Search results
Search for "hydrocarbons" in Full Text gives 153 result(s) in Beilstein Journal of Organic Chemistry.
Development of N-F fluorinating agents and their fluorinations: Historical perspective
- Teruo Umemoto,
- Yuhao Yang and
- Gerald B. Hammond
Beilstein J. Org. Chem. 2021, 17, 1752–1813, doi:10.3762/bjoc.17.123

- powerful fluorinating agent with a high fluorine content, F: 103 g/1 kg (comparison; 54 g/1 kg for Selectfluor). 1-25. N-Fluoro-2,4-dinitroimidazole In 1998, Laali et al. reported the fluorination of polycyclic aromatic hydrocarbons (PAHs) with N-fluoro-2,4-dinitroimidazole (25-1) [87] (Scheme 56). This

Graphical Abstract

Scheme 1: Fluorination with N-F amine 1-1.

Scheme 2: Preparation of N-F amine 1-1.

Scheme 3: Reactions of N-F amine 1-1.

Scheme 4: Synthesis of N-F perfluoroimides 2-1 and 2-2.

Scheme 5: Synthesis of 1-fluoro-2-pyridone (3-1).

Scheme 6: Fluorination with 1-fluoro-2-pyridone (3-1).

Figure 1: Synthesis of N-F sulfonamides 4-1a–g.

Scheme 7: Fluorination with N-F reagent 4-1b,c,f.

Scheme 8: Fluorination of alkenyllithiums with N-F 4-1h.

Scheme 9: Synthesis of N-fluoropyridinium triflate (5-4a).

Scheme 10: Synthetic methods for N-F-pyridinium salts.

Figure 2: Synthesis of various N-fluoropyridinium salts. Note: athis yield was the one by the improved method...

Scheme 11: Fluorination power order of N-fluoropyridinium salts.

Scheme 12: Fluorinations with N-F salts 5-4.

Scheme 13: Fluorination of Corey lactone 5-7 with N-F-bis(methoxymethyl) salt 5-4l.

Scheme 14: Fluorination with NFPy.

Scheme 15: Synthesis of the N-F reagent, N-fluoroquinuclidinium fluoride (6-1).

Scheme 16: Fluorinations achieved with N-F fluoride 6-1.

Scheme 17: Synthesis of N-F imides 7-1a–g.

Scheme 18: Fluorination with (CF3SO2)2NF, 7-1a.

Scheme 19: Fluorination reactions of various substrates with 7-1a.

Scheme 20: Synthesis of N-F triflate 8-1.

Scheme 21: Synthesis of chiral N-fluoro sultams 9-1 and 9-2.

Scheme 22: Fluorination with chiral N-fluoro sultams 9-1 and 9-2.

Scheme 23: Synthesis of saccharin-derived N-fluorosultam 10-2.

Scheme 24: Fluorination with N-fluorosultam 10-2.

Scheme 25: Synthesis of N-F reagent 11-2.

Scheme 26: Fluorination with N-F reagent 11-2.

Scheme 27: Synthesis and reaction of N-fluorolactams 12-1.

Scheme 28: Synthesis of NFOBS 13-2.

Scheme 29: Fluorination with NFOBS 13-2.

Scheme 30: Synthesis of NFSI (14-2).

Scheme 31: Fluorination with NFSI 14-2.

Scheme 32: Synthesis of N-fluorosaccharin (15-1) and N-fluorophthalimide (15-2).

Scheme 33: Synthesis of N-F salts 16-3.

Scheme 34: Fluorination with N-F salts 16-3.

Figure 3: Monofluorination with Selectfluor (16-3a).

Figure 4: Difluorination with Selectfluor (16-3a).

Scheme 35: Transfer fluorination of Selectfluor (16-3a).

Scheme 36: Fluorination of substrates with Selectfluor (16-3a).

Scheme 37: Synthesis of chiral N-fluoro-sultam 17-2.

Scheme 38: Asymmetric fluorination with chiral 17-2.

Figure 5: Synthesis of Zwitterionic N-fluoropyridinium salts 18-2a–h.

Scheme 39: Fluorinating power order of zwitterionic N-fluoropyridinium salts.

Scheme 40: Fluorination with zwitterionic 18-2.

Scheme 41: Activation of salt 18-2h with TfOH.

Scheme 42: Synthesis of NFTh, 19-2.

Scheme 43: Fluorination with NFTh, 19-2.

Scheme 44: Synthesis of 3-fluorobenzo-1,2,3-oxathiazin-4-one 2,2-dioxide (20-2).

Scheme 45: Fluorination with 20-2.

Scheme 46: Synthesis of N-F amide 21-3.

Scheme 47: Fluorination with N-F amide 21-2.

Scheme 48: Synthesis of N,N’-difluorodiazoniabicyclo[2.2.2]octane salts 22-1.

Scheme 49: One-pot synthesis of N,N’-difluoro-1,4-diazoniabicyclo[2.2.2]octane bistetrafluoroborate salt (22-1d...

Figure 6: Fluorination of anisole with 22-1a, d, e.

Scheme 50: Fluorination with N,N’-diF bisBF4 22-1d.

Scheme 51: Synthesis of bis-N-F reagents 23-1–5.

Scheme 52: Fluorination with 23-2, 4, 5.

Figure 7: Synthesis of N,N’-difluorobipyridinium salts 24-2.

Figure 8: Controlled fluorination of N,N’-diF 24-2.

Scheme 53: Fluorinating power of N,N’-diF salts 24-2 and N-F salt 5-4a.

Scheme 54: Fluorination reactions with SynfluorTM (24-2b).

Scheme 55: Additional fluorination reactions with SynfluorTM (24-2b).

Scheme 56: Synthesis of N-F 25-1.

Scheme 57: Fluorination of polycyclic aromatics with 25-1.

Scheme 58: Synthesis of 26-1 and dimethyl analog 26-2.

Scheme 59: Fluorination with reagents 26-1, 26-2, 1-1, and 26-3.

Scheme 60: Synthesis of N-F reagent 27-2.

Scheme 61: Synthesis of chiral N-F reagents 27-6.

Scheme 62: Synthesis of chiral N-F 27-7–9.

Scheme 63: Asymmetric fluorination with 27-6.

Scheme 64: Synthesis of chiral N-F reagents 28-3.

Scheme 65: Asymmetric fluorination with 28-3.

Scheme 66: Synthesis of chiral N-F reagents 28-7.

Figure 9: Asymmetric fluorination with 28-7.

Scheme 67: In situ formation of N-fluorinated cinchona alkaloids with SelectfluorTM.

Scheme 68: Asymmetric fluorination with N-F alkaloids formed in situ.

Scheme 69: Synthesis of N-fluorocinchona alkaloids with Selectfluor.

Scheme 70: Asymmetric fluorination with 30-1–4.

Scheme 71: Transfer fluorination from various N-F reagents.

Figure 10: Asymmetric fluorination of silyl enol ethers.

Scheme 72: Synthesis of N-fluoro salt 32-2.

Scheme 73: Reactivity of N-fluorotriazinium salt 32-2.

Scheme 74: Synthesis of bulky N-fluorobenzenesulfonimide NFBSI 33-3.

Scheme 75: Comparison of NFSI and NFBSI.

Scheme 76: Synthesis of p-substituted N-fluorobenzenesulfonimides 34-3.

Figure 11: Asymmetric fluorination with 34-3 and a chiral catalyst 34-4.

Scheme 77: 1,4-Fluoroamination with Selecfluor and a chiral catalyst.

Figure 12: Asymmetric fluoroamination with 35-5a, b.

Scheme 78: Synthesis of Selectfluor analogs 35-5a, b.

Scheme 79: Synthesis of chiral dicationic DABCO-based N-F reagents 36-5.

Scheme 80: Asymmetric fluorocyclization with chiral 36-5b.

Scheme 81: Synthesis of chiral 37-2a,b.

Scheme 82: Asymmetric fluorination with chiral 37-2a,b.

Scheme 83: Asymmetric fluorination with chiral 37-2b.

Scheme 84: Reaction of indene with chiral 37-2a,b.

Scheme 85: Synthesis of Me-NFSI, 38-2.

Scheme 86: Fluorination of active methine compounds with Me-NFSI.

Scheme 87: Fluorination of malonates with Me-NFSI.

Scheme 88: Fluorination of keto esters with Me-NFSI.

Scheme 89: Synthesis of N-F 39-3 derived from the ethylene-bridged Tröger’s base.

Scheme 90: Fluorine transfer from N-F 39-3.

Scheme 91: Fluorination with N-F 39-3.

Scheme 92: Synthesis of SelectfluorCN.

Scheme 93: Bistrifluoromethoxylation of alkenes using SelectfluorCN.

Figure 13: Synthesis of NFAS 41-2.

Scheme 94: Radical fluorination with different N-F reagents.

Scheme 95: Radical fluorination of alkenes with NFAS 41-2.

Scheme 96: Radical fluorination of alkenes with NFAS 41-2f.

Scheme 97: Decarboxylative fluorination with NFAS 41-2a,f.

Scheme 98: Fluorine plus detachment (FPD).

Figure 14: FPD values of representative N-F reagents in CH2Cl2 and CH3CN (in parentheses). Adapted with permis...

Scheme 99: N-F homolytic bond dissociation energy (BDE).

Figure 15: BDE values of representative N-F reagents in CH3CN. Adapted with permission from ref. [127]. Copyright 2...

Figure 16: Quantitative reactivity scale for popular N-F reagents. Adapted with permission from ref. [138], publish...

Scheme 100: SET and SN2 mechanisms.

Scheme 101: Radical clock reactions.

Scheme 102: Reaction of potassium enolate of citronellic ester with N-F reagents, 10-1, NFSI, and 8-1.

Scheme 103: Reaction of compound IV with Selectfluor (OTf) and NFSI.

Scheme 104: Reaction of TEMPO with Selecfluor.
Design, synthesis and photophysical properties of novel star-shaped truxene-based heterocycles utilizing ring-closing metathesis, Clauson–Kaas, Van Leusen and Ullmann-type reactions as key tools
- Shakeel Alvi and
- Rashid Ali
Beilstein J. Org. Chem. 2021, 17, 1374–1384, doi:10.3762/bjoc.17.96

- Shakeel Alvi Rashid Ali Department of Chemistry, Jamia Millia Islamia, Jamia Nagar, Okhla, New Delhi-110025, India; Phone: +91-7011867613 10.3762/bjoc.17.96 Abstract Design, synthesis and properties of polycyclic aromatic hydrocarbons (PAHs) has historically attracted a considerable

Graphical Abstract

Scheme 1: Retrosynthetic pathways to the pyrrole-based C3-symmetric truxene derivative 6.

Scheme 2: Synthesis of tripyrrolotruxene 6 via cyclotrimerization and RCM as crucial steps.

Scheme 3: Synthesis of star-shaped molecule 6 utilizing the Clauson–Kaas pyrrole strategy.

Scheme 4: Synthesis of truxene derivative 6 involving Ullmann-type cross-coupling reaction.

Scheme 5: Synthesis of imidazole and benzimidazole containing truxene derivatives 14 and 16.

Scheme 6: Construction of truxene-based di- and trioxazole derivatives 21 and 20.

Scheme 7: Synthesis of benzene-bridged rings containing trioxazolotruxene system 25.

Figure 1: Normalized absorption (left); fluorescence spectra (right) of the synthesized truxene derivatives (...
Icilio Guareschi and his amazing “1897 reaction”
- Gian Cesare Tron,
- Alberto Minassi,
- Giovanni Sorba,
- Mara Fausone and
- Giovanni Appendino
Beilstein J. Org. Chem. 2021, 17, 1335–1351, doi:10.3762/bjoc.17.93
- “Guareschi 1897 reaction”, is one of the most unusual reactions in organic chemistry, involving the radical-mediated paradoxical aerobic generation of hydrocarbons in near-neutral water solution. A discussion of the mechanism of this amazing reaction, the only metal-free process that generates hydrocarbons
- , inorganic, and analytical branches and systematically crosses the divide between pure and applied science as well as between the history of chemistry and the personal contributions to its development. Keywords: Guareschi; history of chemistry; hydrocarbons; name reactions; pyridine; Introduction Modern
- so-called “1897 reaction” is not well known [7], despite that the unique mechanistic aspects make it one of the most unusual reactions in organic chemistry, involving the paradoxical radical-mediated aerobic generation of hydrocarbons in water under only slightly basic conditions [8]. Also

Graphical Abstract

Figure 1: Icilio Guareschi (1847–1918). (Source: Annali della Reale Accademia di Agricoltura di Torino 1919, ...

Scheme 1: Vitamin B6 (pyridoxine, 1), gabapentin (2), and thymol (3).

Figure 2: Baliatico (Nursing) by Francesco Scaramuzza (275 cm × 214 cm, Parma, Complesso Museale della Pilott...

Figure 3: Schiff’s fictitious report on the foundation of the Gazzetta Chimica Italiana (Image reproduced fro...

Scheme 2: Reaction of thymol (3) with chloroform under the basic conditions of the Guareschi–Lustgarten react...

Figure 4: The chemistry building of Turin University in a historical picture. Note, that one of the “mysterio...

Scheme 3: Triacetonamine (6) and the related compounds phorone (7), α-eucaine (8), and tropinone (9).

Scheme 4: Taxonomy of the Guareschi pyridone syntheses.

Scheme 5: The catalytic cycle of the “1897 reaction”.

Scheme 6: Resonance forms of the radical 10.

Figure 5: The wet chamber used by Guareschi to restore parchments (Gorrini, G. L'incendio della R. Biblioteca...

Figure 6: The Guareschi mask. (Servizio Chimico Militare. L'opera di Icilio Guareschi precursore della masche...

Figure 7: Guareschi’s bust at the Dipartimento di Scienza e Tecnologia del Farmaco of Turin University. Permi...
Synthetic reactions driven by electron-donor–acceptor (EDA) complexes
- Zhonglie Yang,
- Yutong Liu,
- Kun Cao,
- Xiaobin Zhang,
- Hezhong Jiang and
- Jiahong Li
Beilstein J. Org. Chem. 2021, 17, 771–799, doi:10.3762/bjoc.17.67
- perfluoroalkyl iodide form a halogen-bond adduct, and then light-induced electron transfer happens in order to give a perfluoroalkyl radical. The protocol can realize alkene- and alkyne iodide perfluoroalkylation and C–H perfluoroalkylation of electron-rich heteroaromatic hydrocarbons, providing a novel protocol
- conditions, more attention has been paid to this efficient strategy for C–C-bond formation. In 2015, Yu and colleagues [27] proposed a method for direct C–H trifluoromethylation of aromatic hydrocarbons through an EDA complex. Trifluoromethylated product 61 was synthesized by employing tryptamine derivative

Graphical Abstract

Scheme 1: The electron transfer process in EDA complexes.

Scheme 2: Synthesis of benzo[b]phosphorus oxide 3 initiated by an EDA complex.

Scheme 3: Mechanism of the synthesis of quinoxaline derivative 7.

Scheme 4: Synthesis of imidazole derivative 10 initiated by an EDA complex.

Scheme 5: Synthesis of sulfamoylation product 12 initiated by an EDA complex.

Scheme 6: Mechanism of the synthesis of sulfamoylation product 12.

Scheme 7: Synthesis of indole derivative 22 initiated by an EDA complex.

Scheme 8: Synthesis of perfluoroalkylated pyrimidines 26 initiated by an EDA complex.

Scheme 9: Synthesis of phenanthridine derivative 29 initiated by an EDA complex.

Scheme 10: Synthesis of cis-tetrahydroquinoline derivative 32 initiated by an EDA complex.

Scheme 11: Mechanism of the synthesis of cis-tetrahydroquinoline derivative 32.

Scheme 12: Synthesis of phenanthridine derivative 38 initiated by an EDA complex.

Scheme 13: Synthesis of spiropyrroline derivative 40 initiated by an EDA complex.

Scheme 14: Synthesis of benzothiazole derivative 43 initiated by an EDA complex.

Scheme 15: Synthesis of perfluoroalkyl-s-triazine derivative 45 initiated by an EDA complex.

Scheme 16: Synthesis of indoline derivative 47 initiated by an EDA complex.

Scheme 17: Mechanism of the synthesis of spirocyclic indoline derivative 47.

Scheme 18: Synthesis of cyclobutane product 50 initiated by an EDA complex.

Scheme 19: Mechanism of the synthesis of spirocyclic indoline derivative 50.

Scheme 20: Synthesis of 1,3-oxazolidine compound 59 initiated by an EDA complex.

Scheme 21: Synthesis of trifluoromethylated product 61 initiated by an EDA complex.

Scheme 22: Synthesis of indole alkylation product 64 initiated by an EDA complex.

Scheme 23: Synthesis of perfluoroalkylation product 67 initiated by an EDA complex.

Scheme 24: Synthesis of hydrotrifluoromethylated product 70 initiated by an EDA complex.

Scheme 25: Synthesis of β-trifluoromethylated alkyne product 71 initiated by an EDA complex.

Scheme 26: Mechanism of the synthesis of 2-phenylthiophene derivative 74.

Scheme 27: Synthesis of allylated product 80 initiated by an EDA complex.

Scheme 28: Synthesis of trifluoromethyl-substituted alkynyl product 84 initiated by an EDA complex.

Scheme 29: Synthesis of dearomatized fluoroalkylation product 86 initiated by an EDA complex.

Scheme 30: Mechanism of the synthesis of dearomatized fluoroalkylation product 86.

Scheme 31: Synthesis of C(sp3)–H allylation product 91 initiated by an EDA complex.

Scheme 32: Synthesis of perfluoroalkylation product 93 initiated by an EDA complex.

Scheme 33: Synthesis of spirocyclic indolene derivative 95 initiated by an EDA complex.

Scheme 34: Synthesis of perfluoroalkylation product 97 initiated by an EDA complex.

Scheme 35: Synthesis of alkylated indole derivative 100 initiated by an EDA complex.

Scheme 36: Mechanism of the synthesis of alkylated indole derivative 100.

Scheme 37: Synthesis of arylated oxidized indole derivative 108 initiated by an EDA complex.

Scheme 38: Synthesis of 4-ketoaldehyde derivative 111 initiated by an EDA complex.

Scheme 39: Mechanism of the synthesis of 4-ketoaldehyde derivative 111.

Scheme 40: Synthesis of perfluoroalkylated olefin 118 initiated by an EDA complex.

Scheme 41: Synthesis of alkylation product 121 initiated by an EDA complex.

Scheme 42: Synthesis of acylation product 123 initiated by an EDA complex.

Scheme 43: Mechanism of the synthesis of acylation product 123.

Scheme 44: Synthesis of trifluoromethylation product 126 initiated by an EDA complex.

Scheme 45: Synthesis of unnatural α-amino acid 129 initiated by an EDA complex.

Scheme 46: Synthesis of thioether derivative 132 initiated by an EDA complex.

Scheme 47: Synthesis of S-aryl dithiocarbamate product 135 initiated by an EDA complex.

Scheme 48: Mechanism of the synthesis of S-aryl dithiocarbamate product 135.

Scheme 49: Synthesis of thioether product 141 initiated by an EDA complex.

Scheme 50: Mechanism of the synthesis of borate product 144.

Scheme 51: Synthesis of boronation product 148 initiated by an EDA complex.

Scheme 52: Synthesis of boration product 151 initiated by an EDA complex.

Scheme 53: Synthesis of boronic acid ester derivative 154 initiated by an EDA complex.

Scheme 54: Synthesis of β-azide product 157 initiated by an EDA complex.

Scheme 55: Decarboxylation reaction initiated by an EDA complex.

Scheme 56: Synthesis of amidated product 162 initiated by an EDA complex.

Scheme 57: Synthesis of diethyl phenylphosphonate 165 initiated by an EDA complex.

Scheme 58: Mechanism of the synthesis of diethyl phenylphosphonate derivative 165.

Scheme 59: Synthesis of (Z)-2-iodovinyl phenyl ether 168 initiated by an EDA complex.

Scheme 60: Mechanism of the synthesis of (Z)-2-iodovinyl phenyl ether derivative 168.

Scheme 61: Dehalogenation reaction initiated by an EDA complex.
Synthesis of dibenzosuberenone-based novel polycyclic π-conjugated dihydropyridazines, pyridazines and pyrroles
- Ramazan Koçak and
- Arif Daştan
Beilstein J. Org. Chem. 2021, 17, 719–729, doi:10.3762/bjoc.17.61
- exhibit photophysical properties such as photosensitization [13], fluorescence, and aggregation-induced emission (AIE) [14][15][16]. π-Conjugated polycyclic hydrocarbons (CPHs) containing polycyclic heteroaromatic molecules (PHAs) and aza-polycyclic aromatic hydrocarbons (aza-PAHs) have been attracting

Graphical Abstract

Figure 1: Structures of dibenzosuberenone 1 and pyridazine and pyrrole derivatives.

Figure 2: Structures of s-tetrazines 2a–l.

Scheme 1: Inverse electron-demand Diels–Alder reactions of dibenzosuberenone (1) with tetrazines 2a–l.

Scheme 2: Inverse electron-demand Diels–Alder reactions between dibenzosuberenone 1 and tetrazines 2ka and 2lb...

Scheme 3: Proposed reaction mechanism for the formation of dibenzosuberenone derivatives 3 and 4.

Scheme 4: Proposed mechanism for the formation of 5l.

Scheme 5: Oxidation of dihydropyridazines 3a–f. All reactions were carried in CH2Cl2 at room temperature (4e:...

Scheme 6: Synthesis of pyrrole 10a. a1.34 mmol 4a, Zinc (for 10aa: 6.68 mmol, for 10ab: 13.36 mmol), 10 mL gl...

Scheme 7: Synthesis of pyrrole 10b. a1.21 mmol 4b, 12.10 mmol Zinc, 118 °C, 2 h. b1.13 mmol 10ba, 1.69 mmol K...

Scheme 8: Synthesis of p-quinone methides 13–16. a1.77 mmol 11, 1.77 mmol 2, 5 mL toluene, 80 °C (13a: overni...

Scheme 9: Proposed mechanism for the formation of 13.

Figure 3: UV–vis spectra of 3c–f and 3k in CH3CN at rt (c = 5 μM).

Figure 4: Fluorescence spectra of 3c–f and 3k in CH3CN at rt (c = 5 μM).

Figure 5: Ambient (top) and fluorescence (bottom, under 365 nm UV light) images of 3c–f and 3k in CH3CN.
Valorisation of plastic waste via metal-catalysed depolymerisation
- Francesca Liguori,
- Carmen Moreno-Marrodán and
- Pierluigi Barbaro
Beilstein J. Org. Chem. 2021, 17, 589–621, doi:10.3762/bjoc.17.53

- ], catalytic cracking [56] and gasification [57]. These are usually unselective, high-temperature treatments (300–1000 °C) that may efficiently provide light hydrocarbons or small molecules [58][59]. Chemolytic processes, wherein a chemical reagent is used to achieve depolymerisation, mainly involve solvolysis
- packaging, squeezable bottles, plastic bags and medical devices [140]. PEs (except cross-linked samples) are partially soluble in (hot) aromatic hydrocarbons or in chlorinated solvents [141]. Depolymerisation of PE by catalytic pyrolysis or cracking into liquid fuels was recently reviewed [67][142]. Most of
- these processes are promoted by heterogeneous acid catalysts (e.g., zeolites, alumina, silica) and are usually unselective, resulting in a broad distribution of gas (C3 and C4 hydrocarbons), liquids (cycloparaffins, oligomers, aromatics) and solid products (char, coke) as a consequence of the random

Graphical Abstract

Figure 1: Potential classification of plastic recycling processes. The area covered by the present review is ...

Figure 2: EG produced during glycolytic depolymerisation of PET using DEG + DPG as solvent and titanium(IV) n...

Scheme 1: Simplified representation of the conversion of 1,4-PBD to C16–C44 macrocycles using Ru metathesis c...

Figure 3: Main added-value monomers obtainable by catalytic depolymerisation of PET via chemolytic methods.

Scheme 2: Hydrogenolytic depolymerisation of PET by ruthenium complexes.

Scheme 3: Depolymerisation of PET via catalytic hydrosilylation by Ir(III) pincer complex.

Scheme 4: Catalytic hydrolysis (top) and methanolysis (bottom) reactions of PET.

Scheme 5: Depolymerisation of PET by glycolysis with ethylene glycol.

Figure 4: Glycolysis of PET: evolution of BHET yield over time, with and without zinc acetate catalyst (196 °...

Scheme 6: Potential activated complex for the glycolysis reaction of PET catalysed by metallated ILs and evol...

Scheme 7: One-pot, two-step process for PET repurposing via chemical recycling.

Scheme 8: Synthetic routes to PLA.

Scheme 9: Structures of the zinc molecular catalysts used for PLA-methanolysis in various works. a) See [265], b) ...

Scheme 10: Depolymerisation of PLLA by Zn–N-heterocyclic carbene complex.

Scheme 11: Salalen ligands.

Scheme 12: Catalytic hydrogenolysis of PLA.

Scheme 13: Catalytic hydrosilylation of PLA.

Scheme 14: Hydrogenative depolymerisation of PBT and PCL by molecular Ru catalysts.

Scheme 15: Glycolysis reaction of PCT by diethylene glycol.

Scheme 16: Polymerisation–depolymerisation cycle of 3,4-T6GBL.

Scheme 17: Polymerisation–depolymerisation cycle of 2,3-HDB.

Scheme 18: Hydrogenative depolymerisation of PBPAC by molecular Ru catalysts.

Scheme 19: Catalytic hydrolysis (top), alcoholysis (middle) and aminolysis (bottom) reactions of PBPAC.

Scheme 20: Hydrogenative depolymerisation of PPC (top) and PEC (bottom) by molecular Ru catalysts.

Scheme 21: Polymerisation-depolymerisation cycle of BEP.

Scheme 22: Hydrogenolysis of polyamides using soluble Ru catalysts.

Scheme 23: Catalytic depolymerisation of epoxy resin/carbon fibres composite.

Scheme 24: Depolymerisation of polyethers with metal salt catalysts and acyl chlorides.

Scheme 25: Proposed mechanism for the iron-catalysed depolymerisation reaction of polyethers. Adapted with per...
Helicene synthesis by Brønsted acid-catalyzed cycloaromatization in HFIP [(CF3)2CHOH]
- Takeshi Fujita,
- Noriaki Shoji,
- Nao Yoshikawa and
- Junji Ichikawa
Beilstein J. Org. Chem. 2021, 17, 396–403, doi:10.3762/bjoc.17.35
- gram-scale syntheses of higher-order helicenes, double helical helicenes, and heterohelicenes. Keywords: acid; catalyst; cyclization; fluoroalcohol; helicenes; Introduction Helicenes are a class of polycyclic aromatic hydrocarbons (PAH) that consist of ortho-fused aromatic rings arranged in a helical
- ], which are rarely found in planar aromatic hydrocarbons (e.g., acenes and phenacenes). Therefore, the wide breadth of applications of helicenes as organic optical materials make them interesting synthetic targets. Although several methods for the synthesis of helicenes have been reported, there are still

Graphical Abstract

Scheme 1: Conventional methods for the synthesis of helicenes.

Scheme 2: Brønsted acid-catalyzed cycloaromatization of biaryls bearing an acetal moiety.

Scheme 3: Two strategies for the helicene synthesis via Suzuki–Miyaura coupling/cycloaromatization sequence.

Scheme 4: Synthesis of (a) [5]helicene and (b) [6]helicene.

Scheme 5: Synthesis of helicenes with double helical structures.

Scheme 6: Synthesis of hetero[4]-, [5]-, and [6]helicenes.
On the mass spectrometric fragmentations of the bacterial sesterterpenes sestermobaraenes A–C
- Anwei Hou and
- Jeroen S. Dickschat
Beilstein J. Org. Chem. 2020, 16, 2807–2819, doi:10.3762/bjoc.16.231
- investigation of the electron impact mass spectrometry (EIMS) fragmentation reactions of these sesterterpene hydrocarbons. Keywords: isotopes; mass spectrometry; reaction mechanisms; sesterterpenes; Streptomyces mobaraensis; Introduction The sestermobaraenes A–F (1–6) and sestermobaraol (7) are a series of
- initiate a cationic cyclisation cascade, leading to structurally highly complex and usually polycyclic terpenes in just one enzymatic transformation. The initially formed products are non-functionalised terpene hydrocarbons or, if the terminal cationic intermediate of the cyclisation cascade is trapped by

Graphical Abstract

Figure 1: The structures of the bacterial sesterterpenes sestermobaraenes A–F (1–6) and sestermobaraol (7) fr...

Figure 2: Position-specific mass shift analyses for 1. Carbons that contribute fully to the formation of a fr...

Scheme 1: The EIMS fragmentation mechanisms for 1 explaining the formation of the fragment ions at m/z = 325,...

Scheme 2: The EIMS fragmentation mechanisms for 1 explaining the formation of fragment ions at m/z = 206 and ...

Figure 3: Position-specific mass shift analyses for 2. The carbons that contribute fully to the formation of ...

Scheme 3: The EIMS fragmentation mechanisms for 2 explaining the formation of the fragment ions at m/z = 325,...

Scheme 4: The EIMS fragmentation mechanisms for 2 explaining the formation of the fragment ions at m/z = 203 ...

Figure 4: The position-specific mass shift analyses for 3. Carbons that contribute fully to the formation of ...

Scheme 5: The EIMS fragmentation mechanisms for 3 explaining the formation of the fragment ions at m/z = 325,...

Scheme 6: The EIMS fragmentation mechanisms for 3 explaining the formation of the fragment ion at m/z = 206 a...
Water-soluble host–guest complexes between fullerenes and a sugar-functionalized tribenzotriquinacene assembling to microspheres
- Si-Yuan Liu,
- Xin-Rui Wang,
- Man-Ping Li,
- Wen-Rong Xu and
- Dietmar Kuck
Beilstein J. Org. Chem. 2020, 16, 2551–2561, doi:10.3762/bjoc.16.207

- ][20]. Tribenzotriquinacene (TBTQ) and its derivatives, owing to their unique rigid, C3v-symmetric, concave-convex trifuso-triindane skeleton that consists of three perfectly orthogonally oriented indane wings, bear a similar potential as molecular hosts. TBTQ hydrocarbons are chemically stable and
- shallow cavity of the parent TBTQ hydrocarbons, thus allowing for the inclusion of large guest molecules, such as the fullerenes. Several TBTQ derivatives with extended cavities have been developed by us and other groups. Volkmer et al. designed a series of novel TBTQ-based receptors, 1–3, and studied

Graphical Abstract

Figure 1: Selected TBTQ derivatives 1–5 that bind fullerenes in host–guest complexes.

Scheme 1: Synthetic route to TBTQ-(OG)6.

Figure 2: Fluorescence spectra of TBTQ-(OG)6 (5.0 × 10−6 M) with varying concentrations of (a) C60 and (b) C70...

Figure 3:
Absorption spectra of (a) TBTQ-(OG)6 C60 [TBTQ-(OG)6: 50 μM; C60: 50 μM] and (b) TBTQ-(OG)6
C70 [...

Figure 4:
Absorption spectra of (a) TBTQ-(OG)6 C60 [TBTQ-(OG)6: 50 μM; C60: 50 μM] and (b) TBTQ-(OG)6
C70 [...

Figure 5:
Raman spectra of TBTQ-G6, C60 and TBTQ-G6 C60. Sample solutions of TBTQ-(OG)6 (50 μM) and TBTQ-(OG)...

Figure 6:
Molecular model of the complex TBTQ-(OG)6 C60 in water, as generated by DFT calculations. (a) Side...

Figure 7:
SEM images of (a) C60; (b) TBTQ-(OG)6; (c) and (d) TBTQ-(OG)6 C60 (C60: 1.4 mM; TBTQ-(OG)6: 1.4 mM...
A new method for the synthesis of diamantane by hydroisomerization of binor-S on treatment with sulfuric acid
- Rishat I. Aminov and
- Ravil I. Khusnutdinov
Beilstein J. Org. Chem. 2020, 16, 2534–2539, doi:10.3762/bjoc.16.205
- of diamond-like hydrocarbons, diamantane, in 65% yield by hydroisomerization of the norbornadiene dimer, endo-endo-heptacyclo[8.4.0.02,12.03,8.04,6.05,9.011,13]tetradecane (binor-S) on treatment with concentrated sulfuric acid (98%). In the presence of H2SO4 of lower concentration (75–80%), the
- hydrocarbons and show peculiar chemical behavior. Crude oil is known to be the main natural source of diamondoids. In the oil and gas field exploration, the presence of diamondoids is used to evaluate the field maturity. Whereas the synthesis and chemical reactivity of adamantane, the first member of the
- homologous series, has been poorly studied. The main cause of this situation is the lack of facile methods for its synthesis. In the literature, diamantane (1) is prepared by skeletal isomerization of strained С14Н20 polycyclic hydrocarbons [2][3][4][5][6][7]. In particular, the most suitable initial

Graphical Abstract

Scheme 1: Isomerization of 3а–с to diamantane (1). Reaction conditions: (a) CoBr2·2PPh3–BF3·OEt2, 110 °C, 12 ...

Scheme 2: Isomerization of binor-S (2) to diamantane (1).

Scheme 3: Selective synthesis of tetrahydrobinor-S (3c) from binor-S (2).

Scheme 4: Isomerization of binor-S (2) to hydrocarbons 4а and b.
Synthetic approaches to bowl-shaped π-conjugated sumanene and its congeners
- Shakeel Alvi and
- Rashid Ali
Beilstein J. Org. Chem. 2020, 16, 2212–2259, doi:10.3762/bjoc.16.186

- in materials science and technology. Keywords: buckybowls; heterosumanenes; polyaromatic hydrocarbons; sumanene; synthesis; Review 1 Introduction Over a long period of time, polyaromatic hydrocarbons (PAHs) have attracted a tremendous attention of the scientific community because of their diverse

Graphical Abstract

Figure 1: Representation of corannulene (1) and sumanene (2), the subunits of fullerene (C60).

Scheme 1: Mehta’s unsuccessful effort for the synthesis of sumanene scaffold 2.

Scheme 2: First synthesis of sumanene 2 by Sakurai et al. from norbornadiene 10.

Scheme 3: Synthesis of trimethylsumanene 28 from easily accessible norbornadiene (10).

Scheme 4: Generation of anions 29–31 and the preparation of tris(trimethylsilyl)sumanene 32.

Scheme 5: Synthesis of tri- and hexa-substituted sumanene derivatives.

Scheme 6: Synthesis of bowl-shaped π-extended sumanene derivatives 37a–f.

Scheme 7: Synthesis of monooxasumanene 38, trioxosumanene 40 along with imination of them.

Scheme 8: Synthesis of trimethylsumanenetrione 46 and exo-functionalized products 45a,b.

Scheme 9: Synthesis of bisumanenylidene 47 and sumanene dimer 48 from 2.

Scheme 10: The mono-substitution of 2 to generate diverse mono-sumanene derivatives 49a–d.

Scheme 11: Synthesis of sumanene building block 53 useful for further extension.

Scheme 12: Synthesis of hexafluorosumanene derivative 55 by Sakurai and co-workers.

Scheme 13: Preparation of sumanene-based carbene 60 and its reaction with cyclohexane.

Scheme 14: Barton–Kellogg reaction for the synthesis of sterically hindered alkenes.

Scheme 15: Synthesis of hydroxysumanene 68 by employing Baeyer–Villiger oxidation.

Scheme 16: Synthesis of sumanene derivatives having functionality at an internal carbon.

Scheme 17: Mechanism for nucleophilic substitution reaction at the internal carbon.

Scheme 18: Synthesis of diverse monosubstituted sumanene derivatives.

Scheme 19: Synthesis of di- and trisubstituted sumanene derivatives from sumanene (2).

Scheme 20: Preparation of monochlorosumanene 88 and hydrogenation of sumanene (2).

Scheme 21: The dimer 90 and bissumanenyl 92 achieved from halosumannes.

Scheme 22: Pyrenylsumanene 93 involving the Suzuki-coupling as a key transformation.

Scheme 23: Synthesis of various hexaarylsumanene derivatives using the Suzuki-coupling reaction.

Scheme 24: Synthesis of hexasubstituted sumanene derivatives 96 and 97.

Scheme 25: Synthesis of thioalkylsumanenes via an aromatic nucleophilic substitution reaction.

Scheme 26: Synthesis of tris(ethoxycarbonylethenyl)sumanene derivative 108.

Scheme 27: Synthesis of ferrocenyl-based sumanene derivatives.

Scheme 28: Synthesis of sumanenylferrocene architectures 118 and 119 via Negishi coupling.

Scheme 29: Diosmylation and the synthesis of phenylboronate ester 121 of sumanene.

Scheme 30: Synthesis of the iron-complex of sumanene.

Scheme 31: Synthesis of tri- and mononuclear sumanenyl zirconocene complexes.

Scheme 32: Synthesis of [CpRu(η6-sumanene)]PF6.

Scheme 33: Preparation of sumanene-based porous coordination networks 127 (spherical tetramer units) and 128 (...

Scheme 34: Synthesis of sumanenylhafnocene complexes 129 and 130.

Scheme 35: Synthesis of 134 and 135 along with PdII coordination complex 136.

Scheme 36: Synthesis of alkali metals sumanene complex K7(C21H102−)2(C21H93−)·8THF (137) containing di- and tr...

Scheme 37: The encapsulation of a Cs+ ion between two sumanenyl anions.

Scheme 38: Synthesis of monothiasumanene 140 and dithiasumanene 141 from 139.

Scheme 39: Synthesis of trithiasumanene 151 by Otsubo and his co-workers.

Scheme 40: Synthesis of trithiasumanene derivatives 155 and 156.

Scheme 41: Synthetic route towards hexathiolated trithiasumanenes 158.

Scheme 42: Synthesis of triselenasumanene 160 by Shao and teammates.

Scheme 43: Synthesis of tritellurasumanene derivatives from triphenylene skeletons.

Scheme 44: Synthesis of pyrazine-fused sumanene architectures through condensation reaction.

Scheme 45: Treatment of the trichalcogenasumanenes with diverse oxidative reagents.

Scheme 46: Ring-opening reaction with H2O2 and oxone of heterasumanenes 178 and 179.

Scheme 47: Synthesis of polycyclic compounds from sumanene derivatives.

Scheme 48: Synthesis of diimide-based heterocycles reported by Shao’s and co-workers.

Scheme 49: Synthesis of pristine trichalcogenasumanenes, 151, 205, and 206.

Scheme 50: Synthesis of trichalcogenasumanenes via hexaiodotriphenylene precursor 208.

Scheme 51: Synthesis of trisilasumanenes 214 and 215.

Scheme 52: Synthesis of trisilasumanene derivatives 218 and 219.

Scheme 53: Synthesis of novel trigermasumanene derivative 223.

Scheme 54: An attempt towards the synthesis of tristannasumanene derivative 228.

Scheme 55: Synthesis of triphosphasumanene trisulfide 232 from commercially available 229.

Scheme 56: The doping of sumanene derivatives with chalcogens (S, Se, Te) and phosphorus.

Scheme 57: Synthesis of heterasumanene containing three different heteroatoms.

Scheme 58: Synthesis of trichalcogenasumanene derivatives 240 and 179.

Scheme 59: Preparation of trichalcogenasumanenes 245 and 248.

Scheme 60: Design and synthesis of trichalcogenasumanene derivatives 252 and 178.

Scheme 61: Synthesis of spirosumanenes 264–269 and non-spiroheterasumanenes 258–263.

Scheme 62: Synthesis of sumanene-type hetero polycyclic compounds.

Scheme 63: Synthesis of triazasumanenes 288 and its sulfone congener 287.

Scheme 64: Synthesis of C3-symmetric chiral triaryltriazasumanenes via cross-coupling reaction.

Scheme 65: Synthesis of mononaphthosumanene 293 using Suzuki coupling as a key step.

Scheme 66: Synthesis of di- and trinaphthosumanene derivatives 302–304.

Scheme 67: Synthesis of hemifullerene skeletons by Hirao’s group.

Scheme 68: Design and construction of C70 fragment from a C60 sumanene fragment.
In silico rationalisation of selectivity and reactivity in Pd-catalysed C–H activation reactions
- Liwei Cao,
- Mikhail Kabeshov,
- Steven V. Ley and
- Alexei A. Lapkin
Beilstein J. Org. Chem. 2020, 16, 1465–1475, doi:10.3762/bjoc.16.122
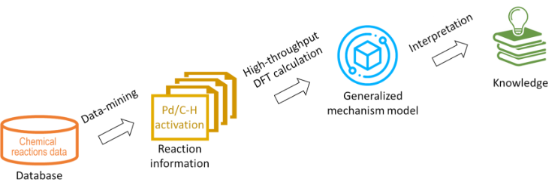
- developing machine learning models for predicting reaction outcomes. C–H activation reactions allow conversion of relatively inexpensive and abundant hydrocarbons into the more sophisticated value-added molecules [11]. With the notion of step-economical and environmentally friendly synthesis, direct

Graphical Abstract

Figure 1: An approximate energy map for the electrophilic aromatic substitution mechanism.

Scheme 1: Schematic representation of the two mechanisms of Pd-catalysed C–H activation reaction considered i...
An overview on disulfide-catalyzed and -cocatalyzed photoreactions
- Yeersen Patehebieke
Beilstein J. Org. Chem. 2020, 16, 1418–1435, doi:10.3762/bjoc.16.118

- also catalyze the formation of four-membered ring intermediates between double or triple bonds and oxygen, and thus converting unsaturated hydrocarbons to carbonyl compounds. Regarding the oxidation of alkynes, Wang reported a method for preparing diaryl-1,2-diketones from diarylalkynes in the presence

Graphical Abstract

Scheme 1: [3 + 2] cyclization catalyzed by diaryl disulfide.

Scheme 2: [3 + 2] cycloaddition catalyzed by disulfide.

Scheme 3: Disulfide-bridged peptide-catalyzed enantioselective cycloaddition.

Scheme 4: Disulfide-catalyzed [3 + 2] methylenecyclopentane annulations.

Scheme 5: Disulfide as a HAT cocatalyst in the [4 + 2] cycloaddition reaction.

Scheme 6: Proposed mechanism of the [4 + 2] cycloaddition reaction using disulfide as a HAT cocatalyst.

Scheme 7: Disulfide-catalyzed ring expansion of vinyl spiro epoxides.

Scheme 8: Disulfide-catalyzed aerobic oxidation of diarylacetylene.

Scheme 9: Disulfide-catalyzed aerobic photooxidative cleavage of olefins.

Scheme 10: Disulfide-catalyzed aerobic oxidation of 1,3-dicarbonyl compounds.

Scheme 11: Proposed mechanism of the disulfide-catalyzed aerobic oxidation of 1,3-dicarbonyl compounds.

Scheme 12: Disulfide-catalyzed oxidation of allyl alcohols.

Scheme 13: Disulfide-catalyzed diboration of alkynes.

Scheme 14: Dehalogenative radical cyclization catalyzed by disulfide.

Scheme 15: Hydrodifluoroacetamidation of alkenes catalyzed by disulfide.

Scheme 16: Plausible mechanism of the hydrodifluoroacetamidation of alkenes catalyzed by disulfide.

Scheme 17: Disulfide-cocatalyzed anti-Markovnikov olefin hydration reactions.

Scheme 18: Disulfide-catalyzed decarboxylation of carboxylic acids.

Scheme 19: Proposed mechanism of the disulfide-catalyzed decarboxylation of carboxylic acids.

Scheme 20: Disulfide-catalyzed decarboxylation of carboxylic acids.

Scheme 21: Disulfide-catalyzed conversion of maleate esters to fumarates and 5H-furanones.

Scheme 22: Disulfide-catalyzed isomerization of difluorotriethylsilylethylene.

Scheme 23: Disulfide-catalyzed isomerization of allyl alcohols to carbonyl compounds.

Scheme 24: Proposed mechanism for the disulfide-catalyzed isomerization of allyl alcohols to carbonyl compound...

Scheme 25: Diphenyl disulfide-catalyzed enantioselective synthesis of ophirin B.

Scheme 26: Disulfide-catalyzed isomerization in the total synthesis of (+)-hitachimycin.

Scheme 27: Disulfide-catalyzed isomerization in the synthesis of (−)-gloeosporone.
Oxime radicals: generation, properties and application in organic synthesis
- Igor B. Krylov,
- Stanislav A. Paveliev,
- Alexander S. Budnikov and
- Alexander O. Terent’ev
Beilstein J. Org. Chem. 2020, 16, 1234–1276, doi:10.3762/bjoc.16.107
- the relatively stable di-tert-butyliminoxyl radical was studied as a reagent in oxidative transformations of various substrates, such as unsaturated hydrocarbons, phenols, amines, and organometallic compounds. A breakthrough in the synthetic use of iminoxyl radicals has occurred in recent years when
- with a C=C double bond was explained by the steric hindrance of the iminoxyl radical. On the other hand, di-tert-butyliminoxyl radical (8) can react with unsaturated hydrocarbons by abstracting the hydrogen atom from the allyl or benzyl position (Scheme 10) [35][45][60][61]. The C-centered radicals

Graphical Abstract

Figure 1: Imine-N-oxyl radicals (IV) discussed in the present review and other classes of N-oxyl radicals (I–...

Figure 2: The products of decomposition of iminoxyl radicals generated from oximes by oxidation with Ag2O.

Scheme 1: Generation of oxime radicals and study of the kinetics of their decay by photolysis of the solution...

Scheme 2: Synthesis of di-tert-butyliminoxyl radical and its decomposition products.

Scheme 3: The proposed reaction pathway of the decomposition of di-tert-butyliminoxyl radical (experimentally...

Scheme 4: Monomolecular decomposition of the tert-butyl(triethylmethyl)oxime radical.

Scheme 5: The synthesis and stability of the most stable dialkyl oxime radicals – di-tert-butyliminoxyl and d...

Scheme 6: The formation of iminoxyl radicals from β-diketones under the action of NO2.

Scheme 7: Synthesis of the diacetyliminoxyl radical.

Scheme 8: Examples of long-living oxime radicals with electron-withdrawing groups and the conditions for thei...

Figure 3: The electronic structure iminoxyl radicals and their geometry compared to the corresponding oximes.

Figure 4: Bond dissociation enthalpies (kcal/mol) of oximes and N,N-disubstituted hydroxylamines calculated o...

Scheme 9: Examples demonstrating the low reactivity of the di-tert-butyliminoxyl radical towards the substrat...

Scheme 10: The reactions of di-tert-butyliminoxyl radical with unsaturated hydrocarbons involving hydrogen ato...

Scheme 11: Possible mechanisms of reaction of di-tert-butyliminoxyl radical with alkenes.

Scheme 12: Products of the reaction between di-tert-butyliminoxyl radical and phenol derivatives.

Scheme 13: The reaction of di-tert-butyliminoxyl radical with amines.

Scheme 14: Reaction of di-tert-butyliminoxyl radicals with organolithium reagents.

Scheme 15: Cross-dehydrogenative C–O coupling of 1,3-dicarbonyl compounds with oximes under the action of mang...

Scheme 16: Cross-dehydrogenative C–O coupling of 1,3-dicarbonyl compounds with oximes under the action of Cu(BF...

Scheme 17: Oxidative C–O coupling of benzylmalononitrile (47) with 3-(hydroxyimino)pentane-2,4-dione (19).

Scheme 18: The proposed mechanism of the oxidative coupling of benzylmalononitrile (47) with diacetyl oxime (19...

Scheme 19: Oxidative C–O coupling of pyrazolones with oximes under the action of Fe(ClO4)3.

Scheme 20: The reaction of diacetyliminoxyl radical with pyrazolones.

Scheme 21: Oxidative C–O coupling of oximes with acetonitrile, ketones, and esters.

Scheme 22: Intramolecular cyclizations of oxime radicals to form substituted isoxazolines or cyclic nitrones.

Scheme 23: TEMPO-mediated oxidative cyclization of oximes with C–H bond cleavage.

Scheme 24: Proposed reaction mechanism of oxidative cyclization of oximes with C–H bond cleavage.

Scheme 25: Selectfluor/Bu4NI-mediated C–H oxidative cyclization of oximes.

Scheme 26: Oxidative cyclization of N-benzyl amidoximes to 1,2,4-oxadiazoles.

Scheme 27: The formation of quinazolinone 73a from 5-phenyl-4,5-dihydro-1,2,4-oxadiazole 74 under air.

Scheme 28: DDQ-mediated oxidative cyclization of thiohydroximic acids.

Scheme 29: Plausible mechanism of the oxidative cyclization of thiohydroximic acids.

Scheme 30: Silver-mediated oxidative cyclization of α-halogenated ketoximes and 1,3-dicarbonyl compounds.

Scheme 31: Possible pathway of one-pot oxidative cyclization of α-halogenated ketoximes and 1,3-dicarbonyl com...

Scheme 32: T(p-F)PPT-catalyzed oxidative cyclization of oximes with the formation of 1,2,4-oxadiazolines.

Scheme 33: Intramolecular cyclization of iminoxyl radicals involving multiple C=C and N=N bonds.

Scheme 34: Oxidative cyclization of β,γ- and γ,δ-unsaturated oximes employing the DEAD or TEMPO/DEAD system wi...

Scheme 35: Cobalt-catalyzed aerobic oxidative cyclization of β,γ-unsaturated oximes.

Scheme 36: Manganese-catalyzed aerobic oxidative cyclization of β,γ-unsaturated oximes.

Scheme 37: Visible light photocatalytic oxidative cyclization of β,γ-unsaturated oximes.

Scheme 38: TBAI/TBHP-mediated radical cascade cyclization of the β,γ-unsaturated oximes.

Scheme 39: TBAI/TBHP-mediated radical cascade cyclization of vinyl isocyanides with β,γ-unsaturated oximes.

Scheme 40: tert-Butylnitrite-mediated oxidative cyclization of unsaturated oximes with the introduction of an ...

Scheme 41: Transformation of unsaturated oxime to oxyiminomethylisoxazoline via the confirmed dimeric nitroso ...

Scheme 42: tert-Butylnitrite-mediated oxidative cyclization of unsaturated oximes with the introduction of a n...

Scheme 43: Synthesis of cyano-substituted oxazolines from unsaturated oximes using the TBN/[RuCl2(p-cymene)]2 ...

Scheme 44: Synthesis of trifluoromethylthiolated isoxazolines from unsaturated oximes.

Scheme 45: Copper-сatalyzed oxidative cyclization of β,γ-unsaturated oximes with the introduction of an azido ...

Scheme 46: TBHP-mediated oxidative cascade cyclization of β,γ-unsaturated oximes and unsaturated N-arylamides.

Scheme 47: Copper-сatalyzed oxidative cyclization of unsaturated oximes with the introduction of an amino grou...

Scheme 48: TEMPO-mediated oxidative cyclization of unsaturated oximes followed by elimination.

Scheme 49: Oxidative cyclization of β,γ-unsaturated oximes with the introduction of a trifluoromethyl group.

Scheme 50: Oxidative cyclization of unsaturated oximes with the introduction of a nitrile group.

Scheme 51: Oxidative cyclization of β,γ-unsaturated oximes to isoxazolines with the introduction of a nitrile ...

Scheme 52: Oxidative cyclization of β,γ-unsaturated oximes to isoxazolines with the introduction of a sulfonyl...

Scheme 53: Oxidative cyclization of β,γ- and γ,δ-unsaturated oximes to isoxazolines with the introduction of a...

Scheme 54: Oxidative cyclization of β,γ-unsaturated oximes to isoxazolines with the introduction of a thiocyan...

Scheme 55: PhI(OAc)2-mediated oxidative cyclization of oximes with C–S and C–Se bond formation.

Scheme 56: PhI(OAc)2-mediated oxidative cyclization of unsaturated oximes accompanied by alkoxylation.

Scheme 57: PhI(OAc)2-mediated cyclization of unsaturated oximes to methylisoxazolines.

Scheme 58: Oxidative cyclization-alkynylation of unsaturated oximes.

Scheme 59: TEMPO-mediated oxidative cyclization of C-glycoside ketoximes to C-glycosylmethylisoxazoles.

Scheme 60: Silver-сatalyzed oxidative cyclization of β,γ-unsaturated oximes with formation of fluoroalkyl isox...

Scheme 61: Oxidative cyclization of β,γ-unsaturated oximes with the formation of haloalkyl isoxazolines.

Scheme 62: Cyclization of β,γ-unsaturated oximes into haloalkyl isoxazolines under the action of the halogenat...

Scheme 63: Synthesis of haloalkyl isoxazoles and cyclic nitrones via oxidative cyclization and 1,2-halogen shi...

Scheme 64: Electrochemical oxidative cyclization of diaryl oximes.

Scheme 65: Copper-сatalyzed cyclization and dioxygenation oximes containing a triple C≡C bond.

Scheme 66: Photoredox-catalyzed sulfonylation of β,γ-unsaturated oximes by sulfonyl hydrazides.

Scheme 67: Oxidative cyclization of β,γ-unsaturated oximes with introduction of sulfonate group.

Scheme 68: Ultrasound-promoted oxidative cyclization of β,γ-unsaturated oximes.
One-pot synthesis of dicyclopenta-fused peropyrene via a fourfold alkyne annulation
- Ji Ma,
- Yubin Fu,
- Junzhi Liu and
- Xinliang Feng
Beilstein J. Org. Chem. 2020, 16, 791–797, doi:10.3762/bjoc.16.72
- an efficient method to develop π-extended aromatic hydrocarbons with cyclopenta moieties. Keywords: alkyne annulation; cyclopenta-fused polycyclic aromatic hydrocarbons; nonplanarity; peropyrene; regioselectivity; Introduction Significant efforts have been recently devoted to the synthesis of
- nonalternant cyclopenta-fused polycyclic aromatic hydrocarbons (CP-PAHs), which represent the topological subunits of fullerenes and exhibit high chemical, physical and biological activities [1][2][3][4][5][6][7][8][9][10]. Thanks to development in organic synthetic methodology, CP-PAHs with peripheral
- as an efficient route to get access to aromatic hydrocarbons with peri-fused five-membered rings [25][26][27]. For instance, the dicyclopenta-fused pyrene derivatives ii and iii (Scheme 1) were successfully synthesized through palladium-catalyzed carbannulation of brominated pyrene with

Graphical Abstract

Scheme 1: Chemical structures of dicyclopenta-fused pyrene derivatives i–iii, peropyrene and the dicyclopenta...

Scheme 2: Synthetic route towards compound 1. a) B2pin2, dtbpy, [Ir(OMe)cod]2, cyclohexane, 70 °C, 20 h, 67%;...

Figure 1: High-resolution MALDI-TOF mass spectrum of 1. Inset: isotopic distribution compared to mass spectru...

Figure 2: Single-crystal X-ray structure of 1. (a) Top view and (b) side view of the (P,P) isomer. c) Crystal...

Figure 3: (a) UV–vis absorption spectra of precursor 5 and 1 in CH2Cl2 solution (10−5 M). Inset: photograph o...

Figure 4: Molecular orbitals of peropyrene derivative 6 and the dicyclopenta-fused peropyrene 1.
Synthesis of C70-fragment buckybowls bearing alkoxy substituents
- Yumi Yakiyama,
- Shota Hishikawa and
- Hidehiro Sakurai
Beilstein J. Org. Chem. 2020, 16, 681–690, doi:10.3762/bjoc.16.66

- equilibrium between the Pd(IV) intermediates through C–H bond activation. Keywords: buckybowl; C70; rearrangement through C–H bond activation; Introduction The study of buckybowls, the bowl-shaped π-conjugated aromatic hydrocarbons corresponding to the fragments of fullerenes, pioneered by the works on

Graphical Abstract

Figure 1: Structure of the target buckybowls 5a–c.

Scheme 1: Synthesis of dialkoxides 5a–c.

Scheme 2: Proposed mechanism of the formation of 5b and 5c.

Figure 2: Crystal structure of 5a. a) ORTEP drawing of the crystallographically independent unit with thermal...

Figure 3: a) Definition of POAV angle (φ). b) Side and c) top view of the molecular skeleton of 1. The double...

Figure 4: Crystal structure of 5b. a) ORTEP drawing of the crystallographically independent unit with thermal...

Figure 5: Crystal structure of 5c. a) ORTEP drawing of the crystallographically independent unit with thermal...

Figure 6: a) UV–vis spectra and b) emission spectra of 1 and dialkoxides 5a–c. For all the spectra, the conce...
Efficient synthesis of 3,6,13,16-tetrasubstituted-tetrabenzo[a,d,j,m]coronenes by selective C–H/C–O arylations of anthraquinone derivatives
- Seiya Terai,
- Yuki Sato,
- Takuya Kochi and
- Fumitoshi Kakiuchi
Beilstein J. Org. Chem. 2020, 16, 544–550, doi:10.3762/bjoc.16.51
- tetrabenzo[a,d,j,m]coronene product indicated its self-assembling behavior in CDCl3. Keywords: C–H arylation; C–O arylation; oxidative cyclization; polycyclic aromatic hydrocarbons; ruthenium catalyst; Introduction Polycyclic aromatic hydrocarbons (PAHs) and their derivatives have attracted much attention

Graphical Abstract

Scheme 1: Projected synthetic routes for 3,6,13,16-tetrasubstituted tetrabenzo[a,d,j,m]coronenes.

Scheme 2: Reported syntheses of 2,7,12,17-tetrasubstituted tetrabenzo[a,d,j,m]coronenes.

Scheme 3: C–H tetraarylation of anthraquinone (1).

Scheme 4: C–H diarylation of 1,4-diarylanthraquinones 5.

Figure 1: Normalized UV–vis absorption spectra of 7aa, 7bb, and 7ba.

Figure 2: Effects of the concentration and the temperature on the 1H NMR spectra of 7aa.
Copper-catalyzed remote C–H arylation of polycyclic aromatic hydrocarbons (PAHs)
- Anping Luo,
- Min Zhang,
- Zhangyi Fu,
- Jingbo Lan,
- Di Wu and
- Jingsong You
Beilstein J. Org. Chem. 2020, 16, 530–536, doi:10.3762/bjoc.16.49
- substituted polycyclic aromatic hydrocarbons (PAHs) is a desired but challenging task. A copper-catalyzed C7–H arylation of 1-naphthamides has been developed by using aryliodonium salts as arylating reagents. This protocol does not need to use precious metal catalysts and tolerates wide variety of functional
- arylation; nonprecious metal catalyst; copper catalysis; polycyclic aromatic hydrocarbons (PAHs); regioselectivity; Introduction Polycyclic aromatic hydrocarbons (PAHs) with rigid planar structure, such as naphthalene, phenanthrene, pyrene and their derivatives, can usually emit relatively strong

Graphical Abstract

Scheme 1: Direct C–H arylation of PAHs.

Scheme 2: Scope of aryliodonium salts. Reaction conditions: 1a (0.2 mmol), 2 (0.3 mmol) in DCE (1 mL) at 70 °...

Scheme 3: Scope of PAHs. Reaction conditions: 1 (0.2 mmol), 2a (0.3 mmol) in DCE (1 mL) at 70 °C under N2 for...

Scheme 4: Proposed catalytic cycle.

Figure 1: a) UV-visible absorption spectra of 4k, 4n and 4o in toluene (1 × 10−5 mol/L). b) Emission spectra ...
Synthesis of triphenylene-fused phosphole oxides via C–H functionalizations
- Md. Shafiqur Rahman and
- Naohiko Yoshikai
Beilstein J. Org. Chem. 2020, 16, 524–529, doi:10.3762/bjoc.16.48
- nature as hybrids of triphenylene and benzo[b]phosphole. Keywords: C–H functionalization; fluorescence; phosphole; polycyclic aromatic hydrocarbons; triphenylene; Introduction The phosphorus-containing five-membered ring, phosphole, has attracted significant attention as a structural motif in π
- extended π-system. These included, in particular, those fused with polycyclic aromatic hydrocarbons (PAHs) for possible applications in organic electronics, bioimaging and sensing, and asymmetric catalysis (Figure 1). To name a few examples, Yamaguchi et al. described synthetic routes to novel phosphorus

Graphical Abstract

Figure 1: Examples of functional molecules based on π-extended phospholes.

Scheme 1: Syntheses of PAH-fused phospholes featuring a 7-hydroxybenzo[b]phosphole as a key intermediate.

Scheme 2: Synthesis of phosphole-fused ortho-teraryl compounds 7.

Scheme 3: Oxidative cyclization of phosphole-fused ortho-teraryl compounds 7 into triphenylene-fused phosphol...

Figure 2: ORTEP drawings of compound 8a (thermal ellipsoids set at 50% probability). a) top view; b) side vie...

Figure 3: UV–vis absorption (solid lines) and fluorescence (dashed lines) spectra of compounds 8a–c.
Six-fold C–H borylation of hexa-peri-hexabenzocoronene
- Mai Nagase,
- Kenta Kato,
- Akiko Yagi,
- Yasutomo Segawa and
- Kenichiro Itami
Beilstein J. Org. Chem. 2020, 16, 391–397, doi:10.3762/bjoc.16.37
- calculations. The spectra revealed a bathochromic shift of absorption bands compared with unsubstituted HBC under the effect of the σ-donation of boryl groups. Keywords: C–H borylation; hexa-peri-hexabenzocoronene; iridium catalyst; X-ray crystallography; Introduction Polycyclic aromatic hydrocarbons (PAHs

Graphical Abstract

Figure 1: C–H functionalization of HBCs. (a) Perchlorinated HBC. (b) Borylated HBC substituted by 2,4,6-trime...

Figure 2: Synthesis of hexaborylated HBC 1. (a) Solvent screening of six-fold C–H borylation of unsubstituted...

Figure 3: The structure of 1 confirmed by X-ray crystallographic analysis. (a) ORTEP drawing of 1 with therma...

Figure 4: Photophysical properties of 1. (a) UV–vis absorption (solid lines) spectra, fluorescence (dotted li...

- initially formed terpene hydrocarbons can subsequently be modified by enzymatic oxidations that often involve radical chemistry. The non-functionalised hydrocarbons are volatile and often exhibit interesting odour properties [3]. As a consequence, these compounds may act as chemical signals such as
Bacterial terpene biosynthesis: challenges and opportunities for pathway engineering
- Eric J. N. Helfrich,
- Geng-Min Lin,
- Christopher A. Voigt and
- Jon Clardy
Beilstein J. Org. Chem. 2019, 15, 2889–2906, doi:10.3762/bjoc.15.283

- Information File 1, Table S2). CYPs are heme-dependent iron proteins that catalyze a wide range of reactions [83][84]. The reactions typically involve substrate radical generation by the activated iron species and subsequent hydroxylation. Terpenes are mainly composed of nonactivated hydrocarbons that are

Graphical Abstract

Figure 1: Examples of bioactive terpenoids.

Figure 2: Repetitive electrophilic and nucleophilic functionalities in terpene and type II PKS-derived polyke...

Figure 3: Abundance and distribution of bacterial terpene biosynthetic gene clusters as determined by genome ...

Figure 4: Terpenoid biosynthesis. Terpenoid biosynthesis is divided into two phases, 1) terpene scaffold gene...

Figure 5: Mechanisms for type I, type II, and type II/type I tandem terpene cyclases. a) Tail-to-head class I...

Figure 6: Functional TC characterization. a) Different terpenes were produced when hedycaryol (18) synthase a...

Figure 7: Selected examples of terpene modification by bacterial CYPs. a) Hydroxylation [89]. b) Carboxylation, h...

Figure 8: Off-target effects observed during heterologous expression of terpenoid BGCs. Unexpected oxidation ...

Figure 9: TC promiscuity and engineering. a) Spata-13,17-diene (39) synthase (SpS) can take C15 and C25 oligo...

Figure 10: Substrate promiscuity and engineering of CYPs. a) Selected examples from using a CYP library to oxi...

Figure 11: Engineering of terpenoid pathways. a) Metabolic network of terpenoid biosynthesis. Toxic intermedia...
Recent advances in transition-metal-catalyzed incorporation of fluorine-containing groups
- Xiaowei Li,
- Xiaolin Shi,
- Xiangqian Li and
- Dayong Shi
Beilstein J. Org. Chem. 2019, 15, 2213–2270, doi:10.3762/bjoc.15.218
- hydrocarbons, substituted cyclic molecules, terpenoids, and steroid derivatives, were selectively fluorinated at some otherwise inaccessible sites, however, in low to moderate yields. On the other hand, the same group [83] developed Mn(salen)Cl as a catalyst for the direct C–H fluorination at benzylic

Graphical Abstract

Scheme 1: The main three strategies of fluorination: nucleophilic, electrophilic and radical fluorination.

Scheme 2: Doyle’s Pd-catalyzed fluorination of allylic chlorides.

Scheme 3: Allylic fluorination of 2- and 3-substituted propenyl esters.

Scheme 4: Regioselective allylic fluorination of cinnamyl phosphorothioate esters.

Scheme 5: Palladium-catalyzed aliphatic C–H fluorination reported by Doyle.

Scheme 6: Pd-catalyzed enantioselective fluorination of α-ketoesters followed by stereoselective reduction to...

Scheme 7: Pd-catalyzed C(sp3)–H fluorination of oxindoles.

Scheme 8: C–H fluorination of 8-methylquinoline derivatives with F− reagents.

Scheme 9: Fluorination of α-cyano acetates reported by van Leeuwen.

Scheme 10: The catalytic enantioselective electrophilic C–H fluorination of α-chloro-β-keto phosphonates.

Scheme 11: Fluorination of unactivated C(sp3)–H bonds directed by the bidentate PIP auxiliary.

Scheme 12: Fluorination of C(sp3)–H bonds at the β-position of carboxylic acids.

Scheme 13: Enantioselective benzylic C–H fluorination with a chiral transient directing group.

Scheme 14: Microwave-heated Pd-catalyzed fluorination of aryl alcohols.

Scheme 15: Fluorination of aryl potassium trifluoroborates.

Scheme 16: C(sp2)–F bond formation using precatalyst [L·Pd]2(cod).

Scheme 17: Pd-catalyzed fluorination of (hetero)aryl triflates and bromides.

Scheme 18: The Pd-catalyzed C–H fluorination of arenes with Selectfluor/NFSI.

Scheme 19: Pd(II)-catalyzed ortho-monofluorination protocol for benzoic acids.

Scheme 20: Pd-catalyzed C(sp2)–H bond fluorination of 2-arylbenzothiazoles.

Scheme 21: Nitrate-promoted fluorination of aromatic and olefinic C(sp2)–H bonds and proposed mechanism.

Scheme 22: Fluorination of oxalyl amide-protected benzylamine derivatives.

Scheme 23: C–H fluorination of benzaldehydes with orthanilic acids as transient directing group.

Scheme 24: Pd(II)-catalyzed aryl C–H fluorination with various directing groups.

Scheme 25: Cu-catalyzed aliphatic, allylic, and benzylic fluorination.

Scheme 26: Cu-catalyzed SN2 fluorination of primary and secondary alkyl bromides.

Scheme 27: Copper-catalyzed fluorination of alkyl triflates.

Scheme 28: Cu-catalyzed fluorination of allylic bromides and chlorides.

Scheme 29: Synthetic strategy for the fluorination of active methylene compounds.

Scheme 30: Fluorination of β-ketoesters using a tartrate-derived bidentate bisoxazoline-Cu(II) complex.

Scheme 31: Highly enantioselective fluorination of β-ketoesters and N-Boc-oxindoles.

Scheme 32: Amide group-assisted site-selective fluorination of α-bromocarbonyl compounds.

Scheme 33: Cu-mediated aryl fluorination reported by Sanford [77].

Scheme 34: Mono- or difluorination reactions of benzoic acid derivatives.

Scheme 35: Cu-catalyzed fluorination of diaryliodonium salts with KF.

Scheme 36: Copper(I)-catalyzed cross-coupling of 2-pyridylaryl bromides.

Scheme 37: AgNO3-catalyzed decarboxylative fluorination of aliphatic carboxylic acids.

Scheme 38: The Mn-catalyzed aliphatic and benzylic C–H fluorination.

Scheme 39: Iron(II)-promoted C–H fluorination of benzylic substrates.

Scheme 40: Ag-catalyzed fluorodecarboxylation of carboxylic acids.

Scheme 41: Vanadium-catalyzed C(sp3)–H fluorination.

Scheme 42: AgNO3-catalyzed radical deboronofluorination of alkylboronates and boronic acids.

Scheme 43: Selective heterobenzylic C–H fluorination with Selectfluor reported by Van Humbeck.

Scheme 44: Fe(II)-catalyzed site-selective fluorination guided by an alkoxyl radical.

Scheme 45: Fluorination of allylic trichloroacetimidates reported by Nguyen et al.

Scheme 46: Iridium-catalyzed fluorination of allylic carbonates with TBAF(t-BuOH)4.

Scheme 47: Iridium-catalyzed asymmetric fluorination of allylic trichloroacetimidates.

Scheme 48: Cobalt-catalyzed α-fluorination of β-ketoesters.

Scheme 49: Nickel-catalyzed α-fluorination of various α-chloro-β-ketoesters.

Scheme 50: Ni(II)-catalyzed enantioselective fluorination of oxindoles and β-ketoesters.

Scheme 51: Scandium(III)-catalyzed asymmetric C–H fluorination of unprotected 3-substituted oxindoles.

Scheme 52: Iron-catalyzed directed C–H fluorination.

Scheme 53: Electrophilic silver-catalyzed Ar–F bond-forming reaction from arylstannanes.

Figure 1: Nucleophilic, electrophilic and radical CF3 sources.

Scheme 54: Cu(I)-catalyzed allylic trifluoromethylation of unactivated terminal olefins.

Scheme 55: Direct copper-catalyzed trifluoromethylation of allylsilanes.

Scheme 56: Cupper-catalyzed enantioselective trifluoromethylation of five and six-membered ring β-ketoesters.

Scheme 57: Cu-catalyzed highly stereoselective trifluoromethylation of secondary propargyl sulfonates.

Scheme 58: Remote C(sp3)–H trifluoromethylation of carboxamides and sulfonamides.

Scheme 59: Trifluoromethylation of allylsilanes with photoredox catalysis.

Scheme 60: Ag-catalyzed decarboxylative trifluoromethylation of aliphatic carboxylic acids in aqueous CH3CN.

Scheme 61: Decarboxylative trifluoromethylation of aliphatic carboxylic acids via combined photoredox and copp...

Scheme 62: Palladium-catalyzed Ar–CF3 bond-forming reaction.

Scheme 63: Palladium-catalyzed trifluoromethylation of arenes with diverse heterocyclic directing groups.

Scheme 64: Pd-catalyzed trifluoromethylation of indoles as reported by Liu.

Scheme 65: Pd-catalyzed trifluoromethylation of vinyl triflates and vinyl nonaflates.

Scheme 66: Pd(II)-catalyzed ortho-trifluoromethylation of aromatic C–H bonds.

Scheme 67: Visible-light-induced Pd(OAc)2-catalyzed ortho-trifluoromethylation of acetanilides with CF3SO2Na.

Scheme 68: CuI-catalyzed trifluoromethylation of aryl- and alkenylboronic acids.

Scheme 69: Cu-catalyzed trifluoromethylation of aryl- and vinylboronic acids.

Scheme 70: Copper-catalyzed trifluoromethylation of α,β-unsaturated carboxylic acids.

Scheme 71: Formation of C(sp2)–CF3 bond catalyzed by copper(I) complex.

Scheme 72: Loh’s Cu(I)-catalyzed trifluoromethylation of enamides and electron-deficient alkenes.

Scheme 73: Copper and iron-catalyzed decarboxylative tri- and difluoromethylation.

Scheme 74: Cu-catalyzed trifluoromethylation of hydrazones developed by Bouyssi.

Scheme 75: Cu(I)-catalyzed trifluoromethylation of terminal alkenes.

Scheme 76: Cu/Ag-catalyzed decarboxylative trifluoromethylation of cinnamic acids.

Scheme 77: Copper-catalyzed direct alkenyl C–H trifluoromethylation.

Scheme 78: Copper(I/II)-catalyzed direct trifluoromethylation of styrene derivatives.

Scheme 79: Regioselective trifluoromethylation of pivalamido arenes and heteroarenes.

Scheme 80: Synthesis of trifluoromethylquinones in the presence of copper(I).

Scheme 81: Oxidative trifluoromethylation of imidazoheterocycles in ionic liquid/water.

Scheme 82: A mild and fast continuous-flow trifluoromethylation of coumarins using a CuI/CF3SO2Na/TBHP system.

Scheme 83: Copper-catalyzed oxidative trifluoromethylation of various 8-aminoquinolines.

Scheme 84: PA-directed copper-catalyzed trifluoromethylation of anilines.

Scheme 85: Trifluoromethylation of potassium vinyltrifluoroborates catalyzed by Fe(II).

Scheme 86: Alkenyl trifluoromethylation catalyzed by Ru(phen)3Cl2 as photocatalyst.

Scheme 87: Ru-catalyzed trifluoromethylation of alkenes by Akita’s group.

Scheme 88: Ir-catalyzed Cvinyl–CF3 bond formation of α,β-unsaturated carboxylic acids.

Scheme 89: Ag(I)-catalyzed denitrative trifluoromethylation of β-nitrostyrenes.

Scheme 90: Photocatalyzed direct trifluoromethylation of aryl and heteroaryl C–H bonds.

Scheme 91: Rhenium (MTO)-catalyzed direct trifluoromethylation of aromatic substrates.

Scheme 92: Trifluoromethylation of unprotected anilines under [Ir(ppy)3] catalyst.

Scheme 93: Oxidative trifluoromethylation of imidazopyridines and imidazoheterocycles.

Scheme 94: Ruthenium-catalyzed trifluoromethylation of (hetero)arenes with trifluoroacetic anhydride.

Scheme 95: Phosphovanadomolybdic acid-catalyzed direct C–H trifluoromethylation.

Scheme 96: Picolinamide-assisted ortho-trifluoromethylation of arylamines.

Scheme 97: A nickel-catalyzed C–H trifluoromethylation of free anilines.

Scheme 98: Cu-mediated trifluoromethylation of terminal alkynes reported by Qing.

Scheme 99: Huang’s C(sp)–H trifluoromethylation using Togni’s reagent.

Scheme 100: Cu-catalyzed methods for trifluoromethylation with Umemoto’s reagent.

Scheme 101: The synthesis of alkynyl-CF3 compounds in the presence of fac-[Ir(ppy)3] under visible-light irradi...

Scheme 102: Pd-catalyzed Heck reaction reported by Reutrakul.

Scheme 103: Difluoromethylation of enamides and ene-carbamates.

Scheme 104: Difluoromethylation of α,β-unsaturated carboxylic acids.

Scheme 105: Copper-catalyzed direct C(sp2)–H difluoroacetylation reported by Pannecoucke and co-workers.

Scheme 106: Difluoroalkylation of aldehyde-derived hydrazones with functionalized difluoromethyl bromides.

Scheme 107: Photoredox-catalyzed C–H difluoroalkylation of aldehyde-derived hydrazones.

Scheme 108: Synergistic ruthenium(II)-catalyzed C–H difluoromethylation reported by Ackermann.

Scheme 109: Visible-light photocatalytic decarboxylation of α,β-unsaturated carboxylic acids.

Scheme 110: Synthesis of difluorinated ketones via S-alkyl dithiocarbamates obtained from acyl chlorides and po...

Scheme 111: Synthesis of aryl and heteroaryl difluoromethylated phosphonates.

Scheme 112: Difluoroalkylation of secondary propargyl sulfonates using Cu as the catalyst.

Scheme 113: Ru(II)-mediated para-selective difluoromethylation of anilides and their derivatives.

Scheme 114: Bulky diamine ligand promoted cross-coupling of difluoroalkyl bromides.

Scheme 115: Copper-catalyzed C3–H difluoroacetylation of quinoxalinones.

Scheme 116: Copper(I) chloride-catalyzed trifluoromethylthiolation of enamines, indoles and β-ketoesters.

Scheme 117: Copper-boxmi-catalyzed asymmetric trifluoromethylthiolation of β-ketoesters.

Scheme 118: Direct Cu-catalyzed trifluoromethylthiolation of boronic acids and alkynes.

Scheme 119: Cu-catalyzed synthesis of α-trifluoromethylthio-substituted ketones.

Scheme 120: Trifluoromethylthiolation reactions promoted by diazotriflone and copper.

Scheme 121: Halide activation of N-(trifluoromethylthio)phthalimide.

Scheme 122: The visible light-promoted trifluoromethylthiolation reported by Glorius.

Scheme 123: Synthesis of α-trifluoromethylthioesters via Goossen’s approach.

Scheme 124: Photoinduced trifluoromethylthiolation of diazonium salts.

Scheme 125: Ag-mediated trifluoromethoxylation of aryl stannanes and arylboronic acids.

Scheme 126: Catalytic (hetero)aryl C–H trifluoromethoxylation under visible light.

Scheme 127: Photoinduced C–H-bond trifluromethoxylation of (hetero)arenes.
Analysis of sesquiterpene hydrocarbons in grape berry exocarp (Vitis vinifera L.) using in vivo-labeling and comprehensive two-dimensional gas chromatography–mass spectrometry (GC×GC–MS)
- Philipp P. Könen and
- Matthias Wüst
Beilstein J. Org. Chem. 2019, 15, 1945–1961, doi:10.3762/bjoc.15.190
- the unlabeled and deuterated compounds, mechanisms for sesquiterpene formation in V. vinifera could be proposed and already known pathways could be confirmed or disproved. For example, the HS-SPME–GC×GC–TOF–MS measurements of fed sample material showed that the tricyclic sesquiterpene hydrocarbons α
- various cyclization reactions [9][10][11][12]. In order to analyze the biosynthetic pathways of sesquiterpene hydrocarbons in grape berries, a method was developed by us using comprehensive two-dimensional gas chromatography (GC×GC) coupled to a time-of-flight mass spectrometer (TOF–MS) after headspace
- isolated exocarp of freshly harvested grapes (Vitis vinifera L.) using the stable isotope-labeled precursors [5,5-2H2]-1-deoxy-ᴅ-xylulose (d2-DOX) and [6,6,6-2H3]-(±)-mevalonolactone (d3-MVL) to unambiguously identify sesquiterpene hydrocarbons. Based on the obtained mass spectra of the genuine and

Graphical Abstract

Figure 1: Contour plot of a HS-SPME–GC×GC–TOF–MS chromatogram (TIC) demonstrating the separation of volatile ...

Figure 2: Sesquiterpene hydrocarbons found in the headspace of Lemberger (Vitis vinifera subsp. vinifera, clo...

Figure 3: Detailed part of the two-dimensional contour plot (Figure 1) to demonstrate the result of a successful feed...

Scheme 1: First steps towards the formation of sesquiterpenes. The (S)-germacradienyl cation can be formed fr...

Scheme 2: Possible biosynthetic pathways of the sesquiterpene hydrocarbons d8-α-copaene, d8-β-copaene, d8-α-c...

Scheme 3: Mechanistic rationale for the generation of the sesquiterpene hydrocarbons δ-cadinene (14), α-copae...

Figure 4: MS spectra of genuine (d0) and deuterium-labeled (d6 and d8) α-cubebene (left panel) after administ...

Scheme 4: Putative formation pathways of the sesquiterpene hydrocarbons α-ylangene (5), β-ylangene (6), β-bou...

Figure 5: MS spectra and expected labeling patterns of A: d0-α-ylangene, B: d8-α-ylangene after administratio...

Figure 6: Expected labeling patterns of deuterium-labeled, aromatic sesquiterpenes after administration of [6...

Figure 7: MS spectra and expected labeling patterns of genuine and deuterium-labeled A: calamenene (isomer) a...

Figure 8: MS spectra and expected labeling patterns of genuine (d0) and deuterium-labeled (d9) β-elemene afte...

Scheme 5: Possible biosynthesis of d9-β-elemene, d9-(+)-valencene and d9-α-guaiene via germacrene A. *An inco...

Scheme 6: Mechanistic rationale for the generation of the sesquiterpene hydrocarbons γ-elemene and selina-3,7...

Figure 9: Mass spectra and associated structural formulas of d0-γ-elemene and d9-γ-elemene after administrati...

Figure 10: MS spectra and expected labeling patterns of genuine (d0) and deuterium-labeled (d9) guaiazulene af...

Scheme 7: Possible synthesis of d9-guaiazulene, d9-δ-elemene, d9-guaia-6,9-diene and d9-δ-selinene via germac...

Scheme 8: Possible biosynthesis of d6-(E)-β-caryophyllene and d5-α-humulene starting from farnesyl pyrophosph...

Figure 11: MS spectra and expected labeling patterns of d0-(E)-β-caryophyllene and d6-(E)-β-caryophyllene afte...
Nanopatterns of arylene–alkynylene squares on graphite: self-sorting and intercalation
- Tristan J. Keller,
- Joshua Bahr,
- Kristin Gratzfeld,
- Nina Schönfelder,
- Marcin A. Majewski,
- Marcin Stępień,
- Sigurd Höger and
- Stefan-S. Jester
Beilstein J. Org. Chem. 2019, 15, 1848–1855, doi:10.3762/bjoc.15.180
- and non-planar coronoid polycyclic aromatic hydrocarbons (i.e., butyloxy-substituted kekulene and octulene derivatives) are found to be able to intercalate into the intramolecular nanopores. Keywords: macrocycles; scanning tunneling microscopy; self-assembled monolayers; self-sorting; solid/liquid
- nanopores from the gas phase [15], the intercalation of organic molecules into nanopores is rather difficult to tailor from scratch, however, with prominent examples [16][17]. Likewise, larger polycyclic aromatic hydrocarbons (PAHs) and nanographenes form robust adsorbate films in a certain size range, and
- –alkynylene macrocycles 1a/b (Figure 1), and the intercalation of solvent molecules and polycyclic aromatic hydrocarbons (PAHs). Our way towards nanoporous quadratic templates containing long (OC16H33) alkoxy side chains on two opposite sides of the square and two shorter (OC10H21, OC6H13) side chains on the

Graphical Abstract

Figure 1: Chemical structures of the molecular squares 1a/b, the kekulene derivative 2, and octulene derivati...

Figure 2: (a)–(c) Scanning tunneling microscopy images, (d)–(f) supramolecular models, and (g)–(l) schematic ...

Figure 3: (a) Overview scanning tunneling microscopy image of a nanopattern of 1a with intermolecularly inter...

Figure 4: (a–c) Scanning tunneling microscopy images of a nanopattern of 1a with intermolecularly intercalate...






