-derived catalyst (deMeQ-Ac) in toluene with moderate 47% yield. The peroxide 306 was transformed into ketone 307 with good 92% yield by using Et3N (Scheme 91) [371].
Scheme 91: The Kornblum–DeLaMare rearrangement in the synthesis of dolabriferol (308).
Scheme 91: The Kornblum–DeLaMare rearrangement in the synthesis of dolabriferol (308).
A sequence consisting of a template-mediated photooxygenation and an acid-catalyzed Kornblum−DeLaMare rearrangement of the intermediate endo-peroxides 310 was used in a one-pot transformation of 3-substituted 2-pyridones 309 into the respective 3-hydroxypyridine-2,6-diones 311 with good enantioselectivity (69–86% ee) (Scheme 92) [372].
Scheme 92: Sequential transformation of 3-substituted 2-pyridones 309 into 3-hydroxypyridine-2,6-diones 311 in one pot.
Scheme 92: Sequential transformation of 3-substituted 2-pyridones 309 into 3-hydroxypyridine-2,6-diones 311 in...
The Kornblum–DeLaMare rearrangement of peroxide 312 into hydroxy enone 313 with high yields and regioselectivity has been reported in the total synthesis of (+)-zeylenol and its congeners (Scheme 93) [373].
Scheme 93: The Kornblum–DeLaMare rearrangement of peroxide 312 into hydroxy enone 313.
Scheme 93: The Kornblum–DeLaMare rearrangement of peroxide 312 into hydroxy enone 313.
The polyfunctionalized carbonyl compounds 317 were prepared via crossover oxidative coupling of ethers 316 with electron-deficient alkenes 315 and vinylarenes 314 in the presence of Co(salen) and TBHP under mild conditions. The transformation involved the combination of a tandem radical reaction and a Kornblum−DeLaMare rearrangement in a one-pot process (Scheme 94) [374].
Scheme 94: The Kornblum–DeLaMare rearrangement in the synthesis of polyfunctionalized carbonyl compounds 317.
Scheme 94: The Kornblum–DeLaMare rearrangement in the synthesis of polyfunctionalized carbonyl compounds 317.
The readily available compounds styrenes 314, amines 318 and perfluoroalkyl iodides 319 were transformed into (Z)-β-perfluoroalkylenaminones 320 via a Co(acac)2/TBHP-promoted multicomponent radical reaction involving sequential fluoroalkylation and Kornblum-DeLaMare rearrangement (Scheme 95) [375].
Scheme 95: The Kornblum–DeLaMare rearrangement in the synthesis of (Z)-β-perfluoroalkylenaminones 320.
Scheme 95: The Kornblum–DeLaMare rearrangement in the synthesis of (Z)-β-perfluoroalkylenaminones 320.
Peroxy products resulted from the reaction of styrenes 314, ethyl diazoacetate (321), and TBHP underwent a Kornblum–DeLaMare rearrangement with formation of γ-ketoester 322 (Scheme 96) [376].
Scheme 96: The Kornblum–DeLaMare rearrangement in the synthesis of γ-ketoester 322.
Scheme 96: The Kornblum–DeLaMare rearrangement in the synthesis of γ-ketoester 322.
The Kornblum–DeLaMare rearrangement is a final step in the total synthesis of the diterpenoids amphilectolide (326) and sandresolide B (328) from a common furan building block 324, which was synthesized from 323. Amphilectolide was obtained through a photooxygenation of 325 in the presence of diisopropylethylamine (DIEA), followed by a one-pot reduction of the intermediate peroxide with sodium borohydride. Sandresolide B was prepared from 327 using tetraphenylporphyrin as a photosensitizer and DBU as a base in 51% yield over two steps (Scheme 97) [377].
Scheme 97: The Kornblum–DeLaMare rearrangement in the synthesis of diterpenoids 326 and 328.
Scheme 97: The Kornblum–DeLaMare rearrangement in the synthesis of diterpenoids 326 and 328.
The total synthesis of the natural products hainanolidol (331) and harringtonolide (332) includes a DBU-promoted Kornblum–DeLaMare rearrangement of endoperoxide 329 to ketone 330 (Scheme 98) [378].
Scheme 98: The synthesis of natural products hainanolidol (331) and harringtonolide (332) from peroxide 329.
Scheme 98: The synthesis of natural products hainanolidol (331) and harringtonolide (332) from peroxide 329.
The reaction of the sodium salts of 1,3-dicarbonyl compounds 333, 334 with endoperoxides 263 and 261a in the presence of an organocatalyst affords the trans-fused butyrolactones 339 and 340 in high yield. The reaction proceeds via the formation of bicycles 335, 336 in the case of method A and 337, 338 in the case of method B (Scheme 99) [379].
Scheme 99: The synthesis of trans-fused butyrolactones 339 and 340.
Scheme 99: The synthesis of trans-fused butyrolactones 339 and 340.
The leucosceptroid A (341) produced leucosceptroid C (343) and its diastereomer in 78% yield (1:1 dr) under the base-induced reduction of the initial endoperoxide intermediate. Irradiation of a solution of leucosceptroid A (341) in an oxygen-saturated dichloromethane solution containing a catalytic amount of tetraphenylporphyrin (TPP) and N,N-diisopropylethylamine cleanly produced 344 (85% yield). The latter compound represents the base-promoted Kornblum–DeLaMare rearrangement product of endoperoxide 342 (Scheme 100) [380].
Scheme 100: The synthesis of leucosceptroid C (343) and leucosceptroid P (344) via the Kornblum–DeLaMare rearrangement.
Scheme 100: The synthesis of leucosceptroid C (343) and leucosceptroid P (344) via the Kornblum–DeLaMare rearra...
It is worth mentioning that the synthesis of 4-hydroxycyclopentenone 343 and litsaverticillols was achieved in a similar way in other works [381-384].
1.5 Dakin oxidation of arylaldehydes or acetophenones
Generally, the Dakin oxidation is a reaction, in which o- or p-hydroxylated benzaldehydes or acetophenones 345 react with hydrogen peroxide in the presence of a base to form o- or p-dihydroxybenzene 346 and carboxylate 347 (Scheme 101) [385,386].
Scheme 101: The Dakin oxidation of arylaldehydes or acetophenones.
Scheme 101: The Dakin oxidation of arylaldehydes or acetophenones.
Actually, the Dakin oxidation is a special case of the Baeyer–Villiger oxidation. Mechanistically, the Dakin oxidation starts with the nucleophilic addition of a hydroperoxide anion to the carbonyl carbon atom of benzaldehyde (348) to form intermediate 349 followed by its rearrangement to phenyl ester 350. The subsequent nucleophilic addition of a hydroxide anion to the carbonyl group of phenyl ester 350 yields intermediate 351, which undergoes a rearrangement accompanied by the elimination of phenoxide anion 352 and carboxylic acid 353. Then, the phenoxide anion 352 deprotonates the carboxylic acid 353 to produce p-dihydroxybenzene (354) and the corresponding carboxylate anion 355 (Scheme 102) [385,387].
Scheme 102: The mechanism of the Dakin oxidation.
Scheme 102: The mechanism of the Dakin oxidation.
The nucleophilic addition of the hydroperoxide to the carbon atom of a carbonyl group and the [1,2]-aryl migration are the two rate-determining reaction steps in the Dakin oxidation process [387]. The total rate of the Dakin oxidation depends on the nucleophilicity of the hydroperoxide, the electrophilicity of the carbonyl carbon atom, the nature of alkyl substituents in the proximity of the carbonyl group, the existence of other functional groups in the aromatic ring, and the alkalinity of the reaction mixture. Generally, hydroxybenzaldehydes are more reactive in the Dakin oxidation than hydroxyacetophenones. This is due to the fact, that the carbonyl carbon atom of ketones is less electrophilic than the carbonyl carbon atom of an aldehyde. Under weakly basic conditions, о-hydroxybenzaldehydes and о-hydroxyacetophenones are oxidized more rapidly than p-hydroxybenzaldehydes and p-hydroxyacetophenones, whereas m-hydroxybenzaldehydes and m-hydroxyacetophenones are unreactive [387]. Electron-donating substituents in the ortho and para positions of the aromatic ring enhance the electron density on the migrating carbon atom thus promoting the [1,2]-aryl migration and accelerating the oxidation. Electron-donating substituents in the meta position have little effect on the electron density on the migrating carbon atom. Electron-withdrawing substituents in the ortho and para positions of the aromatic ring reduce the electron density on the migrating carbon atom, interfering with the [1,2]-aryl migration. The hydroperoxide anion is a more reactive nucleophile than neutral hydrogen peroxide. The reaction rate of the oxidation of hydroxyphenylaldehydes or ketones increases with increasing pH value, however, at pH higher than 13.5 the oxidation does not take place [387].
The efficient oxidation of hydroxylated aldehydes and ketones to hydroquinones and catechols was performed using a complex of urea with hydrogen peroxide as an oxidant [388]. The main advantage of this method is, that the reaction is performed under solvent-free conditions and provides the products in high yields.
A solvent-free Dakin reaction of aromatic aldehydes 356 with m-CPBA resulted in corresponding phenols 357 with high yields within a few minutes (Scheme 103) [389].
Scheme 103: A solvent-free Dakin reaction of aromatic aldehydes 356.
Scheme 103: A solvent-free Dakin reaction of aromatic aldehydes 356.
The phenols 359 were prepared from electron-rich arylaldehydes 358 by a flavin-catalyzed Dakin oxidation under the action of H2O2 and sodium bicarbonate with high yields (Scheme 104) [390].
Scheme 104: The organocatalytic Dakin oxidation of electron-rich arylaldehydes 358.
Scheme 104: The organocatalytic Dakin oxidation of electron-rich arylaldehydes 358.
The flavin-catalyzed Dakin oxidation provides a more selective formation of phenols in comparison with the base-catalyzed rearrangement. The Dakin oxidation of arylaldehydes 361 is performed in the presence of molecular oxygen as the oxidant, a flavin organocatalyst and a Hantzsch ester. The oxidation products, catechols and electron-rich phenols 362, were prepared with 0.1–10 mol % of catalyst, 1 equiv of the Hantzsch ester, and O2 or air in a stoichiometric amount (Scheme 105) [391].
Scheme 105: The Dakin oxidation of electron-rich arylaldehydes 361.
Scheme 105: The Dakin oxidation of electron-rich arylaldehydes 361.
Dakin reactions of benzaldehydes 358 with H2O2 were successfully performed in natural feedstock extract ‘Water Extract of Banana’ (WEB) at room temperature under aerobic conditions in short reaction times. Under these conditions, phenols 359 could be obtained with 90–98% yields (Scheme 106) [392]. The WEB was prepared by extraction of banana ash with distilled water. The authors suggested that the potassium carbonate and sodium carbonate present in the extract serve as the internal base to promote the Dakin oxidation.
Scheme 106: The Dakin oxidation of arylaldehydes 358 in water extract of banana (WEB).
Scheme 106: The Dakin oxidation of arylaldehydes 358 in water extract of banana (WEB).
The Dakin oxidation was applied for the synthesis of indolo[2,1-b]quinazolines 364 from indole-3-carbaldehydes 363. In the first step, the oxidation of indole-3-carbaldehydes 363 with further cyclization leads to isatoic anhydrides 365. Then, the anhydrides 365 react with indole-3-carbaldehydes 363 to produce the target indolo[2,1-b]quinazolines 364 (Scheme 107) [393].
Scheme 107: A one-pot approach towards indolo[2,1-b]quinazolines 364 from indole-3-carbaldehydes 363 through the Dakin oxidation.
Scheme 107: A one-pot approach towards indolo[2,1-b]quinazolines 364 from indole-3-carbaldehydes 363 through th...
The Dakin oxidation is widely used for the synthesis of benzenediols and alkoxyphenols. For example, catechol generated from o-alkoxybenzaldehydes is employed as the starting reagent in the synthesis of catecholamine derivatives [394]. Catechols, for example are substrates in the manufacture of synthetic adhesives and coatings. Their multifaceted reactions with both, organic and inorganic reagents, make catechols widely applied compounds for surface modifications [395].
Vanillin was oxidized under Dakin conditions under formation of 2-methoxyhydroquinone with 97% yield. This vanillin-derivative was used as a building block in the synthesis of bio-based compounds applicable in polymer field [396].
The Dakin oxidation of mixtures of lignin depolymerization products is an important process for increasing the number of hydroxy groups in arene cycles. Then, these byproducts are glycidylated with mixtures of epoxy monomers. The obtained products are interesting compounds for the synthesis of bio-based epoxy thermosets with outstanding thermomechanical indexes [397].
Acid-catalyzed Dakin oxidation: The mechanism of Dakin oxidation under mild acidic conditions is similar to the base-catalyzed mechanism. A 30–35% aqueous H2O2/acid system can be employed as the oxidizing agent to synthesize phenols 367a–c from benzaldehydes 366a–c. The oxidation of 366a using traditional peracids produces a mixture of aryl formate 368 and epoxides 369 and 370 (Scheme 108) and cannot be applied to substrates containing peracid-labile functional groups [398].
Scheme 108: The synthesis of phenols 367a–c from benzaldehydes 366a-c via acid-catalyzed Dakin oxidation.
Scheme 108: The synthesis of phenols 367a–c from benzaldehydes 366a-c via acid-catalyzed Dakin oxidation.
The addition of boric acid to the H2O2/acid system leads to an increase in the yield of phenols 372a–f even in the case of benzaldehydes 371a–c or acetophenones 371d–f containing electron-donating groups in the meta position or electron-withdrawing groups in the ortho or para positions with respect to the carbonyl group (Table 16) [399].
Table 16: Acid-catalyzed Dakin oxidation of benzaldehydes 371a–c and acetophenones 371d–f by H2O2/H3BO3 in THF.
|
|
|||
| Compound | Carbonyl compound | Reaction time, h | Yield, % 372a–f |
|---|---|---|---|
| 371a | R1 = R2 = R3 = R4 = H | 12 | 74 |
| 371b | R1 = OH, R2 = R3 = R4 = H | 7 | 80 |
| 371c | R1 = R2 = R4 = H, R3 = OH | 24 | 90 |
| 371d | R1 = OH, R2 = R3 = H, R4 = Me | 36 | 90 |
| 371e | R1 = R2 = R3 = H, R4 = Me | 24 | 63 |
| 371f | R1 = R2 = H, R3 = NO2, R4 = Me | 48 | 60 |
Presumably, the coordination of the H2O2–aldehyde adduct 373 by the highly polarized boric acid is responsible for the increased yields of phenols 372. The adduct 373 easily eliminates a borate ion with concerted migration of the aryl group giving phenols 372. The migrating rate of the aryl group is higher in comparison with hydride migration and formation of 374 (Scheme 109).
Scheme 109: Possible transformation paths of the highly polarized boric acid coordinated H2O2–aldehyde adduct 373.
Scheme 109: Possible transformation paths of the highly polarized boric acid coordinated H2O2–aldehyde adduct 3...
1.6 Elbs persulfate oxidation of phenols
The Elbs oxidation is the oxidation of phenols 375 with potassium persulfate in the presence of alkali hydroxides to form p-hydroquinones 376 (Scheme 110) [400,401].
Scheme 110: The Elbs oxidation of phenols 375 to hydroquinones.
Scheme 110: The Elbs oxidation of phenols 375 to hydroquinones.
The Elbs oxidation is a multistep process, which commences by the formation of the phenolate anion 377. This is followed by the nucleophilic substitution of peroxide oxygen in the peroxydisulfate ion 378 [402] and the resulting sulfoxy group positioned in the para position (compound 379) is hydrolyzed the with formation of p-hydroquinone 376 (Scheme 111).
Scheme 111: The mechanism of the Elbs persulfate oxidation of phenols 375 affording p-hydroquinones 376.
Scheme 111: The mechanism of the Elbs persulfate oxidation of phenols 375 affording p-hydroquinones 376.
The oxidation of phenols containing electron-donating substituents to dihydroxybenzenes gives products in higher yields compared with phenols containing electron-withdrawing substituents (Table 17) [403-405].
The main drawback of the persulfate-mediated Elbs oxidation of phenols, are the normally observed moderate conversions and yields. Remarkably, under the above Elbs oxidation conditions 5-hydroxy-2-pyridones 381 were prepared from pyridines 380 with good yields (Scheme 112) [406].
Scheme 112: Oxidation of 2-pyridones 380 under Elbs persulfate oxidation conditions.
Scheme 112: Oxidation of 2-pyridones 380 under Elbs persulfate oxidation conditions.
Later, the synthesis of 3-hydroxy-4-pyridone (384) via the Elbs oxidation of 4-pyridone (382) and isolation of 4-pyridone-3-sulfate (383) was described (Scheme 113) [407]. The synthesis of 5-hydroxy-6-bromo-2-pyridone was described under similar conditions [408].
Scheme 113: Synthesis of 3-hydroxy-4-pyridone (384) via an Elbs oxidation of 4-pyridone (382).
Scheme 113: Synthesis of 3-hydroxy-4-pyridone (384) via an Elbs oxidation of 4-pyridone (382).
1.7 Schenck and Smith rearrangements
In 1958, Schenck observed that the storage of 5α-hydroperoxide 385 in chloroform for 3 days results in the shift of the OOH group from the 5α to 7α position and a double-bond migration with formation of 386. This reaction is nowadays known as the Schenck rearrangement (Scheme 114) [409-411].
In 1973, Smith discovered another type of rearrangement of allylic hydroperoxides [412]. The 7α-hydroperoxide 386 underwent a 20–30% isomerization to the 7β-hydroperoxide 387 if a solution of 386 in ethyl acetate was kept at 40 °C for 48 h (Scheme 115). This process is called the Smith rearrangement.
The mechanisms of these, at first glance simple, reactions were systematically investigated 40 years after their discovery.
Three main pathways for the Schenck rearrangement have been proposed (Scheme 116). Path A involves the cyclization resulting in the formation of a carbon-centered radical. Path B comprises the formation of a transition state with the electron density distributed over a cyclic system. Path C proceeds through a dissociation to form an allylic radical and triplet oxygen (Scheme 116) [186,413].
Scheme 116: Three main pathways of the Schenck rearrangement.
Scheme 116: Three main pathways of the Schenck rearrangement.
Path A, the initially considered most favorable pathway, was excluded because the isomerization of hydroperoxides 388 and 389 following this route would lead to a β-scission ring opening of 390 (Scheme 117).
Scheme 117: The isomerization of hydroperoxides 388 and 389.
Scheme 117: The isomerization of hydroperoxides 388 and 389.
However, this process was not observed and none of the possible carbon-centered radicals 390 was trapped by molecular oxygen [414]. Meanwhile, it is known that the dioxacyclopentyl radical 392 formed from 391 is trapped by oxygen to form hydroperoxide 393 (Scheme 118) [415].
Scheme 118: Trapping of dioxacyclopentyl radical 392 by oxygen.
Scheme 118: Trapping of dioxacyclopentyl radical 392 by oxygen.
It was hypothesized that the Schenck rearrangement of peroxide 394 proceeds through a cyclic structure 395 according to the pathway shown in Scheme 119 [414].
Scheme 119: The hypothetical mechanism of the Schenck rearrangement of peroxide 394.
Scheme 119: The hypothetical mechanism of the Schenck rearrangement of peroxide 394.
However, this hypothesis was also rejected because the ESR spectra recorded after the photolysis of 5α- and 7α-hydroperoxides 385 and 386 showed that the tertiary allylperoxyl radical and secondary allylperoxyl radical are separate and distinct species, and that they do not have the common cyclic structure 395 [416].
In a study using labeled isotope 18O2 it was found that the two hydroperoxides 398 and 399 derived from autoxidation of oleic acid (397) underwent the Schenck rearrangement without incorporating dioxygen from the atmosphere (Scheme 120) [417,418]. Later on, Beckwith and Davies confirmed this fact for cholesterol hydroperoxide [416] and the hydroperoxide generated from valencene [419].
Scheme 120: The autoxidation of oleic acid (397) with the use of labeled isotope 18O2.
Scheme 120: The autoxidation of oleic acid (397) with the use of labeled isotope 18O2.
Based on these results, no formation of triplet oxygen occurs in the reaction, thus excluding path C in Scheme 116. Instead, a cyclic transition state (path B, Scheme 116) became more likely, which was confirmed by the stereoselective rearrangement of optically pure olefinic hydroperoxides [420].
However, the study on the rearrangement of hydroperoxides 398, 399 obtained from oleic acid (397) using stereochemical, oxygen-isotopic labeling and solvent viscosity analyses demonstrated that, in hexane, a small amount of atmospheric oxygen is incorporated into the product. The replacement of the solvent by more viscous dodecane and then by octadecane led to a decreased content of atmospheric oxygen in the final product [421,422]. These results provided evidence that the Schenck rearrangement proceeds also through path C in Scheme 116.
Besides, path C was also confirmed by the rearrangement of 18O-labeled hydroperoxide 400 under an atmosphere of 16O2 with formation of isotopomers 401–403 (Scheme 121) [423].
Scheme 121: The rearrangement of 18O-labeled hydroperoxide 400 under an atmosphere of 16O2.
Scheme 121: The rearrangement of 18O-labeled hydroperoxide 400 under an atmosphere of 16O2.
Examples of the Schenck rearrangement are given in Table 18.
Table 18: Examples of the Schenck rearrangement.
| Entry | Allylic isomer A | Allylic isomer B | Ref. |
|---|---|---|---|
| 1 |
404a |
404b |
[424] |
| At 40 °C in non-polar solvents, an approximately equimolar mixture of A and B is formed | |||
| 2 |
405a |
405b |
[414] |
| In hexane, A is rearranged to an equilibrium mixture of ~80% A and ~20% B | |||
| 3 |
406a |
406b |
[425] |
| At 60–70 °C in C6H6 or MeCN in the presence of TBHN or AIBN within 16–22 h, a 50:50 A:B mixture is formed | |||
| 4 |
407a |
407b |
[426] |
| In CCl4 at 40 °C for 141 h, the rearrangement proceeds by 80% | |||
| 5 |
408a |
408b |
[427] |
| In CDCl3, the rearrangement of A into B is completed in 24 h | |||
| 6 |
409a |
409b |
[428] |
| In CDCl3, the rearrangement is completed in 72 h | |||
| 7 |
410a |
410b |
[429] |
| In C6H6 in presence of 10 equiv TBHP and 20 mol % DTBN at 40 °C for 16 h, isomers A and B are formed in equal amounts | |||
| 8 |
411a |
411b |
[430] |
| In CDCl3 the rearrangement is completed in 48 h | |||
| 9 |
412a |
412b |
[431] |
| In CHCl3 for 5 d at room temperature, only partial conversion | |||
| 10 |
413a |
413b |
[432] |
| In CDCl3 the rearrangement is completed after 3–4 weeks; R: CO2H, CO2Me, CH2OH, CH3 | |||
| 11 |
414a |
414b |
|
| In CDCl3 the rearrangement is completed after 2–4 weeks; R: CO2H, CO2Me, CH2OH, CH3 | |||
| 12 |
415a |
415b |
[433] |
| In CDCl3 the rearrangement is completed after 2 d | |||
| 13 |
416a |
416b |
[434] |
| In pyridine for 24 h, R: OH,CH3COO, F, Cl, conversion 12–58% | |||
| 14 |
417a |
417b |
[435] |
| In a 5 M solution of LiClO4 in Et2O the rearrangement is completed in 24 h | |||
| 15 |
418a |
418b |
[436] |
| In CDCl3/D2O, lyophilized PBS buffer at pH 7 for 20 h, the conversion is 14% | |||
| 16 |
419a |
419b |
[136] |
| In CH2Cl2 at −78 °C with BF3·OEt2 (1 mol %) | |||
| 17 |
420a |
420b |
[437] |
| In MeCN/H2O, only partial conversion. | |||
The Schenck rearrangement takes also place with allylic hydroperoxides derived from lipids. The rearrangement of the oleate-derived allylic hydroperoxides (S)-421, and (R)-425 involved free radicals includes the oxygen-centered radicals 422, 423, 426, 427. The E-oleate hydroperoxide (S)-421 transforms into the corresponding (R)-E-product 424 at room temperature with a high (S) → (R) stereoselectivity of more than 97%. A decreased selectivity (~90%) was observed for product 428 obtained from the Z-hydroperoxide (R)-425. In this case, the configurational direction of the reaction was (R) → (R) (Scheme 122) [438].
Scheme 122: The rearrangement of the oleate-derived allylic hydroperoxides (S)-421 and (R)-425.
Scheme 122: The rearrangement of the oleate-derived allylic hydroperoxides (S)-421 and (R)-425.
The Smith rearrangement is a free-radical chain reaction in which atmospheric oxygen may play a greater role than in the Schenck rearrangement. Apparently, the Smith rearrangement proceeds through a dissociation to the allylic radical and 3O2. Presumably, the distance between these active species is large enough to allow an exchange with atmospheric oxygen (Scheme 123). The Schenck and Smith rearrangements are both a consequence of the reversibility of the reaction of allyl radicals with triplet dioxygen and differ mechanistically in the degree of separation of these two components [186]. There are only a few examples of the Smith rearrangement known and some of them are collected in Table 19.
Scheme 123: Mechanisms of Schenck and Smith rearrangements.
Scheme 123: Mechanisms of Schenck and Smith rearrangements.
Table 19: Examples of the Smith rearrangement.
| Entry | Allylic isomer B | Allylic isomer C | Comments | Ref. |
|---|---|---|---|---|
| 1 |
429a |
429b |
In CDCl3 within 259 h, approximately 5% of B was transformed into C | [427] |
| 2 |
430a |
430b |
In CHCl3 at room temperature within 150 h, the B:C ratio reached 1.8:1 | [439] |
| 3 |
431a |
431b |
In CDCl3 at 40 °C within 3.5 h, B is transformed into C by 20%. In EtOAc at 40 °C, the yield of C was 25–30% | [416] |
| In CHCl3 after 5 d at room temperature, only partial conversion | [431] | |||
| 4 |
432a |
432b |
In CDCl3, the B:C ratio reached 1:1.5 | [439] |
In diene or triene-containing systems (433), both the rearrangement and cyclization of allylic peroxyl radicals can take place with formation of 434–436 (Scheme 124) [440].
Scheme 124: The rearrangement and cyclization of 433.
Scheme 124: The rearrangement and cyclization of 433.
1.8 Wieland rearrangement
In 1911 Wieland performed the decomposition of bis(triphenylmethyl)peroxide (437) under an atmosphere of CO2 in boiling xylene for 10 min and obtained the crystalline product 438 in 70% yield (Scheme 125) [441].
The mechanism of the Wieland rearrangement involves the following three steps: Initial formation of O-centered radical A, the rearrangement of radical A into diphenylphenoxymethyl radical C, and the dimerization of radical C [442,443].
Radical 1,2-aryl migrations from silicon or germanium to oxygen is similar to the Wieland rearrangement. The thermal decomposition of either bis(triphenylsilyl) 439 or bis(triphenylgermyl) 441 peroxides leads to the rearranged products 440, 442 in high yields (Scheme 126) [444,445].
Scheme 126: The rearrangement of bis(triphenylsilyl) 439 or bis(triphenylgermyl) 441 peroxides.
Scheme 126: The rearrangement of bis(triphenylsilyl) 439 or bis(triphenylgermyl) 441 peroxides.
2 Unnamed rearrangements of organic peroxides and related processes
2.1 Protic acid-catalyzed rearrangements of organic peroxides and related processes
The oxidative transformation of cyclic ketones 58d and 443a–d in the reaction with hydrogen peroxide in alcohols in the presence of sulfuric acid proceeds through the formation of geminal dihydroperoxides 444a–e. The latter compounds are oxidized to dicarboxylic acids 445a–e followed by their transformation into the corresponding dicarboxylates 446a–e, rather than formation of lactones via the Baeyer–Villiger reaction (Scheme 127) [446].
Scheme 127: The oxidative transformation of cyclic ketones.
Scheme 127: The oxidative transformation of cyclic ketones.
This transformation requires the following key conditions to proceed: a reaction temperature higher than 80 °C, the H2SO4 concentration in the range of 0.2–1.0 mol/L, and a molar ratio of hydrogen peroxide/ketone in the range of 5–10. The corresponding dibutyl esters were prepared in 53−70% yields by oxidation in butanol, which keeps the temperature in the range of 98−106 °C (Table 20).
In a study on the hydroxylation of compounds containing a double bond to the corresponding α-glycols, the tungstic acid-catalyzed reaction of cyclohexene (447) with 90% hydrogen peroxide in methanol, ethanol, or isopropanol afforded the corresponding 2-alkoxycyclohexanols 448a–c in 70, 41, and 21% yields, respectively, as well as the trans-1,2-cyclohexanediols 449a–d (Scheme 128) [447].
Scheme 128: The hydroxylation of cyclohexene (447) in the presence of tungstic acid.
Scheme 128: The hydroxylation of cyclohexene (447) in the presence of tungstic acid.
A detailed study on the hydroxylation of cyclohexene (447) in tert-butanol using 30% hydrogen peroxide showed that in this reaction the formation and rearrangements of 2-hydroperoxyalkanols 451 is involved. The treatment of 2-hydroperoxycyclohexanol (451) with acetone afforded the cyclic peroxide 452. The acid-catalyzed rearrangement of the peroxide 452 gave dialdehyde 453, which further transformed into aldehyde 454. The isolation and characterization of the latter compound was crucial to an understanding of the oxidation of olefins to aldehydes under the action of hydrogen peroxide (Scheme 129).
Scheme 129: The oxidation of cyclohexene (447) under the action of hydrogen peroxide.
Scheme 129: The oxidation of cyclohexene (447) under the action of hydrogen peroxide.
The study of the reactions of various unsaturated molecules with hydrogen peroxide demonstrated that the reaction of butenylacetylacetone 455 with H2O2 at pH 5–6 at 38–40 °C produces 2-methyl-3-hexenoic acid (457). Other possible products 456 resulting from a double-bond oxidation reaction were not observed. Apparently, the formation of carbanion A is the driving force of this reaction. Carbanion A transforms into the symmetrical dihydroxyperoxide B, which subsequently rearranges through a deacetoxylation to finally afford 2-methyl-3-hexenoic acid (457) (Scheme 130) [448].
Scheme 130: The reaction of butenylacetylacetone 455 with hydrogen peroxide.
Scheme 130: The reaction of butenylacetylacetone 455 with hydrogen peroxide.
The oxidation of bridged 1,2,4,5-tetraoxanes 458 upon heating in an acidic medium in the presence of H2O2 is leading to esters 459 (Scheme 131) [449].
Scheme 131: The oxidation of bridged 1,2,4,5-tetraoxanes.
Scheme 131: The oxidation of bridged 1,2,4,5-tetraoxanes.
It is assumed that the reaction of tetraoxanes 458a–f proceeds as an acid-catalyzed oxidative transformation, similar to the Baeyer–Villiger and Hock rearrangements, to yield intermediate A. This is further transformed into esters 459a–f through the oxidation of the CH group and esterification (Scheme 132).
Scheme 132: The proposed mechanism for the oxidation of bridged 1,2,4,5-tetraoxanes.
Scheme 132: The proposed mechanism for the oxidation of bridged 1,2,4,5-tetraoxanes.
In another study [450], the rearrangement of isomeric ozonides was described. Here, the ozonides 460a,b were interconverted and rearranged into the tricyclic monoperoxide 461 under the action of phosphomolybdic acid (PMA). This result is attributable to the protic acid nature of PMA as well as its ability to form peroxo compounds containing M–O–O groups that influence the direction of the reaction (Scheme 133).
Scheme 133: The rearrangement of ozonides.
Scheme 133: The rearrangement of ozonides.
The observed interconversion of ozonides may be useful for the interpretation of the data on the ozonolysis of unsymmetrical unsaturated compounds.
Carboxylic acids 464 were prepared through a camphorsulfonic acid-catalyzed oxidative rearrangement of a 1,2-dioxolane intermediate 463 prepared from malondialdehydes 462 and H2O2 (Scheme 134) [451].
Scheme 134: The acid-catalyzed oxidative rearrangement of malondialdehydes 462 under the action of H2O2.
Scheme 134: The acid-catalyzed oxidative rearrangement of malondialdehydes 462 under the action of H2O2.
2.2 Lewis acid-catalyzed cleavage of peroxides
The Lewis acid-catalyzed cleavage of peroxides follows mainly two pathways: the O–O-bond heterolysis to form an oxycarbenium ion 467 accompanied by the migration of the adjacent substituent, and the acid-catalyzed ionization of the C–O bond to yield carbenium ion 468. The reaction pathway is mainly determined by the nature of the starting compound and the C–O ionization pathway is promoted by the stabilization of the final carbocation, whereas the O–O-bond heterolysis is facilitated by a high migratory ability of the adjacent groups. The fragmentation of dialkyl peroxides 465 and ozonides 466 mainly depends on the nature of the applied Lewis acid. In this way, SnCl4 and BF3·Et2O facilitate the O–O-bond heterolysis (A), whereas TiCl4 promotes the C–O ionization (SN1 mechanism) in tertiary peroxides (B). The formation of ketones 469, 471 and ester 470 is the result of the Lewis acid-catalyzed decomposition of ozonides through the ionization of peroxide, ionization of alkoxide, or oxygen–oxygen heterolysis (C) (Scheme 135) [452].
Scheme 135: Pathways of the Lewis acid-catalyzed cleavage of dialkyl peroxides 465 and ozonides 466.
Scheme 135: Pathways of the Lewis acid-catalyzed cleavage of dialkyl peroxides 465 and ozonides 466.
The TiCl4-promoted rearrangement of (tert-butyldioxy)cyclohexanedienones 472a–d, which are generated by the ruthenium-catalyzed oxidation of phenols with tert-butyl hydroperoxide, provides an efficient route to 2-substituted quinones 473a–d (Table 21) [453,454]. The mechanism of this transformation is depicted in Scheme 136.
Scheme 136: The mechanism of the transformation of (tert-butyldioxy)cyclohexanedienones 472.
Scheme 136: The mechanism of the transformation of (tert-butyldioxy)cyclohexanedienones 472.
In the first step, the coordination of dienone 472 to the Lewis acid gives rise to cation 474. The second step involves a 1,2-alkyl migration to form cation 475. The subsequent deprotonation of the latter affords aromatic intermediate 476. In the final step, trichloro-tert-butoxytitanium is eliminated from intermediate 476 to produce 2-alkylquinones 473.
The transformation of 4-methyl-4-tert-butyldioxycyclohexadienone 472a into 2-methylbenzoquinone (473a) can be used also for the regioselective synthesis of vitamin K3 477 (Scheme 137) [455,456].
Scheme 137: The synthesis of Vitamin K3 from 472a.
Scheme 137: The synthesis of Vitamin K3 from 472a.
The use of SnCl4 or TMSOTf as the catalyst made it possible to prepare trimethylsilyl-substituted cyclic peroxides 479a–d and 480a,b in a cis configuration starting from allyltrimethylsilane and bicyclic [2.2.n]endoperoxides 478a–d (Table 22) [457].
Table 22: Conditions of the synthesis of trimethylsilyl-substituted cyclic peroxides (1,2-dioxanes) 479a–d and 480a,b.
|
|
||||
| Endoperoxide | Equivalents of TMSOTf (or SnCl4) | Reaction time, min | Product/ratio of diastereomers | Total yield, % |
|---|---|---|---|---|
| 478a | 0.033 | 15 | 479a/480a, 1:0 | 54 |
| 478a | 1.0 SnCl4 | 30 | 479a/480a, 1:1 | 53 |
| 478b | 1.1 | 15 | 479b/480b, 1:0.8 | 60 |
| 478c | 1.1 | 40 | 479c, 1:0 | 10 |
| 478d | 1.1 | 40 | 479d, 1:0 | 48 |
The mechanism of this reaction implies that TMSOTf or SnCl4 promote the heterolytic cleavage of the C–O bond in 478d to form carbocation 481d, which is then attacked by allyltrimethylsilane through a chair-like transition state 482d. The subsequent cyclization of 482d through the stabilized carbocation 483d affords silyl-substituted peroxide, 1,2-dioxane 479d, containing the substituent (–CH2SiMe3) in the equatorial position (Scheme 138).
Scheme 138: Proposed mechanism for the transformation of 478d into silylated endoperoxide 479d.
Scheme 138: Proposed mechanism for the transformation of 478d into silylated endoperoxide 479d.
The employment of BF3·Et2O as the catalyst for the rearrangement of hydroperoxide 485, which is generated by the oxidation of steroid 484, enables the opening of the D ring between C-14 and C-16 to form diketone 486 (Scheme 139) [458].
Scheme 139: The rearrangement of hydroperoxide 485 to form diketone 486.
Scheme 139: The rearrangement of hydroperoxide 485 to form diketone 486.
2.3 Rearrangements and related processes of organic peroxides in the presence of bases
The base-catalyzed rearrangement of cyclic peroxides 488a–g, which are prepared by the manganese-catalyzed oxidation of 1- and 1,2-disubstituted cyclopropanols 487a–g, provides a convenient approach to the synthesis of aliphatic and arylaliphatic α,β-epoxy ketones 489a–g. The latter compounds are attractive substrates for the synthesis of for example natural compounds (Scheme 140) [459].
Scheme 140: The base-catalyzed rearrangement of cyclic peroxides 488a–g.
Scheme 140: The base-catalyzed rearrangement of cyclic peroxides 488a–g.
Peroxy hemiketals 491 are the starting reagents in the synthesis of epoxides 492 and aldols 493. Scheme 141 shows the synthesis of epoxides and aldols from inexpensive and readily available α,β-unsubstituted ketones 490 through the intermediate formation of peroxy hemiketal 491 in the presence of a chiral catalyst [460].
Scheme 141: Synthesis of chiral epoxides and aldols from peroxy hemiketals 491.
Scheme 141: Synthesis of chiral epoxides and aldols from peroxy hemiketals 491.
A 1:1 mixture of the diastereomeric hydroperoxides 495a–e was synthesized by ozonolysis of (R)-carvone (494) and in situ trapping with primary alcohols ROH (R = Me, Et, Bu, Pent, Oct). Further cyclization of these hydroperoxides 495a–e using the sodium methanolate/MeOH system results in endoperoxides 496a–e exhibiting antimalarial activity (Scheme 142) [461].
Scheme 142: The multistep transformation of (R)-carvone (494) to endoperoxides 496a–e.
Scheme 142: The multistep transformation of (R)-carvone (494) to endoperoxides 496a–e.
The intramolecular rearrangement of 1,2-dioxetanes 497 containing an aromatic electron-donating substituent is accompanied by emission of light. This process is of special interest for the application in clinical and biological analytical methods, and the synthesis of carbonyl-containing compounds 498 (Table 23) [462-475].
Table 23: Base-catalyzed intramolecular rearrangement of 1,2-dioxetanes.
|
|
|||||
| Entry | X | R | Ar-OH | Reaction conditions | Ref. |
|---|---|---|---|---|---|
| 1 | O | t-Bu |
R1, R2 = H, OMe, CO2Me, CO2H, CH2OH |
TBAF in DMSO at 25 °C for 1 h | [463] |
| 2 | O | t-Bu |
R = H, OMe; Y = O, S |
NaOH in СH3CN/H2O at 45 °C | [464] |
| 3 | O | t-Bu |
|
TBAF in DMSO (NMP or DMF) at 45–100 °C | [465] |
| 4 | O | Me, Et, iPr, iBu | in NMP at 50–100 °C or in TBAF/NMP at 35–60 °C | [466] | |
| 5 | O | t-Bu |
|
TBAF in CH3CN at 45 °C | [467] |
| 6 | NBoc | t-Bu |
3-OH-C6H4
3-OMe-C6H4 6-OH-C10H6 |
TBAF in DMSO at 25 °C | [468,469,473] |
| 7 | S, SO, S(O)2 | t-Bu |
3-OH-C6H4
3-OMe-C6H4 3-OAc-C6H4 |
TBAF in DMSO at 25 °C | [472] |
| 8 | O | t-Bu | HO-phenanthrenyl | TBAF in CH3CN at 45 °C | [474] |
| 9 | O | t-Bu |
R = H, Me, Ph |
TBAF in CH3CN or NaOH in H2O at 45 °C | [475] |
| 10 |
|
TBAF in DMSO at 25 °C | [470] | ||
| 11 |
|
TBAF in THF/DMSO (1:1) at 25 °C | [476] | ||
| 12 |
|
DBU in CH3CN at 25 °C | [477] | ||
| 13 |
|
TBAF in DMSO/PBS buffer | [471] | ||
Catalytic amounts of a sodium bicarbonate are sufficient to induce the decomposition of anthracene endoperoxide 499 to anthraquinone (500) (Scheme 143) [478].
Scheme 143: The decomposition of anthracene endoperoxide 499.
Scheme 143: The decomposition of anthracene endoperoxide 499.
An intramolecular rearrangement of α-azidoperoxides 502 promoted by DBU provides esters 503. The reaction takes place with alkyl, aryl and heteroaryl α-azidoperoxides generated from the corresponding aldehydes 501 (Scheme 144) [479].
Scheme 144: Synthesis of esters 503 from aldehydes 501 via rearrangement of peroxides 502.
Scheme 144: Synthesis of esters 503 from aldehydes 501 via rearrangement of peroxides 502.
There could be two possible paths for base-promoted decomposition of α-azidoperoxides 502 (Scheme 145). The abstraction of the α-hydrogen in the azidoperoxide leads to the direct decomposition of the peroxide bond, which provides acylazide 504 and alkoxide ion 505 (path A). Further, the exchange of the azide moiety in the acylazide with an alkoxide ion generates esters 503. On the other hand, an abstraction of the α-hydrogen in the azidoperoxide leads to a resonance-stabilized intermediate I (path B). Then, an intramolecular 1,2-alkoxy migration of I, via scission of the peroxide bond, followed by cleavage of the C–N bond (intermediate IV) affords the desired ester 503. On basis of control experiments, the reaction is probably following the latter path.
Scheme 145: Two possible paths for the base-promoted decomposition of α-azidoperoxides 502.
Scheme 145: Two possible paths for the base-promoted decomposition of α-azidoperoxides 502.
2.4 Thermal and photochemical transformations of organic peroxides
Story and co-workers discovered that the thermal and photochemical decomposition of cyclic ketone peroxides 506 produces cycloalkanes 507 and cyclic lactones 508 (Scheme 146 and Scheme 147) [480-483]. This transformation is a general method for the synthesis of macrocyclic compounds from readily available starting materials.
Scheme 146: The Story decomposition of cyclic diperoxide 506a.
Scheme 146: The Story decomposition of cyclic diperoxide 506a.
Scheme 147: The Story decomposition of cyclic triperoxide 506b.
Scheme 147: The Story decomposition of cyclic triperoxide 506b.
Examples of the thermal decomposition and photolysis of diperoxide 506a and triperoxide 506b are given in Table 24.
Unsaturated endoperoxides are convenient starting compounds for thermal and photochemical rearrangements. The thermal rearrangement of endoperoxides A into diepoxides B (Scheme 148) is one of the commonly used transformations [353,484,485].
Scheme 148: The thermal rearrangement of endoperoxides A into diepoxides B.
Scheme 148: The thermal rearrangement of endoperoxides A into diepoxides B.
The transformation of peroxide 510 is a key step in the synthesis of the cytotoxic agent stemolide (511) from methyl dehydroabietate (509) (Scheme 149) [486].
Scheme 149: The transformation of peroxide 510 in the synthesis of stemolide (511).
Scheme 149: The transformation of peroxide 510 in the synthesis of stemolide (511).
It was shown that thermal and photochemical transformations of endoperoxides 261g, 263, and 512a–с afford, in addition to diepoxides 513a–e, keto epoxides 514a–e [487-489]. Examples of the thermal decomposition and photolysis of endoperoxides 261g, 263, and 512a–c are given in Table 25.
The possible mechanism of the rearrangement of endoperoxide 261g is shown in Scheme 150. It is supposed that diepoxide 513a and keto epoxide 514a are generated from diradical 516 via cyclization of the diradical or a 1,2-hydride shift, respectively. Since 1,4-cyclohexanedione is not generated from endoperoxide, it can be concluded that the first cyclization of 515 to 1,3-biradical 516 occurs rapidly and the formation of epoxide rings takes place successively rather than simultaneously.
Scheme 150: The possible mechanism of the rearrangement of endoperoxide 261g.
Scheme 150: The possible mechanism of the rearrangement of endoperoxide 261g.
The photooxidation of indene 517 without a sensitizer provides dioxetane 518, in the presence of Rose Bengal, the diepoxyendoperoxide 521 is obtained. Product 521 originates probably from a [2 + 4] addition of singlet oxygen to give 519, followed by rearrangement to diepoxydiene 520, which is capable of adding a second mole of oxygen. The use of meso-tetraphenylporphyrin instead of Rose Bengal leads to the formation of diendiperoxide 522 (Scheme 151) [484].
Scheme 151: The photooxidation of indene 517.
Scheme 151: The photooxidation of indene 517.
Ascaridole (523) was slowly isomerized into isoascaridole (524) under irradiation with visible light (Scheme 152) [490]. Thermal and photochemical isomerization of related endoperoxides have been applied to the syntheses of other ascaridole analogs [491].
Scheme 152: The isomerization of ascaridole (523).
Scheme 152: The isomerization of ascaridole (523).
The diepoxide 526 was obtained in 67% yield by photolysis of 525 with a medium-pressure Hg vapor lamp (Scheme 153) [492].
Scheme 153: The isomerization of peroxide 525.
Scheme 153: The isomerization of peroxide 525.
The thermal transformation of endoperoxides produces mainly bis-epoxides, but can also provide unexpected products such as epoxy ketals. The heating of endoperoxide 236 to 160 °C in toluene affords epoxy ketal 528 in 53% yield through the formation of biradical 527 (Scheme 154) [347].
Scheme 154: The thermal transformation of endoperoxide 355.
Scheme 154: The thermal transformation of endoperoxide 355.
The photooxidation of cyclopentadiene (529) in an alcohol solution in the presence of polymerization inhibitors at a temperature higher than 0 °C gave cis-4,5-epoxy-2-pentenal (531) in 58% yield, cis-1,2,3,4-diepoxycyclopentane (532) as a byproduct (in 7% yield), and polymers instead of the expected peroxide 530 (Scheme 155) [344].
Scheme 155: The photooxidation of cyclopentadiene (529) at a temperature higher than 0 °C.
Scheme 155: The photooxidation of cyclopentadiene (529) at a temperature higher than 0 °C.
The extensive development of methods for the synthesis of cyclopentenones lies in the fact that this structural unit is present in some natural compounds, such as dihydrojasmone, prostaglandins, and rethrolones. The mechanism of thermal decomposition of saturated fulvene endoperoxides 533a–d involves the formation of one of the three intermediates A, B, C, which are precursors to cyclopentenones 534a–d (Table 26) [493].
The replacement of the vinyl group at the exocyclic double bond in the fulvene precursor by a 3-butenyl group and the thermal decomposition of the resulting endoperoxides 535 at 80 °C lead to a [3,4]-sigmatropic shift of the 3-butenyl group and formation of the 5-oxo-6-heptenal derivatives 536. The mechanism of this process involves the formation of epoxide A, which undergoes a [3,4]-shift through the intermediate B [494] (Table 27).
The thermal rearrangement of endoperoxides 538a,b, which are generated by the photooxidation of furanosyl furans 537a,b, selectively affords glycosides 539a,b (Scheme 156) [495].
Scheme 156: The thermal rearrangement of endoperoxides 538a,b.
Scheme 156: The thermal rearrangement of endoperoxides 538a,b.
The methylene blue-sensitized photooxidation of arabinofuranosyl furan 537a as an 1:6 α,β-anomeric mixture at −20 °C followed by warming of the reaction mixture to room temperature produced furanoside 539a as an anomeric mixture in the same molar ratio. The photooxidation of pure β-arabinofuranosyl furan 537a produced exclusively β-furanoside 539a. Based on these data, the intermediate endoperoxide 538a originates from the cycloaddition of 1O2 to the furanosyl furan. The selective thermal rearrangement of endoperoxide 538a, which is similar to the Baeyer–Villiger rearrangement with the retention of the configuration, results in the corresponding O-derivatives.
The thermally unstable endoperoxides 541a–d generated from 2-alkoxyfurans 540a–d rearrange through several pathways depending upon the nature of the substituent at the carbon atom C5 in 541 with formation of 542 or 543 (Table 28) [496].
The rearrangement of endoperoxides 541a–c containing a substituent with a tertiary hydroxy group in the 5 position results in the formation of hydroperoxyoxetanes 542a–c and trace amounts of Z-ketoesters 543a–c. Under the same conditions, the rearrangement of endoperoxide 541d containing a substituent with a secondary hydroxy group in the 5 position produces exclusively the Z-keto ester 543d [497].
Scheme 157: The transformation of peroxides 541.
Scheme 157: The transformation of peroxides 541.
This difference is apparently attributable to the following two factors: (1) the lower nucleophilicity of the secondary hydroxy group compared to the tertiary hydroxy group; (2) the conformer, which would be suitably orientated towards the nucleophilic attack, is sterically unfavored in the case of R2 = H. At −20 °C, the transformation of 541d into a conformational isomer occurs more slowly than the thermal decomposition giving 543d (Scheme 157). Thermal rearrangements of strained cyclic peroxides 544a–d and 546a–e provide a versatile tool for the synthesis of carbonyl compounds 545a–d and 547a–e and heterocyclic systems 548 and 549 (Scheme 158) [498,499].
Scheme 158: The thermal rearrangements of strained cyclic peroxides.
Scheme 158: The thermal rearrangements of strained cyclic peroxides.
The thermal rearrangement of diacyl peroxide 551 was carried out in the synthesis of the C4-epi-lomaiviticin B core 553. Diacyl peroxide 551 was prepared from p-nitroperbenzoic acid (p-NPBA) and the acid chloride of carboxylic acid 550. An ionic Criegee-like rearrangement of peroxide 551 upon heating resulted in the corresponding acyl carbonate species. The reaction of MeOH with this acyl carbonate intermediate provided a single diastereomer of secondary carbinol 552 in 38% yield (Scheme 159) [500].
Scheme 159: The thermal rearrangement of diacyl peroxide 551 in the synthesis of C4-epi-lomaiviticin B core 553.
Scheme 159: The thermal rearrangement of diacyl peroxide 551 in the synthesis of C4-epi-lomaiviticin B core 553....
Two diastereoisomeric dioxindolylalanines 556 were identified after the 1O2 oxidation of tryptophan (554). Mechanistic investigations supported the dioxindolylalanine formation through a dioxetane intermediate 555 (Scheme 160) [501].
Scheme 160: The 1O2 oxidation of tryptophan (554) and rearrangement of dioxetane intermediate 555.
Scheme 160: The 1O2 oxidation of tryptophan (554) and rearrangement of dioxetane intermediate 555.
2.5 Metal-catalyzed transformations of peroxides
This section focuses on transformations of peroxides under the action of the most representative metals used for these types of reactions: Fe(II), Co(II), Ru(II), and Pd(II).
The Fe(II)-promoted activation of peroxides is believed to be involved in the antimalarial activity of a number of peroxides, including the natural product artemisinin. The understanding of the underlying mechanism of the Fe(II)-promoted cleavage of bicyclic peroxides is critical to the design and preparation of more efficient antimalarial peroxides. From this perspective, metal-catalyzed transformations of peroxides are of special interest. It was shown [502] that the reaction of fluorinated cyclic peroxide 557a with FeBr2 in THF proceeds through an intermediate O-centered radical to form epoxy ketone 558a and 1,4-diol 559a. The reaction of 557b with FeCl2(PPh3)2 in CH2Cl2 proceeds in a different manner through an intermediate O-centered radical to yield diepoxide 560b (Scheme 161) [503].
Scheme 161: The Fe(II)-promoted cleavage of aryl-substituted bicyclic peroxides.
Scheme 161: The Fe(II)-promoted cleavage of aryl-substituted bicyclic peroxides.
In a related study investigating the reaction of 557а–с with FeBr2, bis-epoxides 560а–с and epoxy ketones 561а–с were obtained as the major products (Table 29) [504] and the proposed mechanism of the rearrangement of 557a–c is presented in Scheme 162.
Scheme 162: The proposed mechanism of the Fe(II)-promoted rearrangement of 557a–c.
Scheme 162: The proposed mechanism of the Fe(II)-promoted rearrangement of 557a–c.
Both in THF and CH3CN, the intermediate O-centered radical A is generated via an electron transfer from Fe(II) to 557a–c. The transformation of intermediate A can proceed through two different pathways. The first involves the intramolecular addition of an O-centered radical to the double bond in radical A to form C-centered radical B (path A). The second pathway involves an electron transfer from Fe(II) to radical A to give intermediate G (minor path B). The intramolecular electron transfer in intermediate B results in the formation of carbocation C followed by the formation of diepoxide 560a–c and concomitant elimination of Fe(II). The generation of epoxy ketone 561a–c from 560a–c can occur through paths C and D. Paths E and F apparently give rise to 1,4-diol 559a–c and diarylbenzene 562a–c, respectively, from intermediate G.
The reaction of dioxolane 563 with Fe(II) sulfate produces an O-centered radical, and the β-scission of the latter gives a C-centered radical, the oxidation and further cyclization of which yields 564 (Scheme 163) [505].
Scheme 163: The reaction of dioxolane 563 with Fe(II) sulfate.
Scheme 163: The reaction of dioxolane 563 with Fe(II) sulfate.
The monocyclic 1,2-dioxane 565, as opposed to related dioxolane 563, decomposes under the action of Fe(II) with exclusive formation of a 1:1 mixture of products 566 and 567. This is attributed to the fact that the reaction proceeds through 1,5-hydrogen transfer, while β-scission does not occur (Scheme 164) [505].
Scheme 164: Fe(II)-promoted rearrangement of 1,2-dioxane 565.
Scheme 164: Fe(II)-promoted rearrangement of 1,2-dioxane 565.
The reaction of Fe(II) cysteinate with dioxolane 568 produced compounds 569 and 570, which were isolated from the reaction mixture. The formation of methyl acetate 571 was confirmed by GC analysis of the reaction mixture before work-up (Scheme 165) [506].
Scheme 165: Fe(II) cysteinate-promoted rearrangement of 1,2-dioxolane 568.
Scheme 165: Fe(II) cysteinate-promoted rearrangement of 1,2-dioxolane 568.
The reaction of 1,2-dioxanes 572a–c with FeCl2 is accompanied by the formation of lactones 573a,b, which were isolated in the individual state (Scheme 166) [507].
Scheme 166: The transformation of 1,2-dioxanes 572a–c under the action of FeCl2.
Scheme 166: The transformation of 1,2-dioxanes 572a–c under the action of FeCl2.
The reaction of synthetic tetraoxane 574 with Fe(II) cysteinate affords a complex mixture of products. Only one product, 575, could be isolated from the mixture and identified. This was the first work, where the Fe(II)-promoted cleavage of 1,2,4,5-tetraoxane was investigated (Scheme 167) [508].
Scheme 167: Fe(II) cysteinate-promoted transformation of tetraoxane 574.
Scheme 167: Fe(II) cysteinate-promoted transformation of tetraoxane 574.
The hypothesis that this difference in the structure of the reaction products is associated with the rearrangement of intermediate endoperoxides gave impetus to research on the reaction of endoperoxides with transition metal derivatives. It was found that the catalytic rearrangement of endoperoxides using cobalt meso-tetraphenylporphyrin occurs in high yield. Therefore, this is an efficient approach to the synthesis of syn-1,2:3,4-diepoxides from 1,3-dienes under mild conditions. Table 30 summarizes the results of the cobalt(II) tetraphenylporpyrin-catalyzed rearrangement of endoperoxides 576 [509], 578 [510], 580 [511], 582 [512], 584 [353], 586 [513], 588 [514], 590, 592 [515], 594 [516], 596 [517], and 598 [518] which afforded products structurally similar to the diepoxides prepared by thermal rearrangement of endoperoxides (Table 25). All rearrangements were stereospecific and yielded only the syn-diepoxides.
The study of the CoTPP-catalyzed transformation of bicyclic endoperoxides containing non-strained diene moieties demonstrated that the formation of epoxides can be accomplished in yields up to 90–100%, while the side reaction giving epoxy ketones is suppressed. A detailed study on the CoTPP-catalyzed reaction of 600a showed that this reaction affords, in addition to the expected diepoxide 601a, two isomeric epoxy aldehydes 602a and 603a. The reaction of bicyclic endoperoxides 600b,c gives, instead of the expected epoxides 601b,c, exclusively epoxy aldehydes 602b, 603b and the reaction of endoperoxide 600d produces solely the diepoxide 601d (Scheme 168) [519].
Scheme 168: The CoTPP-catalyzed transformation of bicyclic endoperoxides 600a–d.
Scheme 168: The CoTPP-catalyzed transformation of bicyclic endoperoxides 600a–d.
The reaction of epoxy-1,2-dioxanes 604a–d and 606 with Co(II) complexes affords 4-hydroxy-2,3-epoxy ketones 605a–d and 607 in good yields (Scheme 169) [364]. Possibly the selectivity towards the hydroxyketones formation is provided by means of cobalt ions interaction. The obtained compounds are useful synthons in organic synthesis.
Scheme 169: The CoTPP-catalyzed transformation of epoxy-1,2-dioxanes.
Scheme 169: The CoTPP-catalyzed transformation of epoxy-1,2-dioxanes.
The Ru(II)-catalyzed reactions of 1,4-endoperoxide 261g involves the formation of intermediate radicals 608a,b, the structures of which differ from that of the radicals generated by photolysis or thermal decomposition. Ruthenium ions have a considerable effect on the stability and reactivity of radicals, resulting in the selective transformation of peroxides under mild conditions. The reactivity also substantially depends on steric factors (Scheme 170) [503,520,521].
Scheme 170: The Ru(II)-catalyzed reactions of 1,4-endoperoxide 261g.
Scheme 170: The Ru(II)-catalyzed reactions of 1,4-endoperoxide 261g.
The Ru(II)-catalyzed transformation of 1,4-endoperoxide 609 is used as a key step in the synthesis of the natural compound, elyiapyrone A (610) (Scheme 171) [522,523].
Scheme 171: The Ru(II)-catalyzed transformation as a key step in the synthesis of elyiapyrone A (610) from 1,4-endoperoxide.
Scheme 171: The Ru(II)-catalyzed transformation as a key step in the synthesis of elyiapyrone A (610) from 1,4-...
Transformations of endoperoxides catalyzed by variable-valence metals are well studied for metals such as Cu(II), Fe(II), or Co(II), which can initiate the reaction through a one-electron oxidation–reduction mechanism. The decomposition of endoperoxides catalyzed by Ru(II)phosphine complexes also belongs to this type of reaction and the decomposition produces diepoxides as the major products.
The reactions of endoperoxides with Pd(0) proceed through different pathways. Thus, bicyclic 2,3-saturated 1,4-endoperoxides 611a–d are transformed into the corresponding 4-hydroxyketones and 1,4-diols. Bicyclic 2,3-unsaturated 1,4-endoperoxides 530, 261g, 263 produce 4-hydroxyenones, 1,4-diols, and diepoxides. Monocyclic endoperoxides 611e–g are transformed into enones, 1,4-diols, 1,4-diketones, or furan derivatives (Table 31) [524,525].
Table 31: Pd(PPh3)4-catalyzed transformation of 1,4-endoperoxides.
| Endoperoxide | Temperature, °C (time, h) | Products, % | |||
|---|---|---|---|---|---|
|
611a |
17 (3) |
612a, 41 |
613a, 29 |
614a, 20 |
|
|
611b |
60 (5) |
612b, 49 |
613b, 3 |
614b, 37 |
|
|
611c |
60 (10) |
612c, 62 |
613c, 13 |
614c, 25 |
|
|
611d |
65 (15) |
612d, 73 |
614d, 23 |
||
|
530 |
4 (20) |
612e, 54 |
614e, 16 |
||
|
261g |
50–60 (5) |
262g, 42 |
614f, 32 |
513a, 9 |
|
|
263 |
60 (29) |
612f, 45 |
613d, 10 |
614g, 17 |
513b,28 |
|
611e |
60 (39) |
612g, 34 |
614h, 59 |
615, 5 |
|
|
611f |
100 (15) |
612h, 66 |
|||
|
611g |
70 (12) |
612i, 74 |
|||
The reactivity of bicyclic substrates depends on the carbon-ring size. Strained 1,4-endoperoxide derivatives are readily decomposed under the action of Pd(PPh3)4 at room or elevated temperatures, whereas substrates containing larger rings require more severe conditions. Monocyclic substrates are less reactive than bicyclic endoperoxides and require even more harsh conditions.
3 Rearrangements and related processes of important natural and synthetic peroxides
3.1 Antimalarial, antiparasitic, and antitumor peroxides
The extensive development of the chemistry of organic peroxides has been stimulated largely by the isolation of the antimalarial agent artemisinin from leaves of the annual wormwood Artemisia annua in 1972. The structural identification showed that artemisinin contains a cyclic endoperoxide moiety (1,2,4-trioxane ring), which plays a key role in its antimalarial activity [526,527]. The highly reactive and unusual chemical structure, in addition to low yields isolated from natural sources gave impetus to the development of total synthesis methods of artemisinin. Several routes towards the total synthesis of this compound were elaborated and several semisynthetic derivatives were prepared [12,16,528-533]. The high costs of these products stimulated the search for alternative peroxides, which are synthetically easier accessible and less expensive compared with the natural and semisynthetic structures. It was shown that 1,2-dioxolanes [35], 1,2-dioxanes [40], 1,2,4-trioxolanes [534-536], 1,2,4-trioxanes [44], and 1,2,4,5-tetraoxanes [537] exhibit antimalarial activity, which was sometimes higher than that of the parent artemisinin (Scheme 172). As a milestone of this research, arterolane, a fully synthetic 1,2,4-trioxolane was discovered and in 2012, the arterolane-based drug synriam was approved to the market.
Scheme 172: Peroxides with antimalarial activity.
Scheme 172: Peroxides with antimalarial activity.
Although artemisinin has been used in medicine for about three decades, the mechanism of its action remains unclear [538,539]. Two main theories of its antiparasitic action are assumed. In accordance with one theory, the endoperoxide bond is reduced by means of iron ions leading to the formation of oxygen-centered radicals, which are responsible for the initiation of oxidative stress in infected erythrocytes. An alternative theory proposes that specific parasites’ proteins or heme are alkylated by carbon-centered radicals derived from the peroxide [540,541]. In infected human erythrocytes, malaria parasites digest more than 70% of the hemoglobin with formation of globin and heme. After the hydrolysis of globin, the resulting amino acids are used by the parasites for protein synthesis. Malaria parasites detoxify the toxic heme via a heme polymerization process with preparation of hemozoin, which exists in the crystalline form. Parasite metalloproteins, superoxide dismutase and ferredoxin, use a small part of the host’s iron for their construction. In such a manner parasite cells always contain heme iron and non-heme iron, allows for the interaction with artemisinin or other peroxides [542].
Numerous studies on the interaction of iron ions with artemisinin (616) demonstrated that Fe(II) promotes the O–O-bond cleavage via two paths. Thus, Fe(II) may bind to either O1 or O2 in artemisinin (Scheme 173) [542-551]. The interaction of Fe(II) with O1 gives rise to an intermediate oxy radical 617a, which undergoes β-scission to form the primary C-centered radical 617b. The subsequent elimination of Fe(II) is accompanied by the formation of compound 618 containing a tetrahydrofuran ring. The pathway involving the interaction of Fe(II) with O2 affords the O-centered radical 619a. A subsequent [1,5]-H shift results in the formation of the secondary C-centered radical 619b, and the β-scission of the latter produces vinyl ester 620, which can be epoxidated by the resulting high-valent iron-oxo species. Epoxide 621 is finally cyclized to hydroxydeoxoartemisinin 622. The formation of 618 and 622 is evidence in favor of the proposed two pathways of the Fe(II)-promoted transformation of artemisinin. The highly reactive intermediates 617 and 619 apparently lead to the damage of some parasite biomolecules [552].
Scheme 173: The interaction of iron ions with artemisinin (616).
Scheme 173: The interaction of iron ions with artemisinin (616).
The 1,2-dioxanes 623 and 624 exhibiting antimalarial activity were isolated from the Caribbean sponge Plakortis simplex and their reactions with Fe(II) result in compounds 625a,b and 626a,b, respectively (Scheme 174) [553].
Scheme 174: The interaction of FeCl2 with 1,2-dioxanes 623, 624.
Scheme 174: The interaction of FeCl2 with 1,2-dioxanes 623, 624.
Scheme 175 shows the mechanism including the formation of oxygen radicals 627, 629 from cyclic peroxides 623 and 624. The 1,5-rearrangement of the latter produces the alkyl-side chain carbon-centered radicals 628, 630. The reaction of these toxic intermediates with parasite biomolecules determines the biological effect observed for 1,2-dioxanes 623 and 624 (Scheme 175).
Scheme 175: The mechanism of reaction 623 and 624 with Fe(II)Cl2.
Scheme 175: The mechanism of reaction 623 and 624 with Fe(II)Cl2.
Depending on the nature of the substituents in close vicinity of the peroxide group, the bicyclic natural endoperoxides G3-factors 631–633 which are involved in plant defense and extracted from the leaves of Eucalyptus grandis, react with Fe(II) to form different types of products. For instance, treatment of the 631 with Fe(II)SO4, gives rise to 634 in 82% yield. On the other hand the reaction of 632 under the same reaction conditions affords three products 635, 636, and 637 in a 1:1:1 ratio. The fluorinated endoperoxide 633 gives exclusively 638 under these conditions (Scheme 176) [554,555].
Scheme 176: The reaction of bicyclic natural endoperoxides G3-factors 631–633 with FeSO4.
Scheme 176: The reaction of bicyclic natural endoperoxides G3-factors 631–633 with FeSO4.
In the reaction with Fe(II), the natural antimalarial terpene cardamom peroxide 639 isolated from Amomum krervanh Pierre (Siam cardamom) is transformed into acids 640, 641, and 642 (Scheme 177) [164].
Scheme 177: The transformation of terpene cardamom peroxide 639.
Scheme 177: The transformation of terpene cardamom peroxide 639.
However, the cleavage of tetraoxane 643 gives two major products, namely 644 and 645, in yields of 44% and 51%, respectively. The reaction mechanism based on the results of this study is shown in Scheme 178 [556].
Scheme 178: The different ways of the cleavage of tetraoxane 643.
Scheme 178: The different ways of the cleavage of tetraoxane 643.
Presumably, in accordance with the direction from Fe(II) to O2, tetraoxane 646 interacts with iron(II) heme 647. Starting heme 647 reacts within 30 min with formation of three products. The LC–MS study proved the formation of the covalent coupling product 648 formed from heme (mass 616) and the tetroxane-derived secondary C-centered radical. The molecular ion [M]+ of coupling product 648 was observed at m/z 782.3, which is consistent with the prediction (Scheme 179) [537,556].
Scheme 179: The LC–MS analysis of interaction of tetraoxane 646 with iron(II)heme 647.
Scheme 179: The LC–MS analysis of interaction of tetraoxane 646 with iron(II)heme 647.
Under similar conditions, the same alkylated heme adduct was obtained with trioxolanes [557]. Four peaks at m/z 782.3 were detected which were assigned to the four possible regioisomers of alkylated heme adduct 648 as reported for heme–artemisinin adducts [558]. Later, in an initial study dealing with monoclonal antibodies that recognize the alkylation signature (sum of heme and protein alkylation) of synthetic peroxides it was shown that the artemisinins alkylate proteins in P. falciparum [559].
All the above-mentioned transformations involve the homolytic O–O-bond cleavage resulting in the formation of an O-centered radical, which is followed by the rearrangement into a C-centered radical, as a key step. The subsequent transformation of the C-centered radical determines the structure of the final product.
The peroxide, 3,6-epidioxy-1,10-bisaboladiene (EDBD, 649), isolated from wild plants, Cacalia delphiniifolia and Cacalia hastata, possesses cytotoxicity against the human promyelocytic leukemia cell line HL60. It was shown that the mechanism of biological activity of EDBD involves a rearrangement with formation of an unstable C-centered radical intermediate 650, followed by its transformation into product 651 (Scheme 180) [560].
Scheme 180: The rearrangement of 3,6-epidioxy-1,10-bisaboladiene (EDBD, 649).
Scheme 180: The rearrangement of 3,6-epidioxy-1,10-bisaboladiene (EDBD, 649).
3.2 Rearrangement of lipid peroxides
Lipids contained in cell membranes maintain the structure and control of the vital functions of cells. Lipids are the targets of the reactions with reactive oxygen species (ROS) such as various oxygen-centered radicals, which play a key role in several pathological states [561]. Compounds containing double bonds, polyunsaturated fatty acids and esters, cholesterol and its derivatives easily undergo oxidation by action of oxygen-centered radicals (Scheme 181) [562,563].
Rearrangements of organic peroxides play an important role in such biological processes as the synthesis of prostaglandins from fatty acids. Prostaglandins are physiologically active substances produced by the reaction of arachidonic acid (652) with cyclooxygenase (COX) isoenzymes. Prostaglandin G2 (PGG2, 653) containing an endoperoxide fragment undergoes transformations mediated by a series of specific isomerases and synthases with production of PGE2, PGI2, PGD2, PGF2, and TXA2 (Scheme 182) [564-567].
Scheme 182: Biopathway of synthesis of prostaglandins.
Scheme 182: Biopathway of synthesis of prostaglandins.
The formation of the metabolites isoprostanes, neuroprostanes, phytoprostanes, and isofurans 655–657 from fatty acids under autoxidative conditions in vivo involves both the reduction of peroxides and their rearrangements (Scheme 183). These compounds proved to be widespread in nature. Compounds 655–657 display significant biological activities, and the isoprostanes are currently the most reliable indicators of oxidative stress [568-570].
Scheme 183: The reduction and rearrangements of isoprostanes.
Scheme 183: The reduction and rearrangements of isoprostanes.
One of the essential fatty acids, linoleic acid, contains a homoconjugated diene fragment, which is responsible for a specific peroxidation mechanism without the formation of cyclic peroxides. In addition to linoleic acid, its esters are present in the human circulating low-density lipoprotein (LDL). For this reason, the oxidation of linoleic acid esters is of special biomedical interest [566]. A mechanism for linoleate (658) oxidation, which involves hydroperoxyoctadecadienoates (HPODE, 660–662) preparation, is presented in Scheme 184. The first step of the oxidation process is the formation of the carbon-centered pentadienyl radical 659. The reaction of 659 with O2 produces three peroxyl radicals, one of them having a nonconjugated diene part with the oxygen at C-11 position. The two other radicals have Z,E- and E,E-conjugated diene parts with oxygen substituents at the C-9 and C-13 positions. These peroxyl radical intermediates after abstracting hydrogen atoms transform to the hydroperoxyoctadecadienoates (HPODE, 660–662) [570].
Scheme 184: The partial mechanism for linoleate 658 oxidation.
Scheme 184: The partial mechanism for linoleate 658 oxidation.
The Hock cleavage mechanism is a possible route to transform lipid hydroperoxide 663 into smaller carbonyl compounds 664–666, although this transformation seems to occur only in the presence of photosensitizers (Scheme 185) [571].
Scheme 185: The transformation of lipid hydroperoxide.
Scheme 185: The transformation of lipid hydroperoxide.
In mammalian tissues and cells, cholesterol is found to a large extent. One of the main cholesterol functions represents to maintaining the stability of plasma membranes. The oxidation of cholesterol by means of free radical particles is responsible for the initiation of a range of pathological conditions [572,573]. Many processes including the rearrangement of intermediately formed peroxides accompany the oxidation of cholesterol. The major product of 1O2 oxidation of cholesterol (667), cholesterol 5α-hydroperoxide (668), readily forms 5,6-secosterol ketoaldehyde 669 and the product of its intramolecular aldolization 670 through an acid-catalyzed (Hock) cleavage of the C5–C6 bond in 668 (Scheme 186) [67].
Scheme 186: The acid-catalyzed cleavage of the product from free-radical oxidation of cholesterol (667).
Scheme 186: The acid-catalyzed cleavage of the product from free-radical oxidation of cholesterol (667).
3.3 Rearrangement of dioxygenase enzyme–substrate systems
A useful chemical property of most soil bacteria concludes in their capability to oxidize aromatic compounds. This multistep process depends on the structure of dioxygenase enzymes, which utilize molecular oxygen for oxidation [574]. This oxidation has attracted much attention as a green chemistry approach for the conversion of aromatic compounds to water-soluble products and for degradation of lignin [575,576]. The ring cleavage of 1,2-dihydroxybenzene (catechol) is likely the most thoroughly studied reaction which is catalyzed by iron-dependent catechol dioxygenase enzymes [577-579]. The oxidation of catechols 671 and 673 by two types of enzymes – intradiol dioxygenase and extradiol dioxygenase – affords 3-carboxyhexa-2,4-dienedioic acid (672) and 2-hydroxy-6-ketonona-2,4-dienoic acid (674) (Scheme 187) [580,581].
Scheme 187: Two pathways of catechols oxidation.
Scheme 187: Two pathways of catechols oxidation.
A key step in the cleavage of the aromatic ring is the oxygen-atom insertion into the C–C-double bond as the result of a Criegee-like or Hock-like intermediate rearrangement [582,583]. It was demonstrated that, despite the different mechanisms of the initial step of the substrate/molecular oxygen activation, both reactions produce hydroperoxide 675 as the intermediate. This hydroperoxide undergoes Criegee-like or Hock-like rearrangement through different pathways. Intradiol dioxygenase catalyzes the 1,2-acyl migration (path B) and the formation of an intermediate anhydride 677. On the other hand, extradiol dioxygenase catalyzes the 1,2-migration of the alkenyl moiety (path A) through the intermediate formation of lactone 676 (Scheme 188) [584].
Scheme 188: Criegee-like or Hock-like rearrangement of the intermediate hydroperoxide 675 in dioxygenase enzyme–catechol system.
Scheme 188: Criegee-like or Hock-like rearrangement of the intermediate hydroperoxide 675 in dioxygenase enzyme...
Therefore, the catalyst for the O–O-bond cleavage in the Criegee-like intermediate determines the regioselectivity of the catechol oxidation.
A similar rearrangement of the Criegee intermediate with the cleavage of the С–С bond occurs in oxidative cleavage of natural organic pigments, carotinoides 679 by carotenoid cleavage dioxygenases (Scheme 189) [585,586].
Scheme 189: Carotinoides 679 cleavage by carotenoid cleavage dioxygenases.
Scheme 189: Carotinoides 679 cleavage by carotenoid cleavage dioxygenases.
In this section, we considered rearrangements of the most important natural and synthetic peroxides, which proceed or can take place in biological systems. Apparently, there are is a much larger number of biological processes, involving rearrangements of peroxides, which has to be discovered and studied in the future.
Conclusion
The rearrangements of organic peroxides and related processes are covered in the literature in hundreds of publications and several specialized reviews. However, these reviews are limited in scope, narrow in their approach, and do not provide an overall picture of this field of chemistry. The present review is the first to offer a complex analysis of the available data on rearrangements of peroxides published in the last 15−20 years with an excursion to the history of the discovery of particular reactions and transformations. The rearrangements and related processes are classified according to the type of the catalysts used: acid- and base-catalyzed processes, reactions catalyzed by variable-valence metals, photochemical and thermal action. Special emphasis is drawn to current trends in the performance and application of rearrangements of organic peroxides, such as asymmetric synthesis, organocatalysis, and the use of transition metal-peroxo complexes for the preparation of compounds interesting for pharmacological applications. The published data summarized in the review provide, for the first time, an insight into the common and different features of the reaction mechanisms and allow predicting experimental and structural requirements for performing rearrangements with specified results. An analysis of the published data shows that there are numerous new and unnamed processes related to name reactions. The development and investigation of these processes are apparently the future of peroxide chemistry.
Rearrangements of organic peroxides are the key steps in processes such as the Baeyer−Villiger, the Criegee and Hock reactions, the Kornblum−DeLaMare rearrangement, and Dakin and Elbs oxidation reactions. These reactions are widely used in chemistry: The Baeyer−Villiger oxidation is widely used for the synthesis of esters and lactones. The Criegee reaction is employed to transform tertiary alcohols into ketones and aldehydes. The Kornblum−DeLaMare rearrangement is an important tool in the synthesis of γ-hydroxy enones. The Dakin oxidation is applied in the synthesis of phenols from arylaldehydes or aryl ketones and the Elbs persulfate oxidation is used to prepare hydroxyphenols from phenols.
The comprehensive analysis of the published data makes the knotty term "peroxide rearrangement” more exact. Two types of processes are actually included under the term "peroxide rearrangement”: processes that fall under the definition of a classical rearrangement, resulting in the formation of a compound of isomeric structure, and processes, in which the O–O-bond cleavage is followed by the rearrangement of one of the resulting fragments.
The pathways of peroxide rearrangements mainly depend on the type of the catalysts used, the reaction conditions, and the structure of the starting peroxide. Rearrangements can be accompanied by a homolytic or heterolytic O–O-bond cleavage, through the formation of a carbocation (e.g., the Criegee rearrangement), a carbanion (e.g., the Kornblum−DeLaMare rearrangement), or an O-centered radical (e.g., the Wieland rearrangement or rearrangements promoted by variable-valence metals).
In recent years, there has been a growing interest in organic peroxides as a base for the design of antiparasitic and antitumor agents, which led to an extensive search for new classes of peroxides. New compounds and new structural classes play a key role in the development of the chemistry of rearrangements and the performance of related useful transformations of peroxides.
References
-
Ando, W. Organic Peroxides; Wiley: Chichester, 1992.
Return to citation in text: [1] -
Reetz, I.; Yagci, Y.; Mishra, M. K. Initiation of Vinyl Polymerization by Organic Molecules and Nonmetal Initiators. Handbook of Vinyl Polymers; CRC Press: Boca Raton, 2008; pp 27–48.
Return to citation in text: [1] -
Flory, P. J. Principles of Polymer Chemistry; Cornell University Press: Ithaca, New York, 1953.
Return to citation in text: [1] -
Denisov, E. T.; Denisova, T. G.; Pokidova, T. S. Handbook of free radical initiators; John Wiley & Sons: New York, 2005.
Return to citation in text: [1] -
Adam, W. Four-membered ring peroxides: 1,2-dioxetanes and α-peroxylactones. In Peroxides; Patai, S., Ed.; John Wiley & Sons, Ltd.: Chichester, U.K., 1983; pp 829–920. doi:10.1002/9780470771730.ch24
Return to citation in text: [1] -
Bouillon, G.; Lick, C.; Schank, K. Diacyl peroxides, peroxycarboxylic acids and peroxy esters. In Peroxides; Patai, S., Ed.; John Wiley & Sons, Ltd.: Chichester, U.K., 1983; pp 279–309. doi:10.1002/9780470771730.ch10
Return to citation in text: [1] -
Saito, I.; Nittala, S. S. Endoperoxides. In Peroxides; Patai, S., Ed.; John Wiley & Sons, Ltd.: Chichester, U.K., 1983; pp 311–374. doi:10.1002/9780470771730.ch11
Return to citation in text: [1] -
Sheldon, R. A. Synthesis and uses of alkyl hydroperoxides and dialkyl peroxides. In Peroxides; Patai, S., Ed.; John Wiley & Sons, Ltd.: Chichester, U.K., 1983; pp 161–200. doi:10.1002/9780470771730.ch6
Return to citation in text: [1] -
Adam, W. Peroxide Chemistry: Mechanistic and Preparative Aspects of Oxygen Transfer; Wiley-VCH: Weinheim, 2000. doi:10.1002/3527600396
Return to citation in text: [1] -
World Health Organization. World Health Organization. World malaria report 2015; WHO Press, 2005.. http://www.who.int/malaria/publications/world-malaria-report-2015/en/ (accessed July 4, 2016).
Return to citation in text: [1] -
Jefford, C. W. Curr. Top. Med. Chem. 2012, 12, 373–399. doi:10.2174/156802612799362940
Return to citation in text: [1] -
Slack, R. D.; Jacobine, A. M.; Posner, G. H. Med. Chem. Commun. 2012, 3, 281–297. doi:10.1039/c2md00277a
Return to citation in text: [1] [2] -
Muregi, F. W.; Ishih, A. Drug Dev. Res. 2010, 71, 20–32. doi:10.1002/ddr.20345
Return to citation in text: [1] -
Fernández, I.; Robert, A. Org. Biomol. Chem. 2011, 9, 4098–4107. doi:10.1039/c1ob05088e
Return to citation in text: [1] -
Chadwick, J.; Amewu, R. K.; Marti, F.; Bousejra-El Garah, F.; Sharma, R.; Berry, N. G.; Stocks, P. A.; Burrell-Saward, H.; Wittlin, S.; Rottmann, M.; Brun, R.; Taramelli, D.; Parapini, S.; Ward, S. A.; O'Neill, P. M. ChemMedChem 2011, 6, 1357–1361. doi:10.1002/cmdc.201100196
Return to citation in text: [1] -
Opsenica, D. M.; Šolaja, B. A. J. Serb. Chem. Soc. 2009, 74, 1155–1193. doi:10.2298/JSC0911155O
Return to citation in text: [1] [2] -
Ghorai, P.; Dussault, P. H.; Hu, C. Org. Lett. 2008, 10, 2401–2404. doi:10.1021/ol800657m
Return to citation in text: [1] -
Dong, Y. Mini-Rev. Med. Chem. 2002, 2, 113–123. doi:10.2174/1389557024605537
Return to citation in text: [1] -
Vennerstrom, J. L.; Fu, H. N.; Ellis, W. Y.; Ager, A. L., Jr.; Wood, J. K.; Andersen, S. L.; Gerena, L.; Milhous, W. K. J. Med. Chem. 1992, 35, 3023–3027. doi:10.1021/jm00094a015
Return to citation in text: [1] -
Yadav, N.; Sharma, C.; Awasthi, S. K. RSC Adv. 2014, 4, 5469–5498. doi:10.1039/c3ra42513d
Return to citation in text: [1] -
Terent’ev, A. O.; Borisov, D. A.; Yaremenko, I. A. Chem. Heterocycl. Compd. 2012, 48, 55–58. doi:10.1007/s10593-012-0969-3
Return to citation in text: [1] -
Hao, H.-D.; Wittlin, S.; Wu, Y. Chem. – Eur. J. 2013, 19, 7605–7619. doi:10.1002/chem.201300076
Return to citation in text: [1] [2] -
Li, Y.; Hao, H.-D.; Wittlin, S.; Wu, Y. Chem. – Asian J. 2012, 7, 1881–1886. doi:10.1002/asia.201200166
Return to citation in text: [1] -
Boissier, J.; Portela, J.; Pradines, V.; Coslédan, F.; Robert, A.; Meunier, B. C. R. Chim. 2012, 15, 75–78. doi:10.1016/j.crci.2011.11.008
Return to citation in text: [1] -
Keiser, J.; Veneziano, V.; Rinaldi, L.; Mezzino, L.; Duthaler, U.; Cringoli, G. Res. Vet. Sci. 2010, 88, 107–110. doi:10.1016/j.rvsc.2009.05.007
Return to citation in text: [1] -
Keiser, J.; Ingram, K.; Vargas, M.; Chollet, J.; Wang, X.; Dong, Y.; Vennerstrom, J. L. Antimicrob. Agents Chemother. 2012, 56, 1090–1092. doi:10.1128/AAC.05371-11
Return to citation in text: [1] -
Shuhua, X.; Tanner, M.; N'Goran, E. K.; Utzinger, J.; Chollet, J.; Bergquist, R.; Minggang, C.; Jiang, Z. Acta Trop. 2002, 82, 175–181. doi:10.1016/S0001-706X(02)00009-8
Return to citation in text: [1] -
Ingram, K.; Yaremenko, I. A.; Krylov, I. B.; Hofer, L.; Terent’ev, A. O.; Keiser, J. J. Med. Chem. 2012, 55, 8700–8711. doi:10.1021/jm3009184
Return to citation in text: [1] -
Dembitsky, V. M. Eur. J. Med. Chem. 2008, 43, 223–251. doi:10.1016/j.ejmech.2007.04.019
Return to citation in text: [1] -
Dembitsky, V. M.; Gloriozova, T. A.; Poroikov, V. V. Mini-Rev. Med. Chem. 2007, 7, 571–589. doi:10.2174/138955707780859396
Return to citation in text: [1] -
del Sol Jimenez, M.; Garzón, S. P.; Rodriguez, A. D. J. Nat. Prod. 2003, 66, 655–661. doi:10.1021/np030021h
Return to citation in text: [1] -
Posner, G. H.; D’Angelo, J.; MO’Neill, P.; Mercer, A. Expert Opin. Ther. Pat. 2006, 16, 1665–1672. doi:10.1517/13543776.16.12.1665
Return to citation in text: [1] -
Lai, H. C.; Singh, N. P.; Sasaki, T. Invest. New Drugs 2013, 31, 230–246. doi:10.1007/s10637-012-9873-z
Return to citation in text: [1] -
Dwivedi, A.; Mazumder, A.; du Plessis, L.; du Preez, J. L.; Haynes, R. K.; du Plessis, J. Nanomedicine 2015, 11, 2041–2050. doi:10.1016/j.nano.2015.07.010
Return to citation in text: [1] -
Martyn, D. C.; Ramirez, A. P.; Berattie, M. J.; Cortese, J. F.; Patel, V.; Rush, M. A.; Woerpel, K. A.; Clardy, J. Bioorg. Med. Chem. Lett. 2008, 18, 6521–6524. doi:10.1016/j.bmcl.2008.10.083
Return to citation in text: [1] [2] -
Schiaffo, C. E.; Rottman, M.; Wittlin, S.; Dussault, P. H. ACS Med. Chem. Lett. 2011, 2, 316–319. doi:10.1021/ml100308d
Return to citation in text: [1] -
Ingram, K.; Schiaffo, C. E.; Sittiwong, W.; Benner, E.; Dussault, P. H.; Keiser, J. J. Antimicrob. Chemother. 2012, 67, 1979–1986. doi:10.1093/jac/dks141
Return to citation in text: [1] -
Wang, X.; Dong, Y.; Wittlin, S.; Charman, S. A.; Chiu, F. C. K.; Chollet, J.; Katneni, K.; Mannila, J.; Morizzi, J.; Ryan, E.; Scheurer, C.; Steuten, J.; Santo Tomas, J.; Snyder, C.; Vennerstrom, J. L. J. Med. Chem. 2013, 56, 2547–2555. doi:10.1021/jm400004u
Return to citation in text: [1] -
Chaudhary, S.; Sharma, V.; Jaiswal, P. K.; Gaikwad, A. N.; Sinha, S. K.; Puri, S. K.; Sharon, A.; Maulik, P. R.; Chaturvedi, V. Org. Lett. 2015, 17, 4948–4951. doi:10.1021/acs.orglett.5b02296
Return to citation in text: [1] -
O'Neill, P. M.; Stocks, P. A.; Pugh, M. D.; Araujo, N. C.; Korshin, E. E.; Bickley, J. F.; Ward, S. A.; Bray, P. G.; Pasini, E.; Davies, J.; Verissimo, E.; Bachi, M. D. Angew. Chem., Int. Ed. 2004, 43, 4193–4197. doi:10.1002/anie.200453859
Return to citation in text: [1] [2] -
Holla, H.; Labaied, M.; Pham, N.; Jenkins, I. D.; Stuart, K.; Quinn, R. J. Bioorg. Med. Chem. Lett. 2011, 21, 4793–4797. doi:10.1016/j.bmcl.2011.06.059
Return to citation in text: [1] -
Lombardo, M.; Sonawane, D. P.; Quintavalla, A.; Trombini, C.; Dhavale, D. D.; Taramelli, D.; Corbett, Y.; Rondinelli, F.; Fattorusso, C.; Persico, M.; Taglialatela-Scafati, O. Eur. J. Org. Chem. 2014, 1607–1614. doi:10.1002/ejoc.201301394
Return to citation in text: [1] -
Givelet, C.; Bernat, V.; Danel, M.; André-Barrès, C.; Vial, H. Eur. J. Org. Chem. 2007, 3095–3101. doi:10.1002/ejoc.200700086
Return to citation in text: [1] -
Amewu, R.; Gibbons, P.; Mukhtar, A.; Stachulski, A. V.; Ward, S. A.; Hall, C.; Rimmer, K.; Davies, J.; Vivas, L.; Bacsa, J.; Mercer, A. E.; Nixon, G.; Stocks, P. A.; O'Neill, P. M. Org. Biomol. Chem. 2010, 8, 2068–2077. doi:10.1039/b924319d
Return to citation in text: [1] [2] -
Reiter, C.; Fröhlich, T.; Zeino, M.; Marschall, M.; Bahsi, H.; Leidenberger, M.; Friedrich, O.; Kappes, B.; Hampel, F.; Efferth, T.; Tsogoeva, S. B. Eur. J. Med. Chem. 2015, 97, 164–172. doi:10.1016/j.ejmech.2015.04.053
Return to citation in text: [1] -
World Health Organization; Management of severe malaria – A practical handbook, 3rd ed.; WHO Press: 2013. http://www.who.int/malaria/publications/atoz/9789241548526/en/ (accessed July 4, 2016).
Return to citation in text: [1] -
World Health Organization; Guidelines for the treatment of malaria, 3rd ed.; WHO Press, 2015. http://www.who.int/malaria/publications/atoz/9789241549127/en/ (accessed July 4, 2016).
Return to citation in text: [1] -
Johnson, R. A.; Sharpless, K. B. Addition Reactions with Formation of Carbon–Oxygen Bonds: (ii) Asymmetric Methods of Epoxidation. In Comprehensive Organic Synthesis; Fleming, B. M. T., Ed.; Pergamon Press: Oxford, 1991; pp 389–436. doi:10.1016/B978-0-08-052349-1.00196-7
Return to citation in text: [1] -
Baer, H.; Bergamo, M.; Forlin, A.; Pottenger, L. H.; Lindner, J. Propylene Oxide. Ullmann's Encyclopedia of Industrial Chemistry; Wiley-VCH: Weinheim, Germany, 2012. doi:10.1002/14356007.a22_239.pub3
Return to citation in text: [1] -
Sienel, G.; Rieth, R.; Rowbottom, K. T. Epoxides. Ullmann's Encyclopedia of Industrial Chemistry; Wiley-VCH, 2000. doi:10.1002/14356007.a09_531
Return to citation in text: [1] -
Wang, Z. Prilezhaev Reaction. Comprehensive Organic Name Reactions and Reagents; John Wiley & Sons, Inc., 2010; pp 2270–2274. doi:10.1002/9780470638859.conrr513
Return to citation in text: [1] -
Weber, M.; Weber, M.; Kleine-Boymann, M. Phenol. Ullmann's Encyclopedia of Industrial Chemistry; Wiley-VCH, 2000. doi:10.1002/14356007.a19_299.pub2
Return to citation in text: [1] -
Schmidt, R. J. Appl. Catal., A 2005, 280, 89–103. doi:10.1016/j.apcata.2004.08.030
Return to citation in text: [1] -
Weissermel, K.; Arpe, H.-J. Benzene Derivatives. Industrial Organic Chemistry; Wiley-VCH, 2008; pp 337–385. doi:10.1002/9783527619191.ch13
Return to citation in text: [1] -
Ritz, J.; Fuchs, H.; Kieczka, H.; Moran, W. C. Caprolactam. Ullmann's Encyclopedia of Industrial Chemistry; Wiley-VCH, 2000. doi:10.1002/14356007.a05_031
Return to citation in text: [1] -
Vollhardt, K. P. C.; Schore, N. E. Organic Chemistry: Structure and Function, 7th ed.; W. H. Freeman and Company, 2014.
Return to citation in text: [1] -
Poth, U. Drying Oils and Related Products. Ullmann's Encyclopedia of Industrial Chemistry; Wiley-VCH, 2000. doi:10.1002/14356007.a09_055
Return to citation in text: [1] -
Mallégol, J.; Gardette, J.-L.; Lemaire, J. J. Am. Oil Chem. Soc. 2000, 77, 249–255. doi:10.1007/s11746-000-0041-5
Return to citation in text: [1] -
van Gorkum, R.; Bouwman, E. Coord. Chem. Rev. 2005, 249, 1709–1728. doi:10.1016/j.ccr.2005.02.002
Return to citation in text: [1] -
Juita; Dlugogorski, B. Z.; Kennedy, E. M.; Mackie, J. C. Fire Sci. Rev. 2012, 1, No. 3. doi:10.1186/2193-0414-1-3
Return to citation in text: [1] -
Oakley, L. H.; Casadio, F.; Shull, K. R.; Broadbelt, L. J. Appl. Phys. A 2015, 121, 869–878. doi:10.1007/s00339-015-9363-1
Return to citation in text: [1] -
Roda, A. Chemiluminescence and bioluminescence: past, present and future; RSC Press: Cambridge, 2010. doi:10.1039/9781849732024
Return to citation in text: [1] -
Dodeigne, C.; Thunus, L.; Lejeune, R. Talanta 2000, 51, 415–439. doi:10.1016/S0039-9140(99)00294-5
Return to citation in text: [1] -
Ciardelli, F.; Ruggeri, G.; Pucci, A. Chem. Soc. Rev. 2013, 42, 857–870. doi:10.1039/C2CS35414D
Return to citation in text: [1] -
Roda, A.; Mirasoli, M.; Michelini, E.; Di Fusco, M.; Zangheri, M.; Cevenini, L.; Roda, B.; Simoni, P. Biosens. Bioelectron. 2016, 76, 164–179. doi:10.1016/j.bios.2015.06.017
Return to citation in text: [1] -
Takeuchi, C.; Galvé, R.; Nieva, J.; Witter, D. P.; Wentworth, A. D.; Troseth, R. P.; Lerner, R. A.; Wentworth, P., Jr. Biochemistry 2006, 45, 7162–7170. doi:10.1021/bi0604330
Return to citation in text: [1] [2] -
Brinkhorst, J.; Nara, S. J.; Pratt, D. A. J. Am. Chem. Soc. 2008, 130, 12224–12225. doi:10.1021/ja804162d
Return to citation in text: [1] [2] [3] -
Murphy, R. C.; Johnson, K. M. J. Biol. Chem. 2008, 283, 15521–15525. doi:10.1074/jbc.R700049200
Return to citation in text: [1] -
Terao, J.; Minami, Y.; Bando, N. J. Clin. Biochem. Nutr. 2011, 48, 57–62. doi:10.3164/jcbn.11-008FR
Return to citation in text: [1] -
Terao, J. Cholesterol Hydroperoxides and Their Degradation Mechanism. In Lipid Hydroperoxide-Derived Modification of Biomolecules; Kato, Y., Ed.; Springer: Netherlands, 2014; Vol. 77, pp 83–91. doi:10.1007/978-94-007-7920-4_7
Return to citation in text: [1] -
Havaux, M. Plant J. 2014, 79, 597–606. doi:10.1111/tpj.12386
Return to citation in text: [1] -
Milas, N. A.; Mageli, O. L.; Golubovic, A.; Arndt, R. W.; Ho, J. C. J. J. Am. Chem. Soc. 1963, 85, 222–226. doi:10.1021/ja00885a022
Return to citation in text: [1] -
Rieche, A.; Bischoff, C. Chem. Ber. 1962, 95, 77–82. doi:10.1002/cber.19620950115
Return to citation in text: [1] -
Criegee, R. Organic Peroxides; Wiley-Interscience: New York, London, 1972.
Return to citation in text: [1] -
Cocker, W.; Grayson, D. H. J. Chem. Soc., Perkin Trans. 1 1975, 1347–1352. doi:10.1039/P19750001347
Return to citation in text: [1] -
Vinogradova, L. P.; Zav’yalov, S. I. Izv. Akad. Nauk SSSR, Ser. Khim. 1961, 2050–2054.
Chem. Abstr. 1962, 57, 12344c.
Return to citation in text: [1] -
Vinogradova, L. P.; Zav’yalov, S. I. Izv. Akad. Nauk SSSR, Ser. Khim. 1961, 1482–1486.
Chem. Abstr. 1962, 56, 338b.
Return to citation in text: [1] -
Vinogradova, L. P.; Rudenko, B. A.; Zav’yalov, S. I. Izv. Akad. Nauk SSSR, Ser. Khim. 1962, 1436–1441.
Chem. Abstr. 1963, 58, 2378g.
Return to citation in text: [1] -
Rieche, A.; Bischoff, C.; Prescher, D. Chem. Ber. 1964, 97, 3071–3075. doi:10.1002/cber.19640971116
Return to citation in text: [1] -
Rieche, A.; Seyfarth, H. E.; Brand, F. Justus Liebigs Ann. Chem. 1969, 725, 93–98. doi:10.1002/jlac.19697250112
Return to citation in text: [1] -
Wolffenstein, R. Ber. Dtsch. Chem. Ges. 1895, 28, 2265–2269. doi:10.1002/cber.189502802208
Return to citation in text: [1] -
Criegee, R.; Schnorrenberg, W.; Becke, J. Justus Liebigs Ann. Chem. 1949, 565, 7–21. doi:10.1002/jlac.19495650103
Return to citation in text: [1] -
Dickey, F. H.; Rust, F. F.; Vaughan, W. E. J. Am. Chem. Soc. 1949, 71, 1432–1434. doi:10.1021/ja01172a081
Return to citation in text: [1] -
Criegee, R.; Metz, K. Chem. Ber. 1956, 89, 1714–1718. doi:10.1002/cber.19560890720
Return to citation in text: [1] -
Hawkins, E. G. E. J. Chem. Soc. C 1969, 2663–2670. doi:10.1039/j39690002663
Return to citation in text: [1] -
Hawkins, E. G. E. J. Chem. Soc. C 1969, 2671–2677. doi:10.1039/j39690002671
Return to citation in text: [1] -
Hawkins, E. G. E. J. Chem. Soc. C 1969, 2678–2681. doi:10.1039/j39690002678
Return to citation in text: [1] -
Hawkins, E. G. E. J. Chem. Soc. C 1969, 2682–2686. doi:10.1039/j39690002682
Return to citation in text: [1] -
Hawkins, E. G. E. J. Chem. Soc. C 1969, 2686–2691. doi:10.1039/j39690002686
Return to citation in text: [1] -
Hawkins, E. G. E. J. Chem. Soc. C 1969, 2691–2697. doi:10.1039/j39690002691
Return to citation in text: [1] -
Milas, N. A.; Golubović, A. J. Am. Chem. Soc. 1959, 81, 6461–6462. doi:10.1021/ja01533a033
Return to citation in text: [1] -
Rieche, A. Angew. Chem. 1958, 70, 251–266. doi:10.1002/ange.19580700902
Return to citation in text: [1] -
Edwards, J. O.; Sauer, M. C. V. J. Phys. Chem. 1971, 75, 3004–3011. doi:10.1021/j100688a023
Return to citation in text: [1] -
Hine, J.; Redding, R. W. J. Org. Chem. 1970, 35, 2769–2772. doi:10.1021/jo00833a065
Return to citation in text: [1] -
Jefford, C. W.; Jaggi, D.; Kohmoto, S.; Boukouvalas, J.; Bernardinelli, G. Helv. Chim. Acta 1984, 67, 2254–2260. doi:10.1002/hlca.19840670831
Return to citation in text: [1] -
Singh, C. Tetrahedron Lett. 1990, 31, 6901–6902. doi:10.1016/S0040-4039(00)97202-2
Return to citation in text: [1] -
Jefford, C. W.; Li, Y.; Jaber, A.; Boukouvalas, J. Synth. Commun. 1990, 20, 2589–2596. doi:10.1080/00397919008051466
Return to citation in text: [1] -
Antonovskii, V. L.; Fedorova, E. V.; Shtivel, N. E.; Emelin, Y. D. Zh. Org. Khim. 1991, 27, 820–823.
Return to citation in text: [1] -
Avery, M. A.; Gao, F.; Chong, W. K. M.; Hendrickson, T. F.; Inman, W. D.; Crews, P. Tetrahedron 1994, 50, 957–972. doi:10.1016/S0040-4020(01)80810-3
Return to citation in text: [1] -
Rieche, A.; Bischoff, C.; Dietrich, P. Chem. Ber. 1961, 94, 2932–2936. doi:10.1002/cber.19610941115
Return to citation in text: [1] -
Shreibert, A. I.; Chardin, A. P.; Pil’dus, I. E.; Ermarchenko, V. I. Zh. Org. Khim. 1971, 7, 967–971.
Return to citation in text: [1] -
Razuvaev, G. A.; Dodonov, V. A.; Zaburdyaeva, S. N. Zh. Org. Khim. 1968, 4, 1302–1303.
J. Org. Chem. USSR (Eng. Trans.) 1968, 4, 1256–1257.
Return to citation in text: [1] -
Dussault, P. H.; Lee, I. Q. J. Am. Chem. Soc. 1993, 115, 6458–6459. doi:10.1021/ja00067a090
Return to citation in text: [1] -
Dussault, P. H.; Eary, C. T. J. Am. Chem. Soc. 1998, 120, 7133–7134. doi:10.1021/ja9808503
Return to citation in text: [1] -
Dussault, P. H.; Lee, H.-J.; Niu, Q. J. J. Org. Chem. 1995, 60, 784–785. doi:10.1021/jo00109a001
Return to citation in text: [1] -
Kuznetsov, M. L.; Rocha, B. G. M.; Pombeiro, A. J. L.; Shul’pin, G. B. ACS Catal. 2015, 5, 3823–3835. doi:10.1021/acscatal.5b00077
Return to citation in text: [1] -
Wang, C.; Yamamoto, H. J. Am. Chem. Soc. 2014, 136, 1222–1225. doi:10.1021/ja411379e
Return to citation in text: [1] -
Bunge, A.; Hamann, H.-J.; Dietz, D.; Liebscher, J. Tetrahedron 2013, 69, 2446–2450. doi:10.1016/j.tet.2013.01.032
Return to citation in text: [1] -
Jadhav, V. H.; Bande, O. P.; Puranik, V. G.; Dhavale, D. D. Tetrahedron: Asymmetry 2010, 21, 163–170. doi:10.1016/j.tetasy.2010.01.007
Return to citation in text: [1] -
Cussó, O.; Garcia-Bosch, I.; Ribas, X.; Lloret-Fillol, J.; Costas, M. J. Am. Chem. Soc. 2013, 135, 14871–14878. doi:10.1021/ja4078446
Return to citation in text: [1] -
Nieto, N.; Munslow, I. J.; Fernández-Pérez, H.; Vidal-Ferran, A. Synlett 2008, 2856–2858. doi:10.1055/s-0028-1083545
Return to citation in text: [1] -
Burke, C. P.; Shi, Y. J. Org. Chem. 2007, 72, 4093–4097. doi:10.1021/jo070205r
Return to citation in text: [1] -
Lifchits, O.; Mahlau, M.; Reisinger, C. M.; Lee, A.; Farès, C.; Polyak, I.; Gopakumar, G.; Thiel, W.; List, B. J. Am. Chem. Soc. 2013, 135, 6677–6693. doi:10.1021/ja402058v
Return to citation in text: [1] -
Wang, L.; Sha, W.; Dai, Q.; Feng, X.; Wu, W.; Peng, H.; Chen, B.; Cheng, J. Org. Lett. 2014, 16, 2088–2091. doi:10.1021/ol500277u
Return to citation in text: [1] -
Dragan, A.; Kubczyk, T. M.; Rowley, J. H.; Sproules, S.; Tomkinson, N. C. O. Org. Lett. 2015, 17, 2618–2621. doi:10.1021/acs.orglett.5b00953
Return to citation in text: [1] -
Chen, H.-H.; Wang, G.-Z.; Han, J.; Xu, M.-Y.; Zhao, Y.-Q.; Xu, H.-J. Tetrahedron 2014, 70, 212–217. doi:10.1016/j.tet.2013.11.085
Return to citation in text: [1] -
Gogoi, A.; Modi, A.; Guin, S.; Rout, S. K.; Das, D.; Patel, B. K. Chem. Commun. 2014, 50, 10445–10447. doi:10.1039/C4CC04407J
Return to citation in text: [1] -
Wu, X.-F.; Gong, J.-L.; Qi, X. Org. Biomol. Chem. 2014, 12, 5807–5817. doi:10.1039/C4OB00276H
Return to citation in text: [1] -
Zhao, J.; Fang, H.; Han, J.; Pan, Y.; Li, G. Adv. Synth. Catal. 2014, 356, 2719–2724. doi:10.1002/adsc.201400032
Return to citation in text: [1] -
Dai, Q.; Jiang, Y.; Yu, J.-T.; Cheng, J. Synthesis 2015, 48, 329–339. doi:10.1055/s-0035-1560536
Return to citation in text: [1] -
Terent’ev, A. O.; Borisov, D. A.; Chernyshev, V. V.; Nikishin, G. I. J. Org. Chem. 2009, 74, 3335–3340. doi:10.1021/jo900226b
Return to citation in text: [1] -
Li, Y.; Hao, H.-D.; Wu, Y. Org. Lett. 2009, 11, 2691–2694. doi:10.1021/ol900811m
Return to citation in text: [1] -
Opsenica, D. M.; Pocsfalvi, G.; Milhous, W. K.; Šolaja, B. A. J. Serb. Chem. Soc. 2002, 67, 465–471. doi:10.2298/JSC0207465O
Return to citation in text: [1] -
Terent'ev, A. O.; Kutkin, A. V.; Platonov, M. M.; Ogibin, Y. N.; Nikishin, G. I. Tetrahedron Lett. 2003, 44, 7359–7363. doi:10.1016/S0040-4039(03)01844-6
Return to citation in text: [1] -
Žmitek, K.; Zupan, M.; Stavber, S.; Iskra, J. J. Org. Chem. 2007, 72, 6534–6540. doi:10.1021/jo0708745
Return to citation in text: [1] -
Ghorai, P.; Dussault, P. H. Org. Lett. 2008, 10, 4577–4579. doi:10.1021/ol801859c
Return to citation in text: [1] -
Amewu, R.; Stachulski, A. V.; Ward, S. A.; Berry, N. G.; Bray, P. G.; Davies, J.; Labat, G.; Vivas, L.; O'Neill, P. M. Org. Biomol. Chem. 2006, 4, 4431–4436. doi:10.1039/b613565j
Return to citation in text: [1] -
Landenberger, K. B.; Bolton, O.; Matzger, A. J. Angew. Chem., Int. Ed. 2013, 52, 6468–6471. doi:10.1002/anie.201302814
Return to citation in text: [1] -
Climent, E.; Gröninger, D.; Hecht, M.; Walter, M. A.; Martinez-Máñez, R.; Weller, M. G.; Sancenón, F.; Amorós, P.; Rurack, K. Chem. – Eur. J. 2013, 19, 4117–4122. doi:10.1002/chem.201300031
Return to citation in text: [1] -
Terent'ev, A. O.; Borisov, D. A.; Yaremenko, I. A.; Chernyshev, V. V.; Nikishin, G. I. J. Org. Chem. 2010, 75, 5065–5071. doi:10.1021/jo100793j
Return to citation in text: [1] -
Terent'ev, A. O.; Borisov, D. A.; Semenov, V. V.; Chernyshev, V. V.; Dembitsky, V. M.; Nikishin, G. I. Synthesis 2011, 2091–2100. doi:10.1055/s-0030-1260027
Return to citation in text: [1] -
Singh, C.; Srivastav, N. C.; Puri, S. K. Bioorg. Med. Chem. 2004, 12, 5745–5752. doi:10.1016/j.bmc.2004.08.042
Return to citation in text: [1] -
Griesbeck, A. G.; El-Idreesy, T. T.; Höinck, L.-O.; Lex, J.; Brun, R. Bioorg. Med. Chem. Lett. 2005, 15, 595–597. doi:10.1016/j.bmcl.2004.11.043
Return to citation in text: [1] -
Singh, C.; Pandey, S.; Saxena, G.; Srivastava, N.; Sharma, M. J. Org. Chem. 2006, 71, 9057–9061. doi:10.1021/jo061414z
Return to citation in text: [1] -
Liu, Y.-H.; Deng, J.; Gao, J.-W.; Zhang, Z.-H. Adv. Synth. Catal. 2012, 354, 441–447. doi:10.1002/adsc.201100561
Return to citation in text: [1] -
Griesbeck, A. G.; Schlundt, V. Synlett 2011, 2430–2432. doi:10.1055/s-0030-1261225
Return to citation in text: [1] [2] -
Kazakov, D. V.; Kazakova, O. B.; Ishmuratov, G. Yu.; Terent'ev, A. O.; Nikishin, G. I.; Tolstikov, G. A. Dokl. Chem. 2011, 436, 34–38. doi:10.1134/S0012500811010022
Return to citation in text: [1] -
Sashidhara, K. V.; Avula, S. R.; Singh, L. R.; Palnati, G. R. Tetrahedron Lett. 2012, 53, 4880–4884. doi:10.1016/j.tetlet.2012.07.001
Return to citation in text: [1] -
Hall, J. F. B.; Bourne, R. A.; Han, X.; Earley, J. H.; Poliakoff, M.; George, M. W. Green Chem. 2013, 15, 177–180. doi:10.1039/C2GC36711D
Return to citation in text: [1] -
Terent'ev, A. O.; Yaremenko, I. A.; Vil', V. A.; Moiseev, I. K.; Kon'kov, S. A.; Dembitsky, V. M.; Levitsky, D. O.; Nikishin, G. I. Org. Biomol. Chem. 2013, 11, 2613–2623. doi:10.1039/c3ob27239g
Return to citation in text: [1] -
Terent'ev, A. O.; Yaremenko, I. A.; Chernyshev, V. V.; Dembitsky, V. M.; Nikishin, G. I. J. Org. Chem. 2012, 77, 1833–1842. doi:10.1021/jo202437r
Return to citation in text: [1] -
Terent'ev, A. O.; Yaremenko, I. A.; Vil', V. A.; Dembitsky, V. M.; Nikishin, G. I. Synthesis 2013, 45, 246–250. doi:10.1055/s-0032-1317895
Return to citation in text: [1] -
Nesvadba, P. Radical Polymerization in Industry. Encyclopedia of Radicals in Chemistry, Biology and Materials; John Wiley & Sons, 2012. doi:10.1002/9781119953678.rad080
Return to citation in text: [1] -
Moad, G.; Solomon, D. H. The chemistry of radical polymerization, 2nd ed.; Elsevier, 2006.
Return to citation in text: [1] -
Ebewele, R. O. Polymer Science and Technology; CRC Press: Boca Raton, New York, 2000. doi:10.1201/9781420057805
Return to citation in text: [1] -
Mark, H. F. Encyclopedia of Polymer Science and Technology, 3rd ed.; John Wiley & Sons, 2007.
Return to citation in text: [1] -
Matyjaszewski, K.; Davis, T. P. Future Outlook and Perspectives. Handbook of Radical Polymerization; John Wiley & Sons, 2003; pp 895–900. doi:10.1002/0471220450.ch16
Return to citation in text: [1] -
Hao, H.-D.; Li, Y.; Han, W.-B.; Wu, Y. Org. Lett. 2011, 13, 4212–4215. doi:10.1021/ol2015434
Return to citation in text: [1] -
Li, Y.; Hao, H.-D.; Zhang, Q.; Wu, Y. Org. Lett. 2009, 11, 1615–1618. doi:10.1021/ol900262t
Return to citation in text: [1] -
Yan, X.; Chen, J.; Zhu, Y.-T.; Qiao, C. Synlett 2011, 2827–2830. doi:10.1055/s-0031-1289864
Return to citation in text: [1] -
Žmitek, K.; Zupan, M.; Stavber, S.; Iskra, J. Org. Lett. 2006, 8, 2491–2494. doi:10.1021/ol060590r
Return to citation in text: [1] -
Azarifar, D.; Khosravi, K.; Soleimanei, F. Synthesis 2009, 2553–2556. doi:10.1055/s-0029-1217394
Return to citation in text: [1] -
Terent'ev, A. O.; Kutkin, A. V.; Platonov, M. M.; Vorontsov, I. I.; Antipin, M. Yu.; Ogibin, Yu. N.; Nikishin, G. I. Russ. Chem. Bull. 2004, 53, 681–687. doi:10.1023/B:RUCB.0000035657.58776.cc
Return to citation in text: [1] -
Zhang, Q.; Jin, H.-X.; Liu, H.-H.; Wu, Y.-K. Chin. J. Chem. 2006, 24, 1190–1195. doi:10.1002/cjoc.200690222
Return to citation in text: [1] -
Zhang, Q.; Li, Y.; Wu, Y.-K. Chin. J. Chem. 2007, 25, 1304–1308. doi:10.1002/cjoc.200790242
Return to citation in text: [1] -
Terent'ev, A. O.; Kutkin, A. V.; Platonov, M. M.; Starikova, Z. A.; Ogibin, Yu. N.; Nikishin, G. I. Russ. Chem. Bull. 2005, 54, 1214–1218. doi:10.1007/s11172-005-0383-4
Return to citation in text: [1] -
Terent'ev, A. O.; Kutkin, A. V.; Starikova, Z. A.; Antipin, M. Yu.; Ogibin, Yu. N.; Nikishin, G. I. Synthesis 2004, 2356–2366. doi:10.1055/s-2004-831171
Return to citation in text: [1] -
Hamann, H.-J.; Hecht, M.; Bunge, A.; Gogol, M.; Liebscher, J. Tetrahedron Lett. 2011, 52, 107–111. doi:10.1016/j.tetlet.2010.10.151
Return to citation in text: [1] -
Terent'ev, A. O.; Platonov, M. M.; Sonneveld, E. J.; Peschar, R.; Chernyshev, V. V.; Starikova, Z. A.; Nikishin, G. I. J. Org. Chem. 2007, 72, 7237–7243. doi:10.1021/jo071072c
Return to citation in text: [1] -
Terent'ev, A. O.; Platonov, M. M.; Krylov, I. B.; Chernyshev, V. V.; Nikishin, G. I. Org. Biomol. Chem. 2008, 6, 4435–4441. doi:10.1039/b809661a
Return to citation in text: [1] -
Ito, T.; Tokuyasu, T.; Masuyama, A.; Nojima, M.; McCullough, K. J. Tetrahedron 2003, 59, 525–536. doi:10.1016/S0040-4020(02)01556-9
Return to citation in text: [1] -
Terent'ev, A. O.; Borisov, A. M.; Platonov, M. M.; Starikova, Z. A.; Chernyshev, V. V.; Nikishin, G. I. Synthesis 2009, 4159–4166. doi:10.1055/s-0029-1217062
Return to citation in text: [1] -
Eliasen, A. M.; Thedford, R. P.; Claussen, K. R.; Yuan, C.; Siegel, D. Org. Lett. 2014, 16, 3628–3631. doi:10.1021/ol501497y
Return to citation in text: [1] -
Hu, X.; Maimone, T. J. J. Am. Chem. Soc. 2014, 136, 5287–5290. doi:10.1021/ja502208z
Return to citation in text: [1] [2] -
Gamage, N.-D. H.; Stiasny, B.; Stierstorfer, J.; Martin, P. D.; Klapötke, T. M.; Winter, C. H. Chem. Commun. 2015, 51, 13298–13300. doi:10.1039/C5CC05015D
Return to citation in text: [1] -
Starkl Renar, K.; Pečar, S.; Iskra, J. Org. Biomol. Chem. 2015, 13, 9369–9372. doi:10.1039/C5OB01503K
Return to citation in text: [1] -
Rozhko, E.; Solmi, S.; Cavani, F.; Albini, A.; Righi, P.; Ravelli, D. J. Org. Chem. 2015, 80, 6425–6431. doi:10.1021/acs.joc.5b00861
Return to citation in text: [1] -
Hilf, J. A.; Witthoft, L. W.; Woerpel, K. A. J. Org. Chem. 2015, 80, 8262–8267. doi:10.1021/acs.joc.5b01326
Return to citation in text: [1] -
Lu, X.; Deng, L. Org. Lett. 2014, 16, 2358–2361. doi:10.1021/ol500677v
Return to citation in text: [1] -
Fisher, T. J.; Mattson, A. E. Org. Lett. 2014, 16, 5316–5319. doi:10.1021/ol502494h
Return to citation in text: [1] -
Wang, X.; Pan, Y.; Huang, K.-W.; Lai, Z. Org. Lett. 2015, 17, 5630–5633. doi:10.1021/acs.orglett.5b02881
Return to citation in text: [1] -
Yu, H.; Shen, J. Org. Lett. 2014, 16, 3204–3207. doi:10.1021/ol5012168
Return to citation in text: [1] -
Carney, J. M.; Hammer, R. J.; Hulce, M.; Lomas, C. M.; Miyashiro, D. Synthesis 2012, 44, 2560–2566. doi:10.1055/s-0031-1289764
Return to citation in text: [1] -
Yan, X.; Qiao, C.; Guo, Z. Synlett 2013, 24, 502–506. doi:10.1055/s-0032-1318213
Return to citation in text: [1] -
Hurlocker, B.; Miner, M. R.; Woerpel, K. A. Org. Lett. 2014, 16, 4280–4283. doi:10.1021/ol5020015
Return to citation in text: [1] -
Liu, Q.; Wang, W.; Liu, Z.; Wang, T.; Wu, L.; Ge, M. RSC Adv. 2014, 4, 19716–19724. doi:10.1039/C4RA02486A
Return to citation in text: [1] -
Parrish, J. D.; Ischay, M. A.; Lu, Z.; Guo, S.; Peters, N. R.; Yoon, T. P. Org. Lett. 2012, 14, 1640–1643. doi:10.1021/ol300428q
Return to citation in text: [1] -
Wei, W.; Zhang, C.; Xu, Y.; Wan, X. Chem. Commun. 2011, 47, 10827–10829. doi:10.1039/c1cc14602e
Return to citation in text: [1] -
Daeppen, C.; Kaiser, M.; Neuburger, M.; Gademann, K. Org. Lett. 2015, 17, 5420–5423. doi:10.1021/acs.orglett.5b02773
Return to citation in text: [1] -
Kazakova, O. B.; Kazakov, D. V.; Yamansarov, E. Yu.; Medvedeva, N. I.; Tolstikov, G. A.; Suponitsky, K. Yu.; Arkhipov, D. E. Tetrahedron Lett. 2011, 52, 976–979. doi:10.1016/j.tetlet.2010.12.047
Return to citation in text: [1] -
Lau, S.-H.; Galván, A.; Merchant, R. R.; Battilocchio, C.; Souto, J. A.; Berry, M. B.; Ley, S. V. Org. Lett. 2015, 17, 3218–3221. doi:10.1021/acs.orglett.5b01307
Return to citation in text: [1] -
Feng, Y.; Holte, D.; Zoller, J.; Umemiya, S.; Simke, L. R.; Baran, P. S. J. Am. Chem. Soc. 2015, 137, 10160–10163. doi:10.1021/jacs.5b07154
Return to citation in text: [1] -
Yablokov, V. A. Russ. Chem. Rev. 1980, 49, 833–842. doi:10.1070/RC1980v049n09ABEH002509
Usp. Khim. 1980, 49, 1711–1729.
Return to citation in text: [1] [2] -
Dussault, P. Reactions of Hydroperoxides and Peroxides. In Active Oxygen in Chemistry; Foote, C. S.; Valentine, J.; Greenberg, A.; Liebman, J., Eds.; Springer: Netherlands, 1995; Vol. 2, pp 141–203. doi:10.1007/978-94-007-0874-7_5
Return to citation in text: [1] -
Renz, M.; Meunier, B. Eur. J. Org. Chem. 1999, 737–750. doi:10.1002/(SICI)1099-0690(199904)1999:4<737::AID-EJOC737>3.0.CO;2-B
Return to citation in text: [1] [2] [3] -
Davies, A. G. J. Chem. Res. 2009, 533–544. doi:10.3184/030823409X12491375725131
Return to citation in text: [1] [2] [3] -
Syrkin, Ya. K.; Moiseev, I. I. Russ. Chem. Rev. 1960, 29, 193–214. doi:10.1070/RC1960v029n04ABEH001227
Usp. Khim. 1960, 29, 193.
Return to citation in text: [1] -
Ishmuratov, G. Yu.; Legostaeva, Yu. V.; Botsman, L. P.; Tolstikov, G. A. Russ. J. Org. Chem. 2010, 46, 1593–1621. doi:10.1134/S1070428010110011
Return to citation in text: [1] -
Smith, M. B. March's Advanced Organic Chemistry: Reactions, Mechanisms, and Structure, 7th ed.; John Wiley & Sons, Inc., 2013; p 2080.
Return to citation in text: [1] -
Wang, Z. Baeyer–Villiger Oxidation. Comprehensive Organic Name Reactions and Reagents; John Wiley & Sons, 2010; pp 150–155. doi:10.1002/9780470638859.conrr036
Return to citation in text: [1] -
Hassall, C. H. The Baeyer-Villiger Oxidation of Aldehydes and Ketones. In Organic Reactions; Adams, R., Ed.; John Wiley & Sons, Inc.: New York, 2011. doi:10.1002/0471264180.or009.03
Return to citation in text: [1] -
Baeyer, A.; Villiger, V. Ber. Dtsch. Chem. Ges. 1899, 32, 3625–3633. doi:10.1002/cber.189903203151
Return to citation in text: [1] -
Baeyer, A.; Villiger, V. Ber. Dtsch. Chem. Ges. 1900, 33, 858–864. doi:10.1002/cber.190003301153
Return to citation in text: [1] -
Belov, V. N.; Heyfitz, L. A.; Virezub, S. I. Okislenie karbonil'nykh soedineniy perekis'yu vodoroda i nadkislotami (reaktsiya Bayera–Villigera). Reaczii I Metodi issledovaniya organicheskich soedineniy; Goshimizdat: Moscow, 1961; Vol. 10, pp 7–208.
Return to citation in text: [1] [2] -
Krow, G. R. The Baeyer–Villiger Oxidation of Ketones and Aldehydes. In Organic Reactions; Paquette, L. A., Ed.; John Wiley & Sons, Inc., 1993; Vol. 43, pp 251–798. doi:10.1002/0471264180.or043.03
Return to citation in text: [1] [2] -
Strukul, G. Angew. Chem., Int. Ed. 1998, 37, 1198–1209. doi:10.1002/(SICI)1521-3773(19980518)37:9<1198::AID-ANIE1198>3.0.CO;2-Y
Return to citation in text: [1] [2] [3] [4] -
Chida, N.; Tobe, T.; Ogawa, S. Tetrahedron Lett. 1994, 35, 7249–7252. doi:10.1016/0040-4039(94)85373-8
Return to citation in text: [1] -
Butkus, E.; Stončius, S. J. Chem. Soc., Perkin Trans. 1 2001, 1885–1888. doi:10.1039/b103564a
Return to citation in text: [1] -
Doering, W. v. E.; Speers, L. J. Am. Chem. Soc. 1950, 72, 5515–5518. doi:10.1021/ja01168a041
Return to citation in text: [1] -
ten Brink, G.-J.; Arends, I. W. C. E.; Sheldon, R. A. Chem. Rev. 2004, 104, 4105–4124. doi:10.1021/cr030011l
Return to citation in text: [1] [2] -
Lambert, A.; Macquarrie, D. J.; Carr, G.; Clark, J. H. New J. Chem. 2000, 24, 485–488. doi:10.1039/b003161p
Return to citation in text: [1] -
Chang, C. D.; Hellring, S. D. Shape-selective oxidation of cyclic ketones. U.S. Patent US4,870,192, Sept 26, 1989.
Return to citation in text: [1] -
Fischer, J.; Hölderich, W. F. Appl. Catal., A 1999, 180, 435–443. doi:10.1016/S0926-860X(98)00378-0
Return to citation in text: [1] [2] [3] -
Taylor, R. T.; Flood, L. A. J. Org. Chem. 1983, 48, 5160–5164. doi:10.1021/jo00174a003
Return to citation in text: [1] -
Jacobson, S. E.; Mares, F.; Zambri, P. M. J. Am. Chem. Soc. 1979, 101, 6938–6946. doi:10.1021/ja00517a025
Return to citation in text: [1] [2] [3] -
Sandaroos, R.; Goldani, M. T.; Damavandi, S.; Mohammadi, A. J. Chem. Sci. 2012, 124, 871–876. doi:10.1007/s12039-012-0277-6
Return to citation in text: [1] -
Hoelderich, W. P. D.; Fischer, J.; Schindler, G. P. D.; Arntz, D. D. Verfahren zur Herstellung von Lactonen durch Baeyer-Villiger Oxidation. Ger. Patent DE19,745,442 A1, April 22, 1999.
Chem. Abstr. 1999, 130, 281989u.
Return to citation in text: [1] -
Gavagnin, R.; Cataldo, M.; Pinna, F.; Strukul, G. Organometallics 1998, 17, 661–667. doi:10.1021/om970756f
Return to citation in text: [1] [2] -
Bolm, C.; Beckmann, O. Chirality 2000, 12, 523–525. doi:10.1002/(SICI)1520-636X(2000)12:5/6<523::AID-CHIR39>3.0.CO;2-Z
Return to citation in text: [1] -
Phillips, A. M. F.; Romão, C. Eur. J. Org. Chem. 1999, 1767–1770. doi:10.1002/(SICI)1099-0690(199908)1999:8<1767::AID-EJOC1767>3.0.CO;2-S
Return to citation in text: [1] -
Herrmann, W. A.; Fischer, R. W.; Correia, J. D. G. J. Mol. Catal. 1994, 94, 213–223. doi:10.1016/S0304-5102(94)87043-8
Return to citation in text: [1] -
ten Brink, G.-J.; Vis, J.-M.; Arends, I. W. C. E.; Sheldon, R. A. J. Org. Chem. 2001, 66, 2429–2433. doi:10.1021/jo0057710
Return to citation in text: [1] [2] [3] -
Grieco, P. A.; Yokoyama, Y.; Gilman, S.; Ohfune, Y. J. Chem. Soc., Chem. Commun. 1977, 870–871. doi:10.1039/c39770000870
Return to citation in text: [1] -
Jacobson, S. E.; Tang, R.; Mares, F. J. Chem. Soc., Chem. Commun. 1978, 888–889. doi:10.1039/c39780000888
Return to citation in text: [1] [2] -
Uchida, T.; Katsuki, T.; Ito, K.; Akashi, S.; Ishii, A.; Kuroda, T. Helv. Chim. Acta 2002, 85, 3078–3089. doi:10.1002/1522-2675(200210)85:10<3078::AID-HLCA3078>3.0.CO;2-1
Return to citation in text: [1] -
Berkessel, A.; Andreae, M. R. M. Tetrahedron Lett. 2001, 42, 2293–2295. doi:10.1016/S0040-4039(01)00141-1
Return to citation in text: [1] -
McClure, J. D.; Williams, P. H. J. Org. Chem. 1962, 27, 24–26. doi:10.1021/jo01048a005
Return to citation in text: [1] [2] -
Macias-Alonso, M.; Morzycki, J. W.; Iglesias-Arteaga, M. A. Steroids 2011, 76, 317–323. doi:10.1016/j.steroids.2010.12.004
Return to citation in text: [1] [2] -
Carlqvist, P.; Eklund, R.; Hult, K.; Brinck, T. J. Mol. Model. 2003, 9, 164–171. doi:10.1007/s00894-003-0128-y
Return to citation in text: [1] -
Bach, R. D. J. Org. Chem. 2012, 77, 6801–6815. doi:10.1021/jo300727w
Return to citation in text: [1] -
Hitomi, Y.; Yoshida, H.; Tanaka, T.; Funabiki, T. J. Mol. Catal. A: Chem. 2006, 251, 239–245. doi:10.1016/j.molcata.2006.02.002
Return to citation in text: [1] [2] -
Yamabe, S.; Yamazaki, S. J. Org. Chem. 2007, 72, 3031–3041. doi:10.1021/jo0626562
Return to citation in text: [1] -
Hamann, H.-J.; Bunge, A.; Liebscher, J. Chem. – Eur. J. 2008, 14, 6849–6851. doi:10.1002/chem.200800932
Return to citation in text: [1] [2] -
Gordon, N. J.; Evans, S. A., Jr. J. Org. Chem. 1993, 58, 4516–4519. doi:10.1021/jo00069a004
Return to citation in text: [1] -
Enders, D.; Geibel, G.; Osborne, S. Chem. – Eur. J. 2000, 6, 1302–1309. doi:10.1002/(SICI)1521-3765(20000417)6:8<1302::AID-CHEM1302>3.0.CO;2-J
Return to citation in text: [1] -
Iglesias-Arteaga, M. A.; Velazquez-Huerta, G. A.; Mendez-Stivalet, J. M.; Galano, A.; Alvarez-Idaboy, J. R. ARKIVOC 2006, No. vi, 109–126.
Return to citation in text: [1] -
Zhou, G.; Corey, E. J. J. Am. Chem. Soc. 2005, 127, 11958–11959. doi:10.1021/ja054503m
Return to citation in text: [1] -
Sharrna, R.; Nikas, S. P.; Guo, J. J.; Mallipeddi, S.; Wood, J. T.; Makriyannis, A. ACS Med. Chem. Lett. 2014, 5, 400–404. doi:10.1021/ml4005304
Return to citation in text: [1] -
Peris, G.; Miller, S. J. Org. Lett. 2008, 10, 3049–3052. doi:10.1021/ol8010248
Return to citation in text: [1] -
Patil, N. D.; Yao, S. G.; Meier, M. S.; Mobley, J. K.; Crocker, M. Org. Biomol. Chem. 2015, 13, 3243–3254. doi:10.1039/C4OB01771D
Return to citation in text: [1] -
Poladura, B.; Martínez-Castañeda, Á.; Rodríguez-Solla, H.; Llavona, R.; Concellón, C.; del Amo, V. Org. Lett. 2013, 15, 2810–2813. doi:10.1021/ol401143q
Return to citation in text: [1] -
Goodman, M. A.; Detty, M. R. Synlett 2006, 1100–1104. doi:10.1055/s-2006-939692
Return to citation in text: [1] [2] -
Yu, L.; Wu, Y.; Cao, H.; Zhang, X.; Shi, X.; Luan, J.; Chen, T.; Pan, Y.; Xu, Q. Green Chem. 2014, 16, 287–293. doi:10.1039/C3GC41562G
Return to citation in text: [1] [2] -
ten Brink, G.-J.; Vis, J. M.; Arends, I. W. C. E.; Sheldon, R. A. Tetrahedron 2002, 58, 3977–3983. doi:10.1016/S0040-4020(02)00248-X
Return to citation in text: [1] -
Ichikawa, H.; Usami, Y.; Arimoto, M. Tetrahedron Lett. 2005, 46, 8665–8668. doi:10.1016/j.tetlet.2005.10.055
Return to citation in text: [1] [2] -
Zhang, X.; Ye, J.; Yu, L.; Shi, X.; Zhang, M.; Xu, Q.; Lautens, M. Adv. Synth. Catal. 2015, 357, 955–960. doi:10.1002/adsc.201400957
Return to citation in text: [1] [2] -
Yu, L.; Bai, Z.; Zhang, X.; Zhang, X.; Ding, Y.; Xu, Q. Catal. Sci. Technol. 2016, 6, 1804–1809. doi:10.1039/C5CY01395J
Return to citation in text: [1] -
Del Todesco Frisone, M.; Giovanetti, R.; Pinna, F.; Strukul, G. Stud. Surf. Sci. Catal. 1991, 66, 405–410. doi:10.1016/S0167-2991(08)62858-X
Return to citation in text: [1] -
Bolm, C.; Schlingloff, G.; Weickhardt, K. Angew. Chem., Int. Ed. Engl. 1994, 33, 1848–1849. doi:10.1002/anie.199418481
Return to citation in text: [1] -
Bolm, C.; Schlingloff, G.; Bienewald, F. J. Mol. Catal. A: Chem. 1997, 117, 347–350. doi:10.1016/S1381-1169(96)00359-7
Return to citation in text: [1] -
Gusso, A.; Baccin, C.; Pinna, F.; Strukul, G. Organometallics 1994, 13, 3442–3451. doi:10.1021/om00021a019
Return to citation in text: [1] -
Strukul, G.; Varagnolo, A.; Pinna, F. J. Mol. Catal. A: Chem. 1997, 117, 413–423. doi:10.1016/S1381-1169(96)00246-4
Return to citation in text: [1] -
Lopp, M.; Paju, A.; Kanger, T.; Pehk, T. Tetrahedron Lett. 1996, 37, 7583–7586. doi:10.1016/0040-4039(96)01666-8
Return to citation in text: [1] -
Ito, K.; Ishii, A.; Kuroda, T.; Katsuki, T. Synlett 2003, 643–646. doi:10.1055/s-2003-38377
Return to citation in text: [1] -
Petersen, K. S.; Stoltz, B. M. Tetrahedron 2011, 67, 4352–4357. doi:10.1016/j.tet.2011.04.046
Return to citation in text: [1] -
Michelin, R. A.; Sgarbossa, P.; Scarso, A.; Strukul, G. Coord. Chem. Rev. 2010, 254, 646–660. doi:10.1016/j.ccr.2009.09.014
Return to citation in text: [1] -
Bernini, R.; Coratti, A.; Provenzano, G.; Fabrizi, G.; Tofani, D. Tetrahedron 2005, 61, 1821–1825. doi:10.1016/j.tet.2004.12.025
Return to citation in text: [1] -
Wang, Y.; Yokoi, T.; Otomo, R.; Kondo, J. N.; Tatsumi, T. Appl. Catal., A 2015, 490, 93–100. doi:10.1016/j.apcata.2014.11.011
Return to citation in text: [1] -
Luo, H. Y.; Bui, L.; Gunther, W. R.; Min, E.; Román-Leshkov, Y. ACS Catal. 2012, 2, 2695–2699. doi:10.1021/cs300543z
Return to citation in text: [1] -
Kang, Z.; Zhang, X.; Liu, H.; Qiu, J.; Yeung, K. L. Chem. Eng. J. 2013, 218, 425–432. doi:10.1016/j.cej.2012.12.019
Return to citation in text: [1] -
Paul, M.; Pal, N.; Mondal, J.; Sasidharan, M.; Bhaumik, A. Chem. Eng. Sci. 2012, 71, 564–572. doi:10.1016/j.ces.2011.11.038
Return to citation in text: [1] -
Nabae, Y.; Rokubuichi, H.; Mikuni, M.; Kuang, Y.; Hayakawa, T.; Kakimoto, M.-a. ACS Catal. 2013, 3, 230–236. doi:10.1021/cs3007928
Return to citation in text: [1] -
Uyanik, M.; Nakashima, D.; Ishihara, K. Angew. Chem. 2012, 124, 9227–9230. doi:10.1002/ange.201204286
Return to citation in text: [1] -
Uyanik, M.; Ishihara, K. ACS Catal. 2013, 3, 513–520. doi:10.1021/cs300821u
Return to citation in text: [1] -
Nicolaou, K. C.; Yu, R.; Shi, L.; Cai, Q.; Lu, M.; Heretsch, P. Org. Lett. 2013, 15, 1994–1997. doi:10.1021/ol4006689
Return to citation in text: [1] -
Hu, Y.-L.; Li, D.-J.; Li, D.-S. RSC Adv. 2015, 5, 24936–24943. doi:10.1039/C5RA02234G
Return to citation in text: [1] -
House, H. O.; Wasson, R. L. J. Org. Chem. 1957, 22, 1157–1160. doi:10.1021/jo01361a005
Return to citation in text: [1] -
Kaneda, K.; Ueno, S.; Imanaka, T. J. Mol. Catal. A: Chem. 1995, 102, 135–138. doi:10.1016/1381-1169(95)00055-0
Return to citation in text: [1] -
Kaneda, K.; Yamashita, T. Tetrahedron Lett. 1996, 37, 4555–4558. doi:10.1016/0040-4039(96)00902-1
Return to citation in text: [1] -
Pillai, U. R.; Sahle-Demessie, E. J. Mol. Catal. A: Chem. 2003, 191, 93–100. doi:10.1016/S1381-1169(02)00347-3
Return to citation in text: [1] -
Bradley, T. D.; Dragan, A.; Tomkinson, N. C. O. Tetrahedron 2015, 71, 8155–8161. doi:10.1016/j.tet.2015.08.037
Return to citation in text: [1] -
Ito, K. Asymmetric Baeyer–Villiger Oxidation. In Comprehensive Chirality; Carreira, E. M.; Yamamoto, H., Eds.; Elsevier: Amsterdam, 2012; pp 1–35. doi:10.1016/B978-0-08-095167-6.00502-4
Return to citation in text: [1] -
Bryliakov, K. P. Sustainable Asymmetric Oxidations. In Comprehensive Inorganic Chemistry, 2nd ed.; Poeppelmeier, J. R., Ed.; Elsevier: Amsterdam, 2013; pp 625–664. doi:10.1016/B978-0-08-097774-4.00624-0
Return to citation in text: [1] -
Leisch, H.; Morley, K.; Lau, P. C. K. Chem. Rev. 2011, 111, 4165–4222. doi:10.1021/cr1003437
Return to citation in text: [1] -
Mihovilovic, M. D.; Müller, B.; Stanetty, P. Eur. J. Org. Chem. 2002, 3711–3730. doi:10.1002/1099-0690(200211)2002:22<3711::AID-EJOC3711>3.0.CO;2-5
Return to citation in text: [1] -
Mihovilovic, M. D.; Rudroff, F.; Winninger, A.; Schneider, T.; Schulz, F.; Reetz, M. T. Org. Lett. 2006, 8, 1221–1224. doi:10.1021/ol0601040
Return to citation in text: [1] -
Reetz, M. T.; Brunner, B.; Schneider, T.; Schulz, F.; Clouthier, C. M.; Kayser, M. M. Angew. Chem., Int. Ed. 2004, 43, 4075–4078. doi:10.1002/anie.200460272
Return to citation in text: [1] -
Baldwin, C. V. F.; Wohlgemuth, R.; Woodley, J. M. Org. Process Res. Dev. 2008, 12, 660–665. doi:10.1021/op800046t
Return to citation in text: [1] -
Schallmey, A.; Domínguez de María, P.; Bracco, P. Biocatalytic Asymmetric Oxidations in Stereoselective Synthesis. In Stereoselective Synthesis of Drugs and Natural Products; Andrushko, V.; Andrushko, N., Eds.; John Wiley & Sons, Inc.: Hoboken, New Jersey, 2013; Vol. 2. doi:10.1002/9781118596784.ssd036
Return to citation in text: [1] -
Reile, I.; Paju, A.; Müürisepp, A.-M.; Pehk, T.; Lopp, M. Tetrahedron 2011, 67, 5942–5948. doi:10.1016/j.tet.2011.06.036
Return to citation in text: [1] [2] -
Jõgi, A.; Paju, A.; Pehk, T.; Kailas, T.; Müürisepp, A.-M.; Kanger, T.; Lopp, M. Synthesis 2006, 3031–3036. doi:10.1055/s-2006-950193
Return to citation in text: [1] -
Paju, A.; Laos, M.; Jõgi, A.; Päri, M.; Jäälaid, R.; Pehk, T.; Kanger, T.; Lopp, M. Tetrahedron Lett. 2006, 47, 4491–4493. doi:10.1016/j.tetlet.2006.04.013
Return to citation in text: [1] -
Crudden, C. M.; Chen, A. C.; Calhoun, L. A. Angew. Chem., Int. Ed. 2000, 39, 2851–2855. doi:10.1002/1521-3773(20000818)39:16<2851::AID-ANIE2851>3.0.CO;2-Y
Return to citation in text: [1] -
Xu, S.; Wang, Z.; Zhang, X.; Zhang, X.; Ding, K. Angew. Chem., Int. Ed. 2008, 47, 2840–2843. doi:10.1002/anie.200705932
Return to citation in text: [1] [2] -
Xu, S.; Wang, Z.; Li, Y.; Zhang, X.; Wang, H.; Ding, K. Chem. – Eur. J. 2010, 16, 3021–3035. doi:10.1002/chem.200902698
Return to citation in text: [1] [2] -
Xu, S.; Wang, Z.; Zhang, X.; Ding, K. Chin. J. Chem. 2010, 28, 1731–1735. doi:10.1002/cjoc.201090292
Return to citation in text: [1] -
Xu, S.; Wang, Z.; Zhang, X.; Ding, K. Eur. J. Org. Chem. 2011, 110–116. doi:10.1002/ejoc.201001130
Return to citation in text: [1] -
Mazzini, C.; Lebreton, J.; Furstoss, R. J. Org. Chem. 1996, 61, 8–9. doi:10.1021/jo951905b
Return to citation in text: [1] -
Murahashi, S.-I.; Ono, S.; Imada, Y. Angew. Chem., Int. Ed. 2002, 41, 2366–2368. doi:10.1002/1521-3773(20020703)41:13<2366::AID-ANIE2366>3.0.CO;2-S
Return to citation in text: [1] -
van Berkel, W. J. H.; Kamerbeek, N. M.; Fraaije, M. W. J. Biotechnol. 2006, 124, 670–689. doi:10.1016/j.jbiotec.2006.03.044
Return to citation in text: [1] -
Černuchová, P.; Mihovilovic, M. D. Org. Biomol. Chem. 2007, 5, 1715–1719. doi:10.1039/B703175K
Return to citation in text: [1] -
Schulz, F.; Leca, F.; Hollmann, F.; Reetz, M. T. Beilstein J. Org. Chem. 2005, 1, No. 10. doi:10.1186/1860-5397-1-10
Return to citation in text: [1] -
Zhou, L.; Liu, X.; Ji, J.; Zhang, Y.; Hu, X.; Lin, L.; Feng, X. J. Am. Chem. Soc. 2012, 134, 17023–17026. doi:10.1021/ja309262f
Return to citation in text: [1] -
Zhou, L.; Liu, X.; Ji, J.; Zhang, Y.; Wu, W.; Liu, Y.; Lin, L.; Feng, X. Org. Lett. 2014, 16, 3938–3941. doi:10.1021/ol501737a
Return to citation in text: [1] -
Romney, D. K.; Colvin, S. M.; Miller, S. J. J. Am. Chem. Soc. 2014, 136, 14019–14022. doi:10.1021/ja508757g
Return to citation in text: [1] -
Terent'ev, A. O.; Chodykin, S. V. Cent. Eur. J. Chem. 2005, 3, 417–431. doi:10.2478/BF02479272
Return to citation in text: [1] -
Putic, A.; Stecher, L.; Prinz, H.; Müller, K. Eur. J. Med. Chem. 2010, 45, 3299–3310. doi:10.1016/j.ejmech.2010.04.013
Return to citation in text: [1] -
An, G.-i.; Kim, M.; Kim, J. Y.; Rhee, H. Tetrahedron Lett. 2003, 44, 2183–2186. doi:10.1016/S0040-4039(03)00156-4
Return to citation in text: [1] -
Kao, J. P. Y.; Muralidharan, S.; Zavalij, P. Y.; Fletcher, S.; Xue, F.; Rosen, G. M. Tetrahedron Lett. 2014, 55, 3111–3113. doi:10.1016/j.tetlet.2014.04.004
Return to citation in text: [1] -
Wang, Z. Criegee Rearrangement. Comprehensive Organic Name Reactions and Reagents; John Wiley & Sons, 2010; pp 770–774. doi:10.1002/9780470638859.conrr170
Return to citation in text: [1] -
Criegee, R. Ber. Dtsch. Chem. Ges. A 1945, 77, 722–726. doi:10.1002/cber.19450770912
Return to citation in text: [1] -
Hedaya, E.; Winstein, S. Tetrahedron Lett. 1962, 3, 563–567. doi:10.1016/S0040-4039(00)76930-9
Return to citation in text: [1] -
Hedaya, E.; Winstein, S. J. Am. Chem. Soc. 1967, 89, 1661–1672. doi:10.1021/ja00983a023
Return to citation in text: [1] -
Wistuba, E.; Rüchardt, C. Tetrahedron Lett. 1981, 22, 3389–3392. doi:10.1016/S0040-4039(01)81913-4
Return to citation in text: [1] -
Schreiber, S. L.; Liew, W.-F. Tetrahedron Lett. 1983, 24, 2363–2366. doi:10.1016/S0040-4039(00)81926-7
Return to citation in text: [1] -
Goodman, R. M.; Kishi, Y. J. Org. Chem. 1994, 59, 5125–5127. doi:10.1021/jo00097a006
Return to citation in text: [1] -
Ogibin, Y. N.; Terent'ev, A. O.; Kutkin, A. V.; Nikishin, G. I. Tetrahedron Lett. 2002, 43, 1321–1324. doi:10.1016/S0040-4039(01)02368-1
Return to citation in text: [1] -
Baumstark, A. L.; Vasquez, P. C.; Chen, Y.-X. J. Org. Chem. 1994, 59, 6692–6696. doi:10.1021/jo00101a030
Return to citation in text: [1] -
Krasutsky, P. A.; Kolomitsyn, I. V.; Krasutsky, S. G.; Kiprof, P. Org. Lett. 2004, 6, 2539–2542. doi:10.1021/ol049171p
Return to citation in text: [1] [2] -
Krasutsky, P. A.; Kolomitsyn, I. V. ARKIVOC 2005, No. iv, 151–171.
Return to citation in text: [1] [2] -
Srikrishna, A.; Reddy, T. J. Tetrahedron 1998, 54, 11517–11524. doi:10.1016/S0040-4020(98)00672-3
Return to citation in text: [1] -
Srikrishna, A.; Kumar, P. P.; Reddy, T. J. Tetrahedron Lett. 1998, 39, 5815–5818. doi:10.1016/S0040-4039(98)01148-4
Return to citation in text: [1] -
Srikrishna, A.; Anebouselvy, K. J. Org. Chem. 2001, 66, 7102–7106. doi:10.1021/jo0105484
Return to citation in text: [1] [2] -
Srikrishna, A.; Gharpure, S. J.; Kumar, P. P. Tetrahedron Lett. 2000, 41, 3177–3180. doi:10.1016/S0040-4039(00)00325-7
Return to citation in text: [1] -
Srikrishna, A.; Anebouselvy, K.; Reddy, T. J. Tetrahedron Lett. 2000, 41, 6643–6647. doi:10.1016/S0040-4039(00)01107-2
Return to citation in text: [1] -
Srikrishna, A.; Anebouselvy, K. Tetrahedron Lett. 2002, 43, 2769–2771. doi:10.1016/S0040-4039(02)00382-9
Return to citation in text: [1] -
Srikrishna, A.; Mahesh, K. Synlett 2011, 2537–2540. doi:10.1055/s-0030-1260326
Return to citation in text: [1] -
Gándara, Z.; Rivadulla, M. L.; Pérez, M.; Gómez, G.; Fall, Y. Eur. J. Org. Chem. 2013, 5678–5682. doi:10.1002/ejoc.201300528
Return to citation in text: [1] -
Taatjes, C. A.; Shallcross, D. E.; Percival, C. J. Phys. Chem. Chem. Phys. 2014, 16, 1704–1718. doi:10.1039/c3cp52842a
Return to citation in text: [1] -
Chao, W.; Hsieh, J.-T.; Chang, C.-H.; Lin, J. J.-M. Science 2015, 347, 751–754. doi:10.1126/science.1261549
Return to citation in text: [1] -
Liu, F.; Beames, J. M.; Petit, A. S.; McCoy, A. B.; Lester, M. I. Science 2014, 345, 1596–1598. doi:10.1126/science.1257158
Return to citation in text: [1] -
Welz, O.; Savee, J. D.; Osborn, D. L.; Vasu, S. S.; Percival, C. J.; Shallcross, D. E.; Taatjes, C. A. Science 2012, 335, 204–207. doi:10.1126/science.1213229
Return to citation in text: [1] -
Wang, Z. Hock Rearrangement. Comprehensive Organic Name Reactions and Reagents; John Wiley & Sons, 2010; pp 1438–1442. doi:10.1002/9780470638859.conrr321
Return to citation in text: [1] -
Udris, R. J.; Sergeyev, P. G.; Kruzhalov, B. D. Sposob polucheniya gidroperekisejj alkilirovannykh-proizvodnykh benzola ili alicikloaromaticheskikh uglevodorodov. USSR Patent 106,666, Jan 7, 1947.
Return to citation in text: [1] -
Sergeyev, P. G.; Udris, R. J.; Kruzhalov, B. D.; Nyemtsov, B. D. Sposob odnovremennogo polucheniya fenola i acetona. USSR Patent 106,992, Jan 7, 1947.
Return to citation in text: [1] -
Hock, H.; Lang, S. Ber. Dtsch. Chem. Ges. A 1944, 77, 257–264. doi:10.1002/cber.19440770321
Return to citation in text: [1] -
Hock, H.; Kropf, H. Angew. Chem. 1957, 69, 313–321. doi:10.1002/ange.19570691002
Return to citation in text: [1] -
Deno, N. C.; Billups, W. E.; Kramer, K. E.; Lastomirsky, R. R. J. Org. Chem. 1970, 35, 3080–3082. doi:10.1021/jo00834a046
Return to citation in text: [1] -
Olah, G. A.; Parker, D. G.; Yoneda, N. Angew. Chem., Int. Ed. Engl. 1978, 17, 909–931. doi:10.1002/anie.197809091
Return to citation in text: [1] -
Lange, J.-P.; Breed, A. J. M. Catal. Commun. 2002, 3, 25–28. doi:10.1016/S1566-7367(01)00071-1
Return to citation in text: [1] -
Lillie, T. S.; Ronald, R. C. J. Org. Chem. 1985, 50, 5084–5088. doi:10.1021/jo00225a018
Return to citation in text: [1] -
Fisher, T. J.; Dussault, P. H. Tetrahedron Lett. 2010, 51, 5615–5617. doi:10.1016/j.tetlet.2010.08.068
Return to citation in text: [1] -
Anderson, G. H.; Smith, J. G. Can. J. Chem. 1968, 46, 1561–1570. doi:10.1139/v68-256
Return to citation in text: [1] -
Hamann, H.-J.; Liebscher, J. J. Org. Chem. 2000, 65, 1873–1876. doi:10.1021/jo991457y
Return to citation in text: [1] -
Hamann, H.-J.; Liebscher, J. Synlett 2001, 96–98. doi:10.1055/s-2001-9704
Return to citation in text: [1] -
Chan, Y.-Y.; Zhu, C.; Leung, H.-K. J. Am. Chem. Soc. 1985, 107, 5274–5275. doi:10.1021/ja00304a041
Return to citation in text: [1] -
Chan, Y.-Y.; Li, X.; Zhu, C.; Liu, X.; Zhang, Y.; Leung, H.-K. J. Org. Chem. 1990, 55, 5497–5504. doi:10.1021/jo00307a022
Return to citation in text: [1] -
Kabalka, G. W.; Reddy, N. K.; Narayana, C. Tetrahedron Lett. 1993, 34, 7667–7668. doi:10.1016/S0040-4039(00)61534-4
Return to citation in text: [1] -
Boger, D. L.; Coleman, R. S. J. Org. Chem. 1986, 51, 5436–5439. doi:10.1021/jo00376a079
Return to citation in text: [1] -
Zheng, X.; Lu, S.; Li, Z. Org. Lett. 2013, 15, 5432–5435. doi:10.1021/ol402509u
Return to citation in text: [1] [2] -
Zheng, X.; Lv, L.; Lu, S.; Wang, W.; Li, Z. Org. Lett. 2014, 16, 5156–5159. doi:10.1021/ol5025053
Return to citation in text: [1] -
Louillat-Habermeyer, M.-L.; Jin, R.; Patureau, F. W. Angew. Chem., Int. Ed. 2015, 54, 4102–4104. doi:10.1002/anie.201500089
Return to citation in text: [1] -
Boger, D. L.; Coleman, R. S. Tetrahedron Lett. 1987, 28, 1027–1030. doi:10.1016/S0040-4039(00)95902-1
Return to citation in text: [1] -
Amara, Z.; Bellamy, J. F. B.; Horvath, R.; Miller, S. J.; Beeby, A.; Burgard, A.; Rossen, K.; Poliakoff, M.; George, M. W. Nat. Chem. 2015, 7, 489–495. doi:10.1038/nchem.2261
Return to citation in text: [1] -
Wang, Z. Kornblum–Delamare Rearrangement. Comprehensive Organic Name Reactions and Reagents; John Wiley & Sons, Inc., 2010; pp 1675–1678. doi:10.1002/9780470638859.conrr374
Return to citation in text: [1] -
Kornblum, N.; DeLaMare, H. E. J. Am. Chem. Soc. 1951, 73, 880–881. doi:10.1021/ja01146a542
Return to citation in text: [1] -
Mete, E.; Altundaş, R.; Seçen, H.; Balci, M. Turk. J. Chem. 2003, 27, 145–153.
Return to citation in text: [1] -
Akbulut, N.; Balci, M. J. Org. Chem. 1988, 53, 3338–3342. doi:10.1021/jo00249a039
Return to citation in text: [1] -
Kelly, D. R.; Bansal, H.; Morgan, J. J. G. Tetrahedron Lett. 2002, 43, 9331–9333. doi:10.1016/S0040-4039(02)02374-2
Return to citation in text: [1] -
Avery, T. D.; Taylor, D. K.; Tiekink, E. R. T. J. Org. Chem. 2000, 65, 5531–5546. doi:10.1021/jo0002240
Return to citation in text: [1] -
Greatrex, B. W.; Kimber, M. C.; Taylor, D. K.; Tiekink, E. R. T. J. Org. Chem. 2003, 68, 4239–4246. doi:10.1021/jo020700h
Return to citation in text: [1] -
Greatrex, B. W.; Taylor, D. K.; Tiekink, E. R. T. J. Org. Chem. 2004, 69, 2580–2583. doi:10.1021/jo0303315
Return to citation in text: [1] -
Zhang, X.; Khan, S. I.; Foote, C. S. J. Org. Chem. 1993, 58, 7839–7847. doi:10.1021/jo00079a032
Return to citation in text: [1] -
Schulte-Elte, K. H.; Willhalm, B.; Ohloff, G. Angew. Chem. 1969, 81, 1045. doi:10.1002/ange.19690812406
Return to citation in text: [1] [2] -
Staben, S. T.; Linghu, X.; Toste, F. D. J. Am. Chem. Soc. 2006, 128, 12658–12659. doi:10.1021/ja065464x
Return to citation in text: [1] [2] [3] -
Oda, M.; Kitahara, Y. Tetrahedron Lett. 1969, 10, 3295–3296. doi:10.1016/S0040-4039(01)88413-6
Return to citation in text: [1] -
Daştan, A.; Balci, M. Tetrahedron 2006, 62, 4003–4010. doi:10.1016/j.tet.2006.02.026
Return to citation in text: [1] [2] [3] -
Koc, F.; Cadirci, E.; Albayrak, A.; Halici, Z.; Hacimuftuoglu, A.; Suleyman, H. Med. Chem. Res. 2010, 19, 84–93. doi:10.1007/s00044-009-9174-z
Return to citation in text: [1] -
Morita, Y.; Matsumura, E.; Okabe, T.; Shibata, M.; Sugiura, M.; Ohe, T.; Tsujibo, H.; Ishida, N.; Inamori, Y. Biol. Pharm. Bull. 2003, 26, 1487–1490. doi:10.1248/bpb.26.1487
Return to citation in text: [1] -
Balci, M.; Atasoy, B. Tetrahedron Lett. 1984, 25, 4033–4036. doi:10.1016/0040-4039(84)80059-3
Return to citation in text: [1] -
Atasoy, B.; Balci, M. Tetrahedron 1986, 42, 1461–1468. doi:10.1016/S0040-4020(01)87365-8
Return to citation in text: [1] [2] -
Sengül, M. E.; Ceylan, Z.; Balci, M. Tetrahedron 1997, 53, 10401–10408. doi:10.1016/S0040-4020(97)00630-3
Return to citation in text: [1] [2] -
Güney, M.; Ceylan, Z. C.; Daştan, A.; Balci, M. Can. J. Chem. 2005, 83, 227–235. doi:10.1139/v05-046
Return to citation in text: [1] [2] [3] -
Gu, X.; Zhang, W.; Salomon, R. G. J. Org. Chem. 2012, 77, 1554–1559. doi:10.1021/jo201910g
Return to citation in text: [1] -
Kawasumi, M.; Kanoh, N.; Iwabuchi, Y. Org. Lett. 2011, 13, 3620–3623. doi:10.1021/ol201273b
Return to citation in text: [1] -
Paddock, V. L.; Phipps, R. J.; Conde-Angulo, A.; Blanco-Martin, A.; Giró-Mañas, C.; Martin, L. J.; White, A. J. P.; Spivey, A. C. J. Org. Chem. 2011, 76, 1483–1486. doi:10.1021/jo102314w
Return to citation in text: [1] -
Fujishima, T.; Kitoh, F.; Yano, T.; Irie, R. Synlett 2010, 2279–2282. doi:10.1055/s-0030-1258028
Return to citation in text: [1] -
Erden, I.; Öcal, N.; Song, J.; Gleason, C.; Gärtner, C. Tetrahedron 2006, 62, 10676–10682. doi:10.1016/j.tet.2006.07.107
Return to citation in text: [1] -
Wasserman, H. H.; Terao, S. Tetrahedron Lett. 1975, 16, 1735–1738. doi:10.1016/S0040-4039(00)72246-5
Return to citation in text: [1] -
Frimer, A. A. Chem. Rev. 1979, 79, 359–387. doi:10.1021/cr60321a001
Return to citation in text: [1] -
Hewton, C. E.; Kimber, M. C.; Taylor, D. K. Tetrahedron Lett. 2002, 43, 3199–3201. doi:10.1016/S0040-4039(02)00503-8
Return to citation in text: [1] -
Rössle, M.; Werner, T.; Baro, A.; Frey, W.; Christoffers, J. Angew. Chem., Int. Ed. 2004, 43, 6547–6549. doi:10.1002/anie.200461406
Return to citation in text: [1] -
Zhang, F.; Du, P.; Chen, J.; Wang, H.; Luo, Q.; Wan, X. Org. Lett. 2014, 16, 1932–1935. doi:10.1021/ol5004687
Return to citation in text: [1] -
Greatrex, B. W.; Jenkins, N. F.; Taylor, D. K.; Tiekink, E. R. T. J. Org. Chem. 2003, 68, 5205–5210. doi:10.1021/jo0300845
Return to citation in text: [1] [2] -
Zagorski, M. G.; Salomon, R. G. J. Am. Chem. Soc. 1980, 102, 2501–2503. doi:10.1021/ja00527a081
Return to citation in text: [1] -
Zagorski, M. G.; Salomon, R. G. J. Am. Chem. Soc. 1984, 106, 1750–1759. doi:10.1021/ja00318a034
Return to citation in text: [1] -
Nicolaou, K. C.; Totokotsopoulos, S.; Giguère, D.; Sun, Y.-P.; Sarlah, D. J. Am. Chem. Soc. 2011, 133, 8150–8153. doi:10.1021/ja2032635
Return to citation in text: [1] -
Buchanan, G. S.; Cole, K. P.; Tang, Y.; Hsung, R. P. J. Org. Chem. 2011, 76, 7027–7039. doi:10.1021/jo200936r
Return to citation in text: [1] -
Kumar, R. A.; Maheswari, C. U.; Ghantasala, S.; Jyothi, C.; Reddy, K. R. Adv. Synth. Catal. 2011, 353, 401–410. doi:10.1002/adsc.201000580
Return to citation in text: [1] -
Tan, H.; Chen, X.; Liu, Z.; Wang, D. Z. Tetrahedron 2012, 68, 3952–3955. doi:10.1016/j.tet.2012.03.076
Return to citation in text: [1] -
Gesinski, M. R.; Brenzovich, W. E., Jr.; Staben, S. T.; Srinilta, D. J.; Toste, F. D. Tetrahedron Lett. 2015, 56, 3643–3646. doi:10.1016/j.tetlet.2015.02.067
Return to citation in text: [1] -
Wiegand, C.; Herdtweck, E.; Bach, T. Chem. Commun. 2012, 48, 10195–10197. doi:10.1039/c2cc35621j
Return to citation in text: [1] -
Palframan, M. J.; Kociok-Köhn, G.; Lewis, S. E. Chem. – Eur. J. 2012, 18, 4766–4774. doi:10.1002/chem.201104035
Return to citation in text: [1] -
Du, P.; Li, H.; Wang, Y.; Cheng, J.; Wan, X. Org. Lett. 2014, 16, 6350–6353. doi:10.1021/ol503128j
Return to citation in text: [1] -
Liu, C.; Shi, E.; Xu, F.; Luo, Q.; Wang, H.; Chen, J.; Wan, X. Chem. Commun. 2015, 51, 1214–1217. doi:10.1039/C4CC07833K
Return to citation in text: [1] -
Jiang, J.; Liu, J.; Yang, L.; Shao, Y.; Cheng, J.; Bao, X.; Wan, X. Chem. Commun. 2015, 51, 14728–14731. doi:10.1039/C5CC05183E
Return to citation in text: [1] -
Chen, I. T.; Baitinger, I.; Schreyer, L.; Trauner, D. Org. Lett. 2014, 16, 166–169. doi:10.1021/ol403156r
Return to citation in text: [1] -
Zhang, M.; Liu, N.; Tang, W. J. Am. Chem. Soc. 2013, 135, 12434–12438. doi:10.1021/ja406255j
Return to citation in text: [1] -
Priest, J.; Longland, M. R.; Elsegood, M. R. J.; Kimber, M. C. J. Org. Chem. 2013, 78, 3476–3481. doi:10.1021/jo400177j
Return to citation in text: [1] -
Hugelshofer, C. L.; Magauer, T. J. Am. Chem. Soc. 2015, 137, 3807–3810. doi:10.1021/jacs.5b02021
Return to citation in text: [1] -
Kalaitzakis, D.; Triantafyllakis, M.; Alexopoulou, I.; Sofiadis, M.; Vassilikogiannakis, G. Angew. Chem., Int. Ed. 2014, 53, 13201–13205. doi:10.1002/anie.201407477
Return to citation in text: [1] -
Vassilikogiannakis, G.; Stratakis, M. Angew. Chem., Int. Ed. 2003, 42, 5465–5468. doi:10.1002/anie.200352180
Return to citation in text: [1] -
Vassilikogiannakis, G.; Margaros, I.; Montagnon, T. Org. Lett. 2004, 6, 2039–2042. doi:10.1021/ol0493610
Return to citation in text: [1] -
Vassilikogiannakis, G.; Margaros, I.; Montagnon, T.; Stratakis, M. Chem. – Eur. J. 2005, 11, 5899–5907. doi:10.1002/chem.200401311
Return to citation in text: [1] -
Dakin, H. D. Am. Chem. J. 1909, 42, 477–498.
Return to citation in text: [1] [2] -
Wang, Z. Dakin Reaction. Comprehensive Organic Name Reactions and Reagents; John Wiley & Sons, 2010; pp 829–831. doi:10.1002/9780470638859.conrr175
Return to citation in text: [1] -
Hocking, M. B.; Bhandari, K.; Shell, B.; Smyth, T. A. J. Org. Chem. 1982, 47, 4208–4215. doi:10.1021/jo00143a007
Return to citation in text: [1] [2] [3] [4] -
Varma, R. S.; Naicker, K. P. Org. Lett. 1999, 1, 189–192. doi:10.1021/ol990522n
Return to citation in text: [1] -
da Silva, E. T.; Câmara, C. A.; Antunes, O. A. C.; Barreiro, E. J.; Fraga, C. A. M. Synth. Commun. 2008, 38, 784–788. doi:10.1080/00397910701820673
Return to citation in text: [1] -
Chen, S.; Hossain, M. S.; Foss, F. W., Jr. Org. Lett. 2012, 14, 2806–2809. doi:10.1021/ol3010326
Return to citation in text: [1] -
Chen, S.; Foss, F. W., Jr. Org. Lett. 2012, 14, 5150–5153. doi:10.1021/ol302479b
Return to citation in text: [1] -
Saikia, B.; Borah, P.; Barua, N. C. Green Chem. 2015, 17, 4533–4536. doi:10.1039/C5GC01404B
Return to citation in text: [1] -
Abe, T.; Itoh, T.; Choshi, T.; Hibino, S.; Ishikura, M. Tetrahedron Lett. 2014, 55, 5268–5270. doi:10.1016/j.tetlet.2014.07.113
Return to citation in text: [1] -
Jung, M. E.; Lazarova, T. I. J. Org. Chem. 1997, 62, 1553–1555. doi:10.1021/jo962099r
Return to citation in text: [1] -
Faure, E.; Falentin-Daudré, C.; Jérôme, C.; Lyskawa, J.; Fournier, D.; Woisel, P.; Detrembleur, C. Prog. Polym. Sci. 2013, 38, 236–270. doi:10.1016/j.progpolymsci.2012.06.004
Return to citation in text: [1] -
Fache, M.; Darroman, E.; Besse, V.; Auvergne, R.; Caillol, S.; Boutevin, B. Green Chem. 2014, 16, 1987–1998. doi:10.1039/c3gc42613k
Return to citation in text: [1] -
Fache, M.; Boutevin, B.; Caillol, S. Green Chem. 2016, 18, 712–725. doi:10.1039/C5GC01070E
Return to citation in text: [1] -
Matsumoto, M.; Kobayashi, H.; Hotta, Y. J. Org. Chem. 1984, 49, 4740–4741. doi:10.1021/jo00198a037
Return to citation in text: [1] -
Roy, A.; Reddy, K. R.; Mohanta, P. K.; Ila, H.; Junjappat, H. Synth. Commun. 1999, 29, 3781–3791. doi:10.1080/00397919908086017
Return to citation in text: [1] -
Elbs, K. J. Prakt. Chem. 1893, 48, 179–185. doi:10.1002/prac.18930480123
Return to citation in text: [1] -
Wang, Z. Elbs Persulfate Oxidation. Comprehensive Organic Name Reactions and Reagents; John Wiley & Sons, 2010; pp 977–981. doi:10.1002/9780470638859.conrr212
Return to citation in text: [1] -
Behrman, E. J. Beilstein J. Org. Chem. 2006, 2, No. 22. doi:10.1186/1860-5397-2-22
Return to citation in text: [1] -
Behrman, E. C.; Chen, S.; Behrman, E. J. Tetrahedron Lett. 2002, 43, 3221–3224. doi:10.1016/S0040-4039(02)00495-1
Return to citation in text: [1] -
Capdevielle, P.; Maumy, M. Tetrahedron Lett. 1982, 23, 1573–1576. doi:10.1016/S0040-4039(00)87161-0
Return to citation in text: [1] -
Sethna, S. M. Chem. Rev. 1951, 49, 91–101. doi:10.1021/cr60152a002
Return to citation in text: [1] -
Behrman, E. J. Synth. Commun. 2008, 38, 1168–1175. doi:10.1080/00397910701865819
Return to citation in text: [1] -
Behrman, E. J. Chem. Cent. J. 2009, 3, No. 1. doi:10.1186/1752-153X-3-1
Return to citation in text: [1] -
Behrman, E. J. J. Chem. Res. 2014, 38, 121–122. doi:10.3184/174751914X13896383516701
Return to citation in text: [1] -
Schenck, G. O.; Neumüller, O.-A.; Eisfeld, W. Justus Liebigs Ann. Chem. 1958, 618, 202–210. doi:10.1002/jlac.19586180123
Return to citation in text: [1] -
Schenck, G. O.; Neumüller, O.-A.; Eisfeld, W. Angew. Chem. 1958, 70, 595. doi:10.1002/ange.19580701906
Return to citation in text: [1] -
Eistert, B.; Bock, G. Angew. Chem. 1958, 70, 595. doi:10.1002/ange.19580701905
Return to citation in text: [1] -
Teng, J. I.; Kulig, M. J.; Smith, L. L.; Kan, G.; Van Lier, J. E. J. Org. Chem. 1973, 38, 119–123. doi:10.1021/jo00941a024
Return to citation in text: [1] -
Porter, N. A.; Dussault, P. H. Rearrangements of optically pure hydroperoxides. In Free Radicals in Synthesis and Biology; Minisci, F., Ed.; Kluwer Academic Publishers, 1989; pp 407–421. doi:10.1007/978-94-009-0897-0_30
Return to citation in text: [1] -
Brill, W. F. J. Chem. Soc., Perkin Trans. 2 1984, 621–627. doi:10.1039/p29840000621
Return to citation in text: [1] [2] [3] -
Porter, N.; Zuraw, P. J. Chem. Soc., Chem. Commun. 1985, 1472–1473. doi:10.1039/c39850001472
Return to citation in text: [1] -
Beckwith, A. L. J.; Davies, A. G.; Davison, I. G. E.; Maccoll, A.; Mruzek, M. H. J. Chem. Soc., Perkin Trans. 2 1989, 815–824. doi:10.1039/P29890000815
Return to citation in text: [1] [2] [3] -
Porter, N. A.; Wujek, J. S. J. Org. Chem. 1987, 52, 5085–5089. doi:10.1021/jo00232a004
Return to citation in text: [1] -
Beckwith, A. L. J.; Davies, A. G.; Davison, I. G. E.; Maccoll, A.; Mruzek, M. H. J. Chem. Soc., Chem. Commun. 1988, 475–476. doi:10.1039/c39880000475
Return to citation in text: [1] -
Davies, A. G.; Davison, I. G. E. J. Chem. Soc., Perkin Trans. 2 1989, 825–830. doi:10.1039/p29890000825
Return to citation in text: [1] -
Porter, N. A.; Kaplan, J. K.; Dussault, P. H. J. Am. Chem. Soc. 1990, 112, 1266–1267. doi:10.1021/ja00159a068
Return to citation in text: [1] -
Porter, N. A.; Mills, K. A.; Caldwell, S. E.; Dubay, G. R. J. Am. Chem. Soc. 1994, 116, 6697–6705. doi:10.1021/ja00094a027
Return to citation in text: [1] -
Mills, K. A.; Caldwell, S. E.; Dubay, G. R.; Porter, N. A. J. Am. Chem. Soc. 1992, 114, 9689–9691. doi:10.1021/ja00050a076
Return to citation in text: [1] -
Lowe, J. R.; Porter, N. A. J. Am. Chem. Soc. 1997, 119, 11534–11535. doi:10.1021/ja9723038
Return to citation in text: [1] -
Brill, W. F. J. Am. Chem. Soc. 1965, 87, 3286–3287. doi:10.1021/ja01092a077
Return to citation in text: [1] -
Dussault, P. H.; Eary, C. T.; Woller, K. R. J. Org. Chem. 1999, 64, 1789–1797. doi:10.1021/jo981128q
Return to citation in text: [1] -
Dang, H. S.; Davies, A. G.; Davison, I. G. E.; Schiesser, C. H. J. Org. Chem. 1990, 55, 1432–1438. doi:10.1021/jo00292a012
Return to citation in text: [1] -
Avila, D. V.; Davies, A. G.; Davison, I. G. E. J. Chem. Soc., Perkin Trans. 2 1988, 1847–1852. doi:10.1039/p29880001847
Return to citation in text: [1] [2] -
Dang, H.-S.; Davies, A. G. J. Chem. Soc., Perkin Trans. 2 1992, 1095–1101. doi:10.1039/P29920001095
Return to citation in text: [1] -
Dussault, P. H.; Woller, K. R. J. Am. Chem. Soc. 1997, 119, 3824–3825. doi:10.1021/ja970174p
Return to citation in text: [1] -
Nonami, Y.; Baran, J.; Sosnicki, J.; Mayr, H.; Masuyama, A.; Nojima, M. J. Org. Chem. 1999, 64, 4060–4063. doi:10.1021/jo990127a
Return to citation in text: [1] -
Bortolomeazzi, R.; De Zan, M.; Pizzale, L.; Conte, L. S. J. Agric. Food Chem. 1999, 47, 3069–3074. doi:10.1021/jf9812580
Return to citation in text: [1] [2] -
Sy, L.-K.; Ngo, K.-S.; Brown, G. D. Tetrahedron 1999, 55, 15127–15140. doi:10.1016/S0040-4020(99)00987-4
Return to citation in text: [1] -
Ngo, K.-S.; Brown, G. D. Tetrahedron 1999, 55, 14623–14634. doi:10.1016/S0040-4020(99)00938-2
Return to citation in text: [1] -
Ponce, M. A.; Ramirez, J. A.; Galagovsky, L. R.; Gros, E. G.; Erra-Balsells, R. J. Chem. Soc., Perkin Trans. 2 2000, 2351–2358. doi:10.1039/b000144i
Return to citation in text: [1] -
Kinart, W. J.; Kinart, A.; Sendecki, M. Phys. Chem. Liq. 2008, 46, 627–630. doi:10.1080/00319100802072656
Return to citation in text: [1] -
Wentworth, A. D.; Song, B.-D.; Nieva, J.; Shafton, A.; Tripurenani, S.; Wentworth, P., Jr. Chem. Commun. 2009, 3098–3100. doi:10.1039/B821584G
Return to citation in text: [1] -
Chen, D.; Chen, W.; Liu, D.; van Ofwegen, L.; Proksch, P.; Lin, W. J. Nat. Prod. 2013, 76, 1753–1763. doi:10.1021/np400480p
Return to citation in text: [1] -
Pratt, D. A.; Tallman, K. A.; Porter, N. A. Acc. Chem. Res. 2011, 44, 458–467. doi:10.1021/ar200024c
Return to citation in text: [1] -
Dang, H.-S.; Davies, A. G.; Schiesser, C. H. J. Chem. Soc., Perkin Trans. 1 1990, 3, 789–794. doi:10.1039/p19900000789
Return to citation in text: [1] [2] -
Courtneidge, J. L. J. Chem. Soc., Chem. Commun. 1992, 1270–1272. doi:10.1039/c39920001270
Return to citation in text: [1] -
Wieland, H. Ber. Dtsch. Chem. Ges. 1911, 44, 2550–2556. doi:10.1002/cber.19110440380
Return to citation in text: [1] -
Ingold, K. U.; Smeu, M.; DiLabio, G. A. J. Org. Chem. 2006, 71, 9906–9908. doi:10.1021/jo061898z
Return to citation in text: [1] -
DiLabio, G. A.; Ingold, K. U.; Lin, S.; Litwinienko, G.; Mozenson, O.; Mulder, P.; Tidwell, T. T. Angew. Chem., Int. Ed. 2010, 49, 5982–5985. doi:10.1002/anie.201001008
Return to citation in text: [1] -
Dannley, R. L.; Farrant, G. C. J. Org. Chem. 1969, 34, 2432–2437. doi:10.1021/jo01260a037
Return to citation in text: [1] -
Dannley, R. L.; Shubber, A. K. J. Org. Chem. 1971, 36, 3784–3787. doi:10.1021/jo00823a027
Return to citation in text: [1] -
Terent'ev, A. O.; Platonov, M. M.; Kashin, A. S.; Nikishin, G. I. Tetrahedron 2008, 64, 7944–7948. doi:10.1016/j.tet.2008.06.027
Return to citation in text: [1] -
Payne, G. B.; Smith, C. W. J. Org. Chem. 1957, 22, 1682–1685. doi:10.1021/jo01363a042
Return to citation in text: [1] -
Payne, G. J. Org. Chem. 1959, 24, 1830–1832. doi:10.1021/jo01093a633
Return to citation in text: [1] -
Terent'ev, A. O.; Borisov, D. A.; Yaremenko, I. A.; Ogibin, Y. N.; Nikishin, G. I. Synthesis 2010, 1145–1149. doi:10.1055/s-0029-1219225
Return to citation in text: [1] -
Yaremenko, I. A.; Terent'ev, A. O.; Vil', V. A.; Novikov, R. A.; Chernyshev, V. V.; Tafeenko, V. A.; Levitsky, D. O.; Fleury, F.; Nikishin, G. I. Chem. – Eur. J. 2014, 20, 10160–10169. doi:10.1002/chem.201402594
Return to citation in text: [1] -
Yu, X.; Liu, Z.; Xia, Z.; Shen, Z.; Pan, X.; Zhang, H.; Xie, W. RSC Adv. 2014, 4, 53397–53401. doi:10.1039/C4RA11237G
Return to citation in text: [1] -
Dussault, P. H.; Lee, H.-J.; Liu, X. J. Chem. Soc., Perkin Trans. 1 2000, 3006–3013. doi:10.1039/b001391i
Return to citation in text: [1] -
Murahashi, S.-I.; Naota, T.; Miyaguchi, N.; Noda, S. J. Am. Chem. Soc. 1996, 118, 2509–2510. doi:10.1021/ja954009q
Return to citation in text: [1] -
Murahashi, S.-I.; Miyaguchi, N.; Noda, S.; Naota, T.; Fujii, A.; Inubushi, Y.; Komiya, N. Eur. J. Org. Chem. 2011, 5355–5365. doi:10.1002/ejoc.201100740
Return to citation in text: [1] -
Mal, D.; Ray, S.; Sharma, I. J. Org. Chem. 2007, 72, 4981–4984. doi:10.1021/jo062271j
Return to citation in text: [1] -
Murahashi, S.-I.; Fujii, A.; Inubushi, Y.; Komiya, N. Tetrahedron Lett. 2010, 51, 2339–2341. doi:10.1016/j.tetlet.2010.02.134
Return to citation in text: [1] -
O'Neill, P. M.; Rawe, S. L.; Storr, R. C.; Ward, S. A.; Posner, G. H. Tetrahedron Lett. 2005, 46, 3029–3032. doi:10.1016/j.tetlet.2005.03.022
Return to citation in text: [1] -
Afonso, A. Can. J. Chem. 1970, 48, 691–693. doi:10.1139/v70-113
Return to citation in text: [1] -
Kulinkovich, O. G.; Astashko, D. A.; Tyvorskii, V. I.; Ilyina, N. A. Synthesis 2001, 1453–1455. doi:10.1055/s-2001-16089
Return to citation in text: [1] -
Reisinger, C. M.; Wang, X.; List, B. Angew. Chem., Int. Ed. 2008, 47, 8112–8115. doi:10.1002/anie.200803238
Return to citation in text: [1] -
O'Neill, P. M.; Searle, N. L.; Raynes, K. J.; Maggs, J. L.; Ward, S. A.; Storr, R. C.; Park, B. K.; Posner, G. H. Tetrahedron Lett. 1998, 39, 6065–6068. doi:10.1016/S0040-4039(98)01248-9
Return to citation in text: [1] -
Bernat, V.; André, C.; André-Barrès, C. Org. Biomol. Chem. 2008, 6, 454–457. doi:10.1039/b715491g
Return to citation in text: [1] -
Hoshiya, N.; Watanabe, N.; Ijuin, H. K.; Matsumoto, M. Tetrahedron 2006, 62, 12424–12437. doi:10.1016/j.tet.2006.09.108
Return to citation in text: [1] [2] -
Matsumoto, M.; Akimoto, T.; Matsumoto, Y.; Watanabe, N. Tetrahedron Lett. 2005, 46, 6075–6078. doi:10.1016/j.tetlet.2005.07.008
Return to citation in text: [1] [2] -
Matsumoto, M.; Tanimura, M.; Akimoto, T.; Watanabe, N.; Ijuin, H. K. Tetrahedron Lett. 2008, 49, 4170–4173. doi:10.1016/j.tetlet.2008.04.110
Return to citation in text: [1] [2] -
Tanimura, M.; Watanabe, N.; Ijuin, H. K.; Matsumoto, M. J. Org. Chem. 2011, 76, 902–908. doi:10.1021/jo1021822
Return to citation in text: [1] [2] -
Tanimura, M.; Watanabe, N.; Ijuin, H. K.; Matsumoto, M. J. Org. Chem. 2012, 77, 4725–4731. doi:10.1021/jo300417e
Return to citation in text: [1] [2] -
Matsumoto, M.; Suzuki, H.; Sano, Y.; Watanabe, N.; Ijuin, H. K. Tetrahedron Lett. 2008, 49, 5372–5375. doi:10.1016/j.tetlet.2008.06.103
Return to citation in text: [1] [2] -
Matsumoto, M.; Suzuki, H.; Watanabe, N.; Ijuin, H. K.; Tanaka, J.; Tanaka, C. J. Org. Chem. 2011, 76, 5006–5017. doi:10.1021/jo2006945
Return to citation in text: [1] [2] -
Matsumoto, M.; Takamido, Y.; Nomura, K.; Shiono, T.; Watanabe, N.; Ijuin, H. K. Tetrahedron Lett. 2008, 49, 6145–6147. doi:10.1016/j.tetlet.2008.08.024
Return to citation in text: [1] [2] -
Turan, I. S.; Akkaya, E. U. Org. Lett. 2014, 16, 1680–1683. doi:10.1021/ol5003412
Return to citation in text: [1] [2] -
Watanabe, N.; Kikuchi, M.; Maniwa, Y.; Ijuin, H. K.; Matsumoto, M. J. Org. Chem. 2010, 75, 879–884. doi:10.1021/jo902477n
Return to citation in text: [1] [2] -
Watanabe, N.; Sano, Y.; Suzuki, H.; Tanimura, M.; Ijuin, H. K.; Matsumoto, M. J. Org. Chem. 2010, 75, 5920–5926. doi:10.1021/jo101114b
Return to citation in text: [1] [2] -
Koyama, Y.; Watanabe, N.; Ijuin, H. K.; Matsumoto, M. Heterocycles 2015, 90, 462–481. doi:10.3987/COM-14-S(K)43
Return to citation in text: [1] [2] -
Hagiwara, H.; Watanabe, N.; Ijuin, H. K.; Yamada, M.; Matsumoto, M. Heterocycles 2013, 87, 65–78. doi:10.3987/COM-12-12602
Return to citation in text: [1] [2] -
Bastos, E. L.; da Silva, S. M.; Baader, W. J. J. Org. Chem. 2013, 78, 4432–4439. doi:10.1021/jo400426y
Return to citation in text: [1] -
Ciscato, L. F. M. L.; Bartoloni, F. H.; Colavite, A. S.; Weiss, D.; Beckert, R.; Schramm, S. Photochem. Photobiol. Sci. 2014, 13, 32–37. doi:10.1039/C3PP50345C
Return to citation in text: [1] -
Klaper, M.; Wessig, P.; Linker, T. Chem. Commun. 2016, 52, 1210–1213. doi:10.1039/C5CC08606J
Return to citation in text: [1] -
Pramanik, S.; Reddy, R. R.; Ghorai, P. Org. Lett. 2015, 17, 1393–1396. doi:10.1021/acs.orglett.5b00190
Return to citation in text: [1] -
Story, P. R.; Denson, D. D.; Bishop, C. E.; Clark, B. C., Jr.; Farine, J.-C. J. Am. Chem. Soc. 1968, 90, 817–818. doi:10.1021/ja01005a063
Return to citation in text: [1] -
Sanderson, J. R.; Story, P. R. J. Org. Chem. 1974, 39, 3463–3469. doi:10.1021/jo00938a001
Return to citation in text: [1] -
Sanderson, J. R.; Story, P. R.; Paul, K. J. Org. Chem. 1975, 40, 691–695. doi:10.1021/jo00894a006
Return to citation in text: [1] -
Paul, K.; Story, P. R.; Busch, P.; Sanderson, J. R. J. Org. Chem. 1976, 41, 1283–1285. doi:10.1021/jo00869a054
Return to citation in text: [1] -
Boyd, J. D.; Foote, C. S. J. Am. Chem. Soc. 1979, 101, 6758–6759. doi:10.1021/ja00516a051
Return to citation in text: [1] [2] -
Celik, M.; Akbulut, N.; Balci, M. Helv. Chim. Acta 2000, 83, 3131–3138. doi:10.1002/1522-2675(20001220)83:12<3131::AID-HLCA3131>3.0.CO;2-6
Return to citation in text: [1] -
van Tamelen, E. E.; Taylor, E. G. J. Am. Chem. Soc. 1980, 102, 1202–1203. doi:10.1021/ja00523a067
Return to citation in text: [1] -
Carless, H. A. J.; Atkins, R.; Fekarurhobo, G. K. Tetrahedron Lett. 1985, 26, 803–806. doi:10.1016/S0040-4039(00)89142-X
Return to citation in text: [1] -
Maheshwari, K. K.; de Mayo, P.; Wiegand, D. Can. J. Chem. 1970, 48, 3265–3268. doi:10.1139/v70-548
Return to citation in text: [1] -
Wilson, R. M.; Rekers, J. W. J. Am. Chem. Soc. 1981, 103, 206–207. doi:10.1021/ja00391a046
Return to citation in text: [1] -
Ronzani, F.; Costarramone, N.; Blanc, S.; Benabbou, A. K.; Le Bechec, M.; Pigot, T.; Oelgemöller, M.; Lacombe, S. J. Catal. 2013, 303, 164–174. doi:10.1016/j.jcat.2013.04.001
Return to citation in text: [1] -
Delort, E.; Jaquier, A.; Decorzant, E.; Chapuis, C.; Casilli, A.; Frérot, E. Phytochemistry 2015, 109, 111–124. doi:10.1016/j.phytochem.2014.10.023
Return to citation in text: [1] -
El-Mansy, M. F.; Flister, M.; Lindeman, S.; Kalous, K.; Sem, D. S.; Donaldson, W. A. Chem. – Eur. J. 2015, 21, 10886–10895. doi:10.1002/chem.201501274
Return to citation in text: [1] -
Erden, I.; Xu, F.-P.; Drummond, J.; Alstad, R. J. Org. Chem. 1993, 58, 3611–3612. doi:10.1021/jo00066a007
Return to citation in text: [1] -
Erden, I.; Xu, F.-P.; Cao, W.-G. Angew. Chem., Int. Ed. Engl. 1997, 36, 1516–1518. doi:10.1002/anie.199715161
Return to citation in text: [1] -
Cermola, F.; Iesce, M. R.; Buonerba, G. J. Org. Chem. 2005, 70, 6503–6505. doi:10.1021/jo0504159
Return to citation in text: [1] -
Scarpati, R.; Iesce, M. R.; Cermola, F.; Guitto, A. Synlett 1998, 17–25. doi:10.1055/s-1998-1556
Return to citation in text: [1] -
Iesce, M. R.; Cermola, F.; De Lorenzo, F.; Orabona, I.; Graziano, M. L. J. Org. Chem. 2001, 66, 4732–4735. doi:10.1021/jo0102112
Return to citation in text: [1] -
Matsumoto, M.; Murakami, H.; Watanabe, N. Chem. Commun. 1998, 2319–2320. doi:10.1039/a806823b
Return to citation in text: [1] -
Matsumoto, M.; Sano, Y.; Watanabe, N.; Ijuin, H. K. Chem. Lett. 2006, 35, 882–883. doi:10.1246/cl.2006.882
Return to citation in text: [1] -
Lee, A. S.; Shair, M. D. Org. Lett. 2013, 15, 2390–2393. doi:10.1021/ol400832r
Return to citation in text: [1] -
Ronsein, G. E.; de Oliveira, M. C. B.; de Medeiros, M. H. G.; Di Mascio, P. Photochem. Photobiol. Sci. 2011, 10, 1727–1730. doi:10.1039/c1pp05181d
Return to citation in text: [1] -
Posner, G. H.; Tao, X.; Cumming, J. N.; Klinedinst, D.; Shapiro, T. A. Tetrahedron Lett. 1996, 37, 7225–7228. doi:10.1016/0040-4039(96)01625-5
Return to citation in text: [1] -
Suzuki, M.; Ohtake, H.; Kameya, Y.; Hamanaka, N.; Noyori, R. J. Org. Chem. 1989, 54, 5292–5302. doi:10.1021/jo00283a023
Return to citation in text: [1] [2] -
Kamata, M.; Satoh, C.; Kim, H.-S.; Wataya, Y. Tetrahedron Lett. 2002, 43, 8313–8317. doi:10.1016/S0040-4039(02)02024-5
Return to citation in text: [1] -
Bloodworth, A. J.; Shah, A. Tetrahedron Lett. 1995, 36, 7551–7554. doi:10.1016/0040-4039(95)01530-2
Return to citation in text: [1] [2] -
Liu, H.-H.; Jin, H.-X.; Wu, Y.-K. Chin. J. Chem. 2004, 22, 1029–1033. doi:10.1002/cjoc.20040220930
Return to citation in text: [1] -
Persico, M.; Quintavalla, A.; Rondinelli, F.; Trombini, C.; Lombardo, M.; Fattorusso, C.; Azzarito, V.; Taramelli, D.; Parapini, S.; Corbett, Y.; Chianese, G.; Fattorusso, E.; Taglialatela-Scafati, O. J. Med. Chem. 2011, 54, 8526–8540. doi:10.1021/jm201056j
Return to citation in text: [1] -
Liu, H.-H.; Wu, Y.-K.; Shen, X. Chin. J. Chem. 2003, 21, 875–877. doi:10.1002/cjoc.20030210731
Return to citation in text: [1] -
Boyd, J. D.; Foote, C. S.; Imagawa, D. K. J. Am. Chem. Soc. 1980, 102, 3641–3642. doi:10.1021/ja00530a063
Return to citation in text: [1] -
Baran, A.; Bekarlar, M.; Aydin, G.; Nebioglu, M.; Sahin, E.; Balci, M. J. Org. Chem. 2012, 77, 1244–1250. doi:10.1021/jo202494v
Return to citation in text: [1] -
Baran, A.; Cambul, S.; Nebioglu, M.; Balci, M. J. Org. Chem. 2012, 77, 5086–5097. doi:10.1021/jo300655p
Return to citation in text: [1] -
Özen, R.; Kormali, F.; Balci, M.; Atasoy, B. Tetrahedron 2001, 57, 7529–7535. doi:10.1016/S0040-4020(01)00702-5
Return to citation in text: [1] -
Coşkun, A.; Güney, M.; Daştan, A.; Balci, M. Tetrahedron 2007, 63, 4944–4950. doi:10.1016/j.tet.2007.03.145
Return to citation in text: [1] -
Griffen, J. A.; Kenwright, S. J.; Abou-Shehada, S.; Wharry, S.; Moody, T. S.; Lewis, S. E. Org. Chem. Front. 2014, 1, 79–90. doi:10.1039/c3qo00057e
Return to citation in text: [1] -
Özer, G.; Saraçoglu, N.; Balci, M. J. Org. Chem. 2003, 68, 7009–7015. doi:10.1021/jo0345300
Return to citation in text: [1] -
Kiliç, H.; Balci, M. Tetrahedron 2001, 57, 9889–9897. doi:10.1016/S0040-4020(01)01008-0
Return to citation in text: [1] -
Dastan, A.; Saracoglu, N.; Balci, M. Eur. J. Org. Chem. 2001, 3519–3522. doi:10.1002/1099-0690(200109)2001:18<3519::AID-EJOC3519>3.0.CO;2-2
Return to citation in text: [1] -
Adam, W.; Balci, M.; Kiliç, H. J. Org. Chem. 2000, 65, 5926–5931. doi:10.1021/jo000120p
Return to citation in text: [1] -
Sutbeyaz, Y.; Seçen, H.; Balci, M. J. Org. Chem. 1988, 53, 2312–2317. doi:10.1021/jo00245a034
Return to citation in text: [1] -
Durie, A. J.; Slawin, A. M. Z.; Lebl, T.; Kirsch, P.; O'Hagan, D. Chem. Commun. 2011, 47, 8265–8267. doi:10.1039/c1cc13016a
Return to citation in text: [1] -
Sar, A.; Lindeman, S.; Donaldson, W. A. Org. Biomol. Chem. 2010, 8, 3908–3917. doi:10.1039/c004730a
Return to citation in text: [1] -
Barbarow, J. E.; Miller, A. K.; Trauner, D. Org. Lett. 2005, 7, 2901–2903. doi:10.1021/ol050831f
Return to citation in text: [1] -
Miller, A. K.; Trauner, D. Angew. Chem., Int. Ed. 2005, 44, 4602–4606. doi:10.1002/anie.200500488
Return to citation in text: [1] -
Suzuki, M.; Oda, Y.; Hamanaka, N.; Noyori, R. Heterocycles 1990, 30, 517–535. doi:10.3987/COM-89-S45
Return to citation in text: [1] -
Suzuki, M.; Oda, Y.; Noyori, R. Tetrahedron Lett. 1981, 22, 4413–4416. doi:10.1016/S0040-4039(01)82971-3
Return to citation in text: [1] -
The Research Group of Artemisinin Structure. Chin. Sci. Bull. 1977, 22, 142.
Return to citation in text: [1] -
The Qinghaosu Antimalarial Coordinating Research Group. Chin. Med. J. (Beijing, China, Engl. Ed.) 1979, 92, 811–816.
Return to citation in text: [1] -
Chaturvedi, D.; Goswami, A.; Saikia, P. P.; Barua, N. C.; Rao, P. G. Chem. Soc. Rev. 2010, 39, 435–454. doi:10.1039/B816679J
Return to citation in text: [1] -
Chen, H.-J.; Han, W.-B.; Hao, H.-D.; Wu, Y. Tetrahedron 2013, 69, 1112–1114. doi:10.1016/j.tet.2012.11.056
Return to citation in text: [1] -
Opsenica, D. M.; Solaja, B. A. Maced. J. Chem. Chem. Eng. 2012, 31, 137–182.
Return to citation in text: [1] -
Schmid, G.; Hofheinz, W. J. Am. Chem. Soc. 1983, 105, 624–625. doi:10.1021/ja00341a054
Return to citation in text: [1] -
Yadav, J. S.; Thirupathaiah, B.; Srihari, P. Tetrahedron 2010, 66, 2005–2009. doi:10.1016/j.tet.2010.01.051
Return to citation in text: [1] -
Zhu, C.; Cook, S. P. J. Am. Chem. Soc. 2012, 134, 13577–13579. doi:10.1021/ja3061479
Return to citation in text: [1] -
Dong, Y.; Wittlin, S.; Sriraghavan, K.; Chollet, J.; Charman, S. A.; Charman, W. N.; Scheurer, C.; Urwyler, H.; Santo Tomas, J.; Snyder, C.; Creek, D. J.; Morizzi, J.; Koltun, M.; Matile, H.; Wang, X.; Padmanilayam, M.; Tang, Y.; Dorn, A.; Brun, R.; Vennerstrom, J. L. J. Med. Chem. 2010, 53, 481–491. doi:10.1021/jm901473s
Return to citation in text: [1] -
Uhlemann, A.-C.; Wittlin, S.; Matile, H.; Bustamante, L. Y.; Krishna, S. Antimicrob. Agents Chemother. 2007, 51, 667–672. doi:10.1128/AAC.01064-06
Return to citation in text: [1] -
Vennerstrom, J. L.; Arbe-Barnes, S.; Brun, R.; Charman, S. A.; Chiu, F. C. K.; Chollet, J.; Dong, Y.; Dorn, A.; Hunziker, D.; Matile, H.; McIntosh, K.; Padmanilayam, M.; Santo Tomas, J.; Scheurer, C.; Scorneaux, B.; Tang, Y.; Urwyler, H.; Wittlin, S.; Charman, W. N. Nature 2004, 430, 900–904. doi:10.1038/nature02779
Return to citation in text: [1] -
O'Neill, P. M.; Amewu, R. K.; Nixon, G. L.; ElGarah, F. B.; Mungthin, M.; Chadwick, J.; Shone, A. E.; Vivas, L.; Lander, H.; Barton, V.; Muangnoicharoen, S.; Bray, P. G.; Davies, J.; Park, B. K.; Wittlin, S.; Brun, R.; Preschel, M.; Zhang, K.; Ward, S. A. Angew. Chem., Int. Ed. 2010, 49, 5693–5697. doi:10.1002/anie.201001026
Return to citation in text: [1] [2] -
Edikpo, N.; Ghasi, S.; Elias, A.; Oguanobi, N. Mol. Cell. Pharmacol. 2013, 5, 75–89.
Return to citation in text: [1] -
Klonis, N.; Creek, D. J.; Tilley, L. Curr. Opin. Microbiol. 2013, 16, 722–727. doi:10.1016/j.mib.2013.07.005
Return to citation in text: [1] -
Li, J.; Zhou, B. Molecules 2010, 15, 1378–1397. doi:10.3390/molecules15031378
Return to citation in text: [1] -
Robert, A.; Bonduelle, C.; Laurent, S. A.-L.; Meunier, B. J. Phys. Org. Chem. 2006, 19, 562–569. doi:10.1002/poc.1059
Return to citation in text: [1] -
Pereira, M. S. C.; Kiralj, R.; Ferreira, M. M. C. J. Chem. Inf. Model. 2008, 48, 85–98. doi:10.1021/ci700011f
Return to citation in text: [1] [2] -
Creek, D. J.; Chiu, F. C. K.; Prankerd, R. J.; Charman, S. A.; Charman, W. N. J. Pharm. Sci. 2005, 94, 1820–1829. doi:10.1002/jps.20400
Return to citation in text: [1] -
Bousejra-El Garah, F.; Meunier, B.; Robert, A. Eur. J. Inorg. Chem. 2008, 2133–2135. doi:10.1002/ejic.200800129
Return to citation in text: [1] -
Haynes, R. K.; Chan, W. C.; Lung, C.-M.; Uhlemann, A.-C.; Eckstein, U.; Taramelli, D.; Parapini, S.; Monti, D.; Krishna, S. ChemMedChem 2007, 2, 1480–1497. doi:10.1002/cmdc.200700108
Return to citation in text: [1] -
Moles, P.; Oliva, M.; Safont, V. S. J. Phys. Chem. B 2011, 115, 333–346. doi:10.1021/jp1064903
Return to citation in text: [1] -
Navacchia, M. L.; Capobianco, M. L.; D'Angelantonio, M.; Marconi, G. Tetrahedron Lett. 2012, 53, 1296–1299. doi:10.1016/j.tetlet.2012.01.007
Return to citation in text: [1] -
Nosoongnoen, W.; Pratuangdejkul, J.; Sathirakul, K.; Jacob, A.; Conti, M.; Loric, S.; Launay, J.-M.; Manivet, P. Phys. Chem. Chem. Phys. 2008, 10, 5083–5093. doi:10.1039/b804516j
Return to citation in text: [1] -
O'Neill, P. M.; Barton, V. E.; Ward, S. A. Molecules 2010, 15, 1705–1721. doi:10.3390/molecules15031705
Return to citation in text: [1] -
Robert, A.; Benoit-Vical, F.; Meunier, B. Coord. Chem. Rev. 2005, 249, 1927–1936. doi:10.1016/j.ccr.2004.12.022
Return to citation in text: [1] -
Tang, Y.; Dong, Y.; Wang, X.; Sriraghavan, K.; Wood, J. K.; Vennerstrom, J. L. J. Org. Chem. 2005, 70, 5103–5110. doi:10.1021/jo050385+
Return to citation in text: [1] -
Posner, G. H.; Cumming, J. N.; Ploypradith, P.; Oh, C. H. J. Am. Chem. Soc. 1995, 117, 5885–5886. doi:10.1021/ja00126a042
Return to citation in text: [1] -
Taglialatela-Scafati, O.; Fattorusso, E.; Romano, A.; Scala, F.; Barone, V.; Cimino, P.; Stendardo, E.; Catalanotti, B.; Persico, M.; Fattorusso, C. Org. Biomol. Chem. 2010, 8, 846–856. doi:10.1039/B918600J
Return to citation in text: [1] -
André-Barrès, C.; Najjar, F.; Bottalla, A.-L.; Massou, S.; Zedde, C.; Baltas, M.; Gorrichon, L. J. Org. Chem. 2005, 70, 6921–6924. doi:10.1021/jo050439f
Return to citation in text: [1] -
Najjar, F.; Gorrichon, L.; Baltas, M.; André-Barrès, C.; Vial, H. Org. Biomol. Chem. 2005, 3, 1612–1614. doi:10.1039/b503402g
Return to citation in text: [1] -
Bousejra-El Garah, F.; Wong, M. H.-L.; Amewu, R. K.; Muangnoicharoen, S.; Maggs, J. L.; Stigliani, J.-L.; Park, B. K.; Chadwick, J.; Ward, S. A.; O'Neill, P. M. J. Med. Chem. 2011, 54, 6443–6455. doi:10.1021/jm200768h
Return to citation in text: [1] [2] -
Creek, D. J.; Charman, W. N.; Chiu, F. C. K.; Prankerd, R. J.; Dong, Y.; Vennerstrom, J. L.; Charman, S. A. Antimicrob. Agents Chemother. 2008, 52, 1291–1296. doi:10.1128/AAC.01033-07
Return to citation in text: [1] -
Robert, A.; Coppel, Y.; Meunier, B. Inorg. Chim. Acta 2002, 339, 488–496. doi:10.1016/S0020-1693(02)00940-4
Return to citation in text: [1] -
Jourdan, J.; Matile, H.; Reift, E.; Biehlmaier, O.; Dong, Y.; Wang, X.; Mäser, P.; Vennerstrom, J. L.; Wittlin, S. ACS Infect. Dis. 2016, 2, 54–61. doi:10.1021/acsinfecdis.5b00090
Return to citation in text: [1] -
Kimura, K.-i.; Sakamoto, Y.; Fujisawa, N.; Uesugi, S.; Aburai, N.; Kawada, M.; Ohba, S.-i.; Yamori, T.; Tsuchiya, E.; Koshino, H. Bioorg. Med. Chem. 2012, 20, 3887–3897. doi:10.1016/j.bmc.2012.04.025
Return to citation in text: [1] -
Davies, K. J. A. IUBMB Life 2000, 50, 279–289. doi:10.1080/713803728
Return to citation in text: [1] -
Arai, H. Oxidative Modification of Lipoproteins. In Lipid Hydroperoxide-Derived Modification of Biomolecules; Kato, Y., Ed.; Springer: Netherlands, 2014; Vol. 77, pp 103–114. doi:10.1007/978-94-007-7920-4_9
Return to citation in text: [1] -
Mohd Fauzi, N.; Spickett, C. Lipid Oxidation. In Studies on Experimental Toxicology and Pharmacology; Roberts, S. M.; Kehrer, J. P.; Klotz, L.-O., Eds.; Springer International Publishing, 2015; pp 43–79. doi:10.1007/978-3-319-19096-9_4
Return to citation in text: [1] -
Cadet, J.; Di Mascio, P. Peroxides in Biological Systems. PATAI'S Chemistry of Functional Groups; John Wiley & Sons, Ltd., 2009. doi:10.1002/9780470682531.pat0357
Return to citation in text: [1] -
Jahn, U.; Galano, J.-M.; Durand, T. Angew. Chem., Int. Ed. 2008, 47, 5894–5955. doi:10.1002/anie.200705122
Return to citation in text: [1] -
Porter, N. A. J. Org. Chem. 2013, 78, 3511–3524. doi:10.1021/jo4001433
Return to citation in text: [1] [2] -
Ricciotti, E.; FitzGerald, G. A. Arterioscler., Thromb., Vasc. Biol. 2011, 31, 986–1000. doi:10.1161/ATVBAHA.110.207449
Return to citation in text: [1] -
Roberts, L. J.; Fessel, J. P. Chem. Phys. Lipids 2004, 128, 173–186. doi:10.1016/j.chemphyslip.2003.09.016
Return to citation in text: [1] -
Yin, H.; Havrilla, C. M.; Morrow, J. D.; Porter, N. A. J. Am. Chem. Soc. 2002, 124, 7745–7754. doi:10.1021/ja0201092
Return to citation in text: [1] -
Yin, H.; Xu, L.; Porter, N. A. Chem. Rev. 2011, 111, 5944–5972. doi:10.1021/cr200084z
Return to citation in text: [1] [2] -
Itri, R.; Junqueira, H. C.; Mertins, O.; Baptista, M. S. Biophys. Rev. 2014, 6, 47–61. doi:10.1007/s12551-013-0128-9
Return to citation in text: [1] -
Xu, L.; Porter, N. A. Free Radical Res. 2015, 49, 835–849. doi:10.3109/10715762.2014.985219
Return to citation in text: [1] -
Miyoshi, N.; Iuliano, L.; Tomono, S.; Ohshima, H. Biochem. Biophys. Res. Commun. 2014, 446, 702–708. doi:10.1016/j.bbrc.2013.12.107
Return to citation in text: [1] -
Costas, M.; Mehn, M. P.; Jensen, M. P.; Que, L., Jr. Chem. Rev. 2004, 104, 939–986. doi:10.1021/cr020628n
Return to citation in text: [1] -
Sainsbury, P. D.; Mineyeva, Y.; Mycroft, Z.; Bugg, T. D. H. Bioorg. Chem. 2015, 60, 102–109. doi:10.1016/j.bioorg.2015.05.002
Return to citation in text: [1] -
Guzik, U.; Hupert-Kocurek, K.; Wojcieszyńska, D. Intradiol Dioxygenases – The Key Enzymes in Xenobiotics Degradation. In Biodegradation of Hazardous and Special Products; Chamy, R.; Rosenkranz, F., Eds.; InTech, 2013. doi:10.5772/56205
Return to citation in text: [1] -
Komatsuzaki, H.; Shiota, A.; Hazawa, S.; Itoh, M.; Miyamura, N.; Miki, N.; Takano, Y.; Nakazawa, J.; Inagaki, A.; Akita, M.; Hikichi, S. Chem. – Asian J. 2013, 8, 1115–1119. doi:10.1002/asia.201300029
Return to citation in text: [1] -
Arnold, A.; Metzinger, R.; Limberg, C. Chem. – Eur. J. 2015, 21, 1198–1207. doi:10.1002/chem.201405155
Return to citation in text: [1] -
Poureskandari, M.; Safaei, E.; Sajjadi, S. M.; Karimpour, T.; Jaglicic, Z.; Lee, Y.-I. J. Mol. Struct. 2015, 1094, 130–136. doi:10.1016/j.molstruc.2015.04.008
Return to citation in text: [1] -
Borowski, T.; Siegbahn, P. E. M. J. Am. Chem. Soc. 2006, 128, 12941–12953. doi:10.1021/ja0641251
Return to citation in text: [1] -
Mendel, S.; Arndt, A.; Bugg, T. D. H. Biochemistry 2004, 43, 13390–13396. doi:10.1021/bi048518t
Return to citation in text: [1] -
Kovaleva, E. G.; Rogers, M. S.; Lipscomb, J. D. Biochemistry 2015, 54, 5329–5339. doi:10.1021/acs.biochem.5b00709
Return to citation in text: [1] -
Váradi, T.; Pap, J. S.; Giorgi, M.; Párkányi, L.; Csay, T.; Speier, G.; Kaizer, J. Inorg. Chem. 2013, 52, 1559–1569. doi:10.1021/ic302378r
Return to citation in text: [1] -
Xin, M.; Bugg, T. D. H. J. Am. Chem. Soc. 2008, 130, 10422–10430. doi:10.1021/ja8029569
Return to citation in text: [1] -
Harrison, P. J.; Bugg, T. D. H. Arch. Biochem. Biophys. 2014, 544, 105–111. doi:10.1016/j.abb.2013.10.005
Return to citation in text: [1] -
Harrison, P. J.; Newgas, S. A.; Descombes, F.; Shepherd, S. A.; Thompson, A. J.; Bugg, T. D. H. FEBS J. 2015, 282, 3986–4000. doi:10.1111/febs.13400
Return to citation in text: [1]
| 344. | Schulte-Elte, K. H.; Willhalm, B.; Ohloff, G. Angew. Chem. 1969, 81, 1045. doi:10.1002/ange.19690812406 |
| 203. | Fischer, J.; Hölderich, W. F. Appl. Catal., A 1999, 180, 435–443. doi:10.1016/S0926-860X(98)00378-0 |
| 205. | Jacobson, S. E.; Mares, F.; Zambri, P. M. J. Am. Chem. Soc. 1979, 101, 6938–6946. doi:10.1021/ja00517a025 |
| 196. | Strukul, G. Angew. Chem., Int. Ed. 1998, 37, 1198–1209. doi:10.1002/(SICI)1521-3773(19980518)37:9<1198::AID-ANIE1198>3.0.CO;2-Y |
| 214. | Jacobson, S. E.; Tang, R.; Mares, F. J. Chem. Soc., Chem. Commun. 1978, 888–889. doi:10.1039/c39780000888 |
| 236. | Zhang, X.; Ye, J.; Yu, L.; Shi, X.; Zhang, M.; Xu, Q.; Lautens, M. Adv. Synth. Catal. 2015, 357, 955–960. doi:10.1002/adsc.201400957 |
| 237. | Yu, L.; Bai, Z.; Zhang, X.; Zhang, X.; Ding, Y.; Xu, Q. Catal. Sci. Technol. 2016, 6, 1804–1809. doi:10.1039/C5CY01395J |
| 373. | Palframan, M. J.; Kociok-Köhn, G.; Lewis, S. E. Chem. – Eur. J. 2012, 18, 4766–4774. doi:10.1002/chem.201104035 |
| 233. | Yu, L.; Wu, Y.; Cao, H.; Zhang, X.; Shi, X.; Luan, J.; Chen, T.; Pan, Y.; Xu, Q. Green Chem. 2014, 16, 287–293. doi:10.1039/C3GC41562G |
| 372. | Wiegand, C.; Herdtweck, E.; Bach, T. Chem. Commun. 2012, 48, 10195–10197. doi:10.1039/c2cc35621j |
| 502. | Posner, G. H.; Tao, X.; Cumming, J. N.; Klinedinst, D.; Shapiro, T. A. Tetrahedron Lett. 1996, 37, 7225–7228. doi:10.1016/0040-4039(96)01625-5 |
| 369. | Kumar, R. A.; Maheswari, C. U.; Ghantasala, S.; Jyothi, C.; Reddy, K. R. Adv. Synth. Catal. 2011, 353, 401–410. doi:10.1002/adsc.201000580 |
| 498. | Matsumoto, M.; Murakami, H.; Watanabe, N. Chem. Commun. 1998, 2319–2320. doi:10.1039/a806823b |
| 499. | Matsumoto, M.; Sano, Y.; Watanabe, N.; Ijuin, H. K. Chem. Lett. 2006, 35, 882–883. doi:10.1246/cl.2006.882 |
| 368. | Buchanan, G. S.; Cole, K. P.; Tang, Y.; Hsung, R. P. J. Org. Chem. 2011, 76, 7027–7039. doi:10.1021/jo200936r |
| 497. | Iesce, M. R.; Cermola, F.; De Lorenzo, F.; Orabona, I.; Graziano, M. L. J. Org. Chem. 2001, 66, 4732–4735. doi:10.1021/jo0102112 |
| 248. | Wang, Y.; Yokoi, T.; Otomo, R.; Kondo, J. N.; Tatsumi, T. Appl. Catal., A 2015, 490, 93–100. doi:10.1016/j.apcata.2014.11.011 |
| 371. | Gesinski, M. R.; Brenzovich, W. E., Jr.; Staben, S. T.; Srinilta, D. J.; Toste, F. D. Tetrahedron Lett. 2015, 56, 3643–3646. doi:10.1016/j.tetlet.2015.02.067 |
| 501. | Ronsein, G. E.; de Oliveira, M. C. B.; de Medeiros, M. H. G.; Di Mascio, P. Photochem. Photobiol. Sci. 2011, 10, 1727–1730. doi:10.1039/c1pp05181d |
| 370. | Tan, H.; Chen, X.; Liu, Z.; Wang, D. Z. Tetrahedron 2012, 68, 3952–3955. doi:10.1016/j.tet.2012.03.076 |
| 500. | Lee, A. S.; Shair, M. D. Org. Lett. 2013, 15, 2390–2393. doi:10.1021/ol400832r |
| 196. | Strukul, G. Angew. Chem., Int. Ed. 1998, 37, 1198–1209. doi:10.1002/(SICI)1521-3773(19980518)37:9<1198::AID-ANIE1198>3.0.CO;2-Y |
| 239. | Bolm, C.; Schlingloff, G.; Weickhardt, K. Angew. Chem., Int. Ed. Engl. 1994, 33, 1848–1849. doi:10.1002/anie.199418481 |
| 240. | Bolm, C.; Schlingloff, G.; Bienewald, F. J. Mol. Catal. A: Chem. 1997, 117, 347–350. doi:10.1016/S1381-1169(96)00359-7 |
| 241. | Gusso, A.; Baccin, C.; Pinna, F.; Strukul, G. Organometallics 1994, 13, 3442–3451. doi:10.1021/om00021a019 |
| 242. | Strukul, G.; Varagnolo, A.; Pinna, F. J. Mol. Catal. A: Chem. 1997, 117, 413–423. doi:10.1016/S1381-1169(96)00246-4 |
| 243. | Lopp, M.; Paju, A.; Kanger, T.; Pehk, T. Tetrahedron Lett. 1996, 37, 7583–7586. doi:10.1016/0040-4039(96)01666-8 |
| 244. | Ito, K.; Ishii, A.; Kuroda, T.; Katsuki, T. Synlett 2003, 643–646. doi:10.1055/s-2003-38377 |
| 245. | Petersen, K. S.; Stoltz, B. M. Tetrahedron 2011, 67, 4352–4357. doi:10.1016/j.tet.2011.04.046 |
| 246. | Michelin, R. A.; Sgarbossa, P.; Scarso, A.; Strukul, G. Coord. Chem. Rev. 2010, 254, 646–660. doi:10.1016/j.ccr.2009.09.014 |
| 364. | Greatrex, B. W.; Jenkins, N. F.; Taylor, D. K.; Tiekink, E. R. T. J. Org. Chem. 2003, 68, 5205–5210. doi:10.1021/jo0300845 |
| 494. | Erden, I.; Xu, F.-P.; Cao, W.-G. Angew. Chem., Int. Ed. Engl. 1997, 36, 1516–1518. doi:10.1002/anie.199715161 |
| 247. | Bernini, R.; Coratti, A.; Provenzano, G.; Fabrizi, G.; Tofani, D. Tetrahedron 2005, 61, 1821–1825. doi:10.1016/j.tet.2004.12.025 |
| 330. | Zheng, X.; Lu, S.; Li, Z. Org. Lett. 2013, 15, 5432–5435. doi:10.1021/ol402509u |
| 363. | Zhang, F.; Du, P.; Chen, J.; Wang, H.; Luo, Q.; Wan, X. Org. Lett. 2014, 16, 1932–1935. doi:10.1021/ol5004687 |
| 493. | Erden, I.; Xu, F.-P.; Drummond, J.; Alstad, R. J. Org. Chem. 1993, 58, 3611–3612. doi:10.1021/jo00066a007 |
| 238. | Del Todesco Frisone, M.; Giovanetti, R.; Pinna, F.; Strukul, G. Stud. Surf. Sci. Catal. 1991, 66, 405–410. doi:10.1016/S0167-2991(08)62858-X |
| 367. | Nicolaou, K. C.; Totokotsopoulos, S.; Giguère, D.; Sun, Y.-P.; Sarlah, D. J. Am. Chem. Soc. 2011, 133, 8150–8153. doi:10.1021/ja2032635 |
| 496. | Scarpati, R.; Iesce, M. R.; Cermola, F.; Guitto, A. Synlett 1998, 17–25. doi:10.1055/s-1998-1556 |
| 208. | Gavagnin, R.; Cataldo, M.; Pinna, F.; Strukul, G. Organometallics 1998, 17, 661–667. doi:10.1021/om970756f |
| 365. | Zagorski, M. G.; Salomon, R. G. J. Am. Chem. Soc. 1980, 102, 2501–2503. doi:10.1021/ja00527a081 |
| 366. | Zagorski, M. G.; Salomon, R. G. J. Am. Chem. Soc. 1984, 106, 1750–1759. doi:10.1021/ja00318a034 |
| 495. | Cermola, F.; Iesce, M. R.; Buonerba, G. J. Org. Chem. 2005, 70, 6503–6505. doi:10.1021/jo0504159 |
| 374. | Du, P.; Li, H.; Wang, Y.; Cheng, J.; Wan, X. Org. Lett. 2014, 16, 6350–6353. doi:10.1021/ol503128j |
| 253. | Uyanik, M.; Nakashima, D.; Ishihara, K. Angew. Chem. 2012, 124, 9227–9230. doi:10.1002/ange.201204286 |
| 254. | Uyanik, M.; Ishihara, K. ACS Catal. 2013, 3, 513–520. doi:10.1021/cs300821u |
| 255. | Nicolaou, K. C.; Yu, R.; Shi, L.; Cai, Q.; Lu, M.; Heretsch, P. Org. Lett. 2013, 15, 1994–1997. doi:10.1021/ol4006689 |
| 251. | Paul, M.; Pal, N.; Mondal, J.; Sasidharan, M.; Bhaumik, A. Chem. Eng. Sci. 2012, 71, 564–572. doi:10.1016/j.ces.2011.11.038 |
| 252. | Nabae, Y.; Rokubuichi, H.; Mikuni, M.; Kuang, Y.; Hayakawa, T.; Kakimoto, M.-a. ACS Catal. 2013, 3, 230–236. doi:10.1021/cs3007928 |
| 249. | Luo, H. Y.; Bui, L.; Gunther, W. R.; Min, E.; Román-Leshkov, Y. ACS Catal. 2012, 2, 2695–2699. doi:10.1021/cs300543z |
| 250. | Kang, Z.; Zhang, X.; Liu, H.; Qiu, J.; Yeung, K. L. Chem. Eng. J. 2013, 218, 425–432. doi:10.1016/j.cej.2012.12.019 |
| 385. | Dakin, H. D. Am. Chem. J. 1909, 42, 477–498. |
| 387. | Hocking, M. B.; Bhandari, K.; Shell, B.; Smyth, T. A. J. Org. Chem. 1982, 47, 4208–4215. doi:10.1021/jo00143a007 |
| 380. | Hugelshofer, C. L.; Magauer, T. J. Am. Chem. Soc. 2015, 137, 3807–3810. doi:10.1021/jacs.5b02021 |
| 379. | Priest, J.; Longland, M. R.; Elsegood, M. R. J.; Kimber, M. C. J. Org. Chem. 2013, 78, 3476–3481. doi:10.1021/jo400177j |
| 385. | Dakin, H. D. Am. Chem. J. 1909, 42, 477–498. |
| 386. | Wang, Z. Dakin Reaction. Comprehensive Organic Name Reactions and Reagents; John Wiley & Sons, 2010; pp 829–831. doi:10.1002/9780470638859.conrr175 |
| 381. | Kalaitzakis, D.; Triantafyllakis, M.; Alexopoulou, I.; Sofiadis, M.; Vassilikogiannakis, G. Angew. Chem., Int. Ed. 2014, 53, 13201–13205. doi:10.1002/anie.201407477 |
| 382. | Vassilikogiannakis, G.; Stratakis, M. Angew. Chem., Int. Ed. 2003, 42, 5465–5468. doi:10.1002/anie.200352180 |
| 383. | Vassilikogiannakis, G.; Margaros, I.; Montagnon, T. Org. Lett. 2004, 6, 2039–2042. doi:10.1021/ol0493610 |
| 384. | Vassilikogiannakis, G.; Margaros, I.; Montagnon, T.; Stratakis, M. Chem. – Eur. J. 2005, 11, 5899–5907. doi:10.1002/chem.200401311 |
| 258. | Kaneda, K.; Ueno, S.; Imanaka, T. J. Mol. Catal. A: Chem. 1995, 102, 135–138. doi:10.1016/1381-1169(95)00055-0 |
| 259. | Kaneda, K.; Yamashita, T. Tetrahedron Lett. 1996, 37, 4555–4558. doi:10.1016/0040-4039(96)00902-1 |
| 260. | Pillai, U. R.; Sahle-Demessie, E. J. Mol. Catal. A: Chem. 2003, 191, 93–100. doi:10.1016/S1381-1169(02)00347-3 |
| 376. | Jiang, J.; Liu, J.; Yang, L.; Shao, Y.; Cheng, J.; Bao, X.; Wan, X. Chem. Commun. 2015, 51, 14728–14731. doi:10.1039/C5CC05183E |
| 261. | Bradley, T. D.; Dragan, A.; Tomkinson, N. C. O. Tetrahedron 2015, 71, 8155–8161. doi:10.1016/j.tet.2015.08.037 |
| 375. | Liu, C.; Shi, E.; Xu, F.; Luo, Q.; Wang, H.; Chen, J.; Wan, X. Chem. Commun. 2015, 51, 1214–1217. doi:10.1039/C4CC07833K |
| 256. | Hu, Y.-L.; Li, D.-J.; Li, D.-S. RSC Adv. 2015, 5, 24936–24943. doi:10.1039/C5RA02234G |
| 378. | Zhang, M.; Liu, N.; Tang, W. J. Am. Chem. Soc. 2013, 135, 12434–12438. doi:10.1021/ja406255j |
| 257. | House, H. O.; Wasson, R. L. J. Org. Chem. 1957, 22, 1157–1160. doi:10.1021/jo01361a005 |
| 377. | Chen, I. T.; Baitinger, I.; Schreyer, L.; Trauner, D. Org. Lett. 2014, 16, 166–169. doi:10.1021/ol403156r |
| 387. | Hocking, M. B.; Bhandari, K.; Shell, B.; Smyth, T. A. J. Org. Chem. 1982, 47, 4208–4215. doi:10.1021/jo00143a007 |
| 387. | Hocking, M. B.; Bhandari, K.; Shell, B.; Smyth, T. A. J. Org. Chem. 1982, 47, 4208–4215. doi:10.1021/jo00143a007 |
| 217. | McClure, J. D.; Williams, P. H. J. Org. Chem. 1962, 27, 24–26. doi:10.1021/jo01048a005 |
| 219. | Carlqvist, P.; Eklund, R.; Hult, K.; Brinck, T. J. Mol. Model. 2003, 9, 164–171. doi:10.1007/s00894-003-0128-y |
| 200. | ten Brink, G.-J.; Arends, I. W. C. E.; Sheldon, R. A. Chem. Rev. 2004, 104, 4105–4124. doi:10.1021/cr030011l |
| 218. | Macias-Alonso, M.; Morzycki, J. W.; Iglesias-Arteaga, M. A. Steroids 2011, 76, 317–323. doi:10.1016/j.steroids.2010.12.004 |
| 218. | Macias-Alonso, M.; Morzycki, J. W.; Iglesias-Arteaga, M. A. Steroids 2011, 76, 317–323. doi:10.1016/j.steroids.2010.12.004 |
| 225. | Enders, D.; Geibel, G.; Osborne, S. Chem. – Eur. J. 2000, 6, 1302–1309. doi:10.1002/(SICI)1521-3765(20000417)6:8<1302::AID-CHEM1302>3.0.CO;2-J |
| 226. | Iglesias-Arteaga, M. A.; Velazquez-Huerta, G. A.; Mendez-Stivalet, J. M.; Galano, A.; Alvarez-Idaboy, J. R. ARKIVOC 2006, No. vi, 109–126. |
| 392. | Saikia, B.; Borah, P.; Barua, N. C. Green Chem. 2015, 17, 4533–4536. doi:10.1039/C5GC01404B |
| 391. | Chen, S.; Foss, F. W., Jr. Org. Lett. 2012, 14, 5150–5153. doi:10.1021/ol302479b |
| 223. | Hamann, H.-J.; Bunge, A.; Liebscher, J. Chem. – Eur. J. 2008, 14, 6849–6851. doi:10.1002/chem.200800932 |
| 394. | Jung, M. E.; Lazarova, T. I. J. Org. Chem. 1997, 62, 1553–1555. doi:10.1021/jo962099r |
| 224. | Gordon, N. J.; Evans, S. A., Jr. J. Org. Chem. 1993, 58, 4516–4519. doi:10.1021/jo00069a004 |
| 393. | Abe, T.; Itoh, T.; Choshi, T.; Hibino, S.; Ishikura, M. Tetrahedron Lett. 2014, 55, 5268–5270. doi:10.1016/j.tetlet.2014.07.113 |
| 221. | Hitomi, Y.; Yoshida, H.; Tanaka, T.; Funabiki, T. J. Mol. Catal. A: Chem. 2006, 251, 239–245. doi:10.1016/j.molcata.2006.02.002 |
| 388. | Varma, R. S.; Naicker, K. P. Org. Lett. 1999, 1, 189–192. doi:10.1021/ol990522n |
| 223. | Hamann, H.-J.; Bunge, A.; Liebscher, J. Chem. – Eur. J. 2008, 14, 6849–6851. doi:10.1002/chem.200800932 |
| 387. | Hocking, M. B.; Bhandari, K.; Shell, B.; Smyth, T. A. J. Org. Chem. 1982, 47, 4208–4215. doi:10.1021/jo00143a007 |
| 221. | Hitomi, Y.; Yoshida, H.; Tanaka, T.; Funabiki, T. J. Mol. Catal. A: Chem. 2006, 251, 239–245. doi:10.1016/j.molcata.2006.02.002 |
| 390. | Chen, S.; Hossain, M. S.; Foss, F. W., Jr. Org. Lett. 2012, 14, 2806–2809. doi:10.1021/ol3010326 |
| 222. | Yamabe, S.; Yamazaki, S. J. Org. Chem. 2007, 72, 3031–3041. doi:10.1021/jo0626562 |
| 389. | da Silva, E. T.; Câmara, C. A.; Antunes, O. A. C.; Barreiro, E. J.; Fraga, C. A. M. Synth. Commun. 2008, 38, 784–788. doi:10.1080/00397910701820673 |
| 229. | Peris, G.; Miller, S. J. Org. Lett. 2008, 10, 3049–3052. doi:10.1021/ol8010248 |
| 230. | Patil, N. D.; Yao, S. G.; Meier, M. S.; Mobley, J. K.; Crocker, M. Org. Biomol. Chem. 2015, 13, 3243–3254. doi:10.1039/C4OB01771D |
| 227. | Zhou, G.; Corey, E. J. J. Am. Chem. Soc. 2005, 127, 11958–11959. doi:10.1021/ja054503m |
| 228. | Sharrna, R.; Nikas, S. P.; Guo, J. J.; Mallipeddi, S.; Wood, J. T.; Makriyannis, A. ACS Med. Chem. Lett. 2014, 5, 400–404. doi:10.1021/ml4005304 |
| 212. | ten Brink, G.-J.; Vis, J.-M.; Arends, I. W. C. E.; Sheldon, R. A. J. Org. Chem. 2001, 66, 2429–2433. doi:10.1021/jo0057710 |
| 235. | Ichikawa, H.; Usami, Y.; Arimoto, M. Tetrahedron Lett. 2005, 46, 8665–8668. doi:10.1016/j.tetlet.2005.10.055 |
| 234. | ten Brink, G.-J.; Vis, J. M.; Arends, I. W. C. E.; Sheldon, R. A. Tetrahedron 2002, 58, 3977–3983. doi:10.1016/S0040-4020(02)00248-X |
| 235. | Ichikawa, H.; Usami, Y.; Arimoto, M. Tetrahedron Lett. 2005, 46, 8665–8668. doi:10.1016/j.tetlet.2005.10.055 |
| 236. | Zhang, X.; Ye, J.; Yu, L.; Shi, X.; Zhang, M.; Xu, Q.; Lautens, M. Adv. Synth. Catal. 2015, 357, 955–960. doi:10.1002/adsc.201400957 |
| 232. | Goodman, M. A.; Detty, M. R. Synlett 2006, 1100–1104. doi:10.1055/s-2006-939692 |
| 231. | Poladura, B.; Martínez-Castañeda, Á.; Rodríguez-Solla, H.; Llavona, R.; Concellón, C.; del Amo, V. Org. Lett. 2013, 15, 2810–2813. doi:10.1021/ol401143q |
| 212. | ten Brink, G.-J.; Vis, J.-M.; Arends, I. W. C. E.; Sheldon, R. A. J. Org. Chem. 2001, 66, 2429–2433. doi:10.1021/jo0057710 |
| 232. | Goodman, M. A.; Detty, M. R. Synlett 2006, 1100–1104. doi:10.1055/s-2006-939692 |
| 233. | Yu, L.; Wu, Y.; Cao, H.; Zhang, X.; Shi, X.; Luan, J.; Chen, T.; Pan, Y.; Xu, Q. Green Chem. 2014, 16, 287–293. doi:10.1039/C3GC41562G |
| 332. | Louillat-Habermeyer, M.-L.; Jin, R.; Patureau, F. W. Angew. Chem., Int. Ed. 2015, 54, 4102–4104. doi:10.1002/anie.201500089 |
| 331. | Zheng, X.; Lv, L.; Lu, S.; Wang, W.; Li, Z. Org. Lett. 2014, 16, 5156–5159. doi:10.1021/ol5025053 |
| 455. | Mal, D.; Ray, S.; Sharma, I. J. Org. Chem. 2007, 72, 4981–4984. doi:10.1021/jo062271j |
| 456. | Murahashi, S.-I.; Fujii, A.; Inubushi, Y.; Komiya, N. Tetrahedron Lett. 2010, 51, 2339–2341. doi:10.1016/j.tetlet.2010.02.134 |
| 328. | Kabalka, G. W.; Reddy, N. K.; Narayana, C. Tetrahedron Lett. 1993, 34, 7667–7668. doi:10.1016/S0040-4039(00)61534-4 |
| 451. | Yu, X.; Liu, Z.; Xia, Z.; Shen, Z.; Pan, X.; Zhang, H.; Xie, W. RSC Adv. 2014, 4, 53397–53401. doi:10.1039/C4RA11237G |
| 326. | Chan, Y.-Y.; Zhu, C.; Leung, H.-K. J. Am. Chem. Soc. 1985, 107, 5274–5275. doi:10.1021/ja00304a041 |
| 327. | Chan, Y.-Y.; Li, X.; Zhu, C.; Liu, X.; Zhang, Y.; Leung, H.-K. J. Org. Chem. 1990, 55, 5497–5504. doi:10.1021/jo00307a022 |
| 450. | Yaremenko, I. A.; Terent'ev, A. O.; Vil', V. A.; Novikov, R. A.; Chernyshev, V. V.; Tafeenko, V. A.; Levitsky, D. O.; Fleury, F.; Nikishin, G. I. Chem. – Eur. J. 2014, 20, 10160–10169. doi:10.1002/chem.201402594 |
| 330. | Zheng, X.; Lu, S.; Li, Z. Org. Lett. 2013, 15, 5432–5435. doi:10.1021/ol402509u |
| 453. | Murahashi, S.-I.; Naota, T.; Miyaguchi, N.; Noda, S. J. Am. Chem. Soc. 1996, 118, 2509–2510. doi:10.1021/ja954009q |
| 454. | Murahashi, S.-I.; Miyaguchi, N.; Noda, S.; Naota, T.; Fujii, A.; Inubushi, Y.; Komiya, N. Eur. J. Org. Chem. 2011, 5355–5365. doi:10.1002/ejoc.201100740 |
| 329. | Boger, D. L.; Coleman, R. S. J. Org. Chem. 1986, 51, 5436–5439. doi:10.1021/jo00376a079 |
| 452. | Dussault, P. H.; Lee, H.-J.; Liu, X. J. Chem. Soc., Perkin Trans. 1 2000, 3006–3013. doi:10.1039/b001391i |
| 323. | Anderson, G. H.; Smith, J. G. Can. J. Chem. 1968, 46, 1561–1570. doi:10.1139/v68-256 |
| 447. | Payne, G. B.; Smith, C. W. J. Org. Chem. 1957, 22, 1682–1685. doi:10.1021/jo01363a042 |
| 322. | Fisher, T. J.; Dussault, P. H. Tetrahedron Lett. 2010, 51, 5615–5617. doi:10.1016/j.tetlet.2010.08.068 |
| 446. | Terent'ev, A. O.; Platonov, M. M.; Kashin, A. S.; Nikishin, G. I. Tetrahedron 2008, 64, 7944–7948. doi:10.1016/j.tet.2008.06.027 |
| 449. | Terent'ev, A. O.; Borisov, D. A.; Yaremenko, I. A.; Ogibin, Y. N.; Nikishin, G. I. Synthesis 2010, 1145–1149. doi:10.1055/s-0029-1219225 |
| 324. | Hamann, H.-J.; Liebscher, J. J. Org. Chem. 2000, 65, 1873–1876. doi:10.1021/jo991457y |
| 444. | Dannley, R. L.; Farrant, G. C. J. Org. Chem. 1969, 34, 2432–2437. doi:10.1021/jo01260a037 |
| 445. | Dannley, R. L.; Shubber, A. K. J. Org. Chem. 1971, 36, 3784–3787. doi:10.1021/jo00823a027 |
| 343. | Zhang, X.; Khan, S. I.; Foote, C. S. J. Org. Chem. 1993, 58, 7839–7847. doi:10.1021/jo00079a032 |
| 338. | Akbulut, N.; Balci, M. J. Org. Chem. 1988, 53, 3338–3342. doi:10.1021/jo00249a039 |
| 339. | Kelly, D. R.; Bansal, H.; Morgan, J. J. G. Tetrahedron Lett. 2002, 43, 9331–9333. doi:10.1016/S0040-4039(02)02374-2 |
| 464. | Matsumoto, M.; Akimoto, T.; Matsumoto, Y.; Watanabe, N. Tetrahedron Lett. 2005, 46, 6075–6078. doi:10.1016/j.tetlet.2005.07.008 |
| 336. | Kornblum, N.; DeLaMare, H. E. J. Am. Chem. Soc. 1951, 73, 880–881. doi:10.1021/ja01146a542 |
| 337. | Mete, E.; Altundaş, R.; Seçen, H.; Balci, M. Turk. J. Chem. 2003, 27, 145–153. |
| 463. | Hoshiya, N.; Watanabe, N.; Ijuin, H. K.; Matsumoto, M. Tetrahedron 2006, 62, 12424–12437. doi:10.1016/j.tet.2006.09.108 |
| 341. | Greatrex, B. W.; Kimber, M. C.; Taylor, D. K.; Tiekink, E. R. T. J. Org. Chem. 2003, 68, 4239–4246. doi:10.1021/jo020700h |
| 342. | Greatrex, B. W.; Taylor, D. K.; Tiekink, E. R. T. J. Org. Chem. 2004, 69, 2580–2583. doi:10.1021/jo0303315 |
| 466. | Tanimura, M.; Watanabe, N.; Ijuin, H. K.; Matsumoto, M. J. Org. Chem. 2011, 76, 902–908. doi:10.1021/jo1021822 |
| 340. | Avery, T. D.; Taylor, D. K.; Tiekink, E. R. T. J. Org. Chem. 2000, 65, 5531–5546. doi:10.1021/jo0002240 |
| 465. | Matsumoto, M.; Tanimura, M.; Akimoto, T.; Watanabe, N.; Ijuin, H. K. Tetrahedron Lett. 2008, 49, 4170–4173. doi:10.1016/j.tetlet.2008.04.110 |
| 66. | Takeuchi, C.; Galvé, R.; Nieva, J.; Witter, D. P.; Wentworth, A. D.; Troseth, R. P.; Lerner, R. A.; Wentworth, P., Jr. Biochemistry 2006, 45, 7162–7170. doi:10.1021/bi0604330 |
| 460. | Reisinger, C. M.; Wang, X.; List, B. Angew. Chem., Int. Ed. 2008, 47, 8112–8115. doi:10.1002/anie.200803238 |
| 67. | Brinkhorst, J.; Nara, S. J.; Pratt, D. A. J. Am. Chem. Soc. 2008, 130, 12224–12225. doi:10.1021/ja804162d |
| 459. | Kulinkovich, O. G.; Astashko, D. A.; Tyvorskii, V. I.; Ilyina, N. A. Synthesis 2001, 1453–1455. doi:10.1055/s-2001-16089 |
| 335. | Wang, Z. Kornblum–Delamare Rearrangement. Comprehensive Organic Name Reactions and Reagents; John Wiley & Sons, Inc., 2010; pp 1675–1678. doi:10.1002/9780470638859.conrr374 |
| 462. | Bernat, V.; André, C.; André-Barrès, C. Org. Biomol. Chem. 2008, 6, 454–457. doi:10.1039/b715491g |
| 463. | Hoshiya, N.; Watanabe, N.; Ijuin, H. K.; Matsumoto, M. Tetrahedron 2006, 62, 12424–12437. doi:10.1016/j.tet.2006.09.108 |
| 464. | Matsumoto, M.; Akimoto, T.; Matsumoto, Y.; Watanabe, N. Tetrahedron Lett. 2005, 46, 6075–6078. doi:10.1016/j.tetlet.2005.07.008 |
| 465. | Matsumoto, M.; Tanimura, M.; Akimoto, T.; Watanabe, N.; Ijuin, H. K. Tetrahedron Lett. 2008, 49, 4170–4173. doi:10.1016/j.tetlet.2008.04.110 |
| 466. | Tanimura, M.; Watanabe, N.; Ijuin, H. K.; Matsumoto, M. J. Org. Chem. 2011, 76, 902–908. doi:10.1021/jo1021822 |
| 467. | Tanimura, M.; Watanabe, N.; Ijuin, H. K.; Matsumoto, M. J. Org. Chem. 2012, 77, 4725–4731. doi:10.1021/jo300417e |
| 468. | Matsumoto, M.; Suzuki, H.; Sano, Y.; Watanabe, N.; Ijuin, H. K. Tetrahedron Lett. 2008, 49, 5372–5375. doi:10.1016/j.tetlet.2008.06.103 |
| 469. | Matsumoto, M.; Suzuki, H.; Watanabe, N.; Ijuin, H. K.; Tanaka, J.; Tanaka, C. J. Org. Chem. 2011, 76, 5006–5017. doi:10.1021/jo2006945 |
| 470. | Matsumoto, M.; Takamido, Y.; Nomura, K.; Shiono, T.; Watanabe, N.; Ijuin, H. K. Tetrahedron Lett. 2008, 49, 6145–6147. doi:10.1016/j.tetlet.2008.08.024 |
| 471. | Turan, I. S.; Akkaya, E. U. Org. Lett. 2014, 16, 1680–1683. doi:10.1021/ol5003412 |
| 472. | Watanabe, N.; Kikuchi, M.; Maniwa, Y.; Ijuin, H. K.; Matsumoto, M. J. Org. Chem. 2010, 75, 879–884. doi:10.1021/jo902477n |
| 473. | Watanabe, N.; Sano, Y.; Suzuki, H.; Tanimura, M.; Ijuin, H. K.; Matsumoto, M. J. Org. Chem. 2010, 75, 5920–5926. doi:10.1021/jo101114b |
| 474. | Koyama, Y.; Watanabe, N.; Ijuin, H. K.; Matsumoto, M. Heterocycles 2015, 90, 462–481. doi:10.3987/COM-14-S(K)43 |
| 475. | Hagiwara, H.; Watanabe, N.; Ijuin, H. K.; Yamada, M.; Matsumoto, M. Heterocycles 2013, 87, 65–78. doi:10.3987/COM-12-12602 |
| 334. | Amara, Z.; Bellamy, J. F. B.; Horvath, R.; Miller, S. J.; Beeby, A.; Burgard, A.; Rossen, K.; Poliakoff, M.; George, M. W. Nat. Chem. 2015, 7, 489–495. doi:10.1038/nchem.2261 |
| 461. | O'Neill, P. M.; Searle, N. L.; Raynes, K. J.; Maggs, J. L.; Ward, S. A.; Storr, R. C.; Park, B. K.; Posner, G. H. Tetrahedron Lett. 1998, 39, 6065–6068. doi:10.1016/S0040-4039(98)01248-9 |
| 333. | Boger, D. L.; Coleman, R. S. Tetrahedron Lett. 1987, 28, 1027–1030. doi:10.1016/S0040-4039(00)95902-1 |
| 457. | O'Neill, P. M.; Rawe, S. L.; Storr, R. C.; Ward, S. A.; Posner, G. H. Tetrahedron Lett. 2005, 46, 3029–3032. doi:10.1016/j.tetlet.2005.03.022 |
| 274. | Xu, S.; Wang, Z.; Zhang, X.; Zhang, X.; Ding, K. Angew. Chem., Int. Ed. 2008, 47, 2840–2843. doi:10.1002/anie.200705932 |
| 274. | Xu, S.; Wang, Z.; Zhang, X.; Zhang, X.; Ding, K. Angew. Chem., Int. Ed. 2008, 47, 2840–2843. doi:10.1002/anie.200705932 |
| 275. | Xu, S.; Wang, Z.; Li, Y.; Zhang, X.; Wang, H.; Ding, K. Chem. – Eur. J. 2010, 16, 3021–3035. doi:10.1002/chem.200902698 |
| 276. | Xu, S.; Wang, Z.; Zhang, X.; Ding, K. Chin. J. Chem. 2010, 28, 1731–1735. doi:10.1002/cjoc.201090292 |
| 277. | Xu, S.; Wang, Z.; Zhang, X.; Ding, K. Eur. J. Org. Chem. 2011, 110–116. doi:10.1002/ejoc.201001130 |
| 270. | Reile, I.; Paju, A.; Müürisepp, A.-M.; Pehk, T.; Lopp, M. Tetrahedron 2011, 67, 5942–5948. doi:10.1016/j.tet.2011.06.036 |
| 273. | Crudden, C. M.; Chen, A. C.; Calhoun, L. A. Angew. Chem., Int. Ed. 2000, 39, 2851–2855. doi:10.1002/1521-3773(20000818)39:16<2851::AID-ANIE2851>3.0.CO;2-Y |
| 264. | Leisch, H.; Morley, K.; Lau, P. C. K. Chem. Rev. 2011, 111, 4165–4222. doi:10.1021/cr1003437 |
| 265. | Mihovilovic, M. D.; Müller, B.; Stanetty, P. Eur. J. Org. Chem. 2002, 3711–3730. doi:10.1002/1099-0690(200211)2002:22<3711::AID-EJOC3711>3.0.CO;2-5 |
| 266. | Mihovilovic, M. D.; Rudroff, F.; Winninger, A.; Schneider, T.; Schulz, F.; Reetz, M. T. Org. Lett. 2006, 8, 1221–1224. doi:10.1021/ol0601040 |
| 267. | Reetz, M. T.; Brunner, B.; Schneider, T.; Schulz, F.; Clouthier, C. M.; Kayser, M. M. Angew. Chem., Int. Ed. 2004, 43, 4075–4078. doi:10.1002/anie.200460272 |
| 268. | Baldwin, C. V. F.; Wohlgemuth, R.; Woodley, J. M. Org. Process Res. Dev. 2008, 12, 660–665. doi:10.1021/op800046t |
| 269. | Schallmey, A.; Domínguez de María, P.; Bracco, P. Biocatalytic Asymmetric Oxidations in Stereoselective Synthesis. In Stereoselective Synthesis of Drugs and Natural Products; Andrushko, V.; Andrushko, N., Eds.; John Wiley & Sons, Inc.: Hoboken, New Jersey, 2013; Vol. 2. doi:10.1002/9781118596784.ssd036 |
| 351. | Atasoy, B.; Balci, M. Tetrahedron 1986, 42, 1461–1468. doi:10.1016/S0040-4020(01)87365-8 |
| 471. | Turan, I. S.; Akkaya, E. U. Org. Lett. 2014, 16, 1680–1683. doi:10.1021/ol5003412 |
| 270. | Reile, I.; Paju, A.; Müürisepp, A.-M.; Pehk, T.; Lopp, M. Tetrahedron 2011, 67, 5942–5948. doi:10.1016/j.tet.2011.06.036 |
| 271. | Jõgi, A.; Paju, A.; Pehk, T.; Kailas, T.; Müürisepp, A.-M.; Kanger, T.; Lopp, M. Synthesis 2006, 3031–3036. doi:10.1055/s-2006-950193 |
| 272. | Paju, A.; Laos, M.; Jõgi, A.; Päri, M.; Jäälaid, R.; Pehk, T.; Kanger, T.; Lopp, M. Tetrahedron Lett. 2006, 47, 4491–4493. doi:10.1016/j.tetlet.2006.04.013 |
| 350. | Balci, M.; Atasoy, B. Tetrahedron Lett. 1984, 25, 4033–4036. doi:10.1016/0040-4039(84)80059-3 |
| 351. | Atasoy, B.; Balci, M. Tetrahedron 1986, 42, 1461–1468. doi:10.1016/S0040-4020(01)87365-8 |
| 352. | Sengül, M. E.; Ceylan, Z.; Balci, M. Tetrahedron 1997, 53, 10401–10408. doi:10.1016/S0040-4020(97)00630-3 |
| 477. | Ciscato, L. F. M. L.; Bartoloni, F. H.; Colavite, A. S.; Weiss, D.; Beckert, R.; Schramm, S. Photochem. Photobiol. Sci. 2014, 13, 32–37. doi:10.1039/C3PP50345C |
| 352. | Sengül, M. E.; Ceylan, Z.; Balci, M. Tetrahedron 1997, 53, 10401–10408. doi:10.1016/S0040-4020(97)00630-3 |
| 262. | Ito, K. Asymmetric Baeyer–Villiger Oxidation. In Comprehensive Chirality; Carreira, E. M.; Yamamoto, H., Eds.; Elsevier: Amsterdam, 2012; pp 1–35. doi:10.1016/B978-0-08-095167-6.00502-4 |
| 263. | Bryliakov, K. P. Sustainable Asymmetric Oxidations. In Comprehensive Inorganic Chemistry, 2nd ed.; Poeppelmeier, J. R., Ed.; Elsevier: Amsterdam, 2013; pp 625–664. doi:10.1016/B978-0-08-097774-4.00624-0 |
| 353. | Güney, M.; Ceylan, Z. C.; Daştan, A.; Balci, M. Can. J. Chem. 2005, 83, 227–235. doi:10.1139/v05-046 |
| 478. | Klaper, M.; Wessig, P.; Linker, T. Chem. Commun. 2016, 52, 1210–1213. doi:10.1039/C5CC08606J |
| 347. | Daştan, A.; Balci, M. Tetrahedron 2006, 62, 4003–4010. doi:10.1016/j.tet.2006.02.026 |
| 475. | Hagiwara, H.; Watanabe, N.; Ijuin, H. K.; Yamada, M.; Matsumoto, M. Heterocycles 2013, 87, 65–78. doi:10.3987/COM-12-12602 |
| 346. | Oda, M.; Kitahara, Y. Tetrahedron Lett. 1969, 10, 3295–3296. doi:10.1016/S0040-4039(01)88413-6 |
| 474. | Koyama, Y.; Watanabe, N.; Ijuin, H. K.; Matsumoto, M. Heterocycles 2015, 90, 462–481. doi:10.3987/COM-14-S(K)43 |
| 348. | Koc, F.; Cadirci, E.; Albayrak, A.; Halici, Z.; Hacimuftuoglu, A.; Suleyman, H. Med. Chem. Res. 2010, 19, 84–93. doi:10.1007/s00044-009-9174-z |
| 349. | Morita, Y.; Matsumura, E.; Okabe, T.; Shibata, M.; Sugiura, M.; Ohe, T.; Tsujibo, H.; Ishida, N.; Inamori, Y. Biol. Pharm. Bull. 2003, 26, 1487–1490. doi:10.1248/bpb.26.1487 |
| 476. | Bastos, E. L.; da Silva, S. M.; Baader, W. J. J. Org. Chem. 2013, 78, 4432–4439. doi:10.1021/jo400426y |
| 347. | Daştan, A.; Balci, M. Tetrahedron 2006, 62, 4003–4010. doi:10.1016/j.tet.2006.02.026 |
| 470. | Matsumoto, M.; Takamido, Y.; Nomura, K.; Shiono, T.; Watanabe, N.; Ijuin, H. K. Tetrahedron Lett. 2008, 49, 6145–6147. doi:10.1016/j.tetlet.2008.08.024 |
| 275. | Xu, S.; Wang, Z.; Li, Y.; Zhang, X.; Wang, H.; Ding, K. Chem. – Eur. J. 2010, 16, 3021–3035. doi:10.1002/chem.200902698 |
| 467. | Tanimura, M.; Watanabe, N.; Ijuin, H. K.; Matsumoto, M. J. Org. Chem. 2012, 77, 4725–4731. doi:10.1021/jo300417e |
| 278. | Mazzini, C.; Lebreton, J.; Furstoss, R. J. Org. Chem. 1996, 61, 8–9. doi:10.1021/jo951905b |
| 279. | Murahashi, S.-I.; Ono, S.; Imada, Y. Angew. Chem., Int. Ed. 2002, 41, 2366–2368. doi:10.1002/1521-3773(20020703)41:13<2366::AID-ANIE2366>3.0.CO;2-S |
| 345. | Staben, S. T.; Linghu, X.; Toste, F. D. J. Am. Chem. Soc. 2006, 128, 12658–12659. doi:10.1021/ja065464x |
| 472. | Watanabe, N.; Kikuchi, M.; Maniwa, Y.; Ijuin, H. K.; Matsumoto, M. J. Org. Chem. 2010, 75, 879–884. doi:10.1021/jo902477n |
| 280. | van Berkel, W. J. H.; Kamerbeek, N. M.; Fraaije, M. W. J. Biotechnol. 2006, 124, 670–689. doi:10.1016/j.jbiotec.2006.03.044 |
| 281. | Černuchová, P.; Mihovilovic, M. D. Org. Biomol. Chem. 2007, 5, 1715–1719. doi:10.1039/B703175K |
| 282. | Schulz, F.; Leca, F.; Hollmann, F.; Reetz, M. T. Beilstein J. Org. Chem. 2005, 1, No. 10. doi:10.1186/1860-5397-1-10 |
| 344. | Schulte-Elte, K. H.; Willhalm, B.; Ohloff, G. Angew. Chem. 1969, 81, 1045. doi:10.1002/ange.19690812406 |
| 468. | Matsumoto, M.; Suzuki, H.; Sano, Y.; Watanabe, N.; Ijuin, H. K. Tetrahedron Lett. 2008, 49, 5372–5375. doi:10.1016/j.tetlet.2008.06.103 |
| 469. | Matsumoto, M.; Suzuki, H.; Watanabe, N.; Ijuin, H. K.; Tanaka, J.; Tanaka, C. J. Org. Chem. 2011, 76, 5006–5017. doi:10.1021/jo2006945 |
| 473. | Watanabe, N.; Sano, Y.; Suzuki, H.; Tanimura, M.; Ijuin, H. K.; Matsumoto, M. J. Org. Chem. 2010, 75, 5920–5926. doi:10.1021/jo101114b |
| 361. | Hewton, C. E.; Kimber, M. C.; Taylor, D. K. Tetrahedron Lett. 2002, 43, 3199–3201. doi:10.1016/S0040-4039(02)00503-8 |
| 347. | Daştan, A.; Balci, M. Tetrahedron 2006, 62, 4003–4010. doi:10.1016/j.tet.2006.02.026 |
| 359. | Wasserman, H. H.; Terao, S. Tetrahedron Lett. 1975, 16, 1735–1738. doi:10.1016/S0040-4039(00)72246-5 |
| 360. | Frimer, A. A. Chem. Rev. 1979, 79, 359–387. doi:10.1021/cr60321a001 |
| 492. | El-Mansy, M. F.; Flister, M.; Lindeman, S.; Kalous, K.; Sem, D. S.; Donaldson, W. A. Chem. – Eur. J. 2015, 21, 10886–10895. doi:10.1002/chem.201501274 |
| 362. | Rössle, M.; Werner, T.; Baro, A.; Frey, W.; Christoffers, J. Angew. Chem., Int. Ed. 2004, 43, 6547–6549. doi:10.1002/anie.200461406 |
| 356. | Paddock, V. L.; Phipps, R. J.; Conde-Angulo, A.; Blanco-Martin, A.; Giró-Mañas, C.; Martin, L. J.; White, A. J. P.; Spivey, A. C. J. Org. Chem. 2011, 76, 1483–1486. doi:10.1021/jo102314w |
| 484. | Boyd, J. D.; Foote, C. S. J. Am. Chem. Soc. 1979, 101, 6758–6759. doi:10.1021/ja00516a051 |
| 355. | Kawasumi, M.; Kanoh, N.; Iwabuchi, Y. Org. Lett. 2011, 13, 3620–3623. doi:10.1021/ol201273b |
| 487. | Carless, H. A. J.; Atkins, R.; Fekarurhobo, G. K. Tetrahedron Lett. 1985, 26, 803–806. doi:10.1016/S0040-4039(00)89142-X |
| 488. | Maheshwari, K. K.; de Mayo, P.; Wiegand, D. Can. J. Chem. 1970, 48, 3265–3268. doi:10.1139/v70-548 |
| 489. | Wilson, R. M.; Rekers, J. W. J. Am. Chem. Soc. 1981, 103, 206–207. doi:10.1021/ja00391a046 |
| 358. | Erden, I.; Öcal, N.; Song, J.; Gleason, C.; Gärtner, C. Tetrahedron 2006, 62, 10676–10682. doi:10.1016/j.tet.2006.07.107 |
| 491. | Delort, E.; Jaquier, A.; Decorzant, E.; Chapuis, C.; Casilli, A.; Frérot, E. Phytochemistry 2015, 109, 111–124. doi:10.1016/j.phytochem.2014.10.023 |
| 357. | Fujishima, T.; Kitoh, F.; Yano, T.; Irie, R. Synlett 2010, 2279–2282. doi:10.1055/s-0030-1258028 |
| 490. | Ronzani, F.; Costarramone, N.; Blanc, S.; Benabbou, A. K.; Le Bechec, M.; Pigot, T.; Oelgemöller, M.; Lacombe, S. J. Catal. 2013, 303, 164–174. doi:10.1016/j.jcat.2013.04.001 |
| 345. | Staben, S. T.; Linghu, X.; Toste, F. D. J. Am. Chem. Soc. 2006, 128, 12658–12659. doi:10.1021/ja065464x |
| 480. | Story, P. R.; Denson, D. D.; Bishop, C. E.; Clark, B. C., Jr.; Farine, J.-C. J. Am. Chem. Soc. 1968, 90, 817–818. doi:10.1021/ja01005a063 |
| 481. | Sanderson, J. R.; Story, P. R. J. Org. Chem. 1974, 39, 3463–3469. doi:10.1021/jo00938a001 |
| 482. | Sanderson, J. R.; Story, P. R.; Paul, K. J. Org. Chem. 1975, 40, 691–695. doi:10.1021/jo00894a006 |
| 483. | Paul, K.; Story, P. R.; Busch, P.; Sanderson, J. R. J. Org. Chem. 1976, 41, 1283–1285. doi:10.1021/jo00869a054 |
| 479. | Pramanik, S.; Reddy, R. R.; Ghorai, P. Org. Lett. 2015, 17, 1393–1396. doi:10.1021/acs.orglett.5b00190 |
| 345. | Staben, S. T.; Linghu, X.; Toste, F. D. J. Am. Chem. Soc. 2006, 128, 12658–12659. doi:10.1021/ja065464x |
| 486. | van Tamelen, E. E.; Taylor, E. G. J. Am. Chem. Soc. 1980, 102, 1202–1203. doi:10.1021/ja00523a067 |
| 354. | Gu, X.; Zhang, W.; Salomon, R. G. J. Org. Chem. 2012, 77, 1554–1559. doi:10.1021/jo201910g |
| 353. | Güney, M.; Ceylan, Z. C.; Daştan, A.; Balci, M. Can. J. Chem. 2005, 83, 227–235. doi:10.1139/v05-046 |
| 484. | Boyd, J. D.; Foote, C. S. J. Am. Chem. Soc. 1979, 101, 6758–6759. doi:10.1021/ja00516a051 |
| 485. | Celik, M.; Akbulut, N.; Balci, M. Helv. Chim. Acta 2000, 83, 3131–3138. doi:10.1002/1522-2675(20001220)83:12<3131::AID-HLCA3131>3.0.CO;2-6 |
| 406. | Behrman, E. J. Synth. Commun. 2008, 38, 1168–1175. doi:10.1080/00397910701865819 |
| 537. | O'Neill, P. M.; Amewu, R. K.; Nixon, G. L.; ElGarah, F. B.; Mungthin, M.; Chadwick, J.; Shone, A. E.; Vivas, L.; Lander, H.; Barton, V.; Muangnoicharoen, S.; Bray, P. G.; Davies, J.; Park, B. K.; Wittlin, S.; Brun, R.; Preschel, M.; Zhang, K.; Ward, S. A. Angew. Chem., Int. Ed. 2010, 49, 5693–5697. doi:10.1002/anie.201001026 |
| 414. | Brill, W. F. J. Chem. Soc., Perkin Trans. 2 1984, 621–627. doi:10.1039/p29840000621 |
| 164. | Hu, X.; Maimone, T. J. J. Am. Chem. Soc. 2014, 136, 5287–5290. doi:10.1021/ja502208z |
| 415. | Porter, N.; Zuraw, P. J. Chem. Soc., Chem. Commun. 1985, 1472–1473. doi:10.1039/c39850001472 |
| 554. | André-Barrès, C.; Najjar, F.; Bottalla, A.-L.; Massou, S.; Zedde, C.; Baltas, M.; Gorrichon, L. J. Org. Chem. 2005, 70, 6921–6924. doi:10.1021/jo050439f |
| 555. | Najjar, F.; Gorrichon, L.; Baltas, M.; André-Barrès, C.; Vial, H. Org. Biomol. Chem. 2005, 3, 1612–1614. doi:10.1039/b503402g |
| 417. | Porter, N. A.; Wujek, J. S. J. Org. Chem. 1987, 52, 5085–5089. doi:10.1021/jo00232a004 |
| 418. | Beckwith, A. L. J.; Davies, A. G.; Davison, I. G. E.; Maccoll, A.; Mruzek, M. H. J. Chem. Soc., Chem. Commun. 1988, 475–476. doi:10.1039/c39880000475 |
| 537. | O'Neill, P. M.; Amewu, R. K.; Nixon, G. L.; ElGarah, F. B.; Mungthin, M.; Chadwick, J.; Shone, A. E.; Vivas, L.; Lander, H.; Barton, V.; Muangnoicharoen, S.; Bray, P. G.; Davies, J.; Park, B. K.; Wittlin, S.; Brun, R.; Preschel, M.; Zhang, K.; Ward, S. A. Angew. Chem., Int. Ed. 2010, 49, 5693–5697. doi:10.1002/anie.201001026 |
| 556. | Bousejra-El Garah, F.; Wong, M. H.-L.; Amewu, R. K.; Muangnoicharoen, S.; Maggs, J. L.; Stigliani, J.-L.; Park, B. K.; Chadwick, J.; Ward, S. A.; O'Neill, P. M. J. Med. Chem. 2011, 54, 6443–6455. doi:10.1021/jm200768h |
| 416. | Beckwith, A. L. J.; Davies, A. G.; Davison, I. G. E.; Maccoll, A.; Mruzek, M. H. J. Chem. Soc., Perkin Trans. 2 1989, 815–824. doi:10.1039/P29890000815 |
| 556. | Bousejra-El Garah, F.; Wong, M. H.-L.; Amewu, R. K.; Muangnoicharoen, S.; Maggs, J. L.; Stigliani, J.-L.; Park, B. K.; Chadwick, J.; Ward, S. A.; O'Neill, P. M. J. Med. Chem. 2011, 54, 6443–6455. doi:10.1021/jm200768h |
| 412. | Teng, J. I.; Kulig, M. J.; Smith, L. L.; Kan, G.; Van Lier, J. E. J. Org. Chem. 1973, 38, 119–123. doi:10.1021/jo00941a024 |
| 542. | Pereira, M. S. C.; Kiralj, R.; Ferreira, M. M. C. J. Chem. Inf. Model. 2008, 48, 85–98. doi:10.1021/ci700011f |
| 543. | Creek, D. J.; Chiu, F. C. K.; Prankerd, R. J.; Charman, S. A.; Charman, W. N. J. Pharm. Sci. 2005, 94, 1820–1829. doi:10.1002/jps.20400 |
| 544. | Bousejra-El Garah, F.; Meunier, B.; Robert, A. Eur. J. Inorg. Chem. 2008, 2133–2135. doi:10.1002/ejic.200800129 |
| 545. | Haynes, R. K.; Chan, W. C.; Lung, C.-M.; Uhlemann, A.-C.; Eckstein, U.; Taramelli, D.; Parapini, S.; Monti, D.; Krishna, S. ChemMedChem 2007, 2, 1480–1497. doi:10.1002/cmdc.200700108 |
| 546. | Moles, P.; Oliva, M.; Safont, V. S. J. Phys. Chem. B 2011, 115, 333–346. doi:10.1021/jp1064903 |
| 547. | Navacchia, M. L.; Capobianco, M. L.; D'Angelantonio, M.; Marconi, G. Tetrahedron Lett. 2012, 53, 1296–1299. doi:10.1016/j.tetlet.2012.01.007 |
| 548. | Nosoongnoen, W.; Pratuangdejkul, J.; Sathirakul, K.; Jacob, A.; Conti, M.; Loric, S.; Launay, J.-M.; Manivet, P. Phys. Chem. Chem. Phys. 2008, 10, 5083–5093. doi:10.1039/b804516j |
| 549. | O'Neill, P. M.; Barton, V. E.; Ward, S. A. Molecules 2010, 15, 1705–1721. doi:10.3390/molecules15031705 |
| 550. | Robert, A.; Benoit-Vical, F.; Meunier, B. Coord. Chem. Rev. 2005, 249, 1927–1936. doi:10.1016/j.ccr.2004.12.022 |
| 551. | Tang, Y.; Dong, Y.; Wang, X.; Sriraghavan, K.; Wood, J. K.; Vennerstrom, J. L. J. Org. Chem. 2005, 70, 5103–5110. doi:10.1021/jo050385+ |
| 409. | Schenck, G. O.; Neumüller, O.-A.; Eisfeld, W. Justus Liebigs Ann. Chem. 1958, 618, 202–210. doi:10.1002/jlac.19586180123 |
| 410. | Schenck, G. O.; Neumüller, O.-A.; Eisfeld, W. Angew. Chem. 1958, 70, 595. doi:10.1002/ange.19580701906 |
| 411. | Eistert, B.; Bock, G. Angew. Chem. 1958, 70, 595. doi:10.1002/ange.19580701905 |
| 542. | Pereira, M. S. C.; Kiralj, R.; Ferreira, M. M. C. J. Chem. Inf. Model. 2008, 48, 85–98. doi:10.1021/ci700011f |
| 414. | Brill, W. F. J. Chem. Soc., Perkin Trans. 2 1984, 621–627. doi:10.1039/p29840000621 |
| 553. | Taglialatela-Scafati, O.; Fattorusso, E.; Romano, A.; Scala, F.; Barone, V.; Cimino, P.; Stendardo, E.; Catalanotti, B.; Persico, M.; Fattorusso, C. Org. Biomol. Chem. 2010, 8, 846–856. doi:10.1039/B918600J |
| 186. | Davies, A. G. J. Chem. Res. 2009, 533–544. doi:10.3184/030823409X12491375725131 |
| 413. | Porter, N. A.; Dussault, P. H. Rearrangements of optically pure hydroperoxides. In Free Radicals in Synthesis and Biology; Minisci, F., Ed.; Kluwer Academic Publishers, 1989; pp 407–421. doi:10.1007/978-94-009-0897-0_30 |
| 552. | Posner, G. H.; Cumming, J. N.; Ploypradith, P.; Oh, C. H. J. Am. Chem. Soc. 1995, 117, 5885–5886. doi:10.1021/ja00126a042 |
| 408. | Behrman, E. J. J. Chem. Res. 2014, 38, 121–122. doi:10.3184/174751914X13896383516701 |
| 540. | Li, J.; Zhou, B. Molecules 2010, 15, 1378–1397. doi:10.3390/molecules15031378 |
| 541. | Robert, A.; Bonduelle, C.; Laurent, S. A.-L.; Meunier, B. J. Phys. Org. Chem. 2006, 19, 562–569. doi:10.1002/poc.1059 |
| 538. | Edikpo, N.; Ghasi, S.; Elias, A.; Oguanobi, N. Mol. Cell. Pharmacol. 2013, 5, 75–89. |
| 539. | Klonis, N.; Creek, D. J.; Tilley, L. Curr. Opin. Microbiol. 2013, 16, 722–727. doi:10.1016/j.mib.2013.07.005 |
| 416. | Beckwith, A. L. J.; Davies, A. G.; Davison, I. G. E.; Maccoll, A.; Mruzek, M. H. J. Chem. Soc., Perkin Trans. 2 1989, 815–824. doi:10.1039/P29890000815 |
| 289. | Kao, J. P. Y.; Muralidharan, S.; Zavalij, P. Y.; Fletcher, S.; Xue, F.; Rosen, G. M. Tetrahedron Lett. 2014, 55, 3111–3113. doi:10.1016/j.tetlet.2014.04.004 |
| 426. | Dang, H. S.; Davies, A. G.; Davison, I. G. E.; Schiesser, C. H. J. Org. Chem. 1990, 55, 1432–1438. doi:10.1021/jo00292a012 |
| 288. | An, G.-i.; Kim, M.; Kim, J. Y.; Rhee, H. Tetrahedron Lett. 2003, 44, 2183–2186. doi:10.1016/S0040-4039(03)00156-4 |
| 425. | Dussault, P. H.; Eary, C. T.; Woller, K. R. J. Org. Chem. 1999, 64, 1789–1797. doi:10.1021/jo981128q |
| 568. | Roberts, L. J.; Fessel, J. P. Chem. Phys. Lipids 2004, 128, 173–186. doi:10.1016/j.chemphyslip.2003.09.016 |
| 569. | Yin, H.; Havrilla, C. M.; Morrow, J. D.; Porter, N. A. J. Am. Chem. Soc. 2002, 124, 7745–7754. doi:10.1021/ja0201092 |
| 570. | Yin, H.; Xu, L.; Porter, N. A. Chem. Rev. 2011, 111, 5944–5972. doi:10.1021/cr200084z |
| 291. | Criegee, R. Ber. Dtsch. Chem. Ges. A 1945, 77, 722–726. doi:10.1002/cber.19450770912 |
| 428. | Dang, H.-S.; Davies, A. G. J. Chem. Soc., Perkin Trans. 2 1992, 1095–1101. doi:10.1039/P29920001095 |
| 290. | Wang, Z. Criegee Rearrangement. Comprehensive Organic Name Reactions and Reagents; John Wiley & Sons, 2010; pp 770–774. doi:10.1002/9780470638859.conrr170 |
| 427. | Avila, D. V.; Davies, A. G.; Davison, I. G. E. J. Chem. Soc., Perkin Trans. 2 1988, 1847–1852. doi:10.1039/p29880001847 |
| 570. | Yin, H.; Xu, L.; Porter, N. A. Chem. Rev. 2011, 111, 5944–5972. doi:10.1021/cr200084z |
| 285. | Romney, D. K.; Colvin, S. M.; Miller, S. J. J. Am. Chem. Soc. 2014, 136, 14019–14022. doi:10.1021/ja508757g |
| 423. | Lowe, J. R.; Porter, N. A. J. Am. Chem. Soc. 1997, 119, 11534–11535. doi:10.1021/ja9723038 |
| 284. | Zhou, L.; Liu, X.; Ji, J.; Zhang, Y.; Wu, W.; Liu, Y.; Lin, L.; Feng, X. Org. Lett. 2014, 16, 3938–3941. doi:10.1021/ol501737a |
| 421. | Porter, N. A.; Mills, K. A.; Caldwell, S. E.; Dubay, G. R. J. Am. Chem. Soc. 1994, 116, 6697–6705. doi:10.1021/ja00094a027 |
| 422. | Mills, K. A.; Caldwell, S. E.; Dubay, G. R.; Porter, N. A. J. Am. Chem. Soc. 1992, 114, 9689–9691. doi:10.1021/ja00050a076 |
| 560. | Kimura, K.-i.; Sakamoto, Y.; Fujisawa, N.; Uesugi, S.; Aburai, N.; Kawada, M.; Ohba, S.-i.; Yamori, T.; Tsuchiya, E.; Koshino, H. Bioorg. Med. Chem. 2012, 20, 3887–3897. doi:10.1016/j.bmc.2012.04.025 |
| 287. | Putic, A.; Stecher, L.; Prinz, H.; Müller, K. Eur. J. Med. Chem. 2010, 45, 3299–3310. doi:10.1016/j.ejmech.2010.04.013 |
| 414. | Brill, W. F. J. Chem. Soc., Perkin Trans. 2 1984, 621–627. doi:10.1039/p29840000621 |
| 564. | Cadet, J.; Di Mascio, P. Peroxides in Biological Systems. PATAI'S Chemistry of Functional Groups; John Wiley & Sons, Ltd., 2009. doi:10.1002/9780470682531.pat0357 |
| 565. | Jahn, U.; Galano, J.-M.; Durand, T. Angew. Chem., Int. Ed. 2008, 47, 5894–5955. doi:10.1002/anie.200705122 |
| 566. | Porter, N. A. J. Org. Chem. 2013, 78, 3511–3524. doi:10.1021/jo4001433 |
| 567. | Ricciotti, E.; FitzGerald, G. A. Arterioscler., Thromb., Vasc. Biol. 2011, 31, 986–1000. doi:10.1161/ATVBAHA.110.207449 |
| 286. | Terent'ev, A. O.; Chodykin, S. V. Cent. Eur. J. Chem. 2005, 3, 417–431. doi:10.2478/BF02479272 |
| 424. | Brill, W. F. J. Am. Chem. Soc. 1965, 87, 3286–3287. doi:10.1021/ja01092a077 |
| 562. | Arai, H. Oxidative Modification of Lipoproteins. In Lipid Hydroperoxide-Derived Modification of Biomolecules; Kato, Y., Ed.; Springer: Netherlands, 2014; Vol. 77, pp 103–114. doi:10.1007/978-94-007-7920-4_9 |
| 563. | Mohd Fauzi, N.; Spickett, C. Lipid Oxidation. In Studies on Experimental Toxicology and Pharmacology; Roberts, S. M.; Kehrer, J. P.; Klotz, L.-O., Eds.; Springer International Publishing, 2015; pp 43–79. doi:10.1007/978-3-319-19096-9_4 |
| 557. | Creek, D. J.; Charman, W. N.; Chiu, F. C. K.; Prankerd, R. J.; Dong, Y.; Vennerstrom, J. L.; Charman, S. A. Antimicrob. Agents Chemother. 2008, 52, 1291–1296. doi:10.1128/AAC.01033-07 |
| 283. | Zhou, L.; Liu, X.; Ji, J.; Zhang, Y.; Hu, X.; Lin, L.; Feng, X. J. Am. Chem. Soc. 2012, 134, 17023–17026. doi:10.1021/ja309262f |
| 420. | Porter, N. A.; Kaplan, J. K.; Dussault, P. H. J. Am. Chem. Soc. 1990, 112, 1266–1267. doi:10.1021/ja00159a068 |
| 559. | Jourdan, J.; Matile, H.; Reift, E.; Biehlmaier, O.; Dong, Y.; Wang, X.; Mäser, P.; Vennerstrom, J. L.; Wittlin, S. ACS Infect. Dis. 2016, 2, 54–61. doi:10.1021/acsinfecdis.5b00090 |
| 419. | Davies, A. G.; Davison, I. G. E. J. Chem. Soc., Perkin Trans. 2 1989, 825–830. doi:10.1039/p29890000825 |
| 558. | Robert, A.; Coppel, Y.; Meunier, B. Inorg. Chim. Acta 2002, 339, 488–496. doi:10.1016/S0020-1693(02)00940-4 |
| 292. | Hedaya, E.; Winstein, S. Tetrahedron Lett. 1962, 3, 563–567. doi:10.1016/S0040-4039(00)76930-9 |
| 299. | Krasutsky, P. A.; Kolomitsyn, I. V.; Krasutsky, S. G.; Kiprof, P. Org. Lett. 2004, 6, 2539–2542. doi:10.1021/ol049171p |
| 136. | Griesbeck, A. G.; Schlundt, V. Synlett 2011, 2430–2432. doi:10.1055/s-0030-1261225 |
| 585. | Harrison, P. J.; Bugg, T. D. H. Arch. Biochem. Biophys. 2014, 544, 105–111. doi:10.1016/j.abb.2013.10.005 |
| 586. | Harrison, P. J.; Newgas, S. A.; Descombes, F.; Shepherd, S. A.; Thompson, A. J.; Bugg, T. D. H. FEBS J. 2015, 282, 3986–4000. doi:10.1111/febs.13400 |
| 436. | Wentworth, A. D.; Song, B.-D.; Nieva, J.; Shafton, A.; Tripurenani, S.; Wentworth, P., Jr. Chem. Commun. 2009, 3098–3100. doi:10.1039/B821584G |
| 584. | Xin, M.; Bugg, T. D. H. J. Am. Chem. Soc. 2008, 130, 10422–10430. doi:10.1021/ja8029569 |
| 303. | Srikrishna, A.; Anebouselvy, K. J. Org. Chem. 2001, 66, 7102–7106. doi:10.1021/jo0105484 |
| 301. | Srikrishna, A.; Reddy, T. J. Tetrahedron 1998, 54, 11517–11524. doi:10.1016/S0040-4020(98)00672-3 |
| 302. | Srikrishna, A.; Kumar, P. P.; Reddy, T. J. Tetrahedron Lett. 1998, 39, 5815–5818. doi:10.1016/S0040-4039(98)01148-4 |
| 303. | Srikrishna, A.; Anebouselvy, K. J. Org. Chem. 2001, 66, 7102–7106. doi:10.1021/jo0105484 |
| 304. | Srikrishna, A.; Gharpure, S. J.; Kumar, P. P. Tetrahedron Lett. 2000, 41, 3177–3180. doi:10.1016/S0040-4039(00)00325-7 |
| 305. | Srikrishna, A.; Anebouselvy, K.; Reddy, T. J. Tetrahedron Lett. 2000, 41, 6643–6647. doi:10.1016/S0040-4039(00)01107-2 |
| 306. | Srikrishna, A.; Anebouselvy, K. Tetrahedron Lett. 2002, 43, 2769–2771. doi:10.1016/S0040-4039(02)00382-9 |
| 307. | Srikrishna, A.; Mahesh, K. Synlett 2011, 2537–2540. doi:10.1055/s-0030-1260326 |
| 437. | Chen, D.; Chen, W.; Liu, D.; van Ofwegen, L.; Proksch, P.; Lin, W. J. Nat. Prod. 2013, 76, 1753–1763. doi:10.1021/np400480p |
| 1. | Ando, W. Organic Peroxides; Wiley: Chichester, 1992. |
| 2. | Reetz, I.; Yagci, Y.; Mishra, M. K. Initiation of Vinyl Polymerization by Organic Molecules and Nonmetal Initiators. Handbook of Vinyl Polymers; CRC Press: Boca Raton, 2008; pp 27–48. |
| 3. | Flory, P. J. Principles of Polymer Chemistry; Cornell University Press: Ithaca, New York, 1953. |
| 4. | Denisov, E. T.; Denisova, T. G.; Pokidova, T. S. Handbook of free radical initiators; John Wiley & Sons: New York, 2005. |
| 5. | Adam, W. Four-membered ring peroxides: 1,2-dioxetanes and α-peroxylactones. In Peroxides; Patai, S., Ed.; John Wiley & Sons, Ltd.: Chichester, U.K., 1983; pp 829–920. doi:10.1002/9780470771730.ch24 |
| 6. | Bouillon, G.; Lick, C.; Schank, K. Diacyl peroxides, peroxycarboxylic acids and peroxy esters. In Peroxides; Patai, S., Ed.; John Wiley & Sons, Ltd.: Chichester, U.K., 1983; pp 279–309. doi:10.1002/9780470771730.ch10 |
| 7. | Saito, I.; Nittala, S. S. Endoperoxides. In Peroxides; Patai, S., Ed.; John Wiley & Sons, Ltd.: Chichester, U.K., 1983; pp 311–374. doi:10.1002/9780470771730.ch11 |
| 8. | Sheldon, R. A. Synthesis and uses of alkyl hydroperoxides and dialkyl peroxides. In Peroxides; Patai, S., Ed.; John Wiley & Sons, Ltd.: Chichester, U.K., 1983; pp 161–200. doi:10.1002/9780470771730.ch6 |
| 9. | Adam, W. Peroxide Chemistry: Mechanistic and Preparative Aspects of Oxygen Transfer; Wiley-VCH: Weinheim, 2000. doi:10.1002/3527600396 |
| 297. | Ogibin, Y. N.; Terent'ev, A. O.; Kutkin, A. V.; Nikishin, G. I. Tetrahedron Lett. 2002, 43, 1321–1324. doi:10.1016/S0040-4039(01)02368-1 |
| 433. | Ngo, K.-S.; Brown, G. D. Tetrahedron 1999, 55, 14623–14634. doi:10.1016/S0040-4020(99)00938-2 |
| 577. | Komatsuzaki, H.; Shiota, A.; Hazawa, S.; Itoh, M.; Miyamura, N.; Miki, N.; Takano, Y.; Nakazawa, J.; Inagaki, A.; Akita, M.; Hikichi, S. Chem. – Asian J. 2013, 8, 1115–1119. doi:10.1002/asia.201300029 |
| 578. | Arnold, A.; Metzinger, R.; Limberg, C. Chem. – Eur. J. 2015, 21, 1198–1207. doi:10.1002/chem.201405155 |
| 579. | Poureskandari, M.; Safaei, E.; Sajjadi, S. M.; Karimpour, T.; Jaglicic, Z.; Lee, Y.-I. J. Mol. Struct. 2015, 1094, 130–136. doi:10.1016/j.molstruc.2015.04.008 |
| 296. | Goodman, R. M.; Kishi, Y. J. Org. Chem. 1994, 59, 5125–5127. doi:10.1021/jo00097a006 |
| 432. | Sy, L.-K.; Ngo, K.-S.; Brown, G. D. Tetrahedron 1999, 55, 15127–15140. doi:10.1016/S0040-4020(99)00987-4 |
| 575. | Sainsbury, P. D.; Mineyeva, Y.; Mycroft, Z.; Bugg, T. D. H. Bioorg. Chem. 2015, 60, 102–109. doi:10.1016/j.bioorg.2015.05.002 |
| 576. | Guzik, U.; Hupert-Kocurek, K.; Wojcieszyńska, D. Intradiol Dioxygenases – The Key Enzymes in Xenobiotics Degradation. In Biodegradation of Hazardous and Special Products; Chamy, R.; Rosenkranz, F., Eds.; InTech, 2013. doi:10.5772/56205 |
| 299. | Krasutsky, P. A.; Kolomitsyn, I. V.; Krasutsky, S. G.; Kiprof, P. Org. Lett. 2004, 6, 2539–2542. doi:10.1021/ol049171p |
| 300. | Krasutsky, P. A.; Kolomitsyn, I. V. ARKIVOC 2005, No. iv, 151–171. |
| 435. | Kinart, W. J.; Kinart, A.; Sendecki, M. Phys. Chem. Liq. 2008, 46, 627–630. doi:10.1080/00319100802072656 |
| 582. | Kovaleva, E. G.; Rogers, M. S.; Lipscomb, J. D. Biochemistry 2015, 54, 5329–5339. doi:10.1021/acs.biochem.5b00709 |
| 583. | Váradi, T.; Pap, J. S.; Giorgi, M.; Párkányi, L.; Csay, T.; Speier, G.; Kaizer, J. Inorg. Chem. 2013, 52, 1559–1569. doi:10.1021/ic302378r |
| 298. | Baumstark, A. L.; Vasquez, P. C.; Chen, Y.-X. J. Org. Chem. 1994, 59, 6692–6696. doi:10.1021/jo00101a030 |
| 434. | Ponce, M. A.; Ramirez, J. A.; Galagovsky, L. R.; Gros, E. G.; Erra-Balsells, R. J. Chem. Soc., Perkin Trans. 2 2000, 2351–2358. doi:10.1039/b000144i |
| 580. | Borowski, T.; Siegbahn, P. E. M. J. Am. Chem. Soc. 2006, 128, 12941–12953. doi:10.1021/ja0641251 |
| 581. | Mendel, S.; Arndt, A.; Bugg, T. D. H. Biochemistry 2004, 43, 13390–13396. doi:10.1021/bi048518t |
| 29. | Dembitsky, V. M. Eur. J. Med. Chem. 2008, 43, 223–251. doi:10.1016/j.ejmech.2007.04.019 |
| 30. | Dembitsky, V. M.; Gloriozova, T. A.; Poroikov, V. V. Mini-Rev. Med. Chem. 2007, 7, 571–589. doi:10.2174/138955707780859396 |
| 31. | del Sol Jimenez, M.; Garzón, S. P.; Rodriguez, A. D. J. Nat. Prod. 2003, 66, 655–661. doi:10.1021/np030021h |
| 32. | Posner, G. H.; D’Angelo, J.; MO’Neill, P.; Mercer, A. Expert Opin. Ther. Pat. 2006, 16, 1665–1672. doi:10.1517/13543776.16.12.1665 |
| 33. | Lai, H. C.; Singh, N. P.; Sasaki, T. Invest. New Drugs 2013, 31, 230–246. doi:10.1007/s10637-012-9873-z |
| 34. | Dwivedi, A.; Mazumder, A.; du Plessis, L.; du Preez, J. L.; Haynes, R. K.; du Plessis, J. Nanomedicine 2015, 11, 2041–2050. doi:10.1016/j.nano.2015.07.010 |
| 429. | Dussault, P. H.; Woller, K. R. J. Am. Chem. Soc. 1997, 119, 3824–3825. doi:10.1021/ja970174p |
| 572. | Xu, L.; Porter, N. A. Free Radical Res. 2015, 49, 835–849. doi:10.3109/10715762.2014.985219 |
| 573. | Miyoshi, N.; Iuliano, L.; Tomono, S.; Ohshima, H. Biochem. Biophys. Res. Commun. 2014, 446, 702–708. doi:10.1016/j.bbrc.2013.12.107 |
| 24. | Boissier, J.; Portela, J.; Pradines, V.; Coslédan, F.; Robert, A.; Meunier, B. C. R. Chim. 2012, 15, 75–78. doi:10.1016/j.crci.2011.11.008 |
| 25. | Keiser, J.; Veneziano, V.; Rinaldi, L.; Mezzino, L.; Duthaler, U.; Cringoli, G. Res. Vet. Sci. 2010, 88, 107–110. doi:10.1016/j.rvsc.2009.05.007 |
| 26. | Keiser, J.; Ingram, K.; Vargas, M.; Chollet, J.; Wang, X.; Dong, Y.; Vennerstrom, J. L. Antimicrob. Agents Chemother. 2012, 56, 1090–1092. doi:10.1128/AAC.05371-11 |
| 27. | Shuhua, X.; Tanner, M.; N'Goran, E. K.; Utzinger, J.; Chollet, J.; Bergquist, R.; Minggang, C.; Jiang, Z. Acta Trop. 2002, 82, 175–181. doi:10.1016/S0001-706X(02)00009-8 |
| 28. | Ingram, K.; Yaremenko, I. A.; Krylov, I. B.; Hofer, L.; Terent’ev, A. O.; Keiser, J. J. Med. Chem. 2012, 55, 8700–8711. doi:10.1021/jm3009184 |
| 571. | Itri, R.; Junqueira, H. C.; Mertins, O.; Baptista, M. S. Biophys. Rev. 2014, 6, 47–61. doi:10.1007/s12551-013-0128-9 |
| 11. | Jefford, C. W. Curr. Top. Med. Chem. 2012, 12, 373–399. doi:10.2174/156802612799362940 |
| 12. | Slack, R. D.; Jacobine, A. M.; Posner, G. H. Med. Chem. Commun. 2012, 3, 281–297. doi:10.1039/c2md00277a |
| 13. | Muregi, F. W.; Ishih, A. Drug Dev. Res. 2010, 71, 20–32. doi:10.1002/ddr.20345 |
| 14. | Fernández, I.; Robert, A. Org. Biomol. Chem. 2011, 9, 4098–4107. doi:10.1039/c1ob05088e |
| 15. | Chadwick, J.; Amewu, R. K.; Marti, F.; Bousejra-El Garah, F.; Sharma, R.; Berry, N. G.; Stocks, P. A.; Burrell-Saward, H.; Wittlin, S.; Rottmann, M.; Brun, R.; Taramelli, D.; Parapini, S.; Ward, S. A.; O'Neill, P. M. ChemMedChem 2011, 6, 1357–1361. doi:10.1002/cmdc.201100196 |
| 16. | Opsenica, D. M.; Šolaja, B. A. J. Serb. Chem. Soc. 2009, 74, 1155–1193. doi:10.2298/JSC0911155O |
| 17. | Ghorai, P.; Dussault, P. H.; Hu, C. Org. Lett. 2008, 10, 2401–2404. doi:10.1021/ol800657m |
| 18. | Dong, Y. Mini-Rev. Med. Chem. 2002, 2, 113–123. doi:10.2174/1389557024605537 |
| 19. | Vennerstrom, J. L.; Fu, H. N.; Ellis, W. Y.; Ager, A. L., Jr.; Wood, J. K.; Andersen, S. L.; Gerena, L.; Milhous, W. K. J. Med. Chem. 1992, 35, 3023–3027. doi:10.1021/jm00094a015 |
| 20. | Yadav, N.; Sharma, C.; Awasthi, S. K. RSC Adv. 2014, 4, 5469–5498. doi:10.1039/c3ra42513d |
| 21. | Terent’ev, A. O.; Borisov, D. A.; Yaremenko, I. A. Chem. Heterocycl. Compd. 2012, 48, 55–58. doi:10.1007/s10593-012-0969-3 |
| 22. | Hao, H.-D.; Wittlin, S.; Wu, Y. Chem. – Eur. J. 2013, 19, 7605–7619. doi:10.1002/chem.201300076 |
| 23. | Li, Y.; Hao, H.-D.; Wittlin, S.; Wu, Y. Chem. – Asian J. 2012, 7, 1881–1886. doi:10.1002/asia.201200166 |
| 295. | Schreiber, S. L.; Liew, W.-F. Tetrahedron Lett. 1983, 24, 2363–2366. doi:10.1016/S0040-4039(00)81926-7 |
| 431. | Bortolomeazzi, R.; De Zan, M.; Pizzale, L.; Conte, L. S. J. Agric. Food Chem. 1999, 47, 3069–3074. doi:10.1021/jf9812580 |
| 574. | Costas, M.; Mehn, M. P.; Jensen, M. P.; Que, L., Jr. Chem. Rev. 2004, 104, 939–986. doi:10.1021/cr020628n |
| 10. | World Health Organization. World Health Organization. World malaria report 2015; WHO Press, 2005.. http://www.who.int/malaria/publications/world-malaria-report-2015/en/ (accessed July 4, 2016). |
| 293. | Hedaya, E.; Winstein, S. J. Am. Chem. Soc. 1967, 89, 1661–1672. doi:10.1021/ja00983a023 |
| 294. | Wistuba, E.; Rüchardt, C. Tetrahedron Lett. 1981, 22, 3389–3392. doi:10.1016/S0040-4039(01)81913-4 |
| 430. | Nonami, Y.; Baran, J.; Sosnicki, J.; Mayr, H.; Masuyama, A.; Nojima, M. J. Org. Chem. 1999, 64, 4060–4063. doi:10.1021/jo990127a |
| 67. | Brinkhorst, J.; Nara, S. J.; Pratt, D. A. J. Am. Chem. Soc. 2008, 130, 12224–12225. doi:10.1021/ja804162d |
| 43. | Givelet, C.; Bernat, V.; Danel, M.; André-Barrès, C.; Vial, H. Eur. J. Org. Chem. 2007, 3095–3101. doi:10.1002/ejoc.200700086 |
| 40. | O'Neill, P. M.; Stocks, P. A.; Pugh, M. D.; Araujo, N. C.; Korshin, E. E.; Bickley, J. F.; Ward, S. A.; Bray, P. G.; Pasini, E.; Davies, J.; Verissimo, E.; Bachi, M. D. Angew. Chem., Int. Ed. 2004, 43, 4193–4197. doi:10.1002/anie.200453859 |
| 41. | Holla, H.; Labaied, M.; Pham, N.; Jenkins, I. D.; Stuart, K.; Quinn, R. J. Bioorg. Med. Chem. Lett. 2011, 21, 4793–4797. doi:10.1016/j.bmcl.2011.06.059 |
| 42. | Lombardo, M.; Sonawane, D. P.; Quintavalla, A.; Trombini, C.; Dhavale, D. D.; Taramelli, D.; Corbett, Y.; Rondinelli, F.; Fattorusso, C.; Persico, M.; Taglialatela-Scafati, O. Eur. J. Org. Chem. 2014, 1607–1614. doi:10.1002/ejoc.201301394 |
| 38. | Wang, X.; Dong, Y.; Wittlin, S.; Charman, S. A.; Chiu, F. C. K.; Chollet, J.; Katneni, K.; Mannila, J.; Morizzi, J.; Ryan, E.; Scheurer, C.; Steuten, J.; Santo Tomas, J.; Snyder, C.; Vennerstrom, J. L. J. Med. Chem. 2013, 56, 2547–2555. doi:10.1021/jm400004u |
| 39. | Chaudhary, S.; Sharma, V.; Jaiswal, P. K.; Gaikwad, A. N.; Sinha, S. K.; Puri, S. K.; Sharon, A.; Maulik, P. R.; Chaturvedi, V. Org. Lett. 2015, 17, 4948–4951. doi:10.1021/acs.orglett.5b02296 |
| 35. | Martyn, D. C.; Ramirez, A. P.; Berattie, M. J.; Cortese, J. F.; Patel, V.; Rush, M. A.; Woerpel, K. A.; Clardy, J. Bioorg. Med. Chem. Lett. 2008, 18, 6521–6524. doi:10.1016/j.bmcl.2008.10.083 |
| 36. | Schiaffo, C. E.; Rottman, M.; Wittlin, S.; Dussault, P. H. ACS Med. Chem. Lett. 2011, 2, 316–319. doi:10.1021/ml100308d |
| 37. | Ingram, K.; Schiaffo, C. E.; Sittiwong, W.; Benner, E.; Dussault, P. H.; Keiser, J. J. Antimicrob. Chemother. 2012, 67, 1979–1986. doi:10.1093/jac/dks141 |
| 320. | Lange, J.-P.; Breed, A. J. M. Catal. Commun. 2002, 3, 25–28. doi:10.1016/S1566-7367(01)00071-1 |
| 442. | Ingold, K. U.; Smeu, M.; DiLabio, G. A. J. Org. Chem. 2006, 71, 9906–9908. doi:10.1021/jo061898z |
| 443. | DiLabio, G. A.; Ingold, K. U.; Lin, S.; Litwinienko, G.; Mozenson, O.; Mulder, P.; Tidwell, T. T. Angew. Chem., Int. Ed. 2010, 49, 5982–5985. doi:10.1002/anie.201001008 |
| 319. | Olah, G. A.; Parker, D. G.; Yoneda, N. Angew. Chem., Int. Ed. Engl. 1978, 17, 909–931. doi:10.1002/anie.197809091 |
| 441. | Wieland, H. Ber. Dtsch. Chem. Ges. 1911, 44, 2550–2556. doi:10.1002/cber.19110440380 |
| 321. | Lillie, T. S.; Ronald, R. C. J. Org. Chem. 1985, 50, 5084–5088. doi:10.1021/jo00225a018 |
| 314. | Udris, R. J.; Sergeyev, P. G.; Kruzhalov, B. D. Sposob polucheniya gidroperekisejj alkilirovannykh-proizvodnykh benzola ili alicikloaromaticheskikh uglevodorodov. USSR Patent 106,666, Jan 7, 1947. |
| 315. | Sergeyev, P. G.; Udris, R. J.; Kruzhalov, B. D.; Nyemtsov, B. D. Sposob odnovremennogo polucheniya fenola i acetona. USSR Patent 106,992, Jan 7, 1947. |
| 431. | Bortolomeazzi, R.; De Zan, M.; Pizzale, L.; Conte, L. S. J. Agric. Food Chem. 1999, 47, 3069–3074. doi:10.1021/jf9812580 |
| 313. | Wang, Z. Hock Rearrangement. Comprehensive Organic Name Reactions and Reagents; John Wiley & Sons, 2010; pp 1438–1442. doi:10.1002/9780470638859.conrr321 |
| 416. | Beckwith, A. L. J.; Davies, A. G.; Davison, I. G. E.; Maccoll, A.; Mruzek, M. H. J. Chem. Soc., Perkin Trans. 2 1989, 815–824. doi:10.1039/P29890000815 |
| 318. | Deno, N. C.; Billups, W. E.; Kramer, K. E.; Lastomirsky, R. R. J. Org. Chem. 1970, 35, 3080–3082. doi:10.1021/jo00834a046 |
| 440. | Courtneidge, J. L. J. Chem. Soc., Chem. Commun. 1992, 1270–1272. doi:10.1039/c39920001270 |
| 316. | Hock, H.; Lang, S. Ber. Dtsch. Chem. Ges. A 1944, 77, 257–264. doi:10.1002/cber.19440770321 |
| 317. | Hock, H.; Kropf, H. Angew. Chem. 1957, 69, 313–321. doi:10.1002/ange.19570691002 |
| 439. | Dang, H.-S.; Davies, A. G.; Schiesser, C. H. J. Chem. Soc., Perkin Trans. 1 1990, 3, 789–794. doi:10.1039/p19900000789 |
| 308. | Gándara, Z.; Rivadulla, M. L.; Pérez, M.; Gómez, G.; Fall, Y. Eur. J. Org. Chem. 2013, 5678–5682. doi:10.1002/ejoc.201300528 |
| 186. | Davies, A. G. J. Chem. Res. 2009, 533–544. doi:10.3184/030823409X12491375725131 |
| 438. | Pratt, D. A.; Tallman, K. A.; Porter, N. A. Acc. Chem. Res. 2011, 44, 458–467. doi:10.1021/ar200024c |
| 310. | Chao, W.; Hsieh, J.-T.; Chang, C.-H.; Lin, J. J.-M. Science 2015, 347, 751–754. doi:10.1126/science.1261549 |
| 311. | Liu, F.; Beames, J. M.; Petit, A. S.; McCoy, A. B.; Lester, M. I. Science 2014, 345, 1596–1598. doi:10.1126/science.1257158 |
| 312. | Welz, O.; Savee, J. D.; Osborn, D. L.; Vasu, S. S.; Percival, C. J.; Shallcross, D. E.; Taatjes, C. A. Science 2012, 335, 204–207. doi:10.1126/science.1213229 |
| 439. | Dang, H.-S.; Davies, A. G.; Schiesser, C. H. J. Chem. Soc., Perkin Trans. 1 1990, 3, 789–794. doi:10.1039/p19900000789 |
| 309. | Taatjes, C. A.; Shallcross, D. E.; Percival, C. J. Phys. Chem. Chem. Phys. 2014, 16, 1704–1718. doi:10.1039/c3cp52842a |
| 427. | Avila, D. V.; Davies, A. G.; Davison, I. G. E. J. Chem. Soc., Perkin Trans. 2 1988, 1847–1852. doi:10.1039/p29880001847 |
| 196. | Strukul, G. Angew. Chem., Int. Ed. 1998, 37, 1198–1209. doi:10.1002/(SICI)1521-3773(19980518)37:9<1198::AID-ANIE1198>3.0.CO;2-Y |
| 197. | Chida, N.; Tobe, T.; Ogawa, S. Tetrahedron Lett. 1994, 35, 7249–7252. doi:10.1016/0040-4039(94)85373-8 |
| 198. | Butkus, E.; Stončius, S. J. Chem. Soc., Perkin Trans. 1 2001, 1885–1888. doi:10.1039/b103564a |
| 199. | Doering, W. v. E.; Speers, L. J. Am. Chem. Soc. 1950, 72, 5515–5518. doi:10.1021/ja01168a041 |
| 204. | Taylor, R. T.; Flood, L. A. J. Org. Chem. 1983, 48, 5160–5164. doi:10.1021/jo00174a003 |
| 205. | Jacobson, S. E.; Mares, F.; Zambri, P. M. J. Am. Chem. Soc. 1979, 101, 6938–6946. doi:10.1021/ja00517a025 |
| 201. | Lambert, A.; Macquarrie, D. J.; Carr, G.; Clark, J. H. New J. Chem. 2000, 24, 485–488. doi:10.1039/b003161p |
| 202. | Chang, C. D.; Hellring, S. D. Shape-selective oxidation of cyclic ketones. U.S. Patent US4,870,192, Sept 26, 1989. |
| 203. | Fischer, J.; Hölderich, W. F. Appl. Catal., A 1999, 180, 435–443. doi:10.1016/S0926-860X(98)00378-0 |
| 196. | Strukul, G. Angew. Chem., Int. Ed. 1998, 37, 1198–1209. doi:10.1002/(SICI)1521-3773(19980518)37:9<1198::AID-ANIE1198>3.0.CO;2-Y |
| 200. | ten Brink, G.-J.; Arends, I. W. C. E.; Sheldon, R. A. Chem. Rev. 2004, 104, 4105–4124. doi:10.1021/cr030011l |
| 195. | Krow, G. R. The Baeyer–Villiger Oxidation of Ketones and Aldehydes. In Organic Reactions; Paquette, L. A., Ed.; John Wiley & Sons, Inc., 1993; Vol. 43, pp 251–798. doi:10.1002/0471264180.or043.03 |
| 185. | Renz, M.; Meunier, B. Eur. J. Org. Chem. 1999, 737–750. doi:10.1002/(SICI)1099-0690(199904)1999:4<737::AID-EJOC737>3.0.CO;2-B |
| 206. | Sandaroos, R.; Goldani, M. T.; Damavandi, S.; Mohammadi, A. J. Chem. Sci. 2012, 124, 871–876. doi:10.1007/s12039-012-0277-6 |
| 203. | Fischer, J.; Hölderich, W. F. Appl. Catal., A 1999, 180, 435–443. doi:10.1016/S0926-860X(98)00378-0 |
| 207. |
Hoelderich, W. P. D.; Fischer, J.; Schindler, G. P. D.; Arntz, D. D. Verfahren zur Herstellung von Lactonen durch Baeyer-Villiger Oxidation. Ger. Patent DE19,745,442 A1, April 22, 1999.
Chem. Abstr. 1999, 130, 281989u. |
| 506. | Liu, H.-H.; Jin, H.-X.; Wu, Y.-K. Chin. J. Chem. 2004, 22, 1029–1033. doi:10.1002/cjoc.20040220930 |
| 208. | Gavagnin, R.; Cataldo, M.; Pinna, F.; Strukul, G. Organometallics 1998, 17, 661–667. doi:10.1021/om970756f |
| 505. | Bloodworth, A. J.; Shah, A. Tetrahedron Lett. 1995, 36, 7551–7554. doi:10.1016/0040-4039(95)01530-2 |
| 508. | Liu, H.-H.; Wu, Y.-K.; Shen, X. Chin. J. Chem. 2003, 21, 875–877. doi:10.1002/cjoc.20030210731 |
| 507. | Persico, M.; Quintavalla, A.; Rondinelli, F.; Trombini, C.; Lombardo, M.; Fattorusso, C.; Azzarito, V.; Taramelli, D.; Parapini, S.; Corbett, Y.; Chianese, G.; Fattorusso, E.; Taglialatela-Scafati, O. J. Med. Chem. 2011, 54, 8526–8540. doi:10.1021/jm201056j |
| 503. | Suzuki, M.; Ohtake, H.; Kameya, Y.; Hamanaka, N.; Noyori, R. J. Org. Chem. 1989, 54, 5292–5302. doi:10.1021/jo00283a023 |
| 505. | Bloodworth, A. J.; Shah, A. Tetrahedron Lett. 1995, 36, 7551–7554. doi:10.1016/0040-4039(95)01530-2 |
| 504. | Kamata, M.; Satoh, C.; Kim, H.-S.; Wataya, Y. Tetrahedron Lett. 2002, 43, 8313–8317. doi:10.1016/S0040-4039(02)02024-5 |
| 216. | Berkessel, A.; Andreae, M. R. M. Tetrahedron Lett. 2001, 42, 2293–2295. doi:10.1016/S0040-4039(01)00141-1 |
| 217. | McClure, J. D.; Williams, P. H. J. Org. Chem. 1962, 27, 24–26. doi:10.1021/jo01048a005 |
| 214. | Jacobson, S. E.; Tang, R.; Mares, F. J. Chem. Soc., Chem. Commun. 1978, 888–889. doi:10.1039/c39780000888 |
| 215. | Uchida, T.; Katsuki, T.; Ito, K.; Akashi, S.; Ishii, A.; Kuroda, T. Helv. Chim. Acta 2002, 85, 3078–3089. doi:10.1002/1522-2675(200210)85:10<3078::AID-HLCA3078>3.0.CO;2-1 |
| 212. | ten Brink, G.-J.; Vis, J.-M.; Arends, I. W. C. E.; Sheldon, R. A. J. Org. Chem. 2001, 66, 2429–2433. doi:10.1021/jo0057710 |
| 213. | Grieco, P. A.; Yokoyama, Y.; Gilman, S.; Ohfune, Y. J. Chem. Soc., Chem. Commun. 1977, 870–871. doi:10.1039/c39770000870 |
| 205. | Jacobson, S. E.; Mares, F.; Zambri, P. M. J. Am. Chem. Soc. 1979, 101, 6938–6946. doi:10.1021/ja00517a025 |
| 209. | Bolm, C.; Beckmann, O. Chirality 2000, 12, 523–525. doi:10.1002/(SICI)1520-636X(2000)12:5/6<523::AID-CHIR39>3.0.CO;2-Z |
| 210. | Phillips, A. M. F.; Romão, C. Eur. J. Org. Chem. 1999, 1767–1770. doi:10.1002/(SICI)1099-0690(199908)1999:8<1767::AID-EJOC1767>3.0.CO;2-S |
| 211. | Herrmann, W. A.; Fischer, R. W.; Correia, J. D. G. J. Mol. Catal. 1994, 94, 213–223. doi:10.1016/S0304-5102(94)87043-8 |
| 510. | Baran, A.; Bekarlar, M.; Aydin, G.; Nebioglu, M.; Sahin, E.; Balci, M. J. Org. Chem. 2012, 77, 1244–1250. doi:10.1021/jo202494v |
| 509. | Boyd, J. D.; Foote, C. S.; Imagawa, D. K. J. Am. Chem. Soc. 1980, 102, 3641–3642. doi:10.1021/ja00530a063 |
| 511. | Baran, A.; Cambul, S.; Nebioglu, M.; Balci, M. J. Org. Chem. 2012, 77, 5086–5097. doi:10.1021/jo300655p |
| 516. | Kiliç, H.; Balci, M. Tetrahedron 2001, 57, 9889–9897. doi:10.1016/S0040-4020(01)01008-0 |
| 515. | Özer, G.; Saraçoglu, N.; Balci, M. J. Org. Chem. 2003, 68, 7009–7015. doi:10.1021/jo0345300 |
| 518. | Adam, W.; Balci, M.; Kiliç, H. J. Org. Chem. 2000, 65, 5926–5931. doi:10.1021/jo000120p |
| 517. | Dastan, A.; Saracoglu, N.; Balci, M. Eur. J. Org. Chem. 2001, 3519–3522. doi:10.1002/1099-0690(200109)2001:18<3519::AID-EJOC3519>3.0.CO;2-2 |
| 353. | Güney, M.; Ceylan, Z. C.; Daştan, A.; Balci, M. Can. J. Chem. 2005, 83, 227–235. doi:10.1139/v05-046 |
| 512. | Özen, R.; Kormali, F.; Balci, M.; Atasoy, B. Tetrahedron 2001, 57, 7529–7535. doi:10.1016/S0040-4020(01)00702-5 |
| 514. | Griffen, J. A.; Kenwright, S. J.; Abou-Shehada, S.; Wharry, S.; Moody, T. S.; Lewis, S. E. Org. Chem. Front. 2014, 1, 79–90. doi:10.1039/c3qo00057e |
| 513. | Coşkun, A.; Güney, M.; Daştan, A.; Balci, M. Tetrahedron 2007, 63, 4944–4950. doi:10.1016/j.tet.2007.03.145 |
| 55. | Ritz, J.; Fuchs, H.; Kieczka, H.; Moran, W. C. Caprolactam. Ullmann's Encyclopedia of Industrial Chemistry; Wiley-VCH, 2000. doi:10.1002/14356007.a05_031 |
| 56. | Vollhardt, K. P. C.; Schore, N. E. Organic Chemistry: Structure and Function, 7th ed.; W. H. Freeman and Company, 2014. |
| 57. | Poth, U. Drying Oils and Related Products. Ullmann's Encyclopedia of Industrial Chemistry; Wiley-VCH, 2000. doi:10.1002/14356007.a09_055 |
| 58. | Mallégol, J.; Gardette, J.-L.; Lemaire, J. J. Am. Oil Chem. Soc. 2000, 77, 249–255. doi:10.1007/s11746-000-0041-5 |
| 59. | van Gorkum, R.; Bouwman, E. Coord. Chem. Rev. 2005, 249, 1709–1728. doi:10.1016/j.ccr.2005.02.002 |
| 60. | Juita; Dlugogorski, B. Z.; Kennedy, E. M.; Mackie, J. C. Fire Sci. Rev. 2012, 1, No. 3. doi:10.1186/2193-0414-1-3 |
| 61. | Oakley, L. H.; Casadio, F.; Shull, K. R.; Broadbelt, L. J. Appl. Phys. A 2015, 121, 869–878. doi:10.1007/s00339-015-9363-1 |
| 49. | Baer, H.; Bergamo, M.; Forlin, A.; Pottenger, L. H.; Lindner, J. Propylene Oxide. Ullmann's Encyclopedia of Industrial Chemistry; Wiley-VCH: Weinheim, Germany, 2012. doi:10.1002/14356007.a22_239.pub3 |
| 50. | Sienel, G.; Rieth, R.; Rowbottom, K. T. Epoxides. Ullmann's Encyclopedia of Industrial Chemistry; Wiley-VCH, 2000. doi:10.1002/14356007.a09_531 |
| 51. | Wang, Z. Prilezhaev Reaction. Comprehensive Organic Name Reactions and Reagents; John Wiley & Sons, Inc., 2010; pp 2270–2274. doi:10.1002/9780470638859.conrr513 |
| 52. | Weber, M.; Weber, M.; Kleine-Boymann, M. Phenol. Ullmann's Encyclopedia of Industrial Chemistry; Wiley-VCH, 2000. doi:10.1002/14356007.a19_299.pub2 |
| 53. | Schmidt, R. J. Appl. Catal., A 2005, 280, 89–103. doi:10.1016/j.apcata.2004.08.030 |
| 54. | Weissermel, K.; Arpe, H.-J. Benzene Derivatives. Industrial Organic Chemistry; Wiley-VCH, 2008; pp 337–385. doi:10.1002/9783527619191.ch13 |
| 46. | World Health Organization; Management of severe malaria – A practical handbook, 3rd ed.; WHO Press: 2013. http://www.who.int/malaria/publications/atoz/9789241548526/en/ (accessed July 4, 2016). |
| 47. | World Health Organization; Guidelines for the treatment of malaria, 3rd ed.; WHO Press, 2015. http://www.who.int/malaria/publications/atoz/9789241549127/en/ (accessed July 4, 2016). |
| 48. | Johnson, R. A.; Sharpless, K. B. Addition Reactions with Formation of Carbon–Oxygen Bonds: (ii) Asymmetric Methods of Epoxidation. In Comprehensive Organic Synthesis; Fleming, B. M. T., Ed.; Pergamon Press: Oxford, 1991; pp 389–436. doi:10.1016/B978-0-08-052349-1.00196-7 |
| 22. | Hao, H.-D.; Wittlin, S.; Wu, Y. Chem. – Eur. J. 2013, 19, 7605–7619. doi:10.1002/chem.201300076 |
| 44. | Amewu, R.; Gibbons, P.; Mukhtar, A.; Stachulski, A. V.; Ward, S. A.; Hall, C.; Rimmer, K.; Davies, J.; Vivas, L.; Bacsa, J.; Mercer, A. E.; Nixon, G.; Stocks, P. A.; O'Neill, P. M. Org. Biomol. Chem. 2010, 8, 2068–2077. doi:10.1039/b924319d |
| 45. | Reiter, C.; Fröhlich, T.; Zeino, M.; Marschall, M.; Bahsi, H.; Leidenberger, M.; Friedrich, O.; Kappes, B.; Hampel, F.; Efferth, T.; Tsogoeva, S. B. Eur. J. Med. Chem. 2015, 97, 164–172. doi:10.1016/j.ejmech.2015.04.053 |
| 72. | Milas, N. A.; Mageli, O. L.; Golubovic, A.; Arndt, R. W.; Ho, J. C. J. J. Am. Chem. Soc. 1963, 85, 222–226. doi:10.1021/ja00885a022 |
| 73. | Rieche, A.; Bischoff, C. Chem. Ber. 1962, 95, 77–82. doi:10.1002/cber.19620950115 |
| 74. | Criegee, R. Organic Peroxides; Wiley-Interscience: New York, London, 1972. |
| 75. | Cocker, W.; Grayson, D. H. J. Chem. Soc., Perkin Trans. 1 1975, 1347–1352. doi:10.1039/P19750001347 |
| 76. |
Vinogradova, L. P.; Zav’yalov, S. I. Izv. Akad. Nauk SSSR, Ser. Khim. 1961, 2050–2054.
Chem. Abstr. 1962, 57, 12344c. |
| 77. |
Vinogradova, L. P.; Zav’yalov, S. I. Izv. Akad. Nauk SSSR, Ser. Khim. 1961, 1482–1486.
Chem. Abstr. 1962, 56, 338b. |
| 78. |
Vinogradova, L. P.; Rudenko, B. A.; Zav’yalov, S. I. Izv. Akad. Nauk SSSR, Ser. Khim. 1962, 1436–1441.
Chem. Abstr. 1963, 58, 2378g. |
| 79. | Rieche, A.; Bischoff, C.; Prescher, D. Chem. Ber. 1964, 97, 3071–3075. doi:10.1002/cber.19640971116 |
| 80. | Rieche, A.; Seyfarth, H. E.; Brand, F. Justus Liebigs Ann. Chem. 1969, 725, 93–98. doi:10.1002/jlac.19697250112 |
| 81. | Wolffenstein, R. Ber. Dtsch. Chem. Ges. 1895, 28, 2265–2269. doi:10.1002/cber.189502802208 |
| 82. | Criegee, R.; Schnorrenberg, W.; Becke, J. Justus Liebigs Ann. Chem. 1949, 565, 7–21. doi:10.1002/jlac.19495650103 |
| 83. | Dickey, F. H.; Rust, F. F.; Vaughan, W. E. J. Am. Chem. Soc. 1949, 71, 1432–1434. doi:10.1021/ja01172a081 |
| 84. | Criegee, R.; Metz, K. Chem. Ber. 1956, 89, 1714–1718. doi:10.1002/cber.19560890720 |
| 85. | Hawkins, E. G. E. J. Chem. Soc. C 1969, 2663–2670. doi:10.1039/j39690002663 |
| 86. | Hawkins, E. G. E. J. Chem. Soc. C 1969, 2671–2677. doi:10.1039/j39690002671 |
| 87. | Hawkins, E. G. E. J. Chem. Soc. C 1969, 2678–2681. doi:10.1039/j39690002678 |
| 88. | Hawkins, E. G. E. J. Chem. Soc. C 1969, 2682–2686. doi:10.1039/j39690002682 |
| 89. | Hawkins, E. G. E. J. Chem. Soc. C 1969, 2686–2691. doi:10.1039/j39690002686 |
| 90. | Hawkins, E. G. E. J. Chem. Soc. C 1969, 2691–2697. doi:10.1039/j39690002691 |
| 91. | Milas, N. A.; Golubović, A. J. Am. Chem. Soc. 1959, 81, 6461–6462. doi:10.1021/ja01533a033 |
| 92. | Rieche, A. Angew. Chem. 1958, 70, 251–266. doi:10.1002/ange.19580700902 |
| 93. | Edwards, J. O.; Sauer, M. C. V. J. Phys. Chem. 1971, 75, 3004–3011. doi:10.1021/j100688a023 |
| 94. | Hine, J.; Redding, R. W. J. Org. Chem. 1970, 35, 2769–2772. doi:10.1021/jo00833a065 |
| 95. | Jefford, C. W.; Jaggi, D.; Kohmoto, S.; Boukouvalas, J.; Bernardinelli, G. Helv. Chim. Acta 1984, 67, 2254–2260. doi:10.1002/hlca.19840670831 |
| 96. | Singh, C. Tetrahedron Lett. 1990, 31, 6901–6902. doi:10.1016/S0040-4039(00)97202-2 |
| 97. | Jefford, C. W.; Li, Y.; Jaber, A.; Boukouvalas, J. Synth. Commun. 1990, 20, 2589–2596. doi:10.1080/00397919008051466 |
| 98. | Antonovskii, V. L.; Fedorova, E. V.; Shtivel, N. E.; Emelin, Y. D. Zh. Org. Khim. 1991, 27, 820–823. |
| 99. | Avery, M. A.; Gao, F.; Chong, W. K. M.; Hendrickson, T. F.; Inman, W. D.; Crews, P. Tetrahedron 1994, 50, 957–972. doi:10.1016/S0040-4020(01)80810-3 |
| 100. | Rieche, A.; Bischoff, C.; Dietrich, P. Chem. Ber. 1961, 94, 2932–2936. doi:10.1002/cber.19610941115 |
| 101. | Shreibert, A. I.; Chardin, A. P.; Pil’dus, I. E.; Ermarchenko, V. I. Zh. Org. Khim. 1971, 7, 967–971. |
| 102. |
Razuvaev, G. A.; Dodonov, V. A.; Zaburdyaeva, S. N. Zh. Org. Khim. 1968, 4, 1302–1303.
J. Org. Chem. USSR (Eng. Trans.) 1968, 4, 1256–1257. |
| 103. | Dussault, P. H.; Lee, I. Q. J. Am. Chem. Soc. 1993, 115, 6458–6459. doi:10.1021/ja00067a090 |
| 104. | Dussault, P. H.; Eary, C. T. J. Am. Chem. Soc. 1998, 120, 7133–7134. doi:10.1021/ja9808503 |
| 105. | Dussault, P. H.; Lee, H.-J.; Niu, Q. J. J. Org. Chem. 1995, 60, 784–785. doi:10.1021/jo00109a001 |
| 106. | Kuznetsov, M. L.; Rocha, B. G. M.; Pombeiro, A. J. L.; Shul’pin, G. B. ACS Catal. 2015, 5, 3823–3835. doi:10.1021/acscatal.5b00077 |
| 107. | Wang, C.; Yamamoto, H. J. Am. Chem. Soc. 2014, 136, 1222–1225. doi:10.1021/ja411379e |
| 108. | Bunge, A.; Hamann, H.-J.; Dietz, D.; Liebscher, J. Tetrahedron 2013, 69, 2446–2450. doi:10.1016/j.tet.2013.01.032 |
| 109. | Jadhav, V. H.; Bande, O. P.; Puranik, V. G.; Dhavale, D. D. Tetrahedron: Asymmetry 2010, 21, 163–170. doi:10.1016/j.tetasy.2010.01.007 |
| 110. | Cussó, O.; Garcia-Bosch, I.; Ribas, X.; Lloret-Fillol, J.; Costas, M. J. Am. Chem. Soc. 2013, 135, 14871–14878. doi:10.1021/ja4078446 |
| 111. | Nieto, N.; Munslow, I. J.; Fernández-Pérez, H.; Vidal-Ferran, A. Synlett 2008, 2856–2858. doi:10.1055/s-0028-1083545 |
| 112. | Burke, C. P.; Shi, Y. J. Org. Chem. 2007, 72, 4093–4097. doi:10.1021/jo070205r |
| 113. | Lifchits, O.; Mahlau, M.; Reisinger, C. M.; Lee, A.; Farès, C.; Polyak, I.; Gopakumar, G.; Thiel, W.; List, B. J. Am. Chem. Soc. 2013, 135, 6677–6693. doi:10.1021/ja402058v |
| 62. | Roda, A. Chemiluminescence and bioluminescence: past, present and future; RSC Press: Cambridge, 2010. doi:10.1039/9781849732024 |
| 63. | Dodeigne, C.; Thunus, L.; Lejeune, R. Talanta 2000, 51, 415–439. doi:10.1016/S0039-9140(99)00294-5 |
| 64. | Ciardelli, F.; Ruggeri, G.; Pucci, A. Chem. Soc. Rev. 2013, 42, 857–870. doi:10.1039/C2CS35414D |
| 65. | Roda, A.; Mirasoli, M.; Michelini, E.; Di Fusco, M.; Zangheri, M.; Cevenini, L.; Roda, B.; Simoni, P. Biosens. Bioelectron. 2016, 76, 164–179. doi:10.1016/j.bios.2015.06.017 |
| 66. | Takeuchi, C.; Galvé, R.; Nieva, J.; Witter, D. P.; Wentworth, A. D.; Troseth, R. P.; Lerner, R. A.; Wentworth, P., Jr. Biochemistry 2006, 45, 7162–7170. doi:10.1021/bi0604330 |
| 67. | Brinkhorst, J.; Nara, S. J.; Pratt, D. A. J. Am. Chem. Soc. 2008, 130, 12224–12225. doi:10.1021/ja804162d |
| 68. | Murphy, R. C.; Johnson, K. M. J. Biol. Chem. 2008, 283, 15521–15525. doi:10.1074/jbc.R700049200 |
| 69. | Terao, J.; Minami, Y.; Bando, N. J. Clin. Biochem. Nutr. 2011, 48, 57–62. doi:10.3164/jcbn.11-008FR |
| 70. | Terao, J. Cholesterol Hydroperoxides and Their Degradation Mechanism. In Lipid Hydroperoxide-Derived Modification of Biomolecules; Kato, Y., Ed.; Springer: Netherlands, 2014; Vol. 77, pp 83–91. doi:10.1007/978-94-007-7920-4_7 |
| 71. | Havaux, M. Plant J. 2014, 79, 597–606. doi:10.1111/tpj.12386 |
| 364. | Greatrex, B. W.; Jenkins, N. F.; Taylor, D. K.; Tiekink, E. R. T. J. Org. Chem. 2003, 68, 5205–5210. doi:10.1021/jo0300845 |
| 519. | Sutbeyaz, Y.; Seçen, H.; Balci, M. J. Org. Chem. 1988, 53, 2312–2317. doi:10.1021/jo00245a034 |
| 400. | Elbs, K. J. Prakt. Chem. 1893, 48, 179–185. doi:10.1002/prac.18930480123 |
| 401. | Wang, Z. Elbs Persulfate Oxidation. Comprehensive Organic Name Reactions and Reagents; John Wiley & Sons, 2010; pp 977–981. doi:10.1002/9780470638859.conrr212 |
| 40. | O'Neill, P. M.; Stocks, P. A.; Pugh, M. D.; Araujo, N. C.; Korshin, E. E.; Bickley, J. F.; Ward, S. A.; Bray, P. G.; Pasini, E.; Davies, J.; Verissimo, E.; Bachi, M. D. Angew. Chem., Int. Ed. 2004, 43, 4193–4197. doi:10.1002/anie.200453859 |
| 399. | Roy, A.; Reddy, K. R.; Mohanta, P. K.; Ila, H.; Junjappat, H. Synth. Commun. 1999, 29, 3781–3791. doi:10.1080/00397919908086017 |
| 35. | Martyn, D. C.; Ramirez, A. P.; Berattie, M. J.; Cortese, J. F.; Patel, V.; Rush, M. A.; Woerpel, K. A.; Clardy, J. Bioorg. Med. Chem. Lett. 2008, 18, 6521–6524. doi:10.1016/j.bmcl.2008.10.083 |
| 403. | Behrman, E. C.; Chen, S.; Behrman, E. J. Tetrahedron Lett. 2002, 43, 3221–3224. doi:10.1016/S0040-4039(02)00495-1 |
| 404. | Capdevielle, P.; Maumy, M. Tetrahedron Lett. 1982, 23, 1573–1576. doi:10.1016/S0040-4039(00)87161-0 |
| 405. | Sethna, S. M. Chem. Rev. 1951, 49, 91–101. doi:10.1021/cr60152a002 |
| 44. | Amewu, R.; Gibbons, P.; Mukhtar, A.; Stachulski, A. V.; Ward, S. A.; Hall, C.; Rimmer, K.; Davies, J.; Vivas, L.; Bacsa, J.; Mercer, A. E.; Nixon, G.; Stocks, P. A.; O'Neill, P. M. Org. Biomol. Chem. 2010, 8, 2068–2077. doi:10.1039/b924319d |
| 402. | Behrman, E. J. Beilstein J. Org. Chem. 2006, 2, No. 22. doi:10.1186/1860-5397-2-22 |
| 534. | Dong, Y.; Wittlin, S.; Sriraghavan, K.; Chollet, J.; Charman, S. A.; Charman, W. N.; Scheurer, C.; Urwyler, H.; Santo Tomas, J.; Snyder, C.; Creek, D. J.; Morizzi, J.; Koltun, M.; Matile, H.; Wang, X.; Padmanilayam, M.; Tang, Y.; Dorn, A.; Brun, R.; Vennerstrom, J. L. J. Med. Chem. 2010, 53, 481–491. doi:10.1021/jm901473s |
| 535. | Uhlemann, A.-C.; Wittlin, S.; Matile, H.; Bustamante, L. Y.; Krishna, S. Antimicrob. Agents Chemother. 2007, 51, 667–672. doi:10.1128/AAC.01064-06 |
| 536. | Vennerstrom, J. L.; Arbe-Barnes, S.; Brun, R.; Charman, S. A.; Chiu, F. C. K.; Chollet, J.; Dong, Y.; Dorn, A.; Hunziker, D.; Matile, H.; McIntosh, K.; Padmanilayam, M.; Santo Tomas, J.; Scheurer, C.; Scorneaux, B.; Tang, Y.; Urwyler, H.; Wittlin, S.; Charman, W. N. Nature 2004, 430, 900–904. doi:10.1038/nature02779 |
| 396. | Fache, M.; Darroman, E.; Besse, V.; Auvergne, R.; Caillol, S.; Boutevin, B. Green Chem. 2014, 16, 1987–1998. doi:10.1039/c3gc42613k |
| 524. | Suzuki, M.; Oda, Y.; Hamanaka, N.; Noyori, R. Heterocycles 1990, 30, 517–535. doi:10.3987/COM-89-S45 |
| 525. | Suzuki, M.; Oda, Y.; Noyori, R. Tetrahedron Lett. 1981, 22, 4413–4416. doi:10.1016/S0040-4039(01)82971-3 |
| 395. | Faure, E.; Falentin-Daudré, C.; Jérôme, C.; Lyskawa, J.; Fournier, D.; Woisel, P.; Detrembleur, C. Prog. Polym. Sci. 2013, 38, 236–270. doi:10.1016/j.progpolymsci.2012.06.004 |
| 522. | Barbarow, J. E.; Miller, A. K.; Trauner, D. Org. Lett. 2005, 7, 2901–2903. doi:10.1021/ol050831f |
| 523. | Miller, A. K.; Trauner, D. Angew. Chem., Int. Ed. 2005, 44, 4602–4606. doi:10.1002/anie.200500488 |
| 398. | Matsumoto, M.; Kobayashi, H.; Hotta, Y. J. Org. Chem. 1984, 49, 4740–4741. doi:10.1021/jo00198a037 |
| 12. | Slack, R. D.; Jacobine, A. M.; Posner, G. H. Med. Chem. Commun. 2012, 3, 281–297. doi:10.1039/c2md00277a |
| 16. | Opsenica, D. M.; Šolaja, B. A. J. Serb. Chem. Soc. 2009, 74, 1155–1193. doi:10.2298/JSC0911155O |
| 528. | Chaturvedi, D.; Goswami, A.; Saikia, P. P.; Barua, N. C.; Rao, P. G. Chem. Soc. Rev. 2010, 39, 435–454. doi:10.1039/B816679J |
| 529. | Chen, H.-J.; Han, W.-B.; Hao, H.-D.; Wu, Y. Tetrahedron 2013, 69, 1112–1114. doi:10.1016/j.tet.2012.11.056 |
| 530. | Opsenica, D. M.; Solaja, B. A. Maced. J. Chem. Chem. Eng. 2012, 31, 137–182. |
| 531. | Schmid, G.; Hofheinz, W. J. Am. Chem. Soc. 1983, 105, 624–625. doi:10.1021/ja00341a054 |
| 532. | Yadav, J. S.; Thirupathaiah, B.; Srihari, P. Tetrahedron 2010, 66, 2005–2009. doi:10.1016/j.tet.2010.01.051 |
| 533. | Zhu, C.; Cook, S. P. J. Am. Chem. Soc. 2012, 134, 13577–13579. doi:10.1021/ja3061479 |
| 397. | Fache, M.; Boutevin, B.; Caillol, S. Green Chem. 2016, 18, 712–725. doi:10.1039/C5GC01070E |
| 526. | The Research Group of Artemisinin Structure. Chin. Sci. Bull. 1977, 22, 142. |
| 527. | The Qinghaosu Antimalarial Coordinating Research Group. Chin. Med. J. (Beijing, China, Engl. Ed.) 1979, 92, 811–816. |
| 192. | Baeyer, A.; Villiger, V. Ber. Dtsch. Chem. Ges. 1899, 32, 3625–3633. doi:10.1002/cber.189903203151 |
| 193. | Baeyer, A.; Villiger, V. Ber. Dtsch. Chem. Ges. 1900, 33, 858–864. doi:10.1002/cber.190003301153 |
| 194. | Belov, V. N.; Heyfitz, L. A.; Virezub, S. I. Okislenie karbonil'nykh soedineniy perekis'yu vodoroda i nadkislotami (reaktsiya Bayera–Villigera). Reaczii I Metodi issledovaniya organicheskich soedineniy; Goshimizdat: Moscow, 1961; Vol. 10, pp 7–208. |
| 185. | Renz, M.; Meunier, B. Eur. J. Org. Chem. 1999, 737–750. doi:10.1002/(SICI)1099-0690(199904)1999:4<737::AID-EJOC737>3.0.CO;2-B |
| 194. | Belov, V. N.; Heyfitz, L. A.; Virezub, S. I. Okislenie karbonil'nykh soedineniy perekis'yu vodoroda i nadkislotami (reaktsiya Bayera–Villigera). Reaczii I Metodi issledovaniya organicheskich soedineniy; Goshimizdat: Moscow, 1961; Vol. 10, pp 7–208. |
| 195. | Krow, G. R. The Baeyer–Villiger Oxidation of Ketones and Aldehydes. In Organic Reactions; Paquette, L. A., Ed.; John Wiley & Sons, Inc., 1993; Vol. 43, pp 251–798. doi:10.1002/0471264180.or043.03 |
| 183. |
Yablokov, V. A. Russ. Chem. Rev. 1980, 49, 833–842. doi:10.1070/RC1980v049n09ABEH002509
Usp. Khim. 1980, 49, 1711–1729. |
| 503. | Suzuki, M.; Ohtake, H.; Kameya, Y.; Hamanaka, N.; Noyori, R. J. Org. Chem. 1989, 54, 5292–5302. doi:10.1021/jo00283a023 |
| 520. | Durie, A. J.; Slawin, A. M. Z.; Lebl, T.; Kirsch, P.; O'Hagan, D. Chem. Commun. 2011, 47, 8265–8267. doi:10.1039/c1cc13016a |
| 521. | Sar, A.; Lindeman, S.; Donaldson, W. A. Org. Biomol. Chem. 2010, 8, 3908–3917. doi:10.1039/c004730a |
| 190. | Wang, Z. Baeyer–Villiger Oxidation. Comprehensive Organic Name Reactions and Reagents; John Wiley & Sons, 2010; pp 150–155. doi:10.1002/9780470638859.conrr036 |
| 191. | Hassall, C. H. The Baeyer-Villiger Oxidation of Aldehydes and Ketones. In Organic Reactions; Adams, R., Ed.; John Wiley & Sons, Inc.: New York, 2011. doi:10.1002/0471264180.or009.03 |
| 183. |
Yablokov, V. A. Russ. Chem. Rev. 1980, 49, 833–842. doi:10.1070/RC1980v049n09ABEH002509
Usp. Khim. 1980, 49, 1711–1729. |
| 184. | Dussault, P. Reactions of Hydroperoxides and Peroxides. In Active Oxygen in Chemistry; Foote, C. S.; Valentine, J.; Greenberg, A.; Liebman, J., Eds.; Springer: Netherlands, 1995; Vol. 2, pp 141–203. doi:10.1007/978-94-007-0874-7_5 |
| 185. | Renz, M.; Meunier, B. Eur. J. Org. Chem. 1999, 737–750. doi:10.1002/(SICI)1099-0690(199904)1999:4<737::AID-EJOC737>3.0.CO;2-B |
| 186. | Davies, A. G. J. Chem. Res. 2009, 533–544. doi:10.3184/030823409X12491375725131 |
| 187. |
Syrkin, Ya. K.; Moiseev, I. I. Russ. Chem. Rev. 1960, 29, 193–214. doi:10.1070/RC1960v029n04ABEH001227
Usp. Khim. 1960, 29, 193. |
| 188. | Ishmuratov, G. Yu.; Legostaeva, Yu. V.; Botsman, L. P.; Tolstikov, G. A. Russ. J. Org. Chem. 2010, 46, 1593–1621. doi:10.1134/S1070428010110011 |
| 189. | Smith, M. B. March's Advanced Organic Chemistry: Reactions, Mechanisms, and Structure, 7th ed.; John Wiley & Sons, Inc., 2013; p 2080. |
| 114. | Wang, L.; Sha, W.; Dai, Q.; Feng, X.; Wu, W.; Peng, H.; Chen, B.; Cheng, J. Org. Lett. 2014, 16, 2088–2091. doi:10.1021/ol500277u |
| 115. | Dragan, A.; Kubczyk, T. M.; Rowley, J. H.; Sproules, S.; Tomkinson, N. C. O. Org. Lett. 2015, 17, 2618–2621. doi:10.1021/acs.orglett.5b00953 |
| 116. | Chen, H.-H.; Wang, G.-Z.; Han, J.; Xu, M.-Y.; Zhao, Y.-Q.; Xu, H.-J. Tetrahedron 2014, 70, 212–217. doi:10.1016/j.tet.2013.11.085 |
| 117. | Gogoi, A.; Modi, A.; Guin, S.; Rout, S. K.; Das, D.; Patel, B. K. Chem. Commun. 2014, 50, 10445–10447. doi:10.1039/C4CC04407J |
| 118. | Wu, X.-F.; Gong, J.-L.; Qi, X. Org. Biomol. Chem. 2014, 12, 5807–5817. doi:10.1039/C4OB00276H |
| 119. | Zhao, J.; Fang, H.; Han, J.; Pan, Y.; Li, G. Adv. Synth. Catal. 2014, 356, 2719–2724. doi:10.1002/adsc.201400032 |
| 120. | Dai, Q.; Jiang, Y.; Yu, J.-T.; Cheng, J. Synthesis 2015, 48, 329–339. doi:10.1055/s-0035-1560536 |
| 121. | Terent’ev, A. O.; Borisov, D. A.; Chernyshev, V. V.; Nikishin, G. I. J. Org. Chem. 2009, 74, 3335–3340. doi:10.1021/jo900226b |
| 122. | Li, Y.; Hao, H.-D.; Wu, Y. Org. Lett. 2009, 11, 2691–2694. doi:10.1021/ol900811m |
| 123. | Opsenica, D. M.; Pocsfalvi, G.; Milhous, W. K.; Šolaja, B. A. J. Serb. Chem. Soc. 2002, 67, 465–471. doi:10.2298/JSC0207465O |
| 124. | Terent'ev, A. O.; Kutkin, A. V.; Platonov, M. M.; Ogibin, Y. N.; Nikishin, G. I. Tetrahedron Lett. 2003, 44, 7359–7363. doi:10.1016/S0040-4039(03)01844-6 |
| 125. | Žmitek, K.; Zupan, M.; Stavber, S.; Iskra, J. J. Org. Chem. 2007, 72, 6534–6540. doi:10.1021/jo0708745 |
| 126. | Ghorai, P.; Dussault, P. H. Org. Lett. 2008, 10, 4577–4579. doi:10.1021/ol801859c |
| 127. | Amewu, R.; Stachulski, A. V.; Ward, S. A.; Berry, N. G.; Bray, P. G.; Davies, J.; Labat, G.; Vivas, L.; O'Neill, P. M. Org. Biomol. Chem. 2006, 4, 4431–4436. doi:10.1039/b613565j |
| 128. | Landenberger, K. B.; Bolton, O.; Matzger, A. J. Angew. Chem., Int. Ed. 2013, 52, 6468–6471. doi:10.1002/anie.201302814 |
| 129. | Climent, E.; Gröninger, D.; Hecht, M.; Walter, M. A.; Martinez-Máñez, R.; Weller, M. G.; Sancenón, F.; Amorós, P.; Rurack, K. Chem. – Eur. J. 2013, 19, 4117–4122. doi:10.1002/chem.201300031 |
| 130. | Terent'ev, A. O.; Borisov, D. A.; Yaremenko, I. A.; Chernyshev, V. V.; Nikishin, G. I. J. Org. Chem. 2010, 75, 5065–5071. doi:10.1021/jo100793j |
| 131. | Terent'ev, A. O.; Borisov, D. A.; Semenov, V. V.; Chernyshev, V. V.; Dembitsky, V. M.; Nikishin, G. I. Synthesis 2011, 2091–2100. doi:10.1055/s-0030-1260027 |
| 132. | Singh, C.; Srivastav, N. C.; Puri, S. K. Bioorg. Med. Chem. 2004, 12, 5745–5752. doi:10.1016/j.bmc.2004.08.042 |
| 133. | Griesbeck, A. G.; El-Idreesy, T. T.; Höinck, L.-O.; Lex, J.; Brun, R. Bioorg. Med. Chem. Lett. 2005, 15, 595–597. doi:10.1016/j.bmcl.2004.11.043 |
| 134. | Singh, C.; Pandey, S.; Saxena, G.; Srivastava, N.; Sharma, M. J. Org. Chem. 2006, 71, 9057–9061. doi:10.1021/jo061414z |
| 135. | Liu, Y.-H.; Deng, J.; Gao, J.-W.; Zhang, Z.-H. Adv. Synth. Catal. 2012, 354, 441–447. doi:10.1002/adsc.201100561 |
| 136. | Griesbeck, A. G.; Schlundt, V. Synlett 2011, 2430–2432. doi:10.1055/s-0030-1261225 |
| 137. | Kazakov, D. V.; Kazakova, O. B.; Ishmuratov, G. Yu.; Terent'ev, A. O.; Nikishin, G. I.; Tolstikov, G. A. Dokl. Chem. 2011, 436, 34–38. doi:10.1134/S0012500811010022 |
| 138. | Sashidhara, K. V.; Avula, S. R.; Singh, L. R.; Palnati, G. R. Tetrahedron Lett. 2012, 53, 4880–4884. doi:10.1016/j.tetlet.2012.07.001 |
| 139. | Hall, J. F. B.; Bourne, R. A.; Han, X.; Earley, J. H.; Poliakoff, M.; George, M. W. Green Chem. 2013, 15, 177–180. doi:10.1039/C2GC36711D |
| 140. | Terent'ev, A. O.; Yaremenko, I. A.; Vil', V. A.; Moiseev, I. K.; Kon'kov, S. A.; Dembitsky, V. M.; Levitsky, D. O.; Nikishin, G. I. Org. Biomol. Chem. 2013, 11, 2613–2623. doi:10.1039/c3ob27239g |
| 141. | Terent'ev, A. O.; Yaremenko, I. A.; Chernyshev, V. V.; Dembitsky, V. M.; Nikishin, G. I. J. Org. Chem. 2012, 77, 1833–1842. doi:10.1021/jo202437r |
| 142. | Terent'ev, A. O.; Yaremenko, I. A.; Vil', V. A.; Dembitsky, V. M.; Nikishin, G. I. Synthesis 2013, 45, 246–250. doi:10.1055/s-0032-1317895 |
| 143. | Nesvadba, P. Radical Polymerization in Industry. Encyclopedia of Radicals in Chemistry, Biology and Materials; John Wiley & Sons, 2012. doi:10.1002/9781119953678.rad080 |
| 144. | Moad, G.; Solomon, D. H. The chemistry of radical polymerization, 2nd ed.; Elsevier, 2006. |
| 145. | Ebewele, R. O. Polymer Science and Technology; CRC Press: Boca Raton, New York, 2000. doi:10.1201/9781420057805 |
| 146. | Mark, H. F. Encyclopedia of Polymer Science and Technology, 3rd ed.; John Wiley & Sons, 2007. |
| 147. | Matyjaszewski, K.; Davis, T. P. Future Outlook and Perspectives. Handbook of Radical Polymerization; John Wiley & Sons, 2003; pp 895–900. doi:10.1002/0471220450.ch16 |
| 148. | Hao, H.-D.; Li, Y.; Han, W.-B.; Wu, Y. Org. Lett. 2011, 13, 4212–4215. doi:10.1021/ol2015434 |
| 149. | Li, Y.; Hao, H.-D.; Zhang, Q.; Wu, Y. Org. Lett. 2009, 11, 1615–1618. doi:10.1021/ol900262t |
| 150. | Yan, X.; Chen, J.; Zhu, Y.-T.; Qiao, C. Synlett 2011, 2827–2830. doi:10.1055/s-0031-1289864 |
| 151. | Žmitek, K.; Zupan, M.; Stavber, S.; Iskra, J. Org. Lett. 2006, 8, 2491–2494. doi:10.1021/ol060590r |
| 152. | Azarifar, D.; Khosravi, K.; Soleimanei, F. Synthesis 2009, 2553–2556. doi:10.1055/s-0029-1217394 |
| 153. | Terent'ev, A. O.; Kutkin, A. V.; Platonov, M. M.; Vorontsov, I. I.; Antipin, M. Yu.; Ogibin, Yu. N.; Nikishin, G. I. Russ. Chem. Bull. 2004, 53, 681–687. doi:10.1023/B:RUCB.0000035657.58776.cc |
| 154. | Zhang, Q.; Jin, H.-X.; Liu, H.-H.; Wu, Y.-K. Chin. J. Chem. 2006, 24, 1190–1195. doi:10.1002/cjoc.200690222 |
| 155. | Zhang, Q.; Li, Y.; Wu, Y.-K. Chin. J. Chem. 2007, 25, 1304–1308. doi:10.1002/cjoc.200790242 |
| 156. | Terent'ev, A. O.; Kutkin, A. V.; Platonov, M. M.; Starikova, Z. A.; Ogibin, Yu. N.; Nikishin, G. I. Russ. Chem. Bull. 2005, 54, 1214–1218. doi:10.1007/s11172-005-0383-4 |
| 157. | Terent'ev, A. O.; Kutkin, A. V.; Starikova, Z. A.; Antipin, M. Yu.; Ogibin, Yu. N.; Nikishin, G. I. Synthesis 2004, 2356–2366. doi:10.1055/s-2004-831171 |
| 158. | Hamann, H.-J.; Hecht, M.; Bunge, A.; Gogol, M.; Liebscher, J. Tetrahedron Lett. 2011, 52, 107–111. doi:10.1016/j.tetlet.2010.10.151 |
| 159. | Terent'ev, A. O.; Platonov, M. M.; Sonneveld, E. J.; Peschar, R.; Chernyshev, V. V.; Starikova, Z. A.; Nikishin, G. I. J. Org. Chem. 2007, 72, 7237–7243. doi:10.1021/jo071072c |
| 160. | Terent'ev, A. O.; Platonov, M. M.; Krylov, I. B.; Chernyshev, V. V.; Nikishin, G. I. Org. Biomol. Chem. 2008, 6, 4435–4441. doi:10.1039/b809661a |
| 161. | Ito, T.; Tokuyasu, T.; Masuyama, A.; Nojima, M.; McCullough, K. J. Tetrahedron 2003, 59, 525–536. doi:10.1016/S0040-4020(02)01556-9 |
| 162. | Terent'ev, A. O.; Borisov, A. M.; Platonov, M. M.; Starikova, Z. A.; Chernyshev, V. V.; Nikishin, G. I. Synthesis 2009, 4159–4166. doi:10.1055/s-0029-1217062 |
| 163. | Eliasen, A. M.; Thedford, R. P.; Claussen, K. R.; Yuan, C.; Siegel, D. Org. Lett. 2014, 16, 3628–3631. doi:10.1021/ol501497y |
| 164. | Hu, X.; Maimone, T. J. J. Am. Chem. Soc. 2014, 136, 5287–5290. doi:10.1021/ja502208z |
| 165. | Gamage, N.-D. H.; Stiasny, B.; Stierstorfer, J.; Martin, P. D.; Klapötke, T. M.; Winter, C. H. Chem. Commun. 2015, 51, 13298–13300. doi:10.1039/C5CC05015D |
| 166. | Starkl Renar, K.; Pečar, S.; Iskra, J. Org. Biomol. Chem. 2015, 13, 9369–9372. doi:10.1039/C5OB01503K |
| 167. | Rozhko, E.; Solmi, S.; Cavani, F.; Albini, A.; Righi, P.; Ravelli, D. J. Org. Chem. 2015, 80, 6425–6431. doi:10.1021/acs.joc.5b00861 |
| 168. | Hilf, J. A.; Witthoft, L. W.; Woerpel, K. A. J. Org. Chem. 2015, 80, 8262–8267. doi:10.1021/acs.joc.5b01326 |
| 169. | Lu, X.; Deng, L. Org. Lett. 2014, 16, 2358–2361. doi:10.1021/ol500677v |
| 170. | Fisher, T. J.; Mattson, A. E. Org. Lett. 2014, 16, 5316–5319. doi:10.1021/ol502494h |
| 171. | Wang, X.; Pan, Y.; Huang, K.-W.; Lai, Z. Org. Lett. 2015, 17, 5630–5633. doi:10.1021/acs.orglett.5b02881 |
| 172. | Yu, H.; Shen, J. Org. Lett. 2014, 16, 3204–3207. doi:10.1021/ol5012168 |
| 173. | Carney, J. M.; Hammer, R. J.; Hulce, M.; Lomas, C. M.; Miyashiro, D. Synthesis 2012, 44, 2560–2566. doi:10.1055/s-0031-1289764 |
| 174. | Yan, X.; Qiao, C.; Guo, Z. Synlett 2013, 24, 502–506. doi:10.1055/s-0032-1318213 |
| 175. | Hurlocker, B.; Miner, M. R.; Woerpel, K. A. Org. Lett. 2014, 16, 4280–4283. doi:10.1021/ol5020015 |
| 176. | Liu, Q.; Wang, W.; Liu, Z.; Wang, T.; Wu, L.; Ge, M. RSC Adv. 2014, 4, 19716–19724. doi:10.1039/C4RA02486A |
| 177. | Parrish, J. D.; Ischay, M. A.; Lu, Z.; Guo, S.; Peters, N. R.; Yoon, T. P. Org. Lett. 2012, 14, 1640–1643. doi:10.1021/ol300428q |
| 178. | Wei, W.; Zhang, C.; Xu, Y.; Wan, X. Chem. Commun. 2011, 47, 10827–10829. doi:10.1039/c1cc14602e |
| 179. | Daeppen, C.; Kaiser, M.; Neuburger, M.; Gademann, K. Org. Lett. 2015, 17, 5420–5423. doi:10.1021/acs.orglett.5b02773 |
| 180. | Kazakova, O. B.; Kazakov, D. V.; Yamansarov, E. Yu.; Medvedeva, N. I.; Tolstikov, G. A.; Suponitsky, K. Yu.; Arkhipov, D. E. Tetrahedron Lett. 2011, 52, 976–979. doi:10.1016/j.tetlet.2010.12.047 |
| 181. | Lau, S.-H.; Galván, A.; Merchant, R. R.; Battilocchio, C.; Souto, J. A.; Berry, M. B.; Ley, S. V. Org. Lett. 2015, 17, 3218–3221. doi:10.1021/acs.orglett.5b01307 |
| 182. | Feng, Y.; Holte, D.; Zoller, J.; Umemiya, S.; Simke, L. R.; Baran, P. S. J. Am. Chem. Soc. 2015, 137, 10160–10163. doi:10.1021/jacs.5b07154 |
© 2016 Yaremenko et al.; licensee Beilstein-Institut.
This is an Open Access article under the terms of the Creative Commons Attribution License (http://creativecommons.org/licenses/by/2.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
The license is subject to the Beilstein Journal of Organic Chemistry terms and conditions: (http://www.beilstein-journals.org/bjoc)