Abstract
Polycyclic spirobarbiturates containing a pyrrolizidine moiety were synthesized via a one-pot three-component 1,3-dipolar cycloaddition reaction of alloxan, ʟ-proline and N-substituted maleimides. The reaction stereoselectivity was found to depend on the nature of substituents in the maleimide: in most cases the endo-isomer is the main (or only) product, but for some N-arylmaleimides the exo-product is predominant. The proposed mechanism is discussed, and the products were characterized by detailed spectral analysis. The configuration of the stereocenters was determined by X-ray diffraction analysis (XRD) for two adducts, followed by Hirshfeld surface analysis. The antiproliferative effect of the synthesized compounds against cancer cell lines was assessed.
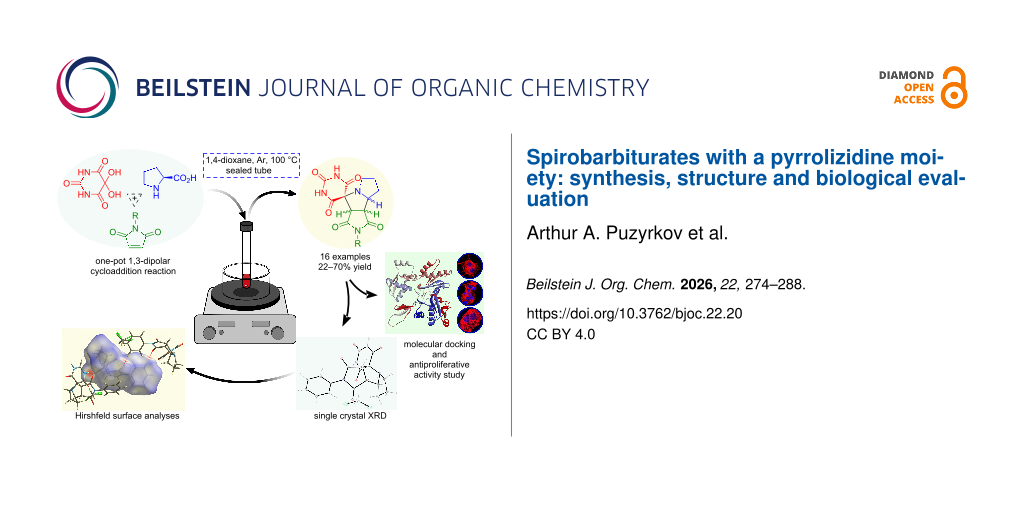
Graphical Abstract
Introduction
The history of barbiturates dates back to 1863, when the young Adolf von Baeyer first synthesized barbituric acid [1,2]. In 1903, the drug marketed under the trade name barbital (veronal©) became commercially available as a sedative [3]. For more than 120 years of barbiturate history a lot has happened. Barbituric acid-based medications were widely used until the 40–50s of the last century. Then, due to accumulating evidence of their addictive potential and adverse effects, most of these substances were eventually phased out of pharmaceutical use. Today, only slightly more than a dozen barbiturate-containing drugs remain in medical practice [4]. Nevertheless, interest in barbiturate derivatives resurged around the 1920s, when the first compounds containing a spirobarbiturate moiety were synthesized [5,6]. By the early 21st century, this research culminated in the approval of the first drug whose active molecule contained a spirobarbiturate scaffold – zoliflodacin© (Scheme 1) [7].
Scheme 1: Biologically active compounds with a spirobarbiturate moiety in their structure [7-12].
Scheme 1: Biologically active compounds with a spirobarbiturate moiety in their structure [7-12].
Due to the significance of spirobarbiturates, numerous approaches have been devised for their synthesis [13-19]. Advances in this area of organic synthesis include the preparation of barbiturates containing spiro-fused cyclohexane, cyclopentane, tetrahydrooxepine, and tetrahydroquinoline moieties via [3 + 2], [4 + 2], and [5 + 2] annulation reactions involving arylidene and alkylidene barbiturates [20-23]. In 2018, Kota and Sreevani prepared spirobarbiturates via a Mo(CO)6-catalyzed intermolecular [2 + 2 + 2] cycloaddition of propargyl halides with dipropargylbarbituric acid [24]. Almeida and colleagues described the formation of spiroindoline barbiturates via thermal rearrangement of 2,1-benzisoxazole barbiturates [25]. Other methods include the DBU-mediated stereospecific cyclopropanation of barbiturate-based olefins with benzyl chlorides, developed by Chang’s group for the synthesis of spirobarbiturate-cyclopropanes [26].
Another interesting structural fragment is the pyrrolizidine heterocycle, which is a system of two condensed pyrrolidine rings sharing a common nitrogen atom. This heterocyclic system occurs naturally in numerous plant-derived alkaloids [27-30]. Notably, most pyrrolizidine alkaloids exhibit severe hepatotoxicity, genotoxicity and cause neurological disorders in humans and animals [31-33]. However, some representatives of these compounds (Scheme 2) find applications in pharmacology [34] due to their antimicrobial (megalanthonine; subulacine N-oxide) [35,36], anti-inflammatory (lindelofidine, benzoic acid ester; paludosine) [37,38], anticancer (ligulachyroine) [39] and antiviral activities (7,7a-diepialexine) [40]. Some pyrrolizidine derivatives are inhibitors of enzymes such as acetylcholinesterase (AChE) (echimidine) [41] or β-N-acetylglucosaminidase (GlcNAcases) (pochonicine) [42].
Scheme 2: Biologically active alkaloids with a pyrrolizidine moiety.
Scheme 2: Biologically active alkaloids with a pyrrolizidine moiety.
Our previous studies have shown that spiro-fused barbiturates containing azabicyclo[3.1.0]hexane and cyclopropa[a]pyrrolizine moieties can be synthesized via 1,3-dipolar cycloaddition between cyclopropene derivatives and azomethine ylides generated in situ from alloxan and α-amino acids [43]. In [43], a mechanism was proposed for the formation of azomethine ylides from alloxan and α-amino acids, which includes a stage of decarboxylation of the intermediate lactone. It is worth noting that generating azomethine ylides via decarboxylation of α-amino acid derivatives is a common practice in synthetic chemistry [44-46]. It was shown in our recent works that spirocyclopropa[a]pyrolizines as well as spiroazabicyclo[3.1.0]hexanes are readily available from cyclopropenes and azomethine ylides [47-49] and some adducts inhibited cancer cell growth in vitro [50,51]. The first reported synthesis of spirobarbiturates employing azomethine ylides (generated in situ from alloxan and α-amino acids) reacted with N-methylmaleimide was published by Ronald Grigg’s group in 1994 (Scheme 3) [52]. The authors reported the synthesis of spirobarbiturates containing various substituents: R’ = iPr (67%), Bn (61%), and Ph (66%).
Scheme 3: Previous studies on the three-component synthesis of spirobarbiturates.
Scheme 3: Previous studies on the three-component synthesis of spirobarbiturates.
In this work, which is a continuation of studies on the synthesis of spirobarbiturates via a [3 + 2] cycloaddition reaction of azomethine ylides, we present for the first time a three-component diastereoselective synthesis of hybrid systems containing spiro-fused cyclic fragments of barbituric acid and pyrrolo[3,4-a]pyrrolizidine. It was found that azomethine ylide, which is generated in situ by condensation of alloxan and ʟ-proline, can react with maleimides to give spirobarbiturate-pyrrolo[3,4-a]pyrrolizidine-1,3-diones in moderate to good yields. We have demonstrated the possibility of using a wide range of N-substituted maleimides as dipolarophiles. All obtained spirocyclic adducts became objects of biological research.
Results and Discussion
The 1,3-dipolar cycloaddition between alloxan (1), ʟ-proline (2), and maleimides 3a–p afforded spirobarbiturates 4a–p as diastereomeric racemic mixtures with 22–70% yields (Scheme 4). The reaction progress is highly sensitive to water presence in the system. It is worth noting that in all cases, a side reaction was observed between alloxan (1) and α-amino acid 2, leading to the formation of murexide via Strecker degradation (Scheme S1, Supporting Information File 1) [53]. This side reaction resulted in decreased yields of the target products. We observed that murexide formation decreased when the reaction was carried out in anhydrous 1,4-dioxane under an inert atmosphere at 100 °C. When using dry acetonitrile as a solvent, the reaction yields were lower, so we decided to use a higher-boiling solvent – 1,4-dioxane.
Scheme 4: Synthesis of racemic spirobarbiturates 4a–p via one-pot three-component reaction of alloxan (1), ʟ-proline (2) and N-substituted maleimides 3.
Scheme 4: Synthesis of racemic spirobarbiturates 4a–p via one-pot three-component reaction of alloxan (1), ʟ-...
It was established that both diastereoselectivity and reaction yields significantly depend on the N-substituent of the maleimide: exclusive formation of endo-products 4a–e (yields up to 52%) was observed when using maleimides with R = H (3a), Me (3b), Ph (3c), p-Tol (3d), or 4-NO2С6H4 (3e) at the nitrogen atom. Maleimides bearing aromatic halogens (3h, 3i, 3j, 3l) or R = Bn (3f), -CH2CH2Ph (3g), 3-NO2С6H4 (3k), and 3-CF3С6H4 (3m) groups yielded mixtures of exo- and endo-adducts. When the reaction was performed with a maleimide containing a chlorine atom at the para-position, a near 1:1 mixture of endo- and exo-isomers 4h (34%) was obtained. For the meta-chloro-substituted maleimide, the exo-isomer 4l (25%) predominated (endo/exo 1:2.5). In contrast, the maleimide bearing two chlorine atoms at the para- and meta-positions preferentially formed the endo-isomer 4i (50%, endo/exo 1:0.77). Substitution of the meta-chlorine with fluorine also favored the exo-isomer (compound 4j, 70%, endo/exo 1:2), similar to 4l. The presence of an electron-withdrawing group at the meta-position of the maleimide’s N-aryl group likely facilitates exo-isomer formation. Reactions with maleimides containing meta-NO2 and meta-CF3 groups were investigated to test this hypothesis. Contrary to expectations, the endo-isomer predominated in these cases (4k, 30%, endo/exo 1:0.14; 4m, 50%, endo/exo 1:0.45). We also synthesized two spirobarbiturates, 4n (59%) and 4o (40%), from maleimides containing electron-donating groups at the meta-position of the aromatic ring. In both cases, the endo-isomers predominated among the cycloadducts. However, using an N-arylmaleimide with an ortho-methoxy group, we obtained product 4p (26%) as diastereomeric mixture, and the exo-isomer became the major product. Notably, the 1H NMR spectrum of product 4p exhibited three doublet signals corresponding to the methine proton in a five-membered ring formed during cycloaddition. This observation indicated the formation of atropisomers in the 1,3-dipolar cycloaddition of N-(2-methoxyphenyl)maleimide (3p) which was previously reported by Awad I. Said et al. [54].
The cycloaddition reactions were performed in sealed tubes under an inert atmosphere at 100 °C in anhydrous 1,4-dioxane with constant stirring for 6–16 h. Reaction progress was monitored by TLC (CH2Cl2/MeOH 25:1) and the spirobarbiturates were isolated from the reaction mixture by preparative TLC or column chromatography. Notably, compounds 4a, 4e, and 4k were purified by crystallization. Detailed synthetic and isolation procedures for all compounds are provided in Supporting Information File 1. We regret to state that when forming a mixture of diastereomers, we were unable to isolate individual products either by chromatography or by recrystallization.
The proposed reaction mechanism is shown in Scheme 5, where it can be seen that the intermediate azomethine ylide, acting as a 1,3-dipole, undergoes a [3 + 2] cycloaddition with N-substituted maleimides 3a–p. The formation of azomethine ylide from alloxan (1) and ʟ-proline (2) was previously studied using DFT calculations [43]. Based on the calculated data, it was assumed that the 1,3-dipole is formed as a result of a multistage sequence: initial generation of a zwitterionic imine intermediate from 1 and 2, followed by intramolecular cyclization to yield a lactone, which undergoes decarboxylation to produce the corresponding azomethine ylide. In our system, we propose this generated ylide reacts with N-substituted maleimides 3a–p such that the transformation predominantly proceeds via the endo-TS, leading to preferential formation of the endo-adduct (upper pathway). The predominance of exo-isomers in some cases probably reflects insufficient stabilization due to secondary orbital interactions in the transition state [43].
Scheme 5: A plausible mechanism of spirobarbiturate formation from alloxan (1), ʟ-proline (2), and N-substituted maleimides 3a–p.
Scheme 5: A plausible mechanism of spirobarbiturate formation from alloxan (1), ʟ-proline (2), and N-substitu...
It should be noted separately that secondary cyclic amino acids such as azetidine-2-carboxylic acid, thiazolidinecarboxylic acid, and pipecolic acid, in combination with alloxan (1) and maleimides 3, did not lead to the formation of the corresponding cycloadducts. In this work, we also used other dipolarophiles. Under the standard reaction conditions (1,4-dioxane, heating with alloxan and ʟ-proline), tetracyanoethylene, chalcone, and dimethyl acetylenedicarboxylate all failed to produce the target spiro adducts – chalcone and dimethyl acetylenedicarboxylate showed no reactivity, while tetracyanoethylene led to complete resinification.
The structure of the products, including the configuration of their stereocenters, was determined using NMR spectroscopy and XRD analysis. endo-Isomers 4a–e exhibited the following characteristic signals in their 1H NMR spectra: multiplets in the range of δ 1.5–4.4 ppm were assigned to the protons of the pyrrolizidine ring system. The methyl singlets of 4b and 4d (δ 2.79 and 2.35 ppm, respectively) also fall within this range.
The methylene protons of the five-membered ring A (Figure 1) in endo-isomers 4a–e resonate as complex multiplets: CH22 and CH23 groups appear at δ 1.65–2.05 ppm, while CH21 is deshielded (δ 2.54–2.70 ppm), consistent with the deshielding effect of the directly attached nitrogen atom on the CH21 group. The methine protons resonate as distinct signals appearing as a ddd for CH4, dd for CH5, and a d for CH6. Chemical shift values, signal multiplicities, and spin–spin coupling constants for the methine protons of all compounds are provided in Supporting Information File 1. Notably, for compounds 4a and 4b containing non-aromatic substituents in the succinimide moiety, the signals of protons CH4 and CH6 overlap, whereas no such overlap occurs with aromatic substituents. The assignments of the methine proton signals for all compounds were established using 1H,13C HSQC NMR spectroscopy. The chemical shift values, signal shapes, and spin–spin coupling constants are provided in Table S1 in Supporting Information File 1. The aromatic protons of compounds 4c–e appear in the downfield region of the spectrum. For product 4c, the aromatic protons resonate as three distinct signals: a broad doublet at δ 7.26 ppm (ortho-protons), along with multiplets at δ 7.40–7.47 ppm (para-proton), and δ 7.48–7.56 ppm (meta-protons). In contrast, compounds 4d and 4e, which contain para-substituted aromatic rings, exhibit two characteristic doublets each: 4d shows signals at δ 7.12 and 7.30 ppm for ortho- and meta-protons respectively (J = 8.2 Hz) and 4e displays resonances at δ 7.58 and 8.41 ppm for ortho- and meta-protons (J = 8.8 Hz). In the 1H NMR spectra, the proton signals of ring A in diastereomers 4f–p overlap and appear at the same chemical shifts as in the endo-isomers 4a–e. The CH4–CH6 protons of diastereomers 4f–p resonate as distinct separate signals. The formation of atropisomers 4p is evidenced by three distinct doublets in the 1H NMR spectrum at δ 4.29 ppm (J = 8.8 Hz), 4.49 ppm (J = 10.3 Hz), and 4.58 ppm (J = 10.3 Hz), assigned to the diastereotopic H6/H6’ methine protons of different atropisomeric forms. The NH protons of the barbiturate ring resonate as broad singlets at δ 11.5–11.7 ppm, while compound 4a shows an additional broad singlet at δ 11.30 ppm corresponding to the succinimide NH group.
Figure 1: Schematic structures of endo- and exo-adducts of spirobarbiturates 4.
Figure 1: Schematic structures of endo- and exo-adducts of spirobarbiturates 4.
In the 13C NMR spectra of endo-adducts 4a–e, the carbon signals of ring A (Figure 1) appear at δC 44.8–45.0 ppm (CH21), 27.3 ppm (CH22), and 25.2 ppm (CH23). Ring B carbons resonate at δC 67.7–67.8 ppm (CH4), 46.3–46.4 ppm (CH5), and 58.2–58.3 ppm (CH6). The carbonyl carbons of ring C and the barbiturate ring appear at δC 150.0, 169.3–169.4, 171.2–171.5, 175.5–176.8, and 176.4–177.9 ppm. The spiro carbon appears at δC 70.1–70.3 ppm. For diastereomers 4f–p, the 13C NMR spectra proved more informative than the 1H NMR spectra, clearly displaying a duplicate set of nearly all signals. The chemical shifts of carbons in rings A, B, and C for endo-4f–p could be unambiguously identified, as they remain virtually unchanged compared to those in the pure endo-isomers 4a–e.
19F NMR analysis of 4j revealed two fluorine signals (δF −114.3 ppm for exo, −114.4 ppm for endo), while the CF3 group in 4m showed analogous splitting (δF −61.2 ppm endo, −61.3 ppm exo).
The structures of all cycloadducts 4a–p were confirmed by 13C DEPT NMR and two-dimensional NMR spectroscopy (HSQC, COSY), supplemented by HRESIMS. Complete spectral data are provided in Supporting Information File 1.
The molecular structures of 4b and 4c were also confirmed by XRD (Figure 2). Both compounds crystallize as racemates, Figure 3 displaying their unit cell fragments. In 4b, the two enantiomers are connected through weak hydrogen bonds (N–H···O 2.612 Å) between the barbiturate rings. In contrast, compound 4c exhibits classical hydrogen bonding (N–H···O 1.878 Å) linking barbiturate moieties of paired enantiomers (see Supporting Information File 1, Tables S2 and S3).
![[1860-5397-22-20-2]](/bjoc/content/figures/1860-5397-22-20-2.png?scale=2.0&max-width=1024&background=FFFFFF)
Figure 2: X-ray crystal structures of compounds 4b (CCDC 2391172, left) and 4c (CCDC 2391171, right).
Figure 2: X-ray crystal structures of compounds 4b (CCDC 2391172, left) and 4c (CCDC 2391171, right).
![[1860-5397-22-20-3]](/bjoc/content/figures/1860-5397-22-20-3.png?scale=2.0&max-width=1024&background=FFFFFF)
Figure 3: Unit cell packing of products 4b (left) and 4c (right).
Figure 3: Unit cell packing of products 4b (left) and 4c (right).
The Hirshfeld surface (HS) analysis provides a robust tool for visualizing and characterizing intermolecular interactions in organic crystal lattices. The calculations of the HS were performed using CrystalExplorer 21.5 [55]. On the Hirshfeld surface, color coding indicates the nature of intermolecular interactions: red highlights contacts shorter than the van der Waals radius, white represents distances equal to the van der Waals radius, and blue denotes contacts longer that the van der Waals radius. Figure 4 shows front and back views of the Hirshfeld surfaces (dnorm) for compounds 4b and 4c.
![[1860-5397-22-20-4]](/bjoc/content/figures/1860-5397-22-20-4.png?scale=2.0&max-width=1024&background=FFFFFF)
Figure 4: HS mapped with dnorm for compounds 4b (left) and 4c (right).
Figure 4: HS mapped with dnorm for compounds 4b (left) and 4c (right).
The Hirshfeld surface for compound 4b with dnorm map plotted (ranging from −0.8499 (red) to 1.4229 (blue) a.u.) is presented in Figure 5. Analysis of the HS revealed the presence of several intermolecular contacts: the barbiturate ring forms hydrogen bonds with water molecules through C=O···H–O and N–H···O–H interactions; the amide group of the barbiturate ring is engaged in a C=O···H–N hydrogen bond with the carbonyl oxygen of the maleimide fragment; additionally, a C=O···H–CH2 contact is observed between the hydrogen atom of the maleimide methyl group and the carbonyl oxygen of an adjacent maleimide ring.
Figure 5: A segment of the crystal structure of compound 4b with the HS (dnorm), showing intermolecular contacts (lengths shown for D···A).
Figure 5: A segment of the crystal structure of compound 4b with the HS (dnorm), showing intermolecular conta...
The crystal structure of compound 4b exists as a solvate. The oxygen atom of a water molecule forms a hydrogen bond with the amide hydrogen of the barbiturate fragment (N–H···O–H = 2.783 Å), while one of the water hydrogens interacts with a carbonyl group of the barbiturate ring (C=O···H–O = 2.867 Å).
The HS for compound 4c with dnorm map plotted (ranging from −0.6673 (red) to 1.4313 (blue) a.u.), is shown in Figure 6. The dnorm map highlights key intermolecular interactions (marked in red), including O···H–N hydrogen bonds between barbiturate rings and O···H–Ph contacts involving maleimide carbonyl oxygens and phenyl ring hydrogens.
Figure 6: A segment of the crystal structure of compound 4c with the HS (dnorm), showing intermolecular contacts (lengths shown for D···A).
Figure 6: A segment of the crystal structure of compound 4c with the HS (dnorm), showing intermolecular conta...
The crystal structure of adduct 4c includes a molecule of dichloromethane. Due to the disorder of the dichloromethane molecule, the exact distances of the corresponding intermolecular interactions cannot be precisely determined. However, the nature of these interactions can be described as follows: one of the hydrogen atoms of dichloromethane contacts with the oxygen atoms of carbonyl groups from two nearby barbiturate rings. The estimated contact distances are C=O···H ≈ 3.6 Å, which is consistent with weak hydrogen-bonding interactions.
In silico analyses of drug-like properties. An in silico analysis was performed to preliminarily determine whether the synthesized spiro-fused adducts have drug-like properties. The physicochemical profile was determined using the free online software SwissADME (http://www.swissadme.ch/). The molecular descriptors were calculated according to Lipinski’s rule of five. This rule was formulated by Ch.A. Lipinski based on the observation that most orally administered drugs are relatively small and moderately lipophilic molecules [56]. By this rule, orally active drugs should not violate more than one of the following criteria: MW – molecular weight: < 500 Da; NHBD – number of hydrogen-bond donors, Log(P) – octanol/water partition coefficient: <5; NHBA – number of hydrogen-bond acceptors, NRotB – number of rotatable bonds: <10 and TPSA – topological polar surface area: <140 Å2. The obtained results are presented in Supporting Information File 1, Table S4.
Since antitumor drugs usually damage healthy cells along with tumor cells, while developing new substances, it is extremely important to pay attention to their ADMET properties: absorption, distribution, metabolism, excretion, and toxicity. In this paper, these were evaluated in silico using an online resource accessible via https://preadmet.webservice.bmdrc.org/. The following ADME descriptors were selected: blood-brain barrier permeability (BBB), human intestinal absorption (HIA), in vitro permeability to Caco-2 cells (Caco2), in vitro binding to plasma proteins (PPB), solubility, and inhibition of CYP2D6. The following were selected as descriptors of toxicity: carcinogenicity for rats and mice, mutagenicity according to the Ames test, and cardiotoxicity by inhibition of hERG in vitro. The results are shown in Supporting Information File 1, Table S5.
As can be seen from the table, the obtained results suggest that the compounds have a good intestinal absorption and medium permeability. However they are expected to have low plasma protein binding and permeation potential in the brain with regard to bioavailability in the CNS.
Antiproliferative activity study. Cancer cells are favorable in vitro models that are widely used in cancer research and drug discovery. In this study, the MTS assay was applied to evaluate the antiproliferative activity of the synthesized compounds against human cervical carcinoma (HeLa), erythroleukemia (K562), and melanoma (Sk-mel-2) cell lines. It was found that the studied spiro-fused adducts reduced the cell proliferation in a time- and concentration-dependent manner. The results of these investigations for 24 h and 72 h are presented in Supporting Information File 1, Figures S77–S79. As can be seen from the obtained results, the compounds generally have a limited antiproliferative effect against these cells in contrast to previously studied compounds that contain a cyclopropane moiety instead of a pyrrolidine one [57] and tryptanthrin, 11H-benzo[4,5]imidazo[1,2-a]indol-11-one or indolin-2-one instead of the barbituric moiety [58-60]. The most interesting effect however, was the increased Sk-mel-2 cell viability under treatment with compounds 4f and 4g at the lowest concentration tested (5 µg/mL).
Actin cytoskeleton changes. It is known that actin plays an important role in vital cellular processes, providing a number of functions such as cell migration, adhesion, and morphogenesis [61,62]. The dynamic reorganization of the actin cytoskeleton, which plays a key role in a variety of cellular functions, directly determines cell motility, and its structural organization can serve as an indicator of the metastatic potential of tumor cells [63-65].
The presence of stress fibers and filopodia-like protrusions were used to evaluate the Sk-mel-2 cells' actin cytoskeleton structure after their co-incubation with spiro-fused adducts 4f, 4g, 4i, 4k, and 4l.
Confocal microscopy showed that co-incubation of Sk-mel-2 cells with adducts 4f, 4g, 4i, 4k, and 4l led to significant changes in the structure of their actin cytoskeleton resulting in stress fibers disappearance (granular actin was found in the cytoplasm of up to 87% cells) and increased number of filopodia-like deformations (up to 61% after co-incubation). Such changes in the cytoskeleton structure may indicate a change in the motor activity of cells. Although compounds 4f and 4g showed increased cell viability, they may have a reduced wound healing ability if applied to non-tumor cells. At the same time, nucleus fragmentation was not observed during the experiment which may indicate the absence of pro-apoptotic activity. Data on actin cytoskeleton structure as well as data demonstrating percentage of cells with filopodia-like deformations and disassembled stress fibers are combined at Figure 7.
![[1860-5397-22-20-7]](/bjoc/content/figures/1860-5397-22-20-7.png?scale=2.0&max-width=1024&background=FFFFFF)
Figure 7: Microscopic images of treated cells and state of the actin cytoskeleton of Sk-mel-2 cells after cultivation with compounds 4f, 4g, 4i, 4k, and 4l (10 μg/mL); p-value <0.05 (*), 0.01 (**), 0.001 (***).
Figure 7: Microscopic images of treated cells and state of the actin cytoskeleton of Sk-mel-2 cells after cul...
Additionally, a scratch-test was performed to study the wound healing ability of these compounds and the results are given in Supporting Information File 1, Figure S80 and Table S6. The measured wound area after 24 h of incubation with compounds 4f, 4g, 4i, 4k and 4l was equal to 25, 19, 31, 21, 23, and 19% for the tested compounds and control sample, respectively. As can be seen from the obtained data the wound healing ability are consistent with the cytoskeleton data. Compounds 4g and 4l, which led to less stress fibers (87 ± 22%, 71 ± 16% vs 46 ± 7% for 4g, 4l vs control, respectively) and nearly the same filopodia-like deformations (35 ± 15% and 36 ± 22% vs 33 ± 17% for 4g and 4l vs control, respectively) as compared to the control, result in low wound healing ability when applied to Sk-mel-2 cells. At the same time, compound 4i which led to slightly less stress fibers (53 ± 3% vs 46 ± 7% for 4i vs control, respectively) and more filopodia-like deformations (57 ± 3% vs 33 ± 17% for 4i vs control, respectively) as compared to the control, result in increased wound healing ability in treated Sk-mel-2 cells.
Molecular docking. To confirm the results obtained, docking simulations were performed for the possible interaction of spiro-adducts with actin, as the most widespread and highly conserved cellular protein. Since the initial determination of the G-actin crystal structure in complex with DNase I, many actin structures have been registered. Taking into account that the conformation of the actin monomer in the described structures is basically the same, the structure of non-muscle β-actin (8DNH, 2.99 Å) obtained by cryo-electron microscopy was used for this study. Docking was performed to both known clefts (hydrophobic or target-binding one and nucleotide, DNaze I-binding).
The structure of the 8DNH protein was retrieved from the protein data bank and the Molegro Virtual Docker 6.0 software was used to prepare it for the docking study [66,67]. The pose organizer and the ligand energy inspector tool were used to examine the docking results. The latter were then tabulated and the docked view was retrieved. Figure 8 and Table S7 in Supporting Information File 1 show the docking results. The analysis of the data obtained, shows that the affinity for the hydrophobic target-binding cleft was always lower in comparison to nucleotide cleft (Rerank Score was found to be from −77 to −108 and from −93 to −125 arbitrary units for both clefts correspondingly, data are collected in Table S7 in Supporting Information File 1). The predicted binding models also revealed that the adducts, while being fitted within the cleft, usually are located in such way that the barbituric moiety is directed to Trp 339, while the aryl substituent of the imide moiety is directed to Lys 212. This result agrees with the data on changes in the actin cytoskeleton and can be caused by a balance disturbance in the processes of actin polymerization and depolymerization that continuously occur in cells.
![[1860-5397-22-20-8]](/bjoc/content/figures/1860-5397-22-20-8.png?scale=2.0&max-width=1024&background=FFFFFF)
Figure 8: Docked view of compounds 4f, 4g, 4i, 4k, and 4l with the target protein (PDB ID: 8DNH).
Figure 8: Docked view of compounds 4f, 4g, 4i, 4k, and 4l with the target protein (PDB ID: 8DNH).
Conclusion
We have developed a novel one-pot three-component method for the synthesis of pyrrolizidine-containing spirobarbiturates via 1,3-dipolar cycloaddition reactions. The study revealed that maleimide derivatives with -H, -Me, -Ph, p-Tol, or 4-NO2С6H4 substituents exclusively form endo-adducts with yields ranging from 22% to 52%, while the use of maleimides bearing -Bn, -CH2CH2Ph, or halogenated aromatic substituents led to a significant decrease in diastereoselectivity, resulting in mixtures of endo- and exo-isomers with yields of 25–70%. Notably, meta-halogen substitution in aromatic maleimides was found to preferentially direct the cycloaddition toward the formation of exo-isomers. The structures of all obtained diastereomers were thoroughly characterized by 1H and 13C NMR spectroscopy. Single crystal XRD analysis of two representative compounds allowed unambiguous determination of the absolute configuration of the spirobarbiturate adducts. Additionally, a detailed analysis of intermolecular interactions in the crystal structure was performed using Hirshfeld surface calculations, providing valuable insights into the packing arrangements of these compounds. The results of antiproliferative activity study showed that generally spiro-adducts have limited effect on the tested cancer cells, while they caused significant changes of Sk-mel-2 cells’ actin cytoskeleton structure leading to the disappearance of stress fibers (granular actin was distributed diffusely in the cytoplasm of treated cells in up to 87%) and changes in the number of filopodia-like deformations (increased up to 61% after cultivation).
Supporting Information
| Supporting Information File 1: General information, experimental procedures, characterization data, X-ray data and biological activity data. | ||
| Format: PDF | Size: 12.1 MB | Download |
Data Availability Statement
All data that supports the findings of this study is available in the published article and/or the supporting information of this article.
References
-
Baeyer, A. Justus Liebigs Ann. Chem. 1863, 127, 199–236. doi:10.1002/jlac.18631270214
Return to citation in text: [1] -
Baeyer, A. Justus Liebigs Ann. Chem. 1864, 130, 129–175. doi:10.1002/jlac.18641300202
Return to citation in text: [1] -
Fischer, E.; von Mering, J. Ther. Ggw. 1903, 44, 97–101.
Return to citation in text: [1] -
López-Muñoz, F.; Ucha-Udabe, R.; Alamo, C. Neuropsychiatr. Dis. Treat. 2005, 1, 329–343.
Return to citation in text: [1] -
Dox, A. W.; Yoder, L. J. Am. Chem. Soc. 1921, 43, 677–684. doi:10.1021/ja01436a040
Return to citation in text: [1] -
Dox, A. W.; Yoder, L. J. Am. Chem. Soc. 1921, 43, 1366–1370. doi:10.1021/ja01439a018
Return to citation in text: [1] -
Taylor, S. N.; Marrazzo, J.; Batteiger, B. E.; Hook, E. W., III; Seña, A. C.; Long, J.; Wierzbicki, M. R.; Kwak, H.; Johnson, S. M.; Lawrence, K.; Mueller, J. N. Engl. J. Med. 2018, 379, 1835–1845. doi:10.1056/nejmoa1706988
Return to citation in text: [1] [2] -
Damião Gouveia, A. C.; Unemo, M.; Jensen, J. S. J. Antimicrob. Chemother. 2018, 73, 1291–1294. doi:10.1093/jac/dky022
Return to citation in text: [1] -
King, S. B.; Stratford, E. S.; Craig, C. R.; Fifer, E. K. Pharm. Res. 1995, 12, 1240–1243. doi:10.1023/a:1016236615559
Return to citation in text: [1] -
Galati, E. M.; Monforte, M. T.; Miceli, N.; Raneri, E. Farmaco 2001, 56, 459–461. doi:10.1016/s0014-827x(01)01062-x
Return to citation in text: [1] -
Chan, P. F.; Srikannathasan, V.; Huang, J.; Cui, H.; Fosberry, A. P.; Gu, M.; Hann, M. M.; Hibbs, M.; Homes, P.; Ingraham, K.; Pizzollo, J.; Shen, C.; Shillings, A. J.; Spitzfaden, C. E.; Tanner, R.; Theobald, A. J.; Stavenger, R. A.; Bax, B. D.; Gwynn, M. N. Nat. Commun. 2015, 6, 10048. doi:10.1038/ncomms10048
Return to citation in text: [1] -
Scott, G. K.; Benz, C. C. Inhibition of proline catabolism for the treatment of cancer and other therapeutic applications. WO Pat. Appl. WO2016077632A2, May 19, 2016.
Return to citation in text: [1] -
Huo, M.; Zhou, J.; Bai, L.; Xu, Q.; Zhou, Z.; Zhou, H.; Liang, G. Tetrahedron 2019, 75, 130752. doi:10.1016/j.tet.2019.130752
Return to citation in text: [1] -
Borisova, Y. G.; Raskil’dina, G. Z.; Zlotskii, S. S. Dokl. Chem. 2017, 476, 201–205. doi:10.1134/s0012500817090014
Return to citation in text: [1] -
Elinson, M. N.; Vereshchagin, A. N.; Korshunov, A. D.; Ryzhkov, F. V.; Egorov, M. P. Heterocycl. Commun. 2017, 23, 85–90. doi:10.1515/hc-2016-0190
Return to citation in text: [1] -
Kashani, E.; Pesyan, N. N.; Şahin, E. J. Chin. Chem. Soc. 2015, 62, 959–967. doi:10.1002/jccs.201500257
Return to citation in text: [1] -
Jiang, B.; Cao, L.-J.; Tu, S.-J.; Zheng, W.-R.; Yu, H.-Z. J. Comb. Chem. 2009, 11, 612–616. doi:10.1021/cc900038g
Return to citation in text: [1] -
Renard, A.; Lhomme, J.; Kotera, M. J. Org. Chem. 2002, 67, 1302–1307. doi:10.1021/jo016194y
Return to citation in text: [1] -
Lomlim, L.; Einsiedel, J.; Heinemann, F. W.; Meyer, K.; Gmeiner, P. J. Org. Chem. 2008, 73, 3608–3611. doi:10.1021/jo702573z
Return to citation in text: [1] -
Gao, X.; Li, Z.; Yang, W.; Liu, Y.; Chen, W.; Zhang, C.; Zheng, L.; Guo, H. Org. Biomol. Chem. 2017, 15, 5298–5307. doi:10.1039/c7ob01034f
Return to citation in text: [1] -
Liu, H.; Liu, Y.; Yuan, C.; Wang, G.-P.; Zhu, S.-F.; Wu, Y.; Wang, B.; Sun, Z.; Xiao, Y.; Zhou, Q.-L.; Guo, H. Org. Lett. 2016, 18, 1302–1305. doi:10.1021/acs.orglett.6b00239
Return to citation in text: [1] -
Zhao, H.-W.; Feng, N.-N.; Guo, J.-M.; Du, J.; Ding, W.-Q.; Wang, L.-R.; Song, X.-Q. J. Org. Chem. 2018, 83, 9291–9299. doi:10.1021/acs.joc.8b01268
Return to citation in text: [1] -
Gao, X.; Zhu, D.; Chen, Y.; Deng, H.; Jiang, F.; Wang, W.; Wu, Y.; Guo, H. Org. Lett. 2020, 22, 7158–7163. doi:10.1021/acs.orglett.0c02508
Return to citation in text: [1] -
Kotha, S.; Sreevani, G. Synthesis 2018, 50, 4883–4888. doi:10.1055/s-0037-1610238
Return to citation in text: [1] -
Soeiro, P. F.; Serrano, J. L.; Paixão, J. A.; Boto, R. E. F.; Silvestre, S.; Almeida, P. Synthesis 2020, 52, 2065–2072. doi:10.1055/s-0039-1690865
Return to citation in text: [1] -
Song, X.; Chang, J.; Zhu, Y.; Zhao, S.; Zhang, M. Synthesis 2019, 51, 899–906. doi:10.1055/s-0037-1609637
Return to citation in text: [1] -
Tábuas, B.; Cruz Barros, S.; Diogo, C.; Cavaleiro, C.; Sanches Silva, A. Toxins 2024, 16, 79. doi:10.3390/toxins16020079
Return to citation in text: [1] -
Al-Subaie, S. F.; Alowaifeer, A. M.; Mohamed, M. E. Foods 2022, 11, 3873. doi:10.3390/foods11233873
Return to citation in text: [1] -
Pomeroy, A. R.; Raper, C. Br. J. Pharmacol. 1971, 41, 683–690. doi:10.1111/j.1476-5381.1971.tb07076.x
Return to citation in text: [1] -
Jayawickreme, K.; Świstak, D.; Ozimek, E.; Reszczyńska, E.; Rysiak, A.; Makuch-Kocka, A.; Hanaka, A. Int. J. Mol. Sci. 2023, 24, 16972. doi:10.3390/ijms242316972
Return to citation in text: [1] -
Chen, T.; Mei, N.; Fu, P. P. J. Appl. Toxicol. 2010, 30, 183–196. doi:10.1002/jat.1504
Return to citation in text: [1] -
Glück, J.; Ebmeyer, J.; Waizenegger, J.; Luckert, C.; Braeuning, A.; Lampen, A.; Hessel-Pras, S. Toxicol. Lett. 2018, 295, S142. doi:10.1016/j.toxlet.2018.06.726
Return to citation in text: [1] -
Jank, B.; Rath, J. Trends Plant Sci. 2017, 22, 191–193. doi:10.1016/j.tplants.2017.01.002
Return to citation in text: [1] -
Wei, X.; Ruan, W.; Vrieling, K. Molecules 2021, 26, 1970. doi:10.3390/molecules26071970
Return to citation in text: [1] -
Reina, M.; Gonzalez-Coloma, A.; Gutierrez, C.; Cabrera, R.; Henriquez, J.; Villarroel, L. J. Nat. Prod. 1998, 61, 1418–1420. doi:10.1021/np980175a
Return to citation in text: [1] -
Singh, B.; Sahu, P. M.; Singh, S. Fitoterapia 2002, 73, 153–155. doi:10.1016/s0367-326x(02)00016-3
Return to citation in text: [1] -
Hoang, L. S.; Tran, M. H.; Lee, J. S.; To, D. C.; Nguyen, V. T.; Kim, J. A.; Lee, J. H.; Woo, M. H.; Min, B. S. Chem. Pharm. Bull. 2015, 63, 481–484. doi:10.1248/cpb.c14-00855
Return to citation in text: [1] -
Huang, S.; Zhou, X.-l.; Wang, C.-j.; Wang, Y.-s.; Xiao, F.; Shan, L.-h.; Guo, Z.-y.; Weng, J. Phytochemistry 2013, 93, 154–161. doi:10.1016/j.phytochem.2013.03.009
Return to citation in text: [1] -
Hua, L.; Chen, J.; Gao, K. Phytochem. Lett. 2012, 5, 541–544. doi:10.1016/j.phytol.2012.05.009
Return to citation in text: [1] -
Taylor, D. L.; Nash, R.; Fellows, L. E.; Kang, M. S.; Tyms, A. S. Antiviral Chem. Chemother. 1992, 3, 273–277. doi:10.1177/095632029200300504
Return to citation in text: [1] -
Benamar, H.; Tomassini, L.; Venditti, A.; Marouf, A.; Bennaceur, M.; Serafini, M.; Nicoletti, M. Nat. Prod. Res. 2017, 31, 1277–1285. doi:10.1080/14786419.2016.1242000
Return to citation in text: [1] -
Usuki, H.; Toyo-oka, M.; Kanzaki, H.; Okuda, T.; Nitoda, T. Bioorg. Med. Chem. 2009, 17, 7248–7253. doi:10.1016/j.bmc.2009.08.052
Return to citation in text: [1] -
Filatov, A. S.; Selivanov, S. I.; Shmakov, S. V.; Larina, A. G.; Boitsov, V. M.; Stepakov, A. V. Synthesis 2022, 54, 1803–1816. doi:10.1055/a-1700-3115
Return to citation in text: [1] [2] [3] [4] -
Kang, Y.; Seidel, D. Org. Lett. 2016, 18, 4277–4279. doi:10.1021/acs.orglett.6b02020
Return to citation in text: [1] -
Zhang, C.; Seidel, D. J. Am. Chem. Soc. 2010, 132, 1798–1799. doi:10.1021/ja910719x
Return to citation in text: [1] -
Zhang, C.; Das, D.; Seidel, D. Chem. Sci. 2011, 2, 233–236. doi:10.1039/c0sc00432d
Return to citation in text: [1] -
Filatov, A. S.; Khoroshilova, O. V.; Larina, A. G.; Boitsov, V. M.; Stepakov, A. V. Beilstein J. Org. Chem. 2022, 18, 769–780. doi:10.3762/bjoc.18.77
Return to citation in text: [1] -
Stepakov, A. V.; Pronina, Y. A.; Filatov, A. S.; Selivanov, S. I.; Kornev, A. A.; Kryukova, M. A.; Ponyaev, A. I.; Boitsov, V. M. Tetrahedron 2024, 151, 133792. doi:10.1016/j.tet.2023.133792
Return to citation in text: [1] -
Pronina, Y. A.; Viktorov, N. B.; Selivanov, S. I.; Kornev, A. A.; Ponyaev, A. I.; Boitsov, V. M.; Stepakov, A. V. Russ. J. Gen. Chem. 2024, 94, 804–823. doi:10.1134/s107036322404008x
Return to citation in text: [1] -
Kornev, A. A.; Shmakov, S. V.; Ponyaev, A. I.; Stepakov, A. V.; Boitsov, V. M. Pharmaceuticals 2024, 17, 1582. doi:10.3390/ph17121582
Return to citation in text: [1] -
Kornev, A. A.; Shmakov, S. V.; Gryschenko, A. M.; Pronina, Y. A.; Ponyaev, A. I.; Stepakov, A. V.; Boitsov, V. M. Int. J. Mol. Sci. 2025, 26, 3474. doi:10.3390/ijms26083474
Return to citation in text: [1] -
Aly, M. F.; El-Nagger, G. M.; El-Emary, T. I.; Grigg, R.; Metwally, S. A. M.; Sivagnanam, S. Tetrahedron 1994, 50, 895–906. doi:10.1016/s0040-4020(01)80804-8
Return to citation in text: [1] -
Strecker, A. Justus Liebigs Ann. Chem. 1862, 123, 363–365. doi:10.1002/jlac.18621230312
Return to citation in text: [1] -
Said, A. I.; El-Emary, T. I. RSC Adv. 2020, 10, 845–850. doi:10.1039/c9ra10039c
Return to citation in text: [1] -
Spackman, P. R.; Turner, M. J.; McKinnon, J. J.; Wolff, S. K.; Grimwood, D. J.; Jayatilaka, D.; Spackman, M. A. J. Appl. Crystallogr. 2021, 54, 1006–1011. doi:10.1107/s1600576721002910
Return to citation in text: [1] -
Lipinski, C. A.; Lombardo, F.; Dominy, B. W.; Feeney, P. J. Adv. Drug Delivery Rev. 2001, 46, 3–26. doi:10.1016/s0169-409x(00)00129-0
Return to citation in text: [1] -
Shmakov, S. V.; Latypova, D. K.; Shmakova, T. V.; Rubinshtein, A. A.; Chukin, M. V.; Zhuravskii, S. G.; Knyazev, N. A.; Stepakov, A. V.; Galagudza, M. M.; Boitsov, V. M. Int. J. Mol. Sci. 2022, 23, 10759. doi:10.3390/ijms231810759
Return to citation in text: [1] -
Latypova, D. K.; Shmakov, S. V.; Pechkovskaya, S. A.; Filatov, A. S.; Stepakov, A. V.; Knyazev, N. A.; Boitsov, V. M. Int. J. Mol. Sci. 2021, 22, 11997. doi:10.3390/ijms222111997
Return to citation in text: [1] -
Filatov, A. S.; Pronina, Y. A.; Selivanov, S. I.; Shmakov, S. V.; Uspenski, A. A.; Boitsov, V. M.; Stepakov, A. V. Int. J. Mol. Sci. 2022, 23, 13202. doi:10.3390/ijms232113202
Return to citation in text: [1] -
Pronina, Y.; Filatov, A.; Shmakov, S.; Selivanov, S.; Kryukova, M.; Spiridonova, D.; Ponyaev, A.; Stepakov, A.; Boitsov, V. J. Org. Chem. 2025, 90, 4926–4945. doi:10.1021/acs.joc.4c03183
Return to citation in text: [1] -
Tojkander, S.; Gateva, G.; Lappalainen, P. J. Cell Sci. 2012, 125, 1855–1864. doi:10.1242/jcs.098087
Return to citation in text: [1] -
Svitkina, T. Cold Spring Harbor Perspect. Biol. 2018, 10, a018267. doi:10.1101/cshperspect.a018267
Return to citation in text: [1] -
Bonello, T. T.; Stehn, J. R.; Gunning, P. W. Future Med. Chem. 2009, 1, 1311–1331. doi:10.4155/fmc.09.99
Return to citation in text: [1] -
Brayford, S.; Schevzov, G.; Vos, J.; Gunning, P. The Role of the Actin Cytoskeleton in Cancer and Its Potential Use as a Therapeutic Target. In The Cytoskeleton in Health and Disease; Schatten, H., Ed.; Springer: New York, NY, USA, 2015; pp 373–391. doi:10.1007/978-1-4939-2904-7_16
Return to citation in text: [1] -
Aseervatham, J. Biology (Basel, Switz.) 2020, 9, 385. doi:10.3390/biology9110385
Return to citation in text: [1] -
RCSB Protein Data Bank. https://www.rcsb.org/ (accessed Jan 29, 2026).
Return to citation in text: [1] -
Thomsen, R.; Christensen, M. H. J. Med. Chem. 2006, 49, 3315–3321. doi:10.1021/jm051197e
Return to citation in text: [1]
| 54. | Said, A. I.; El-Emary, T. I. RSC Adv. 2020, 10, 845–850. doi:10.1039/c9ra10039c |
| 43. | Filatov, A. S.; Selivanov, S. I.; Shmakov, S. V.; Larina, A. G.; Boitsov, V. M.; Stepakov, A. V. Synthesis 2022, 54, 1803–1816. doi:10.1055/a-1700-3115 |
| 43. | Filatov, A. S.; Selivanov, S. I.; Shmakov, S. V.; Larina, A. G.; Boitsov, V. M.; Stepakov, A. V. Synthesis 2022, 54, 1803–1816. doi:10.1055/a-1700-3115 |
| 1. | Baeyer, A. Justus Liebigs Ann. Chem. 1863, 127, 199–236. doi:10.1002/jlac.18631270214 |
| 2. | Baeyer, A. Justus Liebigs Ann. Chem. 1864, 130, 129–175. doi:10.1002/jlac.18641300202 |
| 7. | Taylor, S. N.; Marrazzo, J.; Batteiger, B. E.; Hook, E. W., III; Seña, A. C.; Long, J.; Wierzbicki, M. R.; Kwak, H.; Johnson, S. M.; Lawrence, K.; Mueller, J. N. Engl. J. Med. 2018, 379, 1835–1845. doi:10.1056/nejmoa1706988 |
| 35. | Reina, M.; Gonzalez-Coloma, A.; Gutierrez, C.; Cabrera, R.; Henriquez, J.; Villarroel, L. J. Nat. Prod. 1998, 61, 1418–1420. doi:10.1021/np980175a |
| 36. | Singh, B.; Sahu, P. M.; Singh, S. Fitoterapia 2002, 73, 153–155. doi:10.1016/s0367-326x(02)00016-3 |
| 66. | RCSB Protein Data Bank. https://www.rcsb.org/ (accessed Jan 29, 2026). |
| 67. | Thomsen, R.; Christensen, M. H. J. Med. Chem. 2006, 49, 3315–3321. doi:10.1021/jm051197e |
| 5. | Dox, A. W.; Yoder, L. J. Am. Chem. Soc. 1921, 43, 677–684. doi:10.1021/ja01436a040 |
| 6. | Dox, A. W.; Yoder, L. J. Am. Chem. Soc. 1921, 43, 1366–1370. doi:10.1021/ja01439a018 |
| 37. | Hoang, L. S.; Tran, M. H.; Lee, J. S.; To, D. C.; Nguyen, V. T.; Kim, J. A.; Lee, J. H.; Woo, M. H.; Min, B. S. Chem. Pharm. Bull. 2015, 63, 481–484. doi:10.1248/cpb.c14-00855 |
| 38. | Huang, S.; Zhou, X.-l.; Wang, C.-j.; Wang, Y.-s.; Xiao, F.; Shan, L.-h.; Guo, Z.-y.; Weng, J. Phytochemistry 2013, 93, 154–161. doi:10.1016/j.phytochem.2013.03.009 |
| 4. | López-Muñoz, F.; Ucha-Udabe, R.; Alamo, C. Neuropsychiatr. Dis. Treat. 2005, 1, 329–343. |
| 31. | Chen, T.; Mei, N.; Fu, P. P. J. Appl. Toxicol. 2010, 30, 183–196. doi:10.1002/jat.1504 |
| 32. | Glück, J.; Ebmeyer, J.; Waizenegger, J.; Luckert, C.; Braeuning, A.; Lampen, A.; Hessel-Pras, S. Toxicol. Lett. 2018, 295, S142. doi:10.1016/j.toxlet.2018.06.726 |
| 33. | Jank, B.; Rath, J. Trends Plant Sci. 2017, 22, 191–193. doi:10.1016/j.tplants.2017.01.002 |
| 61. | Tojkander, S.; Gateva, G.; Lappalainen, P. J. Cell Sci. 2012, 125, 1855–1864. doi:10.1242/jcs.098087 |
| 62. | Svitkina, T. Cold Spring Harbor Perspect. Biol. 2018, 10, a018267. doi:10.1101/cshperspect.a018267 |
| 34. | Wei, X.; Ruan, W.; Vrieling, K. Molecules 2021, 26, 1970. doi:10.3390/molecules26071970 |
| 63. | Bonello, T. T.; Stehn, J. R.; Gunning, P. W. Future Med. Chem. 2009, 1, 1311–1331. doi:10.4155/fmc.09.99 |
| 64. | Brayford, S.; Schevzov, G.; Vos, J.; Gunning, P. The Role of the Actin Cytoskeleton in Cancer and Its Potential Use as a Therapeutic Target. In The Cytoskeleton in Health and Disease; Schatten, H., Ed.; Springer: New York, NY, USA, 2015; pp 373–391. doi:10.1007/978-1-4939-2904-7_16 |
| 65. | Aseervatham, J. Biology (Basel, Switz.) 2020, 9, 385. doi:10.3390/biology9110385 |
| 24. | Kotha, S.; Sreevani, G. Synthesis 2018, 50, 4883–4888. doi:10.1055/s-0037-1610238 |
| 26. | Song, X.; Chang, J.; Zhu, Y.; Zhao, S.; Zhang, M. Synthesis 2019, 51, 899–906. doi:10.1055/s-0037-1609637 |
| 57. | Shmakov, S. V.; Latypova, D. K.; Shmakova, T. V.; Rubinshtein, A. A.; Chukin, M. V.; Zhuravskii, S. G.; Knyazev, N. A.; Stepakov, A. V.; Galagudza, M. M.; Boitsov, V. M. Int. J. Mol. Sci. 2022, 23, 10759. doi:10.3390/ijms231810759 |
| 20. | Gao, X.; Li, Z.; Yang, W.; Liu, Y.; Chen, W.; Zhang, C.; Zheng, L.; Guo, H. Org. Biomol. Chem. 2017, 15, 5298–5307. doi:10.1039/c7ob01034f |
| 21. | Liu, H.; Liu, Y.; Yuan, C.; Wang, G.-P.; Zhu, S.-F.; Wu, Y.; Wang, B.; Sun, Z.; Xiao, Y.; Zhou, Q.-L.; Guo, H. Org. Lett. 2016, 18, 1302–1305. doi:10.1021/acs.orglett.6b00239 |
| 22. | Zhao, H.-W.; Feng, N.-N.; Guo, J.-M.; Du, J.; Ding, W.-Q.; Wang, L.-R.; Song, X.-Q. J. Org. Chem. 2018, 83, 9291–9299. doi:10.1021/acs.joc.8b01268 |
| 23. | Gao, X.; Zhu, D.; Chen, Y.; Deng, H.; Jiang, F.; Wang, W.; Wu, Y.; Guo, H. Org. Lett. 2020, 22, 7158–7163. doi:10.1021/acs.orglett.0c02508 |
| 27. | Tábuas, B.; Cruz Barros, S.; Diogo, C.; Cavaleiro, C.; Sanches Silva, A. Toxins 2024, 16, 79. doi:10.3390/toxins16020079 |
| 28. | Al-Subaie, S. F.; Alowaifeer, A. M.; Mohamed, M. E. Foods 2022, 11, 3873. doi:10.3390/foods11233873 |
| 29. | Pomeroy, A. R.; Raper, C. Br. J. Pharmacol. 1971, 41, 683–690. doi:10.1111/j.1476-5381.1971.tb07076.x |
| 30. | Jayawickreme, K.; Świstak, D.; Ozimek, E.; Reszczyńska, E.; Rysiak, A.; Makuch-Kocka, A.; Hanaka, A. Int. J. Mol. Sci. 2023, 24, 16972. doi:10.3390/ijms242316972 |
| 58. | Latypova, D. K.; Shmakov, S. V.; Pechkovskaya, S. A.; Filatov, A. S.; Stepakov, A. V.; Knyazev, N. A.; Boitsov, V. M. Int. J. Mol. Sci. 2021, 22, 11997. doi:10.3390/ijms222111997 |
| 59. | Filatov, A. S.; Pronina, Y. A.; Selivanov, S. I.; Shmakov, S. V.; Uspenski, A. A.; Boitsov, V. M.; Stepakov, A. V. Int. J. Mol. Sci. 2022, 23, 13202. doi:10.3390/ijms232113202 |
| 60. | Pronina, Y.; Filatov, A.; Shmakov, S.; Selivanov, S.; Kryukova, M.; Spiridonova, D.; Ponyaev, A.; Stepakov, A.; Boitsov, V. J. Org. Chem. 2025, 90, 4926–4945. doi:10.1021/acs.joc.4c03183 |
| 13. | Huo, M.; Zhou, J.; Bai, L.; Xu, Q.; Zhou, Z.; Zhou, H.; Liang, G. Tetrahedron 2019, 75, 130752. doi:10.1016/j.tet.2019.130752 |
| 14. | Borisova, Y. G.; Raskil’dina, G. Z.; Zlotskii, S. S. Dokl. Chem. 2017, 476, 201–205. doi:10.1134/s0012500817090014 |
| 15. | Elinson, M. N.; Vereshchagin, A. N.; Korshunov, A. D.; Ryzhkov, F. V.; Egorov, M. P. Heterocycl. Commun. 2017, 23, 85–90. doi:10.1515/hc-2016-0190 |
| 16. | Kashani, E.; Pesyan, N. N.; Şahin, E. J. Chin. Chem. Soc. 2015, 62, 959–967. doi:10.1002/jccs.201500257 |
| 17. | Jiang, B.; Cao, L.-J.; Tu, S.-J.; Zheng, W.-R.; Yu, H.-Z. J. Comb. Chem. 2009, 11, 612–616. doi:10.1021/cc900038g |
| 18. | Renard, A.; Lhomme, J.; Kotera, M. J. Org. Chem. 2002, 67, 1302–1307. doi:10.1021/jo016194y |
| 19. | Lomlim, L.; Einsiedel, J.; Heinemann, F. W.; Meyer, K.; Gmeiner, P. J. Org. Chem. 2008, 73, 3608–3611. doi:10.1021/jo702573z |
| 55. | Spackman, P. R.; Turner, M. J.; McKinnon, J. J.; Wolff, S. K.; Grimwood, D. J.; Jayatilaka, D.; Spackman, M. A. J. Appl. Crystallogr. 2021, 54, 1006–1011. doi:10.1107/s1600576721002910 |
| 7. | Taylor, S. N.; Marrazzo, J.; Batteiger, B. E.; Hook, E. W., III; Seña, A. C.; Long, J.; Wierzbicki, M. R.; Kwak, H.; Johnson, S. M.; Lawrence, K.; Mueller, J. N. Engl. J. Med. 2018, 379, 1835–1845. doi:10.1056/nejmoa1706988 |
| 8. | Damião Gouveia, A. C.; Unemo, M.; Jensen, J. S. J. Antimicrob. Chemother. 2018, 73, 1291–1294. doi:10.1093/jac/dky022 |
| 9. | King, S. B.; Stratford, E. S.; Craig, C. R.; Fifer, E. K. Pharm. Res. 1995, 12, 1240–1243. doi:10.1023/a:1016236615559 |
| 10. | Galati, E. M.; Monforte, M. T.; Miceli, N.; Raneri, E. Farmaco 2001, 56, 459–461. doi:10.1016/s0014-827x(01)01062-x |
| 11. | Chan, P. F.; Srikannathasan, V.; Huang, J.; Cui, H.; Fosberry, A. P.; Gu, M.; Hann, M. M.; Hibbs, M.; Homes, P.; Ingraham, K.; Pizzollo, J.; Shen, C.; Shillings, A. J.; Spitzfaden, C. E.; Tanner, R.; Theobald, A. J.; Stavenger, R. A.; Bax, B. D.; Gwynn, M. N. Nat. Commun. 2015, 6, 10048. doi:10.1038/ncomms10048 |
| 12. | Scott, G. K.; Benz, C. C. Inhibition of proline catabolism for the treatment of cancer and other therapeutic applications. WO Pat. Appl. WO2016077632A2, May 19, 2016. |
| 25. | Soeiro, P. F.; Serrano, J. L.; Paixão, J. A.; Boto, R. E. F.; Silvestre, S.; Almeida, P. Synthesis 2020, 52, 2065–2072. doi:10.1055/s-0039-1690865 |
| 56. | Lipinski, C. A.; Lombardo, F.; Dominy, B. W.; Feeney, P. J. Adv. Drug Delivery Rev. 2001, 46, 3–26. doi:10.1016/s0169-409x(00)00129-0 |
| 41. | Benamar, H.; Tomassini, L.; Venditti, A.; Marouf, A.; Bennaceur, M.; Serafini, M.; Nicoletti, M. Nat. Prod. Res. 2017, 31, 1277–1285. doi:10.1080/14786419.2016.1242000 |
| 39. | Hua, L.; Chen, J.; Gao, K. Phytochem. Lett. 2012, 5, 541–544. doi:10.1016/j.phytol.2012.05.009 |
| 40. | Taylor, D. L.; Nash, R.; Fellows, L. E.; Kang, M. S.; Tyms, A. S. Antiviral Chem. Chemother. 1992, 3, 273–277. doi:10.1177/095632029200300504 |
| 52. | Aly, M. F.; El-Nagger, G. M.; El-Emary, T. I.; Grigg, R.; Metwally, S. A. M.; Sivagnanam, S. Tetrahedron 1994, 50, 895–906. doi:10.1016/s0040-4020(01)80804-8 |
| 53. | Strecker, A. Justus Liebigs Ann. Chem. 1862, 123, 363–365. doi:10.1002/jlac.18621230312 |
| 47. | Filatov, A. S.; Khoroshilova, O. V.; Larina, A. G.; Boitsov, V. M.; Stepakov, A. V. Beilstein J. Org. Chem. 2022, 18, 769–780. doi:10.3762/bjoc.18.77 |
| 48. | Stepakov, A. V.; Pronina, Y. A.; Filatov, A. S.; Selivanov, S. I.; Kornev, A. A.; Kryukova, M. A.; Ponyaev, A. I.; Boitsov, V. M. Tetrahedron 2024, 151, 133792. doi:10.1016/j.tet.2023.133792 |
| 49. | Pronina, Y. A.; Viktorov, N. B.; Selivanov, S. I.; Kornev, A. A.; Ponyaev, A. I.; Boitsov, V. M.; Stepakov, A. V. Russ. J. Gen. Chem. 2024, 94, 804–823. doi:10.1134/s107036322404008x |
| 50. | Kornev, A. A.; Shmakov, S. V.; Ponyaev, A. I.; Stepakov, A. V.; Boitsov, V. M. Pharmaceuticals 2024, 17, 1582. doi:10.3390/ph17121582 |
| 51. | Kornev, A. A.; Shmakov, S. V.; Gryschenko, A. M.; Pronina, Y. A.; Ponyaev, A. I.; Stepakov, A. V.; Boitsov, V. M. Int. J. Mol. Sci. 2025, 26, 3474. doi:10.3390/ijms26083474 |
| 43. | Filatov, A. S.; Selivanov, S. I.; Shmakov, S. V.; Larina, A. G.; Boitsov, V. M.; Stepakov, A. V. Synthesis 2022, 54, 1803–1816. doi:10.1055/a-1700-3115 |
| 44. | Kang, Y.; Seidel, D. Org. Lett. 2016, 18, 4277–4279. doi:10.1021/acs.orglett.6b02020 |
| 45. | Zhang, C.; Seidel, D. J. Am. Chem. Soc. 2010, 132, 1798–1799. doi:10.1021/ja910719x |
| 46. | Zhang, C.; Das, D.; Seidel, D. Chem. Sci. 2011, 2, 233–236. doi:10.1039/c0sc00432d |
| 42. | Usuki, H.; Toyo-oka, M.; Kanzaki, H.; Okuda, T.; Nitoda, T. Bioorg. Med. Chem. 2009, 17, 7248–7253. doi:10.1016/j.bmc.2009.08.052 |
| 43. | Filatov, A. S.; Selivanov, S. I.; Shmakov, S. V.; Larina, A. G.; Boitsov, V. M.; Stepakov, A. V. Synthesis 2022, 54, 1803–1816. doi:10.1055/a-1700-3115 |
© 2026 Puzyrkov et al.; licensee Beilstein-Institut.
This is an open access article licensed under the terms of the Beilstein-Institut Open Access License Agreement (https://www.beilstein-journals.org/bjoc/terms), which is identical to the Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0). The reuse of material under this license requires that the author(s), source and license are credited. Third-party material in this article could be subject to other licenses (typically indicated in the credit line), and in this case, users are required to obtain permission from the license holder to reuse the material.