Search results
Search for "dehydrogenation" in Full Text gives 109 result(s) in Beilstein Journal of Organic Chemistry.
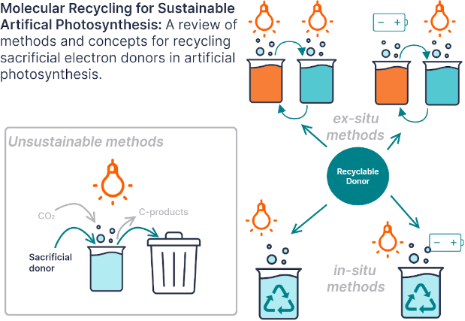
Enabling artificial photosynthesis systems with molecular recycling: A review of photo- and electrochemical methods for regenerating organic sacrificial electron donors
- Grace A. Lowe
Beilstein J. Org. Chem. 2023, 19, 1198–1215, doi:10.3762/bjoc.19.88
- electrochemical hydrogenation methods might be more appropriate [41][42]. There is already work on electrochemical dehydrogenation of LOHCs [76][77]. In one example, DDQ was used to remove hydrogen from secondary amines by oxidizing them, followed by reoxidation of the hydrogenated DDQ at the electrode to

Graphical Abstract

Figure 1: Diagram comparing the two reaction pathways for sacrificial electron donors (SD) in photocatalyzed ...

Figure 2: Diagram showing water-splitting systems developed by Girault, Scanlon, and co-workers that employ i...

Figure 3: Diagram illustrating the transfer of electrons in a photocatalytic particulate suspensions Z-scheme...

Figure 4: A. Structures of the molecules represented in part B. The numbers in brackets correspond to the com...

Figure 5: A. Structures of the molecules represented in part B. The numbers in brackets correspond to the com...
Strategies in the synthesis of dibenzo[b,f]heteropines
- David I. H. Maier,
- Barend C. B. Bezuidenhoudt and
- Charlene Marais
Beilstein J. Org. Chem. 2023, 19, 700–718, doi:10.3762/bjoc.19.51

- of o-nitrotoluene (22) Reduction to 2,2'-diaminobibenzyl (20) Ring-closing via amine condensation Catalytic dehydrogenation 1.1 Oxidative coupling of o-nitrotoluene (22) and reduction to 2,2'-diaminobibenzyl (20) The preparation of dinitrobibenzyl (21) can be achieved by the oxidative coupling of
- [36] via the polyphosphoric acid (PPA) catalysed cyclisation of 2,2'-diaminobibenzyl (20) at elevated temperatures (Scheme 3) [37][38]. 1.3 Catalytic dehydrogenation An early synthesis of 5H-dibenzo[b,f]azepine (1a) involved the gas phase dehydrogenation of 10,11-dihydro-5H-dibenzo[b,f]azepine (2a) to

Graphical Abstract

Figure 1: Dibenzo[b,f]azepine (1a), -oxepine (1b) and -thiepine (1c) as examples of dibenzo[b,f]heteropines (1...

Figure 2: Selected pharmaceuticals with the dibenzo[b,f]azepine skeleton.

Figure 3: Examples of 10,11-dihydrodibenzo[b,f]azepine-based ligands.

Figure 4: The dibenzo[b,f]azepine moiety in dyes with properties suitable for the use in organic light emitti...

Figure 5: Selective bioactive natural products (13–18) containing the dibenzo[b,f]oxepine scaffold and Novart...

Scheme 1: Retrosynthetic approach to 5H-dibenzo[b,f]azepine (1a) from nitrotoluene (22).

Scheme 2: Oxidative coupling of o-nitrotoluene (22) and reduction of 2,2'-dinitrobibenzyl (21) to form 2,2'-d...

Scheme 3: Synthesis of 10,11-dihydro-5H-dibenzo[b,f]azepine (2a) via amine condensation.

Scheme 4: Catalytic reduction of 10,11-dihydro-5H-dibenzo[b,f]azepine (2a).

Scheme 5: The Wagner–Meerwein rearrangement of acridin-9-ylmethanol (23) into 5H-dibenzo[b,f]azepine (1a).

Scheme 6: Oxidative ring expansion of 2-(9-xanthenyl)malonates 24.

Scheme 7: Ring expansion via C–H functionalisation.

Scheme 8: The synthesis of fluorinated 5H-dibenzo[b,f]azepine 38 from isatin (32).

Scheme 9: The synthesis of substituted dibenzo[b,f]azepines 43 from indoles 39.

Scheme 10: Retrosynthetic pathways to dibenzo[b,f]azepines via Buchwald–Hartwig amination.

Scheme 11: Synthesis of dibenzo[b,f]oxepine 54 and -azepine 55 derivatives via (i) Heck reaction and (ii) Buch...

Scheme 12: Double Buchwald–Hartwig amination and thioetherification in the synthesis of tricyclic azepines 60 ...

Scheme 13: Double Buchwald–Hartwig amination towards substituted dibenzoazepines 62.

Scheme 14: Double Buchwald–Hartwig amination towards 10,11-dihydro-5H-dibenzo[b,f]azepine derivatives 71.

Scheme 15: One-pot Suzuki coupling–Buchwald–Hartwig amination.

Scheme 16: One-pot Rh/Pd-catalysed synthesis of dihydropyridobenzazepines.

Scheme 17: A retrosynthetic pathway to dibenzo[b,f]azepines via Mizoroki–Heck reaction.

Scheme 18: One-pot domino Pd-catalyzed Mizoroki–Heck–Buchwald–Hartwig synthesis of dibenzo[b,f]azepines.

Scheme 19: Dibenzo[b,f]thiapine and -oxepine synthesis via SNAr (thio)etherification, Wittig methylenation and...

Scheme 20: A retrosynthetic pathway to dibenzo[b,f]oxepines via Ullmann coupling.

Scheme 21: Ullmann-type coupling in dibenzo[b,f]oxepine synthesis.

Scheme 22: Wittig reaction and Ullmann coupling as key steps in dihydrobenz[b,f]oxepine synthesis.

Scheme 23: Pd-catalysed dibenzo[b,f]azepine synthesis via norbornene azepine intermediate 109.

Scheme 24: A simple representation of olefin metathesis resulting in transalkylidenation.

Scheme 25: Ring-closing metathesis as key step in the synthesis of dibenzo[b,f]heteropines.

Scheme 26: Alkyne–aldehyde metathesis in the synthesis of dibenzo[b,f]heteropines.

Scheme 27: Hydroarylation of 9-(2-alkynylphenyl)-9H-carbazole derivatives.

Scheme 28: Oxidative coupling of bisphonium ylide intermediate to give pacharin (13).

Scheme 29: Preparation of 10,11-dihydrodibenzo[b,f]heteropines via intramolecular Wurtz reaction.

Scheme 30: Phenol deprotonation and intramolecular etherification in the synthesis of bauhinoxepine J.

Figure 6: Functionalisation of dibenzo[b,f]azepine.

Scheme 31: Palladium-catalysed N-arylation of dibenzo[b,f]azepine.

Scheme 32: Cu- and Ni-catalysed N-arylation.

Scheme 33: N-Alkylation of dibenzo[b,f]azepine (1a) and dihydrodibenzo[b,f]azepine (2a).

Scheme 34: Preparation of methoxyiminosilbene.

Scheme 35: Synthesis of oxcarbazepine (153) from methoxy iminostilbene 151.

Scheme 36: Ring functionalisation of dihydrodibenzo[b,f]azepine.
Transition-metal-catalyzed domino reactions of strained bicyclic alkenes
- Austin Pounder,
- Eric Neufeld,
- Peter Myler and
- William Tam
Beilstein J. Org. Chem. 2023, 19, 487–540, doi:10.3762/bjoc.19.38
- oxanorbornenes; however, the latter two substrates did not undergo dehydrogenation, generating cis-selective annulated coumarins (10b and 10d). In 2006, the same group applied this methodology for the total synthesis of arnottin I (10h), a coumarin-type natural product isolated from the bark of the Xanthoxylum
- 10. The selectivity for the non-dehydrogenated coumarins 10d is not understood, but 10b likely does not undergo dehydrogenation because there is no formation of aromaticity to drive the reaction forward. When the bicyclic alkene is substituted unsymmetrically at the bridgehead position, the reaction

Graphical Abstract

Figure 1: Ring-strain energies of homobicyclic and heterobicyclic alkenes in kcal mol−1. a) [2.2.1]-Bicyclic ...

Figure 2: a) Exo and endo face descriptions of bicyclic alkenes. b) Reactivity comparisons for different β-at...

Scheme 1: Ni-catalyzed ring-opening/cyclization cascade of heterobicyclic alkenes 1 with alkyl propiolates 2 ...

Scheme 2: Ni-catalyzed ring-opening/cyclization cascade of heterobicyclic alkenes 8 with β-iodo-(Z)-propenoat...

Scheme 3: Ni-catalyzed two- and three-component difunctionalizations of norbornene derivatives 15 with alkyne...

Scheme 4: Ni-catalyzed intermolecular three-component difunctionalization of oxabicyclic alkenes 1 with alkyn...

Scheme 5: Ni-catalyzed intermolecular three-component carboacylation of norbornene derivatives 15.

Scheme 6: Photoredox/Ni dual-catalyzed coupling of 4-alkyl-1,4-dihydropyridines 31 with heterobicyclic alkene...

Scheme 7: Photoredox/Ni dual-catalyzed coupling of α-amino radicals with heterobicyclic alkenes 30.

Scheme 8: Cu-catalyzed rearrangement/allylic alkylation of 2,3-diazabicyclo[2.2.1]heptenes 47 with Grignard r...

Scheme 9: Cu-catalyzed aminoboration of bicyclic alkenes 1 with bis(pinacolato)diboron (B2pin2) (53) and O-be...

Scheme 10: Cu-catalyzed borylalkynylation of oxabenzonorbornadiene (30b) with B2pin2 (53) and bromoalkynes 62.

Scheme 11: Cu-catalyzed borylacylation of bicyclic alkenes 1.

Scheme 12: Cu-catalyzed diastereoselective 1,2-difunctionalization of oxabenzonorbornadienes 30 for the synthe...

Scheme 13: Fe-catalyzed carbozincation of heterobicyclic alkenes 1 with arylzinc reagents 74.

Scheme 14: Co-catalyzed addition of arylzinc reagents of norbornene derivatives 15.

Scheme 15: Co-catalyzed ring-opening/dehydration of oxabicyclic alkenes 30 via C–H activation of arenes.

Scheme 16: Co-catalyzed [3 + 2] annulation/ring-opening/dehydration domino reaction of oxabicyclic alkenes 1 w...

Scheme 17: Co-catalyzed enantioselective carboamination of bicyclic alkenes 1 via C–H functionalization.

Scheme 18: Ru-catalyzed cyclization of oxabenzonorbornene derivatives with propargylic alcohols for the synthe...

Scheme 19: Ru-catalyzed coupling of oxabenzonorbornene derivatives 30 with propargylic alcohols and ethers 106...

Scheme 20: Ru-catalyzed ring-opening/dehydration of oxabicyclic alkenes via the C–H activation of anilides.

Scheme 21: Ru-catalyzed of azabenzonorbornadiene derivatives with arylamides.

Scheme 22: Rh-catalyzed cyclization of bicyclic alkenes with arylboronate esters 118.

Scheme 23: Rh-catalyzed cyclization of bicyclic alkenes with dienyl- and heteroaromatic boronate esters.

Scheme 24: Rh-catalyzed domino lactonization of doubly bridgehead-substituted oxabicyclic alkenes with seconda...

Scheme 25: Rh-catalyzed domino carboannulation of diazabicyclic alkenes with 2-cyanophenylboronic acid and 2-f...

Scheme 26: Rh-catalyzed synthesis of oxazolidinone scaffolds 147 through a domino ARO/cyclization of oxabicycl...

Scheme 27: Rh-catalyzed oxidative coupling of salicylaldehyde derivatives 151 with diazabicyclic alkenes 130a.

Scheme 28: Rh-catalyzed reaction of O-acetyl ketoximes with bicyclic alkenes for the synthesis of isoquinoline...

Scheme 29: Rh-catalyzed domino coupling reaction of 2-phenylpyridines 165 with oxa- and azabicyclic alkenes 30....

Scheme 30: Rh-catalyzed domino dehydrative naphthylation of oxabenzonorbornadienes 30 with N-sulfonyl 2-aminob...

Scheme 31: Rh-catalyzed domino dehydrative naphthylation of oxabenzonorbornadienes 30 with arylphosphine deriv...

Scheme 32: Rh-catalyzed domino ring-opening coupling reaction of azaspirotricyclic alkenes using arylboronic a...

Scheme 33: Tandem Rh(III)/Sc(III)-catalyzed domino reaction of oxabenzonorbornadienes 30 with alkynols 184 dir...

Scheme 34: Rh-catalyzed asymmetric domino cyclization and addition reaction of 1,6-enynes 194 and oxa/azabenzo...

Scheme 35: Rh/Zn-catalyzed domino ARO/cyclization of oxabenzonorbornadienes 30 with phosphorus ylides 201.

Scheme 36: Rh-catalyzed domino ring opening/lactonization of oxabenzonorbornadienes 30 with 2-nitrobenzenesulf...

Scheme 37: Rh-catalyzed domino C–C/C–N bond formation of azabenzonorbornadienes 30 with aryl-2H-indazoles 210.

Scheme 38: Rh/Pd-catalyzed domino synthesis of indole derivatives with 2-(phenylethynyl)anilines 212 and oxabe...

Scheme 39: Rh-catalyzed domino carborhodation of heterobicyclic alkenes 30 with B2pin2 (53).

Scheme 40: Rh-catalyzed three-component 1,2-carboamidation reaction of bicyclic alkenes 30 with aromatic and h...

Scheme 41: Pd-catalyzed diarylation and dialkenylation reactions of norbornene derivatives.

Scheme 42: Three-component Pd-catalyzed arylalkynylation reactions of bicyclic alkenes.

Scheme 43: Three-component Pd-catalyzed arylalkynylation reactions of norbornene and DFT mechanistic study.

Scheme 44: Pd-catalyzed three-component coupling N-tosylhydrazones 236, aryl halides 66, and norbornene (15a).

Scheme 45: Pd-catalyzed arylboration and allylboration of bicyclic alkenes.

Scheme 46: Pd-catalyzed, three-component annulation of aryl iodides 66, alkenyl bromides 241, and bicyclic alk...

Scheme 47: Pd-catalyzed double insertion/annulation reaction for synthesizing tetrasubstituted olefins.

Scheme 48: Pd-catalyzed aminocyclopropanation of bicyclic alkenes 1 with 5-iodopent-4-enylamine derivatives 249...

Scheme 49: Pd-catalyzed, three-component coupling of alkynyl bromides 62 and norbornene derivatives 15 with el...

Scheme 50: Pd-catalyzed intramolecular cyclization/ring-opening reaction of heterobicyclic alkenes 30 with 2-i...

Scheme 51: Pd-catalyzed dimer- and trimerization of oxabenzonorbornadiene derivatives 30 with anhydrides 268.

Scheme 52: Pd-catalyzed Catellani-type annulation and retro-Diels–Alder of norbornadiene 15b yielding fused xa...

Scheme 53: Pd-catalyzed hydroarylation and heteroannulation of urea-derived bicyclic alkenes 158 and aryl iodi...

Scheme 54: Access to fused 8-membered sulfoximine heterocycles 284/285 via Pd-catalyzed Catellani annulation c...

Scheme 55: Pd-catalyzed 2,2-bifunctionalization of bicyclic alkenes 1 generating spirobicyclic xanthone deriva...

Scheme 56: Pd-catalyzed Catellani-type annulation and retro-Diels–Alder of norbornadiene (15b) producing subst...

Scheme 57: Pd-catalyzed [2 + 2 + 1] annulation furnishing bicyclic-fused indanes 281 and 283.

Scheme 58: Pd-catalyzed ring-opening/ring-closing cascade of diazabicyclic alkenes 130a.

Scheme 59: Pd-NHC-catalyzed cyclopentannulation of diazabicyclic alkenes 130a.

Scheme 60: Pd-catalyzed annulation cascade generating diazabicyclic-fused indanones 292 and indanols 294.

Scheme 61: Pd-catalyzed skeletal rearrangement of spirotricyclic alkenes 176 towards large polycyclic benzofur...

Scheme 62: Pd-catalyzed oxidative annulation of aromatic enamides 298 and diazabicyclic alkenes 130a.

Scheme 63: Accessing 3,4,5-trisubstituted cyclopentenes 300, 301, 302 via the Pd-catalyzed domino reaction of ...

Scheme 64: Palladacycle-catalyzed ring-expansion/cyclization domino reactions of terminal alkynes and bicyclic...

Scheme 65: Pd-catalyzed carboesterification of norbornene (15a) with alkynes, furnishing α-methylene γ-lactone...
Germacrene B – a central intermediate in sesquiterpene biosynthesis
- Houchao Xu and
- Jeroen S. Dickschat
Beilstein J. Org. Chem. 2023, 19, 186–203, doi:10.3762/bjoc.19.18
- ., by heating with sulphur, to the blue azulene derivative 62 (Scheme 16C) [121][122][124][125][126], but the structure elucidation of this compound was only completed in 1936 [127]. Based on a comparison of IR spectra of natural terpenes, their hydrogenation and dehydrogenation products, the correct

Graphical Abstract

Scheme 1: Possible cyclisation modes of FPP.

Scheme 2: Structures of germacrene B (1), germacrene A (2) and hedycaryol (3).

Scheme 3: The chemistry of germacrene B (1). A) Synthesis from germacrone (4), B) the four conformers of 1 es...

Scheme 4: The chemistry of germacrene B (1). A) Cyclisation of 1 to 9 and 10 upon treatment with alumina, B) ...

Scheme 5: Possible cyclisation reactions upon reprotonation of 1. A) Cyclisations to eudesmane sesquiterpenes...

Scheme 6: Cyclisation modes for 1 to the eudesmane skeleton. A) The reprotonation of 1 at C-1 potentially lea...

Scheme 7: The sesquiterpenes derived from cation I1. WMR = Wagner–Meerwein rearrangement.

Scheme 8: The sesquiterpenes derived from cation I1. A) Pyrolysis of 23 to yield 9 and 10, B) deprotonation–r...

Scheme 9: The sesquiterpenes derived from cation I1. A) Acid-catalysed conversion of 18 into 26, B) conversio...

Scheme 10: The sesquiterpenes derived from cation I1. A) Formation of 20 by pyrolysis of 33, B) acid-catalysed...

Scheme 11: The sesquiterpenes derived from cation I2. WMR = Wagner–Meerwein rearrangement.

Scheme 12: The sesquiterpenes derived from cation I2. A) Acid catalysed conversion of 41 into 38, B) dehydrati...

Scheme 13: The sesquiterpenes derived from cation I3. WMR = Wagner–Meerwein rearrangement.

Scheme 14: Cyclisation modes for 1 to the guaiane skeleton. A) The reprotonation of 1 at C-4 potentially leads...

Scheme 15: The sesquiterpenes derived from cations K1, K2 and K4. A) Mechanisms of formation for compounds 53–...

Scheme 16: The sesquiterpenes derived from cations L1–L4. A) Mechanisms of formation for compounds 54, 56, 59 ...
Sequential hydrozirconation/Pd-catalyzed cross coupling of acyl chlorides towards conjugated (2E,4E)-dienones
- Benedikt Kolb,
- Daniela Silva dos Santos,
- Sanja Krause,
- Anna Zens and
- Sabine Laschat
Beilstein J. Org. Chem. 2023, 19, 176–185, doi:10.3762/bjoc.19.17
- condensation of enals 6 with aldehydes 7a or ketones 7b [6][7][8][9][10][11], isomerization of alkynones 8 [12][13][14][15], Horner–Wadsworth–Emmons reaction of unsaturated phosphonates 9 and aldehydes 10 [16][17], and dehydrogenation of enones 11 [18]. Further, Claisen rearrangement of vinyl propargylic

Graphical Abstract

Scheme 1: Examples of biologically active compounds with (2Ε,4E)-unsaturated ketone units.

Scheme 2: Selected examples for the synthesis of conjugated dienones from the literature [6-21].

Scheme 3: Previous work of hydrozirconations with Schwartz's reagent and our work [54,55,57,58,61,62].

Scheme 4: Synthesis of substituted enynes 25f–o via Corey–Fuchs reaction and Hunsdiecker reaction.

Scheme 5: Synthesis of non-natural (a) and natural (b) dienone-containing terpenes: synthesis of β-ionone (3)....
1,4-Dithianes: attractive C2-building blocks for the synthesis of complex molecular architectures
- Bram Ryckaert,
- Ellen Demeyere,
- Frederick Degroote,
- Hilde Janssens and
- Johan M. Winne
Beilstein J. Org. Chem. 2023, 19, 115–132, doi:10.3762/bjoc.19.12
- alternative approaches exist. The direct dehydrogenation of 1,4-dithiane is somewhat cumbersome [25], and, moreover, 1,4-dithiane (1) is surprisingly not available in large quantities at a reasonable cost, in spite of its apparent simplicity. Our lab has found that the synthetic method for dihydrodithiins
- then brominated with an excess of bromination reagent, which effects the 1,2-sulfur-migratory ring expansion, followed by bromination-induced dehydrogenation to the aromatic ring [31]. At the time of writing this review, the resulting benzoannelated dithiane 5 is also commercially available in small

Graphical Abstract

Scheme 1: 1,3-Dithianes as useful synthetic building blocks: a) general synthetic utility (in Corey–Seebach-t...

Scheme 2: Metalation of other saturated heterocycles is often problematic due to β-elimination [16,17].

Scheme 3: Thianes as synthetic building blocks in the construction of complex molecules [18].

Figure 1: a) 1,4-Dithiane-type building blocks that can serve as C2-synthons and b) examples of complex targe...

Scheme 4: Synthetic availability of 1,4-dithiane-type building blocks.

Scheme 5: Dithiins and dihydrodithiins as pseudoaryl groups [36-39].

Scheme 6: Metalation of other saturated heterocycles is often problematic due to β-elimination [40-42].

Figure 2: Reactive conformations leading to β-fragmentation for lithiated 1,4-dithianes and 1,4-dithiin.

Scheme 7: Mild metalation of 1,4-dithiins affords stable heteroaryl-magnesium and heteroaryl-zinc-like reagen...

Scheme 8: Dithiin-based dienophiles and their use in synthesis [33,49-54].

Scheme 9: Dithiin-based dienes and their use in synthesis [55-57].

Scheme 10: Stereoselective 5,6-dihydro-1,4-dithiin-based synthesis of cis-olefins [42,58].

Scheme 11: Addition to aldehydes and applications in stereoselective synthesis.

Figure 3: Applications in the total synthesis of complex target products with original attachment place of 1,...

Scheme 12: Direct C–H functionalization methods for 1,4-dithianes [82,83].

Scheme 13: Known cycloaddition reactivity modes of allyl cations [84-100].

Scheme 14: Cycloadditions of 1,4-dithiane-fused allyl cations derived from dihydrodithiin-methanol 90 [101-107].

Scheme 15: Dearomative [3 + 2] cycloadditions of unprotected indoles with 1,4-dithiane-fused allyl alcohol 90 [30]....

Scheme 16: Comparison of reactivity of dithiin-fused allyl alcohols and similar non-cyclic sulfur-substituted ...

Scheme 17: Applications of dihydrodithiins in the rapid assembly of polycyclic terpenoid scaffolds [108,109].

Scheme 18: Dihydrodithiin-mediated allyl cation and vinyl carbene cycloadditions via a gold(I)-catalyzed 1,2-s...

Scheme 19: Activation mode of ethynyldithiolanes towards gold-coordinated 1,4-dithiane-fused allyl cation and ...

Scheme 20: Desulfurization problems.

Scheme 21: oxidative decoration strategies for 1,4-dithiane scaffolds.
Total synthesis of grayanane natural products
- Nicolas Fay,
- Rémi Blieck,
- Cyrille Kouklovsky and
- Aurélien de la Torre
Beilstein J. Org. Chem. 2022, 18, 1707–1719, doi:10.3762/bjoc.18.181
- opening of the epoxide (15%). From 41 having the correct tetracyclic skeleton, a transient protection followed by Petasis olefination, deprotection, selenide-mediated α,β-dehydrogenation and Mukaiyama oxidation afforded an advanced intermediate 42 bearing most of the target’s functionalities. A sequence

Graphical Abstract

Figure 1: General structure of grayanane natural products.

Scheme 1: Grayanane biosynthesis.

Scheme 2: Matsumoto’s relay approach.

Scheme 3: Shirahama’s total synthesis of (–)-grayanotoxin III.

Scheme 4: Newhouse’s syntheses of fragments 25 and 29.

Scheme 5: Newhouse’s total synthesis of principinol D.

Scheme 6: Ding’s total synthesis of rhodomolleins XX and XXII.

Scheme 7: First key step of Luo’s strategy.

Scheme 8: Luo’s total synthesis of grayanotoxin III.

Scheme 9: Synthesis of principinol E and rhodomollein XX.

Scheme 10: William’s synthetic effort towards pierisformaside C.

Scheme 11: Hong’s synthetic effort towards rhodojaponin III.

Scheme 12: Recent strategies for grayanane synthesis.
Redox-active molecules as organocatalysts for selective oxidative transformations – an unperceived organocatalysis field
- Elena R. Lopat’eva,
- Igor B. Krylov,
- Dmitry A. Lapshin and
- Alexander O. Terent’ev
Beilstein J. Org. Chem. 2022, 18, 1672–1695, doi:10.3762/bjoc.18.179
- activation for hydrogenation of various organic substrates. More recently, SET reactivity of FLP was discovered [155]. The FLP-catalyzed dehydrogenation of N-protected indolines with H2 release [156] is depicted in Scheme 39. According to the proposed mechanism, the reaction starts with a hydride transfer
- heterocyclizations. Electrochemical N-ammonium ylide-catalyzed CH-oxidation. Oxidative dimerization of aryl- and alkenylmagnesium compounds catalyzed by quinonediimines. FLP-catalyzed dehydrogenation of N-substituted indolines. Funding This work was supported by the Russian Science Foundation (Grant no. 21-13-00205).

Graphical Abstract

Scheme 1: Organocatalysis classification used in the present perspective.

Scheme 2: Oxidative processes catalyzed by amines.

Scheme 3: N-Heterocyclic carbene (NHC) catalysis in oxidative functionalization of aldehydes.

Scheme 4: Examples of asymmetric oxidative processes catalyzed by chiral Brønsted acids.

Scheme 5: Asymmetric aerobic α-hydroxylation of lactams under phase-transfer organocatalysis conditions emplo...

Scheme 6: Selective CH-oxidation of methylarenes to aldehydes or carboxylic acids.

Scheme 7: An example of the regioselective CH-amination by a sterically hindered imide-N-oxyl radical precurs...

Scheme 8: CH-amination of ethylbenzene and CH-fluorination of aldehydes catalyzed by N-hydroxybenzimidazoles,...

Scheme 9: Mixed hetero-/homogeneous TiO2/N-hydroxyimide photocatalysis in the selective benzylic oxidation.

Scheme 10: Electrochemical benzylic iodination and benzylation of pyridine by benzyl iodides generated in situ...

Scheme 11: Electrochemical oxidative C–O/C–N coupling of alkylarenes with NHPI. Electrolysis conditions: Const...

Scheme 12: Chemoselective alcohol oxidation catalyzed by TEMPO.

Scheme 13: ABNO-catalyzed oxidative C–N coupling of primary alcohols with primary amines.

Scheme 14: ACT-catalyzed electrochemical oxidation of primary alcohols and aldehydes to carboxylic acids.

Scheme 15: Electrocatalytic oxidation of benzylic alcohols by a TEMPO derivative immobilized on a graphite ano...

Scheme 16: Electrochemical oxidation of carbamates of cyclic amines to lactams and oxidative cyanation of amin...

Scheme 17: Hydrogen atom transfer (HAT) and single-electron transfer (SET) as basic principles of amine cation...

Scheme 18: Electrochemical quinuclidine-catalyzed oxidation involving unactivated C–H bonds.

Scheme 19: DABCO-mediated photocatalytic C–C cross-coupling involving aldehyde C–H bond cleavage.

Scheme 20: DABCO-derived cationic catalysts in inactivated C–H bond cleavage for alkyl radical addition to ele...

Scheme 21: Electrochemical diamination and dioxygenation of vinylarenes catalyzed by triarylamines.

Scheme 22: Electrochemical benzylic oxidation mediated by triarylimidazoles.

Scheme 23: Thiyl radical-catalyzed CH-arylation of allylic substrates by aryl cyanides.

Scheme 24: Synthesis of redox-active alkyl tetrafluoropyridinyl sulfides by unactivated C–H bond cleavage by t...

Scheme 25: Main intermediates in quinone oxidative organocatalysis.

Scheme 26: Electrochemical DDQ-catalyzed intramolecular dehydrogenative aryl–aryl coupling.

Scheme 27: DDQ-mediated cross-dehydrogenative C–N coupling of benzylic substrates with azoles.

Scheme 28: Biomimetic o-quinone-catalyzed benzylic alcohol oxidation.

Scheme 29: Electrochemical synthesis of secondary amines by oxidative coupling of primary amines and benzylic ...

Scheme 30: General scheme of dioxirane and oxaziridine oxidative organocatalysis.

Scheme 31: Dioxirane organocatalyzed CH-hydroxylation involving aliphatic C(sp3)–H bonds.

Scheme 32: Enantioselective hydroxylation of CH-acids catalyzed by chiral oxaziridines.

Scheme 33: Iodoarene-organocatalyzed vinylarene diamination.

Scheme 34: Iodoarene-organocatalyzed asymmetric CH-hydroxylation of benzylic substrates.

Scheme 35: Iodoarene-organocatalyzed asymmetric difluorination of alkenes with migration of aryl or methyl gro...

Scheme 36: Examples of 1,2-diiodo-4,5-dimethoxybenzene-catalyzed electrochemical oxidative heterocyclizations.

Scheme 37: Electrochemical N-ammonium ylide-catalyzed CH-oxidation.

Scheme 38: Oxidative dimerization of aryl- and alkenylmagnesium compounds catalyzed by quinonediimines.

Scheme 39: FLP-catalyzed dehydrogenation of N-substituted indolines.
Cyclometalated iridium complexes-catalyzed acceptorless dehydrogenative coupling reaction: construction of quinoline derivatives and evaluation of their antimicrobial activities
- Hongling Shui,
- Yuhong Zhong,
- Renshi Luo,
- Zhanyi Zhang,
- Jiuzhong Huang,
- Ping Yang and
- Nianhua Luo
Beilstein J. Org. Chem. 2022, 18, 1507–1517, doi:10.3762/bjoc.18.159
- ][42], we found that cyclometalated iridium catalysts can effectively catalyze the dehydrogenation of alcohols to produce carbonyl compounds and hydrogen gas. Therefore, we used cyclometalated iridium complex (TC-6) to catalyze the ADC reaction of o-aminobenzyl alcohols 1 and aryl/heteroaryl/alkyl
- released from the dehydrogenation of 2-aminobenzyl alcohol/1-phenylethanol according to the previous literature [28]. Lastly, the desired product 3aa was obtained by the condensation and cyclization of the aldehyde 5 with acetophenone (6) under base conditions. The potential antimicrobial activity of the

Graphical Abstract

Figure 1: Some new quinoline antibacterial drugs.

Figure 2: Cyclometalated iridium-catalyzed ADC reaction of o-aminobenzyl alcohols and secondary alcohols.

Figure 3: Gram-scale transformations.

Figure 4: Mechanistic investigation.

Figure 5: A speculated possible mechanism.
Cytochrome P450 monooxygenase-mediated tailoring of triterpenoids and steroids in plants
- Karan Malhotra and
- Jakob Franke
Beilstein J. Org. Chem. 2022, 18, 1289–1310, doi:10.3762/bjoc.18.135
- (12) scaffold to 7β-hydroxythalianol, while CYP705A5 is a desaturase and introduces a double bond at C15 [41]. The related Arabidopsis CYP705A1 (also from clan 71) accepts a slightly different scaffold, arabidiol (11), triggering cleavage of the side chain at the same C15 instead of dehydrogenation

Graphical Abstract

Figure 1: Enzyme function of cytochrome P450 monooxygenases (CYPs). A) Typical net reaction of CYPs, resultin...

Figure 2: Phylogenetic distribution of CYPs acting on triterpenoid and steroid scaffolds (red nodes) compared...

Figure 3: CYPs modifying steroid (A), cucurbitacin steroid (B) and tetracyclic triterpene (C) backbones. Subs...

Figure 4: CYPs modifying pentacyclic 6-6-6-6-6 triterpenes. Substructures in grey indicate regions where majo...

Figure 5: CYPs modifying pentacyclic 6-6-6-6-5 triterpenes (A) and unusual triterpenes (B). Substructures in ...

Figure 6: Recent examples of multifunctional CYPs in triterpenoid and steroid metabolism in plants that insta...
Inductive heating and flow chemistry – a perfect synergy of emerging enabling technologies
- Conrad Kuhwald,
- Sibel Türkhan and
- Andreas Kirschning
Beilstein J. Org. Chem. 2022, 18, 688–706, doi:10.3762/bjoc.18.70
- applications. NiO2, on the other hand, was used to achieve the dehydrogenation of amines (to nitriles) and to perform the α,β dehydrogenation of ketones 61. 3.2.3 Using chemically active fixed beds (catalysts): Copper metal in the form of wires or turnings can also be inductively heated when placed inside flow

Graphical Abstract

Figure 1: Inductive heating, a powerful tool in industry and the Life Sciences.

Figure 2: Electric displacement field of a ferromagnetic and superparamagnetic material.

Figure 3: Temperature profiles of reactors heated conventionally and by RF heating (Figure 3 redrawn from [24]).

Scheme 1: Continuous flow synthesis of isopulegol (2) from citronellal (1).

Scheme 2: Dry (reaction 1) and steam (reaction 2) methane reforming.

Scheme 3: Calcination and RF heating.

Scheme 4: The continuously operated “Sabatier” process.

Scheme 5: Biofuel production from biomass using inductive heating for pyrolysis.

Scheme 6: Water electrolysis using an inductively heated electrolysis cell.

Scheme 7: Dimroth rearrangement (reaction 1) and three-component reaction (reaction 2) to propargyl amines 8 ...

Figure 4: A. Flow reactor filled with magnetic nanostructured particles (MagSilicaTM) and packed bed reactor ...

Scheme 8: Claisen rearrangement in flow: A. comparison between conventional heating (external oil bath), micr...

Scheme 9: Continuous flow reactions and comparison with batch reaction (oil bath). A. Pd-catalyzed transfer h...

Scheme 10: Continuous flow reactions and comparison with batch reaction (oil bath). A. pericyclic reactions an...

Scheme 11: Reactions under flow conditions using inductively heated fixed-bed materials serving as stoichiomet...

Scheme 12: Reactions under flow conditions using inductively heated fixed-bed materials serving as catalysts: ...

Scheme 13: Two step flow protocol for the preparation of 1,1'-diarylalkanes 77 from ketones and aldehydes 74, ...

Scheme 14: O-Alkylation, the last step in the multistep flow synthesis of Iloperidone (80) accompanied with a ...

Scheme 15: Continuous two-step flow process consisting of Grignard reaction followed by water elimination bein...

Scheme 16: Inductively heated continuous flow protocol for the synthesis of Iso E Super (88) [91,92].

Scheme 17: Three-step continuous flow synthesis of macrocycles 89 and 90 with musk-like olfactoric properties.
DDQ in mechanochemical C–N coupling reactions
- Shyamal Kanti Bera,
- Rosalin Bhanja and
- Prasenjit Mal
Beilstein J. Org. Chem. 2022, 18, 639–646, doi:10.3762/bjoc.18.64
- moiety in 2,3-dichloro-5,6-dicyano-1,4-benzoquinone (DDQ), it was well established as a hydride transfer reagent in various organic reactions [14][15]. Generally, DDQ assists in dehydrogenation reactions in organic synthesis [16]. In this context, various carbon–heteroatom bond formation reactions such

Graphical Abstract

Figure 1: Our work on mechanochemical C–N coupling reactions using DDQ. The newly formed C–N bonds are shown ...

Figure 2: Scope of the mechanochemical synthesis of substituted benzimidazoles.

Figure 3: Synthesis of quinazolin-4(3H)-one derivatives.

Figure 4: The substrate scope for the synthesis of quinazolin-4(3H)-one derivatives.

Figure 5: a) Control experiment and b) Plausible mechanism.

Figure 6: Large-scale synthesis. a) 1,2-Disubstituted benzimidazoles. b) Substituted quinazolin-4(3H)-ones. R...
Menadione: a platform and a target to valuable compounds synthesis
- Acácio S. de Souza,
- Ruan Carlos B. Ribeiro,
- Dora C. S. Costa,
- Fernanda P. Pauli,
- David R. Pinho,
- Matheus G. de Moraes,
- Fernando de C. da Silva,
- Luana da S. M. Forezi and
- Vitor F. Ferreira
Beilstein J. Org. Chem. 2022, 18, 381–419, doi:10.3762/bjoc.18.43
- 1,3-butadiene, followed by dehydrogenation gave menadione (10). This proved to be a good synthetic route, leading to menadione in approximately 80% overall yield (Scheme 6). Another interesting synthetic approach was reported by Mal and co-workers, who synthetized menadione (10) via a

Graphical Abstract

Figure 1: Natural bioactive naphthoquinones.

Figure 2: Chemical structures of vitamins K.

Figure 3: Redox cycle of menadione.

Scheme 1: Selected approaches for menadione synthesis using silver(I) as a catalyst.

Scheme 2: Methylation approaches for the preparation of menadione from 1,4-naphthoquinone using tert-butyl hy...

Scheme 3: Methylation approach of 1,4-naphthoquinone using i) rhodium complexes/methylboronic acid and ii) bi...

Scheme 4: Synthesis of menadione (10) from itaconic acid.

Scheme 5: Menadione synthesis via Diels–Alder reaction.

Scheme 6: Synthesis of menadione (10) using p-cresol as a synthetic precursor.

Scheme 7: Synthesis of menadione (10) via demethoxycarbonylating annulation of methyl methacrylate.

Scheme 8: Furan 34 used as a diene in a Diels–Alder reaction for the synthesis of menadione (10).

Scheme 9: o-Toluidine as a dienophile in a Diels–Alder reaction for the synthesis of menadione (10).

Scheme 10: Representation of electrochemical synthesis of menadione.

Figure 4: Reaction sites and reaction types of menadione as substrate.

Scheme 11: DBU-catalyzed epoxidation of menadione (10).

Scheme 12: Phase-transfer catalysis for the epoxidation of menadione.

Scheme 13: Menadione epoxidation using a hydroperoxide derived from (+)-norcamphor.

Scheme 14: Enantioselective Diels–Alder reaction for the synthesis of asymmetric quinone 50 catalyzed by a chi...

Scheme 15: Optimized reaction conditions for the synthesis of anthra[9,1-bc]pyranone.

Scheme 16: Synthesis of anthra[9,1-bc]furanone, anthra[9,1-bc]pyridine, and anthra[9,1-bc]pyrrole derivatives.

Scheme 17: Synthesis of derivatives employing protected trienes.

Scheme 18: Synthesis of cyclobutene derivatives of menadione.

Scheme 19: Menadione reduction reactions using sodium hydrosulfite.

Scheme 20: Green methodology for menadiol synthesis and pegylation.

Scheme 21: Menadione reduction by 5,6-O-isopropylidene-ʟ-ascorbic acid under UV light irradiation.

Scheme 22: Selected approaches of menadione hydroacetylation to diacetylated menadiol.

Scheme 23: Thiele–Winter reaction catalyzed by Bi(OTf)3.

Scheme 24: Carbonyl condensation of menadione using resorcinol and a hydrazone derivative.

Scheme 25: Condensation reaction of menadione with thiosemicarbazide.

Scheme 26: Condensation reaction of menadione with acylhydrazides.

Scheme 27: Menadione derivatives functionalized with organochalcogens.

Scheme 28: Synthesis of selenium-menadione conjugates derived from chloromethylated menadione 84.

Scheme 29: Menadione alkylation by the Kochi–Anderson method.

Scheme 30: Menadione alkylation by diacids.

Scheme 31: Menadione alkylation by heterocycles-substituted carboxylic acids.

Scheme 32: Menadione alkylation by bromoalkyl-substituted carboxylic acids.

Scheme 33: Menadione alkylation by complex carboxylic acids.

Scheme 34: Kochi–Anderson method variations for the menadione alkylation via oxidative decarboxylation of carb...

Scheme 35: Copper-catalyzed menadione alkylation via free radicals.

Scheme 36: Nickel-catalyzed menadione cyanoalkylation.

Scheme 37: Iron-catalyzed alkylation of menadione.

Scheme 38: Selected approaches to menadione alkylation.

Scheme 39: Menadione acylation by photo-Friedel–Crafts acylation reported by Waske and co-workers.

Scheme 40: Menadione acylation by Westwood procedure.

Scheme 41: Synthesis of 3-benzoylmenadione via metal-free TBAI/TBHP system.

Scheme 42: Michael-type addition of amines to menadione reported by Kallmayer.

Scheme 43: Synthesis of amino-menadione derivatives using polyalkylamines.

Scheme 44: Selected examples for the synthesis of different amino-substituted menadione derivatives.

Scheme 45: Selected examples of Michael-type addition of complex amines to menadione (10).

Scheme 46: Addition of different natural α-amino acids to menadione.

Scheme 47: Michael-type addition of amines to menadione using silica-supported perchloric acid.

Scheme 48: Indolylnaphthoquinone or indolylnaphthalene-1,4-diol synthesis reported by Yadav et al.

Scheme 49: Indolylnaphthoquinone synthesis reported by Tanoue and co-workers.

Scheme 50: Indolylnaphthoquinone synthesis from menadione by Escobeto-González and co-workers.

Scheme 51: Synthesis of menadione analogues functionalized with thiols.

Scheme 52: Synthesis of menadione-derived symmetrical derivatives through reaction with dithiols.

Scheme 53: Mercaptoalkyl acids as nucleophiles in Michael-type addition reaction to menadione.

Scheme 54: Reactions of menadione (10) with cysteine derivatives for the synthesis of quinoproteins.

Scheme 55: Synthesis of menadione-glutathione conjugate 152 by Michael-type addition.
Recent advances and perspectives in ruthenium-catalyzed cyanation reactions
- Thaipparambil Aneeja,
- Cheriya Mukkolakkal Abdulla Afsina,
- Padinjare Veetil Saranya and
- Gopinathan Anilkumar
Beilstein J. Org. Chem. 2022, 18, 37–52, doi:10.3762/bjoc.18.4
- afforded the products in excellent yields. The authors also conducted various experimental and theoretical studies to analyze the reaction mechanism. The proposed mechanism begins with the oxidative dehydrogenation of the alcohol to afford the aldehyde which undergoes condensation with ammonia to give the
- corresponding imine. Finally, oxidative dehydrogenation results in the formation of the nitrile. Conclusion This review summarizes the recent progress in ruthenium-catalyzed cyanation reactions. Due to the wide application of nitrile compounds in pharmaceutical and biological fields, cyanation reactions have

Graphical Abstract

Scheme 1: Starch-immobilized ruthenium trichloride-catalyzed cyanation of tertiary amines.

Scheme 2: Proposed mechanism for the cyanation of tertiary amines using starch-immobilized ruthenium trichlor...

Scheme 3: Cyanation of tertiary amines using heterogeneous Ru/C catalyst.

Scheme 4: Proposed mechanism for cyanation of tertiary amines using a heterogeneous Ru/C catalyst.

Scheme 5: Ruthenium-carbamato complex-catalyzed oxidative cyanation of tertiary amines.

Scheme 6: Cyanation of tertiary amines using immobilized MCM-41-2N-RuCl3 as the catalyst.

Scheme 7: Cyanation of tertiary amines using RuCl3·nH2O as the catalyst and molecular oxygen as oxidant.

Scheme 8: RuCl3-catalyzed cyanation of tertiary amines using NaCN/HCN and H2O2 as oxidant.

Scheme 9: Proposed mechanism for the ruthenium-catalyzed oxidative cyanation using H2O2.

Scheme 10: Proposed mechanism for the ruthenium-catalyzed aerobic oxidative cyanation.

Scheme 11: RuCl3-catalyzed oxidative cyanation of tertiary amines using acetone cyanohydrin as the cyanating a...

Scheme 12: Cyanation of indoles using K4[Fe(CN)6] as cyano source and Ru(III)-exchanged NaY zeolite (RuY) as c...

Scheme 13: Cyanation of arenes and heteroarenes using a ruthenium(II) catalyst and N-cyano-N-phenyl-p-toluenes...

Scheme 14: Proposed mechanism for the cyanation of arenes and heteroarenes using ruthenium(II) as catalyst and...

Scheme 15: Synthesis of N-(2-cyanoaryl)-7-azaindoles.

Figure 1: Structure of the TiO2-immobilized ruthenium polyazine complex.

Scheme 16: Visible-light-induced oxidative cyanation of aza-Baylis–Hillman adducts.

Scheme 17: Synthesis of 1° alkyl nitriles using [Ru(bpy)3](PF6)2 as the photocatalyst.

Scheme 18: Synthesis of 2° and 3° alkyl nitriles using [Ru(bpy)3](PF6)2 as the photocatalyst.

Scheme 19: Photoredox cross coupling reaction.

Scheme 20: Synthesis of α-amino nitriles from amines via a one-pot strategy.

Scheme 21: Proposed mechanistic pathway for the cyanation of the aldimine intermediate.

Scheme 22: Strecker-type functionalization of N-aryl-substituted tetrahydroisoquinolines under flow conditions....

Scheme 23: One-pot synthesis of α-aminonitriles using RuCl3 as catalyst.

Scheme 24: Synthesis of alkyl nitriles using (Ru(TMHD)3) as the catalyst.

Scheme 25: Synthesis of cyanated isoxazolines from alkenyl oximes catalyzed by [RuCl2(p-cymene)]2 in the prese...

Scheme 26: Proposed mechanism for the synthesis of cyanated isoxazolines from alkenyl oximes.

Scheme 27: Oxidative cyanation of differently substituted alcohols.
Efficient N-arylation of 4-chloroquinazolines en route to novel 4-anilinoquinazolines as potential anticancer agents
- Rodolfo H. V. Nishimura,
- Thiago dos Santos,
- Valter E. Murie,
- Luciana C. Furtado,
- Leticia V. Costa-Lotufo and
- Giuliano C. Clososki
Beilstein J. Org. Chem. 2021, 17, 2968–2975, doi:10.3762/bjoc.17.206
- that, the cyclocondensation [29] of the halogenated anthranilamides 6a,b with benzaldehyde followed by dehydrogenation promoted by iodine gave the corresponding quinazolin-4(3H)-ones 7a,b, which we used in the next step without purification. Finally, chlorination [31] of quinazolin-4(3H)-ones 7a,b by

Graphical Abstract

Figure 1: Some antitumor agents containing the 4-anilinoquinazoline moiety.

Scheme 1: Examples of N-arylation reactions using 4-chloroquinazolines as substrates.

Scheme 2: Synthesis of verubulin analog.

Scheme 3: Synthesis of 4-chloro-6-halo-2-phenylquinazolines 8a and 8b. Conditions: a) NBS, CH3CN, 30 min, 25 ...

Scheme 4: N-Arylation reactions using ortho-, meta-, and para-substituted primary anilines of type 14 followe...

Scheme 5: N-Arylation reactions using 4-chloroquinazoline (16) and 4-chloro-2-methylquinazoline (17) to achie...
Recent advances in the asymmetric phosphoric acid-catalyzed synthesis of axially chiral compounds
- Alemayehu Gashaw Woldegiorgis and
- Xufeng Lin
Beilstein J. Org. Chem. 2021, 17, 2729–2764, doi:10.3762/bjoc.17.185
- accelerating imine formation (I-19), and under the catalysis of a chiral phosphoric acid, intramolecular nucleophilic addition occurs to form I-20, followed by oxidative dehydrogenation with 2,3-dichloro-5,6-dicyano-1,4-benzoquinone (DDQ). In the presence of 10 mol % chiral phosphoric acid CPA 7, the axially

Graphical Abstract

Figure 1: Representative examples of axially chiral biaryls, heterobiaryls, spiranes and allenes as ligands a...

Figure 2: Selected examples of axially chiral drugs and bioactive molecules.

Figure 3: Axially chiral functional materials and supramolecules.

Figure 4: Important chiral phosphoric acid scaffolds used in this review.

Scheme 1: Atroposelective aryl–aryl-bond formation by employing a facile [3,3]-sigmatropic rearrangement.

Scheme 2: Atroposelective synthesis of axially chiral biaryl amino alcohols 5.

Scheme 3: The enantioselective reaction of quinone and 2-naphthol derivatives.

Scheme 4: Enantioselective synthesis of multisubstituted biaryls.

Scheme 5: Enantioselective synthesis of axially chiral quinoline-derived biaryl atropisomers mediated by chir...

Scheme 6: Pd-Catalyzed atroposelective C–H olefination of biarylamines.

Scheme 7: Palladium-catalyzed directed atroposelective C–H allylation.

Scheme 8: Enantioselective synthesis of axially chiral (a) aryl indoles and (b) biaryldiols.

Scheme 9: Asymmetric arylation of indoles enabled by azo groups.

Scheme 10: Proposed mechanism for the asymmetric arylation of indoles.

Scheme 11: Enantioselective synthesis of axially chiral N-arylindoles [38].

Scheme 12: Enantioselective [3 + 2] formal cycloaddition and central-to-axial chirality conversion.

Scheme 13: Organocatalytic atroposelective arene functionalization of nitrosonaphthalene with indoles.

Scheme 14: Proposed reaction mechanism for the atroposelective arene functionalization of nitrosonaphthalenes.

Scheme 15: Asymmetric construction of axially chiral naphthylindoles [65].

Scheme 16: Enantioselective synthesis of axially chiral 3,3’-bisindoles [66].

Scheme 17: Atroposelective synthesis of 3,3’-bisiindoles bearing axial and central chirality.

Scheme 18: Enantioselective synthesis of axially chiral 3,3’-bisindoles bearing single axial chirality.

Scheme 19: Enantioselective reaction of azonaphthalenes with various pyrazolones.

Scheme 20: Enantioselective and atroposelective synthesis of axially chiral N-arylcarbazoles [73].

Scheme 21: Atroposelective cyclodehydration reaction.

Scheme 22: Atroposelective construction of axially chiral N-arylbenzimidazoles [78].

Scheme 23: Proposed reaction mechanism for the atroposelective synthesis of axially chiral N-arylbenzimidazole...

Scheme 24: Atroposelective synthesis of axially chiral arylpyrroles [21].

Scheme 25: Synthesis of axially chiral arylquinazolinones and its reaction pathway [35].

Scheme 26: Synthesis of axially chiral aryquinoline by Friedländer heteroannulation reaction and its proposed...

Scheme 27: Povarov cycloaddition–oxidative chirality conversion process.

Scheme 28: Atroposelective synthesis of oxindole-based axially chiral styrenes via kinetic resolution.

Scheme 29: Synthesis of axially chiral alkene-indole frame works [45].

Scheme 30: Proposed reaction mechanism for axially chiral alkene-indoles.

Scheme 31: Atroposelective C–H aminations of N-aryl-2-naphthylamines with azodicarboxylates.

Scheme 32: Synthesis of brominated atropisomeric N-arylquinoids.

Scheme 33: The enantioselective syntheses of axially chiral SPINOL derivatives.

Scheme 34: γ-Addition reaction of various 2,3-disubstituted indoles to β,γ-alkynyl-α-imino esters.

Scheme 35: Regio- and stereoselective γ-addition reactions of isoxazol-5(4H)-ones to β,γ-alkynyl-α-imino ester...

Scheme 36: Synthesis of chiral tetrasubstituted allenes and naphthopyrans.

Scheme 37: Asymmetric remote 1,8-conjugate additions of thiazolones and azlactones to propargyl alcohols.

Scheme 38: Synthesis of chiral allenes from 1-substituted 2-naphthols [107].
Electrocatalytic C(sp3)–H/C(sp)–H cross-coupling in continuous flow through TEMPO/copper relay catalysis
- Bin Guo and
- Hai-Chao Xu
Beilstein J. Org. Chem. 2021, 17, 2650–2656, doi:10.3762/bjoc.17.178
- through reversible reaction with this cationic species. Conclusion In summary, we have achieved the electrochemical dehydrogenation cross-coupling of tetrahydroisoquinolines with terminal alkynes in continuous flow through Cu/TEMPO relay catalysis. This work demonstrates that continuous-flow

Graphical Abstract

Scheme 1: C(sp3)–H alkynylation of tetrahydroisoquinolines. L* = chiral ligand. TEMPO = 2,2,6,6-tetramethylpi...

Scheme 2: Substrate scope. Reaction conditions: Pt anode, Pt cathode, interelectrode distance 0.25 mm, 1 (0.0...

Scheme 3: Reaction scale-up.

Scheme 4: Proposed mechanism.
Synthesis of new bile acid-fused tetrazoles using the Schmidt reaction
- Dušan Đ. Škorić,
- Olivera R. Klisurić,
- Dimitar S. Jakimov,
- Marija N. Sakač and
- János J. Csanádi
Beilstein J. Org. Chem. 2021, 17, 2611–2620, doi:10.3762/bjoc.17.174
- selective acetylation [43], followed by oxidation. Enones 4 and 8 were prepared by dehydrogenation of corresponding ketones with SeO2 in refluxing acetic acid [44]. Microwave-assisted heating of the reaction mixture in a closed vessel (150 °C) helped in decreasing the reaction time for dehydrogenation

Graphical Abstract

Figure 1: Structures of the steroidal tetrazoles that showed anticancer potential in vitro.

Figure 2: Mechanism of the Schmidt reaction.

Scheme 1: Synthesis of 12-oxo intermediates. Reagents and conditions: a) EtOAc, pTsOH, reflux, 14 h (81%); b)...

Scheme 2: Synthesis of 7-oxo intermediate 11 from chenodeoxycholic acid (9). Reagents and conditions: a) EtOA...

Figure 3: Mercury [51] drawing of the molecular structures of compounds 13 and 14, with labelling of nonhydrogen ...

Figure 4: Dose dependence of the cytotoxicity of tested compounds on treated cell lines. All compounds were t...
Efficient synthesis of polyfunctionalized carbazoles and pyrrolo[3,4-c]carbazoles via domino Diels–Alder reaction
- Ren-Jie Fang,
- Chen Yan,
- Jing Sun,
- Ying Han and
- Chao-Guo Yan
Beilstein J. Org. Chem. 2021, 17, 2425–2432, doi:10.3762/bjoc.17.159
- benzylideneacetone in acetonitrile in the presence of p-TsOH and DDQ resulted in polyfunctionalized carbazoles in satisfactory yields. The reaction mechanism included the DDQ oxidative dehydrogenation of 3-(indol-3-yl)-1,3-diphenylpropan-1-ones to the corresponding 3-vinylindoles, their acid-catalyzed Diels–Alder
- dehydrogenation reaction was carried out in acetonitrile at room temperature. A series of aromatized pyrrolo[3,4-c]carbazoles 4a–l were successfully synthesized by the one-pot two-step reaction and the results are summarized in Table 1. All reactions proceeded smoothly to give the corresponding pyrrolo[3,4-c
- active diene, indole-substituted chalcones. Then, the p-TsOH-catalyzed Diels–Alder reaction of indole-chalcones with second chalcones and sequential aromatization through DDQ dehydrogenation resulted in the polyfunctionalized carbazoles 6a–l in good yields (Table 2). Additionally, the similar reaction

Graphical Abstract

Figure 1: Representative bioactive carbazole derivatives.

Scheme 1: Synthesis of tetrahydropyrrolo[3,4-c]carbazoles 3a and 3b.

Figure 2: Single crystal structure of the isomer 3a.

Figure 3: Single crystal structure of the isomer 3b.

Figure 4: Single crystal structure of the isomer 4g.

Scheme 2: Proposed domino reaction mechanism for the formation of carbazoles 6.
Breaking paracyclophane: the unexpected formation of non-symmetric disubstituted nitro[2.2]metaparacyclophanes
- Suraj Patel,
- Tyson N. Dais,
- Paul G. Plieger and
- Gareth J. Rowlands
Beilstein J. Org. Chem. 2021, 17, 1518–1526, doi:10.3762/bjoc.17.109
- highly oxidizing conditions, dehydrogenation of the resulting cyclohexadienol would give 11.The electron-rich 4-hydroxy[2.2]metaparacyclophane (9) participates in ortho selective nitration to give 5. Nitric acid has previously been used to oxidize phenols to cyclohexadienones [69][70], and a plausible

Graphical Abstract

Figure 1: The common [2.2]cyclophanes.

Scheme 1: Nitration of [2.2]paracyclophane (1) and the synthesis of 4-hydroxy-5-nitro[2.2]metaparacyclophane (...

Figure 2: Crystal structure of 5. Ellipsoids are drawn at a 50% probability level [63-66].

Figure 3: Crystal structure of 6. Ellipsoids are drawn at a 50% probability level [63].

Scheme 2: Possible mechanism for the formation of [2.2]metaparacyclophane 5 and cyclohexadienone cyclophane 6...

Scheme 3: Conjugate addition of methanol and subsequent elimination.

Figure 4: Crystal structure of 14. Ellipsoids are drawn at a 50% probability level [63].

Figure 5: Crystal structure of 15. Ellipsoids are drawn at a 50% probability level [63].

Figure 6: Possible origin of stereoselectivity.
N-tert-Butanesulfinyl imines in the asymmetric synthesis of nitrogen-containing heterocycles
- Joseane A. Mendes,
- Paulo R. R. Costa,
- Miguel Yus,
- Francisco Foubelo and
- Camilla D. Buarque
Beilstein J. Org. Chem. 2021, 17, 1096–1140, doi:10.3762/bjoc.17.86


Graphical Abstract

Scheme 1: General strategy for the enantioselective synthesis of N-containing heterocycles from N-tert-butane...

Scheme 2: Methodologies for condensation of aldehydes and ketones with tert-butanesulfinamides (1).

Scheme 3: Transition models for cis-aziridines and trans-aziridines.

Scheme 4: Mechanism for the reduction of N-tert-butanesulfinyl imines.

Scheme 5: Transition models for the addition of organomagnesium and organolithium compounds to N-tert-butanes...

Scheme 6: Synthesis of 2,2-dibromoaziridines 15 from aldimines 14 and bromoform, and proposed non-chelation-c...

Scheme 7: Diastereoselective synthesis of aziridines from tert-butanesulfinyl imines.

Scheme 8: Synthesis of vinylaziridines 22 from aldimines 14 and 1,3-dibromopropene 23, and proposed chelation...

Scheme 9: Synthesis of vinylaziridines 27 from aldimines 14 and α-bromoesters 26, and proposed transition sta...

Scheme 10: Synthesis of 2-chloroaziridines 28 from aldimines 14 and dichloromethane, and proposed transition s...

Scheme 11: Synthesis of cis-vinylaziridines 30 and 31 from aldimines 14 and bromomethylbutenolide 29.

Scheme 12: Synthesis of 2-chloro-2-aroylaziridines 36 and 32 from aldimines 14, arylnitriles 34, and silyldich...

Scheme 13: Synthesis of trifluoromethylaziridines 39 and proposed transition state of the aziridination.

Scheme 14: Synthesis of aziridines 42 and proposed state transition.

Scheme 15: Synthesis of 1-substituted 2-azaspiro[3.3]heptanes, 1-phenyl-2-azaspiro[3.4]octane and 1-phenyl-2-a...

Scheme 16: Synthesis of 1-substituted 2,6-diazaspiro[3.3]heptanes 48 from chiral imines 14 and 1-Boc-azetidine...

Scheme 17: Synthesis of β-lactams 52 from chiral imines 14 and dimethyl malonate (49).

Scheme 18: Synthesis of spiro-β-lactam 57 from chiral (RS)-N-tert-butanesulfinyl isatin ketimine 53 and ethyl ...

Scheme 19: Synthesis of β-lactam 60, a precursor of (−)-batzelladine D (61) and (−)-13-epi-batzelladine D (62)...

Scheme 20: Rhodium-catalyzed asymmetric synthesis of 3-substituted pyrrolidines 66 from chiral imine (RS)-63 a...

Scheme 21: Asymmetric synthesis of 1,3-disubstituted isoindolines 69 and 70 from chiral imine 67.

Scheme 22: Asymmetric synthesis of cis-2,5-disubstituted pyrrolidines 73 from chiral imine (RS)-71.

Scheme 23: Asymmetric synthesis of 3-hydroxy-5-substituted pyrrolidin-2-ones 77 from chiral imine (RS)-74.

Scheme 24: Asymmetric synthesis of 4-hydroxy-5-substituted pyrrolidin-2-ones 80 from chiral imines 79.

Scheme 25: Asymmetric synthesis of 3-pyrrolines 82 from chiral imines 14 and ethyl 4-bromocrotonate (81).

Scheme 26: Asymmetric synthesis of γ-amino esters 84, and tetramic acid derivative 86 from chiral imines (RS)-...

Scheme 27: Asymmetric synthesis of α-methylene-γ-butyrolactams 90 from chiral imines (Z,SS)-87 and ethyl 2-bro...

Scheme 28: Asymmetric synthesis of methylenepyrrolidines 92 from chiral imines (RS)-14 and 2-(trimethysilylmet...

Scheme 29: Synthesis of dibenzoazaspirodecanes from cyclic N-tert-butanesulfinyl imines.

Scheme 30: Stereoselective synthesis of cyclopenta[c]proline derivatives 103 from β,γ-unsaturated α-amino acid...

Scheme 31: Stereoselective synthesis of alkaloids (−)-angustureine (107) and (−)-cuspareine (108).

Scheme 32: Stereoselective synthesis of alkaloids (−)-pelletierine (112) and (+)-coniine (117).

Scheme 33: Synthesis of piperidine alkaloids (+)-dihydropinidine (122a), (+)-isosolenopsin (122b) and (+)-isos...

Scheme 34: Stereoselective synthesis of the alkaloids(+)-sedamine (125) from chiral imine (SS)-119.

Scheme 35: Stereoselective synthesis of trans-5-hydroxy-6-substituted-2-piperidinones 127 and 129 from chiral ...

Scheme 36: Stereoselective synthesis of trans-5-hydroxy-6-substituted ethanone-2-piperidinones 132 from chiral...

Scheme 37: Stereoselective synthesis of trans-3-benzyl-5-hydroxy-6-substituted-2-piperidinones 136 from chiral...

Scheme 38: Stereoselective synthesis of trans-5-hydroxy-6-substituted 2-piperidinones 139 from chiral imine 138...

Scheme 39: Stereoselective synthesis of ʟ-hydroxypipecolic acid 145 from chiral imine 144.

Scheme 40: Synthesis of 1-substituted isoquinolones 147, 149 and 151.

Scheme 41: Stereoselective synthesis of 3-substituted dihydrobenzo[de]isoquinolinones 154.

Scheme 42: Enantioselective synthesis of alkaloids (S)-1-benzyl-6,7-dimethoxy-1,2,3,4-tetrahydroisoquinoline (...

Scheme 43: Enantioselective synthesis of alkaloids (−)-cermizine B (171) and (+)-serratezomine E (172) develop...

Scheme 44: Stereoselective synthesis of (+)-isosolepnosin (177) and (+)-solepnosin (178) from homoallylamine d...

Scheme 45: Stereoselective synthesis of tetrahydroquinoline derivatives 184, 185 and 187 from chiral imines (RS...

Scheme 46: Stereoselective synthesis of pyridobenzofuran and pyridoindole derivatives 193 from homopropargylam...

Scheme 47: Stereoselective synthesis of 2-substituted 1,2,5,6-tetrahydropyridines 196 from chiral imines (RS)-...

Scheme 48: Stereoselective synthesis of 2-substituted trans-2,6-disubstituted piperidine 199 from chiral imine...

Scheme 49: Stereoselective synthesis of cis-2,6-disubstituted piperidines 200, and alkaloid (+)-241D, from chi...

Scheme 50: Stereoselective synthesis of 6-substituted piperidines-2,5-diones 206 and 1,7-diazaspiro[4.5]decane...

Scheme 51: Stereoselective synthesis of spirocyclic oxindoles 210 from chiral imines (RS)-53.

Scheme 52: Stereoselective synthesis of azaspiro compound 213 from chiral imine 211.

Scheme 53: Stereoselective synthesis of tetrahydroisoquinoline derivatives from chiral imines (RS)-214.

Scheme 54: Stereoselective synthesis of (−)-crispine A 223 from chiral imine (RS)-214.

Scheme 55: Synthesis of (−)-harmicine (228) using tert-butanesulfinamide through haloamide cyclization.

Scheme 56: Stereoselective synthesis of tetraponerines T1–T8.

Scheme 57: Stereoselective synthesis of phenanthroindolizidines 246a and (−)-tylophorine (246b), and phenanthr...

Scheme 58: Stereoselective synthesis of indoline, tetrahydroquinoline and tetrahydrobenzazepine derivatives 253...

Scheme 59: Stereoselective synthesis of (+)-epohelmin A (258) and (+)-epohelmin B (260) from aldimine (RS)-79.

Scheme 60: Stereoselective synthesis of (−)-epiquinamide (266) from chiral aldimine (SS)-261.

Scheme 61: Synthesis synthesis of (–)-hippodamine (273) and (+)-epi-hippodamine (272) using chiral sulfinyl am...

Scheme 62: Stereoselective synthesis of (+)-grandisine D (279) and (+)-amabiline (283).

Scheme 63: Stereoselective synthesis of (−)-epiquinamide (266) and (+)-swaisonine (291) from aldimine (SS)-126....

Scheme 64: Stereoselective synthesis of (+)-C(9a)-epi-epiquinamide (294).

Scheme 65: Stereoselective synthesis of (+)-lasubine II (298) from chiral aldimine (SS)-109.

Scheme 66: Stereoselective synthesis of (−)-epimyrtine (300a) and (−)-lasubine II (ent-302) from β-amino keton...

Scheme 67: Stereoselective synthesis of (−)-tabersonine (310), (−)-vincadifformine (311), and (−)-aspidospermi...

Scheme 68: Stereoselective synthesis of (+)-epohelmin A (258) and (+)-epohelmin B (260) from aldehyde 313 and ...

Scheme 69: Total synthesis of (+)-lysergic acid (323) from N-tert-butanesulfinamide (RS)-1.
Microwave-assisted multicomponent reactions in heterocyclic chemistry and mechanistic aspects
- Shivani Gulati,
- Stephy Elza John and
- Nagula Shankaraiah
Beilstein J. Org. Chem. 2021, 17, 819–865, doi:10.3762/bjoc.17.71

- dehydration forms D and finally 12. The synthesized naphthoacridines 12 with 2,3-diaminonaphthalene produces 14 via dehydration and dehydrogenation. 2 Azepines Azepines are represented by unsaturated seven atom heterocyles with nitrogen replacing a carbon atom. The benzene-fused azepines known as

Graphical Abstract

Figure 1: Marketed drugs with acridine moiety.

Scheme 1: Synthesis of 4-arylacridinediones.

Scheme 2: Proposed mechanism for acridinedione synthesis.

Scheme 3: Synthesis of tetrahydrodibenzoacridinones.

Scheme 4: Synthesis of naphthoacridines.

Scheme 5: Plausible mechanism for naphthoacridines.

Figure 2: Benzoazepines based potent molecules.

Scheme 6: Synthesis of azepinone.

Scheme 7: Proposed mechanism for azepinone formation.

Scheme 8: Synthesis of benzoazulenen-1-one derivatives.

Scheme 9: Proposed mechanism for benzoazulene-1-one synthesis.

Figure 3: Indole-containing pharmacologically active molecules.

Scheme 10: Synthesis of functionalized indoles.

Scheme 11: Plausible mechanism for the synthesis of functionalized indoles.

Scheme 12: Synthesis of spirooxindoles.

Scheme 13: Synthesis of substituted spirooxindoles.

Scheme 14: Plausible mechanism for the synthesis of substituted spirooxindoles.

Scheme 15: Synthesis of pyrrolidinyl spirooxindoles.

Scheme 16: Proposed mechanism for pyrrolidinyl spirooxindoles.

Figure 4: Pyran-containing biologically active molecules.

Scheme 17: Synthesis of functionalized benzopyrans.

Scheme 18: Plausible mechanism for synthesis of benzopyran.

Scheme 19: Synthesis of indoline-spiro-fused pyran derivatives.

Scheme 20: Proposed mechanism for indoline-spiro-fused pyran.

Scheme 21: Synthesis of substituted naphthopyrans.

Figure 5: Marketed drugs with pyrrole ring.

Scheme 22: Synthesis of tetra-substituted pyrroles.

Scheme 23: Mechanism for silica-supported PPA-SiO2-catalyzed pyrrole synthesis.

Scheme 24: Synthesis of pyrrolo[1,10]-phenanthrolines.

Scheme 25: Proposed mechanism for pyrrolo[1,10]-phenanthrolines.

Figure 6: Marketed drugs and molecules containing pyrimidine and pyrimidinones skeletons.

Scheme 26: MWA-MCR pyrimidinone synthesis.

Scheme 27: Two proposed mechanisms for pyrimidinone synthesis.

Scheme 28: MWA multicomponent synthesis of dihydropyrimidinones.

Scheme 29: Proposed mechanism for dihydropyrimidinones.

Figure 7: Biologically active fused pyrimidines.

Scheme 30: MWA- MCR for the synthesis of pyrrolo[2,3-d]pyrimidines.

Scheme 31: Proposed mechanism for pyrrolo[2,3-d]pyrimidines.

Scheme 32: Synthesis of substituted pyrrolo[2,3-d]pyrimidine-2,4-diones.

Scheme 33: Probable pathway for pyrrolo[2,3-d]pyrimidine-2,4-diones.

Scheme 34: Synthesis of pyridopyrimidines.

Scheme 35: Plausible mechanism for the synthesis of pyridopyrimidines.

Scheme 36: Synthesis of dihydropyridopyrimidine and dihydropyrazolopyridine.

Scheme 37: Proposed mechanism for the formation of dihydropyridopyrimidine.

Scheme 38: Synthesis of thiopyrano[4,3-d]pyrimidines.

Scheme 39: Plausible mechanism for the synthesis of thiopyrano[4,3-d]pyrimidines.

Scheme 40: Synthesis of decorated imidazopyrimidines.

Scheme 41: Proposed mechanism for imidazopyrimidine synthesis.

Figure 8: Pharmacologically active molecules containing purine bases.

Scheme 42: Synthesis of aza-adenines.

Scheme 43: Synthesis of 5-aza-7-deazapurines.

Scheme 44: Proposed mechanism for deazapurines synthesis.

Figure 9: Biologically active molecules containing pyridine moiety.

Scheme 45: Synthesis of steroidal pyridines.

Scheme 46: Proposed mechanism for steroidal pyridine.

Scheme 47: Synthesis of N-alkylated 2-pyridones.

Scheme 48: Two possible mechanisms for pyridone synthesis.

Scheme 49: Synthesis of pyridone derivatives.

Scheme 50: Postulated mechanism for synthesis of pyridone.

Figure 10: Biologically active fused pyridines.

Scheme 51: Benzimidazole-imidazo[1,2-a]pyridines synthesis.

Scheme 52: Mechanism for the synthesis of benzimidazole-imidazo[1,2-a]pyridines.

Scheme 53: Synthesis of pyrazolo[3,4-b]pyridine-5-spirocycloalkanedione derivatives.

Scheme 54: Proposed mechanism for spiro-pyridines.

Scheme 55: Functionalized macrocyclane-fused pyrazolo[3,4-b]pyridine derivatives.

Scheme 56: Mechanism postulated for macrocyclane-fused pyrazolo[3,4-b]pyridine.

Scheme 57: Generation of pyrazolo[3,4-b]pyridines.

Scheme 58: Proposed mechanism for the synthesis of pyrazolo[3,4-b]pyridines.

Scheme 59: Proposed mechanism for the synthesis of azepinoindole.

Figure 11: Pharmaceutically important molecules with quinoline moiety.

Scheme 60: Povarov-mediated quinoline synthesis.

Scheme 61: Proposed mechanism for Povarov reaction.

Scheme 62: Synthesis of pyrazoloquinoline.

Scheme 63: Plausible mechanism for pyrazoloquinoline synthesis.

Figure 12: Quinazolinones as pharmacologically significant scaffolds.

Scheme 64: Four-component reaction for dihydroquinazolinone.

Scheme 65: Proposed mechanism for dihydroquinazolinones.

Scheme 66: Synthesis purine quinazolinone and PI3K-δ inhibitor.

Scheme 67: Synthesis of fused benzothiazolo/benzoimidazoloquinazolinones.

Scheme 68: Proposed mechanism for fused benzothiazolo/benzoimidazoloquinazolinones.

Scheme 69: On-water reaction for synthesis of thiazoloquinazolinone.

Scheme 70: Proposed mechanism for the thiazoloquinazolinone synthesis.

Scheme 71: β-Cyclodextrin-mediated synthesis of indoloquinazolinediones.

Scheme 72: Proposed mechanism for synthesis of indoloquinazolinediones.

Figure 13: Triazoles-containing marketted drugs and pharmacologically active molecules.

Scheme 73: Cu(I) DAPTA-catalyzed 1,2,3-triazole formation.

Scheme 74: Mechanism for Cu(I) DAPTA-catalyzed triazole formation.

Scheme 75: Synthesis of β-hydroxy-1,2,3-triazole.

Scheme 76: Proposed mechanism for synthesis of β-hydroxy-1,2,3-triazoles.

Scheme 77: Synthesis of bis-1,2,4-triazoles.

Scheme 78: Proposed mechanism for bis-1,2,4-triazoles synthesis.

Figure 14: Thiazole containing drugs.

Scheme 79: Synthesis of a substituted thiazole ring.

Scheme 80: Synthesis of pyrazolothiazoles.

Figure 15: Chromene containing drugs.

Scheme 81: Magnetic nanocatalyst-mediated aminochromene synthesis.

Scheme 82: Proposed mechanism for the synthesis of chromenes.
[2 + 1] Cycloaddition reactions of fullerene C60 based on diazo compounds
- Yuliya N. Biglova
Beilstein J. Org. Chem. 2021, 17, 630–670, doi:10.3762/bjoc.17.55
- . Catalysis is the next area that is gaining an ever-increasing practical focus in the studies on the use of C60 and fullerene-containing materials [51]. A comparative analysis of the activity of fullerene-containing compounds and noble metals as dehydrogenation catalysts allows to consider the former as
- the substrate on two sites, namely a metal and a fullerene [53]. This allows to level out some negative features that are characteristic of heterogeneous catalysts. Thus, in dehydrogenation processes, catalytic amounts of a coordinated fullerene in metal fullerides act as a hydrogen acceptor in
Biochemistry of fluoroprolines: the prospect of making fluorine a bioelement
- Vladimir Kubyshkin,
- Rebecca Davis and
- Nediljko Budisa
Beilstein J. Org. Chem. 2021, 17, 439–460, doi:10.3762/bjoc.17.40

- entry into the citric acid cycle. The dehydrogenation of proline is involved in numerous biochemical processes. For example, the dehydrogenation of proline linked to an acyl carrier protein makes a first step in the biosynthesis of some neurotoxins from cyanobacteria (ana gene cluster) [68]. The

Graphical Abstract

Figure 1: The structures of the fluoroprolines discussed herein.

Figure 2: The distinction between “the alanine and the proline worlds”. While the polyalanine backbone leads ...

Figure 3: Molecular volume for 20 coded amino acids and fluoroprolines. The COSMO volume was calculated for a...

Figure 4: Comparative analysis of the electrostatic potential for proline and fluoroprolines (electrostatic p...

Figure 5: Experimental logP data for methyl esters of N-acetylamino acids.

Figure 6: The conformational dependence of the proline ring on the fluorination at position 4.

Figure 7: Rotation around the peptidyl-prolyl fragments in polypeptide structures is important for correct ov...

Figure 8: The complex fate of a protein-encoded amino acid in the cell (EF-Tu – elongation factor thermo unst...

Figure 9: Metabolic routes for proline in E. coli. A) Synthesis of proline and B) degradation of proline.

Figure 10: A complete flowchart for the proline incorporation into proteins during ribosomal biosynthesis. A) ...

Figure 11: Amide bond formation capacities of fluoroprolines compared to some coded amino acids measured on ri...

Figure 12: Ribbon representation of the X-ray crystal structures of proteins containing fluoroprolines. A) Enh...

Figure 13: Problems and phenomena associated with the production of a protein-containing proline-to-fluoroprol...

Figure 14: Effects of fluoroprolines on recombinant protein expression using the auxotrophic expression host E...

Figure 15: A) Experimental setup for the incorporation of fluoroprolines into proteins. B) Adaptive laboratory...
Insight into functionalized-macrocycles-guided supramolecular photocatalysis
- Minzan Zuo,
- Krishnasamy Velmurugan,
- Kaiya Wang,
- Xueqi Tian and
- Xiao-Yu Hu
Beilstein J. Org. Chem. 2021, 17, 139–155, doi:10.3762/bjoc.17.15

- between thiol-functionalized β-CD and oleic acid-protected CdS nanocrystals [29]. These spherical CdS–CD nanoparticles could be employed as a photocatalyst for the dehydrogenation of alcohols to aldehydes (at a low concentration of the reactant of 1 mM, ≥92% selectivity) or diols (at a high concentration
- of the reactant of 300 mM, ≥93% selectivity), with H2 liberation being achieved by visible-light irradiation in an aqueous solution. In comparison, CdS–CD was a highly efficient photocatalyst for benzyl alcohol dehydrogenation (77 µmol H2 in 180 h) compared to the CD-free CdS (5.4 µmol H2 in 30 h

Graphical Abstract

Figure 1: Chemical structures of representative macrocycles.

Figure 2: Ba2+-induced intermolecular [2 + 2]-photocycloaddition of crown ether-functionalized substrates 1 a...

Figure 3: Energy transfer system constructed of a BODIPY–zinc porphyrin–crown ether triad assembly bound to a...

Figure 4: The sensitizer 5 was prepared by a flavin–zinc(II)–cyclen complex for the photooxidation of benzyl ...

Figure 5: Enantiodifferentiating Z–E photoisomerization of cyclooctene sensitized by a chiral sensitizer as t...

Figure 6: Structures of the modified CDs as chiral sensitizing hosts. Adapted with permission from [24], Copyrigh...

Figure 7: Supramolecular 1:1 and 2:2 complexations of AC with the cationic β-CD derivatives 16–21 and subsequ...

Figure 8: Construction of the TiO2–AuNCs@β-CD photocatalyst. Republished with permission of The Royal Society...

Figure 9: Visible-light-driven conversion of benzyl alcohol to H2 and a vicinal diol or to H2 and benzaldehyd...

Figure 10: (a) Structures of CDs, (b) CoPyS, and (c) EY. Republished with permission of The Royal Society of C...

Figure 11: Conversion of CO2 to CO by ReP/HO-TPA–TiO2. Republished with permission of The Royal Society of Che...

Figure 12: Thiacalix[4]arene-protected TiO2 clusters for H2 evolution. Reprinted with permission from [37], Copyri...

Figure 13: 4-Methoxycalix[7]arene film-based TiO2 photocatalytic system. Reprinted from [38], Materials Today Chem...

Figure 14: (a) Photodimerization of 6-methylcoumarin (22). (b) Catalytic cycle for the photodimerization of 22...

Figure 15: Formation of a supramolecular PDI–CB[7] complex and structures of monomers and the chain transfer a...

Figure 16: Ternary self-assembled system for photocatalytic H2 evolution (a) and structure of 27 (b). Figure 16 reprodu...

Figure 17: Structures of COP-1, CMP-1, and their substrate S-1 and S-2.

Figure 18: Supramolecular self-assembly of the light-harvesting system formed by WP5, β-CAR, and Chl-b. Reprod...

Figure 19: Photocyclodimerization of AC based on WP5 and WP6.






