Search results
Search for "macrocyclic" in Full Text gives 283 result(s) in Beilstein Journal of Organic Chemistry. Showing first 200.
Anthelmintic drug discovery: target identification, screening methods and the role of open science
Beilstein J. Org. Chem. 2020, 16, 1203–1224, doi:10.3762/bjoc.16.105
- cattle treated annually is hundreds of millions [1]. Treatment of horses, other equids, and companion animals is also a major use of anthelmintics. Anthelmintic drug discovery has been a continued emphasis in the animal health industry, driven by the spread of resistance to the macrocyclic lactones [2
- macrocyclic lactone is an effective microfilaricide but does not kill the adult nematodes. Ivermectin must therefore be administered annually or twice-annually for many years to eliminate the parasite in the population. Progress has been impressive: onchocerciasis has been largely controlled as a public
- action of anthelmintic compounds, since it has been recognised that we do not fully understand how many anthelmintics work – for example the concentrations of macrocyclic lactones that paralyse worms in vitro are much greater than the concentration achieved by effective doses in vivo [147]. Several
The interaction between cucurbit[8]uril and baicalein and the effect on baicalein properties
Beilstein J. Org. Chem. 2020, 16, 71–77, doi:10.3762/bjoc.16.9
- Xiaodong Zhang Jun Xie Zhiling Xu Zhu Tao Qianjun Zhang Key Laboratory of Macrocyclic and Supramolecular Chemistry of Guizhou Province, Guizhou University, Guiyang 550025, China 10.3762/bjoc.16.9 Abstract The host–guest interactions between baicalein (BALE) and cucurbit[8]uril (Q[8]) and the
- , baicalein contains three phenolic hydroxy groups, which are easily oxidized to the quinone derivative and appear green, therefore, it has limited use in pharmaceuticals on account of its poor aqueous solubility and stability [31]. The cucurbit[n]urils (Q[n]s n = 5–8, 10, …) are macrocyclic hosts with a
- hydrophobic rigid cavity [32] (Figure 1). cucurbit[n]urils have a unique combination of properties including rigid highly symmetric structures, relatively large hydrophobic cavities and high thermal and chemical stability [33][34]. Cucurbit[n]urils are a type of macrocyclic drug carrier similar to macrocyclic
Understanding the role of active site residues in CotB2 catalysis using a cluster model
Beilstein J. Org. Chem. 2020, 16, 50–59, doi:10.3762/bjoc.16.7

- different cellular compartments [1][2]. More specifically, the enigmatic class of terpene cyclases is responsible for converting linear aliphatic oligoprenyl diphosphates into various chemically complex macrocyclic products. The resulting terpene scaffolds and their functionalized terpenoid analogues
Starazo triple switches – synthesis of unsymmetrical 1,3,5-tris(arylazo)benzenes
Beilstein J. Org. Chem. 2020, 16, 22–31, doi:10.3762/bjoc.16.4

- their isomerization properties [3][4]. Also, the incorporation of AB units into cyclic [5] or macrocyclic structures can control the switching, depending, i.a., on symmetry and ring strain [6][7][8][9]. By combining these approaches, half-lives can be tuned from milliseconds to years. The incorporation
Synthesis of novel sulfide-based cyclic peptidomimetic analogues to solonamides
Beilstein J. Org. Chem. 2019, 15, 2544–2551, doi:10.3762/bjoc.15.247
- was based on the conservation of the 16-membered macrocyclic scaffold and the apolar tripeptidyl moiety found in the solonamides. Both features are important to guarantee the interference with S. aureus QS [12][13][14][15]. The ester linkage of the lactone core was substituted by the sulfide group
- peptides [40]. Noteworthy, the detected ions A–C always showed the –SCH2CH=NH moiety, confirming the formation of the sulfide group (Schemes S4–S11, Supporting Information File 1). The NMR experiments also confirmed the macrocyclic structure (Supporting Information File 1). The 1H NMR spectra of compounds
Anion-driven encapsulation of cationic guests inside pyridine[4]arene dimers
Beilstein J. Org. Chem. 2019, 15, 2486–2492, doi:10.3762/bjoc.15.241

- , cation binding to pyridinearene is clearly not as strong as with resorcin[4]arene, which is known for its excellent cation receptor properties. A comparison of these two macrocyclic hosts reveals significant differences in their binding properties. Pyridine[4]arene appears to have a better affinity
Self-assembled coordination thioether silver(I) macrocyclic complexes for homogeneous catalysis
Beilstein J. Org. Chem. 2019, 15, 2465–2472, doi:10.3762/bjoc.15.239
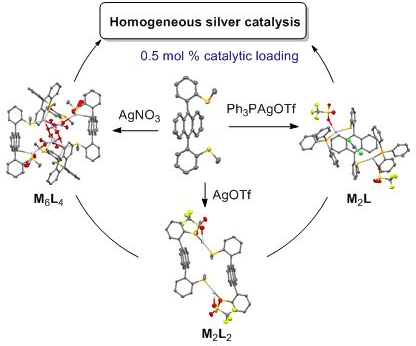
- -atropisomer, as revealed by X-ray diffraction. This alkylaryl thioether ligand (L) formed different macrocyclic complexes by coordination with silver(I) salts depending on the nature of the anion: M2L2 for AgOTf and AgOTFA, M6L4 for AgNO3. A discrete M2L complex was obtained in the presence of bulky PPh3AgOTf
- silver(I) catalysts based on sulfur ligands were reported so far, although alkyl thioethers are soft σ-donor ligands such as crown thioethers that were largely developed as macrocyclic ligands for silver(I) [35][36][37][38][39][40][41][42][43]. Interestingly, depending on their design, these known silver
- /cycloisomerization was previously described in high yields (>95%) using 5 mol % catalyst loadings starting from 2-(alkynyl)quinoline-3-carbaldehyde [60][61] with AgOTf catalyst and starting from 2-alkynylbenzaldehyde derivatives [62] in the presence of a macrocyclic pyridine-tetraaza complex of Ag(I) as a catalyst
Photochromic diarylethene ligands featuring 2-(imidazol-2-yl)pyridine coordination site and their iron(II) complexes
Beilstein J. Org. Chem. 2019, 15, 2428–2437, doi:10.3762/bjoc.15.235
- for realization of light-triggered guest uptake/release [52] and light-controlled interconversion between distinct supramolecular assemblies [53]. Ligand 6 featuring two coordination sites in the heteroaryl moiety and bridge provides unique opportunities to construct novel macrocyclic systems. Our
Effect of ring size on photoisomerization properties of stiff stilbene macrocycles
Beilstein J. Org. Chem. 2019, 15, 2408–2418, doi:10.3762/bjoc.15.233

- of the macrocyclic stiff stilbene diethers, a conformational analysis was undertaken (Figure 6). According to X-ray crystallography, in compound (E)-7 (Scheme 4) the aromatic rings of the two indane units are in the same plane (dihedral angle 180°), whereas in (Z)-7 this angle is 9.1° [21]. In the
- macrocyclic diethers 1a–d, all Z-isomers have a dihedral angle of 12–14°, roughly similar to the one in the crystal structure of (Z)-7. The deviation of this angle from 0° is due to steric interaction between two aromatic protons in position 4 (Figure 9). In the E-isomers, an increasing distortion of the
- stated differently, the obtained yellow oil was purified by CC (pentane/DCM 1:0 to 1:1). The obtained product was dried under high vacuum overnight. Synthesis of macrocyclic stiff stilbene diether (Z)-1a The synthesis followed general procedure B with compound 6a (0.279 g, 0.7 mmol) as starting material
Reversible switching of arylazopyrazole within a metal–organic cage
Beilstein J. Org. Chem. 2019, 15, 2398–2407, doi:10.3762/bjoc.15.232

- photochromism in the spiropyran switch [18]. Other recent studies of photochromic systems within macrocyclic and supramolecular hosts [19] include dihydroazulene switches [20] and red-shifted azobenzenes [21][22] inside cucurbiturils and cyclodextrins. The behavior of light-responsive compounds can also be
1,2,3-Triazolium macrocycles in supramolecular chemistry
Beilstein J. Org. Chem. 2019, 15, 2142–2155, doi:10.3762/bjoc.15.211
- polymers etc. [14][15]. Noncovalent interactions play a dynamic role in the binding mechanism of triazoles as macrocyclic receptors. It has been reported that the combined effects of both an electron lone pair on the nitrogen of the heterocycle and the acidic C5–H proton make 1,2,3-triazoles interesting
- of 1,4-disubstituted 1,2,3- triazole units [19][20][21][22][23][24]. Macrocyclic ring closure can be achieved by the CuAAC of building blocks functionalized with both azide and alkyne, using [1 + 1], [2 + 2], [n + n] strategies depending on how much triazoles are needed to be included in the
- to be very difficult to further proceed. However, the anion binding property of the corresponding bistriazolium macrocyclic part 5·(BF4)2 (Figure 5) has successfully been investigated using 1H NMR titration experiments in CD3CN. The highest binding affinity was found to be almost similar for both BzO
Multiple threading of a triple-calix[6]arene host
Beilstein J. Org. Chem. 2019, 15, 2092–2104, doi:10.3762/bjoc.15.207

- , daisy-chain pseudorotaxanes, olympiadane, Janus rotaxanes [5]) is generally obtained through a template-approach [9] exploiting the threading process between linear (axle) and macrocyclic (wheel) components. In order to synthesize high-order interpenetrated architectures, much attention has been
Attempted synthesis of a meta-metalated calix[4]arene
Beilstein J. Org. Chem. 2019, 15, 1996–2002, doi:10.3762/bjoc.15.195
- yielding transformations to azide and 1,2,3-triazole derivatives which may have application in other areas of research. Keywords: calixarene; inherent chirality; mesoionic carbene; mononitration; ruthenacycle; Introduction Calix[4]arenes are a class of diverse macrocyclic compounds which have been the
Synthesis and anion binding properties of phthalimide-containing corona[6]arenes
Beilstein J. Org. Chem. 2019, 15, 1976–1983, doi:10.3762/bjoc.15.193
- conveniently by means of a macrocyclic condensation reaction between N-functionalized 3,6-dihydroxyphthalimides and 3,6-dichlorotetrazine under mild conditions in a one-pot reaction manner. The novel macrocycles exist as a mixture of rapidly interconvertible conformers in solution while in the solid state they
- adopt the conformation in which three phthalimide units are cis,trans-orientated. Acting as electron-deficient macrocyclic hosts, the synthesized O6-corona[3]arene[3]tetrazines self-regulated conformational structures to complex anions in the gas phase and in the solid state owing to the anion–π
- operational one-pot reaction fashion on the basis of a nucleophilic aromatic substitution reaction [23][24][25][26][27][28][29][30][31]. To prepare functionalized corona[6]arenes using diethyl terephthalate as a starting material, we observed, however, the formation of a mixture of macrocyclic isomers because
Complexation of chiral amines by resorcin[4]arene sulfonic acids in polar media – circular dichroism and diffusion studies of chirality transfer and solvent dependence
Beilstein J. Org. Chem. 2019, 15, 1913–1924, doi:10.3762/bjoc.15.187
- a hydrophobic effect, while the other one has a more directional character, or application of multidentate interaction sites [3]. Macrocyclic compounds with persistent hydrophobic cavities constitute a fundamental class of scaffolds for the construction of supramolecular host–guest complexes in
- cyclodextrines, which, when unmodified, interact with a wide range of hydrophobic guests albeit with low general selectivity and enantioselectivity in particular [6]. In this paper we present a group of synthetic macrocyclic compounds – resorcin[4]arene sulfonic acids (RSAs) and analyze their interactions with
- capable of forming electrostatic interactions. However, in CSAs polar groups are positioned directly on a macrocyclic scaffold, while for RSAs at conformationally labile methylene linkers. Therefore, we think that RSAs are more adaptable than CSAs and are able to enclose guest molecules more tightly (by
Nanopatterns of arylene–alkynylene squares on graphite: self-sorting and intercalation
Beilstein J. Org. Chem. 2019, 15, 1848–1855, doi:10.3762/bjoc.15.180
- the procedure described above) a nanopattern of 1a (5 × 10−7 M), and added a 10−4 M solution of 3 in TCB. STM (Figure 4a) shows – again – bright features in the macrocyclic template of 1a. Again, a slight central depression is visible in the STM image which can attributed to the empty center region of
- images comprising both polymorphs are shown in Supporting Information File 1, Figure S9a and b. Therefore, the PAH coadsorption is a monitor for the alkoxy chain orientation inside the macrocyclic interior (whereas they do not point towards the solution phase here). By adding a solution of 3 at a
Novel macrocycles – and old ones doing new tricks
Beilstein J. Org. Chem. 2019, 15, 1838–1839, doi:10.3762/bjoc.15.178

- special issue in the Beilstein Journal of Organic Chemistry gathers world-renowned experts in the field to share their very recent results and will surely help stimulate macrocyclic chemistry. Wei Jiang and Christoph A. Schalley Shenzhen, Berlin, July 2019
Synthesis of a [6]rotaxane with singly threaded γ-cyclodextrins as a single stereoisomer
Beilstein J. Org. Chem. 2019, 15, 1829–1837, doi:10.3762/bjoc.15.177

- Jason Yin Hei Man Ho Yu Au-Yeung Department of Chemistry, The University of Hong Kong, Pokfulam Road, Hong Kong, P. R. China 10.3762/bjoc.15.177 Abstract A series of hetero [4]-, [5]- and [6]rotaxanes containing both cucurbit[6]uril (CB[6]) and γ-cyclodextrin (γ-CD) as the macrocyclic components
- interested in exploiting the binding capability of γ-CD in the construction of high order [n]rotaxanes and [n]catenanes. Here, we report our work on the synthesis of multiring, hetero[n]rotaxane containing γ-CD and cucurbit[6]uril (CB[6]) as the macrocyclic components in an aqueous medium. Because of the
Complexation of 2,6-helic[6]arene and its derivatives with 1,1′-dimethyl-4,4′-bipyridinium salts and protonated 4,4'-bipyridinium salts: an acid–base controllable complexation
Beilstein J. Org. Chem. 2019, 15, 1795–1804, doi:10.3762/bjoc.15.173

- ; macrocycles; macrocyclic arene; Introduction Macrocyclic host molecules [1][2] play a significant role in host–guest chemistry. Compared with noncyclic molecules, the structures of macrocyclic hosts can greatly enhance the host–guest complexation ability through preorganization. Moreover, cyclic structures
- are also the epitome of complex-binding pockets of enzymes [3]. Macrocyclic arenes including calixarenes [4][5], resorcinarenes [6], cyclotriveratrylenes [7][8], pillararenes [9], biphen[n]arenes [10] and others [11][12] are all composed of hydroxy-substituted aromatic rings bridged by methylene or
- methenyl groups. They have been a kind of important macrocyclic host molecules during the last decades due to their unique structures and a wide range of applications in host–guest chemistry [13][14][15][16][17][18], self-assembly [19], biomedicine [20] and materials science [21][22]. The derivatives of
Host–guest interactions in nor-seco-cucurbit[10]uril: novel guest-dependent molecular recognition and stereoisomerism
Beilstein J. Org. Chem. 2019, 15, 1705–1711, doi:10.3762/bjoc.15.166
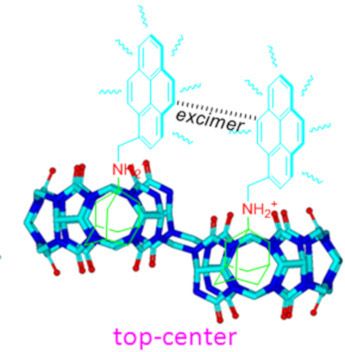
- Xiaodong Zhang Wei Wu Zhu Tao Xin-Long Ni Key Laboratory of Macrocyclic and Supramolecular Chemistry of Guizhou Province, Guizhou University, Guiyang 550025, China 10.3762/bjoc.15.166 Abstract The unique monomer and excimer fluorescence emissions of pyrene were first exploited as distinctly
- because of their varying cavity rigidity and larger portal sizes as compared with those of other macrocyclic hosts [4][5][6][7][8][9][10][11][12][13]. For example, cucurbit[8]uril (Q[8] or CB[8]), a large homologue of the Q[n] family, is unique because of its ability to bind two hetero- and homo-aromatic
Synthesis, enantioseparation and photophysical properties of planar-chiral pillar[5]arene derivatives bearing fluorophore fragments
Beilstein J. Org. Chem. 2019, 15, 1601–1611, doi:10.3762/bjoc.15.164

- guest receptors [4][5]. Planar-chiral macrocyclic molecules are particularly interesting in the context of the host–guest complexation properties [6][7][8]. Pillararenes are typical examples of this type of compounds and have attracted considerable attention due to their facile chemical synthesis and
- quantum yield was significantly decreased to 46.4% compared with 78.2% for Py-6. We ascribed the decreased fluorescence of P5A-Py to the π–π stacking of the Py units caused by the high local concentration of perylene. For P5A-DPA, which also bears two DPA units in one macrocyclic host, the fluorescent
- assigned to the excimer emission of Py (Figure S23, Supporting Information File 1) [54]. Conclusion Two new planar chiral macrocyclic hosts P5A-DPA and P5A-Py were synthesized by grafting two fluorophore pigments (DPA or Py) on pillar[5]arene through CuAAC “click” reaction. The new macrocyclic compounds
An azobenzene container showing a definite folding – synthesis and structural investigation
Beilstein J. Org. Chem. 2019, 15, 1534–1544, doi:10.3762/bjoc.15.156
- chemistry. Here, we present the synthesis of a foldable container consisting of two different types of Lissoclinum macrocyclic peptides which are connected via two azobenzene units. The container is controllable by light: irradiation with UV light causes a switching process to the compact cis,cis-isomer
- are determined by the type and size of the macrocyclic platform, e.g., if all of the amino acid side chains are of the same configuration, they are presented on one face of the macrocycle in a convergent manner. The artificial Lissoclinum cyclopeptide platforms feature C2, C3 and C4 symmetry [3]. So
- -isomer (Figure 8). Conclusion In conclusion, we were able to synthesize a foldable container consisting of two different types of Lissoclinum macrocyclic peptides which are connected via two azobenzene units. The synthesis of this container was achieved by a one pot reaction of the two imidazole
2,3-Dibutoxynaphthalene-based tetralactam macrocycles for recognizing precious metal chloride complexes
Beilstein J. Org. Chem. 2019, 15, 1460–1467, doi:10.3762/bjoc.15.146

- –guest chemistry; macrocycles; molecular recognition; precious metal chloride complexes; Introduction Macrocyclic receptors are the major workhorses in supramolecular chemistry. Design and synthesis of new macrocyclic receptors with new properties is always attractive but is also challenging [1
- interesting new binding properties. During the last six years, our group have developed a series of naphthol-based macrocyclic receptors [24][25][26][27][28][29]. Of them, oxatub[4]arene [30][31] and zorb[4]arene [32][33] show multiple conformations due to the flipping of naphthalenes and thus resulted in a
Complexation of a guanidinium-modified calixarene with diverse dyes and investigation of the corresponding photophysical response
Beilstein J. Org. Chem. 2019, 15, 1394–1406, doi:10.3762/bjoc.15.139

- binding of the analyte gives rise to an extraordinary luminescence response of the reporter dye. Macrocyclic hosts constitute a family of well-studied artificial receptors with a discrete cavity that is selective for complementary binding to certain guests [9]. Modulation of properties of organic
- fluorophores through supramolecular encapsulation using artificial macrocyclic hosts has always been an active research area [10][11][12]. The complexes of macrocycles with luminescent dyes not only act as reporter pairs for sensing [13][14][15], but also offer various applications in bioimaging [16][17
- supramolecular cavities [25]. Calixarenes are the third generation of macrocyclic compounds composed of phenolic units bridged with methylene groups at the o-positions of phenolic hydroxy groups [9]. We have focused on molecular recognition and self-assembly of water-soluble calixarene derivatives for a long
Host–guest interactions between p-sulfonatocalix[4]arene and p-sulfonatothiacalix[4]arene and group IA, IIA and f-block metal cations: a DFT/SMD study
Beilstein J. Org. Chem. 2019, 15, 1321–1330, doi:10.3762/bjoc.15.131

- molecules [27], including some amino acids [28] and proteins [29]. They are also biocompatible: compared to other types of macrocyclic molecules such as cyclodextrins and cucurbiturils (which are also water soluble), p-sulfonatocalix[n]arenes do not exhibit any toxicity, which makes them applicable in





















































































































































































































