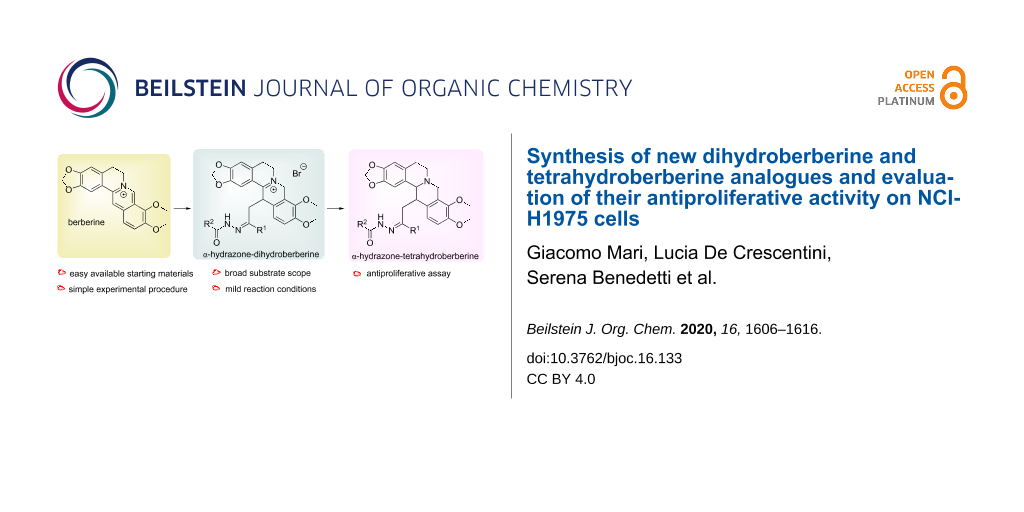
Abstract
Dihydroberberine (DHBER), the partially reduced form of the alkaloid berberine (BER), is known to exhibit important biological activities. Despite this fact, there have been only few studies that concern the biological properties of functionalized DHBER. Attracted by the potentiality of this latter compound, we have realized the preparation of new arylhydrazono-functionalized DHBERs, starting from BER and some α-bromohydrazones. On the other hand, also the fully reduced form of BER, namely tetrahydroberberine (THBER), and its derivatives have proven to present different biological activities. Therefore, the obtained arylhydrazono-functionalized DHBERs were reduced to the corresponding arylhydrazono-THBERs. The antiproliferative activity of both arylhydrazono-DHBERs and -THBERs has been evaluated on NCI-H1975 lung cancer cells.
Graphical Abstract
Introduction
The rhizome of Coptis chinensis Franch. is a common remedy in traditional oriental medicine for the treatment of various inflammatory diseases. The main component of this rhizome is berberine (BER), an alkaloid with numerous pharmacological properties, which include anticancer and anti-inflammatory activities [1]. However, the low bioavailability, poor solubility, and moderate nucleic acid binding affinity constitute a severe limitation in the employ of BER [2-4].
To minimize these drawbacks, its isoquinoline portion was derivatized in positions 9 and 13 that are critical for topoisomerase inhibition and quadruplex structure binding [5-8]. The insertion of a phenyl group or a benzhydryl group linked to position 13 of the BER skeleton, usually causes a geometric propensity for additional stacking-type, noncovalent aromatic interactions with cellular targets forming stronger complexes with nucleic acids than BER [9-12]. In fact, the so obtained functionalized BER shows better anticancer activity [13-19], and an increased DNA and RNA binding efficacy [4,6,9], due to its aromatic interactions with the biological macromolecules [20].
Another interesting and promising derivative is dihydroberberine (DHBER), the reduced form of BER. The enaminic function of this alkaloid is a precious reactive site, employed to insert various electrophilic agents in position 13 of the DHBER skeleton [21-24]. Usually, to remedy the poor stability of the DHBER-derivatives obtained, they are directly oxidized to the corresponding functionalized BERs, relegating to DHBERs the role of intermediates. However, pharmacokinetic studies have shown that, compared with BER [25], DHBER displayed improved adsorption and enhanced bioavailability, as also confirmed by several papers that describe how the gut microbiota converts BER derivatives into its absorbable form DHBER, which has an intestinal absorption rate 5-fold higher than that of BER [26-28].
Currently, DHBER is a drug candidate for the treatment of type 2 diabetes [29,30], can reduce the atherosclerotic plaque size [31], has therapeutic potential for myotonic dystrophy type I due to its central nervous system effects [32], inhibits both hERG current and the expression of hERG protein [33], inhibits the pancreatic lipase [34], shows antiradical, revitalizing and antifibrotic properties for dermatological applications [35], manifests a synergic effect with antibiotics [36], and displays antitumoral activities [37,38].
Unlike BER, there have been few studies that concern the biological properties of functionalized DHBER. Attracted by the potentiality of this latter compound, we have realized the preparation of new arylhydrazono-functionalized DHBERs that were tested in vitro for their biological activity; in particular, we focused our attention on their antiproliferative capacity, due to our previous research experiences on the anticancer properties of both natural and synthetic molecules [39-42]. A common strategy to increase the biological activity of a class of products can be the insertion in the original molecule of different functions with their own peculiar features. Often, the biological properties of the resultant derivatives are not simply attributable to the sum of the characteristics shown by the individual moieties, but synergistic effects can increase their effectiveness, or induce the manifestation of new features. The choice to insert an hydrazonic function on the DHBER skeleton was suggested by its wide range of interesting biological activities like antimicrobial [43], antioxidant [44], analgesic [45], anti-inflammatory [45], antiplatelet [45], anticonvulsant [46], antiprotozoal [47], antidiabetic [48], and antitubercular [49]. Relevant is the role of the hydrazone moiety as antitumor agent [50-53]. An interesting example reported by Ferreira demonstrates that the chemical derivatization of the indole alkaloids dregamine and tabernaemontanine to yield new hydrazone derivatives enhances the apoptosis inducing activity [54].
Another particularly useful feature of the hydrazone group is related by its high tendency to provide solid derivatives. The target of the here reported strategy is to recover as pure products the desired functionalized DHBERs to evaluate their antiproliferative activity directly by precipitation from the reaction medium, avoiding any possible and common subsequent side reactions for this substrate during the purification processes (Scheme 1).
Scheme 1: Preparation of functionalized-BER, -DHBER and -THBER derivatives.
Scheme 1: Preparation of functionalized-BER, -DHBER and -THBER derivatives.
On the other hand, also the fully reduced form of BER, namely tetrahydroberberine (THBER) or canadine, is an isoquinoline alkaloid recurring in several plant species [55-57]. THBER is able to act to the central nervous system as an inhibitor [58], possesses hepatoprotective effects [59], is employed in the protection against cerebral ischemia-reperfusion injury [60], and shows anti-arrhythmic activity [61]. Furthermore, THBER and its derivatives are effective antioxidant agents [62-65].
To extend our investigations in this field, the obtained arylhydrazono-functionalized DHBERs were reduced to the corresponding arylhydrazono-THBERs, whose antiproliferative properties have been evaluated (Scheme 1).
Results and Discussion
Chemistry
The unsubstituted DHBER was prepared by treating BER with 2.5 equivalents of NaBH4 in pyridine at room temperature [66,67], and employed in the reaction with α-bromohydrazone 1a chosen as the representative model, to determine the optimal reaction conditions for the synthesis of the corresponding arylhydrazono-functionalized DHBER 2a. Different solvents, temperatures and molar ratios were investigated (Table 1).
Dichloromethane (DCM), room temperature, and an equimolar ratio have proved to be the best conditions, since the desired product 2a was achieved as a pure pale yellow solid directly from the reaction medium with the best yields (Table 1, entry 7). In tetrahydrofuran (THF), in ethyl acetate (EtOAc), and in alcohols such as methanol, ethanol, isopropanol (MeOH, EtOH, iPrOH) the reaction furnishes a complicate profile, where the desired 2a is detectable only in traces (Table 1, entries 1–5). The monitoring by TLC of the reaction conducted in acetonitrile reveals the formation of derivative 2a, which, however, does not precipitate in the reaction medium (Table 1, entry 6).
Table 1: Reaction conditions optimization.
|
|
||||
| Entrya | Solvent | T (°C) |
Molar ratio
(DHBER/1a) |
Yield of 2a (%)b |
| 1 | THF | Rt | 1:1 | –c |
| 2 | EtOAc | rt | 1:1 | –c |
| 3 | MeOH | rt | 1:1 | –c |
| 4 | EtOH | rt | 1:1 | –c |
| 5 | iPrOH | rt | 1:1 | –c |
| 6 | ACN | rt | 1:1 | –d |
| 7 | DCM | rt | 1:1 | 66 |
| 8 | DCMe | rt | 1:1 | –f |
| 9 | DCM | rt | 1:2 | –c |
| 10 | DCM | 0 | 1:1 | 25 |
| 11 | DCM | reflux | 1:1 | 54 |
aThe reactions were conducted on 0.3 mmol scale in 1.0 mL of solvent. bIsolated yields. cComplicated mixture. d2a is detectable on TLC, but it does not precipitate from the reaction medium. The chromatographic separation does not furnish 2a, probably due to degradation processes that occur in the column. eThe reactions were conducted on 0.3 mmol scale in 0.5 mL of solvent. fThe starting materials DHBER and 1a are not completely dissolved.
The spontaneous precipitation is particularly relevant to prevent any further degradation process. In fact, all attempts to obtain the pure 2a by chromatographic process failed, employing silica gel or aluminum oxide or basified silica gel (by addition of 3% of triethylamine).
Having identified the optimal conditions, the reaction scope was enlarged employing the α-bromohydrazones 1a–n (Scheme 2).
Scheme 2: Substrate scope of hydrazono-DHBER. Reaction conditions: DHBER (1.0 mmol), 1 (1.0 mmol), DCM 3.0 mL, 25 °C. Isolated yields in parentheses.
Scheme 2: Substrate scope of hydrazono-DHBER. Reaction conditions: DHBER (1.0 mmol), 1 (1.0 mmol), DCM 3.0 mL...
All the hydrazono-DHBERs 2a–n directly precipitate as pure products in the reaction medium with yields ranging from poor to good (30–83%). Probably the low affinity of the iminium and hydrazone groups of compounds 2a–n with a medium-low polarity solvent such as DCM causes their direct precipitation.
The mono- and two-dimensional NMR studies confirm that hydrazono-DHBER 2a–n are in the iminium-tautomeric form. For example, the COSY experiment of hydrazono-DHBER 2a clearly indicates the presence of three sets of related signals: the system with the triplet at 5.24 ppm, attributable to the proton bound to the carbon 13, coupled with the multiplet at 2.54–2.60 ppm, attributable to the methylene in position 15, is diagnostic for the structure proposed (highlighted in green, Figure 1). A second set, two doublets at 5.06 and 5.32 ppm, is attributable to the diastereotopic protons bound to carbon in position 8 (highlighted in blue, Figure 1). The last group, three multiplets at 2.54–2.60, 2.93–2.99 and 3.93–4.10 ppm, is ascribable to the protons of the carbons 5 and 6, respectively.
Figure 1: 1H,1H-COSY spectrum of DHBER 2a [65].
Figure 1: 1H,1H-COSY spectrum of DHBER 2a [65].
On the other hand, we have recently demonstrated that some structurally complex THBER analogues, namely pyrrolino-tetrahydroberberines, synthesized by some of us [68], exhibited enhanced antioxidant properties in comparison to THBER against a wide variety of pathophysiologically relevant oxidants such as peroxyl radicals, ferrous ion, and hydrogen peroxide [65].
In continuation of our ongoing interest, the hydrazono-DHBERs 2a–n were further treated with 2.0 equivalents of sodium boron hydride at room temperature in methanol. The reaction furnishes the corresponding hydrazono-THBERs 3a–n in good yields (77–96%, Scheme 3).
Scheme 3: Synthesis of hydrazono-THBERs 3a–n. Reaction conditions: DHBER (0.5 mmol), NaBH4 (2.0 mmol), MeOH, 3.0 mL, 25 °C. Isolated yields in parentheses.
Scheme 3: Synthesis of hydrazono-THBERs 3a–n. Reaction conditions: DHBER (0.5 mmol), NaBH4 (2.0 mmol), MeOH, ...
It is noteworthy that in these conditions, only the iminium moiety is reduced, leaving unchanged the hydrazone function. The presence of the hydrazone moiety is confirmed by the HMBC spectrum in which the signal at 148.9 ppm coupled with the double doublet at 7.66 ppm is attributable to the protons in ortho position of the phenyl bound to the carbon 16 of THBER 3a (Figure 2).
![[1860-5397-16-133-2]](/bjoc/content/figures/1860-5397-16-133-2.png?scale=2.0&max-width=1024&background=FFFFFF)
Figure 2: Detail of the hydrazone signal in the HMBC spectrum of THBER 3a.
Figure 2: Detail of the hydrazone signal in the HMBC spectrum of THBER 3a.
From the comparison between the 1H NMR spectra of DHBER 2a and THBER 3a, it can also be noted as the chemical shifts of 6-C, and 8-C protons, which are the closest to the N-7 nitrogen atom are significantly shifted downfield (assigned by 2D-NMR analysis, see Supporting Information File 1). For example, the two doublets at 5.06 and 5.36 ppm, assigned to the C8 protons of DHBER 2a, are shifted to 3.55 and 4.15 ppm, respectively in THBER 3a (signals highlighted in blue, Figure 3). Similar considerations can be extended to protons bound to C5, C6, (signals highlighted in red, Figure 3), and to protons of 13a-C, 13-C and 15-C (signals highlighted in green, Figure 3).
Figure 3: Comparison between 1H NMR spectra of DHBER 2a and of THBER 3a.
Figure 3: Comparison between 1H NMR spectra of DHBER 2a and of THBER 3a.
It is noteworthy that in the reduction process of the DHBER 2 precursors, a single diastereoisomer of the corresponding THBERs 3 is produced according to 1H and 13C NMR data.
In particular, the NOE correlation between C13−H and C13a−H of THBERs 3a chosen as representative example, reveals that these two hydrogens are in cis position (Figure 4). Therefore, the THBERs 3 show the same configuration of the metabolite cavidine that is the 13-methyltetrahydroprotoberberine alkaloid which occur in various species of Corydalis [69].
Figure 4: NOE correlation between C13−H and C13a−H of THBER 3a.
Figure 4: NOE correlation between C13−H and C13a−H of THBER 3a.
Evaluation of the antiproliferative activity of hydrazono-DHBERs 2 and hydrazono-THBERs 3
As regards the in vitro experiments on lung cancer cells, two different tests were applied to evaluate the effects of hydrazono-DHBERs 2a–n and hydrazono-THBERs 3a–g,i–n [70] on the NCI-H1975 cell proliferation. Both WST-8 [2-(2-methoxy-4-nitrophenyl)-3-(4-nitrophenyl)-5-(2,4-disulfophenyl)-2H-tetrazolium, monosodium salt] (Figure 5) and SRB (sulforhodamine B) (Figure 6) assays revealed that DHBER significantly reduced the cancer cell proliferation as compared to untreated cells both at 24 and 48 h of incubation, accordingly to previous findings on cultured tumor cells. On the contrary, THBER did not affect the NCI-H1975 viability. Among hydrazono-DHBERs, 2m and 2n showed the highest antiproliferative properties, quite comparable to those of DHBER. Referring to hydrazono-THBERs, 3a, 3b, and 3g were the most effective in significantly reducing cancer cell proliferation as compared to THBER and untreated control cells. Overall, hydrazono-DHBERs presented a higher antiproliferative capacity than hydrazono-THBERs, making these compounds particularly of interest from a chemical and biological point of view.
![[1860-5397-16-133-5]](/bjoc/content/figures/1860-5397-16-133-5.png?scale=2.0&max-width=1024&background=FFFFFF)
Figure 5: Effects of 25 µM hydrazono-DHBERs 2a–n (A) and hydrazono-THBERs 3a–g,i–n [70] (B) on NCI-H1975 cell proliferation as evaluated by WST-8 assay (estimating cell metabolic activity) at 24 h and 48 h of treatment. Data are expressed as mean ± SD (n = 3). *p < 0.05 vs CTR (DMSO 0.05%).
Figure 5: Effects of 25 µM hydrazono-DHBERs 2a–n (A) and hydrazono-THBERs 3a–g,i–n [70] (B) on NCI-H1975 cell pro...
![[1860-5397-16-133-6]](/bjoc/content/figures/1860-5397-16-133-6.png?scale=2.0&max-width=1024&background=FFFFFF)
Figure 6: Effects of 25 µM hydrazono-DHBERs 2a–n and hydrazono-THBERs 3a–g,i–n [70] on NCI-H1975 cell proliferation as evaluated by SRB assay (estimating total protein content) at 24 h and 48 h of treatment. Data are expressed as mean ± SD (n = 3). *p < 0.05 vs CTR (DMSO 0.05%).
Figure 6: Effects of 25 µM hydrazono-DHBERs 2a–n and hydrazono-THBERs 3a–g,i–n [70] on NCI-H1975 cell proliferati...
Conclusion
In conclusion, here we report a novel simple, and convenient methodology for easy access to a series of arylhydrazone-functionalized dihydroberberines and tetrahydroberberines. Both the procedures proceed rapidly to completion, under mild reaction conditions, requiring readily available and inexpensive starting materials.
The purification of the hydrazono-dihydroberberines by sole filtration, avoiding any solvent-consuming steps, such as chromatography, extraction or recrystallization, determines a very simple workup. This aspect, together with the total efficiency in atom economy observed, makes this procedure advantageous from both the environmental and the economic point of view.
Also, the regioselective reduction of the iminium moiety that leaves unchanged the hydrazono function requires a simple treatment of the hydrazone dihydroberberines with sodium borohydride producing a new class of tetrahydroberberine analogues with interesting biological properties. Indeed, the investigation on the antiproliferative effects highlighted the ability of some hydrazono-DHBERs and hydrazono-THBERs to induce a significant growth inhibition on the tested cancer cell line, which might deserve future researches.
Experimental
General experimental details: All chemicals and solvents were purchased from commercial suppliers and used as received. α-Bromohydrazones [71] and DHBER [66,67] were prepared as previously reported. Melting points were determined in open capillary tubes and are uncorrected. FTIR spectra were obtained as nujol mulls. All 1H NMR and 13C NMR spectra were recorded at 400 and 100 MHz, respectively. Proton and carbon spectra were referenced internally to solvent signals, using values of δ = 2.50 ppm for proton (middle peak) and δ = 39.50 ppm for carbon (middle peak) in DMSO-d6 and δ = 7.27 ppm for proton and δ = 77.00 ppm for carbon (middle peak) in CDCl3. All coupling constants (J) are given in Hz. All the NH exchanged with D2O. Precoated aluminium oxide plates 0.25 mm were employed for analytical thin-layer chromatography. All new compounds showed satisfactory elemental analysis. Mass spectra were recorded in the ESI and EI modes. The nomenclature was generated using ACD/IUPAC Name (version 3.50, 5 Apr. 1998), Advanced Chemistry Development Inc., Toronto, ON (Canada).
General procedure for the synthesis of hydrazono-dihydroberberines (DHBERs) 2a–n. To a solution in dichloromethane (3.0 mL) of α-bromohydrazones 1a–n [71] (1.0 mmol) was added the unsubstitued dihydroberberine [66,67] (1.0 mmol). The reaction was allowed to stand at room temperature under magnetic stirring until the complete disappearance of the starting materials (TLC monitoring) and the formation of compounds 2a–n that directly precipitated from the reaction medium. Compounds 2a–n were collected by filtration under vacuum and washed with acetone (5.0 mL). Characterization data of 2a, chosen as representative compound, are given below.
13-(2-(2-(tert-Butoxycarbonyl)hydrazono)-2-phenylethyl)-9,10-dimethoxy-5,6,8,13-tetrahydro-[1,3]dioxolo[4,5-g]isoquinolino[3,2-a]isoquinolin-7-ium bromide (2a): 2a was isolated by precipitation in the reaction medium (DCM) in 66% yield (428 mg). Pale yellow amorphous solid; mp: 123–124 °C; 1H NMR (400 MHz, DMSO-d6, 25 °C) δ 1.45 (s, 9H, C(CH3)3), 2.54–2.60 and 2.93–2.99 (2m, 2H, C(5)H2), 3.36–3.43 (m, 2H, C(15)H2), 3.78 (s, 3H, OCH3), 3.83 (s, 3H, OCH3), 3.93–4.10 (m, 2H, C(6)H2), 5.06 (d, J = 19.6 Hz, 1H, C(8)H2), 5.24 (t, J = 6.8 Hz, 1H, CH), 5.32 (d, J = 19.6 Hz, 1H, C(8)H2), 6.25 and 6.27 (2s, 2H, OC(2)H2O), 7.05 (s, 1H, C(14)H), 7.17 (s, 2H, C(12)H and C(11)H), 7.29–7.38 (2m, 3H, ArH), 7.47–7.49 (2m, 2H, ArH), 7.61 (s, 1H, C(4)H), 9.71 (s, 1H, NH); 13C NMR (100 MHz, DMSO-d6, 25 °C) δ 24.7 (t), 27.9 (q), 31.7 (t), 38.1 (d), 50.5 (t), 52.6 (t), 56.0 (q), 60.3 (q), 79.6 (s), 103.1 (t), 108.4 (d), 108.9 (d), 113.6 (d), 118.7 (s), 122.5 (d), 123.0 (s), 123.5 (s), 126.2 (d), 128.2 (d), 128.9 (d), 136.5 (s), 137.0 (s), 143.6 (s), 147.1 (s), 151.5 (s), 152.6 (s), 154.3 (s), 164.0 (s), 172.1 (s); IR (nujol): νmax = 3196, 3039, 1738, 1727, 1700 cm−1; ESIMS (m/z): [M − Br]+ 570; anal. calcd. for C33H36BrN3O6 (650.56): C, 60.92; H, 5.58; N, 6.46; found: C, 61.04; H, 5.54; N, 6.40.
General procedure for the synthesis of hydrazono-tetrahydroberberine (THBERs) 3a–n: To a solution of hydrazono-DHBERs 2a–n (0.2 mmol) in methanol (2.0 mL) at room temperature sodium borohydride (0.4 mmol) was added. The reaction was allowed to stand at room temperature under magnetic stirring until the complete disappearance of the starting hydrazono-DHBERs 2a–n (2.0–3.0 h, TLC monitoring). The reaction solvent was then evaporated under reduced pressure. The crude mixture was then purified by column chromatography on silica gel (elution with cyclohexane/ethyl acetate mixtures) to afford products 3a–n that were crystallized in a mixture of diethyl ether/petroleum ether. Characterization data of 3a, chosen as representative compound, are given below.
tert-Butyl 2-(2-(9,10-dimethoxy-6,8,13,13a-tetrahydro-5H-[1,3]dioxolo[4,5-g]isoquinolino[3,2-a]isoquinolin-13-yl)-1-phenylethylidene)hydrazinecarboxylate (3a): 3a was isolated by chromatographic column on silica gel (ethyl acetate/cyclohexane, 30:70) in 95% yield (109 mg). White amorphous solid; mp: 162–164 °C; 1H NMR (400 MHz, DMSO-d6, 25 °C) δ 1.45 (s, 9H, C(CH3)3), 2.29–2.34 (m, 1H, C(15)H2), 2.62–2.75 (m, 3H, C(15)H2, C(5)H2, C(6)H2), 3.04–3.12 (m, 1H, C(5)H2), 3.23–3.26 (m, 1H, C(6)H2), 3.55 (d, J = 15.6 Hz, 1H, C(8)H2), 3.67 (s, 3H, OCH3), 3.70 (s, 3H, OCH3), 3.84 (brs, 1H, C(13a)H), 3.94–3.98 (m, 1H, C(13)H), 4.15 (d, J = 16.0 Hz, 1H, C(8)H2), 5.99 and 6.00 (2s, 2H, OC(2)H2O), 6.41 (d, J = 8.4 Hz, 1H, C(12)H), 6.57 (d, J = 8.8 Hz, 1H, C(11)H), 6.75 (s, 1H, C(4)H), 7.03 (s, 1H, C(14)H), 7.32–7.38 (m, 3H, ArH), 7.63–7.65 (m, 2H, ArH), 11.04 (brs, 1H, NH); 13C NMR (100 MHz, DMSO-d6, 25 °C) δ 28.0 (q), 28.6 (t), 29.1 (t), 39.9 (d), 50.0 (t), 53.1 (t), 55.4 (q), 59.4 (q), 63.4 (d), 79.1 (s), 100.7 (t), 106.1 (d), 108.1 (d), 110.2 (d), 125.2 (d), 126.0 (d), 127.4 (d), 127.8 (s), 128.0 (d), 128.4 (s), 128.7 (s), 129.1 (s), 138.2 (s), 143.8 (s), 145.7 (s), 146.3 (s), 148.9 (s), 150.1 (s), 152.3 (s); IR (nujol): νmax = 3288, 3129, 1742 cm−1; HRMS–ESI (m/z): [M + H]+: calcd. for C33H38N3O6, 572.2761; found, 572.2814.
Supporting Information
| Supporting Information File 1: Experimental procedures, characterization data, and copies of NMR spectra for compounds 2a–n and 3a–n. | ||
| Format: PDF | Size: 7.6 MB | Download |
| Supporting Information File 2: Antiproliferative evaluation of compounds 2a–n and 3a–g,i–n. | ||
| Format: PDF | Size: 664.0 KB | Download |
References
-
Caliceti, C.; Rizzo, P.; Ferrari, R.; Fortini, F.; Aquila, G.; Leoncini, E.; Zambonin, L.; Rizzo, B.; Calabria, D.; Simoni, P.; Mirasoli, M.; Guardigli, M.; Hrelia, S.; Roda, A.; Cicero, A. F. G. Nutr., Metab. Cardiovasc. Dis. 2017, 27, 552–563. doi:10.1016/j.numecd.2017.04.002
Return to citation in text: [1] -
Tang, J.; Feng, Y.; Tsao, S.; Wang, N.; Curtain, R.; Wang, Y. J. Ethnopharmacol. 2009, 126, 5–17. doi:10.1016/j.jep.2009.08.009
Return to citation in text: [1] -
Tillhon, M.; Guamán Ortiz, L. M.; Lombardi, P.; Scovassi, A. I. Biochem. Pharmacol. 2012, 84, 1260–1267. doi:10.1016/j.bcp.2012.07.018
Return to citation in text: [1] -
Islam, M. M.; Basu, A.; Hossain, M.; Sureshkumar, G.; Hotha, S.; Suresh Kumar, G. DNA Cell Biol. 2011, 30, 123–133. doi:10.1089/dna.2010.1109
Return to citation in text: [1] [2] -
Zhang, S.-L.; Chang, J.-J.; Damu, G. L. V.; Fang, B.; Zhou, X.-D.; Geng, R.-X.; Zhou, C.-H. Bioorg. Med. Chem. Lett. 2013, 23, 1008–1012. doi:10.1016/j.bmcl.2012.12.036
Return to citation in text: [1] -
Basu, A.; Jaisankar, P.; Suresh Kumar, G. Bioorg. Med. Chem. 2012, 20, 2498–2505. doi:10.1016/j.bmc.2012.03.006
Return to citation in text: [1] [2] -
Gornall, K. C.; Samosorn, S.; Tanwirat, B.; Suksamrarn, A.; Bremner, J. B.; Kelso, M. J.; Beck, J. L. Chem. Commun. 2010, 46, 6602–6604. doi:10.1039/c0cc01933j
Return to citation in text: [1] -
Franceschin, M.; Rossetti, L.; D’Ambrosio, A.; Schirripa, S.; Bianco, A.; Ortaggi, G.; Savino, M.; Schultes, C.; Neidle, S. Bioorg. Med. Chem. Lett. 2006, 16, 1707–1711. doi:10.1016/j.bmcl.2005.12.001
Return to citation in text: [1] -
Bhowmik, D.; Hossain, M.; Buzzetti, F.; D’Auria, R.; Lombardi, P.; Kumar, G. S. J. Phys. Chem. B 2012, 116, 2314–2324. doi:10.1021/jp210072a
Return to citation in text: [1] [2] -
Bhowmik, D.; Buzzetti, F.; Fiorillo, G.; Franchini, L.; Syeda, T. M.; Lombardi, P.; Suresh Kumar, G. J. Therm. Anal. Calorim. 2014, 118, 461–473. doi:10.1007/s10973-014-3983-0
Return to citation in text: [1] -
Bhowmik, D.; Buzzetti, F.; Fiorillo, G.; Lombardi, P.; Suresh Kumar, G. Spectrochim. Acta, Part A 2014, 120, 257–264. doi:10.1016/j.saa.2013.09.081
Return to citation in text: [1] -
Bhowmik, D.; Buzzetti, F.; Fiorillo, G.; Orzi, F.; Syeda, T. M.; Lombardi, P.; Suresh Kumar, G. Med. Chem. Commun. 2014, 5, 226–231. doi:10.1039/c3md00254c
Return to citation in text: [1] -
Iwasa, K.; Nanba, H.; Lee, D.-U.; Kang, S.-l. Planta Med. 1998, 64, 748–751. doi:10.1055/s-2006-957572
Return to citation in text: [1] -
Zhang, W.-J.; Ou, T.-M.; Lu, Y.-J.; Huang, Y.-Y.; Wu, W.-B.; Huang, Z.-S.; Zhou, J.-L.; Wong, K.-Y.; Gu, L.-Q. Bioorg. Med. Chem. 2007, 15, 5493–5501. doi:10.1016/j.bmc.2007.05.050
Return to citation in text: [1] -
Ma, Y.; Ou, T.-M.; Hou, J.-Q.; Lu, Y.-J.; Tan, J.-H.; Gu, L.-Q.; Huang, Z.-S. Bioorg. Med. Chem. 2008, 16, 7582–7591. doi:10.1016/j.bmc.2008.07.029
Return to citation in text: [1] -
Ma, Y.; Ou, T.-M.; Tan, J.-H.; Hou, J.-Q.; Huang, S.-L.; Gu, L.-Q.; Huang, Z.-S. Bioorg. Med. Chem. Lett. 2009, 19, 3414–3417. doi:10.1016/j.bmcl.2009.05.030
Return to citation in text: [1] -
Ma, Y.; Ou, T.-M.; Tan, J.-H.; Hou, J.-Q.; Huang, S.-L.; Gu, L.-Q.; Huang, Z.-S. Eur. J. Med. Chem. 2011, 46, 1906–1913. doi:10.1016/j.ejmech.2011.02.020
Return to citation in text: [1] -
Tera, M.; Hirokawa, T.; Okabe, S.; Sugahara, K.; Seimiya, H.; Shimamoto, K. Chem. – Eur. J. 2015, 21, 14519–14528. doi:10.1002/chem.201501693
Return to citation in text: [1] -
Iwasa, K.; Moriyasu, M.; Yamori, T.; Turuo, T.; Lee, D.-U.; Wiegrebe, W. J. Nat. Prod. 2001, 64, 896–898. doi:10.1021/np000554f
Return to citation in text: [1] -
Waters, M. L. Curr. Opin. Chem. Biol. 2002, 6, 736–741. doi:10.1016/s1367-5931(02)00359-9
Return to citation in text: [1] -
Fan, T.-Y.; Wang, Y.-X.; Tang, S.; Hu, X.-X.; Zen, Q.-X.; Pang, J.; Yang, Y.-S.; You, X.-F.; Song, D.-Q. Eur. J. Med. Chem. 2018, 157, 877–886. doi:10.1016/j.ejmech.2018.08.050
Return to citation in text: [1] -
Li, Y.-B.; Zhao, W.-L.; Wang, Y.-X.; Zhang, C.-X.; Jiang, J.-D.; Bi, C.-W.; Tang, S.; Chen, R.-X.; Shao, R.-G.; Song, D.-Q. Eur. J. Med. Chem. 2013, 68, 463–472. doi:10.1016/j.ejmech.2013.07.026
Return to citation in text: [1] -
Zhang, L.; Li, J.; Ma, F.; Yao, S.; Li, N.; Wang, J.; Wang, Y.; Wang, X.; Yao, Q. Molecules 2012, 17, 11294–11302. doi:10.3390/molecules171011294
Return to citation in text: [1] -
Li, Y.-H.; Yang, P.; Kong, W.-J.; Wang, Y.-X.; Hu, C.-Q.; Zuo, Z.-Y.; Wang, Y.-M.; Gao, H.; Gao, L.-M.; Feng, Y.-C.; Du, N.-N.; Liu, Y.; Song, D.-Q.; Jiang, J.-D. J. Med. Chem. 2009, 52, 492–501. doi:10.1021/jm801157z
Return to citation in text: [1] -
Buchanan, B.; Meng, Q.; Poulin, M.-M.; Zuccolo, J.; Azike, C. G.; Gabriele, J.; Baranowski, D. C. PLoS One 2018, 13, e0194979. doi:10.1371/journal.pone.0194979
Return to citation in text: [1] -
Feng, R.; Shou, J.-W.; Zhao, Z.-X.; He, C.-Y.; Ma, C.; Huang, M.; Fu, J.; Tan, X.-S.; Li, X.-Y.; Wen, B.-Y.; Chen, X.; Yang, X.-Y.; Ren, G.; Lin, Y.; Chen, Y.; You, X.-F.; Wang, Y.; Jiang, J.-D. Sci. Rep. 2015, 5, 12155. doi:10.1038/srep12155
Return to citation in text: [1] -
Alolga, R. N.; Fan, Y.; Chen, Z.; Liu, L.-W.; Zhao, Y.-J.; Li, J.; Chen, Y.; Lai, M.-D.; Li, P.; Qi, L.-W. Sci. Rep. 2016, 6, 27671. doi:10.1038/srep27671
Return to citation in text: [1] -
Feng, R.; Zhao, Z.-X.; Ma, S.-R.; Guo, F.; Wang, Y.; Jiang, J.-D. Front. Pharmacol. 2018, 9, 214. doi:10.3389/fphar.2018.00214
Return to citation in text: [1] -
Turner, N.; Li, J.-Y.; Gosby, A.; To, S. W. C.; Cheng, Z.; Miyoshi, H.; Taketo, M. M.; Cooney, G. J.; Kraegen, E. W.; James, D. E.; Hu, L.-H.; Li, J.; Ye, J.-M. Diabetes 2008, 57, 1414–1418. doi:10.2337/db07-1552
Return to citation in text: [1] -
Cheng, Z.; Chen, A.-F.; Wu, F.; Sheng, L.; Zhang, H.-K.; Gu, M.; Li, Y.-Y.; Zhang, L.-N.; Hu, L.-H.; Li, J.-Y.; Li, J. Bioorg. Med. Chem. 2010, 18, 5915–5924. doi:10.1016/j.bmc.2010.06.085
Return to citation in text: [1] -
Chen, J.; Cao, J.; Fang, L.; Liu, B.; Zhou, Q.; Sun, Y.; Wang, Y.; Li, Y.; Meng, S. J. Transl. Med. 2014, 12, 326. doi:10.1186/s12967-014-0326-7
Return to citation in text: [1] -
Herrendorff, R.; Faleschini, M. T.; Stiefvater, A.; Erne, B.; Wiktorowicz, T.; Kern, F.; Hamburger, M.; Potterat, O.; Kinter, J.; Sinnreich, M. J. Biol. Chem. 2016, 291, 17165–17177. doi:10.1074/jbc.m115.710616
Return to citation in text: [1] -
Yu, D.; Lv, L.; Fang, L.; Zhang, B.; Wang, J.; Zhan, G.; Zhao, L.; Zhao, X.; Li, B. PLoS One 2017, 12, e0181823. doi:10.1371/journal.pone.0181823
Return to citation in text: [1] -
Mohammad, M.; Al-masri, I. M.; Issa, A.; Khdair, A.; Bustanji, Y. Med. Chem. Res. 2013, 22, 2273–2278. doi:10.1007/s00044-012-0221-9
Return to citation in text: [1] -
Pietra, D.; Borghini, A.; Bianucci, A. M. Pharmacol. Rep. 2015, 67, 1081–1089. doi:10.1016/j.pharep.2015.04.001
Return to citation in text: [1] -
Zuo, G.-Y.; Li, Y.; Wang, G.-C.; Li, Z.-S.; Han, J. Med. Chem. Res. 2014, 23, 2439–2444. doi:10.1007/s00044-013-0844-5
Return to citation in text: [1] -
Dai, B.; Ma, Y.; Wang, W.; Zhan, Y.; Zhang, D.; Liu, R.; Zhang, Y. J. Cell. Mol. Med. 2017, 21, 2573–2585. doi:10.1111/jcmm.13178
Return to citation in text: [1] -
Kim, I. J.; Park, Y. J.; Kim, J. i.; Lee, K. T.; Kim, S. K. Arch. Pharmacal Res. 1997, 20, 476–479. doi:10.1007/bf02973943
Return to citation in text: [1] -
Catalani, S.; Carbonaro, V.; Palma, F.; Arshakyan, M.; Galati, R.; Nuvoli, B.; Battistelli, S.; Canestrari, F.; Benedetti, S. J. Exp. Clin. Cancer Res. 2013, 32, 63. doi:10.1186/1756-9966-32-63
Return to citation in text: [1] -
Catalani, S.; Palma, F.; Battistelli, S.; Benedetti, S. PLoS One 2017, 12, e0172138. doi:10.1371/journal.pone.0172138
Return to citation in text: [1] -
Catalani, S.; Palma, F.; Battistelli, S.; Nuvoli, B.; Galati, R.; Benedetti, S. Toxicol. In Vitro 2017, 41, 49–55. doi:10.1016/j.tiv.2017.02.008
Return to citation in text: [1] -
Benedetti, S.; Catalani, S.; Palma, F.; Canonico, B.; Luchetti, F.; Galati, R.; Papa, S.; Battistelli, S. Life Sci. 2018, 215, 80–85. doi:10.1016/j.lfs.2018.11.002
Return to citation in text: [1] -
Popiołek, Ł. Med. Chem. Res. 2017, 26, 287–301. doi:10.1007/s00044-016-1756-y
Return to citation in text: [1] -
Kareem, H. S.; Ariffin, A.; Nordin, N.; Heidelberg, T.; Abdul-Aziz, A.; Kong, K. W.; Yehye, W. A. Eur. J. Med. Chem. 2015, 103, 497–505. doi:10.1016/j.ejmech.2015.09.016
Return to citation in text: [1] -
Asif, M.; Husain, A. J. Appl. Chem. 2013, No. 247203. doi:10.1155/2013/247203
Return to citation in text: [1] [2] [3] -
Sridhar, S. K.; Pandeya, S. N.; Stables, J. P.; Ramesh, A. Eur. J. Pharm. Sci. 2002, 16, 129–132. doi:10.1016/s0928-0987(02)00077-5
Return to citation in text: [1] -
Cardona-G, W.; Yepes, A. F.; Herrera-R, A. Curr. Med. Chem. 2018, 25, 3637–3679. doi:10.2174/0929867325666180309111428
Return to citation in text: [1] -
Ramya Rajan, M. P.; Rathikha, R.; Nithyabalaji, R.; Sribalan, R. J. Mol. Struct. 2020, 1216, 128297. doi:10.1016/j.molstruc.2020.128297
Return to citation in text: [1] -
Doğan, H.; Doğan, Ş. D.; Gündüz, M. G.; Krishna, V. S.; Lherbet, C.; Sriram, D.; Şahin, O.; Sarıpınar, E. Eur. J. Med. Chem. 2020, 188, 112035. doi:10.1016/j.ejmech.2020.112035
Return to citation in text: [1] -
Vicini, P.; Incerti, M.; Doytchinova, I. A.; La Colla, P.; Busonera, B.; Loddo, R. Eur. J. Med. Chem. 2006, 41, 624–632. doi:10.1016/j.ejmech.2006.01.010
Return to citation in text: [1] -
Terzioglu, N.; Gürsoy, A. Eur. J. Med. Chem. 2003, 38, 781–786. doi:10.1016/s0223-5234(03)00138-7
Return to citation in text: [1] -
Abadi, A. H.; Eissa, A. A. H.; Hassan, G. S. Chem. Pharm. Bull. 2003, 51, 838–844. doi:10.1248/cpb.51.838
Return to citation in text: [1] -
Kumar, D.; Maruthi Kumar, N.; Ghosh, S.; Shah, K. Bioorg. Med. Chem. Lett. 2012, 22, 212–215. doi:10.1016/j.bmcl.2011.11.031
Return to citation in text: [1] -
Paterna, A.; Borralho, P. M.; Gomes, S. E.; Mulhovo, S.; Rodrigues, C. M. P.; Ferreira, M.-J. U. Bioorg. Med. Chem. Lett. 2015, 25, 3556–3559. doi:10.1016/j.bmcl.2015.06.084
Return to citation in text: [1] -
Zhou, Q.; Fu, Y.-H.; Li, X.-b.; Chen, G.-Y.; Wu, S.-Y.; Song, X.-P.; Liu, Y.-P.; Han, C.-R. Phytochem. Lett. 2015, 11, 296–300. doi:10.1016/j.phytol.2015.01.017
Return to citation in text: [1] -
Wang, L.; Zhang, S.-Y.; Chen, L.; Huang, X.-J.; Zhang, Q.-W.; Jiang, R.-W.; Yao, F.; Ye, W.-C. Phytochem. Lett. 2014, 7, 89–92. doi:10.1016/j.phytol.2013.10.007
Return to citation in text: [1] -
Xuan, B.; Wang, W.; Li, D. X. Acta Pharmacol. Sin. 1994, 15, 133–135.
Return to citation in text: [1] -
Wang, X. L.; Jin, G. Z.; Zhou, T. C. Acta Physiol. Sin. 1994, 46, 505–508.
Return to citation in text: [1] -
He, K.; Gao, J. L.; Zhao, G. S. Chin. Tradit. Herb. Drugs 2007, 38, 1909–1912.
Return to citation in text: [1] -
Xu, Q. X.; Wang, Y. L.; Xie, M. Prog. Pharm. Sci. 1998, 22, 46–49.
Return to citation in text: [1] -
Wang, R. F.; Xu, G. Y.; Hua, W. Y.; Peng, S. X. Chin. J. Med. Chem. 1996, 4, 243–248.
Return to citation in text: [1] -
Correché, E. R.; Andujar, S. A.; Kurdelas, R. R.; Lechón, M. J. G.; Freile, M. L.; Enriz, R. D. Bioorg. Med. Chem. 2008, 16, 3641–3651. doi:10.1016/j.bmc.2008.02.015
Return to citation in text: [1] -
Dai, D.-Z.; Yu, F.; Li, H.-T.; Tang, Y.-q.; An, L.-F.; Huang, W.-L.; Peng, S.-X.; Hao, X.-M.; Zhou, B.-A.; Hu, C.-H. Drug Dev. Res. 1996, 39, 138–146. doi:10.1002/(sici)1098-2299(199610)39:2<138::aid-ddr5>3.0.co;2-q
Return to citation in text: [1] -
Pingali, S.; Donahue, J. P.; Payton-Stewart, F. Acta Crystallogr., Sect. C: Struct. Chem. 2015, 71, 262–265. doi:10.1107/s2053229615004076
Return to citation in text: [1] -
Mari, G.; Catalani, S.; Antonini, E.; De Crescentini, L.; Mantellini, F.; Santeusanio, S.; Lombardi, P.; Amicucci, A.; Battistelli, S.; Benedetti, S.; Palma, F. Bioorg. Med. Chem. 2018, 26, 5037–5044. doi:10.1016/j.bmc.2018.08.038
Return to citation in text: [1] [2] [3] -
Ferraroni, M.; Bazzicalupi, C.; Papi, F.; Fiorillo, G.; Guamán-Ortiz, L. M.; Nocentini, A.; Scovassi, A. I.; Lombardi, P.; Gratteri, P. Chem. – Asian J. 2016, 11, 1107–1115. doi:10.1002/asia.201600116
Return to citation in text: [1] [2] [3] -
Fu, S.; Xie, Y.; Tuo, J.; Wang, Y.; Zhu, W.; Wu, S.; Yan, G.; Hu, H. Med. Chem. Commun. 2015, 6, 164–173. doi:10.1039/c4md00264d
Return to citation in text: [1] [2] [3] -
Mari, G.; De Crescentini, L.; Favi, G.; Lombardi, P.; Fiorillo, G.; Giorgi, G.; Mantellini, F. Asian J. Org. Chem. 2017, 6, 720–727. doi:10.1002/ajoc.201700051
Return to citation in text: [1] -
Iwasa, K.; Gupta, Y. P.; Cushman, M. J. Org. Chem. 1981, 46, 4744–4750. doi:10.1021/jo00336a024
Return to citation in text: [1] -
Hydrazone-THBER 3h was omitted due to its insolubility in the conditions used for the cytotoxicity assays.
Return to citation in text: [1] [2] [3] -
Ciccolini, C.; Mari, G.; Favi, G.; Mantellini, F.; De Crescentini, L.; Santeusanio, S. Molecules 2019, 24, 3785. doi:10.3390/molecules24203785
Return to citation in text: [1] [2]
| 49. | Doğan, H.; Doğan, Ş. D.; Gündüz, M. G.; Krishna, V. S.; Lherbet, C.; Sriram, D.; Şahin, O.; Sarıpınar, E. Eur. J. Med. Chem. 2020, 188, 112035. doi:10.1016/j.ejmech.2020.112035 |
| 50. | Vicini, P.; Incerti, M.; Doytchinova, I. A.; La Colla, P.; Busonera, B.; Loddo, R. Eur. J. Med. Chem. 2006, 41, 624–632. doi:10.1016/j.ejmech.2006.01.010 |
| 51. | Terzioglu, N.; Gürsoy, A. Eur. J. Med. Chem. 2003, 38, 781–786. doi:10.1016/s0223-5234(03)00138-7 |
| 52. | Abadi, A. H.; Eissa, A. A. H.; Hassan, G. S. Chem. Pharm. Bull. 2003, 51, 838–844. doi:10.1248/cpb.51.838 |
| 53. | Kumar, D.; Maruthi Kumar, N.; Ghosh, S.; Shah, K. Bioorg. Med. Chem. Lett. 2012, 22, 212–215. doi:10.1016/j.bmcl.2011.11.031 |
| 54. | Paterna, A.; Borralho, P. M.; Gomes, S. E.; Mulhovo, S.; Rodrigues, C. M. P.; Ferreira, M.-J. U. Bioorg. Med. Chem. Lett. 2015, 25, 3556–3559. doi:10.1016/j.bmcl.2015.06.084 |
| 1. | Caliceti, C.; Rizzo, P.; Ferrari, R.; Fortini, F.; Aquila, G.; Leoncini, E.; Zambonin, L.; Rizzo, B.; Calabria, D.; Simoni, P.; Mirasoli, M.; Guardigli, M.; Hrelia, S.; Roda, A.; Cicero, A. F. G. Nutr., Metab. Cardiovasc. Dis. 2017, 27, 552–563. doi:10.1016/j.numecd.2017.04.002 |
| 13. | Iwasa, K.; Nanba, H.; Lee, D.-U.; Kang, S.-l. Planta Med. 1998, 64, 748–751. doi:10.1055/s-2006-957572 |
| 14. | Zhang, W.-J.; Ou, T.-M.; Lu, Y.-J.; Huang, Y.-Y.; Wu, W.-B.; Huang, Z.-S.; Zhou, J.-L.; Wong, K.-Y.; Gu, L.-Q. Bioorg. Med. Chem. 2007, 15, 5493–5501. doi:10.1016/j.bmc.2007.05.050 |
| 15. | Ma, Y.; Ou, T.-M.; Hou, J.-Q.; Lu, Y.-J.; Tan, J.-H.; Gu, L.-Q.; Huang, Z.-S. Bioorg. Med. Chem. 2008, 16, 7582–7591. doi:10.1016/j.bmc.2008.07.029 |
| 16. | Ma, Y.; Ou, T.-M.; Tan, J.-H.; Hou, J.-Q.; Huang, S.-L.; Gu, L.-Q.; Huang, Z.-S. Bioorg. Med. Chem. Lett. 2009, 19, 3414–3417. doi:10.1016/j.bmcl.2009.05.030 |
| 17. | Ma, Y.; Ou, T.-M.; Tan, J.-H.; Hou, J.-Q.; Huang, S.-L.; Gu, L.-Q.; Huang, Z.-S. Eur. J. Med. Chem. 2011, 46, 1906–1913. doi:10.1016/j.ejmech.2011.02.020 |
| 18. | Tera, M.; Hirokawa, T.; Okabe, S.; Sugahara, K.; Seimiya, H.; Shimamoto, K. Chem. – Eur. J. 2015, 21, 14519–14528. doi:10.1002/chem.201501693 |
| 19. | Iwasa, K.; Moriyasu, M.; Yamori, T.; Turuo, T.; Lee, D.-U.; Wiegrebe, W. J. Nat. Prod. 2001, 64, 896–898. doi:10.1021/np000554f |
| 34. | Mohammad, M.; Al-masri, I. M.; Issa, A.; Khdair, A.; Bustanji, Y. Med. Chem. Res. 2013, 22, 2273–2278. doi:10.1007/s00044-012-0221-9 |
| 66. | Ferraroni, M.; Bazzicalupi, C.; Papi, F.; Fiorillo, G.; Guamán-Ortiz, L. M.; Nocentini, A.; Scovassi, A. I.; Lombardi, P.; Gratteri, P. Chem. – Asian J. 2016, 11, 1107–1115. doi:10.1002/asia.201600116 |
| 67. | Fu, S.; Xie, Y.; Tuo, J.; Wang, Y.; Zhu, W.; Wu, S.; Yan, G.; Hu, H. Med. Chem. Commun. 2015, 6, 164–173. doi:10.1039/c4md00264d |
| 9. | Bhowmik, D.; Hossain, M.; Buzzetti, F.; D’Auria, R.; Lombardi, P.; Kumar, G. S. J. Phys. Chem. B 2012, 116, 2314–2324. doi:10.1021/jp210072a |
| 10. | Bhowmik, D.; Buzzetti, F.; Fiorillo, G.; Franchini, L.; Syeda, T. M.; Lombardi, P.; Suresh Kumar, G. J. Therm. Anal. Calorim. 2014, 118, 461–473. doi:10.1007/s10973-014-3983-0 |
| 11. | Bhowmik, D.; Buzzetti, F.; Fiorillo, G.; Lombardi, P.; Suresh Kumar, G. Spectrochim. Acta, Part A 2014, 120, 257–264. doi:10.1016/j.saa.2013.09.081 |
| 12. | Bhowmik, D.; Buzzetti, F.; Fiorillo, G.; Orzi, F.; Syeda, T. M.; Lombardi, P.; Suresh Kumar, G. Med. Chem. Commun. 2014, 5, 226–231. doi:10.1039/c3md00254c |
| 35. | Pietra, D.; Borghini, A.; Bianucci, A. M. Pharmacol. Rep. 2015, 67, 1081–1089. doi:10.1016/j.pharep.2015.04.001 |
| 65. | Mari, G.; Catalani, S.; Antonini, E.; De Crescentini, L.; Mantellini, F.; Santeusanio, S.; Lombardi, P.; Amicucci, A.; Battistelli, S.; Benedetti, S.; Palma, F. Bioorg. Med. Chem. 2018, 26, 5037–5044. doi:10.1016/j.bmc.2018.08.038 |
| 5. | Zhang, S.-L.; Chang, J.-J.; Damu, G. L. V.; Fang, B.; Zhou, X.-D.; Geng, R.-X.; Zhou, C.-H. Bioorg. Med. Chem. Lett. 2013, 23, 1008–1012. doi:10.1016/j.bmcl.2012.12.036 |
| 6. | Basu, A.; Jaisankar, P.; Suresh Kumar, G. Bioorg. Med. Chem. 2012, 20, 2498–2505. doi:10.1016/j.bmc.2012.03.006 |
| 7. | Gornall, K. C.; Samosorn, S.; Tanwirat, B.; Suksamrarn, A.; Bremner, J. B.; Kelso, M. J.; Beck, J. L. Chem. Commun. 2010, 46, 6602–6604. doi:10.1039/c0cc01933j |
| 8. | Franceschin, M.; Rossetti, L.; D’Ambrosio, A.; Schirripa, S.; Bianco, A.; Ortaggi, G.; Savino, M.; Schultes, C.; Neidle, S. Bioorg. Med. Chem. Lett. 2006, 16, 1707–1711. doi:10.1016/j.bmcl.2005.12.001 |
| 32. | Herrendorff, R.; Faleschini, M. T.; Stiefvater, A.; Erne, B.; Wiktorowicz, T.; Kern, F.; Hamburger, M.; Potterat, O.; Kinter, J.; Sinnreich, M. J. Biol. Chem. 2016, 291, 17165–17177. doi:10.1074/jbc.m115.710616 |
| 61. | Wang, R. F.; Xu, G. Y.; Hua, W. Y.; Peng, S. X. Chin. J. Med. Chem. 1996, 4, 243–248. |
| 2. | Tang, J.; Feng, Y.; Tsao, S.; Wang, N.; Curtain, R.; Wang, Y. J. Ethnopharmacol. 2009, 126, 5–17. doi:10.1016/j.jep.2009.08.009 |
| 3. | Tillhon, M.; Guamán Ortiz, L. M.; Lombardi, P.; Scovassi, A. I. Biochem. Pharmacol. 2012, 84, 1260–1267. doi:10.1016/j.bcp.2012.07.018 |
| 4. | Islam, M. M.; Basu, A.; Hossain, M.; Sureshkumar, G.; Hotha, S.; Suresh Kumar, G. DNA Cell Biol. 2011, 30, 123–133. doi:10.1089/dna.2010.1109 |
| 33. | Yu, D.; Lv, L.; Fang, L.; Zhang, B.; Wang, J.; Zhan, G.; Zhao, L.; Zhao, X.; Li, B. PLoS One 2017, 12, e0181823. doi:10.1371/journal.pone.0181823 |
| 62. | Correché, E. R.; Andujar, S. A.; Kurdelas, R. R.; Lechón, M. J. G.; Freile, M. L.; Enriz, R. D. Bioorg. Med. Chem. 2008, 16, 3641–3651. doi:10.1016/j.bmc.2008.02.015 |
| 63. | Dai, D.-Z.; Yu, F.; Li, H.-T.; Tang, Y.-q.; An, L.-F.; Huang, W.-L.; Peng, S.-X.; Hao, X.-M.; Zhou, B.-A.; Hu, C.-H. Drug Dev. Res. 1996, 39, 138–146. doi:10.1002/(sici)1098-2299(199610)39:2<138::aid-ddr5>3.0.co;2-q |
| 64. | Pingali, S.; Donahue, J. P.; Payton-Stewart, F. Acta Crystallogr., Sect. C: Struct. Chem. 2015, 71, 262–265. doi:10.1107/s2053229615004076 |
| 65. | Mari, G.; Catalani, S.; Antonini, E.; De Crescentini, L.; Mantellini, F.; Santeusanio, S.; Lombardi, P.; Amicucci, A.; Battistelli, S.; Benedetti, S.; Palma, F. Bioorg. Med. Chem. 2018, 26, 5037–5044. doi:10.1016/j.bmc.2018.08.038 |
| 25. | Buchanan, B.; Meng, Q.; Poulin, M.-M.; Zuccolo, J.; Azike, C. G.; Gabriele, J.; Baranowski, D. C. PLoS One 2018, 13, e0194979. doi:10.1371/journal.pone.0194979 |
| 29. | Turner, N.; Li, J.-Y.; Gosby, A.; To, S. W. C.; Cheng, Z.; Miyoshi, H.; Taketo, M. M.; Cooney, G. J.; Kraegen, E. W.; James, D. E.; Hu, L.-H.; Li, J.; Ye, J.-M. Diabetes 2008, 57, 1414–1418. doi:10.2337/db07-1552 |
| 30. | Cheng, Z.; Chen, A.-F.; Wu, F.; Sheng, L.; Zhang, H.-K.; Gu, M.; Li, Y.-Y.; Zhang, L.-N.; Hu, L.-H.; Li, J.-Y.; Li, J. Bioorg. Med. Chem. 2010, 18, 5915–5924. doi:10.1016/j.bmc.2010.06.085 |
| 59. | He, K.; Gao, J. L.; Zhao, G. S. Chin. Tradit. Herb. Drugs 2007, 38, 1909–1912. |
| 21. | Fan, T.-Y.; Wang, Y.-X.; Tang, S.; Hu, X.-X.; Zen, Q.-X.; Pang, J.; Yang, Y.-S.; You, X.-F.; Song, D.-Q. Eur. J. Med. Chem. 2018, 157, 877–886. doi:10.1016/j.ejmech.2018.08.050 |
| 22. | Li, Y.-B.; Zhao, W.-L.; Wang, Y.-X.; Zhang, C.-X.; Jiang, J.-D.; Bi, C.-W.; Tang, S.; Chen, R.-X.; Shao, R.-G.; Song, D.-Q. Eur. J. Med. Chem. 2013, 68, 463–472. doi:10.1016/j.ejmech.2013.07.026 |
| 23. | Zhang, L.; Li, J.; Ma, F.; Yao, S.; Li, N.; Wang, J.; Wang, Y.; Wang, X.; Yao, Q. Molecules 2012, 17, 11294–11302. doi:10.3390/molecules171011294 |
| 24. | Li, Y.-H.; Yang, P.; Kong, W.-J.; Wang, Y.-X.; Hu, C.-Q.; Zuo, Z.-Y.; Wang, Y.-M.; Gao, H.; Gao, L.-M.; Feng, Y.-C.; Du, N.-N.; Liu, Y.; Song, D.-Q.; Jiang, J.-D. J. Med. Chem. 2009, 52, 492–501. doi:10.1021/jm801157z |
| 31. | Chen, J.; Cao, J.; Fang, L.; Liu, B.; Zhou, Q.; Sun, Y.; Wang, Y.; Li, Y.; Meng, S. J. Transl. Med. 2014, 12, 326. doi:10.1186/s12967-014-0326-7 |
| 20. | Waters, M. L. Curr. Opin. Chem. Biol. 2002, 6, 736–741. doi:10.1016/s1367-5931(02)00359-9 |
| 55. | Zhou, Q.; Fu, Y.-H.; Li, X.-b.; Chen, G.-Y.; Wu, S.-Y.; Song, X.-P.; Liu, Y.-P.; Han, C.-R. Phytochem. Lett. 2015, 11, 296–300. doi:10.1016/j.phytol.2015.01.017 |
| 56. | Wang, L.; Zhang, S.-Y.; Chen, L.; Huang, X.-J.; Zhang, Q.-W.; Jiang, R.-W.; Yao, F.; Ye, W.-C. Phytochem. Lett. 2014, 7, 89–92. doi:10.1016/j.phytol.2013.10.007 |
| 57. | Xuan, B.; Wang, W.; Li, D. X. Acta Pharmacol. Sin. 1994, 15, 133–135. |
| 4. | Islam, M. M.; Basu, A.; Hossain, M.; Sureshkumar, G.; Hotha, S.; Suresh Kumar, G. DNA Cell Biol. 2011, 30, 123–133. doi:10.1089/dna.2010.1109 |
| 6. | Basu, A.; Jaisankar, P.; Suresh Kumar, G. Bioorg. Med. Chem. 2012, 20, 2498–2505. doi:10.1016/j.bmc.2012.03.006 |
| 9. | Bhowmik, D.; Hossain, M.; Buzzetti, F.; D’Auria, R.; Lombardi, P.; Kumar, G. S. J. Phys. Chem. B 2012, 116, 2314–2324. doi:10.1021/jp210072a |
| 26. | Feng, R.; Shou, J.-W.; Zhao, Z.-X.; He, C.-Y.; Ma, C.; Huang, M.; Fu, J.; Tan, X.-S.; Li, X.-Y.; Wen, B.-Y.; Chen, X.; Yang, X.-Y.; Ren, G.; Lin, Y.; Chen, Y.; You, X.-F.; Wang, Y.; Jiang, J.-D. Sci. Rep. 2015, 5, 12155. doi:10.1038/srep12155 |
| 27. | Alolga, R. N.; Fan, Y.; Chen, Z.; Liu, L.-W.; Zhao, Y.-J.; Li, J.; Chen, Y.; Lai, M.-D.; Li, P.; Qi, L.-W. Sci. Rep. 2016, 6, 27671. doi:10.1038/srep27671 |
| 28. | Feng, R.; Zhao, Z.-X.; Ma, S.-R.; Guo, F.; Wang, Y.; Jiang, J.-D. Front. Pharmacol. 2018, 9, 214. doi:10.3389/fphar.2018.00214 |
| 39. | Catalani, S.; Carbonaro, V.; Palma, F.; Arshakyan, M.; Galati, R.; Nuvoli, B.; Battistelli, S.; Canestrari, F.; Benedetti, S. J. Exp. Clin. Cancer Res. 2013, 32, 63. doi:10.1186/1756-9966-32-63 |
| 40. | Catalani, S.; Palma, F.; Battistelli, S.; Benedetti, S. PLoS One 2017, 12, e0172138. doi:10.1371/journal.pone.0172138 |
| 41. | Catalani, S.; Palma, F.; Battistelli, S.; Nuvoli, B.; Galati, R.; Benedetti, S. Toxicol. In Vitro 2017, 41, 49–55. doi:10.1016/j.tiv.2017.02.008 |
| 42. | Benedetti, S.; Catalani, S.; Palma, F.; Canonico, B.; Luchetti, F.; Galati, R.; Papa, S.; Battistelli, S. Life Sci. 2018, 215, 80–85. doi:10.1016/j.lfs.2018.11.002 |
| 36. | Zuo, G.-Y.; Li, Y.; Wang, G.-C.; Li, Z.-S.; Han, J. Med. Chem. Res. 2014, 23, 2439–2444. doi:10.1007/s00044-013-0844-5 |
| 68. | Mari, G.; De Crescentini, L.; Favi, G.; Lombardi, P.; Fiorillo, G.; Giorgi, G.; Mantellini, F. Asian J. Org. Chem. 2017, 6, 720–727. doi:10.1002/ajoc.201700051 |
| 37. | Dai, B.; Ma, Y.; Wang, W.; Zhan, Y.; Zhang, D.; Liu, R.; Zhang, Y. J. Cell. Mol. Med. 2017, 21, 2573–2585. doi:10.1111/jcmm.13178 |
| 38. | Kim, I. J.; Park, Y. J.; Kim, J. i.; Lee, K. T.; Kim, S. K. Arch. Pharmacal Res. 1997, 20, 476–479. doi:10.1007/bf02973943 |
| 65. | Mari, G.; Catalani, S.; Antonini, E.; De Crescentini, L.; Mantellini, F.; Santeusanio, S.; Lombardi, P.; Amicucci, A.; Battistelli, S.; Benedetti, S.; Palma, F. Bioorg. Med. Chem. 2018, 26, 5037–5044. doi:10.1016/j.bmc.2018.08.038 |
| 69. | Iwasa, K.; Gupta, Y. P.; Cushman, M. J. Org. Chem. 1981, 46, 4744–4750. doi:10.1021/jo00336a024 |
| 47. | Cardona-G, W.; Yepes, A. F.; Herrera-R, A. Curr. Med. Chem. 2018, 25, 3637–3679. doi:10.2174/0929867325666180309111428 |
| 66. | Ferraroni, M.; Bazzicalupi, C.; Papi, F.; Fiorillo, G.; Guamán-Ortiz, L. M.; Nocentini, A.; Scovassi, A. I.; Lombardi, P.; Gratteri, P. Chem. – Asian J. 2016, 11, 1107–1115. doi:10.1002/asia.201600116 |
| 67. | Fu, S.; Xie, Y.; Tuo, J.; Wang, Y.; Zhu, W.; Wu, S.; Yan, G.; Hu, H. Med. Chem. Commun. 2015, 6, 164–173. doi:10.1039/c4md00264d |
| 48. | Ramya Rajan, M. P.; Rathikha, R.; Nithyabalaji, R.; Sribalan, R. J. Mol. Struct. 2020, 1216, 128297. doi:10.1016/j.molstruc.2020.128297 |
| 45. | Asif, M.; Husain, A. J. Appl. Chem. 2013, No. 247203. doi:10.1155/2013/247203 |
| 66. | Ferraroni, M.; Bazzicalupi, C.; Papi, F.; Fiorillo, G.; Guamán-Ortiz, L. M.; Nocentini, A.; Scovassi, A. I.; Lombardi, P.; Gratteri, P. Chem. – Asian J. 2016, 11, 1107–1115. doi:10.1002/asia.201600116 |
| 67. | Fu, S.; Xie, Y.; Tuo, J.; Wang, Y.; Zhu, W.; Wu, S.; Yan, G.; Hu, H. Med. Chem. Commun. 2015, 6, 164–173. doi:10.1039/c4md00264d |
| 46. | Sridhar, S. K.; Pandeya, S. N.; Stables, J. P.; Ramesh, A. Eur. J. Pharm. Sci. 2002, 16, 129–132. doi:10.1016/s0928-0987(02)00077-5 |
| 71. | Ciccolini, C.; Mari, G.; Favi, G.; Mantellini, F.; De Crescentini, L.; Santeusanio, S. Molecules 2019, 24, 3785. doi:10.3390/molecules24203785 |
| 45. | Asif, M.; Husain, A. J. Appl. Chem. 2013, No. 247203. doi:10.1155/2013/247203 |
| 70. | Hydrazone-THBER 3h was omitted due to its insolubility in the conditions used for the cytotoxicity assays. |
| 45. | Asif, M.; Husain, A. J. Appl. Chem. 2013, No. 247203. doi:10.1155/2013/247203 |
| 71. | Ciccolini, C.; Mari, G.; Favi, G.; Mantellini, F.; De Crescentini, L.; Santeusanio, S. Molecules 2019, 24, 3785. doi:10.3390/molecules24203785 |
| 43. | Popiołek, Ł. Med. Chem. Res. 2017, 26, 287–301. doi:10.1007/s00044-016-1756-y |
| 70. | Hydrazone-THBER 3h was omitted due to its insolubility in the conditions used for the cytotoxicity assays. |
| 44. | Kareem, H. S.; Ariffin, A.; Nordin, N.; Heidelberg, T.; Abdul-Aziz, A.; Kong, K. W.; Yehye, W. A. Eur. J. Med. Chem. 2015, 103, 497–505. doi:10.1016/j.ejmech.2015.09.016 |
| 70. | Hydrazone-THBER 3h was omitted due to its insolubility in the conditions used for the cytotoxicity assays. |
© 2020 Mari et al.; licensee Beilstein-Institut.
This is an Open Access article under the terms of the Creative Commons Attribution License (http://creativecommons.org/licenses/by/4.0). Please note that the reuse, redistribution and reproduction in particular requires that the authors and source are credited.
The license is subject to the Beilstein Journal of Organic Chemistry terms and conditions: (https://www.beilstein-journals.org/bjoc)