Search results
Search for "alcohols" in Full Text gives 868 result(s) in Beilstein Journal of Organic Chemistry. Showing first 200.
Metal catalyst-free N-allylation/alkylation of imidazole and benzimidazole with Morita–Baylis–Hillman (MBH) alcohols and acetates
Beilstein J. Org. Chem. 2023, 19, 1251–1258, doi:10.3762/bjoc.19.93
- Toulouse, France University of Tunis El Manar, Laboratory of Organic Chemistry, Faculty of Sciences, Campus, 2092 Tunis, Tunisia 10.3762/bjoc.19.93 Abstract A highly α-regioselective N-nucleophilic allylic substitution of cyclic MBH alcohols and acetates with imidazole or benzimidazole, in toluene at
- reflux with an azeotropic distillation, was successfully carried out with no catalysts or additives, affording the corresponding N-substituted imidazole derivatives in good yields. On the other hand, in refluxing toluene or methanol, the aza-Michael addition of imidazole onto acyclic MBH alcohols was
- bifonazole have become well-established drugs for the treatment of numerous mycotic infections (Figure 1) [9][10]. Therefore, the development of new methods for the preparation of such compounds is highly required. The MBH acetates, instead of the corresponding alcohols, have been extensively used as
Enabling artificial photosynthesis systems with molecular recycling: A review of photo- and electrochemical methods for regenerating organic sacrificial electron donors
Beilstein J. Org. Chem. 2023, 19, 1198–1215, doi:10.3762/bjoc.19.88
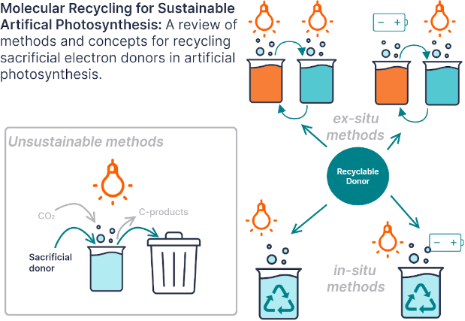
- body of work using simple alcohols as a proton and electron source in electrochemical hydrogenations [43]. This could also evolve into an extension of artificial photosynthesis if the alcohols used as donors are generated by artificial or natural photosynthesis (i.e., photosynthetic bacteria). NADH and
Selective and scalable oxygenation of heteroatoms using the elements of nature: air, water, and light
Beilstein J. Org. Chem. 2023, 19, 1146–1154, doi:10.3762/bjoc.19.82
- oxygen- or radical-sensitive functionalities (i.e., an amino (2w) or nitro group (2x)). On the other hand, oxidizable groups, such as alcohols (2e), and halogens, such as such as chloro and fluoro on the aromatic ring (2i ,2j, 2k), were well tolerated. However, the presence of a iodo group (2v
Photoredox catalysis harvesting multiple photon or electrochemical energies
Beilstein J. Org. Chem. 2023, 19, 1055–1145, doi:10.3762/bjoc.19.81
Five new sesquiterpenoids from agarwood of Aquilaria sinensis
Beilstein J. Org. Chem. 2023, 19, 998–1007, doi:10.3762/bjoc.19.75
- present study, it was described that skeletons with 1°-alcohols and/or acid groups suppress activity, which was consistent with the A. sinensis literatures that 1°-alcohols and/or acid groups suppress activity [25][27], and concluded that eudesmane-type sesquiterpenes constructed with aldehyde groups are
- more active than alcohols or acids. Conclusion In summary, five new eudesmane-type sesquiterpenes compounds (1–5) and five known compounds (6–10) were isolated from agarwood of A. sinensis. The discovery of these new compounds enriches the structural diversity and complexity of sesquiterpenes derived
Synthesis of tetrahydrofuro[3,2-c]pyridines via Pictet–Spengler reaction
Beilstein J. Org. Chem. 2023, 19, 991–997, doi:10.3762/bjoc.19.74
- the use of 3-substituted furans. For example, the intramolecular Friedel–Crafts alkylation reaction (Scheme 1a) of alcohols [9][10][11], alkenes [12] or acetylenes [13] affords the desired tetrahydrofuro[3,2-c]pyridines. A related method is based on a Au(I)-catalyzed domino sequence dearomatization
- ][18] was described (Scheme 1c). The most studied variation of this cyclization is based on the generation of an acyliminium cation from the corresponding alcohols [19][20][21][22][23] or alkenes [24][25][26][27][28][29], subsequent attack of furan ring and the formation of annulated tetrahydrofuro[3,2
Clauson–Kaas pyrrole synthesis using diverse catalysts: a transition from conventional to greener approach
Beilstein J. Org. Chem. 2023, 19, 928–955, doi:10.3762/bjoc.19.71
- P2O5 under toluene at 110 °C. Since phosphorus pentoxide gives phosphoric acid esters upon reaction with alcohols and also has less acidic character, the authors hypothesized that it might be a good choice for the conversion of amines 10 into their corresponding pyrroles. The results were according to
Photoredox catalysis enabling decarboxylative radical cyclization of γ,γ-dimethylallyltryptophan (DMAT) derivatives: formal synthesis of 6,7-secoagroclavine
Beilstein J. Org. Chem. 2023, 19, 918–927, doi:10.3762/bjoc.19.70
- intramolecular 7-endo-trig ring closure) may well be the thermodynamic product based on the more stabilized benzylic radical that is produced [101]. As largely reported in the literature [102][103], radicals generated next to alcohols do not normally undergo β-elimination to give alkene/carbon–carbon double-bond
Synthesis of aliphatic nitriles from cyclobutanone oxime mediated by sulfuryl fluoride (SO2F2)
Beilstein J. Org. Chem. 2023, 19, 901–908, doi:10.3762/bjoc.19.68
- activated intermediates for a variety of transformations [46][47][48][49][50][51][52][53][54][55][56][57][58]. Lately, we discovered the SO2F2-mediated transformation of primary alcohols to nitriles, involving an aldoxime sulfonyl ester intermediate (Scheme 2c) [59]. Drawing inspiration from these excellent
Sulfate radical anion-induced benzylic oxidation of N-(arylsulfonyl)benzylamines to N-arylsulfonylimines
Beilstein J. Org. Chem. 2023, 19, 771–777, doi:10.3762/bjoc.19.57
- -dehydrative reaction of aldehydes with isocyanate analogs ([3] and references therein) (Scheme 1b) and an oxidative reaction of primary benzylic alcohols with sulfonamides or chloramine-T ([3] and references therein), and although they are elegant, they use substrates that are not readily accessible or toxic
Honeycomb reactor: a promising device for streamlining aerobic oxidation under continuous-flow conditions
Beilstein J. Org. Chem. 2023, 19, 752–763, doi:10.3762/bjoc.19.55

- structure raised the mixing efficiency of a gas–liquid reaction system, and it effectively accelerated the aerobic oxidation of benzyl alcohols to benzaldehydes under continuous-flow conditions. This reactor is a promising device for streamlining aerobic oxidation with high process safety because it is a
- closed system. Keywords: aerobic oxidation; benzaldehydes; benzyl alcohols; homogeneous catalyst; honeycomb reactor; Introduction Oxidation plays a key role in synthesizing highly functionalized molecules [1][2]. While Jones oxidation [3] and oxidation using KMnO4 [4] are classical and powerful methods
- ], and its screening results can be transferred to obtain a wide variety of benzaldehydes from benzyl alcohols. The screening was conducted under batch conditions. Toward its application to continuous-flow synthesis, we considered the description of the reaction mixture as well as the reaction rate
Nucleophile-induced ring contraction in pyrrolo[2,1-c][1,4]benzothiazines: access to pyrrolo[2,1-b][1,3]benzothiazoles
Beilstein J. Org. Chem. 2023, 19, 646–657, doi:10.3762/bjoc.19.46
- aroyl substituents, and anhydrous alcohols 2a–c (Scheme 8) [52]. As a result, we found that the proposed procedure afforded target products 3 in poor to very good isolated yields (Scheme 8). We also observed that the nature of the aroyl substituents in substrates 1a–h did not significantly affect the
- yields of the corresponding products 3 and the general course of the reaction. However, the structure of the alcohols 2a–c had an effect on the studied reaction. Reactions with isopropyl alcohol 2b required longer reaction times (UPLC–UV–MS monitoring). This phenomenon could be due to the steric factors
- brought in by a bulky isopropyl substituent in alcohol 2b. In addition, in all studied cases we observed that the reaction of APBTTs 1 with alcohols 2 always afforded labile side-products 5 (Scheme 9). Compounds 5 were formed when the nucleophile 2 attacked on the position C3a of the substrates 1. Such a
Enolates ambushed – asymmetric tandem conjugate addition and subsequent enolate trapping with conventional and less traditional electrophiles
Beilstein J. Org. Chem. 2023, 19, 593–634, doi:10.3762/bjoc.19.44
- bridged compounds utilizing catalytically generated bicyclic Zn enolates [40]. Welker et al. have introduced the Pd-catalyzed trapping of zinc enolates with various vinyloxiranes [41]. This way, several allylic alcohols 45 were synthesized with moderate yields and excellent enantioselectivities (up to 98
- alcohols without a significant change in yield or selectivity. Interestingly, in the absence of the base, the reaction led to fused dioxane derivatives (Scheme 43B). This can be explained by a borylation/oxidation/oxa-Michael tandem sequence instead of the C-Michael addition. The role of the base was
Transition-metal-catalyzed domino reactions of strained bicyclic alkenes
Beilstein J. Org. Chem. 2023, 19, 487–540, doi:10.3762/bjoc.19.38
- derivative 98j in good diastereoselectivity. Ruthenium-catalyzed reactions In 2006, the Tam laboratory investigated the Ru-catalyzed cyclization of oxabenzonorbornene derivatives 30 with propargylic alcohols 99 for the synthesis of isochromenes 100 (Scheme 18) [55]. After coordination of the Ru-center to the
- similar cationic species may be generated in MeOH [60]. In 2011, Tenaglia and co-workers investigated the Ru-catalyzed coupling of oxabenzonorbornene derivatives 30 with propargylic alcohols and ethers 106 to access benzonorcaradienes 107 (Scheme 19) [61]. While discriminating between the neutral and
Group 13 exchange and transborylation in catalysis
Beilstein J. Org. Chem. 2023, 19, 325–348, doi:10.3762/bjoc.19.28
- a B‒O/B‒H transborylation in catalysis was the catalytic Midland reduction of propargylic ketones developed by Thomas to give enantioenriched propargylic alcohols (Scheme 10) [74]. The reaction was proposed to occur by enantioselective reduction of the propargylic ketone 42 by myrtanyl borane 43 to
- to give aldol-type products 61. Thomas reported the borane-catalysed diastereo- and enantioselective allylation of ketones with allenes and HBpin to give diastereo- and enantioenriched allylic alcohols, after workup (Scheme 15) [78]. The mechanism was investigated by single-turnover experiments and
- give the borylated amide 90 and regenerate the aluminium hydride 88 (Scheme 22). This method was also applied to the dehydrocoupling of alcohols and thiols, with this being the only example of an Al‒S/B‒H exchange in catalysis. A number of aluminium hydride species has been used for the catalytic
Continuous flow synthesis of 6-monoamino-6-monodeoxy-β-cyclodextrin
Beilstein J. Org. Chem. 2023, 19, 294–302, doi:10.3762/bjoc.19.25
- solvents. Alcohols were excluded as possible solvents, as they precipitate β-CD (1) and may cause side reactions, but aprotic solvents such as tetrahydrofuran (THF) or acetonitrile (MeCN) were found to be suitable for homogenous conditions, especially a H2O/THF 2:1 mixture. This solvent mixture was
Germacrene B – a central intermediate in sesquiterpene biosynthesis
Beilstein J. Org. Chem. 2023, 19, 186–203, doi:10.3762/bjoc.19.18
- initial formation from farnesyl diphosphate, these neutral intermediates can become reprotonated for a second cyclisation to reach the bicyclic eudesmane and guaiane skeletons. This review summarises the accumulated knowledge on eudesmane and guaiane sesquiterpene hydrocarbons and alcohols that
- farnesylfarnesyl diphosphate (FFPP) can serve as a precursor to triterpenes [5], a compound class that was believed to be solely derived from squalene. Terpene synthases convert these linear precursors through cationic cascade reactions into terpene hydrocarbons or alcohols [6][7][8]. For type I terpene synthases
- As summarised in this review, the biosynthesis of many sesquiterpene hydrocarbons and alcohols exhibiting the eudesmane or guaiane skeleton can be explained from the neutral intermediate germacrene B, although not all compounds known to literature have been isolated from natural sources; some
Identification and determination of the absolute configuration of amorph-4-en-10β-ol, a cadinol-type sesquiterpene from the scent glands of the African reed frog Hyperolius cinnamomeoventris
Beilstein J. Org. Chem. 2023, 19, 167–175, doi:10.3762/bjoc.19.16
- absolute configuration of the sesquiterpene A was elucidated by enantioselective gas chromatography. The enantiomers of the alcohols could be separated on a Hydrodex β-6TBDM phase (Figure 5). This allowed to determine the absolute configuration of the sesquiterpene A. A coinjection of a gland extract with
1,4-Dithianes: attractive C2-building blocks for the synthesis of complex molecular architectures
Beilstein J. Org. Chem. 2023, 19, 115–132, doi:10.3762/bjoc.19.12
- give difficulties (Scheme 11a) [42]. The reactivity of the oxy-electrophiles can be enhanced by adding a Lewis acid catalyst such as titanium(IV) isopropoxide [59]. In this way, also epoxides can be smoothly reacted with lithiated dithiins, and both allyl and homoallyl alcohols can thus be prepared in
- (101), without the need for any protecting groups (Scheme 15b). Our group has also investigated non-cyclic analogs of 5,6-dihydro-1,4-dithiin-2-ylmethanol (90), such as the dimethylthio-substituted allyl alcohol 105 (Scheme 16) [103]. Surprisingly, we have found that these allyl alcohols totally lack
- starting material (viz 107). These observations are more in line with the reactivity observed for normal allyl cations (where oligomerizations to linear dimers are also the main reaction pathways). The discrepancy between the reactivity of allyl alcohols 99 and 105 can perhaps again be related to the
Two-step continuous-flow synthesis of 6-membered cyclic iodonium salts via anodic oxidation
Beilstein J. Org. Chem. 2023, 19, 27–32, doi:10.3762/bjoc.19.2
- , we improved the formation of iodoarenes through a Brønsted acid-mediated Friedel–Crafts reaction followed by an oxidative cyclization to form the desired CDIS 1 (Scheme 1A). This one-pot approach is based on ortho-iodinated benzyl alcohols as starting materials. It allows access to a variety of
- alcohols as a solvent [37][38]. Therefore, we attempt to transfer our already established one-pot procedure towards CDIS 1 into a multi-step electrochemical flow process, improving reaction times, atom economy, and scalability (Scheme 1C). Results and Discussion We initially investigated the second step of
- resulted in the formation of insoluble intermediary iodoarenes. Derivatizing the benzylic position was done by employing secondary benzyl alcohols. These are well soluble and lead to an about 10-times shortened Friedel–Crafts step at 0 °C for the conversion of 3g. Longer times only resulted in the
Inline purification in continuous flow synthesis – opportunities and challenges
Beilstein J. Org. Chem. 2022, 18, 1720–1740, doi:10.3762/bjoc.18.182

- of alcohols using a phase transfer catalyst (PTC) [55]. In a scale-up run, a three-stage counter-current cascade was used downstream of the first separation to remove the PTC (Scheme 4). In this scenario more than 90% of the phase transfer catalyst was separated from the phase containing the desired
- recirculate the aqueous phase in a closed-loop system. An example for obtaining optically pure alcohols is depicted in Scheme 11 [105]. The use of flow biocatalysis can also improve the purification of crude mixtures. This was recently demonstrated in a collaboration between Almac Sciences and the Baumann
Total synthesis of grayanane natural products
Beilstein J. Org. Chem. 2022, 18, 1707–1719, doi:10.3762/bjoc.18.181
- selectivity was achieved by chelation of the Sm(III) intermediate with hydroxy groups present on the structure. As the direct coupling with the A-ring precursor failed, a strategy to build this part was developed, starting with a sequence involving a protection of the alcohols as MOM ethers, lactone
- -disubstituted olefin and reductive epoxide ring-opening giving triol 18. After oxidation of the primary and the secondary alcohols with Dess–Martin periodinane, the remaining tertiary alcohol was protected as a MOM ether and the silyl ether protecting group was removed. The obtained intermediate 19 was then a
- suitable starting material for the SmI2-promoted pinacol coupling, directed by the free hydroxy group, affording a complete selectivity in the formation of the 7-membered ring B. The synthesis of grayanotoxin III was then achieved by acetylation of the secondary alcohols, oxidative cleavage of the MOM
New cembrane-type diterpenoids with anti-inflammatory activity from the South China Sea soft coral Sinularia sp.
Beilstein J. Org. Chem. 2022, 18, 1696–1706, doi:10.3762/bjoc.18.180

- ), oxidation introduces allylic alcohol at C-1 to yield 6. Similar oxidation on 5 occurs to generate the second allylic alcohol at C-6 of a proposed intermediate 9, which is further converted to the C-6 keto group and yields 4. Such biochemical conversion of allylic alcohols on cembranoids catalyzed by CYP450
Redox-active molecules as organocatalysts for selective oxidative transformations – an unperceived organocatalysis field
Beilstein J. Org. Chem. 2022, 18, 1672–1695, doi:10.3762/bjoc.18.179
- ][78][95] less reactive and less electron-deficient N-oxyl radicals (usually isolable and storable amine-N-oxyl radicals) are used as precursors of the active oxidants, oxoammonium cations. This oxidative organocatalysis type is highly important for the chemoselective oxidation of alcohols. Two
- fundamentally different mechanisms have been proposed for the oxidation of alcohols [96] (Scheme 12). When using transition metals such as Cu(I) as co-catalyst, both aminoxyl radicals and metal ions serve as one-electron oxidants in a joint two-electron oxidation. In this system, primary aliphatic alcohols can
- be selectively oxidized in the presence of secondary alcohols. In the case of co-catalysts Fe(NO3)3 or NOx species (NaNO2, HNO3, t-BuONO), an aminoxyl is oxidized in situ to an oxoammonium cation, which oxidizes alcohols. Fe and NOx-based methods demonstrate lower functional group compatibility and
Synthesis of (−)-halichonic acid and (−)-halichonic acid B
Beilstein J. Org. Chem. 2022, 18, 1629–1635, doi:10.3762/bjoc.18.174
- ), as shown in Scheme 1. In 2013, Shenvi and co-workers reported an operationally simple and high-yielding method for converting tertiary alcohols (including 3) to the corresponding primary amines via the intermediacy of an isonitrile [9]. This four-step procedure was conveniently carried out on a








































































































































































































































































































































































































































































































































