Search results
Search for "cyclohexane" in Full Text gives 294 result(s) in Beilstein Journal of Organic Chemistry. Showing first 200.
Steric “attraction”: not by dispersion alone
Beilstein J. Org. Chem. 2018, 14, 1482–1490, doi:10.3762/bjoc.14.125

- steric bulk. In the present work, we quantify this contribution for a diverse set of molecular cores, ranging from unsubstituted benzene and cyclohexane to their derivatives carrying tert-butyl, phenyl, cyclohexyl and adamantyl substituents. While the importance of electrostatic interactions in the
- electrostatic contribution to it [25]. For example, “dispersion dominates and electrostatics commands” is the ‘punch line’ of the 2017 computational study on the σ–σ, σ–π and π–π stacking interactions between benzene, cyclohexane and some of their fluorinated derivatives [26]. The authors show that while
- classes of hydrocarbons (Figure 3): 1. Non-substituted and substituted by comparatively non-bulky Me and t-Bu groups benzene and cyclohexane cores, 6a–c and 9a–c. These systems are associated with relatively small interaction energies, which do not change appreciably upon geometry relaxation. However, in
Recent advances in phosphorescent platinum complexes for organic light-emitting diodes
Beilstein J. Org. Chem. 2018, 14, 1459–1481, doi:10.3762/bjoc.14.124

- configuration ITO/MoO3 (1 nm)/TAPC (65 nm)/mCP (8 nm)/10 (pure/nondoped, 30 nm)/3TPYMB (50 nm)/LiF (1 nm)/Al (120 nm), where TAPC is 1,1-bis[(di-4-tolylamino)phenyl]cyclohexane, and serve either as the hole- or electron-transport layers. A highly efficient yellow-emitting device was obtained with EQE = 25.9
One hundred years of benzotropone chemistry
Beilstein J. Org. Chem. 2018, 14, 1120–1180, doi:10.3762/bjoc.14.98
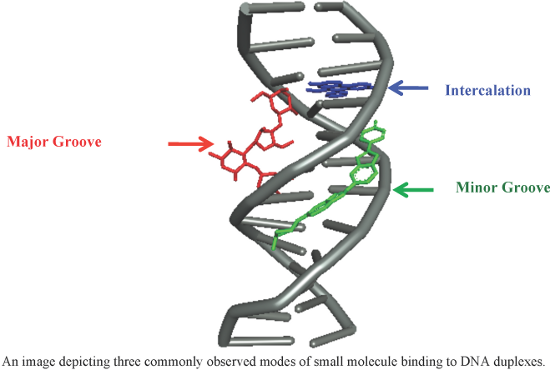
An overview of recent advances in duplex DNA recognition by small molecules
Beilstein J. Org. Chem. 2018, 14, 1051–1086, doi:10.3762/bjoc.14.93
Hypervalent iodine(III)-mediated decarboxylative acetoxylation at tertiary and benzylic carbon centers
Beilstein J. Org. Chem. 2018, 14, 1046–1050, doi:10.3762/bjoc.14.92
- products in good yields. Notably, a carboxyl group on the phenyl ring was inert toward decarboxylation under the oxidation conditions used, allowing the acetate 2e to be successfully synthesized. The acetoxylation of a cyclohexane framework was also achieved, but the yield of the product 2j was somewhat
Volatiles from the xylarialean fungus Hypoxylon invadens
Beilstein J. Org. Chem. 2018, 14, 734–746, doi:10.3762/bjoc.14.62
- acetate. The combined organic phases were dried with MgSO4 and concentrated in vacuo. The residue was purified by column chromatography on silica gel. 2-Methoxy-3-methylbenzaldehyde. Yield: 26 mg (0.17 mmol, 69%), colourless oil. TLC (cyclohexane/ethyl acetate 10:1): Rf = 0.18; 1H NMR (400 MHz, CDCl3, TMS
- solid. TLC (cyclohexane/ethyl acetate 10:1): Rf = 0.18; 1H NMR (400 MHz, CDCl3, TMS) δ 10.38 (s, 1H, CHO), 7.71 (d, 3JH,H = 7.8 Hz, 1H, CH), 6.82 (d, 3JH,H = 7.8 Hz, 1H, CH), 6.77 (s, 1H, CH), 3.90 (s, 3H, OCH3), 2.40 (s, 3H, CH3) ppm; 13C NMR (100 MHz, CDCl3, TMS) δ 189.6 (CHO), 162.0 (Cq), 147.5 (Cq
- . TLC (cyclohexane/ethyl acetate 10:1): Rf = 0.15; 1H NMR (400 MHz, CDCl3, TMS) δ 10.43 (d, 4JH,H = 0.7 Hz, 1H, CHO), 7.61 (d, 4JH,H = 2.4 Hz, 1H, CH), 7.34 (dd, 3JH,H = 8.5 Hz, 4JH,H = 2.4 Hz, 1H, CH), 6.88 (d, 3JH,H = 8.5 Hz, 1H, CH), 3.89 (s, 3H, OCH3), 2.30 (s, 3H, CH3) ppm; 13C NMR (100 MHz, CDCl3
Cobalt-catalyzed directed C–H alkenylation of pivalophenone N–H imine with alkenyl phosphates
Beilstein J. Org. Chem. 2018, 14, 709–715, doi:10.3762/bjoc.14.60
- , the NHC precursor featuring a cyclohexane backbone and 2,6-diethylphenyl groups (L1·HBr), which proved to be the optimal ligand for the C–H arylation of pivalophenone N–H imine as well as for the C–H alkenylation of N-arylimine (Scheme 1a, b) [28][29], was not particularly effective for the present
Stepwise radical cation Diels–Alder reaction via multiple pathways
Beilstein J. Org. Chem. 2018, 14, 704–708, doi:10.3762/bjoc.14.59
- adduct 3 as an approximately cis/trans = 1:4 mixture, a significant difference from the synthetic outcome mentioned above (Scheme 3). Notably, no rearrangement was observed without electricity and the vinyl substituent was found to be essential for the transformation since no cyclohexane 6 was obtained
Liquid-assisted grinding and ion pairing regulates percentage conversion and diastereoselectivity of the Wittig reaction under mechanochemical conditions
Beilstein J. Org. Chem. 2018, 14, 688–696, doi:10.3762/bjoc.14.57
- preference for larger groups to be in equatorial positions in cyclohexane rings (Scheme 2). The E selectivity is driven by both the high concentrations of reactants and a low dielectric constant of the LAG solvent (or no solvent). To evaluate which might be playing a more critical role in the selectivity
D–A–D-type orange-light emitting thermally activated delayed fluorescence (TADF) materials based on a fluorenone unit: simulation, photoluminescence and electroluminescence studies
Beilstein J. Org. Chem. 2018, 14, 672–681, doi:10.3762/bjoc.14.55

- % emitter in CBP (35 nm)/TmPyPB (55 nm)/LiF (1 nm)/Al, where 1,1-bis(4-(di-p-tolylamino)phenyl)cyclohexane (TAPC), 4,4'-bis(9H-carbazol-9-yl)biphenyl (CBP), 1,3,5-tri[(3-pyridyl)-phen-3-yl]benzene (TmPyPB) and LiF play the roles of hole transport layer, host material, electron transport layer and electron
Heterogeneous Pd catalysts as emulsifiers in Pickering emulsions for integrated multistep synthesis in flow chemistry
Beilstein J. Org. Chem. 2018, 14, 648–658, doi:10.3762/bjoc.14.52

- catalyst particles first dispersed in water using an ultrasonic probe (Jencons Vibra-Cell, 5 min, 130 W). The oils chosen are the aliphatic alkane octane (Sigma, >99%, density 0.699 g/cm3), the cyclic alkane cyclohexane (Fisher Scientific, >99%, density 0.774 g/cm3) and the aromatic oil toluene (VWR
- coalescence as well as their average droplet size depend markedly on particle concentration. As an example, the appearance of cyclohexane-in-water emulsions with time for three selected particle concentrations can be seen in Figure 4. This pattern of behaviour is followed with the other two oils also. For
- higher Sn content, surprisingly no stable emulsion was possible using cyclohexane as oil for particle concentrations in water between 0.01 and 2.0 wt % with complete phase separation occurring within 2 h. For octane, emulsion stability to coalescence increased progressively with particle concentration up
An air-stable bisboron complex: a practical bidentate Lewis acid catalyst
Beilstein J. Org. Chem. 2018, 14, 618–625, doi:10.3762/bjoc.14.48
- was washed with DCM twice to afford a yellow solid (581 mg, 51% yield, purity: 85% calculated based on 1H NMR). [Column chromatography on silica gel (EtOAc/cyclohexane 4:1) yielded pure product as an orange solid with a decreased yield (284 mg, 25% yield)]. 1H NMR (400 MHz, THF-d8) δ 9.26 (br s, 2H
- distilled off from the resulting mixture in vacuo. The residue was purified by column chromatography over SiO2 (ethyl acetate/cyclohexane 1:1) to obtain the product. Time-dependent 1H NMR spectra of the air-exposed complex B. ORTEP drawing (50% probability) of complex B. UV–vis spectrum of complex B was
Syn-selective silicon Mukaiyama-type aldol reactions of (pentafluoro-λ6-sulfanyl)acetic acid esters with aldehydes
Beilstein J. Org. Chem. 2018, 14, 373–380, doi:10.3762/bjoc.14.25
- . Among the saturated and unsaturated aliphatic aldehydes, only acetaldehyde gave the expected aldolates (58% yield, ratio 87:13, Table 2, entry 17), while cyclohexane carbaldehyde, acrolein, and crotonaldehyde did not give any aldol products (Table 2, entries 18–20). During the aldol addition, two
Gram-scale preparation of negative-type liquid crystals with a CF2CF2-carbocycle unit via an improved short-step synthetic protocol
Beilstein J. Org. Chem. 2018, 14, 148–154, doi:10.3762/bjoc.14.10
- protocol for the synthesis of multicyclic molecules having a CF2CF2-containing cyclohexadiene or cyclohexane framework in a mesogenic structure. These molecules are promising candidates for vertical alignment (VA)-mode liquid crystal (LC) display devices owing to their large negative dielectric constant
- ; tetrafluorinated cyclohexadiene; tetrafluorinated cyclohexane; Introduction Fluorine-containing organic compounds have attracted much attention in various areas, such as the medicinal, agrochemical, and materials science fields [1][2][3], due to the unique characteristics of the fluorine atom [4][5][6]. It is
- improved methodology enables us to prepare the CF2CF2-containing cyclohexadiene 1a and the corresponding cyclohexane 2c, as selected examples, on the multigram scale. Results and Discussion Improved synthetic design In order to establish an improved synthetic protocol, we initially designed a method for
Preparation and isolation of isobenzofuran
Beilstein J. Org. Chem. 2017, 13, 2659–2662, doi:10.3762/bjoc.13.263
- with a yield of 78% relative to the precursor DMIBF (7, Scheme 3). To further purify isobenzofuran (1), the benzene solution was carefully evaporated and the residue was subjected to column chromatography over silica gel (cyclohexane/ethyl acetate). We were able to isolate IBF (1) as a colorless solid
- (cyclohexane/ethyl acetate 8:2, Rf 0.58) A colorless oil was obtained. Yield: 2.13 g (14.2 mmol, 85%); 1H NMR (500 MHz, 300 K, CDCl3) δ 7.41 (d, 3J = 7.3 Hz, 1H), 7.39–7.33 (m, 2H), 7.27 (d, 3J = 7.4 Hz, 1H), 6.19 (d, 4J = 2.2 Hz, 1H), 5.22 (dd, 2J = 12.7 Hz, 4J = 2.2 Hz, 1H), 5.05 (d, 2J = 12.7 Hz, 1H), 3.44
- and then twice with water. The combined organic layers were dried over magnesium sulfate and the solvent was removed under reduced pressure. The crude product was purified by column chromatography on silica gel (cyclohexane/ethyl acetate 8:2, Rf 0.92). The solvent was carefully evaporated at 20 °C. A
Fluorination of some highly functionalized cycloalkanes: chemoselectivity and substrate dependence
Beilstein J. Org. Chem. 2017, 13, 2364–2371, doi:10.3762/bjoc.13.233
- our synthetic investigations by selecting some cyclohexane β-amino acid esters as model compounds. Thus, diol (±)-8 [23][24][25][26] derived from dihydroxylation of cis-2-aminocyclohex-4-ene carboxylic acid was treated with 1 equiv of Deoxofluor in CH2Cl2. Again, through chemodiscrimination of the
- protocol was applied to commercially available meso-compound 24 having a cyclohexane framework. However, the treatment of 24 with various amounts of Deoxofluor gave only unidentifiable products whereas the reaction in the presence of DBU, afforded ketoester (±)-25 (see also Scheme 6) in 48% yield (Scheme
- -hexane/EtOAc or n-hexane/acetone). X-ray structure of fluorohydrine derivative (±)-5. Fluorination of diol derivative (±)-1. Fluorination of diol derivative (±)-4. Fluorination of diol derivative (±)-6. Fluorination of cyclohexane-derived diol (±)-8. Proposed route for the formation of compounds (±)-10
Dialkyl dicyanofumarates and dicyanomaleates as versatile building blocks for synthetic organic chemistry and mechanistic studies
Beilstein J. Org. Chem. 2017, 13, 2235–2251, doi:10.3762/bjoc.13.221
- starting fumarate [67]. Differently substituted piperazin-2-ones can be efficiently prepared by reacting dialkyl dicyanofumarates E-1 with alkane or cycloalkane-1,2-diamines. For example, the reaction with trans-cyclohexane-1,2-diamine (72) performed in acetonitrile at room temperature for 30 min gave the
Regiodivergent condensation of 5-alkoxycarbonyl-1H-pyrrol-2,3-diones with cyclic ketazinones en route to spirocyclic scaffolds
Beilstein J. Org. Chem. 2017, 13, 2179–2185, doi:10.3762/bjoc.13.218
- the spirocyclization reaction. Our multiple attempts to carry out this transformation with the participation of enolates generated from cyclohexane-1,3-diones 8 (vinylogous carboxylates) in the presence of bases were unsuccessful. This reaction did not proceed in the presence of weak bases (such as
- elusive scaffold. Conclusion In conclusion, we discovered a new mode of cyclocondensations with “inverted” regiochemistry of addition, which involved the acid-catalyzed reaction of 5-alkoxycarbonyl-4-aroyl-1H-pyrrole-2,3-diones with cyclohexane-1,3-diones and lead to the formation of bridged 2,5
New bio-nanocomposites based on iron oxides and polysaccharides applied to oxidation and alkylation reactions
Beilstein J. Org. Chem. 2017, 13, 1982–1993, doi:10.3762/bjoc.13.194
- basic probe molecules applying a pulse chromatographic titration methodology. The catalyst used (≈0.025 g) was fixed inside a tubular stainless steel microreactor (4 mm internal diameter) by Pyrex glass wool. A cyclohexane solution of titrant (0.989 M in PY and 0.686 M in DMPY, respectively) was
NHC-catalyzed cleavage of vicinal diketones and triketones followed by insertion of enones and ynones
Beilstein J. Org. Chem. 2017, 13, 1816–1822, doi:10.3762/bjoc.13.176
- (Scheme 3). When the Z-4ai was dissolved in CDCl3 and monitored by NMR, the ratio of E/Z changed slowly to 38:62 after 21 days. In contrast, the original ratio of 40:60 was quickly attained in the presence of the NHC catalyst A. We demonstrated previously the reaction of cyclohexane-1,2-dione (5a) with
The chemistry and biology of mycolactones
Beilstein J. Org. Chem. 2017, 13, 1596–1660, doi:10.3762/bjoc.13.159
Transition-metal-free one-pot synthesis of alkynyl selenides from terminal alkynes under aerobic and sustainable conditions
Beilstein J. Org. Chem. 2017, 13, 910–918, doi:10.3762/bjoc.13.92

- (bromomethyl)cyclohexane afforded 5e in 56% yield (Table 3, entry 6). The difference in reactivity relative to 5a is probably due to steric hindrance caused by the cyclohexyl ring to the alkynyl anion attack. The basic reaction conditions were responsible for promoting elimination reactions (E2) when secondary
Synthesis of ribavirin 2’-Me-C-nucleoside analogues
Beilstein J. Org. Chem. 2017, 13, 755–761, doi:10.3762/bjoc.13.74
- at 0 °C. Then the mixture was stirred for 12 h at rt and concentrated in vacuum. The residue was purified by flash chromatography (EtOAc/cyclohexane, 3:7 to 1:0) affording the mixture of the corresponding 1-benzyl-4-(2’,3’-O-isopropylidene-2’-C-methyl-β-D-ribofuranosyl)-1,2,3-triazole-5-carboxamide
- for C19H22O5, 330.1467; found, 330.1478. The mixture of carboxamides and palladium chloride (23 mg, 0.13 mmol) in ethanol (10 mL) was hydrogenated with H2 at 4 bar for 48 h. After filtration over Celite and concentration in vacuum, the crude was purified by flash chromatography (cyclohexane/EtOAc 2:8
Fluorinated cyclohexanes: Synthesis of amine building blocks of the all-cis 2,3,5,6-tetrafluorocyclohexylamine motif
Beilstein J. Org. Chem. 2017, 13, 728–733, doi:10.3762/bjoc.13.72
- moieties were then converted to different stereoisomers of the tetrafluorocyclohexyl ring system, and then reductive hydrogenation of the nitrile delivered three amine stereoisomers. It proved necessary to place a methyl group on the cyclohexane ring in order to stabilise the compound against subsequent HF
- elimination. The two all-cis tetrafluorocyclohexyl isomers 5a and 5b constitute facially polarized cyclohexane rings, with fluorines on the electronegative face and hydrogens on the electropositive face. Keywords: all-cis tetrafluorocyclohexane motif; deoxofluorination reactions; fluorinated amines
- ; fluorinated cyclohexanes; Introduction The all-cis-2,3,5,6-tetrafluorocyclohexane 1 ring has been introduced recently as a polarized cyclohexane ring and it has been a focus of our research group to elaborate new building blocks that enable the introduction of this motif into organic discovery programmes
Membrane properties of hydroxycholesterols related to the brain cholesterol metabolism
Beilstein J. Org. Chem. 2017, 13, 720–727, doi:10.3762/bjoc.13.71
- samples The respective amounts of hydroxycholesterols and phospholipids were dissolved in chloroform at the respective molar ratios. The solvent was evaporated and the samples were re-dissolved in cyclohexane. After overnight lyophilization at high vacuum, the obtained fluffy powder was hydrated with 40





































































































































































































































































































































































































































































