Search results
Search for "glycoside" in Full Text gives 155 result(s) in Beilstein Journal of Organic Chemistry.
Semi-synthesis and insecticidal activity of spinetoram J and its D-forosamine replacement analogues
Beilstein J. Org. Chem. 2018, 14, 2321–2330, doi:10.3762/bjoc.14.207
- ether bond at the C17 position under acidic conditions. Synthesis of spinetoram J analogues All carbohydrates and alcohols were activated by CNCCl3 with DBU as catalyst initially to afford glycoside donors, and then the 17-pseudoaglycone of spinetoram J was glycosylated with donors in the presence of
- BF3·(C2H5)2O under Ar gas. Due to the high yields and few byproducts in the synthesis of glycoside donors, the donors could be used directly in the glycosylation without purification. Insecticidal activity The insecticidal activities of synthetic spinetoram J and its analogues were evaluated using
Synthesis of 1,4-imino-L-lyxitols modified at C-5 and their evaluation as inhibitors of GH38 α-mannosidases
Beilstein J. Org. Chem. 2018, 14, 2156–2162, doi:10.3762/bjoc.14.189
- , indolizidines etc. which exhibited remarkable biological activities and are highly interesting as pharmaceutical agents [15][16][17][18][19]. In addition, iminosugars exhibited a powerful inhibitory activity against a wide range of glycoside hydrolases [20][21][22]. One naturally occurring iminosugar, alkaloid
- swainsonine, interferes with the glycosylation pathway where it specifically inhibits GH38 glycoside hydrolases [23][24]. Up to date, swainsonine is the most potent Golgi mannosidase II (GMII) inhibitor. It is known that inhibition of the biosynthesis of complex N-glycans in the Golgi apparatus influences
Synthesis of spirocyclic scaffolds using hypervalent iodine reagents
Beilstein J. Org. Chem. 2018, 14, 1778–1805, doi:10.3762/bjoc.14.152
- spirocyclic glycoside that was launched as antidiabetic agent in 2012 in Japan [15]. Rolapitant (5) is a marketed drug that was approved in 2015 for the treatment of nausea and vomiting [16]. Compound 6 is a spiropyrimidinetrione analogue which is currently in clinical trials for the treatment of gonorrhea
Anomeric modification of carbohydrates using the Mitsunobu reaction
Beilstein J. Org. Chem. 2018, 14, 1619–1636, doi:10.3762/bjoc.14.138
- achieve good results in the Mitsunobu procedure. We have reviewed on the one hand the literature on anomeric esterification, including glycosyl phosphates, and on the other hand glycoside synthesis, including S- and N-glycosides. The mechanistic details of the Mitsunobu reaction are discussed as well as
- circumstances. Keywords: anomeric stereoselectivity; carbohydrates; glycoside synthesis; Mitsunobu reaction; Introduction Fifty years ago, Oyo Mitsunobu reported a preparation of esters from alcohols and carboxylic acids supported by two auxiliary reagents, diethyl azodicarboxylate (DEAD) and
- outcome of the reaction towards β-configured products [53]. In this case, phenolic chromium tricarbonyl complexes of weaker acids such as p-cresol were employed to improve the yield. The challenge of glycoside synthesis using sugars devoid of a C-2 participating group is also highlighted by a total
Glycosylation reactions mediated by hypervalent iodine: application to the synthesis of nucleosides and carbohydrates
Beilstein J. Org. Chem. 2018, 14, 1595–1618, doi:10.3762/bjoc.14.137
- , Tokushima, 770-8505, Japan 10.3762/bjoc.14.137 Abstract To synthesize nucleoside and oligosaccharide derivatives, we often use a glycosylation reaction to form a glycoside bond. Coupling reactions between a nucleobase and a sugar donor in the former case, and the reaction between an acceptor and a sugar
- glycoside bond. In the case of nucleoside synthesis, a coupling reaction between a persilylated nucleobase and a sugar donor is typically used [15][16][17]. On the other hand, the reaction between an acceptor and sugar donor is carried out in the presence of an appropriate activator for oligosaccharide
- the hypervalent iodine-mediated oxidative reactions with theses derivatives provide a different method to build glycosidic bonds. Diversity in glycoside bond forming reactions would contribute to improve the oligosaccharide synthesis. Conclusion The Pummerer-type glycosylation includes oxidation of a
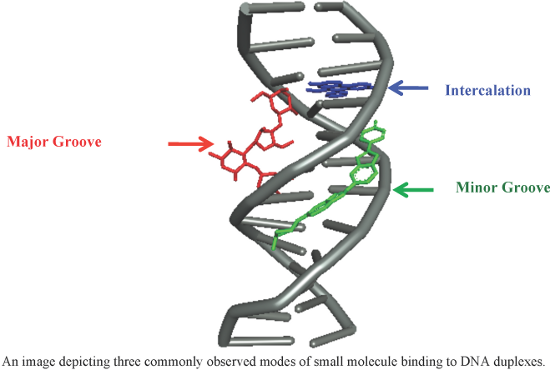
An overview of recent advances in duplex DNA recognition by small molecules
Beilstein J. Org. Chem. 2018, 14, 1051–1086, doi:10.3762/bjoc.14.93
AuBr3-catalyzed azidation of per-O-acetylated and per-O-benzoylated sugars
Beilstein J. Org. Chem. 2018, 14, 682–687, doi:10.3762/bjoc.14.56
- -benzoylated disaccharides needed 2–3 h of heating at 55 °C. Keywords: acylated sugars; azidation; gold(III) bromide; N-glycoside; oxophilicity; Introduction The past few decades had seen the enrichment of transition metal complexes in various glycosylation strategies [1]. In particular, gold complexes with
Aminosugar-based immunomodulator lipid A: synthetic approaches
Beilstein J. Org. Chem. 2018, 14, 25–53, doi:10.3762/bjoc.14.3
- types of protecting groups – allyloxycarbonyl (Alloc), hexyldimethylsilyl (TDS) and benzyl – were sequentially removed to provide the target compound 58. A monophosphoryl lipid A analogue 59 wherein the anomeric center of the proximal GlcN moiety is modified as methyl glycoside was prepared in a similar
- methyl glycoside 59 showed somewhat higher pro-inflammatory activity. Interestingly, attachment of varying 3-O-substitutions at position 3 of the reducing GlcN moiety in MPLA analogue 60 did not enhance the adjuvant activity [107]. Importantly, synthetic MPLA derivatives having variable acylation pattern
Recent applications of click chemistry for the functionalization of gold nanoparticles and their conversion to glyco-gold nanoparticles
Beilstein J. Org. Chem. 2018, 14, 11–24, doi:10.3762/bjoc.14.2
- , oligosaccharides usually bind lectins in a multivalent cooperative fashion. This avidity is significantly greater than the sum of the individual monomeric carbohydrate–protein interactions, and is sometimes referred to as the ‘cluster glycoside’ effect [6]. In order to study biological processes that involve these
Electron-deficient pyridinium salts/thiourea cooperative catalyzed O-glycosylation via activation of O-glycosyl trichloroacetimidate donors
Beilstein J. Org. Chem. 2017, 13, 2385–2395, doi:10.3762/bjoc.13.236
- , the optimized method is also utilized for the regioselective O-glycosylation by using a partially protected acceptor. Keywords: cooperative catalysis; electron-deficient pyridinium salts; O-glycoside; regioselectivity; thiourea; Introduction The glycosidic linkage is the principal bond present in a
- with phosphorus acids) for stereoselective O-glycoside bond formation [30]. Similarly, Galan et al. reported a method for the preparation of 2-deoxyglycosides from glycals under the influence of cooperative catalysis (chiral phosphoric acids/thiourea derivatives) [31]. Encouraged by these reports and
- acceptor 2a in 1:1.1 molar ratio with 10 mol % of 3a in dry DCM at room temperature, the desired O-glycoside 5a was isolated in 56% yield and with poor selectivity (Table 1, entry 2). The use of 25 mol % of 3a was required to drive the reaction to completion with 86% yield (Table 1, entry 3). This result
Diosgenyl 2-amino-2-deoxy-β-D-galactopyranoside: synthesis, derivatives and antimicrobial activity
Beilstein J. Org. Chem. 2017, 13, 2310–2315, doi:10.3762/bjoc.13.227

- the β anomer). Glycosylation of diosgenin with 2 was performed in dichloromethane by a “reverse” procedure: The glycosyl donor was added to the solution of diosgenin and the promoter (silver triflate) [31]. This procedure afforded the expected β glycoside 3 in 80% yield. The structure of 3 was
An efficient synthesis of a C12-higher sugar aminoalditol
Beilstein J. Org. Chem. 2017, 13, 2146–2152, doi:10.3762/bjoc.13.213
- ; Anal. calcd for C76H79N3O11: C, 75.41; H, 6.58; N, 3.47; found: C, 75.41; H, 6.47; N, 3.34. Acetolysis of glycoside 4; synthesis of 5 Azide 4 (0.36 g, 0.3 mmol) was dissolved in ethyl acetate (2.3 mL) to which acetic anhydride (4.6 mL) and sulfuric acid (0.72 mL of the solution: conc. H2SO4 (0.05 mL
Preactivation-based chemoselective glycosylations: A powerful strategy for oligosaccharide assembly
Beilstein J. Org. Chem. 2017, 13, 2094–2114, doi:10.3762/bjoc.13.207
- subsequent acceptor addition then leads to the formation of the glycoside product. As donor activation and glycosylation are carried out in two distinct steps, unique chemoselectivities can be obtained. Successful glycosylation can be performed independent of anomeric reactivities of the building blocks. In
- -active oligoglucosides [28]. A limitation of this glycosyl bromide-mediated selenoglycoside iterative glycosylation is that it is restricted to the formation of 1,2-trans-glycosyl linkages. Furthermore, an additional isomerization step is needed to transform the orthoester to the desired glycoside
- . Preactivation-based iterative glycosylation of 2-pyridyl glycosides O-Unprotected 2-pyridyl glycosyl donors have been utilized in oligosaccharide synthesis [29]. The Ye group reported a preactivation protocol using protected 2-pyridyl donors [30]. The preactivation of 2-pyridyl glycoside 14 was performed using
Intramolecular glycosylation
Beilstein J. Org. Chem. 2017, 13, 2028–2048, doi:10.3762/bjoc.13.201
- (1→4) glycoside.” Indeed, after sequential glycosylation in the presence of TsOH at 50 oC, methanolysis, and per-acetylation, disaccharide 4 was isolated in 20% yield. The authors then very reasonably concluded that “Consequently, the presence of the ester linkage which kept the two sugar moieties in
- close proximity to each other certainly favored the formation of the desired glycoside bond in the above experiment. Thus, this is the first example of the so-called “entropic activation” in glycosidation reaction.” The authors have also projected that the “entropic activation demonstrated in this work
- 72. The latter was then intramolecularly glycosylated in the presence of silver triflate, tin(II) chloride, and 2,6-di-tert-butyl-4-methylpyridine (DTBMP). Finally, the tether was cleaved off using TFA to give pure 1,2-cis glycoside 73 in 63% yield over two steps. An alternative linker was developed
1,3-Dibromo-5,5-dimethylhydantoin as promoter for glycosylations using thioglycosides
Beilstein J. Org. Chem. 2017, 13, 1994–1998, doi:10.3762/bjoc.13.195
- investigated by using a variety of glycosyl donors 4–10 [34][35][36][37][38] containing C-2 participating groups to ensure 1,2-trans-glycoside formation (Table 2). Each glycosylating agent was reacted with D-glucose acceptors 2 (Table 2, entries 1–8) and 11 [39] (Table 2, entries 9–16) with a free hydroxy
Enzymatic synthesis of glycosides: from natural O- and N-glycosides to rare C- and S-glycosides
Beilstein J. Org. Chem. 2017, 13, 1857–1865, doi:10.3762/bjoc.13.180
- Jihen Ati Pierre Lafite Richard Daniellou ICOA UMR CNRS 7311, University of Orléans, rue de Chartres, BP 6759, 45067 Orléans cedex 2, France 10.3762/bjoc.13.180 Abstract Carbohydrate related enzymes, like glycosyltransferases and glycoside hydrolases, are nowadays more easily accessible and are
- of rare or unnatural glycosidic linkages. Keywords: enzyme; glycochemistry; glycoside hydrolase; glycosyltransferase; mechanism; Introduction The role of glycoconjugates is of prime importance, as they are nowadays well known to mediate many biological processes [1]. As a consequence, in a recently
- . Glycoside hydrolases (GHs) or glycosyltransferases (GTs) have been focused on in the search for glycosylation tools, and have been extensively studied for genetic engineering [9][10]. The corresponding compounds have proven useful in many applications ranging from glycosylation of natural products to
Strategies toward protecting group-free glycosylation through selective activation of the anomeric center
Beilstein J. Org. Chem. 2017, 13, 1239–1279, doi:10.3762/bjoc.13.123
- acceptor, with the product of the reaction termed glycoside. Examples of acceptor molecules in nature are other saccharides to form oligosaccharides, nucleobases to form nucleosides, and amino acid side chains to form glycoproteins. The donor is the electrophile in the reaction and, therefore, when
- subject of much debate (Scheme 4B) [21]. The utility of these enzymes is very clear and even extends beyond glycobiology. They are applicable to natural product synthesis as the aglycone of a natural product glycoside can be forged to the saccharide component using either a natural or engineered GT [22
- -glucose in the presence of HCl to provide the methyl glycoside (pathway a, Scheme 6). The reaction proceeds chemoselective at the anomeric position. More recent examples typically use Lewis acids [29][30][31][32][33][34] or microwave irradiation [35][36] to accelerate the reaction. However, shortcomings
Aqueous semisynthesis of C-glycoside glycamines from agarose
Beilstein J. Org. Chem. 2017, 13, 1222–1229, doi:10.3762/bjoc.13.121
- starting material to produce primary, secondary and tertiary C-glycoside glycamines, including mono- and disaccharide structures. The semisynthetic approach utilized was generally based on polysaccharide-controlled hydrolysis followed by reductive amination. All reactions were conducted in aqueous media
- reductive amination from commercially available agar. Initially, in order to obtain the appropriate substrate for the synthesis of the C-glycoside glycamines 3, 7 and 8 (Scheme 2), agarose (1) was submitted to partial hydrolysis to produce disaccharide agarobiose (2, Scheme 1). For this purpose, we
Glycoscience@Synchrotron: Synchrotron radiation applied to structural glycoscience
Beilstein J. Org. Chem. 2017, 13, 1145–1167, doi:10.3762/bjoc.13.114

- of sequenced genomes is paralleled by an increasing number of accession entries for the GTs crystal structures in the PDB, which amounts to 900. Unlike glycoside hydrolases which display a large variety of different folds, the structures of GTs solved today can be clustered in two types of folds (and
- folds. Based on amino acid sequence similarities, polysaccharide lyases have been classified in 24 families [41]. Glycoside hydrolases: The hydrolysis of carbohydrates is the result of the action of a wide spread group of enzymes: the glycosyl hydrolases (GHs). GHs cleave the glycosidic linkage between
A concise and practical stereoselective synthesis of ipragliflozin L-proline
Beilstein J. Org. Chem. 2017, 13, 1064–1070, doi:10.3762/bjoc.13.105
- , 115.7, 73.1, 70.6, 68.4, 62.1, 38.7, 30.0, 27.0; MS (ES+) m/z: 763.44 [M + Na]+. Structure of ipragliflozin L-proline. Stereoselective synthesis of C-aryl glycoside by Lemarie. Stereoselective synthesis of β-C-arylglucoside 5. Synthesis of 1. Synthesis of diastereomer 6’ and 5’. Conditions for the
Cyclodextrins tethered with oligolactides – green synthesis and structural assessment
Beilstein J. Org. Chem. 2017, 13, 779–792, doi:10.3762/bjoc.13.77
- the γ-CD part, which is bigger than other CD homologues (8 glycoside units), to the lower solubility in THF resulting in precipitation of γ-CD-LA species with higher content of dilactate units. This fact is also reflected by the different weight percentage of F1 fractions. The α-CD-LA F1 was on 2%, β
- and small amounts of functionalized CD-LA and L-lactide monomer. All samples showed peaks for the unreacted L-lactide and separated peaks for the protons of the substituted glucopyranose units. In the 1H NMR spectra the substitution pattern of the glycoside rings of cyclodextrins can be followed by
- polymerization more glycoside rings are substituted predominantly at C6. This is also supported by the comparison between the 1H NMR integral ratios of OH2 and OH3 versus OH6 as previously discussed (Supporting Information File 1, Figure S27). Therefore, we may conclude that the lactide ring opening is performed
Total synthesis of a Streptococcus pneumoniae serotype 12F CPS repeating unit hexasaccharide
Beilstein J. Org. Chem. 2017, 13, 164–173, doi:10.3762/bjoc.13.19
- glycosylation of the liberated hydroxy groups. Formation of the β-mannosazide glycoside containing a protected C5 amino linker that serves in the final product as an attachment point for glycan array surfaces or carrier proteins was central to the assembly of trisaccharide 3. To avoid a challenging and often
- C2-participating levulinyl ester protecting group ensured selective formation of the trans-glycoside upon activation of 11 by NIS/TfOH in the presence of the C5 linker to produce glucoside 12 in 70% yield [24]. Cleavage of the C2 levulinyl ester of 12 by treatment with hydrazine acetate furnished 13
Silyl-protective groups influencing the reactivity and selectivity in glycosylations
Beilstein J. Org. Chem. 2017, 13, 93–105, doi:10.3762/bjoc.13.12
- , the ability of the bulky silyl groups to alter the conformation of the glycosyl-donor ring can be used to control the selectivity. Suzuki and collaborators showed that the C-arylation reactions with the 3,4-O-di(tert-butyldiphenylsilyl)-protected acetate 56 led to the α-glycoside 58 with high
- selectivity (Scheme 10). The reason for this selectivity is that the equatorial position is more accessible for attack [44]. However, if different protective groups and even the related TBS group were used, predominantly the β-glycoside 59 was obtained in a 14:1 α:β ratio. A similar conformation-controlled
- -position [55]. Thus the glycoside 68 was obtained in 74% yield as the only isolated product (Scheme 13). Equally remarkable is that the corresponding DTBS-protected galactosamine donors (such as 67) displayed the same selectivity in the presence of the silyl group and thereby overriding the influence of a
Nucleophilic displacement reactions of 5′-derivatised nucleosides in a vibration ball mill
Beilstein J. Org. Chem. 2017, 13, 87–92, doi:10.3762/bjoc.13.11
- ball-milling chemistry on nucleoside substrates has not, to the authors’ knowledge, been demonstrated, despite reports of similar chemistry on glycoside derivatives [16][23] and α-amino acid analogues [24]. We describe here the efficient displacement of 5'-chloride, iodide or tosylate leaving groups
O-Alkylated heavy atom carbohydrate probes for protein X-ray crystallography: Studies towards the synthesis of methyl 2-O-methyl-L-selenofucopyranoside
Beilstein J. Org. Chem. 2016, 12, 2828–2833, doi:10.3762/bjoc.12.282
- ) and ≈2 ppm (13C NMR). To prevent acidic degradation, 6 was treated with palladium on charcoal under a hydrogen atmosphere (Table 1, entries 3–5). In MeOH as the solvent, transglycosylation of the seleno glycoside 6 to its methyl O-glycoside was observed (Table 1, entry 3). Changing the solvent to the
- seleno furanoside 11 were obtained over three steps in 70% and 23% yield, respectively. By this route, the desired seleno glycoside 3 could be successfully synthesized with a high β/α ratio of 18:1. Previously, the tectonins were shown to bind the α-anomer of 2-O-methylated fucoside, and natural





































































































































































































































































































































































































































