Search results
Search for "antioxidant" in Full Text gives 101 result(s) in Beilstein Journal of Organic Chemistry.
Catalyzed and uncatalyzed procedures for the syntheses of isomeric covalent multi-indolyl hetero non-metallides: an account
Beilstein J. Org. Chem. 2021, 17, 2102–2122, doi:10.3762/bjoc.17.137
- displayed high efficiency for activating AhR. Bis(indol-3-yl)selanes possess antioxidant properties. Pioneered by Wilshire [94], their syntheses were studied by Abele [95], Naidu [96], Yang [83], Thurow [97], and Talukdar [98]. The work of Abele in 2004 involved refluxing SeO2 with N-unprotected indole in
On the application of 3d metals for C–H activation toward bioactive compounds: The key step for the synthesis of silver bullets
Beilstein J. Org. Chem. 2021, 17, 1849–1938, doi:10.3762/bjoc.17.126

- effects (85–89, Scheme 31A), such as antioxidant, α-amylase inhibitor, anticancer, and anti-inflammatory activities [164]. The reaction was promoted by non heme-Fe catalyst, behaving similarly to the bioinspired iron catalyst in a redox-selective way. The combination of Fe(phen)3(PF6)3 and tertiary
[2 + 1] Cycloaddition reactions of fullerene C60 based on diazo compounds
Beilstein J. Org. Chem. 2021, 17, 630–670, doi:10.3762/bjoc.17.55
- first gave ester 181 and then acid 182 (Scheme 31), the sodium and potassium salts of which are well soluble in water. The compound has antiviral and anticancer activity as well as pronounced antioxidant properties combined with low toxicity. Diarylmethanofullerene 183 was synthesized according to
Breakdown of 3-(allylsulfonio)propanoates in bacteria from the Roseobacter group yields garlic oil constituents
Beilstein J. Org. Chem. 2021, 17, 569–580, doi:10.3762/bjoc.17.51
- antibacterial, antifungal, antioxidant, anti-inflammatory, and anticancer effects [4]. Later on, also heterocyclic compounds including 2-vinyl-4H-1,3-dithiine (6) and 3-vinyl-3,4-dihydro-1,2-dithiine (7) were discovered [5]. The formation of these volatile sulfur compounds starts from alliin (9) [6], a non
Regioselective chemoenzymatic syntheses of ferulate conjugates as chromogenic substrates for feruloyl esterases
Beilstein J. Org. Chem. 2021, 17, 325–333, doi:10.3762/bjoc.17.30
- preparation of bioactive compounds with potential antioxidant properties [2][3][4][5]. Operating via a two-step serine protease mechanism involving a conserved Ser-His-Asp/Glu catalytic triad [6][7], Faes catalyze the hydrolysis of ester bonds linking hydroxycinnamoyl groups to the glycosyl moieties of plant
Host–guest interaction of cucurbit[8]uril with oroxin A and its effect on the properties of oroxin A
Beilstein J. Org. Chem. 2020, 16, 2332–2337, doi:10.3762/bjoc.16.194

- properties of OA was investigated through comparative experiments. The solubility of OA in water increased 22.47-fold when the concentration of Q[8] was 1 × 10−4 mol·L−1. Q[8] hardly affected the antioxidant capacity of OA, while the cumulative release of OA in gastric juice increased 2.3-fold after forming
- beneficial biological effects of OA, which include antioxidant, antidiabetic, anticancer, antibacterial, anti-inflammatory and antiviral properties [25][26][27][28][29][30]. Herein, we selected Q[8] as a host molecule and investigated its host–guest interactions with OA, as well as its effect on the
- addition of Q[8]. When the concentration of Q[8] was 1.0 × 10−4 mol·L−1, the solubility of OA was increased 22.47-fold. The solubility curve equation was S = 0.01c + 0.0575, R2 = 0.9986. Antioxidant activity OA has strong antioxidant activity and effectively eliminates ABTS+• radicals. If the antioxidant
Synthesis of 1,4-benzothiazinones from acylpyruvic acids or furan-2,3-diones and o-aminothiophenol
Beilstein J. Org. Chem. 2020, 16, 2322–2331, doi:10.3762/bjoc.16.193
- use. Particularly, some enaminones I and II were found to show antioxidant [3], antimycotic [7][8], antimycobacterial [9][10], anti-Alzheimer’s disease (JNK3 inhibitors) [11], platelet aggregation inhibitory [12], antimicrobial [7] and analgesic [13] activities. Enaminones II were reported as
Et3N/DMSO-supported one-pot synthesis of highly fluorescent β-carboline-linked benzothiophenones via sulfur insertion and estimation of the photophysical properties
Beilstein J. Org. Chem. 2020, 16, 1740–1753, doi:10.3762/bjoc.16.146
- products [1][2][3][4] and pharmacologically active compounds endowed with anticancer [5][6][7][8][9], anti-inflammatory, antioxidant, antimalarial, antifungal, and antileishmanial activities (Figure 1) [10][11][12][13]. Notably, this privileged scaffold is incorporated in several marketed drugs such as
- a variety of biological activities such as anticancer [24], inhibition of tyrosine phosphatase 1B, antioxidant properties, etc. [25][26][27]. Due to their numerous applications, they have found diverse uses such as thioindigo-like dyes, photoresponsive devices, and photoswitchable biomolecules [28
Synthesis of new dihydroberberine and tetrahydroberberine analogues and evaluation of their antiproliferative activity on NCI-H1975 cells
Beilstein J. Org. Chem. 2020, 16, 1606–1616, doi:10.3762/bjoc.16.133
- , or induce the manifestation of new features. The choice to insert an hydrazonic function on the DHBER skeleton was suggested by its wide range of interesting biological activities like antimicrobial [43], antioxidant [44], analgesic [45], anti-inflammatory [45], antiplatelet [45], anticonvulsant [46
- activity [61]. Furthermore, THBER and its derivatives are effective antioxidant agents [62][63][64][65]. To extend our investigations in this field, the obtained arylhydrazono-functionalized DHBERs were reduced to the corresponding arylhydrazono-THBERs, whose antiproliferative properties have been
- , namely pyrrolino-tetrahydroberberines, synthesized by some of us [68], exhibited enhanced antioxidant properties in comparison to THBER against a wide variety of pathophysiologically relevant oxidants such as peroxyl radicals, ferrous ion, and hydrogen peroxide [65]. In continuation of our ongoing
4-Hydroxy-3-methyl-2(1H)-quinolone, originally discovered from a Brassicaceae plant, produced by a soil bacterium of the genus Burkholderia sp.: determination of a preferred tautomer and antioxidant activity
Beilstein J. Org. Chem. 2020, 16, 1489–1494, doi:10.3762/bjoc.16.124
- in liquid production cultures, quenched hydroxy radical-induced chemiluminescence emitted by luminol by 86%. Because some Burkholderia species are pathogenic to plants and animals, the above result suggests that 1 is a potential antioxidant to counteract reactive oxygen species-based immune response
- in the host organisms. Keywords: antioxidant; Burkholderia sp; quinolone; soil bacterium; Zn2+ enrichment culture; Findings 4-Hydroxy-2(1H)-quinolone (3) is a unique structural motif mostly found in alkaloids from rutaceous plants (family Rutaceae) [1][2]. This motif has several tautomeric forms
- -hydroxy-3-methyl-2(1H)-quinolone (1, Figure 1). This compound was recently reported from the root of woad (Isatis tinctoria, family Brassicaceae) with no details of structure characterization [17]. Herein we describe the isolation, unequivocal structure characterization, and antioxidant activity of
Synthesis of pyrrolidinedione-fused hexahydropyrrolo[2,1-a]isoquinolines via three-component [3 + 2] cycloaddition followed by one-pot N-allylation and intramolecular Heck reactions
Beilstein J. Org. Chem. 2020, 16, 1225–1233, doi:10.3762/bjoc.16.106
- crispine A isolated from Carduus crispus L has antitumor activity [3]. Erythrina alkaloids have curare-like neuromuscular blocking activities [4], and also antioxidant activity against DPPH free radicals [5]. Lamellarins isolated from marine invertebrates [6] are inhibitors for HIV-1 integrase and also
Synthesis and anticancer activity of bis(2-arylimidazo[1,2-a]pyridin-3-yl) selenides and diselenides: the copper-catalyzed tandem C–H selenation of 2-arylimidazo[1,2-a]pyridine with selenium
Beilstein J. Org. Chem. 2020, 16, 1075–1083, doi:10.3762/bjoc.16.94

- variety of methods [14][15], and many of the targets exhibited biological activity (Figure 1). For example, bis(2-pyridyl) diselenide I has the potential to mitigate oxidative stress and inhibits the AChE activity [16], bis(quinolin-8-yl) diselenide (II) exhibited antioxidant activity in a skin cell model
Reaction of indoles with aromatic fluoromethyl ketones: an efficient synthesis of trifluoromethyl(indolyl)phenylmethanols using K2CO3/n-Bu4PBr in water
Beilstein J. Org. Chem. 2020, 16, 778–790, doi:10.3762/bjoc.16.71
- cruciferous vegetables [15], has a wide range of biomedical applications as an anticancer [16], antioxidant, and antiatherogenic agent [17]. Organofluorine compounds have attracted much attention due to their potential biological applications in medicinal and agricultural sciences. Introducing fluoro groups
The interaction between cucurbit[8]uril and baicalein and the effect on baicalein properties
Beilstein J. Org. Chem. 2020, 16, 71–77, doi:10.3762/bjoc.16.9
- solution using 1H NMR, UV–vis and IR spectroscopy, and DTA. The properties of the BALE–Q[8] inclusion complex, such as stability, solubility, in vitro antioxidant activity and release performance were studied by means of UV–vis spectroscopy. Results and Discussion Host–guest interactions Q[8] and BALE in
- Supporting Information File 1). If the BALE–Q[8] complex significantly reduces its antioxidant activity, this will affect the medicinal value of BALE. Figure 9 shows that Q[8] has no significant influence on the antioxidant activity of BALE. At 0.0005–0.004 mmol·L−1, BALE exhibited a linear increasing
- 3.4 × 105 L·mol−1 (neutral water). The solubility of BALE increased 4.67-fold in the phase-solubility experiment when the concentration of Q[8] was 1 × 10−4 mol·L−1. A study of the UV–vis absorption spectra with time showed that Q[8] significantly increased the stability of BALE. The antioxidant
Construction of trisubstituted chromone skeletons carrying electron-withdrawing groups via PhIO-mediated dehydrogenation and its application to the synthesis of frutinone A
Beilstein J. Org. Chem. 2019, 15, 2958–2965, doi:10.3762/bjoc.15.291
- antibacterical [4], antifungal [5][6], anticancer [7], antioxidant [8], anti-HIV [9], antiulcer, immunostimulator [10], anti-inflammatory [11], as well as biocidal [12], wound-healing [13], and immune-stimulatory activities [14]. For instance, flavoxate [15][16] is a chromone derivative that was employed as an
- of the obtained chromone derivatives was their conversion to chromone-derived natural products. Frutinone A, isolated from the leaves and root bark of Polygala fruticosa, shows various biological activities, including antibacterial, antioxidant, and potent cytochrome P450 1A2 inhibition (CYP1A2, IC50
In water multicomponent synthesis of low-molecular-mass 4,7-dihydrotetrazolo[1,5-a]pyrimidines
Beilstein J. Org. Chem. 2019, 15, 2390–2397, doi:10.3762/bjoc.15.231
- -dihydrotetrazolo[1,5-a]pyrimidine derivatives. Under similar conditions using 4,4,4-trifluoroacetoacetic ester 5-hydroxy-4,5,6,7-tetrahydrotetrazolo[1,5-a]pyrimidines are obtained. The analogous reaction with acetylacetone requires scandium(III) triflate as catalyst. The antioxidant activity of selected compounds
- was assayed with 1,1-diphenyl-2-picrylhydrazyl. Keywords: 5-aminotetrazole; antioxidant activity; 1,3-dicarbonyl compounds; multicomponent synthesis; tetrazolo[1,5-a]pyrimidines; Introduction Tetrazolo[1,5-a]pyrimidines and their partially hydrogenated derivatives are known for their interesting
- biological properties. They have been reported to have anticancer [1], antimicrobial [2][3] and antioxidant [3] activities and to act as inhibitors of hepatitis B virus [4]. The dihydro derivatives of tetrazolo[1,5-a]pyrimidines belong to a bit special kind of dihydropyrimidines due to the strong electron
Archangelolide: A sesquiterpene lactone with immunobiological potential from Laserpitium archangelica
Beilstein J. Org. Chem. 2019, 15, 1933–1944, doi:10.3762/bjoc.15.189
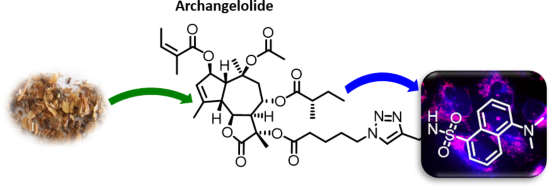
- , antioxidant, antiprotozoal, antiviral and immunobiological activities (reviewed in [1][2]). Two SLs remarkable for their immunobiological potential are archangelolide (1) and trilobolide (2), depicted in Figure 1. Compound 2 exhibits strong induction of nitric oxide (NO) in eukaryotic cells, which in turn
Steroid diversification by multicomponent reactions
Beilstein J. Org. Chem. 2019, 15, 1236–1256, doi:10.3762/bjoc.15.121
- cerebral injection of lipopolysaccharide endotoxin, with some compounds showing antioxidant and antineuroinflammatory activities. The same group employed this 4CR with cholestan-3-one for the construction of 2-amino-3-cyanodihydropyridine scaffold fused to ring A of the cholestane system [45]. Also
Synthesis of polydicyclopentadiene using the Cp2TiCl2/Et2AlCl catalytic system and thin-layer oxidation of the polymer in air
Beilstein J. Org. Chem. 2019, 15, 733–745, doi:10.3762/bjoc.15.69

- through the layers. Consequently, this leads to the transition of the oxidation from a kinetic mode into a diffusive mode. Such structural changes do not occur in a polymer that was stabilized by adding an antioxidant. Keywords: bis(cyclopentadienyl)titanium dichloride; cationic polymerization; oxidation
- occur in the polymer which was stabilized by adding an antioxidant (Agidole-1 in the amount of 0.2% by mass). The infrared spectrum of the thin layer of the stabilized polymer (Figure 17a) does not change and no consumption of double bonds in the polymer can be detected (Figure 17b). Conclusion This
- mode into a diffusion one. These structural changes do not occur in a polymer stabilized by adding an antioxidant in the studied period of time. Experimental Dehydrated toluene, prepared according to a well-known procedure, was used as a solvent [30]. Polymerization of DCPD in toluene was carried out
Synthesis of the aglycon of scorzodihydrostilbenes B and D
Beilstein J. Org. Chem. 2019, 15, 610–616, doi:10.3762/bjoc.15.56
- highly oxygenated aryl rings. Scorzodihydrostilbene E (5) features a dimeric skeleton that originates from an oxidative coupling of compound 1. The natural products exhibited antioxidative activity that was partly stronger than that of the well-known naturally occurring antioxidant resveratrol [5]. In
Synthesis of a tyrosinase inhibitor by consecutive ethenolysis and cross-metathesis of crude cashew nutshell liquid
Beilstein J. Org. Chem. 2018, 14, 2737–2744, doi:10.3762/bjoc.14.252
- display a broad range of biological activities such as antimicrobial [23], antioxidant [24], molluscicidal [25] and antiplaque [26]. Ginkgolic acids, structurally closely related analogues of anacardic acid, have been reported to exhibit tyrosinase inhibitory activity [27]. We herein report a concise
Assembly of fully substituted triazolochromenes via a novel multicomponent reaction or mechanochemical synthesis
Beilstein J. Org. Chem. 2018, 14, 2689–2697, doi:10.3762/bjoc.14.246
- equivalent nitroalkene, 1.2 equivalents of salicylaldehyde and 0.1 equivalents of DABCO as catalyst in the first step at 40 °C, and 2 equivalents of benzyl azide, 2 equivalents of acetic acid, 0.3 equivalents of BHT as antioxidant, 4 Å MS and DMF under argon atmosphere at 120 °C in the second step. Crude NMR
Targeting the Pseudomonas quinolone signal quorum sensing system for the discovery of novel anti-infective pathoblockers
Beilstein J. Org. Chem. 2018, 14, 2627–2645, doi:10.3762/bjoc.14.241

- effects are mediated either through direct PqsR-dependent action or by PqsR-independent mechanisms, which are most likely due to the iron-chelating as well as antioxidant properties of PQS [33]. Furthermore, it has been unravelled that the thioesterase PqsE, whose biosynthetic function is dispensable due
One hundred years of benzotropone chemistry
Beilstein J. Org. Chem. 2018, 14, 1120–1180, doi:10.3762/bjoc.14.98
- ][36]. Purpurogallin (8), which is biogenetically produced by oxidation of pyrogallol, and its analogues (like 9) are natural pigments (Figure 1) [37][38][39][40][41][42][43][44]. Theaflavin (10) and its derivatives, named theaflavins, are antioxidant benzotropones that are formed by the enzymatic
An overview of recent advances in duplex DNA recognition by small molecules
Beilstein J. Org. Chem. 2018, 14, 1051–1086, doi:10.3762/bjoc.14.93
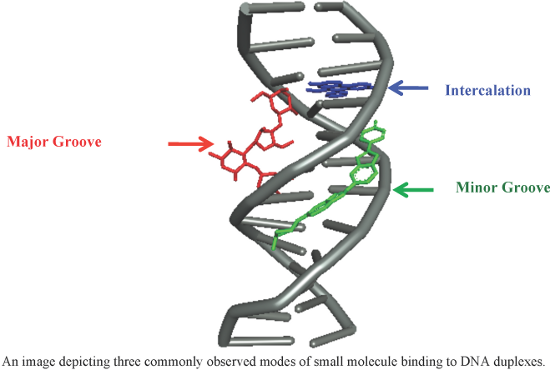
- , dihydrofuranocoumarins are another class of coumarins possessing anticancer activities. Recently, Ahmadi et al. identified several dihydrofuranocoumarins, especially grandivittin (GRA), from Ferulago macrocarpa (Fenzl) Boiss., and their mechanism of minor groove DNA binding and antibacterial, cytotoxic and antioxidant



















































































































































































































































































































































































































































































































