Search results
Search for "diversity" in Full Text gives 482 result(s) in Beilstein Journal of Organic Chemistry. Showing first 200.
Five-component, one-pot synthesis of an electroactive rotaxane comprising a bisferrocene macrocycle
Beilstein J. Org. Chem. 2020, 16, 1564–1571, doi:10.3762/bjoc.16.128
- to perform several functions as artificial molecular switches [1]. The template-directed synthesis of such sophisticated catenane and rotaxane molecular architectures allowed the expansion of chemical diversity and properties. Among these architectures, electroactive rotaxanes have been described to
One-pot synthesis of 1,3,5-triazine-2,4-dithione derivatives via three-component reactions
Beilstein J. Org. Chem. 2020, 16, 1447–1455, doi:10.3762/bjoc.16.120
- anticancer [10][11], antimicrobial [12], antiviral agents [13][14], and eosinophilia inhibitors [15]. Therefore, it is highly desirable to develop efficient and practical synthetic methods for triazinethione architectures and to expand the structure diversity of this class of compounds for medicinal
- molecules. They are quite flexible as possible starting substrates, and through cyclization reactions, can lead to a variety of complex molecules, allowing a wide exploration of scaffolds and substituents diversity [24][25][26][27][28]. In this regard, the use of simple thiourea as an inexpensive
Distinctive reactivity of N-benzylidene-[1,1'-biphenyl]-2-amines under photoredox conditions
Beilstein J. Org. Chem. 2020, 16, 1335–1342, doi:10.3762/bjoc.16.114
- . The developed method can be employed for the synthesis of various bulky vicinal diamines, which are potential ligands in stereoselective synthesis. Keywords: 1,2-diamine; diversity; imine; photocatalysis; visible light; Introduction The selective formation of distinct valuable compounds from the
- product 3a was unambiguously confirmed using X-ray crystallography [57]. To further extend the diversity of the reactivity of the imines in this process, we also attempted the use of a reducing agent in the reaction of 1a. Pleasingly, the reaction of 1a with 2 equivalents of the Hantzsch ester in CH3CN
Highly selective Diels–Alder and Heck arylation reactions in a divergent synthesis of isoindolo- and pyrrolo-fused polycyclic indoles from 2-formylpyrrole
Beilstein J. Org. Chem. 2020, 16, 1320–1334, doi:10.3762/bjoc.16.113
- activity [15][16] and great diversity as building block [17][18]. Among the tetracyclic scaffolds, isoindolo[2,1-a]indoles have attracted a particular interest from the synthetic point of view, either as the target or the motif for designing novel annulation methodologies [19][20][21]. Indeed, isoindole
Oxime radicals: generation, properties and application in organic synthesis
Beilstein J. Org. Chem. 2020, 16, 1234–1276, doi:10.3762/bjoc.16.107
- electron is localized on the N–O fragment (N-oxyl radicals, Figure 1) occupy a special place in organic chemistry due to the increased stability and ease of generation, the diversity of their structures, properties, and applications. Stable N-oxyl radicals (mainly of the aminoxyl type, Figure 1, I) are
Anthelmintic drug discovery: target identification, screening methods and the role of open science
Beilstein J. Org. Chem. 2020, 16, 1203–1224, doi:10.3762/bjoc.16.105
- . elegans. A rich diversity of mutants is available via the Caenorhabditis Genetics Centre [91]. The discovery of RNA interference delivered via feeding worms double-stranded DNA [92] has opened the door to genome-scale gene knockdown in the search for new drug targets. These approaches can expedite the
- researchers agree to share their results. This model follows on from the successful MMV Malaria Box, where 55 groups compiled results from over 290 different assays in a diversity of screens related not only to malaria, but also to other neglected tropical diseases [160]. The Pathogen Box project is currently
A smart deoxyribozyme-based fluorescent sensor for in vitro detection of androgen receptor mRNA
Beilstein J. Org. Chem. 2020, 16, 1135–1141, doi:10.3762/bjoc.16.100

- their binding capability [3] with sensitivities in the range from 1 mM for cocaine to 10 pM for Hg2+, but for many targets it averages in the 1–10 nM range [4]. Although the diversity of NAs is very large, they can be divided into DNA-based biosensors, for example, molecular beacon or TaqMan probes
Palladium-catalyzed regio- and stereoselective synthesis of aryl and 3-indolyl-substituted 4-methylene-3,4-dihydroisoquinolin-1(2H)-ones
Beilstein J. Org. Chem. 2020, 16, 1084–1091, doi:10.3762/bjoc.16.95
- antimetastatic agents [14]. Fittingly, the development of efficient strategies for their construction and peripheral functionalization represents still an active research area aimed to achieve structural diversity [15][16][17][18]. Carbometalations of alkynes constitute a powerful tool for the regio- and
- pleased to report here that this cascade reaction takes place efficiently, resulting in the regio- and stereoselective formation of the poly-substituted isoquinolinones 4 in good to high yield. Applications of this reaction can be relevant for improvements of structure diversity and fine tuning of the
Fabclavine diversity in Xenorhabdus bacteria
Beilstein J. Org. Chem. 2020, 16, 956–965, doi:10.3762/bjoc.16.84
- of the insect by the nematode, the bacteria are released from the nematode gut into the insect hemocoel where they start producing a diversity of different natural products to suppress the immune response and to kill the insects, to defend the carcass against food competitors, and to trigger the
- diversity, selected strains were analyzed both chemically and genomically, and mutants in putative fcl BGCs were generated. Thereby, a list of derivatives was obtained, which was further correlated to the potential fabclavine-producing, but genetically not accessible X. innexi strain. Finally, the
- the anterior midgut of larvae [43]. Consequently, this mode of action could also be possible for fabclavines. Conclusion This study revealed a large chemical diversity for fabclavine derivatives among different Xenorhabdus strains, which is achieved by the promiscuity of single enzymes or domains
Diversity-oriented synthesis of 17-spirosteroids
Beilstein J. Org. Chem. 2020, 16, 880–887, doi:10.3762/bjoc.16.79
- Paris, Palaiseau Cedex, France 10.3762/bjoc.16.79 Abstract A diversity-oriented synthesis (DOS) approach has been used to functionalize 17-ethynyl-17-hydroxysteroids through a one-pot procedure involving a ring-closing enyne metathesis (RCEYM) and a Diels–Alder reaction on the resulting diene, under
- microwave irradiations. Taking advantage of the propargyl alcohol moiety present on commercially available steroids, this classical strategy was applied to mestranol and lynestrenol, giving a collection of new complex 17-spirosteroids. Keywords: diversity-oriented strategy; 17-ethynyl-17-hydroxysteroids
- ; ring-closing enyne metathesis; spirosteroids; steroids; Introduction Diversity-oriented synthesis (DOS) is a powerful approach to access collections of structurally diverse compounds in a few synthetic steps [1][2][3][4][5][6][7]. It can be more relevant when the chemical diversity is centred on
Copper catalysis with redox-active ligands
Beilstein J. Org. Chem. 2020, 16, 858–870, doi:10.3762/bjoc.16.77

- oxidation of alcohols and representative substrate scope. Introduction of H-bonding network in the ligand coordination sphere. Well-defined isatin copper complexes. Catalyst control in the biomimetic phenol ortho-oxidation. Structural diversity accessible by direct functionalization. Copper-catalyzed
Towards triptycene functionalization and triptycene-linked porphyrin arrays
Beilstein J. Org. Chem. 2020, 16, 763–777, doi:10.3762/bjoc.16.70

- seen in Figure 9 highlight the diversity in composition of the four compounds under investigation. The triptycene-linked zinc dimer 9, previously discussed, has a Soret band at 433 nm (23095 cm−1) and two Q bands at 564 nm (17731 cm−1) and 610 nm (16393 cm−1), respectively, while the triptycene-linked
Microwave-assisted efficient and facile synthesis of tetramic acid derivatives via a one-pot post-Ugi cascade reaction
Beilstein J. Org. Chem. 2020, 16, 663–669, doi:10.3762/bjoc.16.63
- medicinal chemistry. Multicomponent reactions (MCRs) [22][23][24] are emerging tools for assembling complex molecules in a rapid and efficient manner. The four-component Ugi reaction followed by a suitable post modification of [25][26] has become a popular research field for diversity-oriented synthesis [27
- , a convertible isonitrile promoted the cyclization, but restricted the scope of structural diversity. For this reason, it is desirable to replace the amide with a better leaving group, an ester group, which will undergo the Dieckmann reaction more effectively to form a five-membered ring. It is worth
Cascade trifluoromethylthiolation and cyclization of N-[(3-aryl)propioloyl]indoles
Beilstein J. Org. Chem. 2020, 16, 657–662, doi:10.3762/bjoc.16.62
- construction of compounds with structural diversity and complexity. In 2014, Wang reported the first radical cascade trifluoromethylthiolation and cyclization of activated alkenes (Scheme 1a) [23]. Afterward, Nevado [24], Hopkinson and Glorius [25], Dagousset and Magnier [26], as well as Fu [27] applied this
Design and synthesis of diazine-based panobinostat analogues for HDAC8 inhibition
Beilstein J. Org. Chem. 2020, 16, 628–637, doi:10.3762/bjoc.16.59

- panobinostat. The reported −log(Kd) values were 8.93, 8.64 and 8.25, respectively, with panobinostat possessing a docking score of 8.47. Considering the effects of structural diversity, TOI3-rev was included in the library and computationally evaluated to determine its potential as an HDAC8 inhibitor. TOI3-rev
Opening up connectivity between documents, structures and bioactivity
Beilstein J. Org. Chem. 2020, 16, 596–606, doi:10.3762/bjoc.16.54

- from patents. However, the 157 million is exceeded by the latest public UniChem release of just under 160 million [12]. In addition, a 2019 scaffold diversity analysis stringently filtered the CAS collection down to only ≈30 million compounds with direct links to literature and patents [13]. Since its
A systematic review on silica-, carbon-, and magnetic materials-supported copper species as efficient heterogeneous nanocatalysts in “click” reactions
Beilstein J. Org. Chem. 2020, 16, 551–586, doi:10.3762/bjoc.16.52
- loss of catalytic activity. Copper anchored on functionalized magnetic nanoparticles (MNPs): efficient and recyclable catalysts for CuAAC reactions MNPs are of significant interest to chemists owing to a number of unique features, including their high structural diversity, large surface area
Architecture and synthesis of P,N-heterocyclic phosphine ligands
Beilstein J. Org. Chem. 2020, 16, 362–383, doi:10.3762/bjoc.16.35

- three metal-centered chiral elements which are conferred upon coordination with a metal [121]. Conclusion In this review, the diversity of phosphine N-heterocyclic ligands and the variety of phosphine skeletons, which includes different five- and six-membered heterocycles and different coordinating
Two antibacterial and PPARα/γ-agonistic unsaturated keto fatty acids from a coral-associated actinomycete of the genus Micrococcus
Beilstein J. Org. Chem. 2020, 16, 297–304, doi:10.3762/bjoc.16.29

- ; Micrococcus; PPAR; Introduction Marine actinobacteria are considered as a potential source for novel natural products with high structural diversity, unique biological activity, and molecular modes of action beneficial to drug development [1][2][3]. Actinobacteria in marine environments are mostly found in
Ultrasonic-assisted unusual four-component synthesis of 7-azolylamino-4,5,6,7-tetrahydroazolo[1,5-a]pyrimidines
Beilstein J. Org. Chem. 2020, 16, 281–289, doi:10.3762/bjoc.16.27
- ]pyrimidines via a cascade of elementary stages that is unusual for such transformations. Thus, we extended the molecular diversity of the compounds obtained by introducing an additional azolyl substituent to the pyrimidine ring. Alternative structures A and B for the tetrahydroazolopyrimidines 4. Molecular
Combination of multicomponent KA2 and Pauson–Khand reactions: short synthesis of spirocyclic pyrrolocyclopentenones
Beilstein J. Org. Chem. 2020, 16, 200–211, doi:10.3762/bjoc.16.23
- approach. The polyfunctional molecular scaffolds were tested on the cyclopentenone reactivity to further expand the skeletal diversity, demonstrating the utility of this combined approach in generating novel spiro compounds as starting material for the generation of chemical libraries. The chemoinformatics
- generation of high-quality small molecule collections, taking advantage of the stereochemical diversity, and of their three-dimensional shape and structural bias to develop lead compounds, specifically in the field of protein–protein interactions [3][4][5][6]. Spiranic rings such as spiroketals are present
- literature [12]. Among the synthetic approaches to improve the quality and quantity of small molecules members of chemical libraries, diversity-oriented synthesis (DOS) [13][14][15][16], has been proposed as a paradigm for developing large collections of structurally diverse small molecules in a way to
The use of isoxazoline and isoxazole scaffolding in the design of novel thiourea and amide liquid-crystalline compounds
Beilstein J. Org. Chem. 2020, 16, 175–184, doi:10.3762/bjoc.16.20
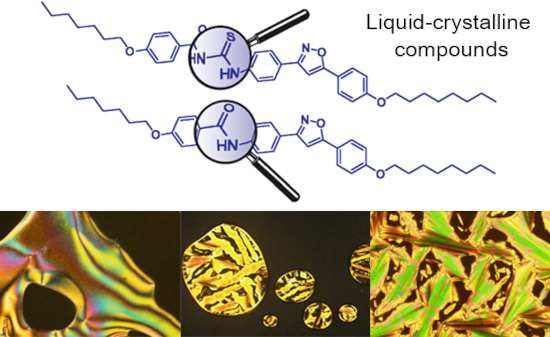
- heterocycle. Keywords: amides; isoxazole; isoxazoline; liquid crystal; thiourea; Introduction Thioureas are a structurally diversified group of organic compounds, with technological applications in different areas. The structural diversity of the thiourea moiety is linked to possibly one or both nitrogen
- differential scanning calorimetry (DSC). Results and Discussion Synthesis In order to generate molecular structural diversity in thioureas, three sets of amines carrying 5-membered isoxazolines/isoxazoles (see Scheme 1) were employed and condensed with acyl isothiocyanates, as outlined in Scheme 2. Amines 3, 4
- between the acyl isothiocyanate 16 and amines 3, 4, 8, 9, 12 and 13 yielded the title compounds 17a–c and 18a–c in 56–62% yields (17a 57%; 17b 56%; 17c 58%; 18a 61%; 18b 60%; 18c 62%). In an attempt to improve the structural diversity of thioureas, potassium cyanate was used as a nucleophile instead of
Potent hemithioindigo-based antimitotics photocontrol the microtubule cytoskeleton in cellulo
Beilstein J. Org. Chem. 2020, 16, 125–134, doi:10.3762/bjoc.16.14

- neurons; the regulation and functioning of these processes is still not satisfactorily understood [1][2][3][4]. The MT cytoskeleton is a finely tuned complex system that is highly conserved through evolution. Direct genetic modifications of tubulin that affect its functions risk causing a diversity of
- development beyond classical small molecule inhibitors; since the spatiotemporal complexity inherent to the diversity of tubulin-dependent cellular processes may finally yield to studies that can leverage high-spatiotemporal-specificity optical control to deliver cell-specific, time-reversible modulation of
Understanding the role of active site residues in CotB2 catalysis using a cluster model
Beilstein J. Org. Chem. 2020, 16, 50–59, doi:10.3762/bjoc.16.7

- comprise the largest and structurally most diverse family of natural products, currently representing over 80,000 reported structures from all kingdoms of life [3]. The largest diversity of terpenoids is reported for the plant kingdom where higher terpenes represent secondary metabolic products, which are
Terpenes
Beilstein J. Org. Chem. 2019, 15, 2966–2967, doi:10.3762/bjoc.15.292

- products. Their remarkable structural complexity and diversity is a result of a unique biosynthetic pathway invented by nature that starts with the formation of only a few acyclic precursors termed oligoprenyl diphosphates. These precursors containing multiple olefinic double bonds can be ionised to



























































































































































































































































































































