Search results
Search for "triazole" in Full Text gives 263 result(s) in Beilstein Journal of Organic Chemistry. Showing first 200.
Tetrathiafulvalene – a redox-switchable building block to control motion in mechanically interlocked molecules
Beilstein J. Org. Chem. 2018, 14, 2163–2185, doi:10.3762/bjoc.14.190

- TTF is preferred as a station for the wheel over the triazole unit. However, after oxidation, the more hydrophilic dication TTF2+ is less favored and the ring moves to the triazole. Both switching states were fully characterized by UV–vis, 1H,1H-NOESY NMR spectroscopy, and cyclic voltammetry. The
Revisiting ring-degenerate rearrangements of 1-substituted-4-imino-1,2,3-triazoles
Beilstein J. Org. Chem. 2018, 14, 2098–2105, doi:10.3762/bjoc.14.184
- James T. Fletcher Matthew D. Hanson Joseph A. Christensen Eric M. Villa Department of Chemistry, Creighton University, 2500 California Plaza, Omaha, NE 68178, U.S.A. 10.3762/bjoc.14.184 Abstract The 1-substituted-4-imino-1,2,3-triazole motif is an established component of coordination compounds
- four imine products via a dynamic equilibrium of condensation, rearrangement and hydrolysis steps. Kinetic studies utilizing 1-(4-nitrophenyl)-1H-1,2,3-triazole-4-carbaldehyde with varying amines showed rearrangement rates sensitive to both steric and electronic factors. Such measurements were
- facilitated by a high throughput colorimetric assay to directly monitor the generation of a 4-nitroaniline byproduct. Keywords: colorimetric assay; condensation; imine exchange; rearrangement; 1,2,3-triazole; Introduction Examples of multidentate chelators comprised of 1,2,3-triazole units have surged in
Synthesis and supramolecular self-assembly of glutamic acid-based squaramides
Beilstein J. Org. Chem. 2018, 14, 2065–2073, doi:10.3762/bjoc.14.180

- concentrations and gel-to-sol transition temperatures were also determined for each case. In addition, the superior squaramide diester gelator was compared with an analogue triazole-based gelator in terms of critical gelation concentration, gelation kinetics and thermal phase transition. Keywords: glutamic acid
- properties of supramolecular gels [33]. Specifically, we exchanged the amide group of N-stearoyl-L-glutamic acid (1, Figure 1), a known LMW gelator [34], by its non-classical isostere [35][36] 1,4-disubstituted 1,2,3-triazole 2 (Figure 1). This approach enabled us to fine-tuning the gelation capacity and the
- due to the delicate equilibrium between metastable gel and thermodynamically stable crystalline phases [39][40]. Squaramide 3 formed stable gels in the same number of solvents than the analogue triazole-based gelator 2, which was previously found to be superior to the amide 1 [33]. Similarly to 3, CGC
Applications of organocatalysed visible-light photoredox reactions for medicinal chemistry
Beilstein J. Org. Chem. 2018, 14, 2035–2064, doi:10.3762/bjoc.14.179
- carbamates, urea and non-basic heterocycles (Scheme 11) [54]. The scope covers a multitude of electron-poor and electron-rich arenes which can be reacted with carbamates, urea, pyrazole and triazole derivatives to furnish aminated products. The authors address the various reactivities observed with respect
- group report are scaffolds and moieties seen in typical medicinal chemistry syntheses. There are numerous examples of amino acid-derived substrates, either as the methoxybenzene electrophile (tyrosine type derivatives) or as the nucleophile (histidine and related structures such as the depicted triazole
Synthesis of new p-tert-butylcalix[4]arene-based polyammonium triazolyl amphiphiles and their binding with nucleoside phosphates
Beilstein J. Org. Chem. 2018, 14, 1980–1993, doi:10.3762/bjoc.14.173

- was elaborated for the naked-eye detection of ADP with a detection limit of 0.5 mM. Keywords: ADP; amphiphile; ATP; calix[4]arene; CuAAC; eosin Y probe; molecular recognition; polydiacetylene; self-assembly; triazole; Introduction During the last two decades many researcher groups have paid much
- target BOC-protected products 9a,b and 11a,b were isolated in good yields. The appearance of a new signal of triazole ring protons and a new set of signals of the methylene group protons between the triazole ring and the tertiary nitrogen atom as well as the signals of the ethylene fragments and BOC
An amphiphilic pseudo[1]catenane: neutral guest-induced clouding point change
Beilstein J. Org. Chem. 2018, 14, 1937–1943, doi:10.3762/bjoc.14.167

- , triazole), 7.15 (s, 2H, phenyl), 6.93 (s, 2H, phenyl), 6.89 (s, 2H, phenyl), 6.80 (s, 2H, phenyl), 6.62 (s, 2H, phenyl), 5.21 (dd, 4H, methylene), 3.50–4.14 (m, 110H, methylene), 3.34–3.36 (m, 24H, methyl), −0.27–0.72 (br, 20H, methylene of alkyl chain) ppm; 13C NMR (125 MHz, CDCl3) δ 150.4, 149.8, 149.7
Efficient catenane synthesis by cucurbit[6]uril-mediated azide–alkyne cycloaddition
Beilstein J. Org. Chem. 2018, 14, 1846–1853, doi:10.3762/bjoc.14.158

- proximity and their cycloaddition will be facilitated. As the cycloaddition is preceded by CB[6] binding, triazole formation will therefore ensure the interlocking of the CB[6]. A [1 + 1] macrocyclization of the diazide and dialkyne will then result in the exclusive formation of the [3]catenane with no
- interactions are preserved under the MS conditions. Inclusion of the triazole in the CB[6] cavity was evidenced by the upfield shifted triazole resonance from 8.5 ppm to 6.4 ppm when compared with that of free triazole in non-interlocked system [22][24][25]. NOE cross peaks between the triazole and CB[6
- upfield shifted triazole 1H NMR resonances of Cat-2–Cat-10 at ca. 6.4 ppm are consistent with the inclusion of the triazole in the CB[6] cavity [22][24][25]. The ESIMS, HRMS and MS2 spectra are all consistent with the [3]catenanes with a similar fragmentation behavior as that of Cat-1. The 1H NMR, 13C NMR
Synthesis of new tricyclic 5,6-dihydro-4H-benzo[b][1,2,4]triazolo[1,5-d][1,4]diazepine derivatives by [3+ + 2]-cycloaddition/rearrangement reactions
Beilstein J. Org. Chem. 2018, 14, 1826–1833, doi:10.3762/bjoc.14.155
- furnishing the tricyclic 1,2,4-triazole-fused 1,4-benzodiazepines. Keywords: 1,4-benzodiazepine (BDZ); cyclization; hydrazones; oxidation; rearrangement; Introduction Heterocyclic compounds comprising a 1,4-benzodiazepine (BDZ) ring have been a topic of continued interest as they exhibit a wide spectrum of
- achieved when a third heterocycle, especially a 1,2,4-triazolo moiety, was attached to the seven-membered ring as part of 1,4-benzodiazepine [13][14]. Among various reported 1,2,4-triazole-annulated 1,4-benzodiazepines, triazolam (I), estazolam (II), alprazolam (III) and pyrazolam (IV) are prominent
- . Conclusion In summary, an appealing series of 1,2,4-triazole-fused 1,4-benzodiazepines 10 and 13 were prepared via the [3+ + 2]-cycloaddition reaction followed by a cationic [1,2]-rearrangement reaction. The procedure is general and has several advantages such as ready availability of starting materials
Synthesis and photophysical studies of a multivalent photoreactive RuII-calix[4]arene complex bearing RGD-containing cyclopentapeptides
Beilstein J. Org. Chem. 2018, 14, 1758–1768, doi:10.3762/bjoc.14.150

- (CuAAC). Note that the triazole moieties that would result from such a cycloaddition are known to be stable towards hydrolysis and protease, which allows their use in a biological environment [72]. For the CuAAC, the use of CuI-generated in situ from a mixture of CuSO4·5H2O and sodium ascorbate is often
Hypervalent organoiodine compounds: from reagents to valuable building blocks in synthesis
Beilstein J. Org. Chem. 2018, 14, 1508–1528, doi:10.3762/bjoc.14.128
- mechanism that first involves the formation of a 2’-iodobiphenyl-2-azide promoted by the copper complex. The latter then catalyzes an intermolecular [3 + 2] cycloaddition with the alkyne. Finally, the copper-triazole moiety inserts intramolecularly into the second Ar–I bond, allowing a ring closure after
Hyper-reticulated calixarene polymers: a new example of entirely synthetic nanosponge materials
Beilstein J. Org. Chem. 2018, 14, 1498–1507, doi:10.3762/bjoc.14.127
- was assessed by means of FTIR and 13C{1H} CP-MAS solid-state NMR techniques, whereas morphological characterization was provided by SEM microghaphy. The materials were proved to possess pH-dependent sequestration abilities, due to the presence of the weakly basic triazole linkers. Sequestration
- ]) between a heptakis(6-azido-6-deoxy)-β-cyclodextrin and a tetrakis(propargyloxy)calix[4]arene [11][31][32]. In this way, a random disposition of the co-monomer units linked by 1,2,3-triazole units is achieved. The obtained CyCaNSs benefit from several advantages. First, the properties of the material can
- polymeric structure and can be subsequently subjected to further chemical transformation, opening the way to a possible post-functionalization [31]. This, in turn, largely improves the possible tunability in the properties of the materials obtained. Finally, the triazole linker present in the structure is
Recent advances in phosphorescent platinum complexes for organic light-emitting diodes
Beilstein J. Org. Chem. 2018, 14, 1459–1481, doi:10.3762/bjoc.14.124

[3 + 2]-Cycloaddition reaction of sydnones with alkynes
Beilstein J. Org. Chem. 2018, 14, 1317–1348, doi:10.3762/bjoc.14.113
- same N3 coordinating both Cu atoms attacks the terminal carbon of the polarized double bond with concerted cleavage of one of the two copper atoms. The copper triazolide formed in this way is then hydrolyzed to the final triazole. The same presumption (Scheme 17) concerning the role of the two Cu atoms
- -phenylbut-1-yne [119] tridentate tris(benzimidazole) ligands completely failed and tris(triazole) ligands gave only poor to moderate yields (16–65%) even at 100 °C, whereas all the bidentate ligands (phenanthrolines L1, L2 and diimidazo[1,2-a:2',1'-c]quinoxalines L3–L6) were found to be more efficient both
Oligonucleotide analogues with cationic backbone linkages
Beilstein J. Org. Chem. 2018, 14, 1293–1308, doi:10.3762/bjoc.14.111
- motifs. Such artificial neutral linkages include, among others, sulfone [14], amide [15][16][17][18][19][20][21][22], triazole [23][24][25][26][27], phosphoramidate [28] and phosphate triester [29] moieties. Using a different approach, positive charges have been introduced into nucleic acid structures
Novel unit B cryptophycin analogues as payloads for targeted therapy
Beilstein J. Org. Chem. 2018, 14, 1281–1286, doi:10.3762/bjoc.14.109

- conjugation of the novel cryptophycin analogues across an appropriate linker to an antibody or peptide. Either a virtually uncleavable triazole (introduced by CuAAC) or scissile ester, carbonate, or carbamate moieties were taken into account. The synthesis of the modified unit B (Scheme 1) started with the
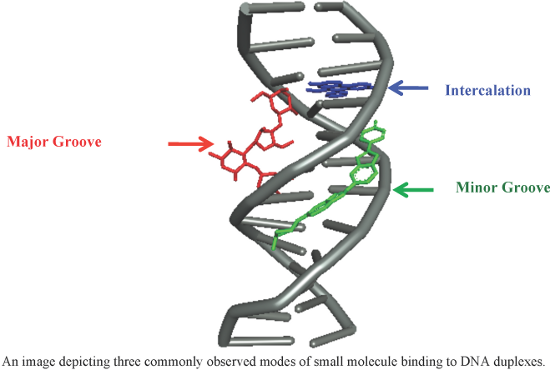
An overview of recent advances in duplex DNA recognition by small molecules
Beilstein J. Org. Chem. 2018, 14, 1051–1086, doi:10.3762/bjoc.14.93
An uracil-linked hydroxyflavone probe for the recognition of ATP
Beilstein J. Org. Chem. 2018, 14, 747–755, doi:10.3762/bjoc.14.63
- precipitated from the solution. The precipitate was filtered, washed with THF thoroughly and dried to yield 264 mg (88%) product as a yellow solid. 1H NMR (500 MHz, DMSO-d6) δ 11.16 (s, 1H, 3-uracil NH), 10.94 (br s, 1H, 1-uracil NH), 8.79 (s, 1H, OH), 8.21 (s, 1H, ArH, triazole), 8.10 (d, J = 8.5 Hz, 2H, 2
- ), 163.64 (C=O6-uracill), 161.90 (CAr-O), 155.98 (CAr-O), 151.16 (C=O2-uracil), 150.85 (C4’-Ar, aniline), 146.17 (CAr-O), 142.98 (CHAr, triazole), 141.70 (CH4-uracil), 136.82 (C-OHchromone), 128.64 (2CH2’-Ar, aniline), 125.96 (CHAr, chromone), 124.83 (CHAr, triazole), 118.06 (CAr, chromone), 115.42 (CAr
Nanoreactors for green catalysis
Beilstein J. Org. Chem. 2018, 14, 716–733, doi:10.3762/bjoc.14.61

- nanoparticles (DSN) have been shown to be highly efficient in the catalysis of olefin hydrogenation and in Suzuki coupling reactions [98][99]. Ornelas et al. entrapped a palladium catalyst with dendrimers containing triazole groups (DSN) (Figure 6) [100]. The aim here was to provide a platform to perform
AuBr3-catalyzed azidation of per-O-acetylated and per-O-benzoylated sugars
Beilstein J. Org. Chem. 2018, 14, 682–687, doi:10.3762/bjoc.14.56
- ], glycoconjugates [30][31][32], N-glycosyl heterocycles [33][34], N-glycosyl triazole [35][36], etc. Glycosyl azides can be accessed from the corresponding glycosyl halides [37][38][39][40] by nucleophilic displacement with NaN3 or using trimethylsilyl azide in the presence of a phase transfer catalyst [41][42][43
Sequential Ugi reaction/base-induced ring closing/IAAC protocol toward triazolobenzodiazepine-fused diketopiperazines and hydantoins
Beilstein J. Org. Chem. 2018, 14, 626–633, doi:10.3762/bjoc.14.49
- multicomponent reactions with intramolecular azide–alkyne cycloaddition (IAAC) for the generation of triazole-fused heterocycles [12][13][14][15][16][17][18][19][20][21][22][23]. In this report we disclose our results on the development of a sequential synthetic approach involving Ugi 4-component reaction (4-CR
- -turns [43][44][45] and are also known to bind to a number of biological targets [46][47][48]. Interestingly, the fusion of a triazole ring to a 1,4-benzodiazepine core has resulted in an increase in the biological activity as evident from the different drugs with triazolobenzodiazepine structure such as
- commenced with the intramolecular azide–alkyne cycloaddition of 5a which furnished the triazole-fused benzodiazepine 8 in 72% yield after 3 hours. The triazolobenzodiazepine 8 was then subjected to base-induced cyclisation with KOH which failed to afford the expected tetracyclic compound 7a. We believe that
Carbohydrate inhibitors of cholera toxin
Beilstein J. Org. Chem. 2018, 14, 484–498, doi:10.3762/bjoc.14.34

- chelating mechanism. Hughes and co-workers synthesised and evaluated bivalent 1,2,3 triazole-linked galactopyranosides 14 and 15 as shown in Figure 7 [48]. They used a piperazine core as central divalent core on to which the galactose units were attached via flexible linkers. They found that these compounds
Recent advances on organic blue thermally activated delayed fluorescence (TADF) emitters for organic light-emitting diodes (OLEDs)
Beilstein J. Org. Chem. 2018, 14, 282–308, doi:10.3762/bjoc.14.18

Synthesis and spectroscopic properties of β-meso directly linked porphyrin–corrole hybrid compounds
Beilstein J. Org. Chem. 2018, 14, 187–193, doi:10.3762/bjoc.14.13
- ) position of substitution (meso or β). So far, corrole macrocyles have been integrated into porphyrin conjugates via anthracene, biphenylene, xanthene, dibenzofuran [16][17][18][19][20], amide [21] and triazole [22][23] linkers. Despite the large number of studies on the synthesis of porphyrin–corrole
Recent applications of click chemistry for the functionalization of gold nanoparticles and their conversion to glyco-gold nanoparticles
Beilstein J. Org. Chem. 2018, 14, 11–24, doi:10.3762/bjoc.14.2
- ; click chemistry; glyco-gold nanoparticles; triazole; Introduction Metal nanoparticles (NPs), with their unique physicochemical properties, have drawn significant interest in recent years, and are expected to form the basis of many biological and technological innovations during the remainder of the
- azide–alkyne Huisgen cycloaddition AuNP surface modification using NCAAC The azide–alkyne Huisgen cycloaddition (AAC) is a 1,3-dipolar cycloaddition between an organic azide and an alkyne that gives triazole products [37][38]. The non-catalysed azide–alkyne Huisgen cycloaddition (NCAAC) is very slow
- , and gives a mixture of 1,4- and 1,5-triazole regioisomers (Scheme 1) [39]. Interest in and applications of the AAC have surged over the past 15 or so years, since the introduction of Cu(I) catalysis, which led to significant improvements in both the regioselectivity and rates of the reaction [40][41






































































































































































































































































































































































































