Search results
Search for "cycloaddition" in Full Text gives 634 result(s) in Beilstein Journal of Organic Chemistry. Showing first 200.
Recent progress in the synthesis of homotropane alkaloids adaline, euphococcinine and N-methyleuphococcinine
Beilstein J. Org. Chem. 2021, 17, 28–41, doi:10.3762/bjoc.17.4
- chemical ecology of Adalia bipunctata and the recent methodologies to obtain adaline (1), euphococcinine (2), and N-methyleuphococcinine (3). Keywords: 9-azabicyclo[3.3.1]nonane; Coccinelid beetles; dipolar cycloaddition; homotropane; ring-closing metathesis; Introduction Coccinellid beetles contain a
- forms. Key (homotropane construction) steps included: i) inter and intramolecular Mannich reaction; ii) double Michael addition in the cyclooctadienone derivative; and iii) intramolecular 1,3-dipolar cycloaddition. As shown in Scheme 1, the azabicyclononane ring is an interesting target for strategies
- dipolar cycloaddition to produce tricyclic isoxazolidines [40]. The synthesis started from 5-hexyn-1-ol (4, Scheme 2). The alcohol was treated with dihydropyran followed by alkylation using butyllithium and then, acetal deprotection, providing the alcohol 5 as a key starting compound for the (±)-adaline
All-carbon [3 + 2] cycloaddition in natural product synthesis
Beilstein J. Org. Chem. 2020, 16, 3015–3031, doi:10.3762/bjoc.16.251

- , stereo-congested, five-membered carbocycles containing all-carbon quaternary center(s) is, at present, a distinct challenge in modern synthetic chemistry, which can be accessed through the all-carbon [3 + 2] cycloaddition. More importantly, the all-carbon [3 + 2] cycloaddition can forge vicinal all
- (inclusive). Keywords: all-carbon; cyclization; [3 + 2] cycloaddition; natural product synthesis; stereocenters; Introduction The highly-substituted, stereo-congested, five-membered carbocycle containing contiguous stereocenters is one of the most common structural features in many structurally complicated
- , the all-carbon [3 + 2] cycloaddition can forge vicinal all-carbon quaternary centers [12] in a single-step operation and provides a direct access to various substituted five-membered carbocycles. These characteristics make the all-carbon [3 + 2] cycloaddition an appealing method and/or strategy in the
Regioselective synthesis of heterocyclic N-sulfonyl amidines from heteroaromatic thioamides and sulfonyl azides
Beilstein J. Org. Chem. 2020, 16, 2937–2947, doi:10.3762/bjoc.16.243
- form can be explained by steric hindrance between the phenyl and the arylsulfonyl groups. Because of the observed evolution of nitrogen and sulfur in every reaction of heterocyclic thioamides and sulfonyl azides it is logic to propose the formation of a thiatriazole ring via [3 + 2] cycloaddition of
Changed reactivity of secondary hydroxy groups in C8-modified adenosine – lessons learned from silylation
Beilstein J. Org. Chem. 2020, 16, 2854–2861, doi:10.3762/bjoc.16.234

- cycloaddition (CuAAC) became very popular [16]. A variant of this, the strain-promoted alkyne–azide cycloaddition (SPAAC) even offers the possibility of in cell application, as applies also to the inverse electron-demand Diels–Alder reaction (IEDDA) [17][18]. In vitro, often a combination of orthogonal methods
Selective and reversible 1,3-dipolar cycloaddition of 6-aryl-1,5-diazabicyclo[3.1.0]hexanes with 1,3-diphenylprop-2-en-1-ones under microwave irradiation
Beilstein J. Org. Chem. 2020, 16, 2679–2686, doi:10.3762/bjoc.16.218
- cycloaddition of in situ-generated azomethine imine under microwave conditions is described. The reaction of 6-aryl-1,5-diazabicyclo[3.1.0]hexanes with 1,3-diphenylprop-2-en-1-ones proceeds regio- and stereoselectively giving mostly good yields of the corresponding perhydropyrazolopyrazoles. The products of the
- reaction undergo cycloreversion under the reaction conditions. Keywords: azomethine imines; cycloaddition; diazabicyclohexanes; diaziridines; regioselectivity; Introduction Cyclic azomethine imines (AMIs) are very useful synthetic blocks for the preparation of diverse dinitrogenated heterocycles by 1,3
- on the catalytic cleavage of the C–N bond in the diaziridine ring under the influence of Lewis acids [23][24]. However, the selectivity of the cycloaddition for thermal or catalytic conditions can be different [25]. It was possible to obtain the cycloaddition products of unstable azomethine imines
Dawn of a new era in industrial photochemistry: the scale-up of micro- and mesostructured photoreactors
Beilstein J. Org. Chem. 2020, 16, 2484–2504, doi:10.3762/bjoc.16.202

Recent developments in enantioselective photocatalysis
Beilstein J. Org. Chem. 2020, 16, 2363–2441, doi:10.3762/bjoc.16.197

- acid so that a Dexter energy transfer process is possible from the photocatalyst to the substrate to promote 155 into its triplet state, which can then cyclise with 156 to give enantioenriched cycloaddition products 158. The selectivity of this reaction is generally low (6 examples, up to 93:7 er, 67
- ] photocycloaddition of quinolone 170. The proposed mechanism proceeds via hydrogen-bonded complex 171, which is sensitised by the pendant benzophenone to its triplet excited state 171*. The following cycloaddition completes the cycle and generates the desired cyclobutane product 173 in excellent conversion but poor
- -deficient alkenes 190 to synthesise cyclobutanes 191 (Scheme 28c) [83]. Recently, Bach et al. also employed this methodology for the intramolecular [2 + 2] cycloaddition of quinolones 192 containing either a pendant alkene or allene to obtain cyclobutanes 193 (Scheme 28d) [84]. Recently, ent-185 was applied
Hierarchically assembled helicates as reaction platform – from stoichiometric Diels–Alder reactions to enamine catalysis
Beilstein J. Org. Chem. 2020, 16, 2338–2345, doi:10.3762/bjoc.16.195

- auxiliary, Lewis acid or catalyst. In the previously reported system two different induction pathways were conceivable: (1) A chiral ligand is located close to the diene and controls the stereochemistry of the cycloaddition. (2) The chiral ligand controls the helicity of the helicate (ΔΔ or ΛΛ) and the
Design and synthesis of a bis-macrocyclic host and guests as building blocks for small molecular knots
Beilstein J. Org. Chem. 2020, 16, 2314–2321, doi:10.3762/bjoc.16.192
- alkyne–azide click cycloaddition as the linking step, and ester saponification as the cutting step [13][21] (Supporting Information File 1). The target trefoil knot using host 1 and guest 2 is shown in Figure 2c. The binding event during the double-threading step was modeled after previous literature
- more than twice as high and this route is more amenable to multigram scale reactions. Azido-bromide 6 can undergo both alkyne–azide click cycloaddition and etherification and the effect of the reaction order on the overall yield was explored next. The click cycloaddition was pursued first and reaction
- changing the order of these steps would be beneficial. Alkylation of terephthalate 12 with azido-bromide 6 using cesium carbonate as the base provided high yields (82%) of core diazide 14 (Scheme 3). Click cycloaddition of 14 with an excess of dialkyne 10 also proceeded in high yields (87%) to give diester
Regioselective cobalt(II)-catalyzed [2 + 3] cycloaddition reaction of fluoroalkylated alkynes with 2-formylphenylboronic acids: easy access to 2-fluoroalkylated indenols
Beilstein J. Org. Chem. 2020, 16, 2193–2200, doi:10.3762/bjoc.16.184
- Tatsuya Kumon Miroku Shimada Jianyan Wu Shigeyuki Yamada Tsutomu Konno Faculty of Molecular Chemistry and Engineering, Kyoto Institute of Technology, Matsugasaki, Sakyo-ku, Kyoto 606-8585, Japan 10.3762/bjoc.16.184 Abstract [2 + 3] cycloaddition reactions of fluorinated alkynes with 2
- -fluoroalkylated indenols, together with a very small amount of 3-fluoroalkylated indanones as side products. Keywords: [2 + 3] cycloaddition; cobalt catalyst; fluorine-containing; indenols; regioselective; Introduction 2,3-Disubstituted indenol derivatives are important compounds possessing high potential due
- . Despite their effective advantages, on the other hand, the reports on cycloaddition reactions with fluorine-containing alkynes for the construction of fluoroalkylated indene derivatives are rare [21][26]. Recently, our group has reported the first practical synthesis of fluoroalkylated indenol derivatives
Reactions of 3-aryl-1-(trifluoromethyl)prop-2-yn-1-iminium salts with 1,3-dienes and styrenes
Beilstein J. Org. Chem. 2020, 16, 2064–2072, doi:10.3762/bjoc.16.173
- cyclobutene and several cationic intermediates and mainly yield 2-(1-phenylvinyl)indenes. In a similar reaction cascade, a fulvene derivative was obtained with 1,4-diphenylbutadiene as the substrate. Keywords: alkynes; aromatic substitution; cyclization; cycloaddition; iminium salts; Introduction In recent
- consider the reactivity of the electrophilic (“electron-poor”) acetylenic bond toward 1,3-dienes, and show how the expected [4 + 2] or [2 + 2] cycloaddition products can enter subsequent cascade reactions toward carbocycles which incorporate a C(CF3)NMe2 structural unit. Results and Discussion The Diels
- –Alder reaction of 1-CF3-substituted propyn-1-iminium salt 1a with cyclopentadiene was carried out in order to assess the dienophilic reactivity of the cation. High conversion into cycloaddition product 2 was observed already within one hour at 0 °C. Because of its high hydrolytic lability, adduct 2 was
Syntheses of spliceostatins and thailanstatins: a review
Beilstein J. Org. Chem. 2020, 16, 1991–2006, doi:10.3762/bjoc.16.166
- strategies that rely on the generation of the C-14 stereocenter by stereoselective C–N bond formation. The Jacobsen group utilized an asymmetric Cr(III)-catalyzed cycloaddition reaction [27] between (2Z,4E)-3-(triethylsilyloxy)-2,4-hexadiene (40) and the aldehyde 41 to generate the 4-silyloxydihydropyran 43
- segment of FR901464 (1) via asymmetric catalysis Similar to their preparation of the C-8–C-15 segment, Jacobsen’s group relied on a Cr(III)-catalyzed silyloxydiene/aldehyde cycloaddition for the C-1–C-7 segment (Scheme 14) [17][18]. The reaction of the protected glycolaldehyde 86 with (1E,3E)-2
- an alkynyl ketone to produce the required stereocenter. Of the syntheses to date, Jacobsen’s use of an asymmetric Cr(III)-catalyzed cycloaddition stands as the most efficient route, in terms of synthetic steps and low catalyst loading, to the (all-cis)-2,3,5,6-tetrasubstituted tetrahydropyran and the
Regiodivergent synthesis of functionalized pyrimidines and imidazoles through phenacyl azides in deep eutectic solvents
Beilstein J. Org. Chem. 2020, 16, 1915–1923, doi:10.3762/bjoc.16.158

- (g) heterogeneous “click” cycloaddition reactions [15] using DESs as environmentally responsible and non-innocent reaction media. Telescoped, one-pot transformations of phenacyl halides to symmetrical 2,5-disubstituted pyrazines (A), through phenacyl azides as intermediates, were also found to take
One-pot synthesis of oxazolidinones and five-membered cyclic carbonates from epoxides and chlorosulfonyl isocyanate: theoretical evidence for an asynchronous concerted pathway
Beilstein J. Org. Chem. 2020, 16, 1805–1819, doi:10.3762/bjoc.16.148
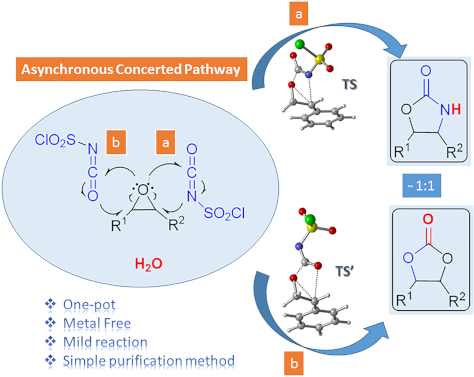
- advantageous: being a one-pot reaction with metal-free reagent, having shorter reaction times, good yields and a very simple purification method. Moreover, using the density functional theory (DFT) method at the M06-2X/6-31+G(d,p) level of theory the mechanism of the cycloaddition reactions has been elucidated
- . The further investigation of the potential energy surfaces associated with two possible channels leading to oxazolidinones and five-membered cyclic carbonates disclosed that the cycloaddition reaction proceeds via an asynchronous concerted mechanism in gas phase and in DCM. Keywords: chlorosulfonyl
- multicomponent reaction of rare-earth metal amides [28], the reaction of CO2 with propargylamines or aziridines [29][30] and the cycloaddition reaction of epoxides with isocyanates [31][32]. On the other hand, for the synthesis of five-membered cyclic carbonates, the cycloaddition of CO2 to epoxides, the
Clickable azide-functionalized bromoarylaldehydes – synthesis and photophysical characterization
Beilstein J. Org. Chem. 2020, 16, 1683–1692, doi:10.3762/bjoc.16.139
- cycloadditions with model alkynes. Besides two ortho- and para-bromo-substituted benzaldehydes, the azide functionalization of a fluorene-based structure will be presented. The copper(I)-catalyzed azide–alkyne cycloaddition (CuAAC) of the so-synthesized azide-functionalized bromocarbaldehydes with terminal
- oxazoline 24, oxazolidine 27 cyclized already during the reaction, caused by the increased basicity of the ring nitrogen. CuAAC reactions of bromocarbaldehydes We further investigated the reactivity of azide-functionalized bromocarbaldehydes 3, 4, and 5 in copper(I)-catalyzed azide–alkyne cycloaddition
Pauson–Khand reaction of fluorinated compounds
Beilstein J. Org. Chem. 2020, 16, 1662–1682, doi:10.3762/bjoc.16.138

- discussed in detail. The focus of this review is to highlight the efforts made in the field of the Pauson–Khand reaction with fluorinated compounds for the preparation of bicyclic derivatives. Review The Pauson–Khand reaction The Pauson–Khand reaction (PKR) formally consists of a [2 + 2 + 1]-cycloaddition
- ’ interest throughout the past decades. Since its discovery, numerous research groups have devoted their efforts to develop alternative ways to expand the scope of the PKR and the combination of fluorinated building blocks with this [2 + 2 + 1] cycloaddition has been efficiently applied in the preparation of
Azidophosphonium salt-directed chemoselective synthesis of (E)/(Z)-cinnamyl-1H-triazoles and regiospecific access to bromomethylcoumarins from Morita–Baylis–Hillman adducts
Beilstein J. Org. Chem. 2020, 16, 1579–1587, doi:10.3762/bjoc.16.130
- ]. Among the known synthetic transformations using functionalized MBH adducts, cycloaddition reactions are challenging and attractive for synthetic organic chemists. In this context, acetate-functionalized Morita–Baylis–Hillman adducts have been extensively utilized over other precursors. For example
- ) acetylation, (ii) azidation, and (iii) cycloaddition to produce IV–VIII. In spite of the broad scope and synthetic utility, it is evident that the multistep synthetic methodology is the only existing module for cycloaddition reactions. Our research group is focused on developing one-pot synthetic
- initiates the allylic rearrangement and thereby facilitates the removal of the crucial phosphonium oxide. The outcome of this process is the structurally relevant azido moiety IIIa, which then undergoes a 1,3-dipolar cycloaddition with the copper acetylide IVa to furnish the 1,4-disubstituted 1,2,3
NHC-catalyzed enantioselective synthesis of β-trifluoromethyl-β-hydroxyamides
Beilstein J. Org. Chem. 2020, 16, 1572–1578, doi:10.3762/bjoc.16.129
- Alyn T. Davies Mark D. Greenhalgh Alexandra M. Z. Slawin Andrew D. Smith EaStCHEM, School of Chemistry, University of St Andrews, North Haugh, St Andrews, Fife KY16 9ST, United Kingdom 10.3762/bjoc.16.129 Abstract The N-heterocyclic carbene (NHC)-catalyzed formal [2 + 2] cycloaddition between α
- moderate to good yield as a single diastereoisomer. Keywords: enantioselective catalysis; formal [2 + 2] cycloaddition; N-heterocyclic carbene; ring opening; trifluoromethyl group; Introduction The trifluoromethyl unit holds a prominent and privileged position within organic chemistry [1][2][3][4][5][6
- paper, this formal [2 + 2] protocol is extended to the use of trifluoroacetophenones to develop an enantioselective route to β-trifluoromethyl-β-hydroxy carboxylic acid derivatives (Figure 2C). Results and Discussion Optimization Optimization of the NHC-catalyzed formal [2 + 2] cycloaddition using the α
Heterogeneous photocatalysis in flow chemical reactors
Beilstein J. Org. Chem. 2020, 16, 1495–1549, doi:10.3762/bjoc.16.125

An overview on disulfide-catalyzed and -cocatalyzed photoreactions
Beilstein J. Org. Chem. 2020, 16, 1418–1435, doi:10.3762/bjoc.16.118

- , mild, and chemoselective radical catalysts that deserve more attention. The present review highlights the recent progress in the field of disulfide-catalyzed and -cocatalyzed photocatalytic reactions for different reaction types. Keywords: cycloaddition; disulfide catalyst; isomerization; oxidation
- , oxidations, or isomerizations, disulfides have increasingly proven their power. Herein, we briefly describe the progress in the field of disulfide-involving photocatalysis in recent years for different reaction types. Review Cycloaddition reactions As early as 1988, Feldman and co-workers reported an example
- of a [3 + 2] cycloaddition reactions under UV irradiation with azobis(isobutyronitrile) (AIBN) as the free radical initiator and phenyl disulfide as the catalyst, in which the three-membered rings containing double bonds and substituted olefins were transformed into five-membered-ring structures with
Recent synthesis of thietanes
Beilstein J. Org. Chem. 2020, 16, 1357–1410, doi:10.3762/bjoc.16.116
- thietane backbones developed during 1966 to 2019. Keywords: cycloaddition; cyclization; ring contraction; ring expansion; thietane; thiotherification; Review 1. Introduction Thietanes are a class of important aliphatic four-membered thiaheterocycles. Some simple alkyl and dialkyl thietanes are components
- accompanied by elimination reactions. 3. Synthesis via cycloadditions Cycloadditions, especially the photochemical [2 + 2] cycloaddition (thia-Paternò–Büchi reaction) of thiones and thioamides with olefins [15][16][17][18], and formal cycloadditions are alternative routes for the construction of thietane
- nm UV light [63]. In 1978, Gotthardt and Nieberl investigated the UV light-induced [2 + 2] cycloaddition reaction of thiones with cyclic alkenes and realized the synthesis of spirothietane derivatives. Under n → π* excitation using Na light, xanthione (196) reacted with acenaphthylene (197), indene
Highly selective Diels–Alder and Heck arylation reactions in a divergent synthesis of isoindolo- and pyrrolo-fused polycyclic indoles from 2-formylpyrrole
Beilstein J. Org. Chem. 2020, 16, 1320–1334, doi:10.3762/bjoc.16.113
- - and pyrrolo-fused polycyclic indoles is herein described, starting from 2-formylpyrrole and employing Diels–Alder and Heck arylation reactions. 3-(N-Benzyl-2-pyrrolyl)acrylates and 4-(pyrrol-2-yl)butenones underwent a highly endo-Diels–Alder cycloaddition with maleimides to furnish octahydropyrrolo
- -polycyclic structures [42], we herein combined both approaches shown in Scheme 1 to design the construction of pentacycles 11, after carrying out an uncommon but highly diastereoselective Diels–Alder cycloaddition followed by a coupling reaction. This route also allowed out the aromatization of the B ring to
- materials, which were accompanied by a decomposition residue. In contrast, maleimides 7b and 7c reacted efficiently to furnish the respective endo/exo adducts 9a–d/10a–d in good to high endo diastereoselectivity and yields (Table 2, entries 1–4). The cycloaddition with 7c led to the best selectivity (vide
[3 + 2] Cycloaddition with photogenerated azomethine ylides in β-cyclodextrin
Beilstein J. Org. Chem. 2020, 16, 1296–1304, doi:10.3762/bjoc.16.110
- decarboxylation and [3 + 2] cycloaddition take place in the ternary complex, whereas such a reactivity from bulky adamantane 3 is less likely. This proof of principle that decarboxylation and cycloaddition can be performed in the β-CD cavity has a significant importance for the design of new supramolecular
- most probably undergoes decarboxylation delivering 1AMY from the S1 state [49]. In CH3CN, 1AMY decays with a rate constant of 2.9 × 106 M−1 s−1, and reacts with methyl acrylate in [3 + 2] cycloaddition with the rate constant 2.7 × 107 M−1 s−1 [49]. Protic solvents such as CH3OH or H2O quench
- azomethyine ylides, giving formal 1,4 H-shifted products. Thus, in the presence of H2O, no cycloaddition products are anticipated. However, it should be probed if the [3 + 2] cycloaddition can compete with the 1,4 H-shift upon irradiation of an inclusion complex. Under our conditions, photodecarboxylation of
Ferrocenyl-substituted tetrahydrothiophenes via formal [3 + 2]-cycloaddition reactions of ferrocenyl thioketones with donor–acceptor cyclopropanes
Beilstein J. Org. Chem. 2020, 16, 1288–1295, doi:10.3762/bjoc.16.109
- thioketones reacted with donor–acceptor cyclopropanes in dichloromethane at room temperature in the presence of catalytic amounts of Sc(OTf)3 yielding tetrahydrothiophene derivatives, products of formal [3 + 2]-cycloaddition reactions, in moderate to high yields. In all studied cases, dimethyl 2
- thioketones to form nearly equal amounts of both diastereoisomeric tetrahydrothiophenes. A low selectivity was also observed in the reaction of a 2-phthalimide-derived cyclopropane with ferrocenyl phenyl thioketone. Keywords: [3 + 2]-cycloaddition reactions; donor–acceptor cyclopropanes; ferrocenyl
- -methanides (thiocarbonyl ylides) with electron-deficient ethylenic dipolarophiles. This method was extensively developed by Huisgen and co-workers in the 1980s [3][4][5]. In the course of these studies, a non-orthodox stepwise mechanism of the 1,3-dipolar cycloaddition was established by experiments
Synthesis of pyrrolidinedione-fused hexahydropyrrolo[2,1-a]isoquinolines via three-component [3 + 2] cycloaddition followed by one-pot N-allylation and intramolecular Heck reactions
Beilstein J. Org. Chem. 2020, 16, 1225–1233, doi:10.3762/bjoc.16.106
- Two kinds of [3 + 2] cycloaddition intermediates generated from the three-component reactions of 2-bromobenzaldehydes and maleimides with amino esters or amino acids were used for a one-pot N-allylation and intramolecular Heck reactions to form pyrrolidinedione-fused hexahydropyrrolo[2,1-a
- ]isoquinolines. The multicomponent reaction was combined with one-pot reactions to make a synthetic method with good pot, atom and step economy. MeCN was used as a preferable green solvent for the reactions. Keywords: [3 + 2] cycloaddition; Heck reaction; hexahydropyrrolo[2,1-a]isoquinoline; one-pot reactions
- stabilized azomethine ylides I-a generated from the condensation of aldehydes and amino esters for making pyrrolidines II-a have been well-reported [23][24][25][26], while the [3 + 2] cycloaddition of the less stable azomethine ylides I-b generated from the reaction of aldehydes and amino acids for






































































































































































































































































































































































































































































































































































