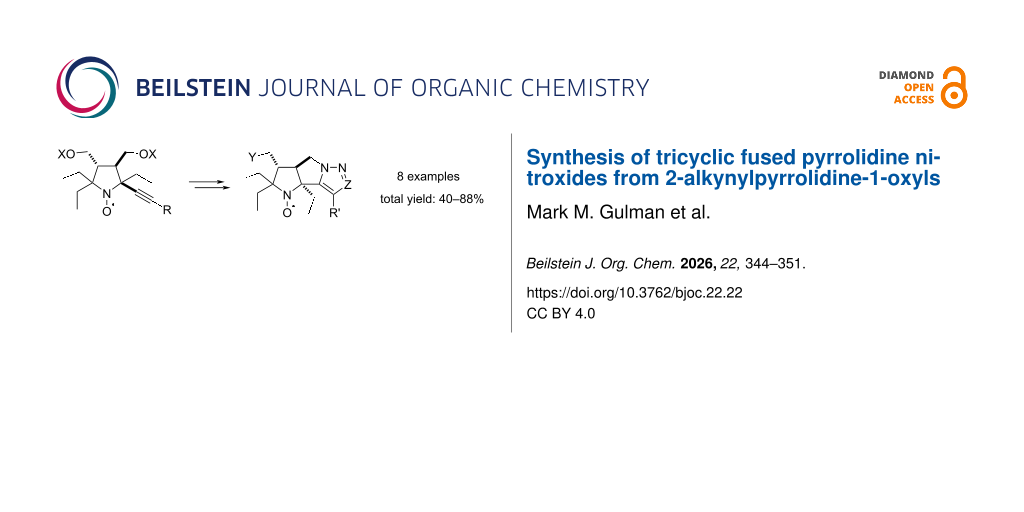
Abstract
Rotational correlation time is a key parameter for organic radical contrast agents (ORCA) for magnetic resonance imaging (MRI). Design of polycyclic systems with incorporated nitroxide moieties in which rotation of the radical separately from the framework is impossible is one of the ways to improve properties of ORCA. Feasibility of the synthesis of rigid 3b,4,5,6,6a,7-hexahydropyrrolo[2',3':3,4]pyrrolo[1,2-c][1,2,3]triazole and 3b,4,5,6,6a,7-hexahydropyrrolo[2',3':3,4]pyrrolo[1,2-b]pyrazole ring systems with incorporated nitroxide moiety from 2-alkynyl-substituted pyrrolidine nitroxides was studied. These nitroxides have been prepared via intramolecular Huisgen cycloaddition or intramolecular alkylation in 2-pyrazolyl derivatives prepared by Michael addition–cyclocondensation of the corresponding alkynones with hydrazine. The reduction kinetics by ascorbate showed that the formation of the rigid tricyclic framework does not lead to a significant increase in stability of the radical center to chemical reduction.
Graphical Abstract
Introduction
Stable nitroxides are functional components of many high-tech materials, such as energy storage and organoelectronics devices [1-8], catalysts [9,10], bioactive coatings and nanoparticles [11,12], organic radical contrast agents (ORCAs) for magnetic resonance imaging (MRI) [13,14], etc. For these applications, numerous nitroxides are incorporated into macromolecular or nanosized supramolecular structures, which modulate nitroxide properties. For example, the efficiency of ORCA (relaxivity) directly depends on the rotational correlation time of the radicals attached to the scaffold [13-15]. Large structures in which rotation of the radical separately from the framework is impossible could be particularly promising. Rigid polycyclic fused systems with incorporated nitroxide moieties could be one of the possible ways to achieve the above feature.
We recently found that the reaction of sterically hindered 3-hydroxymethyl-2-ethynylpyrrolidine-1-oxyls with nucleophilic agents can lead to the formation of condensed systems involving the substituent at position 3 of the pyrrolidine ring [16]. Alkynes are broadly used in the synthesis of various heterocyclic compounds, and participation of neighboring functional groups often leads to formation of complex polycyclic systems [17-19]. In this study, we aimed to construct rigid tricyclic condensed systems with an integrated nitroxyl radical fragment from 3-substituted 2-ethynylpyrrolidine-1-oxyls. The desired tricyclic nitroxides were prepared via intramolecular Huisgen cycloaddition or via one-pot Michael addition–cyclocondensation reaction with hydrazine with subsequent intramolecular alkylation of the resulting pyrazoles.
Results and Discussion
Synthesis
We have earlier reported on the synthesis of 2-alkynylpyrrolidine-1-oxyls 2a–c via addition of the corresponding alkynylmagnesium bromides to nitrone 1 [20]. Radicals 2d and 2e were prepared in analogy to the known procedure using trimethylsilylacetylene and benzyl propargyl ether as the terminal alkynes (Scheme 1). Nitrone 1 was treated with a 10-fold excess of alkynylmagnesium bromide prepared in situ via metalation of trimethylsilylacetylene or benzyl propargyl ether with ethylmagnesium bromide. After quenching, removal of the MOP protecting group, and oxidation by atmospheric oxygen the nitroxides 2d and 2e were isolated in 64% and 66% yields, respectively.
Terminal alkynes can be converted into propargylamines via A3-coupling reaction [21]. In analogy to a literature procedure [22], heating of radical 2a in a mixture of dimethylamine, formalin, and tetrahydrofuran in the presence of copper(II) acetate afforded the corresponding dimethylamino derivative 2f.
To confirm the structure of the novel nitroxides 2d–f, the samples of the radicals were reduced to the corresponding diamagnetic amines using a Zn/CF3COOH system in CD3OD at 63 °C, according to a literature protocol [23] and the 1H NMR spectra were recorded. The spectra showed similarity to those of previously described for radicals 2a–c [20] with characteristic signals of (2R)-2,5,5-triethyl-3,4-bis(hydroxymethyl)pyrrolidines. An additional singlet at 0.21 ppm in the spectrum of 2d was assigned to the hydrogen atoms of the trimethylsilyl group. The spectrum of compound 2e displayed two singlets at 4.33 ppm and 4.62 ppm (2H each), assigned to the methylene groups of the propargyl and benzyl fragments, respectively, and a multiplet of phenyl group in the range of 7.30–7.40 ppm (5H). Successful aminomethylation of the ethynyl group was confirmed by the appearance of two singlets at 3.01 ppm (6H) and 4.28 ppm (2H), attributed to the protons of the dimethylamino group and the methylene protons of the propargyl moiety. The structure of nitroxide 2d was confirmed by single crystal X-ray analysis. (Figure 1, CCDC 2512649).
![[1860-5397-22-22-1]](/bjoc/content/figures/1860-5397-22-22-1.png?scale=2.0&max-width=1024&background=FFFFFF)
Figure 1: X-ray structure of nitroxide 2d.
Figure 1: X-ray structure of nitroxide 2d.
The nitroxides 2a–f were used to synthesize tricyclic nitroxides 4a–f. The mesylation was carried out in the presence of DIPEA in chloroform under reflux (Scheme 2). These conditions ensured complete conversion in 30 minutes.
Scheme 2: Synthesis of mesyl 3a–f and triazole 4a–f derivatives.
Scheme 2: Synthesis of mesyl 3a–f and triazole 4a–f derivatives.
The IR spectra of nitroxides 3a–f exhibit intense absorption bands in the ranges of 1354–1358 cm−1 and 1174–1178 cm−1, corresponding to the asymmetric and symmetric vibrations of the sulfonate group, respectively [24]. The 1H NMR spectra of 3a,b,d–f (Zn/CF3COOH system in CD3OD) showed appearance of a singlet of methanesulfonate hydrogens in the region from 2.78 to 3.17 ppm. The NMR spectra were not recorded for 3c because of heavy resinification upon the sample preparation.
The literature data on reactivity of 5-azidopentyne derivatives in intramolecular Huisgen cycloaddition reactions are contradictory. Some authors successfully obtained 5-azidopentyne derivatives upon nucleophilic substitution at 80 °C in DMF, and additional heating at 170 °C was necessary for cyclization to triazoles to occur [25]. However, there are also examples where the corresponding triazoles were isolated instead of 5-azidopentyne derivatives under the same conditions (DMF, 80 °C) [26].
The nitroxides 3a–f were treated with excess of NaN3 in milder conditions, in DMSO at 60 °C, and a single product was isolated from the reaction mixtures in each case. The IR spectra of the isolated compounds 4a–f showed characteristic absorption bands to the azido group vibrations at 2100–2116 cm−1. Absorption bands in the ranges of 1410–1420 cm−1 and 1170–1190 cm−1 were also observed, which can be assigned to the out-of-plane bending (wagging, ω) and breathing vibrations of the triazole ring, respectively [27]. The X-band EPR spectra of nitroxides 4a–f revealed remarkable difference with those of 3a–f (see Table 1). In analogy to previously reported parameters for 2a–c [20], EPR spectra of trans-3,4-disubstituted 2,2,5-triethyl-5-ethynylpyrrolidine-1-oxyls 2d–f and 3a–f correspond to triplet of doublets pattern with hfc aN = 1.53–1.58 mT at the nitrogen atom of nitroxide group and an additional hfc aH = 0.21–0.23 mT at one of the methylene hydrogens of the ethyl groups [20,28]. The spectra of tricyclic nitroxides 4a–f are characterized with smaller triplet splitting, aN = 1.43–1.46 mT, while hfc on hydrogen reach 0.26–0.31 mT, with the exception of 4b (0.22 mT). The structures of nitroxides 4a, 4b, and 4c were confirmed by single crystal X-ray crystallographic analysis (Figure 2, CCDC 2512650–2512652).
Table 1: Reduction rate constants k2 and EPR spectral parameters of nitroxides 2a–f, 4a–f, 9c.
| Nitroxide |
k2∙101,
M−1s−1 |
aN, mT
(±0.005) |
aH, mT
(±0.005) |
Line width, mT
(±0.001) |
| 2a | 2.38 ± 0.01 | 1.53 | 0.23 | 0.006 |
| 2b | 1.86 ± 0.05 | 1.53 | 0.21 | 0.006 |
| 2c | 1.35 ± 0.05 | 1.54 | 0.21 | 0.006 |
| 2d | 2.50 ± 0.05 | 1.53 | 0.21 | 0.006 |
| 2e | 1.46 ± 0.06 | 1.53 | 0.21 | 0.006 |
| 2f | 2.11 ± 0.05 | 1.53 | 0.23 | 0.006 |
| 4a | 3.13 ± 0.02 | 1.47 | 0.26 | 0.007 |
| 4b | 1.71 ± 0.06 | 1.45 | 0.22 | 0.007 |
| 4c | 3.53 ± 0.05 | 1.45 | 0.26 | 0.007 |
| 4d | 4.39 ± 0.10 | 1.45 | 0.27 | 0.007 |
| 4e | 0.81 ± 0.01 | 1.43 | 0.26 | 0.007 |
| 4f | 1.32 ± 0.01 | 1.46 | 0.31 | 0.006 |
| 9с | 0.85 ± 0.01 | 1.49 | 0.25 | 0.007 |
![[1860-5397-22-22-2]](/bjoc/content/figures/1860-5397-22-22-2.png?scale=2.0&max-width=1024&background=FFFFFF)
Figure 2: X-ray structures of nitroxides 4a–c.
Figure 2: X-ray structures of nitroxides 4a–c.
Similar rigid condensed systems can be constructed on the basis of pyrazole ring. The reaction of conjugated alkynones with hydrazine is a common pathway to pyrazoles [29]. Alkynones, in turn, can be prepared by Sonogashira acylation of terminal alkynes [30]. To prevent formation of mixtures due to incomplete benzoylation of hydroxymethyl groups under the conditions of Sonogashira cross-coupling, nitroxide 2a was treated with acetic anhydride in the presence of sodium acetate, yielding the diacyl derivative 5. Radicals 5 and 3a were heated with benzoyl chloride and triethylamine in toluene in the presence of a catalytic system comprising PPh3, CuI, and Pd(PPh3)2Cl2. This procedure afforded alkynones 6a,b in the yields of 75% and 44%, respectively (Scheme 3).
In the IR spectra of 6a,b intense bands were observed at 2212–2214 and 1645–1647 cm−1, assigned to vibrations of the triple bond, and the conjugated carbonyl group, respectively. The elemental analyses data and high-resolution mass spectra (HRMS) of 6a,b were in agreement with the assigned structure.
Oxidation of propargyl alcohols is another way to α,β-acetylenic carbonyl compounds [31]. Mild oxidation of propargyl alcohol 2c with activated manganese dioxide in tetrahydrofuran gave vinyl ether 8, which was isolated with 50% yield (Scheme 4). The structure of the product 8 was confirmed by X-ray crystallographic analysis (Figure 3, CCDC 2512653). Formation of 8 apparently occurs via cyclization of alkynal 7. The formation of vinyl ethers has been previously described for radicals 2a and 2b, but these cyclizations required heating in the presence of a base [16].
![[1860-5397-22-22-3]](/bjoc/content/figures/1860-5397-22-22-3.png?scale=2.0&max-width=1024&background=FFFFFF)
Figure 3: X-ray structures of nitroxides 8 and 9a.
Figure 3: X-ray structures of nitroxides 8 and 9a.
To obtain pyrazole derivatives, alkynones 6a,b were treated with hydrazine (Scheme 5). Formation of the pyrazole ring in the reaction of 6b was accompanied by partial hydrazinolysis of the acetoxy groups and reduction of the nitroxyl fragment to the corresponding hydroxylamine. Primary products were stirred with a solution of NaOH in methanol under aerobic conditions to achieve complete hydrolysis and regeneration of the nitroxide group. Pyrazole 9a was isolated with an overall yield of 33%, and its structure was confirmed by single crystal X-ray crystallographic analysis (Figure 3, CCDC 2512654).
Scheme 5: Synthesis of pyrazole derivatives 9a–c.
Scheme 5: Synthesis of pyrazole derivatives 9a–c.
Treatment of 6b with hydrazine resulted in the formation of a condensed tricyclic ring system and nitroxide 9b was isolated in 42% yield. This nitroxide was alternatively prepared via mesylation of 9a. The yield of 9b in this reaction depends on excess of the reagent. Heating of 9a with five equivalents of MsCl and excess of DIPEA afforded 9b in 65% yield. When a 1.5-fold excess of mesyl chloride per hydroxy group was used, another nitroxide 9c was isolated in 26% yield along with 9b (43% yield).
The IR spectra of the pyrazole derivatives 9a–c exhibit absorptions at 688–699 cm−1 and 763–770 cm−1, assigned to out-of-plane bending modes of the pyrazole ring [32]. The 1H NMR spectra of the reduction products of pyrazoles 9a–c were recorded after reduction using the Zn/CF3COOH system in CD3OD. The full spectral line shapes were simulated using the gNMR program to assign the signals and to obtain the coupling constants [33]. Direct comparison of the spin–spin coupling constants within the six-spin systems of the non-annulated nitroxide 9a with its annulated counterparts 9b,c revealed reduced dynamic averaging for one of the two methylene groups in 9b,c. The similar values of constants J(1,3), J(2,3), J(4,5), and J(4,6) in 9a reflect rapid, nearly degenerate rotation of its exocyclic methylene groups. In contrast, the disparate values of J(4,5) and J(4,6) in nitroxides 9b,c demonstrate that this methylene fragment has a more fixed conformation, consistent with the annulated structure (Figure 4).
Figure 4: Spin–spin coupling constants for reduced nitroxides 9a–c.
Figure 4: Spin–spin coupling constants for reduced nitroxides 9a–c.
Nitroxides are known to decay in biological systems; the major mechanism is chemical reduction with cellular antioxidants (ascorbic acid and glutathione) and enzymatic systems [34]. The rate of reduction is an important factor for application of a nitroxide as an EPR spin probe or as a component of a MRI contrast agent. The bimolecular rate constants of reduction of radicals 2a–f, 4a–f and 9c with ascorbate were measured with addition of the glutathione system to suppress reverse reaction [35]. The results are given in Table 1. Alkynyl and heteroaromatic derivatives showed no significant differences in the reduction kinetics with the values of the reduction rate constants (0.08–0.44 M−1s−1) comparable in scale to those reported for tetramethyl nitroxides of the pyrrolidine and pyrroline series (0.1–0.3 M−1s−1). These rates are more than two orders of magnitude higher than those of tetraethyl nitroxides of the pyrrolidine series (0.001–0.0001 M−1s−1) [28,29]. This could result from the electron-withdrawing effect of alkyne or heteroaromatic substituent. Moreover, it can be noted that rigid tricyclic systems apparently do not have a screening effect on the radical center.
Conclusion
In this work we showed that formation of rigid tricyclic annulated systems from bifunctional nitroxides bearing alkynyl and another functional group in neighboring positions is feasible. The new tricyclic nitroxyl radicals of the pyrrolidine series annulated with 5,6-dihydro-4H-pyrrolo[1,2-c][1,2,3]triazole or 5,6-dihydro-4H-pyrrolo[1,2-b]pyrazole systems have been synthesized and characterized. Despite these nitroxides showed relatively low stability to reduction, the general strategy suggested here can be applied for the synthesis of various polycyclic systems with incorporated nitroxide.
Acknowledgements
The authors thank the personnel of the Multi-Access Chemical Research Centre SB RAS for having recorded IR, UV, NMR and HRMS spectra, and for having carried out the elemental analysis.
Funding
The work was carried out with support from the Russian Science Foundation within Project No. 23-13-00178, https://rscf.ru/project/23-13-00178/.
Data Availability Statement
All data that supports the findings of this study is available in the published article and/or the supporting information of this article.
References
-
Hansen, K.-A.; Blinco, J. P. Polym. Chem. 2018, 9, 1479–1516. doi:10.1039/c7py02001e
Return to citation in text: [1] -
Chamberland, J. P.; Parent, J. S.; Barz, D. P. J. Chem. Eng. J. 2025, 512, 162051. doi:10.1016/j.cej.2025.162051
Return to citation in text: [1] -
Wilcox, D. A.; Agarkar, V.; Mukherjee, S.; Boudouris, B. W. Annu. Rev. Chem. Biomol. Eng. 2018, 9, 83–103. doi:10.1146/annurev-chembioeng-060817-083945
Return to citation in text: [1] -
Zhang, K.; Lin, X.; Shi, Y.; Oyaizu, K.; Jia, Z. ACS Electrochem. 2025, 1, 123–137. doi:10.1021/acselectrochem.4c00119
Return to citation in text: [1] -
Ji, L.; Shi, J.; Wei, J.; Yu, T.; Huang, W. Adv. Mater. (Weinheim, Ger.) 2020, 32, 1908015. doi:10.1002/adma.201908015
Return to citation in text: [1] -
Xie, Y.; Zhang, K.; Yamauchi, Y.; Oyaizu, K.; Jia, Z. Mater. Horiz. 2021, 8, 803–829. doi:10.1039/d0mh01391a
Return to citation in text: [1] -
Friebe, C.; Schubert, U. S. Top. Curr. Chem. 2017, 375, 19. doi:10.1007/s41061-017-0103-1
Return to citation in text: [1] -
Janoschka, T.; Hager, M. D.; Schubert, U. S. Adv. Mater. (Weinheim, Ger.) 2012, 24, 6397–6409. doi:10.1002/adma.201203119
Return to citation in text: [1] -
Ciriminna, R.; Pagliaro, M. Org. Process Res. Dev. 2010, 14, 245–251. doi:10.1021/op900059x
Return to citation in text: [1] -
Cao, Q.; Dornan, L. M.; Rogan, L.; Hughes, N. L.; Muldoon, M. J. Chem. Commun. 2014, 50, 4524–4543. doi:10.1039/c3cc47081d
Return to citation in text: [1] -
Woehlk, H.; Trimble, M. J.; Mansour, S. C.; Pletzer, D.; Trouillet, V.; Welle, A.; Barner, L.; Hancock, R. E. W.; Barner-Kowollik, C.; Fairfull-Smith, K. E. Polym. Chem. 2019, 10, 4252–4258. doi:10.1039/c9py00690g
Return to citation in text: [1] -
Shashni, B.; Nagasaki, Y. Biomaterials 2018, 178, 48–62. doi:10.1016/j.biomaterials.2018.05.042
Return to citation in text: [1] -
Luo, T.; Wang, B.; Chen, R.; Qi, Q.; Wu, R.; Xie, S.; Chen, H.; Han, J.; Wu, D.; Cao, S. J. Mater. Chem. B 2025, 13, 372–398. doi:10.1039/d4tb02272f
Return to citation in text: [1] [2] -
Guo, S.; Wang, X.; Li, Z.; Pan, D.; Dai, Y.; Ye, Y.; Tian, X.; Gu, Z.; Gong, Q.; Zhang, H.; Luo, K. J. Nanobiotechnol. 2021, 19, 244. doi:10.1186/s12951-021-00990-6
Return to citation in text: [1] [2] -
Laurent, S.; Vander Elst, L.; Muller, R. N. eMagRes 2009. doi:10.1002/9780470034590.emrstm1049
Return to citation in text: [1] -
Gulman, M. M.; Dobrynin, S. A.; Gatilov, Y. V.; Kirilyuk, I. A. Chem. Sustainable Dev. 2024, 32, 460–466.
Return to citation in text: [1] [2] -
Godoi, B.; Schumacher, R. F.; Zeni, G. Chem. Rev. 2011, 111, 2937–2980. doi:10.1021/cr100214d
Return to citation in text: [1] -
Yamamoto, Y. Chem. Soc. Rev. 2014, 43, 1575–1600. doi:10.1039/c3cs60369e
Return to citation in text: [1] -
Neto, J. S. S.; Zeni, G. Tetrahedron 2020, 76, 130876. doi:10.1016/j.tet.2019.130876
Return to citation in text: [1] -
Dobrynin, S. A.; Gulman, M. M.; Morozov, D. A.; Zhurko, I. F.; Taratayko, A. I.; Sotnikova, Y. S.; Glazachev, Y. I.; Gatilov, Y. V.; Kirilyuk, I. A. Molecules 2022, 27, 7626. doi:10.3390/molecules27217626
Return to citation in text: [1] [2] [3] [4] -
Jesin, I.; Nandi, G. C. Eur. J. Org. Chem. 2019, 2704–2720. doi:10.1002/ejoc.201900001
Return to citation in text: [1] -
Mukusheva, G. K.; Toigambekova, N. N.; Savelyev, V. A.; Khlebnikov, A. I.; Burova, L. G.; Afanaseva, S. D.; Nurkenov, O. A.; Kishkentayeva, A. S.; Olzhabayeva, A. S.; Gatilov, Y. V.; Seidakhmetova, R. B.; Evstropov, A. N.; Shults, E. E. Molecules 2025, 30, 4352. doi:10.3390/molecules30224352
Return to citation in text: [1] -
Dobrynin, S. A.; Usatov, M. S.; Zhurko, I. F.; Morozov, D. A.; Polienko, Y. F.; Glazachev, Y. I.; Parkhomenko, D. A.; Tyumentsev, M. A.; Gatilov, Y. V.; Chernyak, E. I.; Bagryanskaya, E. G.; Kirilyuk, I. A. Molecules 2021, 26, 5761. doi:10.3390/molecules26195761
Return to citation in text: [1] -
Pretsch, E.; Bühlmann, P.; Badertscher, M. Structure Determination of Organic Compounds: Tables of Spectral Data; Springer: Berlin, Heidelberg, Germany, 2009. doi:10.1007/978-3-540-93810-1
Return to citation in text: [1] -
Yoshida, Y.; Takizawa, S.; Sasai, H. Tetrahedron Lett. 2011, 52, 6877–6879. doi:10.1016/j.tetlet.2011.09.152
Return to citation in text: [1] -
Coelho, A.; Machado-Rodrigues, C.; Behr, J.-B.; Vasse, J.-L. Org. Lett. 2021, 23, 772–776. doi:10.1021/acs.orglett.0c03998
Return to citation in text: [1] -
Borello, E.; Zecchina, A.; Guglielminotti, E. J. Chem. Soc. B 1969, 307. doi:10.1039/j29690000307
Return to citation in text: [1] -
Polienko, Y. F.; Dobrynin, S. A.; Lomanovich, K. A.; Brovko, A. O.; Bagryanskaya, E. G.; Kirilyuk, I. A. ACS Omega 2023, 8, 38723–38732. doi:10.1021/acsomega.3c06090
Return to citation in text: [1] [2] -
Götzinger, A. C.; Theßeling, F. A.; Hoppe, C.; Müller, T. J. J. J. Org. Chem. 2016, 81, 10328–10338. doi:10.1021/acs.joc.6b01326
Return to citation in text: [1] [2] -
Tohda, Y.; Sonogashira, K.; Hagihara, N. Synthesis 1977, 777–778. doi:10.1055/s-1977-24574
Return to citation in text: [1] -
Gritter, R. J.; Wallace, T. J. J. Org. Chem. 1959, 24, 1051–1056. doi:10.1021/jo01090a006
Return to citation in text: [1] -
Krishnakumar, V.; Jayamani, N.; Mathammal, R. Spectrochim. Acta, Part A 2011, 79, 1959–1968. doi:10.1016/j.saa.2011.05.100
Return to citation in text: [1] -
Rummey, J. M.; Boyce, M. C. J. Chem. Educ. 2004, 81, 762. doi:10.1021/ed081p762
Return to citation in text: [1] -
Kocherginsky, N.; Swartz, H. Nitroxide Spin Labels: Reactions in Biology and Chemistry; CRC Press: Boca Raton, FL, USA, 1995.
Return to citation in text: [1] -
Bobko, A. A.; Kirilyuk, I. A.; Grigor’ev, I. A.; Zweier, J. L.; Khramtsov, V. V. Free Radical Biol. Med. 2007, 42, 404–412. doi:10.1016/j.freeradbiomed.2006.11.007
Return to citation in text: [1]
| 1. | Hansen, K.-A.; Blinco, J. P. Polym. Chem. 2018, 9, 1479–1516. doi:10.1039/c7py02001e |
| 2. | Chamberland, J. P.; Parent, J. S.; Barz, D. P. J. Chem. Eng. J. 2025, 512, 162051. doi:10.1016/j.cej.2025.162051 |
| 3. | Wilcox, D. A.; Agarkar, V.; Mukherjee, S.; Boudouris, B. W. Annu. Rev. Chem. Biomol. Eng. 2018, 9, 83–103. doi:10.1146/annurev-chembioeng-060817-083945 |
| 4. | Zhang, K.; Lin, X.; Shi, Y.; Oyaizu, K.; Jia, Z. ACS Electrochem. 2025, 1, 123–137. doi:10.1021/acselectrochem.4c00119 |
| 5. | Ji, L.; Shi, J.; Wei, J.; Yu, T.; Huang, W. Adv. Mater. (Weinheim, Ger.) 2020, 32, 1908015. doi:10.1002/adma.201908015 |
| 6. | Xie, Y.; Zhang, K.; Yamauchi, Y.; Oyaizu, K.; Jia, Z. Mater. Horiz. 2021, 8, 803–829. doi:10.1039/d0mh01391a |
| 7. | Friebe, C.; Schubert, U. S. Top. Curr. Chem. 2017, 375, 19. doi:10.1007/s41061-017-0103-1 |
| 8. | Janoschka, T.; Hager, M. D.; Schubert, U. S. Adv. Mater. (Weinheim, Ger.) 2012, 24, 6397–6409. doi:10.1002/adma.201203119 |
| 13. | Luo, T.; Wang, B.; Chen, R.; Qi, Q.; Wu, R.; Xie, S.; Chen, H.; Han, J.; Wu, D.; Cao, S. J. Mater. Chem. B 2025, 13, 372–398. doi:10.1039/d4tb02272f |
| 14. | Guo, S.; Wang, X.; Li, Z.; Pan, D.; Dai, Y.; Ye, Y.; Tian, X.; Gu, Z.; Gong, Q.; Zhang, H.; Luo, K. J. Nanobiotechnol. 2021, 19, 244. doi:10.1186/s12951-021-00990-6 |
| 15. | Laurent, S.; Vander Elst, L.; Muller, R. N. eMagRes 2009. doi:10.1002/9780470034590.emrstm1049 |
| 26. | Coelho, A.; Machado-Rodrigues, C.; Behr, J.-B.; Vasse, J.-L. Org. Lett. 2021, 23, 772–776. doi:10.1021/acs.orglett.0c03998 |
| 13. | Luo, T.; Wang, B.; Chen, R.; Qi, Q.; Wu, R.; Xie, S.; Chen, H.; Han, J.; Wu, D.; Cao, S. J. Mater. Chem. B 2025, 13, 372–398. doi:10.1039/d4tb02272f |
| 14. | Guo, S.; Wang, X.; Li, Z.; Pan, D.; Dai, Y.; Ye, Y.; Tian, X.; Gu, Z.; Gong, Q.; Zhang, H.; Luo, K. J. Nanobiotechnol. 2021, 19, 244. doi:10.1186/s12951-021-00990-6 |
| 27. | Borello, E.; Zecchina, A.; Guglielminotti, E. J. Chem. Soc. B 1969, 307. doi:10.1039/j29690000307 |
| 11. | Woehlk, H.; Trimble, M. J.; Mansour, S. C.; Pletzer, D.; Trouillet, V.; Welle, A.; Barner, L.; Hancock, R. E. W.; Barner-Kowollik, C.; Fairfull-Smith, K. E. Polym. Chem. 2019, 10, 4252–4258. doi:10.1039/c9py00690g |
| 12. | Shashni, B.; Nagasaki, Y. Biomaterials 2018, 178, 48–62. doi:10.1016/j.biomaterials.2018.05.042 |
| 24. | Pretsch, E.; Bühlmann, P.; Badertscher, M. Structure Determination of Organic Compounds: Tables of Spectral Data; Springer: Berlin, Heidelberg, Germany, 2009. doi:10.1007/978-3-540-93810-1 |
| 9. | Ciriminna, R.; Pagliaro, M. Org. Process Res. Dev. 2010, 14, 245–251. doi:10.1021/op900059x |
| 10. | Cao, Q.; Dornan, L. M.; Rogan, L.; Hughes, N. L.; Muldoon, M. J. Chem. Commun. 2014, 50, 4524–4543. doi:10.1039/c3cc47081d |
| 25. | Yoshida, Y.; Takizawa, S.; Sasai, H. Tetrahedron Lett. 2011, 52, 6877–6879. doi:10.1016/j.tetlet.2011.09.152 |
| 21. | Jesin, I.; Nandi, G. C. Eur. J. Org. Chem. 2019, 2704–2720. doi:10.1002/ejoc.201900001 |
| 23. | Dobrynin, S. A.; Usatov, M. S.; Zhurko, I. F.; Morozov, D. A.; Polienko, Y. F.; Glazachev, Y. I.; Parkhomenko, D. A.; Tyumentsev, M. A.; Gatilov, Y. V.; Chernyak, E. I.; Bagryanskaya, E. G.; Kirilyuk, I. A. Molecules 2021, 26, 5761. doi:10.3390/molecules26195761 |
| 20. | Dobrynin, S. A.; Gulman, M. M.; Morozov, D. A.; Zhurko, I. F.; Taratayko, A. I.; Sotnikova, Y. S.; Glazachev, Y. I.; Gatilov, Y. V.; Kirilyuk, I. A. Molecules 2022, 27, 7626. doi:10.3390/molecules27217626 |
| 20. | Dobrynin, S. A.; Gulman, M. M.; Morozov, D. A.; Zhurko, I. F.; Taratayko, A. I.; Sotnikova, Y. S.; Glazachev, Y. I.; Gatilov, Y. V.; Kirilyuk, I. A. Molecules 2022, 27, 7626. doi:10.3390/molecules27217626 |
| 17. | Godoi, B.; Schumacher, R. F.; Zeni, G. Chem. Rev. 2011, 111, 2937–2980. doi:10.1021/cr100214d |
| 18. | Yamamoto, Y. Chem. Soc. Rev. 2014, 43, 1575–1600. doi:10.1039/c3cs60369e |
| 19. | Neto, J. S. S.; Zeni, G. Tetrahedron 2020, 76, 130876. doi:10.1016/j.tet.2019.130876 |
| 16. | Gulman, M. M.; Dobrynin, S. A.; Gatilov, Y. V.; Kirilyuk, I. A. Chem. Sustainable Dev. 2024, 32, 460–466. |
| 22. | Mukusheva, G. K.; Toigambekova, N. N.; Savelyev, V. A.; Khlebnikov, A. I.; Burova, L. G.; Afanaseva, S. D.; Nurkenov, O. A.; Kishkentayeva, A. S.; Olzhabayeva, A. S.; Gatilov, Y. V.; Seidakhmetova, R. B.; Evstropov, A. N.; Shults, E. E. Molecules 2025, 30, 4352. doi:10.3390/molecules30224352 |
| 29. | Götzinger, A. C.; Theßeling, F. A.; Hoppe, C.; Müller, T. J. J. J. Org. Chem. 2016, 81, 10328–10338. doi:10.1021/acs.joc.6b01326 |
| 20. | Dobrynin, S. A.; Gulman, M. M.; Morozov, D. A.; Zhurko, I. F.; Taratayko, A. I.; Sotnikova, Y. S.; Glazachev, Y. I.; Gatilov, Y. V.; Kirilyuk, I. A. Molecules 2022, 27, 7626. doi:10.3390/molecules27217626 |
| 20. | Dobrynin, S. A.; Gulman, M. M.; Morozov, D. A.; Zhurko, I. F.; Taratayko, A. I.; Sotnikova, Y. S.; Glazachev, Y. I.; Gatilov, Y. V.; Kirilyuk, I. A. Molecules 2022, 27, 7626. doi:10.3390/molecules27217626 |
| 28. | Polienko, Y. F.; Dobrynin, S. A.; Lomanovich, K. A.; Brovko, A. O.; Bagryanskaya, E. G.; Kirilyuk, I. A. ACS Omega 2023, 8, 38723–38732. doi:10.1021/acsomega.3c06090 |
| 35. | Bobko, A. A.; Kirilyuk, I. A.; Grigor’ev, I. A.; Zweier, J. L.; Khramtsov, V. V. Free Radical Biol. Med. 2007, 42, 404–412. doi:10.1016/j.freeradbiomed.2006.11.007 |
| 28. | Polienko, Y. F.; Dobrynin, S. A.; Lomanovich, K. A.; Brovko, A. O.; Bagryanskaya, E. G.; Kirilyuk, I. A. ACS Omega 2023, 8, 38723–38732. doi:10.1021/acsomega.3c06090 |
| 29. | Götzinger, A. C.; Theßeling, F. A.; Hoppe, C.; Müller, T. J. J. J. Org. Chem. 2016, 81, 10328–10338. doi:10.1021/acs.joc.6b01326 |
| 33. | Rummey, J. M.; Boyce, M. C. J. Chem. Educ. 2004, 81, 762. doi:10.1021/ed081p762 |
| 34. | Kocherginsky, N.; Swartz, H. Nitroxide Spin Labels: Reactions in Biology and Chemistry; CRC Press: Boca Raton, FL, USA, 1995. |
| 16. | Gulman, M. M.; Dobrynin, S. A.; Gatilov, Y. V.; Kirilyuk, I. A. Chem. Sustainable Dev. 2024, 32, 460–466. |
| 32. | Krishnakumar, V.; Jayamani, N.; Mathammal, R. Spectrochim. Acta, Part A 2011, 79, 1959–1968. doi:10.1016/j.saa.2011.05.100 |
| 30. | Tohda, Y.; Sonogashira, K.; Hagihara, N. Synthesis 1977, 777–778. doi:10.1055/s-1977-24574 |
| 31. | Gritter, R. J.; Wallace, T. J. J. Org. Chem. 1959, 24, 1051–1056. doi:10.1021/jo01090a006 |
© 2026 Gulman et al.; licensee Beilstein-Institut.
This is an open access article licensed under the terms of the Beilstein-Institut Open Access License Agreement (https://www.beilstein-journals.org/bjoc/terms), which is identical to the Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by/4.0). The reuse of material under this license requires that the author(s), source and license are credited. Third-party material in this article could be subject to other licenses (typically indicated in the credit line), and in this case, users are required to obtain permission from the license holder to reuse the material.