Search results
Search for "amino acids" in Full Text gives 543 result(s) in Beilstein Journal of Organic Chemistry. Showing first 200.
Photocatalysis with organic dyes: facile access to reactive intermediates for synthesis
- Stephanie G. E. Amos,
- Marion Garreau,
- Luca Buzzetti and
- Jerome Waser
Beilstein J. Org. Chem. 2020, 16, 1163–1187, doi:10.3762/bjoc.16.103
- . Other organic dyes, including several acridinium salts, have been successfully applied in organophotocatalytic decarboxylation protocols. For example, rhodamine 6G (OD14, E(PC+*/PC) ≈ 1.2 V) [42] was used for the photocatalytic decarboxylative azidation of cyclic amino acids and rose bengal (OD15) [43

Graphical Abstract

Figure 1: Selected examples of organic dyes. Mes-Acr+: 9-mesityl-10-methylacridinium, DCA: 9,10-dicyanoanthra...

Scheme 1: Activation modes in photocatalysis.

Scheme 2: Main strategies for the formation of C(sp3) radicals used in organophotocatalysis.

Scheme 3: Illustrative example for the photocatalytic oxidative generation of radicals from carboxylic acids:...

Scheme 4: Illustrative example for the photocatalytic reductive generation of C(sp3) radicals from redoxactiv...

Figure 2: Common substrates for the photocatalytic oxidative generation of C(sp3) radicals.

Scheme 5: Illustrative example for the photocatalytic oxidative generation of radicals from dihydropyridines ...

Scheme 6: Illustrative example for the photocatalytic oxidative generation of C(sp3) radicals from trifluorob...

Scheme 7: Illustrative example for the photocatalytic reductive generation of C(sp3) radicals from benzylic h...

Scheme 8: Illustrative example for the photocatalytic generation of C(sp3) radicals via direct HAT: the cross...

Scheme 9: Illustrative example for the photocatalytic generation of C(sp3) radicals via indirect HAT: the deu...

Scheme 10: Selected precursors for the generation of aryl radicals using organophotocatalysis.

Scheme 11: Illustrative example for the photocatalytic reductive generation of aryl radicals from aryl diazoni...

Scheme 12: Illustrative examples for the photocatalytic reductive generation of aryl radicals from haloarenes:...

Scheme 13: Illustrative example for the photocatalytic reductive generation of aryl radicals from aryl halides...

Scheme 14: Illustrative example for the photocatalytic reductive generation of aryl radicals from arylsulfonyl...

Scheme 15: Illustrative example for the reductive photocatalytic generation of aryl radicals from triaryl sulf...

Scheme 16: Main strategies towards acyl radicals used in organophotocatalysis.

Scheme 17: Illustrative example for the decarboxylative photocatalytic generation of acyl radicals from α-keto...

Scheme 18: Illustrative example for the oxidative photocatalytic generation of acyl radicals from acyl silanes...

Scheme 19: Illustrative example for the oxidative photocatalytic generation of carbamoyl radicals from 4-carba...

Scheme 20: Illustrative example of the photocatalytic HAT approach for the generation of acyl radicals from al...

Scheme 21: General reactivity of a) radical cations; b) radical anions; c) the main strategies towards aryl an...

Scheme 22: Illustrative example for the oxidative photocatalytic generation of alkene radical cations from alk...

Scheme 23: Illustrative example for the reductive photocatalytic generation of an alkene radical anion from al...

Figure 3: Structure of C–X radical anions and their neutral derivatives.

Scheme 24: Illustrative example for the photocatalytic reduction of imines and the generation of an α-amino C(...

Scheme 25: Illustrative example for the oxidative photocatalytic generation of aryl radical cations from arene...

Scheme 26: NCR classifications and generation.

Scheme 27: Illustrative example for the photocatalytic reductive generation of iminyl radicals from O-aryl oxi...

Scheme 28: Illustrative example for the photocatalytic oxidative generation of iminyl radicals from α-N-oxy ac...

Scheme 29: Illustrative example for the photocatalytic oxidative generation of iminyl radicals via an N–H bond...

Scheme 30: Illustrative example for the photocatalytic oxidative generation of amidyl radicals from Weinreb am...

Scheme 31: Illustrative example for the photocatalytic reductive generation of amidyl radicals from hydroxylam...

Scheme 32: Illustrative example for the photocatalytic reductive generation of amidyl radicals from N-aminopyr...

Scheme 33: Illustrative example for the photocatalytic oxidative generation of amidyl radicals from α-amido-ox...

Scheme 34: Illustrative example for the photocatalytic oxidative generation of aminium radicals: the N-aryltet...

Scheme 35: Illustrative example for the photocatalytic oxidative generation of nitrogen-centered radical catio...

Scheme 36: Illustrative example for the photocatalytic oxidative generation of nitrogen-centered radical catio...

Scheme 37: Illustrative example for the photocatalytic oxidative generation of hydrazonyl radical from hydrazo...

Scheme 38: Generation of O-radicals.

Scheme 39: Illustrative examples for the photocatalytic generation of O-radicals from N-alkoxypyridinium salts...

Scheme 40: Illustrative examples for the photocatalytic generation of O-radicals from alkyl hydroperoxides: th...

Scheme 41: Illustrative example for the oxidative photocatalytic generation of thiyl radicals from thiols: the...

Scheme 42: Main strategies and reagents for the generation of sulfonyl radicals used in organophotocatalysis.

Scheme 43: Illustrative example for the reductive photocatalytic generation of sulfonyl radicals from arylsulf...

Scheme 44: Illustrative example of a Cl atom abstraction strategy for the photocatalytic generation of sulfamo...

Scheme 45: Illustrative example for the oxidative photocatalytic generation of sulfonyl radicals from sulfinic...

Scheme 46: Illustrative example for the photocatalytic generation of electronically excited triplet states: th...

Scheme 47: Illustrative example for the photocatalytic generation of electronically excited triplet states: th...
Synthesis of esters of diaminotruxillic bis-amino acids by Pd-mediated photocycloaddition of analogs of the Kaede protein chromophore
- Esteban P. Urriolabeitia,
- Pablo Sánchez,
- Alexandra Pop,
- Cristian Silvestru,
- Eduardo Laga,
- Ana I. Jiménez and
- Carlos Cativiela
Beilstein J. Org. Chem. 2020, 16, 1111–1123, doi:10.3762/bjoc.16.98
- complexes 4. Treatment of 4 with CO in a MeOH/NCMe mixture promoted the methoxycarbonylation of the palladated carbon and the release of the corresponding ortho-functionalized 1,3-diaminotruxillic bis-amino esters 5 as single isomers. Keywords: amino acids; C–H activation; Kaede protein; oxazolones
- ortho-position of the 2,4-aryl rings in the resulting bis-amino acids can be functionalized [30]. Substrates of the (Z)-4-arylidene-2((E)-styryl)-5(4H)-oxazolone type, shown in Figure 2b, are interesting starting materials for the synthesis of new 1,3-diaminotruxillic derivatives. These azlactones are
- findings were also confirmed by analysis of the 13C NMR spectra. Conclusion The synthesis of cyclobutane 1,3-diaminotruxillic bis-amino acids has been achieved with full stereoselectivity (ε-isomer) starting from polyfunctional oxazolones 2 derived from the chromophore of the Kaede protein in three steps

Graphical Abstract

Figure 1: (a) General scheme for truxillic acid derivatives; (b) general scheme for symmetric 1,3-diaminotrux...

Figure 2: (a) (Z)-4-Arylidene-2-aryl-5(4H)-oxazolones used for the synthesis of 1,3-diaminotruxillic derivati...

Figure 3: (Z)-4-Arylidene-2((E)-styryl)-5(4H)-oxazolones 2a–j used in this work and overall reaction scheme.

Figure 4: Molecular drawing of the oxazolone 2c.

Scheme 1: Ortho-palladation of oxazolones 2 by treatment with Pd(OAc)2 and different structures obtained for ...

Scheme 2: [2 + 2] Photocycloaddition of cyclopalladated complexes 3 in solution to give the dinuclear cyclobu...

Figure 5: Molecular drawing of cyclobutane ortho-palladated 4a. Ellipsoids are shown at the 50% probability l...

Scheme 3: Release of the 1,3-diaminotruxillic bis-amino ester derivatives 5 by methoxycarbonylation of the Pd...
Fluorinated phenylalanines: synthesis and pharmaceutical applications
- Laila F. Awad and
- Mohammed Salah Ayoup
Beilstein J. Org. Chem. 2020, 16, 1022–1050, doi:10.3762/bjoc.16.91

- . Incorporation of fluorinated aromatic amino acids into proteins increases their catabolic stability especially in therapeutic proteins and peptide-based vaccines. This review seeks to summarize the different synthetic approaches in the literature to prepare ᴅ- or ʟ-fluorinated phenylalanines and their
- , geometry, conformation, reactivity, and moreover the bioavailability of the analogue [1]. Fluorinated amino acids (FAAs) have considerable industrial and pharmaceutical potential [2]. Also, they have played an important role as enzyme inhibitors as well as therapeutic agents [3][4]. Moreover, they modulate
- membrane permeability and reactivity [11][12][13][14]. The effect of peptide structure and stability has been found to depend on the position and number of fluorine atoms within the amino acid chains [15][16][17]. Incorporation of fluorinated aromatic amino acids into proteins can increase their shelf life

Graphical Abstract

Figure 1: Categories I–V of fluorinated phenylalanines.

Scheme 1: Synthesis of fluorinated phenylalanines via Jackson’s method.

Scheme 2: Synthesis of all-cis-tetrafluorocyclohexylphenylalanines.

Scheme 3: Synthesis of ʟ-4-[sulfono(difluoromethyl)]phenylalanine (nPt: neopentyl, TCE: trichloroethyl).

Scheme 4: Synthesis of ʟ-4-[sulfono(difluoromethyl)]phenylalanine derivatives 17.

Scheme 5: Synthesis of fluorinated Phe analogues from Cbz-protected aminomalonates.

Scheme 6: Synthesis of tetrafluorophenylalanine analogues via the 3-methyl-4-imidazolidinone auxiliary 25.

Scheme 7: Synthesis of tetrafluoro-Phe derivatives via chiral auxiliary 31.

Scheme 8: Synthesis of 2,5-difluoro-Phe and 2,4,5-trifluoro-Phe via Schöllkopf reagent 34.

Scheme 9: Synthesis of 2-fluoro- and 2,6-difluoro Fmoc-Phe derivatives starting from chiral auxiliary 39.

Scheme 10: Synthesis of 2-[18F]FPhe via chiral auxiliary 43.

Scheme 11: Synthesis of FPhe 49a via photooxidative cyanation.

Scheme 12: Synthesis of FPhe derivatives via Erlenmeyer azalactone synthesis.

Scheme 13: Synthesis of (R)- and (S)-2,5-difluoro Phe via the azalactone method.

Scheme 14: Synthesis of 3-bromo-4-fluoro-(S)-Phe (65).

Scheme 15: Synthesis of [18F]FPhe via radiofluorination of phenylalanine with [18F]F2 or [18F]AcOF.

Scheme 16: Synthesis of 4-borono-2-[18F]FPhe.

Scheme 17: Synthesis of protected 4-[18F]FPhe via arylstannane derivatives.

Scheme 18: Synthesis of FPhe derivatives via intermediate imine formation.

Scheme 19: Synthesis of FPhe derivatives via Knoevenagel condensation.

Scheme 20: Synthesis of FPhe derivatives 88a,b from aspartic acid derivatives.

Scheme 21: Synthesis of 2-(2-fluoroethyl)phenylalanine derivatives 93 and 95.

Scheme 22: Synthesis of FPhe derivatives via Zn2+ complexes.

Scheme 23: Synthesis of FPhe derivatives via Ni2+ complexes.

Scheme 24: Synthesis of 3,4,5-trifluorophenylalanine hydrochloride (109).

Scheme 25: Synthesis of FPhe derivatives via phenylalanine aminomutase (PAM).

Scheme 26: Synthesis of (R)-2,5-difluorophenylalanine 115.

Scheme 27: Synthesis of β-fluorophenylalanine via 2-amino-1,3-diol derivatives.

Scheme 28: Synthesis of β-fluorophenylalanine derivatives via the oxazolidinone chiral auxiliary 122.

Scheme 29: Synthesis of β-fluorophenylalanine from pyruvate hemiketal 130.

Scheme 30: Synthesis of β-fluorophenylalanine (136) via fluorination of β-hydroxyphenylalanine (137).

Scheme 31: Synthesis of β-fluorophenylalanine from aziridine derivatives.

Scheme 32: Synthesis of β-fluorophenylalanine 136 via direct fluorination of pyruvate esters.

Scheme 33: Synthesis of β-fluorophenylalanine via fluorination of ethyl 3-phenylpyruvate enol using DAST.

Scheme 34: Synthesis of β-fluorophenylalanine derivatives using photosensitizer TCB.

Scheme 35: Synthesis of β-fluorophenylalanine derivatives using Selectflour and dibenzosuberenone.

Scheme 36: Synthesis of protected β-fluorophenylalanine via aziridinium intermediate 150.

Scheme 37: Synthesis of β-fluorophenylalanine derivatives via fluorination of α-hydroxy-β-aminophenylalanine d...

Scheme 38: Synthesis of β-fluorophenylalanine derivatives from α- or β-hydroxy esters 152a and 155.

Scheme 39: Synthesis of a series of β-fluoro-Phe derivatives via Pd-catalyzed direct fluorination of β-methyle...

Scheme 40: Synthesis of series of β-fluorinated Phe derivatives using quinoline-based ligand 162 in the Pd-cat...

Scheme 41: Synthesis of β,β-difluorophenylalanine derivatives from 2,2-difluoroacetaldehyde derivatives 164a,b....

Scheme 42: Synthesis of β,β-difluorophenylalanine derivatives via an imine chiral auxiliary.

Scheme 43: Synthesis of α-fluorophenylalanine derivatives via direct fluorination of protected Phe 174.

Figure 2: Structures of PET radiotracers of 18FPhe derivatives.

Figure 3: Structures of melfufen (179) and melphalan (180) anticancer drugs.

Figure 4: Structure of gastrazole (JB95008, 181), a CCK2 receptor antagonist.

Figure 5: Dual CCK1/CCK2 antagonist 182.

Figure 6: Structure of sitagliptin (183), an antidiabetic drug.

Figure 7: Structure of retaglpitin (184) and antidiabetic drug.

Figure 8: Structure of evogliptin (185), an antidiabetic drug.

Figure 9: Structure of LY2497282 (186) a DPP-4 inhibitor for the treatment of type II diabetes.

Figure 10: Structure of ulimorelin (187).

Figure 11: Structure of GLP1R (188).

Figure 12: Structures of Nav1.7 blockers 189 and 190.
Fabclavine diversity in Xenorhabdus bacteria
- Sebastian L. Wenski,
- Harun Cimen,
- Natalie Berghaus,
- Sebastian W. Fuchs,
- Selcuk Hazir and
- Helge B. Bode
Beilstein J. Org. Chem. 2020, 16, 956–965, doi:10.3762/bjoc.16.84
- formula [28]. The general structure of the fabclavines is highly conserved and differs only in the specified moieties as shown in Table 1. The NRPS part of the full-length fabclavines harbors six amino acids, whereby the second position (R1) varies between phenylalanine (Phe), histidine (His), and alanine
- of two A-domains A2 and A6 in the NRPSs FclI and FclJ (Figure 1) [22]. Surprisingly, the key residues of these domains are highly conserved or identical, even between strains that differ in the incorporated amino acids (Table S6, Supporting Information File 1). This indicates the involvement of

Graphical Abstract

Figure 1: General biosynthesis of fabclavine Ia (1) in X. budapestensis (A) and representation of a shortened...

Figure 2: Comparison of the fcl BGCs in Xenorhabdus and Photorhabdus strains responsible for the fabclavine b...
Recent applications of porphyrins as photocatalysts in organic synthesis: batch and continuous flow approaches
- Rodrigo Costa e Silva,
- Luely Oliveira da Silva,
- Aloisio de Andrade Bartolomeu,
- Timothy John Brocksom and
- Kleber Thiago de Oliveira
Beilstein J. Org. Chem. 2020, 16, 917–955, doi:10.3762/bjoc.16.83
- using 4 mol % of tetra-n-butylammonium fluoride (TBAF) as an activator of TMSCN (conditions B). Following this second protocol, the primary α-aminonitriles were rapidly prepared in relevant yields (up to 87%) and converted to the corresponding α-amino acids by hydrolysis of the nitrile (Scheme 57

Graphical Abstract

Figure 1: Chemical structures of the porphyrinoids and their absorption spectra: in bold are highlighted the ...

Figure 2: Photophysical and photochemical processes (Por = porphyrin). Adapted from [12,18].

Figure 3: Main dual photocatalysts and their oxidative/reductive excited state potentials, including porphyri...

Scheme 1: Photoredox alkylation of aldehydes with diazo acetates using porphyrins and a Ru complex. aUsing a ...

Scheme 2: Proposed mechanism for the alkylation of aldehydes with diazo acetates in the presence of TPP.

Scheme 3: Arylation of heteroarenes with aryldiazonium salts using TPFPP as photocatalyst, and corresponding ...

Scheme 4: A) Scope with different aryldiazonium salts and enol acetates. B) Photocatalytic cycles and compari...

Scheme 5: Photoarylation of isopropenyl acetate A) Comparison between batch and continuous-flow approaches an...

Scheme 6: Dehalogenation induced by red light using thiaporphyrin (STPP).

Scheme 7: Applications of NiTPP as both photoreductant and photooxidant.

Scheme 8: Proposed mechanism for obtaining tetrahydroquinolines by reductive quenching.

Scheme 9: Selenylation and thiolation of anilines.

Scheme 10: NiTPP as photoredox catalyst in oxidative and reductive quenching, in comparison with other photoca...

Scheme 11: C–O bond cleavage of 1-phenylethanol using a cobalt porphyrin (CoTMPP) under visible light.

Scheme 12: Hydration of terminal alkynes by RhIII(TSPP) under visible light irradiation.

Scheme 13: Regioselective photocatalytic hydro-defluorination of perfluoroarenes by RhIII(TSPP).

Scheme 14: Formation of 2-methyl-2,3-dihydrobenzofuran by intramolecular hydro-functionalization of allylpheno...

Scheme 15: Photocatalytic oxidative hydroxylation of arylboronic acids using UNLPF-12 as heterogeneous photoca...

Scheme 16: Photocatalytic oxidative hydroxylation of arylboronic acids using MOF-525 as heterogeneous photocat...

Scheme 17: Preparation of the heterogeneous photocatalyst CNH.

Scheme 18: Photoinduced sulfonation of alkenes with sulfinic acid using CNH as photocatalyst.

Scheme 19: Sulfonic acid scope of the sulfonation reactions.

Scheme 20: Regioselective sulfonation reaction of arimistane.

Scheme 21: Synthesis of quinazolin-4-(3H)-ones.

Scheme 22: Selective photooxidation of aromatic benzyl alcohols to benzaldehydes using Pt/PCN-224(Zn).

Scheme 23: Photooxidation of benzaldehydes to benzoic acids using Pt or Pd porphyrins.

Scheme 24: Photocatalytic reduction of various nitroaromatics using a Ni-MOF.

Scheme 25: Photoinduced cycloadditions of CO2 with epoxides by MOF1.

Figure 4: Electronic configurations of the species of oxygen. Adapted from [66].

Scheme 26: TPP-photocatalyzed generation of 1O2 and its application in organic synthesis. Adapted from [67-69].

Scheme 27: Pericyclic reactions involving singlet oxygen and their mechanisms. Adapted from [67].

Scheme 28: First scaled up ascaridole preparation from α-terpinene.

Scheme 29: Antimalarial drug synthesis using an endoperoxidation approach.

Scheme 30: Photooxygenation of colchicine.

Scheme 31: Synthesis of (−)-pinocarvone from abundant (+)-α-pinene.

Scheme 32: Seeberger’s semi-synthesis of artemisinin.

Scheme 33: Synthesis of artemisinin using TPP and supercritical CO2.

Scheme 34: Synthesis of artemisinin using chlorophyll a.

Scheme 35: Quercitol stereoisomer preparation.

Scheme 36: Photocatalyzed preparation of naphthoquinones.

Scheme 37: Continuous endoperoxidation of conjugated dienes and subsequent rearrangements leading to oxidized ...

Scheme 38: The Opatz group total synthesis of (–)-oxycodone.

Scheme 39: Biomimetic syntheses of rhodonoids A, B, E, and F.

Scheme 40: α-Photooxygenation of chiral aldehydes.

Scheme 41: Asymmetric photooxidation of indanone β-keto esters by singlet oxygen using PTC as a chiral inducer...

Scheme 42: Asymmetric photooxidation of both β-keto esters and β-keto amides by singlet oxygen using PTC-2 as ...

Scheme 43: Bifunctional photo-organocatalyst used for the asymmetric oxidation of β-keto esters and β-keto ami...

Scheme 44: Mechanism of singlet oxygen oxidation of sulfides to sulfoxides.

Scheme 45: Controlled oxidation of sulfides to sulfoxides using protonated porphyrins as photocatalysts. aIsol...

Scheme 46: Photochemical oxidation of sulfides to sulfoxides using PdTPFPP as photocatalyst.

Scheme 47: Controlled oxidation of sulfides to sulfoxides using SnPor@PAF as a photosensitizer.

Scheme 48: Syntheses of 2D-PdPor-COF and 3D-Pd-COF.

Scheme 49: Photocatalytic oxidation of A) thioanisole to methyl phenyl sulfoxide and B) various aryl sulfides,...

Scheme 50: General mechanism for oxidation of amines to imines.

Scheme 51: Oxidation of secondary amines to imines.

Scheme 52: Oxidation of secondary amines using Pd-TPFPP as photocatalyst.

Scheme 53: Oxidative amine coupling using UNLPF-12 as heterogeneous photocatalyst.

Scheme 54: Synthesis of Por-COF-1 and Por-COF-2.

Scheme 55: Photocatalytic oxidation of amines to imines by Por-COF-2.

Scheme 56: Photocyanation of primary amines.

Scheme 57: Synthesis of ᴅ,ʟ-tert-leucine hydrochloride.

Scheme 58: Photocyanation of catharanthine and 16-O-acetylvindoline using TPP.

Scheme 59: Photochemical α-functionalization of N-aryltetrahydroisoquinolines using Pd-TPFPP as photocatalyst.

Scheme 60: Ugi-type reaction with 1,2,3,4-tetrahydroisoquinoline using molecular oxygen and TPP.

Scheme 61: Ugi-type reaction with dibenzylamines using molecular oxygen and TPP.

Scheme 62: Mannich-type reaction of tertiary amines using PdTPFPP as photocatalyst.

Scheme 63: Oxidative Mannich reaction using UNLPF-12 as heterogeneous photocatalyst.

Scheme 64: Transformation of amines to α-cyanoepoxides and the proposed mechanism.
Synthesis of new asparagine-based glycopeptides for future scanning tunneling microscopy investigations
- Laura Sršan and
- Thomas Ziegler
Beilstein J. Org. Chem. 2020, 16, 888–894, doi:10.3762/bjoc.16.80

- cell surfaces for future investigations by combined preparative mass spectroscopy and scanning tunneling microscopy (STM) using soft-landing electrospray beam deposition (ES-IBD), on metal surfaces. Keywords: amino acids; asparagine; carbohydrates; glycopeptides; peptidomimetics; Introduction
- example, in anti-HIV therapy, MUC1-based antitumor vaccines, or as antibiotics [12][13][14]. Especially glycans bearing noncanonical amino acids, which can only be introduced into a peptide by organic synthesis, are suitable for cancer therapy since they show better resistance to enzymatic degradation in
- comparison with naturally occurring amino acids [14]. Thus, there is still a great effort in finding new potential drugs derived from glycopeptides [4][15]. By using preparative mass spectrometry (pMS) combined with STM on submolecular-resolution peptides and carbohydrates can be investigated regarding their

Graphical Abstract

Scheme 1: Description of the starting materials 1a–f and 2a–f.

Scheme 2: Peptide coupling reactions, including the previous Fmoc cleavage.

Scheme 3: Cleavage of the fully protected peptides 6 and 7.
Photocatalytic deaminative benzylation and alkylation of tetrahydroisoquinolines with N-alkylpyrydinium salts
- David Schönbauer,
- Carlo Sambiagio,
- Timothy Noël and
- Michael Schnürch
Beilstein J. Org. Chem. 2020, 16, 809–817, doi:10.3762/bjoc.16.74
- from amino acids gave a complex reaction mixture, and the desired products could not be isolated. Additionally, alkyl salts derived from primary amines were unreactive. For the more stable allyl radicals, our initially optimized conditions with [Ru(bpy)3]Cl2 in DMA/ACN were again applicable and the

Graphical Abstract

Scheme 1: Examples of photocatalytic C–C bond formation by nucleophilic trapping of a reactive THIQ intermedi...

Figure 1: Kinetic profile for the benzylation of 1 to 3.

Scheme 2: Benzylation of N-phenyl-THIQ.

Scheme 3: Benzylation of substituted N-arylTHIQs.

Scheme 4: Removal of the PMP protecting group.

Scheme 5: Alkylation of N-phenyl-THIQ derivatives. Conditions: a2 mol % [Ir(dtbbpy)(ppy)2]PF6, DMA, 60 h; b2 ...

Scheme 6: Proposed mechanism.
Recent advances in Cu-catalyzed C(sp3)–Si and C(sp3)–B bond formation
- Balaram S. Takale,
- Ruchita R. Thakore,
- Elham Etemadi-Davan and
- Bruce H. Lipshutz
Beilstein J. Org. Chem. 2020, 16, 691–737, doi:10.3762/bjoc.16.67

- give excellent chemical yields with good enantioselectivities for products 57–59 (Scheme 14). Interestingly, this approach could be extended to the synthesis of amino acids using cesium fluoride in the presence of CO2 gas to afford 60 and 61. From the representative examples shown below, it appears

Graphical Abstract

Scheme 1: Pharmaceuticals possessing a silicon or boron atom.

Scheme 2: The first Cu-catalyzed C(sp3)–Si bond formation.

Scheme 3: Conversion of benzylic phosphate 6 to the corresponding silane.

Scheme 4: Conversion of alkyl triflates to alkylsilanes.

Scheme 5: Conversion of secondary alkyl triflates to alkylsilanes.

Scheme 6: Conversion of alkyl iodides to alkylsilanes.

Scheme 7: Trapping of intermediate radical through cascade reaction.

Scheme 8: Radical pathway for conversion of alkyl iodides to alkylsilanes.

Scheme 9: Conversion of alkyl ester of N-hydroxyphthalimide to alkylsilanes.

Scheme 10: Conversion of gem-dibromides to bis-silylalkanes.

Scheme 11: Conversion of imines to α-silylated amines (A) and the reaction pathway (B).

Scheme 12: Conversion of N-tosylimines to α-silylated amines.

Scheme 13: Screening of diamine ligands.

Scheme 14: Conversion of N-tert-butylsulfonylimines to α-silylated amines.

Scheme 15: Conversion of aldimines to nonracemic α-silylated amines.

Scheme 16: Conversion of N-tosylimines to α-silylated amines.

Scheme 17: Reaction pathway [A] and conversion of aldehydes to α-silylated alcohols [B].

Scheme 18: Conversion of aldehydes to benzhydryl silyl ethers.

Scheme 19: Conversion of ketones to 1,2-diols (A) and conversion of imines to 1,2-amino alcohols (B).

Scheme 20: Ligand screening (A) and conversion of aldehydes to α-silylated alcohols (B).

Scheme 21: Conversion of aldehydes to α-silylated alcohols.

Scheme 22: 1,4-Additions to α,β-unsaturated ketones.

Scheme 23: 1,4-Additions to unsaturated ketones to give β-silylated derivatives.

Scheme 24: Additions onto α,β-unsaturated lactones to give β-silylated lactones.

Scheme 25: Conversion of α,β-unsaturated to β-silylated lactams.

Scheme 26: Conversion of N-arylacrylamides to silylated oxindoles.

Scheme 27: Conversion of α,β-unsaturated carbonyl compounds to silylated tert-butylperoxides.

Scheme 28: Catalytic cycle for Cu(I) catalyzed α,β-unsaturated compounds.

Scheme 29: Conversion of p-quinone methides to benzylic silanes.

Scheme 30: Conversion of α,β-unsaturated ketimines to regio- and stereocontrolled allylic silanes.

Scheme 31: Conversion of α,β-unsaturated ketimines to enantioenriched allylic silanes.

Scheme 32: Regioselective conversion of dienedioates to allylic silanes.

Scheme 33: Conversion of alkenyl-substituted azaarenes to β-silylated adducts.

Scheme 34: Conversion of conjugated benzoxazoles to enantioenriched β-silylated adducts.

Scheme 35: Conversion of α,β-unsaturated carbonyl indoles to α-silylated N-alkylated indoles.

Scheme 36: Conversion of β-amidoacrylates to α-aminosilanes.

Scheme 37: Conversion of α,β-unsaturated ketones to enantioenriched β-silylated ketones, nitriles, and nitro d...

Scheme 38: Regio-divergent silacarboxylation of allenes.

Scheme 39: Silylation of diazocarbonyl compounds, (A) asymmetric and (B) racemic.

Scheme 40: Enantioselective hydrosilylation of alkenes.

Scheme 41: Conversion of 3-acylindoles to indolino-silanes.

Scheme 42: Proposed mechanism for the silylation of 3-acylindoles.

Scheme 43: Silyation of N-chlorosulfonamides.

Scheme 44: Conversion of acyl silanes to α-silyl alcohols.

Scheme 45: Conversion of N-tosylaziridines to β-silylated N-tosylamines.

Scheme 46: Conversion of N-tosylaziridines to silylated N-tosylamines.

Scheme 47: Conversion of 3,3-disubstituted cyclopropenes to silylated cyclopropanes.

Scheme 48: Conversion of conjugated enynes to 1,3-bis(silyl)propenes.

Scheme 49: Proposed sequence for the Cu-catalyzed borylation of substituted alkenes.

Scheme 50: Cu-catalyzed synthesis of nonracemic allylic boronates.

Scheme 51: Cu–NHC catalyzed synthesis of α-substituted allylboronates.

Scheme 52: Synthesis of α-chiral (γ-alkoxyallyl)boronates.

Scheme 53: Cu-mediated formation of nonracemic cis- or trans- 2-substituted cyclopropylboronates.

Scheme 54: Cu-catalyzed synthesis of γ,γ-gem-difluoroallylboronates.

Scheme 55: Cu-catalyzed hydrofunctionalization of internal alkenes and vinylarenes.

Scheme 56: Cu-catalyzed Markovnikov and anti-Markovnikov borylation of alkenes.

Scheme 57: Cu-catalyzed borylation/ortho-cyanation/Cope rearrangement.

Scheme 58: Borylfluoromethylation of alkenes.

Scheme 59: Cu-catalyzed synthesis of tertiary nonracemic alcohols.

Scheme 60: Synthesis of densely functionalized and synthetically versatile 1,2- or 4,3-borocyanated 1,3-butadi...

Scheme 61: Cu-catalyzed trifunctionalization of allenes.

Scheme 62: Cu-catalyzed selective arylborylation of arenes.

Scheme 63: Asymmetric borylative coupling between styrenes and imines.

Scheme 64: Regio-divergent aminoboration of unactivated terminal alkenes.

Scheme 65: Cu-catalyzed 1,4-borylation of α,β-unsaturated ketones.

Scheme 66: Cu-catalyzed protodeboronation of α,β-unsaturated ketones.

Scheme 67: Cu-catalyzed β-borylation of α,β-unsaturated imines.

Scheme 68: Cu-catalyzed synthesis of β-trifluoroborato carbonyl compounds.

Scheme 69: Asymmetric 1,4-borylation of α,β-unsaturated carbonyl compounds.

Scheme 70: Cu-catalyzed ACB and ACA reactions of α,β-unsaturated 2-acyl-N-methylimidazoles.

Scheme 71: Cu-catalyzed diborylation of aldehydes.

Scheme 72: Umpolung pathway for chiral, nonracemic tertiary alcohol synthesis (top) and proposed mechanism for...

Scheme 73: Cu-catalyzed synthesis of α-hydroxyboronates.

Scheme 74: Cu-catalyzed borylation of ketones.

Scheme 75: Cu-catalyzed borylation of unactivated alkyl halides.

Scheme 76: Cu-catalyzed borylation of allylic difluorides.

Scheme 77: Cu-catalyzed borylation of cyclic and acyclic alkyl halides.

Scheme 78: Cu-catalyzed borylation of unactivated alkyl chlorides and bromides.

Scheme 79: Cu-catalyzed decarboxylative borylation of carboxylic acids.

Scheme 80: Cu-catalyzed borylation of benzylic, allylic, and propargylic alcohols.
Photocontrolled DNA minor groove interactions of imidazole/pyrrole polyamides
- Sabrina Müller,
- Jannik Paulus,
- Jochen Mattay,
- Heiko Ihmels,
- Veronica I. Dodero and
- Norbert Sewald
Beilstein J. Org. Chem. 2020, 16, 60–70, doi:10.3762/bjoc.16.8
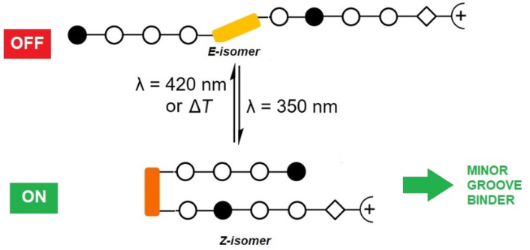
- systematically increased the number of binding motifs, and thus achieved sequence-specific binding [6][7][8][9]. Selectively binding polyamides adopt an antiparallel hairpin structure where a base pair of the DNA is addressed by a pair of the heterocyclic amino acids in the hairpin assembly. Overall, specific
- required because this allowed for an alignment between hydrogen-bonding groups in long polyamides and in the minor groove of DNA [41]. The Fmoc-protected heterocyclic amino acids 2 were obtained from N-methylpyrrole and N-methylimidazole, respectively (Scheme 2A). The N-terminal N-methylpyrrole and N

Graphical Abstract

Scheme 1: Pyrrole–imidazole–azobenzene polyamides and the dsDNA target sequences employed in this study.

Scheme 2: Building blocks required for the synthesis of the photoswitchable Im/Py polyamides. A) Fmoc–Azo–OH 1...

Figure 1: Section of the 1H NMR (600 MHz) spectrum of polyamide P1. A) Initial thermal equilibrium. B) After ...

Figure 2: E/Z isomer ratio of the polyamides P1–P3. Values were obtained from the respective 1H NMR experimen...

Figure 3: Titration experiments of target DNA sequences with P1–P3 in the photostationary Z-state and the the...

Figure 4: Titration of DNA containing single mutations (in bold) with P1–P3 in the photostationary Z-state an...
Understanding the role of active site residues in CotB2 catalysis using a cluster model
- Keren Raz,
- Ronja Driller,
- Thomas Brück,
- Bernhard Loll and
- Dan T. Major
Beilstein J. Org. Chem. 2020, 16, 50–59, doi:10.3762/bjoc.16.7

- relevant, i.e., that have a ligand bound in a reactive configuration and have a fully closed active site. Recently, a crystal structure of the CotB2 enzyme that met these criteria was published [42]. In the current work, we describe the crucial role of the amino acids in the active site on the reaction
- energetics using QM calculations in an active site cluster model. The active site cluster theozyme model [43][44] was constructed from the crystal structure coordinates of active site amino acids, which were presumed to stabilize the carbocations during the reaction cascade. Each reaction step's relevant
- and co-workers [39]. The amino acid cage was constructed from six amino acids, which were located around the substrate and constituted part of the catalytic pocket of the enzyme (PDB-ID 6GGI) [42]. The chosen amino acids were the ones that we presumed stabilized the carbocations the most during the

Graphical Abstract

Scheme 1: Mechanism for formation of cyclooctat-9-en-7-ol, published similarly in [42].

Figure 1: Computed electronic energy profiles (kcal/mol) for the CotB2 cyclase mechanism. The calculations us...

Figure 2: Intermediates A–I in the active site model. Interactions are marked by dashed orange lines, the int...

Figure 3: TS structures TS_A_B–TS_G/H_I in the active site model. Interactions are marked by dashed orange li...

Figure 4: Comparison between gas phase and active site model conformations. A) Intermediate D. B) Intermediat...
Light-controllable dithienylethene-modified cyclic peptides: photoswitching the in vivo toxicity in zebrafish embryos
- Sergii Afonin,
- Oleg Babii,
- Aline Reuter,
- Volker Middel,
- Masanari Takamiya,
- Uwe Strähle,
- Igor V. Komarov and
- Anne S. Ulrich
Beilstein J. Org. Chem. 2020, 16, 39–49, doi:10.3762/bjoc.16.6

- plasma stability in vivo, limited understanding of their mechanisms of action, and high in vivo toxicity. The former two drawbacks are being adequately resolved [9] (e.g., by modifying the peptide backbone [10], macrocyclization [11], or by use of unnatural amino acids [12]), and mechanistic

Graphical Abstract

Figure 1: DAE photoswitch and photoswitchable peptides explored in this study. (A) The reversible photoisomer...

Figure 2: Two versions of the D. rerio embryotoxicity assay for DAE-modified peptides: timelines, peptide pho...

Figure 3: The in vivo toxicity against D. rerio embryos appears to be correlated with the empirical hydrophob...

Figure 4: D. rerio embryotoxicity of GS 1 and the photoswitchable analogues 2–20 correlated with their in vit...

Figure 5: Phototherapeutic cytotoxic action against HeLa cells of GS 1 and its photoswitchable analogues 2–20...
Extension of the 5-alkynyluridine side chain via C–C-bond formation in modified organometallic nucleosides using the Nicholas reaction
- Renata Kaczmarek,
- Dariusz Korczyński,
- James R. Green and
- Roman Dembinski
Beilstein J. Org. Chem. 2020, 16, 1–8, doi:10.3762/bjoc.16.1
- intramolecular versions of the reaction are also highly successful [50][51]. Although the Nicholas reaction has been employed to functionalize biomolecules, including amino acids [52][53], β-lactams [54], steroids [55], and carbohydrates [56][57][58][59][60][61][62], we are unaware of any examples of nucleoside

Graphical Abstract

Scheme 1: Preparation of (2'-deoxy)-5-alkynyluridines 2 and 3, their dicobalt hexacarbonyl derivatives 4 and 5...

Figure 1: Structures of nucleosides 6 and 7, products of the Nicholas reaction.
Why do thioureas and squaramides slow down the Ireland–Claisen rearrangement?
- Dominika Krištofíková,
- Juraj Filo,
- Mária Mečiarová and
- Radovan Šebesta
Beilstein J. Org. Chem. 2019, 15, 2948–2957, doi:10.3762/bjoc.15.290
- diastereoselectivities, and ees up to 86% [26]. Rearrangement of allyl esters of glycine derivatives gave under similar conditions amino acids with a quaternary stereocenter on the β-carbon with high yields and excellent diastereo- as well as enantioselectivity [5]. A reductive rearrangement of allyl esters of acrylic

Graphical Abstract

Scheme 1: Ireland–Claisen rearrangement of allyl esters 1a–c.

Scheme 2: Ireland–Claisen rearrangement of 1c mediated by tertiary amines.

Figure 1: Organocatalysts used in this study. Conditions: typical procedure: 1. Et3N (4.9 equiv), DCM, −60 °C...

Scheme 3: Solvent-free Ireland–Claisen rearrangement of cinnamyl esters.

Figure 2: ωB97X-D/6-31G* calculated uncatalyzed Ireland–Claisen rearrangement of 1c. Charges on allylic oxyge...

Figure 3: ωB97X-D/6-31G* calculated Schreiner thiourea (12)-catalyzed Ireland–Claisen rearrangement of 1c. Ch...

Figure 4: ωB97X-D/6-31G* calculated Ph-thiourea (top) and squaramide-catalyzed (bottom) Ireland–Claisen rearr...

Figure 5: a) Rate of product formation; b) reaction profile without catalyst determined by 1H NMR.
Chemical synthesis of tripeptide thioesters for the biotechnological incorporation into the myxobacterial secondary metabolite argyrin via mutasynthesis
- David C. B. Siebert,
- Roman Sommer,
- Domen Pogorevc,
- Michael Hoffmann,
- Silke C. Wenzel,
- Rolf Müller and
- Alexander Titz
Beilstein J. Org. Chem. 2019, 15, 2922–2929, doi:10.3762/bjoc.15.286
- , i.e., the target for the mutasynthons of this work, is color coded as product of the Arg2 synthetase and in the resulting final argyrin molecule. Designed mutasynthons 9–14 for argyrin biosynthesis. Peptides are based on three amino acids and additionally bear the thioester moiety mimicking the native

Graphical Abstract

Figure 1: Chemical structures of naturally occurring argyrins with potent antipseudomonal activity.

Figure 2: The biosynthetic pathway for argyrin production in Cystobacter sp. SBCb004 (Arg1, radical SAM-depen...

Figure 3: Designed mutasynthons 9–14 for argyrin biosynthesis. Peptides are based on three amino acids and ad...

Scheme 1: Synthesis of tripeptide thioesters. Reagents and conditions: (a) SOCl2, EtOH, 78 °C; (b) IBCF, NMM,...

Scheme 2: Improved synthesis of the tripeptide thioester 14. Reagents and conditions: (a) SOCl2, EtOH, 78 °C;...

Figure 4: Analysis of mutasynthon 14 obtained via the convergent synthetic route by HPLC on a HILIC stationar...
Bacterial terpene biosynthesis: challenges and opportunities for pathway engineering
- Eric J. N. Helfrich,
- Geng-Min Lin,
- Christopher A. Voigt and
- Jon Clardy
Beilstein J. Org. Chem. 2019, 15, 2889–2906, doi:10.3762/bjoc.15.283

- rearrangements, creating the basic hydrocarbon skeleton of a terpene [10][11]. This basic hydrocarbon skeleton is then modified to generate a large number of terpenoid structures, which can be further modified by addition of other building blocks, like sugars, amino acids, or fatty acids [12]. Terpenes are named

Graphical Abstract

Figure 1: Examples of bioactive terpenoids.

Figure 2: Repetitive electrophilic and nucleophilic functionalities in terpene and type II PKS-derived polyke...

Figure 3: Abundance and distribution of bacterial terpene biosynthetic gene clusters as determined by genome ...

Figure 4: Terpenoid biosynthesis. Terpenoid biosynthesis is divided into two phases, 1) terpene scaffold gene...

Figure 5: Mechanisms for type I, type II, and type II/type I tandem terpene cyclases. a) Tail-to-head class I...

Figure 6: Functional TC characterization. a) Different terpenes were produced when hedycaryol (18) synthase a...

Figure 7: Selected examples of terpene modification by bacterial CYPs. a) Hydroxylation [89]. b) Carboxylation, h...

Figure 8: Off-target effects observed during heterologous expression of terpenoid BGCs. Unexpected oxidation ...

Figure 9: TC promiscuity and engineering. a) Spata-13,17-diene (39) synthase (SpS) can take C15 and C25 oligo...

Figure 10: Substrate promiscuity and engineering of CYPs. a) Selected examples from using a CYP library to oxi...

Figure 11: Engineering of terpenoid pathways. a) Metabolic network of terpenoid biosynthesis. Toxic intermedia...
Emission and biosynthesis of volatile terpenoids from the plasmodial slime mold Physarum polycephalum
- Xinlu Chen,
- Tobias G. Köllner,
- Wangdan Xiong,
- Guo Wei and
- Feng Chen
Beilstein J. Org. Chem. 2019, 15, 2872–2880, doi:10.3762/bjoc.15.281
- genes were identified from the transcriptomes. They were designated as PpolyTPS1, PpolyTPS2, PpolyTPS3, and PpolyTPS4. The length of the proteins encoded by PpolyTPS1, PpolyTPS2, PpolyTPS3, and PpolyTPS4s is 334, 347, 353, and 337 amino acids, respectively. Among the four proteins, the highest sequence

Graphical Abstract

Figure 1: Plasmodia of P. polycephalum emit a mixture of volatiles predominated by terpenoids. A) GC chromato...

Figure 2: P. polycephalum contains four terpene synthase genes. A) Multiple sequence alignment of the protein...

Figure 3: PpolyTPS1 and PpolyTPS4 have terpene synthase activities. A) GC chromatogram of sesquiterpenes prod...

Figure 4: Phylogenetic analysis of PpolyTPSs with TPSs from dictyostelid social amoebae (Dictyostelids), the ...
Palladium-catalyzed synthesis and nucleotide pyrophosphatase inhibition of benzo[4,5]furo[3,2-b]indoles
- Hoang Huy Do,
- Saif Ullah,
- Alexander Villinger,
- Joanna Lecka,
- Jean Sévigny,
- Peter Ehlers,
- Jamshed Iqbal and
- Peter Langer
Beilstein J. Org. Chem. 2019, 15, 2830–2839, doi:10.3762/bjoc.15.276
- bonding was found between the oxygen of the benzofuran ring and the hydrogens of Lys291. When compound 6a was docked inside the active pocket of the homology model, it represented π–π stacked and π–alkyl attachment of the indole rings on both sides of the molecule with amino acids His380 and Lys295
- . Suramin was used as a positive control. The 3D illustrations of these docked compounds are shown in Figure 5. The binding was observed in a large area of the modelled ENPP3 protein, inside and over the border of the active pocket, due to the bulky structure of suramin. The amino acids Asn290, Ser292
- 52% in case of ENPP3, amino acids sequence similarity with template, mouse ENPP1. The RMSD for ENPP1 was 0.613 Å and 1.349 Å for ENPP3, over 816 and 811 residues in comparison to the template, respectively. The Ramachandran plot represented promising stereochemical properties as above 98% amino acids

Graphical Abstract

Figure 1: Pharmacologically relevant furoindoles.

Scheme 1: Synthesis of benzo[4,5]furo[3,2-b]indoles 5a–j. Conditions: (i) 1.2 equiv 2-bromophenylboronic acid...

Figure 2: Ortep of 5c (propability of ellipsoids: 45%).

Figure 3: Diindolofurans 6a–e.

Figure 4: Illustration of binding poses of selected inhibitors for the ENPP1 homology model: (a): suramin, (b...

Figure 5: 3D poses of docked selected inhibitors inside homology model of ENPP3. (a): suramin, (b): 5e, (c): ...
Combining the Ugi-azide multicomponent reaction and rhodium(III)-catalyzed annulation for the synthesis of tetrazole-isoquinolone/pyridone hybrids
- Gerardo M. Ojeda,
- Prabhat Ranjan,
- Pavel Fedoseev,
- Lisandra Amable,
- Upendra K. Sharma,
- Daniel G. Rivera and
- Erik V. Van der Eycken
Beilstein J. Org. Chem. 2019, 15, 2447–2457, doi:10.3762/bjoc.15.237
- in the literature supporting the chelation of metal centers by a tetrazole ring during the C–H activation processes, in which the tetrazole may also act as directing group [55][56][57][58][59][60][61]. The closely related triazole ring has also been used for the C–H modification of amino acids and

Graphical Abstract

Figure 1: Bioactive molecules containing a tetrazole, pyridone or isoquinolone ring.

Scheme 1: Approaches for the synthesis of tetrazoles and isoquinolones and their interplay as designed in thi...

Scheme 2: Scope of the Ugi-azide-4CR/deprotection/acylation sequence. Ugi-azide-4CR conducted at the 2.0 mmol...

Scheme 3: Influence of substituents R and R2 on the reaction outcome. For compounds 4k–m the overall yield in...

Scheme 4: Influence of the alkyne and R1 substituent on the reaction outcome.

Scheme 5: Scope of acrylic, heterocyclic and ring-fused N-acylaminomethyl tetrazole substrates.

Scheme 6: Proposed reaction mechanism using substrates 1a and 3a.
Sugar-derived oxazolone pseudotetrapeptide as γ-turn inducer and anion-selective transporter
- Sachin S. Burade,
- Sushil V. Pawar,
- Tanmoy Saha,
- Navanath Kumbhar,
- Amol S. Kotmale,
- Manzoor Ahmad,
- Pinaki Talukdar and
- Dilip D. Dhavale
Beilstein J. Org. Chem. 2019, 15, 2419–2427, doi:10.3762/bjoc.15.234

- peptides from C-3 fluorinated ᴅ-glucofuranoid amino acids and demonstrated their selective anion transport activity [17][18]. In continuation of our interest in sugar-derived cyclic peptides [19], we aimed to synthesize cyclic peptides I and II from the corresponding linear di- and tetrapeptides, however

Graphical Abstract

Figure 1: Oxazolone pseudodipeptide 1 and tetrapeptide 2a.

Scheme 1: Synthesis of linear azido ester dipeptide 5 and tetrapeptide 7.

Scheme 2: Synthesis of oxazolone pseudopeptides 1, 2a and 2b.

Figure 2: Characteristic NOEs of 2a.

Figure 3: DMSO titration study of 2a.

Figure 4: 1H NMR temperature study of 2a.

Figure 5: Optimized helical conformations of (A) 2a, (B) 2b and (C) 9.

Figure 6: Ion transport activity (A) for 1, (B) for 2a, across EYPC-LUVs HPTS.

Figure 7: Cation (A) and anion (B) transport activity of 2a.

Figure 8:
Comparison of the ion transport activity of 2a and 2b at 20 µM across EYPC-LUVslucigenin (A). Conce...
Current understanding and biotechnological application of the bacterial diterpene synthase CotB2
- Ronja Driller,
- Daniel Garbe,
- Norbert Mehlmer,
- Monika Fuchs,
- Keren Raz,
- Dan Thomas Major,
- Thomas Brück and
- Bernhard Loll
Beilstein J. Org. Chem. 2019, 15, 2355–2368, doi:10.3762/bjoc.15.228

- sequences of other bacterial TPSs for the presence of the RY pair, also called “basic pair” [61][62] and their flanking regions, led to the identification of a conserved tryptophan six amino acids upstream of the RY pair (Figure 6) [37]. In CotB2, residues of the motif are located at the end of the C
- binding of GGDP and the cyclization reaction. Based on 2H- as well as 13C-isotope labeling experiments a surprising reaction mechanism has been derived (Scheme 2) [35]. Strong support for the proposed reaction mechanism has been predominantly provided by site-directed mutagenesis of amino acids with an
- . Cyclization is initiated by cleavage of the GGDP diphosphate moiety. After two consecutive cyclization reactions, a dolabellatrienyl cation (A) is generated, stabilized by π-cation interactions with W186 (Scheme 2 and Figure 7). Whereas mutation of this residue to amino acids with aromatic character mainly

Graphical Abstract

Figure 1: CotB1 synthesizes geranylgeranyl diphosphate (GGDP) 3 from the substrates dimethylallyl diphosphate...

Figure 2: The bacterial diterpene synthase CotB2wt·Mg2+3·F-Dola in the closed, active conformation (PDB-ID 6G...

Figure 3: Conformational changes of CotB2 upon ligand binding. Superposition of CotB2’s open (teal), pre-cata...

Figure 4: View into the active site of CotB2wt·Mg2+3·F-Dola [37] superimposed with CotB2wt·Mg2+B·GGSDP [36]. (A) The ...

Figure 5: View into the active site of CotB2wt·Mg2+3·F-Dola [37]. Identical view as in Figure 4. (A) The bound F-Dola rea...

Figure 6: The WXXXXXRY motif in protein sequences of diterpene TPS from different bacteria. Highlighted is th...

Scheme 1: Overview of the altered product portfolio as a result of introduced point mutations in the active s...

Scheme 2: Catalytic mechanism of CotB2, derived from isotope labeling experiments [34,35], density functional theory...

Figure 7: (A) The inner surface of the active site is shown in gray. The bound F-Dola reaction intermediate i...

Scheme 3: Variants of CotB2 open the route to a novel product portfolio with altered cyclic carbon skeletons,...
Isolation of fungi using the diffusion chamber device FIND technology
- Benjamin Libor,
- Henrik Harms,
- Stefan Kehraus,
- Ekaterina Egereva,
- Max Crüsemann and
- Gabriele M. König
Beilstein J. Org. Chem. 2019, 15, 2191–2203, doi:10.3762/bjoc.15.216
- (experiment 1: amino acids/peptone, antibiotics; experiments 2–4: ditto, but in sea water) to yield the desired concentration of roughly 320 000 fungal parts/L. Then, the central plate of FIND (Scheme 1) was lowered into these preparations to fill each chamber with an agar solution, containing in average one

Graphical Abstract

Scheme 1: Design and functional parts of the FIND technology.

Scheme 2: Isolation of fungal strains with the FIND technology. 1. Collection of terrestrial or marine sample...

Figure 1: Secondary metabolites isolated from H. cf. alpina.

Figure 2: a) Significant 1H,1H-COSY and 1H,13C-HMBC correlations for compounds 1 and 2. b) Key NOESY correlat...
Isolation and characterisation of irinans, androstane-type withanolides from Physalis peruviana L.
- Annika Stein,
- Dave Compera,
- Bianka Karge,
- Mark Brönstrup and
- Jakob Franke
Beilstein J. Org. Chem. 2019, 15, 2003–2012, doi:10.3762/bjoc.15.196

- Salt Solution (Gibco) without Ca and Mg, trypsinized and re-suspended in Dulbecco’s modified eagle’s medium that contained 5% fetal bovine serum (FBS; L929, KB-3-1, A549) or Roswell Park Memorial Institute medium that contained 5% FBS, 0.5% Minimum Essential Medium Non-Essential Amino Acids, Gibco (MEM

Graphical Abstract

Figure 1: Withanolides from Physalis peruviana. A) Structures of the newly characterised truncated withanolid...

Figure 2: Key NMR correlations. (A) COSY and HMBC correlations for irinan A (2). (B) COSY and HMBC correlatio...

Figure 3: Structures and biosynthesis of androstanes. (A) Androstane backbone and androsterone (7) as a typic...

Figure 4: Intrinsic reactivity of 4ß-hydroxywithanolide E (1) under acidic/basic and oxidative conditions, re...
Complexation of chiral amines by resorcin[4]arene sulfonic acids in polar media – circular dichroism and diffusion studies of chirality transfer and solvent dependence
- Bartosz Setner and
- Agnieszka Szumna
Beilstein J. Org. Chem. 2019, 15, 1913–1924, doi:10.3762/bjoc.15.187
- protein crystallizations. Due to the interest in interactions with proteins we have selected basic amino acids as chiral bidentate amines to interact with RSAs. We have also studied interactions of RSAs with chiral tetradentate ligands (tetraaminocavitands) in order to evaluate the influence of a chelate
- complexation between 1 and (R or S)-2) or 1:5 stoichiometric ratio (for complexation between 1 and amino acids methyl esters). In all cases except monodentate amines (PheOMe, ValOMe) we observed precipitation of the complexes. Samples [1(PheOMe)2] and [1(ValOMe)2] were obtained by mixing of 1 with PheOMe∙HCl
- with methyl esters of amino acids The complexes of 1 with LysOMe, ArgOMe, and HisOMe precipitate from water solutions at pH 7 (sodium acetate buffer). The complexes are soluble in methanol and DMSO. The composition of the precipitated complexes was determined by integration of signals in 1H NMR spectra

Graphical Abstract

Figure 1: Structures of the compounds used in this study and labelling scheme for NMR spectra.

Figure 2: Spectra of complexes [1(LysOMe)2], [1(ArgOMe)2], [1(HisOMe)2]: 1H NMR (a–g) and ROESY (h–j) in meth...

Figure 3: CD (a) and UV (b) spectra of complexes [1(LysOMe)2], [1(PheOMe)2], [1(ValOMe)2], [1(ArgOMe)2], and [...

Figure 4: DOSY spectra of 1 (a), [1(LysOMe)2] (b), [1(ArgOMe)2] (c), [1(HisOMe)2] (d) and [LysOMe + 1(LysOMe)2...

Figure 5: 1H NMR spectra of 1 (a), LysOMe (b), 1H NMR and DOSY spectra of [1(LysOMe)2] (insets show the shape...

Figure 6: 1H NMR spectra of (R)-2 (a); [1((R)-2] (b); [1 + 1((R)-2] (c) (insets show the shape of signals f, ...

Figure 7: 1H NMR and DOSY spectra of (R)-2 (a); [1(R)-2] (b) (inset show the shape signals f, DMSO-d6, 298K, ...
N-(1-Phenylethyl)aziridine-2-carboxylate esters in the synthesis of biologically relevant compounds
- Iwona E. Głowacka,
- Aleksandra Trocha,
- Andrzej E. Wróblewski and
- Dorota G. Piotrowska
Beilstein J. Org. Chem. 2019, 15, 1722–1757, doi:10.3762/bjoc.15.168

- medications as well as of natural products mostly alkaloids but also sphingoids and ceramides and their 1- and 3-deoxy analogues and several hydroxy amino acids and their precursors. Designed strategies provided new procedures to several drugs and alternative approaches to natural products and proved
- efficiency of a 2-substituted N-(1-phenylethyl)aziridine framework as chiron bearing a chiral auxiliary. Keywords: alkaloids; amino acids; asymmetric synthesis; ceramides; chiral catalysis; chiral pool; N-(1-phenylethyl)aziridine chiron; sphingoids; Introduction The synthesis of enantiomerically pure
- for structural enlargement and at least one stereogenic center which is usually transferred into the final product. To assure the highest possible enantiomeric purity chirons are obtained in most instances from natural products like carbohydrates, amino acids, hydroxy acids or terpenes. The

Graphical Abstract

Figure 1: Examples of three-carbon chirons.

Figure 2: Structures of derivatives of N-(1-phenylethyl)aziridine-2-carboxylic acid 5–8.

Figure 3: Synthetic equivalency of aziridine aldehydes 6.

Scheme 1: Synthesis of N-(1-phenylethyl)aziridine-2-carboxylates 5. Reagents and conditions: a) TEA, toluene,...

Scheme 2: Absolute configuration at C2 in (2S,1'S)-5a. Reagents and conditions: a) 20% HClO4, 80 °C, 30 h the...

Scheme 3: Major synthetic strategies for a 2-ketoaziridine scaffold [R* = (R)- or (S)-1-phenylethyl; R′ = Alk...

Scheme 4: Synthesis of cyanide (2S,1'S)-13. Reagents and conditions: a) NH3, EtOH/H2O, rt, 72 h; b) Ph3P, CCl4...

Scheme 5: Synthesis of key intermediates (R)-16 and (R)-17 for (R,R)-formoterol (14) and (R)-tamsulosin (15)....

Scheme 6: Synthesis of mitotic kinesin inhibitors (2R/S,1'R)-23. Reagents and conditions: a) H2, Pd(OH)2, EtO...

Scheme 7: Synthesis of (R)-mexiletine ((R)-24). Reagents and conditions: a) TsCl, TEA, DMAP, CH2Cl2, rt, 1 h;...

Scheme 8: Synthesis of (−)-cathinone ((S)-27). Reagents and conditions: a) PhMgBr, ether, 0 °C; b) H2, 10% Pd...

Scheme 9: Synthesis of N-Boc-norpseudoephedrine ((1S,2S)-(+)-29) and N-Boc-norephedrine ((1R,2S)-29). Reagent...

Scheme 10: Synthesis of (−)-ephedrine ((1R,2S)-31). Reagents and conditions: a) TfOMe, MeCN then NaBH3CN, rt; ...

Scheme 11: Synthesis of xestoaminol C ((2S,3R)-35), 3-epi-xestoaminol C ((2S,3S)-35) and N-Boc-spisulosine ((2S...

Scheme 12: Synthesis of ʟ-tryptophanol ((S)-41). Reagents and conditions: a) CDI, MeCN, rt, 1 h then TMSI, MeC...

Scheme 13: Synthesis of ʟ-homophenylalaninol ((S)-42). Reagents and conditions: a) NaH, THF, 0 °C to −78 °C, 1...

Scheme 14: Synthesis of ᴅ-homo(4-octylphenyl)alaninol ((R)-47) and a sphingolipid analogue (R)-48. Reagents an...

Scheme 15: Synthesis of florfenicol ((1R,2S)-49). Reagents and conditions: a) (S)-1-phenylethylamine, TEA, MeO...

Scheme 16: Synthesis of natural tyroscherin ((2S,3R,6E,8R,10R)-55). Reagents and conditions: a) I(CH2)3OTIPS, t...

Scheme 17: Syntheses of (−)-hygrine (S)-61, (−)-hygroline (2S,2'S)-62 and (−)-pseudohygroline (2S,2'R)-62. Rea...

Scheme 18: Synthesis of pyrrolidine (3S,3'R)-68, a fragment of the fluoroquinolone antibiotic PF-00951966. Rea...

Scheme 19: Synthesis of sphingolipid analogues (R)-76. Reagents and conditions: a) BnBr, Mg, THF, reflux, 6 h;...

Scheme 20: Synthesis of ᴅ-threo-PDMP (1R,2R)-81. Reagents and conditions: a) TMSCl, NaI, MeCN, rt, 1 h 50 min,...

Scheme 21: Synthesis of the sphingolipid analogue SG-14 (2S,3S)-84. Reagents and conditions: a) LiAlH4, THF, 0...

Scheme 22: Synthesis of the sphingolipid analogue SG-12 (2S,3R)-88. Reagents and conditions: a) 1-(bromomethyl...

Scheme 23: Synthesis of sphingosine-1-phosphate analogues DS-SG-44 and DS-SG-45 (2S,3R)-89a and (2S,3R)-89a. R...

Scheme 24: Synthesis of N-Boc-safingol ((2S,3S)-95) and N-Boc-ᴅ-erythro-sphinganine ((2S,3R)-95). Reagents and...

Scheme 25: Synthesis of ceramide analogues (2S,3R)-96. Reagents and conditions: a) NaBH4, ZnCl2, MeOH, −78 °C,...

Scheme 26: Synthesis of orthogonally protected serinols, (S)-101 and (R)-102. Reagents and conditions: a) BnBr...

Scheme 27: Synthesis of N-acetyl-3-phenylserinol ((1R,2R)-105). Reagents and conditions: a) AcOH, CH2Cl2, refl...

Scheme 28: Synthesis of (S)-linezolid (S)-107. Reagents and conditions: a) LiAlH4, THF, 0 °C to reflux; b) Boc2...

Scheme 29: Synthesis of (2S,3S,4R)-2-aminooctadecane-1,3,4-triol (ᴅ-ribo-phytosphingosine) (2S,3S,4R)-110. Rea...

Scheme 30: Syntheses of ᴅ-phenylalanine (R)-116. Reagents and conditions: a) AcOH, CH2Cl2, reflux, 4 h; b) MsC...

Scheme 31: Synthesis of N-Boc-ᴅ-3,3-diphenylalanine ((R)-122). Reagents and conditions: a) PhMgBr, THF, −78 °C...

Scheme 32: Synthesis of ethyl N,N’-di-Boc-ʟ-2,3-diaminopropanoate ((S)-125). Reagents and conditions: a) NaN3,...

Scheme 33: Synthesis of the bicyclic amino acid (S)-(+)-127. Reagents and conditions: a) BF3·OEt2, THF, 60 °C,...

Scheme 34: Synthesis of lacosamide, (R)-2-acetamido-N-benzyl-3-methoxypropanamide (R)-130. Reagents and condit...

Scheme 35: Synthesis of N-Boc-norfuranomycin ((2S,2'R)-133). Reagents and conditions: a) H2C=CHCH2I, NaH, THF,...

Scheme 36: Synthesis of MeBmt (2S,3R,4R,6E)-139. Reagents and conditions: a) diisopropyl (S,S)-tartrate (E)-cr...

Scheme 37: Synthesis of (+)-polyoxamic acid (2S,3S,4S)-144. Reagents and conditions: a) AD-mix-α, MeSO2NH2, t-...

Scheme 38: Synthesis of the protected 3-hydroxy-ʟ-glutamic acid (2S,3R)-148. Reagents and conditions: a) LiHMD...

Scheme 39: Synthesis of (+)-isoserine (R)-152. Reagents and conditions: a) AcCl, MeCN, rt, 0.5 h then Na2CO3, ...

Scheme 40: Synthesis of (3R,4S)-N3-Boc-3,4-diaminopentanoic acid (3R,4S)-155. Reagents and conditions: a) Ph3P...

Scheme 41: Synthesis of methyl (2S,3S,4S)-4-(dimethylamino)-2,3-dihydroxy-5-methoxypentanoate (2S,3S,4S)-159. ...

Scheme 42: Syntheses of methyl (3S,4S) 4,5-di-N-Boc-amino-3-hydroxypentanoate ((3S,4S)-164), methyl (3S,4S)-4-N...

Scheme 43: Syntheses of (3R,5S)-5-(aminomethyl)-3-(4-methoxyphenyl)dihydrofuran-2(3H)-one ((3R,5S)-168). Reage...

Scheme 44: Syntheses of a series of imidazolin-2-one dipeptides 175–177 (for R' and R'' see text). Reagents an...

Scheme 45: Syntheses of (2S,3S)-N-Boc-3-hydroxy-2-hydroxymethylpyrrolidine ((2S,3S)-179). Reagents and conditi...

Scheme 46: Syntheses of enantiomers of 1,4-dideoxy-1,4-imino-ʟ- and -ᴅ-lyxitols (2S,3R,4S)-182 and (2R,3S,4R)-...

Scheme 47: Synthesis of 1,4-dideoxy-1,4-imino-ʟ-ribitol (2S,3S,4R)-182. Reagents and conditions: a) AcOH, CH2Cl...

Scheme 48: Syntheses of 1,4-dideoxy-1,4-imino-ᴅ-arabinitol (2R,3R,4R)-182 and 1,4-dideoxy-1,4-imino-ᴅ-xylitol ...

Scheme 49: Syntheses of natural 2,5-imino-2,5,6-trideoxy-ʟ-gulo-heptitol ((2S,3R,4R,5R)-184) and its C4 epimer...

Scheme 50: Syntheses of (−)-dihydropinidine ((2S,6R)-187a) (R = C3H7) and (2S,6R)-isosolenopsins (2S,6R)-187b ...

Scheme 51: Syntheses of (+)-deoxocassine ((2S,3S,6R)-190a, R = C12H25) and (+)-spectaline ((2S,3S,6R)-190b, R ...

Scheme 52: Synthesis of (−)-microgrewiapine A ((2S,3R,6S)-194a) and (+)-microcosamine A ((2S,3R,6S)-194b). Rea...

Scheme 53: Syntheses of ʟ-1-deoxynojirimycin ((2S,3S,4S,5R)-200), ʟ-1-deoxymannojirimycin ((2S,3S,4S,5S)-200) ...

Scheme 54: Syntheses of 1-deoxy-ᴅ-galacto-homonojirimycin (2R,3S,4R,5S)-211. Reagents and conditions: a) MeONH...

Scheme 55: Syntheses of 7a-epi-hyacinthacine A1 (1S,2R,3R,7aS)-220. Reagents and conditions: a) TfOTBDMS, 2,6-...

Scheme 56: Syntheses of 8-deoxyhyacinthacine A1 ((1S,2R,3R,7aR)-221). Reagents and conditions: a) H2, Pd/C, PT...

Scheme 57: Syntheses of (+)-lentiginosine ((1S,2S,8aS)-227). Reagents and conditions: a) (EtO)2P(O)CH2COOEt, L...

Scheme 58: Syntheses of 8-epi-swainsonine (1S,2R,8S,8aR)-231. Reagents and conditions: a) Ph3P=CHCOOMe, MeOH, ...

Scheme 59: Synthesis of a protected vinylpiperidine (2S,3R)-237, a key intermediate in the synthesis of (−)-sw...

Scheme 60: Synthesis of a modified carbapenem 245. Reagents and conditions: a) AcOEt, LiHMDS, THF, −78 °C, 1.5...
Recent advances on the transition-metal-catalyzed synthesis of imidazopyridines: an updated coverage
- Gagandeep Kour Reen,
- Ashok Kumar and
- Pratibha Sharma
Beilstein J. Org. Chem. 2019, 15, 1612–1704, doi:10.3762/bjoc.15.165


Graphical Abstract

Figure 1: Various drugs having IP nucleus.

Figure 2: Participation percentage of various TMs for the syntheses of IPs.

Scheme 1: CuI–NaHSO4·SiO2-catalyzed synthesis of imidazo[1,2-a]pyridines.

Scheme 2: Experimental examination of reaction conditions.

Scheme 3: One-pot tandem reaction for the synthesis of 2-haloimidazopyridines.

Scheme 4: Mechanistic scheme for the synthesis of 2-haloimidazopyridine.

Scheme 5: Copper-MOF-catalyzed three-component reaction (3-CR) for imidazo[1,2-a]pyridines.

Scheme 6: Mechanism for copper-MOF-driven synthesis.

Scheme 7: Heterogeneous synthesis via titania-supported CuCl2.

Scheme 8: Mechanism involving oxidative C–H functionalization.

Scheme 9: Heterogeneous synthesis of IPs.

Scheme 10: One-pot regiospecific synthesis of imidazo[1,2-a]pyridines.

Scheme 11: Vinyl azide as an unprecedented substrate for imidazo[1,2-a]pyridines.

Scheme 12: Radical pathway.

Scheme 13: Cu(I)-catalyzed transannulation approach for imidazo[1,5-a]pyridines.

Scheme 14: Plausible radical pathway for the synthesis of imidazo[1,5-a]pyridines.

Scheme 15: A solvent-free domino reaction for imidazo[1,2-a]pyridines.

Scheme 16: Cu-NPs-mediated synthesis of imidazo[1,2-a]pyridines.

Scheme 17: CuI-catalyzed synthesis of isoxazolylimidazo[1,2-a]pyridines.

Scheme 18: Functionalization of 4-bromo derivative via Sonogashira coupling reaction.

Scheme 19: A plausible reaction pathway.

Scheme 20: Cu(I)-catalyzed intramolecular oxidative C–H amidation reaction.

Scheme 21: One-pot synthetic reaction for imidazo[1,2-a]pyridine.

Scheme 22: Plausible reaction mechanism.

Scheme 23: Cu(OAc)2-promoted synthesis of imidazo[1,2-a]pyridines.

Scheme 24: Mechanism for aminomethylation/cycloisomerization of propiolates with imines.

Scheme 25: Three-component synthesis of imidazo[1,2-a]pyridines.

Figure 3: Scope of pyridin-2(1H)-ones and acetophenones.

Scheme 26: CuO NPS-promoted A3 coupling reaction.

Scheme 27: Cu(II)-catalyzed C–N bond formation reaction.

Scheme 28: Mechanism involving Chan–Lam/Ullmann coupling.

Scheme 29: Synthesis of formyl-substituted imidazo[1,2-a]pyridines.

Scheme 30: A tandem sp3 C–H amination reaction.

Scheme 31: Probable mechanistic approach.

Scheme 32: Dual catalytic system for imidazo[1,2-a]pyridines.

Scheme 33: Tentative mechanism.

Scheme 34: CuO/CuAl2O4/ᴅ-glucose-promoted 3-CCR.

Scheme 35: A tandem CuOx/OMS-2-based synthetic strategy.

Figure 4: Biomimetic catalytic oxidation in the presence of electron-transfer mediators (ETMs).

Scheme 36: Control experiment.

Scheme 37: Copper-catalyzed C(sp3)–H aminatin reaction.

Scheme 38: Reaction of secondary amines.

Scheme 39: Probable mechanistic pathway.

Scheme 40: Coupling reaction of α-azidoketones.

Scheme 41: Probable pathway.

Scheme 42: Probable mechanism with free energy calculations.

Scheme 43: MCR for cyanated IP synthesis.

Scheme 44: Substrate scope for the reaction.

Scheme 45: Reaction mechanism.

Scheme 46: Probable mechanistic pathway for Cu/ZnAl2O4-catalyzed reaction.

Scheme 47: Copper-catalyzed double oxidative C–H amination reaction.

Scheme 48: Application towards different coupling reactions.

Scheme 49: Reaction mechanism.

Scheme 50: Condensation–cyclization approach for the synthesis of 1,3-diarylated imidazo[1,5-a]pyridines.

Scheme 51: Optimized reaction conditions.

Scheme 52: One-pot 2-CR.

Scheme 53: One-pot 3-CR without the isolation of chalcone.

Scheme 54: Copper–Pybox-catalyzed cyclization reaction.

Scheme 55: Mechanistic pathway catalyzed by Cu–Pybox complex.

Scheme 56: Cu(II)-promoted C(sp3)-H amination reaction.

Scheme 57: Wider substrate applicability for the reaction.

Scheme 58: Plausible reaction mechanism.

Scheme 59: CuI assisted C–N cross-coupling reaction.

Scheme 60: Probable reaction mechanism involving sp3 C–H amination.

Scheme 61: One-pot MCR-catalyzed by CoFe2O4/CNT-Cu.

Scheme 62: Mechanistic pathway.

Scheme 63: Synthetic scheme for 3-nitroimidazo[1,2-a]pyridines.

Scheme 64: Plausible mechanism for CuBr-catalyzed reaction.

Scheme 65: Regioselective synthesis of halo-substituted imidazo[1,2-a]pyridines.

Scheme 66: Synthesis of 2-phenylimidazo[1,2-a]pyridines.

Scheme 67: Synthesis of diarylated compounds.

Scheme 68: CuBr2-mediated one-pot two-component oxidative coupling reaction.

Scheme 69: Decarboxylative cyclization route to synthesize 1,3-diarylimidazo[1,5-a]pyridines.

Scheme 70: Mechanistic pathway.

Scheme 71: C–H functionalization reaction of enamines to produce diversified heterocycles.

Scheme 72: A plausible mechanism.

Scheme 73: CuI-promoted aerobic oxidative cyclization reaction of ketoxime acetates and pyridines.

Scheme 74: CuI-catalyzed pathway for the formation of imidazo[1,2-a]pyridine.

Scheme 75: Mechanistic pathway.

Scheme 76: Mechanistic rationale for the synthesis of products.

Scheme 77: Copper-catalyzed synthesis of vinyloxy-IP.

Scheme 78: Regioselective product formation with propiolates.

Scheme 79: Proposed mechanism for vinyloxy-IP formation.

Scheme 80: Regioselective synthesis of 3-hetero-substituted imidazo[1,2-a]pyridines with different reaction su...

Scheme 81: Mechanistic pathway.

Scheme 82: CuI-mediated synthesis of 3-formylimidazo[1,2-a]pyridines.

Scheme 83: Radical pathway for 3-formylated IP synthesis.

Scheme 84: Pd-catalyzed urea-cyclization reaction for IPs.

Scheme 85: Pd-catalyzed one-pot-tandem amination and intramolecular amidation reaction.

Figure 5: Scope of aniline nucleophiles.

Scheme 86: Pd–Cu-catalyzed Sonogashira coupling reaction.

Scheme 87: One-pot amide coupling reaction for the synthesis of imidazo[4,5-b]pyridines.

Scheme 88: Urea cyclization reaction for the synthesis of two series of pyridines.

Scheme 89: Amidation reaction for the synthesis of imidazo[4,5-b]pyridines.

Figure 6: Amide scope.

Scheme 90: Pd NPs-catalyzed 3-component reaction for the synthesis of 2,3-diarylated IPs.

Scheme 91: Plausible mechanistic pathway for Pd NPs-catalyzed MCR.

Scheme 92: Synthesis of chromenoannulated imidazo[1,2-a]pyridines.

Scheme 93: Mechanism for the synthesis of chromeno-annulated IPs.

Scheme 94: Zinc oxide NRs-catalyzed synthesis of imidazo[1,2-a]azines/diazines.

Scheme 95: Zinc oxide-catalyzed isocyanide based GBB reaction.

Scheme 96: Reaction pathway for ZnO-catalyzed GBB reaction.

Scheme 97: Mechanistic pathway.

Scheme 98: ZnO NRs-catalyzed MCR for the synthesis of imidazo[1,2-a]azines.

Scheme 99: Ugi type GBB three-component reaction.

Scheme 100: Magnetic NPs-catalyzed synthesis of imidazo[1,2-a]pyridines.

Scheme 101: Regioselective synthesis of 2-alkoxyimidazo[1,2-a]pyridines catalyzed by Fe-SBA-15.

Scheme 102: Plausible mechanistic pathway for the synthesis of 2-alkoxyimidazopyridine.

Scheme 103: Iron-catalyzed synthetic approach.

Scheme 104: Iron-catalyzed aminooxygenation reaction.

Scheme 105: Mechanistic pathway.

Scheme 106: Rh(III)-catalyzed double C–H activation of 2-substituted imidazoles and alkynes.

Scheme 107: Plausible reaction mechanism.

Scheme 108: Rh(III)-catalyzed non-aromatic C(sp2)–H bond activation–functionalization for the synthesis of imid...

Scheme 109: Reactivity and selectivity of different substrates.

Scheme 110: Rh-catalyzed direct C–H alkynylation by Li et al.

Scheme 111: Suggested radical mechanism.

Scheme 112: Scandium(III)triflate-catalyzed one-pot reaction and its mechanism for the synthesis of benzimidazo...

Scheme 113: RuCl3-assisted Ugi-type Groebke–Blackburn condensation reaction.

Scheme 114: C-3 aroylation via Ru-catalyzed two-component reaction.

Scheme 115: Regioselective synthetic mechanism.

Scheme 116: La(III)-catalyzed one-pot GBB reaction.

Scheme 117: Mechanistic approach for the synthesis of imidazo[1,2-a]pyridines.

Scheme 118: Synthesis of imidazo[1,2-a]pyridine using LaMnO3 NPs under neat conditions.

Scheme 119: Mechanistic approach.

Scheme 120: One-pot 3-CR for regioselective synthesis of 2-alkoxy-3-arylimidazo[1,2-a]pyridines.

Scheme 121: Formation of two possible products under optimization of the catalysts.

Scheme 122: Mechanistic strategy for NiFe2O4-catalyzed reaction.

Scheme 123: Two-component reaction for synthesizing imidazodipyridiniums.

Scheme 124: Mechanistic scheme for the synthesis of imidazodipyridiniums.

Scheme 125: CuI-catalyzed arylation of imidazo[1,2-a]pyridines.

Scheme 126: Mechanism for arylation reaction.

Scheme 127: Cupric acetate-catalyzed double carbonylation approach.

Scheme 128: Radical mechanism for double carbonylation of IP.

Scheme 129: C–S bond formation reaction catalyzed by cupric acetate.

Scheme 130: Cupric acetate-catalyzed C-3 formylation approach.

Scheme 131: Control experiments for signifying the role of DMSO and oxygen.

Scheme 132: Mechanism pathway.

Scheme 133: Copper bromide-catalyzed CDC reaction.

Scheme 134: Extension of the substrate scope.

Scheme 135: Plausible radical pathway.

Scheme 136: Transannulation reaction for the synthesis of imidazo[1,5-a]pyridines.

Scheme 137: Plausible reaction pathway for denitrogenative transannulation.

Scheme 138: Cupric acetate-catalyzed C-3 carbonylation reaction.

Scheme 139: Plausible mechanism for regioselective C-3 carbonylation.

Scheme 140: Alkynylation reaction at C-2 of 3H-imidazo[4,5-b]pyridines.

Scheme 141: Two-way mechanism for C-2 alkynylation of 3H-imidazo[4,5-b]pyridines.

Scheme 142: Palladium-catalyzed SCCR approach.

Scheme 143: Palladium-catalyzed Suzuki coupling reaction.

Scheme 144: Reaction mechanism.

Scheme 145: A phosphine free palladium-catalyzed synthesis of C-3 arylated imidazopyridines.

Scheme 146: Palladium-mediated Buchwald–Hartwig cross-coupling reaction.

Figure 7: Structure of the ligands optimized.

Scheme 147: Palladium acetate-catalyzed direct arylation of imidazo[1,2-a]pyridines.

Scheme 148: Palladium acetate-catalyzed mechanistic pathway.

Scheme 149: Palladium acetate-catalyzed regioselective arylation reported by Liu and Zhan.

Scheme 150: Mechanism for selective C-3 arylation of IP.

Scheme 151: Pd(II)-catalyzed alkenylation reaction with styrenes.

Scheme 152: Pd(II)-catalyzed alkenylation reaction with acrylates.

Scheme 153: A two way mechanism.

Scheme 154: Double C–H activation reaction catalyzed by Pd(OAc)2.

Scheme 155: Probable mechanism.

Scheme 156: Palladium-catalyzed decarboxylative coupling.

Scheme 157: Mechanistic cycle for decarboxylative arylation reaction.

Scheme 158: Ligand-free approach for arylation of imidazo[1,2-a]pyridine-3-carboxylic acids.

Scheme 159: Mechanism for ligandless arylation reaction.

Scheme 160: NHC-Pd(II) complex assisted arylation reaction.

Scheme 161: C-3 arylation of imidazo[1,2-a]pyridines with aryl bromides catalyzed by Pd(OAc)2.

Scheme 162: Pd(II)-catalyzed C-3 arylations with aryl tosylates and mesylates.

Scheme 163: CDC reaction for the synthesis of imidazo[1,2-a]pyridines.

Scheme 164: Plausible reaction mechanism for Pd(OAc)2-catalyzed synthesis of imidazo[1,2-a]pyridines.

Scheme 165: Pd-catalyzed C–H amination reaction.

Scheme 166: Mechanism for C–H amination reaction.

Scheme 167: One-pot synthesis for 3,6-di- or 2,3,6-tri(hetero)arylimidazo[1,2-a]pyridines.

Scheme 168: C–H/C–H cross-coupling reaction of IPs and azoles catalyzed by Pd(II).

Scheme 169: Mechanistic cycle.

Scheme 170: Rh-catalyzed C–H arylation reaction.

Scheme 171: Mechanistic pathway for C–H arylation of imidazo[1,2-a]pyridine.

Scheme 172: Rh(III)-catalyzed double C–H activation of 2-phenylimidazo[1,2-a]pyridines and alkynes.

Scheme 173: Rh(III)-catalyzed mechanistic pathway.

Scheme 174: Rh(III)-mediated oxidative coupling reaction.

Scheme 175: Reactions showing functionalization of the product obtained by the group of Kotla.

Scheme 176: Mechanism for Rh(III)-catalyzed oxidative coupling reaction.

Scheme 177: Rh(III)-catalyzed C–H activation reaction.

Scheme 178: Mechanistic cycle.

Scheme 179: Annulation reactions of 2-arylimidazo[1,2-a]pyridines and alkynes.

Scheme 180: Two-way reaction mechanism for annulations reaction.

Scheme 181: [RuCl2(p-cymene)]2-catalyzed C–C bond formation reaction.

Scheme 182: Reported reaction mechanism.

Scheme 183: Fe(III) catalyzed C-3 formylation approach.

Scheme 184: SET mechanism-catalyzed by Fe(III).

Scheme 185: Ni(dpp)Cl2-catalyzed KTC coupling.

Scheme 186: Pd-catalyzed SM coupling.

Scheme 187: Vanadium-catalyzed coupling of IP and NMO.

Scheme 188: Mechanistic cycle.

Scheme 189: Selective C3/C5–H bond functionalizations by mono and bimetallic systems.

Scheme 190: rGO-Ni@Pd-catalyzed C–H bond arylation of imidazo[1,2-a]pyridine.

Scheme 191: Mechanistic pathway for heterogeneously catalyzed arylation reaction.

Scheme 192: Zinc triflate-catalyzed coupling reaction of substituted propargyl alcohols.






