Search results
Search for "Candida albicans" in Full Text gives 55 result(s) in Beilstein Journal of Organic Chemistry.
An overview of recent advances in duplex DNA recognition by small molecules
Beilstein J. Org. Chem. 2018, 14, 1051–1086, doi:10.3762/bjoc.14.93
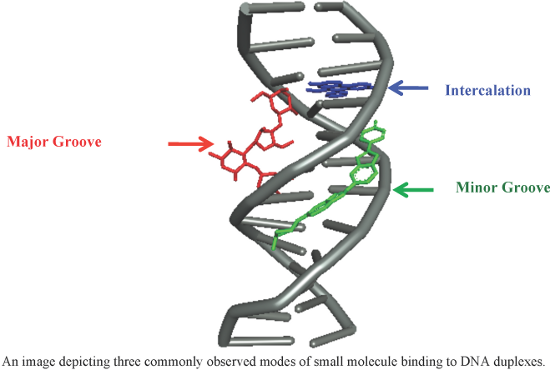
- shown to bind A·T-rich regions preferentially. The compounds containing branched N-alkylpyrrole, hydrophobic N-terminal amide, and especially C-isopropylthiazole (thiazotropsin A as shown in the Figure 4) showed significant antimicrobial activity against MRSA and Candida albicans strains. Thiazotropsin
Herpetopanone, a diterpene from Herpetosiphon aurantiacus discovered by isotope labeling
Beilstein J. Org. Chem. 2017, 13, 2458–2465, doi:10.3762/bjoc.13.242
- performed as previously described [27]. The test organisms included Bacillus subtilis ATCC 6633, Staphylococcus aureus SG 511, Mycobacterium vaccae IMET 10670, Escherichia coli SG 458, Pseudomonas aeruginosa K 799/61, Sporobolomyces salmonicolor SBUG 549, Candida albicans ATCC 14053 and Penicillium notatum
Diosgenyl 2-amino-2-deoxy-β-D-galactopyranoside: synthesis, derivatives and antimicrobial activity
Beilstein J. Org. Chem. 2017, 13, 2310–2315, doi:10.3762/bjoc.13.227

- as well as the HRMS spectrometry confirmed the structures of 11–13 (see Supporting Information File 1 for experimental and NMR data). Evaluation of antimicrobial activity The antimicrobial in vitro activities of 5–13 were tested against two fungal strains: Candida albicans and Candida tropicalis, and
Biosynthetic origin of butyrolactol A, an antifungal polyketide produced by a marine-derived Streptomyces
Beilstein J. Org. Chem. 2017, 13, 441–450, doi:10.3762/bjoc.13.47
- recently, isolation of butyrolactols C and D was presented but the details are not available in public domains [10]. 1 has a broad antimicrobial activity against fungi ranging from Candida albicans to Trichophyton mentagrophytes with comparative activity to nystatin [9]. Despite the uniqueness of the
Interactions between cyclodextrins and cellular components: Towards greener medical applications?
Beilstein J. Org. Chem. 2016, 12, 2644–2662, doi:10.3762/bjoc.12.261

Marine-derived myxobacteria of the suborder Nannocystineae: An underexplored source of structurally intriguing and biologically active metabolites
Beilstein J. Org. Chem. 2016, 12, 969–984, doi:10.3762/bjoc.12.96
- ), Candida albicans (DSM 1665) and Mucor hiemalis (DSM 2656, 33.3 μg mL−1 in each case). Significant cytotoxic activity was revealed for 22 towards SKOV-3 (IC50 = 2.4 μM), KB3-1 (IC50 = 5.3 μM), as well as A431 (IC50 = 8.45 μM), while 23 showed remarkable cytotoxicity against cell lines HUVEC and KB3-1 (IC50
Natural products from microbes associated with insects
Beilstein J. Org. Chem. 2016, 12, 314–327, doi:10.3762/bjoc.12.34

- (e.g., bafilomycin A1 (21) and B1 (22), Figure 7), but also a novel polyunsaturated and polyoxygenated 26-membered macrolactam named sceliphrolactam (23) (Figure 7) [101]. Sceliphrolactam showed strong antifungal activity against amphotericin B-resistant Candida albicans, but its functional role in
Bromotyrosine-derived alkaloids from the Caribbean sponge Aplysina lacunosa
Beilstein J. Org. Chem. 2015, 11, 2334–2342, doi:10.3762/bjoc.11.254
- luteus; Gram-negative: Peumonia aruginosa and Klebsiella pneumonia] and antifungal Candida albicans using microdilution technique. The MIC was defined as lowest concentration that shows 50% growth inhibition after 24 hour incubation. Cytotoxicity assay The cytotoxicity was determined using WST-1 cell
Synthesis, antimicrobial and cytotoxicity evaluation of new cholesterol congeners
Beilstein J. Org. Chem. 2015, 11, 1922–1932, doi:10.3762/bjoc.11.208
- antifungal activities against the filamentous fungal strain Aspergillus flavus (Link) and the yeast forming fungal strain Candida albicans (ATCC 7102) were moderate compared with amphotericin B in vitro. In the cytotoxicity study, this derivative was the most cytotoxic one against the prostate cancer PC3
- that were as active as ampicillin. However, conjugate VI from the previous investigation was still more active than these conjugates [24]. The antifungal activity of selected newly synthesized chalcone conjugates was evaluated in vitro against A. flavus (Link) and Candida albicans (ATCC 7102) similarly
Pyridinoacridine alkaloids of marine origin: NMR and MS spectral data, synthesis, biosynthesis and biological activity
Beilstein J. Org. Chem. 2015, 11, 1667–1699, doi:10.3762/bjoc.11.183
- interesting antimicrobial potency (minimal inhibitory concentration, MIC, below 10 µg/mL) has been assigned to some pyridoacridines such as meridine (56) and ascididemin (42). Compound 56 inhibited the growth of Candida albicans (MIC: 0.2–3.1 µg/mL) and Cryptococcus neoformans (MIC 0.8 µg/mL) as well as that
N-Alkyl derivatives of diosgenyl 2-amino-2-deoxy-β-D-glucopyranoside; synthesis and antimicrobial activity
Beilstein J. Org. Chem. 2015, 11, 869–874, doi:10.3762/bjoc.11.97
- with the longer alkyl chains (13–15, 17) show very weak inhibitory activity against Candida tropicalis. The results presented for Candida albicans resemble those obtained for Candida tropicalis. The growth of this strain of fungi is inhibited at the lowest concentrations by 7·HCl and equally well by 8
Cyclodextrin–polysaccharide-based, in situ-gelled system for ocular antifungal delivery
Beilstein J. Org. Chem. 2014, 10, 2903–2911, doi:10.3762/bjoc.10.308
- lead to loss of vision. Candida albicans is one of the most widespread fungal pathogens involved in fungal keratitis [1][2]. The most common antifungal treatments include the use of polyenes and azoles; however, a significant treatment failure rate exists with these pharmacological agents [3
Aspergiloid I, an unprecedented spirolactone norditerpenoid from the plant-derived endophytic fungus Aspergillus sp. YXf3
Beilstein J. Org. Chem. 2014, 10, 2677–2682, doi:10.3762/bjoc.10.282
- Candida albicans (ATCC 10231) or Fusarium oxysporum f. sp. cubense race 4 at concentrations as high as 20 mg/mL. Conclusion In summary, guided by 1H NMR detection, we isolated and characterized a novel norditerpenoid, aspergiloid I (1), from the liquid fermentation broth of the endophytic Aspergillus sp
- graminearum Schw., Fusarium coeruleum Sacc., Botrytis cinerea Pers., and Fusarium oxysporum f. sp. cubense race 4), and Candida albicans (ATCC 10231), and the antioxidant, acetylcholinesterase (AChE) , α-glucosidase, and topoisomerase IIα inhibitory activities were performed in accordance with the primary
Encapsulation of biocides by cyclodextrins: toward synergistic effects against pathogens
Beilstein J. Org. Chem. 2014, 10, 2603–2622, doi:10.3762/bjoc.10.273

- activity of the biocide alone. In 1993, Lehner et al. reported a very interesting study on the antagonistic effect of the complexation between HP-β-CD and a series of p-hydroxybenzoic acid esters against Candida albicans [43]. The authors investigated the complexation in aqueous solution by solubility
Sacrolide A, a new antimicrobial and cytotoxic oxylipin macrolide from the edible cyanobacterium Aphanothece sacrum
Beilstein J. Org. Chem. 2014, 10, 1808–1816, doi:10.3762/bjoc.10.190
- . Antimicrobial testing against four Gram-positive bacteria (Bacillus subtilis, Micrococcus luteus, Staphylococcus aureus, and Streptomyces lividans), one Gram-negative bacterium (Escherichia coli), two yeasts (Candida albicans and Saccharomyces cerevisiae), and two fungi (Aspergillus oryzae and Penicillum
- strains were grown on agar media (Staphylococcus aureus FDA209P JC-1, Micrococcus luteus ATCC9341, Escherichia coli NIHJ JC-2, Saccharomyces cerevisiae S100, and Candida albicans A9540) or in Sabouraud broth (Penicillum chrysogenum NBRC4626, Streptomyces lividans TK23) overnight and then diluted with
Multicomponent reactions in nucleoside chemistry
Beilstein J. Org. Chem. 2014, 10, 1706–1732, doi:10.3762/bjoc.10.179
- amide resin as the amine-bearing component (Scheme 18) [84]. The synthesis of nikkomycin Z analogs 46 aimed in an examination of their ability to inhibit Candida albicans chitin synthases. The library consisting of 450 analogs 46 was obtained from: (i) reactions involving nucleoside aldehyde 41, Rink
- albicans chitin synthase 1 at the concentration of 10 µM. Among the most active analogs 46a–c, compound 46a showed a comparable activity (IC50 = 6.07 µM) as that determined for nikkomycin Z (IC50 = 9.49 µM). On the other hand, inhibitory activity of this compound toward Candida albicans chitin synthase 2
- (IC50 = 4.78 µM) was significantly lower than that of nikkomycin Z (IC50 = 0.06 µM). The remaining compounds 46 were inactive toward Candida albicans chitin synthase 2. Another approach to the solid-phase synthesis of nucleoside analogs was developed by Sun and Lee (Scheme 19) [85]. The library of 1344
Heronapyrrole D: A case of co-inspiration of natural product biosynthesis, total synthesis and biodiscovery
Beilstein J. Org. Chem. 2014, 10, 1228–1232, doi:10.3762/bjoc.10.121
- coli ATCC 25922, and the fungus Candida albicans ATCC 90028. Conclusion In conclusion, this study illustrates the importance of biosynthetic considerations to both natural products and synthetic chemistry. Biosynthetic hypotheses provide natural product chemists with a framework to challenge the
New sesquiterpene hydroquinones from the Caribbean sponge Aka coralliphagum
Beilstein J. Org. Chem. 2014, 10, 613–621, doi:10.3762/bjoc.10.52
- ) show cytotoxic activity against the L929 mouse fibroblasts, KB-31 epidermoid carcinoma, and the breast cancer cell line MCF7, although none of them showed activity in the antimicrobial assays against Gram-negative bacteria and the fungus Candida albicans. Many of the isolated compounds from Aka
Synthesis of five- and six-membered cyclic organic peroxides: Key transformations into peroxide ring-retaining products
Beilstein J. Org. Chem. 2014, 10, 34–114, doi:10.3762/bjoc.10.6
The regulation and biosynthesis of antimycins
Beilstein J. Org. Chem. 2013, 9, 2556–2563, doi:10.3762/bjoc.9.290
- bioactivity of 5-fluoroantimycins and reported they retained potent antifungal activity against Candida albicans, but were significantly reduced in cytotoxity in a leukemia P388 mouse cell line compared to the parent compounds [25]. Sandy et al. recently showed AntB can accept both acyl-CoAs and acyl-SNACs to
- utilise acyl-SNACs provides the possibility to employ feeding studies to introduce new chemistry at C-8. AntO is a lipase homologue and is predicted to install the N-formyl group resulting in the 3-formamidosalicylate moiety. AntO is required for bioactivity against the human pathogenic fungus, Candida
- albicans (Seipke and Hutchings, unpublished results), but the exact time in which AntO installs the N-formyl group is unclear and requires investigation. Regulation of the ant gene cluster Antimycin biosynthesis is linked to development, as is the case for many Streptomyces secondary metabolites. However
Synthesis and biological activities of the respiratory chain inhibitor aurachin D and new ring versus chain analogues
Beilstein J. Org. Chem. 2013, 9, 1551–1558, doi:10.3762/bjoc.9.176

- (Candida albicans) nor on molds (Mucor hiemalis, Fusarium oxysporum, Alternaria alternata). The natural compound 4 was one of the most active compounds, but also shorter chain analogues 9 and 10 inhibited, for instance, the growth of Staphylococcus aureus at concentrations in the low µg/mL range, whereas
ML212: A small-molecule probe for investigating fluconazole resistance mechanisms in Candida albicans
Beilstein J. Org. Chem. 2013, 9, 1501–1507, doi:10.3762/bjoc.9.171
- Molecular Libraries and Probe Production Centers Network (NIH-MLPCN) screened >300,000 compounds to evaluate their ability to restore fluconazole susceptibility in resistant Candida albicans isolates. Additional counter screens were incorporated to remove substances inherently toxic to either mammalian or
- fungal cells. A substituted indazole possessing the desired bioactivity profile was selected for further development, and initial investigation of structure–activity relationships led to the discovery of ML212. Keywords: antifungal; Candida albicans; chemosensitizer; fluconazole; Molecular Libraries
- chemosensitizers of the pathogenic fungus Candida albicans [11]. The C. albicans clinical isolates used in this study demonstrate a range of resistance to the widely prescribed triazole antimycotic fluconazole (Flc) [12], and the objective was to identify novel small molecules capable of surmounting this inherent
A macrolactonization approach to the total synthesis of the antimicrobial cyclic depsipeptide LI-F04a and diastereoisomeric analogues
Beilstein J. Org. Chem. 2012, 8, 1344–1351, doi:10.3762/bjoc.8.154
- strains of Bacillus (Paenebacillus) and exhibit antifungal and antibacterial activity against a range of clinically relevant species, including Candida albicans, Cryptococcus neoformans, Staphylococcus aureus and Micrococcus luteus [1][2][3][4][5][6][7]. These compounds have a cyclic hexadepsipeptide core
- . The antifungal activity of 1 and 20–23 was evaluated by using a standardised serial dilution sensitivity assay [28] against reference strains of Candida albicans, Cryptococcus neoformans and Aspergillus fumigatus (Table 3). Synthetic LI-F04a was found to exhibit good activity against both C. albicans
Triterpenoid saponins from the roots of Acanthophyllum gypsophiloides Regel
Beilstein J. Org. Chem. 2012, 8, 763–775, doi:10.3762/bjoc.8.87
- VKM Y-1173 (All-Russian Collection of Microorganisms), Candida albicans JCM 1542 (Japan Collection of Microorganisms) were used as test-cultures. Strains were maintained on malt agar slants at 5 °C. S. cerevisiae VKM Y-1173 was grown in YPD medium containing glucose (20 g/L), yeast extract (10 g/L
- ) and peptone (20 g/L; Sigma, USA) under shaking at 30 °C for 24 h. Candida albicans, Filobasidiella neoformans, and Cryptococcus terreus were grown under the same conditions for 48 h in YPD medium containing glucose (20 g/L), yeast extract (4 g/L), peptone (5 g/L). Antifungal activity assay: Sterile 5
Synthesis and antifungal properties of papulacandin derivatives
Beilstein J. Org. Chem. 2012, 8, 732–737, doi:10.3762/bjoc.8.82
- could be obtained, possibly caused by aggregation. This was not a problem for the other three compounds. The final products were tested for inhibition of Candida albicans. No inhibition was observed for the all-trans-retinoic acid derivative 51 and the sorbic acid derivative 48. The MIC for the linoleic
































































































































































































































































































































































































































