Search results
Search for "chromophore" in Full Text gives 154 result(s) in Beilstein Journal of Organic Chemistry.
Diaminoterephthalate–α-lipoic acid conjugates with fluorinated residues
Beilstein J. Org. Chem. 2019, 15, 981–991, doi:10.3762/bjoc.15.96
- indicated that trifluoromethylation is an excellent tool for the detection of surface-bound materials by XPS. Keywords: chromophore; diaminoterephthalate; fluorine surface marker; fluorescence dye; lipoic acid; self-assembled monolayers; Introduction Diaminoterephthalates (DATs) are powerful fluorescence
- dyes [1][2] with outstanding properties such as high quantum yields and pronounced stability against photobleaching [3][4][5]. Although being structurally relatively simple, this class of dyes is so far underrated in the literature. The chromophore, which is accessed from succinyl succinates and
- the first title compound 3 in 71% yield. Due to the electron-withdrawing amide group, the chromophore is relatively electron deficient (absorption at 420 nm and emission at 495 nm). In order to achieve a bathochromic shift of absorption and emission bands, the DAT and ALA moieties should be
Photochemical generation of the 2,2,6,6-tetramethylpiperidine-1-oxyl (TEMPO) radical from caged nitroxides by near-infrared two-photon irradiation and its cytocidal effect on lung cancer cells
Beilstein J. Org. Chem. 2019, 15, 863–873, doi:10.3762/bjoc.15.84

- new caged nitroxides (nitroxide donors) 2a and 2b having the TP-responsive NPBF chromophore and the NIR TP-triggered generation of the 2,2,6,6-tetramethylpiperidine-1-oxyl (TEMPO) radical under atmospheric conditions using these species (Scheme 1). Because free radicals are cytotoxic due to their
- 2a induced cancer cell death in vitro, although no in vivo study was performed because of the low water solubility of 2a. At this point, we cannot rule out generate of ROS by photosensitization of the chromophore in the presence of O2 for the cytotoxicity. Conclusion In the present study, novel caged
- nitroxides 2a and 2b having a TP-responsive chromophore were synthesized, and OP- and TP-induced generation of the TEMPO radical with these species was examined. The quantum yields for generation of the TEMPO radical from 2a and 2b were determined to be 2.5% and 0.8% in benzene, respectively. The quantum
Coordination chemistry and photoswitching of dinuclear macrocyclic cadmium-, nickel-, and zinc complexes containing azobenzene carboxylato co-ligands
Beilstein J. Org. Chem. 2019, 15, 840–851, doi:10.3762/bjoc.15.81
- * transitions of the trans-configured azobenzene chromophore, consistent with literature reports [24][25]. The absorptions below 310 nm (λ = 260 and 300 nm) are attributable to the π–π* transitions within the thiophenolate units of the [Cd2L]2+ fragment, which may be further differentiated as p- and α-bands of
- the parent benzene chromophore utilizing Clar’s notation [26], assuming that the electron-donating alkyl and thiol substituents exert a bathochromic effect. The spectral properties of the nickel complexes differ from those of the zinc and cadmium counterparts in that they exhibit two additional
Application of olefin metathesis in the synthesis of functionalized polyhedral oligomeric silsesquioxanes (POSS) and POSS-containing polymeric materials
Beilstein J. Org. Chem. 2019, 15, 310–332, doi:10.3762/bjoc.15.28
- by Cole-Hamilton [11]. Procedures allowing the syntheses of POSS derivatives with synthetically useful functional groups in multigram quantities have been proposed (Scheme 5). A similar procedure permits the synthesis of a series of vinylbiphenyl chromophore-decorated cubic oligosilsesquioxanes [12
- isolated yields exceeding 60%. Chromophore-functionalized silsesquioxane-core dendrimers were obtained to investigate their photophysical properties [12][14]. In the synthesized compounds chromophore properties were only slightly influenced by the core. The possibility of fine-tuning of the photophysical
- properties of the POSS-based dendritic molecule not only by changing the chromophore but also by providing tailored steric interactions between bridges and/or chromophores was proved [14]. Interestingly, the 4’-vinylbiphenyl-3,5-dicarbaldehyde group modified macromolecule (Figure 5d) displayed the ability to
Olefin metathesis in multiblock copolymer synthesis
Beilstein J. Org. Chem. 2019, 15, 218–235, doi:10.3762/bjoc.15.21
- multiple click reactions of the diazido-telechelic PBD with a dialkynyl-containing azobenzene chromophore. The newly formed triazole moieties can tune and improve the photoresponsive properties of PBD. α,ω-Functional telechelic polymers also can be synthesized by acyclic diene metathesis (ADMET
Rational design of boron-dipyrromethene (BODIPY) reporter dyes for cucurbit[7]uril
Beilstein J. Org. Chem. 2018, 14, 1961–1971, doi:10.3762/bjoc.14.171

- -dipyrromethene (BODIPY) dyes as a new class of fluorophores for the design of reporter dyes for supramolecular host–guest complex formation with cucurbit[7]uril (CB7). The BODIPYs contain a protonatable aniline nitrogen in the meso-position of the BODIPY chromophore, which was functionalized with known binding
- state of the amine lone pair with the BODIPY chromophore. In accordance with this hypothesis, a positive solvatochromism with varying contents of ACN was observed (Figure S18, Supporting Information File 1). The fluorescence quantum yields of the unprotonated BODIPY dyes were determined in 30% (v/v) ACN
- previously noted for water/DMSO mixtures [56]. The attachment of the BODIPY chromophore to the anchor groups thus reduces the binding constant by an additional factor of 10 for the aniline meso-group and by a factor of 1000 for the tetrafluoroaniline group in 4. We ascribe this to steric hindrance between
Synthesis of 9-arylalkynyl- and 9-aryl-substituted benzo[b]quinolizinium derivatives by Palladium-mediated cross-coupling reactions
Beilstein J. Org. Chem. 2018, 14, 1871–1884, doi:10.3762/bjoc.14.161
- < OMe < NMe2) corresponds well with the strength of the donor–acceptor interplay between the electron-donating aryl substituent and the benzo[b]quinolizinium chromophore. The absorption properties depend only slightly on the solvent properties indicating that the corresponding ground state and vertical
Strong binding and fluorescence sensing of bisphosphonates by guanidinium-modified calix[5]arene
Beilstein J. Org. Chem. 2018, 14, 1840–1845, doi:10.3762/bjoc.14.157

- molecules [9]. However, this method will greatly reduce the life of the column [10]. Moreover, the absence of a chromophore in most BPs lead to the employment of derivatization by an UV–vis light-absorbing or fluorescence label for detection [11][12]. However, directly labeling BPs in biological media is
Hyper-reticulated calixarene polymers: a new example of entirely synthetic nanosponge materials
Beilstein J. Org. Chem. 2018, 14, 1498–1507, doi:10.3762/bjoc.14.127
- varied though maintaining a fixed chromophore moiety. The results obtained, expressed in terms of percent of guest absorbed, are summarized in Table 1. As we can notice, the parent p-nitroaniline (1) is absorbed fairly well by CaNSs under neutral conditions, up to an extent comparable with the one
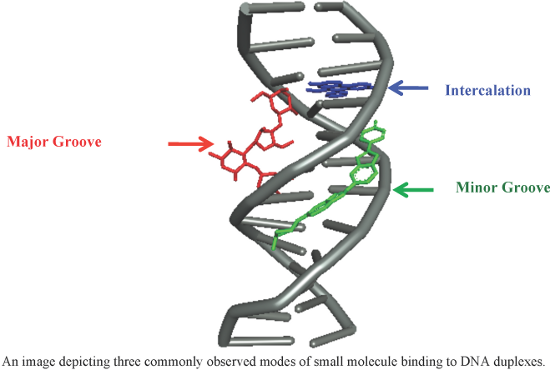
An overview of recent advances in duplex DNA recognition by small molecules
Beilstein J. Org. Chem. 2018, 14, 1051–1086, doi:10.3762/bjoc.14.93
Fluorogenic PNA probes
Beilstein J. Org. Chem. 2018, 14, 253–281, doi:10.3762/bjoc.14.17

- presence of the complementary DNA target as a result of the restricted rotation of the TO chromophore upon its intercalation into the base stack (Figure 19). Considering the bulkiness of the TO dye, it is quite likely that the opposite nucleobase was forced away from the duplex and so did not directly
Synthesis and spectroscopic properties of β-meso directly linked porphyrin–corrole hybrid compounds
Beilstein J. Org. Chem. 2018, 14, 187–193, doi:10.3762/bjoc.14.13
- obtaining multiporphyrin arrays [7][8][9] that could stabilize only metal ions in a bivalent state. To overcome this limitation, porphyrin conjugates with different chromophore groups such as fullerene [10][11][12], BODIPY [13][14][15], corrole [16][17][18][19][20][21][22][23], phthalocyanine [24][25][26
Fluorescent nucleobase analogues for base–base FRET in nucleic acids: synthesis, photophysics and applications
Beilstein J. Org. Chem. 2018, 14, 114–129, doi:10.3762/bjoc.14.7
- match tC and/or tCO. To this end we developed the nitro-version of tC, 7-nitro-1,3-diaza-2-oxophenothiazine (tCnitro) (Scheme 3) [14][38][45]. From UV–vis spectroscopy we showed that it, as do tC and tCO, forms stable, B-form duplexes and stacks firmly inside the DNA. It is a non-emissive chromophore
Polarization spectroscopy methods in the determination of interactions of small molecules with nucleic acids – tutorial
Beilstein J. Org. Chem. 2018, 14, 84–105, doi:10.3762/bjoc.14.5

- chromophore also absorbs in this range, the deconvolution of all contributions is not trivial (see interpretation of results in the chapters of the corresponding methods); b) monitoring changes in polarization spectra at λ > 300 nm for ligands that have corresponding chromophores. This is the most common used
- (ECD) will be discussed first as the most common technique with the least pitfalls. Thus it is most often appropriate for the characterization of the systems of interest. Then, LD follows as a complementary method used to reveal the mutual orientation of the ligand chromophore with respect to the DNA
- that at least the chromophoric portions that are associated with the observed transitions of the compound are not intercalated (inserted between the base pairs, thus being perpendicular to the DNA chiral axis). Instead, the direction of the chromophore transition moment (e.g., the chromophore long axis
Halogen-containing thiazole orange analogues – new fluorogenic DNA stains
Beilstein J. Org. Chem. 2017, 13, 2902–2914, doi:10.3762/bjoc.13.283
- atoms (connected directly to the chromophore or as side groups) on the molar absorptivity and the fluorescence intensity of a series of new TO analogues. Photophysical properties of a series of TO analogues that have fluoro- or trifluoromethyl groups connected to the heterocyclic end groups of the
- (Experimental part) with ethanol as the reaction medium. It is worth noting that replacement of the methanol with the environmentally more benign and less harmful ethanol significantly decreases the formation of blue-colored self-condensation byproducts (probably a chromophore constructed from quinolone and
- quinoline moiety of the chromophore leads to a bathochromic shift of the longest wavelength absorption maximum in all new compounds when compared to TO. The presence of a benzyl substituent in compounds 5a–d doesn’t have significant effect on the maxima position, slightly shifting the transition further
The photodecarboxylative addition of carboxylates to phthalimides as a key-step in the synthesis of biologically active 3-arylmethylene-2,3-dihydro-1H-isoindolin-1-ones
Beilstein J. Org. Chem. 2017, 13, 2833–2841, doi:10.3762/bjoc.13.275
- [41], followed by amination [42] furnishes the desired target compounds. The amino group is introduced in the final step as it would otherwise interfere with the desired photoreaction. In fact, amines are very potent electron donors and are easily oxidized by the excited phthalimide chromophore [43
Binding abilities of polyaminocyclodextrins: polarimetric investigations and biological assays
Beilstein J. Org. Chem. 2017, 13, 2751–2763, doi:10.3762/bjoc.13.271
- values for p-nitroanilines are affected by an induced circular dichroism effect due to the interaction between the dipole moments of the polarized chromophore guest moiety and of the cyclodextrin cavity. Therefore, these parameters provide an estimation of the time-averaged tilt angle between the
Pyrene–nucleobase conjugates: synthesis, oligonucleotide binding and confocal bioimaging studies
Beilstein J. Org. Chem. 2017, 13, 2521–2534, doi:10.3762/bjoc.13.249

- , or oligothymidine, T10, respectively, as template strands were investigated in water. Due to the nearly complete insolubility of the chromophore–nucleobase conjugates in water it is possible to follow this self-assembly simply by UV–vis absorption spectroscopy (Figure 5). Only those chromophore
- –nucleoside conjugates that are bound and assembled along the (dA)10 or T10 template are kept soluble in aqueous media. A higher concentrated stock solution of each chromophore–nucleobase conjugate was prepared in DMSO and added as aliquots to an aqueous solution (2.5 μM) of the (dA)10 or T10 template. The
- chromophore conjugate and allows more efficient self-assembly, whereas the carbonyl group of 3 induces a more rigid conformation that interferes with self-assembly. To gain more insight about self-assembly of 3 and 5, additional titration experiments were done with the double-stranded template (dA)10-T10 that
Structure–property relationships and third-order nonlinearities in diketopyrrolopyrrole based D–π–A–π–D molecules
Beilstein J. Org. Chem. 2017, 13, 2374–2384, doi:10.3762/bjoc.13.235
- , respectively. Commercially available 4-(N,N-dimethylamino)phenylboronic, 4-methoxyphenylboronic, and 2-thienylboronic acids completed with tributyl(5-methoxythiophen-2-yl)stannane, prepared from 2-methoxythiophene [33], were used. Chromophore 4a was also attempted by Suzuki–Miyaura reaction using the
- (ΔλmaxA = 32 nm). However, chromophore 2b with a 4-methoxyphenyl donor possesses almost identical absorption maxima as thiophen-2-yl derivative 3b (λmaxA = 605 nm), implying their similar electron-releasing abilities. A combination of both methoxy and thiophene auxiliary donors as in 4b shifted its
- as follows: Generally largest THG responses were recorded for chromophores with extended π-system in series b. Chromophore 1b bearing a strong N,N-dimethylamino donor and an extended π-system showed the highest third-order NLO response of 130 pm2/V2. However, a comparison of electron donors is less
Synthesis and application of trifluoroethoxy-substituted phthalocyanines and subphthalocyanines
Beilstein J. Org. Chem. 2017, 13, 2273–2296, doi:10.3762/bjoc.13.224
- conjugation. The electronic interaction between the covalently connected chromophore unit leads to important changes in the absorption spectra and so these dimers are expected to serve as attractive building blocks for the construction of multicomponent photoinduced electron transfer supramolecular systems
Remarkable functions of sn-3 hydroxy and phosphocholine groups in 1,2-diacyl-sn-glycerolipids to induce clockwise (+)-helicity around the 1,2-diacyl moiety: Evidence from conformation analysis by 1H NMR spectroscopy
Beilstein J. Org. Chem. 2017, 13, 1999–2009, doi:10.3762/bjoc.13.196

- representative model for the 1,2-diacyl-sn-glycerols, as categorized in Scheme 1. Although the exciton chirality CD methodology is not applicable for these 1,2-diacyl-sn-glycerolipids without an appropriate UV/CD chromophore, 1H NMR spectroscopy will permit the precise determination of their helical
p-tert-Butylthiacalix[4]arenes functionalized by N-(4’-nitrophenyl)acetamide and N,N-diethylacetamide fragments: synthesis and binding of anionic guests
Beilstein J. Org. Chem. 2017, 13, 1940–1949, doi:10.3762/bjoc.13.188
- p-tert-butylthiacalix[4]arene 4 is also of interest because this fragment contains a polar NH group able to interact with anionic substrates and a chromophore fragment necessary for the spectrophotometric detection of the complex formation. In this regard, the alkylation of monosubstituted
Theoretical simulation of the infrared signature of mechanically stressed polymer solids
Beilstein J. Org. Chem. 2017, 13, 1710–1716, doi:10.3762/bjoc.13.165

- Hessian matrix in redundant internal coordinates under the influence of an external mechanical force [75][80]. So far, most studies on infrared spectroscopy of stressed polymers focused on polypropylene [30]. Lacking a pronounced infrared chromophore, however, the spectrum is relatively complicated
Correlation of surface pressure and hue of planarizable push–pull chromophores at the air/water interface
Beilstein J. Org. Chem. 2017, 13, 1099–1105, doi:10.3762/bjoc.13.109

- observed hue of the monolayer starting at 18 mN/m. Although the measured areas per molecule are not fully quantitative [23], a value of 38 Å2 can be assumed for the flipper chromophore in the condensed state. Upon expansion, the hue relaxes back to the initial range. Compared to compression the hue
Cycloheximide congeners produced by Streptomyces sp. SC0581 and photoinduced interconversion between (E)- and (Z)-2,3-dehydroanhydrocycloheximides
Beilstein J. Org. Chem. 2017, 13, 1039–1049, doi:10.3762/bjoc.13.103
- intensities) at 36 h (Figure 5), whereas no changes were detected for the solutions kept in the dark (Figure 5), confirming the interconversion is a photoinduced geometrical isomerization. Compounds 2 and 3 possess a chiral 6-ethylidene-2-cyclohexenone chromophore. To the best of our knowledge, Z/E
- photoisomerizations of compounds with this kind of chromophore had been rarely reported. For better understanding the insights into this reaction, we conducted theoretical investigation using the truncated structures 2a and 3a (Figure 1). At first, the lowest-energy geometries of 2a and 3a in MeOH in the ground (S0
- Φ of these geometries are presented in Table 2. Compound 4 has a structure similar to 2 except for replacement of the C2–C3 double bond by a single bond. Its geometrical isomerization was not observed in the present study, but (E)-2-ethylidenecyclohexanone, which has a similar chromophore to that of








































































































































































































































































































































