Search results
Search for "structure–activity relationship" in Full Text gives 123 result(s) in Beilstein Journal of Organic Chemistry.
Drug targeting to decrease cardiotoxicity – determination of the cytotoxic effect of GnRH-based conjugates containing doxorubicin, daunorubicin and methotrexate on human cardiomyocytes and endothelial cells
Beilstein J. Org. Chem. 2018, 14, 1583–1594, doi:10.3762/bjoc.14.136

- 15 conjugates are also demonstrated in the most important two targets of cardiac tissue (myocytes and endothelium). Data presented in Table 1 provide a good basis for structure–activity relationship analysis of the reported results. Comparison of IC50 values of conjugates possessing no cardiotoxicity
Enantioselective phase-transfer catalyzed alkylation of 1-methyl-7-methoxy-2-tetralone: an effective route to dezocine
Beilstein J. Org. Chem. 2018, 14, 1421–1427, doi:10.3762/bjoc.14.119
- . (Some reports on the non-stereoselective alkylation of 2 were given in references [11][12]). In this paper, several cinchona-derived phase-transfer catalysts were screened for this reaction, and the structure–activity relationship for the catalysis was studied. In addition, optimizations had been made
- ). Subsequently, when the groups substituted at the para-position on the benzyl group were investigated, the structure–activity relationship showed that catalyst C4 (with methyl substituent) did not work for the reaction (Table 1, entry 4) and those with Cl or F (C5 and C6) worked well with an improvement in
Novel unit B cryptophycin analogues as payloads for targeted therapy
Beilstein J. Org. Chem. 2018, 14, 1281–1286, doi:10.3762/bjoc.14.109

- -52 and to better understand the fundamental structure for biological activity, numerous structure–activity relationship studies have been carried out [24][25][26][27][28][29][30][31][32][33][34][35]. However, like cryptophycin-52, the new analogues were not selective against cancer cells making them
An overview of recent advances in duplex DNA recognition by small molecules
Beilstein J. Org. Chem. 2018, 14, 1051–1086, doi:10.3762/bjoc.14.93
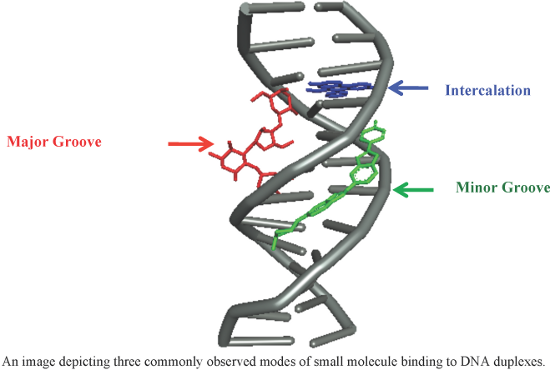
- structure–activity relationship. It has been observed that the number and position of pyrrole rings are crucial for antileukemic activity. The presence of pyrrole rings close to the alkylating BAM moiety is responsible for better cytotoxic activity both in vitro and in vivo, whereas a pyrazole ring in close
A stereoselective and flexible synthesis to access both enantiomers of N-acetylgalactosamine and peracetylated N-acetylidosamine
Beilstein J. Org. Chem. 2018, 14, 856–860, doi:10.3762/bjoc.14.71
- . In contrast to the thoroughly investigated properties of GalNAc, other stereoisomers have not been probed for biological or pharmaceutical activity, yet. Highly flexible synthetic routes are required to access and probe the entire compound class of 2-amino-2-deoxysugars for further structure–activity
- relationship studies. Several strategies for the synthesis of 2-amino sugars have been published so far. In one exemplary straightforward approach, GalNAc was prepared by inverting the stereogenic center at the C-4 position of N-acetylglucosamine (GlcNAc) [10]. However, the necessity of using a 2-amino sugar
Recent advances in synthetic approaches for medicinal chemistry of C-nucleosides
Beilstein J. Org. Chem. 2018, 14, 772–785, doi:10.3762/bjoc.14.65

- ), respiratory syncytial virus (RSV, Pneumoviridae) and the hepatitis-C virus (HCV, Flaviviridae) family [63][65][69]. Through structure activity relationship studies, the 1'-CN compound 4 emerged as a compound with activity against EBOV, HCV and RSV [65][69]. It is active against EBOV in human microvascular
- for structure–activity relationship [71][72]. Synthesis of 1',2'-cyclopentyl C-nucleoside [73]. Functional groups at C1' and C2' were installed and employed for ring cyclization. Anti-influenza C-nucleosides mimicking favipiravir riboside [74]. A. Structure of favipiravir and its riboside, which
Aminosugar-based immunomodulator lipid A: synthetic approaches
Beilstein J. Org. Chem. 2018, 14, 25–53, doi:10.3762/bjoc.14.3
- LPS isolates from wild-type or laboratory-adapted Gram-negative bacteria, the clinical and cellular studies as well as structure–activity relationship investigations using native LPS are complicated and difficult to evaluate. The lipid A content of LPS generally comprises a complex mixture of
The chemistry and biology of mycolactones
Beilstein J. Org. Chem. 2017, 13, 1596–1660, doi:10.3762/bjoc.13.159
- structure and fascinating biology, mycolactones have inspired various total synthesis endeavors and structure–activity relationship studies. Although this review intends to cover all synthesis efforts in the field, special emphasis is given to the comparison of conceptually different approaches and to the
Biomimetic molecular design tools that learn, evolve, and adapt
Beilstein J. Org. Chem. 2017, 13, 1288–1302, doi:10.3762/bjoc.13.125

- Adam and Eve that automate drug development via cycles of quantitative structure–activity relationship (QSAR) learning and biological testing (Figure 3) [16][17][18]. Eve’s selection of compounds was more cost efficient than standard drug screening, and the robotic scientist has identified several new
Structure–efficiency relationships of cyclodextrin scavengers in the hydrolytic degradation of organophosphorus compounds
Beilstein J. Org. Chem. 2017, 13, 417–427, doi:10.3762/bjoc.13.45

- iodosobenzoate group and the methylated oligosaccharide; (4) in case of soman, the degradation is enhanced by a cooperative effect observed between the imidazole and 2-iodosobenzoate when the latter is in close proximity to the macrocycle. A more extended structure–activity relationship study is envisaged to
Posttranslational isoprenylation of tryptophan in bacteria
Beilstein J. Org. Chem. 2017, 13, 338–346, doi:10.3762/bjoc.13.37
- -terminal cysteine (Figure 1B) [9][10]. Each pheromone causes the opposite type of cell to induce the reciprocal conjugation of the heterothallic cells, through the formation of a conjugation tube for mating. A structure–activity relationship study on tremerogen A-10 demonstrated that both the amino acid
- responses in B. subtilis and related bacilli [3]. Structure–activity relationship studies on the ComXRO-E-2 pheromone derived from Bacillus strain RO-E-2, which is a hexapeptide with a geranyl-modified tryptophan residue, revealed that the exact chemical structure of the geranyl group and the absolute
Versatile synthesis of the signaling peptide glorin
Beilstein J. Org. Chem. 2017, 13, 247–250, doi:10.3762/bjoc.13.27

- activate glorin-induced genes in the social amoeba Polysphondylium pallidum was evaluated by quantitative reverse transcription PCR, whereby both compounds showed bioactivity comparable to a glorin standard. This synthetic route will be useful in conducting detailed structure–activity relationship studies
- allow for facile access to glorin derivatives required for structure–activity relationship studies. Eventually, these studies can lead to the construction of various chemical probes to identify the unknown glorin receptor. Syntheses of glorin and glorinamide (Scheme 1) started from commercially
- derivatizations. Glorin, as well as the hydrolytically more stable derivative glorinamide, were shown to display comparable glorin-induced gene expression in Polysphondylium pallidum. In future this synthesis will facilitate the construction of a library of glorin derivatives for a detailed structure–activity
Computational methods in drug discovery
Beilstein J. Org. Chem. 2016, 12, 2694–2718, doi:10.3762/bjoc.12.267

Biomimetic synthesis and HPLC–ECD analysis of the isomers of dracocephins A and B
Beilstein J. Org. Chem. 2016, 12, 2523–2534, doi:10.3762/bjoc.12.247
- biomimetic scheme in order to devise an efficient route to these natural products for structure–activity relationship studies. First the N-acylaminocarbinol reagent was prepared in the form of racemic 5-ethoxypyrrolidine-2-one ((±)-9) by the partial reduction of succinimide (8) with sodium borohydride at 0
Useful access to enantiomerically pure protected inositols from carbohydrates: the aldohexos-5-uloses route
Beilstein J. Org. Chem. 2016, 12, 2343–2350, doi:10.3762/bjoc.12.227
- level is required. For this reason, many research efforts were directed toward the investigation of the structure–activity relationship (SAR) between inositol phosphates and biomacromolecules. These studies require various regio- and stereoisomers of inositol phosphates [6][7] and have prompted the
Enduracididine, a rare amino acid component of peptide antibiotics: Natural products and synthesis
Beilstein J. Org. Chem. 2016, 12, 2325–2342, doi:10.3762/bjoc.12.226
- activity against resistant strains of bacteria and favourable pharmacokinetics. Structure–activity relationship studies of teixobactin suggest that the rare non-proteinogenic amino acid enduracididine, is a key residue for potent antibacterial activity. This observation has driven the need for new
- inspired lead structure. Efficient access to enduracididine will enable ongoing structure–activity relationship studies of teixobactin and other lead compounds, for the development of much needed antibiotic drug candidates. Structures of the enduracididine family of amino acids (1–6). Enduracidin A (7) and
- B (8). Minosaminomycin (9) and related antibiotic kasugamycin (10). Enduracididine-containing compound 11 identified in a cytotoxic extract of Leptoclinides dubius [32]. Mannopeptimycins α–ε (12–16). Regions of the mannopeptimycin structure investigated in structure–activity relationship
Synthesis of the C8’-epimeric thymine pyranosyl amino acid core of amipurimycin
Beilstein J. Org. Chem. 2016, 12, 1765–1771, doi:10.3762/bjoc.12.165
- and its analogues for structure–activity relationship (SAR) studies. Conclusion In summary, we have utilized the skeleton of D-glucose-derived homoallyl alcohol 3 as a chiral podium for efficient synthesis of the 8’R-glycosyl amino acid core of amipurimycin. With this protocol, we have synthesized the
Total synthesis of leopolic acid A, a natural 2,3-pyrrolidinedione with antimicrobial activity
Beilstein J. Org. Chem. 2016, 12, 1624–1628, doi:10.3762/bjoc.12.159
- unusual 2,3-pyrrolidinedione system and developed a synthetic strategy towards new lead compounds with antimicrobial activity. Efforts to synthesize analogues to build a structure–activity relationship (SAR) profile and optimize the activity are underway. Structure of leopolic acid A. Synthesis of
Automated glycan assembly of a S. pneumoniae serotype 3 CPS antigen
Beilstein J. Org. Chem. 2016, 12, 1440–1446, doi:10.3762/bjoc.12.139
- containing oligosaccharides of different lengths and frame shifts [19]. Synthetic oligosaccharide antigens enable structure–activity relationship (SAR) studies of bacterial antigens [20] to better understand antibody binding and help to improve existing vaccine formulations. Two synthetic routes to prepare
Discovery of an inhibitor of the production of the Pseudomonas aeruginosa virulence factor pyocyanin in wild-type cells
Beilstein J. Org. Chem. 2016, 12, 1428–1433, doi:10.3762/bjoc.12.137
- mode of action of 4 and structure–activity relationship studies are ongoing and results will be reported in due course. BHL and OdDHL are two natural AHL-based signaling molecules used by P. aeruginosain quorum sensing. PQS is a natural quinolone signaling molecule also used by P. aeruginosa in quorum
Antibacterial structure–activity relationship studies of several tricyclic sulfur-containing flavonoids
Beilstein J. Org. Chem. 2016, 12, 1065–1071, doi:10.3762/bjoc.12.100
- also established that dithiocarbamic flavanones of type 4 display no such activity. Therefore, only flavonoids 5a–m were tested against Staphylococus aureus (Gram positive) and Escherichia coli (Gram negative) in an attempt to establish an antimicrobial structure–activity relationship. Minimum
- A previously reported class of tricyclic flavonoids has been extended with the synthesis of thirteen new derivatives. These compounds were obtained from the corresponding 3-dithiocarbamic flavanones under acidic conditions. A study of their structure–activity relationship was performed with regard
- –activity relationship study concerning the antibacterial properties of several halogen-substituted tricyclic sulfur-containing flavonoids has been performed. The compounds have been synthesized by cyclocondensation of the corresponding 3-dithiocarbamic flavanones under acidic conditions. The influence of
Marine-derived myxobacteria of the suborder Nannocystineae: An underexplored source of structurally intriguing and biologically active metabolites
Beilstein J. Org. Chem. 2016, 12, 969–984, doi:10.3762/bjoc.12.96
- provided insights into the structure–activity relationship of haliangicin were generated in this study [55]. The huge potential to synthesize novel metabolites in the genus Haliangium is further corroborated by a PCR screening-based study for PKS sequences in H. tepidum among other myxobacteria [56]. The
Muraymycin nucleoside-peptide antibiotics: uridine-derived natural products as lead structures for the development of novel antibacterial agents
Beilstein J. Org. Chem. 2016, 12, 769–795, doi:10.3762/bjoc.12.77
- access to muraymycins and their analogues, some structure–activity relationship (SAR) studies and first insights into muraymycin biosynthesis. It therefore provides an overview on the current state of research, as well as an outlook on possible future developments in this field. Keywords: antibiotics
- ; natural products; nucleosides; peptides; structure–activity relationship; Introduction The treatment of infectious diseases caused by bacteria is a severe issue. With multiresistant bacterial strains rendering well-established therapeutic procedures ineffective, the exploration of novel antimicrobial
- reflect several effects such as target interaction, cellular uptake and potential resistance mechanisms of the microorganism. MIC values are therefore widely used, also in studies on muraymycin analogues (e.g., [22][76][77][78]) and have been the basis of many structure–activity relationship studies (see
Is conformation a fundamental descriptor in QSAR? A case for halogenated anesthetics
Beilstein J. Org. Chem. 2016, 12, 760–768, doi:10.3762/bjoc.12.76

- suggesting that these 2D MD´s can be advantageous over some three-dimensional descriptors. Keywords: conformational analysis; isoflurane; QSAR; theoretical calculations; volatile anesthetics; Introduction Quantitative structure–activity relationship (QSAR) studies try to find a correlation between chemical
A practical way to synthesize chiral fluoro-containing polyhydro-2H-chromenes from monoterpenoids
Beilstein J. Org. Chem. 2016, 12, 648–653, doi:10.3762/bjoc.12.64
- the presence of K10 montmorillonite clay forms chiral heterocyclic compounds with the hexahydro-2H-chromene scaffold 2 (Scheme 1) [1][2][3][4]. Products of these reactions are of interest as many of them exhibit a significant analgesic activity in vivo [2][3][4]. In terms of structure–activity
- relationship studies of hexahydro-2H-chromenes and similar compounds it is important to replace the hydroxy group at the C(5) position by another functional group. Thus, the approaches for synthesis of thio- [5] and nitrogen [6] containing analogous with reasonable yields (50−80%) by using a third component











































































































































































































































































































































