Search results
Search for "tin" in Full Text gives 138 result(s) in Beilstein Journal of Organic Chemistry.
Regioselective addition of Grignard reagents to N-acylpyrazinium salts: synthesis of substituted 1,2-dihydropyrazines and Δ5-2-oxopiperazines
Beilstein J. Org. Chem. 2019, 15, 72–78, doi:10.3762/bjoc.15.8
- , Table 1). Changing the solvent to diethyl ether showed no improvement in the yield of 3a but when THF was used, an excellent yield of 87% was produced (entries 4 and 5, Table 1). Based on our previously developed selective tin hydride reduction of monosubstituted pyrazinium salts [9], we expected the
Stereodivergent approach in the protected glycal synthesis of L-vancosamine, L-saccharosamine, L-daunosamine and L-ristosamine involving a ring-closing metathesis step
Beilstein J. Org. Chem. 2018, 14, 2949–2955, doi:10.3762/bjoc.14.274
- corresponding alkynyl alcohols requires the handling of toxic tin reagents [8][9]. During these last years, ring-closing metathesis (RCM) of vinyl ethers have proved to be an efficient method for the preparation of chiral glycal scaffolds [11][12][13][14][15][16][17][18] as demonstrated in some total syntheses
Learning from B12 enzymes: biomimetic and bioinspired catalysts for eco-friendly organic synthesis
Beilstein J. Org. Chem. 2018, 14, 2553–2567, doi:10.3762/bjoc.14.232

- obtained through isomerization with 1,2-migration of the ester groups. The B12-TiO2 hybrid catalyst can be regarded as a good alternative for conventional radical-involved organic syntheses using tin compounds. Recently, we discovered that the B12 derivative 1 can mediate trifluoromethylation and
Tetrathiafulvalene – a redox-switchable building block to control motion in mechanically interlocked molecules
Beilstein J. Org. Chem. 2018, 14, 2163–2185, doi:10.3762/bjoc.14.190

- -decorated TTF-rotaxanes was achieved by Gao and co-workers [95]. They deposited a thin-film of bistable donor–acceptor rotaxanes on an indium tin oxide (ITO)-coated substrate. A clean electrochemical switching on the substrate was observed with current–voltage curves showing a clear memory effect. The
Synthesis and supramolecular self-assembly of glutamic acid-based squaramides
Beilstein J. Org. Chem. 2018, 14, 2065–2073, doi:10.3762/bjoc.14.180

- = 10 kV). Xerogels, prepared by freeze-drying the corresponding gels, were placed on top of a tin plate and shielded with Pt (40 mA, 30–60 s; film thickness = 5–10 nm). Images were obtained by Servicio General de Apoyo a la Investigación-SAI (Universidad de Zaragoza). Chemical structures of isosteric
Synthesis of chiral 3-substituted 3-amino-2-oxindoles through enantioselective catalytic nucleophilic additions to isatin imines
Beilstein J. Org. Chem. 2018, 14, 1349–1369, doi:10.3762/bjoc.14.114
- diastereomers (>90% de) in both uniformly excellent yields (90–98%) and enantioselectivities (94–99% ee). In only one case of substrate (Ar = 1-Naph), a lower yield (78%) albeit combined with a comparable very good enantioselectivity (93% ee) was obtained. The same year, (S)-BINOL-derived tin dibromide 30 was
- lowest yield (31%). The authors have proposed that the true catalyst of the reaction was a chiral tin iodide methoxide 34 in situ generated from chiral tin bromide 30, two equivalents of NaI and NaOMe (Scheme 10). Enantioselective aza-Morita–Baylis–Hillman reactions The Morita–Baylis–Hillman reaction is
- -isatin imines with silyl ketene imines. Tin-catalyzed Mannich reaction of N-arylisatin imines with an alkenyl trichloroacetate. Aza-Morita–Baylis–Hillman reaction of N-Boc-isatin imines with acrolein catalyzed by β-isocupreidine. Aza-Morita–Baylis–Hillman reaction of N-Boc-isatin imines with acrolein (35
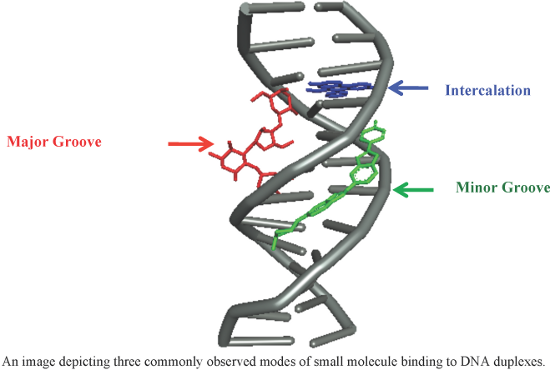
An overview of recent advances in duplex DNA recognition by small molecules
Beilstein J. Org. Chem. 2018, 14, 1051–1086, doi:10.3762/bjoc.14.93
Heterogeneous Pd catalysts as emulsifiers in Pickering emulsions for integrated multistep synthesis in flow chemistry
Beilstein J. Org. Chem. 2018, 14, 648–658, doi:10.3762/bjoc.14.52

- for catalysts rich in tin. Suzuki–Miyaura coupling reactions Batch reactions The catalytic activity of the synthesised catalysts in the Suzuki–Miyaura reaction was investigated using phenylboronic acid 3 in combination with various bromoarenes 4a–e, featuring ortho- and para-substitution of electron
- monitored by high-performance liquid chromatography (HPLC). In general, the catalysts with tin proportions of 0.20, 0.79 and 0.99 proved to be most effective in the desired transformations with exceptional high activities (turn over frequency, TOF > 12,000 h−1) in all tested Suzuki–Miyaura cross-coupling
- termination of the transformation just at the moment of full conversion. In conclusion, Ce0.99−xSnxPd0.01O2−δ (x = 0–0.99) proved to be very active catalysts for Suzuki–Miyaura reactions. Based on the experimental results, the tin and cerium content of the catalysts respectively do not seem to be directly
Mannich base-connected syntheses mediated by ortho-quinone methides
Beilstein J. Org. Chem. 2018, 14, 560–575, doi:10.3762/bjoc.14.43
- 5, the catalyst gave remarkable results at room temperature in short reactions (5–30 minutes) in 90–96% yields. Comparing these results with those achieved by the application of tin dioxide nanoparticles (nano SnO2, Table 1, entry 6), molten salt catalysis affording higher yields in shorter
Aminosugar-based immunomodulator lipid A: synthetic approaches
Beilstein J. Org. Chem. 2018, 14, 25–53, doi:10.3762/bjoc.14.3
Intramolecular glycosylation
Beilstein J. Org. Chem. 2017, 13, 2028–2048, doi:10.3762/bjoc.13.201
- application, glycosyl donor precursors were reacted with phthalic anhydride to afford the corresponding esters. The activation with thionyl chloride was used for tethering the donors to the glycosyl acceptor counterpart and the regioselectivity was controlled using tin-mediated coupling under microwave
- 27 using tin-mediated primary alkylation to afford the tethered pair 28. The latter is then intramolecularly glycosylated in the presence of NIS/TfOH in 93% yield and complete stereoselectivity. The resulting cyclic compound 29 is then subjected to concomitant xylylene tether removal and
- 72. The latter was then intramolecularly glycosylated in the presence of silver triflate, tin(II) chloride, and 2,6-di-tert-butyl-4-methylpyridine (DTBMP). Finally, the tether was cleaved off using TFA to give pure 1,2-cis glycoside 73 in 63% yield over two steps. An alternative linker was developed
The chemistry and biology of mycolactones
Beilstein J. Org. Chem. 2017, 13, 1596–1660, doi:10.3762/bjoc.13.159
Total syntheses of the archazolids: an emerging class of novel anticancer drugs
Beilstein J. Org. Chem. 2017, 13, 1085–1098, doi:10.3762/bjoc.13.108
- synthesis in 44% yield over 3 steps. For the final step the authors decided to follow a Stille coupling protocol established by Fürstner et al. [96] with CuTC as co-catalyst and [Ph2PO2][NBu4] as tin scavenger. Subsequently, the triene 68 could be synthesized in excellent 82% yield. For biological studies
Synthesis of 1-indanones with a broad range of biological activity
Beilstein J. Org. Chem. 2017, 13, 451–494, doi:10.3762/bjoc.13.48

- Friedel–Crafts alkylation has been used by Ahmed et al. for the synthesis of 2-hydroxyindan-1-one derivatives 222 in good yields (Scheme 61) [90]. The same research group used the THP (tetrahydropyranyl) and MOM (methoxymethyl) protected chalcone epoxides and tin(IV) chloride under mild conditions to
The reductive decyanation reaction: an overview and recent developments
Beilstein J. Org. Chem. 2017, 13, 267–284, doi:10.3762/bjoc.13.30
- the formation of 99% of cyclized products (relative yield). The authors proposed a radical chain mechanism similar to that proposed in the reaction with Bu3SnH. While the tin radical was proposed to add to nitrogen [115], here the evidence points more to the addition of the boryl radical 76 on the
Orthogonal protection of saccharide polyols through solvent-free one-pot sequences based on regioselective silylations
Beilstein J. Org. Chem. 2016, 12, 2748–2756, doi:10.3762/bjoc.12.271
- saccharide alcohols [34], the first catalytic tin-mediated procedure for regioselective benzylation/allylation of hydroxy groups incorporated into vicinal diols [35], and three alternative acetalation protocols [36]. Besides the avoided use of solvents, these approaches appear advantageous owing to their
- previously performed better than pyridine in the tin-catalyzed solvent-free regioselective benzylation or allylation of sugars [35]. The silylation rate was not appreciably influenced by tin catalysis (compare entries 2 and 3 in Table 1), although a previous report described the stoichiometric use of
- the adjacent electron-withdrawing benzoyl group. As already described above in Table 1 for mono-silylations, tin catalysis did not affect the double silylation processes as evidenced by the reaction of Table 3, entry 1 that in the presence of 0.1 equiv of Bu2SnO gave 17 in the same yield within the
Facile synthesis of a 3-deazaadenosine phosphoramidite for RNA solid-phase synthesis
Beilstein J. Org. Chem. 2016, 12, 2556–2562, doi:10.3762/bjoc.12.250
- catalysis at elevated pressure (30 psi) in ethanol or N,N-dimethylacetamide, ii) ammonium formiate, Pd/C, in methanol [28], iii) tin(II) chloride, in ethanol [29], iv) thioacetic acid, lutidine, in CH2Cl2 [30], v) triphenylphosphine, in CH2Cl2, aqueous work-up, and finally vi) Mg0 in methanol. Efficient 5
Useful access to enantiomerically pure protected inositols from carbohydrates: the aldohexos-5-uloses route
Beilstein J. Org. Chem. 2016, 12, 2343–2350, doi:10.3762/bjoc.12.227
- the absence of a coordinating metal center (for instance, in the case of tin and zirconium enolates, and of “naked” enolates generated from enolsilanes [42]). In the open-transition-state model, the enolate and the carbonyl group are orientated in an antiperiplanar fashion, maximazing the distance
High performance p-type molecular electron donors for OPV applications via alkylthiophene catenation chromophore extension
Beilstein J. Org. Chem. 2016, 12, 2298–2314, doi:10.3762/bjoc.12.223

- has extremely interesting properties worth further study and leads to three key questions; Synthesis: Can we simplify the synthesis of BTR removing some chromatographic purification steps and use of toxic tin containing Stille condensation reactions? Scale-up: Can we develop a multi-gram synthesis
- large scale use of tin reagents we required the key bis-borylated benzodithiophene (BDT) core 13, which was synthesised from the known BDT core 12 using iridium catalyzed borylation via CH-activation. The bis-borylated product was isolated by precipitation on addition of isopropanol (IPA), and an
Synthesis and characterization of fluorinated azadipyrromethene complexes as acceptors for organic photovoltaics
Beilstein J. Org. Chem. 2016, 12, 1925–1938, doi:10.3762/bjoc.12.182
- (Aldrich), 4-ethynyl-α,α,α-trifluorotoluene (Aldrich), n-butyllithium solution (Aldrich), tributyltin chloride (Fisher), tributyl(phenylethynyl)tin (Aldrich), tetrakis(triphenylphosphine)palladium(0) (Aldrich), n-iodosuccinimide (abbreviated NIS, Aldrich) were used as received. All other reagents and
- : calcd for C32H19F2I2N3, 736.96; found, 735.85. L1: Tributyl(phenylethynyl)tin (318 mg, 0.813 mmol) and L1-ADPI2 (200 mg, 0.271 mmol) was taken into a Schlenk flask (50 mL) which was evacuated and refilled with N2 three times. Dry chlorobenzene (15 mL) was added to the flask using a syringe and stirred
- : Tributyl(phenylethynyl)tin (0.928 g, 2.373 mmol) and L2-ADPI2 (0.500 g, 0.678 mmol) was taken into a Schlenk flask (100 mL), which was evacuated and refilled with N2 three times. Distilled xylenes (50 mL) were added to the flask using a syringe and placed under slight vacuum. Then Pd(PPh3)4 (0.110 g, 14
Ionic liquids as transesterification catalysts: applications for the synthesis of linear and cyclic organic carbonates
Beilstein J. Org. Chem. 2016, 12, 1911–1924, doi:10.3762/bjoc.12.181
- ionic liquid increases the catalytic activity of dibutyltin oxide fourfold probably by forming a highly active tin species where the anion of the ionic liquid acts as a ligand. The developed protocol was further studied for various substituted phenols, proving that electron-donating groups (EDG) at the
TMSBr-mediated solvent- and work-up-free synthesis of α-2-deoxyglycosides from glycals
Beilstein J. Org. Chem. 2016, 12, 1758–1764, doi:10.3762/bjoc.12.164
- -deoxythioglucosides [50]. In the literature, to synthesize 2-deoxythioglycosides, a highly toxic tin hydride reagent was used to produce S-2-deoxysugars from glycosyl bromide through an anomeric glycosyl radical and acetate rearrangement, followed by subsequent thioglycosylation to afford 2-deoxythioglycosides as
Catalytic Chan–Lam coupling using a ‘tube-in-tube’ reactor to deliver molecular oxygen as an oxidant
Beilstein J. Org. Chem. 2016, 12, 1598–1607, doi:10.3762/bjoc.12.156
- proposed a catalytic mechanism and later reported a tin free aryl–amine coupling reaction [6][7]. This major breakthrough made the C–N coupling reaction accessible to a wide range of substrates, including anilines, which did not react very well with the previous conditions. However, despite the
Beta-hydroxyphosphonate ribonucleoside analogues derived from 4-substituted-1,2,3-triazoles as IMP/GMP mimics: synthesis and biological evaluation
Beilstein J. Org. Chem. 2016, 12, 1476–1486, doi:10.3762/bjoc.12.144
- )-hexofuranose (2) was obtained in good yield from 1,2,5-tri-O-acetyl-3-O-benzoyl-6-deoxy-6-diethylphosphono-(α,β)-ribo-(5S)-hexofuranose [15] following a glycosylation procedure using sodium azide as nucleophilic entity and tin(IV) chloride as Lewis acid. Under these conditions, the reaction appeared highly
3,6-Carbazole vs 2,7-carbazole: A comparative study of hole-transporting polymeric materials for inorganic–organic hybrid perovskite solar cells
Beilstein J. Org. Chem. 2016, 12, 1401–1409, doi:10.3762/bjoc.12.134
- at 25 °C in dehydrated CH3CN containing 0.1 M (n-C4H9)4NPF6 in the three electrode cell. The working, reference, and auxiliary electrodes were a glassy carbon electrode, Ag/Ag+/CH3CN/(n-C4H9)4NPF6, and a Pt wire, respectively. Fabrication and measurements of perovskite solar cells Fluorine-doped tin





































































































































































































































































































































































































































































































