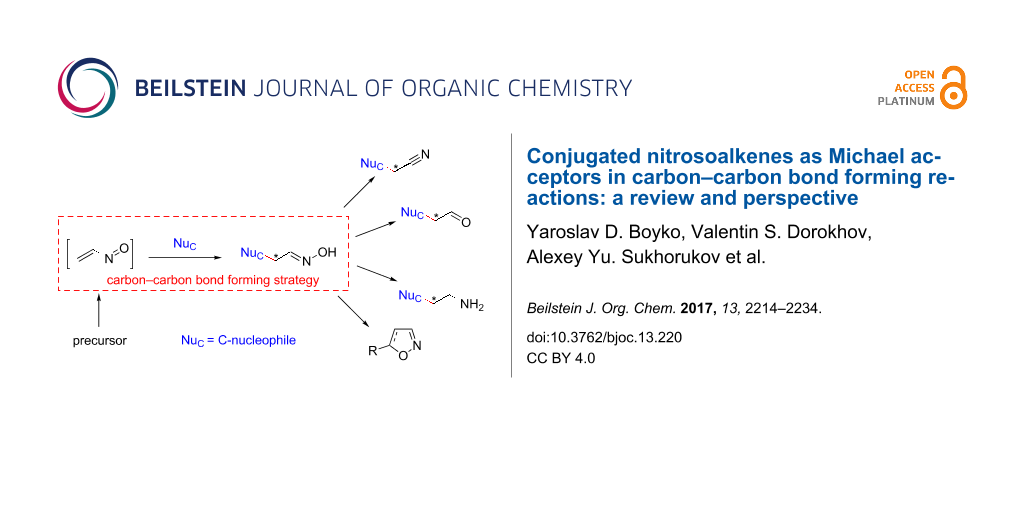
Abstract
Despite of their chemical instability and high reactivity, conjugated nitrosoalkenes are useful intermediates in target-oriented organic synthesis. The present review deals with carbon–carbon bond forming reactions involving Michael addition to α-nitrosoalkenes with a particular focus on recent developments in this methodology and its use in total synthesis.
Graphical Abstract
Introduction
Conjugated nitrosoalkenes (NSA) are close analogs of α-nitroalkenes, which are important Michael acceptors in organic synthesis [1-5]. Unlike the parent α-nitroalkenes, α-nitrosoalkenes are unstable species usually generated in situ, and their synthetic potential has been studied in significantly less detail [6,7]. Because of relatively short lifetimes of α-nitrosoalkenes, conjugate addition of nucleophiles is complicated by side reactions, such as polymerization. Successful coupling of nitrosoalkenes with nucleophiles is highly challenging and depends on many factors such as temperature, concentration, solvent and, especially, the nature of the α-nitrosoalkene precursor. Nevertheless, Michael addition of C-nucleophiles to α-nitrosoalkenes opens access to synthetically valuable α-branched oximes, which can be further utilized as useful intermediates in total synthesis. The high potential of this carbon–carbon bond-forming strategy has been recognized since 1970s in works of Corey, Oppolzer, Gilchrist and others. However, at that time, researchers faced substantial problems associated with low chemoselectivity of reactions with nitrosoalkenes generated in situ from the corresponding α-halooxime precursors as well as with the need of a large access of the nucleophile.
In the recent years, there has been a considerable growth of interest to the conjugate addition of carbon-centered nucleophiles to α-nitrosoalkenes. This is mainly due to the development of new precursors, from which nitrosoalkenes are generated at low stationary concentrations under very mild conditions that allows to suppress side reactions and to use a stoichiometric amount of nucleophile. Under these conditions, Michael addition reactions with nitrosoalkenes can be realized in an intramolecular fashion that opens access to complex bridged and fused carbocyclic frameworks bearing several contiguous stereogenic centers.
In this review, we attempted to summarize up-to-date data on the successful coupling of α-nitrosoalkenes with C-nucleophiles (classified according to a nucleophile type) emphasizing on examples from target-oriented synthesis as well as to analyze growing points in this methodology. The review does not deal with cycloaddition reactions of nitrosoalkenes. For this aspect of nitrosoalkene chemistry, the reader can be referred to earlier general reviews [6-10].
Review
Precursors of conjugated nitrosoalkenes
The way how α-nitrosoalkenes are generated is essential for achieving a successful coupling with a particular nucleophile and for prevention of side reactions. There are several sources of α-nitrosoalkenes as shown in Scheme 1. Conventional precursors of α-nitrosoalkenes are α-halooximes 1 (or halo-substituted nitroso compounds [11,12]), which undergo deprotonation/halide elimination upon treatment with a base [13] (Scheme 1, reaction (1)). A disadvantage of this method is that NSA are generated fast and high stationary concentrations are achieved facilitating polymerization. Furthermore, an excess of nucleophile is needed since it also serves as a base. A milder method was suggested by Denmark, who used α-halooximes silyl ehers 2 as precursors of conjugated nitrosoalkenes upon treatment with TBAF at low temperatures (Scheme 1, reaction (2)) [14-16]. Importantly, the rate of nitrosoalkene formation can be controlled by the bulkiness of the silyl group. Another way of nitrosoalkene generation employs their stable nitrosoacetals of type 3 (N,N-bis(silyloxy)enamines) [17,18]. The latter eliminate (Alk3Si)2O upon the action of Lewis bases generating the corresponding nitrosoalkenes at low stationary concentrations in a controllable manner. Furthermore, synthetic precursors of nitrosoacetals 3 are not halocarbonyl compounds as in the case of oximes 1 and 2, but aliphatic nitro derivatives, which are readily transformed into enamines 3 by double silylation involving an internal redox process [19-21]. This allows the preparation of nitrosoalkenes, which are difficult to access by the first two routes. Tanimoto et al. [22] recently suggested N-siloxysulfonamides 4 as stable sources of unsaturated nitroso compounds (Scheme 1, reaction (4)). However, this approach was used only for the generation of nitrosoallenes so far.
Scheme 1: Precursors of nitrosoalkenes NSA.
Scheme 1: Precursors of nitrosoalkenes NSA.
Addition of C–H acids anions to conjugated nitrosoalkenes
Stabilized enolates and their equivalents are the most studied C-nucleophiles in Michael addition reactions with nitrosoalkenes. A first systematic study in this area was done by Ohno and co-authors [13,23], who reported the addition of diethyl malonate and acetylacetone anions to cyclic nitrosoalkenes NSA1a–c generated from the corresponding α-chloroketones (2-chlorocyclohexanone oxime (1a), 2-chlorocyclooctanone oxime (1b) and 12-chloro-сyclododeca-4,8-dien-1-one oxime (1c)) upon the action of an excess of nucleophile (Scheme 2). It was found that oximes 1b and 1c react both with diethyl malonate and acetylacetone, providing adducts 5 in high to quantitative yields. Six-membered cyclic oxime 1a afforded the corresponding products only in moderate yields owing to the formation of undesired side products and polymerization of the α,β-unsaturated nitroso compound. This early example demonstrates the high dependency of Michael addition to nitrosoalkenes on the substrate structure. However, the need of an excess of nucleophile significantly limits the application of this protocol.
Scheme 2: Reactions of cyclic α-chlorooximes 1 with 1,3-dicarbonyl compounds.
Scheme 2: Reactions of cyclic α-chlorooximes 1 with 1,3-dicarbonyl compounds.
The development of improved methods for the generation of α-nitrosoalkenes have led to more extensive studies of their reactions with enolates. Thus, our group has demonstrated that nitrosoacetals of nitrosoalkenes (N,N-bis(silyloxy)enamines 3) smoothly react with 1,3-dicarbonyl compounds in the presence of TBAF or DBU to give the corresponding oximes 6 in moderate to good yields (Scheme 3) [24]. The process is likely to involve a nitrosoalkene intermediate, which is generated upon the action of TBAF or the anion of the 1,3-dicarbonyl compound on N,N-bis(silyloxy)enamine 3. Unlike malonic acid derivatives and β-keto esters, 1,3-diketones produced only complex mixtures of products in the reaction with N,N-bis(silyloxy)enamines 3 under these conditions.
Scheme 3: C-C-coupling of N,N-bis(silyloxy)enamines 3 with 1,3-dicarbonyl compounds.
Scheme 3: C-C-coupling of N,N-bis(silyloxy)enamines 3 with 1,3-dicarbonyl compounds.
Nitronate anions also react as C-nucleophiles with N,N-bis(silyloxy)enamines 3 producing β-nitrooximes 7 in good yields (Scheme 4) [20,25]. Efficient coupling of DBU-nitronate salts derived from primary nitro compounds with enamines 3 was achieved at –78 °C upon the action of TBAF. However, for less reactive secondary nitronates reactions were carried out at 0 °C without TBAF in order to keep the concentration of reactive nitrosoalkenes lower.
Scheme 4: Reaction of N,N-bis(silyloxy)enamines 3 with nitronate anions.
Scheme 4: Reaction of N,N-bis(silyloxy)enamines 3 with nitronate anions.
In 2010, Weinreb and co-workers developed a general protocol for coupling α-nitrosoalkenes with carboxylic acid esters bearing electron withdrawing substituents (CO2Et, NO2, COCH3, SO2Ph) at the α-position (Scheme 5) [26]. Nitrosoalkenes were generated according to Denmark’s procedure [14] upon the action of TBAF on TBS ethers of α-chlorooximes 2. Importantly, TBS oxime ethers of various halo-substituted aldehydes and ketones including cyclic ones were successfully used as nitrosoalkenes precursors to give oximes 8 in good to high yields.
Scheme 5: Reaction of α-chlorooximes TBS ethers 2 with ester enolates.
Scheme 5: Reaction of α-chlorooximes TBS ethers 2 with ester enolates.
The same authors used the advantage of intramolecular Michael addition of enolates to nitrosoalkenes to construct bridged and fused carbocyclic systems [27]. For example, the proposed strategy of bicyclo[3.2.1]octanone 14 assembly is based on a combination of ring closing metathesis reaction and intramolecular addition of the malonate anion to a nitrosoalkene unit (Scheme 6). On the first stage, readily available chlorodiene 9 was subjected to a metathesis reaction with the second-generation Grubbs’ catalyst. The resulting chlorocyclohexene derivative 10 was oxidized to α-chloroketone 11, which was subsequently transformed into α-chlorooxime TBS ether 12 by standard oximation reaction. The required nitrosoalkene intermediate was generated from α-halo-O-silyloxime 12 upon the action of fluoride anion at −78 °C. It is worth paying attention that the malonate anion is generated prior the addition of TBAF, because of a high instability of the intermediate nitroso compound NSA2. Using this procedure, bicyclic oxime 13 was prepared in quantitative yield from precursor 12. Modification of this strategy allows accessing various bicyclic systems comprising of five-, six- and seven-membered carbo- and heterocycles.
Scheme 6: Assembly of bicyclooctanone 14 via an intramolecular cyclization of nitrosoalkene NSA2.
Scheme 6: Assembly of bicyclooctanone 14 via an intramolecular cyclization of nitrosoalkene NSA2.
Not only malonates, but also other CH-acidic fragments such as dicyanomethane and sulfonyl-, nitro- and phosphorylacetic acid residues can be successfully used in the strategy shown in Scheme 6 [28]. This general synthetic scheme opens access to a variety of substituted bicyclo[2.2.1]heptanes 16 via cyclization of α-chlorooxime TBS ethers 15 as shown in Scheme 7.
Scheme 7: A general strategy for the assembly of bicyclo[2.2.1]heptanes via an intramolecular cyclization of α-chlorooxime TBS ethers 15.
Scheme 7: A general strategy for the assembly of bicyclo[2.2.1]heptanes via an intramolecular cyclization of ...
The stereoselectivity of Michael additions to conjugated nitrosoalkenes is an important issue considering the potential use of this strategy in the synthesis of natural products. Weinreb and co-workers performed a comprehensive investigation of the stereoselectivity of the conjugate addition to α-nitrosoalkenes using both cyclic [29] and acyclic substrates [30]. They have demonstrated that the nucleophilic addition of C-substituted malonic esters 17 to 4-tert-butylnitrosocyclohexene NSA3 (generated either from TBS ether 18 or free oxime 19) proceeds with high stereoselectivity and affords mainly or exclusively the trans-isomers of adducts 20 (Scheme 8) [29]. Expectedly, the stereoselectivity of the Michael addition was not influenced by the method of nitrosoalkene generation as well as by the relative configuration of the α-chlorooxime. The observed stereochemical result can be attributed to an axial attack of the nucleophile on the nitrosoalkene NSA3.
Scheme 8: Stereochemistry of Michael addition to cyclic nitrosoalkene NSA3.
Scheme 8: Stereochemistry of Michael addition to cyclic nitrosoalkene NSA3.
More interestingly, the addition of malonic esters 17 to acyclic nitrosoalkenes NSA4 (generated from oxime TBS ethers 21) resulted exclusively in anti-adducts 22 (Scheme 9) [30]. This stereochemistry is in agreement with the Felkin–Anh model with the most sterically demanding substituent R1 lying in perpendicular plane with respect to the C=C double bond. This pattern is general both for nitrosoalkene NSA4 with phenyl substituent in γ-position (R1 = Ph, Scheme 9) and for the compound with a more sterically demanding neopentyl substituent (R1 = neopentyl, Scheme 9).
Scheme 9: Stereochemistry of Michael addition to acyclic nitrosoalkenes NSA4.
Scheme 9: Stereochemistry of Michael addition to acyclic nitrosoalkenes NSA4.
Similar to nitrosolakenes NSA4, γ-methoxy-substituted nitrosoalkene NSA5 (generated from TBS-oxime ether 23) also gave anti-isomer of adduct 24 (Scheme 10).
Scheme 10: Stereochemistry of Michael addition to γ-alkoxy nitrosoalkene NSA5.
Scheme 10: Stereochemistry of Michael addition to γ-alkoxy nitrosoalkene NSA5.
These model studies demonstrate the high stereoselectivity of the Michael addition to both cyclic and acyclic conjugated nitrosoalkenes possessing asymmetric centers. Yet, the stereochemical induction from a chiral nucleophile as well as asymmetric catalysis still remains to be studied in these reactions.
The high efficiency and stereoselectivity of the enolate addition to nitrosoalkenes make this strategy perspective for application in total synthesis that has been recognized since 1970s. Thus, Oppolzer and co-workers exploited the Michael addition of enolates to nitrosoalkenes in their studies towards syntheses of 3-methoxy-9β-estra(1,3,5(10))trien(11,17)dione (25) (1977) [31] and (+/−)-isocomene (1981) [32]. The key carbon–carbon bond formation step in the synthesis of steroid 25 was the stereoselective reaction of α-bromooxime 26 with lithium enolate 27 to give oxime 28 via the Michael addition to transient nitrosoalkene NSA6 [31]. Oxime adduct 28 existed predominantly in a cyclic 1,2-oxazine form (Scheme 11). Subsequent benzylation of oxime 28 and the thermal retro-[2 + 2]/[4 + 2]-cycloaddition cascade followed by hydrolysis of the oximino group gave target steroid 25 with the desired configuration of all stereogenic centers.
Scheme 11: Oppolzer’s total synthesis of 3-methoxy-9β-estra(1,3,5(10))trien(11,17)dione (25).
Scheme 11: Oppolzer’s total synthesis of 3-methoxy-9β-estra(1,3,5(10))trien(11,17)dione (25).
In the attempted route to (+/−)-isocomene, Oppolzer and co-workers suggested pentalenone 29, accessible by an intramolecular aldol condensation/elimination of diketone 30, as a key precursor. Diketone 30 was prepared in a straightforward manner by C-C-coupling of nitrosoalkene NSA7 (generated in situ from the corresponding α-bromooxime 31) with silyl enolate of 2-methylcyclopentanone, and subsequent deoxygenation of the resulting oxime 32 with CAN. Unfortunately, attempts to convert pentalenone 29 into isocomene failed (Scheme 12) [32].
Scheme 12: Oppolzer’s total synthesis of (+/−)-isocomene.
Scheme 12: Oppolzer’s total synthesis of (+/−)-isocomene.
Recently, Weinreb’s group used the advantage of nitrosoalkenes as Michael acceptors to accomplish total syntheses of several alkaloids, such as (+/−)-alstilobanines A and E, (+/−)-angustilodine [33] and (+/−)-myrioneurinol [34,35] (Figure 1).
Figure 1: Alkaloids synthesized using stereoselective Michael addition to conjugated nitrosoalkenes.
Figure 1: Alkaloids synthesized using stereoselective Michael addition to conjugated nitrosoalkenes.
In the total syntheses of alstilobanines A, E and angustilodine, functionalized indole 33 was initially chosen as a key intermediate [33]. However, attempts of its preparation by Michael addition of substituted indole enolate 34 to nitrosoalkene NSA8, generated in situ either from the corresponding chlorooxime 35 or its TBS ether 36 failed (Scheme 13). The problem was elegantly solved by using indole dianion 37 as both a nucleophile and a base. Reaction of 37 with α-chlorooxime 35 required only one equivalent of nucleophile and produced adduct 38 in 99% yield. Oxime 38 was transformed into a pentacyclic derivative 39, which served as a direct precursor of target alkaloids alstilobanines A, E and angustilodine.
Scheme 13: Weinreb’s total synthesis of alstilobanines A, E and angustilodine.
Scheme 13: Weinreb’s total synthesis of alstilobanines A, E and angustilodine.
The same strategy was employed to construct the tetracyclic core of the apparicine class of indole alkaloids (see Scheme 14) [36]. Here, coupling of indole dianion 40 with chlorooxime 35 (via nitrosoalkene NSA8) furnished the corresponding adduct 41 in 97% yield as a separable 1:1 mixture of diastereomers. Each isomer was subjected to cyclization under reductive conditions to form an indol-fused 1-azabicyclo[4.2.2]decane structure 42.
Scheme 14: Weinreb’s approach to the core structure of apparicine alkaloids.
Scheme 14: Weinreb’s approach to the core structure of apparicine alkaloids.
Conjugate addition of malonate anion to a nitrosoalkene intermediate NSA9 was used as a key stage to construct an important C(7)–C(8) connection in the total synthesis of (+/−)-myrioneurinol (Scheme 15) [34,35]. α-Chloroxime TBS ether 43 (prepared in 10 steps from δ-valerolactam) reacted with a dimethyl malonate anion under Denmark’s conditions via intermediate NSA9 to give the corresponding adduct 44 in 93% yield. Importantly, both stereoisomers obtained were oxime E,Z-isomers with the C-7 relative configuration being the same as in myrioneurinol. Subsequent deoxygenation and reduction of aldoxime followed by transformation of the malonate unit to the formyl group furnished intermediate 45, which was subjected to Wittig olefination. Subsequent aza-Sakurai cyclization of intermediate 46 provided tricyclic derivative 47 as a single stereoisomer, which was then transformed into target myrioneurinol by standard operations.
Scheme 15: Weinreb’s synthesis of (+/−)-myrioneurinol via stereoselective conjugate addition of malonate to nitrosoalkene NSA9.
Scheme 15: Weinreb’s synthesis of (+/−)-myrioneurinol via stereoselective conjugate addition of malonate to ni...
Addition of organometallic reagents to conjugated nitrosoalkenes
The addition of organometallic compounds to conjugated nitrosoalkenes is a promising strategy towards various α-branched oximes. Unfortunately, these reactions are not sufficiently developed to date and only few reports dealing with a successful Michael addition of organometallic compounds to α-nitrosoalkenes were reported so far.
Among the first, Ohno and co-workers studied the addition of Grignard reagents to α-chlorooximes derived from cyclic ketones [23]. In their experiments, 2 equivalents of organomagnesium compound were used with one equivalent needed to transform α-chlorooximes into nitrosoalkene (Scheme 16). Chloro-substituted сyclododeca-4,8-dien-1-one oxime 1c did produce the desired adducts 48 in moderate yields, yet experiments with 2-chlorocyclohexanone oxime (1a) and 2-chlorocyclooctanone oxime (1b) were unsuccessful. This early result demonstrates that classical way to generate conjugated nitrosoalkenes from α-chlorooximes may not be compatible with further conjugate addition of Grignard reagents.
Scheme 16: Reactions of cyclic α-chloro oximes with Grignard reagents.
Scheme 16: Reactions of cyclic α-chloro oximes with Grignard reagents.
Despite of these rather disappointing results, in 1977 Corey successfully employed conjugate addition of 1-lithio-1-butyne to a nitrosoalkene intermediate to introduce the butyl group at C-6 atom in the total synthesis of (+/-)-perhydrohistrionicotoxin (Scheme 17) [37-39]. The key precursor of perhydrohistrionicotoxin, dioxime 51, was prepared by the reaction of α-bromo oxime 49 with 10 equivalents of 1-butyne and 6 equivalents of butyllithium in THF followed by hydrogenation of alkynyl oxime 50. As a result of these transformations, the desired dioxime 51 is obtained in 77% yield. Important to note, that the addition of a nucleophile to nitrosoalkene NSA10 occurred exclusively trans-stereoselective with respect to O-benzyloxy-oxime fragment.
Scheme 17: Corey’s synthesis of (+/−)-perhydrohistrionicotoxin.
Scheme 17: Corey’s synthesis of (+/−)-perhydrohistrionicotoxin.
In their studies towards the total synthesis of erythronolide B (an aglycone of erythromycin B) from intermediate 52, Corey and co-workers suggested the ring opening of α,β-epoxy oximes 53 with Gilman’s reagents as a plausible way to introduce a methyl group at the C-10 position [40] (Scheme 18). Since the addition occurs in α-position and an excess (5 equivalents) of organometallic compound is needed, the reaction is believed to proceed through nitrosoalkene intermediate NSA11. The reaction proved to be efficient yielding the desired β-hydroxyoximes 54 in high yields (80–90%). Interestingly, with cyclohexenone oxime excellent stereoselectivity was observed (exclusively trans isomers were formed), yet substituted cyclohexenone oxime derivatives such as α,β-epoxycarvone and epoxyisophorone oximes produced diastereomeric mixtures of products. Although, it was not reported whether this strategy was helpful in achieving the synthesis of erythronolide B, this result indicates that the organocopper compounds undergo conjugate addition to nitrosoalkenes in much more selective manner as compared to organolithium and organomagnesium reagents.
Scheme 18: Addition of Gilman’s reagents to α,β-epoxy oximes 53.
Scheme 18: Addition of Gilman’s reagents to α,β-epoxy oximes 53.
Later on, Weinreb and co-workers demonstrated that organocopper reagents smoothly react with α-chlorooximes [41]. Thus, the reaction of Gilman’s reagent (2 equivalents) with 4-(tert-butyl)-2-chlorocyclohexan-1-one oxime (19) produced the corresponding α-alkyl- and α-aryl-substituted cyclohexanone oximes 55 in high yields and excellent stereoselectivities (Scheme 19, reaction (1)). After some optimization of the procedure (switching from Gilman’s reagent to aryl lithiocyanocuprates), this method was successfully extended to a wide range of acyclic α-chlorooximes 1 (Scheme 19, reaction (2)). Interestingly, the aryl group, not the cyanide anion is transferred from aryl lithiocyanocuprates to a nitrosoalkene intermediate NSA. However, still two equivalents of the organometallic compound were needed to ensure reaction completion. The resulting α-aryl-substituted aldooximes 56 were without isolation converted into the corresponding nitriles 57 by treatment with DCC in the presence of a py/Et3N mixture.
Scheme 19: Addition of Gilman’s reagents to α-chlorooximes.
Scheme 19: Addition of Gilman’s reagents to α-chlorooximes.
The use of other nitrosoalkene sources (see Scheme 1) in reaction with organometallic compounds is very limited. Thus, Seebach and co-authors reported the addition of organolithium reagents to O-silyl nitronates 58 leading to α-substituted oximes 59 (Scheme 20) [42]. It is likely, that upon treatment with organolithium reagent silyl nitronate 58 is deprotonated to give anion 60, which eliminates the silyloxy anion to form nitrosoalkene NSA12. Conjugate addition of the second equivalent of the organolithium compound furnishes oximes 59. However, oximes 59 are produced in rather poor yields.
Scheme 20: Reaction of silyl nitronate 58 with organolithium reagents via nitrosoalkene NSA12.
Scheme 20: Reaction of silyl nitronate 58 with organolithium reagents via nitrosoalkene NSA12.
In 1996, Trost reported the use of β-ketoxime sulfones 61 in the reaction with lithium acetylides that resulted in formal substitution of the sulfinate group through an elimination–addition mechanism (Scheme 21) [43]. Interestingly, unlike α-halooximes, 1,4-elimination in sulfones 61 to generate nitrosoalkenes NSA13 proceeds only at elevated temperatures (50 °C), that may account for relatively low yields of products 62. Furthermore, the reaction of sulfone 63 with lithium (trimethylsilyl)acetylide furnished only enoxime 64.
Scheme 21: Reaction of β-ketoxime sulfones 61 and 63 with lithium acetylides.
Scheme 21: Reaction of β-ketoxime sulfones 61 and 63 with lithium acetylides.
Apparently, the application of more convenient and selective nitrosoalkene sources (e.g., TBS ethers of α-halooximes 2) in the reaction with organometallic compounds may lead to further developments in this promising methodology.
Addition of arenes to conjugated nitrosoalkenes
Besides common C-nucleophiles, nitrosoalkenes can react with nucleophilic arenes and heteroarenes. Gilchrist and Roberts demonstrated that the reaction of highly electron-rich aromatics with nitrosoalkenes NSA14 affords the corresponding α-aryl-substituted oximes 66 in low to moderate yields as mixtures of regioisomers [44] (Scheme 22). Nitrosoalkenes were generated from the corresponding α-halooximes 65 upon treatment with Na2CO3. The efficiency of the addition reaction depends on both electron enrichment of the aromatic ring and the electron-withdrawing character of the substituent in nitrosoalkene NSA14. 1,3-Dimethoxybenzene gave the corresponding substitution products in highest yield, while anisole proved to be unreactive under these conditions.
Scheme 22: Electrophilic addition of nitrosoalkenes NSA14 to electron-rich arenes.
Scheme 22: Electrophilic addition of nitrosoalkenes NSA14 to electron-rich arenes.
Electron-rich aromatic heterocyclic compounds such as pyrroles and indoles enter reactions with nitrosoalkenes more smoothly [44-55]). The addition of nitrosoalkenes to pyrroles and indoles is a convenient and mild strategy for the functionalization of these heterocyclic systems. As shown in early reports by Gilchrist [44,45,56], oximinoalkylation of these heterocycles with NSA14 provides α-hetaryl-substituted oximes 67 and 68 in high yields and with good regioselectivity (Scheme 23). It should be noted, that the formation of α-heteroaryl oximes 67 and 68 can occur not only via an SEAr mechanism, but also through an alternative [4 + 2]-cycloaddition/elimination pathway (for cycloaddition chemistry of nitrosoalkenes see reviews and accounts: [6-10,57,58]).
Scheme 23: Addition of nitrosoalkenes NSA14 to pyrroles and indoles.
Scheme 23: Addition of nitrosoalkenes NSA14 to pyrroles and indoles.
In the recent work by Palacios and co-workers, the reaction of phosphinyl nitrosoalkenes NSA15 (derived from the corresponding α-bromooximes) with electron-rich nitrogen heterocycles to give adducts 69 is described and the comparison of electrophilic aromatic substitution (1) and cycloaddition (2) routes is discussed [59] (Scheme 24). Quantum-chemical calculations suggest that the [4 + 2]-cycloaddition process (2) is thermodynamically favored for nitrosoalkenes bearing electron-donating group R, whereas the presence of an electron withdrawing group at the same position promotes the electrophilic substitution pathway (1).
Scheme 24: Reaction of phosphinyl nitrosoalkenes NSA15 with indole.
Scheme 24: Reaction of phosphinyl nitrosoalkenes NSA15 with indole.
Pinho e Melo and co-workers developed a one-pot double addition of pyrroles (and indoles) to nitrosoalkenes derived from α,α’-dihalooximes 70 [60-62] (Scheme 25). The synthesis of bis-pyrroles 71 is carried out in water as the solvent. As pointed by the authors, heterogenous conditions can accelerate the rate of reaction compared to organic solvent phase.
Scheme 25: Reaction of pyrrole with α,α’-dihalooximes 70.
Scheme 25: Reaction of pyrrole with α,α’-dihalooximes 70.
The described method of oximinoalkylation of the С-3 position of indoles has a great potential for the synthesis of pharmaceutically relevant compounds and natural products. Thus, de Lera and co-workers described the synthesis of indole-derived psammaplin A analogue 72, which displayed more potent activities than the parent natural product (Scheme 26) [63]. In their synthesis, 5-bromo-1H-indole was converted to the corresponding functionalized oxime 73 upon the action of ethyl bromopyruvate oxime in the presence of Na2CO3 in 60% yield. The adduct was transformed into carboxylic acid 74, which was then used in a double amidation with cystamine to give the target compound 72 after unmasking of oxime. A series of other psammaplin A analogs were prepared in a similar manner.
Scheme 26: Synthesis of indole-derived psammaplin A analogue 72.
Scheme 26: Synthesis of indole-derived psammaplin A analogue 72.
Reduction of the oxime group in oximinoalkylated indoles provides a direct access to various substituted tryptamine derivatives. Thus, reduction of adducts 68 derived from ethyl bromopyruvate oxime and indoles can be considered as a simple and convenient method for the preparation of substituted tryptophanes 75 as shown in Scheme 27 [64].
Scheme 27: Synthesis of tryptophanes by reduction of oximinoalkylated indoles 68.
Scheme 27: Synthesis of tryptophanes by reduction of oximinoalkylated indoles 68.
This synthetic scheme has found several applications in medicinal chemistry. Thus, Park and co-workers successfully employed adducts 68 as intermediates in the synthesis of tumor necrosis factor-α converting enzyme inhibitors 76 [65]. In 2012, Dougherty and co-workers used this approach to prepare 4,7-difluorotryptophan [66].
A similar strategy was successfully applied by Ottenheijm in the total synthesis of an analogue of the tryptophan-containing natural alkaloid neoechinulin B (indole 77) [67,68] (Scheme 28). At the initial stage, N-methylindole was alkylated with ethyl bromopyruvate oxime and sodium carbonate to give adduct 78, which was then transformed into N-hydroxytryptophan derivative 79 using aminolysis and reduction with (CH3)3N·BH3 complex [47,49,69]. Acylation of 79 with pyruvoyl chloride gave amide 80, which was converted into dioxopiperazine 81 upon the action of trifluoroacetic acid. Subsequent water elimination furnished the title neoechinulin B analogue 77.
Scheme 28: Ottenheijm’s synthesis of neoechinulin B analogue 77.
Scheme 28: Ottenheijm’s synthesis of neoechinulin B analogue 77.
Gilchrist [70] employed oximinoalkylation of pyrrole as the initial stage in the synthesis of 1,2-dihydropyrrolizinone antibiotic 82 [71-73] (Scheme 29). The addition of ethyl 2-nitrosoacrylate (generated from ethyl bromopyruvate oxime) to pyrrole under basic conditions afforded product 67a in 61% yield. Subsequent reduction/protection of oxime and selective trifluoroacetylation provided 2,5-disubstituted pyrrole 83, which was cyclized to target dihydropyrrolizin-3-one 82.
Scheme 29: Synthesis of 1,2-dihydropyrrolizinones 82 via addition of pyrrole to ethyl bromopyruvate oxime.
Scheme 29: Synthesis of 1,2-dihydropyrrolizinones 82 via addition of pyrrole to ethyl bromopyruvate oxime.
Kozikowski [74,75] used the advantage of selective oximinoalkylation of indoles to construct the core structure of indolactam-based alkaloids 84 (activators of protein kinase C) as shown in Scheme 30. Employing the same strategy, Webb [76] and Resnick [77] prepared analogues of teleocidin and later Quick [78] accomplished the synthesis of indolactam V.
Scheme 30: Kozikowski’s strategy to indolactam-based alkaloids via addition of indoles to ethyl bromopyruvate oxime.
Scheme 30: Kozikowski’s strategy to indolactam-based alkaloids via addition of indoles to ethyl bromopyruvate ...
Tandem C-nucleophile addition/cyclization processes involving conjugated nitrosoalkenes
With certain C-nucleophiles possessing additionally an electrophilic center a Michael addition to nitrosoalkenes can be followed by a cyclization. These processes are of synthetic value since bioactive five- and six-membered N–O hetereocycles are formed.
A characteristic example of such a process is the Michael addition of cyanide anions to nitrosoalkenes. Ohno and colleagues [79] reported that reaction of NSA derived from oximes of cyclic chloroketones 1 with sodium cyanide led to the corresponding fused 5-aminoisoxazoles 86 rather than the expected α-cyanooximes 85 (Scheme 31). It is believed that the initially formed α-cyanooximes 85 undergo rapid cyclization to isoxazole derivatives 86. Interestingly, in the case of a more sterically hindered 3-chloronorcamphor oxime, the corresponding α-cyanooxime 85a was obtained, which did not undergo cyclization to isoxazole. Considering the fact that cyclic chloroketones are prepared by chloronitrosylation of cyclic olefins, the overall process is a highly straightforward strategy for the conversion of alkenes to bioactive 5-aminoisoxazoles [80].
Scheme 31: Addition of cyanide anion to nitrosoalkenes and subsequent cyclization to 5-aminoisoxazoles 86.
Scheme 31: Addition of cyanide anion to nitrosoalkenes and subsequent cyclization to 5-aminoisoxazoles 86.
Our group employed N,N-bis(silyloxy)enamines 3 as sources of nitrosoalkenes in reaction with trimethylsilylcyanide in the presence of triethylamine as catalyst (Scheme 32) [81]. Interestingly, the initial addition products are intercepted by TMSCN forming stable TMS ethers of α-cyanooximes 87, which can be isolated by vacuum distillation. Mild desilylation of 87 initiates the intramolecular cyclization to 5-aminoisoxazoles 88, which were obtained in moderate to good yields. Following this synthetic scheme, 5-aminoisoxazoles 88 can be readily accessed from available nitroalkanes in three steps.
Scheme 32: Et3N-catalysed addition of trimethylsilyl cyanide to N,N-bis(silyloxy)enamines 3 leading to 5-aminoisoxazoles 88.
Scheme 32: Et3N-catalysed addition of trimethylsilyl cyanide to N,N-bis(silyloxy)enamines 3 leading to 5-amino...
Recently, Tanimoto and co-workers [22] reported the addition of TMSCN to a nitrosoallene intermediate NSA16 generated from allenyl N-siloxysulfonamide 89 in the presence of TBAF and diisopropyl azodicarboxylate. Interestingly, the addition product 90 does not undergo cyclization (Scheme 33).
Scheme 33: Addition of TMSCN to allenyl N-siloxysulfonamide 89.
Scheme 33: Addition of TMSCN to allenyl N-siloxysulfonamide 89.
At the same time, reactions of nitrosoallenes of type NSA16 with malodinitrile and ethyl cyanoacetic ester afforded azafulvene derivatives 91 (Scheme 34) [82]. Remarkably, the formation of five-membered cyclic nitrone through N-attack of the oxime on the cyano group is more preferable over the six-membered N–O heterocycle.
Scheme 34: Reaction of nitrosoallenes NSA16 with malodinitrile and ethyl cyanoacetic ester.
Scheme 34: Reaction of nitrosoallenes NSA16 with malodinitrile and ethyl cyanoacetic ester.
C-Nucleophiles possessing leaving groups have been also used in the construction of N–O heterocycles using nitrosoalkenes as intermediates. An early example is the reaction of sulfonium ylides 92 with α-halo ketoximes 1 leading to isoxazolines 93 reported by Bravo [11] and Gilchrist [83] (Scheme 35). The process is believed to proceed via the generation of a nitrosoalkene intermediate (NSA) followed by a formal [4 + 1]-annulation reaction with ylide (tandem Michael addition/intramolecular nucleophilic substitution of dimethylsulfide by oximate anion in intermediate 94).
Scheme 35: [4 + 1]-Annulation of nitrosoalkenes NSA with sulfonium ylides 92.
Scheme 35: [4 + 1]-Annulation of nitrosoalkenes NSA with sulfonium ylides 92.
The addition of sulfonium ylides to nitrosoalkenes can end up not only with cyclic products, but also with α,β-unsaturated oximes 95, if the elimination of dimethyl sulfide from 94 proceeds faster than the cyclization. The cyclization/elimination selectivity was found be highly dependent on the nature of substituents in ylide 92. Only when R3 was an acyl or phenacyl group, exclusive formation of isoxazoline products 93 was observed.
Triphenylarsonium ylides were also studied in the reaction with nitrosoalkenes, yet lower yields of isoxazolines 93 were obtained as compared to sulfonium ylides [11].
Following the same reaction pattern, Cheng and co-workers [84] applied diazo compounds 96 instead of sulfonium ylides 92 in the reaction with α-bromo ketoximes 1 (Scheme 36). The reaction requires a copper catalyst, which transforms the diazo compound 96 into a metal carbene complex 97. The latter reacts with a nitrosoalkene intermediate NSA (formed from α-bromo ketoxime 1) producing isoxazoline 93 with recovery of the catalyst. Unfortunately, the reaction is complicated by a concurrent [3 + 2]-cycloaddition of diazo compounds 96 to nitrosoalkenes leading to N-nitrosopyrazoles 98 via intermediates 99.
Scheme 36: Reaction of diazo compounds 96 with nitrosoalkenes NSA.
Scheme 36: Reaction of diazo compounds 96 with nitrosoalkenes NSA.
A substantial improvement of this isoxazoline ring-forming strategy was recently introduced by Li et al. [85], who achieved the oxidative [4 + 1]-annulation of nitrosoalkenes with 1,3-dicarbonyl compounds (Scheme 37). Optimized reaction conditions require 2 equivalents of silver carbonate as oxidizer and K2CO3 as a base to generate nitrosoalkene from a halooxime precursor 1. The plausible mechanism involves the initial conjugate addition of dicarbonyl compounds to a nitrosoalkene followed by a silver-mediated radical cyclization of the resulting oximes 6 to final isoxazolines 100. Importantly, the reaction is well-tolerated by various substituents both in halooxime and in dicarbonyl compounds producing isoxazolines 100 in moderate to high yields.
Scheme 37: Tandem Michael addition/oxidative cyclization strategy to isoxazolines 100.
Scheme 37: Tandem Michael addition/oxidative cyclization strategy to isoxazolines 100.
Conclusion
In conclusion, the Michael addition of carbon-centered nucleophiles to conjugated nitrosoalkenes is a promising strategy for the construction of C–C bonds that has been already demonstrated by its use in numerous total syntheses of natural products and pharmaceutically relevant compounds. However, a number of challenges still remain to be addressed in this methodology, namely: (1) the extension of the scope of C-nucleophiles that can be involved in conjugate addition to nitrosoalkenes; (2) the development of general methods for coupling nitrosoalkenes with organometallic reagents; (3) the elaboration of tandem and domino-processes utilizing conjugate addition of C-nucleophiles to nitrosoalkenes and finally, (4) the design of catalytic enantioselective versions of the Michael addition to nitrosoalkenes (for advances in catalytic asymmetric cycloadditions of nitrosoalkenes see [86,87]). The use of highly selective precursors of nitrosoalkenes such as silyl ethers of halooximes, N-vinyl nitrosoacetals and N-vinyl N-siloxysulfonamides definitely holds the key to solving these problems.
References
-
Barrett, A. G. M.; Graboski, G. G. Chem. Rev. 1986, 86, 751. doi:10.1021/cr00075a002
Return to citation in text: [1] -
Denmark, S. E.; Thorarensen, A. Chem. Rev. 1996, 96, 137. doi:10.1021/cr940277f
Return to citation in text: [1] -
Perekalin, V. V.; Lipina, E. S.; Berestovitskaya, V. M.; Efremov, D. A. Nitroalkenes: Conjugated Nitro Compounds; Wiley: New York, NY, 1994.
Return to citation in text: [1] -
Sukhorukov, A. Yu.; Sukhanova, A. A.; Zlotin, S. G. Tetrahedron 2016, 72, 6191. doi:10.1016/j.tet.2016.07.067
Return to citation in text: [1] -
Halimehjani, A. Z.; Namboothiri, I. N. N.; Hooshmand, S. E. RSC Adv. 2014, 4, 48022. doi:10.1039/C4RA08828J
Return to citation in text: [1] -
Gilchrist, T. L. Chem. Soc. Rev. 1983, 12, 53. doi:10.1039/cs9831200053
Return to citation in text: [1] [2] [3] -
Lyapkalo, I. M.; Ioffe, S. L. Russ. Chem. Rev. 1998, 67, 467. doi:10.1070/RC1998v067n06ABEH000406
Return to citation in text: [1] [2] [3] -
Lemos, A. Molecules 2009, 14, 4098. doi:10.3390/molecules14104098
Return to citation in text: [1] [2] -
Reissig, H.-U.; Zimmer, R. 1-Nitrosoalkenes. Science of Synthesis; Georg Thieme Verlag: Stuttgart, 2006; Vol. 33, pp 371 ff.
Return to citation in text: [1] [2] -
Reissig, H.-U.; Zimmer, R. Nitrosoalkenes. Science of Synthesis (Knowledge Update); Georg Thieme Verlag: Stuttgart, 2015; Vol. 2, pp 315 ff.
Return to citation in text: [1] [2] -
Bravo, P.; Gaudiano, G.; Ponti, P. P.; Ticozzi, C. Tetrahedron 1972, 28, 3845. doi:10.1016/S0040-4020(01)93832-3
Return to citation in text: [1] [2] [3] -
Gaonkar, S. L.; Rai, K. M. L. J. Heterocycl. Chem. 2005, 42, 877. doi:10.1002/jhet.5570420520
Return to citation in text: [1] -
Ohno, M.; Naruse, N.; Torimitsu, S.; Okamoto, M. Bull. Chem. Soc. Jpn. 1966, 39, 1119. doi:10.1246/bcsj.39.1119
Return to citation in text: [1] [2] -
Denmark, S. E.; Dappen, M. S. J. Org. Chem. 1984, 49, 798. doi:10.1021/jo00179a011
Return to citation in text: [1] [2] -
Denmark, S. E.; Dappen, M. S.; Sternberg, J. A. J. Org. Chem. 1984, 49, 4741. doi:10.1021/jo00198a038
Return to citation in text: [1] -
Denmark, S. E.; Dappen, M. S.; Sear, N. L.; Jacobs, R. T. J. Am. Chem. Soc. 1990, 112, 3466. doi:10.1021/ja00165a034
Return to citation in text: [1] -
Lesiv, A. V.; Ioffe, S. L.; Strelenko, Y. A.; Tartakovsky, V. A. Helv. Chim. Acta 2002, 85, 3489. doi:10.1002/1522-2675(200210)85:10<3489::AID-HLCA3489>3.0.CO;2-6
Return to citation in text: [1] -
Zhmurov, P. A.; Khoroshutina, Y. A.; Novikov, R. A.; Golovanov, I. S.; Sukhorukov, A. Yu.; Ioffe, S. L. Chem. – Eur. J. 2017, 23, 4570. doi:10.1002/chem.201605390
Return to citation in text: [1] -
Feger, H.; Simchen, G. Liebigs Ann. Chem. 1986, 1456. doi:10.1002/jlac.198619860817
Return to citation in text: [1] -
Dilman, A. D.; Lyapkalo, I. M.; Ioffe, S. L.; Strelenko, Yu. A.; Tartakovsky, V. A. Synthesis 1999, 1767. doi:10.1055/s-1999-3594
Return to citation in text: [1] [2] -
Dilman, A. D.; Tishkov, A. A.; Lyapkalo, I. M.; Ioffe, S. L.; Strelenko, Yu. A.; Tartakovsky, V. A. Synthesis 1998, 181. doi:10.1055/s-1998-2019
Return to citation in text: [1] -
Tanimoto, H.; Yokoyama, K.; Mizutani, Y.; Shitaoka, T.; Morimoto, T.; Nishiyama, Y.; Kakiuchi, K. J. Org. Chem. 2016, 81, 559. doi:10.1021/acs.joc.5b02364
Return to citation in text: [1] [2] -
Ohno, M.; Torimitsu, S.; Naruse, N.; Okamoto, M.; Sakai, I. Bull. Chem. Soc. Jpn. 1966, 39, 1129. doi:10.1246/bcsj.39.1129
Return to citation in text: [1] [2] -
Ustinov, A. V.; Dilman, A. D.; Ioffe, S. L.; Belyakov, P. A.; Strelenko, Yu. A. Russ. Chem. Bull. 2002, 51, 1455. doi:10.1023/A:1020954721491
Return to citation in text: [1] -
Dilman, A. D.; Lyapkalo, I. M.; Ioffe, S. L.; Strelenko, Yu. A.; Tartakovsky, V. A. Russ. Chem. Bull. 2000, 49, 874. doi:10.1007/BF02494712
Return to citation in text: [1] -
Li, P.; Majireck, M. M.; Witek, J. A.; Weinreb, S. M. Tetrahedron Lett. 2010, 51, 2032. doi:10.1016/j.tetlet.2010.02.050
Return to citation in text: [1] -
Korboukh, I.; Kumar, P.; Weinreb, S. M. J. Am. Chem. Soc. 2007, 129, 10342. doi:10.1021/ja074108r
Return to citation in text: [1] -
Kumar, P.; Li, P.; Korboukh, I.; Wang, T. L.; Yennawar, H.; Weinreb, S. M. J. Org. Chem. 2011, 76, 2094. doi:10.1021/jo1024392
Return to citation in text: [1] -
Sengupta, R.; Witek, J. A.; Weinreb, S. M. Tetrahedron 2011, 67, 8229. doi:10.1016/j.tet.2011.08.054
Return to citation in text: [1] [2] -
Witek, J. A.; Weinreb, S. M. Org. Lett. 2011, 13, 1258. doi:10.1021/ol2000793
Return to citation in text: [1] [2] -
Oppolzer, W.; Petrzilka, M.; Bättig, K. Helv. Chim. Acta 1977, 60, 2964. doi:10.1002/hlca.19770600849
Return to citation in text: [1] [2] -
Oppolzer, W.; Bättig, K.; Hudlicky, T. Tetrahedron 1981, 37, 4359. doi:10.1016/0040-4020(81)80001-4
Return to citation in text: [1] [2] -
Feng, Y.; Majireck, M. M.; Weinreb, S. M. J. Org. Chem. 2014, 79, 7. doi:10.1021/jo402495q
Return to citation in text: [1] [2] -
Nocket, A. J.; Weinreb, S. M. Angew. Chem., Int. Ed. 2014, 53, 14162. doi:10.1002/anie.201407810
Return to citation in text: [1] [2] -
Nocket, A. J.; Feng, Y.; Weinreb, S. M. J. Org. Chem. 2015, 80, 1116. doi:10.1021/jo5026404
Return to citation in text: [1] [2] -
Chauhan, P. S.; Weinreb, S. M. J. Org. Chem. 2014, 79, 6389. doi:10.1021/jo501067u
Return to citation in text: [1] -
Corey, E. J.; Petrzilka, M.; Ueda, Y. Helv. Chim. Acta 1977, 60, 2294. doi:10.1002/hlca.19770600720
Return to citation in text: [1] -
Corey, E. J.; Petrzilka, M.; Ueda, Y. Tetrahedron Lett. 1975, 16, 4343. doi:10.1016/S0040-4039(00)91119-5
Return to citation in text: [1] -
Corey, E. J.; Arnett, J. F.; Widiger, G. N. J. Am. Chem. Soc. 1975, 97, 430. doi:10.1021/ja00835a039
Return to citation in text: [1] -
Corey, E. J.; Melvin, L. S., Jr.; Haslanger, M. F. Tetrahedron Lett. 1975, 16, 3117. doi:10.1016/S0040-4039(00)91472-2
Return to citation in text: [1] -
Sengupta, R.; Weinreb, S. M. Synthesis 2012, 44, 2933. doi:10.1055/s-0032-1316984
Return to citation in text: [1] -
Colvin, E. W.; Robertson, A. D.; Seebach, D.; Beck, A. K. J. Chem. Soc., Chem. Commun. 1981, 952. doi:10.1039/C39810000952
Return to citation in text: [1] -
Trost, B. M.; Barrett, D. Tetrahedron 1996, 52, 6903. doi:10.1016/0040-4020(96)00325-0
Return to citation in text: [1] -
Gilchrist, T. L.; Roberts, T. G. J. Chem. Soc., Perkin Trans. 1 1983, 1283. doi:10.1039/p19830001283
Return to citation in text: [1] [2] [3] -
Gilchrist, T. L.; Lingham, D. A.; Roberts, T. G. J. Chem. Soc., Chem. Commun. 1979, 1089. doi:10.1039/C39790001089
Return to citation in text: [1] [2] -
Dauzonne, D.; O'Neil, I. A.; Renaud, A. J. Org. Chem. 1984, 49, 4409. doi:10.1021/jo00197a016
Return to citation in text: [1] -
Plate, R.; Ottenheijm, H. C. J.; Nivard, R. J. F. J. Org. Chem. 1984, 49, 540. doi:10.1021/jo00177a030
Return to citation in text: [1] [2] -
Plate, R.; Akkerman, M. A. J.; Ottenheijm, H. C. J.; Smits, J. M. M. J. Chem. Soc., Perkin Trans. 1 1987, 2481. doi:10.1039/P19870002481
Return to citation in text: [1] -
Hermkens, P. H. H.; van Maarseveen, J. H.; Kruse, C. G.; Scheeren, H. W. Tetrahedron 1988, 44, 6491. doi:10.1016/S0040-4020(01)89838-0
Return to citation in text: [1] [2] -
Zimmer, R.; Reissig, H.-U. J. Org. Chem. 1992, 57, 339. doi:10.1021/jo00027a058
Return to citation in text: [1] -
Guimarães, E.; Lemos, A.; Lopes, M. Phosphorus, Sulfur Silicon Relat. Elem. 2007, 182, 2149. doi:10.1080/10426500701407425
Return to citation in text: [1] -
Lopes, S. M. M.; Lemos, A.; Pinho e Melo, T. M. V. D. Tetrahedron Lett. 2010, 51, 6756. doi:10.1016/j.tetlet.2010.10.095
Return to citation in text: [1] -
Lopes, S. M. M.; Palacios, F.; Lemos, A.; Pinho e Melo, T. M. V. D. Tetrahedron 2011, 67, 8902. doi:10.1016/j.tet.2011.09.051
Return to citation in text: [1] -
Pereira, N. A. M.; Lemos, A.; Serra, A. C.; Pinho e Melo, T. M. V. D. Tetrahedron Lett. 2013, 54, 1553. doi:10.1016/j.tetlet.2013.01.032
Return to citation in text: [1] -
Lopes, S. M. M.; Nunes, S. C. C.; Caratão, C. C.; Pais, A. A. C. C.; Pinho e Melo, T. M. V. D. Monatsh. Chem. 2016, 147, 1565. doi:10.1007/s00706-016-1763-1
Return to citation in text: [1] -
Davies, D. E.; Gilchrist, T. L. J. Chem. Soc., Perkin Trans. 1 1983, 1479. doi:10.1039/P19830001479
Return to citation in text: [1] -
Sukhorukov, A. Yu.; Ioffe, S. L. Chem. Rev. 2011, 111, 5004. doi:10.1021/cr100292w
Return to citation in text: [1] -
Domingo, L. R.; Picher, M. T.; Arroyo, P. Eur. J. Org. Chem. 2006, 2570. doi:10.1002/ejoc.200500978
Return to citation in text: [1] -
de los Santos, J. M.; Rubiales, G.; Sbai, Z. E.; Ochoa de Retana, A. M.; Palacios, F. Org. Biomol. Chem. 2017, 15, 662. doi:10.1039/C6OB02486F
Return to citation in text: [1] -
Grosso, C.; Cardoso, A. L.; Lemos, A.; Varela, J.; Rodrigues, M. J.; Custódio, L.; Barreira, L.; Pinho e Melo, T. M. V. D. Eur. J. Med. Chem. 2015, 93, 9. doi:10.1016/j.ejmech.2015.01.050
Return to citation in text: [1] -
Pereira, N. A. M.; Lopes, S. M. M.; Lemos, A.; Pinho e Melo, T. M. V. D. Synlett 2014, 25, 423. doi:10.1055/s-0033-1340300
Return to citation in text: [1] -
Grosso, C.; Cardoso, A. L.; Rodrigues, M. J.; Marques, C.; Barreira, L.; Lemos, A.; Pinho e Melo, T. M. D. V. Bioorg. Med. Chem. 2017, 25, 1122. doi:10.1016/j.bmc.2016.12.028
Return to citation in text: [1] -
Pereira, R.; Benedetti, R.; Pérez-Rodríguez, S.; Nebbioso, A.; García-Rodríguez, J.; Carafa, V.; Stuhldreier, M.; Conte, M.; Rodríguez-Barrios, F.; Stunnenberg, H. G.; Gronemeyer, H.; Altucci, L.; de Lera, Á. R. J. Med. Chem. 2012, 55, 9467. doi:10.1021/jm300618u
Return to citation in text: [1] -
Li, J. P.; Newlander, K. A.; Yellin, T. O. Synthesis 1988, 73. doi:10.1055/s-1988-27471
Return to citation in text: [1] -
Park, K.; Gopalsamy, A.; Aplasca, A.; Ellingboe, J. W.; Xu, W.; Zhang, Y.; Levin, J. I. Bioorg. Med. Chem. 2009, 17, 3857. doi:10.1016/j.bmc.2009.04.033
Return to citation in text: [1] -
Duffy, N. H.; Lester, H. A.; Dougherty, D. A. ACS Chem. Biol. 2012, 7, 1738. doi:10.1021/cb300246j
Return to citation in text: [1] -
Ottenheijm, H. C. J.; Plate, R.; Noordik, J. H.; Herscheid, J. D. M. J. Org. Chem. 1982, 47, 2147. doi:10.1021/jo00132a032
Return to citation in text: [1] -
Plate, R.; Hermkens, P. H. H.; Smits, J. M. M.; Nivard, R. J. F.; Ottenheijm, H. C. J. J. Org. Chem. 1987, 52, 1047. doi:10.1021/jo00382a014
Return to citation in text: [1] -
Plate, R.; Ottenheijm, H. C. J. Tetrahedron Lett. 1986, 27, 3755. doi:10.1016/S0040-4039(00)83872-1
Return to citation in text: [1] -
Gilchrist, T. L.; Lemos, A. Tetrahedron 1992, 48, 7655. doi:10.1016/S0040-4020(01)90377-1
Return to citation in text: [1] -
Baldwin, J. E.; Lynch, G. P.; Pitlik, J. J. Antibiot. 1991, 44, 1. doi:10.7164/antibiotics.44.1
Return to citation in text: [1] -
Ternansky, R. J.; Draheim, S. E. Tetrahedron Lett. 1990, 31, 2805. doi:10.1016/0040-4039(90)80153-D
Return to citation in text: [1] -
Holmes, R. E.; Neel, D. A. Tetrahedron Lett. 1990, 31, 5567. doi:10.1016/S0040-4039(00)97898-5
Return to citation in text: [1] -
Kozikowski, A. P.; Sato, K.; Basu, A.; Lazo, J. S. J. Am. Chem. Soc. 1989, 111, 6228. doi:10.1021/ja00198a038
Return to citation in text: [1] -
Kozikowski, A. P.; Shum, P. W.; Basu, A.; Lazo, J. S. J. Med. Chem. 1991, 34, 2420. doi:10.1021/jm00112a017
Return to citation in text: [1] -
Webb, R. R., II; Venuti, M. C.; Eigenbrot, C. J. Org. Chem. 1991, 56, 4706. doi:10.1021/jo00015a026
Return to citation in text: [1] -
Pu, J.; Deng, K.; Butera, J.; Chlenov, M.; Gilbert, A.; Kagan, M.; Mattes, J.; Resnick, L. Tetrahedron 2010, 66, 1963. doi:10.1016/j.tet.2010.01.048
Return to citation in text: [1] -
Quick, J.; Saha, B.; Driedger, P. E. Tetrahedron Lett. 1994, 35, 8549. doi:10.1016/S0040-4039(00)78433-4
Return to citation in text: [1] -
Ohno, M.; Naruse, N. Bull. Chem. Soc. Jpn. 1966, 39, 1125. doi:10.1246/bcsj.39.1125
Return to citation in text: [1] -
Stein, P. D.; Floyd, D. M.; Bisaha, S.; Dickey, J.; Girotra, R. N.; Gougoutas, J. Z.; Kozlowski, M.; Lee, V. G.; Liu, E. C.-K. J. Med. Chem. 1995, 38, 1344. doi:10.1021/jm00008a013
Return to citation in text: [1] -
Lesiv, A. V.; Ioffe, S. L.; Strelenko, Yu. A.; Bliznets, I. V.; Tartakovsky, V. A. Mendeleev Commun. 2002, 12, 99. doi:10.1070/MC2002v012n03ABEH001585
Return to citation in text: [1] -
Tanimoto, H.; Shitaoka, T.; Yokoyama, K.; Morimoto, T.; Nishiyama, Y.; Kakiuchi, K. J. Org. Chem. 2016, 81, 8722. doi:10.1021/acs.joc.6b00758
Return to citation in text: [1] -
Faragher, R.; Gilchrist, T. L. J. Chem. Soc., Perkin Trans. 1 1977, 1196. doi:10.1039/p19770001196
Return to citation in text: [1] -
Guo, J.; Gaudette, J.; Cheng, J.-F. Tetrahedron Lett. 2009, 50, 933. doi:10.1016/j.tetlet.2008.12.032
Return to citation in text: [1] -
Liu, Y.-Y.; Yang, X.-H.; Yang, J.; Song, R.-J.; Li, J.-H. Chem. Commun. 2014, 50, 6906. doi:10.1039/c4cc02084g
Return to citation in text: [1] -
Wabnitz, T. C.; Saaby, S.; Jørgensen, K. A. Org. Biomol. Chem. 2004, 2, 828. doi:10.1039/B316518C
Return to citation in text: [1] -
Zhang, Y.; Stephens, D.; Hernandez, G.; Mendoza, R.; Larionov, O. V. Chem. – Eur. J. 2012, 18, 16612. doi:10.1002/chem.201203435
Return to citation in text: [1]
| 36. | Chauhan, P. S.; Weinreb, S. M. J. Org. Chem. 2014, 79, 6389. doi:10.1021/jo501067u |
| 34. | Nocket, A. J.; Weinreb, S. M. Angew. Chem., Int. Ed. 2014, 53, 14162. doi:10.1002/anie.201407810 |
| 35. | Nocket, A. J.; Feng, Y.; Weinreb, S. M. J. Org. Chem. 2015, 80, 1116. doi:10.1021/jo5026404 |
| 23. | Ohno, M.; Torimitsu, S.; Naruse, N.; Okamoto, M.; Sakai, I. Bull. Chem. Soc. Jpn. 1966, 39, 1129. doi:10.1246/bcsj.39.1129 |
| 44. | Gilchrist, T. L.; Roberts, T. G. J. Chem. Soc., Perkin Trans. 1 1983, 1283. doi:10.1039/p19830001283 |
| 45. | Gilchrist, T. L.; Lingham, D. A.; Roberts, T. G. J. Chem. Soc., Chem. Commun. 1979, 1089. doi:10.1039/C39790001089 |
| 46. | Dauzonne, D.; O'Neil, I. A.; Renaud, A. J. Org. Chem. 1984, 49, 4409. doi:10.1021/jo00197a016 |
| 47. | Plate, R.; Ottenheijm, H. C. J.; Nivard, R. J. F. J. Org. Chem. 1984, 49, 540. doi:10.1021/jo00177a030 |
| 48. | Plate, R.; Akkerman, M. A. J.; Ottenheijm, H. C. J.; Smits, J. M. M. J. Chem. Soc., Perkin Trans. 1 1987, 2481. doi:10.1039/P19870002481 |
| 49. | Hermkens, P. H. H.; van Maarseveen, J. H.; Kruse, C. G.; Scheeren, H. W. Tetrahedron 1988, 44, 6491. doi:10.1016/S0040-4020(01)89838-0 |
| 50. | Zimmer, R.; Reissig, H.-U. J. Org. Chem. 1992, 57, 339. doi:10.1021/jo00027a058 |
| 51. | Guimarães, E.; Lemos, A.; Lopes, M. Phosphorus, Sulfur Silicon Relat. Elem. 2007, 182, 2149. doi:10.1080/10426500701407425 |
| 52. | Lopes, S. M. M.; Lemos, A.; Pinho e Melo, T. M. V. D. Tetrahedron Lett. 2010, 51, 6756. doi:10.1016/j.tetlet.2010.10.095 |
| 53. | Lopes, S. M. M.; Palacios, F.; Lemos, A.; Pinho e Melo, T. M. V. D. Tetrahedron 2011, 67, 8902. doi:10.1016/j.tet.2011.09.051 |
| 54. | Pereira, N. A. M.; Lemos, A.; Serra, A. C.; Pinho e Melo, T. M. V. D. Tetrahedron Lett. 2013, 54, 1553. doi:10.1016/j.tetlet.2013.01.032 |
| 55. | Lopes, S. M. M.; Nunes, S. C. C.; Caratão, C. C.; Pais, A. A. C. C.; Pinho e Melo, T. M. V. D. Monatsh. Chem. 2016, 147, 1565. doi:10.1007/s00706-016-1763-1 |
| 44. | Gilchrist, T. L.; Roberts, T. G. J. Chem. Soc., Perkin Trans. 1 1983, 1283. doi:10.1039/p19830001283 |
| 45. | Gilchrist, T. L.; Lingham, D. A.; Roberts, T. G. J. Chem. Soc., Chem. Commun. 1979, 1089. doi:10.1039/C39790001089 |
| 56. | Davies, D. E.; Gilchrist, T. L. J. Chem. Soc., Perkin Trans. 1 1983, 1479. doi:10.1039/P19830001479 |
| 43. | Trost, B. M.; Barrett, D. Tetrahedron 1996, 52, 6903. doi:10.1016/0040-4020(96)00325-0 |
| 44. | Gilchrist, T. L.; Roberts, T. G. J. Chem. Soc., Perkin Trans. 1 1983, 1283. doi:10.1039/p19830001283 |
| 41. | Sengupta, R.; Weinreb, S. M. Synthesis 2012, 44, 2933. doi:10.1055/s-0032-1316984 |
| 42. | Colvin, E. W.; Robertson, A. D.; Seebach, D.; Beck, A. K. J. Chem. Soc., Chem. Commun. 1981, 952. doi:10.1039/C39810000952 |
| 37. | Corey, E. J.; Petrzilka, M.; Ueda, Y. Helv. Chim. Acta 1977, 60, 2294. doi:10.1002/hlca.19770600720 |
| 38. | Corey, E. J.; Petrzilka, M.; Ueda, Y. Tetrahedron Lett. 1975, 16, 4343. doi:10.1016/S0040-4039(00)91119-5 |
| 39. | Corey, E. J.; Arnett, J. F.; Widiger, G. N. J. Am. Chem. Soc. 1975, 97, 430. doi:10.1021/ja00835a039 |
| 40. | Corey, E. J.; Melvin, L. S., Jr.; Haslanger, M. F. Tetrahedron Lett. 1975, 16, 3117. doi:10.1016/S0040-4039(00)91472-2 |
| 6. | Gilchrist, T. L. Chem. Soc. Rev. 1983, 12, 53. doi:10.1039/cs9831200053 |
| 7. | Lyapkalo, I. M.; Ioffe, S. L. Russ. Chem. Rev. 1998, 67, 467. doi:10.1070/RC1998v067n06ABEH000406 |
| 8. | Lemos, A. Molecules 2009, 14, 4098. doi:10.3390/molecules14104098 |
| 9. | Reissig, H.-U.; Zimmer, R. 1-Nitrosoalkenes. Science of Synthesis; Georg Thieme Verlag: Stuttgart, 2006; Vol. 33, pp 371 ff. |
| 10. | Reissig, H.-U.; Zimmer, R. Nitrosoalkenes. Science of Synthesis (Knowledge Update); Georg Thieme Verlag: Stuttgart, 2015; Vol. 2, pp 315 ff. |
| 57. | Sukhorukov, A. Yu.; Ioffe, S. L. Chem. Rev. 2011, 111, 5004. doi:10.1021/cr100292w |
| 58. | Domingo, L. R.; Picher, M. T.; Arroyo, P. Eur. J. Org. Chem. 2006, 2570. doi:10.1002/ejoc.200500978 |
| 59. | de los Santos, J. M.; Rubiales, G.; Sbai, Z. E.; Ochoa de Retana, A. M.; Palacios, F. Org. Biomol. Chem. 2017, 15, 662. doi:10.1039/C6OB02486F |
| 60. | Grosso, C.; Cardoso, A. L.; Lemos, A.; Varela, J.; Rodrigues, M. J.; Custódio, L.; Barreira, L.; Pinho e Melo, T. M. V. D. Eur. J. Med. Chem. 2015, 93, 9. doi:10.1016/j.ejmech.2015.01.050 |
| 61. | Pereira, N. A. M.; Lopes, S. M. M.; Lemos, A.; Pinho e Melo, T. M. V. D. Synlett 2014, 25, 423. doi:10.1055/s-0033-1340300 |
| 62. | Grosso, C.; Cardoso, A. L.; Rodrigues, M. J.; Marques, C.; Barreira, L.; Lemos, A.; Pinho e Melo, T. M. D. V. Bioorg. Med. Chem. 2017, 25, 1122. doi:10.1016/j.bmc.2016.12.028 |
| 70. | Gilchrist, T. L.; Lemos, A. Tetrahedron 1992, 48, 7655. doi:10.1016/S0040-4020(01)90377-1 |
| 71. | Baldwin, J. E.; Lynch, G. P.; Pitlik, J. J. Antibiot. 1991, 44, 1. doi:10.7164/antibiotics.44.1 |
| 72. | Ternansky, R. J.; Draheim, S. E. Tetrahedron Lett. 1990, 31, 2805. doi:10.1016/0040-4039(90)80153-D |
| 73. | Holmes, R. E.; Neel, D. A. Tetrahedron Lett. 1990, 31, 5567. doi:10.1016/S0040-4039(00)97898-5 |
| 67. | Ottenheijm, H. C. J.; Plate, R.; Noordik, J. H.; Herscheid, J. D. M. J. Org. Chem. 1982, 47, 2147. doi:10.1021/jo00132a032 |
| 68. | Plate, R.; Hermkens, P. H. H.; Smits, J. M. M.; Nivard, R. J. F.; Ottenheijm, H. C. J. J. Org. Chem. 1987, 52, 1047. doi:10.1021/jo00382a014 |
| 47. | Plate, R.; Ottenheijm, H. C. J.; Nivard, R. J. F. J. Org. Chem. 1984, 49, 540. doi:10.1021/jo00177a030 |
| 49. | Hermkens, P. H. H.; van Maarseveen, J. H.; Kruse, C. G.; Scheeren, H. W. Tetrahedron 1988, 44, 6491. doi:10.1016/S0040-4020(01)89838-0 |
| 69. | Plate, R.; Ottenheijm, H. C. J. Tetrahedron Lett. 1986, 27, 3755. doi:10.1016/S0040-4039(00)83872-1 |
| 65. | Park, K.; Gopalsamy, A.; Aplasca, A.; Ellingboe, J. W.; Xu, W.; Zhang, Y.; Levin, J. I. Bioorg. Med. Chem. 2009, 17, 3857. doi:10.1016/j.bmc.2009.04.033 |
| 66. | Duffy, N. H.; Lester, H. A.; Dougherty, D. A. ACS Chem. Biol. 2012, 7, 1738. doi:10.1021/cb300246j |
| 63. | Pereira, R.; Benedetti, R.; Pérez-Rodríguez, S.; Nebbioso, A.; García-Rodríguez, J.; Carafa, V.; Stuhldreier, M.; Conte, M.; Rodríguez-Barrios, F.; Stunnenberg, H. G.; Gronemeyer, H.; Altucci, L.; de Lera, Á. R. J. Med. Chem. 2012, 55, 9467. doi:10.1021/jm300618u |
| 64. | Li, J. P.; Newlander, K. A.; Yellin, T. O. Synthesis 1988, 73. doi:10.1055/s-1988-27471 |
| 76. | Webb, R. R., II; Venuti, M. C.; Eigenbrot, C. J. Org. Chem. 1991, 56, 4706. doi:10.1021/jo00015a026 |
| 77. | Pu, J.; Deng, K.; Butera, J.; Chlenov, M.; Gilbert, A.; Kagan, M.; Mattes, J.; Resnick, L. Tetrahedron 2010, 66, 1963. doi:10.1016/j.tet.2010.01.048 |
| 74. | Kozikowski, A. P.; Sato, K.; Basu, A.; Lazo, J. S. J. Am. Chem. Soc. 1989, 111, 6228. doi:10.1021/ja00198a038 |
| 75. | Kozikowski, A. P.; Shum, P. W.; Basu, A.; Lazo, J. S. J. Med. Chem. 1991, 34, 2420. doi:10.1021/jm00112a017 |
| 1. | Barrett, A. G. M.; Graboski, G. G. Chem. Rev. 1986, 86, 751. doi:10.1021/cr00075a002 |
| 2. | Denmark, S. E.; Thorarensen, A. Chem. Rev. 1996, 96, 137. doi:10.1021/cr940277f |
| 3. | Perekalin, V. V.; Lipina, E. S.; Berestovitskaya, V. M.; Efremov, D. A. Nitroalkenes: Conjugated Nitro Compounds; Wiley: New York, NY, 1994. |
| 4. | Sukhorukov, A. Yu.; Sukhanova, A. A.; Zlotin, S. G. Tetrahedron 2016, 72, 6191. doi:10.1016/j.tet.2016.07.067 |
| 5. | Halimehjani, A. Z.; Namboothiri, I. N. N.; Hooshmand, S. E. RSC Adv. 2014, 4, 48022. doi:10.1039/C4RA08828J |
| 13. | Ohno, M.; Naruse, N.; Torimitsu, S.; Okamoto, M. Bull. Chem. Soc. Jpn. 1966, 39, 1119. doi:10.1246/bcsj.39.1119 |
| 27. | Korboukh, I.; Kumar, P.; Weinreb, S. M. J. Am. Chem. Soc. 2007, 129, 10342. doi:10.1021/ja074108r |
| 11. | Bravo, P.; Gaudiano, G.; Ponti, P. P.; Ticozzi, C. Tetrahedron 1972, 28, 3845. doi:10.1016/S0040-4020(01)93832-3 |
| 11. | Bravo, P.; Gaudiano, G.; Ponti, P. P.; Ticozzi, C. Tetrahedron 1972, 28, 3845. doi:10.1016/S0040-4020(01)93832-3 |
| 12. | Gaonkar, S. L.; Rai, K. M. L. J. Heterocycl. Chem. 2005, 42, 877. doi:10.1002/jhet.5570420520 |
| 28. | Kumar, P.; Li, P.; Korboukh, I.; Wang, T. L.; Yennawar, H.; Weinreb, S. M. J. Org. Chem. 2011, 76, 2094. doi:10.1021/jo1024392 |
| 6. | Gilchrist, T. L. Chem. Soc. Rev. 1983, 12, 53. doi:10.1039/cs9831200053 |
| 7. | Lyapkalo, I. M.; Ioffe, S. L. Russ. Chem. Rev. 1998, 67, 467. doi:10.1070/RC1998v067n06ABEH000406 |
| 8. | Lemos, A. Molecules 2009, 14, 4098. doi:10.3390/molecules14104098 |
| 9. | Reissig, H.-U.; Zimmer, R. 1-Nitrosoalkenes. Science of Synthesis; Georg Thieme Verlag: Stuttgart, 2006; Vol. 33, pp 371 ff. |
| 10. | Reissig, H.-U.; Zimmer, R. Nitrosoalkenes. Science of Synthesis (Knowledge Update); Georg Thieme Verlag: Stuttgart, 2015; Vol. 2, pp 315 ff. |
| 26. | Li, P.; Majireck, M. M.; Witek, J. A.; Weinreb, S. M. Tetrahedron Lett. 2010, 51, 2032. doi:10.1016/j.tetlet.2010.02.050 |
| 22. | Tanimoto, H.; Yokoyama, K.; Mizutani, Y.; Shitaoka, T.; Morimoto, T.; Nishiyama, Y.; Kakiuchi, K. J. Org. Chem. 2016, 81, 559. doi:10.1021/acs.joc.5b02364 |
| 6. | Gilchrist, T. L. Chem. Soc. Rev. 1983, 12, 53. doi:10.1039/cs9831200053 |
| 7. | Lyapkalo, I. M.; Ioffe, S. L. Russ. Chem. Rev. 1998, 67, 467. doi:10.1070/RC1998v067n06ABEH000406 |
| 14. | Denmark, S. E.; Dappen, M. S. J. Org. Chem. 1984, 49, 798. doi:10.1021/jo00179a011 |
| 82. | Tanimoto, H.; Shitaoka, T.; Yokoyama, K.; Morimoto, T.; Nishiyama, Y.; Kakiuchi, K. J. Org. Chem. 2016, 81, 8722. doi:10.1021/acs.joc.6b00758 |
| 22. | Tanimoto, H.; Yokoyama, K.; Mizutani, Y.; Shitaoka, T.; Morimoto, T.; Nishiyama, Y.; Kakiuchi, K. J. Org. Chem. 2016, 81, 559. doi:10.1021/acs.joc.5b02364 |
| 24. | Ustinov, A. V.; Dilman, A. D.; Ioffe, S. L.; Belyakov, P. A.; Strelenko, Yu. A. Russ. Chem. Bull. 2002, 51, 1455. doi:10.1023/A:1020954721491 |
| 80. | Stein, P. D.; Floyd, D. M.; Bisaha, S.; Dickey, J.; Girotra, R. N.; Gougoutas, J. Z.; Kozlowski, M.; Lee, V. G.; Liu, E. C.-K. J. Med. Chem. 1995, 38, 1344. doi:10.1021/jm00008a013 |
| 19. | Feger, H.; Simchen, G. Liebigs Ann. Chem. 1986, 1456. doi:10.1002/jlac.198619860817 |
| 20. | Dilman, A. D.; Lyapkalo, I. M.; Ioffe, S. L.; Strelenko, Yu. A.; Tartakovsky, V. A. Synthesis 1999, 1767. doi:10.1055/s-1999-3594 |
| 21. | Dilman, A. D.; Tishkov, A. A.; Lyapkalo, I. M.; Ioffe, S. L.; Strelenko, Yu. A.; Tartakovsky, V. A. Synthesis 1998, 181. doi:10.1055/s-1998-2019 |
| 20. | Dilman, A. D.; Lyapkalo, I. M.; Ioffe, S. L.; Strelenko, Yu. A.; Tartakovsky, V. A. Synthesis 1999, 1767. doi:10.1055/s-1999-3594 |
| 25. | Dilman, A. D.; Lyapkalo, I. M.; Ioffe, S. L.; Strelenko, Yu. A.; Tartakovsky, V. A. Russ. Chem. Bull. 2000, 49, 874. doi:10.1007/BF02494712 |
| 81. | Lesiv, A. V.; Ioffe, S. L.; Strelenko, Yu. A.; Bliznets, I. V.; Tartakovsky, V. A. Mendeleev Commun. 2002, 12, 99. doi:10.1070/MC2002v012n03ABEH001585 |
| 17. | Lesiv, A. V.; Ioffe, S. L.; Strelenko, Y. A.; Tartakovsky, V. A. Helv. Chim. Acta 2002, 85, 3489. doi:10.1002/1522-2675(200210)85:10<3489::AID-HLCA3489>3.0.CO;2-6 |
| 18. | Zhmurov, P. A.; Khoroshutina, Y. A.; Novikov, R. A.; Golovanov, I. S.; Sukhorukov, A. Yu.; Ioffe, S. L. Chem. – Eur. J. 2017, 23, 4570. doi:10.1002/chem.201605390 |
| 78. | Quick, J.; Saha, B.; Driedger, P. E. Tetrahedron Lett. 1994, 35, 8549. doi:10.1016/S0040-4039(00)78433-4 |
| 14. | Denmark, S. E.; Dappen, M. S. J. Org. Chem. 1984, 49, 798. doi:10.1021/jo00179a011 |
| 15. | Denmark, S. E.; Dappen, M. S.; Sternberg, J. A. J. Org. Chem. 1984, 49, 4741. doi:10.1021/jo00198a038 |
| 16. | Denmark, S. E.; Dappen, M. S.; Sear, N. L.; Jacobs, R. T. J. Am. Chem. Soc. 1990, 112, 3466. doi:10.1021/ja00165a034 |
| 13. | Ohno, M.; Naruse, N.; Torimitsu, S.; Okamoto, M. Bull. Chem. Soc. Jpn. 1966, 39, 1119. doi:10.1246/bcsj.39.1119 |
| 23. | Ohno, M.; Torimitsu, S.; Naruse, N.; Okamoto, M.; Sakai, I. Bull. Chem. Soc. Jpn. 1966, 39, 1129. doi:10.1246/bcsj.39.1129 |
| 79. | Ohno, M.; Naruse, N. Bull. Chem. Soc. Jpn. 1966, 39, 1125. doi:10.1246/bcsj.39.1125 |
| 29. | Sengupta, R.; Witek, J. A.; Weinreb, S. M. Tetrahedron 2011, 67, 8229. doi:10.1016/j.tet.2011.08.054 |
| 29. | Sengupta, R.; Witek, J. A.; Weinreb, S. M. Tetrahedron 2011, 67, 8229. doi:10.1016/j.tet.2011.08.054 |
| 30. | Witek, J. A.; Weinreb, S. M. Org. Lett. 2011, 13, 1258. doi:10.1021/ol2000793 |
| 84. | Guo, J.; Gaudette, J.; Cheng, J.-F. Tetrahedron Lett. 2009, 50, 933. doi:10.1016/j.tetlet.2008.12.032 |
| 85. | Liu, Y.-Y.; Yang, X.-H.; Yang, J.; Song, R.-J.; Li, J.-H. Chem. Commun. 2014, 50, 6906. doi:10.1039/c4cc02084g |
| 83. | Faragher, R.; Gilchrist, T. L. J. Chem. Soc., Perkin Trans. 1 1977, 1196. doi:10.1039/p19770001196 |
| 11. | Bravo, P.; Gaudiano, G.; Ponti, P. P.; Ticozzi, C. Tetrahedron 1972, 28, 3845. doi:10.1016/S0040-4020(01)93832-3 |
| 34. | Nocket, A. J.; Weinreb, S. M. Angew. Chem., Int. Ed. 2014, 53, 14162. doi:10.1002/anie.201407810 |
| 35. | Nocket, A. J.; Feng, Y.; Weinreb, S. M. J. Org. Chem. 2015, 80, 1116. doi:10.1021/jo5026404 |
| 33. | Feng, Y.; Majireck, M. M.; Weinreb, S. M. J. Org. Chem. 2014, 79, 7. doi:10.1021/jo402495q |
| 32. | Oppolzer, W.; Bättig, K.; Hudlicky, T. Tetrahedron 1981, 37, 4359. doi:10.1016/0040-4020(81)80001-4 |
| 33. | Feng, Y.; Majireck, M. M.; Weinreb, S. M. J. Org. Chem. 2014, 79, 7. doi:10.1021/jo402495q |
| 32. | Oppolzer, W.; Bättig, K.; Hudlicky, T. Tetrahedron 1981, 37, 4359. doi:10.1016/0040-4020(81)80001-4 |
| 31. | Oppolzer, W.; Petrzilka, M.; Bättig, K. Helv. Chim. Acta 1977, 60, 2964. doi:10.1002/hlca.19770600849 |
| 30. | Witek, J. A.; Weinreb, S. M. Org. Lett. 2011, 13, 1258. doi:10.1021/ol2000793 |
| 86. | Wabnitz, T. C.; Saaby, S.; Jørgensen, K. A. Org. Biomol. Chem. 2004, 2, 828. doi:10.1039/B316518C |
| 87. | Zhang, Y.; Stephens, D.; Hernandez, G.; Mendoza, R.; Larionov, O. V. Chem. – Eur. J. 2012, 18, 16612. doi:10.1002/chem.201203435 |
| 31. | Oppolzer, W.; Petrzilka, M.; Bättig, K. Helv. Chim. Acta 1977, 60, 2964. doi:10.1002/hlca.19770600849 |
© 2017 Boyko et al.; licensee Beilstein-Institut.
This is an Open Access article under the terms of the Creative Commons Attribution License (http://creativecommons.org/licenses/by/4.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
The license is subject to the Beilstein Journal of Organic Chemistry terms and conditions: (http://www.beilstein-journals.org/bjoc)