Search results
Search for "thiazoles" in Full Text gives 51 result(s) in Beilstein Journal of Organic Chemistry.
Reactions of acryl thioamides with iminoiodinanes as a one-step synthesis of N-sulfonyl-2,3-dihydro-1,2-thiazoles
- Vladimir G. Ilkin,
- Pavel S. Silaichev,
- Valeriy O. Filimonov,
- Tetyana V. Beryozkina,
- Margarita D. Likhacheva,
- Pavel A. Slepukhin,
- Wim Dehaen and
- Vasiliy A. Bakulev
Beilstein J. Org. Chem. 2025, 21, 1397–1403, doi:10.3762/bjoc.21.104
- Chemistry for Metals and Molecules, Department of Chemistry, KU Leuven, Celestijnenlaan 200F, Leuven B-3001, Belgium 10.3762/bjoc.21.104 Abstract A one-step method has been developed for the preparation of 2,3-dihydro-2-sulfonyl-3,4,5-substituted 1,2-thiazoles by the reaction of acryl thioamides and
- iminoiodinanes. A library of 31 examples of tetrasubstituted 1,2-thiazoles was thus synthesized in high yields. The effectiveness of the synthesis method for these poorly studied 1,2-thiazoles was confirmed by scaling the reaction using gram amounts of the starting thioamide. Keywords: iminoiodinanes; sulfonyl
- groups; synthesis; 1,2-thiazolines; thioamides; Introduction 1,2-Thiazoles (isothiazoles) exhibit a wide range of biological activity (Figure 1): antipoliovirus [1], anticancer [2][3][4][5][6][7], against Parkinson's disease [8], and diabetes [9][10][11], and are also used as microbiocides [12][13

Graphical Abstract

Figure 1: Representatives of biologically active 1,2-thiazoles.

Scheme 1: Synthesis of 2,5-dihydro-1,2-thiazoles.

Scheme 2: Synthesis of 2,3-dihydro-N-sulfonyl-1,2-thiazoles 3. Conditions: aMethod A: thioamide 1 (1.0 equiv)...

Figure 2: Compound 3aa in thermal ellipsoids 50% probability.
Recent advances in the electrochemical synthesis of organophosphorus compounds
- Babak Kaboudin,
- Milad Behroozi,
- Sepideh Sadighi and
- Fatemeh Asgharzadeh
Beilstein J. Org. Chem. 2025, 21, 770–797, doi:10.3762/bjoc.21.61


Graphical Abstract

Scheme 1: Electrosynthesis of phenanthridine phosphine oxides.

Scheme 2: Electrosynthesis of 1-aminoalkylphosphine oxides.

Scheme 3: Various electrochemical C–P coupling reactions.

Scheme 4: Electrochemical C–P coupling reaction of indolines.

Scheme 5: Electrochemical C–P coupling reaction of ferrocene.

Scheme 6: Electrochemical C–P coupling reaction of acridines with phosphites.

Scheme 7: Electrochemical C–P coupling reaction of alkenes.

Scheme 8: Electrochemical C–P coupling reaction of arenes in a flow system.

Scheme 9: Electrochemical C–P coupling reaction of heteroarenes.

Scheme 10: Electrochemical C–P coupling reaction of thiazoles.

Scheme 11: Electrochemical C–P coupling reaction of indole derivatives.

Scheme 12: Electrosynthesis of 1-amino phosphonates.

Scheme 13: Electrochemical C–P coupling reaction of aryl and vinyl bromides.

Scheme 14: Electrochemical C–P coupling reaction of phenylpyridine with dialkyl phosphonates in the presence o...

Scheme 15: Electrochemical P–C bond formation of amides.

Scheme 16: Electrochemical synthesis of α-hydroxy phosphine oxides.

Scheme 17: Electrochemical synthesis of π-conjugated phosphonium salts.

Scheme 18: Electrochemical phosphorylation of indoles.

Scheme 19: Electrochemical synthesis of phosphorylated propargyl alcohols.

Scheme 20: Electrochemical synthesis of phosphoramidates.

Scheme 21: Electrochemical reaction of carbazole with diphenylphosphine.

Scheme 22: Electrochemical P–N coupling of carbazole with phosphine oxides.

Scheme 23: Electrochemical P–N coupling of indoles with a trialkyl phosphite.

Scheme 24: Electrochemical synthesis of iminophosphoranes.

Scheme 25: Electrochemical P–O coupling of phenols with dialkyl phosphonate.

Scheme 26: Electrochemical P–O coupling of alcohols with diphenylphosphine.

Scheme 27: Electrochemical P–S coupling of thiols with dialkylphosphines.

Scheme 28: Electrochemical thiophosphorylation of indolizines.

Scheme 29: Electrosynthesis of S-heteroaryl phosphorothioates.

Scheme 30: Electrochemical phosphorylation reactions.

Scheme 31: Electrochemical P–Se formation.

Scheme 32: Electrochemical selenation/halogenation of alkynyl phosphonates.

Scheme 33: Electrochemical enantioselective aryl C–H bond activation.
O,S,Se-containing Biginelli products based on cyclic β-ketosulfone and their postfunctionalization
- Kateryna V. Dil and
- Vitalii A. Palchykov
Beilstein J. Org. Chem. 2024, 20, 2143–2151, doi:10.3762/bjoc.20.184
- of condensed thiazoles and tetrazoles. In silico assessment of ADMET parameters shows that most compounds meet the lead-likeness requirements. The biological profiles of new compounds demonstrate high probability levels of activity against the following pathogens/diseases: Candida albicans, Alphis
- the postmodification of the Biginelli products [2]. Both approaches were tested in this work. We used Hantzsch-type thiazole synthesis for postmodification of product 2a. By employing 2-bromoacetophenone, bromomalononitrile and 2-bromo-1-tetralone we obtained condensed thiazoles 3–5 in 67–88% yields

Graphical Abstract

Scheme 1: The general Biginelli reaction (A) and examples of DHMP (B) and thiopyran-1,1-dioxide (C) containin...

Figure 1: Number of aryl-substituted Biginelli-type products and publications as analyzed by Reaxys database....

Scheme 2: Scope of the obtained Biginelli products 2a–q.

Scheme 3: Synthesis of SO2-containing enastron analogue 2r.

Scheme 4: Postmodification of the Biginelli product 2a.

Figure 2: Distribution of compounds 2a–r, 3–7 (log P (y)–MW (x)) through LLAMA software. The chemical structu...
Negishi-coupling-enabled synthesis of α-heteroaryl-α-amino acid building blocks for DNA-encoded chemical library applications
- Matteo Gasparetto,
- Balázs Fődi and
- Gellért Sipos
Beilstein J. Org. Chem. 2024, 20, 1922–1932, doi:10.3762/bjoc.20.168
- Supporting Information File 1). We decided to explore the potential of this methodology for the formation of α-heteroarylacetates. In particular, we were curious to see whether this methodology translates well for five-membered heteroarene substrates (e.g., thiazoles, pyrazoles, imidazoles) which are usually
- Negishi reaction (Supporting Information File 1). Preliminary experiments were carried out with and without blue light irradiation in the PhotoCubeTM photoreactor [45]. These experiments revealed that while the conversion of imidazoles and pyrazoles benefits from irradiation, thiazoles seem to be largely
- conditions in hand, we proceeded with the investigation of the heteroaryl halide scope in batch (Scheme 3). Thiazoles proved to be challenging substrates typically affording the desired products in moderate yields (2b–h). While 2-chlorothiazole led to the production of 2b in 44% yield, 2-bromo-5

Graphical Abstract

Scheme 1: Known and improved synthetic strategies to access α-(hetero)aryl-amino acids.

Scheme 2: Reformatsky reagent production.

Scheme 3: Scope of ethyl heteroarylacetates. Isolated yields are given. *Dark reactions were carried out for ...

Scheme 4: Telescoped flow synthesis of heteroarylacetates.

Scheme 5: Potential routes for the preparation of oximes.

Scheme 6: Oxime group insertion step.

Scheme 7: Amino ester production: general scheme, scope and gram scale experiment. The numbers in brackets re...

Scheme 8: Reactions scheme and results for the on-DNA experiments. The reported values represent the normaliz...
2-Heteroarylethylamines in medicinal chemistry: a review of 2-phenethylamine satellite chemical space
- Carlos Nieto,
- Alejandro Manchado,
- Ángel García-González,
- David Díez and
- Narciso M. Garrido
Beilstein J. Org. Chem. 2024, 20, 1880–1893, doi:10.3762/bjoc.20.163
- studies. Thiazoles: 2-Thiazolylethylamine was characterized as a more selective and potent histamine H1 agonist [70]. Based on this, Govoni et al. [71] analyzed the pharmacological profile of several histamine H1 antagonists, with a section covering thiazole-based compounds. 2-(Thiazol-4-yl)ethylamine (99

Graphical Abstract

Scheme 1: Description of the 2-heteroarylethylamine scope of the present review featuring appropriate heteroa...

Scheme 2: 2-Aminoethylpyridine derivatives with therapeutic activity.

Scheme 3: 2-Aminoethylfuran derivatives with therapeutic activity.

Scheme 4: 2-Aminoethylthiophene derivatives with therapeutic activity, part 1.

Scheme 5: 2-Aminoethylthiophene derivatives with therapeutic activity, part 2.

Scheme 6: 2-Aminoethylthiophene derivatives with therapeutic activity, part 3.

Scheme 7: 2-Aminoethylpyrrole derivatives with therapeutic activity.

Scheme 8: Histamine metabolic pathway.

Scheme 9: 2-Aminoethylimidazole derivatives with therapeutic activity, part 1. Krel is referred as histamine ...

Scheme 10: Conformationally restricted 2-aminoethylimidazole derivatives with therapeutic activity, part 2.

Scheme 11: 2-Aminoethylimidazole derivatives with therapeutic activity, part 3.

Scheme 12: 2-Aminoethylimidazole derivatives with therapeutic activity, part 4.

Scheme 13: 2-Aminoethylpyrazole derivatives with therapeutic activity.

Scheme 14: 2-Aminoethylisoxazole derivatives with therapeutic activity.

Scheme 15: 2-Aminoethylthiazole derivatives with therapeutic activity.

Scheme 16: 2-Aminoethyloxadiazole derivatives with therapeutic activity.

Scheme 17: 2-Aminoethyltriazole derivatives with therapeutic activity.

Scheme 18: 2-Aminoethyloxadiazole derivatives with therapeutic activity.
Syntheses and medicinal chemistry of spiro heterocyclic steroids
- Laura L. Romero-Hernández,
- Ana Isabel Ahuja-Casarín,
- Penélope Merino-Montiel,
- Sara Montiel-Smith,
- José Luis Vega-Báez and
- Jesús Sandoval-Ramírez
Beilstein J. Org. Chem. 2024, 20, 1713–1745, doi:10.3762/bjoc.20.152
- , such as a one-pot four-component synthesis and the use of ionic liquids as solvents [30]. Kanchithalaivan et al. [31] reported a library of 16-spiro pyrrolo[1,2-c][1,3]thiazoles of trans-androsterone and DHEA (49a and 49b, respectively). The syntheses were achieved through the 1,3-dipolar cycloaddition

Graphical Abstract

Figure 1: Steroidal spiro heterocycles with remarkable pharmacological activity.

Scheme 1: Synthesis of the spirooxetanone 2. a) t-BuOK, THF, rt, 16%.

Scheme 2: Synthesis of the 17-spirooxetane derivative 7. a) HC≡C(CH2)2CH2OTBDPS, n-BuLi, THF, BF3·Et2O, −78 °...

Scheme 3: Pd-catalyzed carbonylation of steroidal alkynols to produce α-methylene-β-lactones at C-3 and C-17 ...

Scheme 4: Catalyst-free protocol to obtain functionalized spiro-lactones by an intramolecular C–H insertion. ...

Scheme 5: One-pot procedure from dienamides to spiro-β-lactams. a) 1. Ac2O, DMAP, Et3N, CH2Cl2, 2. malononitr...

Scheme 6: Spiro-γ-lactone 20 afforded from 7α-alkanamidoestrone derivative 17. a) HC≡CCH2OTHP, n-BuLi, THF, –...

Scheme 7: Synthesis of the 17-spiro-γ-lactone 23, a key intermediate to obtain spironolactone. a) Ethyl propi...

Scheme 8: Synthetic pathway to obtain 17-spirodihydrofuran-3(2H)-ones from 17-oxosteroids. a) 1-Methoxypropa-...

Scheme 9: One-pot procedure to obtain 17-spiro-2H-furan-3-one compounds. a) NaH, diethyl oxalate, benzene, rt...

Scheme 10: Synthesis of 17-spiro-2H-furan-3-one derivatives. a) RCH=NOH, N-chlorosuccinimide/CHCl3, 99%; b) H2...

Scheme 11: Intramolecular condensation of a γ-acetoxy-β-ketoester to synthesize spirofuranone 37. a) (CH3CN)2P...

Scheme 12: Synthesis of spiro 2,5-dihydrofuran derivatives. a) Allyl bromide, DMF, NaH, 0 °C to rt, 93%; b) G-...

Scheme 13: First reported synthesis of C-16 dispiropyrrolidine derivatives. a) Sarcosine, isatin, MeOH, reflux...

Scheme 14: Cycloadducts 47 with antiproliferative activity against human cancer cell lines. a) 1,4-Dioxane–MeO...

Scheme 15: Spiropyrrolidine compounds generated from (E)-16-arylidene steroids and different ylides. a) Acenap...

Scheme 16: 3-Spiropyrrolidines 52a–c obtained from ketones 50a–c. a) p-Toluenesulfonyl hydrazide, MeOH, rt; b)...

Scheme 17: 16-Spiropyrazolines from 16-methylene-13α-estrone derivatives. a) AgOAc, toluene, rt, 78–81%.

Scheme 18: 6-Spiroimidazolines 57 synthesized by a one-pot multicomponent reaction. a) R3-NC, T3P®, DMSO, 70 °...

Scheme 19: Synthesis of spiro-1,3-oxazolines 60, tested as progesterone receptor antagonist agents. a) CF3COCF3...

Scheme 20: Synthesis of spiro-1,3-oxazolidin-2-ones 63 and 66a,b. a) RNH2, EtOH, 70 °C, 70–90%; b) (CCl3O)2CO,...

Scheme 21: Formation of spiro 1,3-oxazolidin-2-one and spiro 2-substituted amino-4,5-dihydro-1,3-oxazoles from ...

Scheme 22: Synthesis of diastereomeric spiroisoxazolines 74 and 75. a) Ar-C(Cl)=N-OH, DIPEA, toluene, rt, 74 (...

Scheme 23: Spiro 1,3-thiazolidine derivatives 77–79 obtained from 2α-bromo-5α-cholestan-3-one 76. a) 2-aminoet...

Scheme 24: Method for the preparation of derivative 83. a) Benzaldehyde, MeOH, reflux, 77%; b) thioglycolic ac...

Scheme 25: Synthesis of spiro 1,3-thiazolidin-4-one derivatives from steroidal ketones. a) Aniline, EtOH, refl...

Scheme 26: Synthesis of spiro N-aryl-1,3-thiazolidin-4-one derivatives 91 and 92. a) Sulfanilamide, DMF, reflu...

Scheme 27: 1,2,4-Trithiolane dimers 94a–e selectively obtained from carbonyl derivatives. a) LR, CH2Cl2, reflu...

Scheme 28: Spiro 1,2,4-triazolidin-3-ones synthesized from semicarbazones. a) H2O2, CHCl3, 0 °C, 82–85%.

Scheme 29: Steroidal spiro-1,3,4-oxadiazoline 99 obtained in two steps from cholest-5-en-3-one (97). a) NH2NHC...

Scheme 30: Synthesis of spiro-1,3,4-thiadiazoline 101 by cyclization and diacetylation of thiosemicarbazone 100...

Scheme 31: Mono- and bis(1,3,4-thiadiazolines) obtained from estrane and androstane derivatives. a) H2NCSNHNH2...

Scheme 32: Different reaction conditions to synthesize spiro-1,3,2-oxathiaphospholanes 108 and 109.

Scheme 33: Spiro-δ-lactones derived from ADT and epi-ADT as inhibitors of 17β-HSDs. a) CH≡C(CH2)2OTHP, n-BuLi,...

Scheme 34: Spiro-δ-lactams 123a,b obtained in a five-step reaction sequence. a) (R)-(+)-tert-butylsulfinamide,...

Scheme 35: Steroid-coumarin conjugates as fluorescent DHT analogues to study 17-oxidoreductases for androgen m...

Scheme 36: 17-Spiro estradiolmorpholinones 130 bearing two types of molecular diversity. a) ʟ- or ᴅ-amino acid...

Scheme 37: Steroidal spiromorpholinones as inhibitors of enzyme 17β-HSD3. a) Methyl ester of ʟ- or ᴅ-leucine, ...

Scheme 38: Steroidal spiro-morpholin-3-ones achieved by N-alkylation or N-acylation of amino diols 141, follow...

Scheme 39: Straightforward method to synthesize a spiromorpholinone derivative from estrone. a) BnBr, K2CO3, CH...

Scheme 40: Pyrazolo[4,3-e][1,2,4]-triazine derivatives 152–154. a) 4-Aminoantipyrine, EtOH/DMF, reflux, 82%; b...

Scheme 41: One-pot procedure to synthesize spiro-1,3,4-thiadiazine derivatives. a) NH2NHCSCONHR, H2SO4, dioxan...

Scheme 42: 1,2,4-Trioxanes with antimalarial activity. a) 1. O2, methylene blue, CH3CN, 500 W tungsten halogen...

Scheme 43: Tetraoxanes 167 and 168 synthesized from ketones 163, 165 and 166. a) NaOH, iPrOH/H2O, 80 °C, 93%; ...

Scheme 44: 1,2,4,5-Tetraoxanes bearing a steroidal moiety and a cycloalkane. a) 30% H2O2/CH2Cl2/CH3CN, HCl, rt...

Scheme 45: Spiro-1,3,2-dioxaphosphorinanes obtained from estrone derivatives. a) KBH4, MeOH, THF or CH2Cl2; b)...

Scheme 46: Synthesis of steroidal spiro-ε-lactone 183. a) 1. Jones reagent, acetone, 0 °C to rt, 2. ClCOCOCl, ...

Scheme 47: Synthesis of spiro-2,3,4,7-tetrahydrooxepines 185 and 187 derived from mestranol and lynestrenol (38...
Methyltransferases from RiPP pathways: shaping the landscape of natural product chemistry
- Maria-Paula Schröder,
- Isabel P.-M. Pfeiffer and
- Silja Mordhorst
Beilstein J. Org. Chem. 2024, 20, 1652–1670, doi:10.3762/bjoc.20.147
- encoded in the pbt cluster of P. rosea. The thiopeptide GE2270 encoded by pbt undergoes further regioselective modification through C-methylation of thiazoles. The two rSAM MTs PbtM2 and PbtM3 methylate thiazole E and thiazole D, respectively [64]. PbtM2 and PbtM3 show substantial sequence similarities to

Graphical Abstract

Figure 1: Schematic representation of the different acceptor regions for the methylation of RiPPs discussed i...

Figure 2: Schematic overview of different methylation strategies for amino acids and peptides. There are seve...

Figure 3: Biological methylation. A) Methyl donors from biological systems. The transferred methyl group is h...

Figure 4: Chemical structures of RiPPs with diverse O-, N-, C-, and S-methylations. Amino acids of lassomycin...

Figure 5: The three-dimensional structures of the conventional O-MTs OlvSA (model structure calculated by Col...

Figure 6: Reaction scheme of the PAMT´s catalysis, leading to the enzymatic conversion of aspartate to aspart...

Figure 7: Structural organisation of the OphMA homodimer. A) Schematic representation. The MT domain is colou...

Figure 8: Overview of the protein architectures and core peptide compositions of borosin N-MTs as defined by ...

Figure 9: Radical SAM C-methyltransferases. A) The different rSAM MT classes containing different functional ...

Figure 10: The three-dimensional structures of the rSAM C-MTs TsrM with bound cobalamin and [4Fe-4S] cluster (...
Non-noble metal-catalyzed cross-dehydrogenation coupling (CDC) involving ether α-C(sp3)–H to construct C–C bonds
- Hui Yu and
- Feng Xu
Beilstein J. Org. Chem. 2023, 19, 1259–1288, doi:10.3762/bjoc.19.94
- adjacent C=C bond, various conjugated alkenyl C–H bonds can also be activated to construct functionalized ethers. In 2013, Wang et al. achieved a mild Cu(OTf)2-catalyzed CDC of (benzo)thiazoles with cyclic ethers in the presence of K2S2O8 (Scheme 9) [59]. The catalytic system is also suitable for

Graphical Abstract

Scheme 1: Research progress of coupling reactions and active compounds containing α-C(sp3)-functionalized eth...

Scheme 2: Transition-metal-catalyzed CDC pathways.

Scheme 3: CDC of active methylene compounds in the α-C(sp3) position of ethers.

Scheme 4: InCl3/Cu(OTf)2/NHPI co-catalyzed CDC reaction.

Scheme 5: CDC of cyclic benzyl ethers with aldehydes.

Scheme 6: Cu-catalyzed CDC of (a) unactivated C(sp3)–H ethers with simple ketones and (b) double C(sp3)−H fun...

Scheme 7: Cu-catalyzed CDC of C(sp3)–H/C(sp3)–H bonds.

Scheme 8: Cu-catalyzed synthesis of chiral 2-substituted tetrahydropyrans.

Scheme 9: CDC of thiazole with cyclic ethers.

Scheme 10: Cu(I)-catalyzed oxidative alkenylation of simple ethers.

Scheme 11: Cross-dehydrogenation coupling of isochroman C(sp3)–H bonds with anisole C(sp2)–H bonds.

Scheme 12: Pd(OAc)2/Cu(OTf)2-catalyzed arylation of α-C(sp3)–H bonds of ethers.

Scheme 13: Cu-catalyzed C(sp3)–H/C(sp2)–H activation strategies to construct C(sp3)–C(sp2) bonds.

Scheme 14: Cu(I)-catalyzed C(sp2)–H alkylation.

Scheme 15: Cu-catalyzed C(sp3)–H/C(sp)–H activation to construct C(sp3)–C(sp) bonds (H2BIP: 2,6-bis(benzimidaz...

Scheme 16: Fe-catalyzed CDC reaction pathways.

Scheme 17: Fe2(CO)9-catalyzed functionalization of C–H bonds.

Scheme 18: Ligand-promoted Fe-catalyzed CDC reaction of N-methylaniline with ethers.

Scheme 19: Fe-catalyzed CDC of C(sp3)–H/C(sp3)–H bonds.

Scheme 20: Fe-catalyzed hydroalkylation of α,β-unsaturated ketones with ethers.

Scheme 21: Solvent-free Fe(NO3)3-catalyzed CDC of C(sp3)–H/C(sp2)–H bonds.

Scheme 22: Alkylation of disulfide compounds to afford tetrasubstituted alkenes.

Scheme 23: Fe-catalyzed formation of 1,1-bis-indolylmethane derivatives.

Scheme 24: Alkylation of coumarins and flavonoids.

Scheme 25: Direct CDC α-arylation of azoles with ethers.

Scheme 26: CDC of terminal alkynes with C(sp3)–H bonds adjacent to oxygen, sulfur or nitrogen atoms.

Scheme 27: Alkylation of terminal alkynes.

Scheme 28: Co-catalyzed functionalization of glycine esters.

Scheme 29: Co-catalyzed construction of C(sp2)–C(sp3) bonds.

Scheme 30: Co-catalyzed CDC of imidazo[1,2-a]pyridines with isochroman.

Scheme 31: Co-catalyzed C–H alkylation of (benz)oxazoles with ethers.

Scheme 32: Cobalt-catalyzed CDC between unactivated C(sp2)–H and C(sp3)–H bonds.

Scheme 33: MnO2-catalyzed CDC of the inactive C(sp3)-H.

Scheme 34: Oxidative cross-coupling of ethers with enamides.

Scheme 35: Ni(II)-catalyzed CDC of indoles with 1,4-dioxane.

Scheme 36: Chemo- and regioselective ortho- or para-alkylation of pyridines.

Scheme 37: Asymmetric CDC of 3,6-dihydro-2H-pyrans with aldehydes.

Scheme 38: CDC of heterocyclic aromatics with ethers.

Scheme 39: Indium-catalyzed alkylation of DHPs with 1,3-dicarbonyl compounds.

Scheme 40: Rare earth-metal-catalyzed CDC reaction.

Scheme 41: Visible-light-driven CDC of cycloalkanes with benzazoles.

Scheme 42: Photoinduced alkylation of quinoline with cyclic ethers.

Scheme 43: Photocatalyzed CDC reactions between α-C(sp3)–H bonds of ethers and C(sp2)–H bonds of aromatics.
Enabling artificial photosynthesis systems with molecular recycling: A review of photo- and electrochemical methods for regenerating organic sacrificial electron donors
- Grace A. Lowe
Beilstein J. Org. Chem. 2023, 19, 1198–1215, doi:10.3762/bjoc.19.88
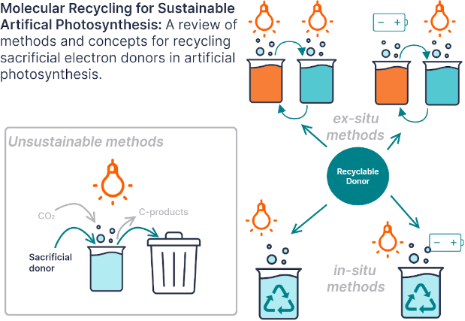

Graphical Abstract

Figure 1: Diagram comparing the two reaction pathways for sacrificial electron donors (SD) in photocatalyzed ...

Figure 2: Diagram showing water-splitting systems developed by Girault, Scanlon, and co-workers that employ i...

Figure 3: Diagram illustrating the transfer of electrons in a photocatalytic particulate suspensions Z-scheme...

Figure 4: A. Structures of the molecules represented in part B. The numbers in brackets correspond to the com...

Figure 5: A. Structures of the molecules represented in part B. The numbers in brackets correspond to the com...
Eschenmoser coupling reactions starting from primary thioamides. When do they work and when not?
- Lukáš Marek,
- Jiří Váňa,
- Jan Svoboda and
- Jiří Hanusek
Beilstein J. Org. Chem. 2023, 19, 808–819, doi:10.3762/bjoc.19.61
- ) without any external thiophile [1][2][5][6]. However, the acidifying effect of the Z-group appears to be not the only prerequisite for successful ECR because in most cases 2-halomalonates [16][17][18][19] and 2-chloroacetoacetates [20] give the corresponding thiazoles and thiazolones, respectively (yields
- ammonium salts [22]. All experiments with salts 6a,b derived from 2b and thiobenzamide or thioacetamide showed that three of four possible products (thiazoles 8a,b, ECR products 9a,b and nitriles; cf. Scheme 3) are formed depending on the reaction conditions specified in Table 1 and Supporting Information
- give the zwitterionic intermediates 6a,b'' necessary for successful ECR. On the other hand, without any base only thiazoles 7a,b are formed probably from isomeric forms 6a,b' of the starting salts 6a,b. In contrast to the tricyclic intermediate 16 formed from oxindole α-thioiminium salt (15), which in

Graphical Abstract

Scheme 1: Eschenmoser coupling reaction between 3-substituted oxindoles and thioamides.

Scheme 2: Possible reactions of α-haloketones, esters and amides with primary thioamides.

Figure 1: Studied α-bromoamides and α-bromolactams.

Scheme 3: Reaction of 4-bromo-1,1-dimethyl-1,4-dihydroisoquinolin-3(2H)-one (2b) with thiobenzamide and thioa...

Scheme 4: Reaction of 4-bromo-1,1-dimethyl-1,4-dihydroisoquinolin-3(2H)-one (2b) with 4’-substituted thiobenz...

Scheme 5: Reaction of 4-bromoisoquinoline-1,3(2H,4H)-dione (3) with thiobenzamide, thioacetamide, and thioben...

Scheme 6: Reaction of N-phenyl- and N-methyl-2-bromo(phenyl)acetamide (4a,b) with thiobenzamide in acetonitri...

Scheme 7: Transformation of salt 15 under kinetic and thermodynamic control conditions [1].

Figure 2: Comparison of energy profiles (relative Gibbs energies at 298 K in kJ·mol−1 for the ECR (right) and...
A one-pot electrochemical synthesis of 2-aminothiazoles from active methylene ketones and thioureas mediated by NH4I
- Shang-Feng Yang,
- Pei Li,
- Zi-Lin Fang,
- Sen Liang,
- Hong-Yu Tian,
- Bao-Guo Sun,
- Kun Xu and
- Cheng-Chu Zeng
Beilstein J. Org. Chem. 2022, 18, 1249–1255, doi:10.3762/bjoc.18.130
- , the in situ generated α-iodoketone was proposed to be the key active species. Keywords: 2-aminothiazoles; electrosynthesis; indirect electrolysis; halide ion; Introduction Thiazoles are prevalent structural motifs in a wide range of natural products [1] and synthetic molecules possessing various
- ], dyes [13], etc. These important features of thiazoles have driven intense interests in their facile synthesis [14][15][16][17]. Among various synthetic routes to the thiazole unit, the Hantzsch condensation of α-halo ketones (dielectrophiles) with various thioureas (dinucleophiles) should be the most
- well-known method (Scheme 1a) [18]. Since active methylene ketones are able to be in situ α-halogenated, the modified Hantzsch condensation of active methylene ketones with thioureas has attracted increasing attention in thiazoles’ synthesis, thereby saving costs and time needed to prepare the required

Graphical Abstract

Scheme 1: Methods for the synthesis of thiazoles using active methylene ketones as starting materials.

Scheme 2: Substrate scope. Reaction conditions: 1 (2 mmol), 2 (1 mmol), NH4I (0.1 mmol), ᴅʟ-alanine (1 mmol),...

Scheme 3: Up-scaling experiment.

Scheme 4: Control experiments.

Scheme 5: The proposed mechanism for the one-pot electrochemical synthesis of 2-aminothiazoles mediated by NH4...
Microwave-assisted multicomponent reactions in heterocyclic chemistry and mechanistic aspects
- Shivani Gulati,
- Stephy Elza John and
- Nagula Shankaraiah
Beilstein J. Org. Chem. 2021, 17, 819–865, doi:10.3762/bjoc.17.71


Graphical Abstract

Figure 1: Marketed drugs with acridine moiety.

Scheme 1: Synthesis of 4-arylacridinediones.

Scheme 2: Proposed mechanism for acridinedione synthesis.

Scheme 3: Synthesis of tetrahydrodibenzoacridinones.

Scheme 4: Synthesis of naphthoacridines.

Scheme 5: Plausible mechanism for naphthoacridines.

Figure 2: Benzoazepines based potent molecules.

Scheme 6: Synthesis of azepinone.

Scheme 7: Proposed mechanism for azepinone formation.

Scheme 8: Synthesis of benzoazulenen-1-one derivatives.

Scheme 9: Proposed mechanism for benzoazulene-1-one synthesis.

Figure 3: Indole-containing pharmacologically active molecules.

Scheme 10: Synthesis of functionalized indoles.

Scheme 11: Plausible mechanism for the synthesis of functionalized indoles.

Scheme 12: Synthesis of spirooxindoles.

Scheme 13: Synthesis of substituted spirooxindoles.

Scheme 14: Plausible mechanism for the synthesis of substituted spirooxindoles.

Scheme 15: Synthesis of pyrrolidinyl spirooxindoles.

Scheme 16: Proposed mechanism for pyrrolidinyl spirooxindoles.

Figure 4: Pyran-containing biologically active molecules.

Scheme 17: Synthesis of functionalized benzopyrans.

Scheme 18: Plausible mechanism for synthesis of benzopyran.

Scheme 19: Synthesis of indoline-spiro-fused pyran derivatives.

Scheme 20: Proposed mechanism for indoline-spiro-fused pyran.

Scheme 21: Synthesis of substituted naphthopyrans.

Figure 5: Marketed drugs with pyrrole ring.

Scheme 22: Synthesis of tetra-substituted pyrroles.

Scheme 23: Mechanism for silica-supported PPA-SiO2-catalyzed pyrrole synthesis.

Scheme 24: Synthesis of pyrrolo[1,10]-phenanthrolines.

Scheme 25: Proposed mechanism for pyrrolo[1,10]-phenanthrolines.

Figure 6: Marketed drugs and molecules containing pyrimidine and pyrimidinones skeletons.

Scheme 26: MWA-MCR pyrimidinone synthesis.

Scheme 27: Two proposed mechanisms for pyrimidinone synthesis.

Scheme 28: MWA multicomponent synthesis of dihydropyrimidinones.

Scheme 29: Proposed mechanism for dihydropyrimidinones.

Figure 7: Biologically active fused pyrimidines.

Scheme 30: MWA- MCR for the synthesis of pyrrolo[2,3-d]pyrimidines.

Scheme 31: Proposed mechanism for pyrrolo[2,3-d]pyrimidines.

Scheme 32: Synthesis of substituted pyrrolo[2,3-d]pyrimidine-2,4-diones.

Scheme 33: Probable pathway for pyrrolo[2,3-d]pyrimidine-2,4-diones.

Scheme 34: Synthesis of pyridopyrimidines.

Scheme 35: Plausible mechanism for the synthesis of pyridopyrimidines.

Scheme 36: Synthesis of dihydropyridopyrimidine and dihydropyrazolopyridine.

Scheme 37: Proposed mechanism for the formation of dihydropyridopyrimidine.

Scheme 38: Synthesis of thiopyrano[4,3-d]pyrimidines.

Scheme 39: Plausible mechanism for the synthesis of thiopyrano[4,3-d]pyrimidines.

Scheme 40: Synthesis of decorated imidazopyrimidines.

Scheme 41: Proposed mechanism for imidazopyrimidine synthesis.

Figure 8: Pharmacologically active molecules containing purine bases.

Scheme 42: Synthesis of aza-adenines.

Scheme 43: Synthesis of 5-aza-7-deazapurines.

Scheme 44: Proposed mechanism for deazapurines synthesis.

Figure 9: Biologically active molecules containing pyridine moiety.

Scheme 45: Synthesis of steroidal pyridines.

Scheme 46: Proposed mechanism for steroidal pyridine.

Scheme 47: Synthesis of N-alkylated 2-pyridones.

Scheme 48: Two possible mechanisms for pyridone synthesis.

Scheme 49: Synthesis of pyridone derivatives.

Scheme 50: Postulated mechanism for synthesis of pyridone.

Figure 10: Biologically active fused pyridines.

Scheme 51: Benzimidazole-imidazo[1,2-a]pyridines synthesis.

Scheme 52: Mechanism for the synthesis of benzimidazole-imidazo[1,2-a]pyridines.

Scheme 53: Synthesis of pyrazolo[3,4-b]pyridine-5-spirocycloalkanedione derivatives.

Scheme 54: Proposed mechanism for spiro-pyridines.

Scheme 55: Functionalized macrocyclane-fused pyrazolo[3,4-b]pyridine derivatives.

Scheme 56: Mechanism postulated for macrocyclane-fused pyrazolo[3,4-b]pyridine.

Scheme 57: Generation of pyrazolo[3,4-b]pyridines.

Scheme 58: Proposed mechanism for the synthesis of pyrazolo[3,4-b]pyridines.

Scheme 59: Proposed mechanism for the synthesis of azepinoindole.

Figure 11: Pharmaceutically important molecules with quinoline moiety.

Scheme 60: Povarov-mediated quinoline synthesis.

Scheme 61: Proposed mechanism for Povarov reaction.

Scheme 62: Synthesis of pyrazoloquinoline.

Scheme 63: Plausible mechanism for pyrazoloquinoline synthesis.

Figure 12: Quinazolinones as pharmacologically significant scaffolds.

Scheme 64: Four-component reaction for dihydroquinazolinone.

Scheme 65: Proposed mechanism for dihydroquinazolinones.

Scheme 66: Synthesis purine quinazolinone and PI3K-δ inhibitor.

Scheme 67: Synthesis of fused benzothiazolo/benzoimidazoloquinazolinones.

Scheme 68: Proposed mechanism for fused benzothiazolo/benzoimidazoloquinazolinones.

Scheme 69: On-water reaction for synthesis of thiazoloquinazolinone.

Scheme 70: Proposed mechanism for the thiazoloquinazolinone synthesis.

Scheme 71: β-Cyclodextrin-mediated synthesis of indoloquinazolinediones.

Scheme 72: Proposed mechanism for synthesis of indoloquinazolinediones.

Figure 13: Triazoles-containing marketted drugs and pharmacologically active molecules.

Scheme 73: Cu(I) DAPTA-catalyzed 1,2,3-triazole formation.

Scheme 74: Mechanism for Cu(I) DAPTA-catalyzed triazole formation.

Scheme 75: Synthesis of β-hydroxy-1,2,3-triazole.

Scheme 76: Proposed mechanism for synthesis of β-hydroxy-1,2,3-triazoles.

Scheme 77: Synthesis of bis-1,2,4-triazoles.

Scheme 78: Proposed mechanism for bis-1,2,4-triazoles synthesis.

Figure 14: Thiazole containing drugs.

Scheme 79: Synthesis of a substituted thiazole ring.

Scheme 80: Synthesis of pyrazolothiazoles.

Figure 15: Chromene containing drugs.

Scheme 81: Magnetic nanocatalyst-mediated aminochromene synthesis.

Scheme 82: Proposed mechanism for the synthesis of chromenes.
Synthesis of (Z)-3-[amino(phenyl)methylidene]-1,3-dihydro-2H-indol-2-ones using an Eschenmoser coupling reaction
- Lukáš Marek,
- Lukáš Kolman,
- Jiří Váňa,
- Jan Svoboda and
- Jiří Hanusek
Beilstein J. Org. Chem. 2021, 17, 527–539, doi:10.3762/bjoc.17.47
- we recently discovered [32][33] a new synthetic pathway involving a rearrangement of 2-aryl-5-(2-aminophenyl)-4-hydroxy-1,3-thiazoles (e.g., 8aa–ad in Scheme 2) leading to the 3-[amino(aryl)methylidene]-1,3-dihydro-2H-indol-2-ones containing an unsubstituted amino group and the oxindole nucleus. In
- the present paper, we want to describe an optimized procedure avoiding the intermediary thiazoles and to demonstrate the usefulness and wide group tolerability of our new synthetic approach for the preparation of a library of substituted phenyl and amino derivatives (Table 1). A number of the
- ) through the intermediary (and in most cases isolable) 2-aryl-5-(2-aminophenyl)-4-hydroxy-1,3-thiazoles 8aa–ad formed from 3-bromooxindole (1a) and various substituted primary aromatic thioamides (e.g., thiobenzamides 2a–d) in acetonitrile (Scheme 2). Although the overall yields of such a two-step

Graphical Abstract

Figure 1: Nintedanib ethanesulfonate.

Scheme 1: The known synthetic strategies leading to 3-(aminomethylidene)oxindoles.

Scheme 2: The possible intermediates and products occurring in the reactions of 3-bromooxindoles with thioben...

Figure 2: The R1 and R2 substitution influence on the isolated yields of products 5aa–ed.

Scheme 3: The Eschenmoser coupling reaction of 3-bromooxindole (1a) with thioacetamides.

Scheme 4: The synthesis of alternative 3-substituted oxindoles and their Eschenmoser coupling reaction with t...
Regioselective synthesis of heterocyclic N-sulfonyl amidines from heteroaromatic thioamides and sulfonyl azides
- Vladimir Ilkin,
- Vera Berseneva,
- Tetyana Beryozkina,
- Tatiana Glukhareva,
- Lidia Dianova,
- Wim Dehaen,
- Eugenia Seliverstova and
- Vasiliy Bakulev
Beilstein J. Org. Chem. 2020, 16, 2937–2947, doi:10.3762/bjoc.16.243
- -arylamino-1,2,3-triazole-4-N-sulfonylcarbimidamides. 2,5-Dithiocarbamoylpyridine reacts with sulfonyl azides to form a pyridine bearing two sulfonyl amidine groups. Keywords: amidines; Dimroth rearrangement; isoxazoles; sulfonyl thiazoles; thioamides; 1,2,3-triazoles; Introduction The biological activity

Graphical Abstract

Figure 1: Examples of biological activity and interesting chemical reactivity of N-sulfonyl amidines.

Figure 2: Data on the synthesis of N′-sulfonylazole-4-carboximidamides.

Scheme 1: Synthesis of 1-alkyl-N-phenyl-N'-(sulfonyl)-1H-1,2,3-triazole-4-carboximidamides 3.

Figure 3: Starting compounds.

Scheme 2: Scope for the reaction of 1-alkyl-1,2,3-triazole-4-carbothioamides 1a–d with azides 2a–f.

Scheme 3: Scope of the reaction of 5-arylamino-1,2,3-triazole-4-carbothioamides 1i–l with azides 2a,c–f.

Scheme 4: Synthesis of 2-aminothiazole-4-N-sulfonyl amidines.

Scheme 5: Synthesis of N-sulfonyl amidines of isoxazolylcarboxylic acid.

Scheme 6: Synthesis of bis(sulfonyl amidines) 3aj–an.

Scheme 7: Plausible mechanism for the reaction of heterocyclic thioamides with sulfonyl azides.
Access to highly substituted oxazoles by the reaction of α-azidochalcone with potassium thiocyanate
- Mysore Bhyrappa Harisha,
- Pandi Dhanalakshmi,
- Rajendran Suresh,
- Raju Ranjith Kumar and
- Shanmugam Muthusubramanian
Beilstein J. Org. Chem. 2020, 16, 2108–2118, doi:10.3762/bjoc.16.178
- -azidochalcones as the precursor [9], we herein report the preparation of highly substituted oxazoles and thiazoles. Oxazoles are ubiquitously found in various natural products [10][11][12][13][14], pharmaceuticals [15][16][17][18], functional materials [19][20] as well as in several organic building blocks [21

Graphical Abstract

Figure 1: Examples of biologically active oxazole and aminothiazole scaffolds.

Scheme 1: Strategies for the synthesis of 2,4,5-trisubstituted oxazole from azirine. a) I2, PPh3; b) NaH, 1H-...

Scheme 2: Scope of the α-azidochalcones. The reactions were carried out at reflux temperature, using 1 (1 mmo...

Scheme 3: Large-scale synthesis of 3i.

Figure 2: Large-scale synthesis of 3i. a) At the start of the reaction, b) after the reaction.

Scheme 4: Acetyl derivative of 3d.

Figure 3: ORTEP diagram of compound 5.

Scheme 5: Synthesis of S-methyl/benzylated products 6 and 7.

Scheme 6: Control experiments.

Scheme 7: Plausible mechanism proposed for the formation of 2,4,5-trisubstituted oxazoles 3.

Scheme 8: Reaction of vinyl azide 1 and 3 with ferric nitrate. Reactions were carried out at reflux temperatu...

Figure 4: X-ray crystal structure of 4h.
When metal-catalyzed C–H functionalization meets visible-light photocatalysis
- Lucas Guillemard and
- Joanna Wencel-Delord
Beilstein J. Org. Chem. 2020, 16, 1754–1804, doi:10.3762/bjoc.16.147

- allowed a significant improvement of the reaction efficiency. The C–H coupling was compatible with a large panel of heteroaromatic substrates including benzoxazoles, oxazoles, thiazoles, and oxadiazoles, as well as non-aromatic oxazolines. Although the initial protocol necessitated the use of a

Graphical Abstract

Figure 1: Concept of dual synergistic catalysis.

Figure 2: Classification of catalytic systems involving two catalysts.

Figure 3: General mechanism for the dual nickel/photoredox catalytic system.

Figure 4: General mechanisms for C–H activation catalysis involving different reoxidation strategies.

Figure 5: Indole synthesis via dual C–H activation/photoredox catalysis.

Figure 6: Proposed mechanism for the indole synthesis via dual catalysis.

Figure 7: Oxidative Heck reaction on arenes via the dual catalysis.

Figure 8: Proposed mechanism for the Heck reaction on arenes via dual catalysis.

Figure 9: Oxidative Heck reaction on phenols via the dual catalysis.

Figure 10: Proposed mechanism for the Heck reaction on phenols via dual catalysis.

Figure 11: Carbazole synthesis via dual C–H activation/photoredox catalysis.

Figure 12: Proposed mechanism for the carbazole synthesis via dual catalysis.

Figure 13: Carbonylation of enamides via the dual C–H activation/photoredox catalysis.

Figure 14: Proposed mechanism for carbonylation of enamides via dual catalysis.

Figure 15: Annulation of benzamides via the dual C–H activation/photoredox catalysis.

Figure 16: Proposed mechanism for the annulation of benzamides via dual catalysis.

Figure 17: Synthesis of indoles via the dual C–H activation/photoredox catalysis.

Figure 18: Proposed mechanism for the indole synthesis via dual catalysis.

Figure 19: General concept of dual catalysis merging C–H activation and photoredox catalysis.

Figure 20: The first example of dual catalysis merging C–H activation and photoredox catalysis.

Figure 21: Proposed mechanism for the C–H arylation with diazonium salts via dual catalysis.

Figure 22: Dual catalysis merging C–H activation/photoredox using diaryliodonium salts.

Figure 23: Direct arylation via the dual catalytic system reported by Xu.

Figure 24: Direct arylation via dual catalytic system reported by Balaraman.

Figure 25: Direct arylation via dual catalytic system reported by Guo.

Figure 26: C(sp3)–H bond arylation via the dual Pd/photoredox catalytic system.

Figure 27: Acetanilide derivatives acylation via the dual C–H activation/photoredox catalysis.

Figure 28: Proposed mechanism for the C–H acylation with α-ketoacids via dual catalysis.

Figure 29: Acylation of azobenzenes via the dual catalysis C–H activation/photoredox.

Figure 30: C2-acylation of indoles via the dual C–H activation/photoredox catalysis.

Figure 31: Proposed mechanism for the C2-acylation of indoles with aldehydes via dual catalysis.

Figure 32: C2-acylation of indoles via the dual C–H activation/photoredox catalysis.

Figure 33: Perfluoroalkylation of arenes via the dual C–H activation/photoredox catalysis.

Figure 34: Proposed mechanism for perfluoroalkylation of arenes via dual catalysis.

Figure 35: Sulfonylation of 1-naphthylamides via the dual C–H activation/photoredox catalysis.

Figure 36: Proposed mechanism for sulfonylation of 1-naphthylamides via dual catalysis.

Figure 37: meta-C–H Alkylation of arenes via visible-light metallaphotocatalysis.

Figure 38: Alternative procedure for meta-C–H alkylation of arenes via metallaphotocatalysis.

Figure 39: Proposed mechanism for meta-C–H alkylation of arenes via metallaphotocatalysis.

Figure 40: C–H borylation of arenes via visible-light metallaphotocatalysis.

Figure 41: Proposed mechanism for C–H borylation of arenes via visible-light metallaphotocatalysis.

Figure 42: Undirected C–H aryl–aryl cross coupling via dual gold/photoredox catalysis.

Figure 43: Proposed mechanism for the undirected C–H aryl–aryl cross-coupling via dual catalysis.

Figure 44: Undirected C–H arylation of (hetero)arenes via dual manganese/photoredox catalysis.

Figure 45: Proposed mechanism for the undirected arylation of (hetero)arenes via dual catalysis.

Figure 46: Photoinduced C–H arylation of azoles via copper catalysis.

Figure 47: Photo-induced C–H chalcogenation of azoles via copper catalysis.

Figure 48: Decarboxylative C–H adamantylation of azoles via dual cobalt/photoredox catalysis.

Figure 49: Proposed mechanism for the C–H adamantylation of azoles via dual catalysis.

Figure 50: General mechanisms for the “classical” (left) and Cu-free variant (right) Sonogoshira reaction.

Figure 51: First example of a dual palladium/photoredox catalysis for Sonogashira-type couplings.

Figure 52: Arylation of terminal alkynes with diazonium salts via dual gold/photoredox catalysis.

Figure 53: Proposed mechanism for the arylation of terminal alkynes via dual catalysis.

Figure 54: C–H Alkylation of alcohols promoted by H-atom transfer (HAT).

Figure 55: Proposed mechanism for the C–H alkylation of alcohols promoted by HAT.

Figure 56: C(sp3)–H arylation of latent nucleophiles promoted by H-atom transfer.

Figure 57: Proposed mechanism for the C(sp3)–H arylation of latent nucleophiles promoted by HAT.

Figure 58: Direct α-arylation of alcohols promoted by H-atom transfer.

Figure 59: Proposed mechanism for the direct α-arylation of alcohols promoted by HAT.

Figure 60: C–H arylation of amines via dual Ni/photoredox catalysis.

Figure 61: Proposed mechanism for the C–H arylation of amines via dual Ni/photoredox catalysis.

Figure 62: C–H functionalization of nucleophiles via excited ketone/nickel dual catalysis.

Figure 63: Proposed mechanism for the C–H functionalization enabled by excited ketones.

Figure 64: Selective sp3–sp3 cross-coupling promoted by H-atom transfer.

Figure 65: Proposed mechanism for the selective sp3–sp3 cross-coupling promoted by HAT.

Figure 66: Direct C(sp3)–H acylation of amines via dual Ni/photoredox catalysis.

Figure 67: Proposed mechanism for the C–H acylation of amines via dual Ni/photoredox catalysis.

Figure 68: C–H hydroalkylation of internal alkynes via dual Ni/photoredox catalysis.

Figure 69: Proposed mechanism for the C–H hydroalkylation of internal alkynes.

Figure 70: Alternative procedure for the C–H hydroalkylation of ynones, ynoates, and ynamides.

Figure 71: Allylic C(sp3)–H activation via dual Ni/photoredox catalysis.

Figure 72: Proposed mechanism for the allylic C(sp3)–H activation via dual Ni/photoredox catalysis.

Figure 73: Asymmetric allylation of aldehydes via dual Cr/photoredox catalysis.

Figure 74: Proposed mechanism for the asymmetric allylation of aldehydes via dual catalysis.

Figure 75: Aldehyde C–H functionalization promoted by H-atom transfer.

Figure 76: Proposed mechanism for the C–H functionalization of aldehydes promoted by HAT.

Figure 77: Direct C–H arylation of strong aliphatic bonds promoted by HAT.

Figure 78: Proposed mechanism for the C–H arylation of strong aliphatic bonds promoted by HAT.

Figure 79: Direct C–H trifluoromethylation of strong aliphatic bonds promoted by HAT.

Figure 80: Proposed mechanism for the C–H trifluoromethylation of strong aliphatic bonds.
Synthesis of new fluorescent molecules having an aggregation-induced emission property derived from 4-fluoroisoxazoles
- Kazuyuki Sato,
- Akira Kawasaki,
- Yukiko Karuo,
- Atsushi Tarui,
- Kentaro Kawai and
- Masaaki Omote
Beilstein J. Org. Chem. 2020, 16, 1411–1417, doi:10.3762/bjoc.16.117
- heteroaromatic systems, especially those comprising two heteroatoms such as pyrazoles [28][29], isoxazoles [30], and thiazoles [31][32]. Recently, we reported the selective fluorination of isoxazoles, to give monofluorinated isoxazoles 3 or trifluorinated isoxazolines 4 in moderate to good yields (Scheme 1) [33

Graphical Abstract

Scheme 1: Selective fluorination of isoxazoles and one-pot synthesis of 4-fluoroisoxazoles.

Scheme 2: One-pot reaction for the synthesis of 3,5-disubstituted 4-fluoroisoxazoles 3. aIsolated yield. bIso...

Figure 1: UV–vis and fluorescence (FL) spectra of compounds 3b and 3c.

Scheme 3: Synthesis of BKIs 6 either from 1,3-diketones 1 or from isoxazoles 2.

Scheme 4: Synthesis of enaminoketones 5 and 8 and their conversion to BKIs (yields refer to isolated yields; a...

Scheme 5: Attempted selective fluorination of BKI 6b.

Scheme 6: Ring-opening reaction of 4-fluoroisoxazoles 3 and their conversion into F-BKIs 9 (yields refer to i...

Figure 2: Photochemical properties comparisons of BKIs and F-BKIs. (a–c) BKI 6b: photograph (a), UV–vis (b), ...
Aldehydes as powerful initiators for photochemical transformations
- Maria A. Theodoropoulou,
- Nikolaos F. Nikitas and
- Christoforos G. Kokotos
Beilstein J. Org. Chem. 2020, 16, 833–857, doi:10.3762/bjoc.16.76
- activation (carbonyl C–H bonds of formamides). Some of the amides or ethers found to be compatible with this method are shown in Scheme 26. A wide range of heteroarenes 114 was also found compatible with this method, including substituted benzothiazole substrates, substituted benzimidazoles, and thiazoles

Graphical Abstract

Scheme 1: Norrish type I and II dissociations.

Scheme 2: Proposed radical pair formation after the photolysis of benzaldehyde (8).

Scheme 3: Aldehydes in the Paterno–Büchi reaction.

Scheme 4: 2,3-Diazabicyclo[2.2.1]hept-2-ene (DBH).

Scheme 5: Dissociation pathways of benzaldehyde.

Scheme 6: Reactions that lead to polarized products detectable by CIDNP.

Scheme 7: MMA (26), DEABP (27), and Michler’s ketone (28).

Scheme 8: Radical intermediates of DEABP.

Scheme 9: Photoinitiated polymerization of monomeric MMA (26) using the quinoxalines 32 and benzaldehyde (8).

Scheme 10: Acetone (4) and formaldehyde (35) as photografting initiators.

Scheme 11: Photografting by employing acetaldehyde (36) as the photoinitiator.

Scheme 12: Proposed photolysis mechanism for aliphatic ketones 44 and formaldehyde (35).

Scheme 13: Initiator 50, reductant 51, and benzaldehyde derivatives 52–54 for the polymerization of the methac...

Scheme 14: Proposed mechanism of the photomediated atom transfer radical polymerization employing the benzalde...

Scheme 15: cis/trans isomerization employing triplet states of photosensitizers.

Scheme 16: Salicylaldehyde (68) forms an internal hydrogen bond.

Scheme 17: Olefin isomerization via energy transfer from a carbonyl compound.

Scheme 18: Mechanistic pathways for the Paterno–Büchi reaction.

Scheme 19: Isomeric oxetanes formed after photochemical addition of aryl aldehydes to 2-butenes.

Scheme 20: Rotation of the C3–C4 bond of the biradical intermediate may lead to all four conformations.

Scheme 21: Photolysis products of benzaldehyde (8) in different solvents. a) In benzene or ethanol. b) In hex-...

Scheme 22: N-tert-Butylbenzamide formation proceeds via a benzoyl radical.

Scheme 23: Photochemical pinacol coupling.

Scheme 24: Photochemical ATRA catalyzed by 4-anisaldehyde (52).

Scheme 25: Proposed triplet sensitization mechanism of the ATRA reaction in the presence of 4-anisaldehyde (52...

Scheme 26: Benzaldehyde-mediated photoredox CDC reaction: compatible amides and ethers.

Scheme 27: Photoredox cross-dehydrogenative coupling (CDC) conditions and proposed reaction mechanism.

Scheme 28: Optimized conditions for the photoredox merger reaction.

Scheme 29: Proposed mechanism for the C(sp3)–H alkylation/arylation of ethers.

Scheme 30: Substrate scope for the photochemical alkylation of ethers.

Scheme 31: C(sp3)–H Functionalization of N-containing molecules.

Scheme 32: Substrate scope for the photochemical alkylation of N-containing molecules.

Scheme 33: Additional products yielded by the photochemical alkylation reaction of N-containing molecules.

Scheme 34: C(sp3)–H functionalization of thioethers.

Scheme 35: Proposed mechanism for the C(sp3)–H alkylation/arylation of N-containing molecules and thioethers.

Scheme 36: Hydroacylation using 4-cyanobenzaldehyde (53) as the photoinitiator.

Scheme 37: Selectivity for the formation of the α,α-disubstituted aldehydes.

Scheme 38: Substrate scope for the photochemical addition of aldehydes to Michael acceptors.

Scheme 39: Proposed mechanism for the hydroacylation of Michael acceptors using 4-cyanobenzaldehyde (53) as th...

Scheme 40: Catalytic arylation of aromatic aldehydes by aryl bromides in which the reaction product acts as th...

Scheme 41: Proposed mechanism for the catalytic arylation of benzaldehydes by aryl bromides in which the react...

Scheme 42: Functionalization of the chiral cyclobutanes 180.

Scheme 43: Optimized reaction conditions and proposed mechanism for the sulfonylcyanation of cyclobutenes.
The reaction of arylmethyl isocyanides and arylmethylamines with xanthate esters: a facile and unexpected synthesis of carbamothioates
- Narasimhamurthy Rajeev,
- Toreshettahally R. Swaroop,
- Ahmad I. Alrawashdeh,
- Shofiur Rahman,
- Abdullah Alodhayb,
- Seegehalli M. Anil,
- Kuppalli R. Kiran,
- Chandra,
- Paris E. Georghiou,
- Kanchugarakoppal S. Rangappa and
- Maralinganadoddi P. Sadashiva
Beilstein J. Org. Chem. 2020, 16, 159–167, doi:10.3762/bjoc.16.18
- reported the synthesis of thiazoles from xanthate esters [31]. In continuation of this ongoing work, we planned to synthesize 5-alkoxy-4-arylthiazoles 3 by the sodium hydride/DMF-mediated reaction of arylmethyl isocyanides 2 with S-alkyl xanthate esters 1 or O-aryl/O-alkyl dithiocarbonates. Unexpectedly

Graphical Abstract

Scheme 1: Synthesis of carbamothioates from xanthate esters and benzyl isocyanides.

Figure 1: Substrate scope for the synthesis of carbamothioates. Reaction conditions for methods A and B: sodi...

Figure 2: ORTEP diagram of O-benzyl (4-fluorobenzyl)carbamothioate (4c).

Figure 3: Rotamers of thionocarbamates 4 (top) and computer-minimized structures of 4c (bottom).

Scheme 2: Proposed general reaction mechanism for the formation of carbamothioates (e.g., 4a) from xanthate e...

Figure 4: Optimized geometries of the reactants, transition states, intermediates, and products of the propos...

Figure 5: Relative energies of the reactants, transition states (TS1–TS3), and intermediates (Int1–Int3) of t...
Reactions of 2-carbonyl- and 2-hydroxy(or methoxy)alkyl-substituted benzimidazoles with arenes in the superacid CF3SO3H. NMR and DFT studies of dicationic electrophilic species
- Dmitry S. Ryabukhin,
- Alexey N. Turdakov,
- Natalia S. Soldatova,
- Mikhail O. Kompanets,
- Alexander Yu. Ivanov,
- Irina A. Boyarskaya and
- Aleksander V. Vasilyev
Beilstein J. Org. Chem. 2019, 15, 1962–1973, doi:10.3762/bjoc.15.191
- reaction [14]. Recently, several hydroxyalkylation reactions followed by alkylation of arenes have been reported involving heterocycle-based superelectrophiles: pyridines, thiazoles, quinolines, isoquinolines, pyrazines, pyrazoles, imidazole and furans, bearing a formyl (carbonyl) group [15][16][17][18][19

Graphical Abstract

Figure 1: Examples of some commercially available pharmaceuticals and agrochemicals containing the benzimidaz...

Figure 2: Formation of cationic species by protonation of 5-formyl-4-methylimidazole in TfOH and their reacti...

Figure 3: Benzimidazoles 1–8 used in this study.

Scheme 1: Reaction of 2-acetylbenzimidazole (2) with TfOH and benzene.

Scheme 2: Reactions of hydroxymethyl-substituted benzimidazole 7 and 8 with TfOH and benzene.

Scheme 3: Reaction mechanism of the formation of compounds 9–11.

Scheme 4: Reaction mechanism of the formation of compounds 12.
Recent advances on the transition-metal-catalyzed synthesis of imidazopyridines: an updated coverage
- Gagandeep Kour Reen,
- Ashok Kumar and
- Pratibha Sharma
Beilstein J. Org. Chem. 2019, 15, 1612–1704, doi:10.3762/bjoc.15.165

- derivatives have been reported by the group of R. K. Reddy [113]. The protocol offered an open air, ligand- and base-free methodology with an extension towards the synthesis of imidazo[1,2-a]pyrazines/pyrimidines and benzo[d]imidazo[2,1-b]thiazoles. For this reaction, they have synthesized the respective

Graphical Abstract

Figure 1: Various drugs having IP nucleus.

Figure 2: Participation percentage of various TMs for the syntheses of IPs.

Scheme 1: CuI–NaHSO4·SiO2-catalyzed synthesis of imidazo[1,2-a]pyridines.

Scheme 2: Experimental examination of reaction conditions.

Scheme 3: One-pot tandem reaction for the synthesis of 2-haloimidazopyridines.

Scheme 4: Mechanistic scheme for the synthesis of 2-haloimidazopyridine.

Scheme 5: Copper-MOF-catalyzed three-component reaction (3-CR) for imidazo[1,2-a]pyridines.

Scheme 6: Mechanism for copper-MOF-driven synthesis.

Scheme 7: Heterogeneous synthesis via titania-supported CuCl2.

Scheme 8: Mechanism involving oxidative C–H functionalization.

Scheme 9: Heterogeneous synthesis of IPs.

Scheme 10: One-pot regiospecific synthesis of imidazo[1,2-a]pyridines.

Scheme 11: Vinyl azide as an unprecedented substrate for imidazo[1,2-a]pyridines.

Scheme 12: Radical pathway.

Scheme 13: Cu(I)-catalyzed transannulation approach for imidazo[1,5-a]pyridines.

Scheme 14: Plausible radical pathway for the synthesis of imidazo[1,5-a]pyridines.

Scheme 15: A solvent-free domino reaction for imidazo[1,2-a]pyridines.

Scheme 16: Cu-NPs-mediated synthesis of imidazo[1,2-a]pyridines.

Scheme 17: CuI-catalyzed synthesis of isoxazolylimidazo[1,2-a]pyridines.

Scheme 18: Functionalization of 4-bromo derivative via Sonogashira coupling reaction.

Scheme 19: A plausible reaction pathway.

Scheme 20: Cu(I)-catalyzed intramolecular oxidative C–H amidation reaction.

Scheme 21: One-pot synthetic reaction for imidazo[1,2-a]pyridine.

Scheme 22: Plausible reaction mechanism.

Scheme 23: Cu(OAc)2-promoted synthesis of imidazo[1,2-a]pyridines.

Scheme 24: Mechanism for aminomethylation/cycloisomerization of propiolates with imines.

Scheme 25: Three-component synthesis of imidazo[1,2-a]pyridines.

Figure 3: Scope of pyridin-2(1H)-ones and acetophenones.

Scheme 26: CuO NPS-promoted A3 coupling reaction.

Scheme 27: Cu(II)-catalyzed C–N bond formation reaction.

Scheme 28: Mechanism involving Chan–Lam/Ullmann coupling.

Scheme 29: Synthesis of formyl-substituted imidazo[1,2-a]pyridines.

Scheme 30: A tandem sp3 C–H amination reaction.

Scheme 31: Probable mechanistic approach.

Scheme 32: Dual catalytic system for imidazo[1,2-a]pyridines.

Scheme 33: Tentative mechanism.

Scheme 34: CuO/CuAl2O4/ᴅ-glucose-promoted 3-CCR.

Scheme 35: A tandem CuOx/OMS-2-based synthetic strategy.

Figure 4: Biomimetic catalytic oxidation in the presence of electron-transfer mediators (ETMs).

Scheme 36: Control experiment.

Scheme 37: Copper-catalyzed C(sp3)–H aminatin reaction.

Scheme 38: Reaction of secondary amines.

Scheme 39: Probable mechanistic pathway.

Scheme 40: Coupling reaction of α-azidoketones.

Scheme 41: Probable pathway.

Scheme 42: Probable mechanism with free energy calculations.

Scheme 43: MCR for cyanated IP synthesis.

Scheme 44: Substrate scope for the reaction.

Scheme 45: Reaction mechanism.

Scheme 46: Probable mechanistic pathway for Cu/ZnAl2O4-catalyzed reaction.

Scheme 47: Copper-catalyzed double oxidative C–H amination reaction.

Scheme 48: Application towards different coupling reactions.

Scheme 49: Reaction mechanism.

Scheme 50: Condensation–cyclization approach for the synthesis of 1,3-diarylated imidazo[1,5-a]pyridines.

Scheme 51: Optimized reaction conditions.

Scheme 52: One-pot 2-CR.

Scheme 53: One-pot 3-CR without the isolation of chalcone.

Scheme 54: Copper–Pybox-catalyzed cyclization reaction.

Scheme 55: Mechanistic pathway catalyzed by Cu–Pybox complex.

Scheme 56: Cu(II)-promoted C(sp3)-H amination reaction.

Scheme 57: Wider substrate applicability for the reaction.

Scheme 58: Plausible reaction mechanism.

Scheme 59: CuI assisted C–N cross-coupling reaction.

Scheme 60: Probable reaction mechanism involving sp3 C–H amination.

Scheme 61: One-pot MCR-catalyzed by CoFe2O4/CNT-Cu.

Scheme 62: Mechanistic pathway.

Scheme 63: Synthetic scheme for 3-nitroimidazo[1,2-a]pyridines.

Scheme 64: Plausible mechanism for CuBr-catalyzed reaction.

Scheme 65: Regioselective synthesis of halo-substituted imidazo[1,2-a]pyridines.

Scheme 66: Synthesis of 2-phenylimidazo[1,2-a]pyridines.

Scheme 67: Synthesis of diarylated compounds.

Scheme 68: CuBr2-mediated one-pot two-component oxidative coupling reaction.

Scheme 69: Decarboxylative cyclization route to synthesize 1,3-diarylimidazo[1,5-a]pyridines.

Scheme 70: Mechanistic pathway.

Scheme 71: C–H functionalization reaction of enamines to produce diversified heterocycles.

Scheme 72: A plausible mechanism.

Scheme 73: CuI-promoted aerobic oxidative cyclization reaction of ketoxime acetates and pyridines.

Scheme 74: CuI-catalyzed pathway for the formation of imidazo[1,2-a]pyridine.

Scheme 75: Mechanistic pathway.

Scheme 76: Mechanistic rationale for the synthesis of products.

Scheme 77: Copper-catalyzed synthesis of vinyloxy-IP.

Scheme 78: Regioselective product formation with propiolates.

Scheme 79: Proposed mechanism for vinyloxy-IP formation.

Scheme 80: Regioselective synthesis of 3-hetero-substituted imidazo[1,2-a]pyridines with different reaction su...

Scheme 81: Mechanistic pathway.

Scheme 82: CuI-mediated synthesis of 3-formylimidazo[1,2-a]pyridines.

Scheme 83: Radical pathway for 3-formylated IP synthesis.

Scheme 84: Pd-catalyzed urea-cyclization reaction for IPs.

Scheme 85: Pd-catalyzed one-pot-tandem amination and intramolecular amidation reaction.

Figure 5: Scope of aniline nucleophiles.

Scheme 86: Pd–Cu-catalyzed Sonogashira coupling reaction.

Scheme 87: One-pot amide coupling reaction for the synthesis of imidazo[4,5-b]pyridines.

Scheme 88: Urea cyclization reaction for the synthesis of two series of pyridines.

Scheme 89: Amidation reaction for the synthesis of imidazo[4,5-b]pyridines.

Figure 6: Amide scope.

Scheme 90: Pd NPs-catalyzed 3-component reaction for the synthesis of 2,3-diarylated IPs.

Scheme 91: Plausible mechanistic pathway for Pd NPs-catalyzed MCR.

Scheme 92: Synthesis of chromenoannulated imidazo[1,2-a]pyridines.

Scheme 93: Mechanism for the synthesis of chromeno-annulated IPs.

Scheme 94: Zinc oxide NRs-catalyzed synthesis of imidazo[1,2-a]azines/diazines.

Scheme 95: Zinc oxide-catalyzed isocyanide based GBB reaction.

Scheme 96: Reaction pathway for ZnO-catalyzed GBB reaction.

Scheme 97: Mechanistic pathway.

Scheme 98: ZnO NRs-catalyzed MCR for the synthesis of imidazo[1,2-a]azines.

Scheme 99: Ugi type GBB three-component reaction.

Scheme 100: Magnetic NPs-catalyzed synthesis of imidazo[1,2-a]pyridines.

Scheme 101: Regioselective synthesis of 2-alkoxyimidazo[1,2-a]pyridines catalyzed by Fe-SBA-15.

Scheme 102: Plausible mechanistic pathway for the synthesis of 2-alkoxyimidazopyridine.

Scheme 103: Iron-catalyzed synthetic approach.

Scheme 104: Iron-catalyzed aminooxygenation reaction.

Scheme 105: Mechanistic pathway.

Scheme 106: Rh(III)-catalyzed double C–H activation of 2-substituted imidazoles and alkynes.

Scheme 107: Plausible reaction mechanism.

Scheme 108: Rh(III)-catalyzed non-aromatic C(sp2)–H bond activation–functionalization for the synthesis of imid...

Scheme 109: Reactivity and selectivity of different substrates.

Scheme 110: Rh-catalyzed direct C–H alkynylation by Li et al.

Scheme 111: Suggested radical mechanism.

Scheme 112: Scandium(III)triflate-catalyzed one-pot reaction and its mechanism for the synthesis of benzimidazo...

Scheme 113: RuCl3-assisted Ugi-type Groebke–Blackburn condensation reaction.

Scheme 114: C-3 aroylation via Ru-catalyzed two-component reaction.

Scheme 115: Regioselective synthetic mechanism.

Scheme 116: La(III)-catalyzed one-pot GBB reaction.

Scheme 117: Mechanistic approach for the synthesis of imidazo[1,2-a]pyridines.

Scheme 118: Synthesis of imidazo[1,2-a]pyridine using LaMnO3 NPs under neat conditions.

Scheme 119: Mechanistic approach.

Scheme 120: One-pot 3-CR for regioselective synthesis of 2-alkoxy-3-arylimidazo[1,2-a]pyridines.

Scheme 121: Formation of two possible products under optimization of the catalysts.

Scheme 122: Mechanistic strategy for NiFe2O4-catalyzed reaction.

Scheme 123: Two-component reaction for synthesizing imidazodipyridiniums.

Scheme 124: Mechanistic scheme for the synthesis of imidazodipyridiniums.

Scheme 125: CuI-catalyzed arylation of imidazo[1,2-a]pyridines.

Scheme 126: Mechanism for arylation reaction.

Scheme 127: Cupric acetate-catalyzed double carbonylation approach.

Scheme 128: Radical mechanism for double carbonylation of IP.

Scheme 129: C–S bond formation reaction catalyzed by cupric acetate.

Scheme 130: Cupric acetate-catalyzed C-3 formylation approach.

Scheme 131: Control experiments for signifying the role of DMSO and oxygen.

Scheme 132: Mechanism pathway.

Scheme 133: Copper bromide-catalyzed CDC reaction.

Scheme 134: Extension of the substrate scope.

Scheme 135: Plausible radical pathway.

Scheme 136: Transannulation reaction for the synthesis of imidazo[1,5-a]pyridines.

Scheme 137: Plausible reaction pathway for denitrogenative transannulation.

Scheme 138: Cupric acetate-catalyzed C-3 carbonylation reaction.

Scheme 139: Plausible mechanism for regioselective C-3 carbonylation.

Scheme 140: Alkynylation reaction at C-2 of 3H-imidazo[4,5-b]pyridines.

Scheme 141: Two-way mechanism for C-2 alkynylation of 3H-imidazo[4,5-b]pyridines.

Scheme 142: Palladium-catalyzed SCCR approach.

Scheme 143: Palladium-catalyzed Suzuki coupling reaction.

Scheme 144: Reaction mechanism.

Scheme 145: A phosphine free palladium-catalyzed synthesis of C-3 arylated imidazopyridines.

Scheme 146: Palladium-mediated Buchwald–Hartwig cross-coupling reaction.

Figure 7: Structure of the ligands optimized.

Scheme 147: Palladium acetate-catalyzed direct arylation of imidazo[1,2-a]pyridines.

Scheme 148: Palladium acetate-catalyzed mechanistic pathway.

Scheme 149: Palladium acetate-catalyzed regioselective arylation reported by Liu and Zhan.

Scheme 150: Mechanism for selective C-3 arylation of IP.

Scheme 151: Pd(II)-catalyzed alkenylation reaction with styrenes.

Scheme 152: Pd(II)-catalyzed alkenylation reaction with acrylates.

Scheme 153: A two way mechanism.

Scheme 154: Double C–H activation reaction catalyzed by Pd(OAc)2.

Scheme 155: Probable mechanism.

Scheme 156: Palladium-catalyzed decarboxylative coupling.

Scheme 157: Mechanistic cycle for decarboxylative arylation reaction.

Scheme 158: Ligand-free approach for arylation of imidazo[1,2-a]pyridine-3-carboxylic acids.

Scheme 159: Mechanism for ligandless arylation reaction.

Scheme 160: NHC-Pd(II) complex assisted arylation reaction.

Scheme 161: C-3 arylation of imidazo[1,2-a]pyridines with aryl bromides catalyzed by Pd(OAc)2.

Scheme 162: Pd(II)-catalyzed C-3 arylations with aryl tosylates and mesylates.

Scheme 163: CDC reaction for the synthesis of imidazo[1,2-a]pyridines.

Scheme 164: Plausible reaction mechanism for Pd(OAc)2-catalyzed synthesis of imidazo[1,2-a]pyridines.

Scheme 165: Pd-catalyzed C–H amination reaction.

Scheme 166: Mechanism for C–H amination reaction.

Scheme 167: One-pot synthesis for 3,6-di- or 2,3,6-tri(hetero)arylimidazo[1,2-a]pyridines.

Scheme 168: C–H/C–H cross-coupling reaction of IPs and azoles catalyzed by Pd(II).

Scheme 169: Mechanistic cycle.

Scheme 170: Rh-catalyzed C–H arylation reaction.

Scheme 171: Mechanistic pathway for C–H arylation of imidazo[1,2-a]pyridine.

Scheme 172: Rh(III)-catalyzed double C–H activation of 2-phenylimidazo[1,2-a]pyridines and alkynes.

Scheme 173: Rh(III)-catalyzed mechanistic pathway.

Scheme 174: Rh(III)-mediated oxidative coupling reaction.

Scheme 175: Reactions showing functionalization of the product obtained by the group of Kotla.

Scheme 176: Mechanism for Rh(III)-catalyzed oxidative coupling reaction.

Scheme 177: Rh(III)-catalyzed C–H activation reaction.

Scheme 178: Mechanistic cycle.

Scheme 179: Annulation reactions of 2-arylimidazo[1,2-a]pyridines and alkynes.

Scheme 180: Two-way reaction mechanism for annulations reaction.

Scheme 181: [RuCl2(p-cymene)]2-catalyzed C–C bond formation reaction.

Scheme 182: Reported reaction mechanism.

Scheme 183: Fe(III) catalyzed C-3 formylation approach.

Scheme 184: SET mechanism-catalyzed by Fe(III).

Scheme 185: Ni(dpp)Cl2-catalyzed KTC coupling.

Scheme 186: Pd-catalyzed SM coupling.

Scheme 187: Vanadium-catalyzed coupling of IP and NMO.

Scheme 188: Mechanistic cycle.

Scheme 189: Selective C3/C5–H bond functionalizations by mono and bimetallic systems.

Scheme 190: rGO-Ni@Pd-catalyzed C–H bond arylation of imidazo[1,2-a]pyridine.

Scheme 191: Mechanistic pathway for heterogeneously catalyzed arylation reaction.

Scheme 192: Zinc triflate-catalyzed coupling reaction of substituted propargyl alcohols.
Synthesis of (macro)heterocycles by consecutive/repetitive isocyanide-based multicomponent reactions
- Angélica de Fátima S. Barreto and
- Carlos Kleber Z. Andrade
Beilstein J. Org. Chem. 2019, 15, 906–930, doi:10.3762/bjoc.15.88
- products has been reviewed [15][16] and this review will focus only on IMCRs. Consecutive IMCRs Synthesis of small-ring heterocycles (tetrazoles, ketopiperazines, imidazoles, imidazolines and thiazoles) The use of consecutive Ugi reactions in the synthesis of heterocycles was first described in 2001 by Ugi

Graphical Abstract

Scheme 1: Comparison between a normal sequential reaction and an MCR.

Scheme 2: Synthesis of tetrazoles and hydantoinimide derivatives by consecutive Ugi reactions [17].

Scheme 3: Synthesis of tetrazole-ketopiperazines by two consecutive Ugi reactions [19].

Scheme 4: Synthesis of acylhydrazino bis(1,5-disubstituted tetrazoles) through two hydrazine-Ugi-azide reacti...

Scheme 5: Synthesis of substituted α-aminomethyltetrazoles through two consecutive Ugi reactions (U-4CR and U...

Scheme 6: Synthesis of tetrazole peptidomimetics by direct use of amino acids in two consecutive Ugi reaction...

Scheme 7: One-pot 8CR based on 3 sequential IMCRs [25].

Scheme 8: Combination of IMCRs for the synthesis of substituted 2H-imidazolines [25].

Scheme 9: 6CR involving a tandem combination of Groebke–Blackburn–Bienaymé and Ugi reaction for the synthesis...

Scheme 10: 5CR involving a tandem combination of Groebke–Blackburn–Bienaymé and Passerini reaction for the syn...

Scheme 11: Synthesis of tubugis via three consecutive IMCRs [27].

Scheme 12: Synthesis of telaprevir through consecutive IMCRs [28].

Scheme 13: Another synthesis of telaprevir through consecutive IMCRs [29].

Scheme 14: a) Synthetic sequence for accessing diverse macrocycles containing the tetrazole nucleus by the uni...

Scheme 15: a) Synthetic sequence for the tetrazolic macrocyclic depsipeptides using a combination of two IMCRs...

Scheme 16: Synthesis of cyclic pentapeptoids by consecutive Ugi reactions [32].

Scheme 17: Synthesis of a cyclic pentapeptoid by consecutive Ugi reactions [32].

Scheme 18: MW-mediated synthesis of a cyclopeptoid by consecutive Ugi reactions [33].

Scheme 19: Synthesis of six cyclic pentadepsipeptoids via consecutive isocyanide-based IMCRs [34].

Scheme 20: Microwave-mediated synthesis of a cyclic heptapeptoid through four consecutive IMCRs [35].

Scheme 21: Macrocyclization of bifunctional building blocks containing diacid/diisonitrile and diamine/diisoni...

Scheme 22: Synthesis of steroid-biaryl ether hybrid macrocycles by MiBs [38].

Scheme 23: Synthesis of biaryl ether-containing macrocycles by MiBs [39].

Scheme 24: Synthesis of natural product-inspired biaryl ether-cyclopeptoid macrocycles [40].

Scheme 25: Synthesis of cholane-based hybrid macrolactams by MiBs [41].

Scheme 26: Synthesis of macrocyclic oligoimine-based DCL using the Ugi-4CR-based quenching approach [42].

Scheme 27: Dye-modified and photoswitchable macrocycles by MiBs [43].

Scheme 28: Synthesis of nonsymmetric cryptands by two sequential double Ugi-4CR-based macrocyclizations [44].

Scheme 29: Synthesis of steroid–aryl hybrid cages by sequential 2- and 3-fold Ugi-4CR-based macrocyclizations [46]....

Scheme 30: Ugi-MiBs approach towards natural product-like macrocycles [47].

Scheme 31: a) Bidirectional macrocyclization of peptides by double Ugi reaction. b) Ugi-4CR for the generation...

Scheme 32: MiBs based on the Passerini-3CR for the synthesis of macrolactones [49].

Scheme 33: Template-driven approach for the synthesis of macrotricycles 170 [50].
Oxidative and reductive cyclization in stiff dithienylethenes
- Michael Kleinwächter,
- Ellen Teichmann,
- Lutz Grubert,
- Martin Herder and
- Stefan Hecht
Beilstein J. Org. Chem. 2018, 14, 2812–2821, doi:10.3762/bjoc.14.259
- case of modified thiazoles [5] or using the strongly donating -PhNMe2 substituent [40]. In these compounds, the first oxidation wave is fully reversible and anodic cyclization occurs only after the second step, forming the open dication (EEC mechanism). By combining strongly electron-donating and

Graphical Abstract

Scheme 1: Combining double bond isomerization (E/Z) and cyclization/cycloreversion (Z/C) in three-state switc...

Scheme 2: Overview of all sDTE and reference DTE compounds investigated in this study. The compound names ind...

Figure 1: Cyclic voltammograms of sDTE66-Me. a) Both E- (black line) and Z-isomer (blue dashed line) display ...

Figure 2: Spectroelectrochemistry of sDTE66-Me. Absorption changes during CV, insets showing the correspondin...

Scheme 3: Proposed mechanism for the oxidative cyclization of sDTE66-Me. Upon two-fold oxidation, both open i...

Figure 3: Anodic peak potentials (Epa) of sDTEs and reference compounds in MeCN. Solid circles refer to the f...

Figure 4: Cyclic voltammograms of sDTE66-PhCN. The reduction of a) E-sDTE66-PhCN (black line) is reversible, ...

Figure 5: Cyclic voltammogram of DTE-PhFluorene. The ring-closed isomer (red dashed line) is formed both unde...

Figure 6: Cyclic voltammograms of Me2NPh-DTE-PhCN displaying separated one-electron anodic and cathodic waves...

Scheme 4: Proposed mechanism to explain the observed selectivity of anodic and cathodic cyclization in sDTE66...
Applications of organocatalysed visible-light photoredox reactions for medicinal chemistry
- Michael K. Bogdos,
- Emmanuel Pinard and
- John A. Murphy
Beilstein J. Org. Chem. 2018, 14, 2035–2064, doi:10.3762/bjoc.14.179
- potential to be of interest if explored further. The good physical chemical properties of thiazoles, as well as their ability to act as isosteres to thiophenes, carbonyls and pyrazoles [49][50] make this scaffold an intriguing novel motif. 2.3 C(sp2)–X bond formation The obvious extension after considering
- thiocyanate functional group, which is a precursor group to many sulfur-containing functional groups, as well as heterocycles such as thiazoles and isothiazoles. The imidazo[1,2-a]pyridine core is a particularly interesting drug-like structure, e.g., electron poor, polar, of low basicity, etc. The scope of

Graphical Abstract

Figure 1: Depiction of the energy levels of a typical organic molecule and the photophysical processes it can...

Figure 2: General catalytic cycle of a photocatalyst in a photoredox organocatalysed reaction. [cat] – photoc...

Figure 3: Structures and names of the most common photocatalysts encountered in the reviewed literature.

Figure 4: General example of a reductive quenching catalytic cycle. [cat] – photocatalyst, [cat]* – photocata...

Figure 5: General example of an oxidative quenching catalytic cycle. [cat] – photocatalyst, [cat]* – photocat...

Scheme 1: Oxidative coupling of aldehydes and amines to amides using acridinium salt photocatalysis.

Figure 6: Biologically active molecules containing a benzamide linkage.

Scheme 2: The photocatalytic reduction of amino acids to produce the corresponding free or protected amines.

Scheme 3: The organocatalysed photoredox base-mediated oxidation of thiols to disulfides.

Scheme 4: C-Terminal modification of peptides and proteins using organophotoredox catalysis.

Scheme 5: The reduction and aryl coupling of aryl halides using a doubly excited photocatalyst (PDI).

Figure 7: Mechanism for the coupling of aryl halides using PDI, which is excited sequentially by two photons.

Scheme 6: The arylation of five-membered heteroarenes using arenediazonium salts under organophotoredox condi...

Scheme 7: The C–H (hetero)arylation of five-membered heterocycles under Eosin Y photocatalysis.

Scheme 8: The C–H sulfurisation of imidazoheterocycles using Eosin B-catalyzed photochemical methods.

Scheme 9: The introduction of the thiocyanate group using Eosin Y photocatalysis.

Scheme 10: Sulfonamidation of pyrroles using oxygen as the terminal oxidant.

Scheme 11: DDQ-catalysed C–H amination of arenes and heteroarenes.

Scheme 12: Photoredox-promoted radical Michael addition reactions of allylic or benzylic carbons.

Figure 8: Proposed mechanistic rationale for the observed chemoselectivities.

Scheme 13: The photocatalytic manipulation of C–H bonds adjacent to amine groups.

Scheme 14: The perylene-catalysed organophotoredox tandem difluoromethylation–acetamidation of styrene-type al...

Figure 9: Examples of biologically active molecules containing highly functionalised five membered heterocycl...

Scheme 15: The [3 + 2]-cycloaddition leading to the formation of pyrroles, through the reaction of 2H-azirines...

Figure 10: Proposed intermediate that determines the regioselectivity of the reaction.

Figure 11: Comparison of possible pathways of reaction and various intermediates involved.

Scheme 16: The acridinium salt-catalysed formation of oxazoles from aldehydes and 2H-azirines.

Scheme 17: The synthesis of oxazolines and thiazolines from amides and thioamides using organocatalysed photor...

Figure 12: Biologically active molecules on the market containing 1,3,4-oxadiazole moieties.

Scheme 18: The synthesis of 1,3,4-oxadiazoles from aldehyde semicarbazones using Eosin Y organophotocatalysis.

Scheme 19: The dimerization of primary thioamides to 1,2,4-thiadiazoles catalysed by the presence of Eosin Y a...

Scheme 20: The radical cycloaddition of o-methylthioarenediazonium salts and substituted alkynes towards the f...

Scheme 21: The dehydrogenative cascade reaction for the synthesis of 5,6-benzofused heterocyclic systems.

Figure 13: Trifluoromethylated version of compounds which have known biological activities.

Scheme 22: Eosin Y-catalysed photoredox formation of 3-substituted benzimidazoles.

Scheme 23: Oxidation of dihydropyrimidines by atmospheric oxygen using photoredox catalysis.

Scheme 24: Photoredox-organocatalysed transformation of 2-substituted phenolic imines to benzoxazoles.

Scheme 25: Visible light-driven oxidative annulation of arylamidines.

Scheme 26: Methylene blue-photocatalysed direct C–H trifluoromethylation of heterocycles.

Scheme 27: Photoredox hydrotrifluoromethylation of terminal alkenes and alkynes.

Scheme 28: Trifluoromethylation and perfluoroalkylation of aromatics and heteroaromatics.

Scheme 29: The cooperative asymmetric and photoredox catalysis towards the functionalisation of α-amino sp3 C–...

Scheme 30: Organophotoredox-catalysed direct C–H amidation of aromatics.

Scheme 31: Direct C–H alkylation of heterocycles using BF3K salts. CFL – compact fluorescent lamp.

Figure 14: The modification of camptothecin, demonstrating the use of the Molander protocol in LSF.

Scheme 32: Direct C–H amination of aromatics using acridinium salts.

Scheme 33: Photoredox-catalysed nucleophilic aromatic substitution of nucleophiles onto methoxybenzene derivat...

Scheme 34: The direct C–H cyanation of aromatics with a focus on its use for LSF.
An overview of recent advances in duplex DNA recognition by small molecules
- Sayantan Bhaduri,
- Nihar Ranjan and
- Dev P. Arya
Beilstein J. Org. Chem. 2018, 14, 1051–1086, doi:10.3762/bjoc.14.93
- significant antibacterial activity against Gram-positive bacterial strains [50]. Several other distamycin analogs were synthesized by replacing one or more pyrrole rings with other heterocycles such as pyrazoles [51], benzofurans [52], thiazoles, thiophenes, imidazole and oxazoles [53] in order to establish a
- reported other conjugates with two thiazoles directly linked via an amide bond, which retained activity to a lesser extent. Baraldi et al. designed and synthesized a novel conjugate 7 by combining naturally occurring antitumor agent distamycin A with the pyrrolo[2,1-c][1,4]benzodiazepine moiety (PBD

Graphical Abstract

Figure 1: A figure showing the hydrogen bonding patterns observed in (a) duplex (b) triplex and (c) quadruple...

Figure 2: (a) Portions of MATα1–MATα2 are shown contacting the minor groove of the DNA substrate. Key arginin...

Figure 3: Chemical structures of naturally occurring and synthetic hybrid minor groove binders.

Figure 4: Synthetic structural analogs of distamycin A by replacing one or more pyrrole rings with other hete...

Figure 5: Pictorial representation of the binding model of pyrrole–imidazole (Py/Im) polyamides based on the ...

Figure 6: Chemical structures of synthetic “hairpin” pyrrole–imidazole (Py/Im) conjugates.

Figure 7: (a) Minor groove complex formation between DNA duplex and 8-ring cyclic Py/Im polyamide (conjugate ...

Figure 8: Telomere-targeting tandem hairpin Py/Im polyamides 23 and 24 capable of recognizing >10 base pairs; ...

Figure 9: Representative examples of recently developed DNA minor groove binders.

Figure 10: Chemical structures of bisbenzamidazoles Hoechst 33258 and 33342 and their synthetic structural ana...

Figure 11: Chemical structures of bisamidines such as diminazene, DAPI, pentamidine and their synthetic struct...

Figure 12: Representative examples of recently developed bisamidine derivatives.

Figure 13: Chemical structures of chromomycin, mithramycin and their synthetic structural analogs 91 and 92.

Figure 14: Chemical structures of well-known naturally occurring DNA binding intercalators.

Figure 15: Naturally occurring indolocarbazole rebeccamycin and its synthetic analogs.

Figure 16: Representative examples of naturally occurring and synthetic derivatives of DNA intercalating agent...

Figure 17: Several recent synthetic varieties of DNA intercalators.

Figure 18: Aminoglycoside (neomycin)–Hoechst 33258/intercalator conjugates.

Figure 19: Chemical structures of triazole linked neomycin dimers and neomycin–bisbenzimidazole conjugates.

Figure 20: Representative examples of naturally occurring and synthetic analogs of DNA binding alkylating agen...

Figure 21: Chemical structures of naturally occurring and synthetic analogs of pyrrolobenzodiazepines.






