Abstract
Recently, it has been shown that the thermochemical cis→trans isomerization of azobenzenes is accelerated by a factor of more than 1000 by electronic coupling to a gold surface via a conjugated system with 11 bonds and a distance of 14 Å. The corresponding molecular architecture consists of a platform (triazatriangulenium (TATA)) which adsorbs on the gold surface, with an acetylene spacer standing upright, like a post in the middle of the platform and the azobenzene unit mounted on top. The rate acceleration is due to a very peculiar thermal singlet–triplet–singlet mechanism mediated by bulk gold. To investigate this mechanism further and to examine scope and limitation of the “spin-switch catalysis” we now prepared analogous diazocine systems. Diazocines, in contrast to azobenzenes, are stable in the cis-configuration. Upon irradiation with light of 405 nm the cis-configuration isomerizes to the trans-form, which slowly returns back to the stable cis-isomer. To investigate the thermal trans→cis isomerization as a function of the conjugation to the metal surface, we connected the acetylene spacer in meta (weak conjugation) and in para (strong conjugation) position. Both isomers form ordered monolayers on Au(111) surfaces.

Graphical Abstract
Introduction
Catalysts increase chemical reaction rates by lowering the activation energies and thus create more favorable reaction pathways [1-4]. However, there are very few reactions which do not follow the classical Eyring theory [5,6]. The rate of these reactions is not dependent on an activation barrier but controlled by quantum mechanical transition probabilities between two quantum states [7-10]. The majority of these quantum chemically forbidden reactions are photochemical processes or transition metal reactions including transitions between spin states or electronic states. We recently discovered a purely organic system in the ground state, whose reaction rate is accelerated from days to seconds by electronic coupling to a bulk gold surface via a conjugated linker over 11 bonds and 14 Å [11]. Thermal cis→trans isomerizations of azobenzenes are usually slow with half-lives of the trans-isomer within the range of hours to days at room temperature (parent azobenzene: 4–5 d at 25 °C) [12]. Rotation around the N=N bond is a symmetry-forbidden process and the slow isomerization proceeds via inversion at the N atoms [13]. The rate of isomerization is temperature dependent and follows a classical Arrhenius type behavior [12]. However, the rate and the mechanism change dramatically if the azobenzenes are electronically coupled to bulk gold [14-17]. To investigate the cis→trans isomerizations of azobenzenes as a function of electronic coupling systematically, we used the so-called platform approach [18]. The azobenzenes are not directly adsorbed on the surface, but covalently mounted on “TATA” (triazatriangulenium) platforms which adsorb on Au(111) surfaces. A spacer, such as an ethynyl group is connected to the central carbon atom like a post and the azobenzene is mounted on top of the spacer. After preparation of an ordered self-assembled monolayer on gold, the azobenzene units are freestanding upright on the surface. The platform defines the lateral distance between next neighbors and provides the free volume for unhindered isomerization of the azobenzene units [19,20]. The length and electronic nature of the spacer units control the distance from the surface and define the electronic coupling with the metal surface [11,18]. With increasing π-conjugation from the azobenzene into the platform, and thus coupling to the gold surface, the activation barrier drops to almost zero (≈8 kJ mol−1) and the frequency factors (log A) become negative [11]. Vanishing barriers and low frequency factors are typical for non-adiabatic reactions [9]. The mechanism was elucidated as a singlet–triplet–singlet spin change process, which is forbidden in solution but mediated by coupling to the conduction band of the bulk gold. We are now exploring scope and limitations of this peculiar spin catalysis. To investigate if the reverse isomerization process from the trans to the cis-configuration would also be accelerated, and to further scrutinize the coupling effects, we prepared analogous diazocine systems. Diazocines are bridged azobenzenes [21]. Imposed by the ring strain of the central eight-membered ring, the cis-configuration (boat conformation) is more stable than the trans-isomer (twist conformation). Upon irradiation with ≈400 nm the cis-form switches to the trans-isomer, and irradiation with ≈500 nm or heating leads back to the cis-form [22]. Hence, the diazocines are quasi reversed azobenzenes that are more stable in their trans-configurations [23].
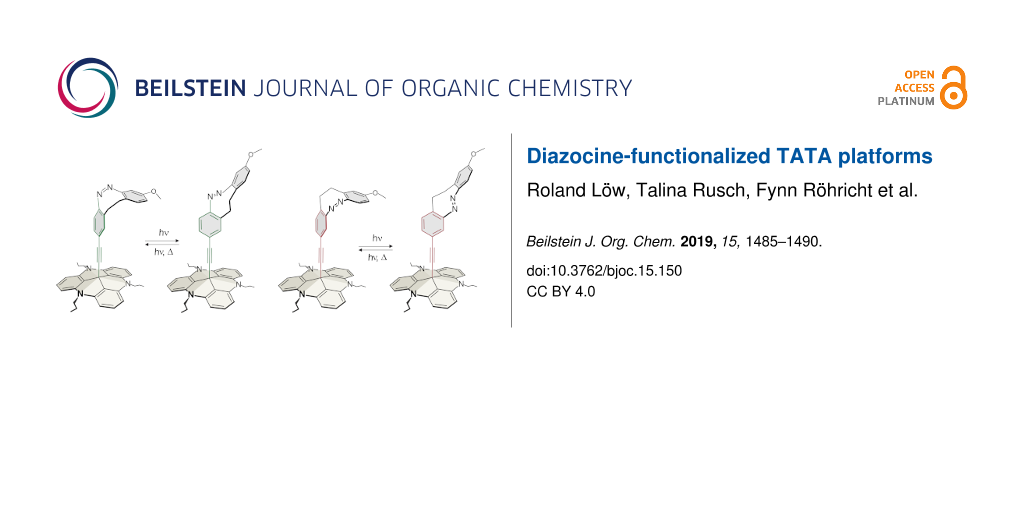
To investigate the electronic coupling effects, we synthesized two diazocine derivatized TATA platforms with ethynyl spacers (diazocine-TATAs). In compound 1 the diazocine is connected to the platform with the ethynyl group in para-position to the azo group, providing a full π-conjugation path of the N=N unit through the ethynyl spacer into the platform. Diazocine-TATA 2 is connected in meta-position and thus interrupting conjugation [24,25]. Both diazocine-TATAs are equipped with methoxy groups, which serve as “reporter units” indicating the configuration of the molecules on metal surfaces [15]. In 1 the OMe group is attached para and in 2 the methoxy group is meta with respect to the azo group. Model calculations predict that the Cphenyl–O bonds in the cis-isomers thus are parallel, and in the trans-isomers orthogonal to the surface (Figure 1). Previous investigations have shown that IRRAS (infrared absorption reflection spectroscopy) in combination with the surface selection rules (stretching mode orthogonal to the surface→high intensity, parallel to the surface→low intensity) is a suitable method to determine the configuration and to measure kinetics on surfaces [15]. The C–O stretching frequencies proved to be ideal reporter signals to determine the configuration and to measure kinetics in monolayers of azo-TATAs on surfaces.
![[1860-5397-15-150-1]](/bjoc/content/figures/1860-5397-15-150-1.png?scale=2.0&max-width=1024&background=FFFFFF)
Figure 1: Structures of diazocine platform molecules (diazocine-TATAs) 1 and 2 in cis (1a, 2a) and trans-configuration (1b, 2b) (octyl side chains are replaced by protons for simplification). The cis-diazocines (1a, 2a) isomerize upon irradiation with 405 nm to the metastable trans-diazocines (1b, 2b) and with 525 nm or thermally back to the cis-diazocines (1a, 2a).
Figure 1: Structures of diazocine platform molecules (diazocine-TATAs) 1 and 2 in cis (1a, 2a) and trans-conf...
Results and Discussion
To obtain information on preferred conformations of 1 and 2 in their cis and trans-configurations and to predict thermodynamic and kinetic stabilities, we performed DFT calculations at the M06-2X/def2-TZVP level of theory (Table 1, for details see Supporting Information File 1, chapter VI). As expected for diazocine-based molecules our calculations predict the cis configuration for both compounds as the thermodynamically most stable isomers. For the corresponding trans-configuration two different conformations were found: the twist and the chair structures. The twist conformation is about 2.5 kcal mol−1 more stable than the chair conformation. Our calculations predict reaction barriers (trans-twist→cis-boat) for both compounds of approximately 23 kcal mol−1 (96 kJ mol−1). Obviously, the TATA platform and the ethynyl spacer have only marginal effects on the isomerization process. Hence, the diazocines 1 and 2 are ideal candidates to investigate the effect of bulk gold as a function of electronic coupling (conjugation) of the azo unit to gold.
Table 1: Calculated quantum chemical energies Erel (M06-2X/def2-TZVP) of the twist and chair conformation of the trans-configuration of para-ethynyl-substituted diazocine 1b (para-diazocine), and meta-diazocine 2b, relative to the boat conformation of the cis-isomers 1a and 2a. ΔH≠ are the calculated reaction barriers (trans-twist→cis- boat). All energies are given in kcal mol−1.
|
Erel
trans twist |
Erel
trans chair |
ΔH≠ | |
| para-diazocine 1 | 7.9 | 10.6 | 22.6 |
| meta-diazocine 2 | 8.0 | 10.3 | 23.0 |
The para-diazocine-TATA 1 was synthesized in a 5-step synthesis route (Scheme 1). Bromotoluene 3 was synthesized as described [26]. In a Sonogashira cross-coupling reaction acetylene-substituted toluene 5 was prepared from bromotoluene 3 with TMS-protected acetylene 4 (95%). The C–C bond formation of 5 and 6 to give dibenzoyl 7 was achieved with potassium butoxide and elemental bromine (9%) according to a literature procedure [27]. The para-ethynyldiazocine 8 was obtained by reduction of both nitro groups, followed by oxidation of the formed hydrazine (16%). The unprotected ethynyldiazocine 8 was deprotonated with potassium hydroxide and connected to the central carbon atom of the TATA platform 9 (synthesized according to Laursen and Krebs [28]) to obtain target para-diazocine mounted on the octyl-substituted TATA platform 1 (99%).
![[1860-5397-15-150-i1]](/bjoc/content/inline/1860-5397-15-150-i1.png?scale=2.0&max-width=1024&background=FFFFFF)
Scheme 1: Synthesis route of para-diazocine platform molecule 1. a) Pd(dppf)Cl2, Cu(I)I, Et3N, 1 h, 60 °C; b) 1: KOt-Bu, THF, 3 min, 0 °C, N2; 2: Br2, 5 min, 0 °C; c) 1: Ba(OH)2, Zn, EtOH, H2O, 4.75 h, reflux; 2: 0.1 M NaOH/MeOH, Cu(II)Cl2, 6 h, rt; d) KOH, THF, 3.5 h, reflux, N2.
Scheme 1: Synthesis route of para-diazocine platform molecule 1. a) Pd(dppf)Cl2, Cu(I)I, Et3N, 1 h, 60 °C; b)...
The synthesis of the meta-diazocine platform molecule 2 was achieved in a 4-step synthesis route (Scheme 2). Nitrotoluene 10 was synthesized as described in literature [29]. The reaction of ethynyltoluene 10 with methoxytoluene 11 gave dibenzoyl 12 (10%) according to the same procedure as for dibenzoyl 7 (Scheme 1). Diazocine 13 was obtained by reduction and oxidation in moderate yields (22%). The reaction of diazocine 13 with the TATA ion 9 gave the target diazocine 2 (88%, Scheme 2).
![[1860-5397-15-150-i2]](/bjoc/content/inline/1860-5397-15-150-i2.png?scale=2.0&max-width=1024&background=FFFFFF)
Scheme 2: Synthesis route of meta-diazocine platform 2. a) 1: KOt-Bu, THF, 3 min, 0 °C, N2; 2: Br2, 5 min, 0 °C, N2; b) 1: Ba(OH)2, Zn, EtOH, H2O, 4.75 h, reflux; 2: 0.1 M NaOH/MeOH, Cu(II)Cl2, 13 h, rt; c) KOH, THF, 2 h, reflux, N2.
Scheme 2: Synthesis route of meta-diazocine platform 2. a) 1: KOt-Bu, THF, 3 min, 0 °C, N2; 2: Br2, 5 min, 0 ...
The photophysical properties and the switching behavior of 1 and 2 were determined in solution (THF). The UV–vis spectra of 1 and 2 are shown before and after irradiation with 405 nm and 525 nm. Both diazocine-TATAs 1 and 2 exhibit similar UV spectra. The n→π* transition of cis-1 appears at 403 nm and at 494 nm in the trans-isomer. The corresponding absorption maxima in diazocine-TATA 2 are 409 nm (cis) and 493 nm (trans) (Figure 2).
![[1860-5397-15-150-2]](/bjoc/content/figures/1860-5397-15-150-2.png?scale=2.0&max-width=1024&background=FFFFFF)
Figure 2: UV–vis spectra of 1 (left) and 2 (right) in THF at room temperature. Black: as synthesized, red: after irradiation with 405 nm, and blue: after irradiation with 525 nm.
Figure 2: UV–vis spectra of 1 (left) and 2 (right) in THF at room temperature. Black: as synthesized, red: af...
The photostationary states of 1 and 2 were determined in toluene-d8 by 1H NMR measurements (Table 2). Optimal wavelengths for the cis→trans isomerization are 405 nm (1: 53% trans, 2: 65% trans). Back-isomerization to the cis-isomer with 525 nm is nearly quantitative. The half-lives (298 K) are similar for both systems (2.12 h for 1 and 2.32 h for 2). The lack of conjugation between the azo function and the ethynyl spacer of 2 yields in a slightly higher half-life, which is in agreement with earlier results [11].
Table 2: Photostationary states (PSS) of para-diazocine-TATA 1 (2.05 mmol/L) and meta-diazocine-TATA 2 (2.27 mmol/L) upon irradiation with light of 405 nm, 525 nm and thermal isomerization half-life (t1/2) determined with 1H NMR spectroscopy (in deuterated toluene). The activation energies (Ea) are calculated from the linear fit of an Arrhenius plot.
| para-diazocine 1 | meta-diazocine 2 | |
|---|---|---|
| PSS (405 nm) | 53% (trans) | 65% (trans) |
| PSS (525 nm) | 93% (cis) | 93% (cis) |
| t1/2 (290.5 K) | 5.27 h | 5.76 h |
| t1/2 (298 K) | 2.12 h | 2.32 h |
| t1/2 (308 K) | 0.69 h | 0.73 h |
| EA (kJ mol−1) | 86.5 | 84.7 |
STM Measurements
The adsorption behavior of the diazocine-TATA molecules on Au(111) surfaces was studied by STM at room temperature (Figure 3). Adlayers of both compounds show a hexagonally ordered superstructure with lattice constants of 1 and 2 being (12.2 ± 0.6) Å and (12.1 ± 0.5) Å, respectively. Additionally, two rotational domains with an angle of (15 ± 4)° are observed. Altogether these parameters are in good agreement with a (√19 × √19) R23.4° superstructure which has been also observed in previous STM investigations of TATA and azobenzene-TATA molecules with octyl ligands [18,20,30].
![[1860-5397-15-150-3]](/bjoc/content/figures/1860-5397-15-150-3.png?scale=2.0&max-width=1024&background=FFFFFF)
Figure 3: STM images (30 × 30 nm², Ubias = 0.3 V, It = 40 pA) of self-assembled monolayers of (a) compound 1 and (b) compound 2 on Au(111).
Figure 3: STM images (30 × 30 nm², Ubias = 0.3 V, It = 40 pA) of self-assembled monolayers of (a) compound 1 ...
Conclusion
In summary, we present the syntheses of two different diazocines mounted on TATA platforms (1, 2). The photochemical switching between the stable cis and metastable trans-isomers was investigated. Upon irradiation with light of 405 nm diazocine-TATAs 1 and 2 convert to their trans-configurations in moderate to good yields. The metastable trans-isomers of 1 and 2 isomerize back to the cis-isomer with half-lives of 2.12 h and 2.32 h at 298 K. The trans→cis activation energies with 86.5 kJ mol−1 for 1 and with 84.7 kJ mol−1 for 2 are similar to the structurally related azobenzenes [11]. Both diazocine-TATAs form highly ordered monolayers on Au(111) surfaces. Further studies will include IRRAS measurements to determine the trans→cis isomerization kinetics on Au(111) surfaces.
Experimental
For detailed experimental procedures, including NMR, UV–vis and MS spectra see Supporting Information File 1, chapters I–IV, for kinetic studies see Supporting Information File 1, chapter V.
Supporting Information
| Supporting Information File 1: Analytical methods, experimental procedures, NMR and UV spectra, kinetic studies and DFT calculations. | ||
| Format: PDF | Size: 2.4 MB | Download |
References
-
Hauffe, K., Ed. Katalyse; DE GRUYTER: Berlin, New York, 1976; p 119. doi:10.1515/9783110829990
Return to citation in text: [1] -
Ostbald, W. Z. Phys. Chem. 1894, 15, 705–706.
Return to citation in text: [1] -
Oyama, S. T.; Somorjai, G. A. J. Chem. Educ. 1988, 65, 765–769. doi:10.1021/ed065p765
Return to citation in text: [1] -
Shriver, D. F.; Atkins, P. W.; Langford, C. H. In Anorganische Chemie, 2nd ed.; Heck, J.; Kaim, W.; Weidenbruch, M., Eds.; Wiley-VCH: Weinheim, Germany, 1997; p 692.
Return to citation in text: [1] -
Eyring, H. J. Chem. Phys. 1935, 3, 107–115. doi:10.1063/1.1749604
Return to citation in text: [1] -
Gowenlock, B. G. Q. Rev., Chem. Soc. 1960, 14, 133–145. doi:10.1039/qr9601400133
Return to citation in text: [1] -
Hauser, A. Coord. Chem. Rev. 1991, 111, 275–290. doi:10.1016/0010-8545(91)84034-3
Return to citation in text: [1] -
Xie, C.-L.; Hendrickson, D. N. J. Am. Chem. Soc. 1987, 109, 6981–6988. doi:10.1021/ja00257a013
Return to citation in text: [1] -
Harvey, J. N. Phys. Chem. Chem. Phys. 2007, 9, 331–343. doi:10.1039/b614390c
Return to citation in text: [1] [2] -
Schreiner, P. R.; Reisenauer, H. P.; Pickard IV, F. C.; Simmonett, A. C.; Allen, W. D.; Mátyus, E.; Császár, A. G. Nature 2008, 453, 906–909. doi:10.1038/nature07010
Return to citation in text: [1] -
Schlimm, A.; Löw, R.; Rusch, T.; Röhricht, F.; Strunskus, T.; Tellkamp, T.; Sönnichsen, F.; Manthe, U.; Magnussen, O.; Tuczek, F.; Herges, R. Angew. Chem., Int. Ed. 2019, 58, 6574–6578. doi:10.1002/anie.201814342
Return to citation in text: [1] [2] [3] [4] [5] -
Hartley, G. S. J. Chem. Soc. 1938, 633–642. doi:10.1039/jr9380000633
Return to citation in text: [1] [2] -
Shinkai, S.; Kusano, Y.; Shigematsu, K.; Manabe, O. Chem. Lett. 1980, 9, 1303–1306. doi:10.1246/cl.1980.1303
Return to citation in text: [1] -
Jung, U.; Schütt, C.; Filinova, O.; Kubitschke, J.; Herges, R.; Magnussen, O. J. Phys. Chem. C 2012, 116, 25943–25948. doi:10.1021/jp310451c
Return to citation in text: [1] -
Jacob, H.; Ulrich, S.; Jung, U.; Lemke, S.; Rusch, T.; Schütt, C.; Petersen, F.; Strunskus, T.; Magnussen, O.; Herges, R.; Tuczek, F. Phys. Chem. Chem. Phys. 2014, 16, 22643–22650. doi:10.1039/c4cp03438d
Return to citation in text: [1] [2] [3] -
Krekiehn, N. R.; Müller, M.; Jung, U.; Ulrich, S.; Herges, R.; Magnussen, O. M. Langmuir 2015, 31, 8362–8370. doi:10.1021/acs.langmuir.5b01645
Return to citation in text: [1] -
Hagen, S.; Kate, P.; Peters, M. V.; Hecht, S.; Wolf, M.; Tegeder, P. Appl. Phys. A: Mater. Sci. Process. 2008, 93, 253–260. doi:10.1007/s00339-008-4831-5
Return to citation in text: [1] -
Baisch, B.; Raffa, D.; Jung, U.; Magnussen, O. M.; Nicolas, C.; Lacour, J.; Kubitschke, J.; Herges, R. J. Am. Chem. Soc. 2009, 131, 442–443. doi:10.1021/ja807923f
Return to citation in text: [1] [2] [3] -
Ulrich, S.; Jung, U.; Strunskus, T.; Schütt, C.; Bloedorn, A.; Lemke, S.; Ludwig, E.; Kipp, L.; Faupel, F.; Magnussen, O.; Herges, R. Phys. Chem. Chem. Phys. 2015, 17, 17053–17062. doi:10.1039/c5cp01447f
Return to citation in text: [1] -
Kuhn, S.; Baisch, B.; Jung, U.; Johannsen, T.; Kubitschke, J.; Herges, R.; Magnussen, O. Phys. Chem. Chem. Phys. 2010, 12, 4481. doi:10.1039/b922882a
Return to citation in text: [1] [2] -
Duval, H. Bull. Soc. Chim. Fr. 1910, 7, 727.
Return to citation in text: [1] -
Siewertsen, R.; Neumann, H.; Buchheim-Stehn, B.; Herges, R.; Näther, C.; Renth, F.; Temps, F. J. Am. Chem. Soc. 2009, 131, 15594–15595. doi:10.1021/ja906547d
Return to citation in text: [1] -
Hartley, G. S. Nature 1937, 140, 281. doi:10.1038/140281a0
Return to citation in text: [1] -
Yaliraki, S. N.; Ratner, M. A. Ann. N. Y. Acad. Sci. 2002, 960, 153–162. doi:10.1111/j.1749-6632.2002.tb03030.x
Return to citation in text: [1] -
Mayor, M.; Büschel, M.; Fromm, K. M.; Lehn, J.-M.; Daub, J. Ann. N. Y. Acad. Sci. 2002, 960, 16–28. doi:10.1111/j.1749-6632.2002.tb03022.x
Return to citation in text: [1] -
Wahhab, A.; Therrien, E. Small Molecule Inhibitors of Protein Arginine methyltransferases. PCT Pat. Appl. WO2008104077 A1, Feb 28, 2007.
Return to citation in text: [1] -
Moormann, W.; Langbehn, D.; Herges, R. Synthesis 2017, 49, 3471–3475. doi:10.1055/s-0036-1590685
Return to citation in text: [1] -
Laursen, B. W.; Krebs, F. C. Chem. – Eur. J. 2001, 7, 1773–1783. doi:10.1002/1521-3765(20010417)7:8<1773::aid-chem17730>3.0.co;2-f
Return to citation in text: [1] -
Park, K.; Lee, B. M. Novel phenylethynyl benzamide glucokinase activator and method for preparing same. PCT Pat. Appl. WO2014112798 A1, Jan 16, 2013.
Return to citation in text: [1] -
Lemke, S.; Ulrich, S.; Claußen, F.; Bloedorn, A.; Jung, U.; Herges, R.; Magnussen, O. M. Surf. Sci. 2015, 632, 71–76. doi:10.1016/j.susc.2014.08.028
Return to citation in text: [1]
| 1. | Hauffe, K., Ed. Katalyse; DE GRUYTER: Berlin, New York, 1976; p 119. doi:10.1515/9783110829990 |
| 2. | Ostbald, W. Z. Phys. Chem. 1894, 15, 705–706. |
| 3. | Oyama, S. T.; Somorjai, G. A. J. Chem. Educ. 1988, 65, 765–769. doi:10.1021/ed065p765 |
| 4. | Shriver, D. F.; Atkins, P. W.; Langford, C. H. In Anorganische Chemie, 2nd ed.; Heck, J.; Kaim, W.; Weidenbruch, M., Eds.; Wiley-VCH: Weinheim, Germany, 1997; p 692. |
| 22. | Siewertsen, R.; Neumann, H.; Buchheim-Stehn, B.; Herges, R.; Näther, C.; Renth, F.; Temps, F. J. Am. Chem. Soc. 2009, 131, 15594–15595. doi:10.1021/ja906547d |
| 11. | Schlimm, A.; Löw, R.; Rusch, T.; Röhricht, F.; Strunskus, T.; Tellkamp, T.; Sönnichsen, F.; Manthe, U.; Magnussen, O.; Tuczek, F.; Herges, R. Angew. Chem., Int. Ed. 2019, 58, 6574–6578. doi:10.1002/anie.201814342 |
| 7. | Hauser, A. Coord. Chem. Rev. 1991, 111, 275–290. doi:10.1016/0010-8545(91)84034-3 |
| 8. | Xie, C.-L.; Hendrickson, D. N. J. Am. Chem. Soc. 1987, 109, 6981–6988. doi:10.1021/ja00257a013 |
| 9. | Harvey, J. N. Phys. Chem. Chem. Phys. 2007, 9, 331–343. doi:10.1039/b614390c |
| 10. | Schreiner, P. R.; Reisenauer, H. P.; Pickard IV, F. C.; Simmonett, A. C.; Allen, W. D.; Mátyus, E.; Császár, A. G. Nature 2008, 453, 906–909. doi:10.1038/nature07010 |
| 5. | Eyring, H. J. Chem. Phys. 1935, 3, 107–115. doi:10.1063/1.1749604 |
| 6. | Gowenlock, B. G. Q. Rev., Chem. Soc. 1960, 14, 133–145. doi:10.1039/qr9601400133 |
| 18. | Baisch, B.; Raffa, D.; Jung, U.; Magnussen, O. M.; Nicolas, C.; Lacour, J.; Kubitschke, J.; Herges, R. J. Am. Chem. Soc. 2009, 131, 442–443. doi:10.1021/ja807923f |
| 11. | Schlimm, A.; Löw, R.; Rusch, T.; Röhricht, F.; Strunskus, T.; Tellkamp, T.; Sönnichsen, F.; Manthe, U.; Magnussen, O.; Tuczek, F.; Herges, R. Angew. Chem., Int. Ed. 2019, 58, 6574–6578. doi:10.1002/anie.201814342 |
| 18. | Baisch, B.; Raffa, D.; Jung, U.; Magnussen, O. M.; Nicolas, C.; Lacour, J.; Kubitschke, J.; Herges, R. J. Am. Chem. Soc. 2009, 131, 442–443. doi:10.1021/ja807923f |
| 14. | Jung, U.; Schütt, C.; Filinova, O.; Kubitschke, J.; Herges, R.; Magnussen, O. J. Phys. Chem. C 2012, 116, 25943–25948. doi:10.1021/jp310451c |
| 15. | Jacob, H.; Ulrich, S.; Jung, U.; Lemke, S.; Rusch, T.; Schütt, C.; Petersen, F.; Strunskus, T.; Magnussen, O.; Herges, R.; Tuczek, F. Phys. Chem. Chem. Phys. 2014, 16, 22643–22650. doi:10.1039/c4cp03438d |
| 16. | Krekiehn, N. R.; Müller, M.; Jung, U.; Ulrich, S.; Herges, R.; Magnussen, O. M. Langmuir 2015, 31, 8362–8370. doi:10.1021/acs.langmuir.5b01645 |
| 17. | Hagen, S.; Kate, P.; Peters, M. V.; Hecht, S.; Wolf, M.; Tegeder, P. Appl. Phys. A: Mater. Sci. Process. 2008, 93, 253–260. doi:10.1007/s00339-008-4831-5 |
| 11. | Schlimm, A.; Löw, R.; Rusch, T.; Röhricht, F.; Strunskus, T.; Tellkamp, T.; Sönnichsen, F.; Manthe, U.; Magnussen, O.; Tuczek, F.; Herges, R. Angew. Chem., Int. Ed. 2019, 58, 6574–6578. doi:10.1002/anie.201814342 |
| 13. | Shinkai, S.; Kusano, Y.; Shigematsu, K.; Manabe, O. Chem. Lett. 1980, 9, 1303–1306. doi:10.1246/cl.1980.1303 |
| 19. | Ulrich, S.; Jung, U.; Strunskus, T.; Schütt, C.; Bloedorn, A.; Lemke, S.; Ludwig, E.; Kipp, L.; Faupel, F.; Magnussen, O.; Herges, R. Phys. Chem. Chem. Phys. 2015, 17, 17053–17062. doi:10.1039/c5cp01447f |
| 20. | Kuhn, S.; Baisch, B.; Jung, U.; Johannsen, T.; Kubitschke, J.; Herges, R.; Magnussen, O. Phys. Chem. Chem. Phys. 2010, 12, 4481. doi:10.1039/b922882a |
| 15. | Jacob, H.; Ulrich, S.; Jung, U.; Lemke, S.; Rusch, T.; Schütt, C.; Petersen, F.; Strunskus, T.; Magnussen, O.; Herges, R.; Tuczek, F. Phys. Chem. Chem. Phys. 2014, 16, 22643–22650. doi:10.1039/c4cp03438d |
| 24. | Yaliraki, S. N.; Ratner, M. A. Ann. N. Y. Acad. Sci. 2002, 960, 153–162. doi:10.1111/j.1749-6632.2002.tb03030.x |
| 25. | Mayor, M.; Büschel, M.; Fromm, K. M.; Lehn, J.-M.; Daub, J. Ann. N. Y. Acad. Sci. 2002, 960, 16–28. doi:10.1111/j.1749-6632.2002.tb03022.x |
| 15. | Jacob, H.; Ulrich, S.; Jung, U.; Lemke, S.; Rusch, T.; Schütt, C.; Petersen, F.; Strunskus, T.; Magnussen, O.; Herges, R.; Tuczek, F. Phys. Chem. Chem. Phys. 2014, 16, 22643–22650. doi:10.1039/c4cp03438d |
| 11. | Schlimm, A.; Löw, R.; Rusch, T.; Röhricht, F.; Strunskus, T.; Tellkamp, T.; Sönnichsen, F.; Manthe, U.; Magnussen, O.; Tuczek, F.; Herges, R. Angew. Chem., Int. Ed. 2019, 58, 6574–6578. doi:10.1002/anie.201814342 |
| 11. | Schlimm, A.; Löw, R.; Rusch, T.; Röhricht, F.; Strunskus, T.; Tellkamp, T.; Sönnichsen, F.; Manthe, U.; Magnussen, O.; Tuczek, F.; Herges, R. Angew. Chem., Int. Ed. 2019, 58, 6574–6578. doi:10.1002/anie.201814342 |
| 18. | Baisch, B.; Raffa, D.; Jung, U.; Magnussen, O. M.; Nicolas, C.; Lacour, J.; Kubitschke, J.; Herges, R. J. Am. Chem. Soc. 2009, 131, 442–443. doi:10.1021/ja807923f |
| 20. | Kuhn, S.; Baisch, B.; Jung, U.; Johannsen, T.; Kubitschke, J.; Herges, R.; Magnussen, O. Phys. Chem. Chem. Phys. 2010, 12, 4481. doi:10.1039/b922882a |
| 30. | Lemke, S.; Ulrich, S.; Claußen, F.; Bloedorn, A.; Jung, U.; Herges, R.; Magnussen, O. M. Surf. Sci. 2015, 632, 71–76. doi:10.1016/j.susc.2014.08.028 |
| 28. | Laursen, B. W.; Krebs, F. C. Chem. – Eur. J. 2001, 7, 1773–1783. doi:10.1002/1521-3765(20010417)7:8<1773::aid-chem17730>3.0.co;2-f |
| 29. | Park, K.; Lee, B. M. Novel phenylethynyl benzamide glucokinase activator and method for preparing same. PCT Pat. Appl. WO2014112798 A1, Jan 16, 2013. |
| 26. | Wahhab, A.; Therrien, E. Small Molecule Inhibitors of Protein Arginine methyltransferases. PCT Pat. Appl. WO2008104077 A1, Feb 28, 2007. |
| 27. | Moormann, W.; Langbehn, D.; Herges, R. Synthesis 2017, 49, 3471–3475. doi:10.1055/s-0036-1590685 |
© 2019 Löw et al.; licensee Beilstein-Institut.
This is an Open Access article under the terms of the Creative Commons Attribution License (http://creativecommons.org/licenses/by/4.0). Please note that the reuse, redistribution and reproduction in particular requires that the authors and source are credited.
The license is subject to the Beilstein Journal of Organic Chemistry terms and conditions: (https://www.beilstein-journals.org/bjoc)