Search results
Search for "arene" in Full Text gives 278 result(s) in Beilstein Journal of Organic Chemistry. Showing first 200.
Multiple threading of a triple-calix[6]arene host
Beilstein J. Org. Chem. 2019, 15, 2092–2104, doi:10.3762/bjoc.15.207

- Fisciano (Salerno), Italy 10.3762/bjoc.15.207 Abstract The synthesis of the triple-calix[6]arene derivative 6 in which three calix[6]arene macrocycles are linked to a central 1,3,5-trimethylbenzene moiety is reported. Derivative 6 is able to give multiple-threading processes in the presence of
- architectures. Historically, the most common components were dialkylammonium ions, as axles, and crown ethers, cyclodextrins, or cucurbiturils, as wheels [1]. With respect to the possible types of wheels, more recently, we have introduced the through-the-annulus threading of simple calix[6]arene hosts with
- -catenanes [25]. On the basis of these results, we were also able to assemble high-order architectures by double-threading of bis-calix[6]arene hosts with ammonium axles [27]. In particular, handcuff (pseudo)rotaxane architectures (e.g., 32+ in Figure 2) [27] were obtained by double-threading of bis-calix[6
Fluorinated azobenzenes as supramolecular halogen-bonding building blocks
Beilstein J. Org. Chem. 2019, 15, 2013–2019, doi:10.3762/bjoc.15.197
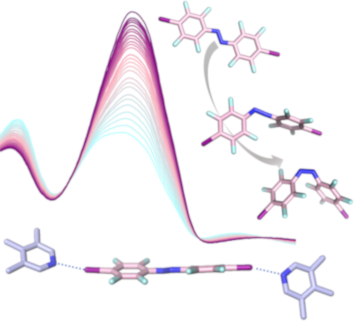
- in the field of crystal engineering [1]. Only few supramolecular capsules were reported so far [29], including the resorcin[4]arene capsules of Diederich and co-workers [21][23], triangular macrocycles assembled by self-complemented halogen bonding [20] and halogen bond templated, polyfluorinated
Attempted synthesis of a meta-metalated calix[4]arene
Beilstein J. Org. Chem. 2019, 15, 1996–2002, doi:10.3762/bjoc.15.195
- Christopher D. Jurisch Gareth E. Arnott Department of Chemistry and Polymer Science, Stellenbosch University, Private Bag X1, Matieland, 7602, South Africa 10.3762/bjoc.15.195 Abstract An evidence for the formation of a rare meta-metalated inherently chiral calix[4]arene is described. Our
- whose chiral nature is a result of selective substitution on the calix[4]arene by various functional groups [9]. In particular, we have focused on the less-studied meta-substituted derivatives [10] whose inherent chirality is more commonly introduced by a suitable directing group [11][12][13][14][15
- this using a pyridyl sulfoxide directing group and palladium as the metal, but in this case the palladium caused a double C–H activation and formed an impressive bridge to the adjacent calix[4]arene aromatic ring [14]. The question for us was whether this bridge formation was a general principle or
Synthesis and anion binding properties of phthalimide-containing corona[6]arenes
Beilstein J. Org. Chem. 2019, 15, 1976–1983, doi:10.3762/bjoc.15.193
- Meng-Di Gu Yao Lu Mei-Xiang Wang MOE Key Laboratory of Bioorganic Phosphorous Chemistry and Chemical Biology, Department of Chemistry, Tsinghua University, Beijing 100084, China 10.3762/bjoc.15.193 Abstract Functionalized O6-corona[3]arene[3]tetrazines were synthesized efficiently and
- adopt the conformation in which three phthalimide units are cis,trans-orientated. Acting as electron-deficient macrocyclic hosts, the synthesized O6-corona[3]arene[3]tetrazines self-regulated conformational structures to complex anions in the gas phase and in the solid state owing to the anion–π
- noncovalent interactions between anions and the tetrazine rings. Keywords: anion–π interactions; coronarenes; host–guest complexation; N-functionalized phthalimides; O6-corona[3]arene[3]tetrazines; Introduction Synthetic macrocycles [1][2] are always attractive and important because they are unique
Complexation of chiral amines by resorcin[4]arene sulfonic acids in polar media – circular dichroism and diffusion studies of chirality transfer and solvent dependence
Beilstein J. Org. Chem. 2019, 15, 1913–1924, doi:10.3762/bjoc.15.187
- chemistry. In the present study we demonstrate that resorcin[4]arene sulfonic acid (RSA) interacts with chiral amines (amino acid derivatives and aminocavitands) to form inclusion complexes and capsules based on electrostatic interactions. The complexes were characterized by circular dichroism and DOSY NMR
- disintegrative for these complexes. This result is quite non-intuitive and worth attention in the context of formation of supramolecular complexes in polar environment, for which DMSO is most often a first-choice solvent. Keywords: cavitands; chirality; macrocycle; resorcin[4]arene; self-assembly
- cyclodextrines, which, when unmodified, interact with a wide range of hydrophobic guests albeit with low general selectivity and enantioselectivity in particular [6]. In this paper we present a group of synthetic macrocyclic compounds – resorcin[4]arene sulfonic acids (RSAs) and analyze their interactions with
Halide metathesis in overdrive: mechanochemical synthesis of a heterometallic group 1 allyl complex
Beilstein J. Org. Chem. 2019, 15, 1856–1863, doi:10.3762/bjoc.15.181

- centroid distance is 2.99 Å, which is typical for K+…(arene) cation–π interactions [22][23]. The enthalpy of binding (∆H°) of an isolated K…(benzene or toluene) unit is almost 80 kJ mol−1 (see calculated value in Table 1, entry 5); the energy is reduced by about 40% when the ring is bound to the neutral
Complexation of 2,6-helic[6]arene and its derivatives with 1,1′-dimethyl-4,4′-bipyridinium salts and protonated 4,4'-bipyridinium salts: an acid–base controllable complexation
Beilstein J. Org. Chem. 2019, 15, 1795–1804, doi:10.3762/bjoc.15.173

- 10.3762/bjoc.15.173 Abstract 2,6-Helic[6]arene and its derivatives were synthesized, and their complexation with 1,1′-dimethyl-4,4′-bipyridinium and protonated 4,4'-bipyridinium salts were investigated in detail. It was found that the helic[6]arene and its derivatives could all form 1:1 complexes with
- both 1,1′-dimethyl-4,4'-bipyridinium salts and protonated 4,4'-bipyridinium salts in solution and in the solid state. Especially, the helic[6]arene and its derivatives containing 2-hydroxyethoxy or 2-methoxyethoxy groups exhibited stronger complexation with the guests than the other helic[6]arene
- with larger counteranions of the guests and in less polar solvents. Furthermore, the switchable complexation between the helic[6]arene and protonated 4,4'-bipyridinium salt could be efficiently controlled by acids and bases. Keywords: 4,4'-bipyridinium salts; complexation; helic[6]arene; hydrogen bond
Recent advances on the transition-metal-catalyzed synthesis of imidazopyridines: an updated coverage
Beilstein J. Org. Chem. 2019, 15, 1612–1704, doi:10.3762/bjoc.15.165

Synthesis, enantioseparation and photophysical properties of planar-chiral pillar[5]arene derivatives bearing fluorophore fragments
Beilstein J. Org. Chem. 2019, 15, 1601–1611, doi:10.3762/bjoc.15.164

- Guojuan Li Chunying Fan Guo Cheng Wanhua Wu Cheng Yang Key Laboratory of Green Chemistry & Technology, College of Chemistry and Healthy Food Evaluation Research Center, Sichuan University, 29 Wangjiang Road, Chengdu, 610064, P. R. China 10.3762/bjoc.15.164 Abstract Planar chiral pillar[5]arene
- derivatives (P5A-DPA and P5A-Py) bearing bulky fluorophores were obtained in high yield by click reaction. The photophysical properties of both compounds were investigated in detail. P5A-DPA with two 9,10-diphenylanthracene (DPA) pigments grafted on the pillar[5]arene showed a high fluorescence quantum yield
- of 89.5%. This is comparable to the monomer DPA-6, while P5A-Py with two perylene (Py) pigments grafted on the pillar[5]arene showed a significantly reduced quantum yield of 46.4% vs 78.2% for the monomer Py-6. The oxygen-through-annulus rotation of the phenolic units was inhibited for both compounds
Different reactivity of phosphorylallenes under the action of Brønsted or Lewis acids: a crucial role of involvement of the P=O group in intra- or intermolecular interactions at the formation of cationic intermediates
Beilstein J. Org. Chem. 2019, 15, 1491–1504, doi:10.3762/bjoc.15.151
- 8–11a (Table 2). The presence of the (MeO)2P=O group in the structures of reaction products makes them more soluble in organic solvents and easy to isolate in preparative reactions. Depending on the structure of the starting arene, allene 1a gave two kinds of reaction products, E-/Z-alkenes 11 and
- equivalent of AlCl3 is coordinated to the central atom of the allene system of the complex 13 and gives intermediate 16. The latter, in the absence of nucleophiles (arene molecules), undergoes deprotonation from the methyl group affording butadiene 14. Hydrolysis of the latter resulted in compounds Z-9 and E
- -10a. Whereas, in the presence of an arene, cation 16 reacts with it leading to species 17. The latter can be protonated with the formation of cation 18. This species may react in two different ways. The first option is it could lead to alkene 19 and finally to compounds 11 upon methanolysis of the
2,3-Dibutoxynaphthalene-based tetralactam macrocycles for recognizing precious metal chloride complexes
Beilstein J. Org. Chem. 2019, 15, 1460–1467, doi:10.3762/bjoc.15.146

- bonds with guests, constituting the main driving forces for molecular recognition. This class of macrocycles, in particular the ones with single arene as sidewalls, have found wide applications in diverse fields: Leigh and co-workers reported many interlocked structures and molecular machines with
- interesting new binding properties. During the last six years, our group have developed a series of naphthol-based macrocyclic receptors [24][25][26][27][28][29]. Of them, oxatub[4]arene [30][31] and zorb[4]arene [32][33] show multiple conformations due to the flipping of naphthalenes and thus resulted in a
- timescale. H3 is diastereotopic and could be split when the conformational interconversion is slow on the NMR timescale, as observed for oxatub[4]arene [30] and zorb[4]arene [32]. When lowering the temperature to 223 K, H3 of 1 was split to two peaks while other peaks become broadened (Figure 1b). This
Complexation of a guanidinium-modified calixarene with diverse dyes and investigation of the corresponding photophysical response
Beilstein J. Org. Chem. 2019, 15, 1394–1406, doi:10.3762/bjoc.15.139

- 300071, P. R. China Research Institute of Petroleum Engineering, Sinopec, Beijing 100101, P. R. China 10.3762/bjoc.15.139 Abstract We herein describe the comprehensive investigation of the complexation behavior of a guanidinium-modified calix[5]arene pentaisohexyl ether (GC5A) with a variety of typical
- fluorescent IDA with guanidinium-modified calix[5]arene pentaisohexyl ether (GC5A) and fluorescein (Fl) as the reporter pair [26]. The ultrasensitive detection is feasible for diagnosing ovarian and other gynecologic cancers at their early stages. Also, we proposed a nanoplatform where the fluorescence and
- photoactivity of photosensitizers (PSs) were annihilated by the complexation of guanidinium-modified calix[5]arene pentadodecyl ether while reactivated by adenosine triphosphate (a cancer biomarker) displacement. This novel supramolecular phototheranostics strategy realized both tumor-selective imaging and
Efficient resolution of racemic crown-shaped cyclotriveratrylene derivatives and isolation and characterization of the intermediate saddle isomer
Beilstein J. Org. Chem. 2019, 15, 1339–1346, doi:10.3762/bjoc.15.133

- . Isolation of the product was then possible through repeated column chromatography. Similarly, the same group was also able to isolate the saddle stereoisomer of a chiral nonamethoxy-substituted CTV in which every arene unit carries three additional substituents through HPLC on a chiral stationary phase [27
Host–guest interactions between p-sulfonatocalix[4]arene and p-sulfonatothiacalix[4]arene and group IA, IIA and f-block metal cations: a DFT/SMD study
Beilstein J. Org. Chem. 2019, 15, 1321–1330, doi:10.3762/bjoc.15.131

- .15.131 Abstract The molecular recognition in aqueous solution is extremely important because most biological processes occur in aqueous solution. Water-soluble members of the calix[n]arene family (e.g., p-sulfonato substituted) can serve as model systems for studying the nature and manner of interactions
- between biological receptors and small ions. The complex formation behavior of water-soluble p-sulfonatocalix[4]arene and thiacalix[4]arene and group IA, IIA and f-block metal cations has been investigated computationally by means of density functional theory computations in the gas phase and in aqueous
- cations (La3+) a supramolecular approach with explicit solvent treatment has been applied in the study of the effect of metal hydration on the complexation process. The La3+ binding to the p-sulfonatocalix[4]arene host molecule (now in the metal’s second coordination shell) is still exergonic as evidenced
N-doped carbon dots covalently functionalized with pillar[5]arenes for Fe3+ sensing
Beilstein J. Org. Chem. 2019, 15, 1262–1267, doi:10.3762/bjoc.15.123

- Engineering, Wuhan University of Science and Technology, Wuhan 430081, P. R. China 10.3762/bjoc.15.123 Abstract Fluorescent N-doped carbon dots (CN-dots) covalently functionalized with carboxylatopillar[5]arene (CP[5]), namely CCDs, have been prepared the first time. Compared with CN-dots without pillarene
- , symmetrical structures, superior host–guest properties, supramolecular assembly characteristics, and versatile functionalities [11][15][16]. Carboxylatopillar[5]arene (CP[5]) is one of the most popular functional pillarenes that has been exploited in many research areas, especially in sensing and detection
Multicomponent reactions (MCRs): a useful access to the synthesis of benzo-fused γ-lactams
Beilstein J. Org. Chem. 2019, 15, 1065–1085, doi:10.3762/bjoc.15.104
- was chemoselective, furnishing in good yields the products resulting from the transfer of the less hindered arene. However, this methodology exhibits two main limitations. First, arenes 86 with electron-withdrawing groups did not react under the optimized conditions and on the other hand, the atom
- economy is quite low, since a half of the diaryliodonium salt 87 is lost in the process. Replacing the arene substrate 86 by ketones, and keeping nearly equal reaction conditions, the same authors have achieved an efficient synthesis of 3-(2-oxopropyl)- or pentacyclic isoindolinones 90 or 92 (Scheme 26
- , that rearranges to afford cationic isoindolinone moiety 95. Then, benzene derivative 86 would react by a Friedel–Crafts type reaction to form diaryl γ-lactam 88. When ketone 89 is used instead of arene 86, the enol form would act as nucleophile and upon reaction with carbocation 95, compounds 90 could
Fabrication, characterization and adsorption properties of cucurbit[7]uril-functionalized polycaprolactone electrospun nanofibrous membranes
Beilstein J. Org. Chem. 2019, 15, 992–999, doi:10.3762/bjoc.15.97

- series of CD-functionalized electrospun nanofibers in the forms of CD-pseudopolyrotaxane [11][12], CDs/polymer [13][14][15], CD-ICs/polymer [16][17], and polymer-free CDs [5][18] or CD-ICs [19] have been fabricated. In addition, calix[8]arene (C[8])/polyacrylonitrile (PAN) composite nanofiber membranes
- [20] and supramolecular polymer nanofibers based on pillar[5]arene [21] also were prepared by the electrospinning technique. Cucurbit[n]urils (CB[n]s, n = 5–8, 10) are a family of pumpkin-shaped cyclic host molecules containing a hydrophobic cavity surrounded by two identical hydrophilic portals [22
Mechanochemistry of supramolecules
Beilstein J. Org. Chem. 2019, 15, 881–900, doi:10.3762/bjoc.15.86
- synthesis of pillar[5]arene-containing [2]rotaxanes (Figure 8). Mixing a 2:1 ratio of pillar[5]arene (wheel) with dodecanedioyl dichloride (axle) in CHCl3 resulted in the formation of pseudorotaxane 16 which was further treated with different amines (stopper) in a stainless-steel jar with 4 steel balls
- (Figure 21a). However, 1,8-dipyridylnaphthalene was used as hydrogen-bond acceptor for the [2 + 2] cycloaddition of fumaric acid derivatives (Figure 21b). In 2017 Purse and co-workers reported the host–guest chemistry of pyrogallo[4]arene (39) hexamers under mechano-milling conditions [94]. A hexameric
- of unsymmetrically substituted azobenzene [58]. a) Schematic representation for the construction of a [2]rotaxane. b) Chiu’s ball-milling approach for the synthesis of [2]rotaxanes. Mechanochemical synthesis of the smallest [2]rotaxane. Solvent-free mechanochemical synthesis of pillar[5]arene
Polyaminoazide mixtures for the synthesis of pH-responsive calixarene nanosponges
Beilstein J. Org. Chem. 2019, 15, 633–641, doi:10.3762/bjoc.15.59
- copolymers (CyCaNSs) [16][17][18], which showed pH-dependent sequestration abilities towards different model organic substrates. The latter NSs were easily synthesized by means of a CuAAC-type reaction [19][20][21] between a heptakisazido-β-cyclodextrin and a tetrakis(propargyloxy)calix[4]arene derivative
- . More recently, we also reported the synthesis of entirely synthetic calixarene nanosponges (CaNSs) [22], which can be prepared by means of the same CuAAC approach, by reacting a tetrakis(propargyloxy)calix[4]arene with a diazidoalkane. Of course, the reaction results in the formation of bis(1,2,3
- ) of polyaminoazide mixtures, which were in turn used for the preparation of CaNSs materials with pH-tunable properties, by reaction with the (5,11,17,23-tetra(tert-butyl))-(25,26,27,28-tetrakis(propargyloxy)calix[4]arene (Ca) under the CuAAC reaction conditions. In turn, the synthon Ca is obtained by
Selective benzylic C–H monooxygenation mediated by iodine oxides
Beilstein J. Org. Chem. 2019, 15, 602–609, doi:10.3762/bjoc.15.55
- intractable products, which were in many cases polyfunctionalized. Additionally, substrates containing arene rings were shown to inhibit aliphatic C–H functionalization via electrophilic trapping of reactive halogen species to form aryl chlorides and iodides. Utilizing NHPI as a less reactive hydrogen atom
- NHPI-catalyzed C–H acetoxylation reaction yielded the ester product 3a in moderate (42%) yield. This contrasts with orthoperiodic acid (H5IO6), which yielded only electrophilic arene iodination. The use of alkali metal iodate salts such as lithium, sodium, and potassium iodate as the terminal oxidant
- decatungstate [58]. Substrates containing tertiary C–H bonds, such as 1-isopropyl-4-methylbenzene (6b), were essentially unreactive under the developed conditions, returning starting alkyl arene with only a trace of acetoxylated product 7b (see Supporting Information File 1, Figure S11) [59]. Previous work on
Dirhodium(II)-catalyzed [3 + 2] cycloaddition of N-arylaminocyclopropane with alkyne derivatives
Beilstein J. Org. Chem. 2019, 15, 542–550, doi:10.3762/bjoc.15.48
- 3,5-dimethyl compound 1d afforded the desired product 3d with an excellent yield of 91% (Table 2, entry 3). We reasoned the electron-withdrawing groups on the arene increased the nitrogen–hydrogen bond dissociation energy (BDEN–H) of compound 1 [21], decreasing the rate of ring opening. Substrates 1i
N-Arylphenothiazines as strong donors for photoredox catalysis – pushing the frontiers of nucleophilic addition of alcohols to alkenes
Beilstein J. Org. Chem. 2019, 15, 52–59, doi:10.3762/bjoc.15.5
- characteristics of the derivatives 1–9 were analyzed and compared (Figure 3). The parent compound N-phenylphenothiazine (1) shows an absorption maximum at 320 nm. Substitution of the arene moiety results in a shift of the absorption maxima due to a change in the HOMO–LUMO gaps. It turned out that the introduction
- found to occur at E(1+·/1) = 0.75 V (vs SCE). The substitution of the arene moiety by one (see 7) or two fluorine substituents (see 8) only leads to a shift in the reduction potential of about 0.06 V. This trend was expected due to the lower electron density of these two N-phenylphenothiazines at the
- arene moiety. However, the effect by the pure σ-acceptor fluorine is not very pronounced. In the case of the 4-NO2 substituted derivative 6 the pronounced influence of the π-acceptor shifts the reduction potential to a value of up to E(6+·/6) = 0.89 V (vs SCE). Substitution of the N-aryl moiety by
Ruthenium-based olefin metathesis catalysts with monodentate unsymmetrical NHC ligands
Beilstein J. Org. Chem. 2018, 14, 3122–3149, doi:10.3762/bjoc.14.292
- converted completely within a few hours into complex 2 due to the formation of an intramolecular carbene–arene bond between the benzylidene carbon atom and the ortho position of the N-phenyl ligand (Figure 3). According to the authors, the mechanism of the reaction that occurs only in the presence of oxygen
- containing a monosubstituted backbone and two different N-aryl groups (202–204, Figure 37) [63]. The idea behind this new category of compounds lied in the possibility of an efficient transfer of chirality from the backbone group to the metal center through a significant twisting of the monosubstituted arene
Synthesis of aryl sulfides via radical–radical cross coupling of electron-rich arenes using visible light photoredox catalysis
Beilstein J. Org. Chem. 2018, 14, 2520–2528, doi:10.3762/bjoc.14.228
- (dtbpy)]PF6 as the photocatalyst. The reaction was carried out under nitrogen under visible-light irradiation at 455 nm. The oxidation potential of this test arene is 1.02 V vs SCE, which allows oxidation by [Ir(dF(CF3)ppy)2(dtbpy)]PF6 having an estimated excited state oxidation potential of 1.21 V vs
- -methoxyphenyl) disulfide was employed as the thiolating agent (3h). With symmetrical arenes, diarylation was observed. The initially formed mono-arylated product is more reactive than the starting material and reacts to the diarylthiol product. When 1-phenyl-1H-pyrrole-2,5-dione was employed as the arene, the
- sulfenylation occurred exclusively at the double bond instead at the arene to give the product 3k in 58% yield and a trace amount of diarylation product of the aromatic ring. When the more difficult to oxidize 2-methoxynaphthalene was used as substrate, the product 3l was obtained in only 30% yield indicating
Calixazulenes: azulene-based calixarene analogues – an overview and recent supramolecular complexation studies
Beilstein J. Org. Chem. 2018, 14, 2488–2494, doi:10.3762/bjoc.14.225

- -containing structures have gained much recent attention. Among the least-studied to date, however, have been the azulene unit-containing calix[4]arene analogues. In 1988 Asao et al. reported the synthesis of the first azulene analogue of the calixarenes, which they called “azulenophane” 2 [14]. They used a





















































































































































































































































































































































































































































































































