Search results
Search for "Pseudomonas" in Full Text gives 114 result(s) in Beilstein Journal of Organic Chemistry.
Impact of Pseudomonas aeruginosa quorum sensing signaling molecules on adhesion and inflammatory markers in endothelial cells
Beilstein J. Org. Chem. 2018, 14, 2580–2588, doi:10.3762/bjoc.14.235

- of Agronomical Sciences and Veterinary Medicine, Faculty of Veterinary Medicine, Bucharest, Romania Institute of Cellular Biology and Pathology Nicolae Simionescu of Romanian Academy, Romania 10.3762/bjoc.14.235 Abstract Pseudomonas aeruginosa relies on the quorum sensing (QS) signaling system as a
- develop anti-QS therapeutic strategies to fight against P. aeruginosa infections. Keywords: adhesion; host–pathogen interaction; inflammation; Pseudomonas; quorum sensing; Introduction Pseudomonas (P.) aeruginosa is an opportunistic pathogen that causes severe and persistent infections in immune
- . Pseudomonas aeruginosa is recognized as the principal pathogen responsible of high morbidity and mortality in patients with cystic fibrosis, one of the most common life-threatening autosomal recessive genetic disease in Northwest European populations, determined by mutations in the cystic fibrosis
The enzymes of microbial nicotine metabolism
Beilstein J. Org. Chem. 2018, 14, 2295–2307, doi:10.3762/bjoc.14.204
- NicA2 instead catalyzes oxidation of the substrate carbon–nitrogen bond as shown in Scheme 4. Cloning and expression of the protein encoded by the nox gene of Pseudomonas sp. HZN6 showed that it also catalyzes oxidation of nicotine to pseudooxynicotine [54]. The sequence of the protein is most similar
- enzyme as shown in Scheme 9, with the hydrolytic step being nonenzymatic. This is essentially the same reaction as that catalyzed by A. nicotinovorans γ-N-methylaminobutyrate demethylating oxidase (Mabo). E. coli expressing the sap gene from Pseudomonas sp. HZN6 will catalyze the NADP+-dependent
- converting 3-succinoylpyrimidine to 6-hydroxy-3-succinoylpyridine [48]. These results support the identification of SpmABC as a molybdopterin enzyme that catalyzes this step in the pathway. Enzymes with this activity do not appear to have been identified as yet for P. putida S5 and Pseudomonas sp. HZN6. HspA
Defining the hydrophobic interactions that drive competence stimulating peptide (CSP)-ComD binding in Streptococcus pneumoniae
Beilstein J. Org. Chem. 2018, 14, 1769–1777, doi:10.3762/bjoc.14.151

- modulators against a multitude of Gram-negative bacterial species, including Pseudomonas aeruginosa, Vibrio fischeri, Vibrio harveyi, Vibrio cholerae, and Acinetobacter baumannii has been conducted [6][7][8][9][10]. Contrary, with the exception of the accessory gene regulator (agr) QS circuitry in
Two new 2-alkylquinolones, inhibitory to the fish skin ulcer pathogen Tenacibaculum maritimum, produced by a rhizobacterium of the genus Burkholderia sp.
Beilstein J. Org. Chem. 2018, 14, 1446–1451, doi:10.3762/bjoc.14.122
- Burkholderia produce many more secondary metabolites than reported, as this group was previously classified into the genus Pseudomonas [8]. In fact, the high capacity of Burkholderia in secondary metabolism is demonstrated by the presence of unique functionalities, such as monocyclic 3-pyrazolone [9], α
- Pseudomonas aeruginosa. A series of chemoecological studies of P. aeruginosa has uncovered multifunctional roles of this quinolone class as antibacterial, antifungal, iron-chelating, and autoinducer agents to assist the survival of the producing organisms [32]. Additionally, drug discovery attempts have
- collected on a Bruker micrOTOF focus mass spectrometer. Collection of Burkholderia strains and broth screening Burkholderia strains were collected by serial dilution plating on Pseudomonas agar supplemented with C-F-C (Oxoid, Basingstoke, England) from rhizosphere soils of Welsh onion and cucumber (Cucumis
An overview of recent advances in duplex DNA recognition by small molecules
Beilstein J. Org. Chem. 2018, 14, 1051–1086, doi:10.3762/bjoc.14.93
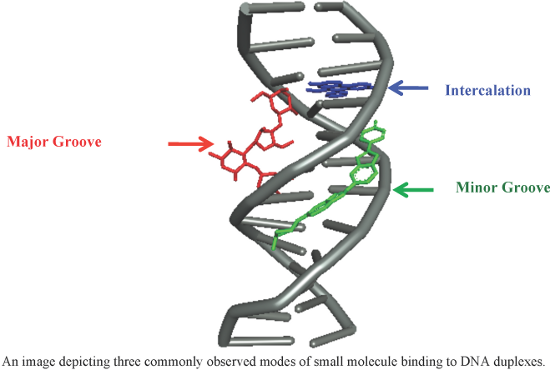
- could facilitate DNA cleavage. Moreover, these complexes showed improved biocidal activity than the free ligands against various bacterial strains such as Staphylococcus aureus, Pseudomonas aeruginosa, Escherichia coli, Staphylococcus epidermidis, and Klebsiella pneumonia. Nair et al. synthesized and
Synthesis of fluoro-functionalized diaryl-λ3-iodonium salts and their cytotoxicity against human lymphoma U937 cells
Beilstein J. Org. Chem. 2018, 14, 364–372, doi:10.3762/bjoc.14.24
- ylides, and (diacyloxyiodo)arenes were also examined for their antibacterial activities against ice nucleation active Pseudomonas syringae, and aryliodonium salts, especially those with electron-withdrawing groups, exhibit higher antibacterial activities [49]. Despite the long history of diaryliodonium
Binding abilities of polyaminocyclodextrins: polarimetric investigations and biological assays
Beilstein J. Org. Chem. 2017, 13, 2751–2763, doi:10.3762/bjoc.13.271
- resistance genes) and the emergence of multidrug resistant strains [59]. In addition, extracellular DNA has been shown to be important for biofilm establishment and maintenance by pathogenic bacteria, such as Pseudomonas aeruginosa and Staphylococcus aureus [60][61][62]. Some other bacteria, such as E. coli
What contributes to an effective mannose recognition domain?
Beilstein J. Org. Chem. 2017, 13, 2584–2595, doi:10.3762/bjoc.13.255

- glycosides on mammalian cell surfaces. After this initial contact, they can infect host cells and form biofilms, both of which are key factors for their survival [9][27][28]. Examples of such opportunistic bacterial species binding to mannosides on host cells include Pseudomonas aeruginosa with its membrane
- additional hydroxy group would not contribute the maximum penalty associated with an isolated one. The cost of desolvating calcium ions (Figure 5). Opportunistic bacteria such as Pseudomonas aeruginosa or Burkholderia cenocepacia have incorporated a second calcium ion into their binding site, coordinating
Herpetopanone, a diterpene from Herpetosiphon aurantiacus discovered by isotope labeling
Beilstein J. Org. Chem. 2017, 13, 2458–2465, doi:10.3762/bjoc.13.242
- performed as previously described [27]. The test organisms included Bacillus subtilis ATCC 6633, Staphylococcus aureus SG 511, Mycobacterium vaccae IMET 10670, Escherichia coli SG 458, Pseudomonas aeruginosa K 799/61, Sporobolomyces salmonicolor SBUG 549, Candida albicans ATCC 14053 and Penicillium notatum
Enzymatic separation of epimeric 4-C-hydroxymethylated furanosugars: Synthesis of bicyclic nucleosides
Beilstein J. Org. Chem. 2017, 13, 2078–2086, doi:10.3762/bjoc.13.205
- hydroxy groups present in different sugars and sugar moieties of synthetic or naturally occurring glycosides, nucleosides, etc. Gotor et al. [11] have reported a lipase-mediated acylation of an equimolecular mixture of D/L-thymidine with acetonoxime levulinate as acylating agent and Pseudomonas cepacia
Glycoscience@Synchrotron: Synchrotron radiation applied to structural glycoscience
Beilstein J. Org. Chem. 2017, 13, 1145–1167, doi:10.3762/bjoc.13.114

New tricks of well-known aminoazoles in isocyanide-based multicomponent reactions and antibacterial activity of the compounds synthesized
Beilstein J. Org. Chem. 2017, 13, 1050–1063, doi:10.3762/bjoc.13.104
- not been studied yet, were examined as an amine component in Ugi-4CR and GBB-3CR. The generated compounds were screened for their biological activity towards Bacillus subtilis, Staphylococcus aureus, Escherichia coli and Pseudomonas aeruginosa. Results and Discussion Since aminoazoles contain an
- -positive) and Escherichia coli (strain 1257), Pseudomonas aeruginosa (strain 1111) (Gram-negative). As it follows from the results obtained several of the substances studied inhibit the growth of test-microorganisms demonstrating a weak antimicrobial effect (Table 7). Generally, the compounds were found to
Synthesis and enzymatic ketonization of the 5-(halo)-2-hydroxymuconates and 5-(halo)-2-hydroxy-2,4-pentadienoates
Beilstein J. Org. Chem. 2017, 13, 1022–1031, doi:10.3762/bjoc.13.101
- formation of the α,β-isomer for the 5-fluoro derivative. Kinetic parameters were also obtained for both sets of compounds in the presence of 4-oxalocrotonate tautomerase (4-OT) from Pseudomonas putida mt-2 and Leptothrix cholodnii SP-6. For 5-halo-2-hydroxymuconates, there are no major differences in the
- pathway steps. This information is also useful to predict the fate of halogenated species once released into the environment. One major route for the degradation of aromatic compounds is the meta-fission pathway [6][7]. The enzymes and reactions of the meta-fission pathway in Pseudomonas putida mt-2 for
Synthesis of 1-indanones with a broad range of biological activity
Beilstein J. Org. Chem. 2017, 13, 451–494, doi:10.3762/bjoc.13.48

O-Alkylated heavy atom carbohydrate probes for protein X-ray crystallography: Studies towards the synthesis of methyl 2-O-methyl-L-selenofucopyranoside
Beilstein J. Org. Chem. 2016, 12, 2828–2833, doi:10.3762/bjoc.12.282
- glycans in pathogens or parasites, numerous other lectins recognize such O-alkylated ligands, e.g., the pilus adhesin from Pseudomonas aeruginosa PAK [31] or PapG from Escherichia coli [32]. In contrast, methylation of lectin ligands can also prevent binding, as observed with O-methylated fucose and
Chemical probes for competitive profiling of the quorum sensing signal synthase PqsD of Pseudomonas aeruginosa
Beilstein J. Org. Chem. 2016, 12, 2784–2792, doi:10.3762/bjoc.12.277

- Michaela Prothiwa David Szamosvari Sandra Glasmacher Thomas Bottcher Department of Chemistry, Konstanz Research School Chemical Biology, University of Konstanz, 78457 Konstanz, Germany 10.3762/bjoc.12.277 Abstract The human pathogen Pseudomonas aeruginosa uses the pqs quorum sensing system to
- coordinate the production of its broad spectrum of virulence factors to facilitate colonization and infection of its host. Hereby, the enzyme PqsD is a virulence related quorum sensing signal synthase that catalyzes the central step in the biosynthesis of the Pseudomonas quinolone signals HHQ and PQS. We
- for the development of customized PqsD inhibitors as well as a chemical toolbox to investigate the activity and active site specificity of the enzyme. Keywords: activity-based probes; PqsD; protein labelling; Pseudomonas aeruginosa; quinolones; Introduction The emergence of multi-drug resistant
Biosynthesis of oxygen and nitrogen-containing heterocycles in polyketides
Beilstein J. Org. Chem. 2016, 12, 1512–1550, doi:10.3762/bjoc.12.148
- clinically important antibiotic against Gram-positive bacteria, which consists of a mixture of pseudomonic acids from Pseudomonas fluorescens NCIMB 10586 with pseudomonic acid A (61) being the main compound (Scheme 9) [37][38][39][40][41][42][43][44]. It belongs to the group of trans-AT-PKS products and the
Discovery of an inhibitor of the production of the Pseudomonas aeruginosa virulence factor pyocyanin in wild-type cells
Beilstein J. Org. Chem. 2016, 12, 1428–1433, doi:10.3762/bjoc.12.137
- , UK Bioinformatics Institute, A*STAR, 30 Biopolis Street, #07-01 Matrix, Singapore 138671 10.3762/bjoc.12.137 Abstract Pyocyanin is a small molecule produced by Pseudomonas aeruginosa that plays a crucial role in the pathogenesis of infections by this notorious opportunistic pathogen. The inhibition
- pathogenicity of P. aeruginosa and there is a wide range of other potential applications where the inhibition of quorum sensing is desirable. Keywords: antibacterial; antivirulence; Pseudomonas aeruginosa; pyocyanin; quorum sensing; Findings The Gram-negative bacterium Pseudomonas aeruginosa is a clinically
- -oxododecanoyl)-L-homoserine lactone (OdDHL) and it’s cognate receptor LasR (Figure 1) [20][21][22]. Interlinking these two AHL signalling systems is a third signaling system utilising a quinolone signalling molecule (termed Pseudomonas quinolone signal, PQS) [20] to form an intricate hierarchical signaling
Muraymycin nucleoside-peptide antibiotics: uridine-derived natural products as lead structures for the development of novel antibacterial agents
Beilstein J. Org. Chem. 2016, 12, 769–795, doi:10.3762/bjoc.12.77
- activity against Gram-positive bacteria, Pseudomonas and M. tuberculosis [48]. The related liposidomycins display good activity against M. phlei, while they are not active against a range of other bacteria [45]. Mode of action To develop an effective antibiotic one needs to choose a target that is
- anti-Pseudomonas agents [115]. These Gram-negative bacteria possess an outer membrane which acts as an additional permeability barrier, making them generally less sensitive to antibacterial agents. In this context, the aforementioned muraymycin analogues (91a, 92a–h) were tested for MraY inhibitory
- activity again, with MraY enzyme from S. aureus (Table 3). However, antibacterial activities against several Pseudomonas strains were moderate to low with MICs between 8 μg/mL and >64 μg/mL. Analogue 92g was the most active congener in this series with MIC values between 8 μg/mL and 32 μg/mL. Compounds 92e
Elucidation of a masked repeating structure of the O-specific polysaccharide of the halotolerant soil bacteria Azospirillum halopraeferens Au4
Beilstein J. Org. Chem. 2016, 12, 636–642, doi:10.3762/bjoc.12.62
- -stoichiometric substituents in the OPS has been reported for phytopathogens Pseudomonas syringae [29] and Xanthomonas campestris [30] as well as for beneficial rhizobacteria, including free-living Azospirillum spp. [31][32][33][34][35] and root-nodulating Rhizobium spp. [24]. Moreover, the degree of acetylation
Biosynthesis of α-pyrones
Beilstein J. Org. Chem. 2016, 12, 571–588, doi:10.3762/bjoc.12.56

- C (57, Figure 14), which have been isolated from different Pseudomonas strains [62][63]. Compounds 55 and 56 had been initially tested positive for antimycobacterial and antiparasitic activities and both inhibited fatty acid biosynthesis [62]. The new derivative 57, possessing a longer eastern acyl
- moiety, was identified in Pseudomonas sp. GM30, and it was subsequently proven by heterologous expression experiments with ketosynthase which is responsible for the biosynthesis of these derivatives [63]. 1.3 Monobenzo-α-pyrones Synthetic derivatives of the natural product 4-hydroxycoumarin are widely
- ]. Recently, the biosynthetic origin of the pseudopyronines A (55) and B (56) in Pseudomonas putida BW11M1 was clarified – and again two chains are fused to yield the final products [86]. Thus, it can be assumed that this mechanism is exemplified quite often in natural products. Therefore, in the next
Natural products from microbes associated with insects
Beilstein J. Org. Chem. 2016, 12, 314–327, doi:10.3762/bjoc.12.34

- potent toxin that can ward of natural predators such as wolf spiders [48]. The initial isolation of pederin (3) included the collection and chemical analysis of 250,000 beetles. Later, the true producer was found to be an endosymbiotic Pseudomonas sp. within the female beetle which was identified by
Highly stable and reusable immobilized formate dehydrogenases: Promising biocatalysts for in situ regeneration of NADH
Beilstein J. Org. Chem. 2016, 12, 271–277, doi:10.3762/bjoc.12.29

- ] reported the optimal pH values were 7.0 for both free FDH and immobilized FDH onto polydopamine-coated iron oxide nanoparticles (PD-IONPs). The optimum pH values of the both free Pseudomonas sp. 101 FDH and its immobilized form onto glyoxylagarose were reported as 7.0 [16]. The temperature–activity
Recent highlights in biosynthesis research using stable isotopes
Beilstein J. Org. Chem. 2015, 11, 2493–2508, doi:10.3762/bjoc.11.271
- study the sulfur source in tropodithietic acid (TDA, 74, Scheme 13) biosynthesis [78]. TDA is a marine antibiotic which was originally isolated from Pseudomonas species [79] showing no observable resistance in important pathogens up to now [80]. The biosynthesis of the tropone core proceeds via the
Biocatalysis for the application of CO2 as a chemical feedstock
Beilstein J. Org. Chem. 2015, 11, 2370–2387, doi:10.3762/bjoc.11.259

- utilise H2 as an electron donor for the reduction of CO2 [118]. Furthermore, there is a growing list of examples of non-acetogenic metallo-FDHs, naturally catalysing formate oxidation, found to also be capable of catalysing CO2 reduction in vitro. FDH from Pseudomonas oxalaticus was the first isolated






















































































































































































































































































































































































































