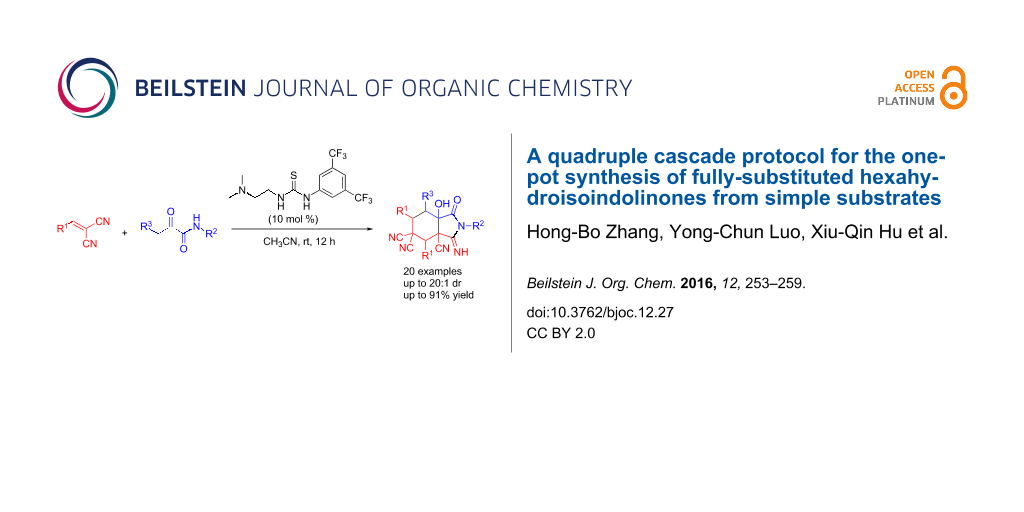
Abstract
A new and efficient synthetic method to obtain fully-substituted hexahydroisoindolinones was developed by using bifunctional tertiary amine-thioureas as powerful catalysts. As far as we know, there is no efficient synthetic method developed toward fully-substituted hexahydroisoindolinones. The products were obtained in good yield and diastereoselectivity. The one-pot cascade quadruple protocol features readily available starting materials, simple manipulation, mild conditions and good atom economy.
Graphical Abstract
Introduction
Isoindolines and their congeners are one kind of the most widespread compounds in nature. They feature not only high biological activity, but also diverse chemical properties [1-16]. Therefore, it is highly desirable to develop efficient methods toward the synthesis of isoindoline derivatives, which is a frontier in organic synthesis.
However, compared with the synthesis of their congeners, the synthesis of fully-substituted hexahydroisoindolinones is much more difficult due to the steric hindrance and the high strain of the molecular architectures [17]. Three methods to synthesize 3-substituted isoindolinones have been developed. The first method was the synthesis of 3-substituted isoindolinones from the corresponding N-methylmaleimides by the Diels–Alder reaction with 1,3-butadiene followed by hydrogenation. The second and the third methods employed the corresponding dicarboxylic acids and the carboxylic acid anhydrides, respectively [17]. To the best of our knowledge, no efficient method toward the synthesis of fully-substituted hexahydroisoindolinones has been developed so far.
The synthesis of complicated molecular structures can now be achieved by organocatalytic cascade reactions [18-33]. By simplifying the experimental procedures and reducing the usage of both solvents and reagents, one-pot reactions can improve the synthesis efficiency and both save time and reduce cost [34]. Although a few types of complicated molecules were generated through multicomponent quadruple cascade reactions, there is no report about the cascade synthesis of isoindolines in the past few decades [35-46], not mention the quadruple cascade synthesis of difficult fully-substituted hexahydroisoindolinones. Previously, we established organocatalytic domino reactions to construct very useful molecular architectures [47-60]. Based on this past experience, we decided to develop a one-pot quadruple protocol to construct this difficult molecular architecture using easily accessible substrates.
Results and Discussion
We initiated this study by using 2-benzylidenemalononitrile (1a) and 2-oxo-N,3-diphenylpropanamide (2a) [61-64] in 0.5 mL of CH3CN in the presence of 10 mol % of DABCO. After 12 h at room temperature, the reaction afforded the expected product rac-3a in 59% yield (Table 1, entry 1). We then tested different catalysts to optimize the reaction. When Et3N was used, the reaction afforded the product with 41% yield (Table 1, entry 2). However, a complex mixture was observed when DBU was used (Table 1, entry 3), while no reaction was observed when K2CO3 was used as the catalyst (Table 1, entry 4). When thioureas were used as the catalysts, we also did not get the expected product (Table 1, entries 5 and 6). Since bifunctional tertiary amine-thioureas have been proved as powerful catalysts that can catalyze a variety of organocascade reactions, we also tested thiourea catalysts, cat-1 to cat-3. Interestingly, the thioureas cat-1 and cat-2 were able to promote the reaction (Table 1, entries 7 and 8), but we obtained an even better yield when the tertiary amine-thiourea cat-3 was used as the catalyst (Table 1, entry 9). All products were racemic even when chiral catalysts were used (see Supporting Information File 1 for details). Next, we performed a solvent screening. As shown in Table 1, when DCM and THF were used as the solvent, the yield of the desired product was 33% and 34%, respectively (Table 1, entries 10 and 11). Only traces of the product were seen when toluene or methanol was used as the solvent (Table 1, entries 12 and 13). Furthermore, raising the reaction temperature was not beneficial for the diastereoselectivity of the reaction (Table 1, entry 14).
Table 1: Screening the reaction conditions.a
|
|
||||
| entry | cat. | solvent | drb | yield [%]c |
|---|---|---|---|---|
| 1 | DABCO | CH3CN | 4:1 | 59 |
| 2 | Et3N | CH3CN | 4:1 | 41 |
| 3 | DBU | CH3CN | n.d. | complex |
| 4 | K2CO3 | CH3CN | n.d. | n.r. |
| 5 | cat-1 | CH3CN | n.d. | n.r. |
| 6 | cat-2 | CH3CN | n.d. | n.r. |
| 7 | DABCOd | CH3CN | 4:1 | 62 |
| 8 | Et3Nd | CH3CN | 5:1 | 52 |
| 9 | cat-3 | CH3CN | 9:1 | 87 |
| 10 | cat-3 | DCM | 4:1 | 33 |
| 11 | cat-3 | THF | 4:1 | 34 |
| 12 | cat-3 | toluene | n.d. | trace |
| 13 | cat-3 | CH3OH | n.d. | trace |
| 14e | cat-3 | CH3CN | 6:1 | 87 |
aUnless otherwise noted, the reactions were carried out with 1a (0.25 mmol, 38.5 mg), 2a (0.1 mmol, 23.9 mg), catalyst (0.01 mmol, 10 mol %) in the indicated solvent (0.5 mL) at rt for 12 h. bDetermined by 1H NMR analysis of the crude products. cColumn chromatography yields. d10 mol % cat-2 was added. eThe reaction was carried out at 35 °C.
With the optimal conditions in hand, we next examined the reaction scope (Table 2). All reactions afforded the corresponding products 3a–t with medium to good yield and diastereoselectivity using the simple protocol at room temperature. To our delight, with our optimized reaction system, various types of substrates 1 showed very good reaction activities. Different types of substrates 1, bearing either electron withdrawing or donating groups in para-, meta- and ortho-positions, gave the desired products in good yield and diastereoselectivity (Table 2, entries 1–10 and 12), although 4-NO2C6H4 gave the product in medium yield due to its poor solubility (Table 2, entry 11). A heteroaromatic substrate such as thiophene could also be successfully employed to afford rac-3 with medium yield and diastereoselectivity (Table 2, entry 13). 3,4-Dichloro-substituted and 3,5-dimethoxy-substituted substrates produced the desired products in 84% and 55% yield with 20:1 and 15:1 diastereoselectivity, respectively (Table 2, entries 14 and 15). When substrates with different R2 and R3 were used in this reaction, the corresponding products were obtained in medium yield and diastereoselectivity (Table 2, entries 16–20). The structure of 3p was determined by X-ray analysis [65]. However, substrates with aliphatic R1, R2 or R3 did not produce the desired products (Table 2, entries 21–26).
Table 2: Substrates scope.a
|
|
|||||
| entry | R1 | R2 | R3 | drb | yield [%]c |
|---|---|---|---|---|---|
| 1 | C6H5 | C6H5 | C6H5 | 9:1 | 87 (3a) |
| 2 | 2-MeC6H4 | C6H5 | C6H5 | >20:1 | 89 (3b) |
| 3 | 3-MeC6H4 | C6H5 | C6H5 | 10:1 | 69 (3c) |
| 4 | 4-OMeC6H4 | C6H5 | C6H5 | 10:1 | 66 (3d) |
| 5 | 2-BrC6H4 | C6H5 | C6H5 | >20:1 | 84 (3e) |
| 6 | 3-ClC6H4 | C6H5 | C6H5 | 4:1 | 72 (3f) |
| 7 | 4-FC6H4 | C6H5 | C6H5 | >20:1 | 82 (3g) |
| 8 | 4-CF3C6H4 | C6H5 | C6H5 | >20:1 | 86 (3h) |
| 9 | 2-NO2C6H4 | C6H5 | C6H5 | >20:1 | 89 (3i) |
| 10 | 3-NO2C6H4 | C6H5 | C6H5 | >20:1 | 91 (3j) |
| 11 | 4-NO2C6H4 | C6H5 | C6H5 | 3:1 | 42 (3k) |
| 12 | 2-naphthalene | C6H5 | C6H5 | >20:1 | 90 (3l) |
| 13 | 2-thiophene | C6H5 | C6H5 | 3:1 | 51 (3m) |
| 14 | 3,4-diClC6H3 | C6H5 | C6H5 | >20:1 | 84 (3n) |
| 15 | 3,5-diOMeC6H3 | C6H5 | C6H5 | 15:1 | 55 (3o) |
| 16 | C6H5 | 4-OMeC6H4 | C6H5 | 4:1 | 56 (3p) |
| 17 | C6H5 | 4-ClC6H4 | C6H5 | >20:1 | 89 (3q) |
| 18 | 2-naphthalene | 4-OMeC6H4 | C6H5 | 14:1 | 88 (3r) |
| 19 | C6H5 | C6H5 | 4-MeC6H4 | 8:1 | 61 (3s) |
| 20 | C6H5 | C6H5 | 4-FC6H4 | 8:1 | 61 (3t) |
| 21 | C6H5(CH2)2 | C6H5 | C6H5 | n.d. | n.r. |
| 22 | CH3(CH2)5 | C6H5 | C6H5 | n.d. | n.r. |
| 23 | C6H5 | CH3(CH2)3 | C6H5 | n.d. | n.r. |
| 24 | C6H5 | CH3CH2 | C6H5 | n.d. | n.r. |
| 25 | C6H5 | C6H5 | H | n.d. | n.r. |
| 26 | C6H5 | C6H5 | CH3 | n.d. | n.r. |
aUnless otherwise noted, the reactions were carried out with 1 (0.25 mmol), 2 (0.1 mmol), cat-3 (3.6 mg, 0.01 mmol, 10 mol %) in CH3CN (0.5 mL) at rt for 12 h. bDetermined by 1H NMR analysis of the crude products. cColumn chromatography yields.
This bifunctional catalysis cascade reaction was also amenable to scale-up. When the reaction was carried out on a 3 mmol scale, the desired product was obtained in 84% yield. Therefore, this method is fast and easy to implement, and it is suitable for large-scale synthesis (Scheme 1).
Scheme 1: An example of scalable synthesis.
Scheme 1: An example of scalable synthesis.
Many isoindolinone skeletons show high biological potential as antihypertensives, anesthetics, etc. [66-68]. The useful hydrolyzed product rac-4a was obtained in 80% yield by treating rac-3a with trifluoroacetic anhydride in DCM (Scheme 2).
Scheme 2: Hydrolysis reaction to produce a useful product.
Scheme 2: Hydrolysis reaction to produce a useful product.
Finally, we propose a mechanism for the reaction. Initially, substrate 1 is activated by catalyst (I), which reacts with substrate 2 via two Michael addition reactions to sequentially produce II and III. Then, IV is generated from III by an aldol reaction. Finally, the product is produced after the nucleophilic reaction, and the catalyst is regenerated (Scheme 3).
Conclusion
In summary, we have developed a one-pot quadruple cascade protocol to obtain fully-substituted hexahydroisoindolinones. This new, synthetic method is simple, efficient and atom-economic. This reaction can be widely used in organic synthesis due to its advantages such as simple operation, availability of raw materials, mild conditions and high efficiency.
Experimental
General procedure for the synthesis of fully-substituted hexahydroisoindolinones
Benzylidenemalononitrile (0.1 mmol), 2-oxo-N,3-diphenylpropanamide (0.25 mmol) and cat-3 (0.01 mmol) were added to a test tube, then CH3CN (0.5 mL) was added to the mixture. The reaction mixture was stirred at 300 rpm at 21 °C in a stoppered carousel tube for 12 h. The solvent was removed in vacuo and the product was purified by silica gel flash column chromatography to give the corresponding product 3.
Supporting Information
| Supporting Information File 1: Experimental procedures, characterization data for all new compounds and X-ray analysis of compound 3. | ||
| Format: PDF | Size: 2.5 MB | Download |
References
-
Jiaang, W.-T.; Chen, Y.-S.; Hsu, T.; Wu, S.-H.; Chien, C.-H.; Chang, C.-N.; Chang, S.-P.; Lee, S.-J.; Chen, X. Bioorg. Med. Chem. Lett. 2005, 15, 687–691. doi:10.1016/j.bmcl.2004.11.023
Return to citation in text: [1] -
Van Goethem, S.; Van der Veken, P.; Dubois, V.; Soroka, A.; Lambeir, A.-M.; Chen, X.; Haemers, A.; Scharpé, S.; De Meester, I.; Augustyns, K. Bioorg. Med. Chem. Lett. 2008, 18, 4159–4162. doi:10.1016/j.bmcl.2008.05.079
Return to citation in text: [1] -
Portevin, B.; Tordjman, C.; Pastoureau, P.; Bonnet, J.; De Nanteuil, G. J. Med. Chem. 2000, 43, 4582–4593. doi:10.1021/jm990965x
Return to citation in text: [1] -
Mancilla, T.; Correa-Basurto, J.; Alavés Carbajal, K. S.; Sanchez Escalante, E. J. T.; Ferrara, J. T. J. Mex. Chem. Soc. 2007, 51, 96–102.
Return to citation in text: [1] -
Kukkola, P. J.; Bilci, N. A.; Ikeler, T. J. Tetrahedron Lett. 1996, 37, 5065–5068. doi:10.1016/0040-4039(96)01018-0
Return to citation in text: [1] -
Kukkola, P. J.; Bilci, N. A.; Ikler, T.; Savage, P.; Shetty, S. S.; DelGrande, D.; Jeng, A. Y. Bioorg. Med. Chem. Lett. 2001, 11, 1737–1740. doi:10.1016/S0960-894X(01)00273-6
Return to citation in text: [1] -
Bare, T. M.; Draper, C. W.; McLaren, C. D.; Pullan, L. M.; Patel, J.; Patel, J. B. Bioorg. Med. Chem. Lett. 1993, 3, 55–60. doi:10.1016/S0960-894X(00)80091-8
Return to citation in text: [1] -
Hamprecht, D.; Micheli, F.; Tedesco, G.; Checchia, A.; Donati, D.; Petrone, M.; Terreni, S.; Wood, M. Bioorg. Med. Chem. Lett. 2007, 17, 428–433. doi:10.1016/j.bmcl.2006.10.029
Return to citation in text: [1] -
Ewing, D. F.; Len, C.; Mackenzie, G.; Petit, J. P.; Ronco, G.; Villa, P. J. Pharm. Pharmacol. 2001, 53, 945–948. doi:10.1211/0022357011776351
Return to citation in text: [1] -
Enders, D.; Narine, A.; Toulgoat, F.; Bisschops, T. Angew. Chem., Int. Ed. 2008, 47, 5661–5665. doi:10.1002/anie.200801354
Return to citation in text: [1] -
Clary, K. N.; Parvez, M.; Back, T. G. Org. Biomol. Chem. 2009, 7, 1226–1230. doi:10.1039/b817954a
Return to citation in text: [1] -
Deniau, E.; Enders, D.; Couture, A.; Grandclaudon, P. Tetrahedron: Asymmetry 2005, 16, 875–881. doi:10.1016/j.tetasy.2005.01.012
Return to citation in text: [1] -
Lamblin, M.; Couture, A.; Deniau, E.; Grandclaudon, P. Synthesis 2006, 1333–1338. doi:10.1055/s-2006-926413
Return to citation in text: [1] -
Kobayashi, K.; Hase, M.; Hashimoto, K.; Fujita, S.; Tanmatsu, M.; Morikawa, O.; Konishi, H. Synthesis 2006, 2493–2496. doi:10.1055/s-2006-942462
Return to citation in text: [1] -
Chen, K.; Pullarkat, S. A. Org. Biomol. Chem. 2012, 10, 6600–6606. doi:10.1039/c2ob25854d
Return to citation in text: [1] -
Zhu, S.; Cao, J.; Wu, L.; Huang, X. J. Org. Chem. 2012, 77, 10409–10415. doi:10.1021/jo301437k
Return to citation in text: [1] -
Barker, M. D.; Dixon, R. A.; Jones, S.; Marsh, B. J. Tetrahedron 2006, 62, 11663–11669. doi:10.1016/j.tet.2006.09.051
Return to citation in text: [1] [2] -
Enders, D.; Grondal, C.; Hűttl, M. R. M. Angew. Chem., Int. Ed. 2007, 119, 1590–1601. doi:10.1002/ange.200603129
Angew. Chem., Int. Ed. 2007, 46, 1570–1581. doi:10.1002/anie.200603129
Return to citation in text: [1] -
Yu, X.; Wang, W. Org. Biomol. Chem. 2008, 6, 2037–2046. doi:10.1039/b800245m
Return to citation in text: [1] -
Grondal, C.; Jeanty, M.; Enders, D. Nat. Chem. 2010, 2, 167–178. doi:10.1038/nchem.539
Return to citation in text: [1] -
Moyano, A.; Rios, R. Chem. Rev. 2011, 111, 4703–4832. doi:10.1021/cr100348t
Return to citation in text: [1] -
Albrecht, Ł.; Jiang, H.; Jørgensen, K. A. Angew. Chem. 2011, 123, 8642–8660. doi:10.1002/ange.201102522
Angew. Chem., Int. Ed. 2011, 50, 8492–8509. doi:10.1002/anie.201102522
Return to citation in text: [1] -
Grossmann, A.; Enders, D. Angew. Chem. 2012, 124, 320–332. doi:10.1002/ange.201105415
Angew. Chem., Int. Ed. 2012, 51, 314–325. doi:10.1002/anie.201105415
Return to citation in text: [1] -
Lu, L.-Q.; Chen, J.-R.; Xiao, W.-J. Acc. Chem. Res. 2012, 45, 1278–1293. doi:10.1021/ar200338s
Return to citation in text: [1] -
Goudedranche, S.; Raimondi, W.; Bugaut, X.; Constantieux, T.; Bonne, D.; Rodriguez, J. Synthesis 2013, 45, 1909–1930. doi:10.1055/s-0033-1338484
Return to citation in text: [1] -
Ramachary, D. B.; Barbas, C. F., III. Chem. – Eur. J. 2004, 10, 5323–5331. doi:10.1002/chem.200400597
Return to citation in text: [1] -
Urushima, T.; Sakamoto, D.; Ishikawa, H.; Hayashi, Y. Org. Lett. 2010, 12, 4588–4591. doi:10.1021/ol1018932
Return to citation in text: [1] -
Ishikawa, H.; Sawano, S.; Yasui, Y.; Shibata, Y.; Hayashi, Y. Angew. Chem., Int. Ed. 2011, 50, 3774–3779. doi:10.1002/anie.201005386
Return to citation in text: [1] -
Roy, S.; Chen, K. Org. Lett. 2012, 14, 2496–2499. doi:10.1021/ol300783e
Return to citation in text: [1] -
Volla, C. M. R.; Atodiresei, I.; Rueping, M. Chem. Rev. 2014, 114, 2390–2431. doi:10.1021/cr400215u
Return to citation in text: [1] -
Cioc, R. C.; Ruijter, E.; Orru, R. V. A. Green Chem. 2014, 16, 2958–2975. doi:10.1039/c4gc00013g
Return to citation in text: [1] -
Pellissier, H. Adv. Synth. Catal. 2012, 354, 237–294. doi:10.1002/adsc.201100714
Return to citation in text: [1] -
Ramón, D. J.; Yus, M. Angew. Chem., Int. Ed. 2005, 44, 1602–1634. doi:10.1002/anie.200460548
Return to citation in text: [1] -
Chauhan, P.; Mahajan, S.; Kaya, U.; Hack, D.; Enders, D. Adv. Synth. Catal. 2015, 357, 253–281. doi:10.1002/adsc.201401003
Return to citation in text: [1] -
Hong, B.-C.; Kotame, P.; Liao, J.-H. Org. Biomol. Chem. 2011, 9, 382–386. doi:10.1039/C0OB00834F
Return to citation in text: [1] -
Rueping, M.; Dufour, J.; Maji, M. S. Chem. Commun. 2012, 48, 3406–3408. doi:10.1039/c2cc00129b
Return to citation in text: [1] -
Zhang, F.-L.; Xu, A.-W.; Gong, Y.-F.; Wei, M.-H.; Yang, X.-L. Chem. – Eur. J. 2009, 15, 6815–6818. doi:10.1002/chem.200900613
Return to citation in text: [1] -
Enders, D.; Wang, C.; Mukanova, M.; Greb, A. Chem. Commun. 2010, 46, 2447–2449. doi:10.1039/c002839h
Return to citation in text: [1] -
Rueping, M.; Haack, K.; Ieawsuwan, W.; Sundén, H.; Blanco, M.; Schoepke, F. R. Chem. Commun. 2011, 47, 3828–3830. doi:10.1039/c1cc10245a
Return to citation in text: [1] -
Enders, D.; Greb, A.; Deckers, K.; Selig, P.; Merkens, C. Chem. – Eur. J. 2012, 18, 10226–10229. doi:10.1002/chem.201201493
Return to citation in text: [1] -
Hong, B.-C.; Kotame, P.; Tsai, C.-W.; Liao, J.-H. Org. Lett. 2010, 12, 776–779. doi:10.1021/ol902840x
Return to citation in text: [1] -
Bertelsen, S.; Johansen, R. L.; Jørgensen, K. A. Chem. Commun. 2008, 3016–3018. doi:10.1039/b806418k
Return to citation in text: [1] -
Jiang, K.; Jia, Z.-J.; Yin, X.; Wu, L.; Chen, Y.-C. Org. Lett. 2010, 12, 2766–2769. doi:10.1021/ol100857s
Return to citation in text: [1] -
Jhuo, D.-H.; Hong, B.-C.; Chang, C.-W.; Lee, G.-H. Org. Lett. 2014, 16, 2724–2727. doi:10.1021/ol501011t
Return to citation in text: [1] -
Raja, A.; Hong, B.-C.; Lee, G.-H. Org. Lett. 2014, 16, 5756–5759. doi:10.1021/ol502821e
Return to citation in text: [1] -
Chang, Y.-P.; Gurubrahamam, R.; Chen, K. Org. Lett. 2015, 17, 2908–2911. doi:10.1021/acs.orglett.5b01040
Return to citation in text: [1] -
Tian, L.; Hu, X.-Q.; Li, Y.-H.; Xu, P.-F. Chem. Commun. 2013, 49, 7213–7215. doi:10.1039/c3cc43755h
Return to citation in text: [1] -
Gu, Y.; Wang, Y.; Yu, T.-Y.; Liang, Y.-M.; Xu, P.-F. Angew. Chem., Int. Ed. 2014, 53, 14128–14131. doi:10.1002/anie.201406786
Return to citation in text: [1] -
Wang, Y.; Yu, T.-Y.; Zhang, H.-B.; Luo, Y.-C.; Xu, P.-F. Angew. Chem., Int. Ed. 2012, 51, 12339–12342. doi:10.1002/anie.201206881
Return to citation in text: [1] -
Wang, Y.; Han, R.-G.; Zhao, Y.-L.; Yang, S.; Xu, P.-F.; Dixon, D. J. Angew. Chem., Int. Ed. 2009, 48, 9834–9838. doi:10.1002/anie.200905014
Return to citation in text: [1] -
Wang, Y.; Yu, D.-F.; Liu, Y.-Z.; Wei, H.; Luo, Y.-C.; Dixon, D. J.; Xu, P.-F. Chem. – Eur. J. 2010, 16, 3922–3925. doi:10.1002/chem.201000059
Return to citation in text: [1] -
Jia, Z.-X.; Luo, Y.-C.; Xu, P.-F. Org. Lett. 2011, 13, 832–835. doi:10.1021/ol103069d
Return to citation in text: [1] -
Tian, L.; Xu, G.-Q.; Li, Y.-H.; Liang, Y.-M.; Xu, P.-F. Chem. Commun. 2014, 50, 2428–2430. doi:10.1039/c3cc49504c
Return to citation in text: [1] -
Jia, Z.-X.; Luo, Y.-C.; Wang, Y.; Chen, L.; Xu, P.-F.; Wang, B. Chem. – Eur. J. 2012, 18, 12958–12961. doi:10.1002/chem.201201362
Return to citation in text: [1] -
Wang, Y.; Luo, Y.-C.; Hu, X.-Q.; Xu, P.-F. Org. Lett. 2011, 13, 5346–5349. doi:10.1021/ol2022092
Return to citation in text: [1] -
Zhao, Y.-L.; Wang, Y.; Cao, J.; Liang, Y.-M.; Xu, P.-F. Org. Lett. 2014, 16, 2438–2441. doi:10.1021/ol5008185
Return to citation in text: [1] -
Zhao, S.; Lin, J.-B.; Zhao, Y.-Y.; Liang, Y.-M.; Xu, P.-F. Org. Lett. 2014, 16, 1802–1805. doi:10.1021/ol500547e
Return to citation in text: [1] -
Lu, H.; Lin, J.-B.; Liu, J.-Y.; Xu, P.-F. Chem. – Eur. J. 2014, 20, 11659–11663. doi:10.1002/chem.201402947
Return to citation in text: [1] -
Gao, T.-P.; Lin, J.-B.; Hu, X.-Q.; Xu, P.-F. Chem. Commun. 2014, 50, 8934–8936. doi:10.1039/C4CC03896G
Return to citation in text: [1] -
Wang, Y.; Lu, H.; Xu, P.-F. Acc. Chem. Res. 2015, 48, 1832–1844. doi:10.1021/acs.accounts.5b00217
Return to citation in text: [1] -
Goudedranche, S.; Pierrot, D.; Constantieux, T.; Bonne, D.; Rodriguez, J. Chem. Commun. 2014, 50, 15605–15608. doi:10.1039/C4CC07731H
Return to citation in text: [1] -
Wang, L.; Ni, Q.; Blümel, M.; Shu, T.; Raabe, G.; Enders, D. Chem. – Eur. J. 2015, 21, 8033–8037. doi:10.1002/chem.201500661
Return to citation in text: [1] -
Joie, C.; Deckers, K.; Enders, D. Synthesis 2014, 46, 799–808. doi:10.1055/s-0033-1340565
Return to citation in text: [1] -
Joie, C.; Deckers, K.; Raabe, G.; Enders, D. Synthesis 2014, 46, 1539–1546. doi:10.1055/s-0033-1340982
Return to citation in text: [1] -
CCDC 1404942 (3p, minor) and CCDC 1409146 (3p, major) contains the supplementary crystallographic data for this paper. These data can be obtained free of charge from The Cambridge Crystallographic Data Centre via http://www.ccdc.cam.ac.uk/data_request/cif.
Return to citation in text: [1] -
Comins, D. L.; Schilling, S.; Zhang, Y. Org. Lett. 2005, 7, 95–98. doi:10.1021/ol047824w
Return to citation in text: [1] -
Comins, D. L.; Hiebel, A.-C. Tetrahedron Lett. 2005, 46, 5639–5642. doi:10.1016/j.tetlet.2005.06.105
Return to citation in text: [1] -
Chitanda, J. M.; Prokopchuk, D. E.; Quail, J. W.; Foley, S. R. Organometallics 2008, 27, 2337–2345. doi:10.1021/om800080e
Return to citation in text: [1]
| 1. | Jiaang, W.-T.; Chen, Y.-S.; Hsu, T.; Wu, S.-H.; Chien, C.-H.; Chang, C.-N.; Chang, S.-P.; Lee, S.-J.; Chen, X. Bioorg. Med. Chem. Lett. 2005, 15, 687–691. doi:10.1016/j.bmcl.2004.11.023 |
| 2. | Van Goethem, S.; Van der Veken, P.; Dubois, V.; Soroka, A.; Lambeir, A.-M.; Chen, X.; Haemers, A.; Scharpé, S.; De Meester, I.; Augustyns, K. Bioorg. Med. Chem. Lett. 2008, 18, 4159–4162. doi:10.1016/j.bmcl.2008.05.079 |
| 3. | Portevin, B.; Tordjman, C.; Pastoureau, P.; Bonnet, J.; De Nanteuil, G. J. Med. Chem. 2000, 43, 4582–4593. doi:10.1021/jm990965x |
| 4. | Mancilla, T.; Correa-Basurto, J.; Alavés Carbajal, K. S.; Sanchez Escalante, E. J. T.; Ferrara, J. T. J. Mex. Chem. Soc. 2007, 51, 96–102. |
| 5. | Kukkola, P. J.; Bilci, N. A.; Ikeler, T. J. Tetrahedron Lett. 1996, 37, 5065–5068. doi:10.1016/0040-4039(96)01018-0 |
| 6. | Kukkola, P. J.; Bilci, N. A.; Ikler, T.; Savage, P.; Shetty, S. S.; DelGrande, D.; Jeng, A. Y. Bioorg. Med. Chem. Lett. 2001, 11, 1737–1740. doi:10.1016/S0960-894X(01)00273-6 |
| 7. | Bare, T. M.; Draper, C. W.; McLaren, C. D.; Pullan, L. M.; Patel, J.; Patel, J. B. Bioorg. Med. Chem. Lett. 1993, 3, 55–60. doi:10.1016/S0960-894X(00)80091-8 |
| 8. | Hamprecht, D.; Micheli, F.; Tedesco, G.; Checchia, A.; Donati, D.; Petrone, M.; Terreni, S.; Wood, M. Bioorg. Med. Chem. Lett. 2007, 17, 428–433. doi:10.1016/j.bmcl.2006.10.029 |
| 9. | Ewing, D. F.; Len, C.; Mackenzie, G.; Petit, J. P.; Ronco, G.; Villa, P. J. Pharm. Pharmacol. 2001, 53, 945–948. doi:10.1211/0022357011776351 |
| 10. | Enders, D.; Narine, A.; Toulgoat, F.; Bisschops, T. Angew. Chem., Int. Ed. 2008, 47, 5661–5665. doi:10.1002/anie.200801354 |
| 11. | Clary, K. N.; Parvez, M.; Back, T. G. Org. Biomol. Chem. 2009, 7, 1226–1230. doi:10.1039/b817954a |
| 12. | Deniau, E.; Enders, D.; Couture, A.; Grandclaudon, P. Tetrahedron: Asymmetry 2005, 16, 875–881. doi:10.1016/j.tetasy.2005.01.012 |
| 13. | Lamblin, M.; Couture, A.; Deniau, E.; Grandclaudon, P. Synthesis 2006, 1333–1338. doi:10.1055/s-2006-926413 |
| 14. | Kobayashi, K.; Hase, M.; Hashimoto, K.; Fujita, S.; Tanmatsu, M.; Morikawa, O.; Konishi, H. Synthesis 2006, 2493–2496. doi:10.1055/s-2006-942462 |
| 15. | Chen, K.; Pullarkat, S. A. Org. Biomol. Chem. 2012, 10, 6600–6606. doi:10.1039/c2ob25854d |
| 16. | Zhu, S.; Cao, J.; Wu, L.; Huang, X. J. Org. Chem. 2012, 77, 10409–10415. doi:10.1021/jo301437k |
| 34. | Chauhan, P.; Mahajan, S.; Kaya, U.; Hack, D.; Enders, D. Adv. Synth. Catal. 2015, 357, 253–281. doi:10.1002/adsc.201401003 |
| 18. |
Enders, D.; Grondal, C.; Hűttl, M. R. M. Angew. Chem., Int. Ed. 2007, 119, 1590–1601. doi:10.1002/ange.200603129
Angew. Chem., Int. Ed. 2007, 46, 1570–1581. doi:10.1002/anie.200603129 |
| 19. | Yu, X.; Wang, W. Org. Biomol. Chem. 2008, 6, 2037–2046. doi:10.1039/b800245m |
| 20. | Grondal, C.; Jeanty, M.; Enders, D. Nat. Chem. 2010, 2, 167–178. doi:10.1038/nchem.539 |
| 21. | Moyano, A.; Rios, R. Chem. Rev. 2011, 111, 4703–4832. doi:10.1021/cr100348t |
| 22. |
Albrecht, Ł.; Jiang, H.; Jørgensen, K. A. Angew. Chem. 2011, 123, 8642–8660. doi:10.1002/ange.201102522
Angew. Chem., Int. Ed. 2011, 50, 8492–8509. doi:10.1002/anie.201102522 |
| 23. |
Grossmann, A.; Enders, D. Angew. Chem. 2012, 124, 320–332. doi:10.1002/ange.201105415
Angew. Chem., Int. Ed. 2012, 51, 314–325. doi:10.1002/anie.201105415 |
| 24. | Lu, L.-Q.; Chen, J.-R.; Xiao, W.-J. Acc. Chem. Res. 2012, 45, 1278–1293. doi:10.1021/ar200338s |
| 25. | Goudedranche, S.; Raimondi, W.; Bugaut, X.; Constantieux, T.; Bonne, D.; Rodriguez, J. Synthesis 2013, 45, 1909–1930. doi:10.1055/s-0033-1338484 |
| 26. | Ramachary, D. B.; Barbas, C. F., III. Chem. – Eur. J. 2004, 10, 5323–5331. doi:10.1002/chem.200400597 |
| 27. | Urushima, T.; Sakamoto, D.; Ishikawa, H.; Hayashi, Y. Org. Lett. 2010, 12, 4588–4591. doi:10.1021/ol1018932 |
| 28. | Ishikawa, H.; Sawano, S.; Yasui, Y.; Shibata, Y.; Hayashi, Y. Angew. Chem., Int. Ed. 2011, 50, 3774–3779. doi:10.1002/anie.201005386 |
| 29. | Roy, S.; Chen, K. Org. Lett. 2012, 14, 2496–2499. doi:10.1021/ol300783e |
| 30. | Volla, C. M. R.; Atodiresei, I.; Rueping, M. Chem. Rev. 2014, 114, 2390–2431. doi:10.1021/cr400215u |
| 31. | Cioc, R. C.; Ruijter, E.; Orru, R. V. A. Green Chem. 2014, 16, 2958–2975. doi:10.1039/c4gc00013g |
| 32. | Pellissier, H. Adv. Synth. Catal. 2012, 354, 237–294. doi:10.1002/adsc.201100714 |
| 33. | Ramón, D. J.; Yus, M. Angew. Chem., Int. Ed. 2005, 44, 1602–1634. doi:10.1002/anie.200460548 |
| 17. | Barker, M. D.; Dixon, R. A.; Jones, S.; Marsh, B. J. Tetrahedron 2006, 62, 11663–11669. doi:10.1016/j.tet.2006.09.051 |
| 17. | Barker, M. D.; Dixon, R. A.; Jones, S.; Marsh, B. J. Tetrahedron 2006, 62, 11663–11669. doi:10.1016/j.tet.2006.09.051 |
| 65. | CCDC 1404942 (3p, minor) and CCDC 1409146 (3p, major) contains the supplementary crystallographic data for this paper. These data can be obtained free of charge from The Cambridge Crystallographic Data Centre via http://www.ccdc.cam.ac.uk/data_request/cif. |
| 61. | Goudedranche, S.; Pierrot, D.; Constantieux, T.; Bonne, D.; Rodriguez, J. Chem. Commun. 2014, 50, 15605–15608. doi:10.1039/C4CC07731H |
| 62. | Wang, L.; Ni, Q.; Blümel, M.; Shu, T.; Raabe, G.; Enders, D. Chem. – Eur. J. 2015, 21, 8033–8037. doi:10.1002/chem.201500661 |
| 63. | Joie, C.; Deckers, K.; Enders, D. Synthesis 2014, 46, 799–808. doi:10.1055/s-0033-1340565 |
| 64. | Joie, C.; Deckers, K.; Raabe, G.; Enders, D. Synthesis 2014, 46, 1539–1546. doi:10.1055/s-0033-1340982 |
| 47. | Tian, L.; Hu, X.-Q.; Li, Y.-H.; Xu, P.-F. Chem. Commun. 2013, 49, 7213–7215. doi:10.1039/c3cc43755h |
| 48. | Gu, Y.; Wang, Y.; Yu, T.-Y.; Liang, Y.-M.; Xu, P.-F. Angew. Chem., Int. Ed. 2014, 53, 14128–14131. doi:10.1002/anie.201406786 |
| 49. | Wang, Y.; Yu, T.-Y.; Zhang, H.-B.; Luo, Y.-C.; Xu, P.-F. Angew. Chem., Int. Ed. 2012, 51, 12339–12342. doi:10.1002/anie.201206881 |
| 50. | Wang, Y.; Han, R.-G.; Zhao, Y.-L.; Yang, S.; Xu, P.-F.; Dixon, D. J. Angew. Chem., Int. Ed. 2009, 48, 9834–9838. doi:10.1002/anie.200905014 |
| 51. | Wang, Y.; Yu, D.-F.; Liu, Y.-Z.; Wei, H.; Luo, Y.-C.; Dixon, D. J.; Xu, P.-F. Chem. – Eur. J. 2010, 16, 3922–3925. doi:10.1002/chem.201000059 |
| 52. | Jia, Z.-X.; Luo, Y.-C.; Xu, P.-F. Org. Lett. 2011, 13, 832–835. doi:10.1021/ol103069d |
| 53. | Tian, L.; Xu, G.-Q.; Li, Y.-H.; Liang, Y.-M.; Xu, P.-F. Chem. Commun. 2014, 50, 2428–2430. doi:10.1039/c3cc49504c |
| 54. | Jia, Z.-X.; Luo, Y.-C.; Wang, Y.; Chen, L.; Xu, P.-F.; Wang, B. Chem. – Eur. J. 2012, 18, 12958–12961. doi:10.1002/chem.201201362 |
| 55. | Wang, Y.; Luo, Y.-C.; Hu, X.-Q.; Xu, P.-F. Org. Lett. 2011, 13, 5346–5349. doi:10.1021/ol2022092 |
| 56. | Zhao, Y.-L.; Wang, Y.; Cao, J.; Liang, Y.-M.; Xu, P.-F. Org. Lett. 2014, 16, 2438–2441. doi:10.1021/ol5008185 |
| 57. | Zhao, S.; Lin, J.-B.; Zhao, Y.-Y.; Liang, Y.-M.; Xu, P.-F. Org. Lett. 2014, 16, 1802–1805. doi:10.1021/ol500547e |
| 58. | Lu, H.; Lin, J.-B.; Liu, J.-Y.; Xu, P.-F. Chem. – Eur. J. 2014, 20, 11659–11663. doi:10.1002/chem.201402947 |
| 59. | Gao, T.-P.; Lin, J.-B.; Hu, X.-Q.; Xu, P.-F. Chem. Commun. 2014, 50, 8934–8936. doi:10.1039/C4CC03896G |
| 60. | Wang, Y.; Lu, H.; Xu, P.-F. Acc. Chem. Res. 2015, 48, 1832–1844. doi:10.1021/acs.accounts.5b00217 |
| 35. | Hong, B.-C.; Kotame, P.; Liao, J.-H. Org. Biomol. Chem. 2011, 9, 382–386. doi:10.1039/C0OB00834F |
| 36. | Rueping, M.; Dufour, J.; Maji, M. S. Chem. Commun. 2012, 48, 3406–3408. doi:10.1039/c2cc00129b |
| 37. | Zhang, F.-L.; Xu, A.-W.; Gong, Y.-F.; Wei, M.-H.; Yang, X.-L. Chem. – Eur. J. 2009, 15, 6815–6818. doi:10.1002/chem.200900613 |
| 38. | Enders, D.; Wang, C.; Mukanova, M.; Greb, A. Chem. Commun. 2010, 46, 2447–2449. doi:10.1039/c002839h |
| 39. | Rueping, M.; Haack, K.; Ieawsuwan, W.; Sundén, H.; Blanco, M.; Schoepke, F. R. Chem. Commun. 2011, 47, 3828–3830. doi:10.1039/c1cc10245a |
| 40. | Enders, D.; Greb, A.; Deckers, K.; Selig, P.; Merkens, C. Chem. – Eur. J. 2012, 18, 10226–10229. doi:10.1002/chem.201201493 |
| 41. | Hong, B.-C.; Kotame, P.; Tsai, C.-W.; Liao, J.-H. Org. Lett. 2010, 12, 776–779. doi:10.1021/ol902840x |
| 42. | Bertelsen, S.; Johansen, R. L.; Jørgensen, K. A. Chem. Commun. 2008, 3016–3018. doi:10.1039/b806418k |
| 43. | Jiang, K.; Jia, Z.-J.; Yin, X.; Wu, L.; Chen, Y.-C. Org. Lett. 2010, 12, 2766–2769. doi:10.1021/ol100857s |
| 44. | Jhuo, D.-H.; Hong, B.-C.; Chang, C.-W.; Lee, G.-H. Org. Lett. 2014, 16, 2724–2727. doi:10.1021/ol501011t |
| 45. | Raja, A.; Hong, B.-C.; Lee, G.-H. Org. Lett. 2014, 16, 5756–5759. doi:10.1021/ol502821e |
| 46. | Chang, Y.-P.; Gurubrahamam, R.; Chen, K. Org. Lett. 2015, 17, 2908–2911. doi:10.1021/acs.orglett.5b01040 |
| 66. | Comins, D. L.; Schilling, S.; Zhang, Y. Org. Lett. 2005, 7, 95–98. doi:10.1021/ol047824w |
| 67. | Comins, D. L.; Hiebel, A.-C. Tetrahedron Lett. 2005, 46, 5639–5642. doi:10.1016/j.tetlet.2005.06.105 |
| 68. | Chitanda, J. M.; Prokopchuk, D. E.; Quail, J. W.; Foley, S. R. Organometallics 2008, 27, 2337–2345. doi:10.1021/om800080e |
© 2016 Zhang et al; licensee Beilstein-Institut.
This is an Open Access article under the terms of the Creative Commons Attribution License (http://creativecommons.org/licenses/by/2.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
The license is subject to the Beilstein Journal of Organic Chemistry terms and conditions: (http://www.beilstein-journals.org/bjoc)