Abstract
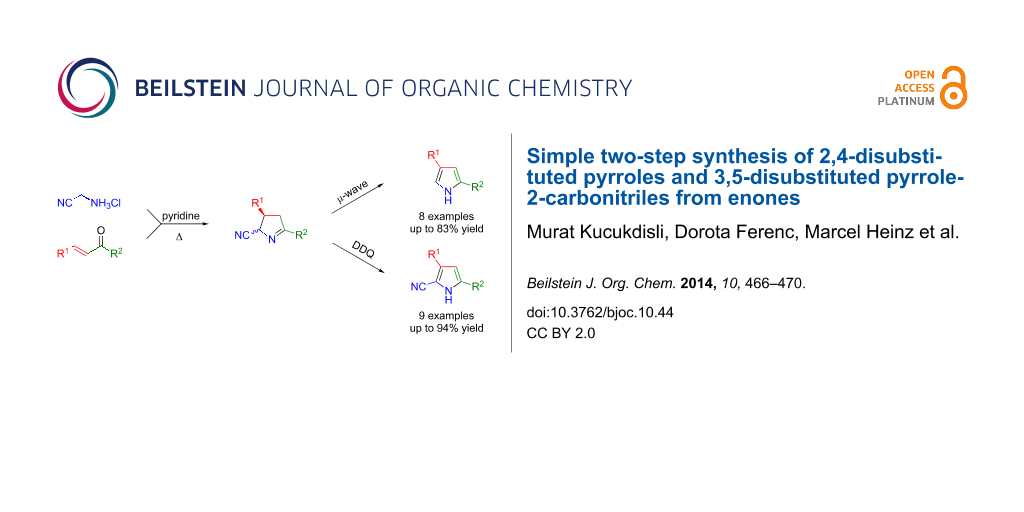
The cyclocondensation of enones with aminoacetonitrile furnishes 3,4-dihydro-2H-pyrrole-2-carbonitriles which can be readily converted to 2,4-disubstituted pyrroles by microwave-induced dehydrocyanation. Alternatively, oxidation of the intermediates produces 3,5-disubstituted pyrrole-2-carbonitriles.
Graphical Abstract
Introduction
Heterocycles are the largest class of organic compounds [1]. Among them, pyrroles have a distinguished position in the chemistry of living organisms due to their close biogenetic connection to the porphyrins, the chlorins, and the corrins. Furthermore, they are regarded as privileged structures by synthetic chemists because of widespread applications in medicinal chemistry [2,3] and materials science [4]. Not surprisingly, numerous synthetic methods [5-27] are still being developed for this compound class although many classical methods such as Hantzsch [28], Knorr [29], Paal–Knorr [30], and Barton–Zard [31] have already found their way into the textbooks of organic chemistry.
α-(Alkylideneamino)nitriles can be used as versatile and readily accessible pronucleophiles, e.g. for the construction of γ-amino acids [32], pyrrolidines [33], as well as pyrroles [34,35]. If an additional conjugated double bond is present, the anions of α-(alkylideneamino)nitriles 3 can undergo an electrocyclic ring closure to furnish 3,4-dihydro-2H-pyrrole-2-carbonitriles 6 after reprotonation [36]. If the products are devoid of an additional substituent in 2-position, the cyclocondensation can be simply effected by refluxing a mixture of the enone 1 and aminoacetonitrile hydrochloride (2) in pyridine. The base-induced dehydrocyanation of compounds 6 does not produce appreciable yields of pyrroles 7 since the abstraction of the most acidic proton leads to a stable anion reluctant to undergo α-elimination of cyanide. Instead, the anions 8 can be added in vinylogous fashion to α,β-unsaturated carbonyls producing intermediates, the exhaustive reduction of which leads to highly substituted pyrrolizidines 9 (Scheme 1).
Scheme 1: Synthesis and conversion of 3,4-dihydro-2H-pyrrole-2-carbonitriles 6.
Scheme 1: Synthesis and conversion of 3,4-dihydro-2H-pyrrole-2-carbonitriles 6.
Results and Discussion
In view of the lacking reactivity of compounds 6 towards base-induced dehydrocyanation to the 2,4-disubstituted pyrroles 7, the reaction was attempted on substrate 6a (R1 = R2 = Ph) under acidic conditions. Several strong and weak acids were screened at temperatures ranging from 0–150 °C. The formation of pyrrole 7a was observed in acetic acid under microwave heating but many byproducts were formed under these conditions, presumably due to oligo- and polymerization of the product. Gratifyingly, yield and purity of pyrrole 7a increased when 6a was heated with microwaves to 250 °C under air cooling of the reaction vessel without any additive under solvent-free conditions. The results of the application of these conditions to cyanopyrrolines 6a–j are summarized in Table 1.
Table 1: Microwave-assisted synthesis of 2,4-disubstituted pyrroles 7a–i.
|
|
|||||
| Entry | R1 | R2 | Yield of 6 (%) | Yield of 7 (%) | Product |
|---|---|---|---|---|---|
| 1 | Ph | Ph | 64a | 81 | 7a |
| 2 | Ph | 2-Naph | 90a | 83 | 7b |
| 3 | Ph | Me | 49a | 43 | 7c |
| 4 | 3-NO2-C6H4 | 4-Cl-C6H4 | 90a | n.r. | 7d |
| 5 | 2,3-Cl2C6H3 | Ph | 82 | 66 | 7e |
| 6 | 2-Br-C6H4 | 2-Naph | 77a | 28 | 7f |
| 7 | 4-CN-C6H4 | Ph | 66b | 32 | 7g |
| 8 | 4-MeO-C6H4 | 4-F-C6H4 | 53a | 61 | 7h |
| 9 | 2-Cl-C6H4 | 4-F-C6H4 | 77a | 38 | 7i |
| 10 | 3,5-(CH3)2C6H3 | Ph | 54 | not tested | – |
aSee [36].bWhen repeated under identical conditions, a higher yield was obtained than reported in [36] (53%).
This compound class comprises important starting materials for the preparation of the BODIPY dyes (4,4-difluoro-4-bora-3a,4a-diaza-s-indacenes) [37,38] as well as their aza-analogues [39], the 4,4-difluoro-4-bora-3a,4a,8-triaza-s-indacenes. Various methods to prepare 2,4-disubstituted pyrroles have been described in the literature. They can be obtained from enones by Michael addition of nitromethane, partial reduction and dehydrogenation of the resulting pyrroline with selenium or sulfur in moderate yields (3 steps) [40,41]. Alternatively, a Nef reaction of the nitromethane adducts gives masked 1,4-dicarbonyls which can be cyclized to compounds 7 in yields of up to 50% (over 4 steps) [42]. Other methods involve the use of stoichiometric zirconocene dichloride [43], the reductive coupling of alkynes to enones followed by ozonolysis and Paal–Knorr cyclization [21] or the reaction of vinylazides with aldehydes or ketones [23]. The most practical approach to a 2,4-disubstituted pyrrole reported to date is the recently disclosed microwave-assisted Stetter reaction of chalcone with carbohydrates as “green” formaldehyde equivalents followed by a microwave-assisted Paal–Knorr cyclization of the resulting 1,4-dicarbonyl [44]. Compared to this elegant strategy, our two-step protocol provides a higher overall yield of compound 7a and uses less expensive reagents.
In addition to the thermal elimination of HCN, intermediates 6 can also be aromatized by dehydrogenation. In this case, the nitrile group remains in the products and 3,5-disubstituted pyrrole-2-carbonitriles 10 are obtained. Tsuge et al. reported the oxidation of pyrroline-3-carboxylates with chloranil [45] while pyrrole-2-carboxylates were obtained from the oxidation of pyrroline-2-carboxylate with chloranil [46,47] or DDQ [48]. Different oxidants were screened for the oxidation of cyanopyrroline 6a to pyrrole 10a and the results were judged by TLC. While chloranil, MnO2, NBS/DBU, and iodine/DBU in toluene or chlorobenzene did not give any conversion, bromotrichloromethane/DBU and DDQ gave positive results. The latter was found to be more suitable since the use of bromotrichloromethane/DBU resulted in many side reactions. Table 2 summarizes the results of the oxidation of 6a–j with DDQ in toluene under reflux.
Table 2: Pyrrole-2-carbonitriles 10a–j.
|
|
||||
| Entry | R1 | R2 | Product | Yield (%) |
|---|---|---|---|---|
| 1 | Ph | Ph | 10a | 94 |
| 2 | Ph | 2-Naph | 10b | 59 |
| 3 | Ph | Me | 10c | – |
| 4 | 3-NO2-C6H4 | 4-Cl-C6H4 | 10d | 65 |
| 5 | 2,3-Cl2-C6H3 | Ph | 10e | 71 |
| 6 | 2-Br-C6H4 | 2-Naph | 10f | 47 |
| 7 | 4-CN-C6H4 | Ph | 10g | 70 |
| 8 | 4-MeO-C6H4 | 4-F-C6H4 | 10h | 77 |
| 9 | 2-Cl-C6H4 | 4-F-C6H4 | 10i | 71 |
| 10 | 3,5-(CH3)2-C6H3 | Ph | 10j | 54 |
The reaction worked smoothly with aryl-substituted compounds. The only failure was observed in case of compound 6c which possesses an alkyl substituent (Table 2, entry 3). As autoxidation and oxidative dealkylation of 2-alkylpyrroles can even be effected by exposure to air [49,50], the lability of 10c towards DDQ oxidation is not very surprising.
Conclusion
Two simple two-step procedures have been developed to convert enones to 2,4-disubstiuted pyrroles and 3,5-disubstituted pyrrole-2-carbonitriles by means of a cyclocondensation with aminoacetonitrile and a microwave-assisted dehydrocyanation or a dehydrogenation with DDQ. These methods provide a simple an efficient entry into these useful compound classes.
Experimental
Typical procedure for the synthesis of cyanopyrrolines 6a–j: The cyanopyrrolines 6a–j were prepared according to an earlier publication [36]. As an illustrative example, compound 6e is prepared as follows: To a solution of (E)-3-(2,3-dichlorophenyl)-1-phenylprop-2-en-1-one (2.20 g, 7.94 mmol, 1e) in pyridine (40 mL) was added aminoacetonitrile hydrochloride (1.10 g, 11.76 mmol, 1.48 equiv, 2). The suspension was heated to reflux for 15 h. The mixture was cooled down, diluted with ethyl acetate, washed with saturated aqueous NaHCO3, and dried over anhydrous Na2SO4. All volatiles were removed in vacuo and the crude product was purified by column chromatography (ethyl acetate/cyclohexane 1:7) to obtain cis/trans-6e (2.05 g, 6.50 mmol, 82%) as a yellow oil.
General procedure for the synthesis of 2,4-disubstituted pyrroles 7a–i: A solution of cyanopyrroline 6a–i in dichloromethane was transferred into a microwave reaction vessel. After removing the solvent in vacuo, the vessel was flushed with argon and closed with a cap. It was irradiated for 30 min (Pmax 180 W, pressure limit 7 bar, temperatures indicated in Supporting Information File 1) in a monomode microwave apparatus under air cooling. The pressurized vessel was opened very carefully inside a well-ventilated hood (caution, hydrogen cyanide!) and the residue was purified by column chromatography.
General procedure for the synthesis of pyrrole-2-carbonitriles 10a–j: A round bottomed flask equipped with a magnetic stir bar was charged with cyanopyrroline 6a–j and DDQ (1.15–1.20 equiv) in toluene (15–20 mL/mmol 6). The reaction mixture was stirred under reflux until the starting material was consumed (TLC, 2–4 h). It was diluted with ethyl acetate and washed with 10% aqueous NaOH. The extracts were dried over MgSO4 and concentrated in vacuo to obtain the crude product which was purified by column chromatography.
Supporting Information
| Supporting Information File 1: Detailed experimental procedures and characterization of compounds 6e, 6j, 7a–i and 10a–j including 1H and 13C NMR spectra. | ||
| Format: PDF | Size: 2.8 MB | Download |
References
-
Gulevich, A. V.; Dudnik, A. S.; Chernyak, N.; Gevorgyan, V. Chem. Rev. 2013, 113, 3084–3213. doi:10.1021/cr300333u
Return to citation in text: [1] -
Sessler, J. L.; Camiolo, S.; Gale, P. A. Coord. Chem. Rev. 2003, 240, 17–55. doi:10.1016/S0010-8545(03)00023-7
Return to citation in text: [1] -
Srivastava, S. K.; Miller, C. N.; Aceto, M. D.; Traynor, J. R.; Lewis, J. W.; Husbands, S. M. J. Med. Chem. 2004, 47, 6645–6648. doi:10.1021/jm040817t
Return to citation in text: [1] -
Groenendaal, L.; Meijer, E. W.; Vekemans, J. A. J. M. In Electronic Materials: The Oligomer Approach; Müllen, K.; Wegner, G., Eds.; Wiley-VCH Verlag GmbH: Weinheim, 2007; pp 235–275.
Return to citation in text: [1] -
Braun, R. U.; Zeitler, K.; Müller, T. J. J. Org. Lett. 2001, 3, 3297–3300. doi:10.1021/ol0165185
Return to citation in text: [1] -
Kel'in, A. V.; Sromek, A. W.; Gevorgyan, V. J. Am. Chem. Soc. 2001, 123, 2074–2075. doi:10.1021/ja0058684
Return to citation in text: [1] -
Fan, X.; Zhang, Y. Tetrahedron Lett. 2002, 43, 1863–1865. doi:10.1016/S0040-4039(02)00108-9
Return to citation in text: [1] -
Rao, H. S. P.; Jothilingam, S.; Scheeren, H. W. Tetrahedron 2004, 60, 1625–1630. doi:10.1016/j.tet.2003.11.087
Return to citation in text: [1] -
Zeng, D. X.; Chen, Y. Synlett 2006, 490–492. doi:10.1055/s-2006-932452
Return to citation in text: [1] -
Lu, L.; Chen, G.; Ma, S. Org. Lett. 2006, 8, 835–838. doi:10.1021/ol052797a
Return to citation in text: [1] -
Bellina, F.; Rossi, R. Tetrahedron 2006, 62, 7213–7256. doi:10.1016/j.tet.2006.05.024
Return to citation in text: [1] -
Kim, Y.; Kim, J.; Park, S. B. Org. Lett. 2009, 11, 17–20. doi:10.1021/ol8022193
Return to citation in text: [1] -
Kiren, S.; Hong, X.; Leverett, C. A.; Padwa, A. Org. Lett. 2009, 11, 1233–1235. doi:10.1021/ol900059e
Return to citation in text: [1] -
Aponick, A.; Li, C.-Y.; Malinge, J.; Marques, E. F. Org. Lett. 2009, 11, 4624–4627. doi:10.1021/ol901901m
Return to citation in text: [1] -
Merkul, E.; Boersch, C.; Frank, W.; Muller, T. J. J. Org. Lett. 2009, 11, 2269–2272. doi:10.1021/ol900581a
Return to citation in text: [1] -
Kramer, S.; Madsen, J. L. H.; Rottländer, M.; Skrydstrup, T. Org. Lett. 2010, 12, 2758–2761. doi:10.1021/ol1008685
Return to citation in text: [1] -
Donohoe, T. J.; Race, N. J.; Bower, J. F.; Callens, C. K. A. Org. Lett. 2010, 12, 4094–4097. doi:10.1021/ol101681r
Return to citation in text: [1] -
Rakshit, S.; Patureau, F. W.; Glorius, F. J. Am. Chem. Soc. 2010, 132, 9585–9587. doi:10.1021/ja104305s
Return to citation in text: [1] -
Estévez, V.; Villacampa, M.; Menéndez, J. C. Chem. Soc. Rev. 2010, 39, 4402–4421. doi:10.1039/b917644f
Return to citation in text: [1] -
Rueping, M.; Parra, A. Org. Lett. 2010, 12, 5281–5283. doi:10.1021/ol102247n
Return to citation in text: [1] -
Thompson, B. B.; Montgomery, J. Org. Lett. 2011, 13, 3289–3291. doi:10.1021/ol201133n
Return to citation in text: [1] [2] -
Zhu, D.; Zhao, J.; Wei, Y.; Zhou, H. Synlett 2011, 2011, 2185–2186. doi:10.1055/s-0030-1261175
Return to citation in text: [1] -
Chen, F.; Shen, T.; Cui, Y.; Jiao, N. Org. Lett. 2012, 14, 4926–4929. doi:10.1021/ol302270z
Return to citation in text: [1] [2] -
Fesenko, A. A.; Shutalev, A. D. J. Org. Chem. 2013, 78, 1190–1207. doi:10.1021/jo302724y
Return to citation in text: [1] -
Zhang, M.; Neumann, H.; Beller, M. Angew. Chem., Int. Ed. 2013, 52, 597–601. doi:10.1002/anie.201206082
Return to citation in text: [1] -
Bharathiraja, G.; Sakthivel, S.; Sengoden, M.; Punniyamurthy, T. Org. Lett. 2013, 15, 4996–4999. doi:10.1021/ol402305b
Return to citation in text: [1] -
Shi, Z.; Suri, M.; Glorius, F. Angew. Chem., Int. Ed. 2013, 52, 4892–4896. doi:10.1002/anie.201300477
Return to citation in text: [1] -
Hantzsch, A. Ber. Dtsch. Chem. Ges. 1890, 23, 1474–1476. doi:10.1002/cber.189002301243
Return to citation in text: [1] -
Knorr, L. Ber. Dtsch. Chem. Ges. 1884, 17, 1635–1642. doi:10.1002/cber.18840170220
Return to citation in text: [1] -
Paal, C. Ber. Dtsch. Chem. Ges. 1885, 18, 367–371. doi:10.1002/cber.18850180175
Return to citation in text: [1] -
Barton, D. H. R.; Kervagoret, J.; Zard, S. Z. Tetrahedron 1990, 46, 7587–7598. doi:10.1016/S0040-4020(01)89069-4
Return to citation in text: [1] -
Bergner, I.; Opatz, T. Synthesis 2007, 918–928. doi:10.1055/s-2007-965937
Return to citation in text: [1] -
Meyer, N.; Opatz, T. Synlett 2004, 787–790. doi:10.1055/s-2004-817774
Return to citation in text: [1] -
Bergner, I.; Opatz, T. J. Org. Chem. 2007, 72, 7083–7090. doi:10.1021/jo070426x
Return to citation in text: [1] -
Schäfer, I.; Opatz, T. Synthesis 2011, 1691–1704. doi:10.1055/s-0030-1260415
Return to citation in text: [1] -
Bergner, I.; Wiebe, C.; Meyer, N.; Opatz, T. J. Org. Chem. 2009, 74, 8243–8253. doi:10.1021/jo901759u
Return to citation in text: [1] [2] [3] [4] -
Treibs, A.; Kreuzer, F.-H. Liebigs Ann. 1968, 718, 208–223. doi:10.1002/jlac.19687180119
Return to citation in text: [1] -
Jameson, L. P.; Dzyuba, S. V. Beilstein J. Org. Chem. 2013, 9, 786–790. doi:10.3762/bjoc.9.89
Return to citation in text: [1] -
Zhao, W.; Carreira, E. M. Chem.–Eur. J. 2006, 12, 7254–7263. doi:10.1002/chem.200600527
Return to citation in text: [1] -
Rogers, M. A. T. J. Chem. Soc. 1943, 590–596. doi:10.1039/JR9430000590
Return to citation in text: [1] -
Kloetzel, M. C.; Pinkus, J. L.; Washburn, R. M. J. Am. Chem. Soc. 1957, 79, 4222–4225. doi:10.1021/ja01572a063
Return to citation in text: [1] -
Hall, M. J.; McDonnell, S. O.; Killoran, J.; O'Shea, D. F. J. Org. Chem. 2005, 70, 5571–5578. doi:10.1021/jo050696k
Return to citation in text: [1] -
Barr, K. J.; Watson, B. T.; Buchwald, S. L. Tetrahedron Lett. 1991, 32, 5465–5468. doi:10.1016/0040-4039(91)80059-F
Return to citation in text: [1] -
Zhang, J.; Xing, C.; Tiwari, B.; Chi, Y. R. J. Am. Chem. Soc. 2013, 135, 8113–8116. doi:10.1021/ja401511r
Return to citation in text: [1] -
Tsuge, O.; Ueno, K.; Kanemasa, S.; Yorozu, K. Bull. Chem. Soc. Jpn. 1986, 59, 1809–1824. doi:10.1246/bcsj.59.1809
Return to citation in text: [1] -
Birouk, M.; Harraga, S.; Panouse-Perrin, J.; Robert, J. F.; Damelincourt, M.; Theobald, F.; Mercier, R.; Panouse, J. J. Eur. J. Med. Chem. 1991, 26, 91–99. doi:10.1016/0223-5234(91)90217-B
Return to citation in text: [1] -
Lakhlifi, T.; Sedqui, A.; Fathi, T.; Laude, B.; Robert, J.-F. Can. J. Chem. 1994, 72, 1417–1423. doi:10.1139/v94-178
Return to citation in text: [1] -
Ezquerra, J.; Pedregal, C.; Rubio, A.; Valenciano, J.; Navio, J. L. G.; Alvarez-Builla, J.; Vaquero, J. J. Tetrahedron Lett. 1993, 34, 6317–6320. doi:10.1016/S0040-4039(00)73741-5
Return to citation in text: [1] -
Metzger, W.; Fischer, H. Liebigs Ann. 1937, 527, 1–37. doi:10.1002/jlac.19375270102
Return to citation in text: [1] -
Smith, E. B.; Jensen, H. B. J. Org. Chem. 1967, 32, 3330–3334. doi:10.1021/jo01286a012
Return to citation in text: [1]
| 1. | Gulevich, A. V.; Dudnik, A. S.; Chernyak, N.; Gevorgyan, V. Chem. Rev. 2013, 113, 3084–3213. doi:10.1021/cr300333u |
| 28. | Hantzsch, A. Ber. Dtsch. Chem. Ges. 1890, 23, 1474–1476. doi:10.1002/cber.189002301243 |
| 37. | Treibs, A.; Kreuzer, F.-H. Liebigs Ann. 1968, 718, 208–223. doi:10.1002/jlac.19687180119 |
| 38. | Jameson, L. P.; Dzyuba, S. V. Beilstein J. Org. Chem. 2013, 9, 786–790. doi:10.3762/bjoc.9.89 |
| 5. | Braun, R. U.; Zeitler, K.; Müller, T. J. J. Org. Lett. 2001, 3, 3297–3300. doi:10.1021/ol0165185 |
| 6. | Kel'in, A. V.; Sromek, A. W.; Gevorgyan, V. J. Am. Chem. Soc. 2001, 123, 2074–2075. doi:10.1021/ja0058684 |
| 7. | Fan, X.; Zhang, Y. Tetrahedron Lett. 2002, 43, 1863–1865. doi:10.1016/S0040-4039(02)00108-9 |
| 8. | Rao, H. S. P.; Jothilingam, S.; Scheeren, H. W. Tetrahedron 2004, 60, 1625–1630. doi:10.1016/j.tet.2003.11.087 |
| 9. | Zeng, D. X.; Chen, Y. Synlett 2006, 490–492. doi:10.1055/s-2006-932452 |
| 10. | Lu, L.; Chen, G.; Ma, S. Org. Lett. 2006, 8, 835–838. doi:10.1021/ol052797a |
| 11. | Bellina, F.; Rossi, R. Tetrahedron 2006, 62, 7213–7256. doi:10.1016/j.tet.2006.05.024 |
| 12. | Kim, Y.; Kim, J.; Park, S. B. Org. Lett. 2009, 11, 17–20. doi:10.1021/ol8022193 |
| 13. | Kiren, S.; Hong, X.; Leverett, C. A.; Padwa, A. Org. Lett. 2009, 11, 1233–1235. doi:10.1021/ol900059e |
| 14. | Aponick, A.; Li, C.-Y.; Malinge, J.; Marques, E. F. Org. Lett. 2009, 11, 4624–4627. doi:10.1021/ol901901m |
| 15. | Merkul, E.; Boersch, C.; Frank, W.; Muller, T. J. J. Org. Lett. 2009, 11, 2269–2272. doi:10.1021/ol900581a |
| 16. | Kramer, S.; Madsen, J. L. H.; Rottländer, M.; Skrydstrup, T. Org. Lett. 2010, 12, 2758–2761. doi:10.1021/ol1008685 |
| 17. | Donohoe, T. J.; Race, N. J.; Bower, J. F.; Callens, C. K. A. Org. Lett. 2010, 12, 4094–4097. doi:10.1021/ol101681r |
| 18. | Rakshit, S.; Patureau, F. W.; Glorius, F. J. Am. Chem. Soc. 2010, 132, 9585–9587. doi:10.1021/ja104305s |
| 19. | Estévez, V.; Villacampa, M.; Menéndez, J. C. Chem. Soc. Rev. 2010, 39, 4402–4421. doi:10.1039/b917644f |
| 20. | Rueping, M.; Parra, A. Org. Lett. 2010, 12, 5281–5283. doi:10.1021/ol102247n |
| 21. | Thompson, B. B.; Montgomery, J. Org. Lett. 2011, 13, 3289–3291. doi:10.1021/ol201133n |
| 22. | Zhu, D.; Zhao, J.; Wei, Y.; Zhou, H. Synlett 2011, 2011, 2185–2186. doi:10.1055/s-0030-1261175 |
| 23. | Chen, F.; Shen, T.; Cui, Y.; Jiao, N. Org. Lett. 2012, 14, 4926–4929. doi:10.1021/ol302270z |
| 24. | Fesenko, A. A.; Shutalev, A. D. J. Org. Chem. 2013, 78, 1190–1207. doi:10.1021/jo302724y |
| 25. | Zhang, M.; Neumann, H.; Beller, M. Angew. Chem., Int. Ed. 2013, 52, 597–601. doi:10.1002/anie.201206082 |
| 26. | Bharathiraja, G.; Sakthivel, S.; Sengoden, M.; Punniyamurthy, T. Org. Lett. 2013, 15, 4996–4999. doi:10.1021/ol402305b |
| 27. | Shi, Z.; Suri, M.; Glorius, F. Angew. Chem., Int. Ed. 2013, 52, 4892–4896. doi:10.1002/anie.201300477 |
| 39. | Zhao, W.; Carreira, E. M. Chem.–Eur. J. 2006, 12, 7254–7263. doi:10.1002/chem.200600527 |
| 4. | Groenendaal, L.; Meijer, E. W.; Vekemans, J. A. J. M. In Electronic Materials: The Oligomer Approach; Müllen, K.; Wegner, G., Eds.; Wiley-VCH Verlag GmbH: Weinheim, 2007; pp 235–275. |
| 36. | Bergner, I.; Wiebe, C.; Meyer, N.; Opatz, T. J. Org. Chem. 2009, 74, 8243–8253. doi:10.1021/jo901759u |
| 2. | Sessler, J. L.; Camiolo, S.; Gale, P. A. Coord. Chem. Rev. 2003, 240, 17–55. doi:10.1016/S0010-8545(03)00023-7 |
| 3. | Srivastava, S. K.; Miller, C. N.; Aceto, M. D.; Traynor, J. R.; Lewis, J. W.; Husbands, S. M. J. Med. Chem. 2004, 47, 6645–6648. doi:10.1021/jm040817t |
| 36. | Bergner, I.; Wiebe, C.; Meyer, N.; Opatz, T. J. Org. Chem. 2009, 74, 8243–8253. doi:10.1021/jo901759u |
| 34. | Bergner, I.; Opatz, T. J. Org. Chem. 2007, 72, 7083–7090. doi:10.1021/jo070426x |
| 35. | Schäfer, I.; Opatz, T. Synthesis 2011, 1691–1704. doi:10.1055/s-0030-1260415 |
| 31. | Barton, D. H. R.; Kervagoret, J.; Zard, S. Z. Tetrahedron 1990, 46, 7587–7598. doi:10.1016/S0040-4020(01)89069-4 |
| 36. | Bergner, I.; Wiebe, C.; Meyer, N.; Opatz, T. J. Org. Chem. 2009, 74, 8243–8253. doi:10.1021/jo901759u |
| 30. | Paal, C. Ber. Dtsch. Chem. Ges. 1885, 18, 367–371. doi:10.1002/cber.18850180175 |
| 29. | Knorr, L. Ber. Dtsch. Chem. Ges. 1884, 17, 1635–1642. doi:10.1002/cber.18840170220 |
| 43. | Barr, K. J.; Watson, B. T.; Buchwald, S. L. Tetrahedron Lett. 1991, 32, 5465–5468. doi:10.1016/0040-4039(91)80059-F |
| 40. | Rogers, M. A. T. J. Chem. Soc. 1943, 590–596. doi:10.1039/JR9430000590 |
| 41. | Kloetzel, M. C.; Pinkus, J. L.; Washburn, R. M. J. Am. Chem. Soc. 1957, 79, 4222–4225. doi:10.1021/ja01572a063 |
| 42. | Hall, M. J.; McDonnell, S. O.; Killoran, J.; O'Shea, D. F. J. Org. Chem. 2005, 70, 5571–5578. doi:10.1021/jo050696k |
| 49. | Metzger, W.; Fischer, H. Liebigs Ann. 1937, 527, 1–37. doi:10.1002/jlac.19375270102 |
| 50. | Smith, E. B.; Jensen, H. B. J. Org. Chem. 1967, 32, 3330–3334. doi:10.1021/jo01286a012 |
| 36. | Bergner, I.; Wiebe, C.; Meyer, N.; Opatz, T. J. Org. Chem. 2009, 74, 8243–8253. doi:10.1021/jo901759u |
| 46. | Birouk, M.; Harraga, S.; Panouse-Perrin, J.; Robert, J. F.; Damelincourt, M.; Theobald, F.; Mercier, R.; Panouse, J. J. Eur. J. Med. Chem. 1991, 26, 91–99. doi:10.1016/0223-5234(91)90217-B |
| 47. | Lakhlifi, T.; Sedqui, A.; Fathi, T.; Laude, B.; Robert, J.-F. Can. J. Chem. 1994, 72, 1417–1423. doi:10.1139/v94-178 |
| 48. | Ezquerra, J.; Pedregal, C.; Rubio, A.; Valenciano, J.; Navio, J. L. G.; Alvarez-Builla, J.; Vaquero, J. J. Tetrahedron Lett. 1993, 34, 6317–6320. doi:10.1016/S0040-4039(00)73741-5 |
| 44. | Zhang, J.; Xing, C.; Tiwari, B.; Chi, Y. R. J. Am. Chem. Soc. 2013, 135, 8113–8116. doi:10.1021/ja401511r |
| 45. | Tsuge, O.; Ueno, K.; Kanemasa, S.; Yorozu, K. Bull. Chem. Soc. Jpn. 1986, 59, 1809–1824. doi:10.1246/bcsj.59.1809 |
| 21. | Thompson, B. B.; Montgomery, J. Org. Lett. 2011, 13, 3289–3291. doi:10.1021/ol201133n |
| 23. | Chen, F.; Shen, T.; Cui, Y.; Jiao, N. Org. Lett. 2012, 14, 4926–4929. doi:10.1021/ol302270z |
© 2014 Kucukdisli et al; licensee Beilstein-Institut.
This is an Open Access article under the terms of the Creative Commons Attribution License (http://creativecommons.org/licenses/by/2.0), which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
The license is subject to the Beilstein Journal of Organic Chemistry terms and conditions: (http://www.beilstein-journals.org/bjoc)