Search results
Search for "thiazole" in Full Text gives 88 result(s) in Beilstein Journal of Organic Chemistry.
Copper-based fluorinated reagents for the synthesis of CF2R-containing molecules (R ≠ F)
- Louise Ruyet and
- Tatiana Besset
Beilstein J. Org. Chem. 2020, 16, 1051–1065, doi:10.3762/bjoc.16.92

- oxazoles (17 examples, up to 87% yield) were difluoromethylated but a variety of other heteroarenes turned out to be suitable such as pyridine, imidazole, benzo[d]thiazole, benzo[b]thiophene, benzo[d]oxazole, thiazole and thiophene derivatives (Scheme 6). Copper-based CF2FG-containing reagents Besides the

Graphical Abstract

Scheme 1: Synthesis of the first isolable (NHC)CuCF2H complexes from TMSCF2H and their application for the sy...

Scheme 2: Pioneer works for the in situ generation of CuCF2H from TMSCF2H and from n-Bu3SnCF2H. Phen = 1,10-p...

Scheme 3: A Sandmeyer-type difluoromethylation reaction via the in situ generation of CuCF2H from TMSCF2H. a ...

Scheme 4: A one pot, two-step sequence for the difluoromethylthiolation of various classes of compounds via t...

Scheme 5: A copper-mediated oxidative difluoromethylation of terminal alkynes via the in situ generation of a...

Scheme 6: A copper-mediated oxidative difluoromethylation of heteroarenes.

Scheme 7: Synthesis of difluoromethylphosphonate-containing molecules using the in situ-generated CuCF2PO(OEt)...

Scheme 8: Synthesis of difluoromethylphosphonate-containing molecules using in situ-generated CuCF2PO(OEt)2 s...

Scheme 9: Synthesis of difluoromethylphosphonate-containing molecules using in situ-generated CuCF2PO(OEt)2 s...

Scheme 10: Synthesis of (diethylphosphono)difluoromethylthiolated molecules using in situ-generated CuCF2PO(OE...

Scheme 11: Access to (diethylphosphono)difluoromethylthiolated molecules via the in situ generation of CuCF2PO...

Scheme 12: Synthesis of (phenylsulfonyl)difluoromethyl-containing molecules via the in situ generation of CuCF2...

Scheme 13: Copper-mediated 1,1-difluoroethylation of diaryliodonium salts by using the in situ-generated CuCF2...

Scheme 14: Pioneer works for the pentafluoroethylation and heptafluoropropylation using a copper-based reagent...

Scheme 15: Pentafluoroethylation of (hetero)aryl bromides using the (Phen)CuCF2CF3 complex. 19F NMR yields wer...

Scheme 16: Synthesis of pentafluoroethyl ketones using the (Ph3P)Cu(phen)CF2CF3 reagent. 19F NMR yields were g...

Scheme 17: Synthesis of (Phen)2Cu(O2CCF2RF) and functionalization of (hetero)aryl iodides.

Scheme 18: Pentafluoroethylation of arylboronic acids and (hetero)aryl bromides via the in situ-generated CuCF2...

Scheme 19: In situ generation of CuCF2CF3 species from a cyclic-protected hexafluoroacetone and KCu(Ot-Bu)2. 19...

Scheme 20: Pentafluoroethylation of bromo- and iodoalkenes. Only examples of isolated compounds were depicted.

Scheme 21: Fluoroalkylation of aryl halides via a RCF2CF2Cu species.

Scheme 22: Synthesis of perfluoroorganolithium copper species or perfluroalkylcopper derivatives from iodoperf...

Scheme 23: Formation of the PhenCuCF2CF3 reagent by means of TFE and pentafluoroethylation of iodoarenes and a...

Scheme 24: Generation of a CuCF2CF3 reagent from TMSCF3 and applications.
Photocontrolled DNA minor groove interactions of imidazole/pyrrole polyamides
- Sabrina Müller,
- Jannik Paulus,
- Jochen Mattay,
- Heiko Ihmels,
- Veronica I. Dodero and
- Norbert Sewald
Beilstein J. Org. Chem. 2020, 16, 60–70, doi:10.3762/bjoc.16.8
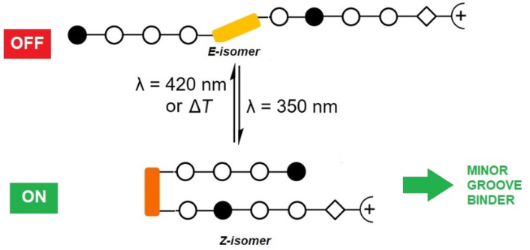
- ligand to examine the propensity of these derivatives to bind to DNA. The known intercalator thiazole orange (TO) was chosen as indicator because it exhibits an intense fluorescence band at 526 nm when bound to DNA, whereas it is only weakly fluorescent in solution. The displacement of TO from its DNA

Graphical Abstract

Scheme 1: Pyrrole–imidazole–azobenzene polyamides and the dsDNA target sequences employed in this study.

Scheme 2: Building blocks required for the synthesis of the photoswitchable Im/Py polyamides. A) Fmoc–Azo–OH 1...

Figure 1: Section of the 1H NMR (600 MHz) spectrum of polyamide P1. A) Initial thermal equilibrium. B) After ...

Figure 2: E/Z isomer ratio of the polyamides P1–P3. Values were obtained from the respective 1H NMR experimen...

Figure 3: Titration experiments of target DNA sequences with P1–P3 in the photostationary Z-state and the the...

Figure 4: Titration of DNA containing single mutations (in bold) with P1–P3 in the photostationary Z-state an...
Thermal stability of N-heterocycle-stabilized iodanes – a systematic investigation
- Andreas Boelke,
- Yulia A. Vlasenko,
- Mekhman S. Yusubov,
- Boris J. Nachtsheim and
- Pavel S. Postnikov
Beilstein J. Org. Chem. 2019, 15, 2311–2318, doi:10.3762/bjoc.15.223

- Supporting Information File 1) were observed at remarkable high Tpeak values (193.9–210.1 °C). Following these results, it has been intriguing to analyze the influence of the heteroatom in the heterocyclic moiety on the thermal decomposition process. The change of one nitrogen atom to sulfur as in thiazole
- model reaction, especially N-substituted triazoles 4 and 5. In contrast, the reactivity of triazole 3 is comparable to that of benzoxazoles 13 and 14 as well as pyrazole 7 and indazole 8. In our view thiazole 12 is the best compromise in this regard since it is thermally even more stable than 7 and 8

Graphical Abstract

Figure 1: General structure of aryl-λ3-iodanes.

Figure 2: Tpeak and ΔHdec-values for a range of N- and O-substituted iodanes.

Figure 3: TGA/DSC curves of (a) benziodoxolone 1, (b) triazole 2 and (c) pyrazole 6.

Figure 4: Decomposition enthalpy (ΔHdec) scale for pseudocyclic tosylates 1–15 and cyclic iodoso species 16 a...

Figure 5: Correlation between the relative reactivity for pseudocyclic NHIs based on the reaction time in the...

Figure 6: Tpeak and ΔHdec values for a range of N- and O-substituted iodanes.

Figure 7: Decomposition enthalpy (ΔHdec) scale for (pseudo)cyclic mesitylen(phenyl)- λ3-iodanes 18–33.

Figure 8: TGA/DSC curves for the benzimidazole based diaryliodonium salt 25.

Figure 9: TGA/DSC curves for the cyclic triazole 32.

Scheme 1: The thermal decomposition of (pseudo)cyclic N-heterocycle-stabilized mesityl(aryl)-λ3-iodanes 25 an...
Click chemistry towards thermally reversible photochromic 4,5-bisthiazolyl-1,2,3-triazoles
- Chenxia Zhang,
- Kaori Morinaka,
- Mahmut Kose,
- Takashi Ubukata and
- Yasushi Yokoyama
Beilstein J. Org. Chem. 2019, 15, 2161–2169, doi:10.3762/bjoc.15.213

- (thiazole) [39], and 10c (imidazole) [40] in non-polar solvents, 1c has a much longer absorption maximum in toluene (Scheme 4, Table 2). It should also be noted that the absorption maximum wavelength is longer when the central ethene moiety is part of the aromatic ring than when it is an isolated ethene in
- AcOEt than in toluene, and the thermal back reaction rate in AcOEt was increased. Conclusion We have synthesized three novel thermally reversible 4,4'-(1-benzyl-1H-1,2,3-triazole-4,5-diyl)bis(5-methyl-2-(4-substituted-phenyl)thiazole)s 1o–3o by Ru(I)-catalysed Huisgen cyclization, which is a type of

Graphical Abstract

Scheme 1: Reaction mechanisms of Huisgen cyclization catalyzed by Cu(I) and Ru(I).

Scheme 2: Synthesis and photochromism of bisthiazolyltriazoles.

Figure 1: Absorption spectral change of triazoles 1o–3o upon irradiation of 313 nm light in MeCN at 28 °C. Li...

Scheme 3: Wavelengths of absorption maxima of the closed forms of bisthienyletenes in hexane [36].

Scheme 4: Photochromism of closely related compounds.

Figure 2: Absorption spectral change of triazoles 1c–3c during the thermal back reaction after 313-nm light i...

Scheme 5: Bond length (a) (in Å) and Mulliken bond order (b) of 1c–3c obtained by DFT calculations. Top numbe...

Scheme 6: Possible reaction mechanism of thermal ring opening of the closed forms.
Identification of optimal fluorescent probes for G-quadruplex nucleic acids through systematic exploration of mono- and distyryl dye libraries
- Xiao Xie,
- Michela Zuffo,
- Marie-Paule Teulade-Fichou and
- Anton Granzhan
Beilstein J. Org. Chem. 2019, 15, 1872–1889, doi:10.3762/bjoc.15.183

- , the widely used fluorescent probes, such as thioflavin T (ThT, Φ = 0.25, in the presence of 22AG/K+ conditions) and thiazole orange (TO, Φ = 0.19 in the presence of 22AG/K+ conditions) [92], and approach the brightest G4-DNA probes developed so far, such as trialryimidazole IZCM-7 (Φ = 0.52, in the

Graphical Abstract

Figure 1: Some di- and mono-styryl dyes previously reported as fluorescent “light-up” probes for G4-DNA and R...

Figure 2: Design of a library of di- and mono-styryl dyes. Counter-ions are omitted for the sake of clarity.

Scheme 1: A, B) General synthesis of A) distyryl and B) mono-styryl dyes via Knoevenagel condensation route. ...

Scheme 2: Synthesis of I3–5 and I15.

Figure 3: Representative absorption spectra of distyryl dyes: A) 1d, B) 1ð, C) 1f, D) 1u, E) 10a and F) 12a i...

Figure 4: Representative absorption spectra of the distyryl dyes (c = 10 µM in MeOH) demonstrating the influe...

Figure 5: Heat map of the relative emission intensity enhancement (I/I0) of styryl dyes and thioflavin T (ThT...

Figure 6: Analysis of the light-up response matrix of the dyes. The average light-up factor of each dye with ...

Figure 7: PC1 vs PC2 plot obtained from the principal component analysis of the light-up data matrix for all ...

Figure 8: Dual-dye conformational analysis of an extended panel of 33 DNA oligonucleotides. This is performed...

Figure 9: Selected probes featuring high fluorimetric response towards G4 structures.

Figure 10: Photographs of solutions of A) distyryl dyes 1p and 1u; B) mono-styryl dyes 17a and 18a, in the abs...
Recent advances on the transition-metal-catalyzed synthesis of imidazopyridines: an updated coverage
- Gagandeep Kour Reen,
- Ashok Kumar and
- Pratibha Sharma
Beilstein J. Org. Chem. 2019, 15, 1612–1704, doi:10.3762/bjoc.15.165


Graphical Abstract

Figure 1: Various drugs having IP nucleus.

Figure 2: Participation percentage of various TMs for the syntheses of IPs.

Scheme 1: CuI–NaHSO4·SiO2-catalyzed synthesis of imidazo[1,2-a]pyridines.

Scheme 2: Experimental examination of reaction conditions.

Scheme 3: One-pot tandem reaction for the synthesis of 2-haloimidazopyridines.

Scheme 4: Mechanistic scheme for the synthesis of 2-haloimidazopyridine.

Scheme 5: Copper-MOF-catalyzed three-component reaction (3-CR) for imidazo[1,2-a]pyridines.

Scheme 6: Mechanism for copper-MOF-driven synthesis.

Scheme 7: Heterogeneous synthesis via titania-supported CuCl2.

Scheme 8: Mechanism involving oxidative C–H functionalization.

Scheme 9: Heterogeneous synthesis of IPs.

Scheme 10: One-pot regiospecific synthesis of imidazo[1,2-a]pyridines.

Scheme 11: Vinyl azide as an unprecedented substrate for imidazo[1,2-a]pyridines.

Scheme 12: Radical pathway.

Scheme 13: Cu(I)-catalyzed transannulation approach for imidazo[1,5-a]pyridines.

Scheme 14: Plausible radical pathway for the synthesis of imidazo[1,5-a]pyridines.

Scheme 15: A solvent-free domino reaction for imidazo[1,2-a]pyridines.

Scheme 16: Cu-NPs-mediated synthesis of imidazo[1,2-a]pyridines.

Scheme 17: CuI-catalyzed synthesis of isoxazolylimidazo[1,2-a]pyridines.

Scheme 18: Functionalization of 4-bromo derivative via Sonogashira coupling reaction.

Scheme 19: A plausible reaction pathway.

Scheme 20: Cu(I)-catalyzed intramolecular oxidative C–H amidation reaction.

Scheme 21: One-pot synthetic reaction for imidazo[1,2-a]pyridine.

Scheme 22: Plausible reaction mechanism.

Scheme 23: Cu(OAc)2-promoted synthesis of imidazo[1,2-a]pyridines.

Scheme 24: Mechanism for aminomethylation/cycloisomerization of propiolates with imines.

Scheme 25: Three-component synthesis of imidazo[1,2-a]pyridines.

Figure 3: Scope of pyridin-2(1H)-ones and acetophenones.

Scheme 26: CuO NPS-promoted A3 coupling reaction.

Scheme 27: Cu(II)-catalyzed C–N bond formation reaction.

Scheme 28: Mechanism involving Chan–Lam/Ullmann coupling.

Scheme 29: Synthesis of formyl-substituted imidazo[1,2-a]pyridines.

Scheme 30: A tandem sp3 C–H amination reaction.

Scheme 31: Probable mechanistic approach.

Scheme 32: Dual catalytic system for imidazo[1,2-a]pyridines.

Scheme 33: Tentative mechanism.

Scheme 34: CuO/CuAl2O4/ᴅ-glucose-promoted 3-CCR.

Scheme 35: A tandem CuOx/OMS-2-based synthetic strategy.

Figure 4: Biomimetic catalytic oxidation in the presence of electron-transfer mediators (ETMs).

Scheme 36: Control experiment.

Scheme 37: Copper-catalyzed C(sp3)–H aminatin reaction.

Scheme 38: Reaction of secondary amines.

Scheme 39: Probable mechanistic pathway.

Scheme 40: Coupling reaction of α-azidoketones.

Scheme 41: Probable pathway.

Scheme 42: Probable mechanism with free energy calculations.

Scheme 43: MCR for cyanated IP synthesis.

Scheme 44: Substrate scope for the reaction.

Scheme 45: Reaction mechanism.

Scheme 46: Probable mechanistic pathway for Cu/ZnAl2O4-catalyzed reaction.

Scheme 47: Copper-catalyzed double oxidative C–H amination reaction.

Scheme 48: Application towards different coupling reactions.

Scheme 49: Reaction mechanism.

Scheme 50: Condensation–cyclization approach for the synthesis of 1,3-diarylated imidazo[1,5-a]pyridines.

Scheme 51: Optimized reaction conditions.

Scheme 52: One-pot 2-CR.

Scheme 53: One-pot 3-CR without the isolation of chalcone.

Scheme 54: Copper–Pybox-catalyzed cyclization reaction.

Scheme 55: Mechanistic pathway catalyzed by Cu–Pybox complex.

Scheme 56: Cu(II)-promoted C(sp3)-H amination reaction.

Scheme 57: Wider substrate applicability for the reaction.

Scheme 58: Plausible reaction mechanism.

Scheme 59: CuI assisted C–N cross-coupling reaction.

Scheme 60: Probable reaction mechanism involving sp3 C–H amination.

Scheme 61: One-pot MCR-catalyzed by CoFe2O4/CNT-Cu.

Scheme 62: Mechanistic pathway.

Scheme 63: Synthetic scheme for 3-nitroimidazo[1,2-a]pyridines.

Scheme 64: Plausible mechanism for CuBr-catalyzed reaction.

Scheme 65: Regioselective synthesis of halo-substituted imidazo[1,2-a]pyridines.

Scheme 66: Synthesis of 2-phenylimidazo[1,2-a]pyridines.

Scheme 67: Synthesis of diarylated compounds.

Scheme 68: CuBr2-mediated one-pot two-component oxidative coupling reaction.

Scheme 69: Decarboxylative cyclization route to synthesize 1,3-diarylimidazo[1,5-a]pyridines.

Scheme 70: Mechanistic pathway.

Scheme 71: C–H functionalization reaction of enamines to produce diversified heterocycles.

Scheme 72: A plausible mechanism.

Scheme 73: CuI-promoted aerobic oxidative cyclization reaction of ketoxime acetates and pyridines.

Scheme 74: CuI-catalyzed pathway for the formation of imidazo[1,2-a]pyridine.

Scheme 75: Mechanistic pathway.

Scheme 76: Mechanistic rationale for the synthesis of products.

Scheme 77: Copper-catalyzed synthesis of vinyloxy-IP.

Scheme 78: Regioselective product formation with propiolates.

Scheme 79: Proposed mechanism for vinyloxy-IP formation.

Scheme 80: Regioselective synthesis of 3-hetero-substituted imidazo[1,2-a]pyridines with different reaction su...

Scheme 81: Mechanistic pathway.

Scheme 82: CuI-mediated synthesis of 3-formylimidazo[1,2-a]pyridines.

Scheme 83: Radical pathway for 3-formylated IP synthesis.

Scheme 84: Pd-catalyzed urea-cyclization reaction for IPs.

Scheme 85: Pd-catalyzed one-pot-tandem amination and intramolecular amidation reaction.

Figure 5: Scope of aniline nucleophiles.

Scheme 86: Pd–Cu-catalyzed Sonogashira coupling reaction.

Scheme 87: One-pot amide coupling reaction for the synthesis of imidazo[4,5-b]pyridines.

Scheme 88: Urea cyclization reaction for the synthesis of two series of pyridines.

Scheme 89: Amidation reaction for the synthesis of imidazo[4,5-b]pyridines.

Figure 6: Amide scope.

Scheme 90: Pd NPs-catalyzed 3-component reaction for the synthesis of 2,3-diarylated IPs.

Scheme 91: Plausible mechanistic pathway for Pd NPs-catalyzed MCR.

Scheme 92: Synthesis of chromenoannulated imidazo[1,2-a]pyridines.

Scheme 93: Mechanism for the synthesis of chromeno-annulated IPs.

Scheme 94: Zinc oxide NRs-catalyzed synthesis of imidazo[1,2-a]azines/diazines.

Scheme 95: Zinc oxide-catalyzed isocyanide based GBB reaction.

Scheme 96: Reaction pathway for ZnO-catalyzed GBB reaction.

Scheme 97: Mechanistic pathway.

Scheme 98: ZnO NRs-catalyzed MCR for the synthesis of imidazo[1,2-a]azines.

Scheme 99: Ugi type GBB three-component reaction.

Scheme 100: Magnetic NPs-catalyzed synthesis of imidazo[1,2-a]pyridines.

Scheme 101: Regioselective synthesis of 2-alkoxyimidazo[1,2-a]pyridines catalyzed by Fe-SBA-15.

Scheme 102: Plausible mechanistic pathway for the synthesis of 2-alkoxyimidazopyridine.

Scheme 103: Iron-catalyzed synthetic approach.

Scheme 104: Iron-catalyzed aminooxygenation reaction.

Scheme 105: Mechanistic pathway.

Scheme 106: Rh(III)-catalyzed double C–H activation of 2-substituted imidazoles and alkynes.

Scheme 107: Plausible reaction mechanism.

Scheme 108: Rh(III)-catalyzed non-aromatic C(sp2)–H bond activation–functionalization for the synthesis of imid...

Scheme 109: Reactivity and selectivity of different substrates.

Scheme 110: Rh-catalyzed direct C–H alkynylation by Li et al.

Scheme 111: Suggested radical mechanism.

Scheme 112: Scandium(III)triflate-catalyzed one-pot reaction and its mechanism for the synthesis of benzimidazo...

Scheme 113: RuCl3-assisted Ugi-type Groebke–Blackburn condensation reaction.

Scheme 114: C-3 aroylation via Ru-catalyzed two-component reaction.

Scheme 115: Regioselective synthetic mechanism.

Scheme 116: La(III)-catalyzed one-pot GBB reaction.

Scheme 117: Mechanistic approach for the synthesis of imidazo[1,2-a]pyridines.

Scheme 118: Synthesis of imidazo[1,2-a]pyridine using LaMnO3 NPs under neat conditions.

Scheme 119: Mechanistic approach.

Scheme 120: One-pot 3-CR for regioselective synthesis of 2-alkoxy-3-arylimidazo[1,2-a]pyridines.

Scheme 121: Formation of two possible products under optimization of the catalysts.

Scheme 122: Mechanistic strategy for NiFe2O4-catalyzed reaction.

Scheme 123: Two-component reaction for synthesizing imidazodipyridiniums.

Scheme 124: Mechanistic scheme for the synthesis of imidazodipyridiniums.

Scheme 125: CuI-catalyzed arylation of imidazo[1,2-a]pyridines.

Scheme 126: Mechanism for arylation reaction.

Scheme 127: Cupric acetate-catalyzed double carbonylation approach.

Scheme 128: Radical mechanism for double carbonylation of IP.

Scheme 129: C–S bond formation reaction catalyzed by cupric acetate.

Scheme 130: Cupric acetate-catalyzed C-3 formylation approach.

Scheme 131: Control experiments for signifying the role of DMSO and oxygen.

Scheme 132: Mechanism pathway.

Scheme 133: Copper bromide-catalyzed CDC reaction.

Scheme 134: Extension of the substrate scope.

Scheme 135: Plausible radical pathway.

Scheme 136: Transannulation reaction for the synthesis of imidazo[1,5-a]pyridines.

Scheme 137: Plausible reaction pathway for denitrogenative transannulation.

Scheme 138: Cupric acetate-catalyzed C-3 carbonylation reaction.

Scheme 139: Plausible mechanism for regioselective C-3 carbonylation.

Scheme 140: Alkynylation reaction at C-2 of 3H-imidazo[4,5-b]pyridines.

Scheme 141: Two-way mechanism for C-2 alkynylation of 3H-imidazo[4,5-b]pyridines.

Scheme 142: Palladium-catalyzed SCCR approach.

Scheme 143: Palladium-catalyzed Suzuki coupling reaction.

Scheme 144: Reaction mechanism.

Scheme 145: A phosphine free palladium-catalyzed synthesis of C-3 arylated imidazopyridines.

Scheme 146: Palladium-mediated Buchwald–Hartwig cross-coupling reaction.

Figure 7: Structure of the ligands optimized.

Scheme 147: Palladium acetate-catalyzed direct arylation of imidazo[1,2-a]pyridines.

Scheme 148: Palladium acetate-catalyzed mechanistic pathway.

Scheme 149: Palladium acetate-catalyzed regioselective arylation reported by Liu and Zhan.

Scheme 150: Mechanism for selective C-3 arylation of IP.

Scheme 151: Pd(II)-catalyzed alkenylation reaction with styrenes.

Scheme 152: Pd(II)-catalyzed alkenylation reaction with acrylates.

Scheme 153: A two way mechanism.

Scheme 154: Double C–H activation reaction catalyzed by Pd(OAc)2.

Scheme 155: Probable mechanism.

Scheme 156: Palladium-catalyzed decarboxylative coupling.

Scheme 157: Mechanistic cycle for decarboxylative arylation reaction.

Scheme 158: Ligand-free approach for arylation of imidazo[1,2-a]pyridine-3-carboxylic acids.

Scheme 159: Mechanism for ligandless arylation reaction.

Scheme 160: NHC-Pd(II) complex assisted arylation reaction.

Scheme 161: C-3 arylation of imidazo[1,2-a]pyridines with aryl bromides catalyzed by Pd(OAc)2.

Scheme 162: Pd(II)-catalyzed C-3 arylations with aryl tosylates and mesylates.

Scheme 163: CDC reaction for the synthesis of imidazo[1,2-a]pyridines.

Scheme 164: Plausible reaction mechanism for Pd(OAc)2-catalyzed synthesis of imidazo[1,2-a]pyridines.

Scheme 165: Pd-catalyzed C–H amination reaction.

Scheme 166: Mechanism for C–H amination reaction.

Scheme 167: One-pot synthesis for 3,6-di- or 2,3,6-tri(hetero)arylimidazo[1,2-a]pyridines.

Scheme 168: C–H/C–H cross-coupling reaction of IPs and azoles catalyzed by Pd(II).

Scheme 169: Mechanistic cycle.

Scheme 170: Rh-catalyzed C–H arylation reaction.

Scheme 171: Mechanistic pathway for C–H arylation of imidazo[1,2-a]pyridine.

Scheme 172: Rh(III)-catalyzed double C–H activation of 2-phenylimidazo[1,2-a]pyridines and alkynes.

Scheme 173: Rh(III)-catalyzed mechanistic pathway.

Scheme 174: Rh(III)-mediated oxidative coupling reaction.

Scheme 175: Reactions showing functionalization of the product obtained by the group of Kotla.

Scheme 176: Mechanism for Rh(III)-catalyzed oxidative coupling reaction.

Scheme 177: Rh(III)-catalyzed C–H activation reaction.

Scheme 178: Mechanistic cycle.

Scheme 179: Annulation reactions of 2-arylimidazo[1,2-a]pyridines and alkynes.

Scheme 180: Two-way reaction mechanism for annulations reaction.

Scheme 181: [RuCl2(p-cymene)]2-catalyzed C–C bond formation reaction.

Scheme 182: Reported reaction mechanism.

Scheme 183: Fe(III) catalyzed C-3 formylation approach.

Scheme 184: SET mechanism-catalyzed by Fe(III).

Scheme 185: Ni(dpp)Cl2-catalyzed KTC coupling.

Scheme 186: Pd-catalyzed SM coupling.

Scheme 187: Vanadium-catalyzed coupling of IP and NMO.

Scheme 188: Mechanistic cycle.

Scheme 189: Selective C3/C5–H bond functionalizations by mono and bimetallic systems.

Scheme 190: rGO-Ni@Pd-catalyzed C–H bond arylation of imidazo[1,2-a]pyridine.

Scheme 191: Mechanistic pathway for heterogeneously catalyzed arylation reaction.

Scheme 192: Zinc triflate-catalyzed coupling reaction of substituted propargyl alcohols.
Synthesis of ([1,2,4]triazolo[4,3-a]pyridin-3-ylmethyl)phosphonates and their benzo derivatives via 5-exo-dig cyclization
- Aleksandr S. Krylov,
- Artem A. Petrosian,
- Julia L. Piterskaya,
- Nataly I. Svintsitskaya and
- Albina V. Dogadina
Beilstein J. Org. Chem. 2019, 15, 1563–1568, doi:10.3762/bjoc.15.159
- ]thiadiazole [2], and benzo[4,5]imidazo[2,1-b]thiazole [3], as well as due to the simultaneous presence of a biologically active phosphorus function in the molecules. Recently, we have shown that the reaction of chloroethynylphosphonates with 2-aminopyridines proceeds through a 5-endo-dig cyclization to form

Graphical Abstract

Scheme 1: Synthetic approaches to [1,2,4]triazolo[4,3-a]pyridines.

Scheme 2: Synthesis of 3-methylphosphonylated [1,2,4]triazolo[4,3-a]pyridines. Reaction conditions: 1 (1 mmol...

Scheme 3: Synthesis of methylphosphonylated 6(8)-nitro-[1,2,4]triazolo[4,3-a]pyridines and 6(8)-nitro-[1,2,4]...

Scheme 4: Acid-promoted Dimroth rearrangement pathway.

Scheme 5: Synthesis of phosphonylated [1,2,4]triazolo[4,3-a]quinolines and [1,2,4]triazolo[3,4-a]isoquinoline...

Scheme 6: Plausible reaction pathway.
Steroid diversification by multicomponent reactions
- Leslie Reguera,
- Cecilia I. Attorresi,
- Javier A. Ramírez and
- Daniel G. Rivera
Beilstein J. Org. Chem. 2019, 15, 1236–1256, doi:10.3762/bjoc.15.121
- , but with the tetrahydropyrane ring having α orientation. A mixture of epimers at position 4' was also obtained when using the C-5 epimer of substrate 33. Asif et al. [36] developed another 3CR for the synthesis of steroidal thiazole derivatives. As shown in Scheme 11, cholestanic ketone 7 was reacted
- with a thiosemicarbazide and 2-bromo-1-phenylethan-1-one under microwave irradiation to form the steroid–thiazole hybrid 35 in very good yield. As previously mentioned, due to the poor reactivity of steroidal ketones and their imine derivatives, most MCRs with ketosteroids described in the literature
- with an alkynyl seco-cholestane [34]. Multicomponent synthesis of steroid–thiazole hybrids from a steroidal ketone [36]. Synthesis of cholanic pseudo-peptide derivatives by novel MCRs based on the reactivity of ynamide [37][38]. Synthesis of steroid-fused pyrimidines and pyrimidones using the Biginelli

Graphical Abstract

Figure 1: Structures of natural steroids of A) animal and B) plant origin.

Scheme 1: Synthesis of a steroidal β-lactam by Ugi reaction of a cholanic aldehyde [14].

Scheme 2: Synthetic route to steroidal 2,5-diketopiperazines based on a diastereoselective Ugi-4CR with an an...

Scheme 3: Multicomponent synthesis of a heterocycle–steroid hybrid using a ketosteroid as carbonyl component [18]....

Scheme 4: Synthesis of peptidomimetic–steroid hybrids using the Ugi-4CR with spirostanic amines and carboxyli...

Scheme 5: Synthesis of azasteroids using the Ugi-4CR with androstanic and pregnanic carboxylic acids [22].

Figure 2: Ugi-4CR-derived library of androstanic azasteroids with diverse substitution patterns at the phenyl...

Scheme 6: Synthesis of 4-azacholestanes by an intramolecular Ugi-4C-3R [26].

Scheme 7: Synthesis of amino acid–steroid hybrid by multiple Ugi-4CR using steroidal isocyanides [29].

Scheme 8: Synthesis of ecdysteroid derivatives by Ugi-4CR using a steroidal isocyanide [30].

Scheme 9: Stereoselective multicomponent synthesis of a steroid–tetrahydropyridine hybrid using a chiral bifu...

Scheme 10: Pd(II)-catalyzed three-component reaction with an alkynyl seco-cholestane [34].

Scheme 11: Multicomponent synthesis of steroid–thiazole hybrids from a steroidal ketone [36].

Scheme 12: Synthesis of cholanic pseudo-peptide derivatives by novel MCRs based on the reactivity of ynamide [37,38].

Scheme 13: Synthesis of steroid-fused pyrimidines and pyrimidones using the Biginelli-3CR [39,42,43].

Scheme 14: Synthesis of steroidal pyridopyrimidines by a reaction sequence comprising a 4CR followed by a post...

Scheme 15: Synthesis of steroid-fused pyrimidines by MCR of 2-hydroxymethylene-3-ketosteroids [46].

Scheme 16: Synthesis of steroid-fused naphthoquinolines by the Kozlov–Wang MCR using ketosteroids [50,51].

Scheme 17: Conjugation of steroids to carbohydrates and peptides by the Ugi-4CR [62,63].

Scheme 18: Solid-phase multicomponent conjugation of peptides to steroids by the Ugi-4CR [64].

Scheme 19: Solid-phase multicomponent conjugation of peptides to steroids by the Petasis-3CR [68].

Scheme 20: Synthesis of steroidal macrobicycles (cages) by multiple multicomponent macrocyclizations based on ...

Scheme 21: One-pot synthesis of steroidal cages by double Ugi-4CR-based macrocyclizations [76].
Microwave-assisted synthesis of N,N-bis(phosphinoylmethyl)amines and N,N,N-tris(phosphinoylmethyl)amines bearing different substituents on the phosphorus atoms
- Erika Bálint,
- Anna Tripolszky,
- László Hegedűs and
- György Keglevich
Beilstein J. Org. Chem. 2019, 15, 469–473, doi:10.3762/bjoc.15.40
- elaborated by us for the synthesis of several (aminomethyl)phosphine oxides [10][11]. As regards α-aminophosphine oxides with different P-substituents, only two different types were reported. Olszewski and co-workers synthesized chiral thiazole-substituted aminophosphine oxides 2 through the Pudovik reaction
- of alkylphenylphosphine oxides and the corresponding aldimine derivatives of thiazole 1 (Scheme 1) [12]. Cherkasov and his group applied the Kabachnik–Fields reaction to synthesize a P-chiral aminophosphine oxide with a 2-pyridyl substituent 3 (Scheme 2) [13]. Bis(aminophosphine oxide) derivatives
- isolated in high yields and fully characterized. Synthesis of chiral thiazole-substituted aminophosphine oxides. Synthesis of a P-chiral aminophosphine oxide containing a 2-pyridyl moiety. Condensation of (octylaminomethyl)dihexylphosphine oxide with paraformaldehyde and di(p-tolyl)phosphine oxide

Graphical Abstract

Scheme 1: Synthesis of chiral thiazole-substituted aminophosphine oxides.

Scheme 2: Synthesis of a P-chiral aminophosphine oxide containing a 2-pyridyl moiety.

Scheme 3: Condensation of (octylaminomethyl)dihexylphosphine oxide with paraformaldehyde and di(p-tolyl)phosp...

Scheme 4: Synthesis of (aminomethyl)phosphine oxides 5–7.

Scheme 5: Synthesis of (aminomethyl)diphenylphosphine oxide (9).

Scheme 6: Synthesis of N,N-bis(phosphinoylmethyl)amines 10a,b, 11a,b and 12a,b bearing different substituents...

Scheme 7: Synthesis of N,N-bis(phosphinoylmethyl)amines 13a–c.

Scheme 8: Synthesis of N,N,N-tris(phosphinoylmethyl)amines 14–17.
Annulation of 1H-pyrrole-2,3-diones by thioacetamide: an approach to 5-azaisatins
- Aleksandr I. Kobelev,
- Ekaterina E. Stepanova,
- Maksim V. Dmitriev and
- Andrey N. Maslivets
Beilstein J. Org. Chem. 2019, 15, 364–370, doi:10.3762/bjoc.15.32
- corresponding 1H-pyrrolo[3,2-c]pyridine-2,3-dione 4l. The most possible explanation of this fact is the steric hindrance introduced by the N-phenyl substituent, which obstructs the thiazole ring opening. Conclusion In summary, we have developed a novel approach to 1H-pyrrolo[3,2-c]pyridine-2,3-diones (5

Graphical Abstract

Scheme 1: Approaches to the synthesis of the 5-azaisatin core.

Scheme 2: Our previous work on the interaction of PBTs 2 with thioamides.

Scheme 3: Interaction of PBTs 2 with thioacetamide.

Scheme 4: Plausible pathways for the formation of compound 4.

Scheme 5: Experiments on the intermolecular trapping of spiro[thiazolo-5,2'-pyrrole] 3a.

Scheme 6: Exploration of the substrate scope.

Scheme 7: Interaction of PBT 2a with N-phenylthioacetamide.
Synthesis of nonracemic hydroxyglutamic acids
- Dorota G. Piotrowska,
- Iwona E. Głowacka,
- Andrzej E. Wróblewski and
- Liwia Lubowiecka
Beilstein J. Org. Chem. 2019, 15, 236–255, doi:10.3762/bjoc.15.22

- precursor in the total synthesis of longicatenamycin A [25]. Syntheses of several modified cephems started from dimethyl (2S,4R)-N-Boc-4-hydroxyglutamate (81) [114]. Protected 4-hydroxyglutamic acids (2S,4S)-66 [75][76] and (2S,4S)-85a [84][85] after installation of the thiazole ring at the C1 terminus were

Graphical Abstract

Figure 1: Structure of L-glutamic acid.

Figure 2: 3-Hydroxy- (2), 4-hydroxy- (3) and 3,4-dihydroxyglutamic acids (4).

Figure 3: Enantiomers of 3-hydroxyglutamic acid (2).

Scheme 1: Synthesis of (2S,3R)-2 from (R)-Garner's aldehyde. Reagents and conditions: a) MeOCH=CH–CH(OTMS)=CH2...

Scheme 2: Synthesis of (2S,3R)-2 and (2S,3S)-2 from (R)-Garner’s aldehyde. Reagents and conditions: a) H2C=CH...

Scheme 3: Two-carbon homologation of the protected L-serine. Reagents and conditions: a) Fmoc-succinimide, Na2...

Scheme 4: Synthesis of di-tert-butyl ester of (2R,3S)-2 from L-serine. Reagents and conditions: a) PhSO2Cl, K2...

Scheme 5: Synthesis of (2R,3S)-2 from O-benzyl-L-serine. Reagents and conditions: a) (CF3CH2O)2P(O)CH2COOMe, ...

Scheme 6: Synthesis of (2S,3R)-2 employing a one-pot cis-olefination–conjugate addition sequence. Reagents an...

Scheme 7: Synthesis of the orthogonally protected (2S,3R)-2 from a chiral aziridine. Reagents and conditions:...

Scheme 8: Synthesis of N-Boc-protected (2S,3R)-2 from D-phenylglycine. Reagents and conditions: a) BnMgCl, et...

Scheme 9: Synthesis of (2S,3R)-2 employing ketopinic acid as chiral auxiliary. Reagents and conditions: a) Br2...

Scheme 10: Synthesis of dimethyl ester of (2S,3R)-2 employing (1S)-2-exo-methoxyethoxyapocamphane-1-carboxylic...

Scheme 11: Synthesis of N-Boc-protected dimethyl ester of (2S,3R)-2 from (S)-N-(1-phenylethyl)thioacetamide. R...

Scheme 12: Synthesis of N-Boc-protected dimethyl ester of (2S,3R)-2 via Sharpless epoxidation. Reagents and co...

Scheme 13: Synthesis of (2S,3S)-2 from the imide 51. Reagents and conditions: a) NaBH4, MeOH/CH2Cl2; b) Ac2O, ...

Scheme 14: Synthesis of (2R,3S)-2 and (2S,3S)-2 from the acetolactam 55 (PMB = p-methoxybenzyl). Reagents and ...

Scheme 15: Synthesis of (2S,3R)-2 from D-glucose. Reagents and conditions: a) NaClO2, 30% H2O2, NaH2PO4, MeCN;...

Figure 4: Enantiomers of 3-hydroxyglutamic acid (3).

Scheme 16: Synthesis of (4S)-4-hydroxy-L-glutamic acid [(2S,4S)-3] by electrophilic hydroxylation. Reagents an...

Scheme 17: Synthesis of all stereoisomers of 4-hydroxyglutamic acid (3). Reagents and conditions: a) Br2, PBr5...

Scheme 18: Synthesis of the orthogonally protected 4-hydroxyglutamic acid (2S,4S)-73. Reagents and conditions:...

Scheme 19: Synthesis of (2S,4R)-4-acetyloxyglutamic acid as a component of a dipeptide. Reagents and condition...

Scheme 20: Synthesis of N-Boc-protected dimethyl esters of (2S,4R)- and (2S,4S)-3 from (2S,4R)-4-hydroxyprolin...

Scheme 21: Synthesis of orthogonally protected (2S,4S)-3 from (2S,4R)-4-hydroxyproline. Reagents and condition...

Scheme 22: Synthesis of the protected (4R)-4-hydroxy-L-pyroglutamic acid (2S,4R)-87 by electrophilic hydroxyla...

Figure 5: Enantiomers of 3,4-dihydroxy-L-glutamic acid (4).

Scheme 23: Synthesis of (2S,3S,4R)-4 from the epoxypyrrolidinone 88. Reagents and conditions: a) MeOH, THF, KC...

Scheme 24: Synthesis of (2S,3R,4R)-4 from the orthoester 92. Reagents and conditions: a) OsO4, NMO, acetone/wa...

Scheme 25: Synthesis of (2S,3S,4S)-4 from the aziridinolactone 95. Reagents and conditions: a) BnOH, BF3·OEt2,...

Scheme 26: Synthesis of (2S,3S,4R)-4 and (2R,3S,4R)-4 from cyclic imides 106. Reagents and conditions: a) NaBH4...

Scheme 27: Synthesis of (2R,3R,4R)-4 and (2S,3R,4R)-4 from the cyclic meso-imide 110. Reagents and conditions:...

Scheme 28: Synthesis of (2S,3S,4S)-4 from the protected serinal (R)-23. Reagents and conditions: a) Ph3P=CHCOO...

Scheme 29: Synthesis of (2S,3S,4S)-4 from O-benzyl-N-Boc-D-serine. Reagents and conditions: a) ClCOOiBu, TEA, ...

Scheme 30: Synthesis of (2S,3S,4R)-127 by enantioselective conjugate addition and asymmetric dihydroxylation. ...

Figure 6: Structures of selected compounds containing hydroxyglutamic motives (in blue).
Oxidative and reductive cyclization in stiff dithienylethenes
- Michael Kleinwächter,
- Ellen Teichmann,
- Lutz Grubert,
- Martin Herder and
- Stefan Hecht
Beilstein J. Org. Chem. 2018, 14, 2812–2821, doi:10.3762/bjoc.14.259
- ][47] as the key intermediate undergoing thermal cyclization or cycloreversion, typically in the context of so-called “ECE” and “EEC” mechanisms, respectively [48]. To contribute to this discussion, nonsymmetrical DAEs bearing two electronically distinct aryl moieties (CF3- and Me-thiazole) [5] or

Graphical Abstract

Scheme 1: Combining double bond isomerization (E/Z) and cyclization/cycloreversion (Z/C) in three-state switc...

Scheme 2: Overview of all sDTE and reference DTE compounds investigated in this study. The compound names ind...

Figure 1: Cyclic voltammograms of sDTE66-Me. a) Both E- (black line) and Z-isomer (blue dashed line) display ...

Figure 2: Spectroelectrochemistry of sDTE66-Me. Absorption changes during CV, insets showing the correspondin...

Scheme 3: Proposed mechanism for the oxidative cyclization of sDTE66-Me. Upon two-fold oxidation, both open i...

Figure 3: Anodic peak potentials (Epa) of sDTEs and reference compounds in MeCN. Solid circles refer to the f...

Figure 4: Cyclic voltammograms of sDTE66-PhCN. The reduction of a) E-sDTE66-PhCN (black line) is reversible, ...

Figure 5: Cyclic voltammogram of DTE-PhFluorene. The ring-closed isomer (red dashed line) is formed both unde...

Figure 6: Cyclic voltammograms of Me2NPh-DTE-PhCN displaying separated one-electron anodic and cathodic waves...

Scheme 4: Proposed mechanism to explain the observed selectivity of anodic and cathodic cyclization in sDTE66...
Targeting the Pseudomonas quinolone signal quorum sensing system for the discovery of novel anti-infective pathoblockers
- Christian Schütz and
- Martin Empting
Beilstein J. Org. Chem. 2018, 14, 2627–2645, doi:10.3762/bjoc.14.241

- the pqs system and 2-(2-hydroxyphenyl)thiazole-4-carbaldehyde used by the iqs system (Figure 1). Strategies addressing las and rhl have been reviewed elsewhere [5][11], while to date one study on iqs inhibition has been reported [23]. Many drug discovery efforts towards effective pathoblockers have

Graphical Abstract

Figure 1: The four quorum sensing systems in P. aeruginosa las, iqs, rhl, and pqs. Abbreviations: OdDHL, N-(3...

Figure 2: Schematic overview of the PQS biosynthesis and involvement of related metabolites and PqsE in virul...

Figure 3: Anthranilic acid (1) and derivatives thereof (2–4).

Figure 4: Crystal structure of 6-FABA-AMP in complex with PqsA.

Figure 5: Structures of substrate mimetic PqsA inhibitors.

Figure 6: Structures and characteristics of prominent classes of PqsD inhibitors.

Figure 7: Comparison of docking poses of three prototypic PqsD inhibitors: benzamidobenzoic acid derivative 12...

Figure 8: Structures and characteristics of hits against PqsD identified through different methods.

Figure 9: HHQ and PQS analogues as PqsD inhibitors and chemical probe used for screening.

Figure 10: Structure of PqsD-targeting biofilm inhibitor derived from linezolid.

Figure 11: Fragment-based PqsE-inhibitors 24–26.

Figure 12: PqsE co-crystal structures. (A) native product 2-ABA; (B–D) hit fragments 24–26.

Figure 13: Structurally diverse PqsBC-inhibitors 27–30.

Figure 14: Native PqsR ligand HHQ (31) which is converted into PQS (32) by PqsH and synthetic inhibitors 33 an...

Figure 15: Quinazolinone inhibitor 36 (QZN).

Figure 16: Crystal structure of QZN (36) in complex with PqsRCBD.

Figure 17: Structures of best fitting compounds 37–40 obtained from docking studies.

Figure 18: Initial hit 21 and optimized compound 42 (M64).

Figure 19: Co-crystal structure of M64 (42) with PqsRLBD.

Figure 20: M64 (42) as the starting point for further optimization leading to 43, which was further modified a...

Figure 21: Hit fragments from the benzamide (47–48) and oxadiazole class (49–51).

Figure 22: Structures of dual inhibitors 52–55.

Figure 23: Sulfonyl pyrimidines 56–58 acting as dual PqsD/PqsR inhibitors.
Applications of organocatalysed visible-light photoredox reactions for medicinal chemistry
- Michael K. Bogdos,
- Emmanuel Pinard and
- John A. Murphy
Beilstein J. Org. Chem. 2018, 14, 2035–2064, doi:10.3762/bjoc.14.179
- the anti-configuration in all cases. In addition, the option for oxidation to the oxazole or thiazole is always enticing as a way of easily accessing a diverse set of molecules. Immediately akin to the oxazole moiety is the oxadiazole heterocycle, which exhibits similar properties. There are several

Graphical Abstract

Figure 1: Depiction of the energy levels of a typical organic molecule and the photophysical processes it can...

Figure 2: General catalytic cycle of a photocatalyst in a photoredox organocatalysed reaction. [cat] – photoc...

Figure 3: Structures and names of the most common photocatalysts encountered in the reviewed literature.

Figure 4: General example of a reductive quenching catalytic cycle. [cat] – photocatalyst, [cat]* – photocata...

Figure 5: General example of an oxidative quenching catalytic cycle. [cat] – photocatalyst, [cat]* – photocat...

Scheme 1: Oxidative coupling of aldehydes and amines to amides using acridinium salt photocatalysis.

Figure 6: Biologically active molecules containing a benzamide linkage.

Scheme 2: The photocatalytic reduction of amino acids to produce the corresponding free or protected amines.

Scheme 3: The organocatalysed photoredox base-mediated oxidation of thiols to disulfides.

Scheme 4: C-Terminal modification of peptides and proteins using organophotoredox catalysis.

Scheme 5: The reduction and aryl coupling of aryl halides using a doubly excited photocatalyst (PDI).

Figure 7: Mechanism for the coupling of aryl halides using PDI, which is excited sequentially by two photons.

Scheme 6: The arylation of five-membered heteroarenes using arenediazonium salts under organophotoredox condi...

Scheme 7: The C–H (hetero)arylation of five-membered heterocycles under Eosin Y photocatalysis.

Scheme 8: The C–H sulfurisation of imidazoheterocycles using Eosin B-catalyzed photochemical methods.

Scheme 9: The introduction of the thiocyanate group using Eosin Y photocatalysis.

Scheme 10: Sulfonamidation of pyrroles using oxygen as the terminal oxidant.

Scheme 11: DDQ-catalysed C–H amination of arenes and heteroarenes.

Scheme 12: Photoredox-promoted radical Michael addition reactions of allylic or benzylic carbons.

Figure 8: Proposed mechanistic rationale for the observed chemoselectivities.

Scheme 13: The photocatalytic manipulation of C–H bonds adjacent to amine groups.

Scheme 14: The perylene-catalysed organophotoredox tandem difluoromethylation–acetamidation of styrene-type al...

Figure 9: Examples of biologically active molecules containing highly functionalised five membered heterocycl...

Scheme 15: The [3 + 2]-cycloaddition leading to the formation of pyrroles, through the reaction of 2H-azirines...

Figure 10: Proposed intermediate that determines the regioselectivity of the reaction.

Figure 11: Comparison of possible pathways of reaction and various intermediates involved.

Scheme 16: The acridinium salt-catalysed formation of oxazoles from aldehydes and 2H-azirines.

Scheme 17: The synthesis of oxazolines and thiazolines from amides and thioamides using organocatalysed photor...

Figure 12: Biologically active molecules on the market containing 1,3,4-oxadiazole moieties.

Scheme 18: The synthesis of 1,3,4-oxadiazoles from aldehyde semicarbazones using Eosin Y organophotocatalysis.

Scheme 19: The dimerization of primary thioamides to 1,2,4-thiadiazoles catalysed by the presence of Eosin Y a...

Scheme 20: The radical cycloaddition of o-methylthioarenediazonium salts and substituted alkynes towards the f...

Scheme 21: The dehydrogenative cascade reaction for the synthesis of 5,6-benzofused heterocyclic systems.

Figure 13: Trifluoromethylated version of compounds which have known biological activities.

Scheme 22: Eosin Y-catalysed photoredox formation of 3-substituted benzimidazoles.

Scheme 23: Oxidation of dihydropyrimidines by atmospheric oxygen using photoredox catalysis.

Scheme 24: Photoredox-organocatalysed transformation of 2-substituted phenolic imines to benzoxazoles.

Scheme 25: Visible light-driven oxidative annulation of arylamidines.

Scheme 26: Methylene blue-photocatalysed direct C–H trifluoromethylation of heterocycles.

Scheme 27: Photoredox hydrotrifluoromethylation of terminal alkenes and alkynes.

Scheme 28: Trifluoromethylation and perfluoroalkylation of aromatics and heteroaromatics.

Scheme 29: The cooperative asymmetric and photoredox catalysis towards the functionalisation of α-amino sp3 C–...

Scheme 30: Organophotoredox-catalysed direct C–H amidation of aromatics.

Scheme 31: Direct C–H alkylation of heterocycles using BF3K salts. CFL – compact fluorescent lamp.

Figure 14: The modification of camptothecin, demonstrating the use of the Molander protocol in LSF.

Scheme 32: Direct C–H amination of aromatics using acridinium salts.

Scheme 33: Photoredox-catalysed nucleophilic aromatic substitution of nucleophiles onto methoxybenzene derivat...

Scheme 34: The direct C–H cyanation of aromatics with a focus on its use for LSF.
An overview of recent advances in duplex DNA recognition by small molecules
- Sayantan Bhaduri,
- Nihar Ranjan and
- Dev P. Arya
Beilstein J. Org. Chem. 2018, 14, 1051–1086, doi:10.3762/bjoc.14.93
- lipophilicity [53]. These structural analogs comprise of branched N-alkyl- and N-cycloalkylpyrroles to test the conformational flexibility towards DNA binding. Hydrophobic N-terminal amides and substituted thiazole replacing pyrrole were installed in order to impart more lipophilicity. All these compounds were
- A has shown much higher affinity than parent distamycin A (preferential selectivity towards G·C sites) due to the presence of an isopropyl-substituted thiazole ring, which makes the molecule more hydrophobic [54]. Recently, a small set of analogs of thiazotropsin was designed and synthesized to
- nicotinamide as shown in the Figure 4 (conjugate 5). Suckling et al. further demonstrated another structural analog of thiazotropsin conjugate 6, a heterocylic triamide containing thiazole carboxylic acid, which showed significant activity (MIC = 63 nM) against Trypanosoma brucei [55]. However, the authors

Graphical Abstract

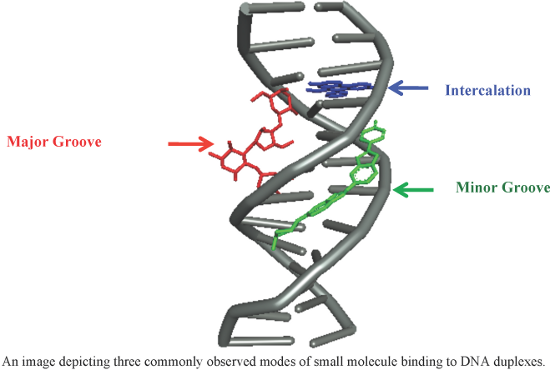
Figure 1: A figure showing the hydrogen bonding patterns observed in (a) duplex (b) triplex and (c) quadruple...

Figure 2: (a) Portions of MATα1–MATα2 are shown contacting the minor groove of the DNA substrate. Key arginin...

Figure 3: Chemical structures of naturally occurring and synthetic hybrid minor groove binders.

Figure 4: Synthetic structural analogs of distamycin A by replacing one or more pyrrole rings with other hete...

Figure 5: Pictorial representation of the binding model of pyrrole–imidazole (Py/Im) polyamides based on the ...

Figure 6: Chemical structures of synthetic “hairpin” pyrrole–imidazole (Py/Im) conjugates.

Figure 7: (a) Minor groove complex formation between DNA duplex and 8-ring cyclic Py/Im polyamide (conjugate ...

Figure 8: Telomere-targeting tandem hairpin Py/Im polyamides 23 and 24 capable of recognizing >10 base pairs; ...

Figure 9: Representative examples of recently developed DNA minor groove binders.

Figure 10: Chemical structures of bisbenzamidazoles Hoechst 33258 and 33342 and their synthetic structural ana...

Figure 11: Chemical structures of bisamidines such as diminazene, DAPI, pentamidine and their synthetic struct...

Figure 12: Representative examples of recently developed bisamidine derivatives.

Figure 13: Chemical structures of chromomycin, mithramycin and their synthetic structural analogs 91 and 92.

Figure 14: Chemical structures of well-known naturally occurring DNA binding intercalators.

Figure 15: Naturally occurring indolocarbazole rebeccamycin and its synthetic analogs.

Figure 16: Representative examples of naturally occurring and synthetic derivatives of DNA intercalating agent...

Figure 17: Several recent synthetic varieties of DNA intercalators.

Figure 18: Aminoglycoside (neomycin)–Hoechst 33258/intercalator conjugates.

Figure 19: Chemical structures of triazole linked neomycin dimers and neomycin–bisbenzimidazole conjugates.

Figure 20: Representative examples of naturally occurring and synthetic analogs of DNA binding alkylating agen...

Figure 21: Chemical structures of naturally occurring and synthetic analogs of pyrrolobenzodiazepines.
Fluorogenic PNA probes
- Tirayut Vilaivan
Beilstein J. Org. Chem. 2018, 14, 253–281, doi:10.3762/bjoc.14.17

- via the formation of high order complexes between PNA–dsDNA was also possible using a combination of PNA probes and CPP [122]. The use of a labeled PNA probe can be avoided by employing a fluorescence dye that can bind to PNA–DNA duplexes or triplexes, such as thiazole orange (TO) [123]. A

Graphical Abstract

Figure 1: The design of classical DNA molecular beacons.

Figure 2: Structures of DNA and selected PNA systems.

Figure 3: Various binding modes of PNA to double stranded DNA including triplex formation, triplex invasion, ...

Figure 4: The design and working principle of the PNA beacons according to (A) Ortiz et al. [41] and (B) Armitage...

Figure 5: The design of "stemless" PNA beacons.

Figure 6: The applications of PNA openers to facilitate the binding of PNA beacons to double stranded DNA [40,47].

Figure 7: The working principle of snap-to-it probes that employed metal chelation to bring the dyes in close...

Figure 8: Examples of pre-formed dye-labeled PNA monomers and functionalizable PNA monomers.

Figure 9: Dual-labeled PNA beacons with end-stacking or intercalating quencher.

Figure 10: The working principle of hybrid PNA-peptide beacons for detection of (A) proteins [80] and (B) protease...

Figure 11: The working principle of binary probes.

Figure 12: The working principle of nucleic acid templated fluorogenic reactions leading to a (A) ligated prod...

Figure 13: Catalytic cycles in fluorogenic nucleic acid templated reactions [90].

Figure 14: The working principle of strand displacement probes.

Figure 15: (A) Examples of CPP successfully used with labeled PNA probes. (B) The use of single-labeled PNA pr...

Figure 16: The concept of PNA–GO platform for DNA/RNA sensing.

Figure 17: Single-labeled fluorogenic PNA probes.

Figure 18: Examples of environment sensitive fluorescent labels that have been incorporated into PNA probes as...

Figure 19: The mechanism of fluorescence change in TO dye.

Figure 20: Fluorescent nucleobases capable of hydrogen bonding that have been incorporated into PNA probes.

Figure 21: Comparison of the designs of the (A) light-up PNA probe and (B) FIT PNA probe.

Figure 22: The structures of TO and its analogues that have successfully been used in FIT PNA probes.

Figure 23: The working principle of dual-labeled FIT PNA probes [222,223].
Halogen-containing thiazole orange analogues – new fluorogenic DNA stains
- Aleksey A. Vasilev,
- Meglena I. Kandinska,
- Stanimir S. Stoyanov,
- Stanislava B. Yordanova,
- David Sucunza,
- Juan J. Vaquero,
- Obis D. Castaño,
- Stanislav Baluschev and
- Silvia E. Angelova
Beilstein J. Org. Chem. 2017, 13, 2902–2914, doi:10.3762/bjoc.13.283
- monomethine cyanine dyes 5a–d, which are analogues of the commercial dsDNA fluorescence binder thiazole orange (TO), have been synthesized. The synthesis was achieved by using a simple, efficient and environmetally benign synthetic procedure to obtain these cationic dyes in good to excellent yields
- fluorescence spectroscopy) and theoretical (DFT and TDDFT calculations) methods. Keywords: cyanine dyes; DFT calculations; green synthesis; nucleic acids; thiazole orange; Introduction Since the discovery by Lee and co-workers [1][2] that the old photographic dye thiazole orange, TO, (Scheme 1) has excellent
- properties as a fluorogenic noncovalent DNA or RNA binder, many representatives of this class of dyes have been developed [3][4][5][6][7]. Thiazole orange does not fluoresce in the free state in solution. Fluorescence appears when the rotation about the monomethine bridge between the two heterocyclic

Graphical Abstract

Scheme 1: Chemical structures of TO and SYBR Green I – commercial monomethine fluorescent dsDNA binders.

Scheme 2: Synthesis of the monofluoro-substituted dye TO-1F. Reagents, conditions and yields: (i) sodium meth...

Scheme 3: Synthesis of new halogen-containing analogues of TO. Reagents, conditions and yields: (i) DMS, 100 ...

Figure 1: Absorption spectra of the studied compounds in MeOH (c = 1 × 10−5 M).

Figure 2: Absorption spectra of dye 5b (c = 5 × 10−7 M) in TE buffer, in the absence and presence of dsDNA (c...

Figure 3: Fluorescence spectra of dye 5a (c = 5 × 10–7 M) in TE buffer and in the presence of increasing conc...

Figure 4: Photostability of dyes TO-7Cl and 5a–d in acetonitrile in the concentration range of 1 × 10−5 M.

Figure 5: B3LYP optimized structures.

Figure 6: Graphical representation of the frontier orbitals (isodensity plot, isovalue = 0.02 a.u.).

Figure 7: Simulated TDPBE0 spectra in methanol.

Figure 8: Electron density (isovalue = 0.002) mapped with electrostatic potential (color scheme: green for ne...
Reactivity of bromoselenophenes in palladium-catalyzed direct arylations
- Aymen Skhiri,
- Ridha Ben Salem,
- Jean-François Soulé and
- Henri Doucet
Beilstein J. Org. Chem. 2017, 13, 2862–2868, doi:10.3762/bjoc.13.278

- employed to prepare 2-heteroarylated selenophenes; whereas, 2,5-dibromoselenophene generally gave 2,5-di(heteroarylated) selenophenes in high yields using both thiazole and thiophene derivatives. Moreover, sequential catalytic C2 heteroarylation, bromination, catalytic C5 arylation reactions allowed the
- allowed the coupling of several heteroaromatics such as thiazole, pyrrole, furan or imidazole derivatives with aryl bromides [36]. 2-Bromoselenophene, which was easily prepared by reaction of selenophene with N-bromosuccinimide [37], and 2-ethyl-4-methylthiazole were employed as model substrates for our
- -dibromoselenophene were found to tolerate both thiazole and thiophene derivatives (Scheme 3). The coupling of 3 equiv of thiazole derivatives with 2,5-dibromoselenophene in the presence of 2 mol % Pd(OAc)2 and KOAc as base gave the corresponding 2,5-diheteroarylated selenophenes 8 and 9 in 78% and 80% yields

Graphical Abstract

Scheme 1: Reported Pd-catalyzed heteroarylations of bromoselenophenes.

Scheme 2: Palladium-catalyzed heteroarylations of 2-bromoselenophene. *: 110 °C

Scheme 3: Palladium-catalyzed 2,5-diheteroarylation of 2,5-dibromoselenophene.

Scheme 4: Synthesis of 2-aryl-5-(heteroaryl)selenophenes.

Scheme 5: Proposed catalytic cycle.
Reagent-controlled regiodivergent intermolecular cyclization of 2-aminobenzothiazoles with β-ketoesters and β-ketoamides
- Irwan Iskandar Roslan,
- Kian-Hong Ng,
- Gaik-Khuan Chuah and
- Stephan Jaenicke
Beilstein J. Org. Chem. 2017, 13, 2739–2750, doi:10.3762/bjoc.13.270
- wastes. Here we describe the extension of this α-bromination shuttle system to 2-aminobenzothiazoles as substrates to synthesize benzo[d]imidazo[2,1-b]thiazoles. The benzo[d]imidazo[2,1-b]thiazole backbone is found in many bioactive molecules and pharmaceutical compounds as evident by its use as
- of benzo[d]imidazo[2,1-b]thiazole is the condensation of 2-aminobenzothiazole with α-halo or tosyloxy ketone [47][48]. The requirement for prior functionalization of the ketone moiety is a drawback, and several more direct methods have been developed in recent years [49][50][51][52]. Zhang et al
- the benzo[d]imidazo[2,1-b]thiazole derivatives via coupling of 2-aminobenzothiazole with the brominated β-ketoesters and amides. Over the past decade, there has been a lot of interest in KOt-Bu-mediated synthesis, especially after Itami’s group showed that KOt-Bu provides a metal-free approach to the

Graphical Abstract

Scheme 1: Two different intermolecular cyclization pathways controlled by reagents used.

Scheme 2: Scope of reaction. Reaction conditions: 1 (1.2 mmol), 2 (1.0 mmol), KOt-Bu (2 mmol), in 3 mL CBrCl3...

Scheme 3: Scope of the reaction. Reaction conditions: 1 (1.0 mmol), 2 (1.5 mmol), In(OTf)3 (0.1 mmol), in 1.5...

Scheme 4: Control experiments.

Figure 1: Proposed mechanism (benzo[d]imidazo[2,1-b]thiazoles).

Figure 2: Proposed mechanism (benzo[4,5]thiazolo[3,2-a]pyrimidin-4-ones).
Mechanochemical synthesis of small organic molecules
- Tapas Kumar Achar,
- Anima Bose and
- Prasenjit Mal
Beilstein J. Org. Chem. 2017, 13, 1907–1931, doi:10.3762/bjoc.13.186
- synthesis under mechanochemical conditions. Phosphonylation of benzothiazole and thiazole derivatives were done with organophosphorus compounds using 3 equiv of Mn(OAc)3·2H2O in a mixer mill for 1.5 h. Benzothiazole or thiazole rings having electron-donating or -withdrawing groups worked efficiently under

Graphical Abstract

Scheme 1: Mechanochemical aldol condensation reactions [48].

Scheme 2: Enantioselective organocatalyzed aldol reactions under mechanomilling. a) Based on binam-(S)-prolin...

Scheme 3: Mechanochemical Michael reaction [51].

Scheme 4: Mechanochemical organocatalytic asymmetric Michael reaction [52].

Scheme 5: Mechanochemical Morita–Baylis–Hillman (MBH) reaction [53].

Scheme 6: Mechanochemical Wittig reactions [55].

Scheme 7: Mechanochemical Suzuki reaction [56].

Scheme 8: Mechanochemical Suzuki–Miyaura coupling by LAG [57].

Scheme 9: Mechanochemical Heck reaction [59].

Scheme 10: a) Sonogashira coupling under milling conditions. b) The representative example of a double Sonogas...

Scheme 11: Copper-catalyzed CDC reaction under mechanomilling [67].

Scheme 12: Asymmetric alkynylation of prochiral sp3 C–H bonds via CDC [68].

Scheme 13: Fe(III)-catalyzed CDC coupling of 3-benzylindoles [69].

Scheme 14: Mechanochemical synthesis of 3-vinylindoles and β,β-diindolylpropionates [70].

Scheme 15: Mechanochemical C–N bond construction using anilines and arylboronic acids [78].

Scheme 16: Mechanochemical amidation reaction from aromatic aldehydes and N-chloramine [79].

Scheme 17: Mechanochemical CDC between benzaldehydes and benzyl amines [81].

Scheme 18: Mechanochemical protection of -NH2 and -COOH group of amino acids [85].

Scheme 19: Mechanochemical Ritter reaction [87].

Scheme 20: Mechanochemical synthesis of dialkyl carbonates [90].

Scheme 21: Mechanochemical transesterification reaction using basic Al2O3 [91].

Scheme 22: Mechanochemical carbamate synthesis [92].

Scheme 23: Mechanochemical bromination reaction using NaBr and oxone [96].

Scheme 24: Mechanochemical aryl halogenation reactions using NaX and oxone [97].

Scheme 25: Mechanochemical halogenation reaction of electron-rich arenes [88,98].

Scheme 26: Mechanochemical aryl halogenation reaction using trihaloisocyanuric acids [100].

Scheme 27: Mechanochemical fluorination reaction by LAG method [102].

Scheme 28: Mechanochemical Ugi reaction [116].

Scheme 29: Mechanochemical Passerine reaction [116].

Scheme 30: Mechanochemical synthesis of α-aminonitriles [120].

Scheme 31: Mechanochemical Hantzsch pyrrole synthesis [121].

Scheme 32: Mechanochemical Biginelli reaction by subcomponent synthesis approach [133].

Scheme 33: Mechanochemical asymmetric multicomponent reaction[134].

Scheme 34: Mechanochemical Paal–Knorr pyrrole synthesis [142].

Scheme 35: Mechanochemical synthesis of benzothiazole using ZnO nano particles [146].

Scheme 36: Mechanochemical synthesis of 1,2-di-substituted benzimidazoles [149].

Scheme 37: Mechanochemical click reaction using an alumina-supported Cu-catalyst [152].

Scheme 38: Mechanochemical click reaction using copper vial [155].

Scheme 39: Mechanochemical indole synthesis [157].

Scheme 40: Mechanochemical synthesis of chromene [158].

Scheme 41: Mechanochemical synthesis of azacenes [169].

Scheme 42: Mechanochemical oxidative C-P bond formation [170].

Scheme 43: Mechanochemical C–chalcogen bond formation [171].

Scheme 44: Solvent-free synthesis of an organometallic complex.

Scheme 45: Selective examples of mechano-synthesis of organometallic complexes. a) Halogenation reaction of Re...

Scheme 46: Mechanochemical activation of C–H bond of unsymmetrical azobenzene [178].

Scheme 47: Mechanochemical synthesis of organometallic pincer complex [179].

Scheme 48: Mechanochemical synthesis of tris(allyl)aluminum complex [180].

Scheme 49: Mechanochemical Ru-catalyzed olefin metathesis reaction [181].

Scheme 50: Rhodium(III)-catalyzed C–H bond functionalization under mechanochemical conditions [182].

Scheme 51: Mechanochemical Csp2–H bond amidation using Ir(III) catalyst [183].

Scheme 52: Mechanochemical Rh-catalyzed Csp2–X bond formation [184].

Scheme 53: Mechanochemical Pd-catalyzed C–H activation [185].

Scheme 54: Mechanochemical Csp2–H bond amidation using Rh catalyst.

Scheme 55: Mechanochemical synthesis of indoles using Rh catalyst [187].

Scheme 56: Mizoroki–Heck reaction of aminoacrylates with aryl halide in a ball-mill [58].

Scheme 57: IBX under mechanomilling conditions [8].

Scheme 58: Thiocarbamoylation of anilines; trapping of reactive aryl-N-thiocarbamoylbenzotriazole intermediate...
Synthesis of the heterocyclic core of the D-series GE2270
- Christophe Berini,
- Thibaut Martin,
- Pierrik Lassalas,
- Francis Marsais,
- Christine Baudequin and
- Christophe Hoarau
Beilstein J. Org. Chem. 2017, 13, 1407–1412, doi:10.3762/bjoc.13.137
- as readily available starting material. Keywords: antibiotic; bromination; BSC; C–H arylation; cross-coupling; Hantzsch synthesis; thiopeptide; Introduction Thiopeptide antibiotics are a class of peptide-derived macrocycles which contain many thiazole and thiazoline units, with almost 90 structures
- thiazole units using cross-coupling reactions and Hantzsch-type condensation to a pyridine central platform. This second strategy has been first initially explored by Kelly in 1991 for the first preparation of heterocyclic core of micrococcinic acid [12][13] but due to the unique and original mode of
- within five Hantzsch thiazole building steps [14]. Nicolaou and Moody reported then the synthesis of the heterocyclic core of GE2270 by using a late-stage [4 + 2] cycloaddition of a sophisticated bithiazolylazadiene with alkynylated thiazole [15][16]. As highly innovative strategy, Bach reported in 2005

Graphical Abstract

Figure 1: Main synthetic strategies towards heterocyclic cores of D-series GE2270 and our present one.

Scheme 1: Synthesis of trithiazolylpyridine 9. Reaction conditions: a) Pd(OAc)2 (5 mol %), CyJohnPhos (10 mol...

Scheme 2: Synthesis of chiral thioamide 16. Reaction conditions: a) SnCl2∙2H2O, dioxane/H2O (1:3), 0 °C to rt...

Scheme 3: Synthesis of the heterocyclic core of the D-series GE2270. Reaction conditions: a) TBDMSOTf, NEt3, ...
Total syntheses of the archazolids: an emerging class of novel anticancer drugs
- Stephan Scheeff and
- Dirk Menche
Beilstein J. Org. Chem. 2017, 13, 1085–1098, doi:10.3762/bjoc.13.108
- characteristic (Z,Z,E)-triene, a thiazole side chain and a characteristic sequence of eight methyl and hydroxy-bearing stereocenters. Synthetic chemistry is of key importance to enhance the supply of these scarce polyketides to fully evaluate the biological potential and develop them as potential drug candidates
- suitable olefination strategy. Subsequently, for connecting the resulting fragment to the thiazole subunit 6 a Heck reaction was envisioned as part of studies advancing this type of Palladium-catalyzed coupling strategies in complex target synthesis [61][62][63][64][65][66]. Finally, a HWE-macrocyclization
- -intermediate [76]. After conversion to amide 28 and thioamide 29 the thiazole 31 was obtained by condensation with bromoester 30. The carbamate was then introduced by activation of the deprotected hydroxy group with carbonyldiimidazole and treatment with methylamine, before the ester was selectively reduced to

Graphical Abstract

Scheme 1: Molecular structures of the archazolids.

Scheme 2: Retrosynthetic analysis of archazolid A by the Menche group.

Scheme 3: Synthesis of north-eastern fragment 5 through a Paterson anti-aldol addition and multiple Still–Gen...

Scheme 4: Synthesis of 4 through an Abiko–Masamune anti-aldol addition.

Scheme 5: Thiazol construction and synthesis of the southern fragment 6.

Scheme 6: Completion of the total synthesis of archazolid A.

Scheme 7: Synthesis of archazolid B (2) by a ring closing Heck reaction of 38.

Scheme 8: Retrosynthetic analysis of archazolid B by the Trauner group.

Scheme 9: Synthesis of acid 40 from Roche ester 41 involving a highly efficient Trost–Alder ene reaction.

Scheme 10: Synthesis of precursor 39 for the projected relay RCM reaction.

Scheme 11: Final steps of Trauner’s total synthesis of archazolid B.

Scheme 12: Overview of the different retrosynthetic approaches for the synthesis of dihydroarchazolid B (3) re...

Scheme 13: Fragment synthesis of 69 towards the total synthesis of 3.

Scheme 14: Organometallic addition of the side chain to access free alcohol 75.
N-Propargylamines: versatile building blocks in the construction of thiazole cores
- S. Arshadi,
- E. Vessally,
- L. Edjlali,
- R. Hosseinzadeh-Khanmiri and
- E. Ghorbani-Kalhor
Beilstein J. Org. Chem. 2017, 13, 625–638, doi:10.3762/bjoc.13.61
- reactions to produce various nitrogen-containing heterocycles. In this review, we highlight the most important developments on the synthesis of thiazole and its derivatives starting from N-propargylamines. This review will be helpful in the development of improved methods for the synthesis of natural and
- ]. As a consequence, many routes for the synthesis of thiazole derivatives are reported in the literature [26][27][28][29][30][31][32][33]. Among them, the Hantzsch thiazole synthesis (condensation of α-haloketones with thioamides) is the most efficient and straight forward procedure [34][35][36][37][38
- important developments on the synthesis of thiazole and its derivatives from N-propargylamines (Figure 3) which will be helpful in the development of improved methods for the synthesis of natural and biologically important compounds. The review is organized by the type of starting materials. Review 1 From N

Graphical Abstract

Figure 1: Selected examples of bioactive thiazole derivatives.

Figure 2: Some natural sources of thiazoles.

Figure 3: Some important thiazole-based compounds derived from N-propargylamines.

Scheme 1: The synthesis of thiazole-2-thiones 3 through the thermal cyclocondensation of N-propargylamines 1 ...

Scheme 2: (a) One-pot synthesis of 2-benzylthiazolo[3,2-a]benzimidazoles 6 through a base-catalyzed cascade r...

Scheme 3: (a) Synthesis of 2-iminothiazolidines 11 from N-propargylamines 9 and isothiocyanates 10. (b) Synth...

Scheme 4: (a) Synthesis of 2-aminothiazoles 17 through the reaction of ethyl 4-aminobut-2-ynoate salts 15 wit...

Scheme 5: Synthesis of 5-(iodomethylene)-3-methylthiazolidines 27 described by Zhou.

Scheme 6: Mechanism that accounts for the formation of 27.

Scheme 7: Clausen’s synthesis of fluorescein thiazolidines 30.

Scheme 8: Synthesis of multiply substituted thiazolidines 33 from N-propargylamines 32 and blocked N-isothioc...

Scheme 9: (a) Microwave-assisted cyclization of N-propargyl thiocarbamate 34. (b) Synthesis of thiazoles 39 t...

Scheme 10: Synthesis of thiazolidines 42 (42’) from the reaction of β-oxodithioesters 40 (40’) with N-propargy...

Scheme 11: Synthesis of 5-(dibromomethyl)thiazoles 44 via halocyclization of N-propargylamines 43 described by...

Scheme 12: Synthesis of dihydrothiazoles 46 through the treatment of N-propargylamides 45 with Lawesson’s reag...

Scheme 13: Synthesis of thiazoles 49 by treatment of silyl-protected N-propargylamines 47 with benzotriazolylt...

Scheme 14: Mechanism proposed to explain the synthesis of 2,5-disubstituted thiazoles 49 developed by Sasmal.

Scheme 15: Mo-catalyzed cyclization of N-propargylthiocarbamate 50.

Scheme 16: (a) DABCO-mediated intramolecular cyclization of N-(propargylcarbamothioyl)amides 53 to the corresp...

Scheme 17: Proposed mechanism for the generation of the iodine-substituted 4H-1,3-thiazines 56 and 4,5-dihydro...

Scheme 18: Au(III)-catalyzed synthesis of 5-alkylidenedihydrothiazoles 58 developed by Stevens.
Derivatives of the triaminoguanidinium ion, 5. Acylation of triaminoguanidines leading to symmetrical tris(acylamino)guanidines and mesoionic 1,2,4-triazolium-3-aminides
- Jan Szabo,
- Julian Greiner and
- Gerhard Maas
Beilstein J. Org. Chem. 2017, 13, 579–588, doi:10.3762/bjoc.13.57
- is supported inter alia by recent theoretical studies using DFT calculations, Natural Bond Orbital analysis and Natural Resonance Theory calculations for mesoionic systems of the 1,3-oxazole, 1,3-diazole and 1,3-thiazole type [39]. As 1,2,4-triazolium-3-aminides have not yet been studied in depth

Graphical Abstract

Scheme 1: Reactions of aminoguanidines with carboxylic acids and acid chlorides. The structural formulae show...

Scheme 2: Threefold N-acylation of triaminoguanidinium chloride (1) with acyl chloride 2b.

Scheme 3: Reaction of 1,2,3-tris(benzylamino)guanidinium salts 4 and 5 with acyl chlorides to give 1,2,3-tris...

Figure 1: Molecular structure of 6b·2C2H5OH in the solid state, with numbering of atoms (ORTEP plot). Selecte...

Scheme 4: Protonation and methylation of 1,2,4-triazolium-3-aminides 7b,c.

Scheme 5: Catalytic hydrogenation/debenzylation of betaines 7.

Figure 2: Left: molecular structure of 8b in the solid state (OLEX2 plot). Right: crystal structure viewed al...

Figure 3: Left: solid-state structure of 9b·H2O (ORTEP plot). Right: centrosymmetric hydrogen-bonded dimer of...

Figure 4: Solid-state structure of mesoionic compound 7a (ORTEP plot); thermal displacement ellipsoids are dr...

Figure 5: UV–vis spectra of 7a–d in chloroform (c = 0.04 mmol L−1); λmax [nm] (ε [L mol−1 cm−1]): 7a: 350 (47...
Synthesis of 1-indanones with a broad range of biological activity
- Marika Turek,
- Dorota Szczęsna,
- Marek Koprowski and
- Piotr Bałczewski
Beilstein J. Org. Chem. 2017, 13, 451–494, doi:10.3762/bjoc.13.48

- quaternary center with high diastereoselectivity as a consequence of the Stetter–Aldol–Aldol (SAA) reaction sequence. The Stetter–Aldol–Aldol conversion of the phthaldialdehyde derivatives 75 and o-formyl substituted chalcones 76 using the thiazole based carbene 78 as a precatalyst allowed to obtain spiro

Graphical Abstract

Figure 1: Biologically active 1-indanones and their structural analogues.

Figure 2: Number of papers about (a) 1-indanones, (b) synthesis of 1-indanones.

Scheme 1: Synthesis of 1-indanone (2) from hydrocinnamic acid (1).

Scheme 2: Synthesis of 1-indanone (2) from 3-(2-bromophenyl)propionic acid (3).

Scheme 3: Synthesis of 1-indanones 5 from 3-arylpropionic acids 4.

Scheme 4: Synthesis of kinamycin (9a) and methylkinamycin C (9b).

Scheme 5: Synthesis of trifluoromethyl-substituted arylpropionic acids 12, 1-indanones 13 and dihydrocoumarin...

Scheme 6: Synthesis of 1-indanones 16 from benzoic acids 15.

Scheme 7: Synthesis of 1-indanones 18 from arylpropionic and 3-arylacrylic acids 17.

Scheme 8: The NbCl5-induced one-step synthesis of 1-indanones 22.

Scheme 9: Synthesis of biologically active 1-indanone derivatives 26.

Scheme 10: Synthesis of enantiomerically pure indatraline ((−)-29).

Scheme 11: Synthesis of 1-indanone (2) from the acyl chloride 30.

Scheme 12: Synthesis of the mechanism-based inhibitors 33 of coelenterazine.

Scheme 13: Synthesis of the indane 2-imidazole derivative 37.

Scheme 14: Synthesis of fluorinated PAHs 41.

Scheme 15: Synthesis of 1-indanones 43 via transition metal complexes-catalyzed carbonylative cyclization of m...

Scheme 16: Synthesis of 6-methyl-1-indanone (46).

Scheme 17: Synthesis of 1-indanone (2) from ester 48.

Scheme 18: Synthesis of benzopyronaphthoquinone 51 from the spiro-1-indanone 50.

Scheme 19: Synthesis of the selective endothelin A receptor antagonist 55.

Scheme 20: Synthesis of 1-indanones 60 from methyl vinyl ketone (57).

Scheme 21: Synthesis of 1-indanones 64 from diethyl phthalate 61.

Scheme 22: Synthesis of 1-indanone derivatives 66 from various Meldrum’s acids 65.

Scheme 23: Synthesis of halo 1-indanones 69.

Scheme 24: Synthesis of substituted 1-indanones 71.

Scheme 25: Synthesis of spiro- and fused 1-indanones 73 and 74.

Scheme 26: Synthesis of spiro-1,3-indanodiones 77.

Scheme 27: Mechanistic pathway for the NHC-catalyzed Stetter–Aldol–Michael reaction.

Scheme 28: Synthesis of 2-benzylidene-1-indanone derivatives 88a–d.

Scheme 29: Synthesis of 1-indanone derivatives 90a–i.

Scheme 30: Synthesis of 1-indanones 96 from o-bromobenzaldehydes 93 and alkynes 94.

Scheme 31: Synthesis of 3-hydroxy-1-indanones 99.

Scheme 32: Photochemical preparation of 1-indanones 103 from ketones 100.

Scheme 33: Synthesis of chiral 3-aryl-1-indanones 107.

Scheme 34: Photochemical isomerization of 2-methylbenzil 108.

Scheme 35: Synthesis of 2-hydroxy-1-indanones 111a–c.

Scheme 36: Synthesis of 1-indanone derivatives 113 and 114 from η6-1,2-dioxobenzocyclobutene complex 112.

Scheme 37: Synthesis of nakiterpiosin (117).

Scheme 38: Synthesis of 2-alkyl-1-indanones 120.

Scheme 39: Synthesis of fluorine-containing 1-indanone derivatives 123.

Scheme 40: Synthesis of 2-benzylidene and 2-benzyl-1-indanones 126, 127 from the chalcone 124.

Scheme 41: Synthesis of 2-bromo-6-methoxy-3-phenyl-1-indanone (130).

Scheme 42: Synthesis of combretastatin A-4-like indanones 132a–s.

Figure 3: Chemical structures of investigated dienones 133 and synthesized cyclic products 134–137.

Figure 4: Chemical structures of 1-indanones and their heteroatom analogues 138–142.

Scheme 43: Synthesis of 2-phosphorylated and 2-non-phosphorylated 1-indanones 147 and 148 from β-ketophosphona...

Scheme 44: Photochemical synthesis of 1-indanone derivatives 150, 153a, 153b.

Scheme 45: Synthesis of polysubstituted-1-indanones 155, 157.

Scheme 46: Synthesis of 1-indanones 159a–g from α-arylpropargyl alcohols 158 using RhCl(PPh3)3 as a catalyst.

Scheme 47: Synthesis of optically active 1-indanones 162 via the asymmetric Rh-catalyzed isomerization of race...

Scheme 48: Mechanism of the Rh-catalyzed isomerization of α-arylpropargyl alcohols 161 to 1-indanones 162.

Figure 5: Chemical structure of abicoviromycin (168) and its new benzo derivative 169.

Scheme 49: Synthesis of racemic benzoabicoviromycin 172.

Scheme 50: Synthesis of [14C]indene 176.

Scheme 51: Synthesis of indanone derivatives 178–180.

Scheme 52: Synthesis of racemic pterosin A 186.

Scheme 53: Synthesis of trans-2,3-disubstituted 1-indanones 189.

Scheme 54: Synthesis of 3-aryl-1-indanone derivatives 192.

Scheme 55: Synthesis of 1-indanone derivatives 194 from 3-(2-iodoaryl)propanonitriles 193.

Scheme 56: Synthesis of 1-indanones 200–204 by cyclization of aromatic nitriles.

Scheme 57: Synthesis of 1,1’-spirobi[indan-3,3’-dione] derivative 208.

Scheme 58: Total synthesis of atipamezole analogues 211.

Scheme 59: Synthesis of 3-[4-(1-piperidinoethoxy)phenyl]spiro[indene-1,1’-indan]-5,5’-diol hydrochloride 216.

Scheme 60: Synthesis of 3-arylindan-1-ones 219.

Scheme 61: Synthesis of 2-hydroxy-1-indanones 222.

Scheme 62: Synthesis of the 1-indanone 224 from the THP/MOM protected chalcone epoxide 223.

Scheme 63: Synthesis of 1-indanones 227 from γ,δ-epoxy ketones 226.

Scheme 64: Synthesis of 2-hydroxy-2-methylindanone (230).

Scheme 65: Synthesis of 1-indanone derivatives 234 from cyclopropanol derivatives 233.

Scheme 66: Synthesis of substituted 1-indanone derivatives 237.

Scheme 67: Synthesis of 7-methyl substituted 1-indanone 241 from 1,3-pentadiene (238) and 2-cyclopentenone (239...

Scheme 68: Synthesis of disubstituted 1-indanone 246 from the siloxydiene 244 and 2-cyclopentenone 239.

Scheme 69: Synthesis of 5-hydroxy-1-indanone (250) via the Diels–Alder reaction of 1,3-diene 248 with sulfoxid...

Scheme 70: Synthesis of halogenated 1-indanones 253a and 253b.

Scheme 71: Synthesis of 1-indanones 257 and 258 from 2-bromocyclopentenones 254.

Scheme 72: Synthesis of 1-indanone 261 from 2-bromo-4-acetoxy-2-cyclopenten-1-one (260) and 1,2-dihydro-4-viny...

Scheme 73: Synthesis of 1-indanone 265 from 1,2-dihydro-7-methoxy-4-vinylnaphthalene (262) and bromo-substitut...

Scheme 74: Synthesis of 1-indanone 268 from dihydro-3-vinylphenanthrene 266 and 4-acetoxy-2-cyclopenten-1-one (...

Scheme 75: Synthesis of 1-indanone 271 from phenylselenyl-substituted cyclopentenone 268.

Scheme 76: Synthesis of 1-indanone 272 from the trienone 270.

Scheme 77: Synthesis of the 1-indanone 276 from the aldehyde 273.

Scheme 78: Synthesis of 1-indanones 278 and 279.

Scheme 79: Synthesis of 1-indanone 285 from octa-1,7-diyne (282) and cyclopentenone 239.

Scheme 80: Synthesis of benz[f]indan-1-one (287) from cyclopentenone 239 and o-bis(dibromomethyl)benzene (286)....

Scheme 81: Synthesis of 3-methyl-substituted benz[f]indan-1-one 291 from o-bis(dibromomethyl)benzene (286) and...

Scheme 82: Synthesis of benz[f]indan-1-one (295) from the anthracene epidioxide 292.

Scheme 83: Synthesis of 1-indanone 299 from homophthalic anhydride 298 and cyclopentynone 297.

Scheme 84: Synthesis of cyano-substituted 1-indanone derivative 301 from 2-cyanomethylbenzaldehyde (300) and c...

Scheme 85: Synthesis of 1-indanone derivatives 303–305 from ketene dithioacetals 302.

Scheme 86: Synthesis of 1-indanones 309–316.

Scheme 87: Mechanism of the hexadehydro-Diels–Alder (HDDA) reaction.

Scheme 88: Synthesis of 1-indenone 318 and 1-indanones 320 and 321 from tetraynes 317 and 319.

Scheme 89: Synthesis of 1-indanone 320 from the triyn 319.

Scheme 90: Synthesis 1-indanone 328 from 2-methylfuran 324.

Scheme 91: Synthesis of 1-indanones 330 and 331 from furans 329.

Scheme 92: Synthesis of 1-indanone 333 from the cycloadduct 332.

Scheme 93: Synthesis of (S)-3-arylindan-1-ones 335.

Scheme 94: Synthesis of (R)-2-acetoxy-1-indanone 338.

Figure 6: Chemical structures of obtained cyclopenta[α]phenanthrenes 339.

Scheme 95: Synthesis of the benzoindanone 343 from arylacetaldehyde 340 with 1-trimethylsilyloxycyclopentene (...






