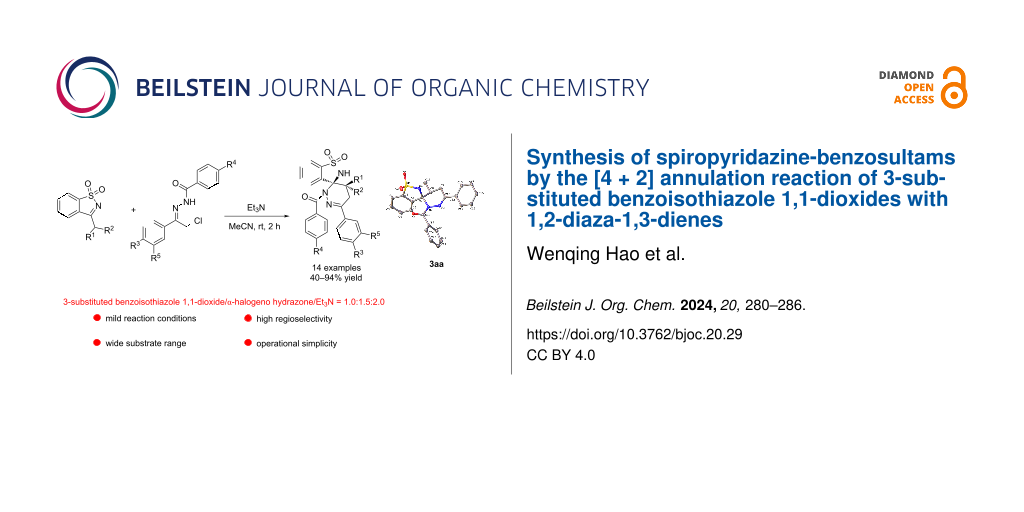
Abstract
A simple and efficient method for the synthesis of spiropyridazine-benzosultams has been developed by means of [4 + 2] annulation reaction of 3-substituted benzoisothiazole 1,1-dioxides with 1,2-diaza-1,3-dienes. This approach displays advantages such as mild reaction conditions, wide substrate range tolerance, simple operation, compatibility with gram-scale preparation.
Graphical Abstract
Introduction
Spirobenzosultams have various biological activities [1-3] such as antiviral, anticancer, antimicrobial, antimalarial, and antileukemia, and are widely used in the pharmaceutical field [4-6]. Pyridazine drugs have also shown high pharmaceutical activity. Many types of pyridazine drugs have been listed for antibacterial, anti-inflammatory, and other purposes [7-10]. Nowadays, a range of transformations to spirobenzosultams have been established using N-sulfonyl ketimines as suitable three-carbon synthons in [3 + 2] and [3 + 3] annulations [11-15]. However, methods using N-sulfonyl ketimines as dienophiles in [4 + 2] annulation reactions to afford biologically important spiro compounds are still limited to date [16]. 1,2-Diaza-1,3-dienes [17-27], which can be readily generated in situ from α-halogeno hydrazones, have been extensively applied in recent years as versatile building blocks in inverse-electron-demand Diels–Alder (IEDDA) reactions [28-30] to construct diverse nitrogen-containing heterocycles.
Inspired by the potential biological activity of pyridazines and in continuation of our work on the synthesis of spirobenzosultams [31-33], we herein report a highly diastereoselective route for the synthesis of spiropyridazine-benzosultams through [4 + 2] annulation reactions of 3-substituted benzoisothiazole 1,1-dioxides with 1,2-diaza-1,3-dienes (Scheme 1).
Scheme 1: Comparision of previous work with this work.
Scheme 1: Comparision of previous work with this work.
Results and Discussion
To initiate our studies, 3-ethylbenzo[d]isothiazole 1,1-dioxide (1a) and α-halogeno hydrazone 2a were selected as the model substrates. Our aim was to explore the possibility of enamine–iminium tautomerism of N-sulfonyl ketimine and its subsequent [4 + 2] annulation reaction with 1,2-diaza-1,3-diene in the presence of Et3N (2.0 equiv) in diethyl ether at room temperature (Table 1, entry 1). However, no product was detected under these conditions. We then replaced diethyl ether with toluene, which resulted in the desired spiropyridazine-benzosultam 3aa with 10% yield and high diastereoselectivity (Table 1, entry 2). Encouraged by this result, we tested several solvents to determine their effect on the [4 + 2] annulation reaction. Acetonitrile showed the best result, providing the highest yield (Table 1, entries 3–7). We also investigated the performance of other organic and inorganic bases, but they did not improve the yield (Table 1, entries 8–12). The structure of spiropyridazine-benzosultam 3aa was determined by 1H NMR, 13C NMR, HRMS analysis and single-crystal X-ray crystallography [33]. Further experiments conducted with different reaction times revealed that the reaction was complete within 2 hours (Table 1, entry 14). We then explored the effect of the temperature on the reaction and found that 25 °C was the most suitable temperature, resulting in a 91% yield (Table 1, entry 14). Lower temperatures of 0 °C and 10 °C led to decreased yields of 24% and 49%, respectively (Table 1, entries 16 and 17). Increasing the temperature beyond 25 °C resulted in the formation of impurities and a decrease in the yield of the target compound (Table 1, entries 18 and 19). Finally, the optimal reaction conditions were determined as follows: 1a (1.0 mmol), 2a (1.5 mmol), Et3N (2.0 mmol), in acetonitrile at 25 °C with stirring for 2 hours (Table 1, entry 14).
Table 1: The effects of solvents, bases, reaction time and temperature on the [4 + 2] annulation reactiona.
|
|
|||||
| Entry | Solvent | Base | Time (h) | Temperature (°C) | Yield (%) |
| 1 | Et2O | Et3N | 1.5 | 25 | 0 |
| 2 | toluene | Et3N | 1.5 | 25 | 10 |
| 3 | DCM | Et3N | 1.5 | 25 | 58 |
| 4 | THF | Et3N | 1.5 | 25 | 62 |
| 5 | MeCN | Et3N | 1.5 | 25 | 70 |
| 6 | DMF | Et3N | 1.5 | 25 | 68 |
| 7 | MeOH | Et3N | 1.5 | 25 | 63 |
| 8 | MeCN | DMAP | 1.5 | 25 | 51 |
| 9 | MeCN | DIPEA | 1.5 | 25 | 59 |
| 10 | MeCN | DBU | 1.5 | 25 | 66 |
| 11 | MeCN | NaH | 1.5 | 25 | 59 |
| 12 | MeCN | Cs2CO3 | 1.5 | 25 | 60 |
| 13 | MeCN | Et3N | 1.0 | 25 | 43 |
| 14 | MeCN | Et3N | 2.0 | 25 | 91 |
| 15 | MeCN | Et3N | 2.5 | 25 | 85 |
| 16 | MeCN | Et3N | 2.0 | 0 | 24 |
| 17 | MeCN | Et3N | 2.0 | 10 | 49 |
| 18 | MeCN | Et3N | 2.0 | 35 | 85 |
| 19 | MeCN | Et3N | 2.0 | 50 | 81 |
aReaction conditions: 1a (1.0 mmol), 2a (1.5 mmol), base (2.0 mmol), solvent (10.0 mL).
With the optimized reaction conditions in hand, we next investigated the scope of the current reaction (Scheme 2). As shown in Scheme 2, a series of α-halogeno hydrazones 2b–l was tested, resulting in the expected spiropyridazine-benzosultams 3ab–al in good to excellent yields with high diastereoselectivity. For N-benzoyl hydrazones 2b–g, the electronic effect of R3 group on the [4 + 2] annulation reaction was significant. Electron-withdrawing groups gave relatively higher yields than electron-donating groups (Scheme 2, 3aa–af). Remarkably, the nitro group gave the corresponding product 3ag in 94% yield. It was found that electron-donating groups afforded relatively higher dr values than electron-withdrawing groups (Scheme 2, 3aa–af). The effect of the R4 group was also detected and a similar result was observed as for the R3 group (Scheme 2, 3ah,ai vs 3aj,ak,al). To further expand the substrate scope, we next tested other 3-substituted benzoisothiazole 1,1-dioxides 1a–c. As seen from Scheme 2, dienophiles with a bulky and branched isopropyl group (1b) could also be employed in the reaction. However, the yield of the corresponding product 3ba was obviously lower than 3aa and 3ca generated from dienophiles bearing linear alkyl groups (Scheme 2, 3ba vs 3aa and 3ca).
Scheme 2: The effects of substituent groups on the [4 + 2] annulation reaction. Reaction conditions: 1 (1.0 mmol), 2 (1.5 mmol), Et3N (2.0 mmol), MeCN (10.0 mL), 25 °C, 2.0 h.
Scheme 2: The effects of substituent groups on the [4 + 2] annulation reaction. Reaction conditions: 1 (1.0 m...
A gram-scale reaction was subsequently conducted to investigate the scalability of the experiment. The reaction of 3-ethylbenzo[d]isothiazole 1,1-dioxide (1a, 1.0 g) and α-halogeno hydrazone 2a (2.1 g) afforded 3aa (2.0 g) in 91% yield (Scheme 3) [34]. Finally, we focused on the transformation of 3aa. When 3aa was treated with KOH and H2O in methanol at 60 °C, the 3,3-disubsitituted-1,2-benzothiazin-4-one 4aa [35] was isolated in 62% yield (Scheme 4).
On the basis of the transformation of 3aa to 4aa, a tentative reaction mechanism is proposed. As shown in Scheme 5, the spiropyridazine-benzosultam 3aa was firstly oxidized to intermediate A. Next, an aziridine was formed with the hydrolysis of the amide bond under basic conditions. Finally, the ring expansion led to intermediate C which was then hydrolyzed to 4aa.
Scheme 5: The reaction mechanism of the reaction from 3aa to 4aa.
Scheme 5: The reaction mechanism of the reaction from 3aa to 4aa.
Conclusion
In conclusion, we have developed a [4 + 2] annulation reaction of 3-substituted benzo[d]isothiazole 1,1-dioxides with 1,2-diaza-1,3-dienes for the efficient preparation of spiropyridazine-benzosultams. The electronic effects of substituents and the influence of steric hindrance on the reaction were explored. The configuration of the product was determined by X-ray single crystal diffraction. This method has the advantages of mild reaction conditions, wide substrate scope, and high regioselectivity.
Supporting Information
| Supporting Information File 1: Experimental part, NMR and HRMS spectra. | ||
| Format: PDF | Size: 3.0 MB | Download |
Data Availability Statement
All data that supports the findings of this study is available in the published article and/or the supporting information to this article. The data generated and analyzed during this study is openly available in Cambridge Crystallographic Data Centre at https://doi.org/10.5517/ccdc.csd.cc2hf5kp (3aa) and https://doi.org/10.5517/ccdc.csd.cc2hf5lq (4aa).
References
-
Scozzafava, A.; Owa, T.; Mastrolorenzo, A.; Supuran, C. T. Curr. Med. Chem. 2003, 10, 925–953. doi:10.2174/0929867033457647
Return to citation in text: [1] -
Zia-ur-Rehman, M.; Choudary, J. A.; Ahmad, S.; Siddiqui, H. L. Chem. Pharm. Bull. 2006, 54, 1175–1178. doi:10.1248/cpb.54.1175
Return to citation in text: [1] -
Valente, C.; Guedes, R. C.; Moreira, R.; Iley, J.; Gut, J.; Rosenthal, P. J. Bioorg. Med. Chem. Lett. 2006, 16, 4115–4119. doi:10.1016/j.bmcl.2006.04.079
Return to citation in text: [1] -
Zhao, Q.-Q.; Hu, X.-Q. Molecules 2020, 25, 4367. doi:10.3390/molecules25194367
Return to citation in text: [1] -
Gennari, C.; Salom, B.; Potenza, D.; Williams, A. Angew. Chem., Int. Ed. Engl. 1994, 33, 2067–2069. doi:10.1002/anie.199420671
Return to citation in text: [1] -
Carson, K. G.; Schwender, C. F.; Shroff, H. N.; Cochran, N. A.; Gallant, D. L.; Briskin, M. J. Bioorg. Med. Chem. Lett. 1997, 7, 711–714. doi:10.1016/s0960-894x(97)00090-5
Return to citation in text: [1] -
Grote, R.; Chen, Y.; Zeeck, A.; Chen, Z.; Zähner, H.; Mischnick-Lübbecke, P.; König, W. A. J. Antibiot. 1988, 41, 595–601. doi:10.7164/antibiotics.41.595
Return to citation in text: [1] -
Bizière, K.; Worms, P.; Kan, J. P.; Mandel, P.; Garattini, S.; Roncucci, R. Drugs Exp. Clin. Res. 1985, 11, 831–840.
Return to citation in text: [1] -
Tan, Y.-S.; Chen, Z.-F.; Liang, H.; Zhang, Y. Acta Crystallogr., Sect. E: Struct. Rep. Online 2005, 61, o1842–o1844. doi:10.1107/s1600536805015576
Return to citation in text: [1] -
Hayashi, F.; Kojima, K.; Fukushige, A.; Sakakibara, Y.; Hattori, T.; Namiki, S.; Namiki, T. Nihon Toseki Igakkai Zasshi 2000, 33, 279–285. doi:10.4009/jsdt.33.279
Return to citation in text: [1] -
Nishimura, T.; Ebe, Y.; Hayashi, T. J. Am. Chem. Soc. 2013, 135, 2092–2095. doi:10.1021/ja311968d
Return to citation in text: [1] -
Reddy, K. N.; Rao, M. V. K.; Sridhar, B.; Reddy, B. V. S. Eur. J. Org. Chem. 2017, 4085–4090. doi:10.1002/ejoc.201700569
Return to citation in text: [1] -
Li, Q.; Yuan, X.; Li, B.; Wang, B. Chem. Commun. 2020, 56, 1835–1838. doi:10.1039/c9cc09621c
Return to citation in text: [1] -
He, X.-L.; Xiao, Y.-C.; Du, W.; Chen, Y.-C. Chem. – Eur. J. 2015, 21, 3443–3448. doi:10.1002/chem.201404550
Return to citation in text: [1] -
Chen, X.; Zhang, J.-Q.; Yin, S.-J.; Li, H.-Y.; Zhou, W.-Q.; Wang, X.-W. Org. Lett. 2015, 17, 4188–4191. doi:10.1021/acs.orglett.5b01951
Return to citation in text: [1] -
Kumar, A. S.; Chauhan, S.; Swamy, K. C. K. Org. Lett. 2021, 23, 1123–1129. doi:10.1021/acs.orglett.1c00076
Return to citation in text: [1] -
Li, Z.; Li, S.; Kan, T.; Wang, X.; Xin, X.; Hou, Y.; Gong, P. Adv. Synth. Catal. 2020, 362, 2626–2631. doi:10.1002/adsc.202000398
Return to citation in text: [1] -
Zhang, X.; Pan, Y.; Wang, H.; Liang, C.; Ma, X.; Jiao, W.; Shao, H. Adv. Synth. Catal. 2021, 363, 459–463. doi:10.1002/adsc.202001375
Return to citation in text: [1] -
Wang, C.; Fang, L.; Wang, Z. Tetrahedron Lett. 2021, 79, 153303. doi:10.1016/j.tetlet.2021.153303
Return to citation in text: [1] -
Shen, L.-W.; Li, T.-T.; You, Y.; Zhao, J.-Q.; Wang, Z.-H.; Yuan, W.-C. J. Org. Chem. 2021, 86, 11472–11481. doi:10.1021/acs.joc.1c00993
Return to citation in text: [1] -
Mlostoń, G.; Urbaniak, K.; Sobiecka, M.; Heimgartner, H.; Würthwein, E.-U.; Zimmer, R.; Lentz, D.; Reissig, H.-U. Molecules 2021, 26, 2544. doi:10.3390/molecules26092544
Return to citation in text: [1] -
Shen, L.-W.; Zhang, Y.-P.; You, Y.; Zhao, J.-Q.; Wang, Z.-H.; Yuan, W.-C. J. Org. Chem. 2022, 87, 4232–4240. doi:10.1021/acs.joc.1c03072
Return to citation in text: [1] -
Sun, Y.; Yang, Z.; Lu, S.-N.; Chen, Z.; Wu, X.-F. Org. Lett. 2022, 24, 6822–6827. doi:10.1021/acs.orglett.2c02746
Return to citation in text: [1] -
Zhou, Y.; Zhou, H.; Xu, J. J. Org. Chem. 2022, 87, 3677–3685. doi:10.1021/acs.joc.1c02581
Return to citation in text: [1] -
Zhang, X.; Zheng, X.; Wang, C. Chin. J. Org. Chem. 2023, 43, 3180–3187. doi:10.6023/cjoc202303031
Return to citation in text: [1] -
Wang, D.-C.; Liu, J.-H.; Guan, Y.-Q.; Guo, H.-M. Org. Lett. 2023, 25, 3543–3547. doi:10.1021/acs.orglett.3c01174
Return to citation in text: [1] -
Zuo, W.-F.; Zhou, J.; Wu, Y.-L.; Fang, H.-Y.; Lang, X.-J.; Li, Y.; Zhan, G.; Han, B. Org. Chem. Front. 2021, 8, 922–927. doi:10.1039/d0qo01422b
Return to citation in text: [1] -
Skrzyńska, A.; Frankowski, S.; Albrecht, Ł. Asian J. Org. Chem. 2020, 9, 1688–1700. doi:10.1002/ajoc.202000332
Return to citation in text: [1] -
Huang, G.; Kouklovsky, C.; de la Torre, A. Chem. – Eur. J. 2021, 27, 4760–4788. doi:10.1002/chem.202003980
Return to citation in text: [1] -
Zhang, F.-G.; Chen, Z.; Tang, X.; Ma, J.-A. Chem. Rev. 2021, 121, 14555–14593. doi:10.1021/acs.chemrev.1c00611
Return to citation in text: [1] -
Cao, G.; Zhou, S.; Teng, D. Tetrahedron 2017, 73, 2329–2333. doi:10.1016/j.tet.2017.03.024
Return to citation in text: [1] -
Li, F.; Cao, G.; Gao, Y.; Teng, D. RSC Adv. 2017, 7, 10816–10820. doi:10.1039/c6ra26543j
Return to citation in text: [1] -
Cao, G.; Long, F.; Zhao, Y.; Wang, Y.; Huang, L.; Teng, D. Tetrahedron 2014, 70, 9359–9365. doi:10.1016/j.tet.2014.10.039
Return to citation in text: [1] [2] -
CCDC-2306572 (3aa) contains the supplementary crystallographic data for this paper. These data can be obtained free of charge from The Cambridge Crystallographic Data Centre via https://www.ccdc.cam.ac.uk/structures. doi:10.5517/ccdc.csd.cc2hf5kp
Return to citation in text: [1] -
CCDC-2306573 (4aa) contains the supplementary crystallographic data for this paper. These data can be obtained free of charge from The Cambridge Crystallographic Data Centre via https://www.ccdc.cam.ac.uk/structures. doi:10.5517/ccdc.csd.cc2hf5lq
Return to citation in text: [1]
| 1. | Scozzafava, A.; Owa, T.; Mastrolorenzo, A.; Supuran, C. T. Curr. Med. Chem. 2003, 10, 925–953. doi:10.2174/0929867033457647 |
| 2. | Zia-ur-Rehman, M.; Choudary, J. A.; Ahmad, S.; Siddiqui, H. L. Chem. Pharm. Bull. 2006, 54, 1175–1178. doi:10.1248/cpb.54.1175 |
| 3. | Valente, C.; Guedes, R. C.; Moreira, R.; Iley, J.; Gut, J.; Rosenthal, P. J. Bioorg. Med. Chem. Lett. 2006, 16, 4115–4119. doi:10.1016/j.bmcl.2006.04.079 |
| 16. | Kumar, A. S.; Chauhan, S.; Swamy, K. C. K. Org. Lett. 2021, 23, 1123–1129. doi:10.1021/acs.orglett.1c00076 |
| 11. | Nishimura, T.; Ebe, Y.; Hayashi, T. J. Am. Chem. Soc. 2013, 135, 2092–2095. doi:10.1021/ja311968d |
| 12. | Reddy, K. N.; Rao, M. V. K.; Sridhar, B.; Reddy, B. V. S. Eur. J. Org. Chem. 2017, 4085–4090. doi:10.1002/ejoc.201700569 |
| 13. | Li, Q.; Yuan, X.; Li, B.; Wang, B. Chem. Commun. 2020, 56, 1835–1838. doi:10.1039/c9cc09621c |
| 14. | He, X.-L.; Xiao, Y.-C.; Du, W.; Chen, Y.-C. Chem. – Eur. J. 2015, 21, 3443–3448. doi:10.1002/chem.201404550 |
| 15. | Chen, X.; Zhang, J.-Q.; Yin, S.-J.; Li, H.-Y.; Zhou, W.-Q.; Wang, X.-W. Org. Lett. 2015, 17, 4188–4191. doi:10.1021/acs.orglett.5b01951 |
| 7. | Grote, R.; Chen, Y.; Zeeck, A.; Chen, Z.; Zähner, H.; Mischnick-Lübbecke, P.; König, W. A. J. Antibiot. 1988, 41, 595–601. doi:10.7164/antibiotics.41.595 |
| 8. | Bizière, K.; Worms, P.; Kan, J. P.; Mandel, P.; Garattini, S.; Roncucci, R. Drugs Exp. Clin. Res. 1985, 11, 831–840. |
| 9. | Tan, Y.-S.; Chen, Z.-F.; Liang, H.; Zhang, Y. Acta Crystallogr., Sect. E: Struct. Rep. Online 2005, 61, o1842–o1844. doi:10.1107/s1600536805015576 |
| 10. | Hayashi, F.; Kojima, K.; Fukushige, A.; Sakakibara, Y.; Hattori, T.; Namiki, S.; Namiki, T. Nihon Toseki Igakkai Zasshi 2000, 33, 279–285. doi:10.4009/jsdt.33.279 |
| 4. | Zhao, Q.-Q.; Hu, X.-Q. Molecules 2020, 25, 4367. doi:10.3390/molecules25194367 |
| 5. | Gennari, C.; Salom, B.; Potenza, D.; Williams, A. Angew. Chem., Int. Ed. Engl. 1994, 33, 2067–2069. doi:10.1002/anie.199420671 |
| 6. | Carson, K. G.; Schwender, C. F.; Shroff, H. N.; Cochran, N. A.; Gallant, D. L.; Briskin, M. J. Bioorg. Med. Chem. Lett. 1997, 7, 711–714. doi:10.1016/s0960-894x(97)00090-5 |
| 33. | Cao, G.; Long, F.; Zhao, Y.; Wang, Y.; Huang, L.; Teng, D. Tetrahedron 2014, 70, 9359–9365. doi:10.1016/j.tet.2014.10.039 |
| 35. | CCDC-2306573 (4aa) contains the supplementary crystallographic data for this paper. These data can be obtained free of charge from The Cambridge Crystallographic Data Centre via https://www.ccdc.cam.ac.uk/structures. doi:10.5517/ccdc.csd.cc2hf5lq |
| 31. | Cao, G.; Zhou, S.; Teng, D. Tetrahedron 2017, 73, 2329–2333. doi:10.1016/j.tet.2017.03.024 |
| 32. | Li, F.; Cao, G.; Gao, Y.; Teng, D. RSC Adv. 2017, 7, 10816–10820. doi:10.1039/c6ra26543j |
| 33. | Cao, G.; Long, F.; Zhao, Y.; Wang, Y.; Huang, L.; Teng, D. Tetrahedron 2014, 70, 9359–9365. doi:10.1016/j.tet.2014.10.039 |
| 28. | Skrzyńska, A.; Frankowski, S.; Albrecht, Ł. Asian J. Org. Chem. 2020, 9, 1688–1700. doi:10.1002/ajoc.202000332 |
| 29. | Huang, G.; Kouklovsky, C.; de la Torre, A. Chem. – Eur. J. 2021, 27, 4760–4788. doi:10.1002/chem.202003980 |
| 30. | Zhang, F.-G.; Chen, Z.; Tang, X.; Ma, J.-A. Chem. Rev. 2021, 121, 14555–14593. doi:10.1021/acs.chemrev.1c00611 |
| 17. | Li, Z.; Li, S.; Kan, T.; Wang, X.; Xin, X.; Hou, Y.; Gong, P. Adv. Synth. Catal. 2020, 362, 2626–2631. doi:10.1002/adsc.202000398 |
| 18. | Zhang, X.; Pan, Y.; Wang, H.; Liang, C.; Ma, X.; Jiao, W.; Shao, H. Adv. Synth. Catal. 2021, 363, 459–463. doi:10.1002/adsc.202001375 |
| 19. | Wang, C.; Fang, L.; Wang, Z. Tetrahedron Lett. 2021, 79, 153303. doi:10.1016/j.tetlet.2021.153303 |
| 20. | Shen, L.-W.; Li, T.-T.; You, Y.; Zhao, J.-Q.; Wang, Z.-H.; Yuan, W.-C. J. Org. Chem. 2021, 86, 11472–11481. doi:10.1021/acs.joc.1c00993 |
| 21. | Mlostoń, G.; Urbaniak, K.; Sobiecka, M.; Heimgartner, H.; Würthwein, E.-U.; Zimmer, R.; Lentz, D.; Reissig, H.-U. Molecules 2021, 26, 2544. doi:10.3390/molecules26092544 |
| 22. | Shen, L.-W.; Zhang, Y.-P.; You, Y.; Zhao, J.-Q.; Wang, Z.-H.; Yuan, W.-C. J. Org. Chem. 2022, 87, 4232–4240. doi:10.1021/acs.joc.1c03072 |
| 23. | Sun, Y.; Yang, Z.; Lu, S.-N.; Chen, Z.; Wu, X.-F. Org. Lett. 2022, 24, 6822–6827. doi:10.1021/acs.orglett.2c02746 |
| 24. | Zhou, Y.; Zhou, H.; Xu, J. J. Org. Chem. 2022, 87, 3677–3685. doi:10.1021/acs.joc.1c02581 |
| 25. | Zhang, X.; Zheng, X.; Wang, C. Chin. J. Org. Chem. 2023, 43, 3180–3187. doi:10.6023/cjoc202303031 |
| 26. | Wang, D.-C.; Liu, J.-H.; Guan, Y.-Q.; Guo, H.-M. Org. Lett. 2023, 25, 3543–3547. doi:10.1021/acs.orglett.3c01174 |
| 27. | Zuo, W.-F.; Zhou, J.; Wu, Y.-L.; Fang, H.-Y.; Lang, X.-J.; Li, Y.; Zhan, G.; Han, B. Org. Chem. Front. 2021, 8, 922–927. doi:10.1039/d0qo01422b |
| 34. | CCDC-2306572 (3aa) contains the supplementary crystallographic data for this paper. These data can be obtained free of charge from The Cambridge Crystallographic Data Centre via https://www.ccdc.cam.ac.uk/structures. doi:10.5517/ccdc.csd.cc2hf5kp |
© 2024 Hao et al.; licensee Beilstein-Institut.
This is an open access article licensed under the terms of the Beilstein-Institut Open Access License Agreement (https://www.beilstein-journals.org/bjoc/terms), which is identical to the Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0). The reuse of material under this license requires that the author(s), source and license are credited. Third-party material in this article could be subject to other licenses (typically indicated in the credit line), and in this case, users are required to obtain permission from the license holder to reuse the material.