Search results
Search for "solubility" in Full Text gives 870 result(s) in Beilstein Journal of Organic Chemistry. Showing first 200.
Biphenylene-containing polycyclic conjugated compounds
Beilstein J. Org. Chem. 2023, 19, 1895–1911, doi:10.3762/bjoc.19.141
- can be attributed to Clar's rule, which considers the increasing number of non-sextet rings throughout the acene series as a contributing factor (Figure 1) [5][6]. Numerous approaches have been developed to address the challenges arising from the instability and solubility issues encountered in acenes
- successfully obtained in 30% yield. In the final step, the target POA-type structure containing naphthazarin 69 was synthesized by aromatization under acidic conditions. The attempts to conduct complexation experiments using BF3·OEt2 to obtain a stable BF2 complex were unsuccessful, primarily due to solubility
- complexation step to isolate stable BF2 complex of 69, likely attributable to solubility issues encountered with substrate 69, prevented the incorporation of the acene backbone into the structure. Consequently, the anticipated red-shift in the absorption spectrum could not be observed as expected. Boron-doped
Anion–π catalysis on carbon allotropes
Beilstein J. Org. Chem. 2023, 19, 1881–1894, doi:10.3762/bjoc.19.140

- fullerenes act as anion–π catalyst without additional activating groups, usually a tethered base to inject a negative charge into the substrate directly on top of the catalytic π surface [69]. The tert-butyl esters in Bingel fullerene 31 serve only to improve solubility and are not expected to participate in
- were not fruitful, mostly due to poor solubility. Anion–π catalysis on carbon nanotubes With fullerenes confirmed as privileged scaffold for induced anion–π catalysis but higher oligomers inaccessible [12][67][80], the obvious next move was to switch to carbon nanotubes. Compared to the sixty free
Aromatic systems with two and three pyridine-2,6-dicarbazolyl-3,5-dicarbonitrile fragments as electron-transporting organic semiconductors exhibiting long-lived emissions
Beilstein J. Org. Chem. 2023, 19, 1867–1880, doi:10.3762/bjoc.19.139
- compounds 6–9 were confirmed by the spectral data and elemental analyses. Remarkably, despite a highly hindered structure, compound 9 exhibits good solubility in non-polar solvents. Photophysical properties The electronic structures of compounds 6–9 in the ground and excited states were investigated by
N-Boc-α-diazo glutarimide as efficient reagent for assembling N-heterocycle-glutarimide diads via Rh(II)-catalyzed N–H insertion reaction
Beilstein J. Org. Chem. 2023, 19, 1841–1848, doi:10.3762/bjoc.19.136
- , the proposed diazo reagent 4 possesses several drawbacks, primarily, insufficient stability during storage and comparatively low solubility in non-polar solvents, as well as complications when isolating it in a pure form. This study focuses on the development of a "diazo" technique to incorporate a
- simplified the process of isolating compound 5, as compared to the prior publication [26], and notably increased its stability. Furthermore, the solubility of the new diazo reagent in non-polar solvents, particularly DCM, was significantly improved, leading to a beneficial impact on the course of Rh(II
A novel recyclable organocatalyst for the gram-scale enantioselective synthesis of (S)-baclofen
Beilstein J. Org. Chem. 2023, 19, 1811–1824, doi:10.3762/bjoc.19.133

- the other components of the reaction mixture. The lipophilic O-alkylated gallic acid unit increases the solubility of the organocatalyst in less polar solvents, such as DCM or toluene but leads to the precipitation of the organocatalyst in polar solvents, including MeOH or MeCN. As a result, the
- addition of trans-β-nitrostyrene (12) and acetylacetone (13). Choosing the best solvent for the reaction is crucial, thus, solubility tests were carried out (Table 1). Since homogeneous catalysts usually exhibit higher activity and selectivity than their heterogeneous counterparts [27], our aim was to
- carry out the catalytic reaction homogeneously. The solubility of the lipophilic catalyst 2 was investigated in ten solvents with low polarity, including a new, bio-based polar aprotic solvent, MeSesamol [33]. The catalyst’s precipitation – which is necessary for its recycling – was tested by adding a
Selectivity control towards CO versus H2 for photo-driven CO2 reduction with a novel Co(II) catalyst
Beilstein J. Org. Chem. 2023, 19, 1766–1775, doi:10.3762/bjoc.19.129

- complex obtainable via a straightforward synthesis, with improved solubility, concerning our previous Co(II) complexes [21]. Thus, the new Co(II) complex bears two 1-benzyl-4-(quinolin-2-yl)-1H-1,2,3-triazole (BzQuTr) units, that were obtained through a copper-catalyzed alkyne–azide cycloaddition (CuAAC
Unprecedented synthesis of a 14-membered hexaazamacrocycle
Beilstein J. Org. Chem. 2023, 19, 1728–1740, doi:10.3762/bjoc.19.126
- with a noticeable increase in the macrocycle yield up to 41% (Table 1, entries 20 and 21). The extremely poor solubility of product 5 in most organic solvents allowed to purify it from all admixtures, including byproduct 6, by a single crystallization from boiling DMF. Thus, pure product 5 was prepared
- , intermediates A, C, D, and hydrazine. Figure 1 shows that the hydrazine-promoted transformation of pyrazolopyrimidine 8 into macrocycle 5 in MeOH is a thermodynamically favorable process. Moreover, the extremely low solubility of macrocycle 5 makes the dimerization of 8 even more preferable. It is noteworthy
Effects of the aldehyde-derived ring substituent on the properties of two new bioinspired trimethoxybenzoylhydrazones: methyl vs nitro groups
Beilstein J. Org. Chem. 2023, 19, 1713–1727, doi:10.3762/bjoc.19.125

- species (ROS) in vitro, which may be another mechanism through which the compound exerts its protective effects in the brain. From a drug development perspective, however, INHHQ has some pharmacological limitations, such as low solubility and certain susceptibility to hydrolysis in a water-rich medium
Quinoxaline derivatives as attractive electron-transporting materials
Beilstein J. Org. Chem. 2023, 19, 1694–1712, doi:10.3762/bjoc.19.124

- , wherein researchers substituted alkoxy chains to enhance solubility and difluoro groups to lower the highest occupied molecular orbital (HOMO) energy level. Qx-derived polymer acceptors have witnessed significant progress in recent years, driven by a contextual understanding of the major issues hindering
- morphology on Qx26 and Qx27, employing indacenodithiophene, Qx and rhodanine as donor, acceptor and end group, respectively. The incorporation of specific side chains facilitated improved thermal stability, solubility, and broad absorption spectra (300–750 nm), narrow bandgaps (1.68–1.74 eV) and PCEs in the
- electronics [51]. Hasegawa and colleagues focused on achieving air-stable n-channel conduction by synthesizing thiadiazole-fused quinoxalineimide derivatives, Qx47–Qx49. The team optimized the molecular packing and solubility by incorporating solubilizing N-substituted alkyl chains. The resulting molecules
A series of perylene diimide cathode interlayer materials for green solvent processing in conventional organic photovoltaics
Beilstein J. Org. Chem. 2023, 19, 1620–1629, doi:10.3762/bjoc.19.119

- the previously reported cyanated N-annulated perylene diimide (CN-PDIN-H), the N-position was functionalized with a benzyl and pentafluorobenzyl group to make CN-PDIN-B and CN-PDIN-FB, respectively. The materials exhibit solubility in the green solvent, ethyl acetate, and thus were processed into thin
- -PDIN-H scaffold was functionalized with a benzyl (CN-PDIN-B) or pentafluorobenzyl group (CN-PDIN-FB). Addition of the benzyl and pentafluorobenzyl groups was to enhance solubility in green solvents, while nitrile functionalization was done to further stabilize the LUMO of the compounds. These materials
- cast from a solution at a concentration of 1 mg/mL in ethyl acetate, where a concentration of 1 mg/mL was used due to CN-PDIN-FB exhibiting a maximum solubility of 1.2 mg/mL (see below). The PDIN-B film has the highest absorbance of 0.037 at its λmax of 535 nm for the 0 → 0 transition and an onset of
Cyclodextrins permeabilize DPPC liposome membranes: a focus on cholesterol content, cyclodextrin type, and concentration
Beilstein J. Org. Chem. 2023, 19, 1570–1579, doi:10.3762/bjoc.19.115

- internal cavity and a hydrophilic outer surface [1]. The common CDs are the native α-CD, β-CD, and γ-CD consisting of 6, 7, and 8 ᴅ-glucopyranose units, respectively. Due to their limited water solubility (especially β-CD), native CDs can be chemically or enzymatically modified (by e.g., alkylation
- , arylation, hydroxypropylation, amination, etherification, etc.) giving rise to synthetic CD derivatives with greater water solubility [2]. Thanks to their unique structure, CDs can offer exclusive advantages by allowing the entrapment of lipophilic molecules inside their inner cavities. This inclusion
- improves the chemical stability and aqueous solubility of the guest molecule and results in most of the cases in the formation of a water-soluble CD–guest complex [3]. Being recognized as non-toxic, biodegradable, and sustainable carriers, CDs have attracted wide interest as potential carriers in different
Complexes of resorcin[4]arene with secondary amines: synthesis, solvent influence on “in-out” structure, and theoretical calculations of non-covalent interactions
Beilstein J. Org. Chem. 2023, 19, 1525–1536, doi:10.3762/bjoc.19.109
- (cavity) and the other directional due to hydrogen bonds (hydroxy groups), leads to the formation of these complexes. Results and Discussion The addition of two equivalents of sec-amines to an ethanol solution of R[4]A results in the formation of a crystalline precipitate with limited solubility in non
- chains, complexes with 1:2 stoichiometry are formed. This stoichiometry is justified by calculations of the acidity of protons of hydroxy groups in the R[4]A molecule. These complexes are highly ionic because of their very low solubility in non-polar solvents. Complexes with higher stoichiometry are not
N-Sulfenylsuccinimide/phthalimide: an alternative sulfenylating reagent in organic transformations
Beilstein J. Org. Chem. 2023, 19, 1471–1502, doi:10.3762/bjoc.19.106

- pyrazolone derivatives with N-thiophthalimides catalyzed by 1 mol % of chiral iminophosphorane organocatalyst was carried out under mild conditions [103]. Solvent control in the procedure can affect the yield of products due to the solubility of the catalysts. Various solvents, such as acetone, ethyl acetate
Functions of enzyme domains in 2-methylisoborneol biosynthesis and enzymatic synthesis of non-natural analogs
Beilstein J. Org. Chem. 2023, 19, 1452–1459, doi:10.3762/bjoc.19.104
- studied. Several 2-methylisoborneol synthases have a proline-rich N-terminal domain of unknown function. The results presented here demonstrate that this domain leads to a reduced enzyme activity, in addition to its ability to increase long-term solubility of the protein. Furthermore, the substrate scope
- combination of domains A and B resulted in a moderately enhanced production of 1 (137 ± 6%). In other words, domain A, especially if it is covalently bound to domain B, serves as a gatekeeper that limits the production of 1 by 2MIBS. Furthermore, a difference was observed in the long-term solubility in the
- elution buffer used for protein purification through Ni2+-NTA affinity chromatography. While domain B alone showed a substantial enzyme precipitation after 12 h at 4 °C, full length 2MIBS did not (Figure 1B), suggesting that the A domain increases enzyme solubility and long-term stability. This effect
Synthesis of ether lipids: natural compounds and analogues
Beilstein J. Org. Chem. 2023, 19, 1299–1369, doi:10.3762/bjoc.19.96
- solubility of the salt. 1.2 Analogues of PAF with modification on sn-1 position PAF is characterized by a C16 or C18 saturated lipid chain at the sn-1 position. A first analogue, reported by Hirth et al. in 1983, consisted in replacing this saturated lipid chains by the mono-unsaturated oleyl ((Z)-octadec-9
Organic thermally activated delayed fluorescence material with strained benzoguanidine donor
Beilstein J. Org. Chem. 2023, 19, 1289–1298, doi:10.3762/bjoc.19.95
- deprotonation the latter with sodium hydride base. The compound shows poor solubility in most common organic solvents with moderate solubility in dichloromethane, 1,2-dichlorobenzene and dimethyl sulfoxide (DMSO). Compound 4BGIPN was characterized by high-resolution mass spectrometry (HRMS), elemental analysis
Enabling artificial photosynthesis systems with molecular recycling: A review of photo- and electrochemical methods for regenerating organic sacrificial electron donors
Beilstein J. Org. Chem. 2023, 19, 1198–1215, doi:10.3762/bjoc.19.88
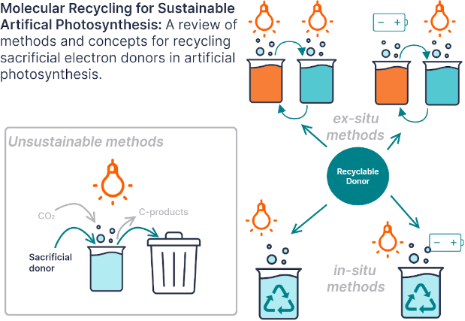
- considered: solubility in the chosen solvent, absorption spectrum, oxidation potential, and the reversibility of the sacrificial donor oxidation. As the sacrificial donor is a reactant in the photoreduction reaction of carbon dioxide, it needs to be highly soluble in the solvent used. It also needs to have a
- . TEMPO and ferrocene derivatives are used to store positive charge in RFB catholytes [59][63]. Both parent compounds undergo reversible one-electron oxidation and re-reduction but had to be modified to improve their solubility. TEMPTMA and both ferrocene derivatives have one or more ammonium groups added
- to the core TEMPO or ferrocene charge-carrying moiety. This both increases the solubility of the species and the oxidation potential in aqueous media. TEMPOL uses an alcohol group to the same effect. Redox couples for RFB catholytes are optimized for increasing the oxidation which decreases their
Exploring the role of halogen bonding in iodonium ylides: insights into unexpected reactivity and reaction control
Beilstein J. Org. Chem. 2023, 19, 1171–1190, doi:10.3762/bjoc.19.86

- include nucleophile σ-hole selectivity, and how ylide structural modifications and intramolecular halogen bonding (e.g., the ortho-effect) can improve ylide stability or solubility, and alter reaction outcomes. Keywords: electron donor–acceptor complex; halogen bonding; σ-holes; iodonium ylides; ortho
- potential to alter the physical properties (e.g., stability, solubility, UV–vis absorption) of an ylide, as well as bias a Lewis base’s σ-hole selectivity through σ-hole blocking, which represent emerging avenues for tuning an ylide’s reactivity and improving its reaction outcomes. Review 1 Halogen bonding
- -substituted iodoarene-derived iodonium ylides for improved solubility and stability [5][6][141]. ortho-Iodoanisole-derived ylide 39 was subject to a rhodium-catalyzed cyclopropanation reaction with styrene, and the results were compared with Charette’s 2009 work on 31 (Scheme 15) [142]. While both ylides were
Selective and scalable oxygenation of heteroatoms using the elements of nature: air, water, and light
Beilstein J. Org. Chem. 2023, 19, 1146–1154, doi:10.3762/bjoc.19.82
- retardants [14]. Sulfoxides are prominent pharmaceutical ingredients, while phosphine oxides improve solubility of corresponding compounds [15] and have applications in catalysis and materials science [16]. Selenoxides find use as oxygen transfer agents and donor ligands in metal catalysis and organic
Photoredox catalysis harvesting multiple photon or electrochemical energies
Beilstein J. Org. Chem. 2023, 19, 1055–1145, doi:10.3762/bjoc.19.81
The effect of dark states on the intersystem crossing and thermally activated delayed fluorescence of naphthalimide-phenothiazine dyads
Beilstein J. Org. Chem. 2023, 19, 1028–1046, doi:10.3762/bjoc.19.79

- , the fs-TA spectroscopy was not used to study their photophysical processes [47]. Femtosecond transient spectra of NI-PTZ-F-O were also studied (Figure 7c and 7d). The solubility of NI-PTZ-F-O in HEX is poor, which leads to a poor transient spectral signal (Figure S35c and S35d in Supporting
CO2 complexation with cyclodextrins
Beilstein J. Org. Chem. 2023, 19, 1021–1027, doi:10.3762/bjoc.19.78
- solubility of this complex in water. X-ray crystallography of the crystals showed a 1:1 complex of CO2 to α-CD with CO2 bound in the center of the wide, secondary rim of the α-CD cavity (Figure 2). Two of the hydroxymethyl side groups on the primary narrow rim are disordered. The disordering was modeled over
- water under a partial pressure of CO2. Using a literature value of the solubility of CO2 in water at 1 bar (34 mM) and Henrys law the more traditional aqueous binding constant Ka = [CD·CO2]/[CD][CO2] in molar terms could be calculated (Table 2). For α-cyclodextrin (1) a Kg of 0.18 bar−1 was obtained – a
- temperature dependence on solubility in water [16]. Therefore, binding of the unipolar CO2 molecule is expected to cause less water H-bond disruption in this host. Conclusion This work shows that CO2 is bound 1:1 by α-cyclodextrins and that the affinity can be improved with a smaller cavity and more
Linker, loading, and reaction scale influence automated glycan assembly
Beilstein J. Org. Chem. 2023, 19, 1015–1020, doi:10.3762/bjoc.19.77
- cleavage is probably influenced by glycan structure, solubility, and aggregation tendency [26]. Lastly, purification of the protected glycan upon cleavage could be affected by the presence or absence of a linker. L1 or L2 were conjugated to Merrifield resins with initial loadings of 0.5 mmol/g and 1.0 mmol
Asymmetric tandem conjugate addition and reaction with carbocations on acylimidazole Michael acceptors
Beilstein J. Org. Chem. 2023, 19, 881–888, doi:10.3762/bjoc.19.65
- solubility in water. Among exceptional properties belongs to easy post-transformation of acylimidazoles to common carbonyl analogs. These tunable properties allow the use of acylimidazoles in chemical biology research, which includes chemical synthesis of proteins/peptides, structure analysis, and functional
- earlier experience, we employed the tropylium ion with the more lipophilic bis(trifluoromethane)sulfinimide (NTf2) anion because of its better solubility in the applied organic solvents than the commercially available tetrafluoroborate (BF4) form [22][23][24]. The cyclic urea DMEU (1,3-dimethyl-2
A fluorescent probe for detection of Hg2+ ions constructed by tetramethyl cucurbit[6]uril and 1,2-bis(4-pyridyl)ethene
Beilstein J. Org. Chem. 2023, 19, 864–872, doi:10.3762/bjoc.19.63

- change in fluorescence intensity can be used for the recognition of Cd2+ and Zn2+ ions [35]. The symmetric tetramethyl cucurbit[6]uril (TMeQ[6]) is one of the earliest characterized modified cucurbit[6]urils [36]. Compared with Q[6], tetramethyl cucurbit[6]uril (TMeQ[6]) has good solubility in water




















































































































































































































































































































































































































































































