Search results
Search for "benzodiazepine" in Full Text gives 32 result(s) in Beilstein Journal of Organic Chemistry.
Total synthesis of asperdinones B, C, D, E and terezine D
Beilstein J. Org. Chem. 2025, 21, 2730–2738, doi:10.3762/bjoc.21.210
- )-configured asperdinones as new members of this small subfamily of 7-prenyltryptophans with appended benzodiazepine-2,5-dione and diketopiperazine units piqued our interest. Herein, we report our efforts toward the total synthesis of asperdinones B, D, C, and E (1–4) and terezine D (6). Considering their
- relatively simple structures, we considered two basic approaches to 4-, 5-, 6-, and 7-prenylated tryptophans as synthetic precursors which could be converted to the intended natural products by cyclization to benzodiazepine-2,5-diones or diketopiperazines. First, is the evident use of tryptophan as a
- ), members of a new class of prenylated 3-indolyl-2,5-benzodiazepine-1,4-diones and diketopiperazine alkaloids, respectively, through Negishi cross-coupling and related C–H functionalization strategies. Importantly, no epimerization was observed during the Negishi coupling or throughout the subsequent steps
Synthesis of triazolo- and tetrazolo-fused 1,4-benzodiazepines via one-pot Ugi–azide and Cu-free click reactions
Beilstein J. Org. Chem. 2025, 21, 2202–2210, doi:10.3762/bjoc.21.167
- /bjoc.21.167 Abstract A one-pot Ugi–azide reaction followed by intramolecular Cu-free azide–alkyne cycloaddition generates a polycyclic scaffold 7 bearing polycyclic triazole, tetrazole, and benzodiazepine rings. This method could be extended for obtaining a more complicated scaffold 8 containing a
- piperazinone ring. Keywords: benzodiazepine; click reaction; multicomponent reaction; one-pot; piperazinone; polycyclic; triazole; tetrazole; Ugi–azide reaction; Introduction Triazole, tetrazole, and benzodiazepine are privileged heterocyclic rings commonly found in drug molecules and functional materials [1
- heterocyclic scaffolds to obtain biologically interesting compounds (Scheme 1) [35][36][37][38][39][40][41]. The development of methods for the synthesis of triazole, tetrazole, piperazinone, and 1,4-benzodiazepine motifs are attractive from both synthetic and medicinal chemistry considerations [42][43][44][45
High-pressure activation for the solvent- and catalyst-free syntheses of heterocycles, pharmaceuticals and esters
Beilstein J. Org. Chem. 2025, 21, 1374–1387, doi:10.3762/bjoc.21.102
- imidazole-based N-heterocyclic carbene (NHC)–CuCl complexes [40]. However, their synthesis is often tainted by the use of toxic reagents and solvents. In addition, when o-phenylenediamine reacts with ketones, the common catalytic methods yield benzodiazepine products [41]. In our case the reaction of o
Silver(I) triflate-catalyzed post-Ugi synthesis of pyrazolodiazepines
Beilstein J. Org. Chem. 2025, 21, 915–925, doi:10.3762/bjoc.21.74
- privileged scaffolds [4][5][6] and are well-known for their use as anxiolytics, hypnotics, and muscle relaxants (Figure 1) [7][8]. Diazepam, sold under the brand name Valium, is among the first marketed medications of the benzodiazepine family [9]. Alprazolam, sold under the brand name Xanax, is a fast
- -acting, potent tranquilizer with moderate duration, belonging to the triazolobenzodiazepine family [10]. It is derived through bioisosteric replacement of the benzodiazepine amide moiety with a triazole ring. Replacements of the benzene ring with thiophene and even with pyrrole are also known. Premazepam
- , which features a pyrrolodiazepine core, has been studied both alone and in combination with diazepam for its antianxiety and sedative effects [11]. Flumazenil comprises an imidazobenzodiazepine scaffold and its intravenous administration is used to treat benzodiazepine overdoses [12][13] and to reverse
Multicomponent reactions driving the discovery and optimization of agents targeting central nervous system pathologies
Beilstein J. Org. Chem. 2024, 20, 3151–3173, doi:10.3762/bjoc.20.261

- organic synthesis, particularly through MCRs, have streamlined the synthesis of benzodiazepines, making it more efficient and environmentally friendly [55]. For example, olanzapine as a benzodiazepine-based clinical drug was appropriately and concisely synthesized via MCR [56][57]. Over the last twenty
- as nitro or azide groups, rather than protected amines. Following the Ugi-4CR, the nitro or azide group is reduced to form the amine, leading to a condensation reaction that results in the formation of the benzodiazepine ring [55]. Pertejo et al. [61] described the diastereselective synthesis of 3
- , isocyanide, and TMSN3, after which the Ugi adduct is treated with an excess of different isocyanates resulting in the corresponding hydantoin derivatives. Finally, under microwave irradiation, the Boc group is removed under acid conditions and the benzodiazepine core is formed after ring closure (Scheme 18
Synthesis of pyrrole-fused dibenzoxazepine/dibenzothiazepine/triazolobenzodiazepine derivatives via isocyanide-based multicomponent reactions
Beilstein J. Org. Chem. 2024, 20, 2870–2882, doi:10.3762/bjoc.20.241
- compounds with antibiotic, antiviral, and anticancer properties that are found in many drugs and natural products [1][2][3][4][5][6]. Pyrroles' biological properties manifest when they are fused to other heterocycles [7][8][9][10][11][12]. Among them, seven-membered heterocycles of the benzodiazepine
- diagnostic and therapeutic procedures [25] or PBOX-6 is a drug for the treatment of depression [26]. Benzothiazepines are known as antidepressants and are molecules with interesting electronic properties (Figure 1) [27]. Accordingly, the synthesis of new derivatives of pyrrole-fused benzodiazepine
- washed with n-hexane and ethyl acetate solvent mixtures (1:3 for 3a and 3d; 2:1 for compounds 5). Representation of distinguished structures of benzodiazepine/benzoxazepine/benzothiazepine with pharmaceutical to electronic applications. The crystal structure of 4h (CCDC 2365305). The DNMR (dynamic
A review of recent advances in electrochemical and photoelectrochemical late-stage functionalization classified by anodic oxidation, cathodic reduction, and paired electrolysis
Beilstein J. Org. Chem. 2024, 20, 2500–2566, doi:10.3762/bjoc.20.214
- efficiently, enabling the LSF of benzodiazepine drugs such as diazepam, halazepam, and prazepam. Mechanistically, the Rh complex activates the C–H bond in phenylpyridine and assists in forming a bond with diphenylphosphine oxide, which, after anodic oxidation, yields the target product (Scheme 39). A chiral
Harnessing unprotected deactivated amines and arylglyoxals in the Ugi reaction for the synthesis of fused complex nitrogen heterocycles
Beilstein J. Org. Chem. 2024, 20, 1758–1766, doi:10.3762/bjoc.20.154
- assayed in our group. Under these conditions, the bis-1,4-benzodiazepines 7 resulting from the reaction of the amino group with the imine on the benzodiazepine were obtained as a single diastereomer. On the basis of the preferred conformation for the 1,4-benzodiazepin-5-ones 5 and 6 [20][22], where the
- order to improve the synthesis of this fused heterocycle, different strategies were tried, e.g., performing the Ugi reaction in different solvents (dichloromethane, ethyl acetate, methanol) to prevent the precipitation of benzodiazepine 5c, conducting the Ugi reaction in boiling methanol or treating the
- benzodiazepine with different bases (caesium carbonate, sodium hydroxide). However, the complete cyclisation to the pyrrolobenzodiazepinone was not achieved, so the described three-step strategy starting from 2-nitrobenzoic acid (Ugi reaction/cyclization to pyrrolidine/reduction sequence) remains as a better
A novel bis-triazole scaffold accessed via two tandem [3 + 2] cycloaddition events including an uncatalyzed, room temperature azide–alkyne click reaction
Beilstein J. Org. Chem. 2022, 18, 1636–1641, doi:10.3762/bjoc.18.175
- reaction conditions were not further optimized (Scheme 1). The structure of tetracyclic product 5a was unequivocally confirmed by 1H and 13C NMR as well as single-crystal X-ray analysis. Compound 5a is representative of the hitherto undescribed bistriazole benzodiazepine scaffold. However, 5,6,7,8
Recent advances in organocatalytic asymmetric aza-Michael reactions of amines and amides
Beilstein J. Org. Chem. 2021, 17, 2585–2610, doi:10.3762/bjoc.17.173
- intramolecular aza-MR by using phase-transfer catalysts. Alkenylated benzamide was used as the substrate in this reaction. The resulting compounds were found to be useful intermediates for the synthesis and development of benzodiazepine-receptor agonists [47]. In 2018, Sallio et al. worked on the same reaction
Effective microwave-assisted approach to 1,2,3-triazolobenzodiazepinones via tandem Ugi reaction/catalyst-free intramolecular azide–alkyne cycloaddition
Beilstein J. Org. Chem. 2021, 17, 678–687, doi:10.3762/bjoc.17.57
- of 1,2,4-triazolobenzodiazepines is the treatment of central nervous system (CNS) disorders. Such drugs as alprazolam and estazolam are used as anxiolytic agents, whereas adinazolam is known as an antidepressant [3]. Benzodiazepine molecules are ligands for GABA-receptors and act as positive
- benzodiazepine skeleton [9]. But these days more and more research groups significantly shift their efforts towards the wide application of a tandem synthetic approach to solve different matters. This approach consists of a combination of several versatile reactions allowing assembling complex molecules just in
- compounds was obtained. Currently, we are working on testing some library representatives for their biological activity. Benzodiazepine-based azolo-containing drugs. Novel potential 1,2,3-triazolobenziadiazepine drugs. Code legend for Ugi products 6 and molecular structure (X-ray analysis) of compound 6aaa
Models of necessity
Beilstein J. Org. Chem. 2020, 16, 1649–1661, doi:10.3762/bjoc.16.137
- molecule is most likely to be protonated or deprotonated and where the aromatic rings should be substituted, all from the Lewis structure. Many cheminformatics practitioners would also be able to write the structure from the SMILES string and medicinal chemists to recognize it as a benzodiazepine and
Multicomponent reactions (MCRs): a useful access to the synthesis of benzo-fused γ-lactams
Beilstein J. Org. Chem. 2019, 15, 1065–1085, doi:10.3762/bjoc.15.104
- hydrazine 114 and sulfur dioxide (Scheme 33). Rearrangement of the so-obtained intermediate 119, through radical 120, would provide oxindole 115. The same acrylamide 113 (R1 = H) has been recently used in another multicomponent synthesis along with CO (23) and benzodiazepine derivative 121 under palladium
- -(2-iodoaryl)acrylamides 113, DABCO·(SO2)2 (69) and hydrazines 114. Proposed mechanism for the preparation of oxindoles 115. Three-component reaction of acrylamide 113, CO (23) and 1,4-benzodiazepine 121. Multicomponent reaction of sulfonylacrylamides 123, aryldiazonium tetrafluoroborates 68 and DABCO
Synthesis of new tricyclic 5,6-dihydro-4H-benzo[b][1,2,4]triazolo[1,5-d][1,4]diazepine derivatives by [3+ + 2]-cycloaddition/rearrangement reactions
Beilstein J. Org. Chem. 2018, 14, 1826–1833, doi:10.3762/bjoc.14.155
- furnishing the tricyclic 1,2,4-triazole-fused 1,4-benzodiazepines. Keywords: 1,4-benzodiazepine (BDZ); cyclization; hydrazones; oxidation; rearrangement; Introduction Heterocyclic compounds comprising a 1,4-benzodiazepine (BDZ) ring have been a topic of continued interest as they exhibit a wide spectrum of
- substances [6]. Thus, for example, chlordiazepoxide (Librium) and the benzodiazepine diazepam (Valium) have been a sedative and hypnotic medication and marked for the treatment of anxiety, insomnia and withdrawal symptoms from alcohol and/or drug abuse by Hoffmann-La Roche since the 1960’s. Benzodiazepines
- achieved when a third heterocycle, especially a 1,2,4-triazolo moiety, was attached to the seven-membered ring as part of 1,4-benzodiazepine [13][14]. Among various reported 1,2,4-triazole-annulated 1,4-benzodiazepines, triazolam (I), estazolam (II), alprazolam (III) and pyrazolam (IV) are prominent
An overview of recent advances in duplex DNA recognition by small molecules
Beilstein J. Org. Chem. 2018, 14, 1051–1086, doi:10.3762/bjoc.14.93
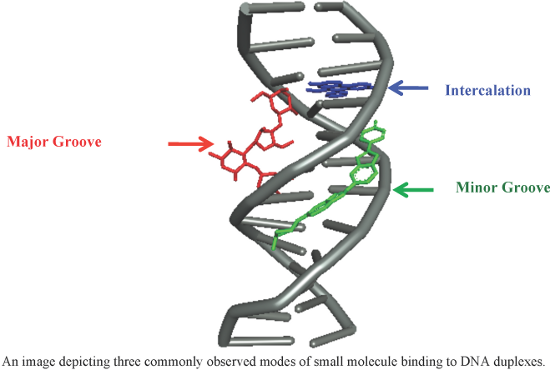
- reported other conjugates with two thiazoles directly linked via an amide bond, which retained activity to a lesser extent. Baraldi et al. designed and synthesized a novel conjugate 7 by combining naturally occurring antitumor agent distamycin A with the pyrrolo[2,1-c][1,4]benzodiazepine moiety (PBD
Sequential Ugi reaction/base-induced ring closing/IAAC protocol toward triazolobenzodiazepine-fused diketopiperazines and hydantoins
Beilstein J. Org. Chem. 2018, 14, 626–633, doi:10.3762/bjoc.14.49
- together all necessary functionalities for further transformations. The Ugi adducts were then subjected to a base-induced ring closing and an intramolecular azide–alkyne cycloaddition reaction in succession to obtain highly fused benzodiazepine frameworks. Keywords: benzodiazepine; 2,5-diketopiperazine
- ) and two ring-closing steps toward triazolobenzodiazepine-fused diketopiperazines and hydantoins. Benzodiazepine derivatives [24][25] form an important class of ‘psychoactive drugs’ which is being extensively used in the treatment of anxiety, insomnia, agitation, seizures, muscle spasms, alcohol
- withdrawal, etc. [26][27][28][29][30][31][32][33][34]. In addition, these azaheterocycles also exhibit anti-inflammatory, antitumor, antiparasitic and anxiolytic activities [35][36][37][38][39][40][41][42]. In particular, 1,4-benzodiazepine derivatives are proposed to serve as a structural mimic of peptide β
Diastereoselective auxiliary- and catalyst-controlled intramolecular aza-Michael reaction for the elaboration of enantioenriched 3-substituted isoindolinones. Application to the synthesis of a new pazinaclone analogue
Beilstein J. Org. Chem. 2018, 14, 593–602, doi:10.3762/bjoc.14.46
- provide 3-substituted isoindolinones in good yields and diastereomeric excesses. This methodology was applied to the asymmetric synthesis of a new pazinaclone analogue which is of interest in the field of benzodiazepine-receptor agonists. Keywords: asymmetric organocatalysis; Aza-Michael reaction; phase
- sedative-hypnotic activities used for the treatment of anxiety by acting as partial agonists at GABAA (γ-aminobutyric acid type A) benzodiazepine receptors [17]. All these studies have highlighted a strong correlation between the compound pharmacological activities and the absolute configurations of their
- then turned our attention to the asymmetric synthesis of a new pazinaclone analogue, which could be of particular interest in the field of benzodiazepine-receptor agonists [8][9][10][11][12][13][14][15][16][17]. Indeed, pazinaclone produces its sedative and anxiolytic effects by acting as a partial
Reagent-controlled regiodivergent intermolecular cyclization of 2-aminobenzothiazoles with β-ketoesters and β-ketoamides
Beilstein J. Org. Chem. 2017, 13, 2739–2750, doi:10.3762/bjoc.13.270
- been found to be biologically active antagonists of adenosine receptors [82], inhibitors of cyclic-AMP-diphosphoesterase [83], and benzodiazepine receptor ligands [84][85]. Reported methods to access this structural motif include annulation between an aromatic amine and acid chloride [86] or via aza
Preparation of imidazo[1,2-a]-N-heterocyclic derivatives with gem-difluorinated side chains
Beilstein J. Org. Chem. 2017, 13, 2115–2121, doi:10.3762/bjoc.13.208
- formation inhibitors, GABA and benzodiazepine receptor agonists, and cardiotonic agents [7][8][9][10]. Further, the biological activities of imidazo[1,2-a]pyridines proved to be strongly depending upon the nature of substituents at C2 and C3 positions. For instance, the 3-aroylimidazo[1,2-a]pyridines 1c
Selective and eco-friendly procedures for the synthesis of benzimidazole derivatives. The role of the Er(OTf)3 catalyst in the reaction selectivity
Beilstein J. Org. Chem. 2016, 12, 2410–2419, doi:10.3762/bjoc.12.235

- reaction of the diamine with ketones affords benzodiazepine as products [29][30]. Next, we investigated the general applicability of our method in the reaction of o-phenylenediamine with several substituted aldehydes using the optimized conditions towards products 1a or 1b, respectively. For this, the best
Multicomponent reactions: A simple and efficient route to heterocyclic phosphonates
Beilstein J. Org. Chem. 2016, 12, 1269–1301, doi:10.3762/bjoc.12.121
- and 1,5-benzodiazepine 95, respectively (Scheme 21). 2.2.7 Isoquinolone-1-phosphonates: From Lewis acid catalyzed 6-endo-dig cyclizations of acetylenic Kabachnik–Fields adducts: A modified Kabachnik–Fields reaction for the synthesis of isoquinoline-1-phosphonate derivatives is the three-component
Bifunctional phase-transfer catalysis in the asymmetric synthesis of biologically active isoindolinones
Beilstein J. Org. Chem. 2015, 11, 2591–2599, doi:10.3762/bjoc.11.279
- , some of which are shown in Figure 1 [4][5][6][7][8]. For example, the enantioenriched isoindolinones 1 and 2 are benzodiazepine-receptor agonists for the treatment of anxiety [4][5][6][7]. Compound (S)-3, developed by Belliotti et al. and known as PD172938, is a potent dopamine D4 ligand [8] while
- mesyl chloride and the subsequent displacement with 1-(3,4-(dimethylphenyl)pyperazine (15) gave 3, the potent dopamine D4 ligand (S)-PD172938, in high overall yield (51%), with 95% ee (Scheme 4). Then, the amide 16, which is of particular interest in the field of benzodiazepine-receptor agonists, was
The synthesis of active pharmaceutical ingredients (APIs) using continuous flow chemistry
Beilstein J. Org. Chem. 2015, 11, 1194–1219, doi:10.3762/bjoc.11.134

- a stream of dilute hydrochloric acid and passed through an inductively heated tubular reactor maintained at 140 °C to furnish benzodiazepine 125 in 88% yield after 30 h processing time. The flow synthesis of olanzapine (121) was completed by directing a mixture of benzodiazepine 125 and N
An overview of the synthetic routes to the best selling drugs containing 6-membered heterocycles
Beilstein J. Org. Chem. 2013, 9, 2265–2319, doi:10.3762/bjoc.9.265

Synthesis and characterization of novel bioactive 1,2,4-oxadiazole natural product analogs bearing the N-phenylmaleimide and N-phenylsuccinimide moieties
Beilstein J. Org. Chem. 2013, 9, 2202–2215, doi:10.3762/bjoc.9.259

- antirhinovirals [11], benzodiazepine receptor partial agonists [12], anti-inflammatory [13], muscarinic agonists [14], serotoninergic (5-HT3) antagonists [15], and growth hormone secretagogues [16]. The maleimide motif is also a useful five-membered heterocycle in pharmacological chemistry. Kratz et al














































































































































































































































































































































































































































































































































































