Abstract
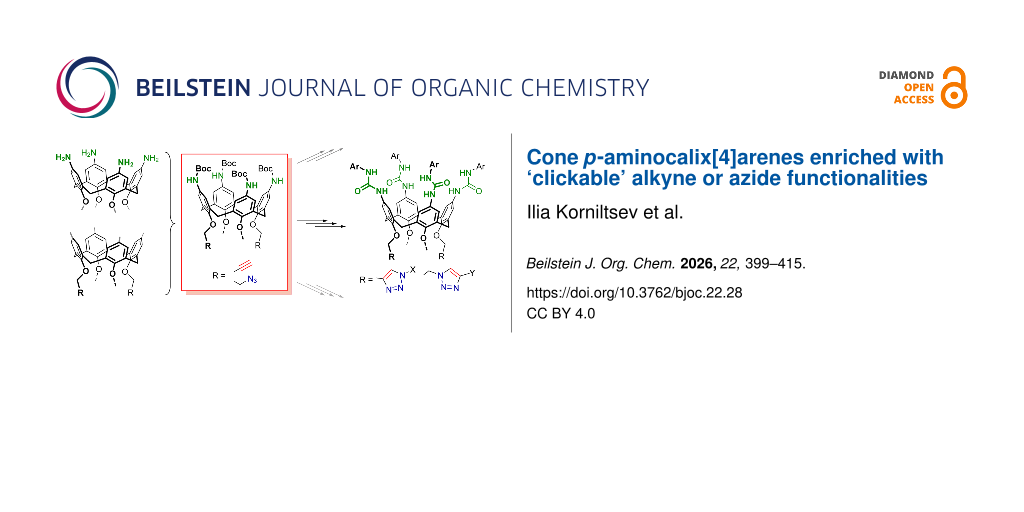
Efficient approaches have been developed for the synthesis of heteromultifunctional cone calix[4]arenes containing four amino groups at the wide rim and one, two or four propargyl or 2-azidoethyl groups at the narrow rim of the macrocycle, which can be used for expanding functionalization of the calixarene core in the well-known amine acylation (or similar reactions) and CuAAC ‘click’ reactions. Two different strategies were implemented to obtain propargylated and 2-azidoethylated p-aminocalixarenes. In the case of propargylated calixarenes, sterically crowding silyl protection was introduced into the alkyne groups of p-tert-butylcalix[4]arene (multiple) propargyl ethers, and the resulting compounds were ipso-nitrated followed by reduction of the nitro groups. To prepare 2-azidoethylated macrocycles, the ipso-nitration/reduction sequence was applied to p-tert-butylcalix[4]arenes containing 2-tosyloxyethyl groups at the narrow rims followed by replacement of the tosyloxy groups with azide ones. In all cases, p-aminocalix[4]arenes were obtained as the readily cleavable tert-butoxycarbonyl (Boc) derivatives, which was crucial for certain transformation and purification steps. To confirm the functionalization capabilities of the five obtained multifunctional calixarenes, they were reacted with excess benzyl azide or phenylacetylene, taken as representatives, under copper(I) catalysis, resulting in the narrow-rim triazolated macrocycles. By removing the Boc protecting groups and involving the free amino groups in reactions with p-tolyl isocyanate, a series of narrow-rim triazolated tetraureacalix[4]arenes was obtained. Examination of the 1H NMR spectra of the tetraureas in CDCl3 showed that in most cases triazole heterocycles do not intervene the formation of homo- and heterodimeric capsules by these compounds. Thus, considering the synthetic value of CuAAC and amine transformations, p-aminocalix[4]arenes enriched with alkyne or azide functionalities can be readily used as multifunctional platforms to obtain even higher functionalized macrocycles. As an example, they can be used for the preparation of sophisticated supramolecular assemblies with homo- or heterodimeric calixarene cores and virtually any functional units attached to them via triazole groups.

Graphical Abstract
Introduction
Considered generally as multifunctional molecular cores, calixarenes in their native forms as cyclic oligophenols actually possess just limited functionalization capabilities unless synthetically more valuable groups are introduced into their structures. Due to the synthetic approaches well-developed in the first decades of calixarene chemistry, these groups can be arranged in a certain number and order at the phenolic oxygen atoms and/or in the aromatic p-positions to them, which form respectively the narrow (lower) and wide (upper) rims of the calixarene macrocycle when it possesses a cone shape. Both the core functionalization and shape control strategies are most developed for calix[4]arene cores, and that is exactly why these cyclic tetramers may be regarded as virtually universal molecular platforms for the construction of unique multifunctional structures capable of diverse supramolecular interactions [1-4].
Amino groups attached directly to all four positions of the wide rim of the cores are among the most important functionalities which can be introduced into calix[4]arene structures either through a wide-rim exhaustive nitration followed by reduction of the nitro groups [5-13], or through azo coupling followed by cleavage of the calixarene azo compounds thus formed [14-17]. (Multi)calix[4]arenes having (dialkyl)amino groups at the wide rims are soluble in aqueous media at physiological pHs and may be used in protein sensing [18], DNA binding/recognition [19-23], and cell transfection [24-26]. Their water-soluble guanidinium derivatives are also biocompatible compounds capable of interacting with proteins, nucleic acids, and polysaccharides [27-31], and may also mimic phosphodiesterase functions [32-35]. Beyond biorelevant structures, p-aminocalix[4]arenes serve as preorganizing platforms that may graft together four functional/receptor units, thus drastically improving their supramolecular interaction capabilities. This property of p-aminocalix[4]arenes has been widely utilized in constructing f-element-targeted extractants for nuclear waste treatment, having four carbamoylmethylphosphine oxide groups introduced to the macrocyclic core through simple acylation of p-aminocalix[4]arenes with the respective phosphorous-containing activated esters. Such grafting of well-known receptor units onto a common platform has led to substantial amplification of the efficiency of lanthanide and actinide extraction from acidic media by carbamoylmethylphosphine oxides along with enrichment of the extraction selectivity [36-45]. Even more impressive is the effect from grafting of four urea groups at the wide rims of calix[4]arene macrocycles achieved by reacting p-aminocalix[4]arenes with aryl- or arylsulfonyl isocyanates. In aprotic media, these compounds, referred to as ‘tetraureacalix[4]arenes’, assemble into well-defined homo- or heterodimeric capsules, which are held together by cyclic belts of hydrogen bonds between interpenetrated urea groups from two molecules and are able to include a single neutral molecule or a trialkylammonium cation inside the joint cavity formed by two cone calix[4]arene macrocycles [46-54]. This phenomenon has been thoroughly investigated, including the effects from substituents in the urea groups and/or at the calixarene narrow rim upon the thermodynamic and kinetic stability of the capsules and the guest exchange processes [55-60]. This has made dimerization of tetraureacalix[4]arenes one of the most explored self-organization processes in calixarene supramolecular chemistry, which has been used in the template synthesis of calixarene-based multi(macrocycles) and multi(catenanes) having impressive topology [61-69].
Over the past two decades, azide and alkyne groups have also appeared among the most synthetically valuable functionalities introduced into calixarene cores due to the ease of their conversion into diverse 1,4-disubstituted 1,2,3-triazole units under CuAAC (copper(I)-catalyzed azide–alkyne cycloaddition) ‘click’ conditions [70-73]. The first implementation of the CuAAC approach for calixarene modification was published shortly after its introduction into chemistry in general, and used cone calix[4]arenes having four propargyl or 2-azidoethyl groups at their narrow rims as CuAAC substrates [74]. Since then, hundreds of publications have reported the application of the CuAAC reaction for the introduction of diverse biorelevant, receptor or sensory functionalities into the structures of calixarenes and related macrocycles [75-78]. Many of them relied on the introduction of propargyl or azidoalkyl groups to the phenolic oxygen atoms for synthesizing the CuAAC-ready calixarene cores. Within this area of research, our studies on the CuAAC reactions applied to cone propargylated and 2-azidoethylated calix[4]arenes revealed their enhanced efficiency, resulting in the preferential formation of exhaustively triazolated macrocycles over mixed triazolated/propargylated or triazolated/2-azidoethylated ones [79,80]. Furthermore, it has been proved that triazolated calixarenes derived from propargylated and 2-azidoethylated precursors possess drastically different complexation abilities towards transition-metal cations [81-84] including the formation of unique inherently dinuclear iridium(III) complexes with cone calix[4]arenes having pairs of 2-(4-aryltriazol-1-yl)ethyl groups at their narrow rims [85]. Regarding the synthetic capabilities of propargyl and 2-azidoethyl groups residing the calixarene core, we have established a series of triazolated calix[4]semitubes in which two or three calix[4]arene macrocycles are connected to each other by pairs of triazole units formed via two-fold CuAAC macrocyclizations [86,87]. We have also developed a straightforward approach to the functionalization of water-soluble p-sulfonatocalix[4]arenes by their enrichment with propargyl groups at the narrow rims followed by CuAAC reactions with various azides [88].
Inspired by the above benefits brought to the calixarene structures by amino groups, the power of CuAAC reactions in building multifunctional architectures, and following the pioneering work on bifunctional “Janus” calix[4]arenes joining propargyl groups and nitro/amino/azide groups in the structures [9], we herein report on the synthesis approaches to a series of cone p-aminocalix[4]arenes having propargyl or 2-azidoethyl groups at the narrow rims (Figure 1). The newly synthesized compounds may be thus treated as novel building blocks in calixarene chemistry capable of step-wise orthogonal transformations into various multifunctional structures and sophisticated supramolecular systems.
![[1860-5397-22-28-1]](/bjoc/content/figures/1860-5397-22-28-1.png?scale=2.0&max-width=1024&background=FFFFFF)
Figure 1: General concept of building multifunctional calix[4]arenes by joining propargyl/2-azidoethyl and p-amino groups on a common platform.
Figure 1: General concept of building multifunctional calix[4]arenes by joining propargyl/2-azidoethyl and p-...
Results and Discussion
In the majority of cases, the introduction of four amino groups into the calix[4]arene wide rim requires exhaustive (ipso-)nitration of the core followed by reduction of the resulting p-nitrocalix[4]arene. Notably, the alternative strategy of utilizing an azo coupling/cleavage sequence has only been applied thus far for the wide-rim modification of narrow-rim unsubstituted calixarenes. Since the exhaustive nitration and reduction steps proceed under relatively drastic conditions, which are poorly compatible with propargyl or 2-azidoethyl functionalities residing at the calixarene narrow rims, these ‘simple’ transformations are not applicable to the available propargylated/2-azidoethylated p-tert-butyl- or p-H-calix[4]arenes. Therefore, the multistep reaction sequences presented below have been developed for the preparation of the targeted multifunctionalized calixarenes.
Propargylated p-aminocalix[4]arenes
Following the published example [9], for the preparation of propargylated p-aminocalix[4]arenes a three-step synthesis strategy was selected, which involved protection of the calixarene propargyl ethers by tert-butyldimethyl-silylation followed by the calixarene wide-rim exhaustive nitration and reduction of the nitro groups. The readily available cone calix[4]arenes 1–5 [89-92] bearing up to four propargyl groups at their narrow rims and n-propyl groups completing the substitution patterns were selected as the starting materials to enable further preparation of the respective mono-, di- and tetrapropargylated calix[4]arene tetraamines and their derivatives.
The silylation of propargyl ethers 1–5 was first attempted using tert-butyldimethylsilyl chloride (TBSCl) and the commercially available lithium bis(trimethylsilyl)amide (LiHMDS), but the latter was found inconvenient for routine syntheses due to high moisture sensitivity which prevented reproducibility of the reaction conditions. To overcome this difficulty, the LiHMDS solution was instead prepared in situ by reacting bis(trimethylsilyl)amine (HMDS) with n-butyllithium in THF immediately before the addition of a propargylated calixarene followed by TBSCl. This allowed us to improve the yield of the known calix[4]arene 6 [9] having four silylated propargyl groups at the narrow rim, and to obtain the respective persilylated propargyl ethers 7–10 from precursors 2–5 (Scheme 1).
![[1860-5397-22-28-i1]](/bjoc/content/inline/1860-5397-22-28-i1.png?scale=2.0&max-width=1024&background=FFFFFF)
Scheme 1: Silylation of the calixarene propargyl ethers. Conditions: i) LiHMDS (HMDS + n-BuLi), TBSCl, THF, −95 °C → rt.
Scheme 1: Silylation of the calixarene propargyl ethers. Conditions: i) LiHMDS (HMDS + n-BuLi), TBSCl, THF, −...
Expectedly, (ipso-)nitration of calixarenes 6–10 by mixtures of fuming nitric acid and glacial acetic acid in dichloromethane solutions could potentially be complicated by both the incomplete conversion of the starting materials and also by formation of side products, due to acid-promoted cleavage and undesired transformations of the TBS-protected propargyl groups and/or due to unwanted oxidation/nitration of the calixarene core [93]. Indeed, nitration of calixarene 6 has been reported to furnish the desired wide-rim exhaustively nitrated calix[4]arene 11 having four TBS-protected propargyl groups at the narrow rim in only moderate yield of 37% [9]. In our hands, according to the NMR spectra of the reaction mixture, under the reported conditions (>30 equiv of fuming HNO3 per calixarene aromatic unit in a 1:1 dichloromethane/acetic acid mixture, from 0 °C → rt, overnight) the nitration of calixarene 6 resulted in a mixture of exhaustively nitrated product 11, partially nitrated calixarenes having nitro and tert-butyl groups at the wide rims, and a large amount of other calixarene side product(s) having broadened and non-interpretable NMR spectra. Upon tuning the reaction conditions, it was found that a more complete nitration of calixarene 6 along with some suppression of the undesired side reactions could be achieved by decreasing the nitric acid excess (to ≈10 equiv per calixarene aromatic unit) and its concentration in dichloromethane (to ≈1 M), reducing the content of acetic acid in the mixture (to 1 mol per mol of HNO3) and extending the room-temperature reaction time to 72 h. Yet, under these conditions, the desired calix[4]arene 11 was obtained in only 31% yield (Scheme 2), and further variations in reagent excess/concentration and reaction time did not significantly improve the yield. Also an attempt to obtain calixarene 11 by nitration of the silylated p-H-calix[4]arene tetrapropargyl ether 7 failed, as no complete conversion of the starting calixarene could be achieved likely due to a slower nitration of unsubstituted calixarene aromatic units in comparison with the ipso-nitration of the corresponding tert-butylated ones. Even larger amounts of side products were formed, thus confirming the involvement of the tightly arranged narrow-rim substituents in calixarene 6 and 7 in undesired side processes under nitration conditions.
![[1860-5397-22-28-i2]](/bjoc/content/inline/1860-5397-22-28-i2.png?scale=2.0&max-width=1024&background=FFFFFF)
Scheme 2: Nitration of calixarenes 6, 8–10. Conditions: i) HNO3 (fuming, 10 equiv per calixarene aromatic unit, c ≈ 1 M), AcOH, dichloromethane, rt, 72 h; ii) HNO3 (fuming, 2.5 equiv per calixarene aromatic unit, c ≈ 0.2 M), AcOH, dichloromethane, rt, 12 h; iii) HNO3 (fuming, 2.5 equiv per calixarene aromatic unit, c ≈ 1 M), AcOH, dichloromethane, rt, 12 h; iv) HNO3 (fuming, 10 equiv per calixarene aromatic unit, c ≈ 2.5 M), AcOH, dichloromethane, rt, 48 h; v) HNO3 (fuming, 2.5 equiv per calixarene aromatic unit, c ≈ 1 M), AcOH, dichloromethane, rt, 48 h.
Scheme 2: Nitration of calixarenes 6, 8–10. Conditions: i) HNO3 (fuming, 10 equiv per calixarene aromatic uni...
In line with this, compounds 8–10 having less sterically hindered narrow-rim substitution patterns were found to be more resistant to side reactions during wide-rim nitration as well as to the target exhaustive nitration itself. These reactions required a higher nitric acid concentration of ≈2.5 M to complete the process and to obtain the p-nitrocalix[4]arenes 15 and 17 in good yield (Scheme 2). Upon further fine-tuning the reaction conditions it was found that some of the partially nitrated products could also be successfully prepared by using smaller amounts of the nitrating agent. Thus, the room-temperature nitration of the silylated p-tert-butylcalix[4]arene ether 8 using 2.5 equiv of HNO3 per calixarene aromatic unit resulted in di- or trinitrated macrocycles 12 and 14 as the major products, when ≈0.2 and ≈1 M nitric acid concentrations were used. Similarly, the wide-rim unsubstituted calixarene 9 was successfully converted into the dinitro-derivative 13 using the dichloromethane-diluted nitric acid/acetic acid mixture. Also, it was found that calixarene 10 could be converted to the trinitrated product 16 under the same conditions as for the synthesis of calixarene 14, with only the reaction time increased (Scheme 2).
The 1D NMR spectra for the dinitrocalix[4]arenes 12 and 13 provided no definitive data on the substitution patterns at their wide-rims except for the presence of two nitro groups in the distal aromatic units. To localize the nitro groups in the structures of calixarenes 12 and 13, 1H,13C HMBC spectra were acquired (Figure 2), which clearly showed that the nitro groups are attached to the propylated aromatic units of the macrocycle in both cases. Notably, the correlations showed that the signals from the nitrated calixarene aromatic units appeared upfield-shifted in the 1H NMR spectra with respect to those from the aromatic units bearing no electron-withdrawing groups. This uncommon signal position may indicate a specific time-averaged conformation of the calixarene core in compounds 12 and 13 due to the presence of the bulky TBS-protected propargyl groups in their structures, in which the nitrated aromatic units experience an additional shielding. This may be also responsible for the selectivity of the two-fold nitration of compounds 8 and 9 leading to calixarenes 12 and 13 rather than to their isomers containing nitro groups in the propargylated aromatic units of the macrocycles.
![[1860-5397-22-28-2]](/bjoc/content/figures/1860-5397-22-28-2.png?scale=2.0&max-width=1024&background=FFFFFF)
Figure 2: Parts of 1H,13C HMBC spectra of calixarenes 12 (a) and 13 (b) recorded in CDCl3 solutions at 600 MHz. Red lines show the key correlations between signals from the calixarene narrow-rim substituents and aromatic units.
Figure 2: Parts of 1H,13C HMBC spectra of calixarenes 12 (a) and 13 (b) recorded in CDCl3 solutions at 600 MH...
There was no doubt in establishing the structure of the trinitrated calix[4]arene 14, as its NMR spectra contained a single set of resonances from propyl groups and two sets of those from the TBS-protected propargyl groups, which indicated clearly the symmetry plane passing through the propargylated aromatic units of the calixarene. Assuming a step-wise nitration of calixarene 8, the dinitrated calixarene 12 must be the immediate precursor of compound 14. The presence of the nitro groups in both of the propylated aromatic units of this calixarene can only be derived from calixarene 12, which is additional evidence for its structure.
The structure of trinitrated calix[4]arene 16 having a single TBS-protected propargyl group at the narrow rim was unambiguously established from X-ray diffraction data. Suitable crystals were collected upon slow evaporation of a dichloromethane/methanol solution of compound 16. Similarly, single crystals of the exhaustively nitrated calix[4]arene 15 having two TBS-protected propargyl groups at the narrow rim were collected, and the molecular structure of this compound was also established (Figure 3) [94]. The results showed clearly, that in calixarene 16 all three propylated aromatic units were nitrated, and the propargylated one still contained a tert-butyl group. Thus, within a step-wise nitration paradigm, the narrow-rim propargylated aromatic units are more stable against nitration when compared to the propylated aromatic units of the calixarene core. This correlates well with the above selectivity of nitration observed for calixarenes 8 and 9 and may be tentatively explained by an electron-withdrawing effect of the propargyl groups.
![[1860-5397-22-28-3]](/bjoc/content/figures/1860-5397-22-28-3.png?scale=2.0&max-width=1024&background=FFFFFF)
Figure 3: Molecular structures of partially nitrated calixarene 16 (a) and exhaustively nitrated calixarene 15 (b). Two projections shown in each case and thermal ellipsoids are drawn at a 50% probability level.
Figure 3: Molecular structures of partially nitrated calixarene 16 (a) and exhaustively nitrated calixarene 15...
However, in the molecular structures of calixarenes 15 and 16, another feature was observed: in both cases the calixarene aromatic units containing the TBS-protected propargyl groups (and the opposed one in the case of compound 16) are extremely flattened, while the other aromatic units are pinched and bring the two nitro groups into very close contact. Similar effects from the TBS-protected propargyl groups may be also expected in calixarenes 6–10 and their partially nitrated products. Thus complex conformational transitions in the calixarene cores may also interfere with the reaction, and, in particular, may hamper the exhaustive nitration of calixarenes 6 and 7 each bearing four TBS-protected propargyl groups at their narrow rims.
The formation of the partially nitrated calix[4]arenes 12–14 and 16 as major reaction products described above was quite unexpected, and these compounds were not used further in this study which was more concerned with calix[4]arenes having four equal substituents at the wide rims. But surely, the partially nitrated calix[4]arenes bearing protected propargyl groups in their structures may be further explored as multifunctional cores capable of other potential applications in calixarene chemistry.
TBS units attached to the propargyl groups of calixarenes appeared to be not bulky enough to protect the alkyne from a Raney-Ni-catalyzed reduction by gaseous hydrogen, as a trial hydrogenation of the nitrated/propargylated calix[4]arene 11 resulted in only trace amounts of the desired p-aminocalix[4]arene 18 among numerous other reaction products. Due to this, a homogeneous reduction using tin(II) chloride in ethanol was used after fine-tuning of the published reaction conditions [9]: the reduction was conducted by gentle heating of a mixture of calixarene 11, SnCl2·2H2O, aqueous HCl and ethanol at 70 °C (instead of prolonged boiling of the reaction mixture) in a closed vessel to prevent the product oxidation. At the work-up step sodium hydroxide was replaced with potassium hydroxide which provided better water solubility of inorganic salts for their extractive removal. As a result, tetraamine 18 having four TBS-protected propargyl groups at the narrow rim was obtained in substantially improved yield and the nitrated calixarenes 15 and 17 were similarly reduced to the corresponding tetraamines 19 and 20 (Scheme 3).
![[1860-5397-22-28-i3]](/bjoc/content/inline/1860-5397-22-28-i3.png?scale=2.0&max-width=1024&background=FFFFFF)
Scheme 3: Reduction of calixarenes 11, 15 and 17. Conditions: i) SnCl2·2H2O, HCl, EtOH, H2O, 70 °C, then KOH, H2O.
Scheme 3: Reduction of calixarenes 11, 15 and 17. Conditions: i) SnCl2·2H2O, HCl, EtOH, H2O, 70 °C, then KOH,...
Chemical modifications of propargylated p-aminocalix[4]arenes may be performed by the amine acylation followed by the CuAAC involving the propargyl groups, or vice versa. Of these two possibilities, the triazole synthesis conducted prior to the target acylation of amines seems to be more convenient, as the acylation may proceed under milder conditions than the CuAAC reaction. Even more important, the amine-derived functional units may interfere or affect the cycloaddition by, for instance, binding copper cations or counter anions, and thus their presence in the structures of calixarenes at the CuAAC step must be avoided. On the other hand, the unmodified amines may also contribute to undesired Cu(I) stabilization and complicate the CuAAC work up in acidic conditions, so the amino groups in calixarenes 18–20 must be deactivated prior to the cycloaddition. Following the above reasons, a two-step replacement of protecting groups in calixarenes 18–20 was performed. First, the amino groups were acylated with di-tert-butyl dicarbonate (Boc2O) and next the TBS-protecting groups were removed from the acetylene units in the Boc-protected tetraamines 21–23 (Scheme 4).
![[1860-5397-22-28-i4]](/bjoc/content/inline/1860-5397-22-28-i4.png?scale=2.0&max-width=1024&background=FFFFFF)
Scheme 4: Protecting group replacement in propargylated p-aminocalix[4]arenes. Conditions: i) Boc2O, Et3N, dichloromethane, rt; ii) H2O, n-Bu4NF·3H2O, THF, rt; iii) H2O, n-Bu4NF·3H2O, THF, 50 °C.
Scheme 4: Protecting group replacement in propargylated p-aminocalix[4]arenes. Conditions: i) Boc2O, Et3N, di...
For the removal of the TBS groups, instead of using an equivalent amount of n-Bu4NF, it was used as a catalyst in a water/THF mixture, which showed excellent efficiency for the room-temperature preparation of the tetrapropargyl ether 24 from its silylated precursor 21. Surprisingly, attempts to apply these conditions to a complete removal of TBS groups in the less sterically hindered propargyl ethers 22 and 23 failed, and large amounts of the starting materials (as well as the partially desilylated product in the case of calixarene 22) remained in the reaction mixtures even after their stirring at room temperature for 72 h. In the case of calixarene 22, heating of the mixture at 50 °C for 48 h was enough to complete the process and p-aminocalix[4]arene 25 having two propargyl groups at the narrow rim was obtained in 97% yield. Still, the deprotection of calixarene 23 to the desired propargyl ether 26 was far from complete even under heating, which significantly reduced the yield of this compound (Scheme 4).
The difference in the efficiency of catalytic removal of the TBS groups observed for calixarenes 21–23 may be due to a reaction rate enhancement upon increasing the number of TBS-protected propargyl groups attached to the same calixarene core. Tentatively, this rate enhancement may be due to entrapment of the catalytic F– and/or water molecule(s) inside a pocket formed by the four TBS-protected propargyl groups in calixarene 21, which thus may be responsible for an autocatalytic process. Of course, this phenomenon needs to be studied in further detail.
2-Azidoethylated p-aminocalix[4]arenes
Due to the instability of azide groups in the presence of reducing agents, a synthesis strategy similar to that described above for the preparation of calixarenes 24–26 involving nitration of calix[4]arenes followed by selective reduction of the nitro groups could not be implemented for the preparation of p-aminocalix[4]arenes bearing 2-azidoethyl groups at their narrow rims. To overcome this, calixarenes 27 [95,96] and 28 [97] decorated with four or two 2-tosyloxyethyl groups which are precursors to the desired 2-azidoethyl groups, were subjected to a nitration/reduction sequence as shown in Scheme 5.
![[1860-5397-22-28-i5]](/bjoc/content/inline/1860-5397-22-28-i5.png?scale=2.0&max-width=1024&background=FFFFFF)
Scheme 5: Synthesis of 2-azidoethylated p-aminocalix[4]arenes. Conditions: i) HNO3 (fuming, 10 equiv per calixarene aromatic unit, c ≈ 2.5 M), AcOH, dichloromethane, rt, 48 h; ii) FeSO4·7H2O, NH4Cl, Zn, EtOH, THF, H2O, 50 °C; iii) Boc2O, Et3N, dichloromethane, rt; iv) NaN3, DMF, 60 °C.
Scheme 5: Synthesis of 2-azidoethylated p-aminocalix[4]arenes. Conditions: i) HNO3 (fuming, 10 equiv per cali...
The ipso-nitration step proceeded smoothly in both cases under the optimized conditions, and the wide-rim pernitrated tetra- and ditosylates 29 and 30 were obtained in good yield. However, the subsequent reduction step was expectedly complicated. On the one hand, even partial reduction of the tosyl groups in calixarenes 29 and 30 might result in poisoning of a metal catalyst, thus hampering or preventing a catalytic hydrogenation of these compounds. On the other hand, in calixarenes 31 and 32, which are immediate products of the selective reduction of the nitro groups in compounds 29 and 30 under neutral or basic conditions, the free amino groups may be alkylated by a neighboring tosylate-containing molecule, resulting in undesired polymeric/oligomeric substances. To avoid this, the reduction must be conducted in acidic medium, and no strong base must be applied during the work-up step. Thus, reduction of calixarenes 29 and 30 with tin(II) chloride, similar to the preparation of propargylated calix[4]arene tetraamines 18–20, proceeds under acidic conditions, but it has limited applicability as a complete removal of a large amount of inorganic by-products could only be achieved using alkaline solutions. This causes the unwanted self-conversions of free tetraamines 31 and 32. Reduction of nitro compounds with activated iron generated from Zn and FeSO4 in the presence of NH4Cl [98] was found to be a good alternative to the above tin(II) chloride reduction. Under these conditions, p-aminocalixarenes 31 and 32 were successfully prepared from the nitrated precursors 29 and 30 using simple filtration to remove the inorganic by-product components from the reaction mixtures. Although the free amines 31 and 32 were pure enough according to their 1H NMR data, these compounds were not fully characterized in order to avoid the risk of self-alkylation of the amines during their storage. Thus, the freshly prepared amines 31 and 32 were immediately treated with Boc2O and the resultant Boc-protected calix[4]arene tetraamines 33 and 34 bearing four or two 2-tosyloxyethyl groups at the narrow rims were obtained in good yields. By replacement of the tosyloxy groups with the azide ones, the desired tetraazidoethyl and diazidoethyl products 35 and 36 were prepared, thus completing the series of bifunctionalized cone calix[4]arenes having four protected amino groups at the wide rims and a CuAAC-ready propargyl (compounds 24–26) or 2-azidoethyl (compounds 35, 36) groups at the narrow rims of the macrocycles.
Some transformations of propargylated/2-azidoethylated p-aminocalix[4]arenes
To evaluate the synthetic potential of the obtained heterobifunctionalized calix[4]arenes, they were subjected to three-step chemical transformations, including the synthesis of triazoles under CuAAC conditions, followed by deprotection of the amino groups, and their conversion to urea moieties (Schemes 6–9).
The propargylated calix[4]arenes 24–26 were reacted with benzyl azide in the presence of CuI activated with triethylamine (20 equiv per Cu, Scheme 6). Under these conditions all three reactions were completed in 72 h at room temperature, and the desired Boc-protected p-aminocalix[4]arenes 37, 38, and 39 having four, two distal or one 1-benzyltriazol-4-ylmethyl moiety at their narrow rims were obtained. The lowered isolated yield of tetratriazole 37 was most likely due to incomplete destruction of its copper complex (although an extraction procedure for complex destruction using Na2S2O3 was applied), which was difficult to separate from the relatively polar free calixarene 37 using column chromatography. In line with this, when toluene-soluble CuI·P(OEt)3 was used as a catalyst (in this case heating was required to complete the cycloaddition), an even lower isolated yield of calixarene 37 (≈35%) was observed, indicating that a large portion of Cu+ was complexed with the calixarene product when no excess competing ligand such as triethylamine was present in the reaction mixture.
![[1860-5397-22-28-i6]](/bjoc/content/inline/1860-5397-22-28-i6.png?scale=2.0&max-width=1024&background=FFFFFF)
Scheme 6: Synthesis of triazolated p-aminocalix[4]arenes 37–39 from the propargylated calixarene precursors 24–26. Conditions: i) benzyl azide, CuI, Et3N, toluene, rt.
Scheme 6: Synthesis of triazolated p-aminocalix[4]arenes 37–39 from the propargylated calixarene precursors 24...
Following our previous findings on the reactivity of 2-azidoethylated calix[4]arenes with arylacetylenes under CuAAC conditions [81], calixarene 35 bearing four 2-azidoethyl groups at the narrow rim was reacted with phenylacetylene under heating using CuI·P(OEt)3 as a catalyst (Scheme 7). In contrast to the synthesis of calixarene 37, there were no problems encountered with the copper complex destruction and the yield of the isomeric calixarene 40 having four 2-(4-phenyltriazol-1-yl)ethyl moieties at the narrow rim was much higher, indicating a weaker binding in the 40/Cu+ system than in the 37/Cu+ one, due to the outer arrangement of the triazole N(3)-atoms relative to the calixarene core in compound 40.
![[1860-5397-22-28-i7]](/bjoc/content/inline/1860-5397-22-28-i7.png?scale=2.0&max-width=1024&background=FFFFFF)
Scheme 7: Synthesis of triazolated p-aminocalix[4]arenes 40 and 41 from the 2-azidoethylated calixarene precursors 35 and 36. Conditions: i) phenylacetylene, CuI·P(OEt)3, toluene, 100 °C; ii) phenylacetylene, CuI, toluene/Et3N, rt.
Scheme 7: Synthesis of triazolated p-aminocalix[4]arenes 40 and 41 from the 2-azidoethylated calixarene precu...
It was established previously, that p-tert-butylcalix[4]arene having two distal 2-azidoethyl groups at the narrow rim could not efficiently react with phenylacetylene under copper(I) catalysis unless a huge excess of Cu+-competing triethylamine is added to the system. This is most likely due to specific interactions of the diazide with polymeric/oligomeric copper(I) phenylacetylide hindering the cycloaddition [81]. To avoid similar issues, the Boc-protected p-aminocalix[4]arene 36 was reacted with phenylacetylene in the presence of CuI using a mixture of toluene and triethylamine (4:1) as solvent. Under these conditions, the reaction proceeded smoothly at room temperature and the desired ditriazole 41 was obtained in high yield (Scheme 7).
The tert-butoxycarbonyl groups in calix[4]arenes 37–41 were cleaved under standard conditions using trifluoroacetic acid, and after basification, free p-aminocalix[4]arenes 42–46 substituted at the narrow rims with either 1-benzyltriazol-4-ylmethyl or 2-(4-phenyltriazol-1-yl)ethyl groups were obtained (Scheme 8). Then, amines 42–46 were reacted with excess p-tolyl isocyanate to afford the corresponding tetraureacalix[4]arenes 47–51 (Scheme 9), the structures of which were unambiguously confirmed by their 1H NMR and 13C NMR spectra obtained from solutions in polar DMSO-d6.
![[1860-5397-22-28-i8]](/bjoc/content/inline/1860-5397-22-28-i8.png?scale=2.0&max-width=1024&background=FFFFFF)
Scheme 8: Removal of Boc protection in calixarenes 37–41; (i) CF3CO2H, dichloromethane, rt, then NaHCO3/H2O.
Scheme 8: Removal of Boc protection in calixarenes 37–41; (i) CF3CO2H, dichloromethane, rt, then NaHCO3/H2O.
![[1860-5397-22-28-i9]](/bjoc/content/inline/1860-5397-22-28-i9.png?scale=2.0&max-width=1024&background=FFFFFF)
Scheme 9: Synthesis of triazolated tetraureacalix[4]arenes 47–51. Conditions: (i) p-tolyl isocyanate, toluene, 60 °C.
Scheme 9: Synthesis of triazolated tetraureacalix[4]arenes 47–51. Conditions: (i) p-tolyl isocyanate, toluene...
Being calix[4]arene tetraureas, compounds 47–51 should form homodimeric capsules in H-bond non-competing solvents (e.g., chloroform) with the inclusion of one of the solvent molecules into the capsules. However, triazole heterocycles attached to the calixarene core can serve as weak hydrogen-bond acceptors [99] and thus may interfere with capsule formation, including the exclusive formation of heterodimeric capsules from the calixarene tetraaryl- and tetraarylsulfonylureas if co-dissolved in a 1:1 molar ratio [51-53]. Furthermore, tetraureas 47–51 contain relatively bulky substituents at the phenolic oxygen atoms, which may influence the conformational behavior of the macrocycle (e.g., pinched cone-to-pinched cone conformational interconversions), thereby favoring or disfavoring the C4v-symmetric ‘ideal’ cone conformation of the calixarene core required for capsule formation [59]. To check this, 1H NMR spectra from the CDCl3 solutions of multifunctionalized calixarenes 47–51 and their 1:1 mixtures with the known O-pentylated tetratosylureacalix[4]arene 52 [51] were acquired and analyzed.
In terms of time-averaged molecular symmetry, tetraureacalix[4]arenes of three types were prepared in this work: Cs-symmetric calixarene 49 with one triazole heterocycle at the narrow rim, C2v-symmetric calixarenes 48 and 51 with pairs of distal triazole groups, and C4v-symmetric tetraureas 47 and 50. Due to the phenomenon of supramolecular chirality caused by the direction of hydrogen bonds in the capsules [100], tetraureacalixarenes of the first type should form two regioisomeric homodimers possessing C1-symmetry or a single heterodimer with a C4v-symmetric tetratosylureacalix[4]arene 52 (each as a pair of enantiomers). Similarly, the C2v-symmetric tetraureacalixarenes of the second type should furnish C2-symmetric homo- or heterodimers as single regioisomers (each as a pair of enantiomers), whereas homo- and heterodimers formed by C4v-symmetric tetraureas of the third type are expected to have S8 (achiral meso) and C4 (as two enantiomers) symmetry, respectively. These features must immediately affect the signal patterns in the 1H NMR spectra of triazolated tetraureacalix[4]arenes and may also complicate their interpretation. Indeed, the 1H NMR spectrum of the triazolated tetraurea 49 obtained from its solution in CDCl3 turned out to be overcomplicated (see Figure 4). The spectral pattern apparently arises from the superposition of signals from two asymmetric homodimers, 492 isomer 1 and 492 isomer 2 (Figure 4a), which are present in a ratio of ≈1:1. The ratio of the isomers cannot be easily obtained from the spectrum except by analyzing the group of the most downfield-shifted signals corresponding to the half of the urea NHs involved in hydrogen bonds. According to the symmetry features discussed above, this group of signals should consist of eight singlets from each of the isomeric homodimers, thus giving sixteen partially overlapping signals (see insert in Figure 4d). Integration of the resolved signals of this group showed that their relative intensities are almost exactly multiples of 1, which is most likely in the case where the entire group is formed by sixteen signals of the same intensity.
![[1860-5397-22-28-4]](/bjoc/content/figures/1860-5397-22-28-4.png?scale=2.0&max-width=1024&background=FFFFFF)
Figure 4: Planar and energy-minimized structures (with CHCl3 molecule included and triazole groups highlighted) of isomeric homodimers of tetraurea 49 (a); structure of tetratosylureacalix[4]arene 52 (b); planar structure of heterodimer 49·52 (c); fragment of the 1H NMR spectrum of calixarene 49 in CDCl3 (d); fragment of the 1H NMR spectrum of an equimolar mixture of calixarenes 49 and 52 in CDCl3 (e). All spectra were acquired using a 600 MHz NMR spectrometer.
Figure 4: Planar and energy-minimized structures (with CHCl3 molecule included and triazole groups highlighte...
In support of this, the energy-minimized structures of two regioisomeric capsules, 492 isomer 1 and 492 isomer 2, each containing a chloroform molecule (Figure 4a, PBE0/def2-SVP, gCP, D4, SMD (chloroform), ORCA 6.0.1 package [101-109]), were obtained and the calculated difference in their full-electron energies was found to be less than 0.1 kcal mol−1, which is well below the error threshold. Thus, in contrast to tetraureas containing several different substituents in the urea groups [100], the presence of one bulky moiety at the narrow rim of the tetraureacalix[4]arene does not lead to a significant difference in the stability of the homodimers formed by compound 49, at least in a chloroform solution. To further confirm the ability of calixarene 49 to form capsules, the 1H NMR spectrum of its equimolar mixture with tetratosylureacalix[4]arene 52 in CDCl3 was also analyzed (Figure 4e). Despite the presence of a second calixarene molecule, the 1H NMR spectrum turned out to be simplified compared to the spectrum of homodimers 492, which is due to the formation of only one heterodimer 49·52. The spectrum clearly shows even more downfield-shifted resonances from the four NH groups involved in hydrogen bonding with the sulfonyl groups, as well as clear signals from the two AB spin systems of the methylene groups attached to the single triazole moiety, in which the protons have become diastereotopic due to the supramolecular chirality of the heterodimeric capsule (see inserts in Figure 4e).
Due to the higher symmetry of both homo- and heterodimeric capsules formed by ditriazolated tetraureas 48 and 51 (Figure 5), the 1H NMR spectra acquired from solutions of these compounds or their mixtures with tetrasulfonylurea 52 in CDCl3 turned out to be much more interpretable. In particular, the spectra of the homodimers (Figure 5b,c) contained four clearly recognizable downfield-shifted singlets from hydrogen-bonded NH groups, and all other signals appearing in the spectra corresponded to chiral C2-symmetric homodimeric capsules in both cases. The 1H NMR spectrum of the equimolar mixture 48/52 (taken as an example) contained one set of signals from the C2-symmetric heterodimer 48·CDCl3·52, but not from the homodimers 48·CDCl3·48 and 52·CDCl3·52, and thus reflected the expected exclusive formation of heterocapsular assemblies, which was thus not hindered by the two triazole heterocycles arranged at the narrow rim of the calixarene core.
![[1860-5397-22-28-5]](/bjoc/content/figures/1860-5397-22-28-5.png?scale=2.0&max-width=1024&background=FFFFFF)
Figure 5: Planar structures of homodimers 482, 512, and heterodimer 48·52 (a); fragment of the 1H NMR spectrum of calixarene 51 in CDCl3 (b); fragment of the 1H NMR spectrum of calixarene 48 in CDCl3 (c); fragment of the 1H NMR spectrum of an equimolar mixture of calixarenes 48 and 52 in CDCl3 (d). All spectra were acquired using a 400 MHz NMR spectrometer.
Figure 5: Planar structures of homodimers 482, 512, and heterodimer 48·52 (a); fragment of the 1H NMR spectru...
Except for a slight broadening of the signals caused by the limited solubility of the compound, the 1H NMR spectrum obtained for a CDCl3 solution of tetraureacalixarene 50 having four 2-(4-phenyltriazol-1-yl)ethyl groups at the narrow rim was in good agreement with the expected spectrum of homodimer 502 possessing S8 symmetry (Figure 6a). Indeed, the spectrum contained two 4J-coupled doublets from the diastereotopic aromatic protons of the calixarene core and just one pair of 2J-coupled doublets from its methylene bridges, two well-distant singlets from the urea NHs participating in the hydrogen-bond belt, one resonance from the triazole protons, as well as only one set of signals from the tolyl and phenyl groups, which confirms the formation of homodimer 502 (Figure 6b).
![[1860-5397-22-28-6]](/bjoc/content/figures/1860-5397-22-28-6.png?scale=2.0&max-width=1024&background=FFFFFF)
Figure 6: Planar structures of homodimers 472, 502, and heterodimer 47·52 (a); fragment of the 1H NMR spectrum of calixarene 50 in CDCl3 (b); fragment of the 1H NMR spectrum of calixarene 47 in CDCl3 (c); fragment of the 1H NMR spectrum of the same sample washed with water (d); fragment of the 1H NMR spectrum of an equimolar mixture of calixarenes 47 and 52 in CDCl3 (e); fragment of the 1H NMR spectrum of the same sample washed with water (f). All spectra were acquired using a 400 MHz NMR spectrometer.
Figure 6: Planar structures of homodimers 472, 502, and heterodimer 47·52 (a); fragment of the 1H NMR spectru...
Quite surprisingly, such a clear pattern was not observed in the 1H NMR spectrum of a CDCl3 solution of tetraurea 47, which is an isomer of calixarene 50, having 1-benzyltriazol-4-ylmethyl groups replacing 2-(4-phenyltriazol-1-yl)ethyl groups, and thus possessing less flexible linkers between the calixarene core and the differently arranged triazole heterocycles. In addition to what could be interpreted as a set of signals from homodimer 472, a set of broadened resonances was observed in the spectrum (Figure 6c). Neither prolonged heating of the sample at 50 °C before the spectrum acquisition, nor measuring the spectrum at elevated temperature resulted in a simplification of the spectral pattern, indicating no significant contribution of any slow rotational/conformational motions to the complexity of the spectrum. Similar additional signals were observed in the 1H NMR spectrum of an equimolar mixture of calixarenes 47 and 52 in CDCl3 (Figure 6e). Reasonably, additional non-covalent interactions possibly occurred between two or more calixarene molecules, partially destroying the dimeric capsules and promoting the formation of more complex assemblies, which was characteristic exclusively of calixarene 47 (of the five calixarene tetraureas studied). Since nothing other than hydrogen bonds involving triazole heterocycles could be proposed for such non-covalent interactions, a drop of methanol-d4 was added to a solution of tetraurea 47 in CDCl3 to break these additional bonds. However, the effect of adding this polar solvent turned out to be too strong, and the resulting spectrum only contained signals from the tetraurea monomer 47, indicating the disruption of intermolecular hydrogen bonds between the urea moieties as well.
Still, it was discovered by chance that simply washing of a solution of calixarene 47 in CDCl3 with water solved the issue. Indeed, the 1H NMR spectrum of the sample obtained after separation of the aqueous phase turned out to be easily interpretable (Figure 6d) and contained a set of signals from homodimer 472 similar to those in the spectrum of homodimer 502. A drastic simplification of the spectrum was also observed for a water-washed solution of a mixture of calixarenes 47 and 52 in CDCl3, and only clear resonances from the heterodimer 47·52 were detected there (Figure 6f). In a separate series of experiments, the 1H NMR spectra of unstable emulsions obtained by shaking solutions of calixarenes 47–51 in CDCl3 with water were rapidly collected. Although lowered-resolution NMR spectra were obtained, in all cases only resonances of the respective homodimers were detected, despite the presence in the spectra of a large signal from the aqueous phase (at ≈4.8 ppm) along with a signal from dissolved water (1.56 ppm). Considering the low solubility of water in chloroform, the stability of the capsules in water-saturated CDCl3 seems reasonable, because the dimers are held together by at least 16 hydrogen bonds between the urea groups. On the other hand, this water content is apparently sufficient to disrupt weaker intermolecular contacts involving triazole heterocycles as hydrogen-bond acceptors. Since the structures of the above mentioned non-capsular aggregate(s) formed by calixarene 47 cannot be drawn based on the available data, it is unclear why only four 1-benzyltriazol-4-ylmethyl groups attached to the narrow rim of the tetraurea calixarene are responsible for the unwanted aggregation, whereas one or two 1-benzyltriazol-4-ylmethyl groups (calixarenes 48, 49) or two or four 2-(4-phenyltriazol-1-yl)ethyl groups (calixarenes 50, 51) do not interfere with the capsule formation.
Due to the mentioned limited solubility of calixarene 50 in CDCl3, it was not possible to conduct an accurate experiment to study any preferential heterodimerization between isomeric tetraureas 47 and 50, which possess a set of narrow rim substituents of different flexibility. However, preliminary NMR data showed that a turbid CDCl3 solution of both calixarenes, taken in a more or less equimolar ratio, contained signals from both homodimers 472 and 502, a set of broadened signals from non-capsular aggregates (readily removed from the spectrum by washing the solution with water) and a set of signals that could be interpreted as those from heterodimer 47·50. Thus, the difference in the spatial preorganization of the four urea groups in calixarenes 47 and 50, caused by the different constraints related to their narrow-rim substituents, is apparently not sufficient to provide any significant selectivity for the formation of homo/heterocapsules.
Finally, since the triazole groups arranged at the narrow rim of the calix[4]arene macrocycle are capable of complexation with transition-metal cations, showing different complexation preferences depending on the triazole attachment mode [81], the attractive idea of combining complexation and capsule formation using triazolated tetraureas was explored. For preliminary NMR complexation experiments, zinc and silver cations were chosen to target 1-benzyltriazol-4-ylmethyl- and 2-(4-phenyltriazol-1-yl)ethyl-decorated calix[4]arenes, respectively, but unfortunately this study was unsuccessful due to solubility issues. Indeed, CD3CN or its mixtures with CDCl3, in which Zn(ClO4)2·6H2O is soluble, turned out to be solvents which were too polar and destroyed dimers 472 or 482, although some changes in the 1H NMR spectra of monomers 47 and 48 could be detected upon addition of the salt. Attempts to conduct the experiment using solid–liquid extraction in neat CDCl3 were also unsuccessful, since no changes in the 1H NMR spectrum of capsule 472 were detected upon the addition of excess solid Zn(ClO4)2·6H2O, even after prolonged storing of the mixture at room temperature or under moderate (55 °C) heating. Although anhydrous AgPF6, which is soluble in non-polar solvents, was used for trial binding experiments with 2-(4-phenyltriazol-1-yl)ethyl-containing capsules 502 and 512, no data on the complexation processes were obtained in these cases either; adding excess salt to solutions of these calixarenes in CDCl3 or in CDCl3/benzene-d6 mixtures resulted in the formation of insoluble precipitates containing the entire portions of the hosts, thus indicating a complexation process but the concentrations of dissolved compounds was too low for NMR analysis. Thus, additional experiments need to be designed to reveal whether the triazolated tetraureacalix[4]arenes can be used to include pairs of triazole-bound cations into supramolecules via intermolecular hydrogen bonding between the urea groups.
Conclusion
We have expanded the functionalization capabilities of cone calix[4]arenes by combining orthogonally transformable functional groups of two types within a single macrocyclic core. Both wide and narrow rims of the calixarene macrocycles were used to introduce four amino groups along with propargyl or 2-azidoethyl groups into the molecules. This allowed for further step-wise functionalization of the core using CuAAC ‘click’ reactions involving alkyne or azide moieties and amine acylation or similar reactions. Five heteromultifunctional N-protected p-aminocalix[4]arenes comprising one/two/four propargyl groups or two/four 2-azidoethyl groups at the narrow rims of the macrocycles were prepared using synthetic strategies that involved wide rim ipso-nitration of prefunctionalized macrocycles (containing either silyl-protected propargyl groups or the synthetic precursors of 2-azidoethyl groups) followed by reduction of the exhaustively nitrated products. During thorough fine-tuning of the nitration conditions, several partially nitrated calix[4]arene propargyl ethers bearing tert-butyldimethylsilyl protecting groups in the alkyne moieties were also obtained and their exact structures were determined using 2D NMR and X-ray diffraction data, contributing to a general understanding of the selectivity of nitration when applied to calixarenes containing bulky narrow-rim substituents.
The functionalization capabilities of the propargylated/2-azidoethylated p-aminocalix[4]arenes were demonstrated by their facile conversion into the narrow-rim triazolated macrocycles of two types (differing in the mutual arrangement of the calixarene core and the triazole heterocycles) and then into the wide-rim tetraurea derivatives. A preliminary study of the aggregation of triazolated calix[4]arene tetraureas showed that the triazole groups, despite their relatively large size and hydrogen-bond accepting ability, have only a minor effect on the formation of homo/heterodimeric capsules by these compounds in chloroform. Thus, a variety of functional units can be introduced into the narrow rims of the developed propargylated/2-azidoethylated calixarenes using CuAAC reactions, and the resulting triazolated calixarenes can be further functionalized at their amino groups, including the preparation of self-assembled capsular aggregates of virtually unlimited complexity.
Supporting Information
| Supporting Information File 1: Synthesis details, copies of 1H and 13C NMR spectra of novel compounds, details of X-ray diffraction measurements and crystal structure data. | ||
| Format: PDF | Size: 5.6 MB | Download |
| Supporting Information File 2: Crystallographic information file for compound 16. | ||
| Format: CIF | Size: 1.6 MB | Download |
| Supporting Information File 3: Crystallographic information file for compound 15. | ||
| Format: CIF | Size: 3.1 MB | Download |
Data Availability Statement
All data that supports the findings of this study is available in the published article and/or the supporting information of this article.
References
-
Asfari, Z.; Böhmer, V.; Harrowfield, J.; Vicens, J., Eds. Calixarenes 2001; Kluwer Academic Publishers: Dordrecht, Netherlands, 2001. doi:10.1007/0-306-47522-7
Return to citation in text: [1] -
Böhmer, V. Calixarenes. In The Chemistry of Phenols; Rappoport, Z., Ed.; PATAI'S Chemistry of Functional Groups; John Wiley & Sons: Chichester, UK, 2003; pp 1369–1454. doi:10.1002/0470857277.ch19
Return to citation in text: [1] -
Vicens, J.; Harrowfield, J.; Baklouti, L., Eds. Calixarenes in the Nanoworld; Springer: Dordrecht, Netherlands, 2007. doi:10.1007/978-1-4020-5022-4
Return to citation in text: [1] -
Neri, P.; Sessler, J. L.; Wang, M.-X., Eds. Calixarenes and Beyond; Springer International Publishing: Cham, Switzerland, 2016. doi:10.1007/978-3-319-31867-7
Return to citation in text: [1] -
Arduini, A.; Mirone, L.; Paganuzzi, D.; Pinalli, A.; Pochini, A.; Secchi, A.; Ungaro, R. Tetrahedron 1996, 52, 6011–6018. doi:10.1016/0040-4020(96)00238-4
Return to citation in text: [1] -
Jakobi, R. A.; Böhmer, V.; Grüttner, C.; Kraft, D.; Vogt, W. New J. Chem. 1996, 20, 493–501.
Return to citation in text: [1] -
Mulder, A.; Auletta, T.; Sartori, A.; Del Ciotto, S.; Casnati, A.; Ungaro, R.; Huskens, J.; Reinhoudt, D. N. J. Am. Chem. Soc. 2004, 126, 6627–6636. doi:10.1021/ja0317168
Return to citation in text: [1] -
Gattuso, G.; Grasso, G.; Marino, N.; Notti, A.; Pappalardo, A.; Pappalardo, S.; Parisi, M. F. Eur. J. Org. Chem. 2011, 5696–5703. doi:10.1002/ejoc.201100812
Return to citation in text: [1] -
Buttress, J. P.; Day, D. P.; Courtney, J. M.; Lawrence, E. J.; Hughes, D. L.; Blagg, R. J.; Crossley, A.; Matthews, S. E.; Redshaw, C.; Bulman Page, P. C.; Wildgoose, G. G. Langmuir 2016, 32, 7806–7813. doi:10.1021/acs.langmuir.6b02222
Return to citation in text: [1] [2] [3] [4] [5] [6] -
Hosseinzadeh, R.; Maliji, F.; Golchoubian, H.; Bekhradnia, A. ChemistrySelect 2019, 4, 3914–3920. doi:10.1002/slct.201900241
Return to citation in text: [1] -
Mattiuzzi, A.; Troian-Gautier, L.; Mertens, J.; Reniers, F.; Bergamini, J.-F.; Lenne, Q.; Lagrost, C.; Jabin, I. RSC Adv. 2020, 10, 13553–13561. doi:10.1039/d0ra01011a
Return to citation in text: [1] -
Gaeta, M.; Rodolico, E.; Fragalà, M. E.; Pappalardo, A.; Pisagatti, I.; Gattuso, G.; Notti, A.; Parisi, M. F.; Purrello, R.; D’Urso, A. Molecules 2021, 26, 704. doi:10.3390/molecules26030704
Return to citation in text: [1] -
Weeden, C.; Hartlieb, K. J.; Lim, L. Y. J. Pharm. Pharmacol. 2012, 64, 1403–1411. doi:10.1111/j.2042-7158.2012.01518.x
Return to citation in text: [1] -
Morita, Y.; Agawa, T.; Kai, Y.; Kanehisa, N.; Kasai, N.; Nomura, E.; Taniguchi, H. Chem. Lett. 1989, 18, 1349–1352. doi:10.1246/cl.1989.1349
Return to citation in text: [1] -
Morita, Y.; Agawa, T.; Nomura, E.; Taniguchi, H. J. Org. Chem. 1992, 57, 3658–3662. doi:10.1021/jo00039a027
Return to citation in text: [1] -
Ak, M. S.; Deligöz, H. J. Inclusion Phenom. Macrocyclic Chem. 2007, 59, 115–123. doi:10.1007/s10847-007-9300-9
Return to citation in text: [1] -
Burilov, V.; Makarov, E.; Mironova, D.; Sultanova, E.; Bilyukova, I.; Akyol, K.; Evtugyn, V.; Islamov, D.; Usachev, K.; Mukhametzyanov, T.; Solovieva, S.; Antipin, I. Int. J. Mol. Sci. 2022, 23, 14889. doi:10.3390/ijms232314889
Return to citation in text: [1] -
Kolusheva, S.; Zadmard, R.; Schrader, T.; Jelinek, R. J. Am. Chem. Soc. 2006, 128, 13592–13598. doi:10.1021/ja064957z
Return to citation in text: [1] -
Zadmard, R.; Schrader, T. Angew. Chem., Int. Ed. 2006, 45, 2703–2706. doi:10.1002/anie.200502946
Return to citation in text: [1] -
Breitkreuz, C. J.; Zadmard, R.; Schrader, T. Supramol. Chem. 2008, 20, 109–115. doi:10.1080/10610270701747040
Return to citation in text: [1] -
Shahgaldian, P.; Sciotti, M. A.; Pieles, U. Langmuir 2008, 24, 8522–8526. doi:10.1021/la801083h
Return to citation in text: [1] -
Blecking, C. J.; Hu, W.; Zadmard, R.; Dasgupta, A.; Schrader, T. Synthesis 2011, 1193–1204. doi:10.1055/s-0030-1259980
Return to citation in text: [1] -
Hu, W.; Blecking, C.; Kralj, M.; Šuman, L.; Piantanida, I.; Schrader, T. Chem. – Eur. J. 2012, 18, 3589–3597. doi:10.1002/chem.201100634
Return to citation in text: [1] -
Lalor, R.; DiGesso, J. L.; Mueller, A.; Matthews, S. E. Chem. Commun. 2007, 4907–4909. doi:10.1039/b712100h
Return to citation in text: [1] -
Lalor, R.; Baillie-Johnson, H.; Redshaw, C.; Matthews, S. E.; Mueller, A. J. Am. Chem. Soc. 2008, 130, 2892–2893. doi:10.1021/ja0782596
Return to citation in text: [1] -
Nault, L.; Cumbo, A.; Pretôt, R. F.; Sciotti, M. A.; Shahgaldian, P. Chem. Commun. 2010, 46, 5581–5583. doi:10.1039/b926025k
Return to citation in text: [1] -
Dudic, M.; Colombo, A.; Sansone, F.; Casnati, A.; Donofrio, G.; Ungaro, R. Tetrahedron 2004, 60, 11613–11618. doi:10.1016/j.tet.2004.09.047
Return to citation in text: [1] -
Sansone, F.; Dudič, M.; Donofrio, G.; Rivetti, C.; Baldini, L.; Casnati, A.; Cellai, S.; Ungaro, R. J. Am. Chem. Soc. 2006, 128, 14528–14536. doi:10.1021/ja0634425
Return to citation in text: [1] -
Huang, Q.; Zhao, H.; Shui, M.; Guo, D.-S.; Wang, R. Chem. Sci. 2020, 11, 9623–9629. doi:10.1039/d0sc03922e
Return to citation in text: [1] -
Hu, X.-Y.; Hu, Z.-Y.; Tian, J.-H.; Shi, L.; Ding, F.; Li, H.-B.; Guo, D.-S. Chem. Commun. 2022, 58, 13198–13201. doi:10.1039/d2cc04963e
Return to citation in text: [1] -
Gasparello, J.; Papi, C.; Zurlo, M.; Volpi, S.; Gambari, R.; Corradini, R.; Casnati, A.; Sansone, F.; Finotti, A. Pharmaceutics 2023, 15, 2121. doi:10.3390/pharmaceutics15082121
Return to citation in text: [1] -
Baldini, L.; Cacciapaglia, R.; Casnati, A.; Mandolini, L.; Salvio, R.; Sansone, F.; Ungaro, R. J. Org. Chem. 2012, 77, 3381–3389. doi:10.1021/jo300193y
Return to citation in text: [1] -
Salvio, R.; Casnati, A.; Mandolini, L.; Sansone, F.; Ungaro, R. Org. Biomol. Chem. 2012, 10, 8941–8943. doi:10.1039/c2ob26688a
Return to citation in text: [1] -
Lisi, D.; Vezzoni, C. A.; Casnati, A.; Sansone, F.; Salvio, R. Chem. – Eur. J. 2023, 29, e202203213. doi:10.1002/chem.202203213
Return to citation in text: [1] -
Vezzoni, C. A.; Casnati, A.; Orlanducci, S.; Sansone, F.; Salvio, R. ChemCatChem 2024, 16, e202301477. doi:10.1002/cctc.202301477
Return to citation in text: [1] -
Arnaud-Neu, F.; Böhmer, V.; Dozol, J.-F.; Grüttner, C.; Jakobi, R. A.; Kraft, D.; Mauprivez, O.; Rouquette, H.; Schwing-Weill, M.-J.; Simon, N.; Vogt, W. J. Chem. Soc., Perkin Trans. 2 1996, 1175–1182. doi:10.1039/p29960001175
Return to citation in text: [1] -
Delmau, L. H.; Simon, N.; Dozol, J.-F.; Eymard, S.; Tournois, B.; Schwing-Weill, M.-J.; Arnaud-Neu, F.; Böhmer, V.; Grüttner, C.; Musigmann, C.; Tunayar, A. Chem. Commun. 1998, 1627–1628. doi:10.1039/a802555j
Return to citation in text: [1] -
Matthews, S. E.; Saadioui, M.; Böhmer, V.; Barboso, S.; Arnaud-Neu, F.; Schwing-Weill, M.-J.; Garcia Carrera, A.; Dozol, J.-F. J. Prakt. Chem. 1999, 341, 264–273. doi:10.1002/(sici)1521-3897(199904)341:3<264::aid-prac264>3.0.co;2-f
Return to citation in text: [1] -
Arduini, A.; Böhmer, V.; Delmau, L.; Desreux, J.-F.; Dozol, J.-F.; Carrera, M. A. G.; Lambert, B.; Musigmann, C.; Pochini, A.; Shivanyuk, A.; Ugozzoli, F. Chem. – Eur. J. 2000, 6, 2135–2144. doi:10.1002/1521-3765(20000616)6:12<2135::aid-chem2135>3.0.co;2-a
Return to citation in text: [1] -
Matthews, S. E.; Parzuchowski, P.; Garcia-Carrera, A.; Grüttner, C.; Dozol, J.-F.; Böhmer, V. Chem. Commun. 2001, 417–418. doi:10.1039/b009679m
Return to citation in text: [1] -
Schmidt, C.; Saadioui, M.; Böhmer, V.; Host, V.; Spirlet, M.-R.; Desreux, J. F.; Brisach, F.; Arnaud-Neu, F.; Dozol, J.-F. Org. Biomol. Chem. 2003, 1, 4089–4096. doi:10.1039/b307929e
Return to citation in text: [1] -
Böhmer, V.; Dozol, J.-F.; Grüttner, C.; Liger, K.; Matthews, S. E.; Rudershausen, S.; Saadioui, M.; Wang, P. Org. Biomol. Chem. 2004, 2, 2327–2334. doi:10.1039/b405602g
Return to citation in text: [1] -
Peters, C.; Braekers, D.; Kroupa, J.; Kasyan, O.; Miroshnichenko, S.; Rudzevich, V.; Böhmer, V.; Desreux, J.-F. Radiochim. Acta 2008, 96, 203–210. doi:10.1524/ract.2008.1480
Return to citation in text: [1] -
May, E. M.; Solovyov, A.; Guo, Y.; Drapailo, A.; Matveev, Y.; Kalchenko, V.; Nitsche, H.; Katz, A. Eur. J. Inorg. Chem. 2016, 4542–4545. doi:10.1002/ejic.201600946
Return to citation in text: [1] -
Putkov, A. E.; Vasiliev, A. N.; Zubavichus, Y. V.; Teterin, Y. A.; Ivanov, K. E.; Trigub, A. L.; Kovalev, V. V.; Vatsouro, I. M. Mendeleev Commun. 2021, 31, 188–190. doi:10.1016/j.mencom.2021.03.014
Return to citation in text: [1] -
Shimizu, K. D.; Rebek, J., Jr. Proc. Natl. Acad. Sci. U. S. A. 1995, 92, 12403–12407. doi:10.1073/pnas.92.26.12403
Return to citation in text: [1] -
Mogck, O.; Böhmer, V.; Vogt, W. Tetrahedron 1996, 52, 8489–8496. doi:10.1016/0040-4020(96)00404-8
Return to citation in text: [1] -
Hamann, B. C.; Shimizu, K. D.; Rebek, J., Jr. Angew. Chem., Int. Ed. Engl. 1996, 35, 1326–1329. doi:10.1002/anie.199613261
Return to citation in text: [1] -
Cho, Y. L.; Rudkevich, D. M.; Shivanyuk, A.; Rissanen, K.; Rebek, J., Jr. Chem. – Eur. J. 2000, 6, 3788–3796. doi:10.1002/1521-3765(20001016)6:20<3788::aid-chem3788>3.0.co;2-y
Return to citation in text: [1] -
Vysotsky, M. O.; Pop, A.; Broda, F.; Thondorf, I.; Böhmer, V. Chem. – Eur. J. 2001, 7, 4403–4410. doi:10.1002/1521-3765(20011015)7:20<4403::aid-chem4403>3.0.co;2-j
Return to citation in text: [1] -
Rudzevich, Y.; Vysotsky, M. O.; Böhmer, V.; Brody, M. S.; Rebek, J., Jr.; Broda, F.; Thondorf, I. Org. Biomol. Chem. 2004, 2, 3080–3084. doi:10.1039/b410462e
Return to citation in text: [1] [2] [3] -
Thondorf, I.; Rudzevich, Y.; Rudzevich, V.; Böhmer, V. Org. Biomol. Chem. 2007, 5, 2775–2782. doi:10.1039/b708679b
Return to citation in text: [1] [2] -
Bolte, M.; Thondorf, I.; Böhmer, V.; Rudzevich, V.; Rudzevich, Y. CrystEngComm 2008, 10, 270–272. doi:10.1039/b716368a
Return to citation in text: [1] [2] -
Rudzevich, V.; Rudzevich, Y.; Böhmer, V. Synlett 2009, 1887–1904. doi:10.1055/s-0029-1217513
Return to citation in text: [1] -
Mogck, O.; Pons, M.; Böhmer, V.; Vogt, W. J. Am. Chem. Soc. 1997, 119, 5706–5712. doi:10.1021/ja970078o
Return to citation in text: [1] -
Vysotsky, M. O.; Böhmer, V. Org. Lett. 2000, 2, 3571–3574. doi:10.1021/ol0064967
Return to citation in text: [1] -
Vysotsky, M. O.; Thondorf, I.; Böhmer, V. Chem. Commun. 2001, 1890–1891. doi:10.1039/b105613c
Return to citation in text: [1] -
Vysotsky, M. O.; Mogck, O.; Rudzevich, Y.; Shivanyuk, A.; Böhmer, V.; Brody, M. S.; Cho, Y. L.; Rudkevich, D. M.; Rebek, J., Jr. J. Org. Chem. 2004, 69, 6115–6120. doi:10.1021/jo049128f
Return to citation in text: [1] -
Vatsouro, I.; Rudzevich, V.; Böhmer, V. Org. Lett. 2007, 9, 1375–1377. doi:10.1021/ol0702775
Return to citation in text: [1] [2] -
Vatsouro, I.; Alt, E.; Vysotsky, M.; Böhmer, V. Org. Biomol. Chem. 2008, 6, 998–1003. doi:10.1039/b719053k
Return to citation in text: [1] -
Vysotsky, M. O.; Bolte, M.; Thondorf, I.; Böhmer, V. Chem. – Eur. J. 2003, 9, 3375–3382. doi:10.1002/chem.200304912
Return to citation in text: [1] -
Wang, L.; Vysotsky, M. O.; Bogdan, A.; Bolte, M.; Böhmer, V. Science 2004, 304, 1312–1314. doi:10.1126/science.1096688
Return to citation in text: [1] -
Vysotsky, M. O.; Bogdan, A.; Wang, L.; Böhmer, V. Chem. Commun. 2004, 1268–1269. doi:10.1039/b402719a
Return to citation in text: [1] -
Bogdan, A.; Vysotsky, M. O.; Ikai, T.; Okamoto, Y.; Böhmer, V. Chem. – Eur. J. 2004, 10, 3324–3330. doi:10.1002/chem.200400195
Return to citation in text: [1] -
Gaeta, C.; Vysotsky, M. O.; Bogdan, A.; Böhmer, V. J. Am. Chem. Soc. 2005, 127, 13136–13137. doi:10.1021/ja054184z
Return to citation in text: [1] -
Molokanova, O.; Vysotsky, M. O.; Cao, Y.; Thondorf, I.; Böhmer, V. Angew. Chem., Int. Ed. 2006, 45, 8051–8055. doi:10.1002/anie.200602166
Return to citation in text: [1] -
Rudzevich, Y.; Cao, Y.; Rudzevich, V.; Böhmer, V. Chem. – Eur. J. 2008, 14, 3346–3354. doi:10.1002/chem.200701694
Return to citation in text: [1] -
Janke, M.; Rudzevich, Y.; Molokanova, O.; Metzroth, T.; Mey, I.; Diezemann, G.; Marszalek, P. E.; Gauss, J.; Böhmer, V.; Janshoff, A. Nat. Nanotechnol. 2009, 4, 225–229. doi:10.1038/nnano.2008.416
Return to citation in text: [1] -
Jaschonek, S.; Schäfer, K.; Diezemann, G. J. Phys. Chem. B 2019, 123, 4688–4694. doi:10.1021/acs.jpcb.9b02676
Return to citation in text: [1] -
Tornøe, C. W.; Christensen, C.; Meldal, M. J. Org. Chem. 2002, 67, 3057–3064. doi:10.1021/jo011148j
Return to citation in text: [1] -
Rostovtsev, V. V.; Green, L. G.; Fokin, V. V.; Sharpless, K. B. Angew. Chem., Int. Ed. 2002, 41, 2596–2599. doi:10.1002/1521-3773(20020715)41:14<2596::aid-anie2596>3.0.co;2-4
Return to citation in text: [1] -
Meldal, M.; Tornøe, C. W. Chem. Rev. 2008, 108, 2952–3015. doi:10.1021/cr0783479
Return to citation in text: [1] -
Singh, M. S.; Chowdhury, S.; Koley, S. Tetrahedron 2016, 72, 5257–5283. doi:10.1016/j.tet.2016.07.044
Return to citation in text: [1] -
Ryu, E.-H.; Zhao, Y. Org. Lett. 2005, 7, 1035–1037. doi:10.1021/ol047468h
Return to citation in text: [1] -
Pineda-Castañeda, H. M.; Rivera-Monroy, Z. J.; Maldonado, M. ACS Omega 2023, 8, 3650–3666. doi:10.1021/acsomega.2c06269
Return to citation in text: [1] -
Sreenivasu Mummidivarapu, V. V.; Joseph, R.; Pulla Rao, C.; Kumar Pathak, R. Coord. Chem. Rev. 2023, 493, 215256. doi:10.1016/j.ccr.2023.215256
Return to citation in text: [1] -
Makarov, E.; Iskhakova, Z.; Burilov, V.; Solovieva, S.; Antipin, I. J. Inclusion Phenom. Macrocyclic Chem. 2023, 103, 319–353. doi:10.1007/s10847-023-01200-6
Return to citation in text: [1] -
Song, M.; Sun, Z.; Han, C.; Tian, D.; Li, H.; Kim, J. S. Chem. – Asian J. 2014, 9, 2344–2357. doi:10.1002/asia.201400024
Return to citation in text: [1] -
Gorbunov, A.; Cheshkov, D.; Kovalev, V.; Vatsouro, I. Chem. – Eur. J. 2015, 21, 9528–9534. doi:10.1002/chem.201500946
Return to citation in text: [1] -
Gorbunov, A.; Kuznetsova, J.; Deltsov, I.; Molokanova, A.; Cheshkov, D.; Bezzubov, S.; Kovalev, V.; Vatsouro, I. Org. Chem. Front. 2020, 7, 2432–2441. doi:10.1039/d0qo00650e
Return to citation in text: [1] -
Gorbunov, A.; Kuznetsova, J.; Puchnin, K.; Kovalev, V.; Vatsouro, I. New J. Chem. 2019, 43, 4562–4580. doi:10.1039/c8nj06464d
Return to citation in text: [1] [2] [3] [4] -
Gorbunov, A.; Sokolova, N.; Kudryashova, E.; Nenajdenko, V.; Kovalev, V.; Vatsouro, I. Chem. – Eur. J. 2016, 22, 12415–12423. doi:10.1002/chem.201601818
Return to citation in text: [1] -
Malakhova, M.; Gorbunov, A.; Lentin, I.; Kovalev, V.; Vatsouro, I. Org. Biomol. Chem. 2022, 20, 8092–8103. doi:10.1039/d2ob01588a
Return to citation in text: [1] -
Syakaev, V. V.; Masliy, A. N.; Podyachev, S. N.; Sudakova, S. N.; Shvedova, A. E.; Lentin, I. I.; Gorbunov, A. N.; Vatsouro, I. M.; Lapaev, D. V.; Mambetova, G. S.; Kovalev, V. V.; Kuznetsov, A. M.; Mustafina, A. R. Inorg. Chim. Acta 2024, 561, 121848. doi:10.1016/j.ica.2023.121848
Return to citation in text: [1] -
Bezzubov, S.; Ermolov, K.; Gorbunov, A.; Kalle, P.; Lentin, I.; Latyshev, G.; Kovalev, V.; Vatsouro, I. Dalton Trans. 2021, 50, 16765–16769. doi:10.1039/d1dt03579g
Return to citation in text: [1] -
Gorbunov, A.; Ozerov, N.; Malakhova, M.; Eshtukov, A.; Cheshkov, D.; Bezzubov, S.; Kovalev, V.; Vatsouro, I. Org. Chem. Front. 2021, 8, 3853–3866. doi:10.1039/d1qo00636c
Return to citation in text: [1] -
Malakhova, M.; Gorbunov, A.; Ozerov, N.; Korniltsev, I.; Ermolov, K.; Bezzubov, S.; Kovalev, V.; Vatsouro, I. Org. Chem. Front. 2022, 9, 3084–3092. doi:10.1039/d2qo00432a
Return to citation in text: [1] -
Gorbunov, A.; Iskandarova, A.; Puchnin, K.; Nenajdenko, V.; Kovalev, V.; Vatsouro, I. Chem. Commun. 2020, 56, 4122–4125. doi:10.1039/d0cc01196g
Return to citation in text: [1] -
Chetcuti, M. J.; Devoille, A. M. J.; Othman, A. B.; Souane, R.; Thuéry, P.; Vicens, J. Dalton Trans. 2009, 2999–3008. doi:10.1039/b821144b
Return to citation in text: [1] -
Gullo, M. C.; Baldini, L.; Casnati, A.; Marchiò, L. Cryst. Growth Des. 2020, 20, 3611–3616. doi:10.1021/acs.cgd.0c00442
Return to citation in text: [1] -
Cecioni, S.; Lalor, R.; Blanchard, B.; Praly, J.-P.; Imberty, A.; Matthews, S. E.; Vidal, S. Chem. – Eur. J. 2009, 15, 13232–13240. doi:10.1002/chem.200901799
Return to citation in text: [1] -
Chang, K.-C.; Chen, C.-Y.; Hsu, C.-Y.; Lee, L.-W.; Chung, W.-S. Analyst 2022, 147, 5105–5112. doi:10.1039/d2an01201d
Return to citation in text: [1] -
Mogck, O.; Böhmer, V.; Ferguson, G.; Vogt, W. J. Chem. Soc., Perkin Trans. 1 1996, 1711–1715. doi:10.1039/p19960001711
Return to citation in text: [1] -
The CIF files have been deposited at the Cambridge Crystallographic Data Centre: CCDC 2431889 (compound 16), 2431890 (compound 15).
Return to citation in text: [1] -
Arnaud-Neu, F.; Browne, J. K.; Byrne, D.; Marrs, D. J.; McKervey, M. A.; O'Hagan, P.; Schwing-Weill, M. J.; Walker, A. Chem. – Eur. J. 1999, 5, 175–186. doi:10.1002/(sici)1521-3765(19990104)5:1<175::aid-chem175>3.0.co;2-p
Return to citation in text: [1] -
Knoblauch, S.; Falana, O. M.; Nam, J.; Roundhill, D. M.; Hennig, H.; Zeckert, K. Inorg. Chim. Acta 2000, 300–302, 328–332. doi:10.1016/s0020-1693(99)00577-0
Return to citation in text: [1] -
Webber, P. R. A.; Cowley, A.; Drew, M. G. B.; Beer, P. D. Chem. – Eur. J. 2003, 9, 2439–2446. doi:10.1002/chem.200204518
Return to citation in text: [1] -
Liu, Y.; Lu, Y.; Prashad, M.; Repič, O.; Blacklock, T. J. Adv. Synth. Catal. 2005, 347, 217–219. doi:10.1002/adsc.200404236
Return to citation in text: [1] -
Bolshchikov, B.; Volkov, S.; Sokolova, D.; Gorbunov, A.; Serebryannikova, A.; Gloriozov, I.; Cheshkov, D.; Bezzubov, S.; Chung, W.-S.; Kovalev, V.; Vatsouro, I. Org. Chem. Front. 2019, 6, 3327–3341. doi:10.1039/c9qo00859d
Return to citation in text: [1] -
Pop, A.; Vysotsky, M. O.; Saadioui, M.; Böhmer, V. Chem. Commun. 2003, 1124–1125. doi:10.1039/b301418e
Return to citation in text: [1] [2] -
Neese, F. Wiley Interdiscip. Rev.: Comput. Mol. Sci. 2022, 12, e1606. doi:10.1002/wcms.1606
Return to citation in text: [1] -
Neese, F.; Wennmohs, F.; Hansen, A.; Becker, U. Chem. Phys. 2009, 356, 98–109. doi:10.1016/j.chemphys.2008.10.036
Return to citation in text: [1] -
Garcia‐Ratés, M.; Neese, F. J. Comput. Chem. 2020, 41, 922–939. doi:10.1002/jcc.26139
Return to citation in text: [1] -
Helmich-Paris, B.; de Souza, B.; Neese, F.; Izsák, R. J. Chem. Phys. 2021, 155, 104109. doi:10.1063/5.0058766
Return to citation in text: [1] -
Neese, F. J. Comput. Chem. 2023, 44, 381–396. doi:10.1002/jcc.26942
Return to citation in text: [1] -
Caldeweyher, E.; Bannwarth, C.; Grimme, S. J. Chem. Phys. 2017, 147, 034112. doi:10.1063/1.4993215
Return to citation in text: [1] -
Caldeweyher, E.; Ehlert, S.; Hansen, A.; Neugebauer, H.; Spicher, S.; Bannwarth, C.; Grimme, S. J. Chem. Phys. 2019, 150, 154122. doi:10.1063/1.5090222
Return to citation in text: [1] -
Caldeweyher, E.; Mewes, J.-M.; Ehlert, S.; Grimme, S. Phys. Chem. Chem. Phys. 2020, 22, 8499–8512. doi:10.1039/d0cp00502a
Return to citation in text: [1] -
Wittmann, L.; Gordiy, I.; Friede, M.; Helmich-Paris, B.; Grimme, S.; Hansen, A.; Bursch, M. Phys. Chem. Chem. Phys. 2024, 26, 21379–21394. doi:10.1039/d4cp01514b
Return to citation in text: [1]
| 9. | Buttress, J. P.; Day, D. P.; Courtney, J. M.; Lawrence, E. J.; Hughes, D. L.; Blagg, R. J.; Crossley, A.; Matthews, S. E.; Redshaw, C.; Bulman Page, P. C.; Wildgoose, G. G. Langmuir 2016, 32, 7806–7813. doi:10.1021/acs.langmuir.6b02222 |
| 95. | Arnaud-Neu, F.; Browne, J. K.; Byrne, D.; Marrs, D. J.; McKervey, M. A.; O'Hagan, P.; Schwing-Weill, M. J.; Walker, A. Chem. – Eur. J. 1999, 5, 175–186. doi:10.1002/(sici)1521-3765(19990104)5:1<175::aid-chem175>3.0.co;2-p |
| 96. | Knoblauch, S.; Falana, O. M.; Nam, J.; Roundhill, D. M.; Hennig, H.; Zeckert, K. Inorg. Chim. Acta 2000, 300–302, 328–332. doi:10.1016/s0020-1693(99)00577-0 |
| 97. | Webber, P. R. A.; Cowley, A.; Drew, M. G. B.; Beer, P. D. Chem. – Eur. J. 2003, 9, 2439–2446. doi:10.1002/chem.200204518 |
| 1. | Asfari, Z.; Böhmer, V.; Harrowfield, J.; Vicens, J., Eds. Calixarenes 2001; Kluwer Academic Publishers: Dordrecht, Netherlands, 2001. doi:10.1007/0-306-47522-7 |
| 2. | Böhmer, V. Calixarenes. In The Chemistry of Phenols; Rappoport, Z., Ed.; PATAI'S Chemistry of Functional Groups; John Wiley & Sons: Chichester, UK, 2003; pp 1369–1454. doi:10.1002/0470857277.ch19 |
| 3. | Vicens, J.; Harrowfield, J.; Baklouti, L., Eds. Calixarenes in the Nanoworld; Springer: Dordrecht, Netherlands, 2007. doi:10.1007/978-1-4020-5022-4 |
| 4. | Neri, P.; Sessler, J. L.; Wang, M.-X., Eds. Calixarenes and Beyond; Springer International Publishing: Cham, Switzerland, 2016. doi:10.1007/978-3-319-31867-7 |
| 19. | Zadmard, R.; Schrader, T. Angew. Chem., Int. Ed. 2006, 45, 2703–2706. doi:10.1002/anie.200502946 |
| 20. | Breitkreuz, C. J.; Zadmard, R.; Schrader, T. Supramol. Chem. 2008, 20, 109–115. doi:10.1080/10610270701747040 |
| 21. | Shahgaldian, P.; Sciotti, M. A.; Pieles, U. Langmuir 2008, 24, 8522–8526. doi:10.1021/la801083h |
| 22. | Blecking, C. J.; Hu, W.; Zadmard, R.; Dasgupta, A.; Schrader, T. Synthesis 2011, 1193–1204. doi:10.1055/s-0030-1259980 |
| 23. | Hu, W.; Blecking, C.; Kralj, M.; Šuman, L.; Piantanida, I.; Schrader, T. Chem. – Eur. J. 2012, 18, 3589–3597. doi:10.1002/chem.201100634 |
| 75. | Pineda-Castañeda, H. M.; Rivera-Monroy, Z. J.; Maldonado, M. ACS Omega 2023, 8, 3650–3666. doi:10.1021/acsomega.2c06269 |
| 76. | Sreenivasu Mummidivarapu, V. V.; Joseph, R.; Pulla Rao, C.; Kumar Pathak, R. Coord. Chem. Rev. 2023, 493, 215256. doi:10.1016/j.ccr.2023.215256 |
| 77. | Makarov, E.; Iskhakova, Z.; Burilov, V.; Solovieva, S.; Antipin, I. J. Inclusion Phenom. Macrocyclic Chem. 2023, 103, 319–353. doi:10.1007/s10847-023-01200-6 |
| 78. | Song, M.; Sun, Z.; Han, C.; Tian, D.; Li, H.; Kim, J. S. Chem. – Asian J. 2014, 9, 2344–2357. doi:10.1002/asia.201400024 |
| 51. | Rudzevich, Y.; Vysotsky, M. O.; Böhmer, V.; Brody, M. S.; Rebek, J., Jr.; Broda, F.; Thondorf, I. Org. Biomol. Chem. 2004, 2, 3080–3084. doi:10.1039/b410462e |
| 18. | Kolusheva, S.; Zadmard, R.; Schrader, T.; Jelinek, R. J. Am. Chem. Soc. 2006, 128, 13592–13598. doi:10.1021/ja064957z |
| 79. | Gorbunov, A.; Cheshkov, D.; Kovalev, V.; Vatsouro, I. Chem. – Eur. J. 2015, 21, 9528–9534. doi:10.1002/chem.201500946 |
| 80. | Gorbunov, A.; Kuznetsova, J.; Deltsov, I.; Molokanova, A.; Cheshkov, D.; Bezzubov, S.; Kovalev, V.; Vatsouro, I. Org. Chem. Front. 2020, 7, 2432–2441. doi:10.1039/d0qo00650e |
| 100. | Pop, A.; Vysotsky, M. O.; Saadioui, M.; Böhmer, V. Chem. Commun. 2003, 1124–1125. doi:10.1039/b301418e |
| 14. | Morita, Y.; Agawa, T.; Kai, Y.; Kanehisa, N.; Kasai, N.; Nomura, E.; Taniguchi, H. Chem. Lett. 1989, 18, 1349–1352. doi:10.1246/cl.1989.1349 |
| 15. | Morita, Y.; Agawa, T.; Nomura, E.; Taniguchi, H. J. Org. Chem. 1992, 57, 3658–3662. doi:10.1021/jo00039a027 |
| 16. | Ak, M. S.; Deligöz, H. J. Inclusion Phenom. Macrocyclic Chem. 2007, 59, 115–123. doi:10.1007/s10847-007-9300-9 |
| 17. | Burilov, V.; Makarov, E.; Mironova, D.; Sultanova, E.; Bilyukova, I.; Akyol, K.; Evtugyn, V.; Islamov, D.; Usachev, K.; Mukhametzyanov, T.; Solovieva, S.; Antipin, I. Int. J. Mol. Sci. 2022, 23, 14889. doi:10.3390/ijms232314889 |
| 70. | Tornøe, C. W.; Christensen, C.; Meldal, M. J. Org. Chem. 2002, 67, 3057–3064. doi:10.1021/jo011148j |
| 71. | Rostovtsev, V. V.; Green, L. G.; Fokin, V. V.; Sharpless, K. B. Angew. Chem., Int. Ed. 2002, 41, 2596–2599. doi:10.1002/1521-3773(20020715)41:14<2596::aid-anie2596>3.0.co;2-4 |
| 72. | Meldal, M.; Tornøe, C. W. Chem. Rev. 2008, 108, 2952–3015. doi:10.1021/cr0783479 |
| 73. | Singh, M. S.; Chowdhury, S.; Koley, S. Tetrahedron 2016, 72, 5257–5283. doi:10.1016/j.tet.2016.07.044 |
| 51. | Rudzevich, Y.; Vysotsky, M. O.; Böhmer, V.; Brody, M. S.; Rebek, J., Jr.; Broda, F.; Thondorf, I. Org. Biomol. Chem. 2004, 2, 3080–3084. doi:10.1039/b410462e |
| 52. | Thondorf, I.; Rudzevich, Y.; Rudzevich, V.; Böhmer, V. Org. Biomol. Chem. 2007, 5, 2775–2782. doi:10.1039/b708679b |
| 53. | Bolte, M.; Thondorf, I.; Böhmer, V.; Rudzevich, V.; Rudzevich, Y. CrystEngComm 2008, 10, 270–272. doi:10.1039/b716368a |
| 5. | Arduini, A.; Mirone, L.; Paganuzzi, D.; Pinalli, A.; Pochini, A.; Secchi, A.; Ungaro, R. Tetrahedron 1996, 52, 6011–6018. doi:10.1016/0040-4020(96)00238-4 |
| 6. | Jakobi, R. A.; Böhmer, V.; Grüttner, C.; Kraft, D.; Vogt, W. New J. Chem. 1996, 20, 493–501. |
| 7. | Mulder, A.; Auletta, T.; Sartori, A.; Del Ciotto, S.; Casnati, A.; Ungaro, R.; Huskens, J.; Reinhoudt, D. N. J. Am. Chem. Soc. 2004, 126, 6627–6636. doi:10.1021/ja0317168 |
| 8. | Gattuso, G.; Grasso, G.; Marino, N.; Notti, A.; Pappalardo, A.; Pappalardo, S.; Parisi, M. F. Eur. J. Org. Chem. 2011, 5696–5703. doi:10.1002/ejoc.201100812 |
| 9. | Buttress, J. P.; Day, D. P.; Courtney, J. M.; Lawrence, E. J.; Hughes, D. L.; Blagg, R. J.; Crossley, A.; Matthews, S. E.; Redshaw, C.; Bulman Page, P. C.; Wildgoose, G. G. Langmuir 2016, 32, 7806–7813. doi:10.1021/acs.langmuir.6b02222 |
| 10. | Hosseinzadeh, R.; Maliji, F.; Golchoubian, H.; Bekhradnia, A. ChemistrySelect 2019, 4, 3914–3920. doi:10.1002/slct.201900241 |
| 11. | Mattiuzzi, A.; Troian-Gautier, L.; Mertens, J.; Reniers, F.; Bergamini, J.-F.; Lenne, Q.; Lagrost, C.; Jabin, I. RSC Adv. 2020, 10, 13553–13561. doi:10.1039/d0ra01011a |
| 12. | Gaeta, M.; Rodolico, E.; Fragalà, M. E.; Pappalardo, A.; Pisagatti, I.; Gattuso, G.; Notti, A.; Parisi, M. F.; Purrello, R.; D’Urso, A. Molecules 2021, 26, 704. doi:10.3390/molecules26030704 |
| 13. | Weeden, C.; Hartlieb, K. J.; Lim, L. Y. J. Pharm. Pharmacol. 2012, 64, 1403–1411. doi:10.1111/j.2042-7158.2012.01518.x |
| 59. | Vatsouro, I.; Rudzevich, V.; Böhmer, V. Org. Lett. 2007, 9, 1375–1377. doi:10.1021/ol0702775 |
| 36. | Arnaud-Neu, F.; Böhmer, V.; Dozol, J.-F.; Grüttner, C.; Jakobi, R. A.; Kraft, D.; Mauprivez, O.; Rouquette, H.; Schwing-Weill, M.-J.; Simon, N.; Vogt, W. J. Chem. Soc., Perkin Trans. 2 1996, 1175–1182. doi:10.1039/p29960001175 |
| 37. | Delmau, L. H.; Simon, N.; Dozol, J.-F.; Eymard, S.; Tournois, B.; Schwing-Weill, M.-J.; Arnaud-Neu, F.; Böhmer, V.; Grüttner, C.; Musigmann, C.; Tunayar, A. Chem. Commun. 1998, 1627–1628. doi:10.1039/a802555j |
| 38. | Matthews, S. E.; Saadioui, M.; Böhmer, V.; Barboso, S.; Arnaud-Neu, F.; Schwing-Weill, M.-J.; Garcia Carrera, A.; Dozol, J.-F. J. Prakt. Chem. 1999, 341, 264–273. doi:10.1002/(sici)1521-3897(199904)341:3<264::aid-prac264>3.0.co;2-f |
| 39. | Arduini, A.; Böhmer, V.; Delmau, L.; Desreux, J.-F.; Dozol, J.-F.; Carrera, M. A. G.; Lambert, B.; Musigmann, C.; Pochini, A.; Shivanyuk, A.; Ugozzoli, F. Chem. – Eur. J. 2000, 6, 2135–2144. doi:10.1002/1521-3765(20000616)6:12<2135::aid-chem2135>3.0.co;2-a |
| 40. | Matthews, S. E.; Parzuchowski, P.; Garcia-Carrera, A.; Grüttner, C.; Dozol, J.-F.; Böhmer, V. Chem. Commun. 2001, 417–418. doi:10.1039/b009679m |
| 41. | Schmidt, C.; Saadioui, M.; Böhmer, V.; Host, V.; Spirlet, M.-R.; Desreux, J. F.; Brisach, F.; Arnaud-Neu, F.; Dozol, J.-F. Org. Biomol. Chem. 2003, 1, 4089–4096. doi:10.1039/b307929e |
| 42. | Böhmer, V.; Dozol, J.-F.; Grüttner, C.; Liger, K.; Matthews, S. E.; Rudershausen, S.; Saadioui, M.; Wang, P. Org. Biomol. Chem. 2004, 2, 2327–2334. doi:10.1039/b405602g |
| 43. | Peters, C.; Braekers, D.; Kroupa, J.; Kasyan, O.; Miroshnichenko, S.; Rudzevich, V.; Böhmer, V.; Desreux, J.-F. Radiochim. Acta 2008, 96, 203–210. doi:10.1524/ract.2008.1480 |
| 44. | May, E. M.; Solovyov, A.; Guo, Y.; Drapailo, A.; Matveev, Y.; Kalchenko, V.; Nitsche, H.; Katz, A. Eur. J. Inorg. Chem. 2016, 4542–4545. doi:10.1002/ejic.201600946 |
| 45. | Putkov, A. E.; Vasiliev, A. N.; Zubavichus, Y. V.; Teterin, Y. A.; Ivanov, K. E.; Trigub, A. L.; Kovalev, V. V.; Vatsouro, I. M. Mendeleev Commun. 2021, 31, 188–190. doi:10.1016/j.mencom.2021.03.014 |
| 55. | Mogck, O.; Pons, M.; Böhmer, V.; Vogt, W. J. Am. Chem. Soc. 1997, 119, 5706–5712. doi:10.1021/ja970078o |
| 56. | Vysotsky, M. O.; Böhmer, V. Org. Lett. 2000, 2, 3571–3574. doi:10.1021/ol0064967 |
| 57. | Vysotsky, M. O.; Thondorf, I.; Böhmer, V. Chem. Commun. 2001, 1890–1891. doi:10.1039/b105613c |
| 58. | Vysotsky, M. O.; Mogck, O.; Rudzevich, Y.; Shivanyuk, A.; Böhmer, V.; Brody, M. S.; Cho, Y. L.; Rudkevich, D. M.; Rebek, J., Jr. J. Org. Chem. 2004, 69, 6115–6120. doi:10.1021/jo049128f |
| 59. | Vatsouro, I.; Rudzevich, V.; Böhmer, V. Org. Lett. 2007, 9, 1375–1377. doi:10.1021/ol0702775 |
| 60. | Vatsouro, I.; Alt, E.; Vysotsky, M.; Böhmer, V. Org. Biomol. Chem. 2008, 6, 998–1003. doi:10.1039/b719053k |
| 81. | Gorbunov, A.; Kuznetsova, J.; Puchnin, K.; Kovalev, V.; Vatsouro, I. New J. Chem. 2019, 43, 4562–4580. doi:10.1039/c8nj06464d |
| 32. | Baldini, L.; Cacciapaglia, R.; Casnati, A.; Mandolini, L.; Salvio, R.; Sansone, F.; Ungaro, R. J. Org. Chem. 2012, 77, 3381–3389. doi:10.1021/jo300193y |
| 33. | Salvio, R.; Casnati, A.; Mandolini, L.; Sansone, F.; Ungaro, R. Org. Biomol. Chem. 2012, 10, 8941–8943. doi:10.1039/c2ob26688a |
| 34. | Lisi, D.; Vezzoni, C. A.; Casnati, A.; Sansone, F.; Salvio, R. Chem. – Eur. J. 2023, 29, e202203213. doi:10.1002/chem.202203213 |
| 35. | Vezzoni, C. A.; Casnati, A.; Orlanducci, S.; Sansone, F.; Salvio, R. ChemCatChem 2024, 16, e202301477. doi:10.1002/cctc.202301477 |
| 61. | Vysotsky, M. O.; Bolte, M.; Thondorf, I.; Böhmer, V. Chem. – Eur. J. 2003, 9, 3375–3382. doi:10.1002/chem.200304912 |
| 62. | Wang, L.; Vysotsky, M. O.; Bogdan, A.; Bolte, M.; Böhmer, V. Science 2004, 304, 1312–1314. doi:10.1126/science.1096688 |
| 63. | Vysotsky, M. O.; Bogdan, A.; Wang, L.; Böhmer, V. Chem. Commun. 2004, 1268–1269. doi:10.1039/b402719a |
| 64. | Bogdan, A.; Vysotsky, M. O.; Ikai, T.; Okamoto, Y.; Böhmer, V. Chem. – Eur. J. 2004, 10, 3324–3330. doi:10.1002/chem.200400195 |
| 65. | Gaeta, C.; Vysotsky, M. O.; Bogdan, A.; Böhmer, V. J. Am. Chem. Soc. 2005, 127, 13136–13137. doi:10.1021/ja054184z |
| 66. | Molokanova, O.; Vysotsky, M. O.; Cao, Y.; Thondorf, I.; Böhmer, V. Angew. Chem., Int. Ed. 2006, 45, 8051–8055. doi:10.1002/anie.200602166 |
| 67. | Rudzevich, Y.; Cao, Y.; Rudzevich, V.; Böhmer, V. Chem. – Eur. J. 2008, 14, 3346–3354. doi:10.1002/chem.200701694 |
| 68. | Janke, M.; Rudzevich, Y.; Molokanova, O.; Metzroth, T.; Mey, I.; Diezemann, G.; Marszalek, P. E.; Gauss, J.; Böhmer, V.; Janshoff, A. Nat. Nanotechnol. 2009, 4, 225–229. doi:10.1038/nnano.2008.416 |
| 69. | Jaschonek, S.; Schäfer, K.; Diezemann, G. J. Phys. Chem. B 2019, 123, 4688–4694. doi:10.1021/acs.jpcb.9b02676 |
| 99. | Bolshchikov, B.; Volkov, S.; Sokolova, D.; Gorbunov, A.; Serebryannikova, A.; Gloriozov, I.; Cheshkov, D.; Bezzubov, S.; Chung, W.-S.; Kovalev, V.; Vatsouro, I. Org. Chem. Front. 2019, 6, 3327–3341. doi:10.1039/c9qo00859d |
| 27. | Dudic, M.; Colombo, A.; Sansone, F.; Casnati, A.; Donofrio, G.; Ungaro, R. Tetrahedron 2004, 60, 11613–11618. doi:10.1016/j.tet.2004.09.047 |
| 28. | Sansone, F.; Dudič, M.; Donofrio, G.; Rivetti, C.; Baldini, L.; Casnati, A.; Cellai, S.; Ungaro, R. J. Am. Chem. Soc. 2006, 128, 14528–14536. doi:10.1021/ja0634425 |
| 29. | Huang, Q.; Zhao, H.; Shui, M.; Guo, D.-S.; Wang, R. Chem. Sci. 2020, 11, 9623–9629. doi:10.1039/d0sc03922e |
| 30. | Hu, X.-Y.; Hu, Z.-Y.; Tian, J.-H.; Shi, L.; Ding, F.; Li, H.-B.; Guo, D.-S. Chem. Commun. 2022, 58, 13198–13201. doi:10.1039/d2cc04963e |
| 31. | Gasparello, J.; Papi, C.; Zurlo, M.; Volpi, S.; Gambari, R.; Corradini, R.; Casnati, A.; Sansone, F.; Finotti, A. Pharmaceutics 2023, 15, 2121. doi:10.3390/pharmaceutics15082121 |
| 98. | Liu, Y.; Lu, Y.; Prashad, M.; Repič, O.; Blacklock, T. J. Adv. Synth. Catal. 2005, 347, 217–219. doi:10.1002/adsc.200404236 |
| 24. | Lalor, R.; DiGesso, J. L.; Mueller, A.; Matthews, S. E. Chem. Commun. 2007, 4907–4909. doi:10.1039/b712100h |
| 25. | Lalor, R.; Baillie-Johnson, H.; Redshaw, C.; Matthews, S. E.; Mueller, A. J. Am. Chem. Soc. 2008, 130, 2892–2893. doi:10.1021/ja0782596 |
| 26. | Nault, L.; Cumbo, A.; Pretôt, R. F.; Sciotti, M. A.; Shahgaldian, P. Chem. Commun. 2010, 46, 5581–5583. doi:10.1039/b926025k |
| 46. | Shimizu, K. D.; Rebek, J., Jr. Proc. Natl. Acad. Sci. U. S. A. 1995, 92, 12403–12407. doi:10.1073/pnas.92.26.12403 |
| 47. | Mogck, O.; Böhmer, V.; Vogt, W. Tetrahedron 1996, 52, 8489–8496. doi:10.1016/0040-4020(96)00404-8 |
| 48. | Hamann, B. C.; Shimizu, K. D.; Rebek, J., Jr. Angew. Chem., Int. Ed. Engl. 1996, 35, 1326–1329. doi:10.1002/anie.199613261 |
| 49. | Cho, Y. L.; Rudkevich, D. M.; Shivanyuk, A.; Rissanen, K.; Rebek, J., Jr. Chem. – Eur. J. 2000, 6, 3788–3796. doi:10.1002/1521-3765(20001016)6:20<3788::aid-chem3788>3.0.co;2-y |
| 50. | Vysotsky, M. O.; Pop, A.; Broda, F.; Thondorf, I.; Böhmer, V. Chem. – Eur. J. 2001, 7, 4403–4410. doi:10.1002/1521-3765(20011015)7:20<4403::aid-chem4403>3.0.co;2-j |
| 51. | Rudzevich, Y.; Vysotsky, M. O.; Böhmer, V.; Brody, M. S.; Rebek, J., Jr.; Broda, F.; Thondorf, I. Org. Biomol. Chem. 2004, 2, 3080–3084. doi:10.1039/b410462e |
| 52. | Thondorf, I.; Rudzevich, Y.; Rudzevich, V.; Böhmer, V. Org. Biomol. Chem. 2007, 5, 2775–2782. doi:10.1039/b708679b |
| 53. | Bolte, M.; Thondorf, I.; Böhmer, V.; Rudzevich, V.; Rudzevich, Y. CrystEngComm 2008, 10, 270–272. doi:10.1039/b716368a |
| 54. | Rudzevich, V.; Rudzevich, Y.; Böhmer, V. Synlett 2009, 1887–1904. doi:10.1055/s-0029-1217513 |
| 81. | Gorbunov, A.; Kuznetsova, J.; Puchnin, K.; Kovalev, V.; Vatsouro, I. New J. Chem. 2019, 43, 4562–4580. doi:10.1039/c8nj06464d |
| 86. | Gorbunov, A.; Ozerov, N.; Malakhova, M.; Eshtukov, A.; Cheshkov, D.; Bezzubov, S.; Kovalev, V.; Vatsouro, I. Org. Chem. Front. 2021, 8, 3853–3866. doi:10.1039/d1qo00636c |
| 87. | Malakhova, M.; Gorbunov, A.; Ozerov, N.; Korniltsev, I.; Ermolov, K.; Bezzubov, S.; Kovalev, V.; Vatsouro, I. Org. Chem. Front. 2022, 9, 3084–3092. doi:10.1039/d2qo00432a |
| 81. | Gorbunov, A.; Kuznetsova, J.; Puchnin, K.; Kovalev, V.; Vatsouro, I. New J. Chem. 2019, 43, 4562–4580. doi:10.1039/c8nj06464d |
| 82. | Gorbunov, A.; Sokolova, N.; Kudryashova, E.; Nenajdenko, V.; Kovalev, V.; Vatsouro, I. Chem. – Eur. J. 2016, 22, 12415–12423. doi:10.1002/chem.201601818 |
| 83. | Malakhova, M.; Gorbunov, A.; Lentin, I.; Kovalev, V.; Vatsouro, I. Org. Biomol. Chem. 2022, 20, 8092–8103. doi:10.1039/d2ob01588a |
| 84. | Syakaev, V. V.; Masliy, A. N.; Podyachev, S. N.; Sudakova, S. N.; Shvedova, A. E.; Lentin, I. I.; Gorbunov, A. N.; Vatsouro, I. M.; Lapaev, D. V.; Mambetova, G. S.; Kovalev, V. V.; Kuznetsov, A. M.; Mustafina, A. R. Inorg. Chim. Acta 2024, 561, 121848. doi:10.1016/j.ica.2023.121848 |
| 101. | Neese, F. Wiley Interdiscip. Rev.: Comput. Mol. Sci. 2022, 12, e1606. doi:10.1002/wcms.1606 |
| 102. | Neese, F.; Wennmohs, F.; Hansen, A.; Becker, U. Chem. Phys. 2009, 356, 98–109. doi:10.1016/j.chemphys.2008.10.036 |
| 103. | Garcia‐Ratés, M.; Neese, F. J. Comput. Chem. 2020, 41, 922–939. doi:10.1002/jcc.26139 |
| 104. | Helmich-Paris, B.; de Souza, B.; Neese, F.; Izsák, R. J. Chem. Phys. 2021, 155, 104109. doi:10.1063/5.0058766 |
| 105. | Neese, F. J. Comput. Chem. 2023, 44, 381–396. doi:10.1002/jcc.26942 |
| 106. | Caldeweyher, E.; Bannwarth, C.; Grimme, S. J. Chem. Phys. 2017, 147, 034112. doi:10.1063/1.4993215 |
| 107. | Caldeweyher, E.; Ehlert, S.; Hansen, A.; Neugebauer, H.; Spicher, S.; Bannwarth, C.; Grimme, S. J. Chem. Phys. 2019, 150, 154122. doi:10.1063/1.5090222 |
| 108. | Caldeweyher, E.; Mewes, J.-M.; Ehlert, S.; Grimme, S. Phys. Chem. Chem. Phys. 2020, 22, 8499–8512. doi:10.1039/d0cp00502a |
| 109. | Wittmann, L.; Gordiy, I.; Friede, M.; Helmich-Paris, B.; Grimme, S.; Hansen, A.; Bursch, M. Phys. Chem. Chem. Phys. 2024, 26, 21379–21394. doi:10.1039/d4cp01514b |
| 85. | Bezzubov, S.; Ermolov, K.; Gorbunov, A.; Kalle, P.; Lentin, I.; Latyshev, G.; Kovalev, V.; Vatsouro, I. Dalton Trans. 2021, 50, 16765–16769. doi:10.1039/d1dt03579g |
| 100. | Pop, A.; Vysotsky, M. O.; Saadioui, M.; Böhmer, V. Chem. Commun. 2003, 1124–1125. doi:10.1039/b301418e |
| 81. | Gorbunov, A.; Kuznetsova, J.; Puchnin, K.; Kovalev, V.; Vatsouro, I. New J. Chem. 2019, 43, 4562–4580. doi:10.1039/c8nj06464d |
| 9. | Buttress, J. P.; Day, D. P.; Courtney, J. M.; Lawrence, E. J.; Hughes, D. L.; Blagg, R. J.; Crossley, A.; Matthews, S. E.; Redshaw, C.; Bulman Page, P. C.; Wildgoose, G. G. Langmuir 2016, 32, 7806–7813. doi:10.1021/acs.langmuir.6b02222 |
| 94. | The CIF files have been deposited at the Cambridge Crystallographic Data Centre: CCDC 2431889 (compound 16), 2431890 (compound 15). |
| 9. | Buttress, J. P.; Day, D. P.; Courtney, J. M.; Lawrence, E. J.; Hughes, D. L.; Blagg, R. J.; Crossley, A.; Matthews, S. E.; Redshaw, C.; Bulman Page, P. C.; Wildgoose, G. G. Langmuir 2016, 32, 7806–7813. doi:10.1021/acs.langmuir.6b02222 |
| 93. | Mogck, O.; Böhmer, V.; Ferguson, G.; Vogt, W. J. Chem. Soc., Perkin Trans. 1 1996, 1711–1715. doi:10.1039/p19960001711 |
| 9. | Buttress, J. P.; Day, D. P.; Courtney, J. M.; Lawrence, E. J.; Hughes, D. L.; Blagg, R. J.; Crossley, A.; Matthews, S. E.; Redshaw, C.; Bulman Page, P. C.; Wildgoose, G. G. Langmuir 2016, 32, 7806–7813. doi:10.1021/acs.langmuir.6b02222 |
| 89. | Chetcuti, M. J.; Devoille, A. M. J.; Othman, A. B.; Souane, R.; Thuéry, P.; Vicens, J. Dalton Trans. 2009, 2999–3008. doi:10.1039/b821144b |
| 90. | Gullo, M. C.; Baldini, L.; Casnati, A.; Marchiò, L. Cryst. Growth Des. 2020, 20, 3611–3616. doi:10.1021/acs.cgd.0c00442 |
| 91. | Cecioni, S.; Lalor, R.; Blanchard, B.; Praly, J.-P.; Imberty, A.; Matthews, S. E.; Vidal, S. Chem. – Eur. J. 2009, 15, 13232–13240. doi:10.1002/chem.200901799 |
| 92. | Chang, K.-C.; Chen, C.-Y.; Hsu, C.-Y.; Lee, L.-W.; Chung, W.-S. Analyst 2022, 147, 5105–5112. doi:10.1039/d2an01201d |
| 88. | Gorbunov, A.; Iskandarova, A.; Puchnin, K.; Nenajdenko, V.; Kovalev, V.; Vatsouro, I. Chem. Commun. 2020, 56, 4122–4125. doi:10.1039/d0cc01196g |
| 9. | Buttress, J. P.; Day, D. P.; Courtney, J. M.; Lawrence, E. J.; Hughes, D. L.; Blagg, R. J.; Crossley, A.; Matthews, S. E.; Redshaw, C.; Bulman Page, P. C.; Wildgoose, G. G. Langmuir 2016, 32, 7806–7813. doi:10.1021/acs.langmuir.6b02222 |
© 2026 Korniltsev et al.; licensee Beilstein-Institut.
This is an open access article licensed under the terms of the Beilstein-Institut Open Access License Agreement (https://www.beilstein-journals.org/bjoc/terms), which is identical to the Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0). The reuse of material under this license requires that the author(s), source and license are credited. Third-party material in this article could be subject to other licenses (typically indicated in the credit line), and in this case, users are required to obtain permission from the license holder to reuse the material.