Search results
Search for "oxidation" in Full Text gives 1278 result(s) in Beilstein Journal of Organic Chemistry. Showing first 200.
A series of perylene diimide cathode interlayer materials for green solvent processing in conventional organic photovoltaics
Beilstein J. Org. Chem. 2023, 19, 1620–1629, doi:10.3762/bjoc.19.119

- properties of the four CILs were probed using solution cyclic voltammetry (CV; Figure 4) and differential pulse voltammetry (DPV; Supporting Information File 1, Figures S27–S30), using dichloromethane as the solvent. For all reversible reduction or oxidation waves, HOMO and LUMO energy levels were determined
- using E1/2 values with Fc/Fc+ as the internal standard. All compounds exhibit two reversible reduction waves, where only PDIN-FB and PDIN-B exhibit a reversible oxidation wave. For CN-PDIN-FB and CN-PDIN-B, the HOMO is estimated using the optical band gap by subtracting the value in eV from the LUMO
Radical chemistry in polymer science: an overview and recent advances
Beilstein J. Org. Chem. 2023, 19, 1580–1603, doi:10.3762/bjoc.19.116
- based on the radical polymerization of catechol derivatives. Catechols are known as easily oxidizable compounds and are prone to undergo oxidation by losing one or two electrons [3]. This way, either semiquinone radicals or o-quinones are formed by single or double-electron oxidation, respectively [4
- ]. The semiquinone radicals formed during the oxidation of catechol can undergo a cross-coupling reaction to form polymers (Scheme 1). One example is the radical polymerization of urushiol. The earliest recorded application of natural radical polymerization can be traced back to the manufacture of
- and in-depth dialogs between the organic and polymer communities. Therefore, future opportunities for polymer science evolution lie in the collaboration of radical chemists in both communities. Used abbreviations in the text and their explanations are collected in Table 2. Oxidation of catechol and
Synthesis and biological evaluation of Argemone mexicana-inspired antimicrobials
Beilstein J. Org. Chem. 2023, 19, 1511–1524, doi:10.3762/bjoc.19.108
- smoothly to the dihydroberberine in the absence of the copper salt [37]. This suggests the Cu2+ may be involved in aiding in the air-oxidation to the fully aromatic berberine core. The prime benefit of the route shown in Scheme 1 is the ease of introducing structural variability, as the precursor is easily
- B2 showed one less aromatic proton than expected and mass spectrometry revealed the presence of an extra oxygen. It was initially thought this unexpected oxidation had occurred at position-8, leading to an 8-oxoberberine variant. However, oxidation at position-8 was questionable (qualitatively) as 8
- -oxoberberine has been reported as a white solid, while our oxidized product maintained the bright yellow color of berberine [38]. This same unexpected oxidation was observed, to varying degrees, in the production of our next two variants wherein the expected products B3 and B5, respectively were isolated as a
N-Sulfenylsuccinimide/phthalimide: an alternative sulfenylating reagent in organic transformations
Beilstein J. Org. Chem. 2023, 19, 1471–1502, doi:10.3762/bjoc.19.106

- nucleophilic attack of TMSN3 to deliver product 11 (Scheme 7). Tian and Chang et al. could synthesize 3‑sulfenylated coumarin compounds 13 by using N-sulfanylsuccinimides 1 under a Lewis acid catalysis system (Scheme 8) [48]. Additionally, oxidation of 3-sulfenylated coumarins utilizing (diacetoxyiodo)benzene
- PhSO2· radical to obtain intermediate III. Radical II underwent oxidation with PhSO2· to form alkenyl cation IV and PhSO2−. At last, H-abstraction from DMF delivered product 71 and the Ni(0) species to continue the catalytic cycle. In 2022, Gao and co-workers introduced a new protocol for the
α-(Aminomethyl)acrylates as acceptors in radical–polar crossover 1,4-additions of dialkylzincs: insights into enolate formation and trapping
Beilstein J. Org. Chem. 2023, 19, 1443–1451, doi:10.3762/bjoc.19.103
- enolate and a new R• that propagates the radical chain (Scheme 1). Initiation occurs upon oxidation of the dialkylzinc reagent by oxygen. The feasibility of such 1,4-addition reactions is fully reliant on the ease of the intermediate enoxyl radical to undergo alkylzinc-group transfer. Secondary α-carbonyl
Synthesis of ether lipids: natural compounds and analogues
Beilstein J. Org. Chem. 2023, 19, 1299–1369, doi:10.3762/bjoc.19.96
- (plasmalogen) is highly sensitive to oxidation and it is reported that this function is even more reactive than unsaturated and polyunsaturated lipid chains [27]. Accordingly, plasmalogens could act as ROS scavenger and thus protect tissues (e.g., brain) from oxidative stress. The replacement of an ester
- oxidation with Br2 and hydrolysis, the bromoethyl phosphate 5.4. Finally, the quaternarization with trimethylamine produced 5.5 and the acetylation produced 5.6 PAF. The intermediate compounds like 6.2 (1-O-alkylglycerol) or the protected secondary alcohol 6.6, either as enantiopure or racemic forms, are
- acylation of lyso-PAF with a series of functionalized carboxylic acid was reported in a series of articles from the group of Salomon [98][99]. This group aimed to identify natural compounds that could be formed by the oxidation of ether lipids featuring a polyunsaturated acyl chain in sn-2 position. This
Organic thermally activated delayed fluorescence material with strained benzoguanidine donor
Beilstein J. Org. Chem. 2023, 19, 1289–1298, doi:10.3762/bjoc.19.95
- benzoguanidine moieties. This explains the ca. 0.2 eV more stabilized LUMO energy level for compound 4BGIPN compared with 4CzIPN. Both 4CzIPN and 4BGIPN exhibit an irreversible oxidation wave observed at +1.25 V for 4BGIPN in THF and +0.94 V for 4CzIPN in MeCN [15]. A higher oxidation potential (Ep) for 4BGIPN
Non-noble metal-catalyzed cross-dehydrogenation coupling (CDC) involving ether α-C(sp3)–H to construct C–C bonds
Beilstein J. Org. Chem. 2023, 19, 1259–1288, doi:10.3762/bjoc.19.94
- . Route b: the α-C(sp3)–H bonds are activated by a combination of transition metals and radical initiators to give the alkyl radicals, which are coupled with other radical receptors to afford the target product. Cu-catalyzed reactions Copper (common oxidation states are +I, +II and +III) has a
- involved in the activation of DDQ by coordinating the carbonyl oxygen atom which leads to an increase in the oxidation activity of DDQ. Subsequently, Li et al. improved the above method, using a mixture of indium and copper salts as a catalyst, NHPI (N-hydroxyphthalimide) as a co-catalyst to achieve the
- has been proved in many reports, and (3) oxidation of radical B to provide the corresponding alkenyl products 48. In recent years, the CDC reaction of alkyl C(sp3)–H substrates with the C(sp2)–H of an aromatic, which allows the construction of highly diverse compounds, has attracted considerable
Radical ligand transfer: a general strategy for radical functionalization
Beilstein J. Org. Chem. 2023, 19, 1225–1233, doi:10.3762/bjoc.19.90
- development. Outside of bioinorganic chemistry, the concept of radical ligand transfer was investigated in early work by Jay Kochi in purely synthetic systems (Scheme 1) [26][27]. Studies on the oxidation of alkyl radicals with earth abundant cupric salts uncovered the ability of simple Cu(II) chloride to
- (XAT) from the alkyl halide reagent and further oxidation of the transient radical to a carbocation by radical polar crossover (RPC), providing two mechanistic pathways to form the ATRA products [32]. While powerful, this approach is inherently incompatible with introducing alternative functionality
- captured via RLT from an in-situ generated iron–azide complex, resulting in net reduction of iron. The competent RLT species can then be regenerated through oxidation by the iodinane species and coordination of another equivalent of azide. This reaction was particularly notable for the wide alkene scope
Enabling artificial photosynthesis systems with molecular recycling: A review of photo- and electrochemical methods for regenerating organic sacrificial electron donors
Beilstein J. Org. Chem. 2023, 19, 1198–1215, doi:10.3762/bjoc.19.88
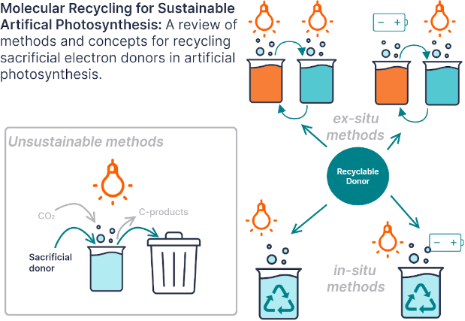
- artificial photosynthesis. Systems for photocatalytic carbon dioxide reduction are optimized using sacrificial electron donors. One strategy for coupling carbon dioxide reduction and water oxidation to achieve artificial photosynthesis is to use a redox mediator, or recyclable electron donor. This review
- decoupled water splitting research, are introduced as alternative recyclable sacrificial electron donors and their oxidation potentials are compared to the redox potentials of some model photosensitizers. The aim of this review is to act as a reference for researchers developing photocatalytic systems with
- photosynthesis systems species other than water are consumed to provide these electrons and protons [3][4]. Ideally these sacrificial donors would be replaced with redox mediators, regenerated using water, or form stable, commercially valuable oxidation products. However, common sacrificial electron donors, such
Two new lanostanoid glycosides isolated from a Kenyan polypore Fomitopsis carnea
Beilstein J. Org. Chem. 2023, 19, 1161–1169, doi:10.3762/bjoc.19.84
- potency against cancer cells in the presence of a 3-OH group. Notably, hydrolysis of the C3-acetoxy group in pachymic acid to tumulosic acid increased the activity of the compound compared to the positive control (cisplatin), in some instances [36]. Concomitantly, the oxidation of the hydroxy group at C-3
New one-pot synthesis of 4-arylpyrazolo[3,4-b]pyridin-6-ones based on 5-aminopyrazoles and azlactones
Beilstein J. Org. Chem. 2023, 19, 1155–1160, doi:10.3762/bjoc.19.83
- are also low in two-stage synthesis methods. The first of them is based on the three-component condensation of aminopyrazoles, Meldrum's acid, and aromatic aldehydes, followed by the oxidation of the intermediate with DDQ [13][16][19] (method B). The second one includes the reaction of an aromatic
Selective and scalable oxygenation of heteroatoms using the elements of nature: air, water, and light
Beilstein J. Org. Chem. 2023, 19, 1146–1154, doi:10.3762/bjoc.19.82
- , 9800 Deinze, Belgium 10.3762/bjoc.19.82 Abstract Sustainable oxidation protocols aim to provide an environmentally friendly and cost-effective method for the production of various chemicals and materials. The development of such protocols can lead to reduced energy consumption, fewer harmful
- byproducts, and increased efficiency in industrial processes. As such, this field of research is of great importance and interest to both academia and industry. This work showcases a sustainable and catalyst-free oxidation method for heteroatoms (e.g., S, P, and Se) using only air, water and light. An
- flow using the HANU flow reactor, indicating scalability and improving safety. Keywords: catalyst-free; flow chemistry; oxygen; photochemistry; sustainable oxidation; Introduction Oxidation reactions are widely used in the chemical industry, but are often problematic due to challenges with
Photoredox catalysis harvesting multiple photon or electrochemical energies
Beilstein J. Org. Chem. 2023, 19, 1055–1145, doi:10.3762/bjoc.19.81
- generation of super-reductants [15] and by Wagenknecht in 2018 for the generation of super-oxidants [16]. Herein, initial excitation of the photocatalyst by a single photon is followed by reduction or oxidation by a sacrificial SET donor (e.g., Et3N [15]) or acceptor (e.g., SF6 [16]) to yield the catalyst
- doublet states which are photoexcited to yield super-oxidants or super-reductants while recycling e-PRC involves the turnover of a ‘standard’ (typically closed-shell) photoredox catalyst (PC) by means of anodic oxidation or cathodic reduction [28][29]. Furthermore, a series of new protocols using
- SCE (1c) were readily reduced and dehalogenated products obtained in excellent yields (70–92%) (Figure 11A). Sodium formate was found to be a more efficient terminal reductant than trialkylamines which the authors attributed to the formation of a carbon dioxide radical anion (CO2•−) upon oxidation of
The effect of dark states on the intersystem crossing and thermally activated delayed fluorescence of naphthalimide-phenothiazine dyads
Beilstein J. Org. Chem. 2023, 19, 1028–1046, doi:10.3762/bjoc.19.79

- capability of the PTZ and NI moieties, respectively, by oxidation of the PTZ unit, or by using different aryl substituents attached to the NI unit. This tuning effect was manifested in the UV–vis absorption and fluorescence emission spectra, e.g., in the change of the charge transfer absorption bands. TADF
- transient absorption spectra showed that the charge separation takes ca. 0.6 ps, and admixtures of locally excited (3LE) state and charge separated (1CS/3CS) states formed (in n-hexane). The subsequent charge recombination from the 1CS state takes ca. 7.92 ns. Upon oxidation of the PTZ unit, the beginning
- the energy ordering of the transient species involved in the TADF photophysical process, the electron-donating strength of the PTZ moiety is lowered by oxidation of the sulfur atom to the corresponding sulfoxide. Conversely, the electron-accepting capability of the NI unit is varied by introducing
Copper-catalyzed N-arylation of amines with aryliodonium ylides in water
Beilstein J. Org. Chem. 2023, 19, 1008–1014, doi:10.3762/bjoc.19.76
- in oxidation, C–C, C–X bond formation, rearrangements, and halogenation reactions [23][24][25]. Due to the nontoxic nature, easier preparation, and handling of the hypervalent iodine reagents, many researchers are attracted to unravel the chemistry and reactivity of these reagents. Amongst different
Aromatic C–H bond functionalization through organocatalyzed asymmetric intermolecular aza-Friedel–Crafts reaction: a recent update
Beilstein J. Org. Chem. 2023, 19, 956–981, doi:10.3762/bjoc.19.72
- reaction between α-naphthol (17) and methyl 2-acetamidoacrylate (18) but promising selectivity was not achieved. The highest enantiomeric excess of 64% was obtained in the presence of P7 as the catalyst (Scheme 6) [30]. In 2018, Reddy and co-workers developed a one pot protocol comprising oxidation and an
- enantioselective aza-Friedel–Crafts addition. In the first step, the DDQ-promoted oxidation of 3-indolinonecarboxylate 22 generated indolenines that performed as the potential electrophiles towards indoles 4. The chiral catalyst effectively assembled the reacting partners in a chiral transition state through H
- ketimines 52 proceeding through C2 functionlization and follow up oxidation to provide 2-substitued indoles 56 which are typically difficult to obtain directly from unsubstituted indoles through electrophilic substitution. The process was catalyzed by the chiral phosphoric acid P17 to install a quaternary
Photoredox catalysis enabling decarboxylative radical cyclization of γ,γ-dimethylallyltryptophan (DMAT) derivatives: formal synthesis of 6,7-secoagroclavine
Beilstein J. Org. Chem. 2023, 19, 918–927, doi:10.3762/bjoc.19.70
- manner due to their intrinsic mildness and broad substrate compatibility [16][17][18][19][20]. This transformative synthetic tool often utilizes direct single-electron transfer (SET) between an electronically excited photoredox catalyst and an organic substrate, resulting in oxidation or reduction, to
- their ability to participate in either redox step of the catalytic cycle [42][43][44][45]. For example, the main use of α-amino acids in syntheses via photoredox catalysis is as readily available precursors of regioselective α-amino radicals by decarboxylative transformations, by oxidation of the
- selectively targeted by photoredox catalysis to enable unprecedented modification of the amino acid. In this context, it is worth mentioning that the single-electron oxidation of the indole moiety in tryptophan provides the radical cation, which enables selective C-radical generation at the weaker benzylic
Intermediates and shunt products of massiliachelin biosynthesis in Massilia sp. NR 4-1
Beilstein J. Org. Chem. 2023, 19, 909–917, doi:10.3762/bjoc.19.69
- reactions (e.g., hydrolysis, esterification, oxidation) might be due to the isolation conditions or they could be attributed to unspecific enzymatic biotransformations. For compound 1, no spontaneous conversion to the ester 2 was observed, even after storage in methanol for two months. In contrast, the
Pyridine C(sp2)–H bond functionalization under transition-metal and rare earth metal catalysis
Beilstein J. Org. Chem. 2023, 19, 820–863, doi:10.3762/bjoc.19.62
- intact under the reaction conditions without any further oxidation. Different oxidants resulted in different products such as the monoarylated product 118 formed in the presence of TBHP as oxidant and the benzylated product 119 was obtained when potassium persulfate was used. Interestingly, aza-fluorene
Eschenmoser coupling reactions starting from primary thioamides. When do they work and when not?
Beilstein J. Org. Chem. 2023, 19, 808–819, doi:10.3762/bjoc.19.61
- ) as a polar aprotic solvent that can be easily distilled-off after the ECR under reduced pressure. The last complication with aromatic primary thioamides concerns their easy oxidation with electron-poor α-haloesters (acetoacetates and α-cyanoacetates). As early as in 1976 Potts and Marshall noticed
- File 1. Only oxidation of thiobenzamide to 3,5-diphenyl-1,2,4-thiadiazole was never observed. Triethylamine (pKa = 10.67 in water, 9.25 in DMF, and 18.5 in MeCN) [22] causes the decomposition of imidothioates 6a,b''' in both polar aprotic solvents (DMF and MeCN) through the elimination route. For
Sulfate radical anion-induced benzylic oxidation of N-(arylsulfonyl)benzylamines to N-arylsulfonylimines
Beilstein J. Org. Chem. 2023, 19, 771–777, doi:10.3762/bjoc.19.57
- with ortho-substituted anilines is also reported. The key features of the protocol include the use of a green oxidant, a short reaction time (30 min), chromatography-free isolation, scalability, and economical, delivering N-arylsulfonylimines in excellent yields of up to 96%. While the oxidation of N
- -aryl(benzyl)amines to N-arylimines using K2S2O8 is reported to be problematic, the oxidation of N-(arylsulfonyl)benzylamines to N-arylsulfonylimines using K2S2O8 has been achieved for the first time. The dual role of the sulfate radical anion (SO4·−), including hydrogen atom abstraction (HAT) and
- single electron transfer (SET), is proposed to be involved in the plausible reaction mechanism. Keywords: arylsulfonylimine; benzylic oxidation; benzyl sulfonamide; K2S2O8; sulfate radical anion; Introduction Among various imine compounds [1], N-arylsulfonylimines are perhaps the most prominent due to
Honeycomb reactor: a promising device for streamlining aerobic oxidation under continuous-flow conditions
Beilstein J. Org. Chem. 2023, 19, 752–763, doi:10.3762/bjoc.19.55

- 10.3762/bjoc.19.55 Abstract We report on the high potential of a honeycomb reactor for the use in aerobic oxidation under continuous-flow conditions. The honeycomb reactor is made of porous material with narrow channels separated by porous walls allowing for high density accumulation in the reactor. This
- structure raised the mixing efficiency of a gas–liquid reaction system, and it effectively accelerated the aerobic oxidation of benzyl alcohols to benzaldehydes under continuous-flow conditions. This reactor is a promising device for streamlining aerobic oxidation with high process safety because it is a
- closed system. Keywords: aerobic oxidation; benzaldehydes; benzyl alcohols; homogeneous catalyst; honeycomb reactor; Introduction Oxidation plays a key role in synthesizing highly functionalized molecules [1][2]. While Jones oxidation [3] and oxidation using KMnO4 [4] are classical and powerful methods
Construction of hexabenzocoronene-based chiral nanographenes
Beilstein J. Org. Chem. 2023, 19, 736–751, doi:10.3762/bjoc.19.54

- dibenzocyclooctyne 8 and tetracyclone 2 in a 91% yield. After a subsequent sequence of deprotection and oxidation, ketone 10 was obtained. Through the oxidative cyclodehydrogenation reaction of 10 in the presence of DDQ and trifluoromethanesulfonic acid (TfOH), a saddle-helix hybrid nanographene 11, bearing an
- . And the derivative 16 was also synthesized by allylic oxidation of compound 15 using selenium dioxide. As helical chiral NGs, helicene 14 and its derivatives 15 and 16 showed highly distorted helical conformation and also exhibited a relatively high isomerization barrier (over 28.9 kcal/mol determined
- nanographenes 96 and 100 using helicene or oxa-helicene as the linkers, respectively [56]. The first π-extension started from the borylated penta-tert-butyl HBC 92. The chemical selective Suzuki−Miyaura cross-coupling reaction between 92 and 93 followed by Scholl oxidation produced compound 94 in an overall 50
Strategies in the synthesis of dibenzo[b,f]heteropines
Beilstein J. Org. Chem. 2023, 19, 700–718, doi:10.3762/bjoc.19.51

- nitrotoluene (22) under alkaline conditions (e.g., O2, KOt-Bu; O2, KOH, MeOH, ethylenediamine, etc.), as reported by Stansbury and Proops [33]. Aerobic oxidation of 22 in alkaline methanol with added ethylenediamine, gave 21 in 36% yield (Scheme 2), which is poor compared to that reported for the p-nitro
- 24 was reported by Cong et al. [45] as a method for the synthesis of substituted dibenzo[b,f]oxepines 25 (Scheme 6). Treatment of the malonate derivative 24 with Mn(OAc)3 in 90% acetic acid gave C-10 carboxylate derivatives of dibenzo[b,f]oxepine 25. The authors proposed a one-electron oxidation of
- Sargent [18] synthesised pacharin (13) using a novel method through oxidation of a bisphosphonium diphenyl ether prepared in situ from dibromide 130 (Scheme 28). On treatment with base and exposure to oxygen, the diylide intermediate underwent oxidative coupling to give the isopropyl-protected dibenzo[b,f












































































































































































































































































































































































































































































































































































































